Abstract
Chemotherapy is the best choice for the vast majority of hepatocellular carcinoma patients at late stage, but few effective chemotherapy drugs are available in clinic. Licochalcone A (LicA) is a new chemotherapy drug inducing apoptosis as Bcl-2 inhibitor, but few studies report on LicA-induced autophagy. This study investigated the phenomenon and mechanisms of LicA-induced autophagy looking for a targeted combination drug. Human hepatocellular carcinoma cells (HCCs) were treated with LicA, to detect markers of autophagy and to investigate the mechanisms. In order to investigate the role of reactive oxygen species (ROS) in LicA-induced autophagy, ROS, glutathione (GSH) and O2− were measured in LicA treated HCCs, and antioxidant N-Acetyl-L-cysteine (NAC) was cotreated with LicA in HCCs, then mechanisms of ROS-induced autophagy was investigated in LicA or LicA combined with NAC treated HCCs. Finally, the LicA-induced apoptosis was detected in LicA combined with NAC treated HCCs. We first report that LicA can induce autophagy through ULK1/Atg13 and ROS pathway in HCCs, suppression of LicA-induced ROS through antioxidant NAC can enhance LicA-induced apoptosis, promoting the function of LicA killing HCCs. LicA can activate the ULK1/Atg13 complex which is upstream of autophagy, additionally, LicA also can promote ROS generation, ROS trigger the expression level of TSC1/2 complex, PRAS40, CTMP, PP2A, PDK1 and Rubicon change, these molecules are upstream of autophagy. In conclusion, LicA can induce autophagy through ULK1/Atg13 and ROS pathway in HCCs, LicA combined with NAC can enhance LicA-induced apoptosis. Our results may provide a novel design for clinical hepatocellular carcinoma therapy trials.
Keywords: licochalcone A, reactive oxygen species, autophagy, hepatocellular carcinoma cells
Introduction
Primary liver cancer is the main diagnosis of hepatocellular carcinoma cell (HCC). The mortality of HCC is ranked second in cancer-related death worldwide, there are 695,900 death and 748,300 new cases every year (1,2). Although the 5-year survival rate is >70% when the patients are diagnosed as early stage and have good prognosis, The vast majority of patients are diagnosed as late stage, and the 5-year survival rate is <16% (1). The treatment of HCC includes surgery and adjuvant chemoradiation (1). Due to the limitation of surgical therapy with tumor size, intra-hepatic metastases and hepatic functional reserve sufficiency, few patients are suitable to receive this treatment (3). Chemotherapy is the final choice of the vast majority of patients, however, the HCC appear moderately tolerated, reducing the effect of chemotherapy (1,3). Therefore, exploring the mechanisms of chemotherapy agents that are tolerated and to select the appropriate adjuvant chemotherapy is very important.
Licochalcone A (LicA) is a chalcone constituent of licorice, it has anti-inflammatory, antitumor and anti-bacterial ability (4). LicA is a potent inhibitor of Bcl-2 protein expression which is the anti-apoptotic proteins in various tumors, LicA also can induce apoptosis in several cancer cell lines contributing to the antitumor effect (5,6). In human HCCs, the migration and invasion can be suppressed by LicA. LicA has anti-inflammatory ability through suppressing nuclear factor-κB (NF-κB) and activator protein-1 (AP-1), and tumor angiogenesis is closely linked to inflammation, LicA can also inhibit tumor angiogenesis (7,8). These results suggest that LicA may have potential as antitumor therapy.
Autophagy is a protective mechanism, many chemotherapy drugs can induce autophagy, and reduce the effect of chemotherapy (9). Few reports exist on LicA inducing autophagy in HCCs, in this study, LicA induced autophagy in HCCs through activating ULK1/Atg13 complex and reactive oxygen species (ROS), ULK1/Atg13 complex is upstream of autophagy, and ROS was upstream of autophagy-related molecule. Moreover, the antioxidant N-Acetyl-L-cysteine (NAC) can inhibit LicA-induced autophagy through suppressing ROS in HCCs, promoting apoptosis and enhancing cell death rate, suggesting LicA may be a good chemotherapy drugs for HCC, especially as co-treatment with an antioxidant.
Materials and methods
Cell culture
Human HCCs HuH7 and HepG2 were purchased from American Type Culture Collection (ATCC, Manassas, VA, USA). The cells were cultured in DMEM conditioned medium, including 2 mM L-glutamine, 100 U/ml penicillin, 100 mg/ml streptomycin and 10% fetal bovine serum (FBS), in the normal culture environment with 37°C and 5% CO2, and all the cell culture regents were purchased from Gibco Laboratories (Grand Island, NE, USA).
Western blot analysis
For each sample, all the cells were lysed for 30 min in lyses buffer (Beyotime, Bejing, China) on ice, and the cell debris was centrifuged at 12,000 × g for 12 min at 4°C, protein concentration were detected by BCA (Beyotime), using 8–15% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) to separate the proteins with 12 V, the separated proteins were blotted onto PVDF membrane, blocking 1 h at room temperature with 5% nonfat drymilk in 0.05% Tween-20 in PBS (PBST), incubating the antibody for 24 h at 4°C with 1:1,000 concentration, washing PVDF membrane 3 times for 10 min in PBST, incubating secondary antibody at 37°C for 1 h with the 1:10,000 concentration, washing PVDF membrane 3 times for 10 min in PBST. All the antibodies were purchased from Santa Cruz Biotechnology, Inc. (Santa Cruz, CA, USA).
Acridine orange (AO) staining
Cells were treated with drugs, slightly washed 3 times with PBS, using 4% faraformaldehyde to fix for 10 min, stained with 1 mg/l AO dye liquor for 20 min at 37°C in the dark, and then observed under fluorescence microscope (Nikon TE2000; Nikon, Tokyo, Japan).
GFP-LC3 transfection
HuH7 and HepG2 cells were transfected with GFP-LC3 vector (GeneChem, Shanghai, China) through Lipofectamine 2000 (Invitrogen Life Technology, Carlsbad, CA, USA). After 18 h, cells were treated with drugs for 24 h, using 4% faraformaldehyde to fix for 10 min, washed 3 times with PBS, and then observed under fluorescence microscope (Nikon TE2000; Nikon).
Co-immunoprecipitation experiments
HepG2 cells were treated with drugs for 12 h, lysing cells in hypotonic lyses buffer (Beyotime) on ice for 30 min, collecting the supernatant to pellet nuclei in whole-cell lysate at a low speed (1,000 × g), preclearing whole-cell lysate with protein A-agarose, adding ULK1 or Atg13 antibodies for 1 h at 4°C. Then the immunoprecipitates were captured on protein A-agarose and detected by immunoblotting with ULK1 or Atg13 antibodies, respectively.
Cell viability assay
Cell viability was detected by cell counting kit-8 (CCK-8) agent. Cells on a 96-well plate were left to attach overnight, then treated with drugs for 24 h. Medium was removed the and washed 3 times with PBS, then 90 µl DMEM and 10 µl CCK-8 was added into each well, and incubated for 1.5 h at 37°C in the dark. OD values were measured by microplate reader at 450 nm.
ROS detection
ROS level was measured by 2,7-dichlorofluo-rescein diacetate (DCFH-DA) (Beyotime). Cells on a 96-well plate were let to attached overnight, then treated with drugs for 24 h. The medium was removed and washed 3 times with PBS, then incubated with DCFH-DA at 37°C for 30 min, measuring the ROS levels through fluorescence microplate reader with 488 nm excitation wavelength and 525 nm emission wavelength.
Cell death assay
Cell death ratio was detected by trypan blue. The cells were treated with drugs, and then digested adding 0.4% (w/v) trypan blue solution into cell suspension at volume ratio 1:9. Counting the dead cells and total cells with a microscope, the dead cells cannot exclude the dye, total death rate = (the number of dead cell/total cell) ×100%.
Transmission electron microscopy (TEM)
Samples were processed with the standard protocol for TEM. TEM was performed on a JEOL 1230 TEM at an accelerating voltage of 80 kV. Images were acquired with an AMT Advantage Plus 2K×2K digital camera connected to the TEM.
Transfection experiments
siRNA transient transfections were performed with Lipofectamine 2000 (Invitrogen Life Technology) according to the manufacturer's protocol. After 36 h transfection, drugs were added, and after an additional 24 h the cells were collected for western blot analysis.
Apoptosis detection
Apoptosis was detected by FACScan flow cytometer. Treating the cell with drugs, cells were incubated with Annexin V-FITC and propidium-iodide (PI) (both from Beyotime) at room temperature for 10 min. FACScan flow cytometer was used to analyses the apoptosis ratio.
Statistic analysis
The data are represented as the mean ± SD from triplicate experiments. Two-way ANOVA was used to analyze the variance of different groups. A threshold of P<0.05 was defined as statistically significant.
Results and Discussion
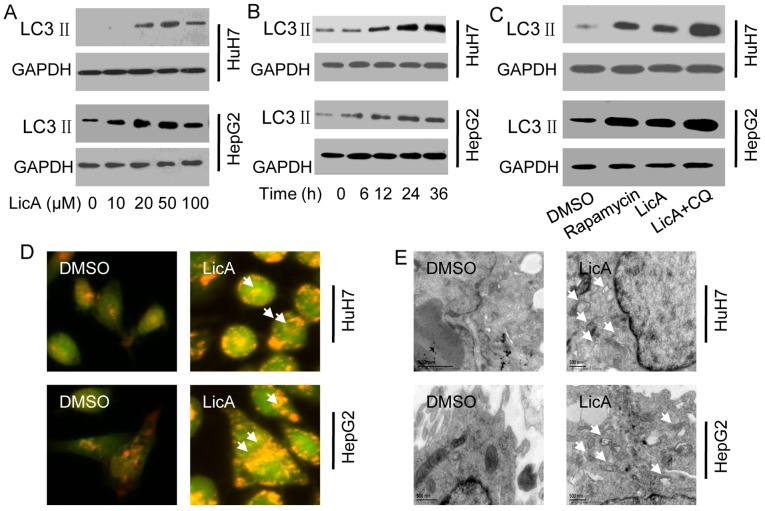
When autophagy occurs, the cytosolic LC3 will be cleaved to short peptide hydrolysis LC3 II which is located on the membrane of autophagosome, therefore, LC3 II is the marker of autophagy that can reflect the level of autophagy. Because the lower PH value around autophagosome, AO can penetrate into acidic organelles and appear as red fluorescence, staining with AO, and the red fluorescence intensity also can reflect the level of autophagy. Moreover, TEM is the gold standard of autophagosome. LicA can significant enhance the expression of LC3 II, the induced ability was time-dependent and dose-dependent, and the best dose was 50 µM, the best time was 24 h (Fig. 1A and B). Compared to the positive control rapamycin treatment, the level of induced-LC3 II has no significant difference, when LicA is co-treated with chloroquine (CQ) which can block the lysosome and engulf the autophagosome and contribute to LC3-II accumulation, the level of induced-LC3 II was significantly increased (Fig. 1C). AO staining show that the red fluorescence intensity of cells which were treated with 50 µM LicA was significant higher than the cells which were treated with 0.1% dimethyl sulfoxide (DMSO) (Fig. 1D). The TEM results also prove that the number of autophagosome in the cells which were treated with 50 µM LicA was significant higher than the cells which were treated with 0.1% DMSO (Fig. 1E). The above results means that LicA can significantly induce autophagy in HCCs.
Figure 1.
Licochalcone A (LicA) induces autophagy in hepatocellular carcinoma cells (HCCs). (A) HCCs were treated with different doses of LicA for 24 h. (B) HCCs were treated with 50 µM LicA for different times. (C) HCCs were treated with 50 µM LicA, 50 µM LicA combined with 40 µM chloroquine (CQ) or 50 µM rapamycin for 24 h. (D) HCCs were treated with 50 µM LicA for 24 h and processed acridine orange (AO) staining (E) and transmission electron microscopy (TEM) (arrows) were examined to evaluate the level of LicA-induced-autophagy.
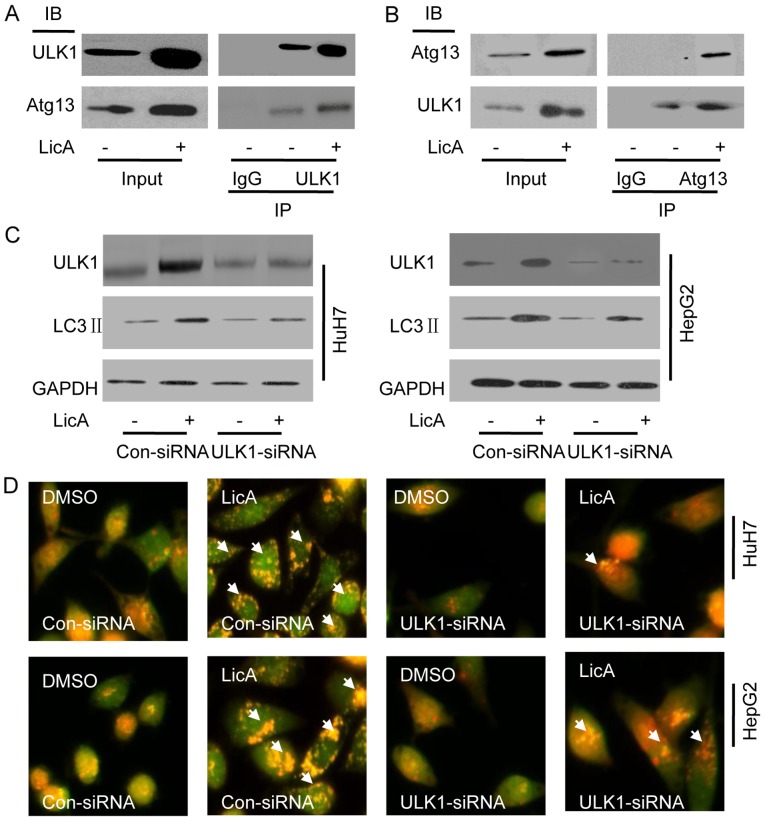
ICo-immunoprecipitation (Co-IP) pull-down assay was used to investigate the mechanism of LicA-induced-autophagy. HepG2 cells were treated with 50 µM LicA or not, immunoprecipitation of ULK1 can pull down Atg13, and immunoprecipitation of Atg13 also can pull down ULK1, that means ULK1 and Atg13 were bound to each other forming a complex in HCCs, and LicA can activate the complex which is upstream of autophagy (Fig. 2A and B). Knocking down ULK1 by ULK1-siRNA in HCCs, the level of LicA-induced LC3 II was significant lower than co-siRNA, but still higher than DMSO treatment (Fig. 2C), similarly, staining with AO, the red fluorescence intensity was significant lower than co-siRNA, but still higher than DMSO treatment (Fig. 2D), thus ULK1/Atg13 complex is one of the regulating molecules of LicA-induced autophagy, not the unique regulating molecule.
Figure 2.
Licochalcone A (LicA) activates ULK1/Atg13 signal pathway. (A and B) HepG2 cells were treated with 50 µM LicA for 12 h. (C) Hepatocellular carcinoma cells (HCCs) were transfected with Con-siRNA or ULK1-siRNA, 36 h later, treated with 0.1% DMSO or 50 µM LicA for 24 h, and then possessing the immunoblot or (D) acridine orange (AO) staining.
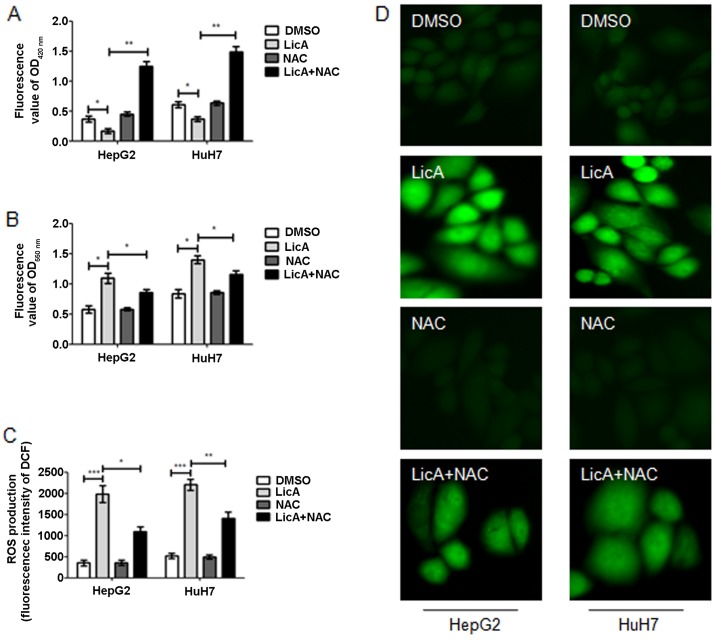
ROS is one of the survival and proliferation regulator factors of cancer cells, and also regulate autophagy. LicA can suppress the glutathione (GSH) generation (Fig. 3A) and promote the O2− generation (Fig. 3B), finally contributing to ROS generation (Fig. 3C and D) in HCCs. Moreover, when HCCs were cotreated with LicA and antioxidant NAC, the level of GSH, O2− and ROS reversed partly (Fig. 3A–D). These results show that LicA can promote ROS generation in HCCs, and the NAC can inhibit the phenomena.
Figure 3.
Licochalcone A (LicA) promotes reactive oxygen species (ROS) generation in hepatocellular carcinoma cells (HCCs). HCCs were pretreated with antioxidant N-Acetyl-L-cysteine (NAC) for 2 h, and treated with 0.1% DMSO or 50 µM LicA for 24 h. (A) Measuring the level of glutathione (GSH) by ELISA kit at 420 nm. (B) The level of O2− was measured by ELISA kit at 550 nm. The level of ROS was measured by 2,7-dichlorofluorescein diacetate (DCFH-DA) using (C) fluorescence microplate reader or (D) fluorescence microscope. *P<0.05, **P<0.01 and ***P<0.005.
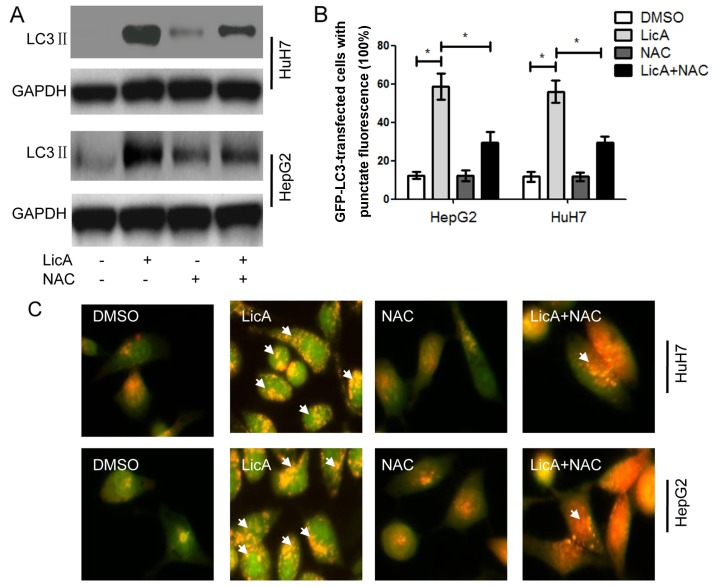
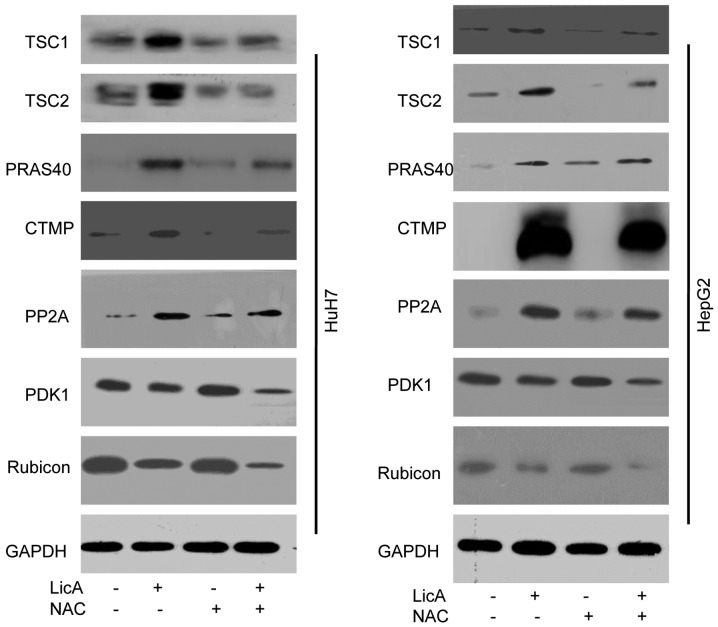
The above results have proved that LicA can promote ROS generation and NAC can reverse the effect of LicA in HCCs. When HCCs were cotreated with LicA and NAC, the level of LicA-induced LC3 II was lower than HCCs treated with only LicA, but also higher than DMSO treatment (Fig. 4A). At the same time the quantity of LC3-GFP puncta and the red fluorescence intensity with AO staining in HCCs was cotreated with LicA and NAC were lower than those treated with LicA alone, but also higher than DMSO treatment (Fig. 4B and C). These results indicated that LicA can induce ROS-related autophagy, and NAC can inhibit the same. Previous studies have reported that ROS was upstream of many molecules, and ROS was not the direct inducing factor of autophagy. In order to investigate the downstream ROS pro-autophagy pathways, other regulated factors were detected in HCCs. Sarbassov et al (10) have reported that TSC1/2 complex was upstream of mTORC1, and mTORC1 was a universal regulator of autophagy (11). So TSC1/2 complex was detected in this work, the results show that TSC1/2 complex can be activated by LicA through ROS pathway, and can be inhibited by NAC that can suppress ROS generation (Fig. 5). PRAS40 is a metabolism protein bound to mTOR complex, and acts as the receptor of mTOR, PRAS40 also can regulate the proliferation in various cell lines (12,13). The results of this work indicated that PRAS40 can be regulated by LicA-induced-ROS in HCCs, and when HCCs were cotreated with LicA and NAC, the change of PRAS40 expression level was inhibited (Fig. 5). Carboxy-terminal modulator protein (CTMP), as an endogenous inhibitor of Akt, also can influence the phosphoinositide 3-kinase (PI3K) pathway through activating protein kinase B (PKB) signaling, leading to insulin signaling regulation (14). These studies support our results that CTMP can regulate autophagy, moreover, CTMP was downstream in LicA-induced autophagy via promoting ROS generation in HCCs, similarly, the phenomenon can be changed by NAC (Fig. 5). Protein phosphatase 2A (PP2A) is a key regulator of cell cycle, also the target for anticancer drugs, PP2A can regulate the autophagy through AMPK phosphorylation. These studies showed PP2A may be an important regulator in LicA-induced autophagy (15,16) the results in our study proved the hypothesis as PP2A acted as the regulator of LicA-induced autophagy through promoting ROS generation, and ROS inhibitor NAC can block this progress (Fig. 5). PDK1 is an AGC kinase of the PKB family which contain a PH domain regulating cell proliferation, apoptosis and differentiation, PDK1 is upstream of Akt, and impair the autophagic effect through targeting activated c-Src (p-Src) (17,18). LicA can inhibit the PDK1 expression through ROS in LicA-induced autophagy, and NAC reversed this (Fig. 5). Rubicon is a subunit of PI3KC3, containing cysteine-rich protein, acting as a regulator of Beclin1-UVRAG-Vps34 complex to regulate autophagy (19). LicA-induced ROS can inhibit Rubicon expression in LicA-induced autophagy, and NAC is the inhibitor of this program (Fig. 5). These results indicated that ROS is the pathway of LicA-induced autophagy in HCCs.
Figure 4.
Licochalcone A (LicA) induces autophagy through reactive oxygen species (ROS) pathway in hepatocellular carcinoma cells (HCCs). HCCs were pretreated with antioxidant N-Acetyl-L-cysteine (NAC) for 2 h, and treated with 0.1% DMSO or 50 µM LicA for 24 h. (A) The expression level of LC3 was detected by immunoblot. (B) HCCs were transfected with GFP-LC3 plasmid, and counting the LC3-GFP puncta positive cells (at least 5 puncta) with fluorescence microscope. (C) acridine orange (AO) staining for detecting autolysosomes. *P<0.05.
Figure 5.
Reactive oxygen species (ROS) has an important role in licochalcone A (LicA)-induced autophagy. Hepatocellular carcinoma cells (HCCs) were pretreated with antioxidant N-Acetyl-L-cysteine (NAC) for 2 h, and treated with 0.1% DMSO or 50 µM LicA for 24 h, the expression of TSC1/2 complex, PRAS40, carboxy-terminal modulator protein (CTMP), protein phosphatase 2A (PP2A), PDK1 and Rubicon were measured by immunoblotting.
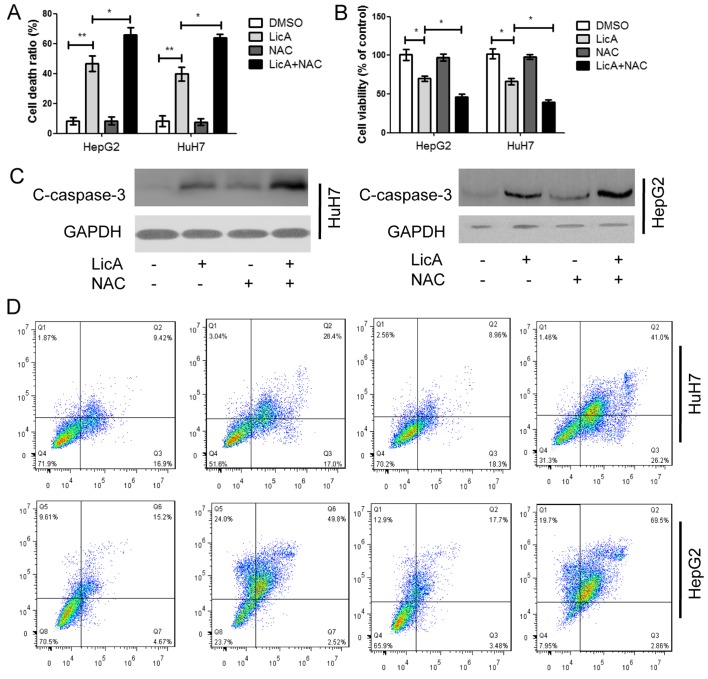
LicA can induce autophagy in HCCs through promoting ROS generation, and autophagy is a protective mechanism for cells, the antioxidant NAC can inhibit ROS generation, moreover, LicA is the activator of apoptosis in various cancer cells. Therefore, in order to investigate the function of ROS-mediated autophagy in LicA-induced apoptosis for HCCs, HCCs were cotreated with LicA and NAC or treated with LicA alone, the cell death ratio and cell viability ratio were detected, LicA can induced cell death and decrease cell viability in HCCs, and NAC can enhance the effect of LicA in killing HCCs (Fig. 6A and B). The apoptosis marker caspase-3 was detected, LicA can promote caspase-3 expression and NAC enhance the effect, which means that NAC could enhance LicA-induced apoptotic in HCCs (Fig. 6C). Annexin V-FITC and PI results indicated that LicA NAC enhanced the LicA-induced cell apoptosis ratio (Fig. 6D). Thus, the above results indicated that suppression of ROS-mediated autophagy induced by LicA enhanced LicA-induced apoptosis.
Figure 6.
Antioxidant N-Acetyl-L-cysteine (NAC) enhances LicA-induced apoptosis. Hepatocellular carcinoma cells were pretreated with antioxidant NAC for 2 h, and treated with 0.1% DMSO or 50 µM LicA for 24 h. (A) Cell death ratio was measured by Trypan blue staining. (B) Cell viability was measured by cell counting kit-8 (CCK-8) kit. (C) Caspase-3 was measured by immunoblotting. (D) Apoptosis was detected by FACScan flow cytometer. *P<0.05 and **P<0.01.
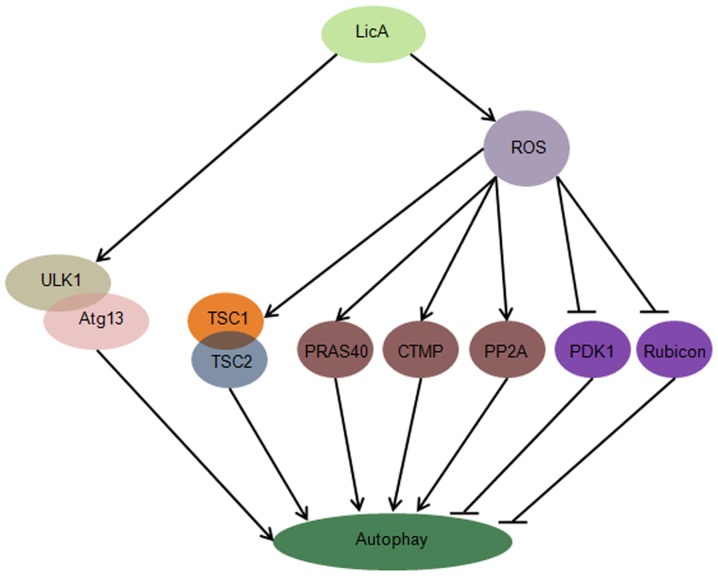
In this study, we first report that LicA can induce autophagy through ULK1/Atg13 and ROS pathway in HCCs, suppression of LicA-induced ROS through antioxidant NAC can enhance LicA-induced apoptosis, promoting the function of LicA killing HCCs. LicA can activate the ULK1/Atg13 complex which is upstream of autophagy, LicA can promote ROS generation, ROS trigger the expression level of TSC1/2 complex, PRAS40, CTMP, PP2A, PDK1 and Rubicon change, these molecules are upstream of autophagy (Fig. 7).
Figure 7.
The pathway of licochalcone A (LicA)-induced autophagy. LicA activates the ULK1/Atg13 complex, and LicA promotes reactive oxygen species (ROS) generation, following TSC1/2 complex, PRAS40, carboxy-terminal modulator protein (CTMP), protein phosphatase 2A (PP2A), PDK1 and Rubicon changed triggering of ROS-mediated autophagy.
LicA has been reported to have antitumor, anti-angiogenesis and anti-inflammatory ability, and considered as Bcl-2 inhibitor (4,5,6,20). LicA can block cell cycle progression through regulating extracellular signal-regulated kinase 1/2 (ERK1/2) and Rb phosphorylation (21). Animal experiments show that, LicA significantly inhibits tumor growth, and reduces the cell nuclear antigen, cyclooxygenase-2 (COX-2) and inducible nitric oxide synthase (iNOS), contributing to survival. Moreover, previously it has been reported that LicA can inhibit matrix metalloproteinase-9 (MMP-9) and vascular endothelial growth factor receptor-2 (VEGFR-2) expression in various cancer cells, indicating that LicA can mediate metabolism and angiogenesis (8,20).
Autophagy is a double membrane structure autophagosome formed in the no ribosome attachment region of reticulum membrane, engulfing the waste proteins or aged organelles, fused with lysosomes and forming autolysosome, degradation its contents. Therefore, autophagy is necessary for cell metabolism, and contributes to cell survival. LicA is the Bcl-2 inhibitor, and Bcl-2 is the anti-apoptosis protein, previously reported have demonstrated that apoptosis and autophagy occur simultaneously. The antitumor function of LicA was mainly through inducing apoptosis. The results have proven that LicA-induced autophagy in HCCs depend on the dose and time. ROS has negative influence on normal cells, but have dual role for cancer cells. ROS can promote tumor growth through inducing DNA damage and activating oncogenes. DeNicola et al reported that Nrf2 was in high expression state in various cancer cells, and Nrf2 could decrease the intracellular ROS generation, remove the free radicals in cells, moreover, cancer cells could not proliferate without Nrf2 expression, therefore, ROS also can inhibit cancer cell proliferation (22). ULK1/Atg13 complex assembled by RB1CC1, FIP200 and Atg101, forms a macroautophagy, and participate in the autophagy mediation through mTOR and AMPK. Our results indicated that LicA could activate the ULK1/Atg13 complex, inducing autophagy in HCCs. Furthermore, LicA also can promote the O2− generation and suppress GSH generation, contribute to ROS generation in HCCs, and the antioxidant NAC can inhibit the ROS generation induced by LicA in HCCs. Previous studies have reported that tuberous sclerosis complex (TSC) is the mutational production of TSC1 or TSC2 gene, occuring in hamartomas and neurological manifestations. TSC1/2 complex can negatively regulate the mTORC1 complex, contribute to protein synthesis, cell growth and autophagy mediation (23). PRAS40 is the substrate of PKB/Akt, it can also bind to mTORC1 complex, possessing various phosphorylation sites, including Thr246 mediated by Akt, Ser183, Ser212 and Ser221 mediated by mTORC1 complex (12). CTMP is an endogenous inhibitor of Akt, a key mediator of insulin signals, and insulin signals mediate autophagy pathway (14,24). PP2A has an important role in the mediation of cell cycle, and PTEN-induced kinase 1 can increase the phosphorylation of PP2A at Y307, moreover, Bcl-2 is downstream of PP2A in PTEN-induced kinase 1 pathway, which participates in the autophagy mediation (15,25). PDK1 is upstream of Akt, and inhibits autophagy through targeting c-Src, Rubicon as a PI3KC3 subunit can communicate with Beclin 1, contribute to autophagosome suppression (17,19). In this study, ROS induced by LicA can activate TSC1/2 complex, PRAS40, CTMP and PP2A signals, and inhibit PDK1 and Rubicon signals, contributing to autophagy progress triggering. Moreover, the antioxidant can inhibit ROS generation in LicA treated HCCs. Cotreatment with LicA and NAC, inhibited and promoting cell death, and enhanced LicA-induced apoptosis in HCCs. Thus, our results may provide a new direction on rational design of clinical trials, LicA and antioxidant NAC may be a good combined regimen in HCC chemotherapy.
Acknowledgments
All the authors are very thankful to Professor Hong Zhao and Wei Yang for their support.
Abbreviations
- LicA
licochalcone A
- ROS
reactive oxygen species
- NAC
N-Acetyl-L-cysteine
- HCCs
human hepatocellular carcinoma cells
- GSH
glutathione
- CTMP
carboxy-terminal modulator protein
- PKB
protein kinase B
- PP2A
protein phosphatase 2A
- TSC
tuberous sclerosis complex
Footnotes
Funding
No funding was received.
Availability of data and material
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
QN, WZ, WD and DZ contributed to the conception and design of the study. QN, WZ, GW, KW and DZ developed the methodology. QN, WZ, JW and CL acquired the data. TY, WL, GW, TZ and KW analyzed and interpreted the data. QN, WZ, WD and DZ wrote, reviewed and revised the manuscript. DZ supervized the study.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin. 2015;65:5–29. doi: 10.3322/caac.21254. [DOI] [PubMed] [Google Scholar]
- 2.Ferlay J, Shin HR, Bray F, Forman D, Mathers C, Parkin DM. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int J Cancer. 2010;127:2893–2917. doi: 10.1002/ijc.25516. [DOI] [PubMed] [Google Scholar]
- 3.Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108. doi: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 4.Yo YT, Shieh GS, Hsu KF, Wu CL, Shiau AL. Licorice and licochalcone-A induce autophagy in LNCaP prostate cancer cells by suppression of Bcl-2 expression and the mTOR pathway. J Agric Food Chem. 2009;57:8266–8273. doi: 10.1021/jf901054c. [DOI] [PubMed] [Google Scholar]
- 5.Lee CK, Son SH, Park KK, Park JH, Lim SS, Kim SH, Chung WY. Licochalcone A inhibits the growth of colon carcinoma and attenuates cisplatin-induced toxicity without a loss of chemotherapeutic efficacy in mice. Basic Clin Pharmacol Toxicol. 2008;103:48–54. doi: 10.1111/j.1742-7843.2008.00238.x. [DOI] [PubMed] [Google Scholar]
- 6.Rafi MM, Rosen RT, Vassil A, Ho CT, Zhang H, Ghai G, Lambert G, DiPaola RS. Modulation of bcl-2 and cytotoxicity by licochalcone-A, a novel estrogenic flavonoid. Anticancer Res. 2000;20:2653–2658. [PubMed] [Google Scholar]
- 7.Kwon HS, Park JH, Kim DH, Kim YH, Park JH, Shin HK, Kim JK. Licochalcone A isolated from licorice suppresses lipopolysaccharide-stimulated inflammatory reactions in RAW264.7 cells and endotoxin shock in mice. J Mol Med (Berl) 2008;86:1287–1295. doi: 10.1007/s00109-008-0395-2. [DOI] [PubMed] [Google Scholar]
- 8.Kim YH, Shin EK, Kim DH, Lee HH, Park JH, Kim JK. Antiangiogenic effect of licochalcone A. Biochem Pharmacol. 2010;80:1152–1159. doi: 10.1016/j.bcp.2010.07.006. [DOI] [PubMed] [Google Scholar]
- 9.Anding AL, Baehrecke EH. Autophagy in cell life and cell death. Curr Top Dev Biol. 2015;114:67–91. doi: 10.1016/bs.ctdb.2015.07.012. [DOI] [PubMed] [Google Scholar]
- 10.Sarbassov DD, Ali SM, Sabatini DM. Growing roles for the mTOR pathway. Curr Opin Cell Biol. 2005;17:596–603. doi: 10.1016/j.ceb.2005.09.009. [DOI] [PubMed] [Google Scholar]
- 11.Høyer-Hansen M, Jäättelä M. AMP-activated protein kinase: A universal regulator of autophagy. Autophagy. 2007;3:381–383. doi: 10.4161/auto.4240. [DOI] [PubMed] [Google Scholar]
- 12.Kazi AA, Lang CH. PRAS40 regulates protein synthesis and cell cycle in C2C12 myoblasts. Mol Med. 2010;16:359–371. doi: 10.2119/molmed.2009-00168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wong CP, Seki A, Horiguchi K, Shoji T, Arai T, Nugroho AE, Hirasawa Y, Sato F, Kaneda T, Morita H. Bisleuconothine a induces autophagosome formation by interfering with AKT-mTOR signaling pathway. J Nat Prod. 2015;78:1656–1662. doi: 10.1021/acs.jnatprod.5b00258. [DOI] [PubMed] [Google Scholar]
- 14.Park J, Li Y, Kim SH, Yang KJ, Kong G, Shrestha R, Tran Q, Park KA, Jeon J, Hur GM, et al. New players in high fat diet-induced obesity: LETM1 and CTMP. Metabolism. 2014;63:318–327. doi: 10.1016/j.metabol.2013.10.012. [DOI] [PubMed] [Google Scholar]
- 15.Yin X, Zhang N, Di W. Regulation of LC3-dependent protective autophagy in ovarian cancer cells by protein phosphatase 2A. Int J Gynecol Cancer. 2013;23:630–641. doi: 10.1097/IGC.0b013e3182892cee. [DOI] [PubMed] [Google Scholar]
- 16.Gong FR, Wu MY, Shen M, Zhi Q, Xu ZK, Wang R, Wang WJ, Zong Y, Li ZL, Wu Y, et al. PP2A inhibitors arrest G2/M transition through JNK/Sp1- dependent down-regulation of CDK1 and autophagy-dependent up-regulation of p21. Oncotarget. 2015;6:18469–18483. doi: 10.18632/oncotarget.4063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Deng Q, Yu X, Xiao L, Hu Z, Luo X, Tao Y, Yang L, Liu X, Chen H, Ding Z, et al. Neoalbaconol induces energy depletion and multiple cell death in cancer cells by targeting PDK1-PI3-K/Akt signaling pathway. Cell Death Dis. 2013;4:e804. doi: 10.1038/cddis.2013.324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Karlsson I, Zhou X, Thomas R, Smith AT, Bonner MY, Bakshi P, Banga AK, Bowen JP, Qabaja G, Ford SL, et al. Solenopsin A and analogs exhibit ceramide-like biological activity. Vasc Cell. 2015;7:5. doi: 10.1186/s13221-015-0030-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhong Y, Wang QJ, Li X, Yan Y, Backer JM, Chait BT, Heintz N, Yue Z. Distinct regulation of autophagic activity by Atg14L and Rubicon associated with Beclin 1-phosphatidylinositol-3-kinase complex. Nat Cell Biol. 2009;11:468–476. doi: 10.1038/ncb1854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kim JK, Shin EK, Park JH, Kim YH, Park JH. Antitumor and antimetastatic effects of licochalcone A in mouse models. J Mol Med (Berl) 2010;88:829–838. doi: 10.1007/s00109-010-0625-2. [DOI] [PubMed] [Google Scholar]
- 21.Park JH, Lim HJ, Lee KS, Lee S, Kwak HJ, Cha JH, Park HY. Anti-proliferative effect of licochalcone A on vascular smooth muscle cells. Biol Pharm Bull. 2008;31:1996–2000. doi: 10.1248/bpb.31.1996. [DOI] [PubMed] [Google Scholar]
- 22.DeNicola GM, Karreth FA, Humpton TJ, Gopinathan A, Wei C, Frese K, Mangal D, Yu KH, Yeo CJ, Calhoun ES, et al. Oncogene-induced Nrf2 transcription promotes ROS detoxification and tumorigenesis. Nature. 2011;475:106–109. doi: 10.1038/nature10189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Di Nardo A, Wertz MH, Kwiatkowski E, Tsai PT, Leech JD, Greene-Colozzi E, Goto J, Dilsiz P, Talos DM, Clish CB, et al. Neuronal Tsc1/2 complex controls autophagy through AMPK-dependent regulation of ULK1. Hum Mol Genet. 2014;23:3865–3874. doi: 10.1093/hmg/ddu101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chen Y, Nie H, Tian L, Tong L, Deng J, Zhang Y, Dong H, Xiong L. Sevoflurane preconditioning-induced neuroprotection is associated with Akt activation via carboxy-terminal modulator protein inhibition. Br J Anaesth. 2015;114:327–335. doi: 10.1093/bja/aeu271. [DOI] [PubMed] [Google Scholar]
- 25.Qi Z, Yang W, Liu Y, Cui T, Gao H, Duan C, Lu L, Zhao C, Zhao H, Yang H. Loss of PINK1 function decreases PP2A activity and promotes autophagy in dopaminergic cells and a murine model. Neurochem Int. 2011;59:572–581. doi: 10.1016/j.neuint.2011.03.023. [DOI] [PubMed] [Google Scholar]