Abstract
Waxy wheat has unique end-use properties; however, its production is limited due mainly to its low grain yield compared with non-waxy wheat. In order to increase its grain yield, it is critical to understand the eco-physiological differences in grain filling between the waxy and non-waxy wheat. In this study, two waxy wheat and two non-waxy wheat cultivars were used to investigate the differences in starch-associated enzymes processes, sucrose and starch dynamics, yield components, and the final grain yield. The results indicated that the mean total grain starch and amylose content, the average 1000-kernel weight and grain yield of the waxy wheat were lower than those of the non-waxy wheat at maturity. The amylose content was significantly and positively correlated with the activity of GBSS (r = 0.80, p < 0.01). Significant positive correlation also exists among activities of AGPase, SSS, GBSS, and SBE, except for GBSS-SBE. In summary, our study has revealed that the reduced conversion of sucrose to starch in the late grain filling stage is the main cause for the low kernel weight and total starch accumulation of the waxy wheat. The reduced conversion also appears to be a factor contributing to the lower grain yield of the waxy wheat.
Introduction
With the growing in health-conscious consumers, there has been an increase in demand for high-quality wheat flour. The endosperm starch content and composition are two critical parameters for evaluating the quality of wheat flour. For conventionally cultivated wheat, namely non-waxy wheat, starch is a mixture of about 75% amylopectin and 25% amylose. To further improve the quality of wheat flour products such as reducing staling in flour products, keeping baking goods fresh for a longer period of time, and improving the palatability of noodles, the replacement of regular wheat flour with waxy wheat flour may provide a promising solution, as waxy wheat grains contain almost 100% amylopectin in their starch1. Common wheat cultivars have three homeologous waxy genes, Wx-A1, Wx-B1 and Wx-D1, and waxy wheat mutants lack all three Wx proteins. The Wx proteins are also known as granule-bound starch synthases, which have a role in the synthesis of amylose1–5. However, due to low grain yield, the waxy wheat has not been widely cultivated in recent years. To improve the current situation, there is an urgent need for improved understanding of the differences in yield performance between the waxy and non-waxy wheat cultivars at the field level.
The endosperm starch content (approximate 70% of the dry weight) not only influences the grain weight and quality6,7, but also reflects the capacity of the sink tissues. Regarding the source-sink relationship, the photoassimilates produced in source tissues as leaf, stem and root were transported into the amyloplast in the form of sucrose, which is the main carbohydrate transported in higher plants. Sucrose is degraded to fructose and uridine diphosphate glucose (UDPG) which are the main precursors of starch synthesis by sucrose synthase (SUS: EC 2.4.1.13)8,9. The ability of the sink tissues to accept and convert photoassimilates can be affected by the sink strength, which is an important limiting factor to wheat grain yield, and the activity of SUS can be considered as an indicator of sink strength8,10,11.
A coordinated series of enzyme-catalyzed reactions in wheat endosperm result in starch synthesis12, including ADPG pyrophosphorylase (AGPase: EC 2.7.7.27), starch branching enzyme (SBE: EC 2.4.1.18), starch debranching enzyme (DBE), granule bound starch synthase (GBSS: EC 2.4.1.21) and soluble starch synthase(SSS: EC 2.4.1.21). GBSS and SSS are the two forms of starch synthase13,14. For catalyzing the first unique step in starch synthesis, AGPase is the rate-limiting enzyme and is the most important determinant of seed sink strength15,16. Starch synthases catalyse the elongation of the linear glucan chains13,17, moreover the different genetic characterization assigns preferential functions for individual isoforms to synthesize amylose or amylopectin. It is generally known that in the endosperm, amylose is synthesized by AGPase and GBSS13. SBE that was formerly known as Q-enzyme plays an important role on amylopectin synthesis, which is the only plant enzyme that can introduce α-1,6-glucosidic linkages into α-polyglucans18,19.
Previous researches on waxy wheat have focused on the end-use quality, composition of the grain and the expression of the Wx genes4,20–22. It is reported that the seed weight of inferior spikelets of rice can be improved by increasing the starch-synthesizing enzymes activities23. In this study, we compared two waxy wheat cultivars with two non-waxy wheat cultivars to investigate the starch content and accumulation, the activities of enzymes responsible for starch biosynthesis in developing grain, 1000-kernel weight and grain yield with the objective to better understand the differences between the wheat varieties and elucidate factors affecting the 1000-kernel weight and grain yield of the waxy wheat.
Materials and Methods
Experimental site
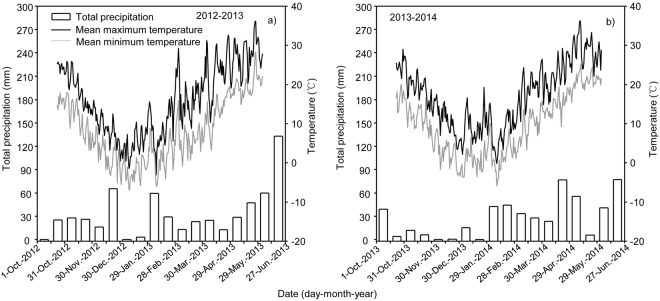
Field experiments were conducted at the Agricultural Experiment Station (32°39′N, 119°25′E) of the Agricultural College of Yangzhou University in China during the winter wheat growing seasons in 2012–2013, and 2013–2014. The field site is situated in a humid subtropical climate zone, with average air temperature of 13 °C to 16 °C, total precipitation of 800 to 1200 mm, total sunshine of 2000 to 2600 h, and a frost-free season of 220 to 240 d. The meteorological data during the wheat growing seasons across the two study years, including temperature and precipitation are shown in Fig. 1. The soil of the site is loamy clay. Pre-planting soil samples were collected prior to any fertilizer applications. The soil before sowing contained 8.38 g kg−1 organic matter, 100.01 mg kg−1 alkali hydrolysable N, 50.11 mg kg−1 Olsen-P, and 149.08 mg kg−1 exchangeable K at 0–20 cm soil depth in 2012, while 19.57 g kg−1 organic matter, 107.97 mg kg−1 alkali hydrolysable N, 33.92 mg kg−1 Olsen-P, and 124.23 mg kg−1 exchangeable K at 0–20 cm soil depth in 2013.
Figure 1.
Mean maximum temperature, mean minimum temperature and total precipitate during the wheat growing season of wheat in 2012–2013 (a) and 2013–2014 (b).
Materials
A total of four wheat cultivars were tested, including two types of waxy wheat cultivars Yangnuomai1 (YN1), Ningnuomai1 (NN1), [which lack functional Wx-A1, Wx-B1, Wx-C1 alleles based on a PCR test (data not shown)] and two non-waxy wheat cultivars Yangmai20 (Y20) and Yangfumai4 (YF4), (contained three functional Wx genes).
Crop management
All plots were supplied with 240 kg N ha−1, applied with the ratio 5:1:2:2 at four stages: pre-sowing, four-leaf stage, jointing stage and booting stage, respectively. Application rate of P2O5 (P) and K2O (K) were both 144 kg ha−1: half the amounts of P and K were applied before sowing and the remaining half were applied at the jointing stage. The seeds were sown at a planting density of 225 × 104 ha−1 with 30 cm row spacing on 3 November of 2012 and 28 October of 2013. Seedling number was maintained at 225 × 104 ha−1 by removing or adding seedlings at the three-leaf stage. Plants were harvested on 3 June of 2013 and 28 May of 2014. Weeds, insects and diseases were controlled as required to avoid yielding loss; other production management practices were based on those used by local farmers.
Sampling and measurements
Measurement of grain yield, components and grain volume
At maturity, an area of 1.2 × 1 m (4 rows included) was harvested manually for the determination of the grain yield. Grain yield was adjusted to 13% moisture. Number of spikes was counted manually for two rows within 1 m. The number of kernels per spike was counted manually to calculate the mean value from 50 continuous spikes. For 1000-kernel weight, 1000 kernels were randomly selected and weighed. The average seed volume was determined via the displacement method24.
Measurement of the starch, amylose, and amylopectin content
The total starch, amylose, and amylopectin contents were determined via the dual wavelength iodine binding method25,26. Wheat grains that were marked flowering on the same day were first ground using a mortar, and the powder was then degreased twice with anhydrous ether. A 100 mg fraction of each sample was used to determine amylose and amylopectin contents. A calibration curve was derived using pure amylose from potato (A0512; Sigma–Aldrich, St. Louis, MO, USA) and pure amylopectin from potato (A8515; Sigma–Aldrich). The sum of amylose and amylopectin contents was designated as the total starch content.
Measurement of the activities of AGPase, GBSS, SBE and SSS
Twenty kernels weighed at different filling stages were tested. AGPase activity was assayed according to Nakamura and Cheng27,28. SSS and GBSS activities were assayed according to Nakamura and Cheng27,28. SBE activity was assayed according to Li et al.29.
Measurement of the sucrose content
The sucrose content was measured by the resorcinol technique30,31. Ten dried spikes those were flowered on the same day were selected and killed at 105 °C for 30 min and then dried at 80 °C for measurements of grain weight and starch content. And then they were weighed, and then the dried endosperm was ground into powder for analysis. Samples of the powdered endosperm prepared as mentioned above (100 mg) were extracted with 8 mL of 80% ethanol at 80 °C for 30 min, followed by two extractions with 8 mL of 80% ethanol. The supernatants were combined and purified by 10 g activated carbon overnight, fixed to a constant volume at 50 ml, and purified by filtering. One-hundred microliters of 2 M sodium hydroxide was added to 0.9 ml filtrate, boiled at 100 °C for 10 min. The mixture was cooled to room temperature, then 1 ml of 0.1% resorcinol and 3 ml of 10 mol/L hydrochloric acid were added. The reaction mixture was boiled at 80 °C for 60 min, cooled to room temperature, and the sucrose content was determined by measuring OD at 500 nm.
Measurement of the activity of Sucrose Synthase (SUS)
The SUS activity was assayed according to Rufty, Doehlert and Wardlaw32–34.
Statistical analysis
All data were checked for normality based on the Kolmogorov–Smirnov test with SAS proc mixed (SAS V.9.1). The effects of year, cultivar and their interaction on test parameters were tested with year, cultivar as fixed effects, year as repeated measurements, and block as a random effect. Differences among least square means (LSMEANS) for all treatment pairs were tested by the LSD procedure at a significance level of P = 0.05. Pearson’s correlation analysis (SAS V.9.1) was performed to establish the relationships among all measured properties. The ANOVA (α = 0.05) was performed to identify significant differences between treatments, multiple comparisons between means (SAS V.9.1) were made using the least significant difference (LSD0.05) test if the F-tests were significant.
Results
Sucrose, total starch, amylose and amylopectin contents in grains among waxy and non-waxy wheat
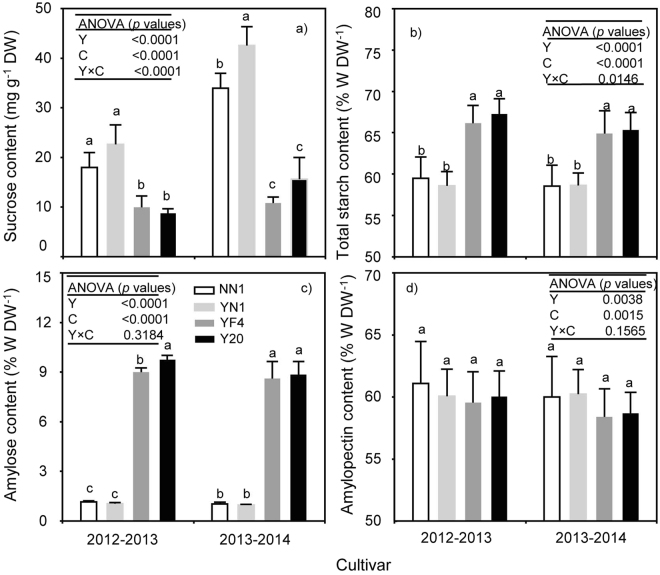
The test found that grain sucrose content was significantly affected by cultivar, year and their interactions (Fig. 2a). Averaged to cultivars, the sucrose content of waxy wheat was 117.67% and 189.82% higher than that of non-waxy wheat (2012–13 and 2013–14, respectively). Similarly, total starch content was strongly governed by cultivar, year and their interactions (Fig. 2b). The waxy wheat had 14.13% and 12.41% lower total starch content in the 2012–13 and 2013–14 growing seasons respectively, when compared to the non-waxy wheat. Separate effect of year and cultivar significantly affected amylose and amylopectin content, while the interaction of year and cultivar to the amylase and amylopectin content was not significant (Fig. 2c,d). The average amylose contents under waxy wheat were 88.09% and 88.46% lower than that in non-waxy wheat in 2012–13 and 2013–14 seasons respectively. No significant differences in amylopectin levels were detected between waxy and non-waxy wheat cultivars.
Figure 2.
Grain content of sucrose (a), total starch (b), amylose (c) and amylopectin (d) in grains between waxy and non-waxy wheats. Values followed by the same letters in each cultivar are not significantly different at p < 0.05 level. Each bar represents the SD value.
Changes in total starch, amylose, and amylopectin accumulation and accumulation rate in grains of waxy and non-waxy wheat
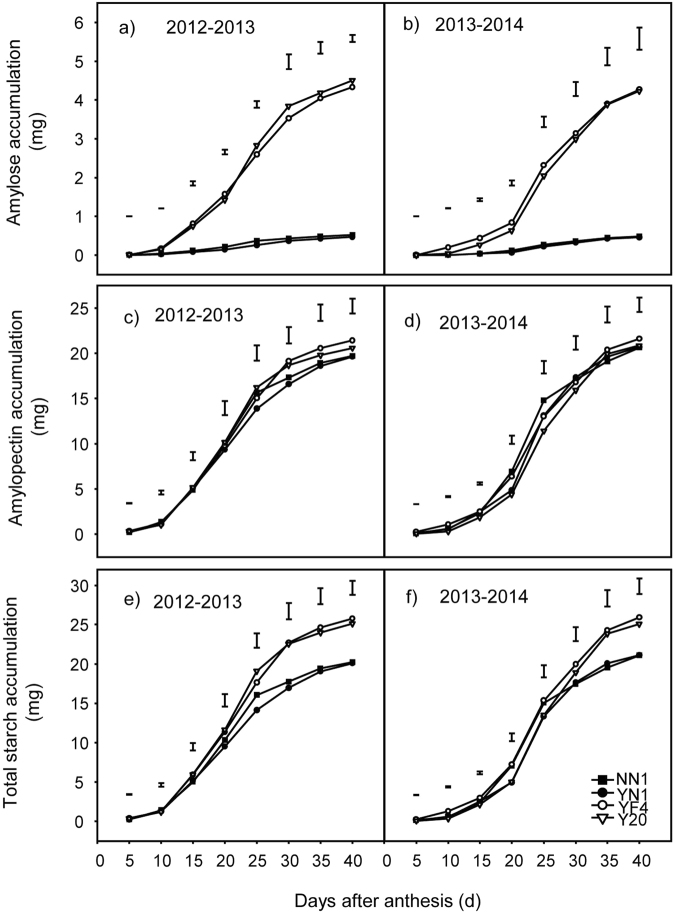
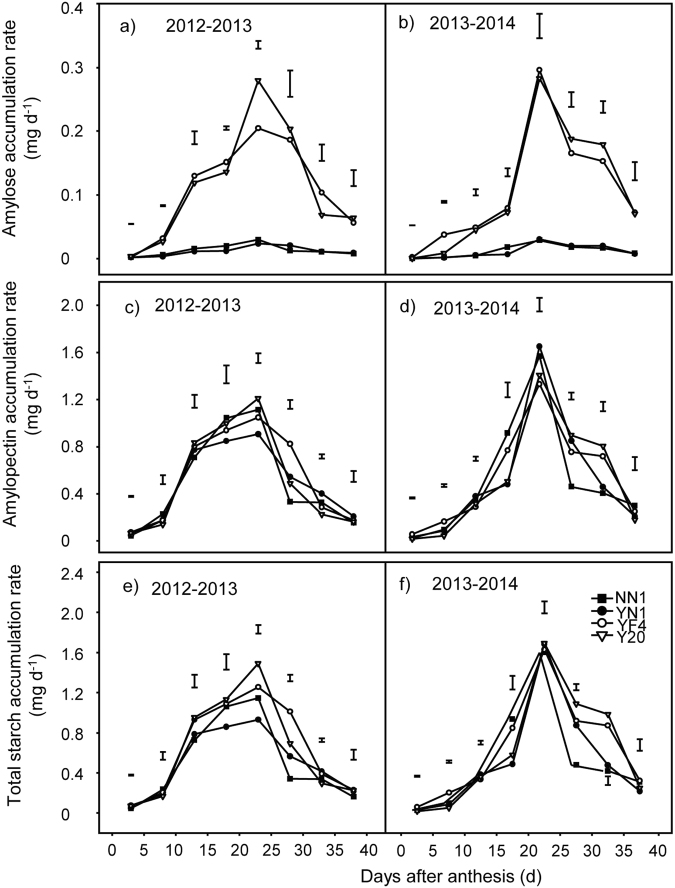
As shown in Fig. 3, the grain total starch, amylose, and amylopectin accumulation in waxy and non-waxy wheat showed overall increases during the grain-filling stage in both test years. The total starch (Fig. 3e,f) and amylose accumulation (Fig. 3a,b) were generally lower in waxy wheat compared with non-waxy wheat, particularly after day 25 after anthesis. There was no significant difference observed on amylopectin accumulation between the waxy and non-waxy wheat over both years (Fig. 3c,d). The greatest rates of accumulation for grain total starch, amylose, and amylopectin occurred 10th–25th days after anthesis, but decreased after the 25th day after anthesis for both types of wheat (Fig. 4). The amylose accumulation rate in waxy wheat was significantly lower than that in non-waxy wheat (Fig. 4a,b). At 25 days after anthesis, the accumulation rate of total starch in waxy wheat was lower than that in non-waxy wheat (Fig. 4e,f). No significant difference was observed in amylopectin accumulation rate between waxy and non-waxy wheat (Fig. 4c,d). At maturity, the rates of accumulation for total starch, amylose, and amylopectin declined and the accumulation of total starch, amylose, and amylopectin reached the maximum levels.
Figure 3.
Changes in grain accumulation of amylose (a,b), amylopectin (c,d) and total starch (e,f) in one kernel between waxy and non-waxy wheats after anthesis. Each bar represents the LSD value at p < 0.05.
Figure 4.
Changes in the accumulation rate of amylose (a,b), the amylopectin (c,d) and total starch (e,f) in one kernel between waxy and non-waxy wheats after anthesis. Each bar represents the LSD value at p < 0.05.
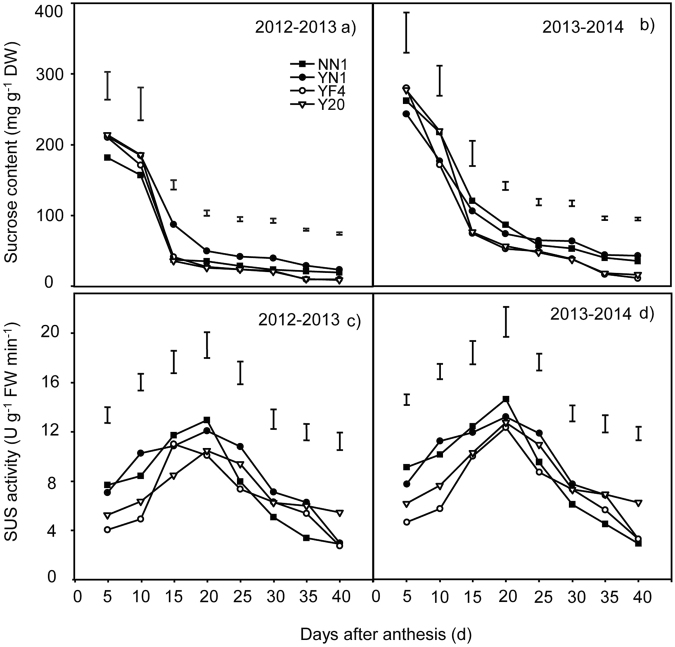
Changes in the sucrose content and SUS activity of grains in waxy and non-waxy wheat
The grain sucrose content of both waxy and non-waxy wheats declined gradually after anthesis during both growing seasons (Fig. 5a,b). The grain sucrose content of waxy wheat tended to be higher than that of non-waxy wheat beginning ten days after anthesis. As shown in Fig. 5c and d, the SUS activities of the waxy wheat cultivars were higher than that of the non-waxy wheat cultivars up to day 20. Afterwards, the SUS activities of all cultivars dropped
Figure 5.
Changes in sucrose content (a), and SUS activity in grains between waxy and non-waxy wheats after anthesis. Each bar represents the LSD value at p < 0.05.
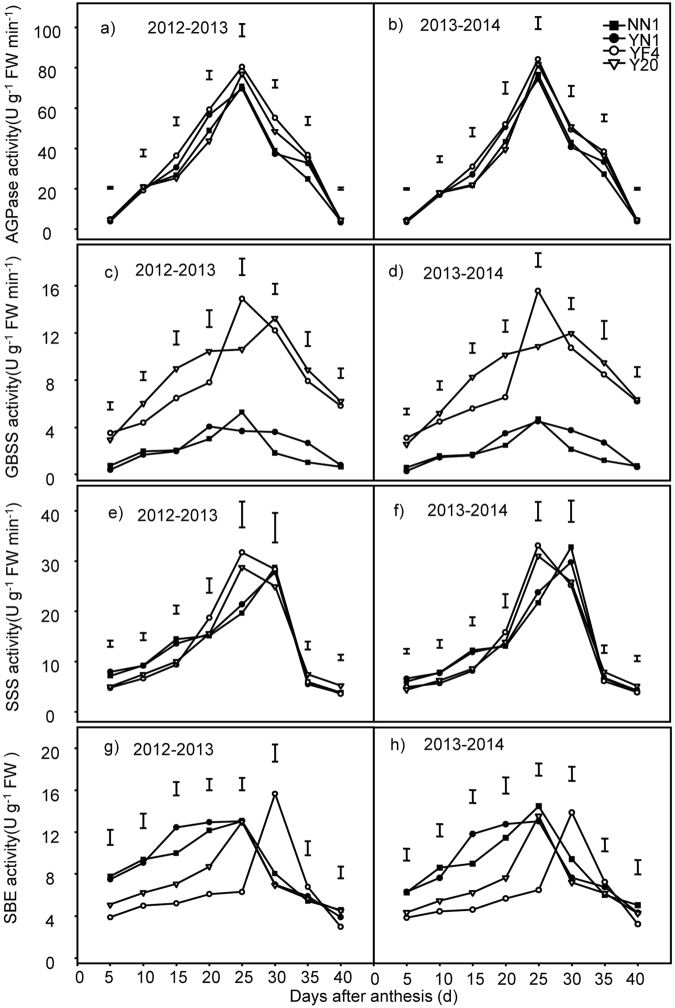
Changes in the AGPase, GBSS, SSS and SBE activities of grains in waxy and non-waxy wheat
For both the waxy and non-waxy wheat cultivars over both years, the activities of four major enzymes (AGPase, GBSS, SBE and SSS) increased during early grain filling period and decreased during the late period after anthesis. The peak activities of the AGPase and GBSS occurred on the 25th day after anthesis; while for SBE and SSS, the peak activities happened during the period of the 25th to 30th day (Fig. 6). The AGPase in waxy wheat were significantly lower than those in non-waxy wheat, especially after the 20th day after anthesis. Throughout the grain filling, the GBSS activities in waxy wheat were significantly lower than those in non-waxy wheat. In contrast, the activity for SBE was higher in waxy wheat than that in non-waxy wheat before the 25th day after anthesis; this trend was reversed after the 25th day after anthesis. The SSS activity was higher in waxy wheat than that in non-waxy wheat between the 20th and the 30th day after anthesis.
Figure 6.
Changes of AGPase (a,b), GBSS (c,d), SSS (e,f) and SBE (g,h) activity in wheat grains between waxy and non-waxy wheats. Each bar represents the LSD value at p < 0.05.
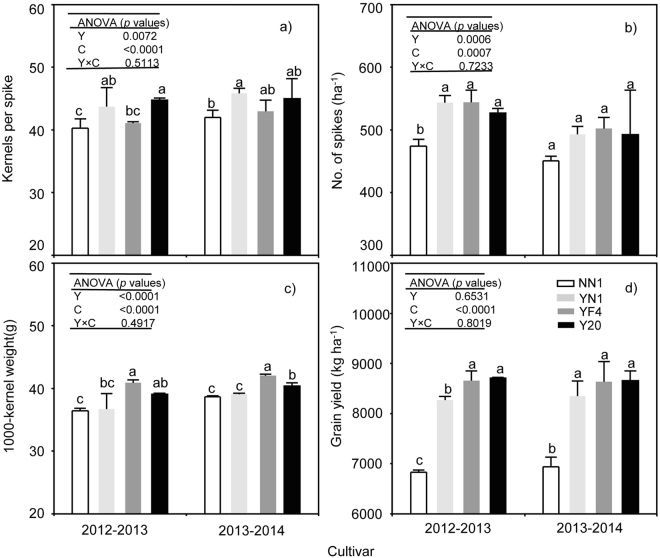
Grain yield, components and grain volume
The year and cultivar significantly affected the number of kernels per spike (py = 0.0072, pc < 0.0001), number of spikes (py = 0.0006, pc = 0.0007) and the 1000-kernel weight (py < 0.0001, pc < 0.0001); however, the interaction between year and cultivar was not significant. For the grain yield, a significant cultivar effect was detected (pc < 0.0001), but not for year and year-cultivar interaction.
The number of kernels per spike in YN1 and Y20 were higher than that in NN1 and YF4 for both test years (Fig. 7a). The lowest value of spike number was observed in NN1 in two years, but there was no significant difference between cultivars in 2013–2014 (Fig. 7b). The 1000-kernel weight and grain yield in waxy wheat were lower than those in non-waxy wheat and in 2013–2014 the difference was significant (Fig. 7c,d). Across cultivars, the kernels per spike, number of spikes, 1000-kernel weight and grain yields of waxy wheat were reduced by 2.22, 5.03, 8.45, 13.02% and 0.11, 5.15, 5.70, 11.62% respectively in 2012–13 and 2013–14 growing seasons, compared with the non-waxy wheat. Trying to verify our hypothesis, we test the grain volume among wheat cultivars in the 2013–2014season,and we found there was no significant difference among wheat cultivars(Fig. 8).
Figure 7.
Kernels per spike (a), Number of spikes (b), 1000-kernel weight (c) and grain yield (d) between waxy and non-waxy wheats. Values followed by the same letters in each cultivar are not significantly different at p < 0.05 level. Each bar represents the LSD value at p < 0.05.
Figure 8.
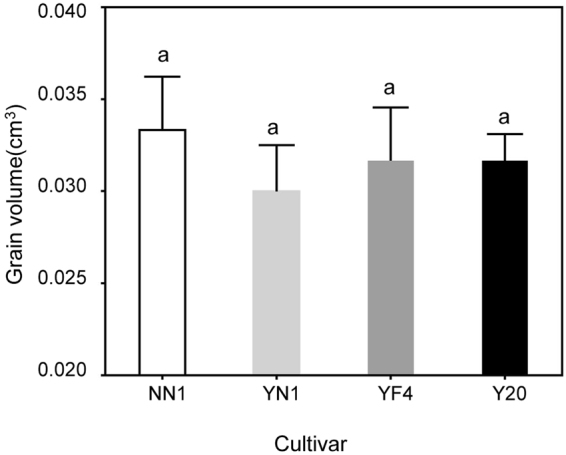
Grain volume between waxy and non-waxy wheats. Values followed by the same letters in each cultivar are not significantly different at p < 0.05 level.
Correlation analysis
The total starch content in grains was not correlated with the 1000-kernel weight, but amylose was significantly and positively correlated with the 1000-kernel weight (Table 1). Amylopectin and sucrose content did not significantly correlate with the 1000-kernel weight. The total starch and amylopectin accumulation rate were significantly and positively linked with the activities of all five enzymes (r > 0.4, p < 0.01), while the amylose content did not significantly correlate with other enzyme activities except for GBSS (Table 2). The total starch content, amylopectin content and amylose accumulation rate were significantly and positively correlated with AGPase, SSS and GBSS activities. In addition, significant correlations were detected between the four enzymatic activities (i.e. AGPase, SSS, GBSS and SBE), with the exception of GBSS-SBE (Table 3).
Table 1.
Pearson Correlation among 1000-kernel weight and contents of sucrose, total starch and starch components in wheat grains.
| 1000-kernel weight | ||
|---|---|---|
| r value | p value | |
| Sucrose | −0.35 | 0.40 |
| Total starch | 0.69 | 0.06 |
| Amylose | 0.75 | 0.03 |
| Amylopectin | −0.63 | 0.09 |
Table 2.
Pearson Correlation analysis on total starch accumulation rate, total starch accumulation with the enzyme activities in grains
| Total starch content | Amylose content | Amylopectin content | Total starch accumulation rate | Amylose accumulation rate | Amylopectin accumulation rate | |
|---|---|---|---|---|---|---|
| SUS | −0.26* | −0.21ns | −0.24ns | 0.43** | 0.06ns | 0.47** |
| AGPase | 0.43** | 0.24ns | 0.43** | 0.90** | 0.60** | 0.90** |
| SSS | 0.36** | 0.18ns | 0.37** | 0.73** | 0.55** | 0.72** |
| GBSS | 0.48** | 0.80** | 0.35** | 0.61** | 0.90** | 0.51** |
| SBE | 0.08ns | −0.13ns | 0.12ns | 0.60** | 0.16ns | 0.65** |
*Significant at p < 0.05.
**Significant at p < 0.01.
nsNot Significant.
Table 3.
Pearson Correlation analysis among activities of the enzyme in grains.
| AGPase | SSS | GBSS | SBE | |
|---|---|---|---|---|
| AGPase | 1 | |||
| SSS | 0.82** | 1 | ||
| GBSS | 0.59** | 0.51** | 1 | |
| SBE | 0.66** | 0.59** | 0.11ns | 1 |
*Significant at p < 0.05.
**Significant at p < 0.01.
nsNot Significant.
Discussion
Grain yield an sink strength
The inherent low grain yield of the waxy wheat was reported in previous study35,36, and it has limited its commercial production. Improving the agronomic performance is critical in order to meet human demands on high-quality wheat products. In practice, grain yield is determined not only by factors such as the number of spikes, number of kernels per spike and 1000-kernel weight but also by the source-sink relationships8. Two key stages, enlargement and grain filling, are responsible for seed size and weight12,37.
The sink strength is formed by sink size (i.e. endosperm cell numbers) and sink activity as indicated by activities of the enzymes such as SUS and AGPase in endosperm, which is closely associated with carbohydrate utilization and storage38. Gleadow et al.39 reported that the endosperm cell number was positively correlated with grain size. Sucrose is the main photoassimilate transported to sink tissues, and then the degradation from sucrose to materials for starch synthesis reflects the sink strength in grains40. Previous studies have shown that the grain dry weight is potentially determined by sink strength in wheat39 and rice38. Enhancement of the sink size and sink activity are two critical factors for increasing sink strength. In this study, there was no significant difference in grain volume between the waxy and non-waxy wheat. However, the 1000-kernel weight of waxy wheat was significantly lower than that of non-waxy wheat.
Sucrose content and SUS activity
The conversion of sucrose to starch in wheat grain was closely related to starch accumulation, for example, high starch accumulation was observed when the conversion of sucrose to starch was high41. Moreover, early studies have found that waxy wheats generally have higher sucrose content and lower transforming ability compared with non-waxy wheat42. The sucrose content is significantly correlated with the starch synthesis in the early period of grain filling (before 25th days after anthesis) in wheat43 and rice44. The SUS plays an important role in sucrose synthesis and degradation in plants, especially in wheat grains; It is generally believed that the main function of the SUS is to catalyze the degradation of sucrose in grains45,46. The decrease of SUS activity leads to lower rate of conversion from sucrose to starch and a high residual sucrose content43, and further resulting in a lower dry matter accumulation characteristic of rice grains47,48. In this study, we recorded significant differences in the sucrose content and SUS activity in grains among waxy and non-waxy wheat cultivars after anthesis. Throughout the grain filling, the grains of waxy wheat had higher sucrose content, but SUS activity in grains was different between the earlier and later stage after anthesis. Our results suggested that waxy wheat had higher sucrose degradation and bioavailability during early grain filling stages, but was lower at the later stage compared with non-waxy wheat. More sucrose and less grain starch synthesis ability could be characteristic of waxy wheat in comparison to non-waxy wheat especially in the late period after anthesis. These results were in agreement with some previous studies conducted in waxy20,42 and non-waxy43,46 wheats.
Starch accumulation and starch-synthesizing enzymes activities
Starch is synthesized and accumulated during the grain-filling process49, which is the major factor influencing both grain yield and quality7,12. The first unique step of the starch synthesis is catalyzed by AGPase, which is considered to be the rate-limiting enzyme because of the positive correlation between the activity and starch accumulation rate8,28,50,51. In this study, AGPase activity increased at the early grain filling stage, leading to increased accumulation of the total starch, amylose and amylopectin, as well as their accumulation rate. We also found that the AGPase activity in waxy wheat was lower than that in non-waxy wheat in the whole grain filling period, resulting in lower total starch content in waxy wheat seeds compared with non-waxy wheat. At 30d after anthesis, the AGPase activity started to decrease, resulting in a reduction in the rate of starch, amylose and amylopectin accumulation. During the later grain filling period, the starch accumulation and accumulation rate of waxy wheat were lower than that of non-waxy wheat. Similar findings have also been reported in previous studies: in other crops higher AGPase activity resulted in higher crop starch biosynthesis and grain yield including maize52, rice53 and wheat16,54.
GBSS is exclusively involved in the synthesis of amylose, where it catalyzes the extension of long glucans within the amylopectin fraction15,17. The absence of waxy protein resulting from waxy mutations in wheat and other cereals could lead to amylose-free starch in endosperm, with low activity of GBSS in waxy wheat1,2,55. GBSS activity determines amylose content in the endosperm of rice56 and wheat57. In this study, the GBSS activity of waxy wheat was significantly lower than that of non-waxy wheat throughout the grain filling period; the amylose accumulation and accumulation rate in waxy wheat were also lower than that in non-waxy wheat. These results indicated that waxy wheat had lower grain starch and amylose synthesis capacity compared with non-waxy wheat, particularly in the case of amylose. Moreover, GBSS is a critical component in the process of amylose formation58; some studies have also reported that reduction of amylose content and GBSS activity has been observed in many species that lack amylase and all of these specifically lack GBSSI activity14. GBSS is a critical enzyme in the process of amylose biosynthesis58 and its activity is closely associated with amylose content as observed in many species that lack amylase and GBSS, specifically the GBSSI activity14.
The SSS and SBE play an important role in the formation of the branched structure of amylopectin molecules17,28,59. Amylose, amylopectin and total starch accumulation rate in wheat grains were significantly and positively correlated with activities of SBE, SSS and GBSS60, and a reduction in enzyme activity appears to be the main factor affecting decreased starch synthesis43. In this study, enzymatic activities of SSS and SBE were higher in waxy wheat than in non-waxy wheat, while amylopectin accumulation and amylopectin accumulation rate was significantly higher in waxy wheat compared with the non-waxy wheat during early grain filling stages. During the late grain filling stage, activities of both enzymes were lower, while the amylopectin accumulation and accumulation rate decreased more quickly in waxy wheat compared with non-waxy wheat. These results were similar to those of Tan et al.61 who reported that lower activities of AGPase, SSS, GBSS and SBE and the lower accumulation of amylose, amylopectin and starch in waxy wheat compared with non-waxy wheat and also in lower starch accumulation cultivar had lower enzyme activities in non-waxy wheat58.The SBE activity did not significantly influence the amylopectin content but significantly positively related to the amylopectin accumulation rate, it is postulated that SBE might not play an important role in the formation of amylopectin, but to its fine structure modification, it is stated in maize for the unique function of SBEI in modulating the branching pattern in normal starch by decreasing local clustering of amylopectin branch points62.
Relationship between enzymes activity and kernel weight
Waxy wheat was found to have lower starch content and higher sugar content than non-waxy wheat22,42, and also lower starch, amylose and amylopectin accumulation and accumulation rate than non-waxy wheat in the grain filling period42,61. It is possible that higher soluble sugar content and lower starch content were due to a low conversion efficiency and the absence of GBSS at the period of grain filling because these two factors have been shown to influence grain weight in waxy rice63 and in waxy wheat20. Higher activities of SBE, APGase, SSS and GBSS are generally attributed to increased kernel weight in wheat, while starch accumulation rate is significantly correlated with activities of SBE, AGPase, SSS and GBSS in both waxy wheat61 and non-waxy wheat41. Seed sink strength and seed yield would all be expected to be enhanced by increasing the AGPase activity16. The activities of AGPase, SSS, GBSS and SUS play important roles in determining individual grain weight in wheat by regulating starch synthesis in grain endosperm64. Further, many studies show that there is a coordinating action in starch biosynthesis among these enzymes60. The four important enzymes for starch biosynthesis played the important role but not the whole starch biosyntheses net.
In this study, grain yield appeared to be influenced by sucrose and starch factors. The total starch and amylose content at maturity in grains of waxy wheat were 13.29 and 88.27% lower respectively than those of non-waxy wheat. The 1000-kernel weight of waxy wheat was 7.05% lower than non-waxy wheat, and the grain yield of waxy wheat was 12.33% lower than that of non-waxy wheat. Reduced activities of AGPase, SSS, GBSS and SBE and a lower accumulation of amylose, amylopectin and starch were also observed during the later stage of grain filling in waxy wheat compared with non-waxy wheat. The amylose content was significantly and positively correlated with GBSS activity, while amylose was significantly and positively correlated with the 1000-kernel weight. The total starch content and amylopectin content were significantly and positively correlated with AGPase, SSS and SBE activities. Among the activities of AGPase, SSS, GBSS and SBE, there were significant correlations, except between the activities of GBSS and SBE.
Conclusions
Our study suggests that low AGPase activity could be the cause of low total starch content in waxy wheat grain, and low GBSS activity could be the contributing factor to insufficient amylose content. During the early grain filling period, no significant difference was found in sink size between the waxy and non-waxy wheat, while higher sucrose degradation and bioavailability and activities of SSS and SBE in grains of waxy wheat were found. Low activities of SSS and SBE in waxy wheat are likely the cause of low amylopectin accumulation in the later stage of grain filling. We speculate that the four enzymes, AGPase, SSS, GBSS and SBE, may have a complementary relationship with regards to starch biosynthesis; thus, the absence of GBSS can reduce the activity of the other three enzymes, leading to low total starch content in grains of waxy wheat. The low activity of SUS during later period of grain filling may have caused the reduced degradation of sucrose and the absence of the precursor of starch in grains of waxy wheat. It appears that weakened starch synthesis in the later stage of grain filling of waxy wheat is likely to be the main cause of low kernel weight and total starch accumulation, which ultimately leads to lower grain yield in waxy wheat.
Acknowledgements
This study was supported by the grants from the National Key Research and Development Program of China (2016YFD0300406), Independent Innovation Program of Jiangsu Province (CX(15)1002), Outstanding Talents Support Program of Yangzhou University, and the Priority Academic Program Development of Jiangsu Higher Education Institutions.We appreciate Dr. Lianne Dwyer [Agriculture and Agri-Food Canada (AAFC)], Dr. Jiali Shang (AAFC), Dr. Linda Langille (AAFC), Prof Min Sun (Shanxi Agricultural University) and Dr. Fei Mo (Lanzhou University) for their help of the latest version of the manuscript.
Author Contributions
W.S.G.,Y.X.P. and Y.Z. conceived the research, designed and performed the experiments, and prepared and revised the manuscript. Y.Z., J.F.D., X.K.Z. and C.Y.L analyzed the data. G.H. and J.M.S. provided the writing and editing of the manuscript. All authors discussed the results and commented on the manuscript.
Competing Interests
The authors declare no competing interests.
Footnotes
Yan Zi and Jinfeng Ding contributed equally to this work.
A correction to this article is available online at https://doi.org/10.1038/s41598-018-25219-9.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Change history
5/1/2018
A correction to this article has been published and is linked from the HTML version of this paper. The error has not been fixed in the paper.
References
- 1.Nakamura T, Yamamori M, Hirano H, Hidaka S, Nagamine T. Production of Waxy (Amylose-Free) Wheats. Molecular and General Genetics MGG. 1995;248:253–259. doi: 10.1007/BF02191591. [DOI] [PubMed] [Google Scholar]
- 2.Yamamori M, Nakamura T, Endo TR, Nagamine T. Waxy Protein Deficiency and Chromosomal Location of Coding Genes in Common Wheat. Theoretical and Applied Genetics. 1994;89:179–184. doi: 10.1007/BF00222461. [DOI] [PubMed] [Google Scholar]
- 3.Yoo S-H, Jane J-l. Structural and Physical Characteristics of Waxy and Other Wheat Starches. Carbohydrate Polymers. 2002;49:297–305. doi: 10.1016/S0144-8617(01)00338-1. [DOI] [Google Scholar]
- 4.Ainsworth C, Clark J, Balsdon J. Expression, Organisation and Structure of the Genes Encoding the Waxy Protein (Granule-bound Starch Synthase) in Wheat. Plant Molecular Biology. 1993;22:67–82. doi: 10.1007/BF00038996. [DOI] [PubMed] [Google Scholar]
- 5.Nakamura T, Yamamori M, Hirano H, Hidaka S. Identification of Three Wx Proteins in Wheat (Triticum Aestivum L.) Biochemical Genetics. 1993;31:75–86. doi: 10.1007/BF02399821. [DOI] [PubMed] [Google Scholar]
- 6.Dale EM, Housley TL. Sucrose Synthase Activity in Developing Wheat Endosperms Differing in Maximum Weight. Plant Physiology. 1986;82:7–10. doi: 10.1104/pp.82.1.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hurkman WJ, et al. Effect of Temperature on Expression of Genes Encoding Enzymes for Starch Biosynthesis in Developing Wheat Endosperm. Plant Science. 2003;164:873–881. doi: 10.1016/S0168-9452(03)00076-1. [DOI] [Google Scholar]
- 8.Tuncel A, Okita TW. Improving Starch Yield in Cereals by Over-Expression of ADPglucose Pyrophosphorylase: Expectations and Unanticipated Outcomes. Plant Science. 2013;211:52–60. doi: 10.1016/j.plantsci.2013.06.009. [DOI] [PubMed] [Google Scholar]
- 9.Keeling PL, Wood JR, Tyson RH, Bridges IG. Starch Biosynthesis in Developing Wheat Grain: Evidence Against the Direct Involvement of Triose Phosphates in the Metabolic Pathway. Plant Physiology. 1988;87:311–319. doi: 10.1104/pp.87.2.311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wang Z, et al. Effects of Source Sink Manipulation on Production and Distribution of Photosynthate after Flowering in Irrigated and Rainfed Wheat. Acta Agronomica Sinica. 1999;25:162–168. [Google Scholar]
- 11.Miralles DJ, Slafer GA. Sink Limitations to Yield in Wheat: How Could It Be Reduced? Journal of Agricultural Science. 2007;145:139–149. doi: 10.1017/S0021859607006752. [DOI] [Google Scholar]
- 12.Emes MJ, et al. Starch Synthesis and Carbon Partitioning in Developing Endosperm. Journal of Experimental Botany. 2003;54:569–575. doi: 10.1093/jxb/erg089. [DOI] [PubMed] [Google Scholar]
- 13.Jeon J-S, Ryoo N, Hahn T-R, Walia H, Nakamura Y. Starch Biosynthesis in Cereal Endosperm. Plant Physiology and Biochemistry. 2010;48:383–392. doi: 10.1016/j.plaphy.2010.03.006. [DOI] [PubMed] [Google Scholar]
- 14.Denyer K, Johnson P, Zeeman S, Smith AM. The Control of Amylose Synthesis. Journal of Plant Physiology. 2001;158:479–487. doi: 10.1078/0176-1617-00360. [DOI] [Google Scholar]
- 15.Preiss, J. Biology and Molecular Biology of Starch Synthesis and Its Regulation. Oxford Surveys of Plant Molecular and Cell Biology (1991).
- 16.Smidansky ED, et al. Enhanced ADP-Glucose Pyrophosphorylase Activity in Wheat Endosperm Increases Seed Yield. Proceedings of the National Academy of Sciences of the United States of America. 2002;99:1724–1729. doi: 10.1073/pnas.022635299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tetlow LJ. Understanding Storage Starch Biosynthesis in Plants: a Means to Quality Improvement. Canadian Journal of Botany. 2006;84:1167–1185. doi: 10.1139/b06-089. [DOI] [Google Scholar]
- 18.Nakamura Y. Towards a Better Understanding of the Metabolic System for Amylopectin Biosynthesis in Plants: Rice Endosperm as a Model Tissue. Plant and Cell Physiology. 2002;43:718–725. doi: 10.1093/pcp/pcf091. [DOI] [PubMed] [Google Scholar]
- 19.Han Y, Sun F-J, Rosales-Mendoza S, Korban SS. Three Orthologs in Rice, Arabidopsis, and Populus Encoding Starch Branching Enzymes (SBEs) are Different From Other SBE Gene Families in Plants. Gene. 2007;401:123–130. doi: 10.1016/j.gene.2007.06.026. [DOI] [PubMed] [Google Scholar]
- 20.Gao D, et al. Characteristics of a Sugar Degradation and Starch Accumulation in Waxy Wheat Grains After Anthesis. Scientia Agricultura Sinica. 2007;40:1890–1897. [Google Scholar]
- 21.Nakamura T, Yamamori M, Hirano H, Hidaka S. Decrease of Waxy (Wx) Protein in Two Common Wheat Cultivars with Low Amylose Content. Plant Breeding. 1993;111:99–105. doi: 10.1111/j.1439-0523.1993.tb00614.x. [DOI] [Google Scholar]
- 22.Ni J, Xu Z, Wang T. Dynamic Changes of GrainSugar, Starch, Protein Contents during Grain Filling Stage in Waxy Wheat. Journal of Triticeae Crops. 2010;30:509–514. [Google Scholar]
- 23.Mohapatra PK, Sarkar RK, Kuanar SR. Starch synthesizing enzymes and sink strength of grains of contrasting rice cultivars. Plant Science. 2009;176:256–263. doi: 10.1016/j.plantsci.2008.11.001. [DOI] [Google Scholar]
- 24.Yan L, et al. Study on Grain Volume and Filling Fullness in Wheat. Acta Agronomica Sinica. 1995;21:637–640. [Google Scholar]
- 25.Zhu T, Jackson DS, Wehling RL, Geera B. Comparison of Amylose Determination Methods and the Development of a Dual Wavelength Iodine Binding Technique. Cereal Chemistry. 2008;85:51–58. doi: 10.1094/CCHEM-85-1-0051. [DOI] [Google Scholar]
- 26.He, Z. Grain Quality and Its Analysis Technology. (Beijing: Agriculture press, 1985).
- 27.Cheng F, Jiang D, Wu P, Shi C. The Dynamic Change of Starch Synthesis Enzymes During the Grain Filling Stage and Effects of Temprature upon It. Acta Agronomica Sinica. 2001;27:201–206. [Google Scholar]
- 28.Nakamura Y, Yuki K, Park S-Y, Ohya T. Carbohydrate Metabolism in the Developing Endosperm of Rice Grains. Plant and Cell Physiology. 1989;30:833–839. doi: 10.1093/oxfordjournals.pcp.a077813. [DOI] [Google Scholar]
- 29.Li T, Shen B, Chen N, Luo Y. Effect of Q-Enzyme on the Chalkiness Formation of Rice Grain. Acta Agronomica Sinica. 1997;23:338–344. [Google Scholar]
- 30.Huber SC. Role of Sucrose-Phosphate Synthase in Partitioning of Carbon in Leaves. Plant Physiology. 1983;71:818–821. doi: 10.1104/pp.71.4.818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Smyth DA, Repetto BM, Seidel NE. Cultivar Differences in Soluble Sugar Content of Mature Rice Grain. Physiologia Plantarum. 1986;68:367–374. doi: 10.1111/j.1399-3054.1986.tb03367.x. [DOI] [Google Scholar]
- 32.Wardlaw IF, Willenbrink J. Carbohydrate Storage and Mobilisation by the Culm of Wheat between Heading and Grain Maturity: the Relation to Sucrose Synthase and Sucrose-Phosphate Synthase. Australian Journal of Plant Physiology. 1994;21:255–271. doi: 10.1071/PP9940255. [DOI] [Google Scholar]
- 33.Rufty TW, Huber SC. Changes in Starch Formation and Activities of Sucrose Phosphate Synthase and Cytoplasmic Fructose-1, 6-Bisphosphatase in Response to Source-Sink Alterations. Plant Physiology. 1983;72:474–480. doi: 10.1104/pp.72.2.474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Doehlert DC, Kuo TM, Felker FC. Enzymes of Sucrose and Hexose Metabolism in Developing Kernels of Two Inbreds of Maize. Plant physiology. 1988;86:1013–1019. doi: 10.1104/pp.86.4.1013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yang H, et al. Effects of Sowing Date, Fertilization Rate and Planting Density on the Quality, Yield and Yield-Contributing Factors of Waxy Wheat Cultivar ‘1718WX’. Journal of Southwest University (Natural Science Edition) 2015;37:12–17. [Google Scholar]
- 36.Yao J, et al. Evaluation and Utilization of Waxy Wheat Germplasm H9908. Journal of Triticeae Crop. 2004;24:116–118. [Google Scholar]
- 37.Briarty L, Hughes C, Evers A. The Developing Endosperm of Wheat—a Stereological Analysis. Annals of Botany. 1979;44:641–658. doi: 10.1093/oxfordjournals.aob.a085779. [DOI] [Google Scholar]
- 38.Liang J, Zhang J, Cao X. Grain Sink Strength May Be Related to the Poor Grain Filling of Indica‐Japonica Rice (Oryza Sativa) Hybrids. Physiologia Plantarum. 2001;112:470–477. doi: 10.1034/j.1399-3054.2001.1120403.x. [DOI] [PubMed] [Google Scholar]
- 39.Gleadow RM, Dalling MJ, Halloran GM. Variation in Endosperm Characteristics and Nitrogen Content in Six Wheat Lines. Australian Journal of Plant Physiology. 1982;9:539–551. doi: 10.1071/PP9820539. [DOI] [Google Scholar]
- 40.Pan Q, Yu Z, Wang Y, Yu S. Effects of Nitrogen Applying Stage on Both Sucrose Synthesis in Flag Leaves and Cleavage in Grains of Wheat. Scientia Agricultura Sinica. 2002;35:771–776. [Google Scholar]
- 41.Li Y, Yu Z, Jiang D, Yu S. Studies on the Dynamic Changes of the Synthesis of Sucrose in the Flag Leaf and Starch in the Grain and Related Enzymes of High-Yielding Wheat. Acta Agronmica Sinica. 2001;27:658–664. [Google Scholar]
- 42.Tan C, et al. Study on the Characteristics of Cucrose and Starch Accumulation in Grains of Different Types Wheat Cultivars. Journal of Yangzhou University (Agricultural and Life Sccience Edition) 2011;32:47–51. [Google Scholar]
- 43.Jiang D, Yu Z, Li Y, Yu S. Dynamic Changes of Enzyme Activities Involving in Starch Synthesis in Superior and Inferior Grains of High-yield Winter Wheat. Scientia Agricultura Sinica. 2002;35:378–383. [Google Scholar]
- 44.Yang J, Su B, Wang Z, Lang Y, Zhu Q. Characteristics and Physiology of Grain-Filling in Intersubspecific Hybrid Rice. Scientia Agricultura Sinica. 1998;31:7–14. [Google Scholar]
- 45.Sowokinos JR, Varns JL. Induction of Sucrose Synthase in Potato Tissue Culture: Effect of Carbon Source and Metabolic Regulators on Sink Strength. Journal of Plant Physiology. 1992;139:672–679. doi: 10.1016/S0176-1617(11)81709-8. [DOI] [Google Scholar]
- 46.Wang B, et al. Photosynthesis, Sucrose Metabolism, and Starch Accumulation in Two NILs of Winter Wheat. Photosynthesis Research. 2015;126:363–373. doi: 10.1007/s11120-015-0126-9. [DOI] [PubMed] [Google Scholar]
- 47.Liang J, Cao X, Zhu Q. Abscisic Acid May Involve in the Regulation of Grain Filling in Water Stressed Rice (Oryza sativa L.) Chinese Journal of Rice Science. 1996;10:29–36. [Google Scholar]
- 48.Liang J, Cao X, Xu S. Effects of Sucrose Concentration and Hormones on the Grain Development of Detached Rice Panicles in Vitro Culture. Chinese Journal of Rice Science. 1993;7:77–82. [Google Scholar]
- 49.Yang J, Zhang J, Wang Z, Xu G, Zhu Q. Activities of Key Enzymes in Sucrose-to-Starch Conversion in Wheat Grains Subjected to Water Deficit during Grain Filling. Plant Physiology. 2004;135:1621–1629. doi: 10.1104/pp.104.041038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Preiss J. Biosynthesis of Starch and Its Regulation. The Biochemistry of Plants. 1988;14:181–254. doi: 10.1016/B978-0-08-092615-5.50012-6. [DOI] [Google Scholar]
- 51.Ballicora MA, Iglesias AA, Preiss J. ADP-glucose Pyrophosphorylase: a Regulatory Enzyme for Plant Starch Synthesis. Photosynthesis Research. 2004;79:1–24. doi: 10.1023/B:PRES.0000011916.67519.58. [DOI] [PubMed] [Google Scholar]
- 52.Li N, Zhang S, Zhao Y, Li B, Zhang J. Over-Expression of AGPase Genes Enhances Seed Weight and Starch Content in Transgenic Maize. Planta. 2011;233:241–250. doi: 10.1007/s00425-010-1296-5. [DOI] [PubMed] [Google Scholar]
- 53.Sakulsingharoj C, et al. Engineering Starch Biosynthesis for Increasing Rice Seed Weight: the Role of the Cytoplasmic ADP-Glucose Pyrophosphorylase. Plant Science. 2004;167:1323–1333. doi: 10.1016/j.plantsci.2004.06.028. [DOI] [Google Scholar]
- 54.Smidansky ED, et al. Expression of a Modified ADP-Glucose Pyrophosphorylase Large Subunit in Wheat Seeds Stimulates Photosynthesis and Carbon Metabolism. Planta. 2007;225:965–976. doi: 10.1007/s00425-006-0400-3. [DOI] [PubMed] [Google Scholar]
- 55.Vrinten PL, Nakamura T. Wheat Granule-Bound Starch Synthase I and II Are Encoded by Separate Genes That Are Expressed in Different Tissues. Plant Physiology. 2000;122:255–264. doi: 10.1104/pp.122.1.255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Umemoto T, Terashima K. Activity of Granule-Bound Starch Synthase is an Important Determinant of Amylose Content in Rice Endosperm. Functional Plant Biology. 2002;29:1121–1124. doi: 10.1071/PP01145. [DOI] [PubMed] [Google Scholar]
- 57.Liu X, et al. Activities of the Enzymes Involved in Starch Synthesis and Starch Accumulation in Grains of Wheat Cultivars GC8901 and SN1391. Scientia Agricultura Sinica. 2005;38:897–903. [Google Scholar]
- 58.Yan S, et al. Activities of Enzymes Involved in Starch Synthesis and Accumulation in Grains of Two Wheat Cultivars with a Different Amylose Content. Acta Agronomica Sinica. 2007;33:84–89. [Google Scholar]
- 59.James MG, Denyer K, Myers AM. Starch Synthesis in the Cereal Endosperm. Current Opinion in Plant Biology. 2003;6:215–222. doi: 10.1016/S1369-5266(03)00042-6. [DOI] [PubMed] [Google Scholar]
- 60.Wang Z, Li W, Qi J, Shi P, Yin Y. Starch Accumulation, Activities of Key Enzyme and Gene Expression in Starch Synthesis of Wheat Endosperm with Different Starch Contents. Journal of Food Science and Technology. 2014;51:419–429. doi: 10.1007/s13197-011-0520-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Tan C, et al. Difference in Expression of Starch Synthase Gene and Starch Synthesis in the Grains of Different Wheat Cultivars. Journal of Triticeae Crops. 2011;31:1063–1070. [Google Scholar]
- 62.Xia H, Yandeaunelson M, Thompson DB, Guiltinan MJ. Deficiency of maize starch-branching enzyme i results in altered starch fine structure, decreased digestibility and reduced coleoptile growth during germination. Bmc Plant Biology. 2011;11:95. doi: 10.1186/1471-2229-11-95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.He Z, Shen Z. Effect of Rice Wx Gene on Substance Accumulation of Grain. Scientia Agricultura Sinica. 1995;28:20–24. [Google Scholar]
- 64.Jiang D, Cao W, Dai T, Jing Q. Activities of Key Enzymes for Starch Synthesis in Relation to Growth of Superior and Inferior Grains on Winter Wheat (Triticum aestivum L.) Spike. Plant Growth Regulation. 2003;41:247–257. doi: 10.1023/B:GROW.0000007500.90240.7d. [DOI] [Google Scholar]