Abstract
Background
The clinical utility of ankle-brachial index (ABI) is not clear in subjects with less severe or calcified vessel. Therefore, we investigated the usefulness of color Doppler ultrasonography for diagnosing peripheral artery disease (PAD) in type 2 diabetes mellitus (T2DM) subjects.
Methods
We analyzed 324 T2DM patients who concurrently underwent ABI and carotid intima-media thickness (CIMT) measurements and color Doppler ultrasonography from 2003 to 2006. The degree of stenosis in patients with PAD was determined according to Jager's criteria, and PAD was defined as grade III (50% to 99% stenosis) or IV stenosis (100% stenosis) by color Doppler ultrasonography. Logistic regression analysis and receiver operating characteristic curve analysis were performed to evaluate the risk factors for PAD in patients with ABI 0.91 to 1.40.
Results
Among the 324 patients, 77 (23.8%) had ABI 0.91 to 1.40 but were diagnosed with PAD. Color Doppler ultrasonography demonstrated that suprapopliteal arterial stenosis, bilateral lesions, and multivessel involvement were less common in PAD patients with ABI 0.91 to 1.40 than in those with ABI ≤0.90. A multivariate logistic regression analysis demonstrated that older age, current smoking status, presence of leg symptoms, and high CIMT were significantly associated with the presence of PAD in patients with ABI 0.91 to 1.40 after adjusting for conventional risk factors. CIMT showed significant power in predicting the presence of PAD in patients with ABI 0.91 to 1.40.
Conclusion
Color Doppler ultrasonography is a useful tool for the detection of PAD in T2DM patients with ABI 0.91 to 1.40 but a high CIMT.
Keywords: Ankle brachial index; Diabetes mellitus, type 2; Peripheral arterial disease; Ultrasonography, Doppler, color
INTRODUCTION
Peripheral artery disease (PAD) is a condition characterized by atherosclerotic occlusive disease of the lower extremities. The prevalence of PAD is increased with advanced age, cigarette smoking, diabetes mellitus (DM), dyslipidemia, and hypertension [1,2,3]. PAD is a major risk factor for coronary and cerebrovascular events, including myocardial infarction, stroke, and death, secondary to systemic atherothrombosis [3,4]. PAD is rarely an isolated condition, but rather a manifestation of systemic atherosclerosis in other vascular beds, including the coronary and carotid arteries in patients with DM [3,5]. PAD is 3- to 4-fold higher in incidence and more extensive but less symptomatic in patients with DM compared with normal subjects [1,6]. Notably, because the rate of limb amputation due to PAD is significantly higher in patients with DM [7], early detection of asymptomatic PAD in patients with DM has important implications in clinical practice.
Although non-invasive and simple diagnostic tests are readily available, PAD remains significantly underestimated and untreated [8], mostly because of the paucity of symptoms and underutilization of screening tools. The American Diabetes Association Consensus Statement recommends that a screening ankle-brachial index (ABI) should be performed in diabetes patients with symptoms or signs of PAD [9]. However, the accuracy of the ABI is controversial when used to evaluate calcified and poorly compressible vessels in elderly patients with medial sclerosis. The ABI may also be falsely negative in symptomatic patients with moderate aortoiliac stenosis or extensive collaterals [10]. Therefore, various imaging tools, such as color Doppler ultrasonography, magnetic resonance angiography (MRA), and computed tomography angiography (CTA) have been used to diagnose suspicious PAD despite a normal ABI [11]. Although all of these tests are non-invasive, color Doppler ultrasonography provides renal protection because contrast dye is not used, compared with MRA or CTA. A previous study also reported that hand-held Doppler has the greatest diagnostic accuracy in patients with PAD, while clinical findings including claudication, femoral bruit, and pulse abnormalities are insufficient to diagnose PAD [12].
Carotid intima-media thickness (CIMT), which is easily assessed by B-mode ultrasound, is an indicator of generalized atherosclerosis and a potentially effective tool to predict vascular complications in patients with DM [13]. In fact, an increased CIMT is related to a higher risk of PAD detected by ABI and claudication [14,15].
This study assessed the utility of color Doppler ultrasonography to detect PAD in type 2 diabetes mellitus (T2DM) patients with ABI 0.91 to 1.40. In addition, we investigated the usefulness of CIMT in predicting the presence of PAD in DM patients with ABI 0.91 to 1.40.
METHODS
Subjects
This was a single center, retrospective study performed at Huh's Diabetes Center in Seoul, Republic of Korea. Among the patients who visited this clinic from January 2003 to December 2006, we analyzed 324 T2DM patients over 50 years of age, and their vascular stenosis was concurrently evaluated by the ABI, color Doppler ultrasonography, and CIMT. Subjects who had a previous history of ketoacidosis, serum fasting C-peptide level ≤0.5 ng/mL, history of cardiovascular disease (CVD) including stroke, myocardial infarction, any coronary revascularization or surgery due to chest pain and hospitalization due to heart failure, history of PAD, or a diagnosis of an orthopedic disease in the lumbar spine were excluded. All subjects were categorized into three groups according to their leg symptoms: the asymptomatic, typical, and atypical symptomatic groups. The typical symptomatic group included patients with symptomatic claudication, resting pain, and necrosis or gangrene, which are compatible with typical symptoms in the Fontaine staging system [16]. The atypical symptomatic group included patients with vague pain, which was difficult to distinguish from symptoms of peripheral neuropathy. This study was approved by the Institutional Review Board of Yonsei University (IRB No. 4-2017-0288). Due to the retrospective nature of this study, the requirement for written informed consent was waived.
Measurement of anthropometric and biochemical parameters
Height (cm) and body weight (kg) were measured to the nearest 0.1 cm and 0.1 kg, respectively, in barefoot subjects wearing light clothing. Body mass index (BMI) was calculated as weight divided by height squared (kg/m2). Waist circumference was measured to the nearest 0.1 cm at the narrowest point between the lower limit of the ribcage and the iliac crest. Blood pressure (BP) was recorded using a mercury sphygmomanometer on the right arm with the subject in the sitting position. Two readings were taken 5 minutes apart, and the mean value was used. All patients were asked about their smoking history and were classified as never-smokers, ex-smokers, or current smokers using a structured questionnaire. Subjects were also asked to refrain from smoking for 24 hours and from consuming alcohol for 7 days before the blood sampling.
After a 14-hour overnight fast, venous blood samples were drawn from the antecubital vein between 7:00 AM and 9:00 AM. Plasma was separated immediately by centrifugation (2,000 rpm, 20 minutes, at 4℃), and biochemical measurements were determined immediately. Fasting plasma concentrations of glucose, total cholesterol, triglycerides, and high density lipoprotein cholesterol and low density lipoprotein cholesterol were measured enzymatically using the Hitachi 747 chemical analyzer (Hitachi, Tokyo, Japan). The glycosylated hemoglobin (HbA1c) value was determined using a high-performance liquid chromatography method (Varient II; Green-cross, Seoul, Korea). Fasting and 2-hour postprandial C-peptide levels were measured by double-antibody radioimmunoassay (DiaSorin, Stillwater, MN, USA).
Ankle-brachial index
A non-invasive screening device (VP-1000; Colin Medical Technology Corp., Komaki, Japan) was used to measure the ABI. All patients were allowed to rest for at least 5 minutes before the measurements. The American College of Cardiology/American Heart Association (ACC/AHA) guidelines [17] recommend selecting the higher of the two arm pressures (brachial) and the higher of the two ankle pressures (anterior tibial/dorsalis pedis or posterior tibial) to calculate the ABI. The lower of the two ABI values was used for the analysis. According to the ACC/AHA recommendations the ABI value was classified as follows: normal, 1.00 to 1.40; borderline, 0.91 to 0.99; abnormal, ≤0.90 (mild, 0.70 to 0.90; moderate, 0.40 to 0.69; severe, <0.40); and poorly compressible >1.40 [17].
Peripheral color Doppler ultrasonography
All Doppler studies were performed by a single radiologist using color Doppler ultrasonography (Logic 7; General Electronics, Milwaukee, WI, USA). Information on the presence and grade of stenosis, post-stenotic turbulence, peak systolic velocity, end diastolic velocity, and the presence of collaterals and their flow characteristics were assessed on color Doppler imaging. The degree of stenosis was graded according to Jager's criteria. Normal, triphasic waveform with thin spectral band. Grade I, 1% to 19% stenosis: normal triphasic flow with normal peak systolic velocity with spectral broadening. Grade II, 20% to 49% stenosis: triphasic waveform with an increase in peak systolic velocity ≥30% with respect to the proximal recording site. Marked spectral broadening. Grade III, 50% to 99% stenosis: monophasic waveform with an increase in peak systolic velocity ≥100% and marked spectral broadening. Distal waveform is abnormal. Grade IV, 100% stenosis: no forward flow detected with altered flow patterns both proximal and distal to the stenosis [18]. Grade III and IV stenoses were defined as PAD in this study.
Carotid intima-media thickness
Both carotid arteries were scanned by a single experienced examiner using high-resolution real-time B mode ultrasonography with a 10 MHZ probe (LOGIQ 7; General Electronics), as previously described [19]. The CIMT was defined as the distance between the lumen-intima and media-adventitia interfaces [20]. CIMT was measured at three points on the far wall in sections (1 cm long) of the common carotid artery in the proximity of the carotid bulb. The mean of six measurements from the right and left carotid arteries was used as the mean CIMT [21].
Statistical analysis
Normally distributed data are expressed as mean±standard deviation, and categorical variables are expressed as percentages. One-way analysis of variance or Student t-test was used to compare continuous variables, and the chi-square test was used to compare categorical variables. Logistic regression analyses were performed using the enter method to identify the risk factors for PAD in patients with ABI 0.91 to 1.40. Receiver operating characteristic (ROC) analysis was performed to assess the utility of CIMT in predicting PAD in symptomatic and asymptomatic patients with ABI 0.91 to 1.40. All statistical tests were two-sided, and analyses were executed using SPSS for Windows version 23.0 (IBM Co., Armonk, NY, USA). A P value <0.05 was considered significant.
RESULTS
Baseline clinical characteristics
A total of 324 patients were analyzed in this study (male: female=192:132). The mean age of all subjects was 62.5±7.3 years, and BMI was 24.5±2.9 kg/m2. Mean DM duration was 11.4±7.3 years, and the mean HbA1c was 8.4%±1.8%.
Table 1 shows the clinical characteristics of the subjects with and without PAD. The PAD group was older, more likely male, and lower in BMI. The PAD group had more current smokers and a longer duration of DM. Other metabolic parameters, including fasting plasma glucose levels, lipid levels, and waist circumference, did not differ between the groups. Systolic and diastolic BP was higher, and leg symptoms were more common, in the PAD group. The proportions of abnormal or borderline ABI were higher in PAD group, compared with non-PAD group. The mean CIMT was also significantly higher in the PAD group, compared with non-PAD group.
Table 1. Clinical characteristics of type 2 diabetes mellitus patients with and without PAD diagnosed by color Doppler ultrasonography (n=324).
| Characteristic | Total | PAD (−) | PAD (+) | P value |
|---|---|---|---|---|
| No. of patients | 324 | 231 (71.3) | 93 (28.7) | |
| Age, yr | 62.5±7.3 | 61.2±6.9 | 65.8±7.2 | <0.001 |
| Male sex | 192 (59.3) | 129 (55.8) | 63 (67.7) | 0.049 |
| BMI, kg/m2 | 24.5±2.9 | 24.7±2.9 | 23.9±2.7 | 0.018 |
| Waist circumference, cm | 84.9±7.6 | 85.1±7.9 | 84.5±6.9 | 0.562 |
| Systolic BP, mm Hg | 141.4±19.3 | 139.9±18.4 | 145.1±21.1 | 0.028 |
| Diastolic BP, mm Hg | 87.1±11.0 | 85.0±12.2 | 87.9±10.4 | 0.031 |
| DM duration, yr | 11.4±7.3 | 10.5±6.6 | 13.5±8.4 | 0.001 |
| Current smoker | 52 (16.0) | 31 (13.4) | 21 (22.6) | 0.002 |
| Use of insulin | 59 (18.2) | 37 (16.0) | 22 (23.7) | 0.107 |
| Use of statins | 50 (15.4) | 33 (14.3) | 17 (18.3) | 0.368 |
| Use of anti-hypertensive drugs | 110 (34.0) | 76 (32.9) | 34 (36.6) | 0.529 |
| HbA1c, % | 8.4±1.8 | 8.4±1.8 | 8.5±1.7 | 0.741 |
| Fasting glucose, mg/dL | 163.3±57.0 | 164.1±56.1 | 161.4±59.6 | 0.701 |
| Total cholesterol, mg/dL | 193.4±41.5 | 193.1±42.6 | 194.2±38.7 | 0.831 |
| Triglycerides, mg/dL | 148.5±95.1 | 153.6±107.0 | 135.8±53.3 | 0.131 |
| HDL-C, mg/dL | 49.6±14.5 | 50.3±14.7 | 48.0±13.8 | 0.183 |
| LDL-C, mg/dL | 113.5±35.5 | 111.4±36.5 | 118.9±32.5 | 0.103 |
| Leg symptoms (yes) | ||||
| Typical | 43 (13.3) | 6 (2.6) | 37 (39.8) | <0.001 |
| Atypical | 31 (9.6) | 19 (8.2) | 12 (12.9) | 0.006 |
| ABI | ||||
| ≤0.90 (abnormal) | 18 (5.6) | 2 (11.1) | 16 (88.9) | <0.001 |
| 0.91–0.99 (borderline) | 15 (4.6) | 7 (46.7) | 8 (53.3) | 0.010 |
| Mean CIMT, mm | 0.94±0.23 | 0.90±0.19 | 1.03±0.27 | <0.001 |
Values are presented as number (%) or mean±standard deviation.
PAD, peripheral artery disease; BMI, body mass index; BP, blood pressure; DM, diabetes mellitus; HbA1c, glycosylated hemoglobin; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; ABI, ankle-brachial index; CIMT, carotid intima-media thickness.
Comparison of anatomical characteristics between the patients with ABI 0.91 to 1.40 vs. ≤0.90 among patients with PAD
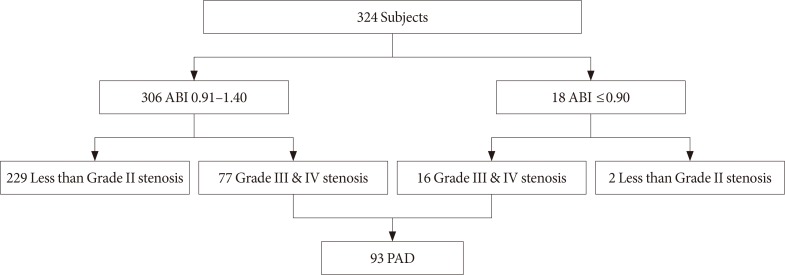
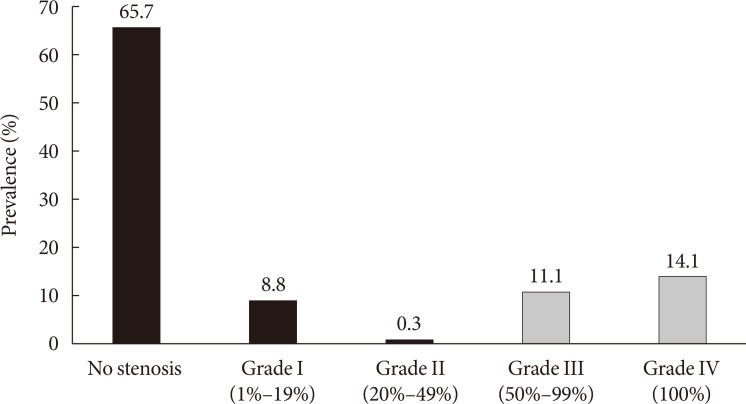
The clinical presentation of PAD according to the ABI and color Doppler ultrasonography results is detailed in Fig. 1. Among the 324 patients, only 18 (5.6%) had an abnormally low ABI (≤0.90) and the other 306 (94.4%) had ABI 0.91 to 1.40. However, color Doppler ultrasonography demonstrated that 93 (28.7%) had PAD. Among the 93 patients diagnosed with PAD by Doppler ultrasonography, only 16 (17.2%) had an abnormally low ABI, and 77 (82.8%) had ABI 0.91 to 1.40. Among the 306 patients with ABI 0.91 to 1.40, 34 (11.1%) had grade III (50% to 99% stenosis) lesions, and 43 (14.1%) had grade IV (100% stenosis) lesions on color Doppler ultrasonography (Fig. 2). These results suggest that the ABI has the potential to underestimate the presence of PAD.
Fig. 1. Flow chart presenting the study subjects. ABI, ankle-brachial index; PAD, peripheral artery disease.
Fig. 2. Degree of stenosis on color Doppler ultrasonography in patients with ankle-brachial index 0.91 to 1.40 (n=306). Grade III and IV stenoses were defined as peripheral artery disease and expressed with gray bars.
Next, we compared the characteristics of the stenotic lesions between ABI 0.91 to 1.40 and ABI ≤0.90 among the patients with PAD (Table 2). Multiple stenotic lesions were less frequent in ABI 0.91 to 1.40 group than in ABI ≤0.90 group. Although stenotic lesions occurred most frequently in the infrapopliteal arteries, such as the anterior and posterior tibial arteries, in both groups, stenosis of the common femoral (14.3% vs. 37.5%, P=0.029) or superficial femoral (22.1% vs. 32.0%, P=0.022) arteries was less frequent in ABI 0.91 to 1.40 group than in ABI ≤0.90 group. Bilateral stenosis was also less common in ABI 0.91 to 1.40 group than in ABI ≤0.90 group (64.9% vs. 93.8%, P=0.022). Collectively, multiple, proximal, and bilateral arterial stenoses were less common in ABI 0.91 to 1.40 group than in ABI ≤0.90 group, suggesting that the ABI alone potentially underestimates PAD if the stenotic lesions are less extensive, distal, or unilateral.
Table 2. Comparison of anatomical characteristics of patients with ABI 0.91 to 1.40 vs. ABI ≤0.90 among the patients diagnosed with PAD by color Doppler ultrasonography (n=93).
| Anatomical characteristic | ABI | ||
|---|---|---|---|
| 0.91–1.40 | ≤0.90 | P value | |
| No. of patients | 77 (82.8) | 16 (17.2) | |
| No. of stenoses | 2 (1–4) | 4 (2–5) | 0.005 |
| Location of stenosis | |||
| Common iliac artery | 8 (10.4) | 3 (18.8) | 0.346 |
| Common femoral artery | 11 (14.3) | 6 (37.5) | 0.029 |
| Superficial femoral artery | 17 (22.1) | 8 (32.0) | 0.022 |
| Deep femoral artery | 10 (13.0) | 4 (25.0) | 0.221 |
| Popliteal artery | 11 (14.3) | 5 (31.3) | 0.102 |
| Anterior tibial artery | 53 (68.8) | 13 (81.3) | 0.319 |
| Posterior tibial artery | 42 (54.5) | 10 (62.5) | 0.560 |
| Bilateral lesions | 50 (64.9) | 15 (93.8) | 0.022 |
Values are presented as number (%) or median (interquartile range).
ABI, ankle-brachial index; PAD, peripheral artery disease.
Comparison of clinical characteristics between the patients with ABI 0.91 to 1.40 vs. ≤0.90 among the patients with PAD
We compared the clinical characteristics between patients with ABI 0.91 to 1.40 versus ≤0.90 among patients with PAD (Table 3). No significant differences in age, sex, or BMI were observed between the groups. In addition, no significant differences in BP, HbA1c, lipid profile, or smoking history were observed between the groups. However, ABI ≤0.90 group had a longer duration of DM, more frequent insulin use, and more frequent leg symptoms compared with those in ABI 0.91 to 1.40 group. Although the mean CIMT was significantly higher in patients with PAD than in those without PAD (1.03±0.27 mm vs. 0.90±0.19 mm, P<0.001) (Table 1), no difference in CIMT was observed between the patients with ABI 0.91 to 1.40 and ≤0.90 among the patients with PAD (Table 3).
Table 3. Comparison of the clinical characteristics of patients with ABI 0.91 to 1.40 vs. ABI ≤0.90 among the patients diagnosed with PAD by color Doppler ultrasonography (n=93).
| Variable | ABI | ||
|---|---|---|---|
| 0.91–1.40 | ≤0.90 | P value | |
| No. of patients | 77 (82.8) | 16 (17.2) | |
| Age, yr | 65.2±7.0 | 68.6±8.0 | 0.084 |
| Male sex | 51 (66.2) | 12 (75.0) | 0.495 |
| BMI, kg/m2 | 24.0±2.7 | 23.1±2.5 | 0.204 |
| Waist circumference, cm | 84.9±7.2 | 82.9±4.8 | 0.324 |
| Systolic BP, mm Hg | 144.1±21.7 | 149.8±17.3 | 0.329 |
| Diastolic BP, mmHg | 81.1±11.1 | 85.8±12.3 | 0.163 |
| DM duration, yr | 12.4±8.0 | 18.9±8.5 | 0.004 |
| Current smoker | 18 (23.4) | 3 (18.8) | 0.687 |
| Use of insulin | 15 (19.5) | 7 (43.8) | 0.038 |
| Use of statins | 12 (15.6) | 5 (31.3) | 0.140 |
| Use of anti-hypertensive drugs | 27 (35.1) | 7 (43.8) | 0.512 |
| HbA1c, % | 8.4±1.6 | 9.1±1.9 | 0.099 |
| Fasting glucose, mg/dL | 156.3±53.9 | 185.8±79.6 | 0.071 |
| Total cholesterol, mg/dL | 193.6±40.2 | 196.7±31.6 | 0.776 |
| Triglycerides, mg/dL | 137.4±55.7 | 129.2±42.0 | 0.581 |
| HDL-C, mg/dL | 48.4±14.0 | 45.9±13.3 | 0.511 |
| LDL-C, mg/dL | 117.6±33.0 | 125.5±30.2 | 0.428 |
| Leg symptoms (typical+atypical) | 37 (48.1) | 12 (75.0) | 0.044 |
| Mean CIMT, mm | 1.02±0.27 | 1.10±0.24 | 0.247 |
Values are presented as number (%) or mean±standard deviation.
ABI, ankle-brachial index; PAD, peripheral artery disease; BMI, body mass index; BP, blood pressure; DM; diabetes mellitus; HbA1c, glycosylated hemoglobin; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; CIMT, carotid intima-media thickness.
Risk factors associated with PAD among the patients with ABI 0.91 to 1.40
The ABI is a more accessible tool for screening PAD in the clinical setting than is color Doppler ultrasonography. However, it has the potential to underestimate the presence of PAD, which may delay proper management. If we could find out the characteristics of the patients with PAD who had ABI 0.91 to 1.40, further vascular evaluation could be recommended for them. Thus, we investigated the risk factors associated with the presence of PAD in the 306 patients with ABI 0.91 to 1.40. Univariate analysis demonstrated that older age, a longer duration of DM, current smoking status, leg symptoms, and a higher mean CIMT were associated with the presence of PAD in patients with ABI 0.91 to 1.40 (Table 4). In the multivariate analysis, we adjusted for age, sex, BMI, systolic BP, DM duration, current smoking status, HbA1c, total cholesterol, presence of leg symptoms, and mean CIMT (Table 4). The results demonstrated that age per 10 years (odds ratio [OR], 1.86; 95% confidence interval [CI], 1.18 to 2.93; P=0.008), current smoking status (OR, 2.62; 95% CI, 1.10 to 6.21; P=0.029), leg symptoms (OR, 7.35; 95% CI; 3.71 to 14.57; P<0.001), and mean CIMT (OR, 4.73; 95% CI, 1.14 to 19.57; P=0.032) were significantly associated with the presence of PAD in these patients (Table 4). After adjusting for age as a categorical variable in the multivariate analysis, age ≥70 years was significantly associated with a higher risk of PAD (OR, 3.08; 95% CI, 1.23 to 7.69; P=0.016) compared with age <60 years. There was no difference in the risk of PAD between the 60 to 69 and <60-year age groups (OR, 2.04; 95% CI, 0.97 to 4.30; P=0.061) (data not shown).
Table 4. Risk factors associated with the presence of PAD in 306 patients with ABI 0.91 to 1.40.
| Variable | Univariate analysis | Multivariate analysisa | ||
|---|---|---|---|---|
| OR (95% CI) | P value | OR (95% CI) | P value | |
| Age (per 10 yr) | 2.23 (1.51–3.28) | <0.001 | 1.86 (1.18–2.93) | 0.008 |
| Male sex | 1.55 (0.90–2.66) | 0.113 | 1.07 (0.53–2.21) | 0.853 |
| BMI, kg/m2 | 0.92 (0.84–1.01) | 0.090 | 0.92 (0.82–1.03) | 0.158 |
| Waist circumference, cm | 1.00 (0.96–1.03) | 0.877 | ||
| Systolic BP (per 10 mm Hg) | 1.12 (0.98–1.28) | 0.096 | 1.02 (0.99–1.06) | 0.085 |
| Diastolic BP (per 10 mm Hg) | 0.84 (0.66–1.06) | 0.145 | ||
| DM duration, yr | 1.04 (1.01–1.07) | 0.042 | 1.02 (0.98–1.06) | 0.402 |
| Current smoker | 1.95 (1.02–3.73) | 0.044 | 2.62 (1.10–6.21) | 0.029 |
| Use of insulin (yes) | 1.30 (0.67–2.53) | 0.445 | ||
| Use of statins (yes) | 1.10 (0.54–2.25) | 0.801 | ||
| Use of anti-hypertensive drugs (yes) | 1.09 (0.63–1.87) | 0.763 | ||
| HbA1c, % | 0.98 (0.85–1.14) | 0.807 | 0.89 (0.74–1.07) | 0.222 |
| Fasting glucose, mg/dL | 1.00 (0.99–1.00) | 0.303 | ||
| Total cholesterol, mg/dL | 1.00 (0.99–1.01) | 0.932 | 1.00 (0.99–1.01) | 0.783 |
| Triglycerides, mg/dL | 1.00 (0.99–1.00) | 0.193 | ||
| HDL-C, mg/dL | 0.99 (0.97–1.01) | 0.306 | ||
| LDL-C, mg/dL | 1.01 (0.99–1.01) | 0.200 | ||
| Leg symptoms (yes) | 7.55 (4.10–13.89) | <0.001 | 7.35 (3.71–14.57) | <0.001 |
| Mean CIMT, mm | 10.46 (3.12–35.03) | <0.001 | 4.73 (1.14–19.57) | 0.032 |
PAD, peripheral artery disease; ABI, ankle-brachial index; OR, odds ratio; CI, confidence interval; BMI, body mass index; BP, blood pressure; DM, diabetes mellitus; HbA1c, glycosylated hemoglobin; HDL-C, high density lipoprotein cholesterol; LDL-C, low density lipoprotein cholesterol; CIMT, carotid intima-media thickness.
aMultivariate model was adjusted for age, sex, BMI, systolic BP, DM duration, current smoking status, HbA1c level, total cholesterol level, leg symptoms, and mean CIMT.
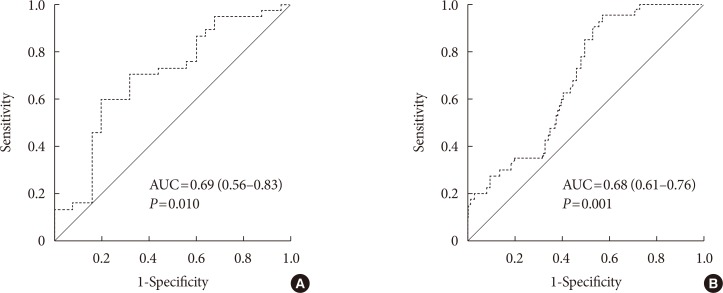
The cut-off CIMT value to predict the presence of PAD was 0.92 mm, with a 62.3% sensitivity and 59.0% specificity. ROC analysis demonstrated that the area under the curve was 0.69 (95% CI, 0.56 to 0.83; P=0.010) for the symptomatic patients (Fig. 3A) and 0.68 (95% CI, 0.61 to 0.76; P=0.001) for the asymptomatic patients (Fig. 3B). Collectively, these results suggest that older age (≥70 years), current smoking status, presence of leg symptoms, high mean CIMT (>0.92 mm) were factors associated with the presence of PAD, even in patients with ABI 0.91 to 1.40.
Fig. 3. Receiver operating characteristic curves for carotid intima-media thickness (CIMT) for predicting peripheral artery disease (PAD) in 306 patients with ankle-brachial index 0.91 to 1.40. (A) The area under the curve for CIMT for predicting PAD in symptomatic type 2 diabetes mellitus (T2DM) patients (n=62). (B) The area under the curve (AUC) for CIMT for predicting PAD in asymptomatic T2DM patients (n=244).
DISCUSSION
PAD is strongly associated with cardiovascular mortality and lower extremity amputation in patients with DM. The use of ABI is commonly recommended in screening for PAD in patients with DM. However, our study demonstrated that 25.2% of patients with DM and ABI 0.91 to 1.40 were diagnosed with PAD by color Doppler ultrasonography. Older age, current smoking status, presence of leg symptoms, and high CIMT were significantly associated with the presence of PAD in patients with ABI 0.91 to 1.40.
The prevalence of PAD detected by the ABI was previously reported to be 20% in a population with DM [22], and reached 29% in older diabetes patients over 50 years of age [23]. An Asian study (PAD-SEARCH) showed that the prevalence of PAD was 17.7%, but this rate varied (12% to 32%) depending on the region. The prevalence of PAD in Koreans over 50 years of age was 12% from the 1980s to the 1990s, which is lower compared with other regions [24]. The difference may be associated with the heterogeneity of the study participants as well as the limitations of the ABI, which can underestimate the prevalence of PAD. Our results also demonstrate different prevalence rates of PAD according to the detection method used (5.6% by ABI vs. 28.7% by color Doppler ultrasonography).
Among subjects who have ABI 0.91 to 1.40 but are diagnosed with PAD, only 39.8% showed typical leg symptoms of PAD in this study. This result supports a previous study that the proportion of asymptomatic PAD is significantly higher among subjects with DM than among normal subjects [25]. Approximately 27% of patients with PAD demonstrate a progression of symptoms over a 5-year period, with limb loss occurring in 4% [26]. Patients with DM and claudication show an overall 20% amputation risk and a 50% 5-year mortality rate [27]. Considering the impact of PAD on the mortality and morbidity of patients with DM, early diagnosis and proper management of PAD are important, even in asymptomatic patients.
ABI value of 0.91 to 0.99 is defined as a borderline [17] and is known to be associated with an increased risk of PAD [28], endothelial dysfunction [29], and hard CVD events [30,31], compared with a normal ABI (1.00 to 1.40). Our study also showed that the proportion of borderline ABI was higher in PAD group compared with non-PAD group (10.4% vs. 3.1%, P=0.010) in Table 1. However, among 77 patients who have ABI 0.91 to 1.40 and PAD, only eight patients (53.3%) had a borderline ABI and the other 69 patients (23.7%) had a normal ABI. This results suggests that not only a borderline ABI but also a normal ABI cannot exclude the possibility of detecting PAD by color Doppler ultrasonography. Since the ABI has limited sensitivity and a large number of DM patients are asymptomatic, we may consider the further vascular evaluation to detect PAD for not only for the symptomatic but also for the asymptomatic patients who have ABI 0.91 to 1.40 and known risk factors.
Color Doppler ultrasonography is a good vascular imaging technology because of its noninvasiveness, safety, and accuracy [32]. It visualizes vessels directly and provides data regarding anatomical localization, the degree of stenosis, and hemodynamics. One study reported that color Doppler ultrasonography has an 80% to 98% sensitivity and 89% to 99% specificity for diagnosis of PAD [33]. A previous study reported that the prevalence of PAD, as detected by color Doppler ultrasonography, was 24% in patients with DM, male predominance, and a tendency for multisegmental and infrapopliteal artery involvement [34].
CIMT measured by high-resolution ultrasonography is a good marker of generalized atherosclerosis [35,36]. CIMT was positively associated with the prevalence of PAD [37], and found to be an independent predictor of peripheral revascularization [38]. Our study also reported that a high CIMT was significantly associated with the presence of PAD in patients with ABI 0.91 to 1.40, independent of leg symptoms or known risk factors such as age, sex, smoking, hypertension, and lipid levels. Thus, color Doppler ultrasonography may be useful for detecting PAD in patients with ABI 0.91 to 1.40 and a high CIMT, regardless of leg symptoms.
This study had several limitations that should be considered. First, the subjects may not be representative of the general population, because they were enrolled from a single diabetes center, and may have had asymptomatic CVD that was not excluded at the enrollment. Second, we could not specify orthopedic diseases in the lumbar spine such as spinal stenosis, any spinal operation, or compression fracture due to lack of the data. Lastly, there were also lack of data on the use of anti-platelet agents, changes in statin or anti-hypertensive medications, even though these factors may affect vascular function. However, the strength of this study is that we identified the utility of color Doppler ultrasonography in detecting PAD, especially in patients with a normal or borderline ABI.
In conclusion, color Doppler ultrasonography could be useful for the detection of PAD among patients with DM and a normal or borderline ABI. Color Doppler ultrasonography could be considered for not only symptomatic but also asymptomatic patients who have ABI 0.91 to 1.40 and a high CIMT.
ACKNOWLEDGMENTS
This work was supported by a grant from the Seoul R&BD Program, Republic of Korea (10526). Kyu Yeon Hur is the recipient of grants from Samsung Medical Center (#CRP1500057).
Footnotes
CONFLICTS OF INTEREST: No potential conflict of interest relevant to this article was reported.
References
- 1.Selvin E, Erlinger TP. Prevalence of and risk factors for peripheral arterial disease in the United States: results from the National Health and Nutrition Examination Survey, 1999-2000. Circulation. 2004;110:738–743. doi: 10.1161/01.CIR.0000137913.26087.F0. [DOI] [PubMed] [Google Scholar]
- 2.Eraso LH, Fukaya E, Mohler ER, 3rd, Xie D, Sha D, Berger JS. Peripheral arterial disease, prevalence and cumulative risk factor profile analysis. Eur J Prev Cardiol. 2014;21:704–711. doi: 10.1177/2047487312452968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Criqui MH, Aboyans V. Epidemiology of peripheral artery disease. Circ Res. 2015;116:1509–1526. doi: 10.1161/CIRCRESAHA.116.303849. [DOI] [PubMed] [Google Scholar]
- 4.Sigvant B, Hasvold P, Kragsterman B, Falkenberg M, Johansson S, Thuresson M, Nordanstig J. Cardiovascular outcomes in patients with peripheral arterial disease as an initial or subsequent manifestation of atherosclerotic disease: results from a Swedish nationwide study. J Vasc Surg. 2017;66:507–514. doi: 10.1016/j.jvs.2017.01.067. [DOI] [PubMed] [Google Scholar]
- 5.Olin JW, Sealove BA. Peripheral artery disease: current insight into the disease and its diagnosis and management. Mayo Clin Proc. 2010;85:678–692. doi: 10.4065/mcp.2010.0133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gregg EW, Sorlie P, Paulose-Ram R, Gu Q, Eberhardt MS, Wolz M, Burt V, Curtin L, Engelgau M, Geiss L 1999-2000 national health and nutrition examination survey. Prevalence of lower-extremity disease in the US adult population >=40 years of age with and without diabetes: 1999-2000 national health and nutrition examination survey. Diabetes Care. 2004;27:1591–1597. doi: 10.2337/diacare.27.7.1591. [DOI] [PubMed] [Google Scholar]
- 7.Shammas AN, Jeon-Slaughter H, Tsai S, Khalili H, Ali M, Xu H, Rodriguez G, Cawich I, Armstrong EJ, Brilakis ES, Banerjee S. Major limb outcomes following lower extremity endovascular revascularization in patients with and without diabetes mellitus. J Endovasc Ther. 2017;24:376–382. doi: 10.1177/1526602817705135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bhatt DL, Steg PG, Ohman EM, Hirsch AT, Ikeda Y, Mas JL, Goto S, Liau CS, Richard AJ, Rother J, Wilson PW REACH Registry Investigators. International prevalence, recognition, and treatment of cardiovascular risk factors in outpatients with atherothrombosis. JAMA. 2006;295:180–189. doi: 10.1001/jama.295.2.180. [DOI] [PubMed] [Google Scholar]
- 9.American Diabetes Association. 10. Microvascular complications and foot care: standards of medical care in diabetes-2018. Diabetes Care. 2018;41(Suppl 1):S105–S118. doi: 10.2337/dc18-S010. [DOI] [PubMed] [Google Scholar]
- 10.Dhaliwal G, Mukherjee D. Peripheral arterial disease: epidemiology, natural history, diagnosis and treatment. Int J Angiol. 2007;16:36–44. doi: 10.1055/s-0031-1278244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.American Diabetes Association. Peripheral arterial disease in people with diabetes. Diabetes Care. 2003;26:3333–3341. doi: 10.2337/diacare.26.12.3333. [DOI] [PubMed] [Google Scholar]
- 12.Khan NA, Rahim SA, Anand SS, Simel DL, Panju A. Does the clinical examination predict lower extremity peripheral arterial disease? JAMA. 2006;295:536–546. doi: 10.1001/jama.295.5.536. [DOI] [PubMed] [Google Scholar]
- 13.Sibal L, Agarwal SC, Home PD. Carotid intima-media thickness as a surrogate marker of cardiovascular disease in diabetes. Diabetes Metab Syndr Obes. 2011;4:23–34. doi: 10.2147/DMSO.S8540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bots ML, Hofman A, Grobbee DE. Common carotid intima-media thickness and lower extremity arterial atherosclerosis. The Rotterdam Study. Arterioscler Thromb. 1994;14:1885–1891. doi: 10.1161/01.atv.14.12.1885. [DOI] [PubMed] [Google Scholar]
- 15.Poredos P, Golob M, Jensterle M. Interrelationship between peripheral arterial occlusive disease, carotid atherosclerosis and flow mediated dilation of the brachial artery. Int Angiol. 2003;22:83–87. [PubMed] [Google Scholar]
- 16.Borssen B, Bergenheim T, Lithner F. The epidemiology of foot lesions in diabetic patients aged 15–50 years. Diabet Med. 1990;7:438–444. doi: 10.1111/j.1464-5491.1990.tb01420.x. [DOI] [PubMed] [Google Scholar]
- 17.Gerhard-Herman MD, Gornik HL, Barrett C, Barshes NR, Corriere MA, Drachman DE, Fleisher LA, Fowkes FG, Hamburg NM, Kinlay S, Lookstein R, Misra S, Mureebe L, Olin JW, Patel RA, Regensteiner JG, Schanzer A, Shishehbor MH, Stewart KJ, Treat-Jacobson D, Walsh ME. 2016 AHA/ACC guideline on the management of patients with lower extremity peripheral artery disease: executive summary. A report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Circulation. 2017;135:e686–e725. doi: 10.1161/CIR.0000000000000470. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jager KA, Phillips DJ, Martin RL, Hanson C, Roederer GO, Langlois YE, Ricketts HJ, Strandness DE., Jr Noninvasive mapping of lower limb arterial lesions. Ultrasound Med Biol. 1985;11:515–521. doi: 10.1016/0301-5629(85)90164-4. [DOI] [PubMed] [Google Scholar]
- 19.Jun JE, Choi YJ, Lee YH, Kim DJ, Park SW, Huh BW, Lee EJ, Jee SH, Hur KY, Choi SH, Huh KB. ApoB/ApoA-I ratio is independently associated with carotid atherosclerosis in type 2 diabetes mellitus with well-controlled LDL cholesterol levels. Korean J Intern Med. 2018;33:138–147. doi: 10.3904/kjim.2017.396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Touboul PJ, Hennerici MG, Meairs S, Adams H, Amarenco P, Bornstein N, Csiba L, Desvarieux M, Ebrahim S, Hernandez Hernandez R, Jaff M, Kownator S, Naqvi T, Prati P, Rundek T, Sitzer M, Schminke U, Tardif JC, Taylor A, Vicaut E, Woo KS. Mannheim carotid intima-media thickness and plaque consensus (2004-2006-2011). An update on behalf of the advisory board of the 3rd, 4th and 5th watching the risk symposia, at the 13th, 15th and 20th European Stroke Conferences, Mannheim, Germany, 2004, Brussels, Belgium, 2006, and Hamburg, Germany, 2011. Cerebrovasc Dis. 2012;34:290–296. doi: 10.1159/000343145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Park SW, Kim SK, Cho YW, Kim DJ, Song YD, Choi YJ, Huh BW, Choi SH, Jee SH, Cho MA, Lee EJ, Huh KB. Insulin resistance and carotid atherosclerosis in patients with type 2 diabetes. Atherosclerosis. 2009;205:309–313. doi: 10.1016/j.atherosclerosis.2008.12.006. [DOI] [PubMed] [Google Scholar]
- 22.Walters DP, Gatling W, Mullee MA, Hill RD. The prevalence, detection, and epidemiological correlates of peripheral vascular disease: a comparison of diabetic and non-diabetic subjects in an English community. Diabet Med. 1992;9:710–715. doi: 10.1111/j.1464-5491.1992.tb01878.x. [DOI] [PubMed] [Google Scholar]
- 23.Hirsch AT, Criqui MH, Treat-Jacobson D, Regensteiner JG, Creager MA, Olin JW, Krook SH, Hunninghake DB, Comerota AJ, Walsh ME, McDermott MM, Hiatt WR. Peripheral arterial disease detection, awareness, and treatment in primary care. JAMA. 2001;286:1317–1324. doi: 10.1001/jama.286.11.1317. [DOI] [PubMed] [Google Scholar]
- 24.Rhee SY, Guan H, Liu ZM, Cheng SW, Waspadji S, Palmes P, Tai TY, Suwanwalaikorn S, Kim YS PAD-SEARCH Study Group. Multi-country study on the prevalence and clinical features of peripheral arterial disease in Asian type 2 diabetes patients at high risk of atherosclerosis. Diabetes Res Clin Pract. 2007;76:82–92. doi: 10.1016/j.diabres.2006.07.029. [DOI] [PubMed] [Google Scholar]
- 25.Ghosh US, Datta S, Banerjee S. Asymptomatic peripheral arterial disease in type 2 diabetes mellitus: prevalence patterns and risk factor associations. Int J Diabetes Dev Ctries. 2011;31:229–238. [Google Scholar]
- 26.Weitz JI, Byrne J, Clagett GP, Farkouh ME, Porter JM, Sackett DL, Strandness DE, Jr, Taylor LM. Diagnosis and treatment of chronic arterial insufficiency of the lower extremities: a critical review. Circulation. 1996;94:3026–3049. doi: 10.1161/01.cir.94.11.3026. [DOI] [PubMed] [Google Scholar]
- 27.Antonopoulos S, Kokkoris S, Stasini F, Mylonopoulou M, Lepeniotis G, Mikros S, Korantzopoulos P, Giannoulis G. High prevalence of subclinical peripheral artery disease in Greek hospitalized patients. Eur J Intern Med. 2005;16:187–191. doi: 10.1016/j.ejim.2005.02.002. [DOI] [PubMed] [Google Scholar]
- 28.Natsuaki C, Inoguchi T, Maeda Y, Yamada T, Sasaki S, Sonoda N, Shimabukuro M, Nawata H, Takayanagi R. Association of borderline ankle-brachial index with mortality and the incidence of peripheral artery disease in diabetic patients. Atherosclerosis. 2014;234:360–365. doi: 10.1016/j.atherosclerosis.2014.03.018. [DOI] [PubMed] [Google Scholar]
- 29.Kajikawa M, Maruhashi T, Iwamoto Y, Iwamoto A, Matsumoto T, Hidaka T, Kihara Y, Chayama K, Nakashima A, Goto C, Noma K, Higashi Y. Borderline ankle-brachial index value of 0.91–0.99 is associated with endothelial dysfunction. Circ J. 2014;78:1740–1745. doi: 10.1253/circj.cj-14-0165. [DOI] [PubMed] [Google Scholar]
- 30.Tanaka S, Kaneko H, Kano H, Matsuno S, Suzuki S, Takai H, Otsuka T, Uejima T, Oikawa Y, Nagashima K, Kirigaya H, Sagara K, Yajima J, Sawada H, Aizawa T, Yamashita T. The predictive value of the borderline ankle-brachial index for long-term clinical outcomes: an observational cohort study. Atherosclerosis. 2016;250:69–76. doi: 10.1016/j.atherosclerosis.2016.05.014. [DOI] [PubMed] [Google Scholar]
- 31.Ovbiagele B. Association of ankle-brachial index level with stroke. J Neurol Sci. 2009;276:14–17. doi: 10.1016/j.jns.2008.08.016. [DOI] [PubMed] [Google Scholar]
- 32.de Vries M, Ouwendijk R, Flobbe K, Nelemans PJ, Kessels AG, Schurink GH, van der Vliet JA, Heijstraten FM, Cuypers PW, Duijm LE, van Engelshoven JM, Hunink MG, de Haan MW. Peripheral arterial disease: clinical and cost comparisons between duplex US and contrast-enhanced MR angiography. A multicenter randomized trial. Radiology. 2006;240:401–410. doi: 10.1148/radiol.2402050223. [DOI] [PubMed] [Google Scholar]
- 33.Collins R, Burch J, Cranny G, Aguiar-Ibanez R, Craig D, Wright K, Berry E, Gough M, Kleijnen J, Westwood M. Duplex ultrasonography, magnetic resonance angiography, and computed tomography angiography for diagnosis and assessment of symptomatic, lower limb peripheral arterial disease: systematic review. BMJ. 2007;334:1257. doi: 10.1136/bmj.39217.473275.55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Bembi V, Singh S, Singh P, Aneja GK, Arya TV, Arora R. Prevalence of peripheral arterial disease in a cohort of diabetic patients. South Med J. 2006;99:564–569. doi: 10.1097/01.smj.0000221624.68378.5d. [DOI] [PubMed] [Google Scholar]
- 35.Lundberg C, Hansen T, Ahlstrom H, Lind L, Wikstrom J, Johansson L. The relationship between carotid intima-media thickness and global atherosclerosis. Clin Physiol Funct Imaging. 2014;34:457–462. doi: 10.1111/cpf.12116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Lorenz MW, Markus HS, Bots ML, Rosvall M, Sitzer M. Prediction of clinical cardiovascular events with carotid intima-media thickness: a systematic review and meta-analysis. Circulation. 2007;115:459–467. doi: 10.1161/CIRCULATIONAHA.106.628875. [DOI] [PubMed] [Google Scholar]
- 37.Pradeepa R, Chella S, Surendar J, Indulekha K, Anjana RM, Mohan V. Prevalence of peripheral vascular disease and its association with carotid intima-media thickness and arterial stiffness in type 2 diabetes: the Chennai urban rural epidemiology study (CURES 111) Diab Vasc Dis Res. 2014;11:190–200. doi: 10.1177/1479164114524584. [DOI] [PubMed] [Google Scholar]
- 38.Bosevski M, Borozanov V, Tosev S, Peovska I, Meskovska-Bongard S, Georgievska-Ismail L. Predictors for peripheral and carotid revascularization in a population-based cohort with type 2 diabetes. Angiology. 2009;60:46–49. doi: 10.1177/0003319708317334. [DOI] [PubMed] [Google Scholar]