Abstract
Chronic cardiovascular diseases are significant health problems. Although current treatment strategies have tremendously improved disease management, up to 30% of these patients cannot be successfully treated with current treatment approaches and new treatment strategies are clearly needed. Gene therapy and therapeutic vascular growth may provide a new treatment option for these patients. Several growth factors, like vascular endothelial growth factors, fibroblast growth factors and hepatocyte growth factor have been tested in clinical trials. However, apart from demonstration of increased vascularity, very few results with clinical significance have been obtained. Problems with gene transfer efficiency, short duration of transgene expression, selection of endpoints, and suboptimal patients for gene therapy have been recognized. Ongoing gene therapy trials have included improvements in study protocols, vector delivery and endpoints, addressing the identified problems. Better, targeted delivery systems and new, more optimal growth factors have been taken to clinical testing. Recent advances in these areas will be discussed and the concept of angiogenic therapy as a sole treatment is re-evaluated. A combination with regenerative therapies or standard revascularization operations might be needed to improve tissue function and clinical benefits.
Keywords: Gene therapy, Angiogenesis, Coronary heart disease, Peripheral arterial disease, Heart failure, Gene delivery, Growth factors
Introduction
Cardiovascular diseases (CVD) represent a major healthcare problem throughout the world and continue to be the leading cause of mortality and morbidity worldwide.1,2 Additionally, many patients are not suitable for current treatments due to e.g. diffuse chronic disease or comorbidities. Thus, there is a clear need to develop novel treatment options that would have an impact not only on the symptoms of the disease but would address the underlying pathological processes.
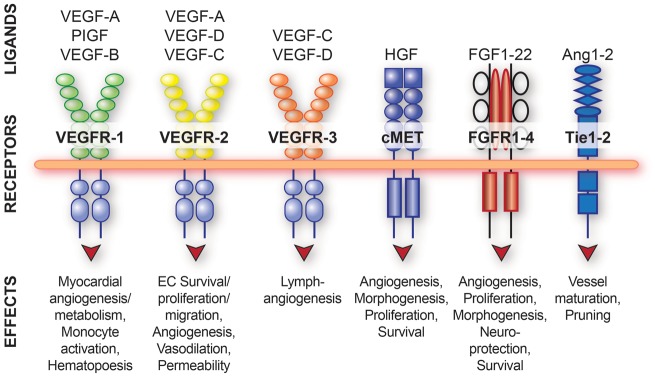
Therapeutic angiogenesis offers a potential approach for improving ischemic tissue function by stimulating blood vessel growth, increasing tissue perfusion and supporting tissue regeneration and recovery.3,4 The potential advantages of local gene-based therapy include: (i) possibility for sustained long-term exposure to therapeutic compounds, (ii) ability to target therapies to specific cell types or tissues, and (iii) decreased risk of systemic side-effects as compared to regular pharmacotherapy. Many angiogenic growth factors and transcription factors have been identified (see5 for review) which also possess other functions related to e.g. cell cycle, proliferation, energy metabolism and survival (Figure 1).
Figure 1.
Growth factor and receptor—families related to blood vessel growth as well as their other functions.
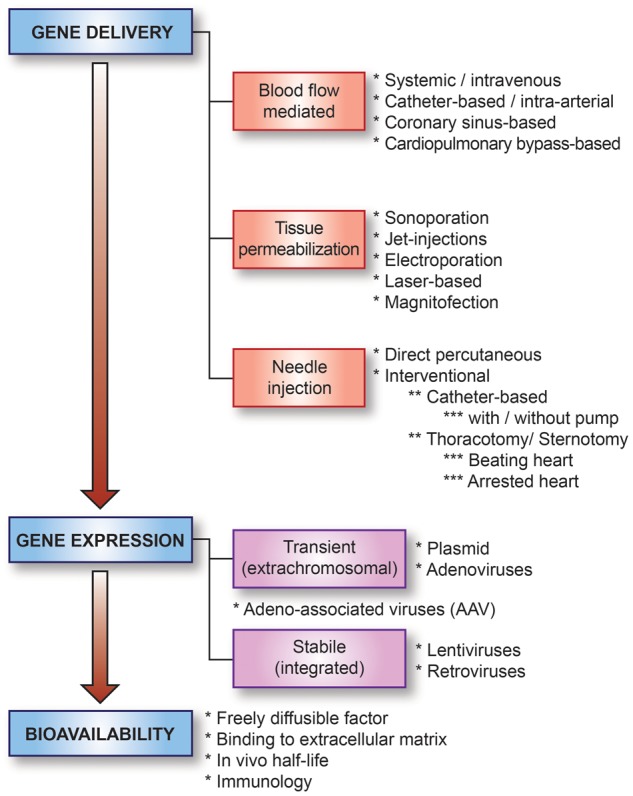
However, the local concentration of angiogenic factors is highly dependent on the used vector system and delivery route.6 Gene delivery approaches can be divided into direct tissue injections and intravascular infusions with or without surgical or catheter-mediated interventions or tissue permeabilization treatments (Figure 2). Gene transfer vectors, carrying the transgene, are usually either of plasmid or viral origin and vary in efficiency and duration of gene expression and immunogenicity (Figure 2) (see5,7 for review). Optimization of these factors should improve possibilities to achieve clinically significant results.
Figure 2.
Methods and factors affecting gene delivery and expression efficacy in clinical trials.
Evaluation of clinical angiogenic gene therapy trials
Ischemic heart disease
Failure to perform complete myocardial revascularization is associated with decreased survival and recurrent angina. Current management strategies for these patients are limited and so-called refractory angina patients make up to 5% of patients in cardiology clinics.7–9 In the past randomized controlled trials (RCTs) such as Euroinject One,10 KAT11, and NORTHERN12 have tested percutaneous intramyocardial delivery of either naked plasmid or adenoviral vascular endothelial growth factor (VEGF)—A in coronary artery disease (CAD). VEGF-A gene therapy has also been combined with cardiac bypass grafting and even performed using epicardial intramyocardial injections via minithoracotomy as a sole therapy.13 However, the results have not been very promising (Supplementary material online, Table S1) except for safety of which over 10 years follow-ups have been conducted with no significant transgene or vector related side effects.14–16
Currently, there are four angiogenic gene therapy trials ongoing in CAD (Table 1). Two trials are testing VEGFs for the treatment of myocardial ischemia. KAT301-trial tests a novel VEGF-D, in 30 patients with refractory angina and no revascularization opportunities (Table 1). This phase I/IIa RCT uses increasing doses of endocardial Ad-injections with electroanatomical targeting of injections using a NOGA© catheter system. Absolute myocardial blood flow will be measured using15 O-radiowater-PET at 3 and 12 months after the therapy. VEGF-D has not been tested in humans before and it stimulates both angiogenesis and lymphangiogenesis, thus representing a new therapeutic vascular growth strategy to treat refractory angina.17 A trial with adenovirus expressing all three major isoforms of human VEGF-A (VEGF-A116A)18 is also planned using intramyocardial injection via thoracotomy (Table 1).
Table 1.
Currently on-going or planned gene therapy trials in coronary artery disease and peripheral arterial disease
| Trial name or ID | Disease | GP | Vector | Delivery | Pat no | Novelty | Status |
|---|---|---|---|---|---|---|---|
| Trials aiming for physiological angiogenesis | |||||||
| NCT01757223 | CAD | VEGF-A116A | Ad | i.my.(tct) | 41 | Expression of 3 different VEGF-A isoforms | Planned |
| NCT00956332 | PAD | VEGF-A+Ang-1 | RV | i.a. | 28 | Ang-1 should stabilize vessels induced by VEGF-A +RV used | Results pending |
| NCT00390767 | PAD | VEGF-A+Ang-1 | RV | i.a. | 12 | Ang-1 should stabilize vessels induced by VEGF-A +RV used | Results pending |
| Trials using therapeutic vascular growth in combination with other treatments | |||||||
| KAT-PAD101 EudraCT2012-001019-22 | PAD | VEGF-DdNdC | Ad | i.m. | 30 | Gene transfer 1-2 days before operation to improve distal runoff from surgical bypass graft in PAD | Recruting |
| Trials using therapeutic vascular growth with aim for reduced side-effects | |||||||
| KAT301 EudraCT2008-003295-22 | CAD | VEGF-DdNdC | Ad | i.my.(cat) | 30 | Stimulation of both angiogenesis and lymphangiogenesis to improve cardiac fluid removal and decrease edema | Results pending |
| Trials with mitogenic and multifunctional growth factors | |||||||
| ASPIRE | CAD | FGF-4 | Ad | i.c.(cat) | 100 | PhaseIII study, 3xdosage compared to previous AGENT-2 trial | Recruiting |
| AWARE | CAD | FGF-4 | Ad | i.c. (cat) | 300 | only women recruited | Planned |
| NCT02276937 | PAD | FGF-2 | SeV | i.m. | 60 | SeV used | Recruting |
| NCT01548378 | PAD | HGF | Pl | i.m. | 200 | A large HGF trial | Results pending |
| NCT02144610 | PAD | HGF | Pl | i.m. | 500 | A large HGF trial | Recruting |
Ad, adenovirus; cat, catheter-mediated; GP, gene product; i.a., intra-arterial; i.c., intracoronary; i.m., intramuscular; i.my.: intramyocardial; Pl, plasmid; RV, retrovirus; SeV, sendaivirus; tct, thoracotomy.
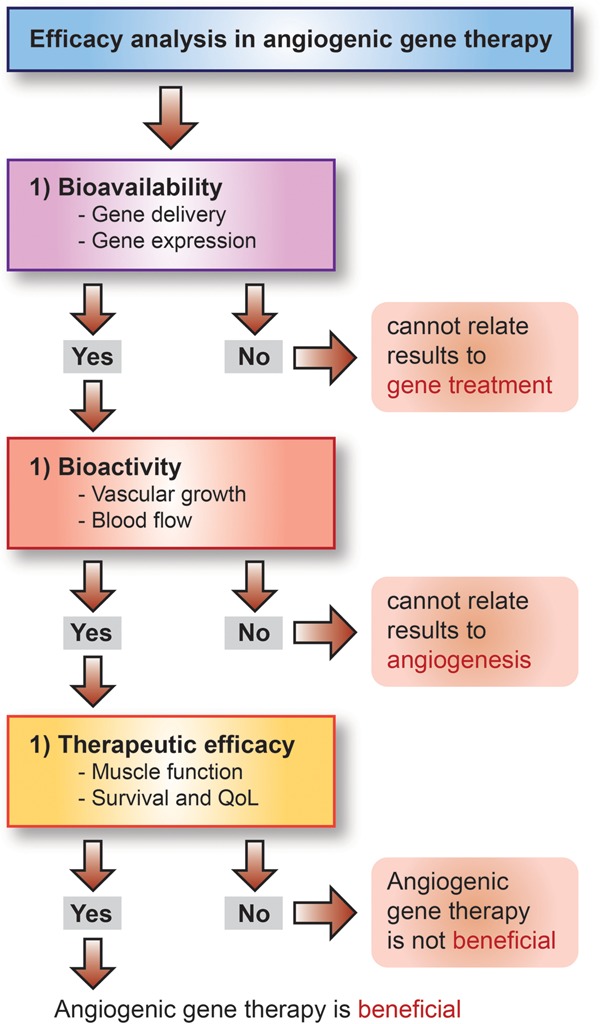
Of other angiogenic factors, intracoronary adenoviral fibroblast growth factor (FGF)-4 has shown significant improvements in post-menopausal women in a series of AGENT-trials.19 In a currently recruiting ASPIRE-trial (Table 1), intracoronary AdFGF-4 will be compared to standard care without a placebo group in an open-labelled design in 100 patients.20 The primary endpoint is the change in reversible perfusion defect size using SPECT imaging at 8 weeks. A randomized, double-blinded, placebo-controlled AWARE-trial is also planned to test intracoronary AdFGF-4 only in women with stable angina. However, the recruitment has not yet started (Table 1). Hepatocyte growth factor (HGF) either in plasmid or adenoviral constructs has been tested in CAD but to date only in small open-label studies.21,22 As the placebo effect is strong in angiogenic therapies,4,23 the RCT-study design is essential for testing efficacy (Figure 3). Additionally, hypoxia inducible factor-1 alpha (Hif-1a) has been combined with coronary bypass grafting in a placebo-controlled study with 13 patients24 but larger studies would be needed to support efficacy.
Figure 3.
Stepwise analysis of gene therapy efficacy in clinical trials.
Heart failure
Despite the fact that CAD is the most common cause of heart failure (HF), gene therapy targets for the treatment of these two diseases differ. Gene therapy in HF has been focused primarily on excitation-contraction coupling and reduction of adverse remodelling regardless of aetiology. Recent large RCTs have used intracoronary injections of adeno-associated virus (AAV)-mediated SERCA2a25 affecting myocardial calcium handling and transendocardial plasmid stromal cell derived factor (SDF)-1 mobilizing stem cells26 but the results have been disappointing. Although inefficient gene expression probably explains the negative findings in the past HF trials,27 a combination with short term angiogenic therapies could also be considered to balance cardiac metabolism and microcirculatory blood flow. For example, initially angiogenic VEGF-B has been shown to also activate expression of genes involved in the regulation of myocardial contractility and metabolism as well as to protect cardiomyocytes from apoptosis and ischemic damage through physiological hypertrophy.28,29 However, the timing and duration of VEGF-B expression seem essential as too long VEGF-B expression can controversially decrease survival.28
Peripheral arterial disease
Peripheral arterial disease (PAD) affects approximately 200 million people worldwide and its prevalence is increasing.30 Current pharmacological treatments are not effective in PAD and not all patients are suitable for operational treatments.31 Locally administrated angiogenic gene therapies have been repeatedly safe and feasible also for severely diseased patients with no other suitable treatment options4,16 (Supplementary material online, Table S1). Even though a plasmid encoding VEGF-A165 ‘Neovasculgen’,32 has been approved for clinical use for the treatment of PAD in Russia, a meta-analysis of RCTs with nearly 1500 treated patients found no consistent benefits of angiogenic gene therapy in PAD.33
RCTs have been able to show some vascular growth effects of both plasmid and Ad-mediated VEGF-A gene therapy in PAD patients but have not found a consistent association of the neovasculature with functional parameters34–36 (Supplementary material online, Table S1). Leaky non-stabile vessels have been blamed for the lack of functional benefits and current efforts have been directed towards improving functionality of the induced neovessels e.g. using a combination of VEGF-A and Angiopoetin (Ang)-1 (Table 1). However, two previous RCTs with plasmid Del-1 (DELTA-1-trial37) and adenoviral Hif-1a/VP16 (WALK-trial38) aiming for more physiological angiogenesis through effects on angiogenic transcription pathways found no beneficial effects on function. Functional improvements were neither found with FGF-1 plasmid in large RCTs TALISMAN39 and TAMARIS.23 A currently on-going trial is testing Sendaivirus40 mediated FGF-2 in PAD (Table 1). As a novel approach for PAD, a combination with surgical revascularization is tested in an ongoing KAT-PAD101-trial with intramuscular adenoviral VEGF-D given 1–2 days prior to vascular surgery to improve distal run-off after the operation (Table 1).
Although clinical trials in PAD patients have largely been unsuccessful, there are some auspicious signs. Significantly increased tissue oxygenation as well as improvements in peak walking time, ulcer healing and reduced amputation rates have been reported by several RCTs using HGF41–43 (Supplementary material online, Table S1). A current RCT is studying the effects of a NL003CLI-II-plasmid expressing two isoforms of HGF on ulcer healing in 200-patients (Table 1).
Key points learned from clinical trials
Patient selection
The meta-analysis of PAD trials showed no difference in the trial results according to disease severity (claudication vs critical ischemia).33 However, the patients may also differ in their comorbidities, such as hypertension or hypercholesterolemia as well as ongoing pharmacological treatments which could have unpredictable effects on vascular growth. Thus, in future trials, patient groups should be better categorized and patient cohorts of responders and non-responders should be studied in more detail to identify factors and biomarkers affecting treatment responsiveness. Knowledge of endogenous levels of vascular growth-associated factors might also be valuable background information.44
Gene delivery
A delivery method which provides effective gene transfer with the capability of global transduction of the treated organ with minimal off-target gene expression would be desired. Intravascular delivery may be limited because of multiple arterial occlusions preventing delivery of the gene to the ischemic areas. At the same time, a considerable proportion of the therapeutic construct usually enters systemic circulation being exposed to the immune system and affecting non-target tissues. Although highly important for targeted gene delivery into the myocardium,10,45 catheter-based approaches even with electromechanical mapping are less specific than direct intramuscular injections to lower limb muscles or intramyocardial delivery during cardiac surgery with an arrested heart, providing better control for the procedure.
Vectors
Regarding its significance, it is surprising how few clinical trials have actually tried to demonstrate adequate gene transfer efficiency in human target tissues.46,47 To achieve consistent positive results, vector development would be essential in targeting gene expression only to treated tissues, escaping the immune system and maintaining long enough transgene expression for biological effects to take place.48,49 Clinically often used plasmids have low immunogenicity but suffer from low transduction efficacy. Adenoviruses cause strong but transient gene expression that may be limited by neutralizing antibodies in some individuals. For longer and more stable gene expression, AAVs and genome-integrating lentiviruses may be considered with AAVs offering also serotype-specific tissue targeting.5 However, it is still debated what is sufficient and long enough gene expression for each particular indication. Past clinical trials have not clearly documented whether the lack of therapeutic effects has been due to the low gene transfer efficiency, insufficient biological availability or low bioactivity of the gene product (Figure 3). Thus, vectors allowing monitoring and regulation of the transgene expression in the target tissues should be developed.
Endpoints
Clinical trial design with appropriate endpoints is of critical importance. Claiming therapeutic effects without demonstration of the presence of the therapeutic protein should be avoided (Figure 3). It would be essential to design and validate surrogate markers for both transduction efficacy and biological activity of the gene product. Objective surrogate endpoint measurements, such as PET, MRI, ECG and ultrasound should be favoured instead of subjective or unspecific measures, such as exercise testing or quality of life questionnaires. Also, the sensitivity of the methods employed should be re-evaluated as for example SPECT is not able to detect angiogenesis in the capillary level vessels. It may also be questioned whether endpoints like overall mortality, can reliably capture potentially significant treatment effects, such as number of hospital admissions or additional interventions in chronically ill aged individuals.
General conclusions and visions for the future
On the basis of the clinical trials in angiogenic gene therapy, there seems to be a clear disconnect between promising preclinical results and disappointments in RCTs.5,33,44 Signs of improved vascularity have been detected after angiogenic gene delivery in some RCTs suggesting efficient gene delivery and biological activity of the transgenes11,34 but translation to functional benefits in the patients lacks convincing evidence (Supplementary material online, Table S1). A re-evaluation is thus needed.
While development of gene delivery vectors is still needed to improve gene transfer efficacy and gene expression in human tissues, the concept of therapeutic vascular growth in the treatment of CVD is evolving. Affecting mainly the capillary level, angiogenic gene therapy might not be sufficient alone to promote revascularization in the scale of a human as compared to a laboratory mouse.6,44 Instead, it might locally enhance distal run-off after surgical or radiological revascularization procedures or improve outcome of incomplete revascularization. Induction of lymphatic growth, to reduce revascularization- and angiogenesis-related oedema, might also be beneficial.50
Additionally, as the recovery potential in animal models may differ significantly from that in the human patients,44 the enhanced capillary level vascularity might not be enough to improve recovery of the severely affected and even necrotic human tissues. The mitogenic and myogenic properties of e.g. HGF working besides angiogenesis might help to restore function and could explain some positive clinical results. Synergy with regenerative gene or cell therapies should also be considered.
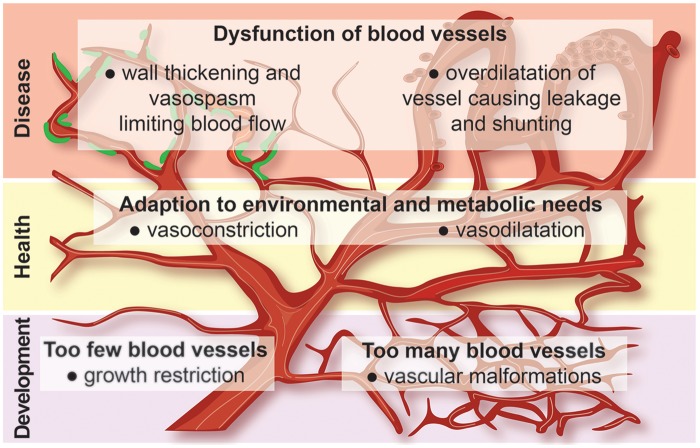
It may also be argued that if tissue ischemia is not cured with angiogenesis, maybe the clinical problem is then not the lack of angiogenesis. Hypoxia initiated vascular growth takes place already in the very early phases of embryonal development and continues its dynamic role as part of the body’s physiological and pathological responses, e.g. in wound healing, throughout life. The dysfunction of blood vessels that is related to different disease conditions (Figure 4) may still possess many unidentified features that could help us to develop novel therapies for CVD. But for establishment of new treatments, preclinical models need to more accurately resemble the chronic nature of human diseases in order to better evaluate the efficacy of novel treatments in humans.44
Key points
Comorbidities and pharmacological treatments might have unpredictable effects on vascular growth. → Patient cohorts of responders and non-responders should be studied to identify those benefitting from the treatments.
Gene transfer should yield high transduction efficiency in the target tissue without off-target side-effects. →Targeted and regulated vectors need to be developed.
Optimal duration and level of gene expression in angiogenic therapies has not yet been established. →Biological availability as well as bioactivity of the gene product need to be studied in more detail in future trials.
Subjective measures, such as exercise testing, may suffer from a significant placebo effect. → Objective surrogate markers need to be developed and validated to measure treatment effects.
Angiogenic gene therapy alone might not be sufficient to promote revascularization in large muscle areas. →Combination with established surgical or catheter-based revascularization procedures should be evaluated. Also, the growth of the whole vascular tree including lymphatics (i.e. therapeutic vascular growth) should be explored.
Increasing capillary level vascularity might not be enough to enhance recovery in severely affected and even necrotic tissues. →Multifunctional growth factors and combination with regenerative therapies should be evaluated.
Figure 4.
The dynamic nature of the vasculature in health, disease and development.
Supplementary material
Supplementary material is available at European Heart Journal online.
Supplementary Material
Acknowledgements
We acknowledge the Gene Therapy Resource Program (GTRP).
Funding
This study was supported by Finnish Academy and Kuopio University Hospital, and by the National Institutes of Health (grant 7-R01 HL083078-10).
Conflict of interest: none declared.
References
- 1. Mozaffarian D, Benjamin EJ, Go AS, Arnett DK, Blaha MJ, Cushman M, de Ferranti S, Després JP, Fullerton HJ, Howard VJ, Huffman MD, Judd SE, Kissela BM, Lackland DT, Lichtman JH, Lisabeth LD, Liu S, Mackey RH, Matchar DB, McGuire DK, Mohler ER, Moy CS, Muntner P, Mussolino ME, Nasir K, Neumar RW, Nichol G, Palaniappan L, Pandey DK, Reeves MJ, Rodriguez CJ, Sorlie PD, Stein J, Towfighi A, Turan TN, Virani SS, Willey JZ, Woo D, Yeh RW, Turner MB.. American heart association statistics committee and stroke statistics subcommittee. heart disease and stroke statistics - 2016 Update. Circulation 2015;131:e29–322. [DOI] [PubMed] [Google Scholar]
- 2. Townsend N, Nichols M, Scarborough P, Rayner M.. Cardiovascular disease in Europe - Epidemiological update 2015. Eur. Heart J 2015;36:2696–2705. [DOI] [PubMed] [Google Scholar]
- 3. Ferrara N, Alitalo K.. Clinical applications of angiogenic growth factors and their inhibitors. Nat Med 1999;5:1359–1364. [DOI] [PubMed] [Google Scholar]
- 4. Yla-Herttuala S, Rissanen TT, Vajanto I, Hartikainen J.. Vascular endothelial growth factors: biology and current status of clinical applications in cardiovascular medicine. J Am Coll Cardiol 2007;49:1015–1026. [DOI] [PubMed] [Google Scholar]
- 5. Rissanen TT, Yla-Herttuala S.. Current status of cardiovascular gene therapy. Mol Ther 2007;15:1233–1247. [DOI] [PubMed] [Google Scholar]
- 6. Markkanen JE, Rissanen TT, Kivelä A, Ylä-Herttuala S.. Growth factor-induced therapeutic angiogenesis and arteriogenesis in the heart–gene therapy. Cardiovasc Res 2005;65:656–664. [DOI] [PubMed] [Google Scholar]
- 7. Henry TD, Satran D, Jolicoeur EM.. Treatment of refractory angina in patients not suitable for revascularization. Nat Rev Cardiol 2014;11:78–95. [DOI] [PubMed] [Google Scholar]
- 8. Henry TD, Satran D, Hodges JS, Johnson RK, Poulose AK, Campbell AR, Garberich RF, Bart BA, Olson RE, Boisjolie CR, Harvey KL, Arndt TL, Traverse JH.. Long-term survival in patients with refractory angina. Eur Heart J 2013;34:2683–2688. [DOI] [PubMed] [Google Scholar]
- 9. Naldini L. Gene therapy returns to centre stage. Nature 2015;526:351–360. [DOI] [PubMed] [Google Scholar]
- 10. Kastrup J, Jorgensen E, Ruck A, Tagil K, Glogar D, Ruzyllo W, Botker HE, Dudek D, Drvota V, Hesse B, Thuesen L, Blomberg P, Gyongyosi M, Sylven C.. Direct intramyocardial plasmid vascular endothelial growth factor-A165 gene therapy in patients with stable severe angina pectoris A randomized double-blind placebo-controlled study: the Euroinject One trial. J Am CollCardiol 2005;45:982–988. [DOI] [PubMed] [Google Scholar]
- 11. Hedman M, Hartikainen J, Syvanne M, Stjernvall J, Hedman A, Kivela A, Vanninen E, Mussalo H, Kauppila E, Simula S, Narvanen O, Rantala A, Peuhkurinen K, Nieminen MS, Laakso M, Yla-Herttuala S.. Safety and feasibility of catheter-based local intracoronary vascular endothelial growth factor gene transfer in the prevention of postangioplasty and in-stent restenosis and in the treatment of chronic myocardial ischemia: phase II results of the Kuopio. Circulation 2003;107:2677–2683. [DOI] [PubMed] [Google Scholar]
- 12. Stewart DJ, Kutryk MJB, Fitchett D, Freeman M, Camack N, Su Y, Siega AD, Bilodeau L, Burton JR, Proulx G, Radhakrishnan S.. VEGF gene therapy fails to improve perfusion of ischemic myocardium in patients with advanced coronary disease: results of the NORTHERN trial. Mol Ther 2009;17:1109–1115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Rosengart TK, Lee LY, Patel SR, Sanborn TA, Parikh M, Bergman GW, Hachamovitch R, Szulc M, Kligfield PD, Okin PM, Hahn RT, Devereux RB, Post MR, Hackett NR, Foster T, Grasso TM, Lesser ML, Isom OW, Crystal RG.. Angiogenesis gene therapy: phase I assessment of direct intramyocardial administration of an adenovirus vector expressing VEGF121 cDNA to individuals with clinically significant severe coronary artery disease. Circulation 1999;100:468–474. [DOI] [PubMed] [Google Scholar]
- 14. Hedman M, Muona K, Hedman A, Kivelä A, Syvänne M, Eränen J, Rantala A, Stjernvall J, Nieminen MS, Hartikainen J, Ylä-Herttuala S.. Eight-year safety follow-up of coronary artery disease patients after local intracoronary VEGF gene transfer. Gene Ther 2009;16:629–634. [DOI] [PubMed] [Google Scholar]
- 15. Rosengart TKBishawi MM, Halbreiner MSHalbreiner MS, Fakhoury M, Finnin EHollmann C, Shroyer AL, Crystal RG. Long-term follow-up of a phase 1 trial of angiogenic gene therapy using direct intramyocardial administration of an adenoviral vector expressing the VEGF121 cDNA for the treatment of diffuse coronary artery disease. Hum Gene Ther 2013;24:203–208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Muona K, Makinen K, Hedman M, Manninen H, Yla-Herttuala S.. 10-year safety follow-up in patients with local VEGF gene transfer to ischemic lower limb. Gene Ther 2012;19:392–395. [DOI] [PubMed] [Google Scholar]
- 17. Hassinen I, Hartikainen J, Hedman A, Kivelä A, Saraste A, Knuuti J, Husso M, Mussalo H, Hedman M, Toivonen P, Heikura T, Ylä-Herttuala S, Adenoviral intramyocardial VEGF-D transfer increases myocardial perfusion in refractory angina patients. Circulation 2015;132:A11987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Kaminsky SM, Quach L, Chen S, Pierre-Destine L, Graaf B, Van De, Monette S, Rosenberg JB, Sondhi D, Hackett NR, Crystal RG.. Safety of direct administration of ADVEGFALL6A+, an adenovirus vector expressing a CDNA/genomic hybrid of human vascular endothelial growth factors, to the ischemic myocardium of rats. Mol Ther 2013;21:S143.. [DOI] [PubMed] [Google Scholar]
- 19. Henry TD, Grines CL, Watkins MW, Dib N, Barbeau G, Moreadith R, Andrasfay T, Engler RL.. Effects of Ad5FGF-4 in patients with angina. An analysis of pooled data from the AGENT-3 and AGENT-4 Trials. J Am Coll Cardiol 2007;50:1038–1046. [DOI] [PubMed] [Google Scholar]
- 20. Kaski JC, Consuegra-Sanchez L.. Evaluation of ASPIRE trial: a Phase III pivotal registration trial, using intracoronary administration of Generx (Ad5FGF4) to treat patients with recurrent angina pectoris. Expert Opin Biol Ther 2013;13:1749–1753. [DOI] [PubMed] [Google Scholar]
- 21. Yang ZJ, Zhang YR, Chen B, Zhang SL, Jia EZ, Wang LS, Zhu TB, Li CJ, Wang H, Huang J, Cao KJ, Ma WZ, Wu B, Wang LS, Wu CT.. Phase i clinical trial on intracoronary administration of Ad-hHGF treating severe coronary artery disease. Mol Biol Rep 2009;36:1323–1329. [DOI] [PubMed] [Google Scholar]
- 22. Kim JS, Hwang HY, Cho KR, Park EA, Lee W, Paeng JC, Lee DS, Kim HK, Sohn DW, Kim KB.. Intramyocardial transfer of hepatocyte growth factor as an adjunct to CABG: phase I clinical study. Gene Ther 2013;20:717–722. [DOI] [PubMed] [Google Scholar]
- 23. Belch J, Hiatt WR, Baumgartner I, Driver IV, Nikol S, Norgren L, Belle EV.. Effect of fibroblast growth factor NV1FGF on amputation and death: A randomised placebo-controlled trial of gene therapy in critical limb ischaemia. Lancet 2011;377:1929–1937. [DOI] [PubMed] [Google Scholar]
- 24. Kilian EG, Sadoni S, Vicol C, Kelly R, Hulst K. V, Schwaiger M, Kupatt C, Boekstegers P, Pillai R, Channon K, Hetzer R, Reichart B.. Myocardial transfection of hypoxia inducible factor-1alpha via an adenoviral vector during coronary artery bypass grafting. - A multicenter phase I and safety study. Circ J 2010;74:916–924. [DOI] [PubMed] [Google Scholar]
- 25. Greenberg B, Butler J, Felker GM, Ponikowski P, Voors AA, Desai AS, Barnard D, Bouchard A, Jaski B, Lyon AR, Pogoda JM, Rudy JJ, Zsebo KM.. Calcium upregulation by percutaneous administration of gene therapy in patients with cardiac disease (CUPID 2): A randomised, multinational, double-blind, placebo-controlled, phase 2b trial. Lancet 2016;387:1178–1186. [DOI] [PubMed] [Google Scholar]
- 26. Chung ES, Miller L, Patel AN, Anderson RD, Mendelsohn FO, Traverse J, Silver KH, Shin J, Ewald G, Farr MJ, Anwaruddin S, Plat F, Fisher SJ, AuWerter AT, Pastore JM, Aras R, Penn MS.. Changes in ventricular remodelling and clinical status during the year following a single administration of stromal cell-derived factor-1 non-viral gene therapy in chronic ischaemic heart failure patients: the STOP-HF randomized Phase II trial. Eur Heart J 2015;36:2228–2238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Halonen PJ, Nurro J, Kuivanen A, Ylä-Herttuala S.. Current gene therapy trials for vascular diseases. Expert Opin Biol Ther 2014;14:327–336. [DOI] [PubMed] [Google Scholar]
- 28. Karpanen T, Bry M, Ollila HM, Seppanen-Laakso T, Liimatta E, Leskinen H, Kivela R, Helkamaa T, Merentie M, Jeltsch M, Paavonen K, Andersson LC, Mervaala E, Hassinen IE, Yla-Herttuala S, Orešič M, Alitalo K.. Overexpression of vascular endothelial growth factor-B in mouse heart alters cardiac lipid metabolism and induces myocardial hypertrophy. Circ Res 2008;103:1018–1026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Lahteenvuo JE, Lahteenvuo MT, Kivela A, Rosenlew C, Falkevall A, Klar J, Heikura T, Rissanen TT, Vahakangas E, Korpisalo P, Enholm B, Carmeliet P, Alitalo K, Eriksson U, Yla-Herttuala S.. Vascular endothelial growth factor-B induces myocardium-specific angiogenesis and arteriogenesis via vascular endothelial growth factor receptor-1- and neuropilin receptor-1-dependent mechanisms. Circulation 2009;119:845–856. [DOI] [PubMed] [Google Scholar]
- 30. Criqui MH, Aboyans V.. Epidemiology of Peripheral Artery Disease. Circ Res 2015;116:1509–1526. [DOI] [PubMed] [Google Scholar]
- 31. Tendera M, Aboyans V, Bartelink M-L, Baumgartner I, Clément D, Collet J-P, Cremonesi A, Carlo MD, Erbel R, Fowkes FGR, Heras M, Kownator S, Minar E, Ostergren J, Poldermans D, Riambau V, Roffi M, Röther J, Sievert H, Sambeek M, Van Zeller T, Bax J, Auricchio A, Baumgartner H, Ceconi C, Dean V, Deaton C, Fagard R, Funck-Brentano C, Hasdai D. et al. ESC Guidelines on the diagnosis and treatment of peripheral artery diseases. Eur Heart J 2011;32: 2851–2906. [DOI] [PubMed] [Google Scholar]
- 32. Deev RV, Bozo IY, Mzhavanadze ND, Voronov DA, Gavrilenko AV, Chervyakov YV, Staroverov IN, Kalinin RE, Shvalb PG, Isaev AA.. pCMV-vegf165 intramuscular gene transfer is an effective method of treatment for patients with chronic lower limb ischemia. J Cardiovasc Pharmacol Ther 2015;20:473–482. [DOI] [PubMed] [Google Scholar]
- 33. Hammer A, Steiner S.. Gene therapy for therapeutic angiogenesis in peripheral arterial disease - a systematic review and meta-analysis of randomized, controlled trials. Vasa 2013;42:331–339. [DOI] [PubMed] [Google Scholar]
- 34. Makinen K, Manninen H, Hedman M, Matsi P, Mussalo H, Alhava E, Yla-Herttuala S.. Increased vascularity detected by digital subtraction angiography after VEGF gene transfer to human lower limb artery: a randomized, placebo-controlled, double-blinded phase II study. Mol Ther 2002;6:127–133. [DOI] [PubMed] [Google Scholar]
- 35. Kusumanto YH, Weel V. V, Mulder NH, Smit AJ, Dungen JJAM. V D, Hooymans JMM, Sluiter WJ, Tio RA, Quax PHA, Gans ROB, Dullaart RPF, Hospers GAP.. Treatment with intramuscular vascular endothelial growth factor gene compared with placebo for patients with diabetes mellitus and critical limb ischemia: a double-blind randomized trial. Hum Gene Ther 2006;17:683–691. [DOI] [PubMed] [Google Scholar]
- 36. Rajagopalan S, Mohler IIIER, Lederman RJ, Mendelsohn FO, Saucedo JF, Goldman CK, Blebea J, Macko J, Kessler PD, Rasmussen HS, Annex BH.. Regional angiogenesis with vascular endothelial growth factor in peripheral arterial disease: a phase II randomized, double-blind, controlled study of adenoviral delivery of vascular endothelial growth factor 121 in patients with disabling intermittent cl. Circulation 2003;108:1933–1938. [DOI] [PubMed] [Google Scholar]
- 37. Grossman PM, Mendelsohn F, Henry TD, Hermiller JB, Litt M, Saucedo JF, Weiss RJ, Kandzari DE, Kleiman N, Anderson RD, Gottlieb D, Karlsberg R, Snell J, Rocha-Singh K.. Results from a phase II multicenter, double-blind placebo-controlled study of Del-1 (VLTS-589) for intermittent claudication in subjects with peripheral arterial disease. Am Heart J 2007;153:874–880. [DOI] [PubMed] [Google Scholar]
- 38. Creager MA, Olin JW, Belch JJF, Moneta GL, Henry TD, Rajagopalan S, Annex BH, Hiatt WR.. Effect of hypoxia-inducible factor-1alpha gene therapy on walking performance in patients with intermittent claudication. Circulation 2011;124: 1765–1773. [DOI] [PubMed] [Google Scholar]
- 39. Nikol S, Baumgartner I, Belle EV, Diehm C, Visoná A, Capogrossi MC, Ferreira-Maldent N, Gallino A, Wyatt MG, Wijesinghe LD, Fusari M, Stephan D, Emmerich J, Pompilio G, Vermassen F, Pham E, Grek V, Coleman M, Meyer F.. Therapeutic angiogenesis with intramuscular NV1FGF improves amputation-free survival in patients with critical limb ischemia. Mol Ther 2008;16:972–978. [DOI] [PubMed] [Google Scholar]
- 40. Yonemitsu Y, Matsumoto T, Maehara Y, Gene Therapy for peripheral arterial disease using Sendai Virus vector: from preclinical studies to the Phase I/IIa clinical trial In: Nagai, ed. Sendai Virus Vector. Japan: Springer; 2013. p185–199. [Google Scholar]
- 41. Powell RJ, Simons M, Mendelsohn FO, Daniel G, Henry TD, Koga M, Morishita R, Annex BH.. Results of a double-blind, placebo-controlled study to assess the safety of intramuscular injection of hepatocyte growth factor plasmid to improve limb perfusion in patients with critical limb ischemia. Circulation 2008;118:58–65. [DOI] [PubMed] [Google Scholar]
- 42. Shigematsu H, Yasuda K, Iwai T, Sasajima T, Ishimaru S, Ohashi Y, Yamaguchi T, Ogihara T, Morishita R.. Randomized, double-blind, placebo-controlled clinical trial of hepatocyte growth factor plasmid for critical limb ischemia. Gene Ther 2010;17:1152–1161. [DOI] [PubMed] [Google Scholar]
- 43. Anghel A, Taranu G, Seclaman E, Rata A, Tamas L, Moldovan H, Ursoniu S, Samoila C, Ionac M, Popa-Wagner A.. Safety of vascular endothelial and hepatocyte growth factor gene therapy in patients with critical limb ischemia. Curr Neurovasc Res 2011;8:183–189. [DOI] [PubMed] [Google Scholar]
- 44. Dragneva G, Korpisalo P, Yla-Herttuala S.. Promoting blood vessel growth in ischemic diseases: challenges in translating preclinical potential into clinical success. Dis Model Mech 2013;6:312–322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Losordo DW, Vale PR, Hendel RC, Milliken CE, Fortuin FD, Cummings N, Schatz RA, Asahara T, Isner JM, Kuntz RE.. Phase 1/2 placebo-controlled, double-blind, dose-escalating trial of myocardial vascular endothelial growth factor 2 gene transfer by catheter delivery in patients with chronic myocardial ischemia. Circulation 2002;105:2012–2018. [DOI] [PubMed] [Google Scholar]
- 46. Laitinen M, Makinen K, Manninen H, Matsi P, Kossila M, Agrawal RS, Pakkanen T, Luoma JS, Viita H, Hartikainen J, Alhava E, Laakso M, Yla-Herttuala S.. Adenovirus-mediated gene transfer to lower limb artery of patients with chronic critical leg ischemia. Hum Gene Ther 1998;9:1481–1486. [DOI] [PubMed] [Google Scholar]
- 47. Puumalainen AM, Vapalahti M, Agrawal RS, Kossila M, Laukkanen J, Lehtolainen P, Viita H, Paljarvi L, Vanninen R, Yla-Herttuala S.. Beta-galactosidase gene transfer to human malignant glioma in vivo using replication-deficient retroviruses and adenoviruses. Hum Gene Ther 1998;9:1769–1774. [DOI] [PubMed] [Google Scholar]
- 48. Hedman M, Hartikainen J, Ylä-Herttuala S.. Progress and prospects: hurdles to cardiovascular gene therapy clinical trials. Gene Ther 2011;18:743–749. [DOI] [PubMed] [Google Scholar]
- 49. Katz MG, Fargnoli AS, Pritchette LA, Bridges CR.. Gene delivery technologies for cardiac applications. Gene Ther 2012;19:659–669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Henri O, Pouehe C, Houssari M, Galas L, Nicol L, Edwards-Lévy F, Henry J-P, Dumesnil A, Boukhalfa I, Banquet S, Schapman D, Thuillez C, Richard V, Mulder P, Brakenhielm E.. Selective stimulation of cardiac lymphangiogenesis reduces myocardial edema and fibrosis leading to improved cardiac function following myocardial infarction. Circulation 2016;133:1484–1497. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.