Abstract
Little is known about survival and hospitalization among alternative-regimen hemodialysis (HD) users compared to thrice-weekly conventional HD patients who have similar characteristics and medical histories. We conducted a cohort study of alternative-regimen HD users and propensity score (PS)-matched controls. Collaborating clinicians identified 101 patients in their programs who used nocturnal HD (NHD) and 44 patients who used short daily HD (SDHD) for 60 days or more. Ten PS-matched control patients for each NHD and SDHD patient were identified from the United States Renal Data System database. Primary outcomes were risk for all-cause mortality and risk for the composite outcome of mortality or major morbid event (AMI or stroke), investigated in Cox proportional hazards models. Risks for all-cause, cardiovascular-related, infection-related, and vascular access-related hospital admissions were also explored. NHD was associated with reduced mortality risk (HR 0.34; 95% CI, 0.21-0.58; P < 0.0001) and with reduced risk for mortality or major morbid event (HR 0.49; 95% CI, 0.31-0.78; P = 0.003) compared to controls. There was a reduced but non-significant risk of death for patients using SDHD compared to controls (HR 0.61; 95% CI, 0.30-1.24; P = 0.17). All-cause and specific hospitalizations did not differ significantly between NHD and SDHD patients and their matched control cohorts. This study provides additional evidence that NHD may improve patient survival.
Keywords: Daily hemodialysis, hospitalization, survival, USRDS
In the United States, the number of programs offering nocturnal HD therapy (NHD) is reported to have increased from 73 in 2004 to 300 in 2008, and the number of programs offering short-duration daily HD therapy (SDHD) is reported to have increased from 37 in 2004 to 480 in 2008.2 There is great interest in the outcomes of patients undergoing these alternative HD regimens, which are believed to be more physiological and better tolerated by patients than conventional thrice-weekly HD.
Alternative HD regimens have the potential to increase longevity and reduce hospitalization as compared to conventional HD (CHD). Unfortunately, selection bias has confounded many reports on outcomes associated with alternative HD regimens.3-8 There is little information about survival and hospitalization outcomes among users of alternative HD regimens compared to controls who have similar demographic characteristics and medical histories.
We used the USRDS database to examine mortality and hospitalization outcomes for patients from multiple programs who used NHD or SDHD for at least 60 days, compared to outcomes for patients with similar characteristics treated by thrice-weekly CHD who were identified by propensity score (PS) matching from the USRDS database. We examined outcomes separately for patients using NHD and SDHD because these are not equivalent therapies.9 The primary research question was the risk for mortality among NHD users compared to CHD controls and among SDHD users compared to CHD controls. We also investigated a composite outcome of risk for mortality or a major morbid event, i.e. acute myocardial infarction (AMI) or stroke, as recommended by Chertow,1 and explored risks for all-cause and specific types of hospitalization.
Results
Approximately 60% of both NHD and SDHD patients dialyzed 6 days/week, with most of the remaining patients dialyzing 5 days/week (Table 1). All NHD and SDHD patients dialyzed at home; 1% of CHD patients used home HD as of January 1, 2003. Most patients converted to an alternative HD regimen from conventional center HD or conventional home HD; two patients converted from continuous ambulatory peritoneal dialysis and continuous cycling peritoneal dialysis, respectively. The mean age at which patients switched to NHD therapy was 52.3 (range 23-81) years, and the mean age at which patients switched to SDHD therapy was 48.2 (range 14-78) years. Patients used NHD or SDHD for a median of 1,102 days (approximately 3 years).
Table 1. Dialysis treatment characteristics.
| NHD (n=101) | SDHD (n=44) | CHD* | |
|---|---|---|---|
| Session time (h) | |||
| Mean (SD) | 7.2 (0.66) | 2.9 (0.58) | 3 |
| Median | 7 | 2.83 | |
| Range | (4.5, 8) | (1.67, 4) | (2.5, 4) |
| Sessions per week (d) | |||
| Mean (SD) | 5.6 (0.68) | 5.4 (0.54) | 3.5 |
| Median | 6 | 5 | |
| Range | (3, 6) | (4, 6) | (2, 4) |
| Blood flow (ml/min; range) | 200-300 | 250-500 | 250-500 |
| Dialysate flow (ml/min; range) | 200-300 | 300-800 | 500-800 |
| Average Kt/V per session | 1.10-1.80 | 0.38-1.26 | 1.12-1.75 |
Abbreviations: NHD, nocturnal hemodialysis; SDHD, short daily hemodialysis; CHD, conventional hemodialysis.
standard hemodialysis prescription
NHD patients and controls were similar on all variables used for PS matching (Table 2). SDHD patients and PS-matched controls were also similar on all variables used for PS matching (Table 2). The majority of patients in both alternative-regimen HD cohorts were male and white. Few patients who initiated alternative HD regimens were characterized by lower income, based on Medicaid coverage at treatment start as an indicator of financial status. Both cohorts had a low average number of cardiovascular conditions at start of ESRD treatment, but patients who initiated NHD were more likely than patients who initiated SDHD to have coronary artery disease at treatment start.
Table 2. Characteristics of NHD study cohort and CHD controls and characteristics of SDHD study cohort and CHD controls.
| NHD (N=101) | CHD Controls (N=1,010) | P | SDHD (N=44) | CHD Controls (N=440) | P | |
|---|---|---|---|---|---|---|
| Age (yr, mean [SD])* | 47.6 (16.3) | 48.0 (17.4) | 0.83 | 41.2 (17.2) | 42.0 (18.5) | 0.78 |
| Male (%) | 65.4 | 67.1 | 0.72 | 72.7 | 75.5 | 0.69 |
| Race (%) | 0.93 | 0.79 | ||||
| Native American | 0.0 | 0.0 | 0.0 | 2.7 | ||
| Black | 23.8 | 23.4 | 20.5 | 20.0 | ||
| White | 76.2 | 76.6 | 70.5 | 70.9 | ||
| Asian | 0.0 | 0.0 | 6.8 | 4.6 | ||
| Other | 0.0 | 0.0 | 2.3 | 1.8 | ||
| Medicaid coverage* | 6.6 | 6.1 | 0.88 | 11.1 | 11.3 | 0.97 |
| Number cardiovascular conditions (mean [SD])* | 0.6 (0.9) | 0.5 (0.9) | 0.57 | 0.4 (0.9) | 0.3 (0.6) | 0.70 |
| CHF (%) | 16.9 | 19.1 | 0.61 | 11.4 | 14.3 | 0.64 |
| CAD (%) | 22.5 | 15.7 | 0.10 | 11.4 | 6.6 | 0.28 |
| CVD (%) | 6.7 | 3.9 | 0.19 | 2.9 | 2.6 | 0.92 |
| PVD (%) | 6.7 | 7.4 | 0.82 | 5.7 | 3.7 | 0.56 |
| BMI (mean [SD])* | 29.2 (8.5) | 29.6 (8.6) | 0.69 | 26.2 (7.3) | 26.3 (6.8) | 0.97 |
| Diabetic ESRD (%) | 27.0 | 25.8 | 0.81 | 25.0 | 25.8 | 0.92 |
| Months on dialysis (mean [SD])† | 55.8 (66.4) | 56.6 (61.6) | 0.90 | 83.7 (87.1) | 88.3 (82.6) | 0.73 |
| All-cause hospital admissions/person-year‡ | 1.1 | 0.9 | 0.55 | 0.6 | 0.7 | 0.64 |
| All-cause hospital days/person-year ‡ | 5.7 | 5.1 | 0.81 | 3.1 | 3.4 | 0.67 |
| Cardiovascular admissions/person-year‡ | 0.4 | 0.3 | 0.52 | 0.1 | 0.1 | 0.87 |
| Cardiovascular hospital days/person-year‡ | 1.9 | 1.5 | 0.70 | 0.4 | 0.4 | 0.98 |
| Vascular access admissions/person-year‡ | 0.07 | 0.06 | 0.82 | 0.1 | 0.1 | 0.93 |
| Vascular access hospital days/person-year‡ | 0.3 | 0.2 | 0.71 | 0.3 | 0.3 | 0.91 |
Abbreviations: NHD, nocturnal hemodialysis; SDHD, short daily hemodialysis; ESRD, end-stage renal disease; CHF, congestive heart failure; CAD, coronary artery disease; CVD, cerebral vascular disease; PVD, peripheral vascular disease; BMI, body mass index.
As of ESRD treatment start, recorded in Medical Evidence Form.
As of conversion to NHD or SDHD; for CHD patients, as of 1/1/03.
During 365 days prior to NHD or SDHD conversion; for CHD patients, during 365 days prior to 1/1/03.
The observed mortality rates for NHD controls and SDHD controls were 147 and 138, respectively, per 1,000 patient years. For NHD patients and SDHD patients, the respective observed mortality rates were 70 and 90 per 1,000 patient years. Multivariable Cox proportional hazards regression analyses indicated that NHD compared to CHD was associated with a significantly reduced hazard for death (hazard ratio 0.34; confidence interval 0.21 to 0.58; P < 0.0001) [Table 3]. There was a reduced but non-significant risk of death for SDHD compared to CHD (hazard ratio 0.61; confidence interval 0.30 to 1.24; P = 0.17) [Table 3]. Corresponding results from Kaplan-Meier analyses of survival among NHD patients and CHD controls, and survival among SDHD patients and CHD controls, are shown in Figure 1 and Figure 2, respectively. Additional multivariable Cox proportional hazards regression analyses indicated that NHD compared to CHD was associated with a significantly reduced hazard for death or major morbid event, i.e. AMI or stroke (hazard ratio 0.49; confidence interval 0.31 to 0.78; P = 0.003). Among SDHD users the hazard ratio for death or major morbid event compared to CHD was 0.73; confidence interval 0.37 to 1.44; P = 0.36.
Table 3. Multivariable Cox proportional hazards models predicting HD patient risk for mortality and risk for composite endpoint of mortality/major morbid event (AMI, stroke).
| NHD cohort and controls | SDHD cohort and controls | |||
|---|---|---|---|---|
| All-cause mortality | Mortality/AMI, stroke | All-cause mortality | Mortality/AMI, stroke | |
| Risk Factor | Hazard Ratio (95% CI) | Hazard Ratio (95% CI) | Hazard Ratio (95% CI) | Hazard Ratio (95% CI) |
| Age (years) | 1.04 (1.03, 1.05)* | 1.04 (1.04, 1.05)* | 1.04 (1.02, 1.05)* | 1.04 (1.03, 1.05)* |
| Male | 1.05 (0.85, 1.30) | 0.98 (0.80, 1.21) | 1.15 (0.81, 1.65) | 1.21 (0.84, 1.75) |
| Black | 1.12 (0.88, 1.44) | 0.95 (0.75, 1.22) | 1.04 (0.77, 1.40) | 1.02 (0.75, 1.38) |
| Medicaid | 1.31 (1.11, 1.53) ‡ | 1.31 (1.12, 1.54) † | 1.64 (0.96, 2.79) | 1.66 (0.94, 2.93) |
| Diabetic ESRD | 1.45 (1.14, 1.85) ‡ | 1.36 (1.07, 1.72) § | 1.53 (1.07, 2.21) § | 1.41 (0.97, 2.05) |
| No. cardiovascular conditions | 1.00 (0.90, 1.12) | 1.05 (0.94, 1.17) | 1.06 (0.85, 1.32) | 1.04 (0.83, 1.31) |
| BMI (kg/m2) | 0.99 (0.98, 1.01) | 0.99 (0.98, 1.01) | 0.99 (0.95, 1.02) | 1.01 (0.97, 1.04) |
| Months on dialysis | 1.00 (0.999, 1.004) | 1.00 (1.00, 1.01) | 1.00 (1.00, 1.01) | 1.00 (0.999, 1.005) |
| Frequent HD | 0.34 (0.21, 0.58)* | 0.49 (0.31, 0.78) ‡ | 0.61 (0.30, 1.24) | 0.73 (0.37, 1.44) |
Abbreviations: CI, confidence interval; ESRD, end-stage renal disease; BMI, body mass index; HD, hemodialysis.
P < 0.0001
P < 0.001
P < 0.005
P < 0.05
Figure 1. Kaplan–Meier analysis of nocturnal hemidiolysis (NHD) therapy patient and conventional hemodialosis (CHD) control survival from baseline.

Patient survival was analyzed as time until death; observations were censored at the end of follow-up or transplantation.
Figure 2. Kaplan–Meier analysis of short-duration daily hemodialysis (SDHD) therapy patient and conventional hemodialysis (CHD) control survival from baseline.
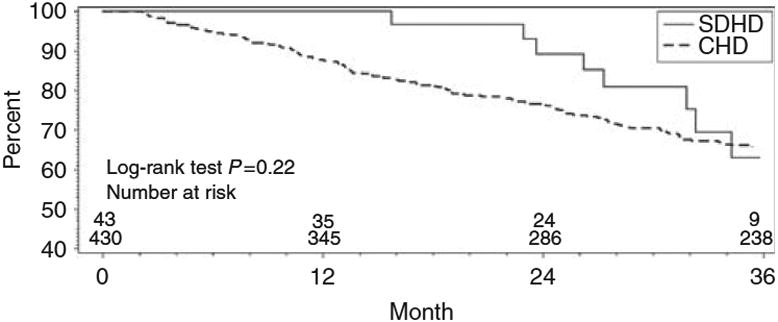
Patient survival was analyzed as time until death; observations were censored at the end of follow-up or transplantation.
Multivariable Cox regression analyses did not show that NHD or SDHD was associated with significantly reduced hazard for all-cause hospitalization, non-vascular access-related hospitalization, cardiovascular hospitalization, or infection-related hospitalization. For hospitalization associated with congestive heart failure specifically, the hazard ratio for NHD compared to CHD was 0.59; confidence interval 0.29 to 1.20; P = 0.15, and the hazard ratio for SDHD compared to CHD was 0.83; confidence interval 0.26 to 2.68; P = 0.75. For vascular access-related hospitalization, the hazard ratio for NHD compared to CHD was 1.34; confidence interval 0.91 to 1.97; P = 0.14, and the hazard ratio for SDHD compared to CHD was 0.70; confidence interval 0.31 to 1.60; P = 0.40.
Discussion
Our study demonstrated a survival benefit of NHD compared to CHD in a relatively modern cohort assembled from multiple centers in the US in which most patients received NHD 5 nights per week. Our results are in general agreement with those of Charra et al.,10 who compared the survival of patients receiving thrice weekly NHD to that of registry patients receiving CHD in Japan, Europe, and the US. The overall 87% survival of the Charra et al. cohort was superior to any of the registries, and the advantage was particularly marked for older patients. Pierratos et al.,11 in a report of 5-year outcomes among 30 patients treated with frequent NHD delivered at home, noted a mortality rate of 2.4% per year, a rate that is substantially lower than that reported by the US Renal Data System for the general HD population.12 [Check Pierratos paper to see if this was their conclusion. Can't really compare this rate directly to ours.]
The improved survival associated with NHD is biologically plausible. Both of the high intensity dialysis cohorts in our study had an apparent immediate survival benefit that could be in part related to patient selection. However, for the NHD cohort, the survival curves continue to diverge throughout the entire follow-up period (Figure 1), suggesting a possible treatment effect. Multiple clinical benefits associated with high intensity HD have been documented. Many of these benefits would be expected to enhance survival, including reduction in left ventricular hypertrophy,16-18 correction of sleep apnea,19 and improved control of hypertension, mineral metabolism, endocrine abnormalities, and anemia.20,21 Improvements in blood pressure,6,22 hemoglobin,23 homocysteine,24 calcium/phosphorus product,6 and Kt/V,22 as well as patient-assessed quality of life6,22 have been identified in patients using high intensity HD by clinicians who contributed to this study.
The current analysis shares some of the limitations of prior studies, including low power as previously mentioned as well as potential confounding resulting from patient selection. Although patient selection strategies differ across centers and over time, they are clearly evident in most reports of high intensity dialysis strategies. Patients starting NHD or SDHD who were identified for our study were younger, less likely to be lower income and less likely to have diabetic ESRD than the overall HD population in the U.S., and were characterized by fewer cardiovascular comorbidities than patients in the overall HD population (data not shown). We used PS matching to identify CHD control patients with demographic, clinical and hospitalization history characteristics similar to high NHD and SDHD users in each of these cohorts. We believe that our propensity score matching, and in particular the incorporation of matching based on prior hospitalization rates, is a significant advance over previous attempts to compare outcomes between patients receiving high intensity dialysis and those receiving CHD. Furthermore, the matching strategy was successful in that it resulted in control groups with similar vintage and prior hospitalization rates. Nevertheless, any matching strategy is only as good as the data available upon which to match, and patients who are considered appropriate for a home-based regimen such as NHD are likely to be different than in-center dialysis patients receiving CHD, raising the strong possibility of residual confounding. Specifically, we lacked data on several important potential predictors of survival and hospitalization, such as type of vascular access, recent laboratory parameters, and patient adherence to dialysis. However, it is important to consider that, although the select nature of patients appropriate for home dialysis makes them difficult to compare to CHD patients, these selection issues reflect actual practice and are not likely to disappear even if a concerted effort is made to increase the use of high intensity dialysis. Similar to peritoneal dialysis, it is likely that a majority of home-based intensive dialysis therapy users will be younger and more stable and have higher socioeconomic status than in-center dialysis patients.
We did not find significantly better survival among SDHD patients when compared to PS-matched CHD control subjects. However, the point estimate of the hazard ratio is in accord with data from Kjellstrand et al.13 and Blagg et al.,14 who reported a lower mortality on SDHD after adjustment for age, sex, race, and the presence of diabetes. Possible reasons for the lack of significantly prolonged survival in our cohort include low statistical power due to the smaller sample size of our study and the low event rate in the control group because of the additional adjustment provided by the propensity score matching that included such factors as pre-conversion hospitalization rates and days. The SDHD cohort assembled for this investigation was in fact younger and healthier than the general US dialysis population, as evidenced by their very low pre-conversion hospitalization rates. Similarly, we did not detect a reduction in hospitalization rates related to changing to either SDHD or NHD, contrary to reports by Ting et al.5 for SDHD and several groups of investigators for NHD,6,7,11,15 all of which included fewer patients than the current investigation. We believe that patient selection could be an important factor limiting our ability to detect changes in hospitalization. Our SDHD and NHD cohorts both had lower hospitalization rates in the year preceding the switch to high intensity dialysis than did previously described cohorts,5-7,11,15 making it less likely that hospitalization would be reduced by intensifying dialysis therapy and limiting power to detect such a change even if present.
The possibility that high intensity HD may be associated with an increase in vascular access complications due to more frequent cannulation has been a concern. Prior evidence about this issue has been equivocal.25 Our results, as well as results from other recent studies, do not indicate that a significant increase in vascular access complications requiring hospitalization accompanies use of high intensity HD. In an RCT recently reported by Culleton et al.,18 complications related to vascular access did not differ significantly over a six-month follow-up between patients randomized to NHD and patients randomized to CHD. In our study, risk for vascular access-related hospital admissions was not significantly associated with NHD use or SDHD use compared to matched controls. The proportion of hospitalization events that were vascular access-related remained a small proportion of the total hospitalization events observed for high intensity dialysis patients, limiting power to detect changes related to treatment type. Furthermore, many vascular access events may be managed without hospital admission and are not counted in these data, making it impossible to rule out an increase in vascular access events.
Vascular access type is an especially important potential confounder because it is clearly associated with survival – better in the case of arteriovenous fistula and worse in the case of tunneled catheter26-29 – and because access type may also be associated with selection for high intensity dialysis. Some groups initially required or preferred tunneled dialysis catheters for NHD. However, a recent report from the International Quotidian Dialysis Registry shows that all types of dialysis access are now in use for high intensity dialysis.30 In this registry, the prevalence of catheter use is 21.1% among patients from the US receiving SDHD at home and 24.6% for patients receiving NHD at home. These numbers are similar to what has been reported in the US CHD cohort from the Dialysis Outcomes and Practice Patterns Study (DOPPS), which varied from 17% during DOPPS I (1996-2000) to 27% in DOPPS II (2002-2003).31 Thus, it does not appear that there is a large difference in access type between patients receiving alternative and conventional dialysis therapies at the national level. However, it is possible that there could be differences between our alternative dialysis and CHD cohorts since this variable was not available for matching.
There is evolving variation in frequency, duration, and setting of treatments. For example, compared to the treatment patterns described in Table 1, it is likely that the median number of SDHD sessions/week is now 6 rather than 5, and NHD patients may be more likely to have 5 rather than 6 sessions/week in order to avoid phosphorus depletion. The International Quotidian Dialysis Registry, developed to track treatment and outcomes in the high intensity dialysis population, has recognized this heterogeneity and has classified patients into five different categories based on treatment length, frequency, and location.30 Thus, although heterogeneity in regimens makes it difficult to compare alternative dialysis regimens with CHD in large numbers of patients, it is reflective of current clinical practice. Furthermore, the heterogeneity in treatment regimens would be likely to obscure rather than exaggerate any differences in survival or hospitalization by increasing variability within the high intensity groups.
A RCT, such as the ongoing Frequent Hemodialysis Network randomized trials of NHD and SDHD, would provide the most definitive evidence about survival benefit associated with high intensity HD. However, these studies are not powered to examine mortality outcomes, and recruiting the number of patients needed for such a study is not considered feasible.33 Given that large-scale trials on alternative-regimen HD vs. CHD may never be conducted,34 observational studies can provide important insight. Culleton et al. recommend that data collection for hospitalizations and vascular access complications should have high priority in future observational studies.18 In addition, evolving technology for frequent HD requires study, as new machines become available and practice standards evolve.33 In the meantime, our study adds to existing evidence that NHD may improve patient survival, which remains the ongoing primary challenge in provision of dialysis therapy.1,33
Methods
Data sources and collection
Eight clinicians with substantial high intensity HD programs were asked in to participate in a special USRDS linked data study. Three of these clinicians expressed interest in the study but decided not to participate due to reported difficulties in obtaining institutional review board approval and/or lack of administrative help needed to furnish the data requested for the study. Requests were also circulated to attendees of the 2001 NIH Daily Dialysis Workshop, but these requests did not yield any additional participants. Thus, five clinicians provided data to the USRDS Coordinating Center for patients who had undergone high intensity HD in their programs over the time period of 1997 to 2005. These datasets included information about the type of frequent HD used (NHD or SDHD), number of days/week treatment, number hours/HD session, start date of frequent HD, and end date if applicable. This paper analyzes outcomes for 101 patients who used NHD and 44 patients who used SDHD for more than 60 days. Most of the SDHD patients used the PHD™ System (Aksys Ltd., Lincolnshire IL).
The USRDS Coordinating Center deidentified the data by assigning unique USRDS patient identifiers before sending the dataset to the USRDS Rehabilitation/Quality of Life Special Studies Center at Emory University. Data for individual patients were then linked with information available in the updated 2007 USRDS Standard Analysis Files (SAFs). In addition, two comparison cohorts of patients using thrice-weekly CHD were identified from the USRDS database using propensity score (PS) analysis. Each patient in both the NHD and SDHD cohorts was matched to 10 CHD patients. The study was approved by the Institutional Review Board of Emory University and by the Committee on Human Research at the University of California, San Francisco.
Patient age, gender, race, and dialysis start date were obtained from the USRDS Patient SAF, along with Medicaid coverage at treatment start (yes/no) as an indicator of lower income status. Clinical characteristics that were documented at treatment start (primary diagnosis of diabetes, number of existing cardiovascular conditions [congestive heart failure, coronary artery disease, history of AMI, cardiac arrest, dysrthythmia, cerebral vascular disease, peripheral vascular disease], and body mass index [BMI]) were also obtained from the Patient SAF. Information about patients' treatment modality prior to start of frequent HD was available in the Treatment History SAF. Patient mortality data were obtained from USRDS Patient files that contained death dates through September 30, 2006.
The Hospital file in the 2007 SAFs was used to identify hospital admissions and days. International Classification of Diseases, Ninth revision, Clinical Modification (ICD-9-CM) codes were used to identify primary diagnoses associated with hospitalization events. Cardiovascular-related hospitalization was identified by ICD-9-CM codes 390-459 (diseases of the circulatory system), 518.4 (acute pulmonary edema), 276.6 (fluid overload), 785 (symptoms involving cardiovascular system), 786.5 (chest pain), and 780.2 (syncope and collapse). Congestive heart failure-related hospitalization was identified by ICD-9-CM codes 398.01, 402.X1, 404.X1, 404.X3, 422, 425, 428. Infection-related hospitalization was identified by ICD-9-CM codes 480-487 (influenza/pneumonia); 038, 790.7, 790.8 (bacteremia/viremia/septicemia); and 472-474.0, 475-477.9, 478.22-478.24, 480-491, 494, 510-511, 513.0, and 518.6 (respiratory infection). Vascular access-related hospitalization was identified by ICD-9-CM codes 996.1, 996.62, and 996.73 (diagnosis codes) and 3895, 3927, 3942, 3943, 3993, 3994, and 8607 (procedure codes). Occurrence of the major morbid events of AMI (ICD-9-CM codes 410, 410.X0, 410.X1) and stroke (ICD-9-CM codes 430-437) was identified by first diagnosis codes attached to hospitalization events in the Hospital file.
Statistical analysis
A PS for initiating high intensity HD was estimated. The baseline was defined as NHD start date or SDHD start date for patients in the two cohorts. Baseline was defined as January 1, 2003 for patients in the comparison cohorts because 2003 was the year in which the largest number of high intensity HD patients initiated this therapy. Because we restricted the high intensity HD patients to persons who used this therapy for at least 60 days after baseline, the population considered for the comparison cohorts was restricted to patients alive 60 days after the baseline date of January 1, 2003. PS scores were based on logistic regression models that included the following covariates: age, gender, race, Medicaid status, diabetic ESRD, number of cardiovascular comorbid conditions, BMI, vintage, number of all-cause, cardiovascular-related, and vascular access-related hospitalizations and number of days of all-cause, cardiovascular-related, and vascular access-related hospitalization occurring within a maximum of 365 days before baseline (start of frequent HD or 2003). The c statistic of the PS model predicting NHD use was 0.56, and the c statistic of the PS model predicting SDHD use was 0.60. We matched the 145 frequent HD patients to CHD patients in a 1:10 ratio. Among them, 129, 11, and 5 frequent HD patients were matched to CHD patients who had a PS that was identical to 4, 3, and 2 digits, respectively.
Baseline characteristics of patients who started NHD (n=101) and matched CHD controls (n = 1010) were compared by t test (continuous variables) and χ2 test (categorical variables). Similarly, baseline characteristics of SDHD patients (n=44) and matched CHD controls (n=440) were compared by t test and χ2 test. The association of dialysis modality (NHD vs. CHD; SDHD vs. CHD) with risk for death, risk for death or major morbid event (AMI, stroke), and risk for hospitalization (all-cause, cardiovascular-related, CHF-related, infection-related, and vascular access-related) was examined in multivariable Cox proportional hazards models. Observations were censored at transplantation or end of follow-up; in the exploratory analyses of risk for hospitalization, observations were also censored at death. For death, the follow-up date was September 30, 2006, the latest date for which mortality outcomes were available. For the composite endpoint (death or major morbid event, i.e. AMI, stroke) and for hospitalization outcomes, the follow-up date was December 31, 2005, the latest date for which hospitalization outcomes were available.
Acknowledgments
The interpretation and reporting of the data presented here are the responsibility of the authors and in no way should be seen as an official policy or interpretation of the United States government. An abstract was presented in poster form at the annual meeting of the American Society of Nephrology; November 3, 2007, in San Francisco CA.
Sources of support: National Institutes of Health contracts N01-DK-1-2471 and HHSN267200715004C, ADB No. N01-DK-7-5004.
Footnotes
Disclosure: CRB is a scientific consultant to DEKA Research and Development Corporation.
References
- 1.Chertow GM. ‘Wishing don't make it so’—why we need a randomized clinical trial of high-intensity hemodialysis. J Am Soc Nephrol. 2001;12:2850–2853. doi: 10.1681/ASN.V12122850. [DOI] [PubMed] [Google Scholar]
- 2.Home Dialysis Central: Home Dialysis Central reports tremendous 4-year growth in #s of home programs. Home Dialysis Central Update. 2008;5(3) available at www.homedialysis.org. [Google Scholar]
- 3.Mohr PE, Neumann PJ, Franco SJ, et al. The case for daily dialysis: its impact on costs and quality of life. Am J Kidney Dis. 2001;37:777–789. doi: 10.1016/s0272-6386(01)80127-x. [DOI] [PubMed] [Google Scholar]
- 4.Mohr PE. The economics of daily dialysis. Adv Ren Replace Ther. 2001;8:273–279. doi: 10.1053/jarr.2001.27592. [DOI] [PubMed] [Google Scholar]
- 5.Ting GO, Kjellstrand C, Freitas T, et al. Long-term study of high-comorbidity ESRD patients converted from conventional to short daily hemodialysis. Am J Kidney Dis. 2003;42:1020–1035. doi: 10.1016/j.ajkd.2003.07.020. [DOI] [PubMed] [Google Scholar]
- 6.Lockridge RS, Jr, Spencer M, Craft V, et al. Nightly home hemodialysis: five and one-half years of experience in Lynchburg, Virginia. Hemodial Int. 2004;8:61–69. doi: 10.1111/j.1492-7535.2004.00076.x. [DOI] [PubMed] [Google Scholar]
- 7.Kumar VA, Ledezma ML, Rasgon SA. Daily home hemodialysis at a health maintenance organization: three-year experience. Hemodial Int. 2007;11:225–230. doi: 10.1111/j.1542-4758.2007.00173.x. [DOI] [PubMed] [Google Scholar]
- 8.Kroeker A, Clark WF, Heidenheim AP, et al. An operating cost comparison between conventional and home quotidian hemodialysis. Am J Kidney Dis. 2003;42(1):S49–S55. doi: 10.1016/s0272-6386(03)00538-9. [DOI] [PubMed] [Google Scholar]
- 9.Schulman G. Daily hemodialysis: the time has come: con. Am J Kidney Dis. 2005;45:798–803. doi: 10.1053/j.ajkd.2005.02.014. [DOI] [PubMed] [Google Scholar]
- 10.Charra B, Calemard E, Ruffet M, et al. Survival as an index of adequacy of dialysis. Kidney Int. 1992;41:1286–1291. doi: 10.1038/ki.1992.191. [DOI] [PubMed] [Google Scholar]
- 11.Pierratos A. Nocturnal home haemodialysis: an update on a 5-year experience. Nephrol Dial Transplant. 1999;14:2835–2840. doi: 10.1093/ndt/14.12.2835. [DOI] [PubMed] [Google Scholar]
- 12.United States Renal Data System: 2008 USRDS Annual Data Report. The National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases; Bethesda, MD: 2008. [Google Scholar]
- 13.Kjellstrand CM, Buoncristiani U, Ting G, et al. Short daily haemodialysis: survival in 415 patients treated for 1006 patient-years. Nephrol Dial Transplant. 2008;23:3283–3289. doi: 10.1093/ndt/gfn210. [DOI] [PubMed] [Google Scholar]
- 14.Blagg CR, Kjellstrand CM, Ting GO, et al. Comparison of survival between short-daily hemodialysis and conventional hemodialysis using the standardized mortality ratio. Hemodial Int. 2006;10:371–374. doi: 10.1111/j.1542-4758.2006.00132.x. [DOI] [PubMed] [Google Scholar]
- 15.Bergman A, Fenton SSA, Richardson RMA, Chan CT. Reduction in cardiovascular related hospitalization with nocturnal home hemodialysis. Clin Nephrol. 2008;69(1):33–39. doi: 10.5414/cnp69033. [DOI] [PubMed] [Google Scholar]
- 16.Chan CT, Floras JS, Miller JA, et al. Regression of left ventricular hypertrophy after conversion to nocturnal hemodialysis. Kidney Int. 2002;61:2235–2239. doi: 10.1046/j.1523-1755.2002.00362.x. [DOI] [PubMed] [Google Scholar]
- 17.Ayus JC, Mizani MR, Achinger SG, et al. Effects of short daily versus conventional hemodialysis on left ventricular hypertrophy and inflammatory markers: a prospective, controlled study. J Am Soc Nephrol. 2005;16:2778–2788. doi: 10.1681/ASN.2005040392. [DOI] [PubMed] [Google Scholar]
- 18.Culleton BF, Walsh M, Klarenbach SW, et al. Effect of frequent nocturnal hemodialysis vs conventional hemodialysis on left ventricular mass and quality of life. JAMA. 2007;298:1291–1299. doi: 10.1001/jama.298.11.1291. [DOI] [PubMed] [Google Scholar]
- 19.Hanly PJ, Pierratos A. Improvement of sleep apnea in patients with chronic renal failure who undergo nocturnal hemodialysis. N Engl J Med. 2001;344:102–107. doi: 10.1056/NEJM200101113440204. [DOI] [PubMed] [Google Scholar]
- 20.Walsh M, Culleton B, Tonelli M, Manns B. A systematic review of the effect of nocturnal hemodialysis on blood pressure, left ventricular hypertrophy, anemia, mineral metabolism, and health-related quality of life. Kidney Int. 2005;67:1500–1508. doi: 10.1111/j.1523-1755.2005.00228.x. [DOI] [PubMed] [Google Scholar]
- 21.Suri RS, Nesrallah GR, Mainra G, et al. Daily hemodialysis: a systematic review. Clin J Am Soc Nephrol. 2006;1:33–42. doi: 10.2215/CJN.00340705. [DOI] [PubMed] [Google Scholar]
- 22.Goldfarb-Rumyantzev AS, Leypoldt JK, Nelson J, et al. A crossover study of short daily haemodialysis. Nephrol Dial Transplant. 2006;21:166–175. doi: 10.1093/ndt/gfi116. [DOI] [PubMed] [Google Scholar]
- 23.Elwell RJ, Hoy CD, Daoui R, et al. Achieving K/DOQI anemia targets in daily nocturnal hemodialysis patients. J Am Soc Nephrol. 2005;16:722A. [Google Scholar]
- 24.Hoy CD. Daily nocturnal hemodialysis—patient experience and clinical update. ESRD Network of New York Annual Meeting; East Elmhurst, NY. October 8, 2004. [Google Scholar]
- 25.Suri RS, Garg AX, Chertow GM, et al. Frequent hemodialysis network (FHN) randomized trials: study design. Kidney Int. 2007;72:349–359. doi: 10.1038/sj.ki.5002032. [DOI] [PubMed] [Google Scholar]
- 26.Dhingra RK, Young EW, Hulbert-Shearon TE, et al. Type of vascular access and mortality in U.S. hemodialysis patients. Kidney Int. 2001;60:1443–1451. doi: 10.1046/j.1523-1755.2001.00947.x. [DOI] [PubMed] [Google Scholar]
- 27.Pastan S, Soucie JM, McClellan WM. Vascular access and increased risk of death among hemodialysis patients. Kidney Int. 2002;62:620–626. doi: 10.1046/j.1523-1755.2002.00460.x. [DOI] [PubMed] [Google Scholar]
- 28.Polkinghorne KR, McDonald SP, Atkins RC, Kerr PG. Vascular access and all-cause mortality: a propensity score analysis. J Am Soc Nephrol. 2004;15:477–486. doi: 10.1097/01.asn.0000109668.05157.05. [DOI] [PubMed] [Google Scholar]
- 29.Astor BC, Eustace JA, Powe NR, et al. Type of vascular access and survival among incident hemodialysis patients: the Choices for Healthy Outcomes in Caring for ESRD (CHOICE) Study. J Am Soc Nephrol. 2005;16:1441–1455. doi: 10.1681/ASN.2004090748. [DOI] [PubMed] [Google Scholar]
- 30.Nesrallah GE, Suri RS, Moist LM, et al. The International Quotidian Dialysis Registry: annual report 2008. Hemodial Int. 2008;12:281–289. doi: 10.1111/j.1542-4758.2008.00268.x. [DOI] [PubMed] [Google Scholar]
- 31.Ethier J, Mendelssohn DC, Elder SJ, et al. Vascular access use and outcomes: an international perspective from the Dialysis Outcomes and Practice Patterns Study. Nephrol Dial Transplant. 2008;23(10):3219–3226. doi: 10.1093/ndt/gfn261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.National Kidney Foundation (NKF) Clinical Practice Guidelines and Clinical Practice Recommendations 2006 Updates Hemodialysis Adequacy. [Accessed 10/2/08];NKF K/DOQI Guidelines. at http://www.kidney.org/Professionals/kdoqi/guideline_upHD_PD_VA/index.htm.
- 33.Kliger AS. Frequent nocturnal hemodialysis—a step forward? JAMA. 2007;298:1331–1333. doi: 10.1001/jama.298.11.1331. [DOI] [PubMed] [Google Scholar]
- 34.Lee CP, Zenios SA, Chertow GM. Cost-effectiveness of frequent in-center hemodialysis. J Am Soc Nephrol. 2008;19:1792–1797. doi: 10.1681/ASN.2008010001. [DOI] [PMC free article] [PubMed] [Google Scholar]


