Abstract
Since cisplatin achieved clinical success, transition metal platinum (Pt) drugs have been effectively used for the treatment of cancer. Iridium (Ir) compounds are considered to be potential alternatives to Pt compounds, as they possess promising anticancer effects with minor side effects. Platelet activation is associated with the metastasis and progression of cancer, and also with arterial thrombosis. Therefore, it is necessary to develop novel, effective antithrombotic agents. An Ir (III)-derived complex, [Ir (Cp*) 1-(2-pyridyl)-3-(3-me-thoxyphenyl)imidazo[1,5-a]pyridine Cl]BF4 (Ir-3), was developed as a novel antiplatelet drug. Ir-3 exerted more potent inhibitory activity on platelet aggregation stimulated by collagen compared with other agonists, including thrombin. In collagen-activated platelets, Ir-3 also inhibited adenosine trisphosphate release, intracellular Ca+2 mobilization and surface P-selectin expression, as well as the phosphorylation of phospholipase Cγ2 (PLCγ2), protein kinase C (PKC), protein kinase B (Akt) and c-Jun N-terminal kinase (JNK) 1, but not p38 mitogen-activated protein kinase or extracellular signal-regulated kinases. Ir-3 did not markedly affect phorbol 12, 13-dibutyrate-stimulated platelet aggregation. Neither the adenylate cyclase inhibitor SQ22536 nor the guanylate cyclase inhibitor 1H-[1, 2, 4] oxadiazolo [4,3-a]quinoxalin-1-one significantly reversed the Ir-3-mediated inhibition of platelet aggregation. Furthermore, Ir-3 had no considerable diminishing effects on OH radical signals in collagen-stimulated platelets or Fenton reaction solution. In conclusion, Ir-3 serves a novel function in the inhibition of platelet aggregation through inhibiting the PLCγ2-PKC cascade, and the subsequent suppression of Akt and JNK1 activation. Therefore, Ir-3 may be a potential novel therapeutic agent for the treatment of thromboembolic disorders, or the interplay between platelets and tumor cells which contributes to tumor cell proliferation and progression.
Keywords: Ir (III)-derived complex, platelet activation, phospholipase Cγ2-protein kinase C cascade, protein kinase B, c-Jun N-terminal kinase 1
Introduction
Platelets are anucleate blood cells which serve crucial functions in thrombosis under physiological and pathological conditions. They are critical for maintaining the integrity of the vascular system and are the first-line defense against hemorrhage. On encountering subendothelial matrix exposed by an injury to a vessel, platelets adhere to the matrix, are activated and become adhesive to other platelets, leading to further aggregation (1). Platelets are involved in the pathogenesis of atherosclerosis-associated diseases, including coronary artery diseases and stroke. During platelet activation, the release of several mediators, including adenosine trisphosphate (ATP) and thromboxane A2, occurs in conjunction with relative intracellular Ca+2 ([Ca+2]i) mobilization, attracting additional platelets towards the injured endothelium and resulting in the thickening of the initial platelet monolayer. Finally, fibrinogen binds to its specific platelet receptor, completing the final common pathway for platelet aggregation.
Platelet activation has also been associated with the key steps of cancer progression, and platelets have been proposed to influence the development of malignancies via regulated events that may trigger the pathobiology of cancer growth (2). Platelets also interact with cancer cells and contribute to the critical steps of cancer metastasis, including tumor cell migration, invasion, and arresting of the tumor cell within the vasculature (2,3). During platelet activation, their contents may be released into the peritumoral space and subsequently enhance tumor cell extravasation and metastasis (4).
The chemically similar platinum (Pt) group elements (PGEs), including Pt, palladium, rhodium, ruthenium, osmium, and iridium (Ir), have exceptional catalytic qualities. These metals are resistant to chemical corrosion over a wide range of temperatures, having a high melting point, high mechanical strength and remarkable ductility (5). Ir is rich in the Earth's crust, with an average mass fraction of 1 µg/kg in crustal rock. In nature, Ir is found in alluvium deposits accompanied by Pt and other PGEs. This metal is obtained from Pt ores, and is also derived as a by-product in nickel mining and industry (6). Various metal complexes have been identified for use in anticancer therapy, leading to an increasing amount of associated research. In the drug development industry, metal complexes represent a highly resourceful platform. Apart from deviations in the metal and oxidation state, metal ions have variable geometries and coordination numbers that allow the modification of their chemical reactivity in terms of kinetics (ligand exchange rates) and thermodynamics (including metal-ligand bond strength and redox potentials). Metals, as well as their ligands, are involved in numerous biological activities, ranging from outer-sphere recognition of target sites to the activity of any released ligands and ligand-centered redox processes (7).
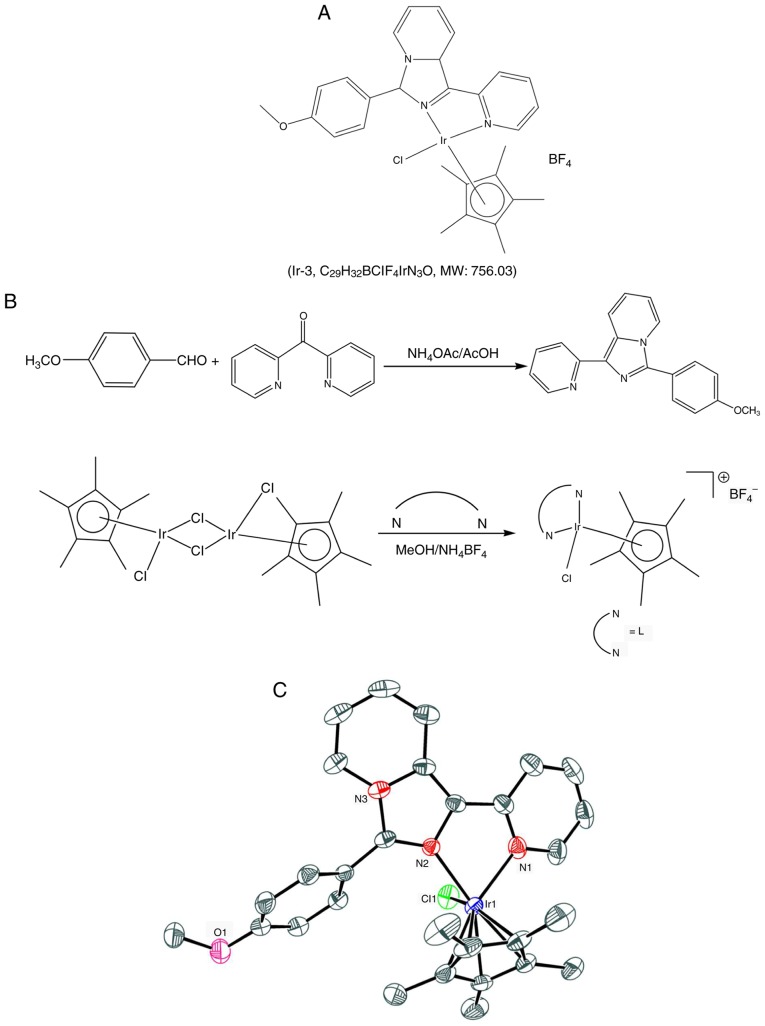
Organometallic Ir (III) complexes are particularly promising in terms of anticancer treatments. Previous research has focused on Ir (III) compounds due to their potential anti-tumor activity and low toxicity towards normal tissues (8,9). Furthermore, Ir complexes exert potent antiangiogenic effects by activating distinct antiangiogenic signaling pathways (8), and antiangiogenic therapy is considered a promising cancer treatment strategy. On the basis of these observations, our group obtained novel biologically active Ir (III) derivatives by developing a new Ir (III) compound, also referred to as Ir-3 (Fig. 1). Although certain experimental and animal studies have demonstrated that Ir-based compounds have potent anticancer activity, to date, no study has investigated their effects on platelet aggregation. The preliminary results of the present study revealed that Ir-3 exhibits potent antiplatelet activity in humans, encouraging further examination of the characteristics and functional activity of Ir-3 in platelet activation. The results of the present study provide valuable evidence for the development of a novel class of Ir-3-based antiplatelet agents.
Figure 1.
A novel, biologically active Ir (III) compound. (A) Chemical structure of Ir-3 and (B) synthesis of the ligand (L) 1-(2-pyridyl)-3-(3-methoxyphenyl) imidazo[1,5-a]pyridine and complex (Ir-3) [Ir(Cp*)(L)Cl]BF4. (C) Oak Ridge Thermal Ellipsoid Plot scheme of Ir-3. Hydrogen atoms and the BF4 anion are omitted for clarity.
Materials and methods
Chemicals and reagents
Thrombin, collagen, arachi-donic acid (AA), luciferin-luciferase, U46619, phorbol 12,13-dibutyrate (PDBu), nitroglycerin (NTG), heparin, prostaglandin E1 (PGE1), 5,5-dimethyl-1-pyrroline N-oxide (DMPO), SQ22536, 1H-[1,2,4]oxadiazolo[4,3-a]quinox-alin-1-one (ODQ), and bovine serum albumin (BSA) were purchased from Sigma-Aldrich; Merck KGaA (Darmstadt, Germany). Fura-2AM was obtained from Molecular Probes; Thermo Fisher Scientific, Inc. (Waltham, MA, USA). Anti-phosphorylated (p)-p38 mitogen-activated protein kinase (MAPK) Thr180/Tyr182 monoclonal antibodies (mAbs; Cat. no. 9211) were purchased from Santa Cruz Biotechnology, Inc. (Dallas, TX, USA). Anti-p38 MAPK (Cat. no. 9217), anti-p-c-Jun N-terminal kinase (JNK; Thr183/Tyr185; Cat. no. 9251), and anti-p44/42 extracellular signal-regulated kinase (ERK) mAbs (Cat. no. 9107), as well as anti-phospholipase Cγ2 (PLCγ2; Cat. no. 3872), anti-p-(Tyr759) PLCγ2 (Cat. no. 3874), anti-p-(Ser) protein kinase C (PKC) substrate (pleckstrin; p-p47; Cat. no. 2261), anti-JNK (Cat. no. 9252), and anti-p-p44/p42 ERK (Thr202/Tyr204) polyclonal antibodies (pAbs; Cat. no. 9101) were purchased from Cell Signaling Technology, Inc. (Danvers, MA, USA). Anti-p-protein kinase B (Akt) (Ser473; Cat. no. 9271) and anti-Akt mAbs (Cat. no. 2920) were purchased from Biovision (Mountain View, CA, USA). An anti-pleckstrin (p47) pAb (Cat. no. GTX17020) was purchased from Gene Tex (Irvine, CA, USA). A Hybond-P polyvinylidene fluoride (PVDF) membrane, an enhanced chemiluminescence western blotting detection reagent, horseradish peroxidase (HRP)-conjugated donkey anti-rabbit immunoglobulin G (IgG; Cat. no. RPN4301) and sheep anti-mouse IgG (Cat. no. RPN4201) were purchased from GE Healthcare Life Sciences (Little Chalfont, UK). A fluorescein isothiocyanate (FITC)-conjugated anti-human CD42P (P-selectin) mAb (Cat. no. 304904) was obtained from Bio Legend, Inc. (San Diego, CA, USA).
Synthesis of 1-(2-pyridyl)-3-(3-methoxyphenyl)imidazo[1,5-a] pyridine (L)
Ammonium acetate (1.93 g, 25 mM), 4-methoxy benzaldehyde (1.02 g, 7.5 mM) and glacial acetic acid (25 ml) were added to a degassed mixture of di-pyridin-2-yl-metha-none (0.9 g, 5 mM), and the mixture was placed in an oil bath maintained at 120°C under a nitrogen atmosphere for 18 h. The reaction mixture was then cooled to room temperature, poured into ice cold water, and extracted with chloroform (3×70 ml). The organic layer was dried over anhydrous sodium sulfate, and then filtered. The residue was chromatographed through silica gel (ethylacetate/hexane = 1:3) to yield a yellow solid. The characteristics of this solid were as follows: Melting point: 105–110°C; 1H nuclear magnetic resonance (NMR; 400 MHz, CDCl3): δ 8.72–8.70 (d, 1H, J=8 Hz), 8.65–8.64 (d, 1H, J=4 Hz), 8.27–8.25 (d, 1H, J=8 Hz), 8.2–8.19 (d, 1H, J=8 Hz), 7.78–7.73 (t, 3H, J=10 Hz), 7.13–7.07 (m, 3H, J=8 Hz), 6.96–6.92 (t, 1H, J=8 Hz), 6.67–6.64 (t, 1H, J=6 Hz), 3.9 (s, 3H); 13C NMR (400 MHz, CDCl3) 160.0, 155.1, 148.9, 138.0, 136.1, 130.1, 129.9, 129.7, 122.5, 121.7, 121.5, 120.8, 120.3, 119.8, 114.4, 113.6, 55.3; ultraviolet-visible spectrophotometry (UV-Vis; λabs, nm): 380, 321, 300, 238; electrospray ionization-mass spectrometry (ESI-MS; m/z): 301 (M+).
Synthesis of [Ir(Cp*)(L)Cl]BF4 (Ir-3)
[Ir(Cp*)(Cl)2]2 (0.16 g, 0.2 mM) in methanol (10 ml) solution was added dropwise to 10 ml methanolic solution of 1-(2-pyridyl)-3-(3-methoxy-phenyl)imidazo[1,5-a]pyridine (L) (0.12 g, 0.4 mM), and the solution was stirred at room temperature for 3 h. Subsequently, NH4BF4 (200 mg, 0.60 mM) was added to the initial pale yellow solution, changing the color to orange. After 24 h, the solution was evaporated and the solid obtained was filtered. The residue was washed with diethyl ether (40 ml) and dried under a vacuum. The desired product was recrystallized from the dichloromethane/hexane mixture, yielding orange micro-crystals. The characteristics of this solid were as follows: 1H NMR (400 MHz, dimethyl sulfoxide [DMSO]-D6): δ 8.82–8.81 (d, 1H, J=4 Hz), 8.52–8.39 (m, 3H), 8.17–8.10 (m, 3H), 7.54–7.47 (m, 2H), 7.31–7.29 (d, 2H, J=8 Hz), 7.15–7.11 (t, 1H, J=8 Hz), 3.90 (s, 3H), 1.29 (s, 15H); UV-Vis (λabs, nm) (ε, M−1 cm−1): 406 (1649), 385 (2372), 367 (1854), 282 (2871), 242 (2881); ESI-MS (m/z): 664.08 [M-BF4]+ (Fig. 1).
Platelet aggregation
The present study was approved by the Institutional Review Board of Taipei Medical University (Taipei, Taiwan; approval no. TMU-JIRB-N201612050) and conformed to the directives of the Declaration of Helsinki. All human volunteers involved in the present study provided written informed consent. Human platelet suspensions were prepared as described previously (10). Human blood was collected from 20 healthy individuals (aged between 20 and 30 years old, 12 females and 8 males) of Taipei Medical University who had taken no drugs or other substances that would interfere with the experiment for at least 14 days prior to collection between July 2017 and August 2017. The collected blood was mixed with an acid-citrate-dextrose solution (9:1, v/v) and centrifuged at 120 × g for 10 min at 37°C to separate the platelet-rich plasma (PRP). Following centrifugation, the PRP was supplemented with 0.5 µM PGE1 and 6.4 IU/ml heparin, incubated for 10 min at 37°C and centrifuged at 500 × g for 10 min at room temperature. The collected pellets were mixed with 5 ml Tyrode's solution, pH 7.3 [containing (mM) NaCl 11.9, KCl 2.7, MgCl2 2.1, NaH2PO4 0.4, NaHCO3 11.9, and glucose 11.1]. Then, apyrase (1.0 U/ml), PGE1 (0.5 µM) and heparin (6.4 IU/ml) were added, and the mixture was incubated for 10 min at 37°C. Following centrifugation of the suspensions at 500 × g for 10 min at room temperature, the washing procedure was repeated. The washed platelets were finally suspended in Tyrode's solution containing BSA (3.5 mg/ml). The final Ca2+ concentration in the Tyrode solution was 1 mM. A platelet aggregation test was performed using a Lumi-Aggregometer (Payton Associates, Scarborough, ON, Canada) as described previously (10). Various concentrations of Ir-3 or a solvent control (0.1% DMSO) were preincubated with platelet suspensions (3.6×108 cells/ml) for 3 min prior to the addition of various concentrations of agonists (1 µg/ml collagen, 0.01 U/ml thrombin, 1 µM U46619 or 120 µM AA). The extent of platelet aggregation was calculated as the percentage with respect to the control (absence of Ir-3) in light transmission units when the reaction had proceeded for 6 min. For an ATP release assay, 20 µl luciferin-luciferase was added 1 min prior to the addition of the agonist, and the amount of ATP released was compared with that released by the control using a Lumi-Aggregometer.
Measurement of [Ca2+]i mobilization using Fura-2AM fluorescence
The [Ca2+] concentration was measured using the calcium sensitive dye Fura-2AM, as described previously (10). In brief, citrated whole blood was centrifuged at 120 × g for 10 min at room temperature, and the supernatant was collected and incubated with 5 µM Fura-2AM for 1 h at 37°C. The Fura-2AM-preincubated platelets were washed with Tyrode's solution and treated with Ir-3 in the presence of 1 mM CaCl2, and then stimulated with collagen (1 µg/ml). Fura-2 fluorescence was measured using excitation wavelengths of 340 and 380 nm and emission at 510 nm with a spectrofluo-rometer (Hitachi FL Spectrophotometer F-4500; Hitachi, Ltd., Tokyo, Japan).
Detection of lactate dehydrogenase
Washed platelets (3.6×108 cells/ml) were preincubated with 20–100 µM Ir-3 or the solvent control (0.1% DMSO) for 20 min at 37°C. Following incubation, supernatant (10 µl) was added to a Fuji Dri-Chem slide lactate dehydrogenase (LDH)-PIII (Fujifilm Holdings Corporation, Tokyo, Japan). The absorbance of the supernatant was measured at 540 nm using a UV-Vis spectrophotometer (UV-160; Shimadzu Corporation, Kyoto, Japan). A maximal value of LDH was recorded in platelets lysed with Triton.
Flow cytometric analysis
Platelet surface P-selectin expression was examined using flow cytometric analysis. Washed platelets were prepared as aforementioned. Aliquots of platelet suspensions (3.6×108 cells/ml) were treated with either the solvent control (0.1% DMSO) or Ir-3 (10 and 20 µM), and FITC-conjugated P-selectin (2 µg/ml) mAbs were then added and incubated for 3 min at 37°C. This was followed by the addition of collagen (1 µg/ml) to trigger platelet activation, for 1 min at 37°C. The suspensions were then assayed for fluorescein-labeled platelets using a flow cytometer (FACScan System; BD Biosciences, San Jose, CA, USA) with FACSuite™ software (version 1.0.5.3841; BD Biosciences). Data were collected from 50,000 platelets per experimental group, and the platelets were identified on the basis of their characteristic forward and orthogonal light-scattering profiles. All experiments were repeated at least three times to ensure reproducibility.
Western blotting
Washed platelets (1.2×109 cells/ml) were preincubated with Ir-3 (10 and 20 µM) or the solvent control (0.1% DMSO) for 3 min at 37°C, and 1 µg/ml collagen was added to trigger platelet activation for 5 min at 37°C. The reaction was then stopped, and the platelets were immediately resuspended in 200 µl lysis buffer [containing (mM) 50 HEPES, 5 EDTA, 50 NaCl and 1% Triton X-100]. Proteins were quantified using an ELISA reader at 570 nm. Samples containing 80 µg of protein were separated through 12% sodium dodecyl sulfate gel electrophoresis, and the proteins were electrotrans-ferred to PVDF membranes using a Bio-Rad semi-dry transfer unit (Bio-Rad Laboratories, Inc., Hercules, CA, USA). The blots were then blocked with Tris-buffered saline in Tween-20 (TBST; 10 mM Tris-base, 100 mM NaCl, and 0.01% Tween-20) containing 5% BSA for 1 h at room temperature, and probed with the aforementioned primary antibodies (diluted 1:1,000 in TBST) for 2 h at 4°C. The membranes were incubated with HRP-conjugated anti-mouse IgG for anti-ERK, anti-p38 and anti-Akt or anti-rabbit IgG for anti-phosphorylated MAPKs, anti-p-Akt, anti-p-PLCγ2, anti-PLCγ2, anti-p-PKC substrate and anti-JNK (diluted 1:3,000 in TBST) for 1 h at 4°C. An enhanced chemiluminescence system was used to detect immunoreactive bands, and their optical density was quantified using Bio-profil Biolight software (version V2000.01; Vilber Lourmat, Marne-la-Vallée, France).
Measurement of OH radical formation in platelet suspensions and Fenton reaction solution through electron spin resonance (ESR) spectrometry
Electron spin resonance spectrometry was performed on a Bruker EMX ESR spectrometer (Bruker Corporation, Billerica, MA, USA) as described previously (11). Platelet suspensions (3.6×108 cells/ml) or Fenton reaction solution (50 µM FeSO4+2 mM H2O2) were preincu-bated with 0.1% DMSO or Ir-3 (10 and 20 µM) for 3 min at room temperature, with or without the addition of 1 µg/ml collagen. Following incubation of the suspensions for 5 min, 100 µM DMPO was added prior to the execution of ESR spectrometry. The ESR spectra were recorded using a quartz flat cell designed for aqueous solutions. The spectrometer was operated at a power of 20 mW, frequency of 9.78 GHz, scan range of 100 G and receiver gain of 5×104. The modulation amplitude was 1 G, the time constant was 164 ms, and scanning was performed for 42 sec, with the spectra being the sum of three scans.
Statistical analysis
The experimental results are expressed as the mean ± standard error of the mean, and are accompanied by the number of observations (n). The n values refer to the number of experiments, and each experiment was conducted using different blood donors. The between-group differences in the experiments were assessed through one-way analysis of variance (ANOVA). When the ANOVA indicated significant differences among the group means, the groups were compared using the Student-Newman-Keuls method. Statistical analyses were performed using SAS (version 9.2; SAS Institute, Inc., Cary, NC, USA). P<0.05 was considered to indicate a statistically significant difference.
Results
Inhibitory effects of Ir-3 on platelet aggregation in washed human platelets
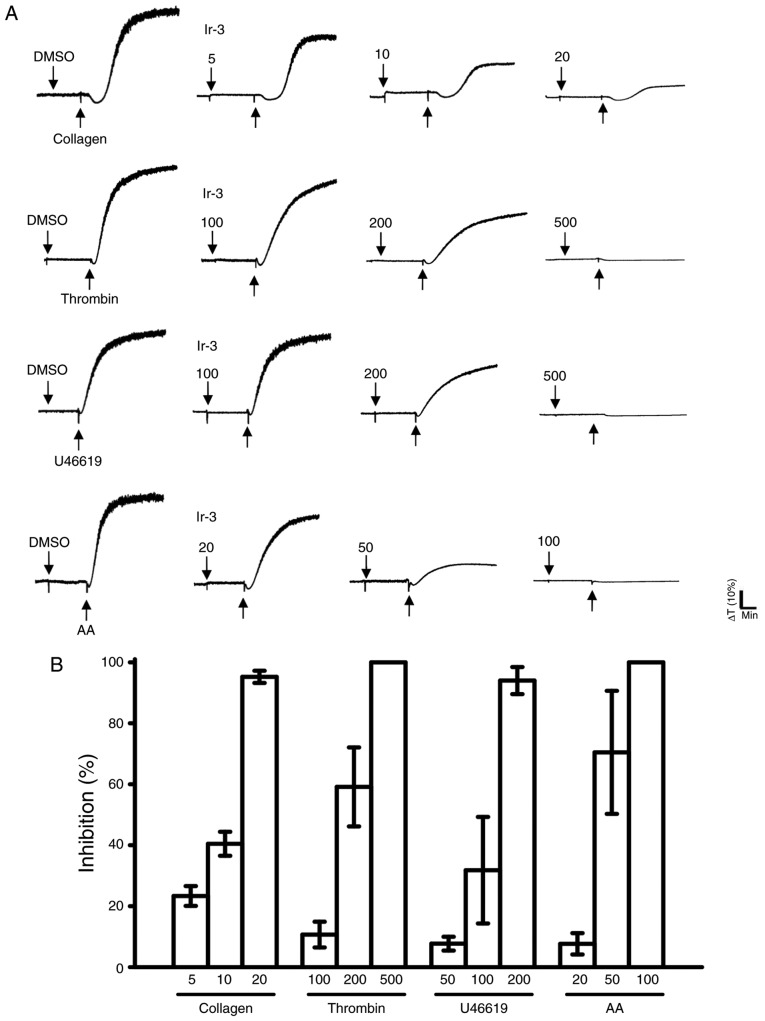
Ir-3 (5–20 µM; Fig. 2) inhibited platelet aggregation in collagen (1 µg/ml)-stimulated human platelets in a concentration-dependent manner. Ir-3 (20–100 µM) demonstrated moderate activity against platelet aggregation in platelets stimulated by 120 µM AA. Furthermore, Ir-3 (100–500 µM) had relatively weak activity against 0.01 U/ml thrombin or 1 µM U46619, a prostaglandin endoperoxide stimulant, indicating that Ir-3 had more potent inhibitory activity against collagen stimulation compared with other agonists (Fig. 2B). The solvent control, 0.1% DMSO, did not affect platelet aggregation (Fig. 2A). Furthermore, aspirin (20, 50 and 100 µM) inhibited platelet aggregation stimulated by 1 µg/ml collagen in a concentration-dependent manner, with a half maximal inhibitory concentration of ~50 µM (n=3; data not shown). Therefore, Ir-3 is ~5 times more potent than aspirin at inhibiting collagen-stimulated platelet aggregation. In the following experiments, 1 µg/ml collagen was used as an agonist for investigating potential inhibitory mechanisms of Ir-3 in human platelets.
Figure 2.
Comparison of the relative inhibitory activity of Ir-3 against platelet aggregation stimulated by various agonists in washed human platelets. (A) Washed human platelets (3.6×108 cells/ml) were pre-incubated with the solvent control (0.1% DMSO) or Ir-3 (5–100 µM) and subsequently treated with 1 µg/ml collagen, 0.01 U/ml thrombin, 1 µM U46619 or 120 µM AA to stimulate platelet aggregation. (B) Concentration-response histograms of Ir-3 against platelet aggregation stimulated by the indicated agonists. Data are presented as the mean ± standard error of the mean (n=3). AA, arachidonic acid.
Effects of Ir-3 on ATP release reaction, relative [Ca+2] mobilization and surface P-selectin expression
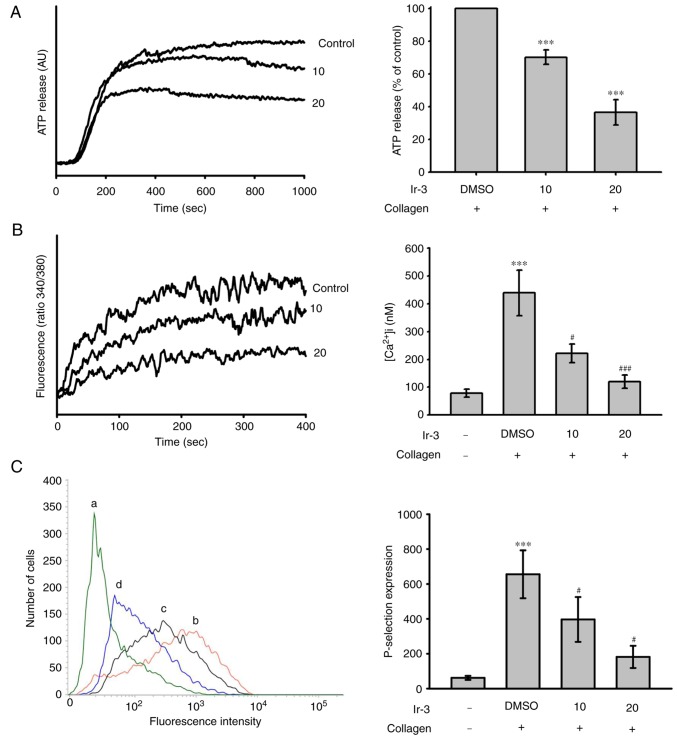
The activation of platelets releases the granular contents, including adenosine diphosphate (ADP)/ATP and Ca+2 from the dense granules, and surface P-selectin from the α-granules, to the external environment where they are involved in substantial platelet aggregation. In the present study, Ir-3 (10 and 20 µM) inhibited the ATP release reaction (10 µM, 33.2±1.1%; 20 µM, 67.8±3.7%; Fig. 3A) and relative [Ca2+]i mobilization (resting control, 78.4±14.3 nM; collagen-stimulated, 438.9±82.6 nM; 10 µM, 221.9±33.3 nM; 20 µM, 119.1±23.8 nM; n=3, Fig. 3B) in platelets stimulated by 1 µg/ml collagen. The corresponding statistical data are presented in the right panels of Fig. 3. In quiescent platelets, P-selectin is located on the inner wall of the α-granules. Platelet activation exposes the inner walls of the granules to the outside of the cell (12). Ir-3 treatment significantly reduced collagen-induced surface P-selectin expression, as demonstrated by the corresponding statistical data in the right panel (resting control, 61.7±11.5; collagen-activated, 655.7±137.3; 10 µM, 396.7±128.8; 20 µM, 181.7±63.8; n=3; Fig. 3C).
Figure 3.
Effects of Ir-3 on ATP release, relative [Ca2+] mobilization and surface FITC-P-selectin expression in human platelets. Washed platelets (3.6×108 cells/ml) were pre-incubated with the solvent control (0.1% DMSO) or Ir-3 (10 and 20 µM), and collagen (1 µg/ml) was added to trigger either (A) the ATP release reaction or (B) relative [Ca2+] mobilization. (C) Washed platelets (3.6×108/ml) were preincubated with the solvent control (0.1% DMSO) or Ir-3 (10 and 20 µM) and FITC-P-selectin (2 µg/ml) for 3 min and then stimulated by collagen (1 µg/ml). The corresponding statistical data are presented in the right panel of each Figure section. Data are presented as the means ± standard error of the mean (n=3). ***P<0.001 vs. the DMSO-treated group in section A or resting control in section B and C; #P<0.05 and ###P<0.001 vs. the DMSO-treated group in sections B and C. ATP, adenosine trisphosphate; FITC, fluorescein isothiocyanate; AU, arbitrary unit.
Influence of Ir-3 on LDH release and cyclic nucleotide formation in washed human platelets
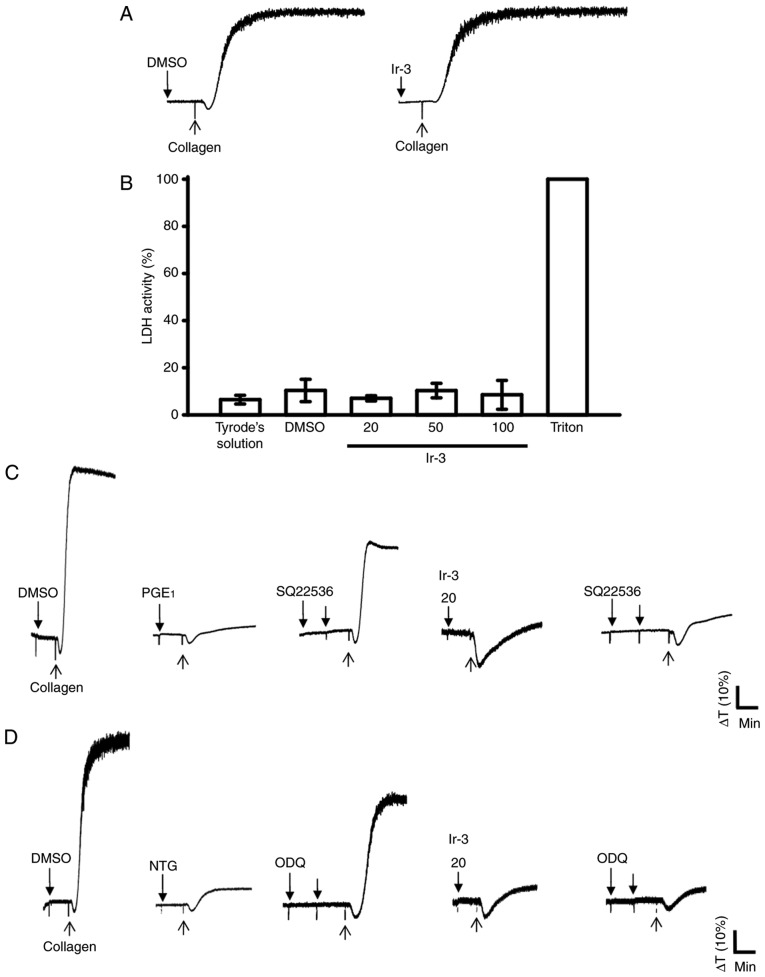
The aggregation curves of platelets pre-incubated with 100 µM Ir-3 for 10 min and washed two times with Tyrode solution were not significantly different from those of platelets pre-incubated with the solvent control (0.1% DMSO) under equivalent conditions (Fig. 4A), preliminarily indicating that the effects of Ir-3 on platelet aggregation are reversible and non-cytotoxic. Furthermore, the LDH results revealed that Ir-3 (20, 50, and 100 µM) incubated with platelets for 20 min did not significantly increase LDH activity or exert cytotoxic effects on platelets (Fig. 4B), demonstrating that Ir-3 does not affect platelet permeability or induce platelet cytolysis. Furthermore, 100 µM SQ22536, an adenylate cyclase inhibitor, and 10 µM ODQ, a guanylate cyclase inhibitor, significantly reversed the inhibition of collagen-induced platelet aggregation mediated by 1 µM PGE1 or 10 µM NTG (Fig. 4C and D). Neither SQ22536 nor ODQ significantly reversed the inhibition of collagen-induced platelet aggregation mediated by 20 µM Ir-3 (Fig. 4C and D), indicating that the mechanisms of Ir-3-mediated inhibition of platelet aggregation are independent of increasing cyclic nucleotide formation.
Figure 4.
Influence of Ir-3 on cytotoxicity, LDH release and cyclic nucleotide formation in human platelets. (A) Washed platelets were pre-incubated with the solvent control (0.1% DMSO) or Ir-3 (100 µM) for 10 min and subsequently washed two times with Tyrode's solution. Collagen (1 µg/ml) was then added to trigger platelet aggregation. (B) Washed platelets (3.6×108/ml) were pre-incubated with the solvent control (0.1% DMSO) or Ir-3 (20, 50, and 100 µM) for 20 min, and a 10-µl aliquot of the supernatant was deposited on a Fuji Dri-Chem slide LDH-PIII. For other experiments, washed platelets (3.6×108 cells/ml) were pre-incubated with (C) 1 µM PGE1, (D) 10 µM NTG, or Ir-3 (20 µM) in the absence or presence of 100 µM SQ22536 or 10 µM ODQ, and were subsequently treated with 1 µg/ml collagen to induce platelet aggregation. Data are presented as the mean ± standard error of the mean (n=3). Profiles in sections A, C and D represent the four independent experiments. LDH, lactate dehydrogenase; PGE1, prostaglandin E1; NTG, nitroglycerin; ODQ, 1H-[1,2,4] oxadiazolo[4,3-a]quinoxalin-1-one.
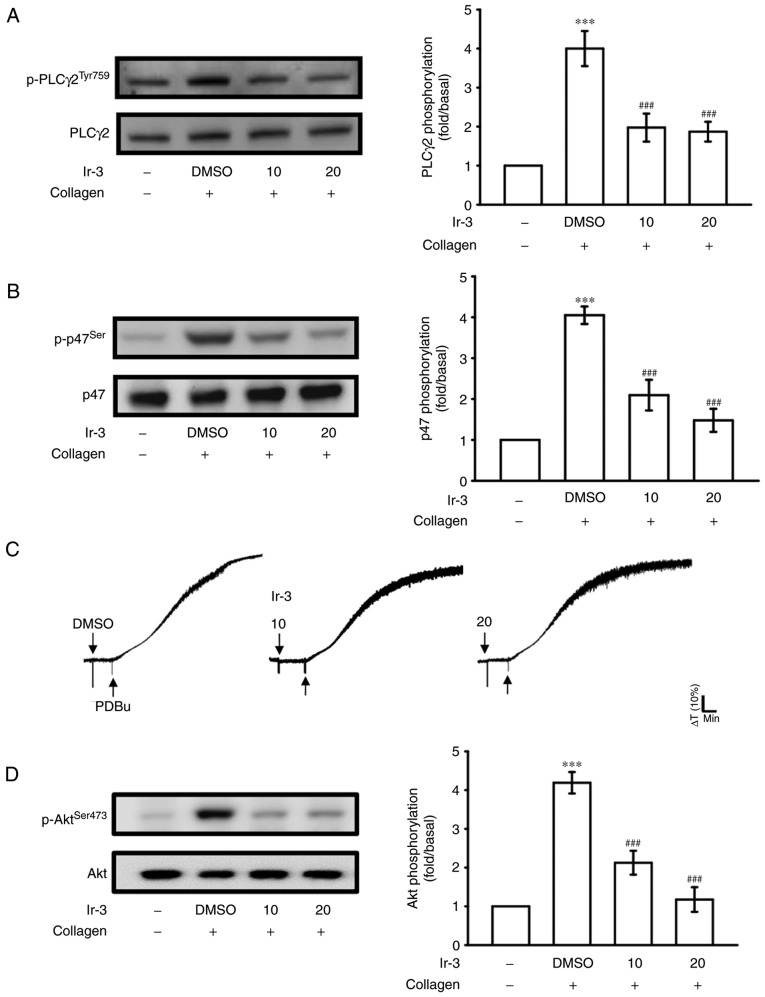
Regulatory character of Ir-3 on the PLCγ2/PKC cascade and Akt activation
PLCs hydrolyze phosphatidylinositol 4,5-bisphosphate to generate the secondary messengers inositol 1,4,5-trisphosphate (IP3) and diacylglycerol (DAG). IP3 triggers relative [Ca2+] mobilization and DAG activates PKC, yielding a ~47-kDa protein that is predominantly phosphorylated (p47 protein; pleckstrin) and leads to the ATP release reaction (13). Fig. 3A and B illustrate the inhibitory effects of Ir-3 against ATP release and relative [Ca2+] mobilization. The influence of Ir-3 on the phosphorylation of the PLCγ2-PKC signaling cascade was then investigated further. At 10 and 20 µM, Ir-3 significantly reduced PLCγ2 phosphorylation and PKC activation (pleckstrin phosphorylation) in collagen-stimulated platelets compared with the DMSO control (Fig. 5A and B). However, Ir-3 had no significant effects on platelet aggregation stimulated by 150 nM PDBu, a PKC activator (Fig. 5C), indicating that Ir-3 does not directly disturb PKC activation but may interfere with upstream regulators of PKC, including PLCγ2. Akt is a serine/threonine-specific protein kinase that serves a key function in multiple cellular processes, including platelet activation, cell proliferation, apoptosis and cell migration (14). Ir-3 (10 and 20 µM) significantly inhibited collagen-induced Akt phosphorylation (Fig. 5D) compared with the DMSO control, demonstrating the involvement of Akt signaling pathway inhibition in Ir-3-mediated inhibition of platelet activation.
Figure 5.
Inhibitory effects of Ir-3 on PLCγ2, PKC, and Akt activation in platelets. Washed platelets were preincubated with the solvent control (0.1% DMSO) or Ir-3 (10 and 20 µM) and subsequently treated with 1 µg/ml collagen or 150 nM PDBu to induce (A) PLCγ2 and (B) PKC activation (pleckstrin phosphorylation) or (C) platelet aggregation and (D) Akt activation. The profiles in section C are representative of four independent experiments. Data are presented as the mean ± standard error of the mean (n=4). ***P<0.001 vs. resting control and ###P<0.001 vs. the DMSO-treated group. PLCγ2, phospholipase Cγ2; PKC, protein kinase C; Akt, protein kinase B.
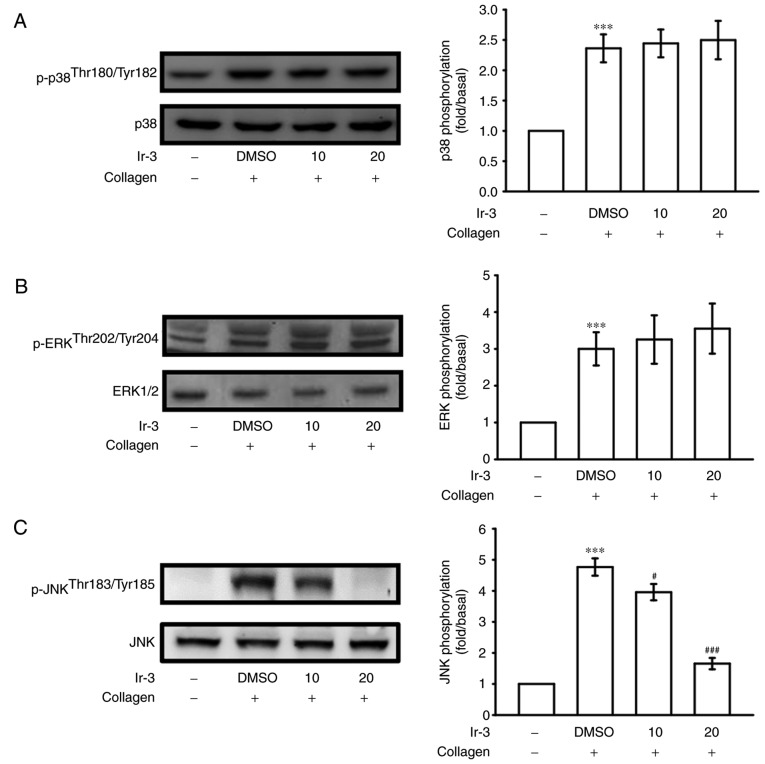
Inhibitory effects of Ir-3 on p38 MAPK, ERKs and JNK1 phosphorylation
To investigate the inhibitory mechanisms of Ir-3 in platelet activation, several signaling molecules of the MAPK phosphorylation pathway were evaluated. MAPKs, including p38 MAPK, ERKs, and JNKs, regulate major cellular responses in eukaryotic organisms and contribute to cell proliferation, migration, differentiation, and apoptosis (15). ERKs, JNK1, and p38 MAPK have been identified in platelets (16). Although collagen-induced p38 MAPK and ERK phosphorylation was unaffected by Ir-3, JNK1 phosphorylation was suppressed by 10 and 20 µM Ir-3 in a concentration-dependent manner (Fig. 6). These results suggest that only JNK1 may be involved in the antiplatelet activity of Ir-3.
Figure 6.
Effects of Ir-3 on p38 MAPK, ERKs, and JNK1 phosphorylation in collagen-activated platelets. Washed platelets (1.2×109 cells/ml) were pretreated with the solvent control (0.1% DMSO) or Ir-3 (10 and 20 µM) and then treated with 1 µg/ml collagen to stimulate platelet activation. Platelets were collected, and their subcellular extracts were analyzed to determine the levels of (A) p38 MAPK (n=3) (B) ERKs (n=3) and (C) JNK1 phosphorylation (n=4). Data are presented as the mean ± standard error of the mean. ***P<0.001 vs. resting control and ###P<0.001 vs. the DMSO-treated group. MAPK, mitogen-activated protein kinase; ERK, extracellular signal-regulated kinase; JNK, c-Jun N-terminal kinase.
Determination of the function of the OH radical (OH·) in the Ir-3-mediated inhibition of platelet aggregation through ESR spectrometry
An ESR signal indicative of OH· formation was observed in collagen-stimulated platelet suspensions and Fenton reaction solution (cell-free system; Fig. 7A and B). A typical OH· signal (aN =aH = 14.8 G) and a long-lived g=2.005 radical detectable using the spin trap DMPO were observed in collagen-stimulated platelets, whereas this signal was not detected in resting platelets (Fig. 7A and B). Treatment with 10 and 20 µM Ir-3 did not notably diminish the OH· signals in collagen-stimulated platelet suspensions or Fenton reaction solution (Fig. 7), suggesting that the Ir-3-mediated inhibition of platelet activation may not be mediated through the reduction of free radical formation.
Figure 7.
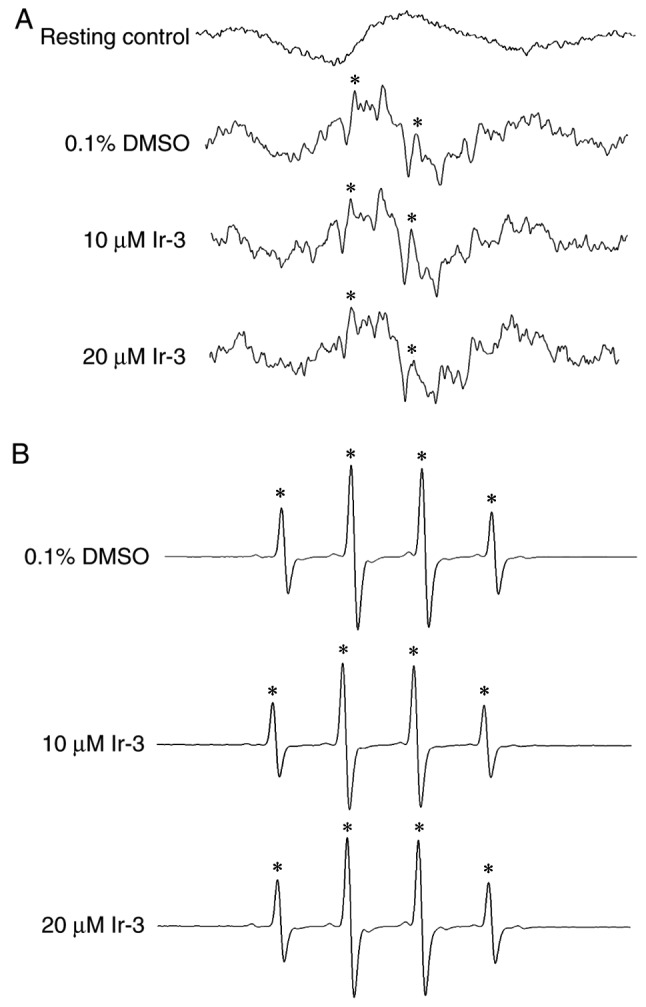
Effect of Ir-3 on OH· formation in human platelets or Fenton reaction solution, investigated through ESR spectrometry. (A) Washed platelets were pre-incubated with Tyrode's solution (resting control) or treated with 0.1% DMSO, 10 or 20 µM Ir-3. Collagen (1 µg/ml) was added to trigger OH· formation. (B) For the cell-free Fenton reaction study, 0.1% DMSO, 10 or 20 µM Ir-3 was pre-incubated with the solution, and DMPO was then added for the ESR experiments. Profiles are representative of four independent experiments. An asterisk (*) indicates OH· formation. OH·, OH radical; ESR, electron spin resonance; DMPO, 5,5-dimethyl-1-pyrroline N-oxide.
Discussion
Platelet activation is associated with thrombotic events in patients with cancer (17). Chemotherapeutics may amplify this effect and stimulate vascular thromboembolic events (VTEs) by aggravating endothelial damage, increasing platelet aggregation and increasing oxidative damage, leading to vascular toxicity (18). Among Pt-based chemotherapeutics, cisplatin has a high prevalence of treatment-associated VTEs (19). Gemcitabine in combination with a Pt-based agent is associated with increased thrombotic and vascular side effects (20,21). Therefore, research has focused on the development of novel metal-based agents for the inhibition of platelet activation to treat vascular disease, reduce toxic side effects and overcome Pt resistance. To the best of our knowledge, the present study is the first to demonstrate that Ir-3, an unique synthetic Ir (III)-derived compound, displays effective antiplatelet activity in human platelets in addition to its antitumor activity.
Platelets adhere to subendothelial matrix proteins, including collagen, thus altering their shape and releasing their granular contents, which include ATP, Ca+2 and P-selectin. Intracellular [Ca2+] mobilize as a result of several agonists, including collagen, thrombin and AA, to phosphorylate the Ca2+/calmodulin-dependent myosin light chain (20 kDa), which is involved in the release of serotonin and ATP (22), and platelet aggregation. Therefore, the inhibition of [Ca2+] mobilization and ATP production are crucial for assessing the antiplatelet effects of a compound. In the present study, Ir-3 inhibited platelet aggregation to different degrees, depending on the agonist used (either collagen, U46619, AA or thrombin), indicating that Ir-3 did not act at the specific individual receptors of these agonists. Therefore, Ir-3 may exert its inhibitory effects on stimulated platelets through a common signaling cascade.
Furthermore, platelet activation by collagen, substantially alters PLC expression, and results in IP3 and DAG production, which activates PKC and consequently induces p47 phosphorylation (13). PKC activation triggers specific responses, assisting in the transmission of activating signals in different cellular compartments. The PLCγ family is comprised of the isozymes PLCγ1 and PLCγ2, with PLCγ2 being involved in collagen-dependent signaling in platelets (23). Ir-3 diminished collagen-induced PLCγ2-PKC activation; however, Ir-3 was not effective on PKC activation as it did not inhibit PDBu-induced platelet aggregation, suggesting that the Ir-3-mediated inhibition of platelet activation involves PLCγ2 downstream signaling. This result also explains how Ir-3 was more efficacious at inhibiting platelet aggregation induced by collagen than that induced by thrombin, U46619, and AA.
Cyclic nucleotides are important modulators of platelet activation (24), and intracellular cyclic AMP- and cyclic GMP are involved in the inhibition of human platelet activation. At elevated levels, cyclic nucleotides prevent several platelet responses and reduce [Ca2+] levels through Ca2+ uptake by the dense tubular system, thereby suppressing PLC and PKC activation (24). Therefore, cyclic AMP and cyclic GMP synergistically inhibit platelet activation. Furthermore, neither SQ22536, an inhibitor of adenylate cyclase, nor ODQ, an inhibitor of guanylate cyclase, significantly reversed the Ir-3-mediated inhibition of collagen-induced platelet aggregation. Therefore, the Ir-3-mediated mechanisms were independent of increasing cyclic nucleotide formation in platelets.
Akt is a downstream molecule of phosphoinositide 3-kinase (PI3K). Akt-deficient mice reported defective effects in agonist-induced platelet activation, suggesting that Akt regulates platelet activation, and such regulation may have consequences concerning thrombosis (14,25). Therefore, selective inhibitors of Akt isoforms or its activating proteins, including individual PI3K isoforms, may be attractive antithrombotic therapy targets (14). In addition, MAPKs are activated by specific MAPK kinases (MEKs). Specifically, MEK1/2, MEK3/6 and MEK4/7 activate ERKs, p38 MAPK and JNKs, respectively (26). Cytosolic phospholipase A2 (cPLA2) is a substrate of p38 MAPK activity induced by various agonists, including von Willebrand factor (vWF) and thrombin (27). Therefore, p38 MAPK is essential for cPLA2 stimulation and AA release (28). This may explain how Ir-3 was less able to inhibit p38 MAPK activation and thrombin- or AA-stimulated platelet aggregation. ERK activation is involved in platelet aggregation but requires prior ATP release, which triggers a P2X1-mediated Ca2+ influx and activates ERKs, thereby increasing the phosphorylation of myosin light-chain kinase (27). JNK1 is the most recently identified MAPK in platelets, and therefore its activation status and function are poorly established. It is activated by several agonists, including thrombin, vWF, collagen, and ADP (27). In addition, a previous study demonstrated that an increased bleeding time, decreased integrin αIIbβ3 activation and severe granule secretion impairment occur in JNK−/− platelets (29). Therefore, the inhibition of JNK phosphorylation may serve an important function in platelet activation. In accordance with these results, the results of the present study demonstrated that Ir-3 markedly inhibited collagen-induced JNK1 phosphorylation.
Reactive oxygen species produced through platelet activation, including hydrogen peroxide and OH·, may affect cells that they come in contact with, for example endothelial cells, thereby increasing platelet reactivity during thrombus formation. Free radical species act as secondary signals that increase [Ca2+] levels during the initial phase of platelet activation, and PKC is involved in the receptor-mediated production of free radicals in platelets (30). In addition, hydrogen peroxide produced by platelets is converted into OH·, because platelet aggregation is inhibited by OH· scavengers (30). The ESR spectrometry results from the present study provide direct evidence that Ir-3 does not significantly reduce OH· formation in collagen-stimulated platelet suspensions and Fenton reaction solution. In addition, Ir tissue distribution and excretion in rats following sub-chronic oral administration of Ir (III) chloride hydrate (1–1,000 ng/ml) for 90 days was previously determined (31). Concerning the distribution, the majority of Ir was located in the kidney and spleen, and smaller amounts were observed in the lungs, liver and brain. Notably, a slight increase of brain Ir levels was observed with increasing doses of Ir, indicating the ability of Ir to cross the blood-brain barrier. Furthermore, the dose-dependent increase of Ir levels in the serum demonstrated that the Ir was distributed in the blood compartment. For excretion, the body clearance of Ir primarily took place by elimination via feces, and this was also dose-associated (31).
In conclusion, the results of the present study demonstrate that the novel Ir-3 compound inhibited platelet activation by inhibiting signaling pathways, including the PLCγ2-PKC cascade, and subsequently suppressing Akt and JNK1 activation. These alterations reduced granule secretion (including ATP release, [Ca2+] levels and surface P-selectin expression) and ultimately inhibited platelet aggregation. However, additional studies are required to investigate the involvement of other unidentified mechanisms of the Ir-3-mediated inhibition of platelet activation. Nevertheless, Ir-3 represents a potential chemotherapeutic agent for the treatment of cancer. In addition, it may be used as an antiplatelet agent for treating thromboembolic disorders or the interplay between platelets and tumor cells which contributes to tumor cell proliferation and progression.
Acknowledgments
The present study was supported by grants from the Ministry of Science and Technology of Taiwan (MOST 104-2622-B-038-003, MOST 104-2320-B-038-045-MY2 and MOST 106-2320-B-038-012), Shin Kong Wu Ho-Su Memorial Hospital-Taipei Medical University (SKH-TMU-103-01), and the University Grants Commission, India (MRP-MAJOR-CHEM-2013-5144; 69/2014 F. No. 10-11/12UGC).
Footnotes
Competing interests
The authors declare that they have no competing interests.
References
- 1.Jayakumar T, Yang CH, Geraldine P, Yen TL, Sheu JR. The pharmacodynamics of antiplatelet compounds in thrombosis treatment. Expert Opin Drug Metab Toxicol. 2016;12:615–632. doi: 10.1080/17425255.2016.1176141. [DOI] [PubMed] [Google Scholar]
- 2.Belloc C, Lu H, Soria C, Fridman R, Legrand Y, Menashi S. The effect of platelets on invasiveness and protease production of human mammary tumor cells. Int J Cancer. 1995;60:413–417. doi: 10.1002/ijc.2910600324. [DOI] [PubMed] [Google Scholar]
- 3.Felding-Habermann B, Ooole TE, Smith JW, Fransvea E, Ruggeri ZM, Ginsberg MH, Hughes PE, Pampori N, Shattil SJ, Saven A, et al. Integrin activation controls metastasis in human breast cancer. Proc Natl Acad Sci USA. 2001;98:1853–1858. doi: 10.1073/pnas.98.4.1853. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Boucharaba A, Serre CM, Gres S, Saulnier-Blache JS, Bordet JC, Guglielmi J, Clezardin P, Peyruchaud O. Platelet-derived lysophosphatidic acid supports the progression of osteolytic bone metastases in breast cancer. J Clin Invest. 2004;114:1714–1725. doi: 10.1172/JCI200422123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Iavicoli I, Cufino V, Corbi M, Goracci M, Caredda E, Cittadini A, Bergamaschi A, Sgambato A. Rhodium and iridium salts inhibit proliferation and induce DNA damage in rat fibroblasts in vitro. Toxicol In Vitro. 2012;26:963–969. doi: 10.1016/j.tiv.2012.03.014. [DOI] [PubMed] [Google Scholar]
- 6.Iavicoli I, Fontana L, Marinaccio A, Alimonti A, Pino A, Bergamaschi A, Calabrese EJ. The effects of iridium on the renal function of female wistar rats. Ecotoxicol Environ Safe. 2011;74:1795–1799. doi: 10.1016/j.ecoenv.2011.06.014. [DOI] [PubMed] [Google Scholar]
- 7.Romero-Canelón I, Sadler PJ. Next-generation metal anticancer complexes: Multitargeting via redox modulation. Inorg Chem. 2013;52:12276–12291. doi: 10.1021/ic400835n. [DOI] [PubMed] [Google Scholar]
- 8.Yellol J, Pérez SA, Buceta A, Yellol G, Donaire A, Szumlas P, Bednarski PJ, Makhloufi G, Janiak C, Espinosa A, Ruiz J. Novel C, N-cyclometalated benzimidazole ruthenium(II) and iridium(III) complexes as antitumor and antiangiogenic agents: A structure-activity relationship study. J Med Chem. 2015;58:7310–7327. doi: 10.1021/acs.jmedchem.5b01194. [DOI] [PubMed] [Google Scholar]
- 9.Schmitt F, Donnelly K, Muenzner JK, Rehm T, Novohradsky V, Brabec V, Kasparkova J, Albrecht M, Schobert R, Mueller T. Effects of histidin-2-ylidene vs. imidazol-2-ylidene ligands on the anticancer and antivascular activity of complexes of ruthenium, iridium, platinum, and gold. J Inorg Biochem. 2016;163:221–228. doi: 10.1016/j.jinorgbio.2016.07.021. [DOI] [PubMed] [Google Scholar]
- 10.Sheu JR, Lee CR, Lin CH, Hsiao G, Ko WC, Chen YC, Yen MH. Mechanisms involved in the antiplatelet activity of staphylococcus aureus lipoteichoic acid in human platelets. Thromb Haemost. 2000;83:777–784. [PubMed] [Google Scholar]
- 11.Chou DS, Hsiao G, Shen MY, Tsai YJ, Chen TF, Sheu JR. ESR spin trapping of a carbon-centered free radical from agonist-stimulated human platelets. Free Radic Biol Med. 2005;39:237–248. doi: 10.1016/j.freeradbiomed.2005.03.011. [DOI] [PubMed] [Google Scholar]
- 12.Harrison P, Cramer EM. Platelet alpha-granules. Blood Rev. 1993;7:52–62. doi: 10.1016/0268-960X(93)90024-X. [DOI] [PubMed] [Google Scholar]
- 13.Singer WD, Brown HA, Sternweis PC. Regulation of eukaryotic phosphatidylinositol-specific phospholipase C and phospholipase D. Annu Rev Biochem. 1997;66:475–509. doi: 10.1146/annurev.biochem.66.1.475. [DOI] [PubMed] [Google Scholar]
- 14.Woulfe DS. Akt signaling in platelet and thrombosis. Expert Rev Hematol. 2010;3:81–91. doi: 10.1586/ehm.09.75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Roux PP, Blenis J. ERK and p38 MAPK-activated protein kinases: A family of protein kinases with diverse biological functions. Microbiol Mol Biol Rev. 2004;68:320–344. doi: 10.1128/MMBR.68.2.320-344.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bugaud F, Nadal-Wollbold F, Levy-Toledano S, Rosa JP, Bryckaert M. Regulation of c-jun-NH2 terminal kinase and extracellular-signal regulated kinase in human platelets. Blood. 1999;94:3800–3805. [PubMed] [Google Scholar]
- 17.Lip GY. Chin BS and Blann A: Cancer and the prothrombotic state. Lancet Oncol. 2002;3:27–34. doi: 10.1016/S1470-2045(01)00619-2. [DOI] [PubMed] [Google Scholar]
- 18.Ferroni P, Della-Morte D, Palmirotta R, McClendon M, Testa G, Abete P, Rengo F, Rundex T, Guadagni F, Roselli M. Platinum-based compounds and risk for cardiovascular toxicity in the elderly: Role of the antioxidants in chemoprevention. Rejuvenation Res. 2011;14:293–308. doi: 10.1089/rej.2010.1141. [DOI] [PubMed] [Google Scholar]
- 19.Jafri M, Protheroe A. Cisplatin-associated thrombosis. Anticancer Drugs. 2008;19:927–929. doi: 10.1097/CAD.0b013e3283100e9c. [DOI] [PubMed] [Google Scholar]
- 20.Barni S, Labianca R, Agnelli G, Bonizzoni E, Verso M, Mandalà M, Brighenti M, Petrelli F, Bianchini C, Perrone T, Gasparini G. Chemotherapy-associated thromboembolic risk in cancer outpatients and effect of nadroparin thromboprophylaxis: Results of a retrospective analysis of the PROTECHT study. J Transl Med. 2011;20(179) doi: 10.1186/1479-5876-9-179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dasanu CA. Gemcitabine: Vascular toxicity and prothrombotic potential. Expert Opin Drug Safe. 2008;7:703–716. doi: 10.1517/14740330802374262. [DOI] [PubMed] [Google Scholar]
- 22.Kaibuchi K, Sano K, Hoshijima M, Takai Y, Nishizuka Y. Phosphatidylinositol turnover in platelet activation; calcium mobilization and protein phosphorylation. Cell Calcium. 1982;3:323–335. doi: 10.1016/0143-4160(82)90020-3. [DOI] [PubMed] [Google Scholar]
- 23.Ragab A, Séverin S, Gratacap MP, Aguado E, Malissen M, Jandrot-Perrus M, Malissen B, Ragab-Thomas J, Payrastre B. Roles of the C-terminal tyrosine residues of LAT in GP VI-induced platelet activation: Insights into the mechanism of PLC gamma 2 activation. Blood. 2007;110:2466–2474. doi: 10.1182/blood-2007-02-075432. [DOI] [PubMed] [Google Scholar]
- 24.Walter U, Eigenthaler M, Geiger J, Reinhard M. Role of cyclic nucleotide-dependent protein kinases and their common substrate VASP in the regulation of human platelets. Adv Exp Med Biol. 1993;344:237–249. doi: 10.1007/978-1-4615-2994-1_19. [DOI] [PubMed] [Google Scholar]
- 25.Chen J, De S, Damron DS, Chen WS, Hay N, Byzova TV. Impaired platelet responses to thrombin and collagen in AKT-1-deficient mice. Blood. 2004;104:1703–1710. doi: 10.1182/blood-2003-10-3428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chang L, Karin M. Mammalian MAP kinase signaling cascades. Nature. 2001;410:37–40. doi: 10.1038/35065000. [DOI] [PubMed] [Google Scholar]
- 27.Adam F, Kauskot A, Rosa JP, Bryckaert M. Mitogen-activated protein kinases in hemostasis and thrombosis. J Thromb Haemost. 2008;6:2007–2016. doi: 10.1111/j.1538-7836.2008.03169.x. [DOI] [PubMed] [Google Scholar]
- 28.Canobbio I, Reineri S, Sinigaglia F, Balduini C, Torti M. A role for p38 MAP kinase in platelet activation by von Willebrand factor. Thromb Haemost. 2004;91:102–110. doi: 10.1160/TH03-02-0083. [DOI] [PubMed] [Google Scholar]
- 29.Adam F, Kauskot A, Nurden P, Sulpice E, Hoylaerts MF, Davis RJ, Rosa JP, Bryckaert M. Platelet JNK1 is involved in secretion and thrombus formation. Blood. 2010;115:4083–4092. doi: 10.1182/blood-2009-07-233932. [DOI] [PubMed] [Google Scholar]
- 30.Wachowicz B, Olas B, Zbikowska HM, Buczyński A. Generation of reactive oxygen species in blood platelets. Platelets. 2002;13:175–182. doi: 10.1080/09533710022149395. [DOI] [PubMed] [Google Scholar]
- 31.Iavicoli I, Fontana L, Bergamaschi A, Conti ME, Pino A, Mattei D, Bocca B, Alimonti A. Sub-chronic oral exposure to iridium (III) chloride hydrate in female wistar rats: Distribution and excretion of the metal. Dose-Response. 2012;10:405–414. doi: 10.2203/dose-response.11-052.Iavicoli. [DOI] [PMC free article] [PubMed] [Google Scholar]