Abstract
Background
Neopterin, a metabolite of GTP, is produced by activated macrophages and is abundantly expressed within atherosclerotic lesions in human aorta and carotid and coronary arteries. We aimed to clarify the influence of neopterin on both vascular inflammation and atherosclerosis, as neither effect had been fully assessed.
Methods and Results
We investigated neopterin expression in coronary artery lesions and plasma from patients with coronary artery disease. We assessed the atheroprotective effects of neopterin in vitro using human aortic endothelial cells, human monocyte‐derived macrophages, and human aortic smooth muscle cells. In vivo experiments included a study of aortic lesions in apolipoprotein E–deficient mice. Neopterin expression in coronary artery lesions and plasma was markedly increased in patients with versus without coronary artery disease. In human aortic endothelial cells, neopterin reduced proliferation and TNF‐α (tumor necrosis factor α)–induced upregulation of MCP‐1 (monocyte chemotactic protein 1), ICAM‐1 (intercellular adhesion molecule 1), and VCAM‐1 (vascular cell adhesion molecule 1). Neopterin attenuated TNF‐α–induced monocyte adhesion to human aortic endothelial cells and the inflammatory macrophage phenotype via NF‐κB (nuclear factor‐κB) downregulation. Neopterin suppressed oxidized low‐density lipoprotein–induced foam cell formation associated with CD36 downregulation and upregulation of ATP‐binding cassette transporters A1 and G1 in human monocyte‐derived macrophages. In human aortic smooth muscle cells, neopterin suppressed angiotensin II–induced migration and proliferation via c‐Src/Raf‐1/ERK1/2 downregulation without inducing apoptosis. Exogenous neopterin administration and endogenous neopterin attenuation with its neutralizing antibody for 4 weeks retarded and promoted, respectively, the development of aortic atherosclerotic lesions in apolipoprotein E–deficient mice.
Conclusions
Our results indicate that neopterin prevents both vascular inflammation and atherosclerosis and may be induced to counteract the progression of atherosclerotic lesions. Consequently, neopterin could be of use as a novel therapeutic target for atherosclerotic cardiovascular diseases.
Keywords: atherosclerosis, endothelial cell, inflammation, macrophage, neopterin, smooth muscle cell
Subject Categories: Atherosclerosis, Coronary Artery Disease, Vascular Biology, Vascular Disease
Clinical Perspective
What Is New?
Neopterin, a GTP metabolite, is produced by activated macrophages and has been regarded as a proinflammatory and proatherosclerotic agent.
Our study challenges this notion, with clear demonstration of neopterin's inhibitory effects toward vascular inflammation and atherosclerosis in in vitro and in vivo experiments.
Neopterin prevents atherogenesis by suppressing inflammatory responses in endothelial cells and macrophages, monocyte–endothelial cell adhesion, macrophage foam cell formation, and the migration and proliferation of vascular smooth muscle cells.
Neopterin increases in coronary lesions and circulating blood in patients with coronary artery disease to counter inflammation and atherosclerosis.
What Are the Clinical Implications?
Neopterin is expected to emerge as a new line of therapy against inflammatory and atherosclerotic cardiovascular diseases such as coronary artery disease, peripheral artery disease, aortitis, aneurysm, and diabetic angiopathy.
Our study may strengthen the clinical utility of neopterin in the diagnosis and treatment of coronary artery disease.
Introduction
Atherosclerosis is a chronic inflammatory response to injury of the arterial wall.1 Endothelial inflammation stimulates the production of proinflammatory cytokines and adhesion molecules such as IL‐6 (interleukin 6), MCP‐1 (monocyte chemotactic protein 1), ICAM‐1 (intercellular adhesion molecule 1), VCAM‐1 (vascular cell adhesion molecule 1), and E‐selectin in endothelial cells (ECs). These effectors encourage monocyte and T‐cell adhesion and infiltration into the neointima lesion, followed by secretion of proinflammatory cytokines such as TNF‐α (tumor necrosis factor α) and INF‐γ (interferon γ) from these cells. Monocyte‐derived macrophages engulf oxidized low‐density lipoprotein (oxLDL) to become foam cells, which are characteristic features of the fatty streak.1, 2 Foam cell formation is characterized by the intracytoplasmic accumulation of cholesterol ester, which depends on the balance between the uptake of oxLDL via CD36 and the efflux of free cholesterol controlled by the ATP‐binding cassette transporters ABCA1 and ABCG1 or SR‐BI (scavenger receptor class B type I).2 The accumulation of cholesterol ester is also regulated by the balance between cholesterol esterification by ACAT‐1 (acyl‐CoA acyltransferase 1) and its hydrolysis by NCEH (neutral cholesterol ester hydrolase).2, 3 The categorization of macrophage phenotype as either pro‐ or anti‐inflammatory (M1 and M2 phenotypes, respectively) has recently focused on foam cell formation and atherosclerosis.4 Other than the accumulation of macrophage foam cells, the migration and proliferation of vascular smooth muscle cells (VSMCs), EC proliferation, and the production of extracellular matrix components such as the collagens, matrix metalloproteinases (MMPs), tissue inhibitor of metalloproteinases (TIMPs), fibronectin, and elastin, all contribute to the development of the atherosclerotic plaque.1, 5
Neopterin, a metabolite of GTP, is produced by activated macrophages following their stimulation by IFN‐γ released by T cells.6, 7 Neopterin is also known as an activation marker for monocytes/macrophages.8 The specific receptors for neopterin have yet to be identified. Previous studies have reported that neopterin has antioxidant properties.9 Its plasma concentration is elevated in patients with coronary artery disease (CAD),10 with neopterin abundantly expressed within atherosclerotic lesions in human coronary and carotid arteries and aorta.11, 12, 13 A previous report showed that neopterin suppressed the expression of ABCA1 and ABCG1 and reduced cholesterol efflux to apolipoprotein A1 and high‐density lipoprotein (HDL) via the LXR‐α (liver X receptor α) pathway in human acute monocytic leukemia THP‐1 cell–derived macrophage foam cells.14 However, the precise effects of neopterin on atherogenesis have yet to be described.
In this study, we aimed to clarify the effects of neopterin on vascular inflammation and atherosclerosis. We initially investigated neopterin expression in the circulating blood and in coronary atherosclerotic lesions in CAD patients. We assessed the effects of neopterin, in vitro, on the inflammatory response, proliferation, and monocyte adhesion in human ECs and the inflammatory phenotype and foam cell formation using human monocyte‐derived macrophages (HMDMs), as well as migration, proliferation, and extracellular matrix production in human aortic smooth muscle cells (HASMCs). In vivo studies focused on the development of atherosclerotic lesions in apolipoprotein E–deficient (Apoe −/−) mice.
Methods
The data that support the findings of this study are available to other researchers on request. All materials used in this study are available commercially from the indicated vendors.
Materials
Neopterin and polyclonal rabbit anti–neopterin antibody were purchased from LKT Laboratories (St. Paul, MN) and My Biosource (San Diego, CA), respectively, for all experiments. The neopterin ELISA kit was purchased from Immuno‐Biological Laboratories (Hamburg, Germany). The ELISA kit shows 100% immunoreactivity with neopterin, 2.0% 7,8‐dihydro‐neopterin, and <0.44% 5,6,7,8‐tetrahydro‐neopterin. The limit of detectable concentration of neopterin is 0.7 nmol/L. Intra‐ and interassay coefficients of variation are 4.3% to 11.7% and 8.8% to 13.8%, respectively.
Collection of Human Blood and Coronary Atherosclerotic Lesions
This study was conducted according to the principles of the Declaration of Helsinki and received prior approval from the ethics committees of Showa University and Tokyo University of Pharmacy and Life Sciences. Written informed consent was obtained from 135 participants before enrollment. Blood was drawn from 92 patients with angiographically proven CAD who were hospitalized at Showa University Hospital (64 men, 28 women; aged 40–82 years), including 22 with unstable angina pectoris, 67 with acute myocardial infarction, 2 with recent myocardial infarction, and 1 with old myocardial infarction; in addition, 43 age‐matched non‐CAD patients (17 men, 26 women; aged 50–82 years) with mild hypertension, diabetes mellitus, and dyslipidemia were recruited because plasma neopterin levels are known to correlate with age.15 Exclusion criteria (confirmed by medical history and routine laboratory examination) included patients with infection and inflammatory disease, as both increase plasma neopterin levels.16 Plasma neopterin levels were measured using the neopterin ELISA kit.
Buffered 10% formalin‐fixed paraffin‐embedded human coronary artery specimens from archive collections of the Japanese National Cerebral and Cardiovascular Center were used for immunohistochemistry. Serial cross‐sections (3–4 μm) of stenotic and nonstenotic coronary arteries from 12 patients with a history of myocardial infarction (8 men, 4 women; aged 60–87 years) and normal coronary arteries from 4 patients (4 men; aged 19–39 years) with dilated cardiomyopathy (as non‐CAD examples) were stained with polyclonal rabbit anti–neopterin antibody.17, 18, 19 Control IgG staining was also performed to exclude nonspecific staining (Figure S1).
Human Monocyte Primary Culture
Human peripheral mononuclear cells were isolated from the blood of 19 healthy volunteers (7 men, 12 women; aged 19–24 years). Ethics approval was received from the Tokyo University of Pharmacy and Life Sciences, and written informed consent was obtained from all participants before blood draws. Monocytes were purified using anti‐CD14 antibody‐conjugated magnetic microbeads. Monocytes were subsequently seeded into 3.5‐cm dishes (1×106 cells/1 mL per dish) for cholesterol esterification assays and immunoblots.17, 18, 19, 20, 21, 22, 23, 24, 25 Cells were incubated at 37°C in a 5% CO2 humidified incubator for 7 days in RPMI‐1640 medium supplemented with 10% human serum, 0.05 mg/mL streptomycin, 50 U/mL penicillin, and the indicated concentrations of neopterin. Media was changed every 3 days.
Cholesterol Esterification Assay
HMDMs were differentiated by 7‐day culture with human serum and the indicated concentrations of neopterin, with incubation for another 19 hours at 37°C in 5% CO2 with the same concentrations of neopterin, together with 50 μg/mL oxLDL in the presence of 100 μmol/L [3H]oleate conjugated with BSA.17, 18, 19, 20, 21, 22, 23 Cellular lipids were extracted, and the radioactivity attributed to cholesterol‐[3H]oleate was determined by thin‐layer chromatography.
Cholesterol Efflux Assay
HMDMs differentiated by 7‐day culture with human serum and the indicated concentrations of neopterin were incubated for 24 hours at 37°C in 5% CO2 with 50 μg/mL oxLDL prelabeled with 74 kBq/mL [3H]cholesterol in RPMI‐1640 medium. [3H]Cholesterol‐labeled macrophages were incubated for 2 hours with serum‐free RPMI‐1640 medium containing 0.1% BSA, and then efflux was initiated by the addition of serum‐free RPMI‐1640 medium containing 0.1% BSA with 50 μg/mL apolipoprotein A1 or HDL.25 After 16‐hour incubation, the medium was collected and centrifuged at 18 000 g for 10 minutes to remove cell debris. Cells were washed twice with PBS and then lysed with 0.3 mL of 1 N NaOH. Radioactivity attributed to [3H]cholesterol in media and cell lysates was measured by liquid scintillation counting. Cholesterol efflux was calculated, as described previously.25
Reverse Transcription–Polymerase Chain Reaction
Human aortic ECs (HAECs) and human umbilical vein ECs (HUVECs) were incubated at 37°C in 5% CO2 for 30 minutes with or without 200 nmol/L neopterin in HuMedia‐EG2 or EGM‐2, respectively. Neopterin and 10 ng/mL TNF‐α were then added to the media for another 4 hours.11 The mRNAs for IL‐6, MCP‐1, ICAM‐1, VCAM‐1, E‐selectin, and GAPDH were detected, as described previously.18, 19, 20, 21, 23
Monocyte Adhesion Assay
Confluent HAECs or HUVECs cultured in 24‐well plates were incubated at 37°C in a 5% CO2 gassed incubator for 16 hours with HuMedia‐EG2 or EGM‐2, respectively, and then pretreated for 30 minutes with or without the indicated concentrations of neopterin, followed by 4‐hour incubation in the presence or absence of 10 ng/mL TNF‐α. Subsequently, THP‐1 cells were labeled with CellTrace calcein red‐orange, with a total of 1×105 cells added to each well of HAEC‐ or HUVEC‐seeded 24‐well plates. After 1 hour, THP‐1 cells bound to HAECs or HUVECs were washed 4 times and then examined by fluorescence microscopy (IX70; Olympus). Their adhesion was assessed using image analysis software (ImageJ; US National Institutes of Health).21, 26
Migration Assay
HASMCs at passages 6 to 8 were seeded into 8‐well culture slides (3×103 cells/250 μL per well). Cells were incubated at 37°C in 5% CO2 for 3 to 5 hours in SmGM‐2. HASMCs were then incubated for 30 minutes with the indicated concentrations of neopterin in serum‐free SmGM‐2, and then for another 16 hours with an indicated concentration of angiotensin II and/or neopterin in the absence or presence of anti–neopterin antibody (25 μL/mL). Cells were photographed at 10‐minute intervals. The average migration distance of 10 cells randomly selected in each well was measured using a time‐lapse cell culture observation system (BIOREVO BZ‐9000 microscope; Keyence).17, 18, 19, 20, 21, 22, 23
Proliferation (Viability) Assay
HAECs, HUVECs, or HASMCs at passages 2 to 8 were seeded into 96‐well plates (1×104 cells/100 μL per well) and incubated for 24 hours in the same media (HuMedia‐EG2, EGM‐2, or SmGM‐2, respectively). Cells were then incubated for another 48 hours in fresh media with the indicated concentrations of neopterin in the absence or presence of anti–neopterin antibody (10 μL/mL) or specific inhibitors of c‐Src (0.1 μmol/L saracatinib) and MEK1 (MAPK/ERK kinase 1) or MEK2 (10 μmol/L SL327). Next, 10µL WST‐8 solution (Cell Count Reagent SF; Nacalai Tesque) was then added to each well.2, 3, 4, 5, 6, 7, 8 After 1 hour of incubation, the quantity of formazan product was determined by reading absorbance at 450 nm using a Sunrise Remote R microplate reader (Tecan).17, 18, 19, 20, 21, 22, 23
Apoptosis Assay
HASMCs were seeded into 12‐well plates (3×105 cells/1 mL per well) and incubated at 37°C in 5% CO2 gassed incubator for 24 hours in the same conditioning medium, followed by 48‐hour incubation with the indicated concentrations of neopterin. Cells were fixed with 4% paraformaldehyde in PBS. TUNEL (terminal deoxynucleotidyl transferase–mediated dUTP‐biotin nick‐end labeling) staining was then performed using an In Situ Apoptosis Detection Kit (Takara Bio), as described previously.20, 21 Nuclei were costained using DAPI (4′,6‐diamidino‐2‐phenylindole). The numbers of TUNEL‐positive cells were counted in 3 fields of view chosen randomly from each sample.
Western Blotting
Aliquots of protein extracts (20 μg) derived from HMDMs, HAECs, HUVECs, and HASMCs were separated by 10% SDS‐PAGE and then immunoblotted with specific antibodies raised against the following proteins: neopterin, ACAT‐1, CD68, E‐selectin, CD36, NCEH, ABCA1, ABCG1, SR‐BI, collagen 1, collagen 3, fibronectin, phosphorylated NF‐κB (nuclear factor‐κB) p65 (Ser529), α‐tubulin, ICAM‐1, MMP‐2, MMP‐9, TIMP‐2, elastin, MARCO (macrophage receptor with collagenous structure), MRC‐1, PPAR‐γ (peroxisome proliferator‐activated receptor γ), phosphorylated ERK1 (extracellular signal‐regulated kinase 1) and ERK2 (Thr202/Tyr204), phosphorylated Akt (protein kinase B; serine/threonine), c‐Src, SMemb (nonmuscle myosin heavy chain B), Raf‐1, LXR‐α, PI3K (phosphoinositide 3‐kinase), Bcl‐2, Bax (Bcl‐2 associated X protein), VCAM‐1, and β‐actin.17, 18, 19, 20, 21, 22, 23
PAGE Zymography
The activities of MMP‐2 and MMP‐9 in culture supernatants of HASMCs incubated for 48 hours in serum‐free SmGM‐2 with the indicated concentrations of neopterin were determined using a gelatin‐zymography kit, as described previously.18, 20, 21
Animal Experiments
Animal experiments were performed in accordance with the Guide for the Care and Use of Laboratory Animals published by the US National Research Council, with protocols approved by the institutional animal care and use committee of Tokyo University of Pharmacy and Life Sciences. A total of 37 male spontaneously hyperlipidemic Apoe −/− mice in 2 strains, C57BL/6 (KOR/StmSlc‐Apoe shl mice) and BALB/c (KOR/StmSlc‐Apoe shl mice), were purchased from Japan SLC (Hamamatsu, Japan). Mice were fed a high‐cholesterol diet containing 16.5% fat, 1.25% cholesterol, and 0.5% sodium cholate, starting at 13 weeks of age.17, 18, 20, 21, 22 Experiment 1 was performed to evaluate the effects of exogenous neopterin administration on atherogenesis in 26 Apoe −/− mice (C57BL/6). At 17 weeks of age, 10 mice were injected intraperitoneally with vehicle and 8 mice were injected intraperitoneally with neopterin (1.2 μmol/kg per day), daily for 4 weeks. Doses of neopterin were selected on the basis of previous data.27, 28, 29, 30 The remaining 8 mice were euthanized at 17 weeks of age as preinjection controls. Experiment 2 was performed to evaluate the effects of endogenous neopterin attenuation on atherogenesis in 11 Apoe −/− mice (BALB/c). At 17 weeks of age, 6 mice were infused with vehicle and 5 mice were infused with anti–neopterin antibody (2.5 μL/kg per hour), using osmotic minipumps, for 4 weeks. Pumps were implanted subcutaneously into the dorsum and replaced once every 2 weeks under medetomidine–midazolam–butorphanol anesthesia.
Animal Measurements
Four weeks after commencing administration into Apoe −/− mice, systolic and diastolic blood pressures were measured using the indirect tail‐cuff method. Blood samples were collected after a 4‐hour fast. Plasma levels of glucose and total cholesterol were measured by enzymatic methods, and HDL cholesterol was measured by a precipitation method.17, 18, 20, 21, 22 Plasma levels of neopterin and PTX‐3 (pentraxin 3) were measured by ELISA.
Assessment of Atherosclerotic Lesions
Before and 4 weeks after commencing administration, Apoe −/− mice were euthanized by exsanguination (total blood collection) under deep anesthesia.17, 18, 20, 21, 22 The whole aorta was washed by perfusion with PBS and fixed with 4% paraformaldehyde. The aorta was then excised from the aortic root to the abdominal area, and connective and adipose tissues were carefully removed. The entire aorta and cross‐sections of the aortic root were stained with Oil Red O for the assessment of atherosclerotic lesions.2, 3, 5, 6, 7 The immunohistochemical expression of neopterin, IFN‐γ, MCP‐1, and PTX‐3 was detected by specific antibodies raised against neopterin, IFN‐γ, MCP‐1, and PTX‐3, respectively. ECs, monocytes/macrophages, CD4+ T cells, CD8+ T cells, and VSMCs in atheromatous plaques were visualized by staining with specific antibodies raised against podocalyxin, MOMA‐2 (monocyte/macrophage‐2), CD4, CD8, and α‐smooth muscle actin, respectively.17, 18, 20, 21, 22 Control IgG stainings were also performed to exclude any nonspecific stainings within atheromatous plaques (Figure S2). These areas of the aortic wall were traced by an investigator blind to the treatment and then quantified by image analysis (Photoshop [Adobe] and ImageJ). In addition, high levels of PTX‐3 expression and the increased ratio of macrophage contents (μm2)/VSMC contents (μm2) within the atheromatous plaques were regarded as indexes of plaque instability.21, 31
Statistical Analyses
Normality was tested with the Kolmogorov–Smirnov test. Normally distributed data are presented as mean±SEM and nonnormally distributed data as median (25th, 75th percentiles). Comparisons between 2 groups were made using the unpaired Student t test or nonparametric Mann–Whitney U test if data were not normally distributed. Multiple comparisons were made among ≥3 groups using 1‐way ANOVA followed by the Bonferroni post hoc test. Categorical variables are presented as frequencies and were analyzed using the Fisher exact test. Statistical analyses were performed using Statview‐J 5.0 (SAS Institute). A value of P<0.05 was considered to be statistically significant.
Results
Expression of Neopterin in Coronary Artery Lesions and Plasma in Non‐CAD and CAD Patients
Neopterin expression levels were assessed in human coronary arteries from 4 non‐CAD and 12 CAD patients. Little expression of neopterin was observed in normal and nonstenotic coronary arteries in a non‐CAD patient and a stable CAD patient, respectively (Figure 1A and 1B). In the coronary arteries from stable and unstable CAD patients, neopterin was expressed at high levels in macrophage foam cells within fatty streaks and both early and advanced atherosclerotic plaques (Figure 1C and 1D). The degree of neopterin expression became markedly higher in accordance with the progression of atherosclerostic plaques.
Figure 1.

Expression of neopterin in coronary atherosclerotic lesions and its plasma levels in patients with CAD. Coronary artery staining with an anti–neopterin antibody is shown in representative cases of (A) a non‐CAD patient (male, aged 31 years) and (B through D) CAD patients: (B) female, aged 83 years; (C) male, aged 70 years; (D) male, aged 62 years. Coronary arteries were not the culprit artery in acute coronary events. Scale bar=500 μm. E, Box plot graph of plasma neopterin levels measured by ELISA in 43 non‐CAD and 92 CAD patients. Values shown are median (center rule in box) with 25th percentile (lower rule of box), 75th percentile (upper rule in box), 10th percentile (lower short bars), and 90th percentile (upper short bars). *P<0.05. ACS indicates acute coronary syndrome; CAD, coronary arteries disease; DCM, dilated cardiomyopathy; OMI, old myocardial infarction.
Next, plasma neopterin levels were compared for 43 non‐CAD and 92 CAD patients. As listed in Table 1, sex (male ratio) and the prevalence of smoking and diabetes mellitus was significantly greater and the prevalence of hypertension, dyslipidemia, and angiotensin II receptor blocker use was significantly lower in CAD versus non‐CAD patients. Plasma HDL‐cholesterol levels were significantly lower and plasma levels of high‐sensitivity C‐reactive protein were significantly greater in CAD versus non‐CAD patients (Table 1). Age; the use of antidiabetic drugs, angiotensin‐converting enzyme inhibitors, calcium‐channel blockers, β‐blockers, statins, and fibrates; and plasma low‐density lipoprotein–cholesterol levels did not differ significantly between CAD and non‐CAD patients(Table 1). As shown in Figure 1E, plasma neopterin levels were significantly elevated in CAD versus non‐CAD patients (12.3±1.0 versus 9.7±0.6, P<0.05). Among CAD patients, plasma neopterin levels tended to be higher in those with acute coronary syndrome compared with those with recent and prior myocardial infarction (12.4±1.0 versus 8.4±1.5 nmol/L, P=0.4620).
Table 1.
Patient Characteristics
| Non‐CAD Patients (n=43) | CAD Patients (n=92) | |
|---|---|---|
| Age, y | 64.3±1.3 | 67.6±1.1 |
| Male, % | 39.5 | 69.6* |
| Smoking, % | 25.6 | 69.6† |
| Diabetes mellitus, % | 11.6 | 28.3‡ |
| Hypertension, % | 100.0 | 66.3† |
| Dyslipidemia, % | 51.2 | 30.4‡ |
| Antidiabetic drugs, % | 7.0 | 14.1 |
| ARBs, % | 62.8 | 32.6* |
| ACE inhibitors, % | 2.3 | 3.3 |
| Calcium‐channel blockers, % | 51.2 | 33.7 |
| β‐blockers, % | 9.3 | 9.8 |
| Statins, % | 32.6 | 23.9 |
| Fibrates, % | 11.6 | 7.6 |
| LDL cholesterol, mg/dL | 122.2±4.8 | 118.2±3.8 |
| HDL cholesterol, mg/dL | 59.3±2.3 | 45.0±1.3† |
| hs‐CRP, mg/dL | 0.1±0.0 | 1.2±0.3§ |
Values are mean±SEM or %. ACE indicates angiotensin‐converting enzyme; ARB, angiotensin II receptor blocker; CAD, coronary artery disease; HDL, high‐density lipoprotein; hs‐CRP, high‐sensitivity C‐reactive protein; LDL, low‐density lipoprotein.
*P<0.005, † P<0.0001, ‡ P<0.05, § P<0.01 vs non‐CAD patients.
Effects of Neopterin on Proliferation and Inflammatory Response in Human ECs
Neopterin significantly suppressed the proliferation of HAECs, with a maximal effect observed at 1 nmol/L (Figure 2A). Neopterin had no effect on mRNA expression for IL‐6, MCP‐1, ICAM‐1, VCAM‐1, or E‐selectin, unlike TNF‐α (10 ng/mL), which significantly enhanced their expression levels (Figure 2B). However, neopterin at 200 nmol/L significantly suppressed TNF‐α–induced mRNA expression of MCP‐1, ICAM‐1, and VCAM‐1 but not IL‐6 or E‐selectin (Figure 2B). Immunoblots revealed that treatment with TNF‐α (10 ng/mL) significantly increased expression at the protein level of ICAM‐1, VCAM‐1, and E‐selectin, as well as NF‐κB phosphorylation in HAECs, with these effects of ICAM‐1, VCAM‐1, and NF‐κB, but not E‐selectin, significantly suppressed by the addition of neopterin (Figure 2C through 2F). The alterations identified at the protein level were consistent with changes in mRNA expression (Figure 2B).
Figure 2.

Effects of neopterin on proliferation, inflammatory response, and monocyte adhesion in HAECs. A, HAEC proliferation was determined by WST‐8 assay after 48‐h incubation in HuMedia‐EG2 supplemented with 5% FBS and the indicated concentrations of neopterin (n=5). *P<0.005, † P<0.05 vs 0 nmol/L of neopterin. B, mRNA expression of IL‐6, MCP‐1, ICAM‐1, VCAM‐1, and E‐selectin as analyzed by reverse transcription–polymerase chain reaction. HAECs were pretreated with or without neopterin (200 nmol/L) for 30 minutes and then incubated with neopterin (200 nmol/L) plus TNF‐α (10 ng/mL) for 4 h. Representative images are shown; the graph at the right side indicates densitometry data following normalization relative to GAPDH (n=5). *P<0.005, † P<0.05, ‡ P<0.0001 vs corresponding control; § P<0.05 vs corresponding TNF‐α. C through F, HAECs treated as described above were harvested and subject to immunoblot to evaluate ICAM‐1, VCAM‐1, E‐selectin, and NF‐κB protein expression (n=5). Upper panels show representative immunoblots with densitometry data after normalization relative to GAPDH shown beneath. G, Confluent HAECs were incubated in HuMedia‐EG2 supplemented with 0.5% FBS for 16 h and then pretreated for 30 minutes with the indicated concentrations of neopterin, followed by 4‐h incubation in the presence or absence of TNF‐α (10 ng/mL). Subsequently, calcein red‐orange–labeled THP‐1 monocytes were plated on the HAEC monolayer and incubated for 1 h. After washing, the adherent cells were observed by fluorescence microscopy (n=5). Baseline (1‐fold)=30,755.4±3828.2 pixels. C through G, *P<0.005, ‡ P<0.0001 vs 0 nmol/L of neopterin; § P<0.05, || P<0.005, # P<0.01, ¶ P<0.001, **P<0.0001 vs 10 ng/mL of TNF‐α. FBS indicates fetal bovine serum; HAEC, human aortic endothelial cell; ICAM‐1, intercellular adhesion molecule 1; IL‐6, interleukin 6; MCP‐1, monocyte chemotactic protein 1; NF‐κB, nuclear factor‐κB; TNF‐α, tumor necrosis factor α; VCAM‐1, vascular cell adhesion molecule 1.
The same results applied to HUVECs are shown in Figure S3A through S3F. These findings indicate that neopterin may downregulate proinflammatory cytokines and adhesion molecules associated with suppression of NF‐κB phosphorylation.
Effects of Neopterin on Human Monocyte Adhesion to Human ECs
Exposure of HAECs to TNF‐α (10 ng/mL) for 4 hours resulted in a 98‐fold increase in THP‐1 cell adhesion compared with untreated controls (Figure 2G). This effect was significantly reduced by neopterin at 10 to 200 nmol/L, with maximal reduction by 85% at 200 nmol/L (Figure 2G).
Likewise, neopterin significantly suppressed TNF‐α–stimulated adhesion of THP‐1 cells to HUVECs at 10 to 200 nmol/L (Figure S3G).
Effects of Neopterin on Inflammatory Phenotypes in HMDMs
After 3 to 6 days of primary culture, the differentiation of human monocytes into macrophages was confirmed by increased protein expression of CD68, a macrophage differentiation marker (Figure 3A). Neopterin did not affect monocyte differentiation into macrophages; however, neopterin significantly decreased the protein expression of MARCO, an M1 marker, and increased that of MRC‐1, an M2 marker, on day 3 and 6 (Figure 3A). Likewise, neopterin suppressed NF‐κB phosphorylation and increased PPAR‐γ expression on the corresponding day (Figure 3A).
Figure 3.
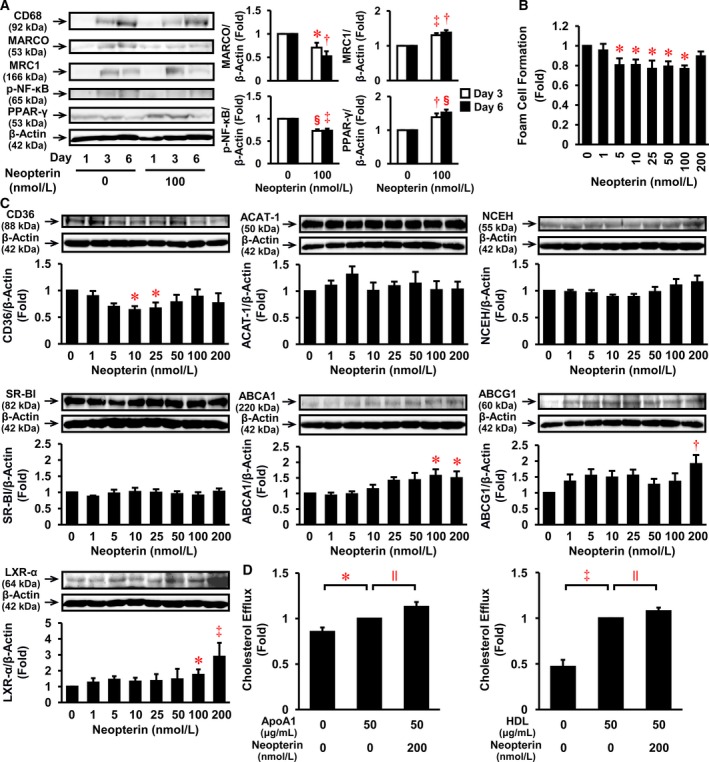
Effects of neopterin on inflammatory phenotype, foam cell formation, cholesterol efflux, and relevant protein expression in HMDMs. A, Human monocytes were incubated for the indicated times with or without neopterin (100 nmol/L). Cells were harvested and subject to immunoblot for CD68 (a macrophage differentiation marker; n=5), MARCO (an M1 macrophage marker; n=4), MRC‐1 (an M2 macrophage marker; n=5), p‐NF‐κB (n=5), PPAR‐γ (n=5), or β‐actin (n=5). The graphs show the expressions of MARCO, MRC1, p‐NF‐κB, and PPAR‐γ on day 3 and 6. B, Human monocytes were incubated for 7 days with the indicated concentrations of neopterin (0–200 nmol/L), followed by a 19‐h incubation with 50 μg/mL oxLDL in the presence of 100 μmol/L [3H]oleate. Foam cell formation was determined from reads of the intracellular radioactivity of cholesterol‐[3H]oleate (n=5). Baseline of control=3.97±0.53 nmol/mg cell protein. C, Before the addition of oxLDL, HMDMs were harvested for immunoblot analyses of CD36, ACAT‐1, NCEH, SR‐BI, ABCA1, ABCG1, LXR‐α, or β‐actin (n=6). Upper panels show representative immunoblots, with densitometry data following normalization to β‐actin beneath. A through C, *P<0.05, † P<0.005, ‡ P<0.0005, § P<0.0001 vs 0 nmol/L of neopterin. D, HMDMs were incubated for 24 h with oxLDL (50 μg/mL) labeled with [3H]cholesterol, and for further 16 h with ApoA1 (50 μg/mL) or HDL (50 μg/mL) in serum‐free medium. Radioactivity attributed to [3H]cholesterol was measured in both the cell media and lysate (n=4 for ApoA1, n=3 for HDL). ApoA1 or HDL‐mediated efflux in the absence of neopterin was normalized to 1‐fold. Baseline (1‐fold)=16.5±3.2% (ApoA1) and =33.8±4.9% (HDL). *P<0.05, ‡ P<0.0005, || P<0.05. ABCA1 indicates ATP‐binding cassette transporter A1; ABCG1, ATP‐binding cassette transporters G1; ACAT‐1, acyl‐CoA acyltransferase‐1; ApoA1, apolipoprotein A1; HDL, high‐density lipoprotein; HMDM, human monocyte‐derived macrophage; oxLDL, oxidized low‐density lipoprotein; LXR‐α, liver X receptor α; NCEH, neutral cholesterol ester hydrolase; p‐NF‐κB, phosphorylated nuclear factor‐κB; PPAR‐γ, peroxisome proliferator‐activated receptor γ; SR‐BI, scavenger receptor class B type I.
Effects of Neopterin on Foam Cell Formation, Cholesterol Efflux, and Relevant Protein Expression in HMDMs
Neopterin significantly suppressed oxLDL‐induced foam cell formation at 5 to 100 nmol/L, with maximal reduction by 23% at 25 and 100 nmol/L in HMDMs (Figure 3B). As shown in Figure 3C, neopterin at 10 and 25 nmol/L significantly reduced CD36 expression by 37% and 33%, respectively. In addition, neopterin significantly increased ABCA1 expression by 1.6‐ and 1.5‐fold at 100 and 200 nmol/L, respectively. The latter concentration (200 nmol/L) also induced a 1.9‐fold increase in ABCG1 expression (Figure 3C). Neopterin significantly increased LXR‐α expression by ≈2.9‐fold at 200 nmol/L, although no significant effects were identified for ACAT‐1, NCEH, or SR‐BI expression (Figure 3C). In addition, we assessed the effects of neopterin on cholesterol efflux in HMDMs to apolipoprotein A1 (ABCA1 function) and HDL (ABCG1 function). Exposure to neopterin at 200 nmol/L significantly increased cholesterol efflux to both apolipoprotein A1 and HDL, with increases of 13% and 8%, respectively (Figure 3D).
Effects of Neopterin on Migration, Proliferation, Apoptosis, and Relevant Signal Transduction in HASMCs
Treatment with angiotensin II (500 nmol/L) significantly increased HASMC migration (Figure 4A), with this effect suppressed by neopterin at 10 and 100 nmol/L (Figure 4A). The anti–neopterin antibody significantly canceled neopterin's effect (Figure 4B). Neopterin at 50 to 200 nmol/L significantly suppressed HASMC proliferation (Figure 4C). The suppressive effect of neopterin (200 nmol/L) on HASMC proliferation was significantly canceled by anti–neopterin antibody, a c‐Src inhibitor saracatinib, and a MEK1/2 inhibitor SL327 (Figure 4D). However, neopterin's significant effect on apoptosis was not evident at 200 nmol/L (Figure 4E).
Figure 4.

Effects of neopterin on migration, proliferation, apoptosis, and relevant signal transduction in HASMCs. A and B, Migration was determined in 10 cells per plate using a BIOREVO BZ‐9000 microscope in serum‐free SmGM‐2, with the indicated concentrations of AngII and/or neopterin in the absence or presence of anti–neopterin Ab (25 μL/mL). Independent experimental replicates were performed (n=4 for A, n=7 for B). *P<0.01, † P<0.0005, ‡ P<0.0001, § P<0.05. C and D, The proliferation of HASMCs was determined by WST‐8 assay following 48‐h incubation in SmGM‐2 supplemented with 5% FBS and the indicated concentrations of neopterin in the absence or presence of anti–neopterin Ab (10 μL/mL), c‐Src inhibitor (saracatinib, 0.1 μmol/L), and MEK1/2 inhibitor (SL327, 10 μmol/L). Independent experimental replicates were performed (n=5 for C, n=7 for D). *P<0.01, § P<0.05, || P<0.0001 vs control (0 nmol/L of neopterin); # P<0.005, ¶ P<0.05 vs vehicle of neopterin. E, HASMCs were stained using the TUNEL method (green) after 48‐h incubation in SmGM‐2 supplemented with 5% FBS and neopterin (0, 200 nmol/L). DAPI was incorporated as a nuclear costain (blue). The graph indicates the percentage of apoptotic cells (n=3). Scale bar=100 μm. F, HASMCs were incubated in SmGM‐2 supplemented with 5% FBS and neopterin (0, 200 nmol/L) for 48 h. The effects of neopterin on proliferative phenotype and intracellular signaling were assessed by immunoblot (n=4 for p‐NF‐κB and p‐Akt; n=5 for SMemb, p‐ERK1/2, and PI3K; n=6 for c‐Src and Raf‐1). § P<0.05 vs 0 nmol/L of neopterin. Ab indicates antibody; AngII, angiotensin II; DAPI, 4′,6‐diamidino‐2‐phenylindole; FBS, fetal bovine serum; HASMC, human aortic smooth muscle cell; MEK, MAPK/ERK kinase; p‐Akt, phosphorylated Akt; p‐ERK1/2, phosphorylated extracellular signal‐regulated kinase 1 and 2; p‐NF‐κB, phosphorylated nuclear factor‐κB; PI3K, phosphoinositide 3‐kinase; SmGM‐2, Smooth Muscle Growth Medium‐2; TUNEL, terminal deoxynucleotidyl transferase‐mediated dUTP‐biotin nick‐end labeling.
We evaluated time‐dependent effects of neopterin on SMemb, a proliferative phenotype marker of VSMCs, and several signal transductions in HASMCs (Figure S4). Concentration‐dependent effects on only molecules observed with the positive changes (Figure S4) are shown in Figure 4F. The treatment with neopterin at 200 nmol/L for 48 hours significantly suppressed SMemb, c‐Src, and Raf‐1 expression and ERK1/2 phosphorylation, without a significant influence of NF‐κB phosphorylation, but increased PI3K expression and Akt phosphorylation (Figure 4F).
Effects of Neopterin on Extracellular Matrix Expression in HASMCs
Neopterin significantly increased protein expression of fibronectin, MMP‐2, and TIMP‐2 in a concentration‐dependent manner in HASMCs (Figure 5A). However, neopterin did not significantly alter the expression of collagen 1 or 3, elastin, or MMP‐9 (Figure 5A). In addition, neopterin significantly enhanced the activity of MMP‐2, but not MMP‐9, in HASMCs (Figure 5B).
Figure 5.

Effects of neopterin on extracellular matrix expression in HASMCs. A, HASMCs were incubated for 24 h in serum‐free SmGM‐2, with the indicated concentrations of neopterin and then harvested for immunoblot analyses of collagen 1, collagen 3, fibronectin, elastin, MMP‐2, MMP‐9, TIMP‐2, and α‐tubulin. Representative data showing protein expression (upper panels) with densitometry following normalization relative to α‐tubulin (lower panels, n=4). B, MMP‐2 and MMP‐9 activity. HASMCs were incubated for 24 h in serum‐free SmGM‐2 with the indicated concentrations of neopterin. MMP‐2 and MMP‐9 activity in culture supernatants was then determined by gelatin zymography (n=4). *P<0.001, † P<0.0005, ‡ P<0.05 vs 0 nmol/L of neopterin. HASMC indicates human aortic smooth muscle cell; MMP, matrix metalloproteinase; SmGM‐2, Smooth Muscle Growth Medium‐2; TIMP, tissue inhibitor of metalloproteinase.
Effects of Neopterin on Atherosclerotic Lesion Development in Apoe −/− Mice
In experiment 1, there were no significant differences in food intake, body weight, systolic and diastolic blood pressures, or plasma levels of glucose, total cholesterol, non‐HDL cholesterol, and HDL cholesterol among the 3 groups of 17‐ and 21‐week‐old Apoe −/− mice (Table 2). In Apoe −/− mice aged 21 versus 17 weeks, aortic atherosclerotic lesions in both the entire surface area and the cross‐sectional area of the root (plaque size) significantly increased by 4.5‐ and 2‐fold, respectively (Figure 6A, 6B, 6D, 6E, 6K, and 6L). Chronic administration of neopterin, demonstrated by a ≈1.4‐fold increase in plasma neopterin levels (Table 2), significantly retarded the development of the aortic atherosclerotic lesion area and atheromatous plaque size by 35% and 17%, respectively (Figure 6B, 6C, 6E, 6F, 6K, and 6L). In experiment 2, chronic infusion of anti–neopterin antibody significantly enhanced the development of aortic atherosclerotic lesion area and atheromatous plaque size by 1.4‐ and 1.3‐fold, respectively (Figure 6G through 6J, 6M, and 6N).
Table 2.
Characteristics and Laboratory Data of Apoe −/− Mice
| 17W Control (n=8) | 21W Control (n=10) | 21W Neopterin (n=8) | |
|---|---|---|---|
| Food intake, g/d | 4.4±0.2 | 4.4±0.5 | 4.6±0.1 |
| Body weight, g | 27.1±0.7 | 29.2±0.8 | 27.4±0.8 |
| Systolic blood pressure, mm Hg | 115.0±1.6 | 113.3±4.8 | 103.6±4.5 |
| Diastolic blood pressure, mm Hg | 95.0±1.6 | 86.4±7.3 | 85.1±3.3 |
| Glucose, mg/dL | 285.8±39.9 | 256.8±36.5 | 288.9±15.2 |
| Total cholesterol, mg/dL | 1686.6±82.9 | 1764.3±33.5 | 1613.1±95.7 |
| Non‐HDL cholesterol, mg/dL | 1679.0±83.3 | 1753.4±37.2 | 1606.7±95.6 |
| HDL cholesterol, mg/dL | 5.6±0.5 | 7.0±0.9 | 6.4±0.4 |
| PTX‐3, ng/mL | 108.9±7.7 | 204.4±15.3a | 143.5±9.3† |
| Neopterin, nmol/L | 1.28±0.21 | 1.36±0.19 | 1.93±0.10‡ |
Values are mean±SEM. HDL indicates high‐density lipoprotein; PTX‐3, pentraxin‐3; 17W, 17 weeks of age; 21W, 21 weeks of age.
P<0.0001 vs 17W control.
† P<0.01, ‡ P<0.05 vs 21W control.
Figure 6.

Effects of exogenous neopterin administration and endogenous neopterin attenuation on the development of atherosclerotic lesions in Apoe −/− mice. Experiment 1: 10 and 8 Apoe −/− mice (C57BL/6) at 17 weeks of age were intraperitoneally injected for 4 weeks with either vehicle (control) or neopterin (1.2 μmol/kg per day), respectively. Eight Apoe −/− mice (C57BL/6) at 17 weeks of age were euthanized before injection. Experiment 2: 6 and 5 Apoe −/− mice (BALB/c) at 17 weeks of age were infused for 4 weeks with either vehicle or anti–neopterin antibody (2.5 μL/kg per h), respectively, using osmotic minipumps. The aortic surface (A through C, G, and H) and cross‐sections of the aortic root (D through F, I, and J) were stained with Oil Red O. Comparisons of atherosclerotic lesion area (K and M) and atheromatous plaque size (L and N) between the groups. *P<0.0001, † P<0.05. Scale bar=200 μm. Apoe −/− indicates apolipoprotein E deficient; 17W, 17 weeks of age; 21W, 21 weeks of age.
In experiment 1, the expression of neopterin within atheromatous plaques of the aortic root significantly increased with age (17–21 weeks of age) in Apoe −/− mice (Figure 7A and 7D), which was in part consistent with the localization of macrophages (Figure 7A). Furthermore, neopterin levels were markedly (significantly) increased in the whole aortic wall, including atheromatous plaques and cusps of aortic valve, with neopterin injection (Figure 7A and 7D). In atheromatous plaques, monocyte/macrophage and CD8+ T‐cell infiltration (Figures 7A, 7B and 8A, 8C), VSMC content (Figure 7A and 7C), IFN‐γ, MCP‐1, and PTX‐3 expression (Figure 7A and 7E through 7G), as well as plasma PTX‐3 levels significantly increased with age (Table 2). CD4+ T cells were infiltrated in paralleled with CD8+ T cells in atheromatous plaques (Figure 8A through 8C). Chronic administration of neopterin significantly retarded monocyte/macrophage and CD8+ T cell infiltration (Figures 7A, 7B and 8A, 8C), VSMC content (Figure 7A and 7C), and MCP‐1 and PTX‐3 expression (Figure 7A, 7F, and 7G), but not IFN‐γ (Figure 7A and 7E), within atheromatous plaques, and decreased plasma PTX‐3 levels (Table 2). Neopterin administration tended to decrease CD4+ T cell infiltration in atheromatous plaques (Figure 8A and 8B). No change in IFN‐γ expression was associated with the incomplete suppression of CD4+ T cell content in atheromatous plaques. In addition, neopterin administration significantly reduced the ratio of macrophage/VSMC content within atheromatous plaques (Figure 9).
Figure 7.

Changes in plaque phenotype with exogenous neopterin administration and colocalization of endogenous neopterin within atheromatous plaques. Experiment 1: At 17 weeks of age, 8 Apoe −/− mice (C57BL/6) were euthanized before injection, and 10 and 8 Apoe −/− mice were intraperitoneally injected daily for 4 weeks with either vehicle (control) or neopterin (1.2 μmol/kg per day), respectively. A, Atheromatous plaques of the aortic root were stained with Oil Red O and specific antibodies against podocalyxin (an endothelial cell marker), MOMA‐2 (a monocyte/macrophage marker), α‐SMA (a VSMC marker), neopterin, interferon‐γ, MCP‐1, or PTX‐3. Hematoxylin was used for nuclear staining. Comparisons of MOMA‐2 (B), α‐SMA (C), neopterin (D), interferon‐γ (E), MCP‐1 (F), or PTX‐3 (G) among the 3 groups. *P<0.005, † P<0.05, ‡ P<0.0001, § P<0.0005, || P<0.001. Scale bar=200 μm. Apoe −/− indicates apolipoprotein E deficient; MCP‐1, monocyte chemotactic protein 1; MOMA‐2, monocyte/macrophage‐2; α‐SMA, α‐smooth muscle actin; PTX‐3, pentraxin 3; 17W, 17 weeks of age; 21W, 21 weeks of age; VSMC, vascular smooth muscle cell.
Figure 8.

Effects of exogenous neopterin administration on T‐cell infiltration within atheromatous plaque in Apoe −/− mice. In experiment 1 shown in Figure 7, atheromatous plaques were stained with specific antibodies against CD4 or CD8. Hematoxylin was used for nuclear staining. A, Arrows show CD4+ T cells or CD8+ T cells. Scale bar=50 μm. B and C, Comparisons of CD4+ and CD8+ cells among the 3 groups. *P<0.05. Apoe −/− indicates apolipoprotein E deficient; 17W, 17 weeks of age; 21W, 21 weeks of age.
Figure 9.

Effects of exogenous neopterin administration on atheromatous plaque stability in Apoe −/− mice. In experiment 1 shown in Figure 7, the ratio of macrophage (μm2)/VSMC content (μm2) within atheromatous plaques was compared among 3 groups of 17‐week old mice (n=8) and 21‐week‐old mice administered intraperitoneally vehicle (n=9) or neopterin (1.2 μmol/kg per day; n=8). *P<0.05. Apoe −/− indicates apolipoprotein E deficient; 17W, 17 weeks of age; 21W, 21 weeks of age; VSMC, vascular smooth muscle cell.
Discussion
This study demonstrated the increased expression of neopterin in coronary artery lesions and its plasma levels in CAD patients compared with non‐CAD patients; these levels are consistent with previous reports.10, 11, 13, 32, 33 In addition, we provide the first evidence showing that neopterin suppresses vascular inflammation and atherosclerosis. Neopterin reduces the inflammatory responses of both ECs and monocytes/macrophages and monocyte–EC adhesion. Macrophage foam cell formation and the migration and proliferation of VSMCs are all suppressed by neopterin. Neopterin increased protein expression of fibronectin, MMP‐2, and TIMP‐2 in VSMCs; this could correlate with inflammatory vascular remodeling. The reason why neopterin can exert multiple effects in all 3 vascular cells could be explained from the possible presence of its own receptors in these cells. Each change with neopterin in a variety of cellular and molecular phenomena in vitro seems to be weak, but united, all exert a greatly and visibly protective effect of neopterin against atherogenesis in vivo. Ultimately, exogenous neopterin administration retards the development of aortic atherosclerotic lesions with reduced monocyte/macrophage and T‐cell infiltration and lowered PTX‐3 levels in the aortic tissue and plasma in Apoe −/− mice. In addition, endogenous neopterin attenuation with chronic infusion of anti–neopterin antibody accelerates the development of atherosclerotic lesions in Apoe −/− mice. Collectively, these findings suggest that high levels of neopterin expression in coronary lesions may be attributed to endogenous upregulation of neopterin to counteract the progression of atherosclerosis.
These results concerning the effects of neopterin on macrophage foam cell formation conflict with previous data. Yan et al14 showed that extremely high concentrations of neopterin (100 μmol/L) decreased ABCA1 mRNA expression and cholesterol efflux in THP‐1 monocyte‐derived macrophage foam cells. This time‐ and concentration‐dependent effect was mediated by LXR‐α. THP‐1 cells were treated with the PKC (protein kinase C) agonist phorbol‐12‐myristate‐13‐acetate to provoke their differentiation into macrophages. The cell culture medium was then replaced with a serum‐free medium containing oxLDL to encourage full differentiation into foam cells. In our study, neopterin significantly increased protein expression of ABCA1 and ABCG1 (via LXR‐α) as well as cholesterol efflux to apolipoprotein A1 and HDL, leading to the suppression of oxLDL‐induced foam cell formation in primary cultured HMDMs. Our experimental protocol used standard medium containing 10% human sera and may, together with the different cell types and neopterin doses involved, explain the discrepancies between data sets. In addition, Gieseg and colleagues showed the antioxidant and antiapoptotic effects of 7,8‐dihydro‐neopterin at extremely high concentrations of ≥100 μmol/L.34, 35, 36 Compared with these studies,14, 34, 35, 36 our experiments used primary HMDMs under more physiologic conditions, using more conceivable concentrations of neopterin in human plasma.
The specific receptors for neopterin have yet to be identified. The signal transduction pathways for neopterin have also received scant attention. During their differentiation into HMDMs, neopterin decelerates M1 phenotype acquisition and accelerates expression of the M2 phenotype associated with NF‐κB downregulation and PPAR‐γ upregulation.4 Neopterin suppresses inflammatory responses associated with NF‐κB downregulation in human ECs. In general, foam cell formation and CD36 expression in macrophages and the migration and proliferation of VSMCs are regulated by the PKC/ERK1/2 pathway.37, 38, 39 The expression of fibronectin and MMP is mediated by the Akt pathway.40, 41 The present study suggests that neopterin suppresses VSMC proliferation via the c‐Src/Raf‐1/ERK1/2 pathway and increases fibronectin, MMP‐2, and TIMP‐2 expression, probably via the PI3K/Akt pathway.
The physiologic relevance of the neopterin concentrations used in the present in vitro and in vivo experiments warrants further discussion. First, the concentrations of neopterin needed to influence multiple HAEC, HUVEC, HMDM, and HASMC responses were relatively high (maximum ≈40‐fold) compared with human plasma concentrations (≈5–10 nmol/L). In the vascular wall, macrophages generate large amounts of neopterin in an autocrine/paracrine manner.6, 11, 12 A previous study showed that local levels of other vasoactive agents, such as serotonin in obstructive arteries, were increased by ≈33‐fold with coronary events.42 Consequently, it is not surprising that local levels of neopterin were increased in a fashion similar to other vasoactive agents. Second, the concentration of neopterin in serum obtained from a healthy human volunteer was 8.5 nmol/L in the present study. Therefore, the 10% concentration added to culture medium for HMDMs (0.85 nmol/L) was minor compared with the concentrations of neopterin added. Third, the concentrations of neopterin differed in terms of the inflammatory responses that they provoked in HAECs and HUVECs and their influence on monocyte adhesion, macrophage foam cell formation and effector expression, HASMC migration and proliferation, and extracellular matrix production. This likely reflects the different cell types used and their intracellular signaling pathways. In addition, the adequate concentrations of neopterin differed in inducing foam cell formation and related ABCG1 expression in HMDMs. This is mostly dependent on the difference in the presence or absence of oxLDL and their concerned signal transductions. Neopterin at greater than adequate concentrations might lead to the downregulation of the receptor (unknown) and intracellular signals, leading to the biphasic effects of neopterin. Finally, plasma neopterin levels in Apoe −/− mice infused with exogenous neopterin became significantly higher by ≈1.4‐fold than those of Apoe −/− mice infused with saline, which were not as high as expected. Because the half‐life of neopterin in circulating blood is 90 minutes,43 plasma neopterin might be somewhat reduced by renal excretion within 4 hours after the last intraperitoneal injection in Apoe −/− mice.
In this study, we demonstrated the atheroprotective effects of neopterin in vitro and in vivo. Other studies, together with this one, have shown that neopterin levels in the circulating blood and its expression in atherosclerotic lesions from carotid and coronary arteries are elevated in patients with CAD.10, 11, 13, 32, 33, 44 Furthermore, circulating neopterin levels are higher in acute coronary syndrome (unstable CAD) than in chronic coronary syndrome (stable CAD).33 Discrepancies between the anti‐atherosclerotic and anti‐inflammatory effects of neopterin in vitro and in vivo and the high expression levels of neopterin in CAD patients may reflect the different phases of inflammatory disease, as described for IL‐6.45 IL‐6 plays a crucial anti‐inflammatory role by controlling local and systemic acute inflammatory responses in the acute phase, whereas its inflammatory cytokine role takes over in the chronic phase.45 If nothing else, exogenous neopterin may contribute to the prevention and treatment of vascular inflammation and atherosclerosis, with endogenous neopterin needed to protect against the progression of vascular inflammation and atherosclerosis. Further analyses with overexpression and knockout of neopterin in Apoe −/− mice are required to clarify the roles of endogenous neopterin in atherogenesis.
This study investigated the roles of neopterin in both pathophysiology and treatment of atherosclerosis. Because neopterin can exert atheroprotective effects in all 3 types of vascular cells, as with incretins, adiponectin, omentin 1, urocortin 1, catestatin 1, and tumor necrosis factor–stimulated gene 6,16, 17, 19, 46, 47, 48 neopterin is a convincing candidate as a therapeutic agent against atherosclerosis. Further studies are needed to determine pharmacodynamics and pharmacokinetics of neopterin in animals and humans.
Methodological Limitations
We acknowledge that the limited sample size and the multiple statistical analyses using 1‐way ANOVA with the Bonferroni post hoc test might have led to type I errors (false positive). Concentration‐dependent effects of neopterin were evaluated in 3 to 8 samples. In these cases, the significant difference values should be set with a cutoff at P<0.017 (0.05/n=3) to P<0.006 (0.05/n=8) to handle false discovery rates from multiple testing. Further determinations are needed to analyze the precise concentration‐dependent effects of neopterin.
We used 2 strains of Apoe −/− mice (C57BL/6 and BALB/c) because Apoe −/− (C57BL/6) mice have been temporarily unavailable from our favorite breeder company. C57BL/6 and BALB/c mice are prototypical Th1‐ and Th2‐type mouse strains, respectively. Although CD4+ T cells isolated from Apoe −/− (BALB/c) mice produce high levels of IL‐4 (a Th2‐type cytokine) and low levels of IFN‐γ (a Th1‐type cytokine), CD4+ T cells from Apoe −/− (C57BL/6) mice secrete high levels of IFN‐γ and low levels of IL‐4 under stimuli of phorbol 12‐myristate 13‐acetate and ionomycin.49 Consequently, the progression of aortic atherosclerotic lesions is relatively moderate in Apoe −/− (BALB/c) mice compared with Apoe −/− (C57BL/6) mice.49 We have prepared strain‐matched Apoe −/− mice administered vehicle as a control (21 week old); therefore, data in the independent protocol of experiments are reliable. However, it is possible that the atherogenic effects of anti–neopterin antibody might be underestimated in Apoe −/− (BALB/c) mice compared with Apoe −/− (C57BL/6) mice.
Conclusion
The results of the present study indicate that neopterin prevents atherogenesis by suppressing inflammatory responses in ECs and macrophages, proliferation in ECs and VSMCs, and macrophage foam cell formation. Clinically, the results presented offer an extended therapeutic window in which to combat atherosclerosis and restenosis after coronary angioplasty. Future studies are needed to identify the roles of neopterin in other inflammatory cardiovascular diseases such as ischemia–reperfusion injury, aortitis, aneurysm, and diabetic angiopathy.
Sources of Funding
This work was supported in part by a Grant‐in‐Aid for Young Scientists (B) (25860418 to Sato) and Grants‐in‐Aid for Scientific Research (C) (16K08943 to Sato and 26460659 to Watanabe) from the Japan Society for the Promotion of Science (JSPS), and a Grant‐in‐Aid for JSPS Fellows (DC2, 17J02716 to Shirai).
Disclosures
None.
Supporting information
Figure S1. Immunohistochemical staining of neopterin and IgG control in coronary atherosclerotic lesions from patients with coronary artery disease.
Figure S2. Immunohistochemical staining of IgG controls.
Figure S3. Effects of neopterin on proliferation, inflammatory response, and monocyte adhesion in human umbilical vein endothelial cells.
Figure S4. Time‐dependent effects of neopterin on intracellular signal transductions in human aortic smooth muscle cells.
Acknowledgments
We thank Dr Fumiko Itoh for her kind support, and San Francisco Edit for their assistance in editing this article.
(J Am Heart Assoc. 2018;7:e007359 DOI: 10.1161/JAHA.117.007359.)29420219
This work was presented in part at the American Heart Association's Scientific Sessions, November 7–11, 2015, in Orlando, FL; at the 65th Annual Scientific Session of the American College of Cardiology, April 2 to 4, 2016, in Chicago, IL; and at the European Society of Cardiology Congress, August 27–31, 2016, in Rome, Italy.
References
- 1. Hansson GK, Libby P. The immune response in atherosclerosis: a double‐edged sword. Nat Rev Immunol. 2006;6:508–519. [DOI] [PubMed] [Google Scholar]
- 2. Yu XH, Fu YC, Zhang DW, Yin K, Tang CK. Foam cells in atherosclerosis. Clin Chim Acta. 2013;424:245–252. [DOI] [PubMed] [Google Scholar]
- 3. Igarashi M, Osuga J, Uozaki H, Sekiya M, Nagashima S, Takahashi M, Takase S, Takanashi M, Li Y, Ohta K, Kumagai M, Nishi M, Hosokawa M, Fledelius C, Jacobsen P, Yagyu H, Fukayama M, Nagai R, Kadowaki T, Ohashi K, Ishibashi S. The critical role of neutral cholesterol ester hydrolase 1 in cholesterol removal from human macrophages. Circ Res. 2010;107:1387–1395. [DOI] [PubMed] [Google Scholar]
- 4. Paoli FD, Staels B, Chinetti‐Gbaguidi G. Macrophage phenotypes and their modulation in atherosclerosis. Circ J. 2014;78:1775–1781. [DOI] [PubMed] [Google Scholar]
- 5. Obikane H, Abiko Y, Ueno H, Kusumi Y, Esumi M, Mitsumata M. Effect of endothelial cell proliferation on atherogenesis: a role of p21Sdi/Cip/Waf1 in monocyte adhesion to endothelial cells. Atherosclerosis. 2010;212:116–122. [DOI] [PubMed] [Google Scholar]
- 6. Huber C, Batchelor JR, Fuchs D, Hausen A, Lang A, Niederwieser D, Reibnegger G, Swetly P, Troppmair J, Wachter H. Immune response‐associated production of neopterin. Release from macrophages primarily under control of interferon‐gamma. J Exp Med. 1984;160:310–316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Schroecksnadel K, Frick B, Winkler C, Fuchs D. Crucial role of interferon‐γ and stimulated macrophages in cardiovascular disease. Curr Vasc Pharmacol. 2006;4:205–213. [DOI] [PubMed] [Google Scholar]
- 8. Fuchs D, Weiss G, Wachter H. Neopterin, biochemistry and clinical use as a marker for cellular immune reactions. Int Arch Allergy Immunol. 1993;101:1–6. [DOI] [PubMed] [Google Scholar]
- 9. Kojima S, Icho T, Kajiwara Y, Kubota K. Neopterin as an endogenous antioxidant. FEBS Lett. 1992;304:163–166. [DOI] [PubMed] [Google Scholar]
- 10. Murr C, Grammer TB, Meinitzer A, Kleber ME, März W, Fuchs D. Immune activation and inflammation in patients with cardiovascular disease are associated with higher phenylalanine to tyrosine ratios: the Ludwigshafen Risk and Cardiovascular Health Study. J Amino Acids. 2014;2014:783730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Adachi T, Naruko T, Itoh A, Komatsu R, Abe Y, Shirai N, Yamashita H, Ehara S, Nakagawa M, Kitabayashi C, Ikura Y, Ohsawa M, Yoshiyama M, Haze K, Ueda M. Neopterin is associated with plaque inflammation and destabilisation in human coronary atherosclerotic lesions. Heart. 2007;93:1537–1541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Sugioka K, Naruko T, Hozumi T, Nakagawa M, Kitabayashi C, Ikura Y, Shirai N, Matsumura Y, Ehara S, Ujino K, Itoh A, Haze K, Becker AE, Yoshiyama M, Ueda M. Elevated levels of neopterin are associated with carotid plaques with complex morphology in patients with stable angina pectoris. Atherosclerosis. 2010;208:524–530. [DOI] [PubMed] [Google Scholar]
- 13. van Dijk RA, Rijs K, Wezel A, Hamming JF, Kolodgie FD, Virmani R, Schaapherder AF, Lindeman JH. Systematic evaluation of the cellular innate immune response during the process of human atherosclerosis. J Am Heart Assoc. 2016;5:e002860 DOI: 10.1161/JAHA.115.002860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Yan JQ, Tan CZ, Wu JH, Zhang DC, Chen JL, Zeng BY, Jiang YP, Nie J, Liu W, Liu Q, Dai H. Neopterin negatively regulates expression of ABCA1 and ABCG1 by the LXRα signaling pathway in THP‐1 macrophage‐derived foam cells. Mol Cell Biochem. 2013;379:123–131. [DOI] [PubMed] [Google Scholar]
- 15. Grammer TB, Fuchs D, Boehm BO, Winkelmann BR, Maerz W. Neopterin as a predictor of total and cardiovascular mortality in individuals undergoing angiography in the Ludwigshafen Risk and Cardiovascular Health Study. Clin Chem. 2009;55:1135–1146. [DOI] [PubMed] [Google Scholar]
- 16. Sheldon J, Riches PG, Soni N, Jurges E, Gore M, Dadian G, Hobbs JR. Plasma neopterin as an adjunct to C‐reactive protein in assessment of infection. Clin Chem. 1991;37:2038–2042. [PubMed] [Google Scholar]
- 17. Watanabe K, Watanabe R, Konii H, Shirai R, Sato K, Matsuyama T, Ishibashi‐Ueda H, Koba S, Kobayashi Y, Hirano T, Watanabe T. Counteractive effects of omentin‐1 against atherogenesis. Cardiovasc Res. 2016;110:118–128. [DOI] [PubMed] [Google Scholar]
- 18. Watanabe R, Watanabe H, Takahashi Y, Kojima M, Konii H, Watanabe K, Shirai R, Sato K, Matsuyama T, Ishibashi‐Ueda H, Iso Y, Koba S, Kobayashi Y, Hirano T, Watanabe T. Atheroprotective effects of tumor necrosis factor‐stimulated gene‐6. JACC Basic Transl Sci. 2016;1:496–509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Naito C, Hashimoto M, Watanabe K, Shirai R, Takahashi Y, Kojima M, Watanabe R, Sato K, Iso Y, Matsuyama T, Suzuki H, Ishibashi‐Ueda H, Watanabe T. Facilitatory effects of fetuin‐A on atherosclerosis. Atherosclerosis. 2016;246:344–351. [DOI] [PubMed] [Google Scholar]
- 20. Hasegawa A, Sato K, Shirai R, Watanabe R, Yamamoto K, Watanabe K, Nohtomi K, Hirano T, Watanabe T. Vasoprotective effects of urocortin 1 against atherosclerosis in vitro and in vivo. PLoS One. 2014;9:e0110866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Sato K, Shirai R, Hontani M, Shinooka R, Hasegawa A, Kichise T, Yamashita T, Yoshizawa H, Watanabe R, Matsuyama T, Ishibashi‐Ueda H, Koba S, Kobayashi Y, Hirano T, Watanabe T. Potent vasoconstrictor kisspeptin‐10 induces atherosclerotic plaque progression and instability: reversal by its receptor GPR54 antagonist. J Am Heart Assoc. 2017;6:e005790 DOI: 10.1161/JAHA.117.005790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Konii H, Sato K, Kikuchi S, Okiyama H, Watanabe R, Hasegawa A, Yamamoto K, Itoh F, Hirano T, Watanabe T. Stimulatory effects of cardiotrophin 1 on atherosclerosis. Hypertension. 2013;62:942–950. [DOI] [PubMed] [Google Scholar]
- 23. Yamamoto K, Tajima Y, Hasegawa A, Takahashi Y, Kojima M, Watanabe R, Sato K, Shichiri M, Watanabe T. Contrasting effects of stanniocalcin‐related polypeptides on macrophage foam cell formation and vascular smooth muscle cell migration. Peptides. 2016;82:120–127. [DOI] [PubMed] [Google Scholar]
- 24. Watanabe T, Nishio K, Kanome T, Matsuyama T, Koba S, Sakai T, Sato K, Hongo S, Nose K, Ota H, Kobayashi Y, Katagiri T, Shichiri M, Miyazaki A. Impact of salusin‐α and ‐β on human macrophage foam cell formation and coronary atherosclerosis. Circulation. 2008;117:638–648. [DOI] [PubMed] [Google Scholar]
- 25. Xu G, Watanabe T, Iso Y, Koba S, Sakai T, Nagashima M, Arita S, Hongo S, Ota H, Kobayashi Y, Miyazaki A, Hirano T. Preventive effects of heregulin‐β1 on macrophage foam cell formation and atherosclerosis. Circ Res. 2009;105:500–510. [DOI] [PubMed] [Google Scholar]
- 26. Inoue K, Kobayashi M, Yano K, Miura M, Izumi A, Mataki C, Doi T, Hamakubo T, Reid PC, Hume DA, Yoshida M, Aird WC, Kodama T, Minami T. Histone deacetylase inhibitor reduces monocyte adhesion to endothelium through the suppression of vascular cell adhesion molecule‐1 expression. Arterioscler Thromb Vasc Biol. 2006;26:2652–2659. [DOI] [PubMed] [Google Scholar]
- 27. Kanbe E, Hatta Y, Tsuboi I, Harada T, Koshinaga M, Inoue T, Aizawa S. Effects of neopterin on the hematopoietic microenvironment of senescence‐accelerated mice (SAM). Biol Pharm Bull. 2006;29:43–48. [DOI] [PubMed] [Google Scholar]
- 28. Aisaki K, Tsuboi I, Harada T, Oshima H, Yamashita A, Hirabayashi Y, Kanno J, Inoue T, Aizawa S. Neopterin, inflammation‐associated product, prolongs erythropoiesis suppression in aged SAMP1 mice due to senescent stromal‐cell impairment. Exp Biol Med (Maywood). 2012;237:279–286. [DOI] [PubMed] [Google Scholar]
- 29. Balogh A, Mittermayr M, Schlager A, Balogh D, Schobersberger W, Fuchs D, Margreiter J. Mechanism of neopterin‐induced myocardial dysfunction in the isolated perfused rat heart. Biochim Biophys Acta. 2005;1724:17–22. [DOI] [PubMed] [Google Scholar]
- 30. Icho T, Kojima S, Hayashi M, Kajiwara Y, Kitabatake K, Kubota K. Suppression of ischemia‐reperfusion injury in murine models by neopterins. Toxicol Appl Pharmacol. 1995;130:27–31. [DOI] [PubMed] [Google Scholar]
- 31. Shindo A, Tanemura H, Yata K, Hamada K, Shibata M, Umeda Y, Asakura F, Toma N, Sakaida H, Fujisawa T, Taki W, Tomimoto H. Inflammatory biomarkers in atherosclerosis: pentraxin 3 can become a novel marker of plaque vulnerability. PLoS One. 2014;9:e100045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Liu ZY, Li YD. Relationship between serum neopterin levels and coronary heart disease. Genet Mol Res. 2013;12:4222–4229. [DOI] [PubMed] [Google Scholar]
- 33. Schumacher M, Halwachs G, Tatzber F, Fruhwald FM, Zweiker R, Watzinger N, Eber B, Wilders‐Truschnig M, Esterbauer H, Klein W. Increased neopterin in patients with chronic and acute coronary syndromes. J Am Coll Cardiol. 1997;30:703–707. [DOI] [PubMed] [Google Scholar]
- 34. Gieseg SP, Amit Z, Yang YT, Shchepetkina A, Katouah H. Oxidant production, oxLDL uptake, and CD36 levels in human monocyte‐derived macrophages are downregulated by the macrophage‐generated antioxidant 7,8‐dihydroneopterin. Antioxid Redox Signal. 2010;13:1525–1534. [DOI] [PubMed] [Google Scholar]
- 35. Baird SK, Reid L, Hampton MB, Gieseg SP. OxLDL induced cell death is inhibited by the macrophage synthesised pterin, 7,8‐dihydroneopterin, in U937 cells but not THP‐1 cells. Biochim Biophys Acta. 2005;1745:361–369. [DOI] [PubMed] [Google Scholar]
- 36. Shchepetkina AA, Hock BD, Miller A, Kennedy MA, Gieseg SP. Effect of 7,8‐dihydroneopterin mediated CD36 downregulation and oxidant scavenging on oxidised low‐density lipoprotein induced cell death in human macrophages. Int J Biochem Cell Biol. 2017;87:27–33. [DOI] [PubMed] [Google Scholar]
- 37. Lin CS, Lin FY, Ho LJ, Tsai CS, Cheng SM, Wu WL, Huang CY, Lian CH, Yang SP, Lai JH. PKCδ signalling regulates SR‐A and CD36 expression and foam cell formation. Cardiovasc Res. 2012;95:346–355. [DOI] [PubMed] [Google Scholar]
- 38. Blanc A, Pandey NR, Srivastava AK. Synchronous activation of ERK 1/2, p38MAPK and PKB/Akt signaling by H2O2 in vascular smooth muscle cells: potential involvement in vascular disease. Int J Mol Med. 2003;11:229–234. [PubMed] [Google Scholar]
- 39. Hong H, McCullough CM, Stegemann JP. The role of ERK signaling in protein hydrogel remodeling by vascular smooth muscle cells. Biomaterials. 2007;28:3824–3833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Kim JH, Lee TY, Park J, Ha H, Kang SW, Kim YS. Effects of tautomycetin on proliferation and fibronectin secretion in vascular smooth muscle cells and glomerular mesangial cells. Transplant Proc. 2005;37:1959–1961. [DOI] [PubMed] [Google Scholar]
- 41. Seo KW, Lee SJ, Ye BH, Kim YW, Bae SS, Kim CD. Mechanical stretch enhances the expression and activity of osteopontin and MMP‐2 via the Akt1/AP‐1 pathways in VSMC. J Mol Cell Cardiol. 2015;85:13–24. [DOI] [PubMed] [Google Scholar]
- 42. Leosco D, Fineschi M, Pierli C, Fiaschi A, Ferrara N, Bianco S, Longobardi G, Pisani E, Bravi A, Rengo F. Intracoronary serotonin release after high‐pressure coronary stenting. Am J Cardiol. 1999;84:1317–1322. [DOI] [PubMed] [Google Scholar]
- 43. Fuchs D, Stahl‐Hennig C, Gruber A, Murr C, Hunsmann G, Wachter H. Neopterin—its clinical use in urinalysis. Kidney Int. 1994;47:S8–S11. [PubMed] [Google Scholar]
- 44. Ragab M, Hassan H, Zaytoun T, Refai W, Rocks B, Elsammak M. Evaluation of serum neopterin, high‐sensitivity C‐reactive protein and thiobarbituric acid reactive substances in Egyptian patients with acute coronary syndrome. Exp Clin Cardiol. 2005;10:250–255. [PMC free article] [PubMed] [Google Scholar]
- 45. Xing Z, Gauldie J, Cox G, Baumann H, Jordana M, Lei XF, Achong MK. IL‐6 is an antiinflammatory cytokine required for controlling local or systemic acute inflammatory responses. J Clin Invest. 1998;101:311–320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Nagashima M, Watanabe T, Terasaki M, Tomoyasu M, Nohtomi K, Kim‐Kaneyama J, Miyazaki A, Hirano T. Native incretins prevent the development of atherosclerotic lesions in apolipoprotein E knockout mice. Diabetologia. 2011;54:2649–2659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47. Kojima M, Ozawa N, Mori Y, Takahashi Y, Watanabe‐Kominato K, Shirai R, Watanabe R, Sato K, Matsuyama T, Ishibashi‐Ueda H, Koba S, Kobayashi Y, Hirano T, Watanabe T. Catestatin prevents macrophage‐driven atherosclerosis but not arterial injury‐induced neointimal hyperplasia. Thromb Haemost. 2018;118:182–194. [DOI] [PubMed] [Google Scholar]
- 48. Watanabe T, Sato K, Itoh F, Noguchi Y, Fujimoto K, Koyama T, Shichiri M. Emerging roles for vasoactive peptides in diagnostic and therapeutic strategies against atherosclerotic cardiovascular diseases. Curr Protein Pept Sci. 2013;14:472–480. [DOI] [PubMed] [Google Scholar]
- 49. Schulte S, Sukhova GK, Libby P. Genetically programmed biases in Th1 and Th2 immune responses modulate atherogenesis. Am J Pathol. 2008;172:1500–1508. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1. Immunohistochemical staining of neopterin and IgG control in coronary atherosclerotic lesions from patients with coronary artery disease.
Figure S2. Immunohistochemical staining of IgG controls.
Figure S3. Effects of neopterin on proliferation, inflammatory response, and monocyte adhesion in human umbilical vein endothelial cells.
Figure S4. Time‐dependent effects of neopterin on intracellular signal transductions in human aortic smooth muscle cells.


