Abstract
Medroxyprogesterone 17-acetate (MPA) combined with human menopausal gonadotropin (hMG) has been effectively used for ovarian stimulation in clinical practice. However, the molecular mechanism of MPA + hMG treatment in follicular development is poorly described. Here we performed a study to investigate the impact of MPA + hMG on ovarian stimulation utilizing a mouse model in vivo. Forty female BALB/C mice were randomly divided into four groups of 10 each and treated during ciestrus stage and continued for 5 days: control group, MPA group, hMG group, and MPA + hMG group. Morphological and molecular biology methods were used for detecting serum hormones and ovarian function. MPA + hMG group exhibited increasing follicle stimulating hormone (FSH), antral follicle, FSH receptor (FSHR) and phosphorylated mammal target of rapamycin (p-mTOR), and decreasing luteinizing hormone (LH), estradiol (E2), progesterone (P), corpus luteum, phosphoinositide 3-kinase (PI3K), Akt and mTOR compared with control group. In contrast, MPA + hMG group showed reduced FSH, LH, E2, P, corpus luteum, LH receptor (LHR), and activated PI3K,/Akt/mTOR pathway compared with hMG group (P < 0.05). Collectively, these data definitively established that MPA plus hMG may modulate the hormone, hormone receptor and PI3K/Akt/mTOR signaling pathway to influence follicular development in the mouse ovary. Our study provides overwhelming support for MPA + hMG as an effective treatment for infertility in women.
Introduction
Infertility is one of the most common diseases in the world. Currently, more and more patients have to seek help from assisted reproductive therapy (ART) to have their own child1. The administration of gonadotropin-releasing hormone (GnRH) analogues, including GnRH agonists and GnRH antagonists, is used for preventing premature LH surges in pituitary desensitization among infertile patients in conventional controlled ovarian hyperstimulation2. However, this therapy has proven to have some limits because of the increased incidence of ovarian hyperstimulation syndrome (OHSS) by GnRH agonists and the rate of premature LH surges (0.34–38.3%) via GnRH antagonists3,4. Owing to the “freeze-all” strategies, a new ovarian stimulation regimen, MPA combined with hMG treatment was proposed by Dr. Kuang to inhibit premature LH surges and reduce the incidence of OHSS during follicular phase5. Our previous clinical studies have demonstrated that the MPA + hMG treatment is successful used in patients with normal ovarian response, advanced maternal age, low ovarian response, or polycystic ovary syndrome (PCOS)6, however, the molecular mechanism is not understood.
P is widely used for menstrual disorders, hormone replacement therapy and luteal support for pregnancy7 Animal research found that P improves follicular viability by increasing the levels of vascular endothelial growth factor and granulosa cell proliferation in large follicles and promotes the maturation of fish oocytes by promoting germinal vesicle breakdown8–10. In clinic, MPA is used as an alternative to GnRH analog to suppress a premature LH surge and avoid a low response of the hypothalamic-pituitary-ovarian axis (HPOA) during the follicular phase11. Unlike dydrogesterone, MPA does not disturb the measurement of serum progesterone. And the the administration of P during follicular phase had no negative effect on oocyte retrieval rates of the hMG + MPA treatment cycles based on frozen embryo transfer12. This suggests that MPA + hMG treatment is crucial for ovarian stimulation.
In most physiologic contexts, the PI3K/Akt signaling pathway is an important regulatory factor for cell proliferation and the initiation of oocyte growth13. Ovarian follicular growth is dependent on the growth and proliferation of granulosa cells. Indeed, it was found that FSH (the main component of hMG) could promote the rapid activation of PI3K pathway in ovarian granulosa cells, and PI3K catalyzes the production of PIP3 in the plasma membrane, leading to membrane recruitment, phosphorylation, and activation of downstream branched chain kinase Akt14. Meanwhile, it is known that Akt causes mTOR activation through a variety of mechanisms15. Activation of mTOR synergistically stimulate the growth of follicles16.
Our previous data indicate that MPA + hMG treatment does not impair the outcome of IVF/ICSI for FET. Based on our own and others’ work, we developed a mouse model of MPA + hMG treatment to investigate the role of MPA + hMG in follicular development and hypothesized that MPA + hMG may promote follicular development by regulating ovarian hormones, hormone receptors and PI3K/Akt/mTOR signaling pathways.
Results
Ovary weight index
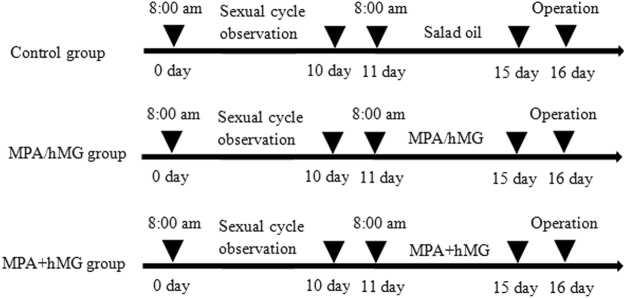
After successful establishment of the MPA + hMG mice model, both ovaries of the mice were removed and weighed (Fig. 1). The ovarian weight index of each group is presented in Table 1. The ovarian wet weights and ovarian weight indexes are not significantly different between all groups (P > 0.05).
Figure 1.
The establishment of MPA + hMG mice model.
Table 1.
The ovarian weight index of each group.
| Group | Number | Ovarian wet weight (mg) | Ovary index (%) | P-value |
|---|---|---|---|---|
| Control group | 10 | 9.72 ± 0.81 | 0.049 ± 0.041 | 0.2439 |
| MPA group | 10 | 10.22 ± 1.17 | 0.052 ± 0.058 | 0.3094 |
| hMG group | 10 | 10.74 ± 1.22 | 0.055 ± 0.061 | 0.2617 |
| MPA + hMG group | 10 | 11.66 ± 0.67 | 0.058 ± 0.033 | — |
Plus–minus values represent the mean ± SD. MPA, medroxyprogesterone acetate; hMG, human menopausal gonadotropin. Ovarian index (%) = ovarian wet weight (mg)/body weight (g) × 100%. P-value represents MPA + hMG group vs Control group, MPA group or hMG group.
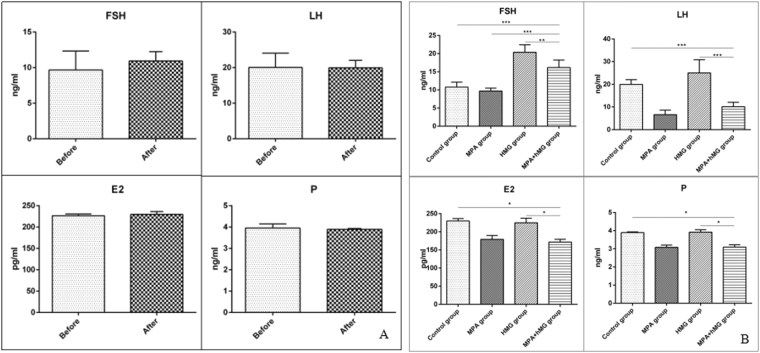
Serum hormone levels
To determine the effect of MPA + hMG on serum FSH, LH, E2, and P levels, serum was collected on the first day and after 5 days of treatment and these values were analyzed. In the control group, there are no significant changes in serum hormones before or after administration of salad oil (p-value is 0.3224, 0.9543, 0.3389 and 0.4665 in FSH, LH, E2, and P levels, respectively) (Fig. 2A). In the MPA + hMG group, there is a significantly increase in FSH level and decreases in LH, E2 and P levels compared to the control group (P < 0.05), meanwhile, FSH level is higher in MPA + hMG group than the MPA group. The levels of FSH, LH, E2, and P are all decreased in MPA + hMG group compared with the hMG group (P < 0.05) (Fig. 2B).
Figure 2.
The changes in serum FSH, LH, E2 and P levels. (A) The level of serum FSH, LH, E2 and P in control group before and after 5 days; (B) The levels of serum FSH, LH, E2 and P were detected after 5 days. *Represents MPA + hMG group vs control group, MPA group or hMG group. *P < 0.05, **P < 0.01, ***P < 0.001.
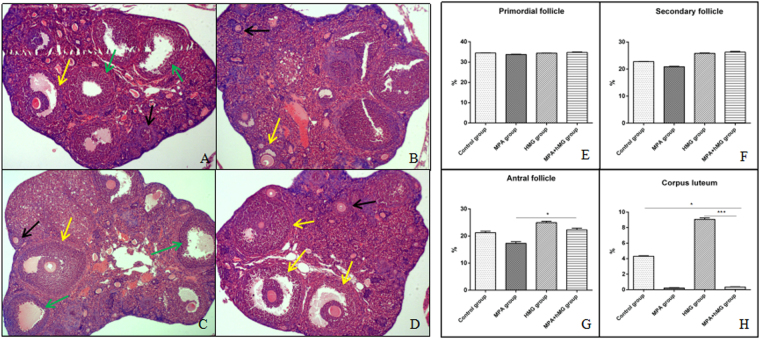
Effects of MPA + hMG on ovarian follicle development
In the control group (Fig. 3A), visible primordial follicles, greater volumes of the large follicles, thicker granulosa cells, and the limited amount of follicular atresia are observed; however, visible corpus luteum development is more pronounced than in the MPA + hMG group. In the MPA group (Fig. 3B), the number and volume of antral follicles are decreased compared to the control group; and there not appear newly formed corpus luteum in the MPA group. Greater volumes of the large follicles, thicker granulosa cells and corpus luteum are observed in the hMG group (Fig. 3C). In MPA + hMG group (Fig. 3D), the number of antral follicles and follicular volume is increased, granulosa cells are thicker, and the number of atresic follicles is decreased compared to the control group (P < 0.05); and there not appear newly formed corpus luteum compared with the control group and hMG group. There are not significantly different in the percentage of primordial follicles and secondary follicles between all groups (P > 0.05) (Fig. 3E,F). The percentage of antral follicles in MPA + hMG group is higher than that in MPA group (Fig. 3G). Meanwhile, corpus luteum is lower in MPA + hMG group compared to the control and hMG groups (Fig. 3H).
Figure 3.
Effects of MPA + hMG on ovarian follicle development. Histologic sections were prepared from the ovaries of each group 24 hours after MPA or hMG treatment and stained with H&E, a detailed histopathological examination was performed under a light microscope noting each of the ovarian components. (A) Normal ovary of a control mouse. (B) The ovary with decreasing antral follicle and corpus luteum after MPA treatment. (C) The ovary with increasing corpus luteum after hMG treatment. (D) The ovary with increasing antral follicle and decreasing corpus luteum after MPA + hMG treatment. (E) The percentage of primordial follicle in four groups after 5 days of treatment. (F) The percentage of secondary follicle in four groups after 5 days of treatment. (G) The percentage of antral follicle in four groups after 5 days of treatment. (H) The percentage of corpus luteum in four groups after 5 days of treatment. Black arrows represent secondary follicle, yellow arrows represent antral follicle, green arrows represent corpus luteum. *Represents MPA + hMG group vs control group, MPA group or hMG group. *P < 0.05, ***P < 0.001.
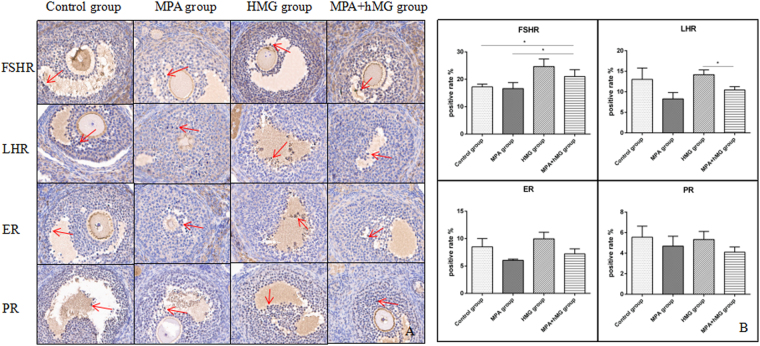
The effects of treatment on ovarian FSHR, LHR, ER, and PR expression level
In the MPA + hMG group, there is a significant increase in FSHR expression level compared to the control group and MPA group (P < 0.05), and a decreased FSHR and LHR expression level compared with the hMG group (P < 0.05). Although the ER in MPA group is lower compared to MPA + hMG group, it is not significantly different between them (p = 0.0623). There is no significant differences of between all groups (P > 0.05) (Fig. 4).
Figure 4.
The effects of treatment on ovarian FSHR, LHR, ER, and PR expression level. (A) The FSHR, LHR, ER, and PR were detected by immunohistochemistry, red arrows represent the expression of receptors. (B) The positive rate of FSHR, LHR, ER, and PR in four groups. *Represents MPA + hMG group vs control group, MPA group or hMG group. *P < 0.05.
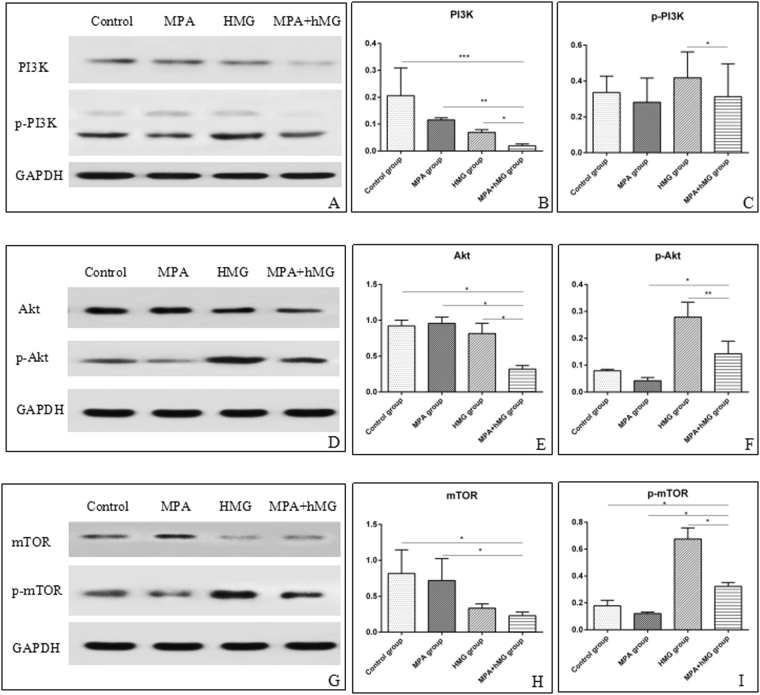
The effects of treatment on the PI3K/Akt/mTOR signaling pathway in ovarian tissue
To investigate whether the PI3K/Akt/mTOR pathway is activated by MPA + hMG treatment, we have assayed the phosphorylation of PI3K, Akt, and mTOR by western blot in ovarian tissues. Interestingly, quantification of protein expression shows that MPA + hMG causes a reduction in expression of PI3K, Akt and mTOR compared to the control group and MPA group, and the decreased PI3K and Akt in MPA + hMG group compared to the hMG group. While increased expression of p-Akt and p-mTOR in MPA + hMG group compared to the MPA group and a higher p-mTOR in MPA + hMG group than that in control group (P < 0.05). On the contrary, the expression of p-PI3K, p-Akt and p-mTOR is significantly decreased in the MPA + hMG group compared to the hMG group (P < 0.05) (Fig. 5).
Figure 5.
The effects of treatment on the PI3K/AKT/mTOR signaling pathway in ovarian tissue. Ten samples were used for each western blot assay, and each sample was measured in triplicate. (A–C) Western analyses of PI3K and p-PI3K, and their statistical analysis in all groups. (D–F) Western analyses of AKT and p-Akt,, and their statistical analysis in all groups. (G–I) Western analyses of mTOR and p-mTOR,, and their statistical analysis in all groups. *Represents MPA + hMG group vs control group, MPA group or hMG group. *P < 0.05.
Discussion
The benefits of MPA + hMG treatment in ART to improve success rates of IVF/ICSI was recently discovered and confirmed by Dr. Kuang followed by embryo cryopreservation. However, the mechanism(s) of MPA + hMG treatment remained unclear. In this study, we generated a BALB/C mouse model of clinical MPA + hMG treatment to delineate whether this treatment have positive effects on follicular development, through regulation of ovarian hormones, their receptors and the involvement of the PI3K/Akt/mTOR pathway on follicular development during ovarian hyperstimulation. This investigation demonstrated that the serum FSH level in MPA + hMG group was higher than the control group and MPA group, and lower than the hMG group. Meanwhile, reductions of serum LH, E2 and P were observed in MPA + hMG group when compared to the control group and hMG group. Studies found that the changes of plasma FSH, E2, and P in ovarian tissues were influenced by the effect of exogenous P on the pituitary under controlled ovarian hyperstimulation17, and hMG can significantly increase plasma FSH and LH levels18. In addition, the rapid increase in E2 produces a GnRH impulse and LH surges via positive feedback of the hypothalamus and pituitary in follicular development19, while P has potent anti-GnRH activity exerted at the hypothalamic level. Thus, it diminishes the sensitivity of GnRH-stimulated LH release and inhibits FSH-stimulated E2 production by controlling FSH induction of the aromatase enzyme20,21. Thus, the MPA + hMG group still did not have a higher E2 levels during the activating PI3K/Akt/mTOR pathway. Another study found that MPA is mainly as effective as oral contraceptives to inhibit ovulation22, leading to the decreased of E2 and P levels in MPA + hMG group compared with the control group and hMG group. According to these results, we hypothesize that the elevated FSH and decreased LH, E2 and P were mainly attributed to the effect of hMG on FSH as well as anti-GnRH effects of the hypothalamus and pituitary mediated by MPA administration.
In the MPA + hMG group and the MPA group, the treatment of MPA resulted in remarkable ovulation inhibition and no new corpus luteum due to the use of exogenous P as a contraceptive is associated with suppression of ovulation for a longer period23. Interestingly, some studies suggested that the MPA treatment suppressed the dominant follicle more than the physiological dose during the growing phase24, there are still a number of dominant follicles in the MPA + hMG group, which may be due to the FSH, as the main component of hMG and maintenance of FSH during follicular development can result in granulosa cell proliferation and oocyte development and reduce the negative effects of MPA on dominant follicles25.
Analyses of ovarian hormone receptor demonstrated that a higher FSHR was showed in the MPA + hMG group than the control group and MPA group. In the current study, FSH upregulates the expression of FSHR, and the alterations in the FSHR may contribute to the variability of ovarian response to FSH26. In addition, high levels or activity of FSHR may affect the intensity of FSH, and in turn, the increased feedback regulation of FSHR may be stimulated by FSH27,28. Moreover, P increased the response of granulosa cells to FSH via increased cAMP, which plays an important role in folliculogenesis and follicle survival29. The fact that we observed increased FSH and FSHR in the MPA + hMG group suggested that those results might lead to the improvement of follicular growth by hMG and increased sensitivity of granulosa cells to FSH via MPA. Moreover, Karlsson et al. showed that the activation of LHR, upon binding LH in ovarian granulosa initiates signaling cascades that regulate transcription of genes necessary for ovulation and luteinization30. In our study we showed a lower LHR and non-ovulation in MPA + hMG group compared to the hMG group, which due to the P can participate in down-regulation of the LHR gene expression31.
Edson MA et al. found that FSH substantially activated the PI3K/Akt pathway in ovarian granulosa cells, and this pathway plays critical roles in folliculogenesis, including the activation of follicles and maturation32, and reduced fertility were found in an Akt1-knockout females, resulting in reduced numbers of antral follicles33. Sun et al. used transient treatment with mTOR and PI3K stimulators in vitro and observed a synergistic effect on follicular development34. Increasing evidence suggests the involvement of PI3K/Akt/mTOR in cell proliferation, follicular growth and development, maturation, and periodic ovulation, which is activated in oocytes and granulosa35–37. However, there are still limited studies that have been developed to determine whether MPA + hMG can affect follicle growth, development and maturation. In order to explore the molecular mechanisms underlying the regulatory function of MPA + hMG on follicle development, PI3K/Akt/mTOR pathway was examined in mouse ovaries. Most importantly, we found that the levels of p-Akt and p-mTOR in the MPA + hMG group were significantly higher compared to the control and MPA groups. Similar to previous studies, in this study, hMG was added to stimulate PI3K/Akt/mTOR signal transduction in granulosa cells38. As studies showed that FSH activated the PI3K pathway including increased Akt and mTOR, which predominantly occurs in the nucleus and cytoplasm39,40. This indicates that hMG can increase p-Akt and p-mTOR by activating the PI3K/Akt/mTOR signalling pathway. Our results clearly demonstrated that the development of follicles via activation of the PI3K/Akt/mTOR pathway relied on the appropriate dose of MPA combined with hMG. To our knowledge, there are no published animal trials of MPA + hMG in ovarian stimulation including an analysis of its effect on the ovary microenvironment and PI3K/Akt/mTOR signaling pathway.
Although the ovarian index was higher in the MPA + hMG group than in the other three groups, there was no statistically significant difference between the groups. This may be due to the inhibition of ovulation in MPA group, multiple follicles development but no ovulation in the hMG group, and reduced the effect of multiple follicles development and inhibition of ovulation via MPA in MPA + hMG group were presented. Therefore, the number of follicles and ovulation may affect the size of ovarian index, although in the activated PI3K/Akt/mTOR pathway.
In conclusion, our results demonstrated that follicular development in MPA + hMG treated mice were associated with activation of the PI3K/Akt/mTOR pathway and the variation of serum hormone and hormone receptor via the synergistic effect of hMG and MPA. Moreover, our findings provide a theoretical basis to establish a new protocol for ovarian stimulation for infertile patients.
Methods
Animal treatment
Female, 6 week-old BALB/C mice were obtained from laboratory animal center, Shanghai Jiaotong University School of Medicine and kept under standard conditions: environmental temperature 20 ± 2, relative humidity 60%~80%, and on light of 14 hours one day. Water and food were available ad libitum. Forty female mice were randomly divided into four groups including: control group, MPA group, hMG group and MPA + hMG group.
Experimental design
The female mice cycle was observed at 8 am every day and sexual cycle was divided by the characteristic of cellular changes and selected the normal cycle mice as the experimental object. According to the principle of vaginal exfoliated cells (leukocytes, nucleated cells and keratinocytes): proestrus stage, 17–21 hours, lots of nucleated epithelium cells and a small amount of keratinocytes; estrus stag, 9–15 hours, keratinocytes; metestyus stage, 10–14 hours, leukocytes, nucleated cells and keratinocytes; ciestrus stage, 60–70 hours, only the presence of white blood cells. The control group received 0.25 ml/day salad oil via gavage; the MPA group conducted 3.0 mg/kg·d MPA (Zhejiang Xianju Co. Ltd, Zhejiang, China) via gavage; 5 IU/d hMG (Anhui Fengyuan Pharmaceutical Co. Ltd, Hefei, China) was used for hMG group via intraperitoneal injection; and the MPA + hMG group administered 3.0 mg/kg·d MPA combined with 5 IU/day hMG via gavage and intraperitoneal injection, respectively. All animals were treated for 5 days and vaginal smears were taken to determine the estrous cycle continuously.
Ovary index calculation
Animals were sacrificed after 24 hours of MPA or/and hMG administration for 5 days. Their ovaries (bilateral) were collected carefully. The fascia and adipose tissue was removed from the ovaries and the wet weight of the ovaries were measured. Ovary index was calculated according to the following formula: ovarian index = ovarian wet weight (mg)/body weight (g) × 100%.
Hormonal assay
Orbital blood collection were performed before treatmnet and blood collection of enucleation were performed after 5 days of treatment. Then they were kept at room temperature for 2 hours and centrifuged at 2000 g for 30 minutes. Subsequently, serum were transferred into 1.5 mL polypropylene tubes, and stored at −20 °C. Serum FSH, LH, E2 and P levels were determined using an Euzyme Linked Immimosorbent Assay Kit (Yinggong Corporation, Shanghai, China) and measured the absorbance at 450 nm.
Histological examination
The ovaries were fixed in 4% polysorbate solution for 24 hours. Dehydration, embedding, slicing (thickness:5 um) and hematoxylin & eosin staining were performed, number of follicles at all levels were observed under an 10x magnification optical microscope. The classification of follicles refers to the classification of Myers41. Only follicles containing an oocyte with a visible nucleus were counted to avoid double counting.The percentage of the number of follicles at each stage on total number of oocytes was calculated.
Tissue microarray immunohistochemistry analysis
Mouse ovaries were fixed in 10% buffered formalin for paraffin embedding and arranged by array, with an aperture of 2 mm, spacing 2 mm. FSHR (1:500, anti-mouse, Abcam, United States), LHR (1:500, anti-mouse, Boster, China), E receptor (ER) (1:400, anti-mouse, Abcam, United States) and P receptor (PR) (1:200, anti-mouse, Abcam, United States) were used to via immunohistochemistry staining. Finally, positive area analysis was performed using positive cell number expression points and according to the formula and the positive expression rate of the slice: positive rate (%) = positive number of expression/total number of cases × 100%.
Western blotting analysis
After the mice were sacrificed, ovaries from different mice in each group were harvested and stored at −80 °C. Subsequently the protein was extracted using triple detergent lysis buffer, the expression of PI3K (CST, 1:1000), p-PI3K (CST, 1:1000), AKT (CST, 1:1000), p-Akt(CST, 1:2000), mTOR(CST, 1:1000) and p-mTOR(CST, 1:1000) was detected by western-blotting.
Statistical Analysis
GraphPad prism 5 software was used for statistical analysis. Data was presented as the mean ± standard deviation (SD). Statistical analysis of normal distribution was performed on two independent samples. The Mann-Whitney U test was used to measure the non-normal distribution. The data were analysed by Chi-square. P < 0.05 was considered statistically significant. Assays were performed at least three times independently.
Data availability
See ‘Availability of materials and data’ section for more information
Ethical approval
The ethical review committee for animal experiments at the Shanghai Jiaotong University School of Medicine approved the use of mice for this study. Principles of laboratory animal care were followed and all procedures were conducted according to the guidelines established by the National Institutes of Health, and every effort was made to minimize suffering.
Acknowledgements
We thank Dr. Chen Xu for providing laboratory equipment in the Department of Microorganism, Shanghai Jiaotong University School of Medicine. We would like to thank not only the doctors and nurses, but also our patients and their families who assisted with recruitment for this study. This work was supported by grants from the Science and Technology Commission of Shanghai Municipality (Nos 15411953000, 15411964500 and 14411964300); the National Natural Science Foundation of China (No. 81571397) and Merck Serono Reproductive Medicine fund.
Author Contributions
All authors contributed to planning, writing and revising the article. W.R.C. and J.X. conceived and designed the study. X.W.W. performed the experiments, collected data, performed statistical analysis and wrote the paper. Y.P.K. wrote the article and created the figures with the assistance of L.X.Z., B.F.Y., Q.J.C., Y.L.F., Z.Y., Y.F. and H.Y.G. recruited the volunteers and patients. Q.F.L. critically revised the article for important intellectual content.
Competing Interests
The authors declare no competing interests.
Footnotes
Xiaowei Wen and Jun Xie jointly supervised this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Yanping Kuang, Email: kuangyanp@126.com.
Weiran Chai, Email: chaiwrsh@126.com.
References
- 1.Meng QQ, et al. Infertility rate in married couples of reproductive age in China: a systematic review and meta-analysis. Zhonghua Liu Xing Bing Xue Za Zhi. 2013. 2013;34:826–31. [PubMed] [Google Scholar]
- 2.Sills ES, et al. Balancing selected medication costs with total number of daily injections: a preference analysis of GnRH-agonist and antagonist protocols by IVF patients. Reprod Biol Endocrinol. 2012;30(10):67. doi: 10.1186/1477-7827-10-67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Fleming R, et al. A new systematic treatment for infertile women with abnormal hormone profiles. Br J Obstet Gynaecol. 1982;89:80–83. doi: 10.1111/j.1471-0528.1982.tb04642.x. [DOI] [PubMed] [Google Scholar]
- 4.Bosch E, et al. Premature luteinization during gonadotropin-releasing hormone antagonist cycles and its relationship with in vitro fertilization outcome. Fertil Steril. 2003;80:1444–1449. doi: 10.1016/j.fertnstert.2003.07.002. [DOI] [PubMed] [Google Scholar]
- 5.Kuang Y, et al. Medroxyprogesterone acetate is an effective oral alternative for preventing premature luteinizing hormone surges in women undergoing controlled ovarian hyperstimulation for in vitro fertilization. Fertil Steril. 2015;10:62–70. doi: 10.1016/j.fertnstert.2015.03.022. [DOI] [PubMed] [Google Scholar]
- 6.Massin N. New stimulation regimens: endogenous and exogenous progesterone use to block the LH surge during ovarian stimulation for IVF. Hum Reprod Update. 2017;23:211–220. doi: 10.1093/humupd/dmw047. [DOI] [PubMed] [Google Scholar]
- 7.Rizner TL, et al. Selectivity and potency of the retroprogesterone dydrogesterone in vitro. Steroids. 2011;76:607–615. doi: 10.1016/j.steroids.2011.02.043. [DOI] [PubMed] [Google Scholar]
- 8.Christensen AC, Haresign W, Khalid M. Progesterone exposure of seasonally anoestrous ewes alters the expression of angiogenic growth factors in preovulatory follicles. Theriogenology. 2014;81:358–367. doi: 10.1016/j.theriogenology.2013.10.005. [DOI] [PubMed] [Google Scholar]
- 9.Abdel-Ghani MA, Shimizu T, Suzuki H. Expression pattern of vascular endothelial growth factor in canine folliculogenesis and its effect on the growth and development of follicles after ovarian organ culture. Reprod Domest Anim. 2014;49:734–739. doi: 10.1111/rda.12357. [DOI] [PubMed] [Google Scholar]
- 10.Nagahama Y, Adachi S. Identification of maturation-inducing steroid in a teleost, the amago salmon (Oncorhynchus rhodurus) Dev Biol. 1985;109:428–435. doi: 10.1016/0012-1606(85)90469-5. [DOI] [PubMed] [Google Scholar]
- 11.Schreiber JR, Nakamura K, Truscello AM, Erickson GF. Progestins inhibit FSH-induced functional LH receptors in cultured rat granulosa cells. Mol Cell Endocrinol. 1982;25:13–24. doi: 10.1016/0303-7207(82)90174-5. [DOI] [PubMed] [Google Scholar]
- 12.Lu X, et al. Elevated progesterone on the trigger day does not impair the outcome of Human Menotrophins Gonadotrophin and Medroxyprogesterone acetate treatment cycles. Sci Rep. 2016;6:31112. doi: 10.1038/srep31112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Cully M, You H, Levine AJ, Mak TW. Beyond PTEN mutations: the PI3K pathway as an integrator of multiple inputs during tumorigenesis. Nat Rev Cancer. 2006;6:184–192. doi: 10.1038/nrc1819. [DOI] [PubMed] [Google Scholar]
- 14.Dupont J, Reverchon M, Cloix L, Froment P, Ramé C. Involvement of adipokines, AMPK, PI3K and the PPAR signaling pathways in ovarian follicle development and cancer. Int J Dev Biol. 2012;56:959–967. doi: 10.1387/ijdb.120134jd. [DOI] [PubMed] [Google Scholar]
- 15.Reddy P, et al. Oocyte-specific deletion of Pten causes premature activation of the primordial follicle pool. Science. 2008;319:611–613. doi: 10.1126/science.1152257. [DOI] [PubMed] [Google Scholar]
- 16.Fan HY, Liu Z, Cahill N, Richards JS. Targeted disruption of Pten in ovarian granulosa cells enhances ovulation and extends the life span of luteal cells. Mol Endocrinol. 2008;22:2128–2140. doi: 10.1210/me.2008-0095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Carlström K, Pschera H, Lunell NO. Serum levels of oestrogens, progesterone, follicle-stimulating hormone and sex-hormone-binding globulin during simultaneous vaginal administration of 17 beta-oestradiol and progesterone in the pre- and post-menopause. Maturitas. 1988;10:307–316. doi: 10.1016/0378-5122(88)90066-7. [DOI] [PubMed] [Google Scholar]
- 18.Orvieto R, et al. HMG improves IVF outcome in patients with high basal FSH/LH ratio: a preliminary study. Reprod Biomed Online. 2009;18:205–208. doi: 10.1016/S1472-6483(10)60257-X. [DOI] [PubMed] [Google Scholar]
- 19.Evans NP, Dahl GE, Padmanabhan V, Thrun LA, Karsch FJ. Estradiol requirements for induction and maintenance of the gonadotropin-releasing hormone surge: implications for neuroendocrine processing of the estradiol signal. Endocrinology. 1997;138:5408–5014. doi: 10.1210/endo.138.12.5558. [DOI] [PubMed] [Google Scholar]
- 20.Janovick JA, Conn PM. Progesterone diminishes the sensitivity of gonadotropin-releasing hormone-stimulated luteinizing hormone (LH) release and protects an LH pool from desensitization: actions opposed by cholera toxin. Endocrinology. 1996;137:1823–1827. doi: 10.1210/endo.137.5.8612520. [DOI] [PubMed] [Google Scholar]
- 21.Couzinet B, et al. The antigonadotropic activity of a 19-nor-progesterone derivative is exerted both at the hypothalamic and pituitary levels in women. J Clin Endocrinol Metab. 1999;84:4191–4196. doi: 10.1210/jcem.84.11.6162. [DOI] [PubMed] [Google Scholar]
- 22.Heikinheimo O, Gordon K, Williams RF, Hodgen GD. Inhibition of ovulation by progestin analogs (agonists vs antagonists): preliminary evidence for different sites and mechanisms of actions. Contraception. 1996;53:55–64. doi: 10.1016/0010-7824(95)00255-3. [DOI] [PubMed] [Google Scholar]
- 23.Dias LM, et al. Effect of a new device for sustained progesterone release on the progesterone concentration, ovarian follicular diameter, time of ovulation and pregnancy rate of ewes. Anim Reprod Sci. 2015;155:56–63. doi: 10.1016/j.anireprosci.2015.01.016. [DOI] [PubMed] [Google Scholar]
- 24.Adams GP, Matteri RL, Ginther OJ. Effect of progesterone on ovarian follicles, emergence of follicular waves and circulating follicle-stimulating hormone in heifers. J Reprod Fertil. 1992;96:627–640. doi: 10.1530/jrf.0.0960627. [DOI] [PubMed] [Google Scholar]
- 25.Hernández-Coronado CG, et al. Sphingosine-1-phosphate, regulated by FSH and VEGF, stimulates granulosa cell proliferation. Gen Comp Endocrinol. 2016;236:1–8. doi: 10.1016/j.ygcen.2016.06.029. [DOI] [PubMed] [Google Scholar]
- 26.Karakaya C, et al. Follicle-stimulating hormone receptor (FSHR) alternative skipping of exon 2 or 3 affects ovarian response to FSH. Mol Hum Reprod. 2014;20:630–643. doi: 10.1093/molehr/gau024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.La Marca A, et al. The combination of genetic variants of the FSHB and FSHR genes affects serum FSH in women of reproductive age. Hum Reprod. 2013;28:1369–1374. doi: 10.1093/humrep/det061. [DOI] [PubMed] [Google Scholar]
- 28.Cui H, et al. FSH stimulates lipid biosynthesis in chicken adipose tissue by upregulating the expression of its receptor FSHR. J Lipid Res. 2012;53:909–917. doi: 10.1194/jlr.M025403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhang P, et al. In vitro effect of cyclic adenosine 3′, 5′-monophosphate (cAMP) on early human ovarian follicles. J Assist Reprod Genet. 2004;21:301–306. doi: 10.1023/B:JARG.0000043704.10845.87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Karlsson AB, et al. Luteinizing hormone receptor-stimulated progesterone production by preovulatory granulosa cells requires protein kinase A-dependent activation/dephosphorylation of the actin dynamizing protein cofilin. Mol Endocrinol. 2010;24:1765–1781. doi: 10.1210/me.2009-0487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.El-Hefnawy T, Huhtaniemi I. Progesterone can participate in down-regulation of the luteinizing hormone receptor gene expression and function in cultured murine Leydig cells. Mol Cell Endocrinol. 1998;137:127–138. doi: 10.1016/S0303-7207(98)00002-1. [DOI] [PubMed] [Google Scholar]
- 32.Edson MA, Nagaraja AK, Matzuk MM. The mammalian ovary from genesis to revelation. Endocr Rev. 2009;30:624–712. doi: 10.1210/er.2009-0012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Brown C, et al. Subfertility caused by altered follicular development and oocyte growth in female mice lacking PKB alpha/Akt1. Biol Reprod. 2010;82:246–256. doi: 10.1095/biolreprod.109.077925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Sun X, et al. New strategy for in vitro activation of primordial follicles with mTOR and PI3K stimulators. Cell Cycle. 2015;14:721–731. doi: 10.1080/15384101.2014.995496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yang XL, Lin FJ, Guo YJ, Shao ZM, Ou ZL. Gemcitabine resistance in breast cancer cells regulated by PI3K/AKT-mediated cellular proliferation exerts negative feedback via the MEK/MAPK and mTOR pathways. Onco Targets Ther. 2014;7:1033–1942. doi: 10.2147/OTT.S63145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sobinoff AP, Mahony M, Nixon B, Roman SD, McLaughlin EA. Understanding the Villain: DMBA-induced preantral ovotoxicity involves selective follicular destruction and primordial follicle activation through PI3K/Akt and mTOR signaling. Toxicol Sci. 2011;123:563–575. doi: 10.1093/toxsci/kfr195. [DOI] [PubMed] [Google Scholar]
- 37.Song T. et al. Expression of p-Akt in ovarian serous carcinoma and its association with proliferation and apoptosis. 7, 59–64 (2013). [DOI] [PMC free article] [PubMed]
- 38.Alam H, et al. Follicle-stimulating hormone activation of hypoxia-inducible factor-1 by the phosphatidylinositol 3-kinase/AKT/Ras homolog enriched in brain (Rheb)/mammalian target of rapamycin (mTOR) pathway is necessary for induction of select protein markers of follicular differentiation. J Biol Chem. 2004;279:19431–19440. doi: 10.1074/jbc.M401235200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Xu CL, Lu XL, Yan XN, Wang HL, Chen SQ. Effects of PI3K/Akt/NF-κB signal pathway on FSH facilitation on cell proliferation and invasion by human epithelial ovarian cancer. Zhonghua Fu Chan Ke Za Zhi. 2012;47:134–138. [PubMed] [Google Scholar]
- 40.Chen YJ, et al. Interplay of PI3K and cAMP/PKA signaling, and rapamycin-hypersensitivity in TGFbeta1 enhancement of FSH-stimulated steroidogenesis in rat ovarian granulosa cells. J Endocrinol. 2007;192:405–419. doi: 10.1677/JOE-06-0076. [DOI] [PubMed] [Google Scholar]
- 41.Myers M, Britt KL, Wreford NG, Ebling FJ, Kerr JB. Methods for quantifying follicular numbers within the mouse ovary. Reproduction. 2004;127:569–580. doi: 10.1530/rep.1.00095. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
See ‘Availability of materials and data’ section for more information