Abstract
Low back pain (LBP) is one of the most common musculoskeletal diseases in the world. The incidence is ~70% in adults and many of them suffer from disability. Recently, intervertebral disc degeneration (IDD) has been deemed as a main cause of LBP. The present study aimed to investigate the potentials of growth and differentiation factor-5 (GDF-5) in IDD. The protein levels of prostaglandin-E2 (PGE2), tumor necrosis factor (TNF)-α and interleukin (IL)-1β in culture medium were evaluated by ELISA. mRNA and protein expression levels in nucleus pulposus (NP) cells were evaluated by reverse transcription-quantitative polymerase chain reaction (RT-qPCR) and western blotting, respectively. Griess reaction was applied to test the nitric oxide (NO) concentration in the culture supernatant. The expression levels of inducible NO synthase (iNOS) and cyclooxygenase-2 (COX-2) in NP cells were measured by RT-qPCR. Collagen-II, aggrecan, IκBα and phosphorylated (p)-p65 expression levels were detected by western blotting. Compared with the control group, protein expression levels of TNF-α, IL-1β and PGE2, and NO concentration in culture medium were upregulated by LPS, which were significantly repressed by GDF-5 overexpression (P<0.05). Additionally, GDF-5 overexpression reduced lipopolysaccharide-induced upregulation of TNF-α, IL-1β, iNOS, COX-2, collagen-II, aggrecan, IκBα and p-p65 expression levels in NP cells.
Keywords: growth and differentiation factor-5, intervertebral disc degeneration, inflammatory factors, nuclear factor-κB signaling pathway
Introduction
Low back pain (LBP) is one of the most common musculoskeletal diseases in the world, with an incidence of ~70% in adults (1). Unfortunately, many of those with LBP suffer from disability (1). Multiple causes could lead to LBP; however, intervertebral disc degeneration (IDD) and disc herniation are reported to be the two most common diagnoses and targets for intervention (2), with IDD being deemed as the main cause of LBP (3,4). IDD is a multifactorial process that is characterized by cellular and biochemical changes in the disc tissue, consequently generating structural failure (5).
A normal disc is composed of two types of tissues, the nucleus pulposus (NP) and the annulus fibrosus (AF), and they serve different roles in load bearing. Gelatinous NP is predominantly composed of type II collagen and proteoglycans, and its relatively higher water content (compared to AF tissues) is responsible for its resistance to compressive forces and hydrostatic pressurization (6,7). In degeneration, loss of proteoglycans and water signal intensity is detected by changes in disc height and T2-weighted magnetic resonance imaging (MRI) signal in the NP (8), which may lead to redistribution of load onto fibrochondrocyte-like cells in the AF (9).
As IDD is associated with normal aging, many people with IDD indications on MRI do not suffer with pain or disability (10,11). However, current treatments for IDD, including surgery, steroid injection and physical therapy, treat symptoms and not disc structure/function regeneration.
Inflammation is correlated with IDD and it involves various cells and molecules (12). Multiple genes were reported to be associated with genetic predisposition to IDD, including the inflammatory genes cyclooxygenase (COX)-2, interleukin (IL)1-α, IL1-β and IL-6 (13).
The GDF-5 gene was demonstrated to be a susceptibility gene for IDD (14,15) and defects in this gene result in abnormalities of collagen and proteoglycan discs in mice (16). However, there was no report regarding overexpression of GDF-5 in inhibiting inflammatory factors released by intervertebral disc cells. Therefore, the present study aimed to demonstrate this hypothesis in vitro by analyzing the levels of nuclear factor (NF)-κB and inflammatory factors.
Materials and methods
Reagents and animal ethics
Lipopolysaccharide (LPS) was purchased from Sigma-Aldrich (Merck KGaA, Darmstadt, Germany). pRL-TK plasmids were purchased from Promega Corporation (Madison, WI, USA). A total of 30 sprague Dawley (SD) rats (6–8 weeks old, 250–300 g), regardless of their gender (24 male, 6 female), were purchased from Animal Research Center of Beijing University of Chinese Medicine (Beijing, China). All the rats were maintained at 27±3°C and a humidity of 60±15% with a 12 h light/dark cycle with free access to water and food. Then the rats were euthanized by abdominal injection of a lethal dose (90 mg/kg) of pentobarbital sodium (Sigma-Aldrich; Merck KGaA). All animal work was performed in accordance with relevant national and international guidelines and approved by the Animal Experimental Ethical Committee of Beijing University of Chinese Medicine (Beijing, China).
NP cell isolation and culture
Following sacrifice, NP cells were isolated from the lumbar spines of adult SD rats. The spines were separated between each of the lumbar discs and, subsequently, a sterile scalpel blade was applied to extract the NP completely. NP was rinsed with PBS to remove other cells, followed by digestion with trypsin and collagenase at 37°C for 25 min. NP cells were incubated in high-glucose Dulbecco's modified Eagle's medium (Gibco; Thermo Fisher Scientific, Inc., Waltham, MA, USA) supplemented with 10% fetal bovine serum (Gibco; Thermo Fisher Scientific, Inc.), 100 U/ml penicillin and 100 mg/ml streptomycin, cultured up to passage 2–3 at 37°C. In order to create an IDD model group, cells were stimulated with 10 mg/ml LPS at 37°C for 24 h. Cells cultured with normal medium were used as controls and cells treated with LPS + GDF-5 were termed the LPS + GDP-5 group.
ELISA assessments
The levels of prostaglandin-E2 (PGE-2), tumor necrosis factor (TNF)-α and IL-1β in the culture medium were assessed using a Rat TNF-α Quantikine ELISA kit (cat. no. RTA00) or Rat IL-1β/IL-1F2 Quantikine ELISA kit (cat. no. RLB00; R&D Systems, Inc., Minneapolis, MN, USA), according to the protocols of the manufacturer.
Measurement of nitric oxide (NO) concentration
NO concentration was measured using Griess reagent (Sigma-Aldrich; Merck KGaA). In brief, NP cells were transfected with 100 ng/ml GDF-5 plasmid (Guangzhou Ribobio Co., Ltd., Guangzhou, China) using the Lipofectamine™ 2000 kit (Thermo Fisher Scientific, Inc.) for 2 h followed by incubation with 10 mg/ml LPS at 37°C for 24 h. Culture supernatant (50 µl) from each group was incubated with the same volume (50 µl) of Griess reagent at room temperature for 15 min in a 96-well plate. Subsequently, the absorbance at 540 nm was read. The standard curve was made by NaNO2.
Reverse transcription-quantitative polymerase chain reaction (RT-qPCR)
Total RNA from NP cells was isolated using TRIzol reagent (Invitrogen; Thermo Fisher Scientific, Inc.) according to the manufacturer's protocol. RT was conducted from 1 mg RNA with a 1st Strand cDNA Synthesis kit (Takara Biotechnology Co., Ltd., Dalian, China) to obtain first-strand cDNA.
The relative gene expression levels were determined using qPCR. qPCR was conducted with a SYBR Premix Ex Taq kit (Takara Biotechnology Co., Ltd.) on an ABI Prism 7500 Fast Real-Time PCR system (Applied Biosystems; Thermo Fisher Scientific, Inc.), according to the manufacturer's protocol. The primer sequences used were as follows: Inducible NO synthase (iNOS) forward, 5′-ACACAGTGTCGCTGGTTTGA-3′ and reverse, 5′-AGAAACTTCCAGGGGCAAGC-3′; COX-2 forward, 5′-ATCAGAACCGCATTGCCTCT-3′ and reverse, 5′-GCCAGCAATCTGTCTGGTGA-3′; TNF-α forward, 5′-GACCCTCACACTCAGATCATCTT-3′ and reverse, 5′-CCACTTGGTGGTTTGCTACGA-3′; IL-1β forward, 5′-TGAAATGCCACCTTTTGACAG-3′ and reverse, 5′-CCACAGCCACAATGAGTGATAC-3′; and β-actin forward, 5′-AACCTTCTTGCAGCTCCTCCG-3′ and reverse, 5′-CCATACCCACCATCACACCCT-3′. β-actin was used as an internal control. The thermocycling conditions of PCR were as follows: An initial denaturation at 95°C for 10 min, followed by an amplification cycle consisting of three steps; denaturation at 95°C for 15 sec, annealing at 58°C for 30 sec and elongation at 72°C for 30 sec. Triplicate Cq values were averaged and the relative expression levels were determined using the 2−ΔΔCq method (17).
Western blotting
For the analysis of pathway-related protein levels, NP cells were stimulated with 10 mg/ml LPS at 37°C for 30 min. For collagen II and aggrecan protein levels, NP cells were cultured with 10 mg/ml LPS at 37°C for 5 days. Subsequently, cells were washed with PBS and total protein was extracted with radioimmunoprecipitation assay lysis buffer (Beyotime Institute of Biotechnology, Haimen, China). The protein concentration was assessed using a BCA Protein Assay kit (Thermo Fisher Scientific, Inc.). Protein samples (20 mg) were separated by 10% SDS-PAGE and transferred onto polyvinylidene fluoride membranes (EMD Millipore, Billerica, MA, USA). Membranes were blocked with 5% fat-free milk at room temperature for 1 h. Subsequently, the membranes were incubated with primary antibodies against phosphorylated (p)-IκB (cat. no. 2859), p-p65 (cat. no. 3033), TNF-α (cat. no. 13377), IL-1β (cat. no. 12703) (1:1,000; Cell Signaling Technology, Inc., Danvers, MA, USA), aggrecan (cat. no. ab3773) and collagen-II (cat. no. ab188570) (all 1:1,000; Abcam, Cambridge, UK) at 4°C overnight. Following three washes with Tris-buffered saline with Tween-20, the membranes were incubated with horseradish peroxidase (HRP)-labeled Goat Anti-Rabbit Immunoglobulin G (IgG; cat. no. A0208; Beyotime Institute of Biotechnology) or HRP-labeled Goat Anti-Mouse IgG (cat. no. A0216; Beyotime Institute of Biotechnology) that were conjugated with IRDye 800CW at room temperature for 1 h. Immunoreactive bands were detected using an Odyssey infrared imaging system (LI-COR Biosciences, Lincoln, NE, USA). β-actin (cat. no. 4970; 1:2,000; Cell Signaling Technology, Inc.) was used as a control. Quantification was performed using ImageJ software version 1.43 (National Institutes of Health, Bethesda, MD, USA).
Statistical analysis
All experiments were repeated at least three times. Data were presented as the mean ± standard deviation. Statistical analyses were conducted using one-way analysis of variance, followed with Duncan's post hoc test. SPSS v. 19.0 (IBM Corp., Armonk, NY, USA) was used for statistical analyses. P<0.05 was considered to indicate a statistically significant difference.
Results
GDF-5 overexpression inhibits LPS-induced elevation of TNF-α and IL-1β in culture medium
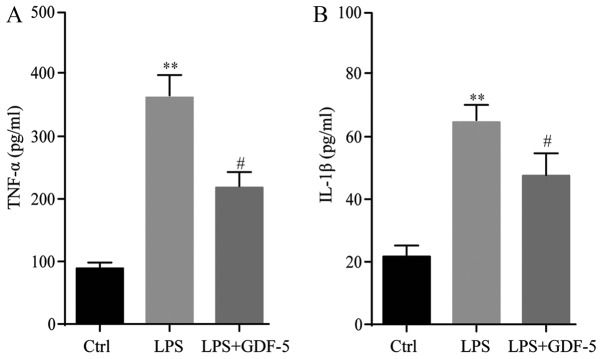
The protein levels of TNF-α and IL-1β in culture medium were evaluated by ELISA. Results demonstrated that in the control group, the expression level of TNF-α was only 95 pg/ml, while in the LPS-induced IDD model group, the TNF-α concentration was significantly elevated (P<0.01) compared with the levels in the control group (361 pg/ml). This level was significantly repressed to 203 pg/ml by GDF-5 overexpression (P<0.05; Fig. 1A). The changes to the IL-1β level were similar to that of TNF-α. In the control group, IL-1β was only at a level of 23 pg/ml and in the IDD group this level was significantly higher than that in the control group (63 pg/ml; P<0.01). This level was significantly reduced to 46 pg/ml following GDF-5 overexpression (P<0.05; Fig. 1B).
Figure 1.
GDF-5 overexpression inhibits LPS-induced secretion of TNF-α and IL-1β from nucleus pulposus cells. In the LPS group (model of intervertebral disc degeneration), protein expression levels of (A) TNF-α and (B) IL-1β were significantly higher than those in the Ctrl group, and these levels were significantly repressed by GDF-5 overexpression. **P<0.01 vs. Ctrl group; #P<0.05 vs. LPS group. GDF-5, growth and differentiation factor-5; LPS, lipopolysaccharide; TNF-α, tumor necrosis factor-α; IL-1β, interleukin-1β; Ctrl, control.
GDF-5 overexpression represses LPS-induced elevation of TNF-α and IL-1β in NP cells
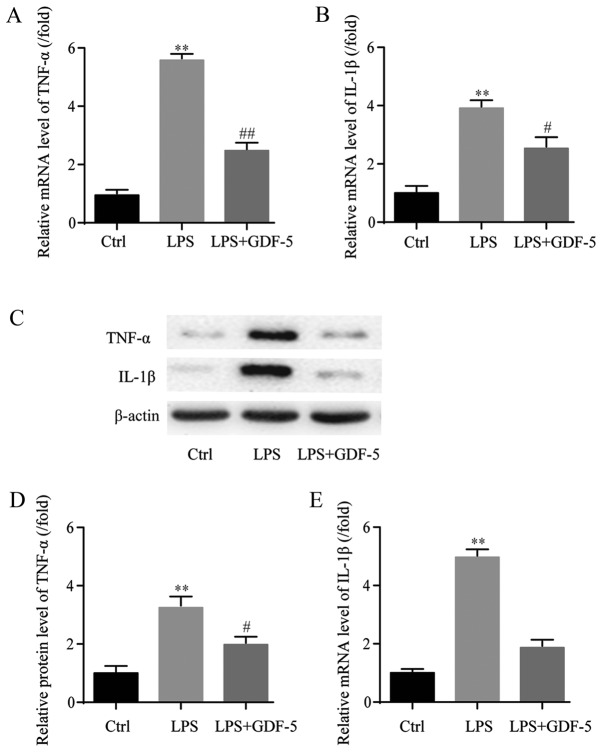
mRNA expression levels of TNF-α and IL-1β were assessed in NP cells by RT-qPCR. In the LPS-induced IDD model group, the mRNA expression level of TNF-α was ~6-fold higher that in the control group (P<0.01) and GDF-5 overexpression significantly inhibited TNF-α to near the normal level (P<0.01; Fig. 2A). Similarly, in the IDD group, the mRNA expression level of IL-1β was significantly increased by almost 4-fold compared with the level in the control group (P<0.01), which was significantly decreased by GDF-5 overexpression (P<0.05; Fig. 2B).
Figure 2.
GDF-5 overexpression represses LPS-induced production of TNF-α and IL-1β from nucleus pulposus cells. In the LPS group (model of intervertebral disc degeneration), the mRNA level of (A) TNF-α was ~5 folds higher than that in the Ctrl group and GDF-5 overexpression significantly inhibited TNF-α elevation. (B) The mRNA expression level of IL-1β demonstrated similar trends. (C) Western blotting was used to determine that protein expression levels of TNF-α and IL-1β. In the LPS group, results demonstrated that (D) TNF-α and (E) IL-1β protein expression levels were significantly higher than those in the Ctrl group. GDF-5 overexpression inhibited this overexpression. **P<0.01 vs. Ctrl group; #P<0.05 and ##P<0.01 vs. LPS group. GDF-5, growth and differentiation factor-5; LPS, lipopolysaccharide; TNF-α, tumor necrosis factor-α; IL-1β, interleukin-1β; Ctrl, control.
Following this, the protein expression levels of TNF-α and IL-1β in NP cells were evaluated by western blotting (Fig. 2C). In the IDD model group, the TNF-α protein expression level was significantly higher compared with the level in the control group (P<0.01). Furthermore, GDF-5 overexpression significantly inhibited the level of TNF-α (P<0.05; Fig. 2D). As for the IL-1β protein expression level, in the IDD model group, IL-1β was also significantly upregulated compared with the level in the control group (P<0.01). This level was suppressed by GDF-5 overexpression; however, not to a significant level (Fig. 2E).
GDF-5 overexpression decreases LPS-induced production of PGE2 and NO in NP cells
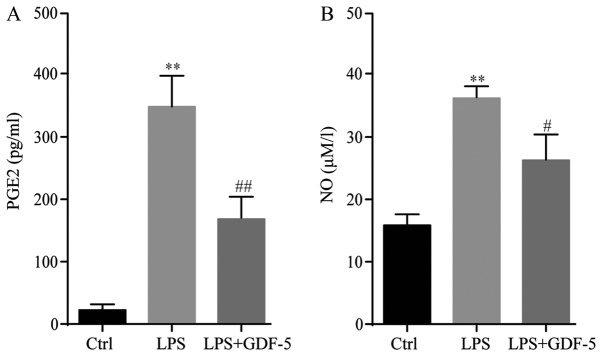
ELISA demonstrated that, in the control group, the PGE2 level was only ~20 pg/ml. In the IDD model group, the PGE2 level was significantly increased by ~17-fold following LPS stimulation compared with the level in the control group (P<0.01). GDF-5 overexpression significantly inhibited LPS-induced PGE2 production to ~156 pg/ml in the culture medium (P<0.01; Fig. 3A).
Figure 3.
GDF-5 overexpression decreases LPS-induced production of PGE2 and NO in nucleus pulposus cells. In the Ctrl group, the levels of (A) PGE2 and (B) NO were very low; however, these were significantly increased following LPS stimulation. GDF-5 overexpression inhibited LPS-induced PGE2 and NO production. **P<0.01 vs. Ctrl group; #P<0.05 and ##P<0.01 vs. LPS group. GDF-5, growth and differentiation factor-5; LPS, lipopolysaccharide; PGE2, prostaglandin-E2; NO, nitric oxide; Ctrl, control.
Results of the Griess reaction indicated that, in the culture supernatant of the control group, the NO concentration was 16 µM/l, whereas in the LPS-stimulated cells, this level increased significantly to 36 µM/l in the IDD group (P<0.01). GDF-5 overexpression significantly inhibited LPS-induced NO production to 25 µM/l in NP cells (P<0.05; Fig. 3B).
GDF-5 overexpression decreases LPS-induced production of COX-2 and iNOS in NP cells
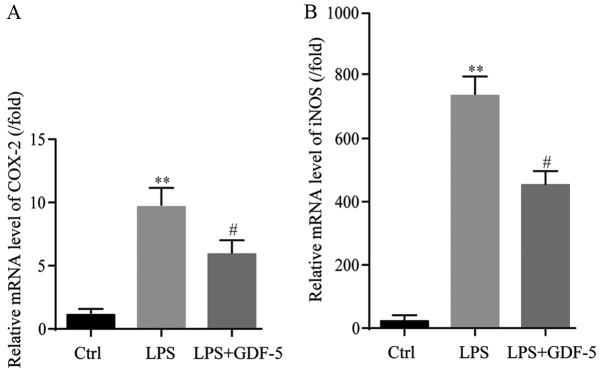
RT-qPCR indicated that LPS induced an ~10-fold increase in gene expression of COX-2 in comparison with the level in the control group (P<0.01). This increase induced by LPS treatment was significantly reduced (by 6-fold) by GDF-5 overexpression (P<0.05; Fig. 4A).
Figure 4.
GDF-5 overexpression decreases LPS-induced production of COX-2 and iNOS in nucleus pulposus cells. (A) LPS induced gene expression of COX-2 in comparison with the Ctrl group, which was reversed by 6 folds by GDF-5 overexpression. (B) Regarding gene expression of iNOS, LPS increased gene expression levels of iNOS compared with those in the Ctrl group and GDF-5 overexpression reversed LPS-induced gene expression of iNOS. **P<0.01 vs. Ctrl group; #P<0.05 vs. LPS group. GDF-5, growth and differentiation factor-5; LPS, lipopolysaccharide; COX-2, cyclooxygenase-2; iNOS, inducible nitric oxide synthase; Ctrl, control.
Results from RT-qPCR demonstrated that LPS significantly increased gene expression levels of iNOS, by 770-fold of that in the control group (P<0.01). GDF-5 overexpression significantly reduced the LPS-induced gene expression level of iNOS by ~421-fold (P<0.05; Fig. 4B).
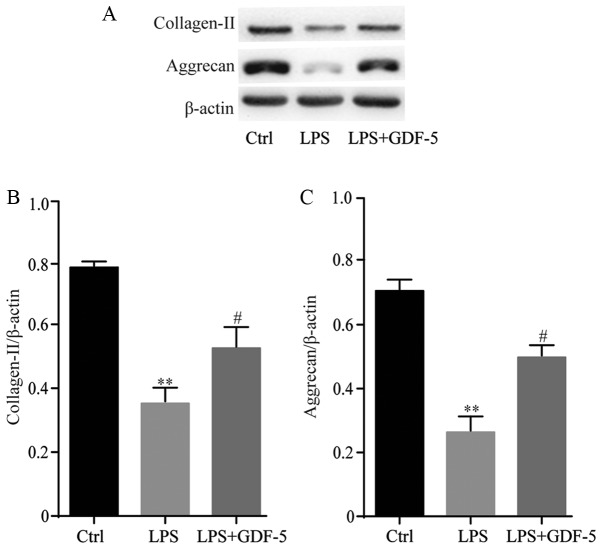
GDF-5 overexpression protects NP cells from LPS-induced matrix degradation
GDF-5 overexpression effectively inhibited plentiful matrix-degrading enzymes, which were induced by LPS; therefore, analysis was performed to determine whether GDF-5 overexpression antagonized LPS-induced matrix degradation of NP cells. NP cells were stimulated with 10 mg/ml LPS in the presence or absence of GDF-5 overexpression for 5 days, followed by western blot analysis. Results indicated that stimulation of NP cells with LPS significantly reduced collagen-II and aggrecan content compared with the levels in the control cells (P<0.01). GDF-5 overexpression significantly inhibited the decrease of collagen-II and aggrecan compared with that in LPS group (P<0.05; Fig. 5).
Figure 5.
GDF-5 overexpression protects NP cells from LPS-induced matrix degradation. (A) Western blotting results for the protein expression levels of collagen-II and aggrecan. Stimulation of NP cells with LPS significantly reduced (B) collagen-II and (C) aggrecan content. GDF-5 overexpression significantly inhibited the decrease of collagen-II and aggrecan compared with that in the LPS group. **P<0.01 vs. Ctrl group; #P<0.05 vs. LPS group. NP, nucleus pulposus; GDF-5, growth and differentiation factor-5; LPS, lipopolysaccharide; Ctrl, control.
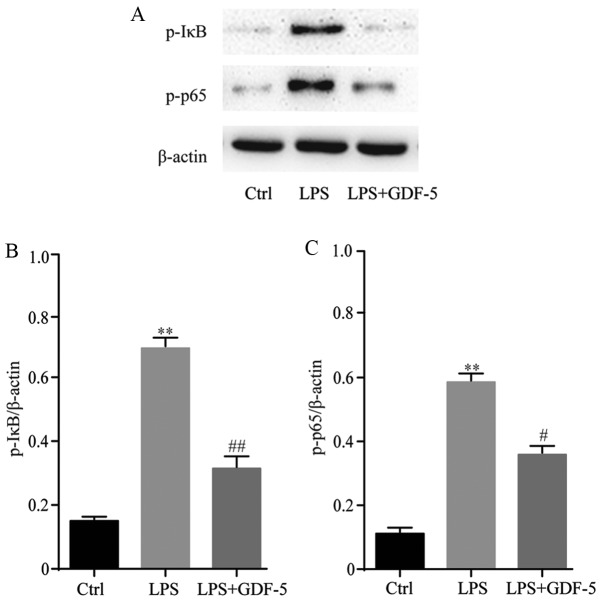
GDF-5 overexpression prevents NF-κB over-activation from LPS-stimulated NP cells
NP cells were pretreated with GDF-5 plasmid for 2 h and then stimulated with 10 mg/ml LPS for 24 h. Subsequently, western blotting was performed to evaluate the mechanism of GDF-5 on LPS-treated NP cells (Fig. 6A). GDF-5 significantly inhibited the phosphorylation of IκB (P<0.01) and p65 (P<0.05) induced by LPS (Fig. 6B and C). These results demonstrated that GDF-5 significantly inhibited the activation of the NF-κB pathway induced by LPS.
Figure 6.
GDF-5 overexpression prevents NF-κB over-activation in LPS-stimulated nucleus pulposus cells. (A) Western blotting results for the protein expression levels of p-IκB and p-p65. Quantitative analysis of western blotting data demonstrated that GDF-5 significantly inhibited the phosphorylation of (B) IκB and (C) p65 induced by LPS, indicating that GDF-5 inhibited the activation of the NF-κB pathway induced by LPS. **P<0.01 vs. Ctrl group; #P<0.05 and ##P<0.01 vs. LPS group. GDF-5, growth and differentiation factor-5; LPS, lipopolysaccharide; Ctrl, control; NF-κB, nuclear factor-κB; p, phosphorylated.
Discussion
LBP is one of the most frequent musculoskeletal diseases worldwide and results from IDD (1,3,4). In IDD, cellular and biochemical deficits and structural failure occur in the discs (5). However, a large quantity of patients with IDD exhibit neither pain nor disability (10,11). GDF-5 defects have been reported to lead to abnormalities of collagen and proteoglycan in discs in mice (16). Current treatments for IDD treat symptoms but not disc structure/function regeneration. Therefore, it is necessary to explore possible effective therapies for IDD.
Inflammation is correlated with IDD (12); furthermore, various inflammatory genes have been demonstrated to be correlated with IDD, including COX-2, IL-1α, IL-1β and IL-6 (13). The present study utilized ELISA to determine levels of TNF-α and IL-1β in the culture medium, meanwhile, the mRNA and protein levels of TNF-α and IL-1β in NP cells were evaluated by RT-qPCR and western blotting, respectively. Results indicated that in culture medium and NP cells, TNF-α and IL-1β levels were significantly increased in the LPS-stimulated group, and GDF-5 significantly decreased the aforementioned symptoms.
Mounting evidences have suggested that NO and PGE2 serve crucial roles in the modulation of cellular metabolism of discs and the pathology of IDD (18–20). PGE2 was found to be involved in the progression of sciatica (21). Additionally, NO was discovered to contribute to the development of radiculopathy (22). In the present study, ELISA and Griess reactions were performed to assess the levels of PGE2 in culture medium and NO in NP cells, respectively. Results demonstrated that GDF-5 overexpression significantly inhibited LPS-induced upregulation of PGE2 and NO expression levels.
Furthermore, RT-qPCR was conducted to evaluate the mRNA expression levels of COX-2 and iNOS in NP cells. Results indicated that, GDF-5 overexpression significantly reversed LPS-induced elevation of gene expression of COX-2 and iNOS. Taken together, these results demonstrated that GDF-5 attenuated LPS-induced IDD via inhibiting the production and release of inflammatory factors; however, the possible molecular mechanisms remain to be fully elucidated.
NP is predominantly composed of collagen-II and aggrecan, and the decrease of collagen-II is associated with disc degeneration (23,24). The present study demonstrated that GDF-5 prevented the loss of collagen-II and aggrecan that was induced by LPS. The NF-κB pathway modulates inflammation (25,26). The results of the present study suggested that GDF-5 inhibited phosphorylation of IκB and p65, which was stimulated by LPS in NP cells. However, further research performed in vivo is required to confirm this hypothesis.
In conclusion, the present study demonstrated that GDF-5 possessed anti-inflammatory and anti-degenerative effects on LPS-induced IDD via inhibition of NF-κB pathway activation in NP cells; therefore, GDF-5 may be a potential novel agent for treating IDD in the future.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The analyzed data sets generated during the present study are available from the corresponding author on reasonable request.
Authors' contributions
LS wrote the manuscript and interpreted the data. YW analyzed the data and revised the manuscript. LH searched the literature and collected the data. HZ designed the study.
Ethics approval and consent to participate
All animal work was performed in accordance with relevant national and international guidelines and approved by the Animal Experimental Ethical Committee of Beijing University of Chinese Medicine (Beijing, China).
Consent for publication
Not applicable.
Competing interests
All authors have no conflict of interest to declare.
References
- 1.Andersson GB. Epidemiological features of chronic low-back pain. Lancet. 1999;354:581–585. doi: 10.1016/S0140-6736(99)01312-4. [DOI] [PubMed] [Google Scholar]
- 2.Deyo RA, Weinstein JN. Low back pain. N Engl J Med. 2001;344:363–370. doi: 10.1056/NEJM200102013440508. [DOI] [PubMed] [Google Scholar]
- 3.Luoma K, Riihimäki H, Luukkonen R, Raininko R, Viikari-Juntura E, Lamminen A. Low back pain in relation to lumbar disc degeneration. Spine (Phila Pa 1976) 2000;25:487–492. doi: 10.1097/00007632-200002150-00016. [DOI] [PubMed] [Google Scholar]
- 4.Takatalo J, Karppinen J, Niinimäki J, Taimela S, Näyhä S, Mutanen P, Sequeiros RB, Kyllönen E, Tervonen O. Does lumbar disc degeneration on magnetic resonance imaging associate with low back symptom severity in young Finnish adults? Spine (Phila Pa 1976) 2011;36:2180–2189. doi: 10.1097/BRS.0b013e3182077122. [DOI] [PubMed] [Google Scholar]
- 5.Adams MA, Roughley PJ. What is intervertebral disc degeneration, and what causes it? Spine (Phila Pa 1976) 2006;31:2151–2161. doi: 10.1097/01.brs.0000231761.73859.2c. [DOI] [PubMed] [Google Scholar]
- 6.Antoniou J, Steffen T, Nelson F, Winterbottom N, Hollander AP, Poole RA, Aebi M, Alini M. The human lumbar intervertebral disc: Evidence for changes in the biosynthesis and denaturation of the extracellular matrix with growth, maturation, ageing, and degeneration. J Clin Invest. 1996;98:996–1003. doi: 10.1172/JCI118884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Ohshima H, Tsuji H, Hirano N, Ishihara H, Katoh Y, Yamada H. Water diffusion pathway, swelling pressure, and biomechanical properties of the intervertebral disc during compression load. Spine (Phila Pa 1976) 1989;14:1234–1244. doi: 10.1097/00007632-198911000-00017. [DOI] [PubMed] [Google Scholar]
- 8.Mwale F, Iatridis JC, Antoniou J. Quantitative MRI as a diagnostic tool of intervertebral disc matrix composition and integrity. Eur Spine J. 2008;17(Suppl 4):S432–S440. doi: 10.1007/s00586-008-0744-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Iatridis JC, Nicoll SB, Michalek AJ, Walter BA, Gupta MS. Role of biomechanics in intervertebral disc degeneration and regenerative therapies: What needs repairing in the disc and what are promising biomaterials for its repair? Spine J. 2013;13:243–262. doi: 10.1016/j.spinee.2012.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Boden SD, McCowin PR, Davis DO, Dina TS, Mark AS, Wiesel S. Abnormal magnetic-resonance scans of the cervical spine in asymptomatic subjects. A prospective investigation. J Bone Joint Surg Am. 1990;72:1178–1184. doi: 10.2106/00004623-199072080-00008. [DOI] [PubMed] [Google Scholar]
- 11.Jensen MC, Brant-Zawadzki MN, Obuchowski N, Modic MT, Malkasian D, Ross JS. Magnetic resonance imaging of the lumbar spine in people without back pain. N Engl J Med. 1994;331:69–73. doi: 10.1056/NEJM199407143310201. [DOI] [PubMed] [Google Scholar]
- 12.Risbud MV, Shapiro IM. Role of cytokines in intervertebral disc degeneration: Pain and disc content. Nat Rev Rheumatol. 2014;10:44–56. doi: 10.1038/nrrheum.2013.160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mayer JE, Iatridis JC, Chan D, Qureshi SA, Gottesman O, Hecht AC. Genetic polymorphisms associated with intervertebral disc degeneration. Spine J. 2013;13:299–317. doi: 10.1016/j.spinee.2013.01.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Williams FM, Popham M, Hart DJ, de Schepper E, Bierma-Zeinstra S, Hofman A, Uitterlinden AG, Arden NK, Cooper C, Spector TD, et al. GDF5 single-nucleotide polymorphism rs143383 is associated with lumbar disc degeneration in northern european women. Arthritis Rheum. 2011;63:708–712. doi: 10.1002/art.30169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mu J, Ge W, Zuo X, Chen Y, Huang C. Analysis of association between il-1β, casp-9, and gdf5 variants and low-back pain in chinese male soldier: Clinical article. J Neurosurg Spine. 2013;19:243–247. doi: 10.3171/2013.4.SPINE12782. [DOI] [PubMed] [Google Scholar]
- 16.Li X, Leo BM, Beck G, Balian G, Anderson GD. Collagen and proteoglycan abnormalities in the GDF-5-deficient mice and molecular changes when treating disk cells with recombinant growth factor. Spine (Phila Pa 1976) 2004;29:2229–2234. doi: 10.1097/01.brs.0000142427.82605.fb. [DOI] [PubMed] [Google Scholar]
- 17.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 18.Wang IC, Ueng SW, Lin SS, Niu CC, Yuan LJ, Su CI, Chen CH, Chen WJ. Effect of hyperbaric oxygenation on intervertebral disc degeneration: An in vitro study with human lumbar nucleus pulposus. Spine (Phila Pa 1976) 2011;36:1925–1931. doi: 10.1097/BRS.0b013e3181feebde. [DOI] [PubMed] [Google Scholar]
- 19.Takada T, Nishida K, Maeno K, Kakutani K, Yurube T, Doita M, Kurosaka M. Intervertebral disc and macrophage interaction induces mechanical hyperalgesia and cytokine production in a herniated disc model in rats. Arthtitis Rheum. 2012;64:2601–2610. doi: 10.1002/art.34456. [DOI] [PubMed] [Google Scholar]
- 20.Hou G, Lu H, Chen M, Yao H, Zhao H. Oxidative stress participates in age-related changes in rat lumbar intervertebral discs. Arch Gerontol Geriatr. 2014;59:665–669. doi: 10.1016/j.archger.2014.07.002. [DOI] [PubMed] [Google Scholar]
- 21.England S, Bevan S, Docherty RJ. PGE2 modulates the tetrodotoxin-resistant sodium current in neonatal rat dorsal root ganglion neurones via the cyclic AMP-protein kinase A cascade. J Physiol. 1996;495:429–440. doi: 10.1113/jphysiol.1996.sp021604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lee SJ, Kim TU, Park JS, Ra JY. Inhibition of nitric oxide mediated protein nitration: Therapeutic implications in experimental radiculopathy. Spine (Phila Pa 1976) 2013;38:1749–1753. doi: 10.1097/BRS.0b013e3182a085d9. [DOI] [PubMed] [Google Scholar]
- 23.Shi Z, Gu T, Xin H, Wu J, Xu C, Zhang C, He Q, Ruan D. Intervention of rAAV-hTERT-transducted nucleus pulposus cells in early stage of intervertebral disc degeneration: A study in canine model. Tissue Eng Part A. 2015;21:2186–2194. doi: 10.1089/ten.tea.2014.0408. [DOI] [PubMed] [Google Scholar]
- 24.Kozaci LD, Guner A, Oktay G, Guner G. Alterations in biochemical components of extracellular matrix in intervertebral disc herniation: Role of MMP-2 and TIMP-2 in type II collagen loss. Cell Biochem Funct. 2006;24:431–436. doi: 10.1002/cbf.1250. [DOI] [PubMed] [Google Scholar]
- 25.Tak PP, Firestein GS. NF-kappaB: A key role in inflammatory diseases. J Clin Invest. 2001;107:7–11. doi: 10.1172/JCI11830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Berenbaum F. Signaling transduction: Target in osteoarthritis. Curr Opin Rheumatol. 2004;16:616–622. doi: 10.1097/01.bor.0000133663.37352.4a. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The analyzed data sets generated during the present study are available from the corresponding author on reasonable request.