Abstract
Background
Betatrophin is a newly identified hormone derived from the liver and adipose tissue, which has been suggested to regulate glucose and lipid metabolism. Circulating levels of betatrophin are altered in various metabolic diseases, although the results are inconsistent. We aimed to examine whether betatrophin is a useful biomarker in predicting the development of diabetes.
Methods
A nested case-control study was performed using a prospective Chungju Metabolic disease Cohort Study. During a 4-year follow-up period, we analyzed 167 individuals who converted to diabetes and 167 non-converters, who were matched by age, sex, and body mass index. Serum betatrophin levels were measured by an ELISA (enzyme-linked immunosorbent assay).
Results
Baseline serum betatrophin levels were significantly higher in the converter group compared to the non-converter group (1,315±598 pg/mL vs. 1,072±446 pg/mL, P<0.001). After adjusting for age, sex, body mass index, fasting plasma glucose, systolic blood pressure, total cholesterol, and family history of diabetes, the risk of developing diabetes showed a stepwise increase across the betatrophin quartile groups. Subjects in the highest baseline quartile of betatrophin levels had more than a threefold higher risk of incident diabetes than the subjects in the lowest quartile (relative risk, 3.275; 95% confidence interval, 1.574 to 6.814; P=0.010). However, no significant relationships were observed between serum betatrophin levels and indices of insulin resistance or β-cell function.
Conclusion
Circulating levels of betatrophin could be a potential biomarker for predicting new-onset diabetes. Further studies are needed to understand the underlying mechanism of this association.
Keywords: Betatrophin, Biomarker, Case-control study, Risk
INTRODUCTION
With a steady increase in the prevalence and associated comorbidities, diabetes has become one of the most significant socioeconomic burdens worldwide [1,2]. Due to the rising population at risk for diabetes [2,3], effective treatment and prevention measures have attracted much interest, recently. Although blood glucose and glycosylated hemoglobin levels are powerful predictors of diabetes development, there have been considerable efforts to improve the prediction model using several different biomarkers. However, few of these novel circulating and genetic biomarkers have proven valuable thus far [4,5]. From the pathophysiologic point of view, a compensatory increase in pancreatic β-cell mass or function in reOriginal sponse to insulin resistance precedes glucose intolerance [6]. In this context, biomarkers reflecting these processes may enhance the identification of individuals in the subclinical stages of diabetes progression.
Dynamic cross-talk between organs is fundamental in maintaining metabolic homeostasis and in the process of developing metabolic diseases [7]. Recently, a liver-derived β-cell growth factor has been suggested to induce β-cell proliferation against insulin resistant state in a liver-specific insulin receptor knockout mouse model [8]. Another study identified a liver and adipose tissue expressed hormone, betatrophin (also known as angiopoietin-like 8 [ANGPTL8], lipasin, TD26, and refeeding induced in fat and liver [RIFL]), which correlated with β-cell proliferation in the mouse model of pharmacologically induced severe insulin resistance [9]. Identification of the hormone betatrophin led to various human studies, which aimed to elucidate its association with metabolic states including diabetes. Many studies have shown differences in the circulating levels of betatrophin in the context of diabetes, insulin resistance, obesity, metabolic syndrome, or dyslipidemia; however, the results were inconclusive [10,11,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28]. The relationship between circulating betatrophin concentrations and the risk of future diabetes is still unknown. Therefore, this study aimed to examine the predictive value of serum betatrophin levels in incident diabetes using a nested case-control design in a prospective cohort study.
METHODS
Study subjects
The Institutional Review Board at The Catholic University of Korea approved this study (No. KCMC070T076, KC14SISI0885) and written informed consent was obtained from all participants. The Chungju Metabolic disease Cohort (CMC) study is a community-based longitudinal study of metabolic diseases, including diabetes, metabolic syndrome, and osteoporosis, in a population aged 40 years and over living in the rural area of Chungju, Korea [29,30]. A total of 334 districts were selected by stratified random cluster sampling, and participants were enrolled in the first phase of the study conducted in 2003 to 2006. Follow-up of the participants occured every 4 years (the second phase in 2007 to 2010 and the third phase in 2011 to 2014). There were 2,624 individuals who participated in both the second and third phases of the study. The present study was a nested case-control study, using the second phase as baseline and the third phase as follow-up. Among non-diabetic subjects at baseline, we identified 213 individuals (8.1%) who developed diabetes during the 4-year follow-up period. After excluding subjects lacking laboratory or anthropometric data and those without available serum samples, 167 individuals who developed diabetes were included in the analysis. For each of these individuals, we selected an individual from the non-converter group with a 1:1 matching propensity scores. Controls were matched for age, sex, and body mass index (BMI).
Study protocol
Investigators and survey personnel were trained in the study protocol prior to carrying out physical examinations and administering the questionnaire. Anthropometric measurements were performed with the participants wearing light clothing, and their height, weight, and waist circumference (WC) were measured to the nearest 0.1 cm and 0.1 kg per standardized methods. Blood pressure (BP) was measured twice on the right upper arm while the subjects were in a seated position after 5 minutes of rest, and the average was recorded. Hypertension was defined on the basis of the Joint National Committee 7 report as ≥140 (systolic BP)/90 (diastolic BP) mm Hg or when the subjects reported using antihypertensive medications. Detailed data on the lifestyle behavior and medical history of the participants were obtained by interview, and included inquieries concerning the subjects' alcohol consumption (yes/no) and cigarette smoking (none, ex-smoker, or current smoker). Regular exercise was defined as three or more times per week for at least 30 minutes per session.
Biochemical assays and calculations
Blood samples were collected after the participants had fasted overnight for at least 12 hours and analyzed at a central laboratory (Neodin Medical Institute, Seoul, Korea). Fasting plasma glucose (FPG) was measured using the hexokinase method, and serum creatinine was measured using an enzymatic method. Estimated glomerular filtration rate (eGFR) was calculated using the abbreviated Modification of Diet in Renal Disease formula: 175×serum Cr (mg/dL)−1.154×Age (yr)−0.203×(0.742 if female). Serum total cholesterol (TC) and triglycerides (TG) were measured using enzymatic colorimetric tests. High density lipoprotein cholesterol was measured using a selective inhibition method, and low density lipoprotein cholesterol was calculated using the Friedewald formula. Serum high-sensitivity C-reactive protein levels were measured using a particle enhanced immunoturbidometric assay. Serum insulin levels were measured using a radioimmunoassay kit (Izotop, Budapest, Hungary), and the intra- and inter-assay coefficient of variance (CV) were <5%. The homeostasis model assessment estimate of β-cell function (HOMA-β) and insulin resistance (HOMA-IR) were calculated using the following formulae: 20×fasting insulin (mIU/L)/[FPG (mmol/L)–3.5] and FPG (mmol/L)×fasting insulin (mIU/L)/22.5, respectively. Serum betatrophin levels were determined using a commercially available human enzyme-linked immunosorbent assay (ELISA) kit (Wuhan EIAab Science, Wuhan, China; Catalog No. E11644h) per the manufacturer's instructions. The intra- and inter-assay CV were 7% and 12%, respectively.
Definition of diabetes mellitus and metabolic syndrome
Diabetes and prediabetes (impaired fasting glucose and/or impaired glucose tolerance) were defined according to the American Diabetes Association criteria. In the early period of the baseline study, an oral glucose tolerance test (OGTT) was performed if the FPG level exceeded 5.6 mmol/L. Beginning in September 2009, however, every participant underwent an OGTT, regardless of their FPG level. Every participant also underwent an OGTT for the diagnosis of glucose tolerance status during the follow-up period. Metabolic syndrome was defined using the American Heart Association/National Heart, Lung and Blood Institute criteria with modification of the WC value according to the World Health Organization–Asian Pacific region criteria for abdominal obesity as previously described [30].
Statistical analysis
Data are expressed as the mean±standard deviation (SD), as medians (interquartile range) or as proportions. The characteristics of the subgroups were compared using Student t-tests, chi-squared tests or analysis of variance using the general linear model (GLM) procedure. Spearman's correlation analysis was used to examine the relationships between serum betatrophin concentrations and other parameters. A log-binomial regression model using the GENMOD procedure was performed to calculate the relative risk (RR) of serum betatrophin in predicting the development of diabetes. The variables with significant difference between non-converter and converter at baseline or known risk factors of diabetes were selected as covariates. RR and 95% confidence interval values of incident diabetes for the second through fourth quartiles were calculated using the lowest quartile as the reference category. All statistical analyses were performed using SAS software version 9.4 (SAS Institute Inc., Cary, NC, USA). A P value <0.05 was considered significant.
RESULTS
The mean age and BMI were 69.7±7.4 years and 22.9±2.9 kg/m2 for men and 67.3±8.5 years and 25.7±3.4 kg/m2 for women, respectively. Table 1 shows a comparison of participant characteristics according to their glucose tolerance status at follow-up. At baseline, the subjects who had converted to diabetes had significantly higher levels of FPG and TC and a higher percentage of family history of diabetes compared to the non-converter group. Serum insulin levels, HOMA-IR, and HOMA-β were not significantly different between the two groups. At follow-up, the glucose levels obtained from OGTT, fasting insulin levels, and HOMA-IR were significantly higher in the converter group.
Table 1. Characteristics of participants according to their diabetes status at follow-up.
| Characteristic | Non-converter (n=167) | DM converter (n=167) | P value |
|---|---|---|---|
| Baseline | |||
| Age, yr | 68.1±8.1 | 68.2±8.3 | 0.873 |
| Male sex, % | 36.5 | 36.5 | 1.000 |
| Height, cm | 155.5±8.4 | 154.8±8.3 | 0.465 |
| Weight, kg | 59.8±8.9 | 59.1±9.6 | 0.458 |
| Waist circumference, cm | 90.0±8.5 | 89.9±9.4 | 0.933 |
| BMI, kg/m2 | 24.8±3.4 | 24.6±4.0 | 0.754 |
| Systolic BP, mm Hg | 144.3±19.5 | 146.0±18.6 | 0.406 |
| Diastolic BP, mm Hg | 85.3±10.0 | 84.5±10.3 | 0.461 |
| FPG, mmol/L | 5.32±0.54 | 5.52±0.58 | 0.001 |
| Fasting insulin, pmol/L | 25.5 (14.2–46.9) | 31.4 (17.3–44.8) | 0.252 |
| HOMA-IR | 0.91 (0.47–1.57) | 1.09 (0.61–1.55) | 0.126 |
| HOMA-β | 41.7 (24.2–73.6) | 46.3 (25.4–72.0) | 0.734 |
| Total cholesterol, mmol/L | 4.78±0.78 | 5.00±0.95 | 0.021 |
| Triglycerides, mmol/L | 1.41 (0.95–2.13) | 1.56 (1.02–2.22) | 0.464 |
| HDL-C, mmol/L | 1.21±0.30 | 1.27±0.37 | 0.120 |
| LDL-C, mmol/L | 2.82±0.73 | 2.94±0.85 | 0.178 |
| Serum creatinine, µmol/L | 64.4±14.4 | 65.2±14.6 | 0.648 |
| eGFR, mL/min/1.73 m2 | 93.5±20.9 | 91.9±19.2 | 0.465 |
| hsCRP, mg/L | 0.12 (0.07–0.23) | 0.11 (0.07–0.25) | 0.730 |
| Betatrophin, pg/mL | 1,072±446 | 1,315±598 | <0.001 |
| Hypertension, % | 46.1 | 55.7 | 0.080 |
| Family history of DM, % | 10.8 | 20.1 | 0.019 |
| Smoking, % | 0.891 | ||
| None | 66.5 | 64.0 | |
| Ex-smoker | 22.7 | 24.0 | |
| Current | 10.8 | 12.0 | |
| Alcohol drinking, % | 58.1 | 41.3 | 0.002 |
| Regular exercise, % | 7.8 | 7.2 | 0.835 |
| Follow-up | |||
| FPG, mmol/L | 5.18±0.52 | 5.99±1.12 | <0.001 |
| Glucose30, mmol/L | 9.21±1.72 | 10.96±2.17 | <0.001 |
| Glucose120, mmol/L | 7.68±1.74 | 12.68±2.57 | <0.001 |
| Fasting insulin, pmol/L | 28.0 (13.3–47.6) | 44.3 (16.1–64.8) | 0.001 |
| HOMA-IR | 0.96 (0.39–1.59) | 1.66 (0.55–2.49) | <0.001 |
| HOMA-β | 59.4 (21.5–95.0) | 59.9 (23.0–83.6) | 0.526 |
Values are presented as mean±standard deviation or median (interquartile range).
DM, diabetes mellitus; BMI, body mass index; BP, blood pressure; FPG, fasting plasma glucose; HOMA-IR, homeostasis model assessment of insulin resistance; HOMA-β, homeostasis model assessment estimate of β-cell function; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; eGFR, estimated glomerular filtration rate; hsCRP, high-sensitivity C-reactive protein.
Table 2 shows the mean betatrophin concentrations according to the baseline characteristic subgroups. Serum betatrophin concentrations were significantly higher in older, men, non-obese, non-abdominally obese, and hypertensive subjects compared to their counterparts. There were no significant differences between the subgroups according to metabolic syndrome, TG levels, eGFR, alcohol drinking, or the degree of insulin resistance. We calculated correlation coefficients between baseline serum betatrophin concentrations and glucose parameters in the baseline and during the follow-up period. At baseline, there were no significant correlations between betatrophin levels and FPG or insulin levels. Betatrophin levels were positively correlated with glucose levels at 120 minutes after OGTT (r=0.201, P<0.001) and negatively correlated with insulin levels at 30 minutes after OGTT (r=−0.187, P=0.006) at follow-up. However, no significant relationships were observed between betatrophin levels and HOMA-IR, HOMA-β or insulin/glucose ratio. The correlation between betatrophin levels and lipid profiles was also insignificant.
Table 2. Serum betatrophin concentrations according to characteristics of participants.
| Characteristic | No. | Mean±SD, pg/mL | P value |
|---|---|---|---|
| Age, yra | <0.001 | ||
| <70 | 171 | 1,064±552 | |
| ≥70 | 163 | 1,329±494 | |
| Sex | <0.001 | ||
| Men | 122 | 1,358±627 | |
| Women | 212 | 1,099±459 | |
| BMI, kg/m2 | 0.009 | ||
| <25 | 175 | 1,266±581 | |
| ≥25 | 159 | 1,113±480 | |
| WC, cm | 0.032 | ||
| <80 (W), 90 (M) | 49 | 1,347±583 | |
| ≥80 (W), 90 (M) | 285 | 1,167±529 | |
| Metabolic syndrome | 0.130 | ||
| No | 115 | 1,255±532 | |
| Yes | 219 | 1,161±543 | |
| Triglycerides, mmol/L | 0.274 | ||
| <1.69 | 194 | 1,220±576 | |
| ≥1.69 | 140 | 1,156±487 | |
| eGFR, mL/min/1.73 m2 | 0.124 | ||
| <60 | 12 | 1,429±490 | |
| ≥60 | 322 | 1,185±541 | |
| Hypertension | 0.026 | ||
| No | 164 | 1,126±527 | |
| Yes | 170 | 1,258±547 | |
| Alcohol drinking | 0.625 | ||
| No | 168 | 1,208±481 | |
| Yes | 166 | 1,179±596 | |
| HOMA-IRa | 0.142 | ||
| <1.0 | 182 | 1,233±541 | |
| ≥1.0 | 152 | 1,146±537 |
BMI, body mass index; WC, waist circumference; W, women; M, men; eGFR, estimated glomerular filtration rate; HOMA-IR, homeostasis model assessment of insulin resistance.
aSubgroups were divided according to the approximate mean or median value of the study population.
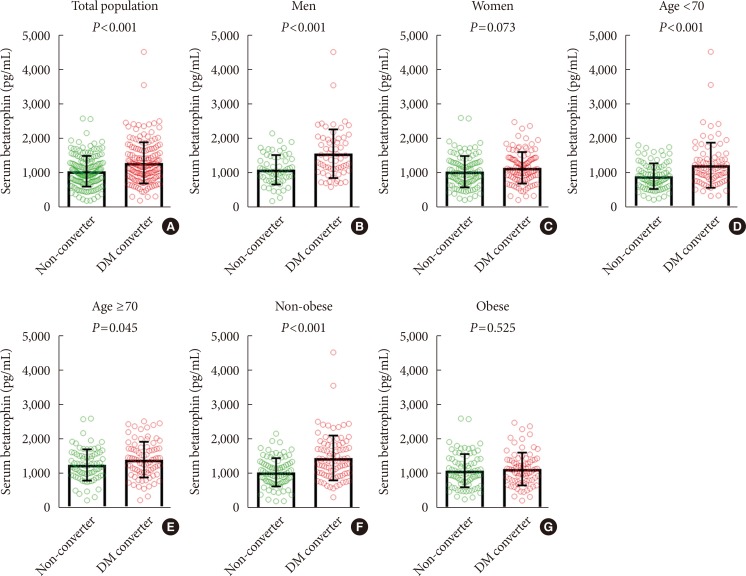
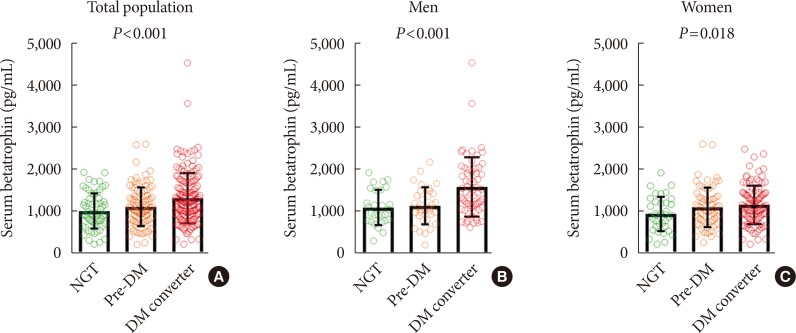
Baseline serum betatrophin levels were significantly higher in the converter group than in the non-converter group (1,315±598 pg/mL vs. 1,072±446 pg/mL, P<0.001). When subgroup analyses were performed, significant differences in betatrophin levels between the non-converter and converter groups were noted in both subjects <70 years of age (909±368 pg/mL vs. 1,225±657 pg/mL, P<0.001) and ≥70 years of age (1,250±456 pg/mL vs. 1,405±520 pg/mL, P=0.045), men (1,124±428 pg/mL vs. 1,591±707 pg/mL, P<0.001), and non-obese (1,057±409 pg/mL vs. 1,473±651 pg/mL, P<0.001) groups, but not in women or obese groups (Fig. 1). However, when the non-converter group was further divided into normal glucose tolerance (NGT) and prediabetes groups, a significant stepwise increase in baseline serum betatrophin levels were observed in both men (NGT, 1,103±419 pg/mL; prediabetes, 1,143±441 pg/mL; diabetes converter, 1,591±707 pg/mL; P for trend <0.001) and women (NGT, 939±409 pg/mL; prediabetes, 1,096±471 pg/mL; diabetes converter, 1,155±458 pg/mL; P for trend=0.018) (Fig. 2).
Fig. 1. Baseline serum betatrophin levels according to diabetes status at follow-up in the total population (A), men (B), women (C), <70 years-old (D), ≥70 years-old (E), non-obese (F), and obese (G) group. DM, diabetes mellitus.
Fig. 2. Baseline serum betatrophin levels according to glucose tolerance status at follow-up in the total population (A), men (B), and women (C). NGT, normal glucose tolerance; Pre-DM, prediabetes mellitus; DM, diabetes mellitus.
To evaluate the role of serum betatrophin in predicting the development of diabetes, we performed log-binomial regression analyses (Table 3). In the crude analysis, there was a statistically significant association between higher betatrophin concentrations and an increased risk of incident diabetes. After adjusting for age, sex, and BMI (model 1) and further adjusting for fasting glucose, systolic BP, TC, and family history of diabetes (model 2), this association remained significant. These results suggest that subjects with serum betatrophin concentrations in the highest quartile have more than a threefold increased risk of developing diabetes in the future.
Table 3. Risk of developing diabetes according to quartiles of serum betatrophin concentrations.
| No. of converter (%) | Crude | Model 1a | Model 2b | |
|---|---|---|---|---|
| Q1 | 33 (39.8) | 1 (reference) | 1 (reference) | 1 (reference) |
| Q2 | 37 (44.1) | 1.193 (0.645–2.207) | 1.301 (0.690–2.452) | 1.362 (0.698–2.657) |
| Q3 | 46 (55.4) | 1.884 (1.016–3.491) | 2.167 (1.122–4.183) | 2.063 (1.036–4.107) |
| Q4 | 51 (60.7) | 2.342 (1.259–4.355) | 2.854 (1.427–5.708) | 3.275 (1.574–6.814) |
| P value | 0.023 | 0.025 | 0.012 | 0.010 |
Values are presented as relative risks (95% confidence interval).
aModel 1: Adjusted for age, sex, and body mass index, bModel 2: Adjusted for model 1+fasting glucose, systolic blood pressure, total cholesterol, and family history of diabetes.
DISCUSSION
In this nested case-control study from a population-based prospective cohort, we found that baseline serum betatrophin levels were significantly higher in the diabetes converter group compared to the non-converter group. There was a stepwise increase in the risk of developing diabetes according to the serum betatrophin levels. After 4 years of follow-up, subjects in the highest baseline quartile of betatrophin levels had more than a threefold higher risk of incident diabetes than the subjects in the lowest quartile. Our findings indicate a possible role of circulating betatrophin as an additional biomarker for identifying individuals at high risk of developing diabetes.
'Betatrophin' was first named by the Melton group [9]. Microarray analysis of mouse liver treated with S961, a peptide that binds to insulin receptors and antagonizes insulin signaling, identified an upregulated gene which they called betatrophin. Betatrophin mRNA was expressed in mouse liver and adipose tissue, whereas liver was the primary organ that expressed betatrophin in human. They also showed that betatrophin encodes a liver-secreted protein leading to pancreatic β-cell proliferation, β-cell mass expansion, and improved glucose tolerance in mice. Although this attractive finding caught attention as a possible candidate of regenerative therapy for diabetes, subsequent studies strongly argued the effect of betatrophin in similar settings [31,32,33,34]. ANGPTL8-deficient mice showed normal β-cell expansion in response to insulin resistance and no alterations in glucose homeostasis [31,32]. Furthermore, ANGPTL8 overexpression did not increase β-cell replication in mice [32,34]. The effect of S961 treatment, which induces betatrophin gene expression and β-cell replication in mice, was not replicated in human β-cells [33]. A recent study with a different approach examined the effect of betatrophin-overexpressed adipose-derived mesenchymal stem cells and demonstrated that co-culture of human islets with these cells increased islet proliferation and insulin secretion [35]. Although the role of betatrophin is still being debated, the original article by the Melton group was recently retracted due to a lack of reproducibility.
Despite dissapointing observations in animal studies, many association studies in humans suggest a possible role of betatrophin in glucose and lipid metabolism. Circulating betatrophin levels were generally higher in metabolically disturbed states, such as type 1 diabetes mellitus [10,21], type 2 diabetes mellitus (T2DM) [11,13,15,16,17,18,21,22,24,25,36], overweight [19] or obesity [23], metabolic syndrome [37], and nonalcoholic fatty liver disease [38]. However, contradictory results were also reported with either no difference or lower levels of betatrophin in subjects with T2DM, obesity, and dyslipidemia when compared to control groups [12,14,19,26]. Correlation analyses between circulating betatrophin levels and clinical parameters in various studies also yielded inconsistent results. The role of betatrophin in TG metabolism is evidenced by preclinical studies. Overexpression of betatrophin in the liver increased TG levels, whereas knockout mice had lower TG levels, which was linked to lipoprotein lipase activity [31,32,39]. In humans, several investigations showed a positive correlation between serum betatrophin and TG levels [16,18,21], while we and others [10] found no such relationship. Whether betatrophin levels are associated with insulin resistance or β-cell function is also a matter of debate. Our results showed no correlation, which is in line with several studies [12,16,27], while other studies suggested either a positive correlation with the insulin resistance index [17,19] or a negative correlation with insulin secretory capacity [20].
The reason for these inconsistencies remains unclear. A meta-analysis suggested a possible difference in the relationship between betatrophin levels and T2DM according to ethnicity [28]. Different ELISA kits, which use antibodies against the N-terminus or C-terminus of betatrophin, detect either the full-length form or both the full-length and the cleaved form, respectively [40]. Although it is unclear which form of betatrophin is functional and how the proteolytic cleavage is controlled, it should be noted that use of different ELISA kits could lead to contrasting results. The inconsistency among studies might also arise from the small number of samples with different clinical characteristics or selection bias. Because the methods used to measure the degree of insulin resistance in previous studies were not the gold standard, further studies using more precise methods and a larger study population are needed to draw definitive conclusions.
To the best of our knowledge, our analysis is the first prospective study to examine the predictive value of betatrophin levels in identifying subjects at high-risk of developing diabetes. In another prospective study, Wang et al. [27] observed that the baseline betatrophin levels were inversely correlated with incident metabolic syndrome in 153 chinese adults after a 3.5-year follow-up. Interestingly, the odds ratio for high blood glucose (≥5.6 mmol/L) component was significantly lower in subjects with three higher quartiles of betatrophin when compared to the lowest quartile group in men, but not in women.
Some limitations should be acknowledged. Because our cohort consisted mainly of middle aged or elderly Koreans, the ability to generalize these findings to other populations of different ages and ethnicities is limited. In particular, several studies revealed a positive correlation between betatrophin levels and age [15,16,21,27]. Furthermore, the type of diabetes could not be determined due to the lack of C-peptide or autoantibody data, but we assume that the majority of the cases would be T2DM based on the age of the study population. Lastly, the HOMA index is limited in its ability to precisely reflect the degree of insulin resistance and β-cell function, although it is practical for large epidemiologic studies.
In conclusion, we propose that the circulating level of betatrophin could be a potential biomarker for predicting new-onset diabetes. Further studies are needed to understand the underlying mechanism of this association and to resolve remaining contradictory issues.
ACKNOWLEDGEMENTS
The authors wish to thank all the team members and survey personnel of the Chungju Metabolic disease Cohort study and the officers in Chungju Health Center for continued support.
Footnotes
CONFLICTS OF INTEREST: This study was supported by the grants from Yuhan Co. through the Catholic Medical Center Research Foundation. The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Koo BK, Moon MK. Are we in the same risk of diabetes mellitus? Gender- and age-specific epidemiology of diabetes in 2001 to 2014 in the Korean population. Diabetes Metab J. 2016;40:175–181. doi: 10.4093/dmj.2016.40.3.175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chen L, Magliano DJ, Zimmet PZ. The worldwide epidemiology of type 2 diabetes mellitus: present and future perspectives. Nat Rev Endocrinol. 2011;8:228–236. doi: 10.1038/nrendo.2011.183. [DOI] [PubMed] [Google Scholar]
- 3.Tabak AG, Herder C, Rathmann W, Brunner EJ, Kivimaki M. Prediabetes: a high-risk state for diabetes development. Lancet. 2012;379:2279–2290. doi: 10.1016/S0140-6736(12)60283-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Herder C, Kowall B, Tabak AG, Rathmann W. The potential of novel biomarkers to improve risk prediction of type 2 diabetes. Diabetologia. 2014;57:16–29. doi: 10.1007/s00125-013-3061-3. [DOI] [PubMed] [Google Scholar]
- 5.Echouffo-Tcheugui JB, Dieffenbach SD, Kengne AP. Added value of novel circulating and genetic biomarkers in type 2 diabetes prediction: a systematic review. Diabetes Res Clin Pract. 2013;101:255–269. doi: 10.1016/j.diabres.2013.03.023. [DOI] [PubMed] [Google Scholar]
- 6.Cho JH, Kim JW, Shin JA, Shin J, Yoon KH. β-Cell mass in people with type 2 diabetes. J Diabetes Investig. 2011;2:6–17. doi: 10.1111/j.2040-1124.2010.00072.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Shirakawa J, De Jesus DF, Kulkarni RN. Exploring inter-organ crosstalk to uncover mechanisms that regulate β-cell function and mass. Eur J Clin Nutr. 2017;71:896–903. doi: 10.1038/ejcn.2017.13. [DOI] [PubMed] [Google Scholar]
- 8.El Ouaamari A, Kawamori D, Dirice E, Liew CW, Shadrach JL, Hu J, Katsuta H, Hollister-Lock J, Qian WJ, Wagers AJ, Kulkarni RN. Liver-derived systemic factors drive β cell hyperplasia in insulin-resistant states. Cell Rep. 2013;3:401–410. doi: 10.1016/j.celrep.2013.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yi P, Park JS, Melton DA. Betatrophin: a hormone that controls pancreatic β cell proliferation. Cell. 2013;153:747–758. doi: 10.1016/j.cell.2013.04.008. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 10.Espes D, Lau J, Carlsson PO. Increased circulating levels of betatrophin in individuals with long-standing type 1 diabetes. Diabetologia. 2014;57:50–53. doi: 10.1007/s00125-013-3071-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Espes D, Martinell M, Carlsson PO. Increased circulating betatrophin concentrations in patients with type 2 diabetes. Int J Endocrinol. 2014;2014:323407. doi: 10.1155/2014/323407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Fenzl A, Itariu BK, Kosi L, Fritzer-Szekeres M, Kautzky-Willer A, Stulnig TM, Kiefer FW. Circulating betatrophin correlates with atherogenic lipid profiles but not with glucose and insulin levels in insulin-resistant individuals. Diabetologia. 2014;57:1204–1208. doi: 10.1007/s00125-014-3208-x. [DOI] [PubMed] [Google Scholar]
- 13.Fu Z, Berhane F, Fite A, Seyoum B, Abou-Samra AB, Zhang R. Elevated circulating lipasin/betatrophin in human type 2 diabetes and obesity. Sci Rep. 2014;4:5013. doi: 10.1038/srep05013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gomez-Ambrosi J, Pascual E, Catalan V, Rodriguez A, Ramirez B, Silva C, Gil MJ, Salvador J, Fruhbeck G. Circulating betatrophin concentrations are decreased in human obesity and type 2 diabetes. J Clin Endocrinol Metab. 2014;99:E2004–E2009. doi: 10.1210/jc.2014-1568. [DOI] [PubMed] [Google Scholar]
- 15.Hu H, Sun W, Yu S, Hong X, Qian W, Tang B, Wang D, Yang L, Wang J, Mao C, Zhou L, Yuan G. Increased circulating levels of betatrophin in newly diagnosed type 2 diabetic patients. Diabetes Care. 2014;37:2718–2722. doi: 10.2337/dc14-0602. [DOI] [PubMed] [Google Scholar]
- 16.Abu-Farha M, Abubaker J, Al-Khairi I, Cherian P, Noronha F, Hu FB, Behbehani K, Elkum N. Higher plasma betatrophin/ANGPTL8 level in type 2 diabetes subjects does not correlate with blood glucose or insulin resistance. Sci Rep. 2015;5:10949. doi: 10.1038/srep10949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chen X, Lu P, He W, Zhang J, Liu L, Yang Y, Liu Z, Xie J, Shao S, Du T, Su X, Zhou X, Hu S, Yuan G, Zhang M, Zhang H, Liu L, Wang D, Yu X. Circulating betatrophin levels are increased in patients with type 2 diabetes and associated with insulin resistance. J Clin Endocrinol Metab. 2015;100:E96–E100. doi: 10.1210/jc.2014-2300. [DOI] [PubMed] [Google Scholar]
- 18.Gao T, Jin K, Chen P, Jin H, Yang L, Xie X, Yang M, Hu C, Yu X. Circulating betatrophin correlates with triglycerides and postprandial glucose among different glucose tolerance statuses: a case-control study. PLoS One. 2015;10:e0133640. doi: 10.1371/journal.pone.0133640. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Guo K, Lu J, Yu H, Zhao F, Pan P, Zhang L, Chen H, Bao Y, Jia W. Serum betatrophin concentrations are significantly increased in overweight but not in obese or type 2 diabetic individuals. Obesity (Silver Spring) 2015;23:793–797. doi: 10.1002/oby.21038. [DOI] [PubMed] [Google Scholar]
- 20.Tokumoto S, Hamamoto Y, Fujimoto K, Yamaguchi E, Okamura E, Honjo S, Ikeda H, Wada Y, Hamasaki A, Koshiyama H. Correlation of circulating betatrophin concentrations with insulin secretion capacity, evaluated by glucagon stimulation tests. Diabet Med. 2015;32:653–656. doi: 10.1111/dme.12696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yamada H, Saito T, Aoki A, Asano T, Yoshida M, Ikoma A, Kusaka I, Toyoshima H, Kakei M, Ishikawa SE. Circulating betatrophin is elevated in patients with type 1 and type 2 diabetes. Endocr J. 2015;62:417–421. doi: 10.1507/endocrj.EJ14-0525. [DOI] [PubMed] [Google Scholar]
- 22.Yi M, Chen RP, Yang R, Guo XF, Zhang JC, Chen H. Betatrophin acts as a diagnostic biomarker in type 2 diabetes mellitus and is negatively associated with HDL-cholesterol. Int J Endocrinol. 2015;2015:479157. doi: 10.1155/2015/479157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Abu-Farha M, Sriraman D, Cherian P, AlKhairi I, Elkum N, Behbehani K, Abubaker J. Circulating ANGPTL8/betatrophin is increased in obesity and reduced after exercise training. PLoS One. 2016;11:e0147367. doi: 10.1371/journal.pone.0147367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Al-Daghri NM, Rahman S, Sabico S, Amer OE, Wani K, Ansari MG, Al-Attas OS, Kumar S, Alokail MS. Circulating betatrophin in healthy control and type 2 diabetic subjects and its association with metabolic parameters. J Diabetes Complications. 2016;30:1321–1325. doi: 10.1016/j.jdiacomp.2016.05.023. [DOI] [PubMed] [Google Scholar]
- 25.Chen CC, Susanto H, Chuang WH, Liu TY, Wang CH. Higher serum betatrophin level in type 2 diabetes subjects is associated with urinary albumin excretion and renal function. Cardiovasc Diabetol. 2016;15:3. doi: 10.1186/s12933-015-0326-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gomez-Ambrosi J, Pascual-Corrales E, Catalan V, Rodriguez A, Ramirez B, Romero S, Vila N, Ibanez P, Margall MA, Silva C, Gil MJ, Salvador J, Fruhbeck G. Altered concentrations in dyslipidemia evidence a role for ANGPTL8/betatrophin in lipid metabolism in humans. J Clin Endocrinol Metab. 2016;101:3803–3811. doi: 10.1210/jc.2016-2084. [DOI] [PubMed] [Google Scholar]
- 27.Wang H, Lai Y, Han C, Liu A, Fan C, Wang H, Zhang H, Ding S, Teng W, Shan Z. The effects of serum ANGPTL8/betatrophin on the risk of developing the metabolic syndrome: a prospective study. Sci Rep. 2016;6:28431. doi: 10.1038/srep28431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Yue S, Wu J, Zhang J, Liu L, Chen L. The relationship between betatrophin levels in blood and T2DM: a systematic review and meta-analysis. Dis Markers. 2016;2016:9391837. doi: 10.1155/2016/9391837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lee SH, Rhee M, Yang HK, Ha HS, Lee JH, Kwon HS, Park YM, Yim HW, Kang MI, Lee WC, Son HY, Yoon KH. Serum preadipocyte factor 1 concentrations and risk of developing diabetes: a nested case-control study. Diabet Med. 2016;33:631–638. doi: 10.1111/dme.12871. [DOI] [PubMed] [Google Scholar]
- 30.Lee SH, Yang HK, Ha HS, Lee JH, Kwon HS, Park YM, Yim HW, Kang MI, Lee WC, Son HY, Yoon KH. Changes in metabolic health status over time and risk of developing type 2 diabetes: a prospective cohort study. Medicine (Baltimore) 2015;94:e1705. doi: 10.1097/MD.0000000000001705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wang Y, Quagliarini F, Gusarova V, Gromada J, Valenzuela DM, Cohen JC, Hobbs HH. Mice lacking ANGPTL8 (Betatrophin) manifest disrupted triglyceride metabolism without impaired glucose homeostasis. Proc Natl Acad Sci U S A. 2013;110:16109–16114. doi: 10.1073/pnas.1315292110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gusarova V, Alexa CA, Na E, Stevis PE, Xin Y, Bonner-Weir S, Cohen JC, Hobbs HH, Murphy AJ, Yancopoulos GD, Gromada J. ANGPTL8/betatrophin does not control pancreatic beta cell expansion. Cell. 2014;159:691–696. doi: 10.1016/j.cell.2014.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Jiao Y, Le Lay J, Yu M, Naji A, Kaestner KH. Elevated mouse hepatic betatrophin expression does not increase human β-cell replication in the transplant setting. Diabetes. 2014;63:1283–1288. doi: 10.2337/db13-1435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cox AR, Lam CJ, Bonnyman CW, Chavez J, Rios JS, Kushner JA. Angiopoietin-like protein 8 (ANGPTL8)/betatrophin overexpression does not increase beta cell proliferation in mice. Diabetologia. 2015;58:1523–1531. doi: 10.1007/s00125-015-3590-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Sun LL, Liu TJ, Li L, Tang W, Zou JJ, Chen XF, Zheng JY, Jiang BG, Shi YQ. Transplantation of betatrophin-expressing adipose-derived mesenchymal stem cells induces β-cell proliferation in diabetic mice. Int J Mol Med. 2017;39:936–948. doi: 10.3892/ijmm.2017.2914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Li S, Liu D, Li L, Li Y, Li Q, An Z, Sun X, Tian H. Circulating betatrophin in patients with type 2 diabetes: a meta-analysis. J Diabetes Res. 2016;2016:6194750. doi: 10.1155/2016/6194750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Abu-Farha M, Abubaker J, Al-Khairi I, Cherian P, Noronha F, Kavalakatt S, Khadir A, Behbehani K, Alarouj M, Bennakhi A, Elkum N. Circulating angiopoietin-like protein 8 (betatrophin) association with HsCRP and metabolic syndrome. Cardiovasc Diabetol. 2016;15:25. doi: 10.1186/s12933-016-0346-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Lee YH, Lee SG, Lee CJ, Kim SH, Song YM, Yoon MR, Jeon BH, Lee JH, Lee BW, Kang ES, Lee HC, Cha BS. Association between betatrophin/ANGPTL8 and non-alcoholic fatty liver disease: animal and human studies. Sci Rep. 2016;6:24013. doi: 10.1038/srep24013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Ren G, Kim JY, Smas CM. Identification of RIFL, a novel adipocyte-enriched insulin target gene with a role in lipid metabolism. Am J Physiol Endocrinol Metab. 2012;303:E334–E351. doi: 10.1152/ajpendo.00084.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fu Z, Abou-Samra AB, Zhang R. An explanation for recent discrepancies in levels of human circulating betatrophin. Diabetologia. 2014;57:2232–2234. doi: 10.1007/s00125-014-3346-1. [DOI] [PubMed] [Google Scholar]