Abstract
Correlation of the surface physicochemical properties of nanoparticles with their interactions with biosystems provides key foundational data for nanomedicine. We report here the systematic synthesis of 2, 4, and 6 nm core gold nanoparticles (AuNP) featuring neutral (zwitterionic), anionic, and cationic headgroups. The cellular internalization of these AuNPs was quantified, providing a parametric evaluation of charge and size effects. Contrasting behavior was observed with these systems: with zwitterionic and anionic particles, uptake decreased with increasing AuNP size, whereas with cationic particles uptake increased with increasing particle size. Through mechanistic studies of the uptake process we can attribute these opposing trends to a surface-dictated shift in uptake pathways. Zwitterionic NPs are primarily internalized through passive diffusion, while the internalization of cationic and anionic NPs is dominated by multiple endocytic pathways. Our study demonstrates that size and surface charge interact in an interrelated fashion to modulate nanoparticle uptake into cells, providing an engineering tool for designing nanomaterials for specific biological applications.
Keywords: endocytosis pathway, passive fusion, size-dependent, surface charge, sub-10 nm gold nanoparticles
Graphical Abstract

The physicochemical properties of nanoparticles, in particular size,1–5 shape,6–8 surface charge9–12 and functionality,13–15 comprehensively determine the interactions of NPs with physiological systems.16,17 Recent studies have generally attempted to correlate the NPs-cell interactions with the physical and structural properties of NPs on a case-by-case basis, looking at one parameter while keeping others constant. For example, the cellular uptake of nanoparticles was found to be strongly size-dependent, with 50 nm nanoparticles exhibiting the highest cellular uptake efficiency among a set of AuNP ranged in 10–100 nm.7 In similar fashion, we and other groups have found that the internalization of NPs is dictated by chemical functionalities on nanoparticle surface.9,15
Exploring NP behavior in biological contexts along a single structural parametric axis is clearly informative. Interplay is expected, however, between structural factors, e.g. size and surface properties in terms of the biological properties of nanomaterials. Examining the collective impact of these parameters in cellular internalization is crucial for generating effective tools for biomedical applications in delivery. For example, Chan et al. studied the roles of nanoparticle size (15, 30, 60, and 90 nm) and surface ligand grafting density on serum protein adsorption, showing how these attributes differentiated the pathway and efficiency of nanoparticle uptake by macrophage cells.18 In another study, Toth et al. explored the effects of NP charge and size on the cellular uptake by changing the polymer coatings of AuNPs of 5, 10, and 20 nm sizes.14
Smaller (sub-10 nm) AuNP provide a particularly important set of particles for biomedical applications.3, 19–22 Pragmatically, these particles provide high payload-to-carrier ratios, with smaller particles in this regime capable of renal clearance.23 On a fundamental level, these particles are on a size scale commensurate with proteins, and can be engineered to provide highly biomimetic surfaces.24 Practically, however, the small size of these NPs makes parametric evaluation of surface chemistry challenging, as polymeric coatings will induce more dynamic changes in size and dynamic properties to smaller particles.
Here, we report the parametric fabrication of 2, 4, and 6 nm core AuNP featuring zwitterionic, anionic and cationic surface ligands. We correlated the cellular internalization efficiency of these particles and observed a striking difference in uptake trends. With cationic particles, increasing particle size resulted in increasing uptake. The opposite trend was observed for neutral zwitterionic and anionic particles, where uptake efficiency decreased with particle diameter. Mechanistic studies provide insight into these opposing trends. With cationic particles, active endocytotic pathways were responsible for the bulk of NP uptake. With the zwitterionic particles, however, passive diffusion appeared to be the predominant mechanism. More interestingly, small sized anionic NP (2, and 4 nm) entered cells through endocytic pathways to that of same sized cationic particles, while large sized anionic particles (6 nm) mainly displayed caveolae/lipid raft-mediated pathway. The results reported here demonstrate the importance of the interplay between size and surface functionality on dictating nanoparticle-cell interactions, and offer insight into engineering NP systems with predictable cell interactions.
RESULTS AND DISCUSSION
Fabrication and characterization of sub-10 nm AuNP
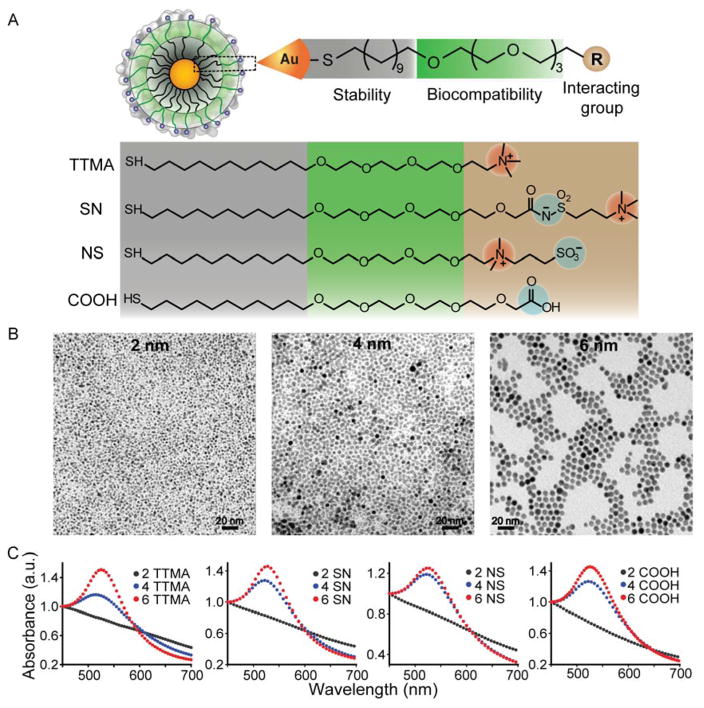
We designed structurally related NPs that featuring different surface charges to probe the effect of NP surface functionality on the cellular uptake of sub-10 nm AuNP. We first synthesized a set of cationic and anionic sub-10 nm AuNP coated with thioalkyl tetra(ethylene glycol)ated trimethyl ammonium (TTMA), and carboxylate ligands (COOH), respectively. The tetra(ethylene glycol) spacer was added between the terminal positively or negatively charged group and the hydrophobic alkyl chain to enhance NP stability, and to improve the biocompatibility of AuNP. The third category of sub-10 nm AuNP in this study was coated with zwitterionic ligands. We generated two subfamilies featuring different positioning of the charged moieties (Figure 1A), the first has positive charges in outermost layer (SN), while the second ligand has innermost layer of positive charge (NS).
Figure 1.
Characterization of as-synthesized gold nanoparticles with different sizes (2, 4 and 6 nm) and surface modification (cationic: TTMA, zwitterionic: SN and NS, and anionic: COOH). (A) General structure of AuNP, with either interactive cationic and anionic or “stealth” zwitterionic headgroups appended to a non-interacting OEG-functionalized interior. (B) TEM images of 2, 4, and 6 nm core diameter AuNPs. (C) UV-Vis absorbance spectra of the each of the particle families as a function of core size.
The particles were fabricated using 2 nm NPs protected with 1-dodecanethiol synthesized according to the Brust method,25 while 4 nm and 6 nm AuNP were grown from 2 nm dodecanethiol AuNP using heat-induced size evolution.26 These AuNP were then functionalized with TTMA, COOH or zwitterionic ligands via place exchange reactions. The successful NP surface functionalization was analyzed and confirmed using laser desorption/ionization mass spectrometry (LDI-MS, Figure S1). A molecular peak observed at m/z value of 422.3 was ascribed to TTMA on NP surface, while peaks at 438.3, 530.2 and 601.1 were attributed to COOH, NS and SN ligands, respectively. The number of ligands on each particle surface was determined by matrix-assisted laser desorption/ionization mass spectrometry (MALDI-MS, Table S1). The size and morphology of all NPs were further characterized using transmission electron microscopy (TEM) (Figure 1B) and dynamic light scattering (DLS) analysis. As shown in Table 1, the hydrodynamic size of AuNP increased gradually when the core size of NP was increased from 2 nm to 6 nm. Meanwhile, the functionalization of AuNP with TTMA, COOH, or zwitterionic surface ligands had minor effect on AuNP size. Zeta potential measurements confirmed the positive, negative, and neutral charges of TTMA, COOH, and zwitterionic ligand-coated AuNP, respectively (Table 1). As expected, UV spectra of AuNP indicated a red-shifted surface plasma resonance (SPR) peak when NP core size was increased from 2 to 6 nm, with no significant SPR band difference was observed between the same size AuNP coated with TTMA, COOH, and zwitterionic ligands (Figure 1C).
Table 1.
Dynamic light scattering analysis and zeta-potential measurements of as-synthesized nanoparticles in 5 mM phosphate buffer (pH = 7.4).
| Nanoparticles | Hydrodynamic size (nm) | Zeta-potential (mV) |
|---|---|---|
| 2 TTMA | 6.7 ± 0.4 | 18.2 ± 0.8 |
| 4 TTMA | 8.6 ± 0.6 | 19.3 ± 0.3 |
| 6 TTMA | 10.1 ± 1.1 | 17.5 ± 0.6 |
| 2 SN | 6.7 ± 0.2 | −3.6 ± 0.7 |
| 4 SN | 8.9 ± 0.2 | −4.8 ± 0.6 |
| 6 SN | 12.5 ± 0.4 | −6.1 ± 0.4 |
| 2 NS | 6.1 ± 0.4 | −3.7 ± 0.3 |
| 4 NS | 7.7 ± 0.2 | −7.9 ±1.4 |
| 6 NS | 10.6 ± 0.8 | −10.0 ± 1.2 |
| 2 COOH | 7.0 ± 0.2 | −41.2 ± 1.7 |
| 4 COOH | 8.2 ± 1.2 | −42.8 ± 1.2 |
| 6 COOH | 9.5 ±1.3 | −44.6 ± 1.6 |
Cellular uptake of AuNPs
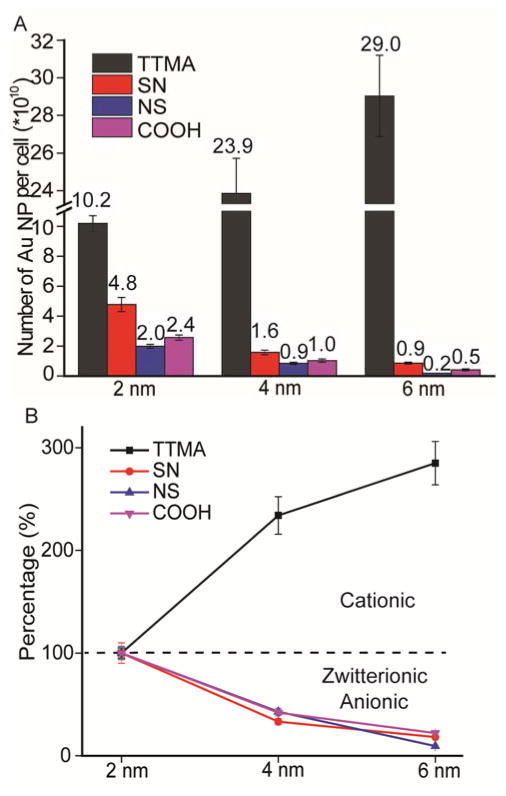
Our uptake studies used the widely employed human cervical carcinoma (HeLa) cell line, allowing the correlation of current study with prior reports. Preliminary studies indicated a low cytotoxicity of AuNP to HeLa Cells at a NP concentration of 25 nM, this concentration was then fixed for the following NP uptake study (Figure S2). The cellular internalization of AuNP was studied by exposing NPs to HeLa cells, followed by extensive washing to remove NPs adsorbed to the cell surface. AuNP uptake was quantified using inductively coupled plasma mass spectrometry (ICP-MS), with the mass of intracellular gold converted to the AuNP number using a previously reported method.7 As expected, all cationic AuNP featuring TTMA surface ligands enter cells in a significantly higher efficiency than that of neutral zwitterionic and anionic AuNP (Figure 2A). Interestingly, zwitterionic NPs with positive outmost layer charge (Au-SN) entered cells more efficiently than NPs with negative outmost layer charge (Au-NS), and anionic Au-COOH NPs, indicating the charge orientation of zwitterionic AuNP significantly affected NP internalization.
Figure 2.
(A) Cellular uptake of as-synthesized gold nanoparticles with different size and varied surface charge by HeLa cells after 3 h incubation in serum-free media. The amount of gold per cell was determined by ICP-MS. Number of gold nanoparticles inside cells was also indicated above the histogram bar. (B) Size-dependent uptake trend of AuNP. The uptake efficiency of 4 nm and 6 nm NP was normalized to that of 2 nm NP featuring the same surface charge. Mean values ± standard deviation, N = 3.
While there were no major surprises when looking at the functionality axis alone, parametric variation of charge and size generated unexpected interrelationships. For the cationic particles, increasing particle size from 2 nm to 6 nm increased uptake efficiency 3-fold (Figure 2B), a trend consistent with Chan’s study of larger AuNPs.18 A completely opposite trend was observed for both zwitterionic particles (NS and SN) and anionic particles, where increasing the core size decreased uptake, with 2 nm AuNPs uptaken >10-fold more efficiently than their 6 nm counterparts. This distinctive size dependence indicates that both particle size and surface functionality need to be considered together when designing carriers for cellular delivery.
Having found the size and surface charge dependence of the cellular uptake of AuNP in the absence of serum, we investigated further the uptake of these NP in complete cell culture medium containing 10% FBS, with the aim of understanding the effects of serum on NP uptake. For this purpose, HeLa cells were treated with NPs for 3 h or 24 h in the presence of 10% FBS. The treatment of NPs (25 nM) had negligible cytotoxicity, with all treated cells having >90% viability 24 h post-treatment (Figure S3). The cellular uptake efficiency of different NP was similarly quantified using ICP-MS. As shown in Figure S4A, all NPs were internalized less efficiently in the presence of serum compared to that in serum-free medium after 3 h of treatment. Our results are similar to previous reports of multiple particles that showed decreased cellular uptake in the presence of serum, possibly due to the nonspecific binding of proteins to the cell membrane, a competitive process that slows the uptake of NPs when serum proteins are present.7, 27 Notably, similar size and charge-determined cellular uptake trends were still observed for both zwitterionic and anionic NPs in the presence and absence of serum, while cationic NPs exhibited a significantly different trend. With 4 nm Au-TTMA NPs showed higher cellular uptake efficiency than that of 2 nm and 6 nm NPs in the presence of serum. In addition, the incubation of NPs with cells for 24 h showed similar size and charge-dependent cellular uptake, with enhanced number of NPs internalized to cells (Figure S4B). The different uptake trend of cationic NPs in the presence and absence of serum protein probably resulted from the distinct protein corona formation on the different sized particle surface, suggesting an indirect correlation between surface chemistry and biological response.28
Energy-dependent cellular uptake pathways of sub-10 nm AuNPs
The opposite trends in size dependencies for neutral and charged particle uptake strongly suggested different mechanisms may be involved for the internalization of these NPs. Previous studies have shown that energy-dependent endocytosis is the predominant pathway for nanoparticle internalization, a process that can be suppressed through reduced temperature and ATP depletion.29, 30 Cooling cells to 4°C only had minor effects on the uptake of any of the 2 nm particles (Figure 3, and Figure S5). For the cellular uptake of all NPs at reduced temperature, we observed strong size dependence. 2 nm TTMA particles internalized much more efficiently than that of 6 nm TTMA NPs. With zwitterionic and anionic particles, little change of uptake was observed for the 2 nm and 4 nm NPs. The behavior of the 6 nm particles differed, however, with a modest decrease observed for the SN particle and a much larger decrease observed with the NS analog and COOH particles. Qualitatively similar results were observed with pre-treatment of cells with sodium azide (NaN3, 10 mM)/2-deoxyglucose (DOG 50 mM) to deplete ATP, followed by AuNP exposure at 4 °C or 37 °C for ATP-depleted cells. Significantly, none of the 2 nm particles showed substantially different uptake under any of the conditions, strongly suggesting a different pathway was involved in the uptake of 2 nm NPs.
Figure 3.
Uptake efficiency of (A) Au-TTMA NP, (B) Au-COOH NP, (C) Au-SN NP, and (D) Au-NS NP by HeLa cells in the absence or presence of sodium azide (NaN3, 10 mM) and deoxy-glucose (DOG, 50 mM) at 4 °C or 37 °C after 3 h incubation in serum-free media. The cellular uptake efficiency of NPs (25 nM) was normalized to cells without inhibition treatment. Mean values ± standard deviation, N = 3. * p < 0.05, * * p < 0.01 compared to each normalized control.
Endocytosis inhibition of sub-10 nm AuNP
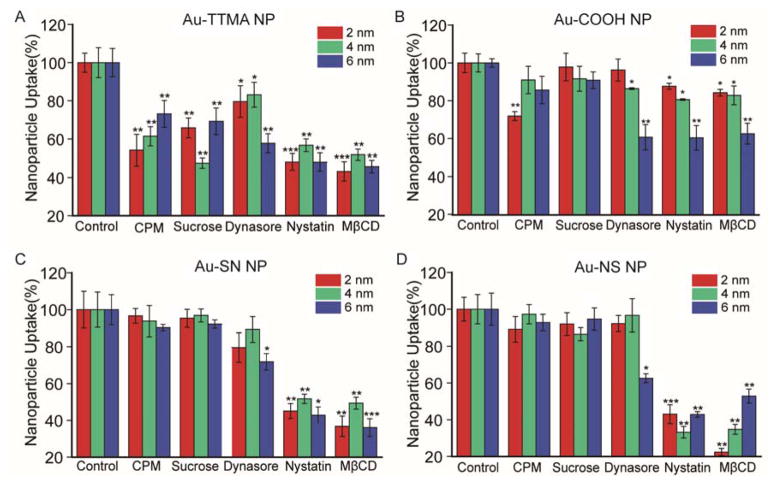
We next compared the effects of endocytosis inhibitors on NP uptake. We first studied the effects of chlorpromazine (CPM) and sucrose, which inhibit clathrin-mediated endocytosis.31–35 As shown in Figure 4, CPM (28 nM) and sucrose (450 mM) pre-treatments significantly inhibited the internalization of all TTMA-coated AuNPs. However, negligible inhibition effect was observed for zwitterionic and anionic AuNP with similar CPM and sucrose pre-treatment, with no dependence on NP size, but with the exception that modest inhibition effect was observed for 2 nm Au-COOH NPs with CPM treatment.
Figure 4.
Uptake efficiency of (A) Au-TTMA NP, (B) Au-COOH NP, (C) Au-SN NP, and (D) Au-NS NP by HeLa cells in the presence of different endocytosis inhibitors. HeLa cells were treated with 25 nM NP for 3 h incubation in serum-free media. Mean values ± standard deviation, N = 3. * p < 0.05, * * p < 0.01, * * * p < 0.001 compared to each normalized control.
We next pre-treated cells with dynasore, an effective inhibitor of dynamin-dependent endocytosis.36 We found that dynasore pre-treatment significantly inhibited the internalization of all Au-TTMA NP and 6 nm zwitterionic AuNPs (Figure 4 and Table 2). A strong size dependence was also observed for anionic NPs, 2 nm Au-COOH particles were less inhibited by dynasore than that of 6 nm NPs. These studies collectively show that TTMA particles and 2 nm Au-COOH particles are internalized through multiple endocytotic pathways, while dynamin-mediated endocytosis is the only operative endocytotic mechanism for zwitterionic and 6 nm anionic particles, a completely unexpected outcome.
Table 2.
Summary of cellular uptake inhibition of NP in presence of endocytic inhibitors. + p < 0.05, + + p < 0.01, + + + p < 0.001 through unpaired t-test between normalized control and inhibitor-treated groups. − no significant inhibition.
| Inhibitors | Function | 2 nm | 4 nm | 6 nm | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
| |||||||||||||
| TTMA | SN | NS | COOH | TTMA | SN | NS | COOH | TTMA | SN | NS | COOH | ||
| 4°C | Slow down metabolism | − | − | − | − | − | − | − | − | ++ | + | ++ | ++ |
| NaN3+DOG | ATP depletion | − | − | − | − | + | − | − | − | ++ | + | + | + |
| NaN3+DOG+4°C | ATP depletion/Slow down metabolism | + | − | − | − | ++ | + | + | − | ++ | ++ | ++ | ++ |
| Chlorpromazine (CPM) | Inhibits Rho GTPase(Clathrin) | ++ | − | − | ++ | ++ | − | − | − | ++ | − | − | − |
| Sucrose | Inhibitor of clathrin-mediated endocytosis | ++ | − | − | − | ++ | − | − | − | ++ | − | − | − |
| Dynasore | Inhibits Dynamin-GTPase | − | − | − | − | + | − | − | + | ++ | + | + | ++ |
| Nystatin | Inhibition of raft formation | +++ | ++ | +++ | + | ++ | ++ | ++ | + | ++ | + | ++ | ++ |
| MβCD | Cholesterol depletion/caveolae | +++ | ++ | ++ | + | ++ | ++ | ++ | + | ++ | +++ | ++ | ++ |
Finally, we studied the uptake difference of AuNP in the presence of cholesterol depletion agents that inhibit lipid raft-mediated uptake, in particular both membrane fusion processes and caveolae-dependent endocytosis.20, 37–39 The pre-treatment of cells with nystatin or methyl-β-cyclodextrin (MβCD) dramatically reduced uptake of all particles, including the 2 nm and 4 nm zwitterionic NPs that were unresponsive to other inhibitors (Figure 4 and Table 2). To exclude the possibility that these inhibitors may have interfered with the uptake of particles by inducing toxicity to cells, we measured the viability of HeLa cells treated with inhibitors for 3 h, and have not found obvious toxicity (Figure S6).
Taken together, it is clear that the interplay of particle size and coverage plays a complex role in determining NP uptake. With cationic particles, multiple endocytic pathways, including clathrin- and caveolae/lipid raft-mediated endocytosis were observed (Table 2 and Scheme 1). The zwitterionic NPs provided more specific mechanisms of uptake, with membrane fusion being the predominant mechanism for 2 nm and 4 nm zwitterionic NPs. In contrast, 6 nm zwitterionic NP were prone to enter cells through caveolae/lipid raft-mediated uptake, indicating a significant size-induced switching of the internalization mechanisms of NPs. Interestingly, 2 and 4 nm anionic particles displayed a similar multiple endocytic pathways to that of same sized cationic particles, while 6 nm anionic particles were internalized in caveolae/lipid raft-mediated pathway.
Scheme 1.

Interplay of size and surface functionality on the cellular uptake pathway of AuNP.
A key feature of our study is the importance of surface coverage in determining cell uptake efficiency and mechanism. The distinct energy-independent cellular uptake of 2 nm and 4 nm zwitterionic is consistent with the previous report by Alexander-Katz et al. that for small size NP (diameters ≤ 4 nm),40 the monolayer contains a large amount of free volume that maximizes the ligand fluctuations and minimizes the energy barrier to “snorkeling”, facilitating the energy-independent membrane fusion process for NPs to enter cells. The 6 nm zwitterionic and anionic NPs are above the size threshold for membrane fusion so these NPs mainly enter cells via dynamin-dependent endocytosis pathway.
CONCLUSIONS
In summary, we have shown that there is a strong interrelationship between NP size and surface functionality on the cellular uptake of sub-10 nm gold nanoparticles. We have found that relatively small changes of NP core size were sufficient to induce dramatic changes in NP internalization efficiency and mechanism. These studies demonstrate that variation of particle size and coverage can be used synergistically to control cell uptake processes, an important design tool for bionanotechnology applications.
METHODS
Cell culture
HeLa cells were purchased from American Type Culture Collection (ATCC, Manassas, VA) and maintained in Low-glucose Dulbecco’s modified Eagle’s medium (DMEM) with 10% fetal bovine serum (FBS) and 1% antibiotic (100 U/mL penicillin and 100μg/mL streptomycin). And the cells were cultured at 37 °C under a humidified atmosphere of 5% CO2.
Nanoparticle uptake and inhibition studies
HeLa cells were seeded in a 48 well plate at a density of ~ 2 × 104 cells/well 24 h a day before experiment. At the day of incubation, cells were washed three time with phosphate buffered saline (PBS) and pre-incubated with following endocytic inhibitors in serum free media for 1 h at 37 °C, methyl-β-cyclodextrin (10 mM), nystatin (180 nM), sodium azide (NaN3, 10 mM) + 2-deoxyglucose (DOG, 50 mM), dynasore (80 μM), chlorpromazine hydrochloride (28 nM), and sucrose (450 mM). All inhibitors were purchased from Sigma except for dynasore was obtained from Fisher Scientific, and the concentrations of the inhibitors were used according to previous reports.15, 30
After 1 h, nanoparticles were added at a concentration of 25 nM and incubated for another 3 h at 37 °C in the presence of the inhibitors. Untreated cells were used as negative control, and cells treated with only NPs in the absence of inhibitors were used as positive control. After incubation, cells were washed five times with PBS buffer. Cell lysis buffer (250 μL per well) was used to lyse the cell, with the cell culture plate kept at room temperature on a vibrator for 30 min. In addition, a freeze/thaw cycle was also adopted to further facilitate the cell lysis process. After that, cells were digested with 0.5 mL of fresh aqua regia for 10 min. The digested samples were further processed for ICP-MS analysis to determine the intracellular amount of gold. Each cell uptake experiment was done in triplicate, and each replicate was measured 5 times by ICP-MS.
Temperature and energy dependent pathways study
HeLa cells were seeded in a 48 well plate at a density of 2 × 104 cells/well a day before experiment. At the day of incubation, cells were washed three times with PBS buffer and pre-treated with 10 mM NaN3 and 50 mM DOG at 4 °C for 1 hour, then HeLa cell were incubated with 25 nM Au NPs in the presence of NaN3/DOG at 4 °C for another 3 hours.
Sample preparation for ICP-MS and ICP-MS measurements
For the quantitative determination of Au content in the cellular uptake study, the cell lysates were digested with 0.5 mL fresh aqua regia composed of hydrochloric acid and nitric acid (3:1, v/v) (highly corrosive and must be used with extreme caution!) for 10 min.15, 41, 42 Then each digested sample was diluted to 10 mL with de-ionized water. A series of gold standard solutions (20, 10, 5, 2, 1, 0.5, 0.2, and 0 ppb) were prepared at the same time. Each gold standard solution also contained 5% aqua regia. The gold standard solutions and cellular uptake sample solutions were measured on a Perkin-Elmer NexION 300X ICP mass spectrometer.
Particle uptake efficiency (%) was calculated based on the following equation:
And inhibition efficiency (%) was calculated by the following equation:
Supplementary Material
Acknowledgments
This work was supported by the grants from the NIH (EB014277), National Distinguished Young Scholars grant from NSFC (31225009) and financial support from China Scholarship Council. T. M. is grateful to the Japan Society for the Promotion of Sciences (JSPS) for a Postdoctoral Fellowship for Research Abroad.
Footnotes
Conflicts of Interest: The authors declare no competing financial interests.
Supporting Information Available: Nanoparticle synthesis and characterization, and additional experimental details and figures. This material is available free of charge via the Internet at http://pubs.acs.org.
REFERENCES AND NOTES
- 1.Jiang W, Kim BYS, Rutka JT, Chan WCW. Nanoparticle-Mediated Cellular Response is Size-Dependent. Nat Nanotech. 2008;3:145–150. doi: 10.1038/nnano.2008.30. [DOI] [PubMed] [Google Scholar]
- 2.Lundqvist M, Stigler J, Elia G, Lynch I, Cedervall T, Dawson KA. Nanoparticle Size and Surface Properties Determine the Protein Corona with Possible Implications for Biological Impacts. P Natl Acad Sci U S A. 2008;105:14265–14270. doi: 10.1073/pnas.0805135105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhang SL, Li J, Lykotrafitis G, Bao G, Suresh S. Size-Dependent Endocytosis of Nanoparticles. Adv Mater. 2009;21:419–424. doi: 10.1002/adma.200801393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Huo SD, Ma HL, Huang KY, Liu J, Wei T, Jin SB, Zhang JC, He ST, Liang XJ. Superior Penetration and Retention Behavior of 50 nm Gold Nanoparticles in Tumors. Cancer Res. 2013;73:319–330. doi: 10.1158/0008-5472.CAN-12-2071. [DOI] [PubMed] [Google Scholar]
- 5.Ma X, Wu Y, Jin S, Tian Y, Zhang X, Zhao Y, Yu L, Liang XJ. Gold Nanoparticles Induce Autophagosome Accumulation through Size-Dependent Nanoparticle Uptake and Lysosome Impairment. ACS Nano. 2011;5:8629–8639. doi: 10.1021/nn202155y. [DOI] [PubMed] [Google Scholar]
- 6.Yang K, Ma YQ. Computer Simulation of the Translocation of Nanoparticles with Different Shapes across A Lipid Bilayer. Nat Nanotech. 2010;5:579–583. doi: 10.1038/nnano.2010.141. [DOI] [PubMed] [Google Scholar]
- 7.Chithrani BD, Ghazani AA, Chan WCW. Determining the Size and Shape Dependence of Gold Nanoparticle Uptake into Mammalian Cells. Nano Lett. 2006;6:662–668. doi: 10.1021/nl052396o. [DOI] [PubMed] [Google Scholar]
- 8.Jin S, Ma X, Ma H, Zheng K, Liu J, Hou S, Meng J, Wang PC, Wu X, Liang XJ. Surface Chemistry-Mediated Penetration and Gold Nanorod Thermotherapy in Multicellular Tumor Spheroids. Nanoscale. 2013;5:143–146. doi: 10.1039/c2nr31877f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Han HS, Martin JD, Lee J, Harris DK, Fukumura D, Jain RK, Bawendi M. Spatial Charge Configuration Regulates Nanoparticle Transport and Binding Behavior in Vivo. Angew Chem Int Edit. 2013;52:1414–1419. doi: 10.1002/anie.201208331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cho EC, Xie JW, Wurm PA, Xia YN. Understanding the Role of Surface Charges in Cellular Adsorption versus Internalization by Selectively Removing Gold Nanoparticles on the Cell Surface with A I-2/KI Etchant. Nano Lett. 2009;9:1080–1084. doi: 10.1021/nl803487r. [DOI] [PubMed] [Google Scholar]
- 11.Verma A, Stellacci F. Effect of Surface Properties on Nanoparticle-Cell Interactions. Small. 2010;6:12–21. doi: 10.1002/smll.200901158. [DOI] [PubMed] [Google Scholar]
- 12.Arvizo RR, Rana S, Miranda OR, Bhattacharya R, Rotello VM, Mukherjee P. Mechanism of Anti-Angiogenic Property of Gold Nanoparticles: Role of Nanoparticle Size and Surface Charge. Nanomed Nanotech. 2011;7:580–587. doi: 10.1016/j.nano.2011.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kostarelos K, Lacerda L, Pastorin G, Wu W, Wieckowski S, Luangsivilay J, Godefroy S, Pantarotto D, Briand JP, Muller S, et al. Cellular Uptake of Functionalized Carbon Nanotubes is Independent of Functional Group and Cell Type. Nat Nanotech. 2007;2:108–113. doi: 10.1038/nnano.2006.209. [DOI] [PubMed] [Google Scholar]
- 14.Liang M, Lin IC, Whittaker MR, Minchin RF, Monteiro MJ, Toth I. Cellular Uptake of Densely Packed Polymer Coatings on Gold Nanoparticles. ACS Nano. 2010;4:403–413. doi: 10.1021/nn9011237. [DOI] [PubMed] [Google Scholar]
- 15.Saha K, Kim ST, Yan B, Miranda OR, Alfonso FS, Shlosman D, Rotello VM. Surface Functionality of Nanoparticles Determines Cellular Uptake Mechanisms in Mammalian Cells. Small. 2013;9:300–305. doi: 10.1002/smll.201201129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Albanese A, Tang PS, Chan WCW. The Effect of Nanoparticle Size, Shape, and Surface Chemistry on Biological Systems. Annu Rev Biomed Eng. 2012;14:1–16. doi: 10.1146/annurev-bioeng-071811-150124. [DOI] [PubMed] [Google Scholar]
- 17.Saha K, Bajaj A, Duncan B, Rotello VM. Beauty is Skin Deep: A Surface Monolayer Perspective on Nanoparticle Interactions with Cells and Biomacromolecules. Small. 2011;7:1903–1918. doi: 10.1002/smll.201100478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Walkey CD, Olsen JB, Guo HB, Emili A, Chan WCW. Nanoparticle Size and Surface Chemistry Determine Serum Protein Adsorption and Macrophage Uptake. J Am Chem Soc. 2012;134:2139–2147. doi: 10.1021/ja2084338. [DOI] [PubMed] [Google Scholar]
- 19.Khlebtsov N, Dykman L. Biodistribution and Toxicity of Engineered Gold Nanoparticles: A Review of in Vitro and in Vivo Studies. Chem Soc Rev. 2011;40:1647–1671. doi: 10.1039/c0cs00018c. [DOI] [PubMed] [Google Scholar]
- 20.Jiang Y, Tang R, Duncan B, Jiang Z, Yan B, Mout R, Rotello VM. Direct Cytosolic Delivery of siRNA Using Nanoparticle-Stabilized Nanocapsules. Angew Chem Int Edit. 2015;54:506–510. doi: 10.1002/anie.201409161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Huo SD, Jin SB, Ma XW, Xue XD, Yang KN, Kumar A, Wang PC, Zhang JC, Hu ZB, Liang XJ. Ultrasmall Gold Nanoparticles as Carriers for Nucleus-Based Gene Therapy Due to Size-Dependent Nuclear Entry. ACS Nano. 2014;8:5852–5862. doi: 10.1021/nn5008572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Huang KY, Ma HL, Liu J, Huo SD, Kumar A, Wei T, Zhang X, Jin SB, Gan YL, Wang PC, et al. Size-Dependent Localization and Penetration of Ultrasmall Gold Nanoparticles in Cancer Cells, Multicellular Spheroids, and Tumors in Vivo. ACS Nano. 2012;6:4483–4493. doi: 10.1021/nn301282m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhou C, Long M, Qin YP, Sun XK, Zheng J. Luminescent Gold Nanoparticles with Efficient Renal Clearance. Angew Chem Int Edit. 2011;50:3168–3172. doi: 10.1002/anie.201007321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.De M, You CC, Srivastava S, Rotello VM. Biomimetic Interactions of Proteins with Functionalized Nanoparticles: A Thermodynamic Study. J Am Chem Soc. 2007;129:10747–10753. doi: 10.1021/ja071642q. [DOI] [PubMed] [Google Scholar]
- 25.Brust M, Walker M, Bethell D, Schiffrin DJ, Whyman R. Synthesis of Thiol-Derivatised Gold Nanoparticles in A Two-Phase Liquid-Liquid System. J Chem Soc, Chem Commun. 1994:801–802. [Google Scholar]
- 26.Teranishi T, Hasegawa S, Shimizu T, Miyake M. Heat-Induced Size Evolution of Gold Nanoparticles in the Solid State. Adv Mater. 2001;13:1699–1701. [Google Scholar]
- 27.Kim CS, Le ND, Xing Y, Yan B, Tonga GY, Kim C, Vachet RW, Rotello VM. The Role of Surface Functionality in Nanoparticle Exocytosis. Adv Healthc Mater. 2014;3:1200–1202. doi: 10.1002/adhm.201400001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Monopoli MP, Aberg C, Salvati A, Dawson KA. Biomolecular Coronas Provide the Biological Identity of Nanosized Materials. Nat Nanotechnol. 2012;7:779–86. doi: 10.1038/nnano.2012.207. [DOI] [PubMed] [Google Scholar]
- 29.Kim JS, Yoon TJ, Yu KN, Noh MS, Woo M, Kim BG, Lee KH, Sohn BH, Park SB, Lee JK, Cho MH. Cellular Uptake of Magnetic Nanoparticle is Mediated through Energy-Dependent Endocytosis in A549 Cells. J Vet Sci. 2006;7:321–326. doi: 10.4142/jvs.2006.7.4.321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang LM, Liu Y, Li W, Jiang XM, Ji YL, Wu XC, Xu LG, Qiu Y, Zhao K, Wei TT, et al. Selective Targeting of Gold Nanorods at the Mitochondria of Cancer Cells: Implications for Cancer Therapy. Nano Lett. 2011;11:772–780. doi: 10.1021/nl103992v. [DOI] [PubMed] [Google Scholar]
- 31.McMahon HT, Boucrot E. Molecular Mechanism and Physiological Functions of Clathrin-Mediated Endocytosis. Nat Rev Mol Cell Bio. 2011;12:517–533. doi: 10.1038/nrm3151. [DOI] [PubMed] [Google Scholar]
- 32.Stuart AD, Brown TDK. Entry of Feline Calicivirus is Dependent on Clathrin-Mediated Endocytosis and Acidification in Endosomes. J Virol. 2006;80:7500–7509. doi: 10.1128/JVI.02452-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Liu Y, Kim YJ, Ji M, Fang J, Siriwon N, Zhang LI, Wang P. Enhancing Gene Delivery of Adeno-Associated Viruses by Cell-Permeable Peptides. Mol Ther Methods Clin Dev. 2014;1:1–12. doi: 10.1038/mtm.2013.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Madani F, Lindberg S, Langel Ü, Futaki S, Gräslund A. Mechanisms of Cellular Uptake of Cell-Penetrating Peptides. J Biophy. 2011;2011:1–10. doi: 10.1155/2011/414729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Zhu J, Liao L, Zhu LN, Zhang P, Guo K, Kong JL, Ji C, Liu BH. Size-Dependent Cellular Uptake Efficiency, Mechanism, and Cytotoxicity of Silica Nanoparticles toward HeLa Cells. Talanta. 2013;107:408–415. doi: 10.1016/j.talanta.2013.01.037. [DOI] [PubMed] [Google Scholar]
- 36.Macia E, Ehrlich M, Massol R, Boucrot E, Brunner C, Kirchhausen T. Dynasore, A Cell-Permeable Inhibitor of Dynamin. Dev Cell. 2006;10:839–850. doi: 10.1016/j.devcel.2006.04.002. [DOI] [PubMed] [Google Scholar]
- 37.Nabi IR, Le PU. Caveolae/raft-Dependent Endocytosis. J Cell Biol. 2003;161:673–677. doi: 10.1083/jcb.200302028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Rodal SK, Skretting G, Garred O, Vilhardt F, Van Deurs B, Sandvig K. Extraction of Cholesterol with Methyl-b-cyclodextrin Perturbs Formation of Clathrin-Coated Endocytic Vesicles. Mol Biol Cell. 1999;10:961–974. doi: 10.1091/mbc.10.4.961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Verma A, Uzun O, Hu YH, Hu Y, Han HS, Watson N, Chen SL, Irvine DJ, Stellacci F. Surface-Structure-Regulated Cell-Membrane Penetration by Monolayer-Protected Nanoparticles. Nat Mater. 2013;12:376–376. doi: 10.1038/nmat2202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Van Lehn RC, Atukorale PU, Carney RP, Yang YS, Stellacci F, Irvine DJ, Alexander-Katz A. Effect of Particle Diameter and Surface Composition on the Spontaneous Fusion of Monolayer-Protected Gold Nanoparticles with Lipid Bilayers. Nano Lett. 2013;13:4060–4067. doi: 10.1021/nl401365n. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Zhu ZJ, Yeh YC, Tang R, Yan B, Tamayo J, Vachet RW, Rotello VM. Stability of Quantum Dots in Live Cells. Nat Chem. 2011;3:963–968. doi: 10.1038/nchem.1177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Liu X, Atwater M, Wang J, Huo Q. Extinction Coefficient of Gold Nanoparticles with Different Sizes and Different Capping Ligands. Colloids Surf B Biointerfaces. 2007;58:3–7. doi: 10.1016/j.colsurfb.2006.08.005. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.