Abstract
Gastric diseases, including peptic ulcer disease and gastric cancer, are highly prevalent in human beings. Despite this, the cellular biology of the stomach remains poorly understood relative to other gastrointestinal organs such as the liver, intestine, and colon. In particular, little is known about the molecular basis of stomach development and the differentiation of gastric lineages. Although animal models are useful for studying gastric development, function, and disease, there are major structural and physiological differences in human stomachs that render these models insufficient. To look at gastric development, function, and disease in a human context, a model system of the human stomach is imperative. This review details how this was achieved through the directed differentiation of human pluripotent stem cells in a 3-dimensional environment into human gastric organoids (HGOs). Similar to previous work that has generated human intestine, colon, and lung tissue in vitro, HGOs were generated in vitro through a step-wise differentiation designed to mimic the temporal-spatial signaling dynamics that control stomach development in vivo. HGOs can be used for a variety of purposes, including genetic modeling, drug screening, and potentially even in future patient transplantation. Moreover, HGOs are well suited to study the development and interactions of nonepithelial cell types, such as endothelial, neuronal, and mesenchymal, which remain almost completely unstudied. This review discusses the basics of stomach morphology, function, and developmental pathways involved in generating HGOs. We also highlight important gaps in our understanding of how epithelial and mesenchymal interactions are essential for the development and overall function of the human stomach.
Keywords: Organoids, Pluripotent Stem Cells, Directed Differentiation, Endoderm, Gastric Development
Abbreviations used in this paper: BMP, bone morphogenetic protein; e, embryonic day; ECL, enterochromaffin-like; ENS, enteric nervous system; ENCC, enteric neural crest cell; GI, gastrointestinal; HDGC, hereditary diffuse gastric cancer; HGO, human gastric organoid; hPSC, human pluripotent stem cell; PSC, pluripotent stem cell; Shh, Sonic hedgehog; 3-D, 3-dimensional
Summary.
This review discusses the basics of stomach morphology, function, and development, and explains the technical process of generating human gastric organoids. Important gaps in understanding why epithelial-mesenchymal interactions are essential for the development and overall function of the human stomach are highlighted.
The stomach’s main role is to digest macronutrients. Chemical and mechanical digestion breaks down and prepares macronutrients for proper absorption once they are emptied into the intestine. The stomach also plays a part in regulating satiety.1 A diverse set of endocrine cells secrete a variety of hormones and neuropeptides, including gastrin, ghrelin, and somatostatin, to regulate appetite and gastric acid secretion.
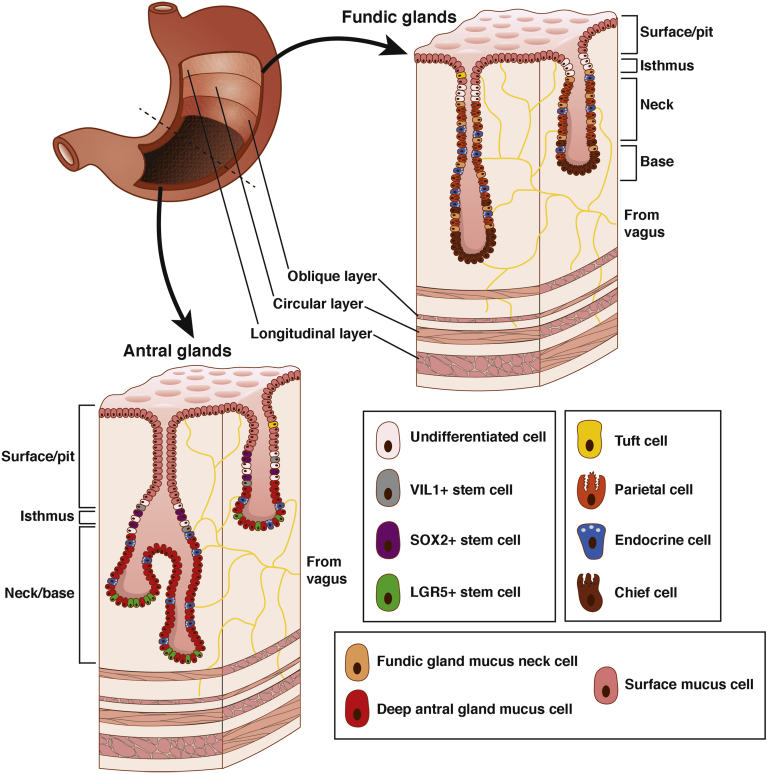
The 2 main regions of the human stomach are the proximal corpus/fundus and the distal antrum1, 2, 3 (Figure 1). In the corpus, the epithelium is organized into fundic glands that contain mucus-producing cells along the surface and within the gland, acid-secreting parietal cells, pepsinogen-secreting chief cells, stem cells, endocrine cells, and rare tuft cells.2 The antrum gland unit also contains mucus cells along the surface and within the gland, endocrine cells, including gastrin-producing cells that are unique to the antrum, and rare tuft cells, but has far fewer parietal and chief cells.4 It does, however, contain LGR5+ stem cells that give rise to all other cell types within the antrum.2 Both gland units are organized into 3 basic sections: a deep base, medial isthmus, and surface pit. Although undifferentiated stem cells make up the isthmus of both fundic and antral glands, the isthmus is nearer to the surface in fundic glands and nearer to the base in antral glands. Parietal, chief, and endocrine cells reside in the base of fundic glands, whereas endocrine and LGR5+ stem cells, along with rare parietal and chief cells, reside in the base of antral glands. Rare tuft cells can be found on the surface of both types of glands.1, 2
Figure 1.
Morphology and germ layer contributions of the adult stomach with specific cell types of the antral and fundic glands. The adult stomach comprises an endoderm-derived epithelial lining surrounded by mesoderm-derived smooth muscle and ectoderm-derived nerves and glia. Fundic glands are comprised of chief cells (brown) in the base, parietal (orange), endocrine (blue), and mucus (tan) cells throughout the neck, stem cells (white) in the isthmus, and mucus (pink) and tuft (yellow) cells on the surface. Antral glands comprise endocrine (blue), mucus (red), and LGR5+ (green) stem cells within the base, VIL1+ (grey), SOX2+ (purple), and other (white) stem cells in the isthmus, and mucus (pink) and tuft (yellow) cells along the surface. Myenteric neurons innervating the submucosa and muscle layers of the stomach not only stimulate hormonal secretion, including gastrin, somatostatin, and histamine, to aid in chemical digestion, but also play a role in regulating muscular contraction to aid in mechanical digestion.
Stomach Morphology Across Species
Historically, functional studies of gastric development, lineage commitment, and disease have predominantly used murine models. However, stomach morphology and regionalization differ greatly across species.1, 2 Unlike the human stomach, the murine stomach contains an additional region, the forestomach, that is anterior to the corpus/fundus and contains a stratified squamous, rather than glandular, epithelium.2 In addition, parietal cells are absent from murine antral gland units.1 It is likely that the embryonic development of the stomach similarly differs across species.1 Finally, the pathophysiology of gastric diseases often is hard to recapitulate in murine models.5, 6, 7 Thus, to effectively study human gastric development, physiology, and disease, it is imperative to use a human model system.
Gastric Epithelium and Mesenchyme
Epithelial Function and Development
The stomach’s main role of breaking down orally ingested macronutrients is accomplished, in part, through the secretion of gastric acid, proteases, hormones, and neuropeptides from cells within the stomach’s epithelial lining. However, because of the constant stress of this chemical digestion, the gastric epithelium must be highly regenerative. Gastric cell lineages have very different life spans; mucus cells on the surface only live for approximately 3–5 days, while chief cells may survive for several months.8 To support this epithelial turnover, the gastric epithelium contains stem and progenitor cells. There are several notable differences between the gastric stem cells of the fundus and the antrum.8, 9, 10 In the antrum, lineage-tracing studies in mice suggest that there are 2 multipotent stem cell populations—Lgr5+ stem cells in the base of the gland and Sox2+ stem cells in the isthmus—that differentiate almost exclusively into endocrine cells, surface and deep base mucus cells, and tuft cells.2 Vil1+ cells, found in the isthmus of antral glands, can act as stem cells during mucosal damage and repair.2 More seems to be known about these identified antral stem cells compared with their fundic counterparts. In the fundus, there are a variety of markers for stem cells that reside in the isthmus and give rise to the 5 principal cell types of the fundus mentioned earlier.2 Lineage tracing in adult mice showed that Sox2+ cells in the glandular stomach are able to self-renew and differentiate into chief, endocrine, mucous, and parietal cells.1, 11, 12 However, recent findings have shown that Sox2 itself may be dispensable for gastric stem cells and epithelial self-renewal.13 Other markers for stem cells within the fundic region of the stomach include Runx1, Axin2, and Lrig1.10, 14, 15 There is also evidence that Lgr5+, Mist1+, and Troy+ chief cells can act as reserve stem cells during cellular damage and repair.16, 17, 18, 19
All of the endocrine cells in the epithelium of both the fundus and the antrum arise from a common Ascl1+ (Mash1+) progenitor.2 In the antrum, the transcription factor Neurog3, downstream of Ascl1, is required to specify all endocrine cells: gastrin-secreting G cells, somatostatin-secreting D cells, ghrelin-secreting X/A cells, histamine-secreting enterochromaffin-like (ECL) cells, and serotonin-secreting enterochromaffin (EC) cells.20, 21 These cell types are absent in Neurog3 knockouts.20, 21 Neurog3-/- mice also have a drastically smaller stomach and disorganized gastric epithelium when compared with wild-type mice at P3.21 Other transcription factors downstream of Neurog3, such as NeuroD1 and Pax6, work in coordination with Neurog3 to specify endocrine cells that reside not only in the stomach, but also in the intestine.20, 21 However, in the fundus, even though lineage-tracing experiments show that Neurog3+ cells can give rise to most of the fundic endocrine cells, there are still X/A, ECL, and EC cells present in Neurog3-/- mice, suggesting that other factors may play a role in the specification of fundic endocrine cells.2, 20
Mesenchymal Function and Development
Although the gastric epithelium is often the focal point for discussions of stomach function and disease, the stomach is developmentally derived from and requires all 3 germ layers for proper function.1 The glandular epithelium of the stomach is surrounded by layers of smooth muscle in which a neural plexus is embedded. Moreover, there are many additional cell and tissue types including supporting stromal cells, immune cells, pericytes, and an endothelial plexus. Although the epithelial cells are derived from the endoderm, smooth muscle and vascular cells are derived from the mesoderm and the enteric neurons and glial cells are derived from the ectoderm.1 Communication between the cell types from all 3 germ layers is essential for the coordination of gastric function.2 For example, regulation of gastric acid secretion involves vagal afferent nerves that innervate the gastric mesenchyme (Figure 1).22, 23, 24, 25, 26, 27, 28, 29, 30 Myenteric neurons stimulate not only parietal cells to directly secrete acid, but also G cells, D cells, and ECL cells to indirectly influence gastric acid secretion. Enteric neurons and glial cells, which eventually form the 2 concentric rings of our enteric nervous system (ENS), also play a role in other key digestive functions such as gut motility and muscular contractility, which aid in the stomach’s mechanical digestion.
This multigerm layer interaction occurs not only during adult stages; the inductive cross-talk between the stomach epithelium and its underlying mesenchyme begin at early stages of organ development and establishes proper epithelial and mesenchymal regionalization.1, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47 A number of key morphogens play a role in this regionalization, including WNTs, bone morphogenetic proteins (BMPs), Sonic hedgehog (Shh), and members of the HOX and SOX gene families.32, 43 These signaling pathways also play a role in the later differentiation of gastric mesenchyme into smooth muscle cells, which, in turn, is suggested to influence the regionalization of the epithelium through interactions with the ENS.32 These mesenchymal-epithelial interactions not only establish regionalization and direct differentiation, but also play a role in the epithelial homeostasis of adult stomach function.32
Epithelial-Mesenchymal Interactions
Early regional patterning and Wnt signaling
Early regional patterning of the stomach involves the regulation of Wnt signaling. In the presumptive fundic region, active epithelial Wnt signaling initiates and maintains fundic epithelial fate.48 In contrast, low Wnt signaling in the antral region is necessary for both the development of stomach-specific epithelium and maintenance of the stomach-intestinal boundary.1, 33 Wnt inhibition in the antrum is mediated non–cell autonomously by Barx1, a homeobox transcription factor restricted to the stomach mesenchyme during gut organogenesis that not only specifies gastric epithelial identity but also controls smooth muscle formation within the gastric mesenchyme.33, 34 Loss of Barx1 in the mesenchyme of recombinant tissue cultures prevents stomach epithelial differentiation and induces intestine-specific genes.33 In addition, Barx1-/- mouse embryos show intestinal gene expression within a highly disorganized gastric epithelium.33 Barx1, through the regulation of mesenchymal expression of 2 Wnt-signaling antagonists, secreted frizzled-related protein 1 and 2, inhibits Wnt signaling around the antral-duodenal boundary and prospective gastric endoderm to permit the development of stomach-specific epithelium.1, 33
Barx1 plays a role in the development of not only the gastric epithelium, but also its mesenchyme; it induces the expansion of gastric smooth muscle by promoting the proliferation of myogenic progenitors over other subepithelial cells.34 In fact, ectopic Barx1 expression in the presumptive intestinal region induces the smooth muscle to develop more thickly, similar to gastric smooth muscle.34 In the stomach mesenchyme, Barx1 activates a group of secondary transcription factors, including Isl1, Six2, and Pitx1/2, that are known to control myogenesis and play a role in smooth muscle development.34 Isl1 and Six2, in particular, have been shown to play a role in the formation of the pyloric sphincter at the same antral-duodenal boundary where Barx1 maintains gastric epithelial identity.34, 40, 45, 49, 50, 51, 52, 53, 54
One of the best-studied interactions between the epithelium and mesenchyme involves BMP signaling (along with fibroblast growth factor 10), from the mesenchyme, regulating gastric epithelial development, while epithelial-derived Hedgehog ligands control mesenchymal differentiation.1
Epithelial Shh signaling
Shh, an important endoderm signaling molecule expressed throughout the gastrointestinal (GI) tract, specifies early GI patterning, controls mesenchymal growth, and directs later gastric gland formation.42, 47, 55 In the stomach, Shh-responsive progenitors begin to give rise to smooth muscle cells and myofibroblasts in gut mesenchyme starting on embryonic day (e)11, and by e12.5 there is a rapid expansion of mesenchymal tissue. However, Shh-/- stomachs at e12.5 show very little mesenchyme and a complete absence of smooth muscle.55 Shh induces Bmp4 in the mesoderm of the midgut and hindgut, but not in the stomach region of the foregut to limit the growth of intestinal and colonic mesenchyme, while preserving a thicker layer of gastric mesenchyme.41, 43, 45 Indeed, ectopic Bmp4 expression in the stomach results in reduced and disorganized mesenchymal growth as well as reduced smooth muscle differentiation.41, 43, 45
Interestingly, Shh-/- embryos also show a significant decrease in Tuj1+ neurons.55, 56 Recent studies have suggested that the reduction in smooth muscle, as seen in many mutant models, including Shh-/- embryos, may not solely be a direct result of the genetic mutations, but also an indirect effect of reduced enteric neurons. Ablating enteric neural crest cells (ENCCs) in chick embryos induces sustained activation of the BMP and Notch pathways in the stomach mesenchyme and impairs smooth muscle development.57 In addition, reducing the number of ENCCs leads to the transdifferentiation of the stomach mesenchyme into a mixed stomach-intestinal phenotype.57 Therefore, ENCCs may act as a mediator in the mesenchymal-epithelial development of the stomach mesenchyme.
Mesenchymal Bmp signaling
Although Bmp4 is known to be critical for development of the gut mesoderm,40, 41, 43, 45, 58, 59 other members of the BMP signaling pathway are involved in different aspects of gut development; Bmp7 is involved in stomach gland formation,60 and Bmp2 is involved in the maturation of the enteric neuronal cells.61, 62 These BMP signaling molecules are dynamically expressed across different tissue layers of the GI tract and, thus, play active roles in the interactions between different gut layers.39 Bmp4, expressed very early in gut development before colonization of the ENS, also plays a role in early ENS patterning.39, 43 Inhibition of BMP activity by an overexpression of Noggin within the developing gut inhibits normal ENCC migration and leads to hypoganglionosis and failure to form enteric ganglion.39
Additional mesenchymal transcription factors
There are other gastric mesenchymal transcription factors that are expressed in a temporally and spatially dynamic fashion, such as Bapx1 (Nkx3-2) and Hoxa5, which also drive mesenchymal-epithelial signaling to promote proper regionalization and specification of the stomach.1, 2, 31, 32 Bapx1 is expressed in the splanchnic mesoderm and plays roles in gut regionalization and muscle formation.63, 64 Hoxa5 is expressed in a dynamic fashion across the gut mesenchyme and, by setting up proper expression domains of key signaling molecules, acts to regionalize and specify the stomach; its loss of function causes defects in gastric epithelial specification as well as misexpression of Shh and Fgf10.31
Congenital Defects and Disease
Pyloric Sphincter Development
The pyloric sphincter is a functional gate at the stomach-intestinal junction that inhibits duodenogastric reflux, and abnormalities in either pyloric development or contractile function can cause reflux of intestinal contents into the stomach, increasing the risk of gastric metaplasia and cancer.54 Pyloric abnormalities also are linked to congenital defects, such as the relatively common hypertrophic pyloric stenosis.50, 53, 54 Isl1 and its direct downstream target Gata3 are highly expressed in pyloric smooth muscle, and deficiency in either leads to an almost complete absence of pyloric longitudinal muscle at e18.5 in mice, impairing pyloric smooth muscle formation and sphincter formation.50, 54 In addition to Isl1, Gata3, and Six2, several other key genes, including Bmp4, Nkx2.5, and Sox9 are known to play a role in pyloric sphincter formation.49, 50, 51 Bmp4 up-regulates Nkx2.5, while Gata3 and Nkx2.5 independently up-regulate Sox9.45, 52 Loss of either Gata3 or Nkx2.5 abrogates Sox9 expression and impairs gastric smooth muscle development.50, 53 Sox9 is expressed not only in pyloric mesenchyme but also in the endoderm, serving to also specify the pyloric sphincter epithelium.40, 52
H pylori and Our Limitations of Understanding Gastric Disease
There are many diseases that affect the stomach, such as gastroenteritis, gastroparesis, gastritis, peptic ulcer disease, and stomach cancer. The latter 3 all have well-known associations with the bacilli bacterium Helicobacter pylori. This pathogen colonizes the stomach of approximately half of the world’s population, and although infection is usually asymptomatic, it can lead to some of the aforementioned diseases, of which gastritis is most common.65 This disease is characterized by inflammation, irritation, or erosion of the stomach lining.65 People with H pylori also have a 10%–20% chance of developing ulcers and a 1%–2% risk of developing gastric cancer.66 In fact, 1 cohort study involving patients with ulcers showed that 2.9% of patients infected with H pylori developed gastric cancer, whereas none of the uninfected patients showed any cancer progression.67 Of patients with ulcers, 95% of those with duodenal ulcers and 80% of those with gastric ulcers have H pylori infection.68 Ulcers typically arise in the corpus-antrum transition zone as well as in the duodenum, where it is referred to as peptic ulcer disease.66, 69 There are currently a broad range of mammalian model systems used to study H pylori pathogenesis, ranging from rodent to primate-based models.70 These animal models are useful for identifying the cellular and molecular factors that allow pathogens to survive and thrive in gastric mucosa.65 However, none of these models replicate exact human pathophysiology and any conclusions drawn from these studies may not directly translate to understanding the human gastric disease.65
Human gastric organoids (HGOs), derived from human pluripotent stem cells (hPSCs), offer a new alternative to studying stomach development, physiology, and diseases in a human-specific context. Later, we discuss the differentiation of HGOs from PSCs in vitro, and how it is dependent on the fundamental knowledge of the signaling pathways that control stomach development in vivo. We also discuss the importance of mesenchyme and neural crest cells for the generation of more functional organoids and how organoid-based platforms ideally might be suited to study the reciprocal interactions of these varied cell types during development of the human stomach.
Generating Gastric Tissue In Vitro
Deriving Definitive Endoderm
To generate gastric tissues from hPSCs in vitro, it is important to utilize and temporally mimic signaling pathways that control stomach development in vivo (Figure 2). The first step in this process is to direct the hPSCs into a state of gastrulation, which in the embryo is a process by which the 3 germ layers, the endoderm, mesoderm, and ectoderm, are specified.71 Directing hPSCs specifically into the endoderm lineage is achieved by activation of the nodal/transforming growth factor-β signaling pathway using the commercially available nodal mimetic activin A.72, 73 Although nodal signaling is necessary and sufficient to induce both endoderm and mesoderm, high levels of nodal signaling promote the formation of endoderm, whereas lower levels promote mesoderm.71, 73, 74 High nodal signaling activates a transcriptional network that includes Sox17 and Foxa2, which marks definitive endoderm.71 The functional requirements of this endoderm-specific transcriptional network have been studied in Xenopus, mouse, and zebrafish.74, 75, 76, 77, 78, 79 In hPSC cultures, activation of nodal signaling via activin A for 3 days directs hPSCs into a Sox17+ and Foxa2+ layer of definitive endoderm.80
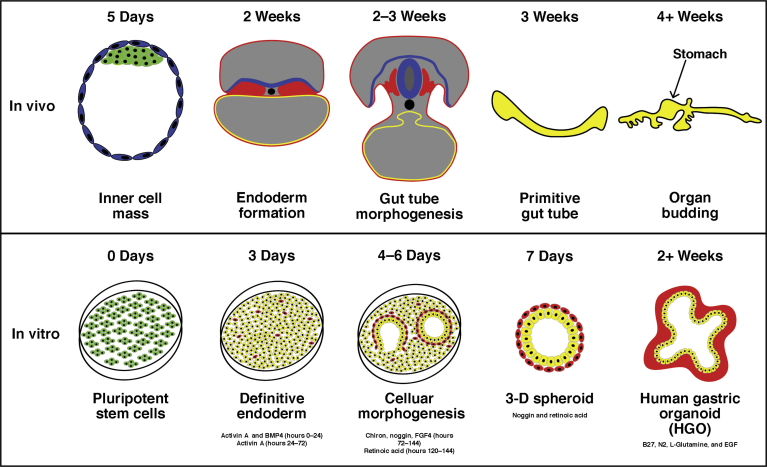
Figure 2.
In vitro growth of human gastric organoids mimics in vivo development of the human stomach. During human embryonic development, human embryonic stem cells of the inner cell mass of the blastocyst have the potential to form all cells from the 3 germ layers. hPSC cultures are derived from cells of the inner cell mass. All 3 germ layers are well defined by 2 weeks of human embryonic development, including the endoderm. The equivalent step of this process in vitro is completed just after 3 days in culture. In vivo, between 2 and 3 weeks of human embryonic development, the endoderm undergoes a morphogenetic process that gives rise to the primitive gut tube. In vitro, during days 4 and 5 of the protocol, definitive endoderm, defined by the protocol’s third day, elicits a spontaneous 2-dimensional to 3-D transformation, forming spheroids, which corresponds with the human primitive gut tube that is formed and closed by 3 weeks in human development. In vivo, by 4 weeks, organs will begin to bud from the primitive gut tube and continue to mature and develop over embryonic time. The equivalent process in the in vitro protocol allows for the growth and maturation of the 3D spheroids into organoids over the course of approximately 4 weeks in culture. EGF, epidermal growth factor; FGF, fibroblast growth factor.
Patterning the Endoderm Along the A-P Axis
By the end of gastrulation in chick, mouse, and human beings, the definitive endoderm is a monolayer of cells that is not yet committed to specific organ lineages. One of the first critical steps toward acquiring organ identity, in this case the stomach, involves patterning the endoderm along the anterior-posterior axis.81 In addition, the postgastrula endoderm undergoes morphogenesis from a 2-dimensional sheet of cells into a 3-dimensional (3-D) primitive gut tube.81 Patterning of the endoderm and the early gut tube involves 4 major signaling pathways: Wnt, Fgf, Bmp, and retinoic acid.6, 35, 82 Endoderm initially is patterned into broad anterior and posterior domains that give rise to the 3 main regions of the developing gut tube: the anterior foregut and the more posterior midgut and hindgut.48 The foregut, from which the stomach derives, is marked by the expression of the transcription factor Sox2, whereas the midgut and hindgut express the transcription factor Cdx2.48, 83, 84, 85, 86 Specification of the anterior endoderm that gives rise to the foregut in Xenopus occurs through synergistic inhibition of Wnt and Bmp.35, 82, 87, 88, 89 Once the foregut is formed it is subdivided further into anterior and posterior foregut. Posterior foregut development requires retinoic acid signaling; genetic loss-of-function of the retinoic acid synthesizing enzyme Raldh in mice resulted in defective patterning and morphogenesis of the posterior foregut.89 Conversely, the hindgut requires high levels of Wnt, Bmp, and Fgf4.35, 88, 90
3-D Morphogenesis and Generation of Fundic Organoids
Therefore, the creation of HGOs in vitro require similar temporal manipulation of these pathways. To generate foregut tissue (spheroids) from hPSC-derived definitive endoderm, BMP signaling is inhibited by noggin, resulting in the activation of SOX2 expression and repression of CDX2 expression.6 However, morphogenesis from 2-dimensional endoderm cultures into gut tube spheroids requires activation of WNT and fibroblast growth factor 4.6 To promote PDX1 positive antral identity, the resulting 3-D spheroids still need further inhibition of BMP and activation of retinoic acid, as well as activation of epidermal growth factor signaling. Together, this is sufficient to promote the formation of antral-specific organoids.6
The generation of fundic organoids was considerably more challenging because the signaling pathways that pattern the fundus were unknown. As described earlier, posterior foregut organoids and mouse genetics were used to identify that fundic specification required canonical Wnt signaling. The profundic role of Wnt is separate from its earlier developmental role as a repressor of anterior endoderm, exemplifying the concept that signaling pathways have distinct roles at different stages of development. Sustained activation of WNT signaling in posterior foregut cultures is sufficient to promote a fundic epithelial fate by suppressing PDX1 expression, resulting in the formation of fundic organoids that expressed mucus and chief cell markers. However, this was not sufficient to promote differentiation of parietal cells, which required an additional step involving inhibition of MEK and activation of BMP signaling pathways in the final stages of culture (days 30–34).48
Organoid Epithelium and Mesenchyme
Both fundic and antral human gastric organoids have a glandular epithelium and a layer of mesenchyme. The epithelium contains all gastric cell types including surface and glandular mucus cells, all known endocrine cells, and chief cells that secrete pepsinogen. Importantly, fundic organoids contain proton pump–expressing parietal cells capable of secreting acid in response to histamine. However, HGOs are not equivalent to adult gastric tissue. HGOs are relatively fetal in nature; the mesenchyme is not robust and does not contain significant smooth muscle in vitro. In addition, these tissues do not contain vasculature or an ENS.6, 48 Although these are current limitations of these systems, these are not insurmountable.
Current and Future Applications of HGOS
Organoids in Basic Research
Gastric organoids have proven to be an excellent model to study human stomach development and physiology.6, 48, 81, 91 Organoid cultures are ideally suited to investigate signaling pathways that regulate gastric patterning, morphogenesis, and the poorly understood process of cell subtype specification. In fact, the essential role for epithelial Wnt signaling in establishing and maintaining a fundic epithelial fate initially was discovered using HGOs, then validated using mouse genetic approaches.48 Given the presence of mesenchyme in HGOs, they also can be used to study the epithelial-mesenchymal interactions that drive gastric development. In addition, the advent of gene editing tools, such as CRISPR/Cas9, have allowed researchers to generate fluorescent reporter lines that are used to study specific cell populations, their lineage relationships, and how various pathogenic agents affect them.92 Finally, infection of HGOs with a pathogenic form of H pylori triggers a rapid signaling response and a concomitant stimulation of epithelial proliferation that we can use in the laboratory to study bacterial infection and disease progression.6
Organoid Uses in Personalized and Regenerative Medicine
Gene editing and disease modeling
Gene editing technologies, such as CRISPR/Cas9, have allowed for the generation and/or correction of disease-causing genetic mutations in human pluripotent stem cells (PSCs). In the context of a patient with a genetically caused disease, the discovery of human induced PSCs now allows for the generation of PSC lines through the reprogramming of the patient’s own somatic cells.93 By using these cell lines, scientists can generate diseased and genetically corrected organoid tissues, allowing for unprecedented studies of human disease mechanisms.93, 94 One use of this approach would be to study unappreciated gastric pathologies that arise in patients with complex syndromic birth defects. For example, 10% of patients in the neonatal intensive care unit have severe birth defects. Given the fact that gastric abnormalities are rarely life threatening in this context, it seems likely that gastric defects remain unidentified. Patient-derived organoids are ideally suited to perform deep phenotyping on such patients and would allow for identification of subtle pathologies in newborns and possibly prevent severe complications later in life.
Organoids for disease diagnostics and therapy
Although birth defects are one example of using induced PSC-derived organoids to diagnose pathologies, HGOs ideally are suited to study hereditary forms of gastric cancer. Hereditary diffuse gastric cancer (HDGC) is a rare form of diffuse gastric cancer in which the genetic cause is known only in less than half the patients. The most common cause of HDGC is mutations in E-cadherin, however, in >60% of cases the causative mutations are not known, and the only treatment options are close monitoring or complete gastrectomy. HGOs could be used to diagnose patients who are at risk for HDGC in cases in which the causative genes remain unidentified.
Tissue engineering
Information derived from organoid-based diagnostics could predict cancer and save lives. In the case of HDGC, organoids could indicate that preventative measures, such as complete gastrectomy, should be taken to prevent cancer progression. Although many people do live without a stomach, the quality of life is impacted negatively by the many complications associated with complete gastrectomy. In cases such as this, patient-specific gastric organoids represent an exciting starting point for organ-replacement therapies. However, efforts to engineer replacement tissues face significant challenges. The stomach is 10 cm wide and 30 cm long, while gastric organoids are 2–3 mm in diameter, highlighting the need for engineering new tissue growth technologies for scale-up. One option would be to grow organoids larger in vivo, as has been done with intestinal organoids that reach centimeters in size.95 Another hurdle is tissue complexity. HGOs only contain an epithelium and mesenchyme and are lacking the enteric nervous system that is essential for many gastric functions. This hurdle recently has been overcome in the context of human intestinal organoids (see later).96
Drug testing and discovery
Gastric toxicity is a new common side effect of some drugs.94 HGOs are also a novel in vitro tool for drug screening. Commercially available drugs used to block acid production in patients are effective at inhibiting acid secretion in fundic HGOs, suggesting that HGOs would be an effective tool for the identification of new drugs that control acid production. Adverse reactions to pharmaceuticals are a major concern and create significant issues for patients, health care providers, and drug companies.97 This technology would provide the industry with a high-throughput way of testing drug efficacy and toxicity.
This patient-specific, personalized medicine approach has the potential to revolutionize health care and cure genetic issues that to date are impossible to fix through traditional means. However, an important hurdle remains because HGOs are still relatively fetal and simple in nature. Adding additional complexity to enhance the physiological function of HGOs will be necessary to make these types of treatments possible.
Engineering Additional Complexity to Enhance Functionality
Gastric organoids currently are composed of a simple epithelium with a relatively small mesenchymal component. The next wave of research is dedicated to incorporating additional cellular components to HGOs with the aim of increasing their complexity and viability. Previous work with human intestinal organoids successfully has incorporated a functional ENS within the mesenchyme of the organoid.96, 98 Moving forward, this will be necessary to accomplish with HGOs.
Additional work is being conducted by various laboratories to introduce a vascular system to human lung organoids, by means of recombining endothelial cells, as well as to fully introduce an immune system to human colonic and liver organoids.99, 100, 101 Engineering different germ layers and additional cell types within hPSC-derived GI organoids will be essential in creating the soundest models of human biology and physiology.
Conclusions
Human PSC-derived organoids have allowed for unique and unprecedented studies of human development and will continue to be an invaluable tool in research laboratories. Because organoids are engineered to more closely recapitulate in vivo biology, their clinical uses will expand to include diagnosing patient pathologies and screening pharmacologic drugs. Patient-specific organoids will be invaluable as we enter the era of personalized medicine.
Acknowledgment
The authors thank the members of the Wells and Zorn Laboratories of Cincinnati Children's Hospital Medical Center for continued support and advice.
Footnotes
Author contributions Alexandra K. Eicher was responsible for major drafting of the manuscript; H. Matthew Berns was responsible for minor drafting and editing of the manuscript; and James M. Wells provided critical revision for intellectual content and obtained funding.
Conflicts of interest The authors disclose no conflicts.
Funding Supported by National Institutes of Health grants R01DK092456, U19AI116491, U18EB021780, and U01DK103117 (J.M.W., A.K.E., and H.M.B.).
References
- 1.Willet S.G., Mills J.C. Stomach organ and cell lineage differentiation: from embryogenesis to adult homeostasis. Cell Mol Gastroenterol Hepatol. 2016;2:546–559. doi: 10.1016/j.jcmgh.2016.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kim T.H., Shivdasani R.A. Stomach development, stem cells and disease. Development. 2016;143:554–565. doi: 10.1242/dev.124891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.McCracken K.W., Wells J.M. Mechanisms of embryonic stomach development. Semin Cell Dev Biol. 2017;66:36–42. doi: 10.1016/j.semcdb.2017.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Choi E., Roland J.T., Barlow B.J., O'Neal R., Rich A.E., Nam K.T., Shi C., Goldenring J.R. Cell lineage distribution atlas of the human stomach reveals heterogeneous gland populations in the gastric antrum. Gut. 2014;63:1711–1720. doi: 10.1136/gutjnl-2013-305964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Huch M., Koo B.K. Modeling mouse and human development using organoid cultures. Development. 2015;142:3113–3125. doi: 10.1242/dev.118570. [DOI] [PubMed] [Google Scholar]
- 6.McCracken K.W., Cata E.M., Crawford C.M., Sinagoga K.L., Schumacher M., Rockich B.E., Tsai Y.H., Mayhew C.N., Spence J.R., Zavros Y., Wells J.M. Modelling human development and disease in pluripotent stem-cell-derived gastric organoids. Nature. 2014;516:400–404. doi: 10.1038/nature13863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Dedhia P.H., Bertaux-Skeirik N., Zavros Y., Spence J.R. Organoid models of human gastrointestinal development and disease. Gastroenterology. 2016;150:1098–1112. doi: 10.1053/j.gastro.2015.12.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mills J.C., Shivdasani R.A. Gastric epithelial stem cells. Gastroenterology. 2011;140:412–424. doi: 10.1053/j.gastro.2010.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Katano T., Ootani A., Mizoshita T., Tanida S., Tsukamoto H., Ozeki K., Kataoka H., Joh T. Gastric mesenchymal myofibroblasts maintain stem cell activity and proliferation of murine gastric epithelium in vitro. Am J Pathol. 2015;185:798–807. doi: 10.1016/j.ajpath.2014.11.007. [DOI] [PubMed] [Google Scholar]
- 10.Matsuo J., Kimura S., Yamamura A., Koh C.P., Hossain M.Z., Heng D.L., Kohu K., Voon D.C., Hiai H., Unno M., So J.B., Zhu F., Srivastava S., Teh M., Yeoh K.G., Osato M., Ito Y. Identification of stem cells in the epithelium of the stomach corpus and antrum of mice. Gastroenterology. 2017;152:218–231.e14. doi: 10.1053/j.gastro.2016.09.018. [DOI] [PubMed] [Google Scholar]
- 11.Karam S.M., Leblond C.P. Dynamics of epithelial cells in the corpus of the mouse stomach. I. Identification of proliferative cell types and pinpointing of the stem cell. Anat Rec. 1993;236:259–279. doi: 10.1002/ar.1092360202. [DOI] [PubMed] [Google Scholar]
- 12.Arnold K., Sarkar A., Yram M.A., Polo J.M., Bronson R., Sengupta S., Seandel M., Geijsen N., Hochedlinger K. Sox2(+) adult stem and progenitor cells are important for tissue regeneration and survival of mice. Cell Stem Cell. 2011;9:317–329. doi: 10.1016/j.stem.2011.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sarkar A., Huebner A.J., Sulahian R., Anselmo A., Xu X., Flattery K., Desai N., Sebastian C., Yram M.A., Arnold K., Rivera M., Mostoslavsky R., Bronson R., Bass A.J., Sadreyev R., Shivdasani R.A., Hochedlinger K. Sox2 suppresses gastric tumorigenesis in mice. Cell Rep. 2016;16:1929–1941. doi: 10.1016/j.celrep.2016.07.034. [DOI] [PubMed] [Google Scholar]
- 14.Sigal M., Logan C.Y., Kapalczynska M., Mollenkopf H.J., Berger H., Wiedenmann B., Nusse R., Amieva M.R., Meyer T.F. Stromal R-spondin orchestrates gastric epithelial stem cells and gland homeostasis. Nature. 2017;548:451–455. doi: 10.1038/nature23642. [DOI] [PubMed] [Google Scholar]
- 15.Choi E., Lantz T.L., Vlacich G., Keeley T.M., Samuelson L.C., Coffey R.J., Goldenring J.R., Powell A.E. Lrig1+ gastric isthmal progenitor cells restore normal gastric lineage cells during damage recovery in adult mouse stomach. Gut. 2017 doi: 10.1136/gutjnl-2017-313874. Epub 2017/08/18. https://doi.org/10.1136/gutjnl-2017-313874. PubMed PMID: 28814482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Stange D.E., Koo B.K., Huch M., Sibbel G., Basak O., Lyubimova A., Kujala P., Bartfeld S., Koster J., Geahlen J.H., Peters P.J., van Es J.H., van de Wetering M., Mills J.C., Clevers H. Differentiated Troy+ chief cells act as reserve stem cells to generate all lineages of the stomach epithelium. Cell. 2013;155:357–368. doi: 10.1016/j.cell.2013.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Barker N., Huch M., Kujala P., van de Wetering M., Snippert H.J., van Es J.H., Sato T., Stange D.E., Begthel H., van den Born M., Danenberg E., van den Brink S., Korving J., Abo A., Peters P.J., Wright N., Poulsom R., Clevers H. Lgr5(+ve) stem cells drive self-renewal in the stomach and build long-lived gastric units in vitro. Cell Stem Cell. 2010;6:25–36. doi: 10.1016/j.stem.2009.11.013. [DOI] [PubMed] [Google Scholar]
- 18.Leushacke M., Tan S.H., Wong A., Swathi Y., Hajamohideen A., Tan L.T., Goh J., Wong E., Denil S., Murakami K., Barker N. Lgr5-expressing chief cells drive epithelial regeneration and cancer in the oxyntic stomach. Nat Cell Biol. 2017;19:774–786. doi: 10.1038/ncb3541. [DOI] [PubMed] [Google Scholar]
- 19.Hayakawa Y., Ariyama H., Stancikova J., Sakitani K., Asfaha S., Renz B.W., Dubeykovskaya Z.A., Shibata W., Wang H., Westphalen C.B., Chen X., Takemoto Y., Kim W., Khurana S.S., Tailor Y., Nagar K., Tomita H., Hara A., Sepulveda A.R., Setlik W., Gershon M.D., Saha S., Ding L., Shen Z., Fox J.G., Friedman R.A., Konieczny S.F., Worthley D.L., Korinek V., Wang T.C. Mist1 expressing gastric stem cells maintain the normal and neoplastic gastric epithelium and are supported by a perivascular stem cell niche. Cancer Cell. 2015;28:800–814. doi: 10.1016/j.ccell.2015.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jenny M., Uhl C., Roche C., Duluc I., Guillermin V., Guillemot F., Jensen J., Kedinger M., Gradwohl G. Neurogenin3 is differentially required for endocrine cell fate specification in the intestinal and gastric epithelium. EMBO J. 2002;21:6338–6347. doi: 10.1093/emboj/cdf649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lee C.S., Perreault N., Brestelli J.E., Kaestner K.H. Neurogenin 3 is essential for the proper specification of gastric enteroendocrine cells and the maintenance of gastric epithelial cell identity. Genes Dev. 2002;16:1488–1497. doi: 10.1101/gad.985002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Browning K.N., Travagli R.A. Central nervous system control of gastrointestinal motility and secretion and modulation of gastrointestinal functions. Compr Physiol. 2014;4:1339–1368. doi: 10.1002/cphy.c130055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Browning K.N., Verheijden S., Boeckxstaens G.E. The vagus nerve in appetite regulation, mood, and intestinal inflammation. Gastroenterology. 2017;152:730–744. doi: 10.1053/j.gastro.2016.10.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Swartz E.M., Browning K.N., Travagli R.A., Holmes G.M. Ghrelin increases vagally mediated gastric activity by central sites of action. Neurogastroenterol Motil. 2014;26:272–282. doi: 10.1111/nmo.12261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Roosen L., Boesmans W., Dondeyne M., Depoortere I., Tack J., Vanden Berghe P. Specific hunger- and satiety-induced tuning of guinea pig enteric nerve activity. J Physiol. 2012;590:4321–4333. doi: 10.1113/jphysiol.2012.231134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yakabi K., Ro S., Onouhi T., Tanaka T., Ohno S., Miura S., Johno Y., Takayama K. Histamine mediates the stimulatory action of ghrelin on acid secretion in rat stomach. Dig Dis Sci. 2006;51:1313–1321. doi: 10.1007/s10620-005-9002-3. [DOI] [PubMed] [Google Scholar]
- 27.Taniguchi H., Ariga H., Zheng J., Ludwig K., Takahashi T. Effects of ghrelin on interdigestive contractions of the rat gastrointestinal tract. World J Gastroenterol. 2008;14:6299–6302. doi: 10.3748/wjg.14.6299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lowette K., Tack J., Vanden Berghe P. Role of corticosterone in the murine enteric nervous system during fasting. Am J Physiol Gastrointest Liver Physiol. 2014;307:G905–G913. doi: 10.1152/ajpgi.00233.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Norlen P., Ericsson P., Kitano M., Ekelund M., Hakanson R. The vagus regulates histamine mobilization from rat stomach ECL cells by controlling their sensitivity to gastrin. J Physiol. 2005;564:895–905. doi: 10.1113/jphysiol.2005.082677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Goswami C., Shimada Y., Yoshimura M., Mondal A., Oda S., Tanaka T., Sakai T., Sakata I. Motilin stimulates gastric acid secretion in coordination with ghrelin in suncus murinus. PLoS One. 2015;10:e0131554. doi: 10.1371/journal.pone.0131554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Aubin J., Dery U., Lemieux M., Chailler P., Jeannotte L. Stomach regional specification requires Hoxa5-driven mesenchymal-epithelial signaling. Development. 2002;129:4075–4087. doi: 10.1242/dev.129.17.4075. [DOI] [PubMed] [Google Scholar]
- 32.Le Guen L., Marchal S., Faure S., de Santa Barbara P. Mesenchymal-epithelial interactions during digestive tract development and epithelial stem cell regeneration. Cell Mol Life Sci. 2015;72:3883–3896. doi: 10.1007/s00018-015-1975-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kim B.M., Buchner G., Miletich I., Sharpe P.T., Shivdasani R.A. The stomach mesenchymal transcription factor Barx1 specifies gastric epithelial identity through inhibition of transient Wnt signaling. Dev Cell. 2005;8:611–622. doi: 10.1016/j.devcel.2005.01.015. [DOI] [PubMed] [Google Scholar]
- 34.Jayewickreme C.D., Shivdasani R.A. Control of stomach smooth muscle development and intestinal rotation by transcription factor BARX1. Dev Biol. 2015;405:21–32. doi: 10.1016/j.ydbio.2015.05.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.De Santa Barbara P., Williams J., Goldstein A.M., Doyle A.M., Nielsen C., Winfield S., Faure S., Roberts D.J. Bone morphogenetic protein signaling pathway plays multiple roles during gastrointestinal tract development. Dev Dyn. 2005;234:312–322. doi: 10.1002/dvdy.20554. [DOI] [PubMed] [Google Scholar]
- 36.de Santa Barbara P., van den Brink G.R., Roberts D.J. Molecular etiology of gut malformations and diseases. Am J Med Genet. 2002;115:221–230. doi: 10.1002/ajmg.10978. [DOI] [PubMed] [Google Scholar]
- 37.Doyle A.M., Roberts D.J., Goldstein A.M. Enteric nervous system patterning in the avian hindgut. Dev Dyn. 2004;229:708–712. doi: 10.1002/dvdy.20011. [DOI] [PubMed] [Google Scholar]
- 38.Faure S., de Santa Barbara P., Roberts D.J., Whitman M. Endogenous patterns of BMP signaling during early chick development. Dev Biol. 2002;244:44–65. doi: 10.1006/dbio.2002.0579. [DOI] [PubMed] [Google Scholar]
- 39.Goldstein A.M., Brewer K.C., Doyle A.M., Nagy N., Roberts D.J. BMP signaling is necessary for neural crest cell migration and ganglion formation in the enteric nervous system. Mech Dev. 2005;122:821–833. doi: 10.1016/j.mod.2005.03.003. [DOI] [PubMed] [Google Scholar]
- 40.Moniot B., Biau S., Faure S., Nielsen C.M., Berta P., Roberts D.J. de Santa Barbara P. SOX9 specifies the pyloric sphincter epithelium through mesenchymal-epithelial signals. Development. 2004;131:3795–3804. doi: 10.1242/dev.01259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Nielsen C., Murtaugh L.C., Chyung J.C., Lassar A., Roberts D.J. Gizzard formation and the role of Bapx1. Dev Biol. 2001;231:164–174. doi: 10.1006/dbio.2000.0151. [DOI] [PubMed] [Google Scholar]
- 42.Nielsen C.M., Williams J., van den Brink G.R., Lauwers G.Y., Roberts D.J. Hh pathway expression in human gut tissues and in inflammatory gut diseases. Lab Invest. 2004;84:1631–1642. doi: 10.1038/labinvest.3700197. [DOI] [PubMed] [Google Scholar]
- 43.Roberts D.J., Smith D.M., Goff D.J., Tabin C.J. Epithelial-mesenchymal signaling during the regionalization of the chick gut. Development. 1998;125:2791–2801. doi: 10.1242/dev.125.15.2791. [DOI] [PubMed] [Google Scholar]
- 44.Roberts D.J. Molecular mechanisms of development of the gastrointestinal tract. Dev Dyn. 2000;219:109–120. doi: 10.1002/1097-0177(2000)9999:9999<::aid-dvdy1047>3.3.co;2-y. [DOI] [PubMed] [Google Scholar]
- 45.Smith D.M., Nielsen C., Tabin C.J., Roberts D.J. Roles of BMP signaling and Nkx2.5 in patterning at the chick midgut-foregut boundary. Development. 2000;127:3671–3681. doi: 10.1242/dev.127.17.3671. [DOI] [PubMed] [Google Scholar]
- 46.van Den Brink G.R., de Santa Barbara P., Roberts D.J. Development. Epithelial cell differentiation–a Mather of choice. Science. 2001;294:2115–2116. doi: 10.1126/science.1067751. [DOI] [PubMed] [Google Scholar]
- 47.van den Brink G.R., Hardwick J.C., Nielsen C., Xu C., ten Kate F.J., Glickman J., van Deventer S.J., Roberts D.J., Peppelenbosch M.P. Sonic hedgehog expression correlates with fundic gland differentiation in the adult gastrointestinal tract. Gut. 2002;51:628–633. doi: 10.1136/gut.51.5.628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.McCracken K.W., Aihara E., Martin B., Crawford C.M., Broda T., Treguier J., Zhang X., Shannon J.M., Montrose M.H., Wells J.M. Wnt/beta-catenin promotes gastric fundus specification in mice and humans. Nature. 2017;541:182–187. doi: 10.1038/nature21021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kiefer S.M., Robbins L., Rauchman M. Conditional expression of Wnt9b in Six2-positive cells disrupts stomach and kidney function. PLoS One. 2012;7:e43098. doi: 10.1371/journal.pone.0043098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Prakash A., Udager A.M., Saenz D.A., Gumucio D.L. Roles for Nkx2–5 and Gata3 in the ontogeny of the murine smooth muscle gastric ligaments. Am J Physiol Gastrointest Liver Physiol. 2014;307:G430–G436. doi: 10.1152/ajpgi.00360.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Self M., Geng X., Oliver G. Six2 activity is required for the formation of the mammalian pyloric sphincter. Dev Biol. 2009;334:409–417. doi: 10.1016/j.ydbio.2009.07.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Theodosiou N.A., Tabin C.J. Sox9 and Nkx2.5 determine the pyloric sphincter epithelium under the control of BMP signaling. Dev Biol. 2005;279:481–490. doi: 10.1016/j.ydbio.2004.12.019. [DOI] [PubMed] [Google Scholar]
- 53.Udager A.M., Prakash A., Saenz D.A., Schinke M., Moriguchi T., Jay P.Y., Lim K.C., Engel J.D., Gumucio D.L. Proper development of the outer longitudinal smooth muscle of the mouse pylorus requires Nkx2-5 and Gata3. Gastroenterology. 2014;146:157–165.e10. doi: 10.1053/j.gastro.2013.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Li Y., Pan J., Wei C., Chen J., Liu Y., Liu J., Zhang X., Evans S.M., Cui Y., Cui S. LIM homeodomain transcription factor Isl1 directs normal pyloric development by targeting Gata3. BMC Biol. 2014;12:25. doi: 10.1186/1741-7007-12-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Mao J., Kim B.M., Rajurkar M., Shivdasani R.A., McMahon A.P. Hedgehog signaling controls mesenchymal growth in the developing mammalian digestive tract. Development. 2010;137:1721–1729. doi: 10.1242/dev.044586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Nagy N., Barad C., Graham H.K., Hotta R., Cheng L.S., Fejszak N., Goldstein A.M. Sonic hedgehog controls enteric nervous system development by patterning the extracellular matrix. Development. 2016;143:264–275. doi: 10.1242/dev.128132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Faure S., McKey J., Sagnol S., de Santa Barbara P. Enteric neural crest cells regulate vertebrate stomach patterning and differentiation. Development. 2015;142:331–342. doi: 10.1242/dev.118422. [DOI] [PubMed] [Google Scholar]
- 58.Smith D.M., Tabin C.J. BMP signalling specifies the pyloric sphincter. Nature. 1999;402:748–749. doi: 10.1038/45439. [DOI] [PubMed] [Google Scholar]
- 59.Roberts D.J., Johnson R.L., Burke A.C., Nelson C.E., Morgan B.A., Tabin C. Sonic hedgehog is an endodermal signal inducing Bmp-4 and Hox genes during induction and regionalization of the chick hindgut. Development. 1995;121:3163–3174. doi: 10.1242/dev.121.10.3163. [DOI] [PubMed] [Google Scholar]
- 60.Narita T., Saitoh K., Kameda T., Kuroiwa A., Mizutani M., Koike C., Iba H., Yasugi S. BMPs are necessary for stomach gland formation in the chicken embryo: a study using virally induced BMP-2 and Noggin expression. Development. 2000;127:981–988. doi: 10.1242/dev.127.5.981. [DOI] [PubMed] [Google Scholar]
- 61.Lo L., Sommer L., Anderson D.J. MASH1 maintains competence for BMP2-induced neuronal differentiation in post-migratory neural crest cells. Curr Biol. 1997;7:440–450. doi: 10.1016/s0960-9822(06)00191-6. [DOI] [PubMed] [Google Scholar]
- 62.Pisano J.M., Colon-Hastings F., Birren S.J. Postmigratory enteric and sympathetic neural precursors share common, developmentally regulated, responses to BMP2. Dev Biol. 2000;227:1–11. doi: 10.1006/dbio.2000.9876. [DOI] [PubMed] [Google Scholar]
- 63.Rainbow R.S., Won H.K., Zeng L. The role of Nkx3.2 in chondrogenesis. Front Biol (Beijing) 2014;9:376–381. doi: 10.1007/s11515-014-1321-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Tribioli C., Lufkin T. The murine Bapx1 homeobox gene plays a critical role in embryonic development of the axial skeleton and spleen. Development. 1999;126:5699–5711. doi: 10.1242/dev.126.24.5699. [DOI] [PubMed] [Google Scholar]
- 65.Salama N.R., Hartung M.L., Müller A. Life in the human stomach: persistence strategies of the bacterial pathogen Helicobacter pylori. Nat Rev Microbiol. 2013;11:385–399. doi: 10.1038/nrmicro3016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Kusters J.G., van Vliet A.H.M., Kuipers E.J. Pathogenesis of Helicobacter pylori infection. Clin Microbiol Rev. 2006;19:449–490. doi: 10.1128/CMR.00054-05. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Uemura N., Okamoto S., Yamamoto S., Matsumura N., Yamaguchi S., Yamakido M., Taniyama K., Sasaki N., Schlemper R.J. Helicobacter pylori infection and the development of gastric cancer. N Engl J Med. 2001;345:784–789. doi: 10.1056/NEJMoa001999. [DOI] [PubMed] [Google Scholar]
- 68.Kuipers E.J., Thijs J.C., Festen H.P. The prevalence of Helicobacter pylori in peptic ulcer disease. Aliment Pharmacol Ther. 1995;9(Suppl 2):59–69. [PubMed] [Google Scholar]
- 69.Van Zanten S.J., Dixon M.F., Lee A. The gastric transitional zones: neglected links between gastroduodenal pathology and helicobacter ecology. Gastroenterology. 1999;116:1217–1229. doi: 10.1016/s0016-5085(99)70025-9. [DOI] [PubMed] [Google Scholar]
- 70.O'Rourke J.L., Lee A. Animal models of Helicobacter pylori infection and disease. Microbes Infect. 2003;5:741–748. doi: 10.1016/s1286-4579(03)00123-0. [DOI] [PubMed] [Google Scholar]
- 71.Zorn A.M., Wells J.M. Vertebrate endoderm development and organ formation. Annu Rev Cell Dev Biol. 2009;25:221–251. doi: 10.1146/annurev.cellbio.042308.113344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Spence J.R., Mayhew C.N., Rankin S.A., Kuhar M.F., Vallance J.E., Tolle K., Hoskins E.E., Kalinichenko V.V., Wells S.I., Zorn A.M., Shroyer N.F., Wells J.M. Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature. 2011;470:105–109. doi: 10.1038/nature09691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Wells J.M., Spence J.R. How to make an intestine. Development. 2014;141:752–760. doi: 10.1242/dev.097386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Green J.B., Smith J.C. Graded changes in dose of a Xenopus activin A homologue elicit stepwise transitions in embryonic cell fate. Nature. 1990;347:391–394. doi: 10.1038/347391a0. [DOI] [PubMed] [Google Scholar]
- 75.Shen M.M. Nodal signaling: developmental roles and regulation. Development. 2007;134:1023–1034. doi: 10.1242/dev.000166. [DOI] [PubMed] [Google Scholar]
- 76.Clements D., Friday R.V., Woodland H.R. Mode of action of VegT in mesoderm and endoderm formation. Development. 1999;126:4903–4911. doi: 10.1242/dev.126.21.4903. [DOI] [PubMed] [Google Scholar]
- 77.Aoki T.O., David N.B., Minchiotti G., Saint-Etienne L., Dickmeis T., Persico G.M., Strahle U., Mourrain P., Rosa F.M. Molecular integration of casanova in the Nodal signalling pathway controlling endoderm formation. Development. 2002;129:275–286. doi: 10.1242/dev.129.2.275. [DOI] [PubMed] [Google Scholar]
- 78.Ben-Haim N., Lu C., Guzman-Ayala M., Pescatore L., Mesnard D., Bischofberger M., Naef F., Robertson E.J., Constam D.B. The nodal precursor acting via activin receptors induces mesoderm by maintaining a source of its convertases and BMP4. Dev Cell. 2006;11:313–323. doi: 10.1016/j.devcel.2006.07.005. [DOI] [PubMed] [Google Scholar]
- 79.Hagos E.G., Dougan S.T. Time-dependent patterning of the mesoderm and endoderm by Nodal signals in zebrafish. BMC Dev Biol. 2007;7:22. doi: 10.1186/1471-213X-7-22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.D'Amour K.A., Agulnick A.D., Eliazer S., Kelly O.G., Kroon E., Baetge E.E. Efficient differentiation of human embryonic stem cells to definitive endoderm. Nat Biotechnol. 2005;23:1534–1541. doi: 10.1038/nbt1163. [DOI] [PubMed] [Google Scholar]
- 81.Sinagoga K.L., Wells J.M. Generating human intestinal tissues from pluripotent stem cells to study development and disease. EMBO J. 2015;34:1149–1163. doi: 10.15252/embj.201490686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.McLin V.A., Rankin S.A., Zorn A.M. Repression of Wnt/beta-catenin signaling in the anterior endoderm is essential for liver and pancreas development. Development. 2007;134:2207–2217. doi: 10.1242/dev.001230. [DOI] [PubMed] [Google Scholar]
- 83.Dufort D., Schwartz L., Harpal K., Rossant J. The transcription factor HNF3beta is required in visceral endoderm for normal primitive streak morphogenesis. Development. 1998;125:3015–3025. doi: 10.1242/dev.125.16.3015. [DOI] [PubMed] [Google Scholar]
- 84.Kinkel M.D., Eames S.C., Alonzo M.R., Prince V.E. Cdx4 is required in the endoderm to localize the pancreas and limit beta-cell number. Development. 2008;135:919–929. doi: 10.1242/dev.010660. [DOI] [PubMed] [Google Scholar]
- 85.Martinez Barbera J.P., Clements M., Thomas P., Rodriguez T., Meloy D., Kioussis D., Beddington R.S. The homeobox gene Hex is required in definitive endodermal tissues for normal forebrain, liver and thyroid formation. Development. 2000;127:2433–2445. doi: 10.1242/dev.127.11.2433. [DOI] [PubMed] [Google Scholar]
- 86.Chawengsaksophak K., de Graaff W., Rossant J., Deschamps J., Beck F. Cdx2 is essential for axial elongation in mouse development. Proc Natl Acad Sci U S A. 2004;101:7641–7645. doi: 10.1073/pnas.0401654101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Davenport C., Diekmann U., Budde I., Detering N., Naujok O. Anterior-posterior patterning of definitive endoderm generated from human embryonic stem cells depends on the differential signaling of retinoic acid, Wnt-, and BMP-signaling. Stem Cell. 2016;34:2635–2647. doi: 10.1002/stem.2428. [DOI] [PubMed] [Google Scholar]
- 88.Stevens M.L., Chaturvedi P., Rankin S.A., Macdonald M., Jagannathan S., Yukawa M., Barski A., Zorn A.M. Genomic integration of Wnt/beta-catenin and BMP/Smad1 signaling coordinates foregut and hindgut transcriptional programs. Development. 2017;144:1283–1295. doi: 10.1242/dev.145789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Wang Z., Dolle P., Cardoso W.V., Niederreither K. Retinoic acid regulates morphogenesis and patterning of posterior foregut derivatives. Dev Biol. 2006;297:433–445. doi: 10.1016/j.ydbio.2006.05.019. [DOI] [PubMed] [Google Scholar]
- 90.Rankin S.A., Kormish J., Kofron M., Jegga A., Zorn A.M. A gene regulatory network controlling hhex transcription in the anterior endoderm of the organizer. Dev Biol. 2011;351:297–310. doi: 10.1016/j.ydbio.2010.11.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.McCauley H.A., Wells J.M. Pluripotent stem cell-derived organoids: using principles of developmental biology to grow human tissues in a dish. Development. 2017;144:958–962. doi: 10.1242/dev.140731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Zhang F., Wen Y., Guo X. CRISPR/Cas9 for genome editing: progress, implications and challenges. Hum Mol Genet. 2014;23:R40–R46. doi: 10.1093/hmg/ddu125. [DOI] [PubMed] [Google Scholar]
- 93.Takahashi K., Tanabe K., Ohnuki M., Narita M., Ichisaka T., Tomoda K., Yamanaka S. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell. 2007;131:861–872. doi: 10.1016/j.cell.2007.11.019. [DOI] [PubMed] [Google Scholar]
- 94.Drubin D.G., Hyman A.A. Stem cells: the new “model organism”. Mol Biol Cell. 2017;28:1409–1411. doi: 10.1091/mbc.E17-03-0183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Watson C.L., Mahe M.M., Munera J., Howell J.C., Sundaram N., Poling H.M., Schweitzer J.I., Vallance J.E., Mayhew C.N., Sun Y., Grabowski G., Finkbeiner S.R., Spence J.R., Shroyer N.F., Wells J.M., Helmrath M.A. An in vivo model of human small intestine using pluripotent stem cells. Nat Med. 2014;20:1310–1314. doi: 10.1038/nm.3737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Workman M.J., Mahe M.M., Trisno S., Poling H.M., Watson C.L., Sundaram N., Chang C.F., Schiesser J., Aubert P., Stanley E.G., Elefanty A.G., Miyaoka Y., Mandegar M.A., Conklin B.R., Neunlist M., Brugmann S.A., Helmrath M.A., Wells J.M. Engineered human pluripotent-stem-cell-derived intestinal tissues with a functional enteric nervous system. Nat Med. 2017;23:49–59. doi: 10.1038/nm.4233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Carr D.F., Ayehunie S., Davies A., Duckworth C.A., French S., Hall N., Hussain S., Mellor H.R., Norris A., Park B.K., Penrose A., Pritchard D.M., Probert C.S., Ramaiah S., Sadler C., Schmitt M., Shaw A., Sidaway J.E., Vries R.G., Wagoner M., Pirmohamed M. Towards better models and mechanistic biomarkers for drug-induced gastrointestinal injury. Pharmacol Ther. 2017;172:181–194. doi: 10.1016/j.pharmthera.2017.01.002. [DOI] [PubMed] [Google Scholar]
- 98.Brugmann S.A., Wells J.M. Building additional complexity to in vitro-derived intestinal tissues. Stem Cell Res Ther. 2013;4(Suppl 1):S1. doi: 10.1186/scrt362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Munera J.O., Wells J.M. Generation of gastrointestinal organoids from human pluripotent stem cells. Methods Mol Biol. 2017;1597:167–177. doi: 10.1007/978-1-4939-6949-4_12. [DOI] [PubMed] [Google Scholar]
- 100.Aurora M., Spence J.R. hPSC-derived lung and intestinal organoids as models of human fetal tissue. Dev Biol. 2016;420:230–238. doi: 10.1016/j.ydbio.2016.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Takebe T., Sekine K., Enomura M., Koike H., Kimura M., Ogaeri T., Zhang R.R., Ueno Y., Zheng Y.W., Koike N., Aoyama S., Adachi Y., Taniguchi H. Vascularized and functional human liver from an iPSC-derived organ bud transplant. Nature. 2013;499:481–484. doi: 10.1038/nature12271. [DOI] [PubMed] [Google Scholar]