Abstract
Osteoporosis develops when the rate of osteoclastic bone breakdown (resorption) exceeds that of osteoblastic bone formation, which leads to loss of BMD and deterioration of bone structure and strength. Osteoporosis increases the risk of fragility fractures, a cause of substantial morbidity and mortality, especially in elderly patients. This imbalance between bone formation and bone resorption is brought about by natural ageing processes, but is frequently exacerbated by a number of pathological conditions. Of importance to the aetiology of osteoporosis are findings over the past two decades attesting to a deep integration of the skeletal system with the immune system (the immuno–skeletal interface (ISI)). Although protective of the skeleton under physiological conditions, the ISI might contribute to bone destruction in a growing number of pathophysiological states. Although numerous research groups have investigated how the immune system affects basal and pathological osteoclastic bone resorption, recent findings suggest that the reach of the adaptive immune response extends to the regulation of osteoblastic bone formation. This Review examines the evolution of the field of osteoimmunology and how advances in our understanding of the ISI might lead to novel approaches to prevent and treat bone loss, and avert fractures.
Osteoporosis results in a significant increase in the risk of fragility fractures. Almost 50% of women and 30% of men over the age of 50 years will suffer a fracture1. With an upward trend in life expectancy, the worldwide incidence of fracture is expected to increase to 6 million by 2050 (REF. 2). Fractures can have devastating consequences, and are often associated with crippling disability requiring rehabilitation in up to 75% of individuals, loss of independence and a marked decline in quality of life3,4. In the case of hip fractures, mortality of 24–33% is common in the first year following a fracture2,4–6.
Although bone loss is a consequence of natural ageing, numerous disease states accelerate this process leading to skeletal damage at a relatively early age. Such conditions include postmenopausal osteoporosis in women, autoimmune diseases including rheumatoid arthritis, periodontal infection (a leading cause of tooth loss), hyperparathyroidism resulting from tumours or impaired calcium absorption, infection with human immunodeficiency virus (HIV) and the combination antiretroviral therapy (cART) used in HIV treatment7,8.
In inflammation, activated immune cells promote bone destruction by stimulating bone-resorbing osteoclasts. However, under physiological conditions the immune system might actually protect the skeleton. Consequently, disruption of immune function, such as occurs in HIV infection, might promote bone loss9,10. Furthermore, new findings suggest that certain pharmacological stimuli including teriparatide, an intermittently administered fragment of parathyroid hormone (PTH)11–14 and abatacept (cytotoxic T-lymphocyte-associated protein 4 (CTLA4)-Ig), a pharmacological co-stimulation inhibitor that renders T cells dormant (anergic)15, promote or intensify osteoblastic bone formation, in part, through T cells. This paradoxical inter-relationship between the immune system and the skeletal system results from an immuno–skeletal interface (ISI), in which immune cells and their mediators, which regulate physiological immune responses to infection, serve a dual function as skeletal regulators.
In this Review, we examine evidence for the involvement of the ISI in skeletal physiology and pathophysiology and discuss how our understanding of the ISI might lead to novel approaches to prevent and/or treat bone loss by targeting upstream inflammatory and/or immunological cascades beyond classic downstream bone formation and bone resorption pathways. This Review is written predominantly from the perspective of osteoimmunology and is based on work over the past 2 decades. Lack of validation in human systems and variable outcomes in some animal models have, however, led to disagreements concerning the importance of immune cells in some models. Furthermore, many of the concepts discussed here have yet to be formally adopted into mainstream bone biology. Nonetheless, extraordinary progress is being made in dissecting the ISI and evidence continues to accumulate in support of a role for immune cells in both physiological and pathophysiological bone states.
Physiological bone turnover
Role of the immune system
Osteoclastogenesis and the RANK–RANKL–OPG axis
The skeleton is constantly renewed by the coordinated removal of worn bone by osteoclasts and the deposition of new bone by osteoblasts16. Whereas osteoblasts develop from cells of mesenchymal origin, precursors of osteoclasts have, since the 1970s, been recognized to derive from monocytes and macrophages (that is, cells of haematopoietic origin)17–19. However, the exact mechanism for converting a monocyte into an osteoclast has remained elusive for over 2 decades.
In 1997, osteoprotegerin (OPG), a potent inhibitor of osteoclast differentiation, was discovered20,21. This discovery was quickly followed by that of its ligand, osteoprotegerin ligand (OPGL)22,23, a potent inducer of osteoclast differentiation. OPGL, also known as TNF-related activation-induced cytokine (TRANCE)24,25, is now referred to in the bone field, by convention, as receptor activator of NF-κB (RANK) ligand (RANKL)26. Interestingly, RANKL was first described as a T-cell-derived cytokine mediating T-cell proliferation and dendritic cell functions26. Besides this immunological role, RANKL is now considered the key downstream effector cytokine required for osteoclast formation and activity.
In the presence of permissive concentrations of macrophage colony-stimulating factor (M-CSF), a survival factor for cells of the monocyte–macrophage–osteoclast lineage, RANKL binds to its receptor (RANK) on osteoclast precursors and promotes their differentiation into pre-osteoclasts, which fuse together into mature bone resorbing osteoclasts. OPG is a physiological decoy receptor of RANKL27,28 and the RANKL to OPG ratio is a key determinant of osteoclast differentiation and bone resorption in the body.
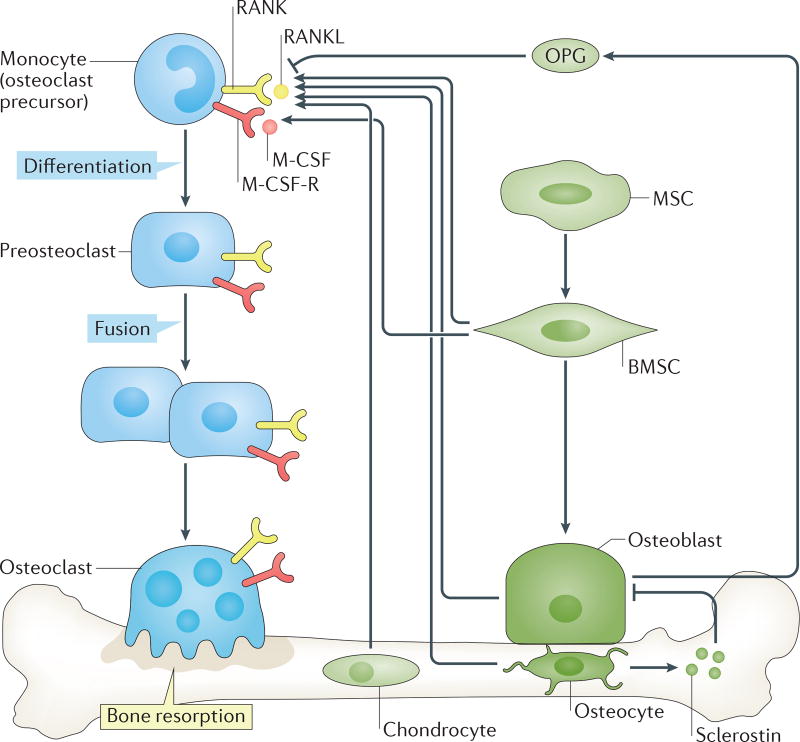
Although several cell types have the potential to make RANKL and OPG, under physiological conditions, osteoblasts and/or their precursors (bone marrow stromal cells) are generally regarded as the dominant source of both RANKL and OPG. Some studies have suggested, however, that during bone remodelling chondrocytes and osteocytes are also essential sources of RANKL29,30 and, as discussed later in the text, B cells might also be a key source of OPG. The osteocyte, a terminally differentiated osteoblast embedded in bone matrix, is also a key source of sclerostin, a Wnt pathway antagonist that serves to decrease bone formation and has become a bone anabolic therapeutic target31. The discovery of the RANK–RANKL–OPG pathway (FIG. 1) has revolutionized our understanding of osteoclast biology and established a basis for understanding the ISI.
Figure 1. Role of the RANK–RANKL–OPG system in physiological osteoclast formation.
Osteoclast precursors derived from cells of the monocyte lineage express receptor activator of NF-κB (RANK), the receptor for the key osteoclastogenic cytokine RANK ligand (RANKL). In the presence of permissive concentrations of macrophage colony-stimulating factor 1 (M-CSF), which binds to its receptor M-CSF-R (commonly known as c-fms), RANKL promotes differentiation of monocytes into pre-osteoclasts that fuse into multinucleated mature bone resorbing osteoclasts. Osteoprotegerin (OPG) is a decoy receptor for RANKL and moderates its activity. Bone-forming osteoblasts derive from mesenchymal stem cells (MSCs) that differentiate into pluripotent progenitors, often referred to as bone marrow stromal cells (BMSCs). Under appropriate conditions, BMSCs differentiate into osteoblasts that are capable of synthesizing and mineralizing bone matrix. Both BMSCs and osteoblasts are considered a major source of RANKL and OPG. Some osteoblasts might further differentiate into osteocytes that remain encapsulated within bone and continue to sense the microenvironment and secrete factors that regulate bone turnover such as RANKL and the inhibitor of bone formation, sclerostin.
B cells — a key source of OPG and potent regulators of bone turnover
In 1998, human B cells were reported to secrete OPG32. A decade later, murine bone marrow B-cell subsets (precursors, immature, mature and plasma cells) were shown to be collectively responsible for 64% of total bone marrow OPG, with mature B cells alone accounting for almost 40%33. OPG production in mature B cells was further demonstrated to be important for bone homeostasis in vivo, as B-cell-deficient mice had low BMD owing to increased bone resorption and a marked deficit in bone marrow OPG. Transplantation of B cells into B-cell-deficient mice normalized OPG production and prevented elevated bone resorption and loss of bone mass33.
T cells — key regulators of OPG production in B cells and basal bone turnover
T cells are a cornerstone of adaptive immunity and via secreted factors and surface bound co-stimulatory molecules they regulate the function of antigen presenting cells (APCs) and B-cell-directed humoral immunity34. Evidence for the involvement of T cells in physiological bone remodelling emerged as far back as 1983, when an increase in bone resorption was reported in T-cell-deficient nude mice35. This increased bone resorption was attributed to an osteoclastogenic factor, probably secreted by B cells or macrophages in response to T-cell deficiency35. Similarly, co-culture of macrophages and activated T cells resulted in the production of a soluble osteoclastogenic factor, which led to speculation that bone remodelling might be regulated, in part, by cells of the immune system36.
Importantly, studies of OPG production by human B cells revealed that ligation of the CD40 receptor on B cells results in upregulation of OPG mRNA expression32. The counterpart of CD40, CD40 ligand (CD40-L), is highly expressed by activated T cells, which suggests a link between B-cell–T-cell interactions and OPG production, and the potential for both B cells and T cells to regulate the basal RANKL to OPG ratio. Additional support for a role of T cells in the regulation of OPG levels came from studies in which T-cell subsets (CD4+ and CD8+) were depleted in mice using neutralizing antibodies37. Culture of T-cell-depleted bone marrow ex vivo with an osteoclastogenic stimulus (vitamin D3) markedly increased osteoclast formation. Osteoclastogenesis was reversed by indomethacin, suggesting the involvement of prostaglandin; however, T-cell depletion also induced complete suppression of OPG production37. Furthermore, in a mouse model of periodontal infection, depletion of B cells intensified alveolar bone loss, suggesting that B cells produce antiresorptive factors38.
Consistent with a role of lymphocytes in the regulation of basal osteoclastogenesis, elevated bone resorption and diminished BMD has been reported in T-cell-deficient nude mice, which was associated with a deficiency in the production of OPG by B cells33. Moreover, genetic deletion of T cells, or the genes encoding the CD40 and CD40-L co-stimulatory molecules, increased bone resorption and bone deterioration in mice, and was accompanied by a considerable decline in OPG production by B cells33. In support of a role of CD40 and/or CD40-L in bone turnover in humans, polymorphisms in the Kozak sequence of the CD40 gene promoter are associated with low BMD in postmenopausal women39. Furthermore, mutations in CD40LG (which encodes human CD40-L) are associated with increased bone resorption, low BMD and osteoporotic fractures in X-linked hyper-IgM syndrome40.
Another co-stimulatory molecule involved in the activation of T cells is CD28. CTLA4 is an inhibitor of CD28 activation and is secreted by cells of the T-cell lineage41. CTLA4 binds to the CD28 ligands, CD80 (also known as B7.1) and CD86 (also known as B7.2), expressed by APCs41. Interestingly, CTLA4 directly inhibits RANKL-induced and TNF-induced osteoclast formation in vitro41. As monocytes — the precursors of osteoclasts — also function as APCs, they express CD80 and CD86 molecules. The binding of CTLA4 to these ligands leads to the generation of intracellular signals that induce activation of indoleamine 2,3-dioxygenase (IDO) in osteoclast precursors, thereby leading to tryptophan degradation and induction of apoptosis42. Consequently, CD80, CD86 and IDO-deficient mice have increased osteoclastogenesis and an osteopenic phenotype42. CTLA4 might, therefore, have an important role in the physiological regulation of bone resorption and preservation of bone mass42.
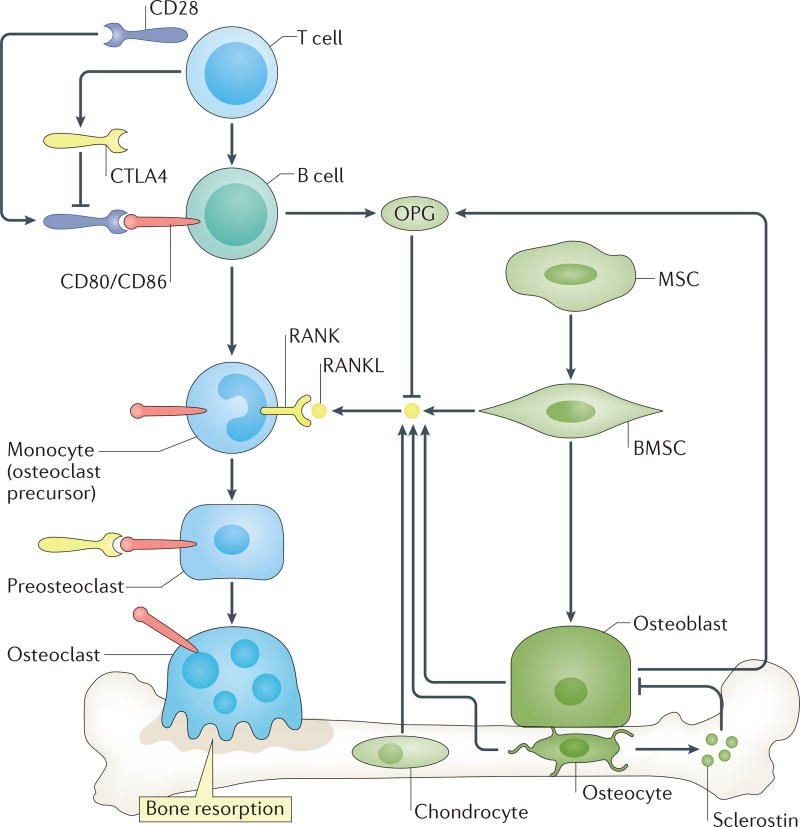
Taken together, current evidence supports the notion that both B and T lymphocytes moderate physiological bone resorption through regulation of the RANKL to OPG ratio, as well as via direct effects of CTLA4 on osteoclast precursors and, therefore, contribute to the maintenance of peak BMD. This immunocentric model of immune cell involvement in basal osteoclast regulation is shown in FIG. 2.
Figure 2. Role of the immuno–skeletal interface in physiological osteoclast formation.
Under physiological conditions, B cells, regulated by co-stimulatory interactions with T cells, supplement levels of osteoprotegerin (OPG) in the bone marrow. OPG decoys receptor activator of NF-κB ligand (RANKL), derived from multiple sources, moderating osteoclast resorption and stabilizing bone remodelling. Certain T-cell subpopulations including regulatory T (Treg) cells further secrete cytotoxic T-lymphocyte protein 4 (CTLA4), which can bind to CD80/CD86 on osteoclast precursors, thereby promoting apoptosis and diminishing bone resorption. BMSC, bone marrow stromal cell; MSC, mesenchymal stem cell; RANK, receptor activator of NF-κB.
Pathological bone turnover
Role of the immune system
Immune disruption and bone loss in HIV infection
If lymphocytes protect the skeleton, bone loss would be predicted to accompany the acquired immunodeficiency syndrome (AIDS) associated with HIV infection, as HIV results in depletion of CD4+ T cells as well as B-cell dysfunction through loss of co-stimulation and/or direct viral attack43,44. In fact, bone loss is common in patients infected with HIV45–50 and nearly two thirds of patients infected with HIV exhibit osteopenia and about 15% have osteoporosis51–53.
A dramatic increase in fracture incidence has been demonstrated in HIV-infected individuals over a wide age range in both sexes54. Additional studies have reported an ~2–4% overall increase in fractures and up to a ninefold increase in the incidence of hip fracture54–60. Not all studies, however, have found an increased incidence of fractures with HIV infection61. Nevertheless, almost 50% of individuals living with HIV infection in the USA are over the age of 50 years62, and the HIV population worldwide is entering an age demographic in which fracture incidence is substantially increased. Concerns are therefore mounting as to a looming epidemic of fractures and the need for preventive measures63.
Defining the cause of bone loss in this population has been extremely difficult owing, in part, to traditional risk factors for osteoporosis being common among HIV-infected individuals. These risk factors include low BMI, AIDS-associated pathologies such as muscle wasting, kidney disease, hypogonadism, vitamin D insufficiency or deficiency, smoking, and recreational drug and alcohol use64,65. Furthermore, considerable confusion exists regarding whether bone loss is a result of HIV infection, related to traditional risk factors, a consequence of cART, or a combination of these factors46,47,51,66–68.
Development of the HIV-1 transgenic rat model, which develops many of the pathologies characteristic of human AIDS69, was a major scientific advance facilitating the study of HIV-mediated bone loss in the absence of confounding factors associated with HIV infection in humans. BMD and bone structure are markedly reduced in the HIV-1 transgenic rat due to elevated bone resorption and an increase in the ratio of RANKL to OPG10,70. Furthermore, OPG production by B cells was reduced, exacerbated by substantial B-cell production of RANKL10. These studies demonstrated, for the first time, the potential involvement of the ISI in HIV-associated bone loss.
In 2014, these preclinical data were translated back to the human system in a study involving 58 HIV− and 62 cART-naive HIV+ individuals9. As expected, increased bone resorption, low BMD and, indeed, osteopenia was common in the HIV-infected individuals. Importantly, as in the rat model, B-cell expression of RANKL was increased markedly while B-cell expression of OPG was diminished. These changes remained statistically significantly associated with HIV status following multivariate analysis adjusting for age, sex, race, BMI, smoking, alcohol consumption and fracture history. Furthermore, the RANKL to OPG ratio in B cells was significantly associated with BMD and BMD-derived T-scores and/or Z-scores at the total hip and femoral neck, two common sites of fracture in humans9. These findings are consistent, in part, with those of another study, in which serum levels of OPG (but not RANKL) were significantly associated with lumbar spine, total hip and femoral neck Z-scores in cART-naive patients71.
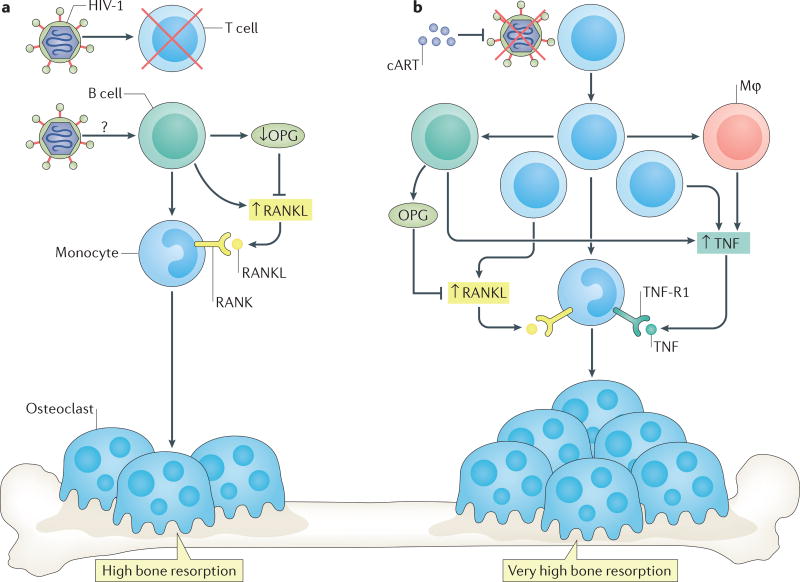
Although bone loss in HIV-infected individuals is probably multifactorial, the aforementioned data demonstrate an immunological underpinning that probably contributes to bone loss and the high fracture rates in HIV-infected individuals (immunocentric model of HIV-induced bone loss; FIG. 3a).
Figure 3. Bone loss associated with HIV infection and initiated by cART.
a | The immunodeficiency characteristic of HIV infection leads to damage and depletion of T cells and B cells, which, in B cells, reduces production of osteoprotegerin (OPG) and increases production of receptor activator of NF-κB ligand (RANKL). In turn, the RANKL to OPG ratio is increased and results in bone loss. b | T-cell activation and repopulation following initiation of combination antiretroviral therapy (cART) leads to secretion of osteoclastogenic cytokines including RANKL and tumour necrosis factor (TNF) from T cells and other immune components such as B cells and macrophages. Secretion of these factors increases bone loss beyond that already induced by the imbalance of RANKL to OPG in B cells caused by HIV infection. Mφ, macrophage; RANK, receptor activator of NF-κB; TNF-R1, TNF receptor 1.
Inflammatory bone loss associated with HIV-1-antiretroviral therapy
cART is recommended for all patients following a diagnosis of HIV and is effective in reversing many of the sequelae of HIV infection and AIDS. Paradoxically, however, skeletal deterioration continues unabated and in many cases actually becomes markedly worse46,64,72–75. Although not all studies have found decreased BMD and, in fact, some have reported increased or stable BMD76,77, in general most studies report a 2–6% loss of BMD, predominantly within the first 2 years of initiation of cART73 that probably exacerbates the bone loss already sustained by those with chronic HIV infection.
cART comprises combinations of different drugs, usually from two or more drug classes. This wide range of drug combinations, used concomitantly, has confounded comparison of the effects of individual constituent antiretroviral drugs on bone. Furthermore, accurate modelling of the effects of cART in cell culture systems in vitro78–80 and in murine models in vivo81 has been difficult, as the effects of cART generally fail to replicate the pattern of bone loss observed in humans infected with HIV. Owing to the different modes of action and different biochemical pathways affected by different cART regimens, one particular drug or drug class would be expected to be responsible for bone loss. Although effect sizes can vary and cART regimens containing tenofovir disoproxil fumarate result in the most bone loss, paradoxically, bone loss has been reported for all drug classes. Consequently, the evidence suggests that a major component of bone loss is independent of the particular cART regimen used82,83,66.
T-cell repopulation and subsequent immune reactivation following T-cell recovery has been hypothesized to be a common denominator between cART regimens, generating an inflammatory state favourable for osteoclastic bone loss. In fact, chronic inflammation and immune activation are known to persist in patients on cART and might contribute to end-organ damage such as end stage renal and liver disease, cardiovascular disease, neurocognitive disorders and, indeed, bone disease84.
Activated T cells produce several osteoclastogenic cytokines including RANKL26,24 and TNF, a cytokine that promotes RANKL expression by osteoblasts85 and directly synergizes with RANKL to amplify osteoclastogenesis86 and bone resorption87. A marked increase in plasma levels of RANKL and TNF has been reported in some, but not all, patients following initiation of ART, together with a significant correlation between the magnitude of CD4+ T-cell recovery and bone resorption88. These findings suggest that a more aggressive T-cell repopulation drives higher rates of bone resorption88 and supports a contribution from an inflammatory state involving T-cell activation and osteoclastogenic cytokine production in cART-induced bone loss. However, such changes in serum factors have not been observed in all studies and, despite a significant increase in bone resorption in patients initiating cART, one study reported a significant decline in levels of RANKL89. The reason for the discrepancies between studies is presently unclear; however, circulating factors might not reflect actual conditions at sites of bone turnover and might need to be interpreted with caution.
To further test the notion of immune reconstitution causing bone loss we mimicked this process in the context of immunodeficiency in an animal model by syngeneic T-cell reconstitution of T-cell deficient mice88. T-cell reconstitution caused a marked increase in bone resorption and declines in BMD and trabecular and cortical microarchitecture. Although T cells were the primary instigator of bone loss and produced both RANKL and TNF, reactivation of adaptive immunity further led to RANKL and/or TNF secretion by other immune components including B cells and monocytes/macrophages.
These data support the concept that inflammatory reactions associated with T-cell reconstitution following cART initiation might significantly contribute to the bone loss common to all cART regimens. Our data do not, however, exclude additional direct effects of cART on bone cells. In humans, bone loss is probably multifactorial and inflammatory events might only be one contributing factor. This inflammatory component of cART-induced bone loss is presented in FIG. 3b.
Therapies and interventions to prevent bone loss associated with HIV and cART
Given the relatively young age of affected individuals and the inability of traditional bone densitometry to predict fractures in patients under the age of 50 years90, the implications of osteopenia and even osteoporosis in these individuals were, until recently, unclear. This situation resulted in a conservative approach to intervention, even though clinical studies have demonstrated efficacy of antiresorptive drugs such as zoledronic acid in increasing and maintaining BMD in patients with HIV receiving cART91,92 and of alendronate for treating low BMD93. Guidelines currently recommend screening by dual-energy X-ray absorptiometry (DXA) of men aged ≥50 years, postmenopausal women, patients with a history of fragility fracture, patients receiving chronic glucocorticoid treatment and patients at high risk of falls, and initiation of anti-osteoporotic interventions when indicated73,94.
Despite these recommendations, bone mass, once lost, is difficult to restore with antiresorptive therapies owing to a simultaneous drop in bone formation observed with almost all antiresorptive drugs95,96, which leads to suppression of bone turnover. Administration of a long-lasting antiresorptive prophylaxis to patients at the time of initiating cART has been proposed to block bone loss during the period when resorption is most intense88. As a proof of concept, zoledronic acid was demonstrated to ameliorate bone loss associated with T-cell reconstitution in mice, without evidence of impediment to early T-cell reconstitution97. The results of a phase II clinical study to test this concept of prophylaxis against bone loss in treatment naive HIV-infected patients initiating cART have now been published98. These data reveal that a single administration of zoledronic acid significantly protect patients through the first 48 weeks, the period when ART-induced bone loss is most pronounced, at the lumbar spine, hip and femoral neck98.
Although a direct antiresorptive approach will probably have benefits for bone status and reduce the risk of fracture, the underlying immuno–skeletal imbalance is not corrected. Targeting inflammation might consequently be an important long-term direction for more inclusively dealing with bone loss as well as other end-organ complications associated with cART that are all exacerbated, if not instigated, by a persistent inflammatory state. Obviously, care is needed when targeting the immune system, especially in the context of prior immunodeficiency. However, immunosuppressive drugs, such as etanercept and infliximab that block TNF activity and action, respectively99,100, B-cell-abating antibodies, such as rituximab101 and co-stimulation inhibitors, such as abatacept102, have all been successfully used to blunt inflammation with direct or indirect benefits for bone turnover in rheumatoid arthritis. Such agents might find future applications in ameliorating inflammation in other disease states (including HIV infection), in which inflammation persists and continues to damage the skeleton even when viral replication is successfully suppressed by cART.
Immune activation and bone loss in rheumatoid arthritis
Rheumatoid arthritis is an autoimmune disease that affects 2% of adult individuals, which results in chronic inflammation in the synovial membrane of affected joints and leads to chronic pain and fatigue and, ultimately, permanent disability and increased mortality103. In terms of the ISI, rheumatoid arthritis might be the most well accepted condition in which inflammation underlies bone loss. Although rheumatoid arthritis results in severe joint damage, global bone loss resulting from disruption of the systemic control of bone remodelling is common104. In rheumatoid arthritis, activated T cells and B cells infiltrate the synovial membrane and initiate and sustain an inflammatory state. However, these activated immune cells are thought to also drive systemic bone loss associated with rheumatoid arthritis primarily by secretion of RANKL and TNF103–108.
TNF is a key protagonist of both inflammation and bone loss in rheumatoid arthritis and overexpression of TNF in mice is commonly used to model the joint and systemic bone destruction observed in patients with the disease109,110. Increased production of IL-7, a master regulator of T-cell differentiation, maturation and activity111, has long been associated with adult and juvenile rheumatoid arthritis112 and administration of IL-7 causes expansion of T cells and B cells and aggravates inflammation in collagen-induced arthritis, a mouse model of rheumatoid arthritis113. Furthermore, blockade of IL-7 inhibits collagen-induced arthritis by suppressing T-cell activation and inflammatory cytokine production114.
Regulatory T cells (Treg cells) are a subset of potent immunosuppressive/anti-inflammatory T cells. Treg cells are mainly CD4+, but can be CD8+ and are characterized by expression of CD25 and FOXP3 (REF. 115). The balance between effector T cells and Treg cells is central to immune homeostasis115. In vivo studies in humans and animal models suggest that Treg cells can suppress inflammation-induced bone loss116,117. In fact, a strong inverse correlation has been reported between the number of circulating Treg cells and levels of bone resorption markers in patients with rheumatoid arthritis, which suggests that Treg cells might moderate bone destruction in rheumatoid arthritis in vivo117.
Although Treg cells would be expected to diminish immune activation and, thus, moderate downstream inflammatory bone loss, interestingly, in vitro co-cultures of purified human118 and murine119 Treg cells with osteoclast precursors revealed inhibitory effects of Treg cells on RANKL-induced osteoclastogenesis. This finding suggested that a direct inhibitory effect exists, independent of the anti-inflammatory actions of Treg cells and has been attributed to production of the anti-osteoclastogenic cytokines TGF-β and IL-4118. By contrast, another study concluded that although TGF-β, IL-4 and IL-10 probably contributed, the dominant effect of Treg cells is mediated by production of CTLA4 (REF. 119).
Treg cells further mediate potent protective effects against bone destruction in the TNF transgenic mouse model of rheumatoid arthritis117. Although Treg cells diminished clinical signs of arthritis, the investigators concluded that the dominant bone-protective effect of Treg cells was probably mediated by direct effects on osteoclastogenesis rather than by suppression of inflammation117. This conclusion is supported by findings of increased basal BMD in wild-type mice harbouring increased numbers of Treg cells117, in Foxp3 overexpressing transgenic mice and in T-cell–deficient RAG-1 null mice reconstituted with Treg cells120.
Although RANKL is considered to be the key effector of osteoclastogenesis, a novel RANKL-independent T-cell-secreted cytokine, threonine synthase-like 2 (originally named secreted osteoclastogenic factor of activated T cells; SOFAT) has been identified and speculated to exacerbate inflammation and/or bone loss in rheumatoid arthritis121. Little is known about the action of SOFAT in rheumatoid arthritis or any other bone disease; however, enhanced SOFAT gene expression and protein production has been reported in periodontal tissues of patients with chronic periodontal disease122, a bacterial-induced inflammatory response resulting in gingival soft tissue and alveolar bone destruction (a cause of tooth loosening and loss)116. Injection of SOFAT induced osteoclast formation in periodontal ligaments of mice in vivo122.
Therapies and novel interventions to prevent bone loss in rheumatoid arthritis
As the role of the immune system in the aetiology of rheumatoid arthritis is so well established and owing to the debilitating nature and high morbidity the disease imparts, the rheumatology field has pioneered the use of novel immunomodulatory drugs for blocking inflammation and protection of systemic and local bone erosion. Targeting of inflammatory cascades by blocking TNF effectively alleviates inflammation and bone loss in mouse models of rheumatoid arthritis123 and TNF antagonists are approved for the treatment of patients with rheumatoid arthritis99. In addition, as immune activation is involved in the aetiology of rheumatoid arthritis, powerful immunomodulatory drugs that target T-cell co-stimulation are now being used in intractable cases of rheumatoid arthritis. One such drug is abatacept, a pharmaceutical based on natural CTLA4 that dampens inflammatory responses. Furthermore, Treg cells might suppress osteoclastogenic bone loss not only by dampening upstream inflammatory cascades, but also by mediating potent direct production of anti-osteoclastogenic factors.
Similar to rheumatoid arthritis, blocking Treg function in a murine model of periodontal infection increased inflammation and bone loss124. Furthermore, blocking Treg cell migration to inflammatory sites by deletion of the Treg chemoattractant factor CCR4, or its ligand CCL22, resulted in an increase in inflammatory bone loss125. By contrast, controlled release of CCL22 at sites of inflammation recruited Treg cells and mitigated bone resorption in both murine and canine models of periodontal disease116. Although the contribution of direct and indirect mechanisms to bone turnover in humans remains to be studied, harnessing the anti-osteoclastogenic activities of Treg cells might be a promising strategy to protect the skeleton in inflammatory disease states. The rheumatoid arthritis field will probably continue to lead the way in the discovery of novel immunomodualtory therapeutics for reversing inflammation and bone disease.
Role of the immuno–skeletal interface in oestrogen deficiency-induced bone loss
Bone loss in postmenopausal osteoporosis is a complex process with a multifactorial aetiology. An increase in the pituitary hormone, follicle-stimulating hormone (FSH), might be an early event instigating an increase in bone resorption126. The dominant effect, however, is generally ascribed to ovarian steroid deficiency following menopause, most notably oestrogen, a potent anti-inflammatory and bone protective factor8. Oestrogen deficiency leads to an increase in bone resorption through increased osteoclastogenesis, decreased osteoclast apoptosis and increased osteoclast activity127. Oestrogen protects bone via numerous complex effects on bone turnover, some mediated directly on the basal bone homeostatic machinery. These effects, which boost bone formation, include reducing self-renewal of osteoblast progenitors128, decreasing stromal cell production of OPG129, increasing production of M-CSF130 and RANKL131, and decreasing the anti-anabolic factor sclerostin132. Interestingly, oestrogen is also potently immunosuppressive133 and over the past 2 decades an immunocentric aspect to oestrogen-deficiency bone loss has emerged suggesting potent permissive or additive effects of the ISI to the classic perspective.
B cells and oestrogen deficiency bone loss
Intensive studies have elucidated unexpected putative roles of the ISI in oestrogen deficiency bone loss. Clinical studies have documented significant increases in RANKL production by both B cells and T cells derived from postmenopausal women134. The recognition that oestrogen deficiency upregulates B lymphopoiesis135, along with evidence that the potent B-lymphopoietic cytokine IL-7 is an inducer of bone loss, suggested a possible role for B cells in the aetiology of postmenopausal osteoporosis136. A 2012 study using conditional B-cell RANKL knock-out mice documented considerable protection from vertebral, but not femoral, bone loss137 and, thus, suggested a contributory role of B cell RANKL to the trabecular bone loss component in ovariectomy.
T-cells and oestrogen deficiency bone loss
The first empirical evidence for a supporting role of T cells in oestrogen deficiency bone loss came from studies showing that T-cell deficient nude mice were resistant to ovariectomy-induced bone loss86. Although T cells are known to make RANKL, TNF production rather than RANKL was key to ovariectomy-induced bone loss in mice as evidenced by TNF and TNF type I receptor null mice being protected. Furthermore, the conditional knockout of TNF in T-cells using an adoptive transfer model, produced a mouse that was resistant to ovariectomy-induced bone loss and suggested that T cells are the required source of TNF necessary for bone loss138. The dispensability of RANKL production by mouse T cells has been independently verified using advanced genetic models in which RANKL was conditionally ablated in T-cells137.
Extensive mechanistic studies have further unravelled the basis for production of TNF by T cells and have revealed that loss of oestrogen initiates a multipronged attack on the ISI. This attack begins with an increase in reactive oxygen species, which leads to CD80 receptor expression on dendritic cells139. In addition, enhanced production of IFN-γ upregulates major histocompatibility complex (MHC) class II (MHCII) antigen expression on macrophages by stimulating the class II transactivator (CIITA) transcription factor140. These actions culminate in an increased sensitivity of APCs to T-cell-mediated co-stimulatory signals and T cells to prevailing antigens promoting T-cell activation and leading to TNF production. In vitro co-culture studies suggest that oestrogen might promote secretion of the anti-inflammatory cytokine IL-10 and of TGF-β from Treg cells141. Indeed, conditional blockade of TGF-β production by T cells renders them completely insensitive to the bone-sparing effects of oestrogen142.
As osteoclasts are derived from monocytes/macrophages, cells that function as APCs, osteoclasts have, unsurprisingly, been reported to retain a capacity to present antigens to other immune cells and can activate T cells143. Interestingly, while presentation of antigens by dendritic cells induced formation of cytolytic CD8+ T cells, presentation of antigens by osteoclasts induced a regulatory T-cell phenotype in CD8+ T cells115. Importantly, transplantation of osteoclast-induced Treg cells into ovariectomized mice prevented the increase in osteoclast number, bone resorption and bone loss. These data suggest a negative feedback by osteoclasts involving differentiation of Treg cells from CD8+ T-cells115.
Much of the immunocentric basis for oestrogen deficiency bone loss is predicated on the notion of APC-mediated immune responses, which in classical immunological terms are expected to be antigen-driven processes; indeed, ablating antigen presentation protects mice from ovariectomy-induced bone loss140. Although a persistent low-level assault by weak self-antigens and foreign antigens exists even in healthy animals and humans, the nature of the antigens involved in ovariectomy-induced bone loss is still unclear. However, the gastrointestinal tracts of humans and animals are colonized by vast numbers of bacteria and other microorganisms commonly referred to as the gut microbiota. Treatment with the probiotic Lactobacillus reuteri markedly protects against ovariectomy-induced bone resorption and loss and supresses the expansion of CD4+ T cells in the bone marrow144. Furthermore, Lactobacillus species, either singly or as a mixture of three strains, protected ovariectomized mice from cortical bone loss, reduced levels of markers of bone resorption and inflammation (TNF and IL-1β, respectively) and prevented a decline in the number of Treg cells145. These studies suggest that the antigens pertinent to immune responses causing bone loss in ovariectomy models might be derived, in part, from the gut microbiota. Indeed, in germ-free mice, sex steroid deficiency fails to increase levels of osteoclastogenic cytokines, stimulate bone resorption or cause trabecular bone loss146.
A further consequence of declining TGF-β and increasing hepatic IGF-1 (REF. 147) production is increased production of IL-7 (REF. 8), a potent inducer of T-cell RANKL production and osteoclastic bone loss136,148,149. In addition to these signals, an increase in IL-17-producing TH17 T cells further stimulates osteoblastic RANKL production and promotes bone loss150. Transplantation of human exfoliated deciduous teeth (SHED) stem cells have been reported to protect mice from ovariectomy-induced bone loss by inducing apoptosis of activated TH1 and TH17 T cells151. Furthermore, suppression of B lymphopoeisis and of TNF-secreting senescent T cells by daidzein prevents the increase in bone marrow TNF and bone loss associated with ovariectomy in mice152. Furthermore, an antibody against IL-17 suppressed ovariectomy-induced CD4+ T-cell proliferation, reversed T-cell senescence (characterized by loss of CD28) and induced expansion of Treg cells, leading to protection from bone loss. Neutralization of TNF or RANKL also blocked bone loss, but not the upstream immunological events, thereby suggesting a key direct role for IL-17 in regulating T-cell changes associated with ovariectomy153.
Interestingly, both IFN-γ and IL-7 have also been reported to directly suppress osteoclast formation154,155. Although systemic overexpression of IL-7 resulted in osteolysis156, osteoblastic overproduction of IL–7 in the bone marrow only, led to a small increase in bone volume in female mice, but a nonsignificant decline in male mice157. Although osteoclast numbers were increased ex vivo, the increased bone mass might, in part, be a consequence of suppressive effects of IL-7 on bone formation. Supporting evidence for this suggestion comes from the observation that IL-7 suppressed bone formation in calvarial organ cultures and when injected into mice158. IL-7 knockout mice are also reported to have a marked increase in osteoblast surface157, which suggests complex effects on bone formation and resorption. The effects of IL-7 on OPG production in B cells are presently unknown but might account, in part, for some of the suppressive effects on osteoclastogenesis.
However, under inflammatory conditions, such as during oestrogen deficiency, the balance of indirect stimulatory effects of IFN-γ and IL-7 on the skeleton, mediated through the ISI, seem to overwhelm the direct inhibitory effects on osteoclast differentiation, leading to net bone loss. Additional support for the pro-osteoclastogenic effect of IFN-γ in vivo comes from studies reporting that IFN-γ administration induces bone resorption and ameliorates osteopetrosis in mice159,160 and humans161.
Independent verification of a role for T cells in ovariectomy-induced bone loss came from a study showing a failure of T-cell-deficient NIH-III beige nude mice to increase osteoclast numbers and loose bone mass, events rescued by adoptive transfer of T cells. This study further reported that T cells stimulated apoptosis of osteoblast precursors, thereby contributing to the decline in bone formation that accentuates oestrogen deficiency bone loss162.
Not all studies have found key roles for T cells in ovariectomy-induced bone loss, and in some studies only selective protection from bone loss in immunocompromised mice has been seen. Studies in T-cell-deficient Rowett athymic nude rats failed to identify protection from trabecular bone loss quantified by histomorphometry163. Whether T cells are dispensable for ovariectomy-induced bone loss in the rat or whether the outcome was due to the incomplete T-cell deficiency in the rat model is unknown. Even though in immunocompromised mice adoptive transfer of T cells leads to only partial reconstitution (typically around 50%) of T cells, full rescue of bone loss associated with ovariectomy is possible, which suggests that even an incomplete complement of T cells might be permissive in some models.
In another study, variable outcomes were observed in different T-cell-deficient mice strains including nude, RAG-2 null (missing both T cells and B cells) and T cell receptor α null mice. Trabecular bone loss was still observed in these models but cortical bone loss was prevented in all mouse strains except RAG-2 null mice164. BALBc nude mice have also been reported to lose trabecular but not cortical bone volume165.
The reason for the discrepancies in outcomes between these studies remains unclear, although subtle differences in experimental conditions such as residual T cells, animal diets, the gut microbiota, environmental factors and hormonal status could all be contributing factors. In nude mice, age might be of particular importance as extra-thymic maturation of T cells leads to partial recovery of the T cell population. Although a compelling body of work exists to support a role for T cells in ovariectomy-induced bone loss, several aspects remain to be clarified.
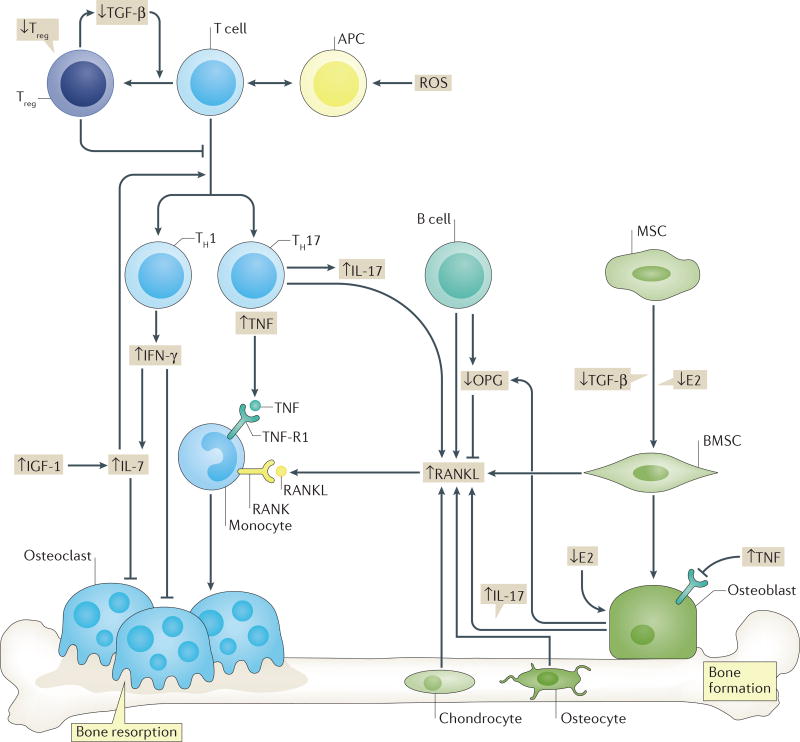
Data supporting an immunocentric basis for postmenopausal bone loss in humans remain relatively sparse; however, RANKL levels have been reported to be upregulated by bone marrow B cells and T cells from postmenopausal women in comparison with those from premenopausal and oestrogen treated postmenopausal controls134. Moreover, markedly elevated T-cell production of TNF and RANKL has been reported in women with postmenopausal osteoporosis compared with that in healthy postmenopausal women166. Furthermore, young patients (mean age 46.3 years) undergoing elective surgical ovariectomy were found to have increased T-cell activation and thymic hypertrophy compared with control individuals167. This model of ISI involvement in oestrogen-induced bone loss is presented in FIG. 4.
Figure 4. Augmentation of oestrogen deficiency bone loss by changes in the immuno–skeletal interface.
Oestrogen decline leads to complex direct and indirect effects on bone cells including suppression of bone formation, a reduction in osteoprotegerin (OPG) production and an increase in production of receptor activator of NF-κB ligand (RANKL) by osteoblasts and/or bone marrow stromal cells (BMSCs). T-cell activation and expansion following oestrogen decline amplifies these events though secretion of the proinflammatory cytokine tumour necrosis factor (TNF), which synergizes with prevailing RANKL and thereby upregulates osteoclastic bone resorption. TNF production is the net result of a complex immunological realignment following oestrogen withdrawal and involves a cascade of events that include downregulation of transforming growth factor β (TGF-β) production and upregulation of insulin-like growth factor 1 (IGF-1) production. These events promote a decline in the number of regulatory T (Treg) cells and increased production of IFN-γ by type 1 T helper (TH1) cells and IL-17-producing type 17 T helper (TH17) cells. IL-17 promotes RANKL production by osteoblasts whereas IFN-γ increases antigen presenting cell (APC) activity. Increased IL-7 production facilitates multiple T-cell processes including proliferation and survival, and T-cell activation. The net result is an expansion of T cells and production of TNF. Although B cells might contribute to bone loss, the mechanism is presently unclear. E2, oestradiol; MSC, mesenchymal stem cell; RANK, receptor activator of NF-κB. ROS, reactive oxygen species; TNF-R1, TNF receptor 1.
Therapies and novel interventions to prevent oestrogen deficiency bone loss
Postmenopausal osteoporosis has been proposed to exhibit many of the classic hallmarks of an inflammatory disease8, which has important ramifications for future therapeutic strategies to ameliorate bone loss. As oestrogen deficiency seems to instigate a chronic inflammatory state propitious for bone loss, breaking the inflammatory cascade at any one of multiple nexus points seems to be effective in alleviating bone loss, at least in animal models138,142,147,158,168,169. Indeed, pharmacological or genetic ablation of IL-1 and TNF138,168, IL-7 (REF. 158), IFN-γ169 and IGF-1 (REF. 147), or overexpression of TGF-β by somatic gene therapy142, are all effective in preventing ovariectomy-induced bone loss in mice. Interestingly, IL-7 knockout mice are protected from cortical, but not trabecular, bone loss following ovariectomy170 whereas IL-17 receptor knockout mice have markedly more ovariectomy-induced bone loss than controls owing to the inhibitory effects of IL-17 on adipogenesis and leptin expression171. Targeting these pathways therapeutically is, therefore, complicated and can lead to complex and unexpected outcomes. Furthermore, overproduction of Treg cells protects ovariectomized mice from bone resorption and bone loss120.
Consistent with a role of antigens in the inflammatory aspect of ovariectomy-induced bone loss, treatment of oestrogen-deficient mice with probiotics, but not E. coli, reduces gut permeability, diminishes adsorption of bacterial antigens into the circulation and dampens intestinal and bone marrow inflammation and bone loss146. Blunting the development of inflammation by use of probiotics or by regulating the gut microbiota could be a simple and fairly cheap alternative to pharmacological measures and is an important future direction in the field.
Finally, one osteoimmunological target, RANKL itself, has been successfully targeted pharmacologically and an antibody against RANKL (denosumab) has been approved by the FDA for fracture prevention172. However. future therapies for postmenopausal osteoporosis might involve selective targeting of upstream inflammatory cascades.
Hyperparathyroidism-induced bone loss and the role of the immune system
Serum calcium levels are tightly regulated in the body and even small decrements in serum-ionized calcium lead to secretion of PTH, which drives skeletal catabolism to release calcium stores. Excessive production of PTH leads to hyperparathyroidism and severe skeletal deterioration173. PTH promotes bone resorption by complex mechanisms that involve increased production of RANKL and decreased production of OPG in cells of the osteoblast lineage174. However, studies performed more than 15 years ago revealed that nude mice deficient in T cells are protected from the catabolic effects of hyperparathyroidism, which suggests a permissive role for T cells175.
Investigations have confirmed a lack of bone catabolic activity of PTH in T-cell-deficient T-cell receptor β knockout mice, as well as following conditional deletion of the PTH receptor in T cells13,176. Bone loss in T-cell receptor β knockout mice was rescued following adoptive transfer of T cells176. Mechanistically, these data suggest that T cells provide a source of TNF that augments the catabolic activity of PTH by amplifying RANKL signalling and promoting the expression of CD40 on osteoblast precursors. Activated T cells deliver proliferative and survival signals though CD40L, the counterpart of CD40, further facilitating production of RANKL and diminishing levels of OPG174–177.
Therapies and novel interventions for bone loss in hyperparathyroidism
The finding that T-cells are involved in bone loss in hyperparathyroidism again suggests the potential for novel interventions involving immunosuppression. A proof of concept has been published, in which ablation of antigen presentation in mice, by silencing either class I or class II MHC interactions with T cells, prevents cortical bone loss whereas the co-stimulation inhibitor CTLA4-Ig likewise silences the catabolic activity of PTH177.
Bone formation
Role of the immune system
Bone anabolic activity of RANKL
Although RANKL is the key osteoclastogenic cytokine, administration of low concentrations of RANKL promotes CD8+ Treg cell development in ovariectomized mice by stimulation of Notch ligand (DLL4) production in osteoclasts and production of the co-stimulatory molecule CD200 (REF. 178). The magnitude of this effect was sufficient to prevent increased bone resorption and bone loss. Another interesting finding from this study was a potent bone anabolic effect of low-dose RANKL, which led to increased bone formation and mineral apposition178.
Whether RANKL can be administered in a manner capable of eliciting anti-osteoclastogenic effects and/or bone anabolic effects remains unclear. However, with an improved understanding of the mechanisms involved, these pathways might provide important new therapeutic targets in the future. The mechanisms underlying the anabolic effect of RANKL are presently unclear and might, in part, involve a reduction in levels of anti-osteoblastogenic cytokines such as TNF178. An alternative explanation could be production of CTLA4 by Treg cells, a molecule with unexpected bone anabolic potential.
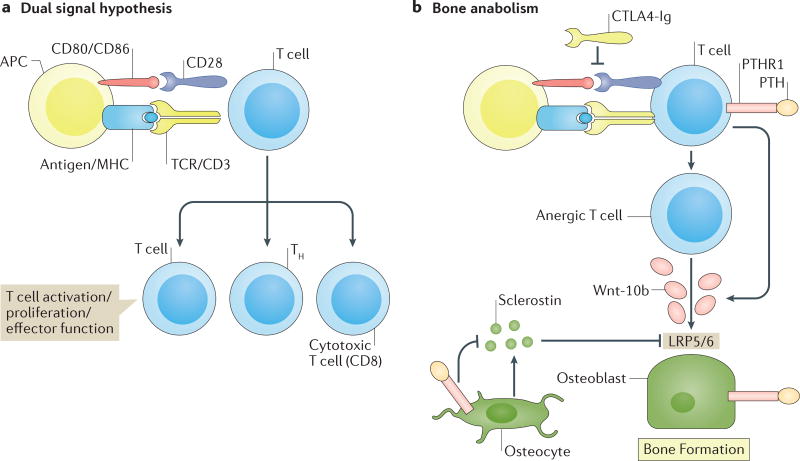
Bone anabolic activity of CTLA4-Ig and anergic T cells
Little is known about the effects of T cells on basal bone turnover. In healthy mice, CTLA4-Ig induces a robust gain in BMD and in trabecular and cortical bone mass, surprisingly, a consequence of increased bone formation15. Mechanistically, CTLA4-Ig seems to promote bone formation as a quirk of its suppressive activity on T-cell activation. In the dual signal hypothesis of T-cell activation, engagement of antigens presented by APCs to the T-cell receptor initiates an anergic signal. Reversal of this signal by a second co-stimulatory signal from the APC (directed via CD80 and CD86 ligands to the CD28 receptor on the T cell) is necessary for T-cell activation to occur. Natural CTLA4 and pharmacological CTLA4-Ig block this second signal committing the T cell to an anergic state179. This inhibition favours the production of cAMP, which mediates upregulation of the Wnt10b promoter15. Wnt-10b, an anabolic Wnt pathway ligand, promotes osteoblast differentiation and activation, which stimulates bone formation. Whether CTLA4-Ig can promote bone formation in humans through effects on the ISI remains to be examined but, if validated, CTLA4-Ig might have application as a novel bone anabolic agent for promoting bone formation in patients with osteoporosis.
Bone anabolic effects of PTH
Chronic delivery of PTH, such as in the setting of hyperparathyroidism and thyroid tumours, leads to osteoclastic bone loss. However, when PTH is delivered in an intermittent and/or pulsatile fashion, a robust bone anabolic response is induced in animal models and in humans that completely offsets bone resorption and leads to a net gain of bone. This phenomenon, which remains to be explained mechanistically, has led to the development of teriparatide, the first bone anabolic agent approved by the FDA in the USA for osteoporosis therapy180.
PTH has been extensively studied in animal models and humans and it’s anabolic actions are the result of increased osteoblast proliferation and differentiation, activation of quiescent lining osteoblasts, which increase osteoblast lifespan as a consequence of diminished apoptosis, and downregulation of levels of sclerostin (a Wnt receptor antagonist) in osteocytes180. A central molecular mechanism involved in PTH-induced bone formation involves activation of Wnt signalling in the osteoblast. Interestingly, data from our group suggest that an important source of the Wnt ligand necessary for PTH-induced bone anabolism is the T cell13,14. These conclusions are supported by studies demonstrating muted anabolic responses to intermittent PTH administration in T-cell deficient T-cell receptor β knockout mice14 and in mice specifically lacking the PTH receptor in T cells13. Furthermore, the Wnt ligand secreted by T cells that promotes bone formation in response to PTH has been shown to be Wnt-10b, as mice with T cells incapable of producing Wnt-10b fail to promote bone formation following PTH administration14.
Although the molecular mechanisms of PTH action on T cells are still under investigation, as Wnt10b is also elicited by CTLA4-Ig-induced T-cell anergy15, a convergence of the same intracellular pathways is probable. The dual-signal hypothesis of physiological T-cell activation and the proposed mechanisms by which CTLA4-Ig and PTH promote bone anabolism are presented in FIG. 5.
Figure 5. T-cell anergy promotes Wnt-10b secretion and bone anabolism.
a | In the dual signal hypothesis of T-cell activation, engagement of an antigen presented by an antigen presenting cell (APC) to the T-cell receptor (TCR) initiates an anergic signal that requires a second co-stimulatory signal from the APC directed via CD80/CD86 ligands to the CD28 receptor on the T cell, for T-cell activation, proliferation and differentiation into T helper cells (TH) or cytotoxic CD8 T cells to proceed. b | Pharmacological blockade of the second co-stimulatory signal commits the T cell to an anergic state that favours the release of protein Wnt-10b (Wnt-10b), an anabolic Wnt pathway ligand that promotes osteoblast differentiation and activation, thereby stimulating bone formation. Intermittent binding of parathyroid hormone (PTH) to its receptor (PTHR1) ‘hijacks’ the T-cell machinery, promotes release of Wnt-10b and amplifies the direct effects of PTH on bone cells. These effects include differentiation, proliferation, activation, generation of anti-apoptotic signals and downregulation of osteocyte production of sclerostin, a Wnt receptor (LRP5/6) antagonist, which leads to potent anabolic effects on bone. CTLA4-Ig, cytotoxic T-lymphocyte protein 4-Ig; MHC, major histocompatibility complex.
Intermittent PTH administration — the first anabolic therapy for osteoporosis
From a therapeutic perspective, teriparatide has been an important development in the prevention of fracture, as current antiresorptive medications that form the standard of care effectively suppress bone degradation but lead to a state of low bone turnover that is inefficient at recovering lost bone mass and can eventually damage bone. However, limitations including high costs and an inconvenient daily subcutaneous delivery has limited the broad use of teriparatide, and novel anabolic agents are urgently needed.
Although new therapies based on modulation of the Wnt signalling pathway, such as the sclerostin-targeting antibody romosozumab31 are currently under development or undergoing clinical trials, modulation of the ISI and the potential of T cells to secrete Wnt-10b might eventually be a novel mechanism to promote bone formation. Interestingly, because the immune system is a prominent source of anti-anabolic factors including TNF, anti-inflammatory agents might possess bone anabolic properties. Mice deficient in TNF or its type I receptor (p55) exhibit a markedly elevated basal peak BMD as a consequence of enhanced bone formation181. This effect is probably a consequence of diminished nuclear factor κB signalling in osteoblasts, which potently suppresses bone formation. Although TNF is a potent stimulator of osteoclastic bone resorption at high pathological concentrations, low-dose TNF is sufficient to suppress bone formation181,182. Although in these studies all sources of TNF were ablated, TNF in T cells is a key mediator of bone loss in oestrogen deficiency and a key driver of tissue destruction and bone loss in rheumatoid arthritis and with cART. Indeed, anti-inflammatory pharmaceuticals targeting TNF can ameliorate bone loss, in part, by promoting bone formation in patients with rheumatoid arthritis100. Selective targeting of TNF production in T cells has never been investigated in humans but could be an area of future investigation as the ability to selectively target specific cell populations continues to evolve.
Conclusions
As the field of osteoimmunology continues to evolve and mature, new developments are expected. Work to date has provided new unexpected explanations for bone loss involving the ISI in conditions as seemingly diametrically opposed as inflammation and immunosuppression. A rapidly growing list of skeletal disease states now exists that might involve, in part, defects in the ISI. This list includes classic inflammatory diseases such as rheumatoid arthritis, novel inflammatory conditions such as postmenopausal osteoporosis and cART-induced bone loss, and yet to be classified immuno–skeletal diseases such has hyperparathyroidism. In addition, disruption of basic immuno–skeletal homeostatic processes might contribute to the bone loss associated with HIV infection.
The evolution of anti-osteoporotic therapeutics might eventually include upstream immune targets, rather than the classic downstream effectors of bone resorption that are currently the standard of care. The identification of anabolic functions deeply encoded within T-cell responses is providing new insights into the mechanisms of action of anabolic PTH, whereas pharmacologic manipulation of T cells by agents such as abatacept might be exploited to promote bone formation and add bulk to the skeleton to offset the deleterious effects of ageing and bone disease.
At the present time, the use of potent immunomodualtory biologics have been mainly reserved for cases of rheumatoid arthritis refractory to other therapies. Although well tolerated in most patients, the risks of immunosuppression and potential adverse effects such as reduced ability to fight infections (for example, tuberculosis and histoplasmosis) and increased susceptibility to certain cancers, remain a concern. The potential risk to reward ratio of targeting inflammatory cascades to alleviate conditions such as postmenopausal bone loss is currently unclear and existing therapeutic strategies still focus almost exclusively on the final effectors of osteoclastogenesis, osteoblastogenesis, and the osteoclasts and osteoblasts themselves. As the field progresses and an improved understanding of the integration between immune and skeletal functions emerges, new targets for amelioration of bone loss will probably be identified.
Key points.
Receptor activator of NF-κB ligand (RANKL) and osteoprotegerin (OPG) are key downstream effectors of bone resorption; tumour necrosis factor (TNF) might synergize with RANKL to superinduce osteoclastic bone resorption
B cells, regulated by T cells, are a key source of basal OPG whereas activated T cells and B cells are key sources of TNF and RANKL in inflammatory conditions
Short-term antiresorptive therapies might safely prevent bone loss associated with combination antiretroviral therapy
Anergic and parathyroid-hormone-treated T cells secrete the protein Wnt-10b, which promotes bone formation
Novel therapeutic strategies targeting the immune system might promote bone formation and decrease bone resorption to manage osteoporotic bone loss and prevent fracture
Acknowledgments
The authors gratefully acknowledge research support from the Biomedical Laboratory Research and Development Service of the Veteran’s Affairs Office of Research and Development (Grant BX000105 to M.N.W.) and National Institutes of Health grants from the National Institute of Arthritis and Musculoskeletal and Skin Diseases (AR059364, AR056090 and AR053607) and the National Institute on Aging (AG040013) to M.N.W. and I.O.
Footnotes
Author contributions
Both authors researched data for the article, made substantial contributions to discussions of the content, and edited and/or reviewed the manuscript before submission. M.N.W. wrote the manuscript.
Competing interests statement
M.N.W. has filed a patent on the use of abatacept for stimulating bone anabolism. I.O. declares no competing interests.
References
- 1.Eisman JA, et al. Making the first fracture the last fracture: ASBMR task force report on secondary fracture prevention. J. Bone Miner. Res. 2012;27:2039–2046. doi: 10.1002/jbmr.1698. [DOI] [PubMed] [Google Scholar]
- 2.World Health Organization. Prevention and management of osteoporosis. WHO; 2003. [PubMed] [Google Scholar]
- 3.Kates SL, Kates OS, Mendelson DA. Advances in the medical management of osteoporosis. Injury. 2007;38:S17–S23. doi: 10.1016/j.injury.2007.08.007. [DOI] [PubMed] [Google Scholar]
- 4.Johnell O, Kanis JA. An estimate of the worldwide prevalence and disability associated with osteoporotic fractures. Osteoporos. Int. 2006;17:1726–1733. doi: 10.1007/s00198-006-0172-4. [DOI] [PubMed] [Google Scholar]
- 5.Bass E, French DD, Bradham DD, Rubenstein LZ. Risk-adjusted mortality rates of elderly veterans with hip fractures. Ann. Epidemiol. 2007;17:514–519. doi: 10.1016/j.annepidem.2006.12.004. [DOI] [PubMed] [Google Scholar]
- 6.Lewis JR, Hassan SK, Wenn RT, Moran CG. Mortality and serum urea and electrolytes on admission for hip fracture patients. Injury. 2006;37:698–704. doi: 10.1016/j.injury.2006.04.121. [DOI] [PubMed] [Google Scholar]
- 7.Ofotokun I, McIntosh E, Weitzmann MN. HIV: inflammation and bone. Curr. HIV/AIDS Rep. 2012;9:16–25. doi: 10.1007/s11904-011-0099-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Weitzmann MN, Pacifici R. Estrogen deficiency and bone loss: an inflammatory tale. J. Clin. Invest. 2006;116:1186–1194. doi: 10.1172/JCI28550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Titanji K, et al. Dysregulated B cell expression of RANKL and OPG correlates with loss of bone mineral density in HIV infection. PLoS Pathog. 2014;10:e1004497. doi: 10.1371/journal.ppat.1004497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vikulina T, et al. Alterations in the immuno–skeletal interface drive bone destruction in HIV-1 transgenic rats. Proc. Natl Acad. Sci. USA. 2010;107:13848–13853. doi: 10.1073/pnas.1003020107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Robinson JW, et al. T cell expressed CD40L potentiates the bone anabolic activity of intermittent PTH treatment. J. Bone Miner. Res. 2014;30:695–705. doi: 10.1002/jbmr.2394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Li JY, et al. The sclerostin-independent bone anabolic activity of intermittent PTH treatment is mediated by T-cell-produced Wnt10b. J. Bone Miner. Res. 2014;29:43–54. doi: 10.1002/jbmr.2044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bedi B, et al. Silencing of parathyroid hormone (PTH) receptor 1 in T cells blunts the bone anabolic activity of PTH. Proc. Natl Acad. Sci. USA. 2012;109:E725–E733. doi: 10.1073/pnas.1120735109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Terauchi M, et al. T lymphocytes amplify the anabolic activity of parathyroid hormone through Wnt10b signaling. Cell Metab. 2009;10:229–240. doi: 10.1016/j.cmet.2009.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Roser-Page S, Vikulina T, Zayzafoon M, Weitzmann MN. CTLA-4Ig-induced T cell anergy promotes Wnt-10b production and bone formation in a mouse model. Arthritis Rheumatol. 2014;66:990–999. doi: 10.1002/art.38319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Manolagas SC. Birth and death of bone cells: basic regulatory mechanisms and implications for the pathogenesis and treatment of osteoporosis. Endocr. Rev. 2000;21:115–137. doi: 10.1210/edrv.21.2.0395. [DOI] [PubMed] [Google Scholar]
- 17.Walker DG. Bone resorption restored in osteopetrotic mice by transplants of normal bone marrow and spleen cells. Science. 1975;190:784–785. doi: 10.1126/science.1105786. [DOI] [PubMed] [Google Scholar]
- 18.Walker DG. Congenital osteopetrosis in mice cured by parabiotic union with normal siblings. Endocrinology. 1972;91:916–920. doi: 10.1210/endo-91-4-916. [DOI] [PubMed] [Google Scholar]
- 19.Buring K. On the origin of cells in heterotopic bone formation. Clin. Orthop. Relat. Res. 1975;110:293–301. doi: 10.1097/00003086-197507000-00040. [DOI] [PubMed] [Google Scholar]
- 20.Simonet WS, et al. Osteoprotegerin: a novel secreted protein involved in the regulation of bone density. Cell. 1997;89:309–319. doi: 10.1016/s0092-8674(00)80209-3. [DOI] [PubMed] [Google Scholar]
- 21.Tsuda E, et al. Isolation of a novel cytokine from human fibroblasts that specifically inhibits osteoclastogenesis. Biochem. Biophys. Res. Commun. 1997;234:137–142. doi: 10.1006/bbrc.1997.6603. [DOI] [PubMed] [Google Scholar]
- 22.Lacey DL, et al. Osteoprotegerin ligand is a cytokine that regulates osteoclast differentiation and activation. Cell. 1998;93:165–176. doi: 10.1016/s0092-8674(00)81569-x. [DOI] [PubMed] [Google Scholar]
- 23.Matsuzaki K, et al. Osteoclast differentiation factor (ODF) induces osteoclast-like cell formation in human peripheral blood mononuclear cell cultures. Biochem. Biophys. Res. Commun. 1998;246:199–204. doi: 10.1006/bbrc.1998.8586. [DOI] [PubMed] [Google Scholar]
- 24.Wong BR, et al. TRANCE (tumor necrosis factor [TNF]-related activation-induced cytokine), a new TNF family member predominantly expressed in T cells, is a dendritic cell-specific survival factor. J. Exp. Med. 1997;186:2075–2080. doi: 10.1084/jem.186.12.2075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wong BR, et al. TRANCE is a novel ligand of the tumor necrosis factor receptor family that activates c-Jun N-terminal kinase in T cells. J. Biol. Chem. 1997;272:25190–25194. doi: 10.1074/jbc.272.40.25190. [DOI] [PubMed] [Google Scholar]
- 26.Anderson DM, et al. A homologue of the TNF receptor and its ligand enhance T-cell growth and dendritic-cell function. Nature. 1997;390:175–179. doi: 10.1038/36593. [DOI] [PubMed] [Google Scholar]
- 27.Teitelbaum SL. Bone resorption by osteoclasts. Science. 2000;289:1504–1508. doi: 10.1126/science.289.5484.1504. [DOI] [PubMed] [Google Scholar]
- 28.Khosla S. Minireview: the OPG/RANKL/RANK system. Endocrinology. 2001;142:5050–5055. doi: 10.1210/endo.142.12.8536. [DOI] [PubMed] [Google Scholar]
- 29.Xiong J, et al. Matrix-embedded cells control osteoclast formation. Nat. Med. 2011;17:1235–1241. doi: 10.1038/nm.2448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Nakashima T, et al. Evidence for osteocyte regulation of bone homeostasis through RANKL expression. Nat. Med. 2011;17:1231–1234. doi: 10.1038/nm.2452. [DOI] [PubMed] [Google Scholar]
- 31.McClung MR, et al. Romosozumab in postmenopausal women with low bone mineral density. N. Engl. J. Med. 2014;370:412–420. doi: 10.1056/NEJMoa1305224. [DOI] [PubMed] [Google Scholar]
- 32.Yun TJ, et al. OPG/FDCR-1, a TNF receptor family member, is expressed in lymphoid cells and is up-regulated by ligating CD40. J. Immunol. 1998;161:6113–6121. [PubMed] [Google Scholar]
- 33.Li Y, et al. B cells and T cells are critical for the preservation of bone homeostasis and attainment of peak bone mass in vivo. Blood. 2007;109:3839–3848. doi: 10.1182/blood-2006-07-037994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Surh CD, Sprent J. Homeostasis of naive and memory T cells. Immunity. 2008;29:848–862. doi: 10.1016/j.immuni.2008.11.002. [DOI] [PubMed] [Google Scholar]
- 35.Gyarmati J, et al. Alterations of the connective tissue in nude mice. Thymus. 1983;5:383–392. [PubMed] [Google Scholar]
- 36.Horowitz M, Vignery A, Gershon RK, Baron R. Thymus-derived lymphocytes and their interactions with macrophages are required for the production of osteoclast-activating factor in the mouse. Proc. Natl Acad. Sci. USA. 1984;81:2181–2185. doi: 10.1073/pnas.81.7.2181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Grcevic D, Lee SK, Marusic A, Lorenzo JA. Depletion of CD4 and CD8 T lymphocytes in mice in vivo enhances 1, 25- dihydroxyvitamin D3-stimulated osteoclast-like cell formation in vitro by a mechanism that is dependent on prostaglandin synthesis. J. Immunol. 2000;165:4231–4238. doi: 10.4049/jimmunol.165.8.4231. [DOI] [PubMed] [Google Scholar]
- 38.Klausen B, Hougen HP, Fiehn NE. Increased periodontal bone loss in temporarily B lymphocyte-deficient rats. J. Periodontal Res. 1989;24:384–390. doi: 10.1111/j.1600-0765.1989.tb00887.x. [DOI] [PubMed] [Google Scholar]
- 39.Pineda B, Laporta P, Hermenegildo C, Cano A, Garcia-Perez MA. A C > T polymorphism located at position-1 of the Kozak sequence of CD40 gene is associated with low bone mass in Spanish postmenopausal women. Osteoporos. Int. 2007;19:1147–1152. doi: 10.1007/s00198-007-0536-4. [DOI] [PubMed] [Google Scholar]
- 40.Lopez-Granados E, et al. Osteopenia in X-linked hyper-IgM syndrome reveals a regulatory role for CD40 ligand in osteoclastogenesis. Proc. Natl Acad. Sci. USA. 2007;104:5056–5061. doi: 10.1073/pnas.0605715104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Axmann R, et al. CTLA-4 directly inhibits osteoclast formation. Ann. Rheum. Dis. 2008;67:1603–1609. doi: 10.1136/ard.2007.080713. [DOI] [PubMed] [Google Scholar]
- 42.Bozec A, et al. T cell costimulation molecules CD80/86 inhibit osteoclast differentiation by inducing the IDO/tryptophan pathway. Sci. Transl Med. 2014;6:235ra260. doi: 10.1126/scitranslmed.3007764. [DOI] [PubMed] [Google Scholar]
- 43.Moir S, Fauci AS. B cells in HIV infection and disease. Nat. Rev. Immunol. 2009;9:235–245. doi: 10.1038/nri2524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Moir S, et al. Evidence for HIV-associated B cell exhaustion in a dysfunctional memory B cell compartment in HIV-infected viremic individuals. J. Exp. Med. 2008;205:1797–1805. doi: 10.1084/jem.20072683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Aukrust P, et al. Decreased bone formative and enhanced resorptive markers in human immunodeficiency virus infection: indication of normalization of the bone-remodeling process during highly active antiretroviral therapy. J. Clin. Endocrinol. Metab. 1999;84:145–150. doi: 10.1210/jcem.84.1.5417. [DOI] [PubMed] [Google Scholar]
- 46.Tebas P, et al. Accelerated bone mineral loss in HIV-infected patients receiving potent antiretroviral therapy. AIDS. 2000;14:F63–F67. doi: 10.1097/00002030-200003100-00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Jain RG, Lenhard JM. Select HIV protease inhibitors alter bone and fat metabolism ex vivo. J. Biol. Chem. 2002;277:19247–19250. doi: 10.1074/jbc.C200069200. [DOI] [PubMed] [Google Scholar]
- 48.Mora S, et al. Bone mineral loss through increased bone turnover in HIV-infected children treated with highly active antiretroviral therapy. AIDS. 2001;15:1823–1829. doi: 10.1097/00002030-200109280-00011. [DOI] [PubMed] [Google Scholar]
- 49.Huang JS, Wilkie SJ, Sullivan MP, Grinspoon S. Reduced bone density in androgen-deficient women with acquired immune deficiency syndrome wasting. J. Clin. Endocrinol. Metab. 2001;86:3533–3539. doi: 10.1210/jcem.86.8.7728. [DOI] [PubMed] [Google Scholar]
- 50.Mondy K, Tebas P. Emerging bone problems in patients infected with human immunodeficiency virus. Clin. Infect. Dis. 2003;36:S101–S105. doi: 10.1086/367566. [DOI] [PubMed] [Google Scholar]
- 51.Brown TT, Qaqish RB. Antiretroviral therapy and the prevalence of osteopenia and osteoporosis: a meta-analytic review. AIDS. 2006;20:2165–2174. doi: 10.1097/QAD.0b013e32801022eb. [DOI] [PubMed] [Google Scholar]
- 52.Bonjoch A, et al. High prevalence of and progression to low bone mineral density in HIV-infected patients: a longitudinal cohort study. AIDS. 2010;24:2827–2833. doi: 10.1097/QAD.0b013e328340a28d. [DOI] [PubMed] [Google Scholar]
- 53.Sharma A, Flom PL, Weedon J, Klein RS. Prospective study of bone mineral density changes in aging men with or at risk for HIV infection. AIDS. 2010;24:2337–2345. doi: 10.1097/QAD.0b013e32833d7da7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Triant VA, Brown TT, Lee H, Grinspoon SK. Fracture prevalence among human immunodeficiency virus (HIV)-infected versus non-HIV-infected patients in a large U.S. healthcare system. J. Clin. Endocrinol. Metab. 2008;93:3499–3504. doi: 10.1210/jc.2008-0828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Prior J, et al. Fragility fractures and bone mineral density in HIV positive women: a case–control population-based study. Osteoporos. Int. 2007;18:1345–1353. doi: 10.1007/s00198-007-0428-7. [DOI] [PubMed] [Google Scholar]
- 56.Young B, Dao CN, Buchacz K, Baker R, Brooks JT. Increased rates of bone fracture among HIV-infected persons in the HIV Outpatient Study (HOPS) compared with the US general population, 2000–2006. Clin. Infect. Dis. 2011;52:1061–1068. doi: 10.1093/cid/ciq242. [DOI] [PubMed] [Google Scholar]
- 57.Womack JA, et al. Increased risk of fragility fractures among HIV infected compared to uninfected male veterans. PLoS ONE. 2011;6:e17217. doi: 10.1371/journal.pone.0017217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Guerri-Fernandez R, et al. HIV infection is strongly associated with hip fracture risk, independently of age, gender, and comorbidities: a population-based cohort study. J. Bone Miner. Res. 2013;28:1259–1263. doi: 10.1002/jbmr.1874. [DOI] [PubMed] [Google Scholar]
- 59.Prieto-Alhambra D, et al. HIV infection and its association with an excess risk of clinical fractures: a nationwide case–control study. J. Acquir. Immune Def. Syndr. 2014;66:90–95. doi: 10.1097/QAI.0000000000000112. [DOI] [PubMed] [Google Scholar]
- 60.Sharma A, et al. Increased fracture incidence in middle-aged HIV-infected and HIV-uninfected women: updated results from the women’s interagency HIV study. J. Acquir. Immune Defic. Syndr. 2015;70:54–61. doi: 10.1097/QAI.0000000000000674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Yin MT, et al. Fracture incidence in HIV-infected women: results from the Women’s Interagency HIV Study. AIDS. 2010;24:2679–2686. doi: 10.1097/QAD.0b013e32833f6294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Vance DE, McGuinness T, Musgrove K, Orel NA, Fazeli PL. Successful aging and the epidemiology of HIV. Clin. Interv. Aging. 2011;6:181–192. doi: 10.2147/CIA.S14726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Amorosa V, Tebas P. Bone disease and HIV infection. Clin. Infect. Dis. 2006;42:108–114. doi: 10.1086/498511. [DOI] [PubMed] [Google Scholar]
- 64.Ofotokun I, Weitzmann MN. HIV-1 infection and antiretroviral therapies: risk factors for osteoporosis and bone fracture. Curr. Opin. Endocrinol. Diabetes Obes. 2010;17:523–529. doi: 10.1097/MED.0b013e32833f48d6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ofotokun I, Weitzmann MN. HIV and bone metabolism. Discov. Med. 2011;11:385–393. [PMC free article] [PubMed] [Google Scholar]
- 66.Bruera D, Luna N, David DO, Bergoglio LM, Zamudio J. Decreased bone mineral density in HIV-infected patients is independent of antiretroviral therapy. AIDS. 2003;17:1917–1923. doi: 10.1097/00002030-200309050-00010. [DOI] [PubMed] [Google Scholar]
- 67.Dube MP, et al. Prospective, intensive study of metabolic changes associated with 48 weeks of amprenavir-based antiretroviral therapy. Clin. Infect. Dis. 2002;35:475–481. doi: 10.1086/341489. [DOI] [PubMed] [Google Scholar]
- 68.Knobel H, Guelar A, Vallecillo G, Nogues X, Diez A. Osteopenia in HIV-infected patients: is it the disease or is it the treatment? AIDS. 2001;15:807–808. doi: 10.1097/00002030-200104130-00022. [DOI] [PubMed] [Google Scholar]
- 69.Reid W, et al. An HIV-1 transgenic rat that develops HIV-related pathology and immunologic dysfunction. Proc. Natl Acad. Sci. USA. 2001;98:9271–9276. doi: 10.1073/pnas.161290298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Lafferty MK, et al. Elevated suppressor of cytokine signaling-1 (SOCS-1): a mechanism for dysregulated osteoclastogenesis in HIV transgenic rats. Pathog. Dis. 2014;71:81–89. doi: 10.1111/2049-632X.12117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Brown TT, et al. Body composition, soluble markers of inflammation, and bone mineral density in antiretroviral therapy-naive HIV-1-infected individuals. J. Acquir. Immune Defic. Syndr. 2013;63:323–330. doi: 10.1097/QAI.0b013e318295eb1d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Thomas J, Doherty SM. HIV infection — a risk factor for osteoporosis. J. Acquir. Immune Defic. Syndr. 2003;33:281–291. doi: 10.1097/00126334-200307010-00001. [DOI] [PubMed] [Google Scholar]
- 73.McComsey GA, et al. Bone disease in HIV infection: a practical review and recommendations for HIV care providers. Clin. Infect. Dis. 2010;51:937–946. doi: 10.1086/656412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Yin MT, et al. Short-term bone loss in HIV-infected premenopausal women. J. Acquir. Immune Defic. Syndr. 2010;53:202–208. doi: 10.1097/QAI.0b013e3181bf6471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Bolland MJ, Grey A. HIV and low bone density: responsible party, or guilty by association? IBMS BoneKEy. 2011;8:7–15. [Google Scholar]
- 76.Nolan D, et al. Stable or increasing bone mineral density in HIV-infected patients treated with nelfinavir or indinavir. AIDS. 2001;15:1275–1280. doi: 10.1097/00002030-200107060-00009. [DOI] [PubMed] [Google Scholar]
- 77.Bolland MJ, et al. Stable bone mineral density over 6 years in HIV-infected men treated with highly active antiretroviral therapy (HAART) Clin. Endocrinol. (Oxf.) 2012;76:643–648. doi: 10.1111/j.1365-2265.2011.04274.x. [DOI] [PubMed] [Google Scholar]
- 78.Gibellini D, et al. Analysis of the effects of specific protease inhibitors on OPG/RANKL regulation in an osteoblast-like cell line. New Microbiol. 2010;33:109–115. [PubMed] [Google Scholar]
- 79.Grigsby IF, Pham L, Gopalakrishnan R, Mansky LM, Mansky KC. Downregulation of Gnas, Got2 and Snord32a following tenofovir exposure of primary osteoclasts. Biochem. Biophys. Res. Commun. 2010;391:1324–1329. doi: 10.1016/j.bbrc.2009.12.039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Grigsby IF, et al. Tenofovir treatment of primary osteoblasts alters gene expression profiles: implications for bone mineral density loss. Biochem. Biophys. Res. Commun. 2010;394:48–53. doi: 10.1016/j.bbrc.2010.02.080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Wang MW, et al. The HIV protease inhibitor ritonavir blocks osteoclastogenesis and function by impairing RANKL-induced signaling. J. Clin. Invest. 2004;114:206–213. doi: 10.1172/JCI15797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Brown TT, et al. Loss of bone mineral density after antiretroviral therapy initiation, independent of antiretroviral regimen. J. Acquir. Immune Defic. Syndr. 2009;51:554–561. doi: 10.1097/QAI.0b013e3181adce44. [DOI] [PubMed] [Google Scholar]
- 83.Piso RJ, Rothen M, Rothen JP, Stahl M. Markers of bone turnover are elevated in patients with antiretroviral treatment independent of the substance used. J. Acquir. Immune Defic. Syndr. 2011;56:320–324. doi: 10.1097/QAI.0b013e31820cf010. [DOI] [PubMed] [Google Scholar]
- 84.Deeks SG, Tracy R, Douek DC. Systemic effects of inflammation on health during chronic HIV infection. Immunity. 2013;39:633–645. doi: 10.1016/j.immuni.2013.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Hofbauer LC, et al. Interleukin-1β and tumor necrosis factor-α, but not interleukin-6, stimulate osteoprotegerin ligand gene expression in human osteoblastic cells. Bone. 1999;25:255–259. doi: 10.1016/s8756-3282(99)00162-3. [DOI] [PubMed] [Google Scholar]
- 86.Cenci S, et al. Estrogen deficiency induces bone loss by enhancing T-cell production of TNF-α. J. Clin. Invest. 2000;106:1229–1237. doi: 10.1172/JCI11066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Fuller K, Murphy C, Kirstein B, Fox SW, Chambers TJ. TNFα potently activates osteoclasts, through a direct action independent of and strongly synergistic with RANKL. Endocrinology. 2002;143:1108–1118. doi: 10.1210/endo.143.3.8701. [DOI] [PubMed] [Google Scholar]
- 88.Ofotokun I, et al. Antiretroviral therapy induces a rapid increase in bone resorption that is positively associated with the magnitude of immune reconstitution in HIV infection. AIDS. 2016;30:405–414. doi: 10.1097/QAD.0000000000000918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Brown TT, Ross AC, Storer N, Labbato D, McComsey GA. Bone turnover, osteoprotegerin/RANKL and inflammation with antiretroviral initiation: tenofovir versus non-tenofovir regimens. Antivir. Ther. 2011;16:1063–1072. doi: 10.3851/IMP1874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Hui SL, Slemenda CW, Johnston CC., Jr Age and bone mass as predictors of fracture in a prospective study. J. Clin. Invest. 1988;81:1804–1809. doi: 10.1172/JCI113523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Bolland MJ, et al. Annual zoledronate increases bone density in highly active antiretroviral therapy-treated human immunodeficiency virus-infected men: a randomized controlled trial. J. Clin. Endocrinol. Metab. 2007;92:1283–1288. doi: 10.1210/jc.2006-2216. [DOI] [PubMed] [Google Scholar]
- 92.Bolland MJ, et al. Effects of intravenous zoledronate on bone turnover and bone density persist for at least five years in HIV-infected men. J. Clin. Endocrinol. Metab. 2012;97:1922–1928. doi: 10.1210/jc.2012-1424. [DOI] [PubMed] [Google Scholar]
- 93.McComsey GA, et al. Alendronate with calcium and vitamin D supplementation is safe and effective for the treatment of decreased bone mineral density in HIV. AIDS. 2007;21:2473–2482. doi: 10.1097/QAD.0b013e3282ef961d. [DOI] [PubMed] [Google Scholar]
- 94.Brown TT, et al. Recommendations for evaluation and management of bone disease in HIV. Clin. Infect. Dis. 2015;60:1242–1251. doi: 10.1093/cid/civ010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.McClung MR, et al. Denosumab in postmenopausal women with low bone mineral density. N. Engl. J. Med. 2006;354:821–831. doi: 10.1056/NEJMoa044459. [DOI] [PubMed] [Google Scholar]
- 96.Reid IR, et al. Intravenous zoledronic acid in postmenopausal women with low bone mineral density. N. Engl. J. Med. 2002;346:653–661. doi: 10.1056/NEJMoa011807. [DOI] [PubMed] [Google Scholar]
- 97.Ofotokun I, et al. Role of T-cell reconstitution in HIV-1 antiretroviral therapy-induced bone loss. Nat. Commun. 2015;6:8282. doi: 10.1038/ncomms9282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ofotokun I, et al. A single dose zoledronic acid infusion prevents antiretroviral therapy-induced bone loss in treatment-naive HIV-infected patients: a phase IIb trial. Clin. Infect. Dis. 2016 doi: 10.1093/cid/ciw331. http://dx.doi.org/10.1093/cid/ciw331. [DOI] [PMC free article] [PubMed]
- 99.Chopin F, et al. Long term effects of Infliximab on bone and cartilage turnover markers in patients with rheumatoid arthritis. Ann. Rheum. Dis. 2007;67:353–357. doi: 10.1136/ard.2007.076604. [DOI] [PubMed] [Google Scholar]
- 100.Seriolo B, Paolino S, Sulli A, Ferretti V, Cutolo M. Bone metabolism changes during anti-TNF-α therapy in patients with active rheumatoid arthritis. Ann. NY Acad. Sci. 2006;1069:420–427. doi: 10.1196/annals.1351.040. [DOI] [PubMed] [Google Scholar]
- 101.Wheater G, et al. Suppression of bone turnover by B-cell depletion in patients with rheumatoid arthritis. Osteoporos. Int. 2011;22:3067–3072. doi: 10.1007/s00198-011-1607-0. [DOI] [PubMed] [Google Scholar]
- 102.Genovese MC, et al. Abatacept for rheumatoid arthritis refractory to tumor necrosis factor α inhibition. N. Engl. J. Med. 2005;353:1114–1123. doi: 10.1056/NEJMoa050524. [DOI] [PubMed] [Google Scholar]
- 103.Edwards JC, et al. Efficacy of B-cell-targeted therapy with rituximab in patients with rheumatoid arthritis. N. Engl. J. Med. 2004;350:2572–2581. doi: 10.1056/NEJMoa032534. [DOI] [PubMed] [Google Scholar]
- 104.Schett G, David JP. The multiple faces of autoimmune-mediated bone loss. Nat. Rev. Endocrinol. 2010;6:698–706. doi: 10.1038/nrendo.2010.190. [DOI] [PubMed] [Google Scholar]
- 105.Fournier C. Where do T cells stand in rheumatoid arthritis? Joint Bone Spine. 2005;72:527–532. doi: 10.1016/j.jbspin.2004.12.012. [DOI] [PubMed] [Google Scholar]
- 106.Kong YY, et al. Activated T cells regulate bone loss and joint destruction in adjuvant arthritis through osteoprotegerin ligand. Nature. 1999;402:304–309. doi: 10.1038/46303. [DOI] [PubMed] [Google Scholar]
- 107.He X, Kang AH, Stuart JM. Anti-human type II collagen CD19+ B cells are present in patients with rheumatoid arthritis and healthy individuals. J. Rheumatol. 2001;28:2168–2175. [PubMed] [Google Scholar]
- 108.Yanaba K, et al. B cell depletion delays collagen-induced arthritis in mice: arthritis induction requires synergy between humoral and cell-mediated immunity. J. Immunol. 2007;179:1369–1380. doi: 10.4049/jimmunol.179.2.1369. [DOI] [PubMed] [Google Scholar]
- 109.Li P, Schwarz EM. The TNF-α transgenic mouse model of inflammatory arthritis. Springer Semin. Immunopathol. 2003;25:19–33. doi: 10.1007/s00281-003-0125-3. [DOI] [PubMed] [Google Scholar]
- 110.Schett G, et al. Osteoprotegerin protects against generalized bone loss in tumor necrosis factor-transgenic mice. Arthritis Rheum. 2003;48:2042–2051. doi: 10.1002/art.11150. [DOI] [PubMed] [Google Scholar]
- 111.Fry TJ, Mackall CL. Interleukin-7: master regulator of peripheral T-cell homeostasis? Trends Immunol. 2001;22:564–571. doi: 10.1016/s1471-4906(01)02028-2. [DOI] [PubMed] [Google Scholar]
- 112.De Benedetti F, et al. Elevated circulating interleukin-7 levels in patients with systemic juvenile rheumatoid arthritis. J. Rheumatol. 1995;22:1581–1585. [PubMed] [Google Scholar]
- 113.Hartgring SA, Willis CR, Bijlsma JW, Lafeber FP, van Roon JA. Interleukin-7-aggravated joint inflammation and tissue destruction in collagen-induced arthritis is associated with T-cell and B-cell activation. Arthritis Res. Ther. 2012;14:R137. doi: 10.1186/ar3870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Hartgring SA, et al. Blockade of the interleukin-7 receptor inhibits collagen-induced arthritis and is associated with reduction of T cell activity and proinflammatory mediators. Arthritis Rheum. 2010;62:2716–2725. doi: 10.1002/art.27578. [DOI] [PubMed] [Google Scholar]
- 115.Buchwald ZS, et al. Osteoclast-induced Foxp3+ CD8 T-cells limit bone loss in mice. Bone. 2013;56:163–173. doi: 10.1016/j.bone.2013.05.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Glowacki AJ, et al. Prevention of inflammation-mediated bone loss in murine and canine periodontal disease via recruitment of regulatory lymphocytes. Proc. Natl Acad. Sci. USA. 2013;110:18525–18530. doi: 10.1073/pnas.1302829110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Zaiss MM, et al. Regulatory T cells protect from local and systemic bone destruction in arthritis. J. Immunol. 2010;184:7238–7246. doi: 10.4049/jimmunol.0903841. [DOI] [PubMed] [Google Scholar]
- 118.Kim YG, et al. Human CD4+CD25+ regulatory T cells inhibit the differentiation of osteoclasts from peripheral blood mononuclear cells. Biochem. Biophys. Res. Commun. 2007;357:1046–1052. doi: 10.1016/j.bbrc.2007.04.042. [DOI] [PubMed] [Google Scholar]
- 119.Zaiss MM, et al. Treg cells suppress osteoclast formation: a new link between the immune system and bone. Arthritis Rheum. 2007;56:4104–4112. doi: 10.1002/art.23138. [DOI] [PubMed] [Google Scholar]
- 120.Zaiss MM, et al. Increased bone density and resistance to ovariectomy-induced bone loss in FoxP3-transgenic mice based on impaired osteoclast differentiation. Arthritis Rheum. 2010;62:2328–2338. doi: 10.1002/art.27535. [DOI] [PubMed] [Google Scholar]
- 121.Rifas L, Weitzmann MN. A novel T cell cytokine, secreted osteoclastogenic factor of activated T cells, induces osteoclast formation in a RANKL-independent manner. Arthritis Rheumatol. 2009;60:3324–3335. doi: 10.1002/art.24877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Jarry CR, et al. Secreted osteoclastogenic factor of activated T cells (SOFAT), a novel osteoclast activator, in chronic periodontitis. Hum. Immunol. 2013;74:861–866. doi: 10.1016/j.humimm.2013.04.013. [DOI] [PubMed] [Google Scholar]
- 123.Redlich K, et al. Repair of local bone erosions and reversal of systemic bone loss upon therapy with anti-tumor necrosis factor in combination with osteoprotegerin or parathyroid hormone in tumor necrosis factor-mediated arthritis. Am. J. Pathol. 2004;164:543–555. doi: 10.1016/S0002-9440(10)63144-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Garlet GP, et al. Regulatory T cells attenuate experimental periodontitis progression in mice. J. Clin. Periodontol. 2010;37:591–600. doi: 10.1111/j.1600-051X.2010.01586.x. [DOI] [PubMed] [Google Scholar]
- 125.Araujo-Pires AC, et al. IL-4/CCL22/CCR4 axis controls regulatory T-cell migration that suppresses inflammatory bone loss in murine experimental periodontitis. J. Bone Miner. Res. 2015;30:412–422. doi: 10.1002/jbmr.2376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Zhu LL, et al. Blocking antibody to the β-subunit of FSH prevents bone loss by inhibiting bone resorption and stimulating bone synthesis. Proc. Natl Acad. Sci. USA. 2012;109:14574–14579. doi: 10.1073/pnas.1212806109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Clowes JA, Riggs BL, Khosla S. The role of the immune system in the pathophysiology of osteoporosis. Immunol. Rev. 2005;208:207–227. doi: 10.1111/j.0105-2896.2005.00334.x. [DOI] [PubMed] [Google Scholar]
- 128.Di Gregorio GB, et al. Attenuation of the self-renewal of transit-amplifying osteoblast progenitors in the murine bone marrow by 17 β-estradiol. J. Clin. Invest. 2001;107:803–812. doi: 10.1172/JCI11653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Szulc P, Hofbauer LC, Heufelder AE, Roth S, Delmas PD. Osteoprotegerin serum levels in men: correlation with age, estrogen, and testosterone status. J. Clin. Endocrinol. Metab. 2001;86:3162–3165. doi: 10.1210/jcem.86.7.7657. [DOI] [PubMed] [Google Scholar]
- 130.Srivastava S, et al. Estrogen blocks M-CSF gene expression and osteoclast formation by regulating phosphorylation of Egr-1 and its interaction with Sp-1. J. Clin. Invest. 1998;102:1850–1859. doi: 10.1172/JCI4561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Srivastava S, et al. Estrogen decreases osteoclast formation by down-regulating receptor activator of NF-κB ligand (RANKL)-induced JNK activation. J. Biol. Chem. 2001;276:8836–8840. doi: 10.1074/jbc.M010764200. [DOI] [PubMed] [Google Scholar]
- 132.Kim BJ, et al. TNF-α mediates the stimulation of sclerostin expression in an estrogen-deficient condition. Biochem. Biophys. Res. Commun. 2012;424:170–175. doi: 10.1016/j.bbrc.2012.06.100. [DOI] [PubMed] [Google Scholar]
- 133.Salem ML. Estrogen, a double-edged sword: modulation of TH1- and TH2-mediated inflammations by differential regulation of TH1/TH2 cytokine production. Curr. Drug Targets Inflamm. Allergy. 2004;3:97–104. doi: 10.2174/1568010043483944. [DOI] [PubMed] [Google Scholar]
- 134.Eghbali-Fatourechi G, et al. Role of RANK ligand in mediating increased bone resorption in early postmenopausal women. J. Clin. Invest. 2003;111:1221–1230. doi: 10.1172/JCI17215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Masuzawa T, et al. Estrogen deficiency stimulates B lymphopoiesis in mouse bone marrow. J. Clin. Invest. 1994;94:1090–1097. doi: 10.1172/JCI117424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Miyaura C, et al. Increased B-lymphopoiesis by interleukin 7 induces bone loss in mice with intact ovarian function: similarity to estrogen deficiency. Proc. Natl Acad. Sci. USA. 1997;94:9360–9365. doi: 10.1073/pnas.94.17.9360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Onal M, et al. Receptor activator of nuclear factor κB ligand (RANKL) protein expression by B lymphocytes contributes to ovariectomy-induced bone loss. J. Biol. Chem. 2012;287:29851–29860. doi: 10.1074/jbc.M112.377945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Roggia C, et al. Up-regulation of TNF-producing T cells in the bone marrow: a key mechanism by which estrogen deficiency induces bone loss in vivo. Proc. Natl Acad. Sci. USA. 2001;98:13960–13965. doi: 10.1073/pnas.251534698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Grassi F, et al. Oxidative stress causes bone loss in estrogen-deficient mice through enhanced bone marrow dendritic cell activation. Proc. Natl Acad. Sci. USA. 2007;104:15087–15092. doi: 10.1073/pnas.0703610104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Cenci S, et al. Estrogen deficiency induces bone loss by increasing T cell proliferation and lifespan through IFN-γ-induced class II transactivator. Proc. Natl Acad. Sci. USA. 2003;100:10405–10410. doi: 10.1073/pnas.1533207100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Luo CY, Wang L, Sun C, Li DJ. Estrogen enhances the functions of CD4+CD25+Foxp3+ regulatory T cells that suppress osteoclast differentiation and bone resorption in vitro. Cell. Mol. Immunol. 2011;8:50–58. doi: 10.1038/cmi.2010.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Gao Y, et al. Estrogen prevents bone loss through transforming growth factor β signaling in T cells. Proc. Natl Acad. Sci. USA. 2004;101:16618–16623. doi: 10.1073/pnas.0404888101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Li H, et al. Cross talk between the bone and immune systems: osteoclasts function as antigen-presenting cells and activate CD4+ and CD8+ T cells. Blood. 2010;116:210–217. doi: 10.1182/blood-2009-11-255026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Britton RA, et al. Probiotic L. reuteri treatment prevents bone loss in a menopausal ovariectomized mouse model. J. Cell. Physiol. 2014;229:1822–1830. doi: 10.1002/jcp.24636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Ohlsson C, et al. Probiotics protect mice from ovariectomy-induced cortical bone loss. PLoS ONE. 2014;9:e92368. doi: 10.1371/journal.pone.0092368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Li JY, et al. Sex steroid deficiency-associated bone loss is microbiota dependent and prevented by probiotics. J. Clin. Invest. 2016 doi: 10.1172/JCI86062. http://dx.doi.org/10.1172/JCI86062. [DOI] [PMC free article] [PubMed]
- 147.Lindberg MK, et al. Liver-derived IGF-I is permissive for ovariectomy-induced trabecular bone loss. Bone. 2006;38:85–92. doi: 10.1016/j.bone.2005.07.027. [DOI] [PubMed] [Google Scholar]
- 148.Weitzmann MN, Cenci S, Rifas L, Brown C, Pacifici R. Interleukin-7 stimulates osteoclast formation by up-regulating the T-cell production of soluble osteoclastogenic cytokines. Blood. 2000;96:1873–1878. [PubMed] [Google Scholar]
- 149.Toraldo G, Roggia C, Qian WP, Pacifici R, Weitzmann MN. IL-7 induces bone loss in vivo by induction of receptor activator of nuclear factor κB ligand and tumor necrosis factor α from T cells. Proc. Natl Acad. Sci. USA. 2003;100:125–130. doi: 10.1073/pnas.0136772100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Tyagi AM, et al. Estrogen deficiency induces the differentiation of IL-17 secreting Th17 cells: a new candidate in the pathogenesis of osteoporosis. PLoS ONE. 2012;7:e44552. doi: 10.1371/journal.pone.0044552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Liu Y, et al. Transplantation of SHED prevents bone loss in the early phase of ovariectomy-induced osteoporosis. J. Dent. Res. 2014;93:1124–1132. doi: 10.1177/0022034514552675. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Tyagi AM, et al. Daidzein prevents the increase in CD4+CD28null T cells and B lymphopoesis in ovariectomized mice: a key mechanism for anti-osteoclastogenic effect. PLoS ONE. 2011;6:e21216. doi: 10.1371/journal.pone.0021216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Tyagi AM, et al. Enhanced immunoprotective effects by anti-IL-17 antibody translates to improved skeletal parameters under estrogen deficiency compared with anti-RANKL and anti-TNF-α antibodies. J. Bone Miner. Res. 2014;29:1981–1992. doi: 10.1002/jbmr.2228. [DOI] [PubMed] [Google Scholar]
- 154.Takayanagi H, et al. T-cell-mediated regulation of osteoclastogenesis by signalling cross-talk between RANKL and IFN-γ. Nature. 2000;408:600–605. doi: 10.1038/35046102. [DOI] [PubMed] [Google Scholar]
- 155.Lee SK, Kalinowski JF, Jastrzebski SL, Puddington L, Lorenzo JA. Interleukin-7 is a direct inhibitor of in vitro osteoclastogenesis. Endocrinology. 2003;144:3524–3531. doi: 10.1210/en.2002-221057. [DOI] [PubMed] [Google Scholar]
- 156.Valenzona HO, Pointer R, Ceredig R, Osmond DG. Prelymphomatous B cell hyperplasia in the bone marrow of interleukin-7 transgenic mice: precursor B cell dynamics, microenvironmental organization and osteolysis. Exp. Hematol. 1996;24:1521–1529. [PubMed] [Google Scholar]
- 157.Aguila HL, et al. Osteoblast-specific overexpression of human interleukin-7 rescues the bone mass phenotype of interleukin-7-deficient female mice. J. Bone Miner. Res. 2012;27:1030–1042. doi: 10.1002/jbmr.1553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Weitzmann MN, Roggia C, Toraldo G, Weitzmann L, Pacifici R. Increased production of IL-7 uncouples bone formation from bone resorption during estrogen deficiency. J. Clin. Invest. 2002;110:1643–1650. doi: 10.1172/JCI15687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Rodriguiz RM, Key LL, Jr, Ries WL. Combination macrophage-colony stimulating factor and interferon-γ administration ameliorates the osteopetrotic condition in microphthalmic (mi/mi) mice. Pediatr. Res. 1993;33:384–389. doi: 10.1203/00006450-199304000-00014. [DOI] [PubMed] [Google Scholar]
- 160.Alam I, et al. Interferon γ, but not calcitriol improves the osteopetrotic phenotypes in ADO2 mice. J. Bone Miner. Res. 2015;30:2005–2013. doi: 10.1002/jbmr.2545. [DOI] [PubMed] [Google Scholar]
- 161.Key LL, Jr, et al. Long-term treatment of osteopetrosis with recombinant human interferon γ. N. Engl. J. Med. 1995;332:1594–1599. doi: 10.1056/NEJM199506153322402. [DOI] [PubMed] [Google Scholar]
- 162.Yamaza T, et al. Pharmacologic stem cell based intervention as a new approach to osteoporosis treatment in rodents. PLoS ONE. 2008;3:e2615. doi: 10.1371/journal.pone.0002615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Sass DA, et al. The role of the T-lymphocyte in estrogen deficiency osteopenia. J. Bone Miner. Res. 1997;12:479–486. doi: 10.1359/jbmr.1997.12.3.479. [DOI] [PubMed] [Google Scholar]
- 164.Lee SK, et al. T Lymphocyte deficient mice lose trabecular bone mass with ovariectomy. J. Bone Miner. Res. 2006;21:1704–1712. doi: 10.1359/jbmr.060726. [DOI] [PubMed] [Google Scholar]
- 165.Cho SW, et al. Human adipose tissue-derived stromal cell therapy prevents bone loss in ovariectomized nude mouse. Tissue Eng. Part A. 2012;18:1067–1078. doi: 10.1089/ten.TEA.2011.0355. [DOI] [PubMed] [Google Scholar]
- 166.D’Amelio P, et al. Estrogen deficiency increases osteoclastogenesis up-regulating T cells activity: a key mechanism in osteoporosis. Bone. 2008;43:92–100. doi: 10.1016/j.bone.2008.02.017. [DOI] [PubMed] [Google Scholar]
- 167.Adeel S, et al. Bone loss in surgically ovariectomized premenopausal women is associated with T lymphocyte activation and thymic hypertrophy. J. Investig. Med. 2013;61:1178–1183. doi: 10.231/JIM.0000000000000016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Kimble RB, et al. Simultaneous block of interleukin-1 and tumor necrosis factor is required to completely prevent bone loss in the early postovariectomy period. Endocrinology. 1995;136:3054–3061. doi: 10.1210/endo.136.7.7789332. [DOI] [PubMed] [Google Scholar]
- 169.Gao Y, et al. IFN-γ stimulates osteoclast formation and bone loss in vivo via antigen-driven T cell activation. J. Clin. Invest. 2007;117:122–132. doi: 10.1172/JCI30074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Lee SK, et al. Interleukin-7 influences osteoclast function in vivo but is not a critical factor in ovariectomy-induced bone loss. J. Bone Miner. Res. 2006;21:695–702. doi: 10.1359/jbmr.060117. [DOI] [PubMed] [Google Scholar]
- 171.Goswami J, Hernandez-Santos N, Zuniga LA, Gaffen SL. A bone-protective role for IL-17 receptor signaling in ovariectomy-induced bone loss. Eur. J. Immunol. 2009;39:2831–2839. doi: 10.1002/eji.200939670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Cummings SR, et al. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N. Engl. J. Med. 2009;361:756–765. doi: 10.1056/NEJMoa0809493. [DOI] [PubMed] [Google Scholar]
- 173.Pacifici R. T cells: critical bone regulators in health and disease. Bone. 2010;47:461–471. doi: 10.1016/j.bone.2010.04.611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Tawfeek H, et al. Disruption of PTH receptor 1 in T cells protects against PTH-induced bone loss. PLoS ONE. 2010;5:e12290. doi: 10.1371/journal.pone.0012290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Hory BG, et al. Absence of response to human parathyroid hormone in athymic mice grafted with human parathyroid adenoma, hyperplasia or parathyroid cells maintained in culture. J. Endocrinol. Invest. 2000;23:273–279. doi: 10.1007/BF03343723. [DOI] [PubMed] [Google Scholar]
- 176.Gao Y, et al. T cells potentiate PTH-induced cortical bone loss through CD40L signaling. Cell Metab. 2008;8:132–145. doi: 10.1016/j.cmet.2008.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Bedi B, et al. Inhibition of antigen presentation and T cell costimulation blocks PTH-induced bone loss. Ann. NY Acad. Sci. 2010;1192:215–221. doi: 10.1111/j.1749-6632.2009.05216.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Buchwald ZS, et al. A bone anabolic effect of RANKL in a murine model of osteoporosis mediated through FoxP3+ CD8 T-Cells. J. Bone Miner. Res. 2015;30:1508–1522. doi: 10.1002/jbmr.2472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Sayegh MH. Finally, CTLA4Ig graduates to the clinic. J. Clin. Invest. 1999;103:1223–1225. doi: 10.1172/JCI6952. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Pacifici R. Role of T cells in the modulation of PTH action: physiological and clinical significance. Endocrine. 2013;44:576–582. doi: 10.1007/s12020-013-9960-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Li Y, et al. Endogenous TNFα lowers maximum peak bone mass and inhibits osteoblastic Smad activation through NF-κB. J. Bone Miner. Res. 2007;22:646–655. doi: 10.1359/jbmr.070121. [DOI] [PubMed] [Google Scholar]
- 182.Gilbert LC, Chen H, Lu X, Nanes MS. Chronic low dose tumor necrosis factor-α (TNF) suppresses early bone accrual in young mice by inhibiting osteoblasts without affecting osteoclasts. Bone. 2013;56:174–183. doi: 10.1016/j.bone.2013.06.002. [DOI] [PubMed] [Google Scholar]