Abstract
Breast tumour progression results from the advancement of the disease to a metastatic phenotype. Rac1 and Cdc42 belong to the Rho family of genes that, together with their downstream effectors, Wiskott–Aldrich Syndrome protein-family verprolin-homologous protein 2 (WAVE2) and Arp2/3, assume a vital part in cytoskeletal rearrangement and the arrangement of film projections that advance malignant cell relocation and invasion. Mangiferin is a characteristic polyphenolic compound from Mangifera indica L. (Anacardiaceae), ordinarily referred to as mango, that is consumed worldwide as a natural product, including culinary and seasoning applications. Mangiferin delays breast malignancy development and progression by inhibiting different signalling pathways required in mitogenic signalling and metastatic progression. Studies were performed to analyse the impact of mangiferin on Rac1/WAVE2 flagging, relocation and invasion in highly metastatic human MDA-MB-231 mammary cells. Additional studies led to the observation that comparative treatment with mangiferin caused marked reduction in tumour cell movement and invasion. Taken together, these discoveries demonstrate that mangiferin treatment adequately hinders Rac1/WAVE2 flagging and diminishes metastatic phenotypic expression in malignant mammary cells, indicating that mangiferin may provide a benefit as a novel restorative approach in the treatment of metastatic breast cancer.
Keywords: Migration, Invasion, Mangiferin, Rac1, Wiskott–Aldrich Syndrome protein-family verprolin-homologous protein 2 (WAVE2)
Introduction
Breast malignancy is a noteworthy medical issue and one of the primary reasons for tumour growth-related mortality in women (Dubey et al. 2015). By far, most breast tumour growth results from the progression of the metastatic phenotype, which is described as malignant cells separating from the essential tumour and spreading to distal destinations in the body—for example, the bone, brain, encompassing lymph hubs, liver, and lungs (Escudero-Esparza et al. 2012; Ishihara et al. 2013; Ko et al. 2014). Breast tumour metastasis is a complex and multistep process that incorporates morphological changes, separation from the cellular layer, expanded versatility and invasion into encompassing tissues, intravasation, flow, bond, extravasation, and development at distal destinations (Yamaguchi and Condeelis 2007). An underlying step in the metastasis procedure includes the epithelial-to-mesenchymal transition (EMT), where neoplastic epithelial cells lose their cell extremity and acquire mesenchymal-like versatility that permits them to attack encompassing tissues (Ozdamar et al. 2005; Kalluri and Weinberg 2009). This amoeboid-like development requires the arrangement of cell projections that result from the transformation of monomeric globular actin (G-actin) into polymerized filamentous actin (Foerster et al. 2014).
The amoeboid type of tumour cell relocation is described by adjusted morphology and broad RhoA flagging. Tumour cells can switch between these 2 development types depending on their movement by repressing Rho/ROCK flagging (Symons and Segall 2009). Additionally, promotion of the mesenchymal phenotype that leads to a noteworthy increment in the quantity of melanoma cells moving through CECs (Circulating Endothelial Cells) could be investigated (Wilhelm et al. 2014). Here, we investigated melanoma and breast malignancy cells regarding mesenchymal versus amoeboid relocation through the brain endothelium. Whether tumour cells promote Rho/ROCK or Rac-subordinate transendothelial movement is clinically significant because inhibitors of both Rho/ROCK (e.g. fasudil) and Rac pathways are increasingly being used as providing potential benefit. Wiskott–Aldrich Syndrome protein-family verprolin-homologous protein 2 (WAVE2) and the Arp2/3 complex are downstream effectors of Rac1/Cdc42 that prompt the development of layer bulges that are required for tumour cell development (Kurisu and Takenawa 2010; Yokotsuka et al. 2011). Among the few members of the WAVE group of proteins, WAVE2 has appeared to be the most critical in directing actin polymerization, lamellipodia development and mesenchymal-like versatility in different malignant cell types (Takenawa and Miki 2001).
Mangiferin, 1,3,6,7-tetrahydroxyxanthone-C2-beta-d-glucoside, a type of xanthone derivative and C-glucosylxanthone, is a monomeric compound from plants of the Anacardiaceae and Gentianaceae families. Dar et al. (2005) reported that mangiferin exhibited various natural benefits, including cell reinforcement and anti-tumour (Guha et al. 1996), antibacterial, antiviral and immunomodulatory activities (Duang et al. 2011). Additionally, previous studies have revealed that mangiferin displays antineoplastic effects on prostate malignancy (Garcia-Rivera et al. 2011), colon cancer (Noratto et al. 2010), leukaemia (Percival et al. 2006), and lung cancer (Rajendran et al. 2015). It has already been proposed that mangiferin can hinder the expansion of K562 leukaemia cells and initiate cell apoptosis using the downregulation of NF-κB action. Additionally, Cheng et al. (2007) reported that mangiferin hinders telomerase activity in K562 cells in a time- and concentration-dependent manner, induces apoptosis, and upregulates mRNA and protein levels. Telomerase is a reverse transcriptase enzyme that carries its own RNA molecule which is used as a template when it elongates telomeres. In the present study, we investigated the effect of mangiferin on Rac1/WAVE2 inhibition and invasion in MDA-MB-231 mammary tumour cells in vitro.
Materials and methods
Reagents and antibodies
Chemicals and reagents were obtained from Sigma-Aldrich (St. Louis, MO, USA), unless otherwise indicated. Anti-actin was purchased from Santa Cruz Biotechnology (Dallas, TX, USA). Goat anti-rabbit rabbit secondary antibody (Ab) was acquired from Perkin Elmer, Inc. Biosciences (Boston, MA, USA). Cultrex® BME Cell Invasion Assay Kit was acquired from Trevigen (Gaithersburg, MD, USA).
Cell lines and culture conditions
Human MDA-MB-231 breast cancer cells were purchased from American Type Culture Collection (Manassas, VA, USA). MDA-MB-231 breast epithelial cancer cells were grown in RPMI-1640 medium supplemented with 10% foetal bovine serum supplemented with 100 U/mL penicillin, 0.1 mg/mL streptomycin, and 10 μg/mL insulin and were maintained at 37 °C in a humidified incubator provided with 95% air and 5% CO2. MCF-7 cells, obtained from the American Type Culture Collection, were cultivated in a 1:1 mixture of DMEM and Ham’s F-12 medium supplemented with 5% (vol:vol) FBS. For experiments, cells were separated and collected for stock culture by first washing in sterile Ca2+ and Mg2+ phosphate-buffered saline (PBS), then incubating the cells in 0.05% trypsin containing 0.025% ethylenediaminetetraacetic acid (EDTA) in PBS for 5 min at 37 °C. Cells were then centrifuged, resuspended in medium and counted using a haemocytometer.
Experimental treatments
Mangiferin was first dissolved in 70% DMSO and then was added to sterile 10% BSA in water, followed by incubation at 37 °C overnight with gentle shaking as described previously (Higgs and Pollard 1999). This mangiferin stock solution was then used to prepare different concentrations in the test media.
Measurement of cell viability
For cell viability examination, MDA-MB-231 and MCF-7 cells were seeded at a density of 1 × 104 cells/well in 96-well culture plates (5 replicates/condition). The next day, cells were divided into different treatment groups according to the concentration of mangiferin (10–50 μM). Cells were grown in the respective test media, which were replaced every other day for 4 d. For cytotoxicity experiments, MDA-MB-231 and MCF-7 cells were seeded at a density of 1 × 104 cells/well in 96-well culture plates (5 replicates/condition), maintained in control media and were allowed to grow for 3 day (roughly up to 70% confluency). Cells were then divided into different treatment groups and were treated with 10–50 μM mangiferin for 24 h. The following day, cell viability was determined using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) colorimetric assay as performed previously (Desai et al. 2016; Lucato et al. 2015). Briefly, cells were incubated at 37 °C for 3 h in control media containing 0.5 mg/mL MTT. Thereafter, medium was removed, the formed formazan crystals were dissolved in dimethyl sulfoxide (DMSO; 100 μL/well), and the optical density of each specimen was measured at 570 nm using a microplate reader (SpectraCount; Packard BioScience Company, Meriden, CT, USA). The quantity of cells/well was computed against standards arranged by plating known cell densities at the beginning of experimentation.
Western blot analysis
MDA-MB-231 cells were plated at 1 × 106 cells/100-mm culture plate. The following day, the cells were treated with medium containing the respective mangiferin concentrations for 4 d. Thereafter, the cells were washed with PBS, trypsin was utilized to detach cells, and lysates were collected for further analysis. The Bio-Rad protein assay (Bio-Rad, Hercules, CA, USA) was utilized to determine the protein concentration in each sample. Then electrophoresis using sodium dodecyl sulfate (SDS)–polyacrylamide minigels was performed. The SDS-PAGE electrophoresis conditions were as follows: 12% gel, 90 V for 90 min, and 25 µg/protein loaded per well. Proteins from each gel were then transferred at 30 V for 12–16 h at 4 °C onto a solitary polyvinylidene fluoride film (DuPont, Boston, MA, USA). The membranes were then blocked and incubated with the respective primary antibodies diluted 1:1000 to 1:5000 as depicted already. The membranes were then washed and incubated with the respective horseradish peroxide-conjugated secondary antibody diluted 1:5000 for 1 h. Proteins were visualized by chemiluminescence (Pierce, Rockford, IL, USA), and images were acquired using the Syngene Imaging System (Fredrick, MD, USA). Actin expression was used as a loading control. Analyses were performed at least 3 times, and representative images are shown in the figures.
Immunocytochemical fluorescence staining
MDA-MB-231 cells were plated at 2 × 104 cells/chamber in an 8-well chamber glass slide (BD Falcon, San Jose, CA, USA) and were maintained in medium. The following day, the medium was removed, cells were washed in PBS, and then they were incubated with their respective test media (10 μM mangiferin) for 4 d. Thereafter, cells were washed, resuspended in 4% paraformaldehyde/PBS and then permeabilized with 0.2% Triton X-100/PBS as described previously. The cells were then washed, blocked with 5% goat serum, and then were incubated with the primary Ab against WAVE2 (1:1000) at 4 °C overnight (Iwaya et al. 2007). The following day, cells were washed and then were incubated with Alexa Flour 488-conjugated secondary Ab (1:5000) (Iwaya et al. 2007). Slides were washed and mounted with Vectashield medium containing 4′-6-diamidino-2-phenylindole (DAPI) (Vector Laboratories Inc., Burlingame, CA, USA) (Iwaya et al. 2007). An LSM Pascal confocal microscope (Carl Zeiss Microimaging Inc, Thornwood, NY, USA) was then used to capture fluorescent images.
Invasion assay
Tumour cell invasion was determined using the Cultrex® 96-well basement membrane extract (BME) invasion assay system according to the manufacturer’s instructions (Trevigen Inc., Gaithersburg, MD, USA). MDA-MB-231 cells were plated at a thickness of 1 × 106 cells/100-mm culture plate and were maintained in their respective control media for 3 day (approximately up to 70% confluence). Subsequently, cells were washed in sterile PBS, incubated in serum-free medium and starved for 24 h. The following day, cells were trypsinized, washed, and counted, and 5 × 104 cells/well (5 wells/group) were plated on the top layer of the 96-well plate in 50 μL of medium containing 10 μM mangiferin, and RPMI medium containing 10% FBS was used as a chemo attractant. Next, 120 μL of the control or test medium was added to the basement chamber of the plate. After 24 h of incubation period, the top assembly of cells was then washed, and analysed at 485 nm (excitation), 520 nm (emission). The fluorescence intensity of each sample was then measured against a standard to determine the percent cell invasion as described in the guidelines of the unit.
Migration assay
MDA-MB-231 cells were plated in 24-well plates (5 replicates/group) and were incubated overnight to form a sub-confluent cell monolayer. Wounds were then made by making a scratch with a sterile 200-μL micropipette tip. The medium was then removed, cells were washed, and then they were fed with their respective serum-free-test medium containing 10 μM mangiferin and 10 ng/mL EGF as a mitogen. After 24 h, the media were removed, cells were washed in ice-cold PBS, fixed in − 20 °C methanol, and then stained with Giemsa reagent. Images were visualized using a phase-contrast microscope (Nikon Eclipse TS100 inverted microscope; Nikon Instruments Inc., Melville, NY, USA).
Statistical analysis
Differences among treatment groups were determined using one-way ANOVA followed by Dunnett’s t test. p < 0.05 was considered to be statistically significant compared with their respective control groups.
Results
Growth inhibitory effects of mangiferin on human mammary epithelial cells
After treatment for 4 d, mangiferin induced a dose-responsive inhibition in the growth of MDA-MB-231 and MCF-7 tumour cells compared with their respective vehicle-treated control groups (Fig. 1A1, A2). The IC50 dose for mangiferin was determined to be 10 μM for MDA-MB-231 and MCF-7 cells. After 24 h of treatment, mangiferin induced a dose-responsive reduction in the viability of MDA-MB-231 and MCF-7 breast cancer cells compared with that of control cells (Fig. 1B1, B2).
Fig. 1.
Growth inhibitory and cytotoxic effects of mangiferin on highly malignant MDA-MB-231 and MCF-7 cells. a All cells (MDA-MB-231) were initially plated at a density of 1 × 104 cells/well (6 wells/group) in 96-well plates. Cells were allowed to attach for a 24-h period and then were subjected to their specific treatments for 4 d. b All cells (MCF-7) were initially plated at a density of 1 × 104 cells/well (6 wells/group) in 96-well plates and were maintained in medium for 3 days (approximately 70% confluence). Cells were then exposed to their respective treatments for 24 h. The MTT colorimetric assay was used to count the viable cell number. Vertical bars indicate the mean cell number ± SEM in each treatment group. *p < 0.05 compared with their respective treated control group
Effects of mangiferin on Rac1/WAVE2 signalling protein expression in mammary tumour cells
Western blot analysis shows that, after 4 days of treatment with 0–10 μM mangiferin, MDA-MB-231 mammary tumour cells displayed a dose-responsive decrease in Rac1/Cdc42, phospho-Rac1/Cdc42, WAVE2, Arp2, and Arp3 compared with cells in their control groups (Fig. 2).
Fig. 2.
Western blot analysis of mangiferin effects on Rac1/WAVE2 signalling proteins in highly metastatic mammary tumour cells. Western blot analysis of Rac1/Cdc42, phospho-Rac1/Cdc42, Wave2, Arp2, and Arp3 (a). Actin bands were visualized to ensure equal sample loading. Western blots are representative images obtained in experiments that were repeated 3 or more times. (b) The integrated optical density (arbitrary units) of each band was normalized to their corresponding actin and control treatment bands. Vertical bars represent the protein levels in individual treatment groups ± SEM compared with their respective vehicle-treated control group. *p < 0.05 compared with their respective vehicle-treated control group
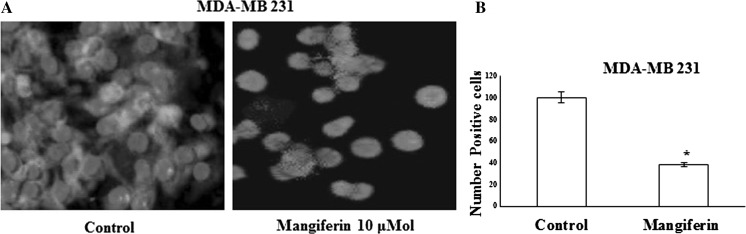
Effects of mangiferin on WAVE2 immunofluorescent staining in mammary tumour cells
MDA-MB-231 (Fig. 3) cells in their respective control groups displayed a moderate level of positive WAVE2 immunofluorescent staining. Treatment with 10 μM mangiferin resulted in nearly complete elimination of positive WAVE2 immunofluorescent staining (Fig. 3a). Image analysis of fluorescence photomicrographs showed that treatment with 10 μM mangiferin resulted in a significant decrease in positive WAVE2 staining in MDA-MB-231 (Fig. 3b) mammary tumour cells compared with cells in their respective treated control groups.
Fig. 3.
Effects of mangiferin on WAVE2 immunofluorescent staining in highly metastatic MDA-MB-231 mammary tumour cells. The green staining indicates positive staining for WAVE2 (a), while the blue colour represents counterstaining of cell nuclei with DAPI. b Quantitative image analysis of treatment effects on the percentage of MDA-MB-231 mammary tumour cells displaying positive WAVE2 staining compared with the total number of cells within each treatment group. Vertical bars represent the percentage of positive WAVE2 stained cells ± SEM in each treatment group. *p < 0.05 compared with their respective vehicle-treated control group. Cells were counted manually in five photomicrographs selected randomly in each chamber for all treatment groups. (Color figure online)
Effects of mangiferin on mammary tumour cell migration
The migration assay was used to determine the effects of mangiferin on MDA-MB-231 breast cancer cell motility. Cells in the MDA-MB-231 control groups displayed nearly complete wound closure, whereas treatment with 10 μM mangiferin resulted in a large reduction in cell migration after 24 h (Fig. 4a). Quantitative analysis determined that mangiferin treatment significantly inhibited breast cancer cell migration by nearly 62% (MDA-MB-231) compared with cells in their respective control groups (Fig. 4b).
Fig. 4.
a Effects of mangiferin on MDA-MB-231 migration and cell invasion of the basement membrane extract (BME) layer. b Vertical bars represent the wound closure in each treatment group and determined relative to the wound distance at the start of the experiment. The distance migrated was calculated in three or more randomly selected fields per treatment group. *p < 0.05 compared with the respective treated control group. c The vertical bar represents the percent invasion ± SEM. *p < 0.05 compared with the respective vehicle-treated control group
Effects of mangiferin on mammary tumour cell invasion
The cell invasion assay was used to determine the effects of mangiferin on MDA-MB-231 breast cancer cell invasion. The results showed that MDA-MB-231 cells in the treated control groups displayed nearly complete invasion into the BME layer, whereas treatment with 10 μM mangiferin caused an approximate 65% reduction in MDA-MB-231 cell invasion into the BME layer compared with cells in their respective treated control groups (Fig. 4c).
Discussion
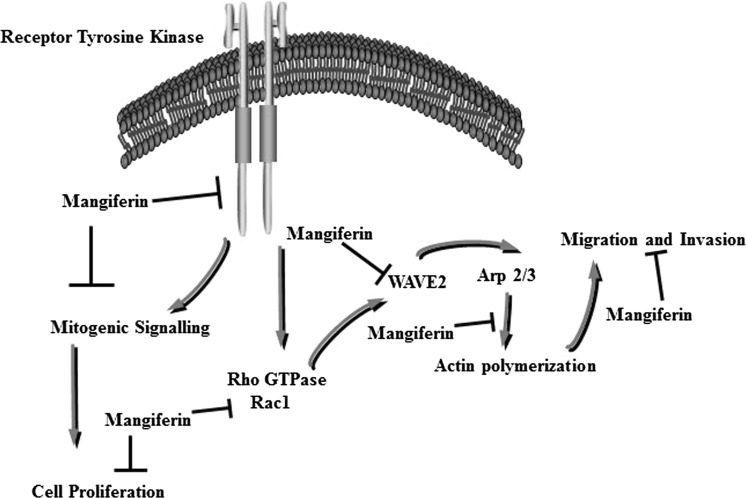
Results from these studies demonstrate that treatment with 4 μM mangiferin induced a significant inhibition in the growth of the highly metastatic MDA-MB-231 mammary tumour cells. However, treatment with higher doses of mangiferin had little or no effect on the growth or viability of immortalized normal human MCF-10 A mammary epithelial cells (data not shown), indicating that cancer cells display selective sensitivity to the antiproliferative effects of mangiferin compared with normal, noncancerous cells. The anticancer effects of mangiferin have previously been described in detail (Rajendran et al. 2013, 2015). Specifically, studies have shown that breast cancer cells exposed to growth-inhibiting doses of mangiferin significantly activated ERα,β and their downstream effectors, including mitogen-activated protein kinase (MAPK), phosphatidylinositol 3-kinase (PI3K)/Akt, Stat, and nuclear factor kappa B (NF-κB) (Wilkinson et al. 2015; Jang et al. 2016; Suchal et al. 2016; Dou et al. 2014; Jeong et al. 2014). Moreover, mangiferin inhibition of c-Met receptor flagging was specifically associated with reduced EMT, as demonstrated by the increased expression of epithelial markers such as E-cadherin, β-catenin, and cytokeratins 8/18 and reduction in vimentin expression, a mesenchymal marker (Ayoub et al. 2011). Thus, Rac1/WAVE2 plays an important role in advancing EMT and metastasis in different tumour cell types (Higgs and Pollard 1999) (Fig. 5).
Fig. 5.
Schematic illustration of the apparent mechanisms involved in mediating the anti-proliferative and anti-metastatic effects of mangiferin
Discoveries in the present study demonstrate that mangiferin treatment causes a noteworthy inhibition of the Rac1-WAVE2-Arp2/3 signalling pathway in MDA-MB-231 breast cancer cells. Immunofluorescence concentrates affirmed these discoveries and demonstrated that comparative treatment with mangiferin brought about a huge lessening in positive WAVE2 staining in these metastatic breast cancer cell lines. Rac1/WAVE2-dependent actin polymerization is required for cytoskeletal-mediated cellular protrusions, membrane ruffling and mobility (Higgs and Pollard 1999; Upadhyaya and van Oudenaarden 2004). Mangiferin treatment fundamentally repressed MDA-MB-231 breast cancer cell growth. Moreover, comparable treatment with mangiferin reduced the viability of these cells as demonstrated by the wound healing assay. These findings suggest that the inhibition of Rac1/WAVE2 flagging by mangiferin is related to reduced cytoskeletal changes, as demonstrated by cell viability and invasion in metastatic breast cancer cells (Saenz-Narciso et al. 2016). Previous studies have investigated the role of WAVE proteins in actin polymerization, membrane protrusions and mobility in breast cancer cells (Desai et al. 2016; Lucato et al. 2015). Investigation of the three WAVE protein expression levels in human breast tumour biopsies indicated abnormal amounts of all WAVE proteins in breast tumours; however, only WAVE2 expression was associated with a highly metastatic phenotype and poor prognosis in patients, although increases in WAVE1 and WAVE3 demonstrated no such connection. Different studies demonstrated that Arp2 and WAVE2 are co-expressed in a high rate of intrusive ductal breast carcinomas but not in adjacent normal tissue (Iwaya et al. 2007). Taken together, the findings in the present study suggest that the inhibitory effects of mangiferin on malignant MDA-MB-231 breast cancer cell migration and invasion appears to be mediated by suppression in Rac1/WAVE2 signalling and indicate that mangiferin may provide some benefit in the treatment of metastatic breast cancer.
References
- Ayoub NM, Bachawal SV, Sylvester PW. Gamma-Tocotrienol inhibits HGF-dependent mitogenesis and Met activation in highly malignant mammary tumour cells. Cell Prolif. 2011;44:516–526. doi: 10.1111/j.1365-2184.2011.00785.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cheng P, Peng ZG, Yang J, Song SJ. The effect of mangiferin on telomerase activity and apoptosis in leukemic K562 cells. Zhong Yao Cai. 2007;30:306–309. [PubMed] [Google Scholar]
- Dar A, Faizi S, Naqvi S, Roome T, Zikr-ur-Rehman S, Ali M, et al. Analgesic and antioxidant activity of mangiferin and its derivatives: the structure activity relationship. Biol Pharm Bull. 2005;28:596–600. doi: 10.1248/bpb.28.596. [DOI] [PubMed] [Google Scholar]
- Desai K, Nair MG, Prabhu JS, Vinod A, Korlimarla A, Rajarajan S, et al. High expression of integrin beta6 in association with the Rho-Rac pathway identifies a poor prognostic subgroup within HER2 amplified breast cancers. Cancer Med. 2016;5:2000–2011. doi: 10.1002/cam4.756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dou W, Zhang J, Ren G, Ding L, Sun A, Deng C, et al. Mangiferin attenuates the symptoms of dextran sulfate sodium-induced colitis in mice via NF-kappaB and MAPK signaling inactivation. Int Immunopharmacol. 2014;23:170–178. doi: 10.1016/j.intimp.2014.08.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duang XY, Wang Q, Zhou XD, Huang DM. Mangiferin: a possible strategy for periodontal disease to therapy. Med Hypotheses. 2011;76:486–488. doi: 10.1016/j.mehy.2010.11.029. [DOI] [PubMed] [Google Scholar]
- Dubey AK, Gupta U, Jain S. Breast cancer statistics and prediction methodology: a systematic review and analysis. Asian Pac J Cancer Prev APJCP. 2015;16:4237–4245. doi: 10.7314/APJCP.2015.16.10.4237. [DOI] [PubMed] [Google Scholar]
- Escudero-Esparza A, Jiang WG, Martin TA. Claudin-5 is involved in breast cancer cell motility through the N-WASP and ROCK signalling pathways. J Exp Clin Cancer Res CR. 2012;31:43. doi: 10.1186/1756-9966-31-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foerster F, Braig S, Moser C, Kubisch R, Busse J, Wagner E, et al. Targeting the actin cytoskeleton: selective antitumor action via trapping PKCvarepsilon. Cell Death Dis. 2014;5:e1398. doi: 10.1038/cddis.2014.363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Rivera D, Delgado R, Bougarne N, Haegeman G, Berghe WV. Gallic acid indanone and mangiferin xanthone are strong determinants of immunosuppressive anti-tumour effects of Mangifera indica L. bark in MDA-MB231 breast cancer cells. Cancer Lett. 2011;305:21–31. doi: 10.1016/j.canlet.2011.02.011. [DOI] [PubMed] [Google Scholar]
- Guha S, Ghosal S, Chattopadhyay U. Antitumor, immunomodulatory and anti-HIV effect of mangiferin, a naturally occurring glucosylxanthone. Chemotherapy. 1996;42:443–451. doi: 10.1159/000239478. [DOI] [PubMed] [Google Scholar]
- Higgs HN, Pollard TD. Regulation of actin polymerization by Arp2/3 complex and WASp/Scar proteins. J Biol Chem. 1999;274:32531–32534. doi: 10.1074/jbc.274.46.32531. [DOI] [PubMed] [Google Scholar]
- Ishihara D, Dovas A, Hernandez L, Pozzuto M, Wyckoff J, Segall JE, et al. Wiskott-Aldrich syndrome protein regulates leukocyte-dependent breast cancer metastasis. Cell Rep. 2013;4:429–436. doi: 10.1016/j.celrep.2013.07.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwaya K, Norio K, Mukai K. Coexpression of Arp2 and WAVE2 predicts poor outcome in invasive breast carcinoma. Mod Pathol. 2007;20:339–343. doi: 10.1038/modpathol.3800741. [DOI] [PubMed] [Google Scholar]
- Jang JH, Lee KH, Jung HK, Sim MO, Kim TM, Woo KW, et al. Anti-inflammatory effects of 6′-O-acetyl mangiferin from Iris rossii Baker via NF-kappab signal blocking in lipopolysaccharide-stimulated RAW 264.7 cells. Chem Biol Interact. 2016;257:54–60. doi: 10.1016/j.cbi.2016.07.029. [DOI] [PubMed] [Google Scholar]
- Jeong JJ, Jang SE, Hyam SR, Han MJ, Kim DH. Mangiferin ameliorates colitis by inhibiting IRAK1 phosphorylation in NF-kappaB and MAPK pathways. Eur J Pharmacol. 2014;740:652–661. doi: 10.1016/j.ejphar.2014.06.013. [DOI] [PubMed] [Google Scholar]
- Kalluri R, Weinberg RA. The basics of epithelial-mesenchymal transition. J Clin Investig. 2009;119:1420–1428. doi: 10.1172/JCI39104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ko HS, Kim JS, Cho SM, Lee HJ, Ahn KS, Kim SH, et al. Urokinase-type plasminogen activator expression and Rac1/WAVE-2/Arp2/3 pathway are blocked by pterostilbene to suppress cell migration and invasion in MDA-MB-231 cells. Bioorg Med Chem Lett. 2014;24:1176–1179. doi: 10.1016/j.bmcl.2013.12.115. [DOI] [PubMed] [Google Scholar]
- Kurisu S, Takenawa T. WASP and WAVE family proteins: friends or foes in cancer invasion? Cancer Sci. 2010;101:2093–2104. doi: 10.1111/j.1349-7006.2010.01654.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lucato CM, Halls ML, Ooms LM, Liu HJ, Mitchell CA, Whisstock JC, et al. The phosphatidylinositol (3,4,5)-trisphosphate-dependent Rac exchanger 1.Ras-related C3 botulinum toxin substrate 1 (P-Rex1.Rac1) complex reveals the basis of Rac1 activation in breast cancer cells. J Biol Chem. 2015;290:20827–20840. doi: 10.1074/jbc.M115.660456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Noratto GD, Bertoldi MC, Krenek K, Talcott ST, Stringheta PC, Mertens-Talcott SU. Anticarcinogenic effects of polyphenolics from mango (Mangifera indica) varieties. J Agric Food Chem. 2010;58:4104–4112. doi: 10.1021/jf903161g. [DOI] [PubMed] [Google Scholar]
- Ozdamar B, Bose R, Barrios-Rodiles M, Wang HR, Zhang Y, Wrana JL. Regulation of the polarity protein Par6 by TGFbeta receptors controls epithelial cell plasticity. Science. 2005;307:1603–1609. doi: 10.1126/science.1105718. [DOI] [PubMed] [Google Scholar]
- Percival SS, Talcott ST, Chin ST, Mallak AC, Lounds-Singleton A, Pettit-Moore J. Neoplastic transformation of BALB/3T3 cells and cell cycle of HL-60 cells are inhibited by mango (Mangifera indica L.) juice and mango juice extracts. J Nutr. 2006;136:1300–1304. doi: 10.1093/jn/136.5.1300. [DOI] [PubMed] [Google Scholar]
- Rajendran P, Jayakumar T, Nishigaki I, Ekambaram G, Nishigaki Y, Vetriselvi J, et al. Immunomodulatory effect of mangiferin in experimental animals with benzo(a)pyrene-induced lung carcinogenesis. Int J Biomed Sci IJBS. 2013;9:68–74. [PMC free article] [PubMed] [Google Scholar]
- Rajendran P, Rengarajan T, Nandakumar N, Divya H, Nishigaki I. Mangiferin in cancer chemoprevention and treatment: pharmacokinetics and molecular targets. J Receptor Signal Transduct Res. 2015;35:76–84. doi: 10.3109/10799893.2014.931431. [DOI] [PubMed] [Google Scholar]
- Saenz-Narciso B, Gomez-Orte E, Zheleva A, Gastaca I, Cabello J. Control of developmental networks by Rac/Rho small GTPases: how cytoskeletal changes during embryogenesis are orchestrated. BioEssays News Rev Mol Cell Dev Biol. 2016;38:1246–1254. doi: 10.1002/bies.201600165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Suchal K, Malik S, Gamad N, Malhotra RK, Goyal SN, Ojha S, et al. Mangiferin protect myocardial insults through modulation of MAPK/TGF-beta pathways. Eur J Pharmacol. 2016;776:34–43. doi: 10.1016/j.ejphar.2016.02.055. [DOI] [PubMed] [Google Scholar]
- Symons M, Segall JE. Rac and Rho driving tumor invasion: who’s at the wheel? Genome Biol. 2009;10:213. doi: 10.1186/gb-2009-10-3-213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takenawa T, Miki H. WASP and WAVE family proteins: key molecules for rapid rearrangement of cortical actin filaments and cell movement. J Cell Sci. 2001;114:1801–1809. doi: 10.1242/jcs.114.10.1801. [DOI] [PubMed] [Google Scholar]
- Upadhyaya A, van Oudenaarden A. Actin polymerization: forcing flat faces forward. Curr Biol CB. 2004;14:R467–R469. doi: 10.1016/j.cub.2004.06.010. [DOI] [PubMed] [Google Scholar]
- Wilhelm I, Fazakas C, Molnar J, Hasko J, Vegh AG, Cervenak L, et al. Role of Rho/ROCK signaling in the interaction of melanoma cells with the blood-brain barrier. Pigment Cell Melanoma Res. 2014;27:113–123. doi: 10.1111/pcmr.12169. [DOI] [PubMed] [Google Scholar]
- Wilkinson AS, Taing MW, Pierson JT, Lin CN, Dietzgen RG, Shaw PN, et al. Estrogen modulation properties of mangiferin and quercetin and the mangiferin metabolite norathyriol. Food Funct. 2015;6:1847–1854. doi: 10.1039/C5FO00133A. [DOI] [PubMed] [Google Scholar]
- Yamaguchi H, Condeelis J. Regulation of the actin cytoskeleton in cancer cell migration and invasion. Biochem Biophys Acta. 2007;1773:642–652. doi: 10.1016/j.bbamcr.2006.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yokotsuka M, Iwaya K, Saito T, Pandiella A, Tsuboi R, Kohno N, et al. Overexpression of HER2 signaling to WAVE2-Arp2/3 complex activates MMP-independent migration in breast cancer. Breast Cancer Res Treat. 2011;126:311–318. doi: 10.1007/s10549-010-0896-x. [DOI] [PubMed] [Google Scholar]