Abstract
Previous work has established a role for CD40-mediated signals in eliciting helper-dependent CD8+ T cell responses. Here we investigated the effects of in vivo CD40 stimulation on the survival and function of tumor-specific CD8+ T cells in a mouse melanoma model system. We found that agonistic anti-CD40 antibody treatment alone of tumor-bearing mice accelerated the deletion of tumor-antigen-specific T cells. However, long-term survival and function of tumor-antigen-specific T cells could be achieved when viral immunization with tumor antigen and anti-CD40 treatment were combined. This rescue of CD8+ T cells could not be easily replicated by inflammatory or antigen-specific stimuli alone, demonstrating the specificity of signals that regulate the deletion or survival of tumor-specific T cells. These results demonstrate that opposing effects can be elicited by CD40 stimulation in vivo and suggest the need for caution in using this treatment for cancer patients.
Keywords: melanoma, MHC tetramer
T cells play an important role in immunity to tumors, particularly melanomas. T cells specific for melanoma antigens can often be isolated from the tumor site, lymphoid tissue, or the blood of melanoma patients (1–3). These T cells have various ranges of functional activity, but in some cases their presence correlates with a better clinical prognosis for the patient (4–7). For example, melanoma patients who develop vitiligo, a T cell-mediated autoimmune destruction of normal melanocytes, demonstrate a greater ability to control their tumor growth (8–11).
Despite the importance of T cells in tumor immunity, we still know very little about the induction of tumor-specific T cells and the signals that are necessary to sustain their activation and effector function. Melanoma-specific T cells are frequently isolated from patients but are often functionally unresponsive (12–16). In humans, where we are unable to follow the progression of the disease from its earliest stages of development, it is difficult to determine either the causes of this functional inactivation or the intervention(s) necessary to prevent it.
The activation state of the antigen-presenting cell (APC) has been shown to have a dramatic effect on CD8+ T cell responses. Three groups simultaneously demonstrated that a helper-dependent CD8+ T cell response could be converted to a helper-independent response simply by activating the APC with antibodies against CD40 (17–19). Because suboptimal tumor antigen presentation is speculated to be a problem in tumor-bearing hosts, several groups have used CD40 stimulation in tumor model systems in an attempt to augment failed or weak CD8+ T cell responses (20–22). While CD40 stimulation has shown some promise, the precise conditions under which this approach is most effective have not been fully defined. Indeed, a recent report suggests that agonistic anti-CD40 antibodies can be immunosuppressive (23), exemplifying the need for careful assessment of this therapeutic intervention, particularly in the case of a tumor-bearing host where the immune response against the tumor may be minimal to begin with.
We have developed a melanoma model system that allows the tracking of endogenously generated tumor-specific T cells in vivo during the course of tumor growth. Mice were challenged with the murine melanoma tumor line B16 transfected with the gene for ovalbumin (B16ova). The CD8+ T cell responses to the dominant ovalbumin epitope SIINFEKL (ova8) were followed with the use of a class I Kb/ova8 tetrameric staining reagent. In these studies we demonstrated that tumor-specific T cells expanded and migrated to tumor tissues. We further demonstrated that agonistic antibodies against CD40 actually enhanced the deletion of antigen-specific CD8+ T cells, and this deletion could be prevented by vaccination with tumor antigen.
Materials and Methods
Tumor Cell Lines and Mouse Injections.
The B16-ovalbumin (B16ova) and B16-neomycin (B16neo) cell lines were kindly provided by Richard Duke (University of Colorado Health Sciences Center, Denver). These cell lines were made by lipofection of the B16-F10 cell line with constructs encoding the full-length ovalbumin gene with the neomycin-resistance selection gene (B16ova) or with the neomycin-resistance selection gene alone (B16neo) under the control of the cytomegalovirus long terminal repeat promoter. These tumor cells were cultured in complete media containing 750 μg/ml G418. Before injection into mice, the cells were trypsinized for 5 min at 37°C, washed with complete media and balanced salt solution (Earle's BSS), and resuspended in BSS at 1 × 106 cells per milliliter. Six- to 12-week-old C57BL/6J (B6) female mice from the Jackson Laboratory were anesthetized with Avertin, their rear flanks were shaved, and they were injected with 1 × 105 tumor cells intradermally.
Virus and DNA Vaccination.
Vaccinia virus (VV) (kindly provided by Tom Mitchell, University of Louisville, Louisville, KY) was propagated in and titrated by plaque assay on cultured 143B osteosarcoma cells as described (24). Mice were challenged i.v. with 2–4 × 106 plaque-forming units of VV encoding ovalbumin (VVova) (25) or influenza virus nucleoprotein (VV-NP) (25).
The ovalbumin gene was subcloned into an expression vector containing the tissue plasminogen activator leader sequence for secretion, along with a cytomegalovirus promoter and the bovine growth hormone polyadenylation sequence (a kind gift from Keith Rushlow, Heska Corp., Fort Collins, CO). Plasmid DNA was prepared by a modified alkaline lysis procedure followed by glycol precipitation as described (26). Mice were injected with 50 μg of total plasmid DNA in a total volume of 200 μl. Equal amounts of DNA were injected bilaterally into the quadriceps muscles of mice anesthetized with Avertin.
Monoclonal Antibodies.
The antibodies used in these studies were 1C10 (anti-CD40), XMG1.2 (anti-IFNγ), GK1.5 (anti-CD4), and 20LC-11.1 (anti-DR1 used as a control rat antibody). The respective hybridomas were grown in serum-free conditions, and each antibody was purified on a protein G column. After elution in a glycine⋅HCl buffer and neutralization with a Tris buffer, the purified antibodies were dialyzed into PBS and injected i.p. into tumor-bearing hosts. Two hundred micrograms of anti-CD40 antibody (27) was injected 7–10 days after initial tumor challenge and, in the cases where noted, every 7 days thereafter. In vivo depletion of CD4+ T cells was performed by the weekly injection of 500 μg of anti-CD4 (28). In vivo blocking of IFNγ was performed by the weekly injection of 2–3 mg XMG1.2. This treatment was shown to be effective in blocking the effect of IFNγ in vivo in previous studies (29, 30).
Cell Preparation, Tetramers, and Cell Staining.
After sacrifice of the animals at various times, the draining nodes (periaortic, inguinal, axillary, and brachial), spleen, and tumor tissues were removed and homogenized into single-cell suspensions. In the case of spleen and tumor, the red blood cells were lysed by brief treatment with ammonium chloride buffer followed by washing with BSS. All cells were finally suspended in complete SMEM, and total cell numbers were determined with a Coulter Counter.
Anti-CD8-APC, CD44-FITC, B220-Cychrome, IAb-biotin, and streptavidin-Cychrome were all purchased from PharMingen. Kb covalently linked by the C terminus to a peptide tag which is a substrate for BirA was produced in insect cells, biotinylated, and bound to phycoerythrin-streptavidin as described (31, 32). A 5–10 M excess of SIINFEKL peptide (ova8, ovalbumin residues 257–264) or SIYRYYGL peptide (which, in the context of Kb class I, activates T cells bearing the 2C T cell antigen receptor) was added directly to newly constructed Kb–phycoerythrin-streptavidin tetramer for at least 30 min at 4°C. Tetramer staining was performed as described (32). The Kb/SIYRYYGL tetramer (i.e., same MHC, wrong peptide) and/or Kb/ova8 staining of cells from mice bearing a non-ovalbumin-expressing tumor (B16neo) was used to establish the background tetramer staining of experimental samples. Each batch of Kb/ova8 tetramer was tested and normalized for binding to naive OT1 transgenic T cells, which are specific for Kb/ova8, before use in experiments (32).
Cells were stained with tetramer for 1 h and then treated with GolgiStop (brefeldin A) in the presence of ovalbumin peptide for 4–6 h in complete medium at 37°C. The cells were then stained for CD8 and IFNγ according to the Becton Dickinson protocol for intracellular cytokine staining.
Four-color fluorescence-activated cell sorter data were collected on a Becton Dickinson FACSCalibur flow cytometer and analyzed with cellquest software.
In Vitro Restimulation and 51Cr Release Lytic Assays.
Single-cell suspensions were made from the spleens of experimental mice, placed at 3–4 × 106 cells per milliliter in 12-well plates, and incubated in the presence of peptide and 10% Rat Con-A supernatant in complete minimal essential medium (MEM) for 5 days. The cells were then washed and plated at increasing cell densities in 96-well plates. 51Cr lytic assays were performed as described (33) with the use of EL4 tumor cells (H-2b) as targets ± the ova8 peptide.
Results
T Cell Responses in Tumor-Bearing Hosts Are Detectable but Transient and Nonfunctional.
Many human tumors express mutated gene products or gene products that are normally expressed only within a developing embryo (34–38). Because of a lack of central tolerance of these so-called tumor neo-antigens, epitopes from these gene products can serve as tumor rejection antigens. In an effort to approximate this kind of tumor rejection antigen, we used the B16 melanoma cell line transfected with the gene for ovalbumin (B16ova) as a representative tumor neo-antigen.
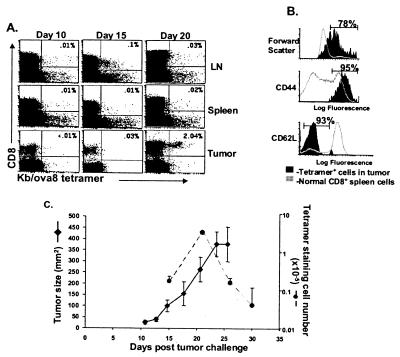
Mice were injected with B16ova intradermally, and at various times thereafter spleen, lymph nodes, and tumor tissue were removed, and the cells were stained with the Kb/ova8 tetramer to identify all T cells directed against the dominant epitope of the ovalbumin tumor neo-antigen. Despite the development of palpable tumors by day 7–10, tumor-specific T cells were not detectable until about day 15 (Fig. 1A). This population of CD8+ T cells was usually very small and was seen in either the lymph nodes or spleen. Tumor-specific T cells were more readily detectable within the tumor itself, but usually not until after day 20. Forty to eighty percent of the ova8-specific T cells in the tumor were blasts (high forward scatter) at this time point, suggesting that clonal expansion occurred within the tumor tissue (Fig. 1B). These cells demonstrated an activated phenotype of high CD44 and low L-selectin expression (Fig. 1B). Despite this activated phenotype, little if any ova8-specific lytic activity could be detected either directly ex vivo (data not shown) or after in vitro restimulation (see Fig. 4A).
Figure 1.
ova8-specific T cells are detectable by tetramer staining in B16ova-bearing mice. B6 mice were injected intradermally with 1 × 105 B16ova cells. At the times indicated in A, mice were killed and the cells were isolated from lymph nodes, spleen, and tumor. (A and B) The cells were stained with anti-CD8, anti-IAb, and either Kb/ova8-tetramer or control Kb-tetramer. Plots shown were gated on all live, IAb− events, and the percentages given in the upper right quadrant are of the total CD8+ T cells. The settings for determining Kb/ova8-tetramer staining were obtained by staining cells with control tetramer (not shown). Results are representative of five separate experiments. (B) All histograms are gated on Kb/ova8-tetramer+ events from tumor tissue 20 days after initial tumor challenge, stained as described in A. (C) Data from two separate experiments were plotted for tumor size (♦, left axis) vs. number of tetramer-staining CD8+ T cells (●, right axis). Tumor size was measured in two dimensions with a caliper; error bars represent the standard error between six individual mice. The numbers of tetramer-staining T cells were calculated from the percentage of tetramer-staining cells, as shown in the upper right quadrant in A and the total cell number as determined with a Coulter Counter.
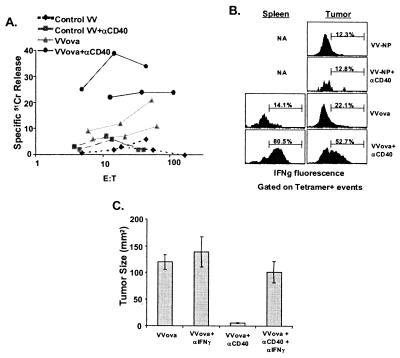
Figure 4.
VVova and anti-CD40 treatment results in enhanced lytic activity and IFNγ production from tumor-specific CD8+ T cells. Mice were challenged with tumor and treated with VVova and/or anti-CD40 as in Fig. 3. (A) Twenty-five days after initial tumor challenge, spleens were removed from the different treated groups and restimulated with 5 μg/ml ovalbumin peptide in vitro for 5 days. The cells were washed, and 51Cr release lytic assays were performed as described in Materials and Methods. Cells from two mice per treatment group are shown and are representative of two separate experiments performed. (B) Twenty-five days after initial tumor challenge, spleen and tumor tissue were removed from the different groups, and the cells were stained and analyzed for the expression of intracellular IFNγ in tetramer-staining cells as described for Fig. 2. The results are representative of four independent experiments. (C) Blocking IFNγ in VVova + anti-CD40-treated tumor-bearing mice eliminates the inhibition of tumor growth normally seen with this treatment. Tumor-bearing mice were treated with anti-CD40 and/or VVova as in Fig. 3. Seven days after VVova immunization, mice were injected with 3 mg of XMG1.2 (anti-IFNγ) or control anti-DR4 i.p. every 7 days. Twenty-five days after initial tumor challenge, tumors from two to three mice per group were measured by caliper. The error bars indicate the standard error of tumor size within a given treatment group. The data are representative of three experiments performed.
The number of ova8-specific T cells within the tumor declined over time as the tumor increased in size until the animal was killed, suggesting that the process of deletion of this T cell population was limited only by the life span of the host (Fig. 1C). The growing tumor still expressed ovalbumin, based on the ability of naive OT1 transgenic T cells (specific for the Kb/ova8 epitope) to proliferate within the tumor when transferred into tumor-bearing hosts at late time points (data not shown), which suggested that the loss of Kb/ova8-specific T cells at late time points was not due to the outgrowth of a tumor-antigen-loss variant. In addition, the decline in T cells within the tumor was not caused by their migration to other sites such as lung and liver; although tumor-specific T cells could be detected in these peripheral tissues (data not shown) they were only detectable at such sites at times when they were also detectable in the tumor. Therefore the data suggest that initial T cell activation occurred at early time points within the lymphoid tissue (Fig. 1A) followed by a trafficking to and clonal expansion of the cells within the peripheral tumor compartment (Fig. 1B). Despite this clonal expansion, the tumor-specific T cells were nonfunctional, and their decline in number indicated their progressive deletion (Fig. 1C).
This model system is remarkably consistent with what little is known about tumor-specific T cell responses from human patient samples. Similar to our model system, the melanoma-specific T cells detectable by tetramer staining in blood or tissue samples from patients often show an activated phenotype but no functional response when assessed for lytic activity or cytokine production (12–16). In addition, whereas we originally designed these experiments to assess the activity of T cells directed against a representative tumor neo-antigen (ovalbumin), we were also recently able to confirm these findings for T cells directed against the endogenous B16 tumor antigen TRP2. The kinetics of the response of TRP2-specific T cells, as assessed by tetramer staining in B16 tumor-bearing mice, is remarkably similar to the ovalbumin-specific response in terms of expansion and deletion (data not shown) and further supports the physiological relevance of this model system (R.M.K. and S.D., unpublished results).
Agonistic Anti-CD40 Antibody Treatment Accelerates the Deletion of Tumor-Specific CD8+ T Cells.
CD40 is a molecule expressed on APCs which, when stimulated with agonistic antibodies, can dramatically affect CD8+ T cell responses (17–19). A number of groups have used anti-CD40 treatment in studies of tumor growth and rejection and have reported significant benefits from this treatment, particularly with respect to inhibiting the long-term deletion of tumor-antigen-specific T cells (21, 22). In those studies, anti-CD40 treatment was accompanied by some form of tumor-antigen immunization in the presence of adjuvant (e.g., peptide in complete Freund's adjuvant or virus), to enhance tumor-antigen expression and presentation to a level that was detectable by the specific T cells.
Using MHC tetrameric reagents, we were able to detect an ovalbumin T cell response in tumor-bearing mice, suggesting that ova8 antigen presentation was occurring to a significant extent in mice challenged with tumor alone. It therefore seemed possible that tumor-antigen immunization was unnecessary and that anti-CD40 treatment alone of tumor-bearing mice might prevent the deletion and/or rescue the function of the tumor-specific T cell response. If true, this treatment with anti-CD40 alone might be of significant clinical benefit, alleviating the need to determine a priori the primary tumor-antigen candidates in any given patient and essentially allowing the host to focus its responses on whatever tumor antigens it is already presenting. We therefore treated mice with anti-CD40 alone 7 days after initial tumor challenge and assessed the T cell response as before.
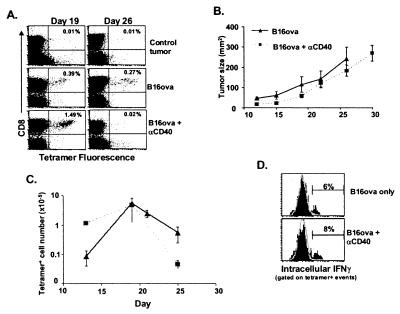
Shortly after the anti-CD40 treatment, we observed an increase in the numbers of Kb/ova8-specific T cells within the tumor by comparison with controls (Fig. 2A), indicating that this treatment did have an effect on the developing T cell response. However, in the long term, the anti-CD40 treatment actually accelerated deletion of the Kb/ova8-specific T cells (Fig. 2 A and C). Moreover, even the T cells present during the early expansion immediately after anti-CD40 treatment did not demonstrate significant IFNγ production (Fig. 2D) or lytic activity (data not shown and see Fig. 4A), a result that was consistent with the continued growth of the tumor (Fig. 2B) and further indicated that anti-CD40 stimulation alone was deficient for signals necessary for a functional T cell response.
Figure 2.
Anti-CD40 treatment accelerates the deletion of tumor-specific T cells. Mice were challenged with 1 × 105 B16ova cells intradermally. Seven days later, when the tumor was palpable, mice were injected with 200 μg of either anti-CD40 or control antibody i.p. (A) At the times indicated, the tumor was removed, stained, and analyzed as described in the legend of Fig. 1. (B and C) Tumor-bearing mice were injected with 200 μg of anti-CD40 i.p. on day 7 after initial tumor challenge and every 7 days thereafter (i.e., days 14 and 21). The data were analyzed as described in the legend of Fig. 1C. (D) Fifteen days after initial tumor challenge and 8 days after anti-CD40 treatment, cells from the tumors of control and anti-CD40-treated mice were stained as described in Materials and Methods for intracellular IFNγ. The data shown (solid histograms) have been gated on all class II−, CD8+, tetramer+ events. The background (open histogram) is from gating on all non-tetramer-staining CD8+ T cells in the tumor. These results are representative of three separate experiments.
T Cell Deletion Is Prevented by Coimmunization with Anti-CD40 and Ovalbumin-Expressing VV.
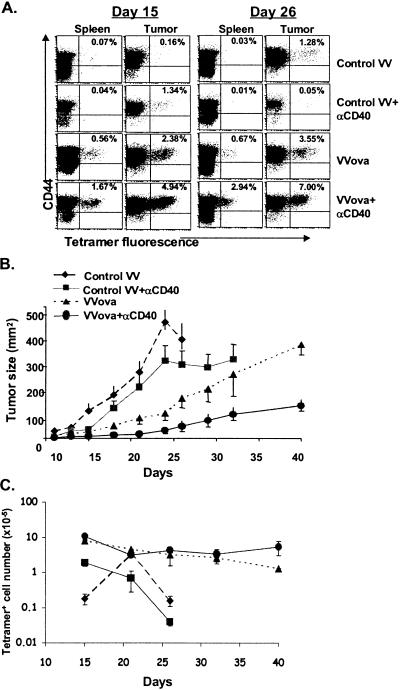
As mentioned, other groups have demonstrated that anti-CD40 treatment of tumor-bearing hosts significantly augments a tumor-specific T cell response when the host is coimmunized in some fashion with tumor antigen (21, 22). By using VVova, we determined whether tumor-antigen immunization in our model system would similarly prevent the deletion of tumor-specific T cells. B16ova tumor-bearing mice were injected on day 7 with VVova with or without the addition of anti-CD40 antibody treatment. Control VV-immunized mice showed little difference from nonimmunized (control) mice with respect to tumor growth and expansion of Kb/ova8-specific T cells (Fig. 3A). Interestingly, immunization of mice with control virus and anti-CD40 showed little difference from mice treated with anti-CD40 only, demonstrating that the inflammatory signals induced by the viral infection were not sufficient to prevent the anti-CD40-enhanced deletion of ova8-specific T cells. Immunization of a tumor-bearing host with VVova alone resulted in a significant expansion of ova8-specific T cells in both the spleen and tumor (Fig. 3 A and C). Initially the expansion of T cells in VVova-immunized hosts correlated with a dramatic reduction in tumor size compared with control virus-immunized hosts (Fig. 3B). Despite this initial expansion of T cells, however, the ova8-specific T cell number gradually declined (Fig. 3 A and C; note the logarithmic scale), and the tumor resumed growth (Fig. 3B) until sacrifice of the host was necessary.
Figure 3.
Virus immunization with tumor antigen inhibits anti-CD40-mediated deletion of tumor-specific T cells and elicits effective tumor growth control. B6 mice were injected with 1 × 105 B16ova cells intradermally. Seven days later mice were injected with the indicated combinations of anti-CD40 (i.p.), the control virus VV-NP (i.v.), and VVova (i.v.). (A) Cells were isolated from spleen and tumor on the days given and stained and analyzed as in Fig. 1. (B) Tumor sizes were measured by caliper on four to six mice per group. The error bars represent the calculated standard error between at least four mice per group. (C) The total number of tetramer-staining cells was calculated as in Fig. 1. Error bars represent the standard error between four and six mice per time point per group. The results are representative of three separate experiments.
In contrast, the combination of anti-CD40 treatment and VVova immunization resulted in an increase in the percentage of tumor-antigen-specific T cells over VVova infection alone (Fig. 3A). Moreover, no discernible decrease in ova8-specific T cell number occurred over 5 weeks after immunization (Fig. 3C). Anti-CD40 treatment of VVova-immunized mice also resulted in long-term inhibition of tumor growth (Fig. 2B). Consistent with data from previous reports (21, 22), this treatment induced significant lytic activity (Fig. 4A) and IFNγ production by the tumor-specific T cells (Fig. 4B). The IFNγ production appeared to be the primary cause of the tumor growth inhibition observed, as demonstrated by in vivo anti-IFNγ antibody blocking experiments (Fig. 4C). Thus our model system is consistent with other experimental model systems (21, 22) as well as with observed clinical data on the progression of tumor-specific T cell responses in patients (12–16).
Antigenic Immunization or Inflammation Alone Is Not Sufficient to Rescue T Cell Function.
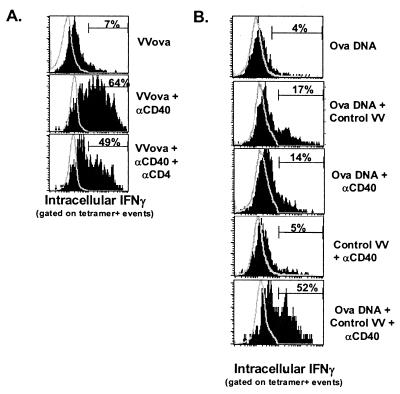
Because it was apparent that anti-CD40 treatment during challenge with VVova was capable of rescuing both T cell survival and function (Fig. 3), we attempted to determine what cellular, inflammatory, and antigenic factors played a role in this rescue. CD4+ T cells have been shown to be necessary for enhancing the survival and function of CD8+ T cells and/or the host, particularly after vaccination in tumor model systems (39–41). However, CD4 cells did not appear to play a significant role in mediating the rescue seen by VVova and anti-CD40 treatment, as CD4 depletion of these tumor-bearing hosts demonstrated T cell function (Fig. 5A) and tumor growth control (data not shown) comparable to those of the non-CD4-depleted controls. It therefore appeared that some combination of antigenic and inflammatory stimuli may have acted directly upon the CD8+ T cells. Neither a control virus infection alone, to provide non-antigen-specific inflammatory conditions as in Fig. 3, nor an increased antigen load alone, in the form of ovalbumin DNA immunization, was able to replicate the ability of VVova and anti-CD40 treatment to rescue T cell function (Fig. 5B). Only when tumor-bearing mice were treated with a combination of ovalbumin DNA immunization, control virus, and anti-CD40, a situation that essentially recapitulates VVova and anti-CD40 treatment, was T cell function rescued (Fig. 5B). Therefore, the accelerated tolerance and deletion brought on by anti-CD40 treatment alone appeared to be dominant in most cases, whereas preventing this deletion was accomplished solely by the expression of specific antigen in the context of a viral infection.
Figure 5.
Rescue of tumor-specific CD8+ T cell function is CD4+ T cell-independent and requires both antigenic immunization and virally mediated inflammation. (A) Mice were injected with 500 μg of anti-CD4 antibody (GK1.5) i.p. 1 day before B16ova tumor challenge and every 7 days thereafter. Mice were then immunized as in Fig. 3 with VVova and anti-CD40, and the IFNγ production of ova8-specific T cells in the tumor was assessed 25 days after initial tumor challenge as described in Fig. 2. Staining spleen cells with anti-CD4 (RM4-4) indicated that CD4 cells had been depleted (less than 0.2% staining; data not shown). Open histograms are of total CD8+ T cells from within the tumor, and closed histograms are of tetramer+, CD8+ cells. (B) Seven days after B16ova challenge, mice were treated with combinations of control VV, intramuscular ovalbumin DNA immunization, and/or anti-CD40. Eighteen days later, tumor tissue was removed, and the IFNγ production of tetramer-staining CD8+ T cells was assessed as in Fig. 2. The results are representative of two independent experiments.
Discussion
It is well known that the activation state of the APC significantly affects the success of a T cell response. Our data suggest, however, that the success of a T cell response may depend upon more than simple activation of antigen-bearing APC. Stimulation of APC via CD40 has been shown to have a positive effect upon the activation of APC and, as a result, on the development and effector function of CD8+ T cells in a variety of model systems (17–19). These effects on the generation of CD8+ T cells have made anti-CD40 treatment seem particularly useful for tumor immunotherapy. Indeed, we (Fig. 2) and others (21, 22) have found that in the appropriate immunization context, agonistic CD40 antibody treatment can induce tumor regression and activation of tumor-specific T cells in tumor-bearing hosts. However, CD40 stimulation alone in B16ova-bearing mice unexpectedly resulted in a more rapid deletion of the tumor-specific T cells. This finding demonstrated that, rather than immunity, anti-CD40 stimulation in this context enhanced systemic tolerance of a tumor-specific antigen.
Whereas we have demonstrated this effect of CD40 treatment in a tumor model system, another group has documented the ability of an agonistic anti-CD40 antibody to mediate T cell deletion in an autoimmunity model system (23), suggesting that this effect of agonistic CD40-mediated stimulation is not limited to our experimental protocol. It is worth noting that we used a rather late time point of anti-CD40 treatment compared with that of other reports (21, 22), so possibly the anti-CD40 was administered too late to prevent the death of the tumor-reactive cells. However, the anti-CD40 in our protocol did initially increase the numbers of tumor-specific T cells for 7–10 days after treatment, so this seems unlikely.
It is not clear what signals are responsible, in the cascade of signals that a virus such as vaccinia induces, for synergizing with anti-CD40 to supply the T cells with the proper stimuli for long-term survival and IFNγ production. It is likely that proinflammatory cytokines such as IL-12 (42–44), tumor necrosis factor-γ (45–48), or the recently described IL-23 (49) may be involved, and they may not be elicited by anti-CD40 treatment alone. This hypothesis is supported by a recent report demonstrating that whereas anti-CD40 treatment of dendritic cells in vitro elicits IL-12 production, anti-CD40 treatment alone in vivo does not (50). However, it is unlikely that the expression of a single cytokine such as IL-12 will make up the entire difference between a functional or nonfunctional T cell response. For example, preliminary experiments using intramuscular IL-12 DNA injections to provide long-term IL-12 expression in vivo in tumor-bearing hosts has demonstrated that although this treatment does promote the survival and/or expansion of tumor-specific T cells, it has little effect on promoting their ability to produce IFNγ (R.K. and S.D., unpublished results). It is likely that an integration of the type, quantity, and timing of cytokine(s) expression during antigen presentation is necessary for inducing a long-lived, functional T cell response.
In conclusion, our results demonstrate that the activation of APC through CD40 alone is not sufficient to elicit a long-lived T cell response in a tumor-bearing host unless it is coupled to a tumor antigen/inflammatory/adjuvant immunization. Clinical trials evaluating anti-CD40 treatment have been proposed for a number of different cancer types. Our data suggest that caution must be used in treating these patients with anti-CD40 and that continuing research must focus on understanding what other stimuli act in concert with this APC stimulatory pathway to promote either tolerance or immunity.
Acknowledgments
We thank Fran Crawford, Chris Wagner, and Tracy DeBlieck for technical assistance and Brian Schaefer for assistance in reviewing the manuscript. This work was supported by a Postdoctoral Fellowship Grant from the Cancer Research Institute (R.M.K.) and U.S. Public Health Service Grants AI-17134, AI-18785, AI-22295, and CA86224-01.
Abbreviations
- APC
antigen-presenting cell
- B16ova
murine melanoma tumor line B16 transfected with the gene for ovalbumin
- B16neo
B16-neomycin
- VV
vaccinia virus
- VVova
VV encoding ovalbumin
- VV-NP
VV encoding influenza virus nucleoprotein
References
- 1.thor Straten P, Becker J C, Guldberg P, Zeuthen J. Cancer Immunol Immunother. 1999;48:386–395. doi: 10.1007/s002620050591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.thor Straten P, Guldberg P, Gronbaek K, Hansen M R, Kirkin A F, Seremet T, Zeuthen J, Becker J C. J Immunol. 1999;163:443–447. [PubMed] [Google Scholar]
- 3.Clark W H. Br J Cancer. 1991;64:631–644. doi: 10.1038/bjc.1991.375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mihm M C, Jr, Clemente C G, Cascinelli N. Lab Invest. 1996;74:43–47. [PubMed] [Google Scholar]
- 5.Bartoli C, Bono A, Clemente C, Del Prato I D, Zurrida S, Cascinelli N. Cancer (Philadelphia) 1996;77:888–892. doi: 10.1002/(sici)1097-0142(19960301)77:5<888::aid-cncr12>3.0.co;2-#. [DOI] [PubMed] [Google Scholar]
- 6.Clemente C G, Mihm M C, Jr, Bufalino R, Zurrida S, Collini P, Cascinelli N. Cancer (Philadelphia) 1996;77:1303–1310. doi: 10.1002/(SICI)1097-0142(19960401)77:7<1303::AID-CNCR12>3.0.CO;2-5. [DOI] [PubMed] [Google Scholar]
- 7.Halliday G M, Patel A, Hunt M J, Tefany F J, Barnetson R S. World J Surg. 1995;19:352–358. doi: 10.1007/BF00299157. [DOI] [PubMed] [Google Scholar]
- 8.Becker J C, Guldberg P, Zeuthen J, Brocker E B, Straten P T. J Invest Dermatol. 1999;113:1033–1038. doi: 10.1046/j.1523-1747.1999.00805.x. [DOI] [PubMed] [Google Scholar]
- 9.Ho R C. Cancer (Philadelphia) 1995;75:735–741. doi: 10.1002/1097-0142(19950115)75:2+<735::aid-cncr2820751418>3.0.co;2-q. [DOI] [PubMed] [Google Scholar]
- 10.Kawakami Y, Rosenberg S A. Immunol Res. 1996;15:179–190. doi: 10.1007/BF02918248. [DOI] [PubMed] [Google Scholar]
- 11.Yee C, Thompson J A, Roche P, Byrd D R, Lee P P, Piepkorn M, Kenyon K, Davis M M, Riddell S R, Greenberg P D. J Exp Med. 2000;192:1637–1644. doi: 10.1084/jem.192.11.1637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Romero P, Dunbar P R, Valmori D, Pittet M, Ogg G S, Rimoldi D, Chen J L, Lienard D, Cerottini J C, Cerundolo V. J Exp Med. 1998;188:1641–1650. doi: 10.1084/jem.188.9.1641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Anichini A, Molla A, Mortarini R, Tragni G, Bersani I, Di Nicola M, Gianni A M, Pilotti S, Dunbar R, Cerundolo V, et al. J Exp Med. 1999;190:651–667. doi: 10.1084/jem.190.5.651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lee P P, Yee C, Savage P A, Fong L, Brockstedt D, Weber J S, Johnson D, Swetter S, Thompson J, Greenberg P D, et al. Nat Med. 1999;5:677–685. doi: 10.1038/9525. [DOI] [PubMed] [Google Scholar]
- 15.Pittet M J, Valmori D, Dunbar P R, Speiser D E, Lienard D, Lejeune F, Fleischhauer K, Cerundolo V, Cerottini J C, Romero P. J Exp Med. 1999;190:705–715. doi: 10.1084/jem.190.5.705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yee C, Savage P A, Lee P P, Davis M M, Greenberg P D. J Immunol. 1999;162:2227–2234. [PubMed] [Google Scholar]
- 17.Schoenberger S P, Toes R E, van der Voort E I, Offringa R, Melief C J. Nature (London) 1998;393:480–483. doi: 10.1038/31002. [DOI] [PubMed] [Google Scholar]
- 18.Ridge J P, Di Rosa F, Matzinger P. Nature (London) 1998;393:474–478. doi: 10.1038/30989. [DOI] [PubMed] [Google Scholar]
- 19.Bennett S R, Carbone F R, Karamalis F, Flavell R A, Miller J F, Heath W R. Nature (London) 1998;393:478–480. doi: 10.1038/30996. [DOI] [PubMed] [Google Scholar]
- 20.Schultze J L, Gribben J G, Nadler L M. Curr Opin Oncol. 1998;10:542–547. doi: 10.1097/00001622-199811000-00011. [DOI] [PubMed] [Google Scholar]
- 21.Diehl L, den Boer A T, Schoenberger S P, van der Voort E I, Schumacher T N, Melief C J, Offringa R, Toes R E. Nat Med. 1999;5:774–779. doi: 10.1038/10495. [DOI] [PubMed] [Google Scholar]
- 22.Sotomayor E M, Borrello I, Tubb E, Rattis F M, Bien H, Lu Z, Fein S, Schoenberger S, Levitsky H I. Nat Med. 1999;5:780–787. doi: 10.1038/10503. [DOI] [PubMed] [Google Scholar]
- 23.Mauri C, Mars L T, Londei M. Nat Med. 2000;6:673–679. doi: 10.1038/76251. [DOI] [PubMed] [Google Scholar]
- 24.Mitchell T, Kappler J, Marrack P. J Immunol. 1999;162:4527–4535. [PubMed] [Google Scholar]
- 25.Restifo N P, Bacik I, Irvine K R, Yewdell J W, McCabe B J, Anderson R W, Eisenlohr L C, Rosenberg S A, Bennink J R. J Immunol. 1995;154:4414–4422. [PMC free article] [PubMed] [Google Scholar]
- 26.Liu Y, Liggitt D, Zhong W, Tu G, Gaensler K, Debs R. J Biol Chem. 1995;270:24864–24870. doi: 10.1074/jbc.270.42.24864. [DOI] [PubMed] [Google Scholar]
- 27.Heath A W, Wu W W, Howard M C. Eur J Immunol. 1994;24:1828–1834. doi: 10.1002/eji.1830240816. [DOI] [PubMed] [Google Scholar]
- 28.Mielke M E, Ehlers S, Hahn H. Infect Immun. 1988;56:1920–1925. doi: 10.1128/iai.56.8.1920-1925.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Smith D, Hansch H, Bancroft G, Ehlers S. Immunology. 1997;92:413–421. doi: 10.1046/j.1365-2567.1997.00384.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Appelberg R, Castro A G, Pedrosa J, Silva R A, Orme I M, Minoprio P. Infect Immun. 1994;62:3962–3971. doi: 10.1128/iai.62.9.3962-3971.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Crawford F, Kozono H, White J, Marrack P, Kappler J. Immunity. 1998;8:675–682. doi: 10.1016/s1074-7613(00)80572-5. [DOI] [PubMed] [Google Scholar]
- 32.Kedl R M, Rees W A, Hildeman D A, Schaefer B, Mitchell T, Kappler J, Marrack P. J Exp Med. 2000;192:1105–1114. doi: 10.1084/jem.192.8.1105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kedl R M, Mescher M F. J Immunol. 1997;159:650–663. [PubMed] [Google Scholar]
- 34.Sahin U, Tureci O, Pfreundschuh M. Curr Opin Immunol. 1997;9:709–716. doi: 10.1016/s0952-7915(97)80053-2. [DOI] [PubMed] [Google Scholar]
- 35.Tureci O, Sahin U, Pfreundschuh M. Mol Med Today. 1997;3:342–349. doi: 10.1016/s1357-4310(97)01081-2. [DOI] [PubMed] [Google Scholar]
- 36.Pinilla-Ibarz J, Cathcart K, Scheinberg D A. Blood Rev. 2000;14:111–120. doi: 10.1054/blre.2000.0127. [DOI] [PubMed] [Google Scholar]
- 37.Offringa R, Vierboom M P, van der Burg S H, Erdile L, Melief C J. Ann NY Acad Sci. 2000;910:223–236. doi: 10.1111/j.1749-6632.2000.tb06711.x. [DOI] [PubMed] [Google Scholar]
- 38.Abrams S I, Hand P H, Tsang K Y, Schlom J. Semin Oncol. 1996;23:118–134. [PubMed] [Google Scholar]
- 39.Hung K, Hayashi R, Lafond-Walker A, Lowenstein C, Pardoll D, Levitsky H. J Exp Med. 1998;188:2357–2368. doi: 10.1084/jem.188.12.2357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Overwijk W W, Lee D S, Surman D R, Irvine K R, Touloukian C E, Chan C C, Carroll M W, Moss B, Rosenberg S A, Restifo N P. Proc Natl Acad Sci USA. 1999;96:2982–2987. doi: 10.1073/pnas.96.6.2982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Toes R E, Ossendorp F, Offringa R, Melief C J. J Exp Med. 1999;189:753–756. doi: 10.1084/jem.189.5.753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Schmidt C S, Mescher M F. J Immunol. 1999;163:2561–2567. [PubMed] [Google Scholar]
- 43.Parmiani G, Rivoltini L, Andreola G, Carrabba M. Immunol Lett. 2000;74:41–44. doi: 10.1016/s0165-2478(00)00247-9. [DOI] [PubMed] [Google Scholar]
- 44.Becker J C, Brocker E B. Exp Dermatol. 1994;3:283–289. doi: 10.1111/j.1600-0625.1994.tb00290.x. [DOI] [PubMed] [Google Scholar]
- 45.Pape K A, Kearney E R, Khoruts A, Mondino A, Merica R, Chen Z M, Ingulli E, White J, Johnson J G, Jenkins M K. Immunol Rev. 1997;156:67–78. doi: 10.1111/j.1600-065x.1997.tb00959.x. [DOI] [PubMed] [Google Scholar]
- 46.Pape K A, Khoruts A, Mondino A, Jenkins M K. J Immunol. 1997;159:591–598. [PubMed] [Google Scholar]
- 47.Vella A T, McCormack J E, Linsley P S, Kappler J W, Marrack P. Immunity. 1995;2:261–270. doi: 10.1016/1074-7613(95)90050-0. [DOI] [PubMed] [Google Scholar]
- 48.Gorelik L, Bar-Dagan Y, Mokyr M B. J Immunol. 1996;156:4298–4308. [PubMed] [Google Scholar]
- 49.Oppmann B, Lesley R, Blom B, Timans J C, Xu Y, Hunte B, Vega F, Yu N, Wang J, Singh K, et al. Immunity. 2000;13:715–725. doi: 10.1016/s1074-7613(00)00070-4. [DOI] [PubMed] [Google Scholar]
- 50.Schulz O, Edwards D A, Schito M, Aliberti J, Manickasingham S, Sher A, Reise S. Immunity. 2000;13:453–462. doi: 10.1016/s1074-7613(00)00045-5. [DOI] [PubMed] [Google Scholar]