Abstract
Systemic lupus erythematosus (SLE), an autoimmune disease of unknown etiology, is characterized by the production of autoantibodies and end-organ damage. Lupus nephritis affects up to 70% of patients with SLE and is the most critical predictor of morbidity and mortality. The immunopathogenesis of SLE is complex and most clinical trials of biologics targeting immune cells or their mediators have failed to show efficacy in SLE patients. It has therefore become increasingly clear that additional, local factors give rise to the inflammation and organ damage. In this review, we describe recent advances in the role of renal resident cells, including podocytes, mesangial cells, and epithelial cells, in the pathogenesis of lupus nephritis.
Keywords: Systemic lupus erythematosus, Autoimmune diseases, Lupus nephritis, Podocytes
INTRODUCTION
Systemic lupus erythematosus (SLE) is an autoimmune disease that affects multiple organs and has significant mortality. Its etiology is multifactorial and includes both environmental and genetic influences. Despite considerable progress in research into the pathogenesis of SLE, through the use of genetic variant identification, murine models, gene expression studies, and epigenetic analysis, the exact pathogenesis of SLE remains poorly understood [1,2]. Nevertheless, contributions of various immunologic abnormalities to the pathogenesis of SLE have been confirmed, including the defective clearance of apoptotic cells, a loss of tolerance to self-antigens, aberrant activation of T- and B-cells, altered cytokine profiles, such as those of type I interferon (IFN), the involvement of neutrophilic extracellular traps, and pathogenic autoantibody production [3,4]. The serologic hallmark of SLE is the production of autoantibodies, which are mostly directed against nuclear antigens, including double-stranded DNA (dsDNA), small nuclear ribonucleoproteins, and nucleosomes, but also against antigens located in the cytoplasm, on the cell surface, and secreted by the cell [1,5].
Renal involvement, characterized by immune complex deposition, inflammation, and scarring of the glomeruli and interstitium, is the most common and severe clinical manifestation of SLE. However, only a few autoantibodies have been found to specifically contribute to the disease-related injury seen in mice and humans, including anti-blood cell antibodies causing cytopenia, anti-dsDNA antibodies causing nephritis, and anti-phospholipid antibodies causing fetal distress [6]. From a clinical perspective, most trials of biologics targeting immune cells have failed to show efficacy in SLE patients. For example, rituximab, a genetically engineered chimeric monoclonal IgG1-κ antibody targeting CD20 on B-cells, was expected to show efficacy in SLE patients because it directly targets B-cells, which are responsible for autoantibody production. However, two large phase III clinical trials did not verify the clinical efficacy of rituximab in patients with non-renal lupus and lupus nephritis [7,8]. In addition, the use of congenic mice has shown that distinct chromosomal regions determine the development of autoimmunity and chronic kidney damage [9]. Another study demonstrated that SLE patients in clinical remission continue to produce elevated levels of self-reactive and polyreactive antibodies [10]. These findings suggest that autoimmunity and tissue damage are independent processes. Thus, to achieve a comprehensive understanding of the pathogenesis of lupus, greater attention will need to be paid to tissue resident cells and the tissue-specific factors involved in the tissue damage process.
Recent evidence supports a direct role for renal resident cells, including podocytes, mesangial cells, and tubular epithelial cells, in the development of lupus nephritis. In this review, we provide an overview of the role of renal resident cells, and especially podocytes, in the pathogenesis of lupus nephritis.
THE ROLE OF PODOCYTES
Podocytes are highly specialized cells that reside on the visceral side of Bowman’s capsule and wrap around glomerular capillaries. They are essential components of the glomerular filtration apparatus and are critical for the maintenance of renal function [11]. By expressing markers such as synaptopodin, nephrin, podocin, and Wilms’ tumor protein, podocytes play an important role in maintaining the integrity of the glomerular filtration barrier. Animal and human studies have demonstrated a strong correlation between defects in podocyte anchoring and several glomerular abnormalities, verifying the role of podocytes in the onset of renal diseases. In addition, extensive human genetic studies have indicated that monogenic mutations in podocyte proteins are associated with specific renal pathologic phenotypes. This capacity of podocyte injury or loss to initiate glomerular damage may be related to the development of chronic kidney disease [12]. Disruption of the glomerular filtration barrier allows passage of large molecules through the glomerulus, which, together with apoptotic debris, can activate tubular interstitial cells and resident dendritic cells to initiate the pathways leading to chronic inflammatory disease [13].
Immune cell-like features of glomerular podocytes
In addition to helping to maintain the glomerular filtration barrier, podocytes exhibit the properties of immune cells (Table 1) [14-25] and may be involved in adaptive immunity. For example, podocytes express CD80, which costimulates lymphocytes; mice lacking CD80 are protected from lipopolysaccharide (LPS)-induced nephrotic syndrome, suggesting a link between podocyte CD80 expression and proteinuria [14]. Podocytes also express major histocompatibility complex (MHC) class II [15] and neonatal Fc receptor, a receptor protein of antigen-presenting lymphocytes [16]. Goldwich et al. [26] reported that deleting MHC class II exclusively on podocytes prevents the induction of experimental anti-glomerular basement membrane nephritis. Podocytes ingest the soluble antigens that activate CD4+ T cells and cross-present exogenous antigen on MHC class I molecules to CD8+ T cells, which suggests that podocytes participate in the antigen-specific activation of adaptive immune responses [26].
Table 1.
Immune molecules present in podocytes
| Name | Function | Sources | References |
|---|---|---|---|
| CD80 (B7-1) | Costimulatory molecule | Human, mouse | [14] |
| CD86 | Costimulatory molecule | Human | [22] |
| CaMK4 | T-cell signaling molecule | Human | [22] |
| MHC class II | Antigen presentation | Rat | [15] |
| FcRn | Receptor protein in APC | Human, mouse | [16,22] |
| TLRs | PRR | Human, mouse | [17,24] |
| RAGE | PRR | Human, mouse | [18,19] |
| NLRP3 inflammasome | To switch on the inflammatory process | Human, mouse | [21,23] |
| IL-23 | Proinflammatory cytokine | Mouse | [20] |
| IL-6 | Proinflammatory cytokine | Mouse | [25] |
CaMK4, calcium/calmodulin-dependent protein kinase IV; MHC, major histocompatibility complex; FcRn, neonatal Fc receptor; APC, antigen-presenting cell; TLR, Toll-like receptor; PRR, pattern-recognition receptor; RAGE, receptor for advanced glycation endproducts; NLRP3, Nod-like receptor protein 3; IL, interleukin.
Podocytes also express diverse genes required for the innate immune response, including those encoding the pattern recognition receptors (PRRs) that sense both pathogen-associated molecular patterns and damage-associated molecular patterns (DAMPs). Toll-like receptors (TLRs) are the most well-known PRRs. Isolated glomeruli express TLR1–TLR9 and TLR11 mRNA, with the highest levels of expression being those of TLR3 and TLR4 [17]. In vitro treatment of cells with TLR4 ligands, such as LPS and fibrinogen, promotes a marked induction of chemokines [17]. The receptor for advanced glycation endproducts (RAGE) is a PRR involved in several innate immune responses. RAGE can use DAMPs, including advanced glycation endproducts and high-mobility group box protein 1, as its ligands. RAGE is expressed in podocytes and up-regulated in both human and mouse glomerular diseases [18]. In a murine model of adriamycin-induced glomerulosclerosis, RAGE was shown to mediate podocyte injury [19].
In addition, podocytes produce the proinflammatory cytokine interleukin 23 (IL-23) [20], which is a potent stimulator of renal inflammation. IL-23 promotes the differentiation of T-cells into Th17 cells, γδ T-cells, and double-negative T-cells [27]. The resulting stimulation of neutrophil infiltration into the tubular interstitium leads to the establishment of chronic inflammation.
The role of podocytes in lupus nephritis
Proteinuria is the most important feature of lupus nephritis and its development is associated with podocyte dysfunction. Therefore, the development of lupus nephritis likely accompanies podocyte injury. Indeed, podocyte injury is common in patients with lupus nephritis, as shown in a large cohort study of patients with renal-biopsy-proven lupus nephritis [28].
Several lines of evidence support the role of podocytes in the pathogenesis of lupus nephritis [14,21-24,29,30]. Podocyte expression of costimulatory molecules, such as CD80, correlates with the severity of human lupus nephritis [14]. TLR9 is up-regulated within the glomeruli of patients with lupus nephritis, but not in normal kidneys [24]. Ichinose et al. [22] showed that immunoglobulin G (IgG) from lupus nephritis patients enters podocytes, where it up-regulates calcium/calmodulin-dependent protein kinase IV (CaMK4), which is followed by the increased expression of genes linked to podocyte damage and T-cell activation. This finding suggests the targeted inhibition of CaMK4 in podocytes as a therapeutic strategy in lupus nephritis [22].
The major component of inflammasomes, Nod-like receptor protein 3 (NLRP3), the apoptosis-associated protein ASC (apoptosis-associated speck-like protein containing a CARD), and caspase 1, are also expressed in podocytes [23]. NLRP3-expressing inflammasomes are activated in the podocytes of lupus-prone mice, and in those of patients with lupus nephritis. In the former, the inhibition of NLRP3 ameliorates proteinuria, renal histologic lesions, and podocyte foot process effacement [21], suggesting that NLRP3 activation is involved in the pathogenesis of podocyte injury and in the development of proteinuria in lupus nephritis.
THE ROLE OF MESANGIAL CELLS
Mesangial cells are specialized cells in the kidney that make up the mesangium of the glomerulus. Together with the mesangial matrix, they form the vascular pole of the renal corpuscle. The main function of mesangial cells is to remove trapped residues and aggregated protein from the basement membrane; thus, keeping it and the glomerular filtration apparatus free of debris [31].
Mesangial cells have also been implicated in the pathogenesis of lupus nephritis [32]. Studies have shown that mesangial cells express TLRs [33,34], and when stimulated with TLR-3 ligand they produce type I IFN [33], the major cytokine in the development of SLE. The production of type I IFN by renal resident cells, including mesangial cells, may aggravate autoimmune kidney diseases [33,35]. Anti-dsDNA antibodies are essential to the diagnosis of SLE, and their levels correlate with disease activity. Yung et al. [36,37] demonstrated that anti-dsDNA antibodies bind to mesangial cells and trigger downstream inflammatory and fibrotic pathways, including the protein kinase C (PKC) and mitogen-activated protein kinase (MAPK) signaling pathways, in addition to increasing both the secretion of proinflammatory cytokines and matrix protein deposition, all of which contribute to pathologic processes in the renal parenchyma.
Mesangial cells produce IL-6, and probably additional cytokines, which could independently contribute to glomerulonephritis. Mesangial cells lacking CaMK4 produce decreased amounts of IL-6 [38]. An early study showed that IL-6 on its own can drive the development of glomerulonephritis [39,40].
THE ROLE OF RENAL TUBULAR EPITHELIAL CELLS
Renal tubular epithelial cells are another type of renal resident cell likely to be involved in the pathophysiology of lupus nephritis. For example, renal tubular epithelial cells produce pathogenic cytokines, including type I IFN [41] and B-cell activating factor (BAFF) [42], both of which are very important to the development of SLE. BAFF expression in the kidney of lupus-prone mice correlates with disease activity. In biopsies of patients with diffuse proliferative lupus nephritis, BAFF expression on tubular epithelial cells correlates with the histopathological activity index [42]. In addition, renal tubular epithelial cells from patients with lupus nephritis express the co-stimulatory molecule B7-H4, suggesting the ability of these cells to activate T-cells [43].
The incubation of renal tubular epithelial cells with human anti-dsDNA antibodies leads to the sequential up-regulation of tumor necrosis factor α, IL-1β, and IL-6 [44], indicating a contribution by these cells to inflammatory processes in the tubulointerstitium in lupus nephritis.
CONCLUSIONS
Conventional therapy for SLE has been based on non-specific immunosuppressants, which have limited clinical efficacy and cause severe adverse events. As our understanding of the immunopathogenesis of SLE has progressed, new classes of drugs and biologics that target immune cells in patients with this disease have been developed. However, their clinical efficacy is unsatisfactory and none of the biologics has proven to be effective in the treatment of lupus nephritis, a common manifestation and critical determinant in the prognosis of SLE patients.
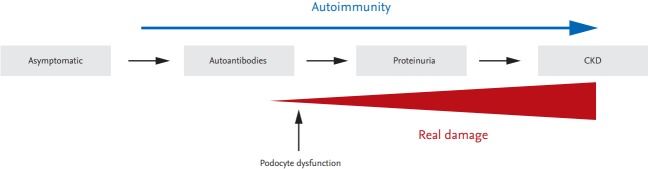
Given that autoimmunity can occur independently of tissue injury mechanisms, it is necessary to understand the process that alters resident cell function. Podocytes are an essential component of the glomerular filtration barrier and their dysfunction is directly connected with the initiation of glomerulonephritis. Therefore, the identification of a common intracellular pathway leading to both immune cell aberration and podocyte dysfunction will aid in the development of therapeutics targeting both immune cells and podocytes in a novel and effective therapeutic strategy (Fig. 1).
Figure 1.
Proposed model of the development of lupus nephritis. Autoimmunity persists from the asymptomatic stage to the stage marked by chronic kidney disease (CKD). Podocyte dysfunction ,may already occur at an early stage of the kidney damage process, ultimately leading to overt proteinuria and CKD. Identification of a common intracellular pathway that mediates not only aberrant immune cell activation related to autoimmunity but also podocyte dysfunction, will lead to a better understanding of the pathogenesis of lupus nephritis.
Acknowledgments
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education, ICT and Future Planning (2015R1A2A2A01003886).
Footnotes
No potential conflict of interest relevant to this article was reported.
REFERENCES
- 1.Tsokos GC. Systemic lupus erythematosus. N Engl J Med. 2011;365:2110–2121. doi: 10.1056/NEJMra1100359. [DOI] [PubMed] [Google Scholar]
- 2.Tsokos GC, Lo MS, Costa Reis P, Sullivan KE. New insights into the immunopathogenesis of systemic lupus erythematosus. Nat Rev Rheumatol. 2016;12:716–730. doi: 10.1038/nrrheum.2016.186. [DOI] [PubMed] [Google Scholar]
- 3.Kim JM, Park SH, Kim HY, Kwok SK. A plasmacytoid dendritic cells-type I interferon axis is critically implicated in the pathogenesis of systemic lupus erythematosus. Int J Mol Sci. 2015;16:14158–14170. doi: 10.3390/ijms160614158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Moulton VR, Suarez-Fueyo A, Meidan E, Li H, Mizui M, Tsokos GC. Pathogenesis of human systemic lupus erythematosus: a cellular perspective. Trends Mol Med. 2017;23:615–635. doi: 10.1016/j.molmed.2017.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Lisnevskaia L, Murphy G, Isenberg D. Systemic lupus erythematosus. Lancet. 2014;384:1878–1888. doi: 10.1016/S0140-6736(14)60128-8. [DOI] [PubMed] [Google Scholar]
- 6.Crispin JC, Liossis SN, Kis-Toth K, et al. Pathogenesis of human systemic lupus erythematosus: recent advances. Trends Mol Med. 2010;16:47–57. doi: 10.1016/j.molmed.2009.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Merrill JT, Neuwelt CM, Wallace DJ, et al. Efficacy and safety of rituximab in moderately-to-severely active systemic lupus erythematosus: the randomized, double- blind, phase II/III systemic lupus erythematosus evaluation of rituximab trial. Arthritis Rheum. 2010;62:222–233. doi: 10.1002/art.27233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rovin BH, Furie R, Latinis K, et al. Efficacy and safety of rituximab in patients with active proliferative lupus nephritis: the Lupus Nephritis Assessment with Rituximab study. Arthritis Rheum. 2012;64:1215–1226. doi: 10.1002/art.34359. [DOI] [PubMed] [Google Scholar]
- 9.Ge Y, Jiang C, Sung SS, et al. Cgnz1 allele confers kidney resistance to damage preventing progression of immune complex-mediated acute lupus glomerulonephritis. J Exp Med. 2013;210:2387–2401. doi: 10.1084/jem.20130731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Yurasov S, Tiller T, Tsuiji M, et al. Persistent expression of autoantibodies in SLE patients in remission. J Exp Med. 2006;203:2255–2261. doi: 10.1084/jem.20061446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Perico L, Conti S, Benigni A, Remuzzi G. Podocyte-actin dynamics in health and disease. Nat Rev Nephrol. 2016;12:692–710. doi: 10.1038/nrneph.2016.127. [DOI] [PubMed] [Google Scholar]
- 12.Xia H, Bao W, Shi S. Innate immune activity in glomerular podocytes. Front Immunol. 2017;8:122. doi: 10.3389/fimmu.2017.00122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kurts C, Panzer U, Anders HJ, Rees AJ. The immune system and kidney disease: basic concepts and clinical implications. Nat Rev Immunol. 2013;13:738–753. doi: 10.1038/nri3523. [DOI] [PubMed] [Google Scholar]
- 14.Reiser J, von Gersdorff G, Loos M, et al. Induction of B7-1 in podocytes is associated with nephrotic syndrome. J Clin Invest. 2004;113:1390–1397. doi: 10.1172/JCI20402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Coers W, Brouwer E, Vos JT, et al. Podocyte expression of MHC class I and II and intercellular adhesion molecule-1 (ICAM-1) in experimental pauci-immune crescentic glomerulonephritis. Clin Exp Immunol. 1994;98:279–286. doi: 10.1111/j.1365-2249.1994.tb06138.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Akilesh S, Huber TB, Wu H, et al. Podocytes use FcRn to clear IgG from the glomerular basement membrane. Proc Natl Acad Sci U S A. 2008;105:967–972. doi: 10.1073/pnas.0711515105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Banas MC, Banas B, Hudkins KL, et al. TLR4 links podocytes with the innate immune system to mediate glomerular injury. J Am Soc Nephrol. 2008;19:704–713. doi: 10.1681/ASN.2007040395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tanji N, Markowitz GS, Fu C, et al. Expression of advanced glycation end products and their cellular receptor RAGE in diabetic nephropathy and nondiabetic renal disease. J Am Soc Nephrol. 2000;11:1656–1666. doi: 10.1681/ASN.V1191656. [DOI] [PubMed] [Google Scholar]
- 19.Guo J, Ananthakrishnan R, Qu W, et al. RAGE mediates podocyte injury in adriamycin-induced glomerulosclerosis. J Am Soc Nephrol. 2008;19:961–972. doi: 10.1681/ASN.2007101109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Goto K, Kaneko Y, Sato Y, et al. Leptin deficiency down-regulates IL-23 production in glomerular podocytes resulting in an attenuated immune response in nephrotoxic serum nephritis. Int Immunol. 2016;28:197–208. doi: 10.1093/intimm/dxv067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Fu R, Guo C, Wang S, et al. Podocyte activation of NLRP3 inflammasomes contributes to the development of proteinuria in lupus nephritis. Arthritis Rheumatol. 2017;69:1636–1646. doi: 10.1002/art.40155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ichinose K, Ushigusa T, Nishino A, et al. Lupus nephritis IgG induction of calcium/calmodulin-dependent protein kinase IV expression in podocytes and alteration of their function. Arthritis Rheumatol. 2016;68:944–952. doi: 10.1002/art.39499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhang C, Boini KM, Xia M, et al. Activation of Nod-like receptor protein 3 inflammasomes turns on podocyte injury and glomerular sclerosis in hyperhomocysteinemia. Hypertension. 2012;60:154–162. doi: 10.1161/HYPERTENSIONAHA.111.189688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Papadimitraki ED, Tzardi M, Bertsias G, Sotsiou E, Boumpas DT. Glomerular expression of toll-like receptor-9 in lupus nephritis but not in normal kidneys: implications for the amplification of the inflammatory response. Lupus. 2009;18:831–835. doi: 10.1177/0961203309103054. [DOI] [PubMed] [Google Scholar]
- 25.Lee SJ, Borsting E, Decleves AE, Singh P, Cunard R. Podocytes express IL-6 and lipocalin 2/neutrophil gelatinase-associated lipocalin in lipopolysaccharide-induced acute glomerular injury. Nephron Exp Nephrol. 2012;121:e86–e96. doi: 10.1159/000345151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Goldwich A, Burkard M, Olke M, et al. Podocytes are nonhematopoietic professional antigen-presenting cells. J Am Soc Nephrol. 2013;24:906–916. doi: 10.1681/ASN.2012020133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Suarez-Fueyo A, Bradley SJ, Klatzmann D, Tsokos GC. T cells and autoimmune kidney disease. Nat Rev Nephrol. 2017;13:329–343. doi: 10.1038/nrneph.2017.34. [DOI] [PubMed] [Google Scholar]
- 28.Wang Y, Yu F, Song D, Wang SX, Zhao MH. Podocyte involvement in lupus nephritis based on the 2003 ISN/RPS system: a large cohort study from a single centre. Rheumatology (Oxford) 2014;53:1235–1244. doi: 10.1093/rheumatology/ket491. [DOI] [PubMed] [Google Scholar]
- 29.Ikuma D, Hiromura K, Kajiyama H, et al. The correlation of urinary podocytes and podocalyxin with histological features of lupus nephritis. Lupus. 2017 Jan 1; doi: 10.1177/0961203317734918. [Epub]. https://doi.org/10.1177/0961203317734918. [DOI] [PubMed] [Google Scholar]
- 30.Dos Santos M, Poletti PT, Milhoransa P, Monticielo OA, Veronese FV. Unraveling the podocyte injury in lupus nephritis: clinical and experimental approaches. Semin Arthritis Rheum. 2017;46:632–641. doi: 10.1016/j.semarthrit.2016.10.005. [DOI] [PubMed] [Google Scholar]
- 31.Schlondorff D, Banas B. The mesangial cell revisited: no cell is an island. J Am Soc Nephrol. 2009;20:1179–1187. doi: 10.1681/ASN.2008050549. [DOI] [PubMed] [Google Scholar]
- 32.Ka SM, Cheng CW, Shui HA, et al. Mesangial cells of lupus- prone mice are sensitive to chemokine production. Arthritis Res Ther. 2007;9:R67. doi: 10.1186/ar2226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Flur K, Allam R, Zecher D, et al. Viral RNA induces type I interferon-dependent cytokine release and cell death in mesangial cells via melanoma-differentiation-associated gene-5: implications for viral infection-associated glomerulonephritis. Am J Pathol. 2009;175:2014–2022. doi: 10.2353/ajpath.2009.080585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Patole PS, Pawar RD, Lech M, et al. Expression and regulation of Toll-like receptors in lupus-like immune complex glomerulonephritis of MRL-Fas(lpr) mice. Nephrol Dial Transplant. 2006;21:3062–3073. doi: 10.1093/ndt/gfl336. [DOI] [PubMed] [Google Scholar]
- 35.Fairhurst AM, Xie C, Fu Y, et al. Type I interferons produced by resident renal cells may promote end-organ disease in autoantibody-mediated glomerulonephritis. J Immunol. 2009;183:6831–6838. doi: 10.4049/jimmunol.0900742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Yung S, Cheung KF, Zhang Q, Chan TM. Anti-dsDNA antibodies bind to mesangial annexin II in lupus nephritis. J Am Soc Nephrol. 2010;21:1912–1927. doi: 10.1681/ASN.2009080805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yung S, Chan TM. Anti-dsDNA antibodies and resident renal cells: their putative roles in pathogenesis of renal lesions in lupus nephritis. Clin Immunol. 2017;185:40–50. doi: 10.1016/j.clim.2016.09.002. [DOI] [PubMed] [Google Scholar]
- 38.Ichinose K, Rauen T, Juang YT, et al. Cutting edge: calcium/ calmodulin-dependent protein kinase type IV is essential for mesangial cell proliferation and lupus nephritis. J Immunol. 2011;187:5500–5504. doi: 10.4049/jimmunol.1102357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Tsantikos E, Maxwell MJ, Putoczki T, et al. Interleukin- 6 trans-signaling exacerbates inflammation and renal pathology in lupus-prone mice. Arthritis Rheum. 2013;65:2691–2702. doi: 10.1002/art.38061. [DOI] [PubMed] [Google Scholar]
- 40.Ryffel B, Car BD, Gunn H, Roman D, Hiestand P, Mihatsch MJ. Interleukin-6 exacerbates glomerulonephritis in (NZB x NZW)F1 mice. Am J Pathol. 1994;144:927–937. [PMC free article] [PubMed] [Google Scholar]
- 41.Castellano G, Cafiero C, Divella C, et al. Local synthesis of interferon-alpha in lupus nephritis is associated with type I interferons signature and LMP7 induction in renal tubular epithelial cells. Arthritis Res Ther. 2015;17:72. doi: 10.1186/s13075-015-0588-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Schwarting A, Relle M, Meineck M, et al. Renal tubular epithelial cell-derived BAFF expression mediates kidney damage and correlates with activity of proliferative lupus nephritis in mouse and men. Lupus. 2017 Jan 1; doi: 10.1177/0961203317717083. [Epub]. https://doi.org/10.1177/0961203317717083. [DOI] [PubMed] [Google Scholar]
- 43.Chen Y, Yang C, Xie Z, et al. Expression of the novel co-stimulatory molecule B7-H4 by renal tubular epithelial cells. Kidney Int. 2006;70:2092–2099. doi: 10.1038/sj.ki.5001867. [DOI] [PubMed] [Google Scholar]
- 44.Yung S, Tsang RC, Sun Y, Leung JK, Chan TM. Effect of human anti-DNA antibodies on proximal renal tubular epithelial cell cytokine expression: implications on tubulointerstitial inflammation in lupus nephritis. J Am Soc Nephrol. 2005;16:3281–3294. doi: 10.1681/ASN.2004110917. [DOI] [PubMed] [Google Scholar]