Like in other eukaryotic organisms, mitogen-activated protein (MAP) kinase cascades play important roles in response to host and environmental signals in fungal pathogens. In general, mitogen-activated protein kinase (MAPK) is activated by phosphorylation at the well-conserved threonine-x-tyrosine (TXY) motif by mitogen-activated protein kinase (MEK), which is in turn activated by mitogen-activated protein kinase (MEKK). The budding yeast Saccharomyces cerevisiae has five MAPK pathways that regulate mating, invasive growth, cell wall integrity, osmoregulation, and ascospore formation. Except for ascosporogenesis-specific MAPK sporulation-specific mitogen-activated protein kinase (Smk1), other yeast MAPKs are conserved in plant-pathogenic ascomycetes to regulate different infection and developmental processes, which is the focus of this review. In phytopathogenic basidiomycetes, MAPKs have only been well characterized in Ustilago maydis.
The Fus3/Kss1 orthologs regulate appressorium formation and other infection processes
Most filamentous ascomycetes have only one ortholog of yeast Fus3 and Kss1 MAPKs that function downstream from Ste11–Ste7 in the pheromone response and filamentation pathways. In over 20 plant pathogenic fungi characterized, this MAPK is important for plant infection [1,2]. In the rice blast fungus Magnaporthe oryzae, pathogenicity MAP kinase 1 (PMK1) is essential for appressorium formation and invasive growth (Fig 1A). Its ortholog is also required for appressorium formation in all the other appressorium-forming pathogens studied (Table 1). Expression of its orthologs from fungi such as Colletotrichum lagenarium and Puccinia striiformis rescues the appressorium formation defect of pmk1 mutant, indicating the well-conserved nature of this MAPK (S1 Table). In non-appressorium-forming pathogens, this pathway is also important for plant penetration and infectious growth in various fungi, such as biotrophic Claviceps purpurea, hemibiotrophic Mycosphaerella graminicola, and necrotrophic Stagonospora nodorum [1,2]. kss1 mutants of the multihost pathogen Fusarium oxysporum are nonpathogenic on tomato plants but fully pathogenic in a murine model system [3], indicating that this MAPK plays different roles in plant and animal pathogenesis. In U. maydis, although Kpp6 plays a more important role in appressorium penetration than Kpp2, both kpp2 and kpp6 mutants are attenuated in virulence, and kpp2 kpp6 double mutants are nonpathogenic [4]. Another putative MAPK, Crk1, that was first identified as a homolog of yeast Ime2 also regulates morphogenesis and plant infection in U. maydis [5].
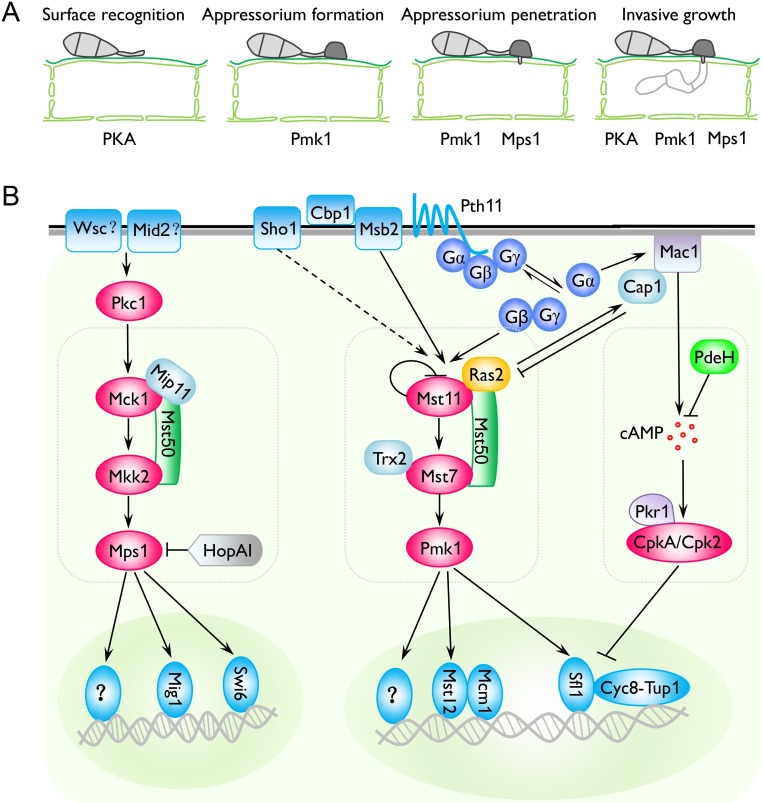
Fig 1. The Pmk1 and Mps1 pathways and their relationship with cAMP signaling in Magnaporthe oryzae.
A. Distinct and overlapping functions of the cAMP-PKA pathway and Pmk1 and Mps1 MAPK cascades during plant infection. B. Physical and chemical signals known to trigger appressorium formation include surface hydrophobicity and hardness, cutin monomers, plant surface waxes, and primary alcohols. Msb2, Sho1, Pth11, and Cbp1 are involved in recognizing extracellular or surface signals to activate the downstream cAMP-PKA pathway and Mst11-Mst7-Pmk1 MAPK cascade. Both the trimeric G-proteins and Ras monomeric G-proteins are functionally related to these two pathways that regulate appressorium formation, penetration, and invasive growth. Although its upstream sensors have not been characterized, the Bck1-Mkk2-Mps1 cascade likely functions downstream from PKC and is important for sporulation, appressorium penetration, and pathogenesis via downstream Mig1, Swi6, and possibly other transcription factors. The adapter protein Mst50 is involved in both Pmk1 and Mps1 pathways. Mip11 is a RACK protein that interacts with both Mst50 and Mck1. Pmk1 positively regulates Mst12, Mcm1, Sfl1, and likely other transcription factors during different infection processes. Together with the Cyc8-Tup1 corepressor complex, Sfl1 also functions as a transcriptional repressor for hyphal growth. cAMP, cyclic adenosine monophosphate; MAPK, MAP kinase; PKA, protein kinase A; PKC, protein kinase C; RACK, receptor for activated C kinase.
Table 1. MAP kinases characterized in plant pathogenic fungi.
| Fungal species | Orthologs of yeast | ||
|---|---|---|---|
| Fus3/Kss1 | Slt2 | Hog1 | |
| Alternaria alternata* | Fus3 | Slt2 | |
| A. brassicicola* | Amk1 | ||
| Bipolaris oryzae | Bmk1 | Srm1 | |
| Blumeria graminis* | Mpk1 | ||
| Botrytis cinerea* | Bmp1 | Bmp3 | Sak1 |
| Claviceps purpurea | Cpmk1 | ||
| Cochliobolus heterostrophus* | Chk1 | ||
| Cochliobolus sativus* | Fus3 | Slt2 | Cshog1 |
| Colletotrichum gloeosporioides* | CgMk1 | Cgl-Slt2 | |
| Colletotrichum higginsianum* | ChMK1 | ||
| Colletotrichum lagenarium* | Cmk1 | Maf1 | |
| Colletotrichum orbiculare* | Osc1 | ||
| Fusarium graminearum | Gpmk1 | Mgv1 | FgHog1 |
| F. oxysporum | Fmk1 | Mpk1 | |
| F. verticillioides | Mk1 | ||
| Heterobasidion annosum | HaHog1 | ||
| Magnaporthe oryzae* | Pmk1 | Mps1 | Osm1 |
| Mycosphaerella graminicola | Fus3 | MgSlt2 | MgHog1 |
| Puccinia striiformis | Mapk1 | ||
| Pyrenophora teres* | Ptk1 | ||
| Sclerotinia sclerotiorum* | Smk1 | Smk3 | |
| Setosphaeria turcica* | Stk2 | ||
| Stagonospora nodorum | Mak2 | ||
| Ustilaginoidea virens | UvHog1 | ||
| Ustilago maydis | Kpp2 Kpp6 | UmHog1 | |
| Verticillium dahliae* | Vmk1 | VdHog1 | |
* Pathogens that form appressoria during plant infection.
Abbreviations: MAP, mitogen-activated protein.
Unlike its conserved role in pathogenesis, the functions of this MAPK in other development processes vary among different fungi [1,2]. For example, the Fus3 ortholog is important for deoxynivalenol production in F. graminearum and fumonisin biosynthesis in F. verticillioides, indicating a regulatory role in secondary metabolism. Whereas this MAPK is important for conidiation in Alternaria brassicicola and conidium germination in C. lagenarium, its ortholog regulates pycnidium formation and sclerotium development in M. graminicola and Sclerotinia sclerotiorum (S1 Table). In A. alternata, AsFus3 is important for conidiation and copper fungicide resistance.
In M. oryzae, Pmk1 is activated by its upstream MEK Mst7 and MEKK Mst11 (Fig 1B). Without a yeast Ste5 ortholog, Mst50 functions as an adaptor protein for the Mst11-Mst7 interaction, but Mst7 interacts with Pmk1 via its MAPK-docking site [1]. The formation of Mst7 homodimers involves the thioredoxins and is important for Pmk1 activation [6]. Besides its intramolecular self-inhibitory binding, Mst11 also interacts with Ras proteins via the Ras-association domain for Pmk1 activation [7,8]. Upstream components of this MAPK pathway also have been characterized in other fungal pathogens [1,9]. In U. maydis, the Ste50 ortholog Ubc2 functions as an adaptor protein for the Kpp4–Fuz7–Kpp2/Kpp6 cascade [10]. Orthologs of Ste12, a downstream transcription factor of yeast Fus3 and Kss1, also play a critical role in development and pathogenesis in fungal pathogens [1]. In M. oryzae and C. lagenarium, deletion of Ste12 ortholog results in the loss of pathogenicity and defects in appressorium penetration.
The Slt2 cell wall integrity pathway also has a conserved role in pathogenesis
The MAPK cascade orthologous to the yeast Bck1–Mkk1/Mkk2–Slt2 is conserved in filamentous ascomycetes to regulate cell wall integrity (CWI) and pathogenesis [1,2]. In M. oryzae, Mps1 is essential for plant infection, and mps1 mutant is defective in appressorium penetration. Expression of HopAI, a Pseudomonas MAPK–inactivating effector, strongly affects Mps1 phosphorylation and virulence [11]. The CWI MAPK pathway is also important for pathogenesis in other plant pathogens (Table 1). However, its role in appressorium formation or initial penetration varies among different fungi. Although its ortholog is important for early stages of appressorium development in C. lagenarium and Colletotrichum gloeosporioides, Mps1 is dispensable for appressorium formation in M. oryzae. In M. graminicola, MgSlt2 mutant is normal in stomata penetration but defective in developing invasive hyphae.
Beside their conserved roles in infection and cell wall integrity, Slt2 orthologs also are involved in regulating different biological processes in plant pathogenic fungi. For example, the mps1 mutant is normal in growth rate but has severe defects in aerial hyphal growth and conidiation in M. oryzae. In S. sclerotiorum, smk3 mutant is reduced in sclerotium formation but increased in aerial hyphal growth [12]. Whereas M. graminicola Mgslt2 mutant is hypersensitive to azole fungicides, Botrytis cinerea bmp3 mutant has increased sensitivity to paraquat and fludioxonil (S1 Table).
A number of upstream and downstream components of the CWI pathway have been functionally characterized in M. oryzae, F. graminearum, and other fungi [1,9]. Whereas most plant pathogens have a single MAPK, MEK, and MEKK, a species-specific duplication of the MEKK is observed in F. oxysporum and two rust fungi. The bck1 mutant of Cryphonectria parasitica has severe growth defects and often produces fast-growing sectors although the underlying mechanism is not clear [13]. For the downstream targets of the CWI MAPK cascade, the orthologs of yeast Rlm1 and Swi6 are conserved in filamentous ascomycetes. In M. oryzae, the mig1 mutant deleted of the MCM1/AGAMOUS/DEFICIENS/SRF (MADS) box transcription factor orthologous to Rlm1 is normal in vegetative growth and the formation of melanized appressoria but defective in the differentiation and growth of invasive hyphae. The Moswi6 mutant deleted of the transcriptional regulator orthologous to yeast Swi6 is defective in cell wall integrity, hyphal growth, and appressorium penetration [1].
The osmoregulation pathway plays a species-specific role in pathogenesis
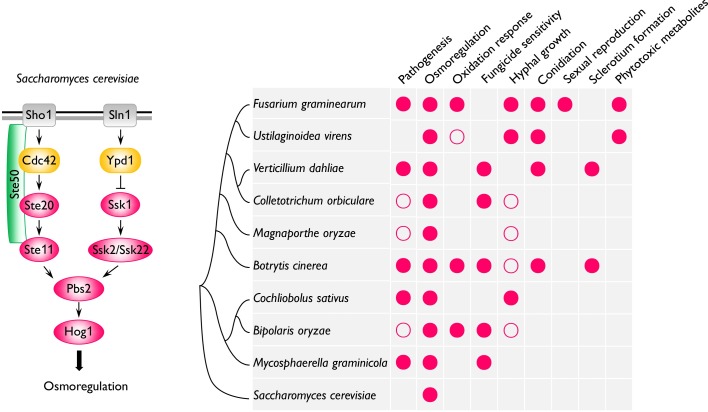
Unlike the other two MAPKs with the threonine-glutamate-tyrosine (TEY) phosphorylation sites, the Hog1 or OS-2 ortholog with the threonine-glycine-tyrosine (TGY) phosphorylation sites is not important for pathogenesis in all the plant pathogenic fungi (Fig 2). In M. oryzae, Osm1 is dispensable for appressorium turgor generation and pathogenesis. Hog1 ortholog is also dispensable for plant infection in Cochliobolus orbiculare and Bipolaris oryzae [1,2]. However, mutants blocked in this pathway are defective in plant infection in B. cinerea, M. graminicola, and other fungi (S1 Table). Whereas Mghog1 mutant is nonpathogenic and defective in the yeast-like-to-hyphal growth switch in M. graminicola, deletion of this MAPK reduces the production of phytotoxic metabolites in F. graminearum and Ustilaginoidea virens [1,14]. The Cochliobolus sativus Cshog1 mutant is normal in root infection but significantly reduced in virulence on barley leaves [15]. Therefore, the function of this MAPK pathway in pathogenesis may be not only species-specific but also tissue-specific.
Fig 2. Functional diversity of the HOG pathway in yeast and plant-pathogenic fungi.
Schematic model of the HOG pathway that is mainly involved in hyperosmoregulation in S. cerevisiae. Although lacking redundant MEK kinases, filamentous ascomycetes generally have orthologs of all these conserved components of the Hog1 pathway, including MEKK, MEK, and upstream phosphorelay and sensor proteins. Besides its conserved role in osmoregulation, this pathway has species-specific functions in pathogenesis, vegetative growth, fungicide sensitivity, sexual and asexual development, and responses to oxidative, cell wall, and other stresses in different plant pathogenic fungi. Filled and empty circles indicate that the Hog1/OS-2 kinase is important or dispensable, respectively, for specific functions characterized in different fungi. HOG, high-osmolarity glycerol; MEK, MAPK kinase; MEKK, MEK kinase.
In phytopathogenic fungi, the Hog1 pathway also plays a species-specific role in growth and development, such as the regulation of perithecium formation in F. graminearum and microsclerotium formation in Verticillium dahliae (S1 Table). In B. cinerea, Sak1 positively controls conidiation but negatively regulates sclerotium development. Nevertheless, in general, this TGY MAPK is important for oxidative stress and responsible for sensitivity to phenylpyrrole fungicides, although its role in response to cell wall and other stresses may vary among different fungi. The upstream MEK and MEKK and downstream transcription factors of the Hog1 pathway also have been characterized in several fungal pathogens [1,2,16]. Deletion of FgPbs2 and FgSsk22 results in the same defects as Fgos2 mutant in F. graminearum [9], in which the ATF/CREB transcription factor FgAtf1 interacts with FgOs2 in the nucleus under osmotic stress and constitutive expression of FgATF1 almost fully complements Fgos2 defects in osmoregulation and pathogenesis [17].
Upstream receptors or sensors
Unlike plants and animals, fungi lack receptor kinases or receptor-like kinases. However, in comparison with yeast, G-protein–coupled receptor (GPCR) genes are expanded in plant pathogens, such as 76 and 116 putative GPCR genes in M. oryzae and F. graminearum, respectively. In M. oryzae, Pth11, a noncanonical GPCR with the CFEM motif, is involved in surface recognition for appressorium formation [18]. It is internalized and transported to dynamic tubulovesicular endosomal compartments with its downstream signaling components [1,19]. In plant pathogens, CFEM domain–containing GPCRs are often induced during plant infection [20]. In F. oxysporum, the ortholog of yeast Ste2 pheromone receptor appears to be involved in the sensing of α-pheromone, peroxidase, and other host compounds [21]. However, deletion of Ste2 ortholog has no obvious effect on virulence.
The other two receptor-like genes functioning upstream from MAPK pathways in plant pathogens are orthologous to yeast Msb2 and Sho1 [1,2]. In U. maydis and F. oxysporum, Msb2 plays a major and Sho1 plays a minor role in activating downstream MAPKs and pathogenesis. In M. oryzae, the Momsb2 Mosho1 mutant rarely forms appressoria on artificial hydrophobic surfaces but still develops appressoria in response to plant surface waxes and primary alcohols. MoMsb2 is functionally related to another mucin-like protein, MoCbp1, because Momsb2 Mocbp1 mutant is defective in Pmk1 activation and non-pathogenic [22]. The Msb2 ortholog is also important for plant infection in V. dahliae but not in B. cinerea, although Bmp1 MAPK is activated in a Msb2-dependent manner [23].
No receptors have been characterized for the CWI pathway in plant pathogenic fungi, although they have Wsc1 and Mid1 homologs. In contrast, components of the two-component phosphorelay system upstream from the Hog1 cascade have been characterized in several fungal pathogens [1,2]. In M. oryzae, the two histidine kinases—MoSln1 and MoHik1—differ in sensing salt and sugar stresses, but both of them are important for full virulence. MoYpd1, the only intermediate signal transfer protein, is important for pathogenesis and hyperactivation of Osm1 in response to fungicides and osmotic stress.
Cross-talking among different MAPK pathways
In Cochliobolus heterostrophus, Chk1 and Mps1 coregulate several downstream targets, such as the Colletotrichum melanin regulation (CMR1) transcription factor and melanin biosynthesis genes. Hog1 plays an opposite role in the regulation of some Chk1 targets, although it has overlapping functions with Chk1 during plant infection [24]. In F. oxysporum, both Fmk1 and Mpk1 regulate responses to cell wall and heat stresses, and Hog1 likely negatively controls the activation of Fmk1 and Mpk1 [25]. In M. oryzae, besides its role in the Pmk1 pathway, Mst50 interacts with Mck1 and Mkk2, and the mst50 mutant is defective in Mps1 phosphorylation under cell wall stress. Deletion of MST50 also affects Osm1 activation in response to hyperosmotic stress, and Hik1 interacts with Mst50 [26].
Because the cyclic adenosine monophosphate–protein kinase A (cAMP-PKA) pathway also regulates various developmental and infection processes, cross-talking between cyclic adenosine monophosphate (cAMP) signaling and MAPK cascades must occur and likely involve different mechanisms in plant pathogenic fungi [1,2,27]. In S. cerevisiae, inhibition of β-1,3-glucan synthesis led to the activation of the CWI pathway and suppression of protein kinase A (PKA) signaling [28]. In U. maydis, the Prf1 transcription factor functions downstream from both the cAMP-PKA and MAPK pathways. In M. oryzae, MoRas2 functions upstream from both cAMP signaling and Pmk1 cascade (Fig 1B). Loss-of-function mutations in MoSfl1, a Pmk1-interacting transcription factor, suppress the growth defects of cpkA cpk2 mutant [29].
Overall, plant pathogenic fungi must properly respond to host and environmental signals throughout the infection cycle. Further characterization of cross-talking among MAPK cascades or their relationships with other signaling pathways will lead to better understanding of the regulatory network involved in the regulation of different infection processes. Another important area is to identify and characterize the upstream receptors by systematic characterization of GPCRs. The expansion of GPCR genes in plant pathogenic fungi strongly suggests their importance for the recognition of extracellular cues, although many of them may have overlapping functions. Furthermore, MAPK pathways may regulate other biological processes that have not been well studied in plant pathogens, such as response to volatile signals.
Supporting information
(DOCX)
Funding Statement
This work was supported by grants from USDA NIFA (Award number: 2013-68004-20378), National Natural Science Foundation of China (No. 31772114), and Northwest A & F University Young Talent program. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Li G, Zhou X, Xu JR (2012) Genetic control of infection-related development in Magnaporthe oryzae. Curr Opin Microbiol 15: 678–684. doi: 10.1016/j.mib.2012.09.004 [DOI] [PubMed] [Google Scholar]
- 2.Turra D, Segorbe D, Di Pietro A (2014) Protein kinases in plant-pathogenic fungi: conserved regulators of infection. Annu Rev Phytopathol 52: 267–288. doi: 10.1146/annurev-phyto-102313-050143 [DOI] [PubMed] [Google Scholar]
- 3.Ortoneda M, Guarro J, Madrid MP, Caracuel Z, Roncero MIG, et al. (2004) Fusarium oxysporum as a multihost model for the genetic dissection of fungal virulence in plants and mammals. Infect Immun 72: 1760–1766. doi: 10.1128/IAI.72.3.1760-1766.2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Di Stasio M, Brefort T, Mendoza-Mendoza A, Munch K, Kahmann R (2009) The dual specificity phosphatase Rok1 negatively regulates mating and pathogenicity in Ustilago maydis. Mol Microbiol 73: 73–88. doi: 10.1111/j.1365-2958.2009.06747.x [DOI] [PubMed] [Google Scholar]
- 5.Garrido E, Voss U, Muller P, Castillo-Lluva S, Kahmann R, et al. (2004) The induction of sexual development and virulence in the smut fungus Ustilago maydis depends on Crk1, a novel MAPK protein. Genes Dev 18: 3117–3130. doi: 10.1101/gad.314904 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhang S, Jiang C, Zhang Q, Qi L, Li C, et al. (2016) Thioredoxins are involved in the activation of the PMK1 MAP kinase pathway during appressorium penetration and invasive growth in Magnaporthe oryzae. Environ Microbiol 18: 3768–3784. doi: 10.1111/1462-2920.13315 [DOI] [PubMed] [Google Scholar]
- 7.Zhou XY, Zhao XH, Xue CY, Dai YF, Xu JR (2014) Bypassing both surface attachment and surface recognition requirements for appressorium formation by overactive Ras signaling in Magnaporthe oryzae. Mol Plant Microbe In 27: 996–1004. [DOI] [PubMed] [Google Scholar]
- 8.Qi LL, Kim Y, Jiang C, Li Y, Peng YL, et al. (2015) Activation of Mst11 and feedback inhibition of germ tube growth in Magnaporthe oryzae. Mol Plant Microbe In 28: 881–891. [DOI] [PubMed] [Google Scholar]
- 9.Wang C, Zhang S, Hou R, Zhao Z, Zheng Q, et al. (2011) Functional analysis of the kinome of the wheat scab fungus Fusarium graminearum. PLoS Pathog 7: e1002460 doi: 10.1371/journal.ppat.1002460 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Klosterman SJ, Martinez-Espinoza AD, Andrews DL, Seay JR, Gold SE (2008) Ubc2, an ortholog of the yeast Ste50p adaptor, possesses a basidiomycete-specific carboxy terminal extension essential for pathogenicity independent of pheromone response. Mol Plant Microbe In 21: 110–121. [DOI] [PubMed] [Google Scholar]
- 11.Zhang X, Liu W, Li Y, Li G, Xu JR (2017) Expression of HopAI interferes with MAP kinase signalling in Magnaporthe oryzae. Environ Microbiol 19: 4190–4204. doi: 10.1111/1462-2920.13884 [DOI] [PubMed] [Google Scholar]
- 12.Bashi ZD, Gyawali S, Bekkaoui D, Coutu C, Lee L, et al. (2016) The Sclerotinia sclerotiorum Slt2 mitogen-activated protein kinase ortholog, SMK3, is required for infection initiation but not lesion expansion. Can J Microbiol 62: 836–850. doi: 10.1139/cjm-2016-0091 [DOI] [PubMed] [Google Scholar]
- 13.Kim JM, Lee JG, Yun SH, So KK, Ko YH, et al. (2016) A mutant of the Bck1 homolog from Cryphonectria parasitica resulted in sectorization with an impaired pathogenicity. Mol Plant Microbe In 29: 268–276. [DOI] [PubMed] [Google Scholar]
- 14.Zheng DW, Wang Y, Han Y, Xu JR, Wang CF (2016) UvHOG1 is important for hyphal growth and stress responses in the rice false smut fungus Ustilaginoidea virens. Sci Rep 6: 24824 doi: 10.1038/srep24824 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Leng YQ, Zhong SB (2015) The role of mitogen-activated protein (MAP) kinase signaling components in the fungal development, stress response and virulence of the fungal cereal pathogen Bipolaris sorokiniana. PLoS ONE 10: e0128291 doi: 10.1371/journal.pone.0128291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pareek M, Rajam MV (2017) RNAi-mediated silencing of MAP kinase signalling genes (Fmk1, Hog1, and Pbs2) in Fusarium oxysporum reduces pathogenesis on tomato plants. Fungal Biol 121: 775–784. doi: 10.1016/j.funbio.2017.05.005 [DOI] [PubMed] [Google Scholar]
- 17.Van Nguyen T, Kroger C, Bonnighausen J, Schafer W, Bormann J (2013) The ATF/CREB transcription factor Atf1 is essential for full virulence, deoxynivalenol production, and stress tolerance in the cereal pathogen Fusarium graminearum. Mol Plant Microbe In 26: 1378–1394. [DOI] [PubMed] [Google Scholar]
- 18.Wilson RA, Talbot NJ (2009) Under pressure: investigating the biology of plant infection by Magnaporthe oryzae. Nat Rev Microbiol 7: 185–195. doi: 10.1038/nrmicro2032 [DOI] [PubMed] [Google Scholar]
- 19.Kou YJ, Tan YH, Ramanujam R, Naqvi NI (2017) Structure-function analyses of the Pth11 receptor reveal an important role for CFEM motif and redox regulation in rice blast. New Phytol 214: 330–342. doi: 10.1111/nph.14347 [DOI] [PubMed] [Google Scholar]
- 20.Xu XH, Li GP, Li L, Su ZZ, Chen C (2017) Genome-wide comparative analysis of putative Pth11-related G protein-coupled receptors in fungi belonging to Pezizomycotina. BMC Microbiol 17: 166 doi: 10.1186/s12866-017-1076-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Turra D, El Ghalid M, Rossi F, Di Pietro A (2015) Fungal pathogen uses sex pheromone receptor for chemotropic sensing of host plant signals. Nature 527: 521-+. doi: 10.1038/nature15516 [DOI] [PubMed] [Google Scholar]
- 22.Wang GH, Li GT, Zhang SJ, Jiang C, Qin J, et al. (2015) Activation of the signalling mucin MoMsb2 and its functional relationship with Cbp1 in Magnaporthe oryzae. Environ Microbiol 17: 2969–2981. doi: 10.1111/1462-2920.12847 [DOI] [PubMed] [Google Scholar]
- 23.Leroch M, Mueller N, Hinsenkamp I, Hahn M (2015) The signalling mucin Msb2 regulates surface sensing and host penetration via BMP1 MAP kinase signalling in Botrytis cinerea. Mol Plant Pathol 16: 787–798. doi: 10.1111/mpp.12234 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Igbaria A, Lev S, Rose MS, Lee BN, Hadar R, et al. (2008) Distinct and combined roles of the MAP kinases of Cochliobolus heterostrophus in virulence and stress responses. Mol Plant Microbe In 21: 769–780. [DOI] [PubMed] [Google Scholar]
- 25.Segorbe D, Di Pietro A, Perez-Nadales E, Turra D (2017) Three Fusarium oxysporum mitogen-activated protein kinases (MAPKs) have distinct and complementary roles in stress adaptation and cross-kingdom pathogenicity. Mol Plant Pathol 18: 912–924. doi: 10.1111/mpp.12446 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li GT, Zhang X, Tian H, Choi YE, Tao WA, et al. (2017) MST50 is involved in multiple MAP kinase signaling pathways in Magnaporthe oryzae. Environ Microbiol 19: 1959–1974. doi: 10.1111/1462-2920.13710 [DOI] [PubMed] [Google Scholar]
- 27.Guo L, Breakspear A, Zhao G, Gao L, Kistler HC, et al. (2016) Conservation and divergence of the cyclic adenosine monophosphate-protein kinase A (cAMP-PKA) pathway in two plant-pathogenic fungi: Fusarium graminearum and F. verticillioides. Mol Plant Pathol 17: 196–209. doi: 10.1111/mpp.12272 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Garcia R, Bravo E, Diez-Muniz S, Nombela C, Rodriguez-Pena JM, et al. (2017) A novel connection between the Cell Wall Integrity and the PKA pathways regulates cell wall stress response in yeast. Sci Rep 7: 5703 doi: 10.1038/s41598-017-06001-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Li Y, Zhang X, Hu S, Liu H, Xu JR (2017) PKA activity is essential for relieving the suppression of hyphal growth and appressorium formation by MoSfl1 in Magnaporthe oryzae. PLoS Genet 13: e1006954 doi: 10.1371/journal.pgen.1006954 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
(DOCX)