Abstract
Textile electromyography (EMG) electrodes embedded in clothing allow muscle excitation to be recorded in previously inaccessible settings; however, their ability to accurately and reliably measure EMG during dynamic tasks remains largely unexplored. To quantify the validity and reliability of textile electrodes, 16 recreationally active males completed two identical testing sessions, within which three functional movements (run, cycle and squat) were performed twice: once wearing EMG shorts (measuring quadriceps, hamstrings and gluteals myoelectric activity) and once with surface EMG electrodes attached to the vastus lateralis, biceps femoris and gluteus maximus. EMG signals were identically processed to provide average rectified EMG (normalized to walking) and excitation length. Results were compared across measurement systems and demonstrated good agreement between the magnitude of muscle excitation when EMG activity was lower, but agreement was poorer when excitation was higher. The length of excitation bursts was consistently longer when measured using textile vs. surface EMG electrodes. Comparable between-session (day-to-day) repeatability was found for average rectified EMG (mean coefficient of variation, CV: 42.6 and 41.2%) and excitation length (CV: 12.9 and 9.8%) when using textile and surface EMG, respectively. Additionally, similar within-session repeatability (CV) was recorded for average rectified EMG (13.8 and 14.1%) and excitation length (13.0 and 12.7%) for textile and surface electrodes, respectively. Generally, textile EMG electrodes appear to be capable of providing comparable muscle excitation information and reproducibility to surface EMG during dynamic tasks. Textile EMG shorts could therefore be a practical alternative to traditional laboratory-based methods allowing muscle excitation information to be collected in more externally-valid training environments.
Key points.
Muscle excitation (normalized average rectified EMG) during functional tasks was generally comparable across the textile EMG and traditional surface EMG systems
Excitation lengths tend to be longer when collected using textile electrodes compared with traditional surface electrodes
Reproducibility is similar across the two systems
Textile EMG electrodes can provide a practical alternative to traditional surface EMG, which may allow greater opportunity to collect muscle excitation information in externally-valid, field-based environments, such as normal training situations
Key words: EMG, lower-limb, myoelectric activity, reliability, validity
Introduction
Electrodes attached to the skin surface are widely used to measure the electrical excitation of superficial muscles (electromyography, EMG) during dynamic activities and can provide insight into the control and execution of movement across a wide variety of settings (Häkkinen et al., 2001; Morin et al., 2015; Rose and McGill, 2005). This technique has a potentially important role in neuromuscular rehabilitation; for example, by providing information (biofeedback) to patients in real-time (Giggins et al., 2013). Moreover, surface EMG is a key tool in musculoskeletal modelling either as input for EMG-driven models (Lloyd and Besier, 2003) or as validation criteria for the outputs (e.g. Cazzola et al., 2017).
Myoelectric signals can, however, be more difficult to record in settings outside of controlled laboratory trials and the practical application of this measurement technique may therefore be restricted. For instance, EMG electrodes can impede movement in some cases or detach from the skin altogether, and wires may need to be carefully secured in order to minimize movement artefact (Clancy et al., 2002). Additionally, it is very difficult to record from exactly the same motor units on two different occasions (Burden, 2008), introducing inevitable inconsistencies in longitudinal EMG data. Although normalization can account for these discrepancies to an extent (Burden, 2010), there remains fundamental variability in the signals recorded from one day to the next (Albertus-Kajee et al., 2010; Albertus-Kajee et al., 2011). It is important that such variation is quantified and taken into account at the inception of study design, with particular care taken when attempting to monitor changes over time to avoid the misinterpretation of any observed effect (Hopkins, 2000).
The development of textile electrodes embedded in clothing, which do not require the careful placement of sensors or wires and seem to allow unrestricted movement (Scilingo et al., 2005), could provide a solution to the aforementioned limitations of traditional surface electrodes. Textile electrodes cover a larger area of the muscle to surface EMG and thus, muscle excitation of whole muscle groups rather than individual muscles are obtained. Nonetheless, such technology undoubtedly improves the ease of EMG measurement (Lintu et al., 2005) and could potentially allow myoelectric activity patterns to be collected in previously inaccessible but more externally-valid settings, such as elite training environments, rehabilitative settings or normal daily living conditions. In fact, textile EMG electrodes embedded in shorts have previously been used to characterize muscle excitation when performing normal daily activities (Pesola et al., 2016; Tikkanen et al., 2013; Tikkanen et al., 2016), to assess the efficacy of an intervention targeted at reducing sedentary time in office workers (Pesola et al., 2014) and to quantify the effect of different exercise interventions on daily (in)activity (Finni et al., 2016).
Previous studies using textile electrodes embedded in shorts have demonstrated close agreement with traditional surface EMG signals, and similar (or even superior) day-to-day reproducibility during relatively static tasks such as bilateral isometric knee extensions (Finni et al., 2007) and lying down, quiet standing and a half-squat hold (Tikkanen et al., 2013). However, the ability of textile electrodes to accurately and reliably measure muscle excitation during more dynamic, and potentially more functionally relevant, movements is yet to be determined. Thus, the aims of this study were to investigate the agreement between EMG signals measured during cyclical, dynamic activities using traditional surface EMG and textile electrodes, and to compare both the within- and between-session reliability of the myoelectric activity patterns acquired using these two measurements systems.
Methods
Participants
Sixteen healthy, recreationally active males (25 ± 5 yrs, 1.84 ± 0.06 m, 82 ± 12 kg; mean ± SD) participated in this study. Participants were injury-free and were asked to perform their normal training and/or activity routines during the study period, but to avoid abnormally vigorous lower-limb exercise in the 24-hour period before each trial. Prior to testing, a local research ethics committee provided ethical approval for the study and participants provided written informed consent.
Testing procedures
Participants visited the laboratory on two occasions within one week (typically 24 hours apart). On each occasion, participants performed exactly the same two sets of four cyclical movements: walking on a treadmill, running on a treadmill, cycling on a cycle ergometer and a repeated squatting exercise. These exercises were chosen to encompass a broad range of movement conditions (e.g. speed and impacts) and patterns of lower-limb muscle excitation. One set of these exercises was performed with traditional surface electrodes attached to the gluteal, vasti and hamstring muscles and the other was performed whilst wearing EMG shorts. The order of measurement techniques was randomized between participants, but was consistent within participants across the two days.
Participants firstly walked on a treadmill (Powerjog JX200, Sport Engineering, UK) at a self-selected pace for two minutes (4 ± 1 km/hr; mean ± SD) to warm-up, with EMG signals collected for the entire second minute and used to normalize the data from subsequent exercises. Participants then increased the pace of the treadmill to a self-selected jogging speed (9 ± 2 km/hr; mean ± SD) which was maintained for five minutes. EMG data were collected for the entire final minute. For both the walking and running trials, step frequency was noted using a metronome and inter-trial consistency was ensured during subsequent trials. Five minutes of cycling exercise was then conducted on a friction-loaded cycle ergometer (Monark 824E, Monark Exercise AB, Sweden) against a resistance which allowed the participants to maintain a cadence of 60 rpm (2.0 ± 0.5 kg; mean ± SD). Myoelectric activity was again collected across the final minute. Finally, a repeated squatting exercise was performed for one minute with EMG data collected throughout. A lightweight plastic dowel was held across the shoulders and participants lowered to a predetermined depth (approximately 90° knee angle) before returning to standing. This was repeated for the entire minute at frequency of 0.3 Hz, indicated by a metronome.
EMG data collection
Traditional surface electrodes: In the traditional surface electromyography trials, three wireless electrodes (Delsys Trigno, Delsys Inc., USA) sampling at 2000 Hz were attached (Delsys adhesive interface) to the gluteus maximus, vastus lateralis and biceps femoris of the right leg only. Sensors were placed directly superficial to the muscle belly, according to the locations and orientations recommended by SENIAM (Hermens et al., 1999). Where necessary, the skin was shaved, before being lightly abraded and cleansed using alcohol wipes. Raw data were collected and stored using Delsys EMGworks software (version 4.1.05; Delsys Inc., USA).
Textile electrodes: In the crossover trial, muscle excitation was assessed using EMG shorts (Myonear Pro, Myontec, Finland), the conductive electrodes and the associated wires of which are integrated into the fabric. These electrodes cover three main muscle groups: the gluteals, vasti and hamstrings. Two sizes of shorts were available (XL and XXL) and the best fit was chosen for each participant. Appropriate sizing of the shorts is crucial as necessary contact between the electrodes and skin is required to avoid the introduction of movement artefact during dynamic activities. Additionally, a small amount of water was applied to the electrodes before the participant put on the shorts to ensure adequate signal conduction, as previously recommended (Finni et al., 2007). EMG signals were transmitted wirelessly to a laptop and collected at 1000 Hz within MegaWin software (Mega Electronics Ltd., Finland). The data relating to the right leg only were used in subsequent analyses.
EMG data processing
All exported electromyograms were analyzed within MATLAB R2013b (MathWorks Inc., USA). Typical raw EMG traces of the vasti obtained using the traditional and textile electrode systems are provided in Figure 1.
Figure 1.
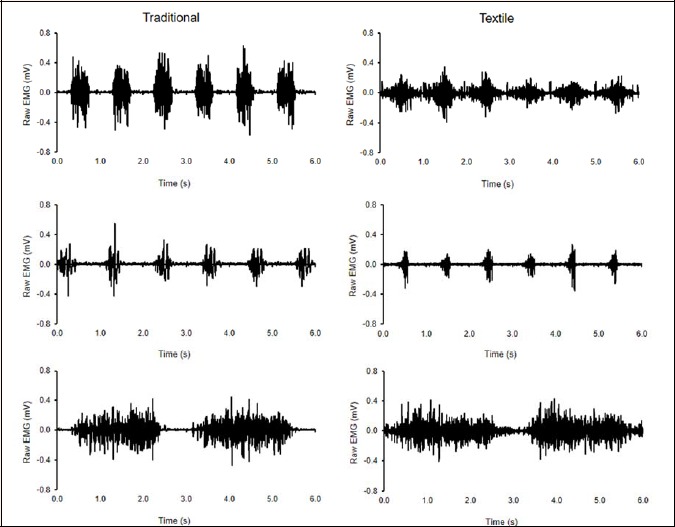
Example of raw EMG recordings for the vasti from one participant using the traditional (left) and textile (right) electrodes during the cycling (top row), running (middle row) and squatting (bottom row) exercises.
Each trial was firstly filtered with a second-order Butterworth band pass (20-300 Hz, derived through exploration of the frequency domain) filter, before being rectified and averaged over 50-ms windows. For each muscle in each trial the onset and end of 10 myoelectric activity bursts were visually identified by the same researcher as a clear departure from and return to the background signal, respectively (Hodges and Bui, 1996). These 10 bursts approximately occurred at the mid-point of the 1-minute collection period. The signals were then integrated across each excitation burst, and divided by the excitation duration to yield average rectified EMG. For each trial, a mean average rectified EMG was calculated across the 10 excitation bursts for each muscle. Due to the instability of EMG signals at maximum or near maximum levels (Bamman et al., 1997; Heinonen et al., 1994; Yang and Winter, 1983), it has been recommended that EMG amplitudes are normalized to activities with submaximal force levels (De Luca, 1997). Thus, for the running, cycling and squatting trials, the average rectified EMG values were then normalized with respect to the walking average rectified EMG trials. Previously, good reliability has been shown for EMG data during walking, which has been attributed to the well-learned, habitual nature of walking gait (Chung and Giuliani, 1997).
Statistical analysis
Group means and standard deviations (SD) were computed for the length of excitation bursts and normalized average rectified EMG recorded by each measurement technique during each movement. Effect sizes (Cohen’s D, mean difference divided by pooled standard deviation) were used in conjunction with 90% confidence intervals (CI) to assess for differences across the two measurement techniques. A threshold for a practically important effect size was set at 0.2 (Hopkins et al., 2009) with values between -0.2 and +0.2 signifying a trivial effect, and clear and unclear effects defined using 90% CI, as previously suggested (Batterham and Hopkins, 2006). Confidence intervals were computed using a published spreadsheet (Hopkins, 2007). Bland-Altman plots were used to assess the agreement between the normalized average rectified EMG measured by the two methods, with the limits of agreement set at 2SD. Heteroscedascity tests were used to check for a relationship between the difference in the two methods and the size of the measured excitation. Within-session variability of the normalized average rectified EMG was assessed across the 10 excitation bursts using coefficients of variation (CV%). Day-to-day reliability was assessed using log-transformed typical error of the measurements (TEMs, %), as previously advocated (Hopkins, 2000), and intraclass correlation coefficients (ICC), using a published spreadsheet (Hopkins, 2015). Uncertainty in TEM estimates was expressed as 90% CI.
Results
The average length of the excitation bursts recorded by the textile electrodes was generally higher than that of the traditional surface EMG across each muscle and movement (Figure 2), except for the hamstrings during the running trial (effect size = -0.08). For all three movements, the normalized average rectified EMG were similar across the two measurement techniques, except for the gluteals during cycling where the textile electrodes provided a lower magnitude of excitation than that with traditional surface electrodes (effect size = -0.58, ; 90% CI = -1.1 to -0.09; Figure 3).
Figure 2.
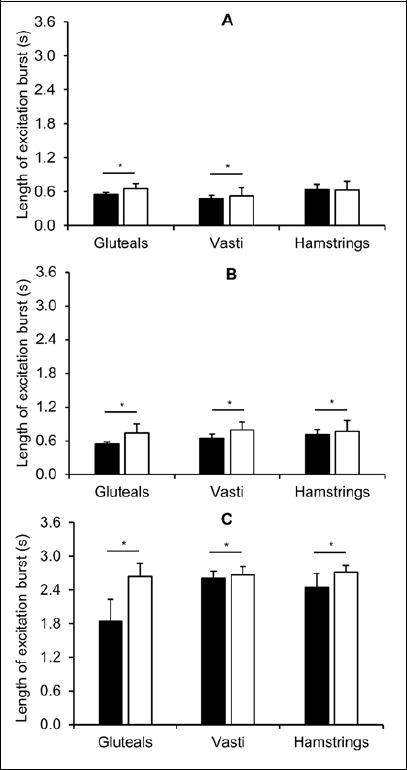
Length of excitation bursts (mean ± SD) during the run (A), cycle (B) and squat (C) exercise measured using traditional surface electromyography (black bars) and textile electrodes (white bars). *denotes longer excitation burst in the textile electrode trial compared with traditional surface electrode trial.
Figure 3.
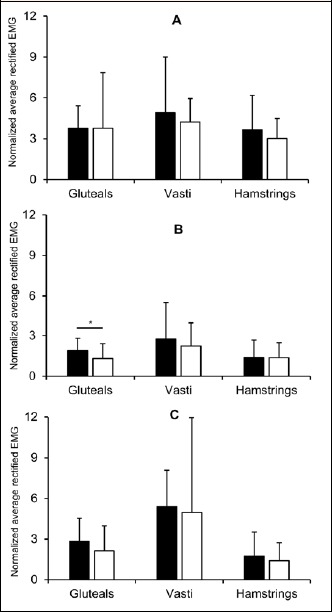
Average rectified EMG across one excitation burst (mean ± SD) during the run (A), cycle (B) and squat (C) exercise measured using traditional surface electromyography (black bars) and textile electrodes (white bars). *denotes higher magnitude of excitation in the traditional surface electrode trial compared with textile electrode trial.
Bland-Altman plots suggested good agreement between the excitation lengths values observed in the traditional surface and textile electrode trials (Figure 4). Normalized average rectified EMG values were generally in good agreement when the excitation levels were lower (Figure 5). There was some evidence of heteroscedasticity within a number of these Bland-Altman plots. Specifically, the three plots for the running trials and the plot for the vasti average rectified EMG during cycling appeared to be heteroscedastic (r ranged from -0.64 to -0.44). Additionally, all excitation length plots for the hamstrings exhibited some heteroscedascity (r ranged from -0.64 to 0.67), as well as the plots for gluteal excitation during squatting and cycling exercise (r = 0.49 and -0.91 respectively). All other plots did not exhibit heteroscedasticity.
Figure 4.
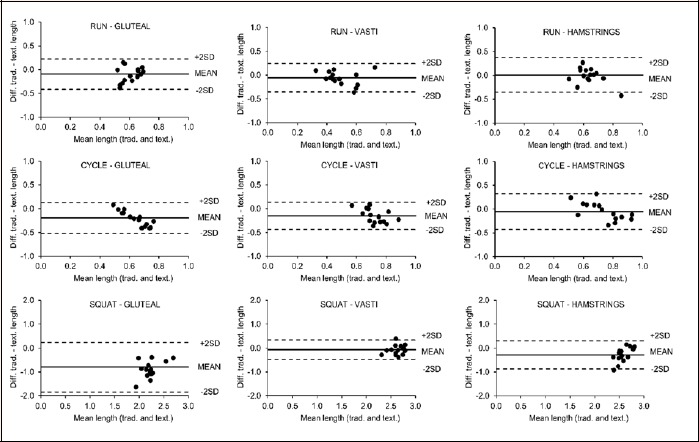
Bland-Altman plots for the lengths of excitation bursts during running, cycling and squatting measured using textile (text.) and traditional surface (trad.) EMG electrodes. Each point represents one participant, solid lines denote mean differences and dotted lines denote ±2SD.
Figure 5.
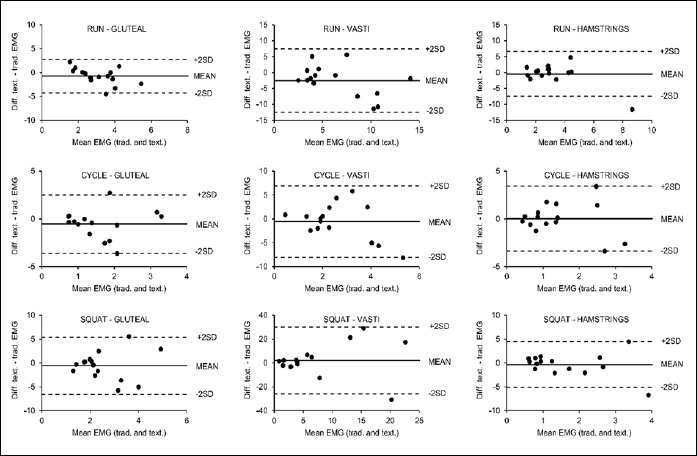
Bland-Altman plots for the normalized average rectified EMG during running, cycling and squatting measured using textile (text.) and traditional surface (trad.) EMG electrodes. Each point represents one participant, solid lines denote mean differences and dotted lines denote ±2SD.
The within-session CV% in the length of excitation bursts and the normalized average rectified EMG revealed similar variability in the traditional surface EMG trials compared with the textile electrodes (Tables 1 and 2). The only differences were observed in the within-session reliability of the length of excitation and normalized average rectified EMG for the hamstring muscles during running and cycling, respectively, with greater variability observed in the textile EMG conditions compared with the surface EMG.
Table 1.
Within-session variability (CV%; mean ± SD) of the length of excitation across 10 excitation bursts during running, cycling and squatting exercise for traditional surface and textile EMG electrodes.
| Traditional surface EMG electrodes | Textile EMG electrodes | ||
|---|---|---|---|
| Run | Gluteals | 14.2 (4.2) | 11.6 (4.9) |
| Vasti | 17.3 (6.0) | 18.9 (6.1) | |
| Hamstrings | 12.3 (4.5) | 17.4 (6.6) * | |
| Cycle | Gluteals | 17.2 (5.9) | 15.6 (5.0) |
| Vasti | 11.8 (3.8) | 14.2 (7.9) | |
| Hamstrings | 14.5 (4.3) | 16.2 (6.2) | |
| Squat | Gluteals | 10.8 (5.3) | 8.7 (3.8) |
| Vasti | 7.0 (2.4) | 6.4 (1.6) | |
| Hamstrings | 9.2 (2.5) | 7.7 (2.9) | |
* denotes higher variability in the textile vs. traditional surface EMG electrodes
Table 2.
Within-session variability (CV%; mean ± SD) of the normalized average rectified EMG signal across 10 excitation bursts during running, cycling and squatting exercise for traditional surface and textile EMG electrodes.
| Traditional surface EMG electrodes | Textile EMG electrodes | ||
|---|---|---|---|
| Run | Gluteals | 16.5 (6.5) | 14.4 (4.6) |
| Vasti | 20.3 (7.7) | 16.5 (5.8) | |
| Hamstrings | 13.4 (4.2) | 12.6 (5.5) | |
| Cycle | Gluteals | 17.4 (6.8) | 16.7 (5.7) |
| Vasti | 13.7 (5.4) | 15.1 (5.9) | |
| Hamstrings | 13.2 (4.1) | 17.4 (6.8) * | |
| Squat | Gluteals | 13.7 (5.2) | 12.5 (5.5) |
| Vasti | 9.8 (4.4) | 8.3 (3.3) | |
| Hamstrings | 8.8 (4.7) | 10.4 (4.6) | |
* denotes higher variability in the textile vs. traditional surface EMG electrodes
The between-session reliability statistics (TEMs in % and 90% CI) of the excitation bursts and normalized average rectified EMG are presented in Figures 6 and 7, respectively. The only differences in reliability observed between the two measurement techniques was for the gluteal electrodes during the running trials, in which the TEMs were higher in the textile electrode trials compared with the traditional surface EMG trials. Additionally, ICCs are provided in Table 3 and appeared to be higher for the textile electrodes embedded in shorts compared with the traditional electrodes.
Figure 6.
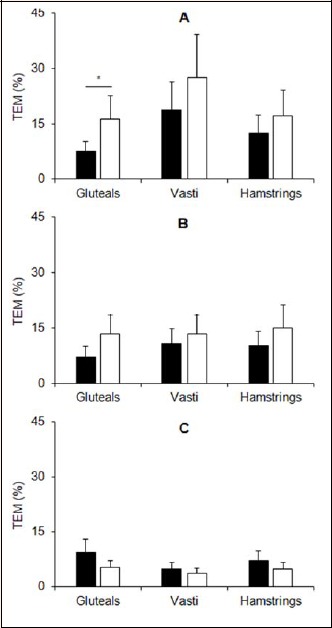
Day-to-day reproducibility (typical error of measurement, TEM) of the length of excitation bursts for running (A), cycling (B) and squatting (C) exercise measured by traditional surface (black bars) and textile (white bars) EMG electrodes. Error bars indicate 90% confidence intervals. *denotes higher TEM in the textile vs. traditional EMG electrodes.
Figure 7.
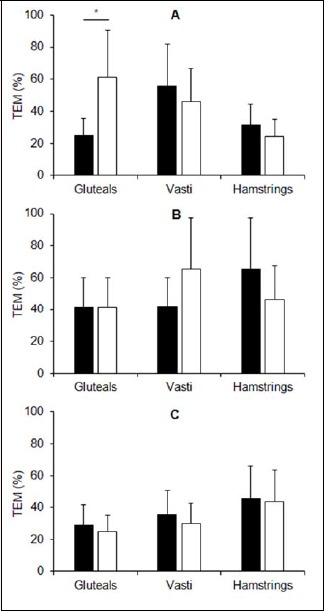
Day-to-day reproducibility (typical error of measurement, TEM) of the normalized average rectified EMG for running (A), cycling (B) and squatting (C) exercise measured by traditional surface (black bars) and textile (white bars) EMG electrodes. Error bars indicate 90% confidence intervals. *denotes higher TEM in the textile vs. traditional EMG electrodes.
Table 3.
Intraclass correlation coefficients of the normalized average rectified EMG signal across 10 excitation bursts during running, cycling and squatting exercise for traditional surface and textile EMG electrodes.
| Traditional surface EMG electrodes | Textile EMG electrodes | ||
|---|---|---|---|
| Run | Gluteals | .46 | .81 |
| Vasti | .44 | .64 | |
| Hamstrings | .58 | .96 | |
| Cycle | Gluteals | .83 | .89 |
| Vasti | .51 | .87 | |
| Hamstrings | .73 | .92 | |
| Squat | Gluteals | .77 | .87 |
| Vasti | .46 | .94 | |
| Hamstrings | .49 | .83 | |
Discussion
Textile electrodes embedded in clothing have specifically been developed to allow muscle excitation to be recorded with ease in more ecologically valid settings, for example collecting real-time data on-field during sports training. The validity and reliability of textile EMG has previously been reported for isometric or low-velocity movements (Finni et al., 2007; Tikkanen et al., 2013). However, whether accurate and reproducible myoelectric activity can be measured with textile EMG during dynamic tasks (which more closely reflect those performed in applied on-field environments) had previously not been investigated. The current study showed that the magnitude of muscle excitation (normalized average rectified EMG) across all three cyclical, dynamic tasks were similar when measured using the textile electrodes and the traditionally-used surface EMG, except for the gluteal muscles during cycling (Figure 3). Thus, overall it appears that similar information regarding the magnitude of the muscle excitation bursts can be obtained from both of the measurement techniques. However, the Bland-Altman plots (Figure 5) suggested that measurement systems were not in close agreement when the measured magnitude of excitation was high (above 5 times the average rectified EMG during walking). As greater differences between systems were found at mean values markedly greater than the norm, it is plausible that they could be attributed to measurement artefact (for example the shorts moving relative to the skin during dynamic activities), which could produce spurious values.
There were some differences in the length of excitation bursts measured by the different systems with generally longer periods of excitation measured by the textile electrodes compared with the traditional surface EMG, except for the hamstrings during running, which were not different across methods (Figure 2). This could be due to the greater area of contact between the textile electrodes and the skin, compared with the small measurement area of the traditional surface electrodes. Consequently, textile electrodes are likely to measure a greater number of motor units from a greater number of muscles which may be recruited for a longer period of time collectively, compared with traditional surface EMG. This highlights an important consideration and limitation of the current textile-embedded electrodes, which relates to the inability to measure precise muscles (e.g. vastus lateralis) within the overall muscle group (e.g. the vasti). Additionally, the differences in temporal resolution between the systems or the signal processing that is internal to the textile electrode system could also contribute to these differences.
In line with previous findings during isometric exercise (Finni et al., 2007), there appeared to be similar within-session (Table 2) and day-to-day reliability for the normalized average rectified EMG (Figure 7) and excitation burst length (Figure 6) measured via traditional surface and textile electrodes. However, during the more dynamic activities (i.e. running) the hamstrings and gluteal muscle excitations appeared more variable in the textile electrode trials compared with the traditional electrode trials. This perhaps indicates a loss of contact between the shorts and the participant’s skin, which is likely to introduce variability through the detection of non-physiological signals. This problem has been raised previously for isometric tasks (Finni et al., 2007) and for treadmill walking and running (Tikkanen et al., 2014), and the need for the shorts to tightly fit the participant remains a potential drawback to this method as garments are limited to a small range of sizes.
The day-to-day variability of the textile electrodes measured during dynamic activities in the current study was greater than that previously measured during isometric knee extensions (Finni et al., 2007). Additionally, the current study revealed muscle excitation during the slowest velocity movement (squatting) to be the most reproducible out of all three functional exercises (Figures 6 and 7). Previous findings suggest the reliability of EMG measurements is higher for squat jumps compared to countermovement jumps (Ball and Scurr, 2010). Moreover, muscle excitation during isometric or low velocity contractions have consistently been found to be more repeatable than those during dynamic, higher velocity tasks. For example, using traditional surface EMG electrodes, within-session coefficients of variation of 18 to 27% have been demonstrated for the vasti excitation during isometric contraction (Finni et al., 2007) and 11 to 22% for hip abductor muscles during low velocity rehabilitation exercises (Bolgla and Uhl, 2007). Conversely during running, Guidetti et al. (1996) found mean intra-individual variability of the EMG profiles to range from 42 to 66%. It is likely that these higher velocity movements involving impacts (such as the running in the current study) are less repeatable than lower velocity movements (for example the squatting exercise) due to the introduction of movement artefact, which is considered to be the most troublesome source of noise in EMG signals (De Luca et al., 2010). Alternatively, as the squatting exercise was conducted last and the running was first in the sequence of exercises in the current study, it may be that there was improved conductivity between the skin and electrodes (the importance of which has previously been highlighted, Finni et al. (2007)) during the latter exercises due to the participant sweating. It is of course also possible that some of the between-day variation observed could be attributed to genuine differences in muscle excitation due to day-to-day variation in the movement coordination. However, as these movements are reasonably closed motor skills which the participants were well-accustomed to, and the data were averaged across 10 excitation bursts, it is perceived that the variation identified in the current study is predominantly measurement error.
The observed high ICCs for certain measures in the current study may be perceived to indicate good agreement between repeated tests for both the textile (ICCs ranged from 0.64 to 0.96) and the surface EMG electrode (ICCs ranged from 0.46 to 0.83), as shown in Table 3. However, the typical error of the measurements are considerable in some cases. This demonstrates the importance of quantifying the likely error magnitude (typical errors) rather than solely using correlational analyses, which can appear artificially high if the spread of data is wide, as suggested by Weir (2005). In fact, the high reliability observed in previous studies (ICCs of 0.73 to 0.97, for example) could be explained in part by the wide variation in the observed normalized EMG patterns during daily tasks (Tikkanen et al., 2013).
The length of excitation bursts certainly appears to be more reproducible than the magnitude of the excitation bursts themselves (Figures 6 and 7, respectively). However, it is important to note that no output variable for either EMG collection method displayed high reliability, particularly in comparison to other methods routinely used to analyze the execution of movement. For instance, Ball and Scurr (2010) previously showed physical performance test outputs (sprint times, squat jump height and one-repetition maximum strength) to be considerably more reliable to that of the EMG signals collected during those tests. In fact, it has long been acknowledged that EMG data are inherently variable and substantial measurement errors are present (Yang and Winter, 1983), particularly when recorded during dynamic conditions (Guidetti et al., 1996; Karamanidis et al., 2004; Smoliga et al., 2010). These previous findings, alongside those of the current study, highlight the importance to carefully consider the design of studies which look to assess for differences in EMG signals between groups or changes in muscle excitation across time. Specifically, researchers should take into consideration the fact that large sample sizes, multiple trials and/or very large effects or changes are required in order to confidently detect true, clear differences in normalized average rectified EMG during dynamic tasks.
Conclusions
Textile electrodes embedded in shorts appeared to provide comparable lower limb muscle excitation data to traditional surface EMG, especially when the recorded excitation levels were lower. Additionally, the textile electrodes seem to provide similar reproducibility to the traditional surface EMG in most cases. As surface EMG is a widely accepted method to assess muscle excitation in both clinical and sporting settings, the textile electrodes appear to provide a practical alternative to acquiring muscle excitation data. Thus, textile-embedded electrodes can potentially provide opportunities to study movement control outside of controlled laboratory conditions. However, particularly when collected in dynamic situations, experimental artefacts are likely to influence the data acquired which appears to be inherently variable, regardless of the measurement technique adopted. This warrants consideration when interpreting differences between groups or changes in muscle excitation across time to ensure that signal is not confused with noise and that the correct conclusions are drawn.
Acknowledgements
All authors contributed to data collection and manuscript preparation. No funding to declare. The authors report no conflicts of interest. The reported experiments comply with the current laws of the country in which they were performed.
Biographies

Steffi COLYER
Employment
Research Associate, Department for Health, University of Bath, UK.
Degree
PhD
Research interests
Improving performance with motion analysis, understanding responses to training and technique intervention.
E-mail: S.Colyer@bath.ac.uk

Polly MCGUIGAN
Employment
Lecturer, Department for Health, University of Bath, UK.
Degree
PhD
Research interests
Understanding musculoskeletal function to prevent injury.
E-mail: M.P.McGuigan@bath.ac.uk
References
- Albertus-Kajee Y., Tucker R., Derman W., Lambert M.I. (2010) Alternative methods of normalising EMG during cycling. Journal of Electromyography and Kinesiology 20, 1036-1043. [DOI] [PubMed] [Google Scholar]
- Albertus-Kajee Y., Tucker R., Derman W., Lamberts R.P., Lambert M.I. (2011) Alternative methods of normalising EMG during running. Journal of Electromyography and Kinesiology 21, 579-586. [DOI] [PubMed] [Google Scholar]
- Ball N., Scurr J. (2010) An assessment of the reliability and standardisation of tests used to elicit reference muscular actions for electromyographical normalisation. Journal of Electromyography and Kinesiology 20, 81-88. [DOI] [PubMed] [Google Scholar]
- Bamman M.M., Ingram S.G., Caruso J.F., Greenisen S.C. (1997) Evaluation of surface electromyography during maximal voluntary contraction. Journal of Strength and Conditioning Research 11, 68-72. [Google Scholar]
- Batterham A.M., Hopkins W.G. (2006) Making meaningful inferences about magnitudes. International Journal of Sports Physiology and Performance 1, 50-57. [PubMed] [Google Scholar]
- Bolgla L.A., Uhl T.L. (2007) Reliability of electromyographic normalization methods for evaluating the hip musculature. Journal of Electromyography and Kinesiology 17, 102-111. [DOI] [PubMed] [Google Scholar]
- Burden A. (2008) Surface Electromyography. In: Biomechanical Evaluation of Movement in Sport and Exercise: The British Association of Sport and Exercise Sciences Guidelines. Eds: Payton C.J., Bartlett R.M. Abingdon, Oxon: Routledge; 77-102. [Google Scholar]
- Burden A. (2010) How should we normalize electomyograms obtained from healthy participants? What we have learned from over 25 years of research. Journal of Electromyography and Kinesiology 20, 1023-1035. [DOI] [PubMed] [Google Scholar]
- Cazzola D., Holsgrove T., Preatoni E., Gill H.S., Trewartha G. (2017) Cervical spine injuries: A whole-body musculoskeletal model for the analysis of spinal loading. PloS One 12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung S.H., Giuliani C.A. (1997) Within- and between-session consistency of electromyographic temporal patterns of walking in non-disabled older adults. Gait and Posture 6, 110-118. [Google Scholar]
- Clancy E.A., Morin E.L., Merletti R. (2002) Sampling, noise-reduction and amplitude estimation issues in surface electromyography. Journal of Electromyography and Kinesiology 12, 1-16. [DOI] [PubMed] [Google Scholar]
- De Luca C.J. (1997) The use of surface electromyography in biomechanics. Journal of Applied Biomechanics 13. [Google Scholar]
- De Luca C.J., Gilmore D.L., Kuznetsov K., Roy S.H. (2010) Filtering the surface EMG signal: Movement artifact and baseline noise contamination. Journal of Biomechanics 43, 1573-1579. [DOI] [PubMed] [Google Scholar]
- Finni T., Hu M., Kettunen P., Vilavuo T., Cheng S. (2007) Measurement of EMG activity with textile electrodes embedded into clothing. Physiological Measurement 28, 1405-1419. [DOI] [PubMed] [Google Scholar]
- Finni T., Uusi-Vähälä M., Pesola A.J., Taipale R.S. (2016) Do running and strength exercises reduce daily muscle inactivity time? AIMS Public Health 3, 702-721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giggins O.M., Persson U.M., Caulfield B. (2013) Biofeedback in rehabilitation. Journal of Neuroengineering and Rehabilitation 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guidetti L., Rivellini G., Figura F. (1996) EMG patterns during running: Intra- and inter-individual variability. Journal of Electromyography and Kinesiology 6, 37-48. [DOI] [PubMed] [Google Scholar]
- Häkkinen K., Pakarinen A., Kraemer W.J., Häkkinen A., Valkeinen H., Alén M. (2001) Selective muscle hypertrophy, changes in EMG and force, and serum hormones during strength training in older women. Journal of Applied Physiology 91, 569-580. [DOI] [PubMed] [Google Scholar]
- Heinonen A., Sievanen H., Viitasalo J., Pasanen P.O., Vuori I. (1994) Reproducibility of computer measurement of maximal isometric strength and electromyography in sedentary middle-aged women. European Journal of Applied Physiology and Occupational Physiology 68, 310-314. [DOI] [PubMed] [Google Scholar]
- Hermens H., Freriks B., Merletti R., Stegeman D., Blok J., Rau G., Disselhorst-Klug C., Hägg G. (1999) SENIAM 8: European recommendations for surface electromyography. Enschede, Holland: Roessingh Research and Development. [Google Scholar]
- Hodges P.W., Bui B.H. (1996) A comparison of computer-based methods for the determination of onset of muscle contraction using electromyography. Electroencephalography and Clinical Neurophysiology 101, 511-519. [DOI] [PubMed] [Google Scholar]
- Hopkins W.G. (2000) Measures of reliability in sports medicine and science. Sports Medicine 30, 1-15. [DOI] [PubMed] [Google Scholar]
- Hopkins W.G. (2007) A spreadsheet for deriving a confidence interval, mechanistic inference and clinical inference from a p value. Sportscience 11, 16-20, Available from URL: www.sportsci.org/2007/wghinf.htm [Google Scholar]
- Hopkins W.G. (2015) Spreadsheets for analysis of validity and reliability. Sportscience 19, 36-42, Available from: www.sportsci.org/2015/ValidRely.htm [Google Scholar]
- Hopkins W.G., Marshall S.W., Batterham A., Hanin J. (2009) Progressive statistics for studies in sports medicine and exercise science. Medicine and Science in Sports and Exercise 41, 3-12. [DOI] [PubMed] [Google Scholar]
- Karamanidis K., Arampatzis A., Brüggemann G.P. (2004) Reproducbility of electromyography and ground reaction force during various running techniques. Gait and Posture 19, 115-123. [DOI] [PubMed] [Google Scholar]
- Lintu N., Holopainen J., Hänninen O. (2005). Usability of textile-integrated electrodes for EMG measurements. In: Ambience Tampere, Finland. [Google Scholar]
- Lloyd D.G., Besier T.F. (2003) An EMG-driven musculoskeletal model to estimate muscle forces and knee joint moments in vivo. Journal of Biomechanics 36, 765-776. [DOI] [PubMed] [Google Scholar]
- Morin J.B., Gimenez P., Edouard P., Arnal P., Jiménez-Reyes P., Samozino P. M. B., Mendiguchia J. (2015) Sprint acceleration mechanics: The major role of hamstrings in horizontal force production. Frontiers in Physiology 6, 1-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pesola A.J., Laukkanen A., Haakana P., Havu M., Sääkslahti A., Sipilä S., Finni T. (2014) Muscle inactivity and activity patterns after sedentary time-targeted randomized controlled trial. Medicine and Science in Sports and Exercise 46, 2122-2131. [DOI] [PubMed] [Google Scholar]
- Pesola A.J., Laukkanen A., Tikkanen O., Finni T. (2016) Heterogeneity of muscle activity during sedentary behavior. Applied Physiology Nutrition and Metabolism 41, 1155-1162. [DOI] [PubMed] [Google Scholar]
- Rose J., McGill K.C. (2005) Neuromuscular activation and motor-unit firing characteristics in cerabral palsy. Developmental Medicine and Child Neurology 47, 329-336. [DOI] [PubMed] [Google Scholar]
- Scilingo E.P., Gemignani A., Paradiso R., Taccini N., Ghelarducci B., De Rossi D. (2005) Performance evaluation of sensing fabrics for monitoring physiological and biomechanical variables. IEEE Transactions on Information Technology in Biomedicine 9, 345-352. [DOI] [PubMed] [Google Scholar]
- Smoliga J.M., Myers J.B., Redfern M.S., Lephart S.M. (2010) Reliability and precision of EMG in leg, torso, and arm muscles during running. Journal of Electromyography and Kinesiology 20, e1-e9. [DOI] [PubMed] [Google Scholar]
- Tikkanen O., Haakana P., Pesola A., Häkkinen K., Rantalainen T., Havu M., Pullinen T., Finni T. (2013) Muscle activity and inactivity periods during normal daily life. PloS One 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tikkanen O., Kärkkäinen S., Haakana P., Kallinen M., Pullinen T., Finni T. (2014) EMG, heart rate, and accelerometer as estimators of energy expenditure in locomotion. Medicine and Science in Sports and Exercise 46, 1831-1839. [DOI] [PubMed] [Google Scholar]
- Tikkanen O., Sipilä S., Kuula A.-S., Pesola A., Haakana P., Finni T. (2016) Muscle activity during daily life in the older people. Aging Clinical and Experimental Research 28, 713-720. [DOI] [PubMed] [Google Scholar]
- Weir J.P. (2005) Quantifying test-retest reliability using the intraclass correlation coefficient and the SEM. Journal of Strength and Conditioning Research 19, 231-240. [DOI] [PubMed] [Google Scholar]
- Yang J.F., Winter D.A. (1983) Electromyography reliability in maximal and submaximal isometric contractions. Archives of Physical Medicine and Rehabilitation 64, 417-420. [PubMed] [Google Scholar]


