Summary
Many Siglecs function as inhibitory receptors on innate and adaptive immune cells and may contribute to the attenuation of immune responses to tumors. Siglec 9 on neutrophils and Siglec 7 on NK cells are prominent examples of inhibitory Siglecs that can potentially dampen anti-tumor immunity. CD169 is a Siglec that may function as an adhesion molecule and a facilitator of the recognition and internalization of sialic acid decorated apoptotic bodies and exosomes derived from tumors. It can potentially contribute to both the attenuation as well as the facilitation of anti-tumor immunity. Siglecs have been best studied in the tumor context in animal models of cancer. Modulators of Siglec function are likely to be developed and investigated clinically in a cancer context over the next few years.
Keywords: cancer/tumor immunology, hypersialylation, immune evasion, immune regulation, inhibitory receptor, sialic acid, Siglec
1 | INTRODUCTION
The diversity found within carbohydrates is immense when one considers the number of ways different monosaccharides can be linked together and form branched structures within complex glycans, the attachment of various functional groups to sugar carbon atoms, alterations in stereochemistry, and postsynthetic chemical modifications on available hydroxyl groups. Complementing this diversity is the fact that carbohydrates can be recognized by cells through a variety of receptors that mediate events integral to immune responses, including pathogen recognition, cell adhesion and migration, and the regulation of immune cell signaling. Sialic acid, a monosaccharide that decorates glycans found on vertebrate cells, is of interest to immunologists for a number of reasons, largely related to cell migration and cell signaling. In this review, we will describe the recognition of sialic acid containing ligands by unique Sialic acid-binding immunoglobulin-type lectins (Siglecs) found on the surfaces of both innate and adaptive immune cells. In this way, sialic acid is involved in discrimination between self and non-self and in the regulation of immune cells. However, pathogens and tumor cells have evolved to evade the immune system by increasing their surface expression of sialic acids in order to engage inhibitory Siglecs or to evade complement. In this review, we will specifically discuss the nuanced interactions between cancer cells and distinct Siglecs and their effect on immune cell regulation.
Sialic acids represent a structurally diverse family of nine-carbon sugars consisting of a pyranose ring bound to a carboxylic group and a three-carbon exocyclic side chain (Figure 1). The carboxylic carbon is numbered C-1 while the ring includes C-2 through C-6, and the side chain includes C-7 to C-9. These bulky, electronegatively charged monosaccharides are derived from a neuraminic acid (Neu) backbone that can be N-acetylated at C-5 in the cytosol to form N-acetyl neuraminic acid (Neu5Ac), the metabolic precursor to sialic acid. Biosynthesized Neu5Ac or sialic acid that has been recycled from the lysosome must first enter the nucleus where it is converted to cytidine monosphophate-sialic acid (CMP-Sia) by a nuclear enzyme, CMP-Sia synthetase. After returning to the cytosol, cytidine monophospho-Neu5Ac hydroxylase (CMAH) can subsequently convert CMP-Neu5Ac into CMP-N-glycolyl neuraminic acid (CMP-Neu5Gc), another CMP-Sia, with the addition of a hydroxyl group1,2 However, this irreversible hydroxyl addition does not occur in humans because of a 92 base pair deletion in exon 6 of CMAH.3
FIGURE 1.

The chemical structure of neuraminic acid, Neu5Ac, and Neu5Gc. Based on figure 1 in Schauer (2009). This figure details the various sites at which chemical alterations can be made to sialic acid that are relevant to recognition by Siglecs
With the help of an active transporter, CMP-Sia enters the Golgi compartment where various sialyltransferases can transfer the sialic acid moiety to the termini of glycoconjugates, including N-linked or O-linked glycan chains on proteins and glycolipids. The anomeric form of sialic acid covalently attached to glycans is the α anomer, in which the carboxylic acid of the sialic acid is depicted as being oriented upward. In contrast most soluble sialic acid exists as the β anomer. Thus, the covalent linkages formed between the C-2 hydroxyl group of a sialic acid and the C-3 or C-6 positions of the preterminal galactose or N-acetylgalactosamine, are referred to as an α2-3 or α2-6 linkage respectively. If a second sialic acid is added to the glycan chain, the C-2 hydroxyl group of the terminal sialic acid is linked to the C-8 hydroxyl group of the preterminal sialic acid in an α2-8 linkage.4 In addition to either an N-acetyl or N-glycolyl group at C-5, sialic acids are diverse in regards to modifications to their hydroxyl groups at carbon positions 4, 7, 8, or 9. For example, N-acetyl-9-O-acetylneuraminic acid (Neu5Ac9Ac) is the most frequent postsynthetic change in humans.1
Siglecs are cell surface transmembrane receptors that contain 2–17 extracellular Ig domains. Sialic acid binds to the N-terminal Ig V-set domain and typically induces a conformational change within the nine β-strands that encompass the domain. Some N-terminal C-2 domains that follow the V-set domain may also contribute to ligand binding. Siglec are classified as I-type lectins because the lectin domain has an Ig-fold structure. Many of the lectins of importance in the immune system are calcium-dependent C-type lectins. Unlike C-type lectins, Siglecs are not pattern recognition receptors in that they are not involved in the production of a proinflammatory or antiviral response. Most Siglecs bind sialic acid ligands either in-cis (on the same cell) or in-trans (on a neighboring cell or on a microbe) and provide inhibitory signals to immune cells. The cytoplamic portion of most Siglecs contains immunoreceptor tyrosine-based inhibitory motifs (ITIMs), which provide inhibitory signals by recruiting the tyrosine phosphatases SHP1 and SHP2. Some Siglecs, such as Siglec 14, 15, and 16, associate with the DAP12 adaptor that contains cytosolic immunotyrosine based activating motifs (ITAMs). These receptor complexes typically activate cells by recruiting Syk. Activating Siglecs are frequently paired with inhibitory counterparts. Siglec-5 is an inhibitory Siglec that has similar ligand binding properties to Siglec-14, an activating Siglec. They are both generally expressed on the same cell types. Similarly Siglec-11 is an inhibitory Siglec that is paired with Siglec-16, its activating counterpart.
The Siglec family was established following the discovery of sialoadhesin, CD22, CD33 and MAG/Siglec-4. They are now separated into distinct groups. Group 1 consists of Siglecs like sialoadhesin, CD22, MAG, and Siglec-15 that are conserved across mammals. Group 2 consists of CD33-related Siglecs that vary from species to species. Humans express more CD33-related Siglecs than rodents do, due to the loss of Siglec genes in rodents.5
Considering the abundance of Siglec ligands on the surface of tumor cells and the inhibitory nature of many Siglecs, it may be of interest in the context of cancer immunotherapy to consider altering the levels of sialylation on tumor cells or to target Siglecs using blocking antibodies in addition to the use of established checkpoint blockade modalities such as anti-PD-1 and anti-CTLA4.
2 | MODULATION OF THE INNATE IMMUNE SYSTEM BY TUMOR HYPERSIALYLATION
Like pathogens that have evolved to evade immune surveillance by decorating their surface with sialic acid glycoconjugates that can inhibit immune cells that express cognate inhibitory Siglecs, there is evidence that tumor cell hypersialylation may modulate immune responses although the outcome of interactions between Siglec receptors and their ligands may depend on cellular context and microenvironment. In fact, there is an association between levels of tumor glycosylation and metastatic potential.6 Tumor cells display altered glycosylation patterns compared to non-malignant cells as a result of increased expression of β-galactoside α2,6-sialyltransferase 1 (ST6Gal-1) that adds α-2,6 sialic acid to the termini of N-glycans and of α-N-acetyltransferase 1 (ST6GalNAc-I). ST6Gal-I is overexpressed in malignant cells of colon, breast, and ovarian cancers downstream of Ras oncogene signaling. In addition to changes in expression of individual sialyltransferases, surface glycans on some human cancer cells contain higher levels of Neu5Gc, a sialic acid that is not synthesized by humans.7 However, dietary sources of Neu5Gc can be metabolized and displayed on epithelial cells.8,9 Malignant epithelial cells also express membrane-bound and secreted mucins, which predominantly contain O-linked glycans with altered glycosylation patterns. Finally, while levels of 9-O-acetyl sialic acid have been shown to be downregulated in some cancers, other tumors, such as colorectal cancers, melanomas, and acute lymphoblastic leukemia (ALL) cells express high levels of 9-O-acetylated sialic acid residues. In particular, disialyl-ganglioside GD3 on melanomas displays terminal 9-O-acetylated sialic acid. Among other functions, these changes in tumor cell glycosylation play a role in mediating interactions between Siglec receptors and tumor cells; sialic acid expression on tumor cells can serve to inhibit innate immune cell responses within the context of anti-tumor immunity. The role of Siglecs in the cancer environment is likely to be complex since Siglecs may also attenuate tumorigenesis by contributing to the inhibition of tumor-promoting inflammation.7,10,11
Siglec-7 (p75/AIRM1) and Siglec-9 are members of the CD33-related Siglec family. The amino acid sequence of Siglec-9 is about 80% identical to the Siglec-7 sequence suggesting a gene duplication event during the evolution of these two Siglecs; they are localized on chromosome 19 with other CD33-related Siglecs.12–14 However, they exhibit distinct expression profiles among immune cell populations.15 Siglec-7 has been found on granulocytes and CD56+CD38+CD16+CD45RA NK cells.13,16 Siglec-9 is expressed on granulocytes, B cells, CD56+ NK cells, and weakly on minor subsets of CD8+ and CD4+ T cells.14 Siglec-7 and -9 are both expressed on bone marrow precursor cells, monocytes, and monocyte-derived macrophages while they are not detected on monocyte-derived dendritic cells.17,18 Expression of Siglec-7 and -9 may vary between mucosal immune cells and cells in human peripheral blood.19 The functionally equivalent ortholog of Siglec-9 in mice is Siglec-E, whose expression is limited to granulocytes and mononuclear phagocytes. Siglec-E exhibits a preference for α2,8-linked disialic acid over α2,3- and α2,6-linked sialic acids.20
The extracellular portions of Siglec-7 and Siglec-9 contain three Ig-like domains while the cytoplasmic tail has one tyrosine residue within an ITIM and another tyrosine residue in a motif that is similar to one found in the SH2-domain containing SLAM-associated protein (SAP) binding site on the signaling lymphocyte activating molecule (SLAM), an immunomodulatory receptor.12–14,16 Thus, the membrane-proximal ITIM provides Siglec-7 and -9 expressing cells with the capacity for inhibitory signaling.21 In fact, one group discovered Siglec-7 while investigating the inhibition of NK cell cytotoxicity in an in vitro cell killing assay.13 However, after tyrosine phosphorylation of the ITIM, Siglec-7 recruits the SH2 domain-containing SHP-1 and SHP-2 tyrosine phophatases less efficiently than Siglec-9; this is because of a structurally different ITIM sequence.22,23 Cross-linking of Siglec-9 in neutrophils in an inflammatory environment has been shown to induce both apoptotic and caspase -independent cell death of the autologous cells secondary to the generation of reactive oxygen species (ROS).24 In immunoprecipitation analyses, binding of DAP proteins to Siglec-7 is not detected perhaps due to the lack of basic amino acids in its transmembrane domain; these residues are typically present in other Siglecs that recruit adaptors of the DAP family.25 Aside from being an inhibitory receptor, Siglec-9 also possesses endocytic capabilities.26
Both Siglec-7 and Siglec-9 recognize α2,3- and α2,6-linked sialic acids in humans.14–16 Siglec-7 preferentially binds to α2-6-linked sialic acids, but the underlying glycan structure of the ligand may also affect recognition.12 Unlike Siglec-9, Siglec-7 has a preference for α2, 8-disialic acid that is found on the GD3 ganglioside and contains a unique sugar-binding domain with a preference for branched α2,6-sialylated glycans.27–30 These findings may be of interest in the field of cancer immunotherapy considering the high levels of expression of cell surface and secreted GD3 in some tumors, such as melanomas and renal cell carcinoma.31,32 Siglec-9 also prefers an underlying Galβ1-4GlcNAc structure that is typically found in N-linked glycans. However, Siglec-9 can nevertheless still recognize other glycan structures, such as those found in O-linked glycans and glycosphingolipids.14
3 | SIGLECS AND THE REGULATION OF IMMUNE RESPONSES MEDIATED BY MYELOID CELLS
Human carcinoma cells, both cell lines or tumors examined histologically, express Siglec-9 ligands that may either be in the form of α2,3-or α2,6-sialic acids as discussed in the previous section. Furthermore, an immune cell infiltrate containing Siglec-9 expressing cells has been observed in histological sections of human cancer samples but these cells are absent in normal tissue. In an in vitro co-culture assay, interactions of sialic acid ligands on tumor cells with Siglec-9 on neutrophils inhibited neutrophil activation as measured by ROS production (Figure 2). However, in the presence of a Siglec-9 blocking antibody, a neutrophil effector response was restored. Analysis in vivo showed that neutrophils in Siglec-E deficient mice mount an anti-tumor response to intravenously injected carcinoma cells while transgenic expression of Siglec-9 in these mice reverses this phenotype. Although subcutaneous injection of 3-methylcholanthrene (MCA), a carcinogen, into Siglec-E deficient mice resulted in enhanced immune surveillance, once tumors were established, an immune cell infiltrate consisting of tumor-promoting M2 polarized macrophages supported tumor growth. Siglec-9 was shown to inhibit M2 macrophage polarization in tumor co-cultures.33 Thus, before considering the use of Siglec blockade in immunotherapy, the differing functions of a Siglec based on its expression on multiple cell types must be studied further, and the kinetics of innate immune cell responses in the tumor setting deserve further investigation.
FIGURE 2.

Interaction of sialic acid on tumor cells with Siglec-9 on neutrophils may result in the inhibition of signaling and reduced ROS production
Many cancers, such as prostrate and colorectal cancer, can upregulate the extracellular matrix expression or secretion of lectin galactoside-binding soluble 3 binding protein (LGALS3BP or Mac-2 binding protein). This heavily N-glycosylated protein contains sialic acid ligands for human CD33-related Siglecs, such as Siglec-9, and has a weak affinity for murine Siglec-E.34 These tumor proteins were shown to inhibit neutrophil-mediated tumor killing in co-culture assays measuring extracellular ROS formation while the addition of a blocking antibody induces higher cytotoxicity of target cells. Alternatively, LGALS3BP downregulation can increase tumor cell apoptosis due to loss of Siglec-9 regulation on neutrophil activity.34
In the same fashion as Siglec-9-mediated regulation of immune responses, the cross-linking of Siglec-7 leads to the inhibition of myelomonocytic cells—an effect that may be of interest in the context of the attenuation of chronic myeloid leukemia cell survival and proliferation.35
Besides regulation of immune cells within the tumor infiltrate, Siglec ligands on cancer cells can function as counter-receptors and generate activation signals to these malignant cells that may enhance cancer progression and metastasis. Siglec-9 binding carbohydrates are expressed on various membrane proteins on astrocytomas and are also secreted by these tumor cells. Siglec-9 on immune cells can ligate these sialic containing counter-receptors on cancer cells and deliver activating signals. Moreover, the transfection of Siglec-9 into a monocytic cell line and subsequent co-culture with an astrocytoma cell line results in the degradation of focal adhesion kinase (FAK), which is a regulator of cell migration and adhesion, along with the degradation of Akt and other signaling molecules. These astrocytoma cells that were exposed to either monocytes expressing high levels of Siglec-9 or soluble Siglec-9 showed increased motility compared to cells co-cultured with low levels of Siglec-9 suggesting that Siglec-9 might provide activation signals in trans to cancer cells in addition to mediating inhibitory signals in cis in immune cells.36
Similarly, the binding of Siglec-9 to the MUC1 mucin expressed by tumor cells, including human colon, pancreas, and breast tumor tissues, may activate cancer signaling pathways involved in tumor progression.37 Along with positive histological staining for MUC1 in human tumor tissue, a Siglec-9 positive immune cell infiltrate is observed in malignant tissue and is absent in non-malignant tissue. Accordingly, MUC1 expression on cancer cells is often associated with poor prognosis in patients. MUC1 contains sialylated O-glycans that bind to Siglec-9. The interaction of Siglec-9 with MUC1 leads to the recruitment of β-catenin, an activator of cell cycle progression, to the C-terminal domain of the mucin and prevents the phosphorylation of β-catenin that marks it for proteosomal degradation. Thus, Siglec-9 interactions promote the stability of β-catenin and contribute to malignancy.37 MUC1 on tumor cells leads to the immunoregulation of Siglec-9-expressing monocyte-derived dendritic that lose their ability to function as antigen presenting cells as shown by the loss of costimulatory, antigen presentation molecules, and markers of a mature phenotype.38,39 DCs cultured with MUC1 display markers associated with immature cells and express higher levels of the anti-inflammatory cytokine IL-10 coupled with an inability to produce IL-12.39 Moreover, in macrophages, there is evidence for a role for Siglec-9 in upregulating IL-10 production and conversely suppressing TNF-α production in the context of contemporaneous TLR stimulation.40
Although Siglec-7 has been previously characterized as an inhibitory receptor, there is evidence to suggest that it has activating qualities solely in human monocytes. After exposing PBMC to a cross-linking monoclonal antibody directed against Siglec-7, the monocyte population was specifically activated and intracellularly expressed proinflammatory cytokines and chemokines while the blocking of Siglec-7 on these cells led to a decrease in the inflammatory response. The activation of the monocyte population following Siglec-7 engagement may be due to the activation of ERK, a MAP kinase involved in the regulation of pro-inflammatory gene transcription.41 These experiments suggest that Siglec-7 on monocytes may promote inflammation in addition to potentially inhibiting monocyte activation.
4 | SIGLECS AND SIGNALING IN NK CELLS
NK cells, a class of innate lymphoid cells, are important in the elimination of tumor cells through the production of pro-inflammatory cytokines, such as IFNγ and TNF, and by their specialized cytolytic functions. When NK cells are depleted or their activity is impaired in mice, tumors grow and metastasize at a greater rate. In humans, deficiencies in NK cells are associated with higher rates of cancer.42 In fact, NK cell numbers are considered a positive prognostic in cancer patients.
Activation of NK cells occurs through the recognition of a number of poorly defined ligands including cell stress markers, through FcgRIII activation when an antibody coated target is recognized, and generally occurs in the context of “missing-self” based on reduced or altered expression of MHC class I on the target cell. After activation, the NK cell releases perforin and granzymes that lead to the apoptosis of the target cell. In addition, NK cells can secrete IFNγ, TNF, GM-CSF, and MIP-1α which contribute to a proinflammatory state.43 Inhibitory receptors on NK cells create a balance of activating and inhibitory signals. NK cells use various inhibitory receptors, such as the human killer-cell immunoglobulin-like receptors (KIRs) and leukocyte immunoglobulin-like receptor 1 (LIR1) in humans and the murine Ly49 C-type-lectin-like receptor family to monitor the expression of MHC class I molecules on potential targets. These inhibitory receptors contain cytosolic ITIM tyrosines that can be phosphorylated and recruit protein tyrosine phosphatases. However, NK cells also utilize MHC class I-independent inhibitory receptors that are capable of recognizing ligands found on normal cells. Here, we will discuss the roles of Siglec-7 and Siglec-9 in humans and Siglec-E in mice as MHC class I-independent inhibitory receptors on NK cells.
There are two major sub-populations of NK cells in humans: CD56dimCD16+ cells and CD56brightCD16− cells. The CD56dimCD16+ NK cells are found in peripheral blood and spleen, express perforins and granzymes and are considered more mature, while the CD56brightCD16+ NK cells are found in lymph node and tonsil, secrete various cytokines, and have limited cytotoxic capabilities. While Siglec-7 is highly expressed on all NK cells, Siglec-9 is selectively expressed on CD56dim NK cells, which may have differentiated from CD56bright NK cells; this subset of Siglec-9+CD56dim NK cells display markers of a mature phenotype and exhibit reduced cytotoxic function in the presence of ligand-expressing tumor cells in vitro and in vivo. Although the removal of sialic acid ligands restored NK cell cytotoxicity and cytokine production in culture,44 in contrast NK cells treated with sialidase exhibit inhibited killing of target cells transfected with ganglioside GD3 synthase and this impaired killing is dependent on Siglec-7 engagement.27 In addition to GD3, Siglec-7 on NK cells can also bind to the disialylated ganglioside DSGb5 expressed on renal cell carcinoma cells45,46 (Figure 3). Just as in the studies investigating interactions of NK cells with GD3, sialidase treatment of NK cells was necessary to remove masking cis interactions with endogenous ligands before in vitro cytotoxicity assays could be performed. In these assays, NK cells exhibited reduced cytotoxicity against renal cell carcinoma cells expressing high levels of DSGb5 implying that Siglec dependent impaired activity of NK cells may play a role in this cancer’s metastasis.46
FIGURE 3.
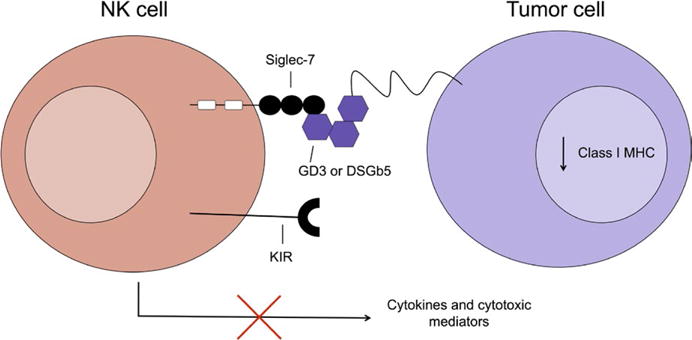
Interaction of sialic acid ligands on tumors cells and Siglec-7 on NK cells may dampen NK cell activation in the tumor context
Similar to MUC1 interactions with neutrophils, MUC16 found on epithelial ovarian cancer cells can provide the tumor with immune protection. Soluble MUC16 is capable of binding to subsets of CD56dim NK cells, B cells, and monocytes via Siglec-9. MUC16 glycans contain α2,3-linked sialic acid, which serves as the ligand for Siglec-9. As a result of interactions with cell surface MUC16, Siglec-9 promotes tumor cell and immune cell adhesion events that may benefit the tumor through immune regulation. Perhaps, soluble MUC16 even suppresses an immune response prior to immune cells having direct contact with tumor cells.47 Cell membrane expressed MUC16 can prevent the formation of an immunological synapse between NK cells and cancer cells, a necessary event for the cytotoxic function of NK cells, and soluble MUC16 caused the downregulation of the activating Fcγ receptor, CD16, on NK cells.48,49 Although Siglec-9 was not studied in regards to synapse formation with MUC16+ tumor cells or CD16 expression, the inhibitory molecule likely participates in the suppression of NK cell anti-tumor immunity.
Using a glycocalyx engineering approach whereby synthetic sialylated glycopolymers can be incorporated into cell membranes, Hudak et al. showed that cell killing by NK cells in vitro was inhibited in the presence of sialylated glycopolymers that was dependent on their density on target cells. The presence of blocking antibodies against Siglec-7 reduced this inhibitory effect. In co-culture experiments with target cells lacking sialylated polymers, minimal tyrosine phosphorylation occurred. In contrast there was a dramatic increase in phosphorylation levels and increased SHP-1 recruitment in NK cells cultured with sialylated target cells. Finally, this group also showed that there was increased NK cell killing of various carcinoma cell lines that were treated with sialidase. The ability of cancer cells to evade “natural” NK cell mediated killing was restored with the addition of the synthetic sialylated glycopolymers. Furthermore, tumor cell lines coated with sialylated glycopolymers were provided with increased protection against antibody-dependent cellular cytotoxicity when cultured with therapeutic monoclonal antibodies. In these studies a hypersialylated Burkitt’s lymphoma B cell line, Daudi, was incubated with a humanized anti-CD22 antibody and primary NK cells, and hypersialylated SK-BR-3 breast adenocarcinoma cells and NCI-N87 gastrric carcinoma cells were incubated with commercial Herceptin (a monoclonal antibody against HER2/Neu) and NK cells. Hypersialylation reduced tumor killing suggesting a role for Siglec-7 in the modulation of the NK cell response to administered therapy.50
Unmasking of Siglec-7 and -9, which may be bound to ligands on the NK cell itself, may be necessary for these receptors to inhibit NK cell function. This unmasking may occur due to stimuli found in the tumor microenvironment that reduce the expression of the sialic acid ligands on the NK cells themselves. Further research into the expression of cis ligands on NK cells themselves as well as the nature of their regulation of Siglec receptors needs to be undertaken.51 However, some experiments have shown that with any native cis ligand interactions, ligand expression on tumor cells still elicited an inhibition of NK cell cytotoxicity.44,50 Additionally, altered NK cell function due to MHC class I binding inhibitory receptors versus Siglecs needs to be investigated further especially considering CD56dim NK cells express KIRs as well as Siglec-9.52 Siglecs may play a more prominent role in cancers wherein MHC class I expression is downregulated. In such cases, the tumor cells may utilize Siglecs to evade anti-tumor immune responses.
5 | SIGLECS AND THE CAPTURE OF TUMOR-DERIVED VESICLES
CD169, also known as Siglec-1 and sialoadhesin, was originally called sheep erythrocyte receptor.53 It strongly recognizes NeuAcα2-3Gal on N- and O-linked glycans and glycolipids.54 CD169 binds to sialic acid with low affinity, therefore CD169 ligands must be heavily sialylated and multimeric to overcome this low binding affinity and bind with a functionally relevant avidity. Sialoadhesin/Siglec-1 will primarily be referred to as CD169 in this review. CD169 is a macrophage adhesion molecule primarily expressed on marginal metallophillic macrophages in the marginal zone of the spleen and on subcapsular sinus and medullary macrophages of lymph nodes. In addition, bone marrow resident macrophages in mice express CD169 while lower levels of this Siglec are found on liver, lung, and intestinal macrophages. The receptor is not found on monocytes.55 CD169 is highly conserved between human and mouse.56
Both human and mouse CD169 contain 17 Ig-like extracellular domains, as a result of which the extracellular portion of this molecule is substantially longer than the extracellular domains of other Siglecs.56,57 The length of the extracellular domain may allow CD169 to avoid cis sialic acid interactions.58 The short cytoplasmic tail lacks tyrosine-based motifs. Therefore, CD169 probably does not alter the activation state of its resident cell. Instead, CD169 functions in cell-cell interactions and binds to sialylated pathogens and structures. CD169 undergoes clathrin-mediated endocytosis whereby the receptor can transport sialic acid-bound molecules into the cell.59 Considering the short length of the CD169 cytoplasmic tail, an adaptor molecule probably exists to assist in receptor-mediated endocytosis. Although CD169 is not a phagocytic receptor, it may interact with other macrophage surface molecules that achieve phagocytosis, and it was shown in one study that wildtype bone marrow-derived macrophages show enhanced phagocytosis of sialylated bacteria compared to cells from CD169-deficient mice.60 CD169 can also mediate the binding of CD169+ macrophages from mouse lymph node or spleen to lymphocytes. In particular, purified CD169 bound more strongly to activated T cells than naïve T cells and strongest to a TK-1 lymphoma cell line suggesting that higher levels or increased availability of CD169 ligands may be a feature of some tumor cells.61
Like Siglec-9, CD169 is capable of binding to MUC1 expressed on breast cancer cell lines as shown by in vitro binding assays.62 A high density of CD169+ macrophages in the regional lymph nodes of colorectal and endometrial carcinoma as well as malignant melanoma patients is associated with a better clinical prognosis that is perhaps due to an increase in the amount of cytotoxic immune cells that have infiltrated the tumor.63,64 In patients with endometrial carcinoma, a high CD169+ cell density in the regional lymph node of the tumor is correlated with higher CD8+ T cell and NK cell counts in the tumor tissue.63–65 The number of intra-tumoral CD169 expressing cells also positively correlates with the number of CD8+ T cells present in the tumor and also with patient survival.66 Conversely, the subcapsular sinus macrophages in regional lymph nodes of patients that exhibit advanced-stage endometrial carcinoma or metastatic carcinoma express lower levels of CD169 or are completely negative for this marker.64
Tumor-infiltrating macrophages have been shown to express lower levels of CD169 compared to non-tumor tissues from the same patients. The reason for this downregulation may be the production of TGF-β and anti-inflammatory cytokines by the tumor or by the macrophages themselves in response to their interactions with the tumor. Macrophage exposure to TGF-β, specifically, has been shown to induce the downregulation of CD169 in a dose-dependent manner.66 CD169 expression can be upregulated in monocyte-derived macrophages after stimulation in culture with type I and type II interferons or lipopolysaccharide.63,65,67,68 Primary monocytes and THP-1 cells, a monocytic cell line, also express CD169 upon interferon exposure.69 In regional lymph nodes, CD169+ macrophages in the marginal zone localize near CD169-cells that produce IFNα.65
CD169-deficient mice display a minimal phenotype characterized by a small increase in CD8+ T cells and a decrease in B cells in the secondary lymphoid organs.70 Contradictorily, ST3GalI-deficient mice, which lack the ligand for CD169, exhibit decreased numbers of CD8+ T cells due to apoptotic loss in the spleen.71 CD4+ T cell populations remained unaltered perhaps due to high levels of 9-O acetylation of sialic acid, which renders this terminal sugar incapable of being recognized by CD169. These mice also exhibit impaired B cell signaling.70 When CD4+Foxp3+ T cells are activated in vitro, they readily express ligands for CD169 and at higher levels than Foxp3− effector CD4 T cells suggesting there is a possibility that these regulatory CD4+ T cells might interact with CD169+ macrophages. The CD4+ effector T cells that do express CD169 ligands are highly activated cells. This ligand-expressing behavior is also replicated in vivo in a model of murine lupus.72 The implications of CD169 ligand expression on regulatory T cells and some effector CD4+ T cells are unknown. However, bone marrow-derived macrophages that have been induced by IFNα to express CD169 can endocytose OVA antigen packaged in sialylated liposomes and subsequently drive the proliferation of antigen-specific CD4+ T cells in vitro. Less CD4+ T cell proliferation occurs when helper T cells are co-cultured with these macrophages and free OVA or OVA packaged in naked liposomes.73 Counter-receptors to CD169 exist on T lymphocytes. These include various glycoforms of the sialomucin CD43 and P-selectin glycoprotein ligand 1 (CD162).74 Adhesion of purified CD169 to T cells varies depending on activation state, with CD169 binding to activated T cells with a higher affinity than T cells that were not stimulated in culture.61 CD169 binding has also been shown to be involved in the receptor-mediated endocytosis of lipid antigens that results in presentation on CD1d to invariant NKT cells.75
It has recently been shown that CD169 on macrophages in the draining lymph nodes of virally infected mice can bind to gangliosides on the lipid membranes of cell-free murine leukemia virus (MLV) and human immunodeficiency virus (HIV) providing a method of viral capture. After CD169-dependent binding, the macrophage can then hold MLV particles in vesicle-like compartments attached to the cell membrane. Whether CD169 is mechanistically involved in viral internalization is unknown. In this process, however, MLV seems to hijack the cell until it can be delivered to permissive lymphocytes, specifically B-1 cells, for subsequent infection via the formation of a synapse involving the generation of a complex membranous protrusion by the macrophage.76 Other studies have also observed CD169-mediated trans-infection of HIV-1 and MLV.67,68,77 The capacity of CD169 to initiate this unique method of viral capture, transport, and trans-infection raises the possibility that the Siglec may be involved in the capture of other pathogens and immunogenic molecules coated by sialic acids. For example, CD169+ bone marrow derived macrophages can bind and assist in the phagocytosis of Campylobacter jejuni that display GD1a- and GM1-like structures. TNF-α and IFN-β are produced by the macrophages in response.78
Tumor cells have the ability to secrete extracellular vesicles (EVs) that purposefully or inadvertently communicate with immune cells although it is not known whether tumor cells secrete more EVs than non-tumor cells.79 EVs include exosomes and other types of vesicles that are unique based on the method of their generation and secretion.80 In addition to disposing of unwanted cellular material and creating a pro-metastatic environment through the transfer of oncogenic proteins and nucleic acids to healthy cells, vesicles secreted by tumors may interact with immune cells through endocytosis, phagocytosis, or membrane fusion that allows for tumor protein degradation and antigen presentation.79,80 Larger EVs are probably phagocytosed while small vesicles may be internalized by other methods like receptor-mediated endocytosis.79 However, the nature of the interaction between tumor-derived EVs and immune cells is not completely understood because EVs carrying tumor peptides to dendritic cells could, instead of being inhibitory, actually induce an antitumor immune response.79
B cell-derived exosomes that express α2,3-linked sialic acids co-localize with CD169+ macrophages in vivo and directly bind to CD169 in vitro. Furthermore, exosomes that were intravenously injected into CD169−/− mice aberrantly distributed into the outer marginal zone sinus and red pulp of the spleen from the circulation, and exosomes that traveled through the lymphatic system entered deeper into the lymph node cortex of CD169 deficient mice while the barrier functions of the subcaspsular sinus and marginal zone macrophages were not impaired. These studies suggest that CD169 is important for the capture of exosomes and possibly of other EVs that are also decorated with sialic acid containing ligands for this Siglec. Although there is no difference in CD4+ and CD8+ T cell proliferation in wildtype versus CD169−/− mice in response to protein-or peptide-pulsed exosomes, CD169 may be directly involved in inhibiting the immune response to antigen-containing exosomes or the promotion of cancer because this Siglec captures exosomes that contain tumor antigens and prevents them from accessing specific lymphocyte clones.81 Further studies are necessary to explore whether CD169-dependent exosome capture contributes in some circumstances to enhanced cytotoxic T cell anti-tumor responses.
In addition to exosomes, apoptotic vesicles derived from primary B cells, EL4 lymphoma cells, and B16 melanoma cells all express α2,3-linked sialic acids.82 EL4 lymphoma-derived apoptotic vesicles as well as B cell and B16 melanoma derived apoptotic vesicles prominently co-localized with CD169 in murine spleen sections in vitro, and EL4-derived apoptotic vesicles co-localized with CD169+ macrophages to a greater extent than outer marginal zone and red pulp macrophages in vivo. Furthermore, spleen sections from CD169 deficient mice did not exhibit any interaction with EL4 apoptotic vesicles.82 Apoptotic vesicles provide a debris clearance mechanism while also providing an important source of foreign and self-antigens, including tumor antigens. After injection of apoptotic vesicles derived from antigen-pulsed tumor cells, the mouse immune system is capable of mounting an enhanced cytotoxic response to antigen-pulsed target cells. This response is further enhanced in CD169−/− mice suggesting a role for CD169 in the capture of apoptotic vesicles and attenuation of the cytotoxic immune response.82
Recently, in vivo mouse experiments have provided evidence for the importance of CD169+ macrophages in the subcapsular sinus of tumor-draining lymph nodes in preventing the spread of melanoma tumor-derived EVs deeper into the lymph node and limiting the development of vesicle-B cell interactions that lead to tumor-promoting autoantibody production.83 Auto-antibodies in the tumor context are often against mutant or overexpressed oncogenes and tumor suppressors84,85 and may also be against “passenger” neo-antigens in tumors. The penetration of tumor-derived vesicles in the lymph node in the absence of CD169+ subcapsular sinus cells is similar to the phenotype seen in CD169 deficient mice that have intact sub-capsular sinus macrophages.81 Tumor-derived vesicles from melanomas travel in a cell-independent manner through the lymphatics and into the draining lymph node where they predominantly interact with CD169+ subcapsular sinus macrophages. Although the vesicles may bind to CD169+ macrophages, they do not fuse with the CD169+ cells as shown by the absence of horizontal gene or protein transfer, suggesting that these subcapsular sinus macrophages may act as physical barriers or filters.83 However, this does not preclude the possibility of endocytosis or phagocytosis of the tumor-derived vesicles. In fact, it was shown previously that some CD169+ macrophages in the lymph node can phagocytose dead tumor cells based on a flow assay while dendritic cells were incapable of accomplishing this function.86 Given these findings, CD169+ subcapsular sinus macrophages may modulate anti-tumor immunity by creating a physical barrier preventing B cell responses that facilitate tumorigenesis.
Dead tumor cells that arise during tumor growth or anti-tumor therapy are capable of generating an anti-tumor immune response. In experiments where dead tumor cells were subcutaneously injected into mice, these cells traveled through the lymphatics to the draining lymph node where they could be phagocytosed by CD169+ macrophages utilizing a mechanism that is partly dependent on phophatidylserine recognition on the surface of the apoptotic cells.86 There may be other cellular interactions involved in apoptotic tumor cell uptake, and interactions with tumor-derived vesicles may engage the immune system differently. Ultimately, this process of subcapsular sinus macrophage phagocytosis of tumor cells may allow these cells to cross-present tumor antigen to CD8+ T cells in the draining lymph nodes and induce anti-tumor immunity that is impaired in mice lacking CD169+ macrophages.86
Although CD169 is often used as a marker for the subcapsular sinus macrophage population, the cell surface molecule itself could be involved in tumor-derived vesicle capture considering the abundance of CD169 ligands on EVs.
These recent studies provide support for CD169 as a mediator of EV capture and it may also be thus implicated in the dampening of cytotoxic T cell responses to antigen or the prevention of autoantibody production by preventing exosomes from contacting B cells in the lymph node as described by Pucci et al.83 In this latter study examining the fate of B16-F10 melanoma tumor cells, tumor-derived EVs induced antibodies that promoted tumorigenesis, but this tumor-promoting activity could be abrogated by the CD169+ subcapsular sinus macrophages that captured antigens before they activated B cells in draining lymph nodes.83 Cytotoxic T cell responses are a key component of antitumor immunity, and CD169+ macrophages could suppress the induction of this response by capturing antigen-containing vesicles in the lymph node presumably before they can be captured by lymph node resident DCs for cross-presentation in the T cell zone to CD8+ T cells (Figure 4).
FIGURE 4.

CD169 on subcapsular sinus macrophages prevents tumor derived exosomes decorated with sialic acid from accessing B cells that make pro-tumorigenic antibodies and possibly from accessing naive CD8+ T cells as well
However, in the context of the tumor microenvironment, CD169 may also be positively involved in inducing a cytotoxic T cell response. CD169+ monocyte-derived macrophages induce greater granzyme B, IFN-γ, and IL-2 production in co-cultured CD8+ T cells compared to co-cultures with CD169− cells. Despite the possible presence of other confounding variables that may influence these cellular interactions, the addition of recombinant human CD169 resulted in similar findings. In addition, the application of a CD169-blocking antibody in the culture system resulted in a significant decrease in the production of CD8+ T cell associated effector molecules.66 While the number of CD169+ macrophages is lower in intra-tumoral tissue than in non-tumoral tissue probably due to the influence of the tumor microenvironment, the presence of a CD169+ tumor infiltrate population is associated with a positive clinical outcome. The presence of a CD169+ macrophage infiltrate in tumors was shown to enhance CD8+ T cell proliferation, cytotoxicity, and cytokine production dependent on the interaction of CD169 with the CD43 sialomucin on T lymphocytes that serves as a ligand for this Siglec. CD169 induced signaling in T cells may contribute to CD8+ T cell responses in a co-stimulatory manner and this possibility deserves further investigation.
6 | TUNING B CELL RECEPTOR SIGNALING WITHIN THE TUMOR ENVIRONMENT
CD22, a B-cell specific Siglec (Siglec-2) found in humans and mice, is expressed in the cytoplasm of progenitor B cells and pre-B cells and its surface expression coincides with the appearance of IgD on the cell surface, but the surface expression of this Siglec is lost upon plasma cell differentiation.87 This Siglec possesses an extracellular domain containing seven Ig-like domains and a cytoplasmic tail that contains six tyrosine residues.88,89 Three of these tyrosine residues are found within immunoreceptor tyrosine-based inhibitory motifs (ITIMs), which contain an (I/L/V/S)XYXX(L/V) intracytoplasmic domain. Thus, along with FcyRIIb, CD22 can inhibit B cell receptor (BCR) signaling through the recruitment of inhibitory signaling proteins to its cytoplasmic tail.90,91 Upon BCR cross-linking, CD22 associates with the BCR and is tyrosine phosphorylated.92–94 The protein tyrosine phosphatase SHP-1 then binds to the receptor’s phosphorylated ITIM through its two Src homology 2 (SH2) domains and becomes activated.92,95–97 Thus, CD22 can attenuate BCR signal transduction events by recruiting and activating SHP-1.95 Indeed, the loss of this receptor in CD22 deficient mice results in elevated intracellular free calcium levels and increased proliferation in response to BCR cross-linking as well as an expanded peritoneal B-1 cell population.87,98–101 The phosphorylation of CD22 tyrosines also leads to the recruitment of the adapter Grb2.102 Paradoxically, however, the tyrosine kinase Syk and phospholipase C-γ1 (Plcg1) can also dock onto phosphorylated tyrosine residues to form a complex with CD22 suggesting a counterbalance to the SHP-1 mediated signaling might exist.97 In addition, the src-related non-receptor tyrosine kinase Lyn is required for the tyrosine phosphorylation of CD22 and SHP-1 recruitment and suppression of BCR signaling.103–105
The N-terminus of CD22 specifically interacts with N-linked oligosaccharide structures that terminate in accessible α2-6 linked sialic acids.106–110 CD22 binds to a number of potential ligands that include CD45, membrane IgM and CD22 itself. 9-O-acetylation of sialic acids on CD22 ligands, expression of β-galactoside α2,6-sialyltransferase (ST6Gal I) by the B cell itself, or sialylated cis ligands have been shown to mask CD22 ligands.107,111–114 B cell lymphoma cells express high amounts of cell surface CD22 ligands that can abrogate the lectin function of CD22.112
CD22 undergoes constitutive internalization or endocytosis and degradation in late endosomes or lysosomes with no apparent recycling back to the cell surface regardless of BCR cross-linking.115 This lectin can also potentially mediate adhesion events involving erythrocytes, T lymphocytes, B lymphocytes, neutrophils, and monocytes.88,116,117 A study has also implicated CD22 as a homing receptor specifically in the movement of mature B cells into the bone marrow because of decreased numbers of bone marrow B cells in CD22 deficient mice and the presence of CD22 ligand found on sinusoidal endothelial cells of the bone marrow specifically.118 B cells within the bone marrow contains about a two to five times greater number of unmasked CD22 compared to lymph node or splenic B cells.119
In addition to increased proliferation, B cells from CD22 deficient mice exhibit heightened autoantibody production that may result from the absence of the CD22 receptor’s inhibitory signals during antigen recognition by the BCR.98,120 Although young CD22 deficient mice only exhibit increased IgM production possibly due to the B1 population expansion, as the mice age, they produce somatically mutated and high affinity autoreactive IgG.120 This is interesting to note in regards to work characterizing autoantibodies as a major factor in driving the pro-tumoral response.121–123 Immune complexes that are against intracellular tumor associated antigens presumably do not prime immune responses against tumors but instead might facilitate the differentiation of myeloid cells in the tumor milieu into myeloid-derived suppressor cells, thus promoting tumorigenesis.121,124
In the context of B cell malignancies, such as ALL, CD22 has become a therapeutic target. The most promising therapy involves the use of an anti-CD22 monoclonal antibody conjugated to a cytotoxic antibiotic agent that is internalized by the receptor on cancer cells and causes apoptosis of the cell. In a recent phase 3 clinical trial, patients receiving anti-CD22 antibody treatment underwent higher rates of complete remission than those receiving standard chemotherapy.125,126
7 | SIGLECS AND T CELL ACTIVATION
Not only do Siglecs provide inhibitory signals to their autologous cells, but they may also instruct the inhibition of adaptive immunity through the induction of regulatory T cells that in turn may dampen T cell responses to tumor cells. Although most T cells do not express Siglecs, T cell glycosylation can change throughout T cell development as well as after activation providing various levels of Siglec ligands depending on the cell state, and different subsets of T cells display distinct glycosylation patterns.127 It was recently shown that recognition of sialylated antigen by mouse dendritic cells created a suppressive environment. When ovalbumin was modified to include α2,3 or α2,6-linked sialic acid and then used to pulse splenic CD11c+ DCs or bone marrow derived dendritic cells that were subsequently co-cultured with naïve CD4+ T cells, there was an increase in FOXP3+CD4+ T reg cell differentiation that was 2–5 fold greater compared to control OVA pulsed cells. In addition, the adoptive transfer of sialylated peptide-pulsed dendritic cells caused a dampened effector T cell response in vivo and the induction of tolerance. The sialylation of antigen enhanced its uptake by dendritic cells. Indeed, Siglec-E was a major player in inducing this type of immune response. Siglec E-deficient dendritic cells in mice did not endocytose the sialylated antigen to the extent of Siglec E-sufficient cells, and they did not attenuate effector T cell function.128
Conversely, after activation, murine CD4+ and CD8+ T cells have been shown to upregulate the expression of Siglec-E ligands compared to ligands for other Siglecs. Early after activation, murine T cells shift from expressing N-acetyl-neuraminic acid (Neu-5Ac) to predominantly expressing N-glycolyl-neuraminic acid (Neu-5Gc) on N-glycans due to elevated levels of CMP-NeuAc hydroxylase, the enzyme involved in this glycan conversion. This is particularly relevant since Siglec-E displays a preference for Neu-5Gc over Neu-5Ac.129 Although the reasons for the glycan remodeling and perhaps selection for Siglec-E ligands are unknown, these events may promote immune regulation whereby Siglec-E on antigen presenting cells (Siglec-E is expressed primarily on dendritic cells and macrophages) can directly interact with sialic acid ligands on already activated T cells to promote inhibition of further stimulation or alter other signaling processes. Perhaps the Siglec-E positive cells could dampen effector responses and lead to an increase in Treg cells after interactions with activated T cells expressing NeuGc as described above. The interaction between Siglec-E and ligands on T cells may also affect cell trafficking.
Plasmacytoid dendritic cells (pDCs) are part of the initial innate immune defense against viral infections and promote antiviral immunity through the production of type I IFN. Siglec-H, expressed exclusively on pDCs, was shown to modulate the function of these cells in vivo in mice Specifically, Siglec-H deficient mice exhibit enhanced NFκB and IKK-α activity as well as increased cytokine production, providing evidence that Siglec-H may perhaps function as an inhibitory receptor in pDCs. However, Siglec-H lacks ITIM motifs, and is transported to the cell surface along with the ITAM-containing DAP12 adapter. Its inhibitory functions are therefore likely not to be direct. Siglec-H deficiency caused a decrease in expression of MHC class I and II and the B7 costimulatory molecules on pDCs leading to an inhibited capacity of these cells to activate antigen-specific CD8+ T cells through cross-presentation as well as an attenuated ability to activate antigen-specific CD4+ T cells.130 Siglec H may therefore functionally represent an activating receptor required for pDC function in the context of tumor immunity.
Although rare, a subset of human T cells express Siglec-10, which has been shown to act as an inhibitory receptor when it binds soluble CD52, which contains a sialylated N-glycan. CD52 is also expressed on a subset of CD4+ T cells, CD52hiCD4+ T cells. Siglec-10 probably binds any molecule with the appropriate sialic acid containing ligand. Upon binding to CD52, Siglec-10 inhibited the phosphorylation of Lck and Zap70 tyrosine residues that are normally required for appropriate TCR signaling.131 Siglecs-7 and/or -9 are also expressed on about 2%–18% of CD4+ and CD8+ T cells in the peripheral blood.15,16,23 When Jurkat cells are transfected with Siglec-7 and Siglec-9 and subjected to pervanadate treatment or TCR engagement, these ITIM-harboring inhibitory receptors co-localized with the T cell receptor. Siglec-7 and Siglec-9 both contributed to the recruitment of SHP-1, which mediates the dephosphorylation of the tyrosine phosphorylated CD3 complex and zeta ITAM tyrosines and results in reduced NFAT-mediated transcriptional activation.23
A subset of T cells in the periphery express the GD3 ganglioside and anti-GD3 monoclonal antibodies have the ability to cause the proliferation of these cells as well as expansion of the GD3+ T cell population.132 Purified CD169 protein is capable of binding to TK-1 lymphoma T cells in a sialic acid-dependent manner. Specifically, one of the glycan ligands for CD169 in this context is CD43, a sialomucin on T cells, and this may facilitate T cell-macrophage interactions.74 Additionally, it has been demonstrated that sialoadhesin/CD169 (described above), which is expressed on macrophages in inflamed organs in autoimmune mice, negatively regulated the expansion of Tregs as inferred from studies in sialoadhesin-deficient mice.133
8 | CONCLUSIONS
Although T cells do not widely express sialic-acid binding receptors, other immune cells responsible for promoting or inhibiting tumor immunity express unique Siglecs or combinations of Siglecs that can alter their levels of activation or their adhesion properties. Tumor cells abundantly express sialic acid moieties on their cell surfaces providing ligands for Siglec-expressing immune cells within the tumor microenvironment. In addition, tumor cells release sialic acid coated apoptotic bodies during the natural course of the tumor or as a result of chemotherapy and other treatments. Living tumor cells can also release exosomes that contain sialic acid on their surfaces. Although Siglecs may have evolved to prevent autoimmunity, the Siglec class of lectins represents a possible target for tumor therapy or to generally modulate the immune response.
Many Siglecs, such as Siglec-9 on monocytes, neutrophils, and a subpopulation of NK cells, as well as Siglec-7 on NK cells, contain ITIM motifs in their cytosolic portions, and these Siglecs generally inhibit innate immunity to cancer cells. Indeed there are several studies showing a negative correlation between the presence of certain sialic acid moieties on tumor cells and a positive clinical outcome, and the addition of antibodies that block Siglecs in experimental tumor models improves immune effector responses. The same effect can also be achieved by the removal of sialic acids by sialidase. In the case of tumor cells that have downregulated class I MHC expression, many inhibitory receptors on NK cells that recognize class I MHC are no longer capable of effectively reducing immune activation and the release of cytotoxic molecules and cytokines. However, if the NK cell also expresses Siglec-7 and/or -9 then this may provide the sialic acid expressing tumor cell with an alternative strategy for NK cell inhibition. Hypersialylation on tumor cells may be an adaptation that evolved to not only induce inhibitory signals during innate immune cell activation, but sialic acid mediated ligation of Siglecs may also induce pro-survival signals in the tumor; positive roles for Siglecs in anti-tumor immunity, however, remain poorly explored.
The abundance of tumor-derived vesicles that can circulate through the lymphatics may allow Siglec expressing cells in secondary lymphoid organs to interact with tumors without actually being part of the immune cell infiltrate in the tumor. For example, macrophages that reside in the sub-capsular and medullary sinuses of the lymph node and in the marginal zone of the spleen express CD169. Although CD169 is typically used solely as a marker for these cells located at the interface between antigen accumulation and lymphocyte activation, this Siglec may bind to tumor-derived vesicles entering the draining lymph node and prevent the vesicles from interacting with tumor facilitating B cells located further within the follicle. CD169 may participate in the endocytosis of these vesicles and allow macrophages to either degrade the contents or enhance the capture, processing and presentation of the antigen on MHC class II molecules in order to enhance immune activation.
The CD22 inhibitory signaling pathway in B cells is very well-studied. However, the role of the humoral response in the production of pro-tumoral autoantibodies or generating an anti-tumor response requires further examination. In the case of B cell malignancies, therapeutic antibodies that target CD22 in a stimulatory fashion have been developed. The development of liposomes expressing CD22 ligands in proximity to protein antigen has also been used to target BCR signaling on certain autoreactive cells.101
Acknowledgments
This work was supported by grants AI064930 and AI110495 from the NIH and a grant from the Mizutani Foundation.
Footnotes
This article is part of a series of reviews covering Checkpoint Inhibitors and Cancer Immunotherapy appearing in Volume 276 of Immunological Reviews.
CONFLICT OF INTEREST
The authors have no conflicts of interest.
References
- 1.Schauer R. Sialic acids as regulators of molecular and cellular interactions. Curr Opin Struct Biol. 2009;19:507–514. doi: 10.1016/j.sbi.2009.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Varki A, Schauer R. Sialic Acids. In: Varki A, Cummings RD, Esko JD, Freeze HH, Stanley P, Bertozzi CR, Hart GR, Etzler ME, editors. Essentials of Glycobiology. Cold Spring Harbor, NY: Cold Spring Harbor Press; 2009. pp. 199–218. Chapter 14. [PubMed] [Google Scholar]
- 3.Varki A. Multiple changes in sialic acid biology during human evolution. Glycoconj J. 2009;26:231–245. doi: 10.1007/s10719-008-9183-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Pillai S, Netravali IA, Cariappa A, Mattoo H. Siglecs and immune regulation. Annu Rev Immunol. 2012;30:357–392. doi: 10.1146/annurev-immunol-020711-075018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Macauley MS, Crocker PR, Paulson JC. Siglec-mediated regulation of immune cell function in disease. Nat Rev Immunol. 2014;14:653–666. doi: 10.1038/nri3737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hakomori S. Tumor malignancy defined by aberrant glycosylation and sphingo(glyco)lipid metabolism. Cancer Res. 1996;56:5309–5318. [PubMed] [Google Scholar]
- 7.Pearce OMT, Läubli H. Sialic acids in cancer biology and immunity. Glycobiology. 2016;26:111–128. doi: 10.1093/glycob/cwv097. [DOI] [PubMed] [Google Scholar]
- 8.Tangvoranuntakul P, Gagneux P, Diaz S, et al. Human uptake and incorporation of an immunogenic nonhuman dietary sialic acid. Proc Natl Acad Sci USA. 2003;100:12045–12050. doi: 10.1073/pnas.2131556100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hedlund M, Padler-Karavani V, Varki NM, Varki A. Evidence for a human-specific mechanism for diet and antibody-mediated inflammation in carcinoma progression. Proc Natl Acad Sci USA. 2008;105:18936–18941. doi: 10.1073/pnas.0803943105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Varki A, Kannagi R, Toole BP. Glycosylation changes in cancer. In: Varki A, Cummings RD, Esko JD, Freeze HH, Stanley P, Bertozzi CR, Hart GR, Etzler ME, editors. Essentials of Glycobiology. Cold Spring Harbor, NY: Cold Spring Harbor Press; 2009. pp. 617–632. Chapter 44. [PubMed] [Google Scholar]
- 11.Swindall AF, Londoño-Joshi AI, Schultz MJ, Fineberg N, Buchsbaum DJ, Bellis SL. ST6Gal-I protein expression is upregulated in human epithelial tumors and correlates with stem cell markers in normal tissues and colon cancer cell lines. Cancer Res. 2013;73:2368–2378. doi: 10.1158/0008-5472.CAN-12-3424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Angata T, Varki A. Siglec-7: A sialic acid-binding lectin of the immunoglobulin superfamily. Glycobiology. 2000;10:431–438. doi: 10.1093/glycob/10.4.431. [DOI] [PubMed] [Google Scholar]
- 13.Falco M, Biassoni R, Bottino C, et al. Identification and molecular cloning of p75/AIRM1, a novel member of the sialoadhesin family that functions as an inhibitory receptor in human natural killer cells. J Exp Med. 1999;190:793–802. doi: 10.1084/jem.190.6.793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Angata T, Varki A. Cloning, characterization, and phylogenetic analysis of siglec-9, a new member of the CD33-related group of siglecs. Evidence for co-evolution with sialic acid synthesis pathways. J Biol Chem. 2000;275:22127–22135. doi: 10.1074/jbc.M002775200. [DOI] [PubMed] [Google Scholar]
- 15.Zhang JQ, Nicoll G, Jones C, Crocker PR. Siglec-9, a novel sialic acid binding member of the immunoglobulin superfamily expressed broadly on human blood leukocytes. J Biol Chem. 2000;275:22121–22126. doi: 10.1074/jbc.M002788200. [DOI] [PubMed] [Google Scholar]
- 16.Nicoll G, Ni J, Liu D, et al. Identification and characterization of a novel siglec, siglec-7, expressed by human natural killer cells and monocytes. J Biol Chem. 1999;274:34089–34095. doi: 10.1074/jbc.274.48.34089. [DOI] [PubMed] [Google Scholar]
- 17.Lock K, Zhang J, Lu J, Lee SH, Crocker PR. Expression of CD33-related siglecs on human mononuclear phagocytes, monocyte-derived dendritic cells and plasmacytoid dendritic cells. Immunobiology. 2004;209:199–207. doi: 10.1016/j.imbio.2004.04.007. [DOI] [PubMed] [Google Scholar]
- 18.Nguyen DH, Ball ED, Varki A. Myeloid precursors and acute myeloid leukemia cells express multiple CD33-related Siglecs. Exp Hematol. 2006;34:728–735. doi: 10.1016/j.exphem.2006.03.003. [DOI] [PubMed] [Google Scholar]
- 19.Miyazaki K, Sakuma K, Kawamura YI, et al. Colonic epithelial cells express specific ligands for mucosal macrophage immunosuppressive receptors siglec-7 and -9. J Immunol. 2012;188:4690–4700. doi: 10.4049/jimmunol.1100605. [DOI] [PubMed] [Google Scholar]
- 20.Zhang JQ, Biedermann B, Nitschke L, Crocker PR. The murine inhibitory receptor mSiglec-E is expressed broadly on cells of the innate immune system whereas mSiglec-F is restricted to eosinophils. Eur J Immunol. 2004;34:1175–1184. doi: 10.1002/eji.200324723. [DOI] [PubMed] [Google Scholar]
- 21.Avril T, Floyd H, Lopez F, Vivier E, Crocker PR. The membrane-proximal immunoreceptor tyrosine-based inhibitory motif is critical for the inhibitory signaling mediated by Siglecs-7 and -9, CD33-related Siglecs expressed on human monocytes and NK cells. J Immunol. 2004;173:6841–6849. doi: 10.4049/jimmunol.173.11.6841. [DOI] [PubMed] [Google Scholar]
- 22.Yamaji T, Mitsuki M, Teranishi T, Hashimoto Y. Characterization of inhibitory signaling motifs of the natural killer cell receptor Siglec-7: Attenuated recruitment of phosphatases by the receptor is attributed to two amino acids in the motifs. Glycobiology. 2005;15:667–676. doi: 10.1093/glycob/cwi048. [DOI] [PubMed] [Google Scholar]
- 23.Ikehara Y, Ikehara SK, Paulson JC. Negative regulation of T cell receptor signaling by Siglec-7 (p70/AIRM) and Siglec-9. J Biol Chem. 2004;279:43117–43125. doi: 10.1074/jbc.M403538200. [DOI] [PubMed] [Google Scholar]
- 24.von Gunten S, Yousefi S, Seitz M, et al. Siglec-9 transduces apoptotic and nonapoptotic death signals into neutrophils depending on the proinflammatory cytokine environment. Blood. 2005;106:1423–1431. doi: 10.1182/blood-2004-10-4112. [DOI] [PubMed] [Google Scholar]
- 25.Mitsuki M, Nara K, Yamaji T, et al. Siglec-7 mediates nonapoptotic cell death independently of its immunoreceptor tyrosine-based inhibitory motifs in monocytic cell line U937. Glycobiology. 2010;20:395–402. doi: 10.1093/glycob/cwp195. [DOI] [PubMed] [Google Scholar]
- 26.Biedermann B, Gil D, Bowen DT, Crocker PR. Analysis of the CD33-related siglec family reveals that Siglec-9 is an endocytic receptor expressed on subsets of acute myeloid leukemia cells and absent from normal hematopoietic progenitors. Leuk Res. 2007;31:211–220. doi: 10.1016/j.leukres.2006.05.026. [DOI] [PubMed] [Google Scholar]
- 27.Nicoll G, Avril T, Lock K, Furukawa K, Bovin N, Crocker PR. Ganglioside GD3 expression on target cells can modulate NK cell cytotoxicity via siglec-7-dependent and -independent mechanisms. Eur J Immunol. 2003;33:1642–1648. doi: 10.1002/eji.200323693. [DOI] [PubMed] [Google Scholar]
- 28.Ito A, Handa K, Withers DA, Satoh M, Hakomori S. Binding specificity of siglec7 to disialogangliosides of renal cell carcinoma: Possible role of disialogangliosides in tumor progression. FEBS Lett. 2001;498:116–120. doi: 10.1016/s0014-5793(01)02476-0. [DOI] [PubMed] [Google Scholar]
- 29.Miyazaki K, Ohmori K, Izawa M, et al. Loss of disialyl Lewis(a), the ligand for lymphocyte inhibitory receptor sialic acid-binding immunoglobulin-like lectin-7 (Siglec-7) associated with increased sialyl Lewis(a) expression on human colon cancers. Cancer Res. 2004;64:4498–4505. doi: 10.1158/0008-5472.CAN-03-3614. [DOI] [PubMed] [Google Scholar]
- 30.Yamaji T, Teranishi T, Alphey MS, Crocker PR, Hashimoto Y. A small region of the natural killer cell receptor, Siglec-7, is responsible for its preferred binding to alpha 2,8-disialyl and branched alpha 2,6-sialyl residues. A comparison with Siglec-9. J Biol Chem. 2002;277:6324–6332. doi: 10.1074/jbc.M110146200. [DOI] [PubMed] [Google Scholar]
- 31.Urmacher C, Cordon-Cardo C, Houghton AN. Tissue distribution of GD3 ganglioside detected by mouse monoclonal antibody R24. Am J Dermatopathol. 1989;11:577–581. doi: 10.1097/00000372-198912000-00013. [DOI] [PubMed] [Google Scholar]
- 32.Pukel CS, Lloyd KO, Travassos LR, Dippold WG, Oettgen HF, Old LJ. GD3, a prominent ganglioside of human melanoma. Detection and characterisation by mouse monoclonal antibody. J Exp Med. 1982;155:1133–1147. doi: 10.1084/jem.155.4.1133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Läubli H, Pearce OMT, Schwarz F, et al. Engagement of myelomonocytic Siglecs by tumor-associated ligands modulates the innate immune response to cancer. Proc Natl Acad Sci USA. 2014;111:14211–14216. doi: 10.1073/pnas.1409580111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Läubli H, Alisson-Silva F, Stanczak MA, et al. Lectin galactoside-binding soluble 3 binding protein (LGALS3BP) is a tumor-associated immunomodulatory ligand for CD33-related Siglecs. J Biol Chem. 2014;289:33481–33491. doi: 10.1074/jbc.M114.593129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Vitale C, Romagnani C, Falco M, et al. Engagement of p75/AIRM1 or CD33 inhibits the proliferation of normal or leukemic myeloid cells. Proc Natl Acad Sci USA. 1999;96:15091–15096. doi: 10.1073/pnas.96.26.15091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sabit I, Hashimoto N, Matsumoto Y, Yamaji T, Furukawa K, Furukawa K. Binding of a sialic acid-recognizing lectin Siglec-9 modulates adhesion dynamics of cancer cells via calpain-mediated protein degradation. J Biol Chem. 2013;288:35417–35427. doi: 10.1074/jbc.M113.513192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tanida S, Akita K, Ishida A, et al. Binding of the sialic acid-binding lectin, Siglec-9, to the membrane mucin, MUC1, induces recruitment of β-catenin and subsequent cell growth. J Biol Chem. 2013;288:31842–31852. doi: 10.1074/jbc.M113.471318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Ohta M, Ishida A, Toda M, et al. Immunomodulation of monocyte-derived dendritic cells through ligation of tumor-produced mucins to Siglec-9. Biochem Biophys Res Commun. 2010;402:663–669. doi: 10.1016/j.bbrc.2010.10.079. [DOI] [PubMed] [Google Scholar]
- 39.Rughetti A, Pellicciotta I, Biffoni M, et al. Recombinant tumor-associated MUC1 glycoprotein impairs the differentiation and function of dendritic cells. J Immunol. 2005;174:7764–7772. doi: 10.4049/jimmunol.174.12.7764. [DOI] [PubMed] [Google Scholar]
- 40.Ando M, Tu W, Nishijima K-I, Iijima S. Siglec-9 enhances IL-10 production in macrophages via tyrosine-based motifs. Biochem Biophys Res Commun. 2008;369:878–883. doi: 10.1016/j.bbrc.2008.02.111. [DOI] [PubMed] [Google Scholar]
- 41.Varchetta S, Brunetta E, Roberto A, et al. Engagement of Siglec-7 receptor induces a pro-inflammatory response selectively in monocytes. PLoS ONE. 2012;7:e45821. doi: 10.1371/journal.pone.0045821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Morvan MG, Lanier LL. NK cells and cancer: You can teach innate cells new tricks. Nat Rev Cancer. 2016;16:7–19. doi: 10.1038/nrc.2015.5. [DOI] [PubMed] [Google Scholar]
- 43.Waldhauer I, Steinle A. NK cells and cancer immunosurveillance. Oncogene. 2008;27:5932–5943. doi: 10.1038/onc.2008.267. [DOI] [PubMed] [Google Scholar]
- 44.Jandus C, Boligan KF, Chijioke O, et al. Interactions between Siglec-7/9 receptors and ligands influence NK cell-dependent tumor immunosurveillance. J Clin Invest. 2014;124:1810–1820. doi: 10.1172/JCI65899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Saito S, Orikasa S, Satoh M, Ohyama C, Ito A, Takahashi T. Expression of globo-series gangliosides in human renal cell carcinoma. Jpn J Cancer Res. 1997;88:652–659. doi: 10.1111/j.1349-7006.1997.tb00433.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Kawasaki Y, Ito A, Withers DA, et al. Ganglioside DSGb5, preferred ligand for Siglec-7, inhibits NK cell cytotoxicity against renal cell carcinoma cells. Glycobiology. 2010;20:1373–1379. doi: 10.1093/glycob/cwq116. [DOI] [PubMed] [Google Scholar]
- 47.Belisle JA, Horibata S, Jennifer GAA, et al. Identification of Siglec-9 as the receptor for MUC16 on human NK cells, B cells, and monocytes. Mol Cancer. 2010;9:118. doi: 10.1186/1476-4598-9-118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Gubbels JAA, Felder M, Horibata S, et al. MUC16 provides immune protection by inhibiting synapse formation between NK and ovarian tumor cells. Mol Cancer. 2010;9:11. doi: 10.1186/1476-4598-9-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Patankar MS, Jing Y, Morrison JC, et al. Potent suppression of natural killer cell response mediated by the ovarian tumor marker CA125. Gynecol Oncol. 2005;99:704–713. doi: 10.1016/j.ygyno.2005.07.030. [DOI] [PubMed] [Google Scholar]
- 50.Hudak JE, Canham SM, Bertozzi CR. Glycocalyx engineering reveals a Siglec-based mechanism for NK cell immunoevasion. Nat Chem Biol. 2014;10:69–75. doi: 10.1038/nchembio.1388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Avril T, North SJ, Haslam SM, Willison HJ, Crocker PR. Probing the cis interactions of the inhibitory receptor Siglec-7 with alpha2,8-disialylated ligands on natural killer cells and other leukocytes using glycan-specific antibodies and by analysis of alpha2,8-sialyltransferase gene expression. J Leukoc Biol. 2006;80:787–796. doi: 10.1189/jlb.1005559. [DOI] [PubMed] [Google Scholar]
- 52.Jacobs R, Hintzen G, Kemper A, et al. CD56bright cells differ in their KIR repertoire and cytotoxic features from CD56dim NK cells. Eur J Immunol. 2001;31:3121–3127. doi: 10.1002/1521-4141(2001010)31:10<3121::aid-immu3121>3.0.co;2-4. [DOI] [PubMed] [Google Scholar]
- 53.Crocker PR, Werb Z, Gordon S, Bainton DF. Ultrastructural localization of a macrophage-restricted sialic acid binding hemagglutinin, SER, in macrophage-hematopoietic cell clusters. Blood. 1990;76:1131–1138. [PubMed] [Google Scholar]
- 54.Crocker PR, Kelm S, Dubois C, et al. Purification and properties of sialoadhesin, a sialic acid-binding receptor of murine tissue macrophages. EMBO J. 1991;10:1661–1669. doi: 10.1002/j.1460-2075.1991.tb07689.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Crocker PR, Gordon S. Mouse macrophage hemagglutinin (sheep erythrocyte receptor) with specificity for sialylated glycoconjugates characterized by a monoclonal antibody. J Exp Med. 1989;169:1333–1346. doi: 10.1084/jem.169.4.1333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hartnell A, Steel J, Turley H, Jones M, Jackson DG, Crocker PR. Characterization of human sialoadhesin, a sialic acid binding receptor expressed by resident and inflammatory macrophage populations. Blood. 2001;97:288–296. doi: 10.1182/blood.v97.1.288. [DOI] [PubMed] [Google Scholar]
- 57.Crocker PR, Mucklow S, Bouckson V, et al. Sialoadhesin, a macrophage sialic acid binding receptor for haemopoietic cells with 17 immunoglobulin-like domains. EMBO J. 1994;13:4490–4503. doi: 10.1002/j.1460-2075.1994.tb06771.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Munday J, Floyd H, Crocker PR. Sialic acid binding receptors (siglecs) expressed by macrophages. J Leukoc Biol. 1999;66:705–711. doi: 10.1002/jlb.66.5.705. [DOI] [PubMed] [Google Scholar]
- 59.Delputte PL, Van Gorp H, Favoreel HW, et al. Porcine sialoadhesin (CD169/Siglec-1) is an endocytic receptor that allows targeted delivery of toxins and antigens to macrophages. PLoS ONE. 2011;6:e16827. doi: 10.1371/journal.pone.0016827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Jones C, Virji M, Crocker PR. Recognition of sialylated meningococcal lipopolysaccharide by siglecs expressed on myeloid cells leads to enhanced bacterial uptake. Mol Microbiol. 2003;49:1213–1225. doi: 10.1046/j.1365-2958.2003.03634.x. [DOI] [PubMed] [Google Scholar]
- 61.van den Berg TK, Brevé JJ, Damoiseaux JG, et al. Sialoadhesin on macrophages: Its identification as a lymphocyte adhesion molecule. J Exp Med. 1992;176:647–655. doi: 10.1084/jem.176.3.647. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Nath D, Hartnell A, Happerfield L, et al. Macrophage-tumour cell interactions: Identification of MUC1 on breast cancer cells as a potential counter-receptor for the macrophage-restricted receptor, sialoadhesin. Immunology. 1999;98:213–219. doi: 10.1046/j.1365-2567.1999.00827.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ohnishi K, Komohara Y, Saito Y, et al. CD169-positive macrophages in regional lymph nodes are associated with a favorable prognosis in patients with colorectal carcinoma. Cancer Sci. 2013;104:1237–1244. doi: 10.1111/cas.12212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ohnishi K, Yamaguchi M, Erdenebaatar C, et al. Prognostic significance of CD169-positive lymph node sinus macrophages in patients with endometrial carcinoma. Cancer Sci. 2016;107:846–852. doi: 10.1111/cas.12929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Saito Y, Ohnishi K, Miyashita A, et al. Prognostic significance of CD169+ lymph node sinus macrophages in patients with malignant melanoma. Cancer Immunol Res. 2015;3:1356–1363. doi: 10.1158/2326-6066.CIR-14-0180. [DOI] [PubMed] [Google Scholar]
- 66.Zhang Y, Li J-Q, Jiang Z-Z, Li L, Wu Y, Zheng L. CD169 identifies an anti-tumour macrophage subpopulation in human hepatocellular carcinoma. J Pathol. 2016;239:231–241. doi: 10.1002/path.4720. [DOI] [PubMed] [Google Scholar]
- 67.Pino M, Erkizia I, Benet S, et al. HIV-1 immune activation induces Siglec-1 expression and enhances viral trans-infection in blood and tissue myeloid cells. Retrovirology. 2015;12:37. doi: 10.1186/s12977-015-0160-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Erikson E, Wratil PR, Frank M, et al. Mouse Siglec-1 mediates trans-infection of surface-bound murine leukemia virus in a sialic acid N-Acyl side chain-dependent manner. J Biol Chem. 2015;290:27345–27359. doi: 10.1074/jbc.M115.681338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Rempel H, Calosing C, Sun B, Pulliam L. Sialoadhesin expressed on IFN-induced monocytes binds HIV-1 and enhances infectivity. PLoS ONE. 2008;3:e1967. doi: 10.1371/journal.pone.0001967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Oetke C, Vinson MC, Jones C, Crocker PR. Sialoadhesin-deficient mice exhibit subtle changes in B- and T-cell populations and reduced immunoglobulin M levels. Mol Cell Biol. 2006;26:1549–1557. doi: 10.1128/MCB.26.4.1549-1557.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Priatel JJ, Chui D, Hiraoka N, et al. The ST3Gal-I sialyltransferase controls CD8+ T lymphocyte homeostasis by modulating O-glycan biosynthesis. Immunity. 2000;12:273–283. doi: 10.1016/s1074-7613(00)80180-6. [DOI] [PubMed] [Google Scholar]
- 72.Kidder D, Richards HE, Ziltener HJ, Garden OA, Crocker PR. Sialoadhesin ligand expression identifies a subset of CD4+Foxp3− T cells with a distinct activation and glycosylation profile. J Immunol. 2013;190:2593–2602. doi: 10.4049/jimmunol.1201172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Chen WC, Kawasaki N, Nycholat CM, et al. Antigen delivery to macrophages using liposomal nanoparticles targeting sialoadhesin/CD169. PLoS ONE. 2012;7:e39039. doi: 10.1371/journal.pone.0039039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.van den Berg TK, Nath D, Ziltener HJ, et al. Cutting edge: CD43 functions as a T cell counterreceptor for the macrophage adhesion receptor sialoadhesin (Siglec-1) J Immunol. 2001;166:3637–3640. doi: 10.4049/jimmunol.166.6.3637. [DOI] [PubMed] [Google Scholar]
- 75.Kawasaki N, Vela JL, Nycholat CM, et al. Targeted delivery of lipid antigen to macrophages via the CD169/sialoadhesin endocytic pathway induces robust invariant natural killer T cell activation. Proc Natl Acad Sci USA. 2013;110:7826–7831. doi: 10.1073/pnas.1219888110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Sewald X, Ladinsky MS, Uchil PD, et al. Retroviruses use CD169-mediated trans-infection of permissive lymphocytes to establish infection. Science. 2015;350:563–567. doi: 10.1126/science.aab2749. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Jobe O, Trinh HV, Kim J, et al. Effect of cytokines on Siglec-1 and HIV-1 entry in monocyte-derived macrophages: The importance of HIV-1 envelope V1V2 region. J Leukoc Biol. 2016;99:1089–1106. doi: 10.1189/jlb.2A0815-361R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Klaas M, Crocker PR. Sialoadhesin in recognition of self and non-self. Semin Immunopathol. 2012;34:353–364. doi: 10.1007/s00281-012-0310-3. [DOI] [PubMed] [Google Scholar]
- 79.Colombo M, Raposo G, Théry C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu Rev Cell Dev Biol. 2014;30:255–289. doi: 10.1146/annurev-cellbio-101512-122326. [DOI] [PubMed] [Google Scholar]
- 80.Tkach M, Théry C. Communication by extracellular vesicles: Where we are and where we need to go. Cell. 2016;164:1226–1232. doi: 10.1016/j.cell.2016.01.043. [DOI] [PubMed] [Google Scholar]
- 81.Saunderson SC, Dunn AC, Crocker PR, McLellan AD. CD169 mediates the capture of exosomes in spleen and lymph node. Blood. 2014;123:208–216. doi: 10.1182/blood-2013-03-489732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Black LV, Saunderson SC, Coutinho FP, et al. The CD169 sialoadhesin molecule mediates cytotoxic T-cell responses to tumour apoptotic vesicles. Immunol Cell Biol. 2016;94:430–438. doi: 10.1038/icb.2015.111. [DOI] [PubMed] [Google Scholar]
- 83.Pucci F, Garris C, Lai CP, et al. SCS macrophages suppress melanoma by restricting tumor-derived vesicle-B cell interactions. Science. 2016;352:242–246. doi: 10.1126/science.aaf1328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Lu H, Goodell C, Disis ML. Humoral Immunity against tumor-associated antigens as potential biomarkers for the early diagnosis of cancer. J Proteome Res. 2008;7:1388–1394. doi: 10.1021/pr700818f. [DOI] [PubMed] [Google Scholar]
- 85.Scanlan MJ, Chen YT, Williamson B, et al. Characterization of human colon cancer antigens recognized by autologous antibodies. Int J Cancer. 1998;76:652–658. doi: 10.1002/(sici)1097-0215(19980529)76:5<652::aid-ijc7>3.0.co;2-p. [DOI] [PubMed] [Google Scholar]
- 86.Asano K, Nabeyama A, Miyake Y, et al. CD169-positive macrophages dominate antitumor immunity by crosspresenting dead cell-associated antigens. Immunity. 2011;34:85–95. doi: 10.1016/j.immuni.2010.12.011. [DOI] [PubMed] [Google Scholar]
- 87.Pezzutto A, Dörken B, Moldenhauer G, Clark EA. Amplification of human B cell activation by a monoclonal antibody to the B cell-specific antigen CD22, Bp 130/140. J Immunol. 1987;138:98–103. [PubMed] [Google Scholar]
- 88.Wilson GL, Fox CH, Fauci AS, Kehrl JH. cDNA cloning of the B cell membrane protein CD22: A mediator of B-B cell interactions. J Exp Med. 1991;173:137–146. doi: 10.1084/jem.173.1.137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Torres RM, Law CL, Santos-Argumedo L, et al. Identification and characterization of the murine homologue of CD22, a B lymphocyte-restricted adhesion molecule. J Immunol. 1992;149:2641–2649. [PubMed] [Google Scholar]
- 90.Muta T, Kurosaki T, Misulovin Z, Sanchez M, Nussenzweig MC, Ravetch JV. A 13-amino-acid motif in the cytoplasmic domain of Fc gamma RIIB modulates B-cell receptor signalling. Nature. 1994;369:340. doi: 10.1038/369340a0. [DOI] [PubMed] [Google Scholar]
- 91.D’Ambrosio D, Hippen KL, Minskoff SA, et al. Recruitment and activation of PTP1C in negative regulation of antigen receptor signaling by Fc gamma RIIB1. Science. 1995;268:293–297. doi: 10.1126/science.7716523. [DOI] [PubMed] [Google Scholar]
- 92.Schulte RJ, Campbell MA, Fischer WH, Sefton BM. Tyrosine phosphorylation of CD22 during B cell activation. Science. 1992;258:1001–1004. doi: 10.1126/science.1279802. [DOI] [PubMed] [Google Scholar]
- 93.Leprince C, Draves KE, Geahlen RL, Ledbetter JA, Clark EA. CD22 associates with the human surface IgM-B-cell antigen receptor complex. Proc Natl Acad Sci USA. 1993;90:3236–3240. doi: 10.1073/pnas.90.8.3236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Peaker CJ, Neuberger MS. Association of CD22 with the B cell antigen receptor. Eur J Immunol. 1993;23:1358–1363. doi: 10.1002/eji.1830230626. [DOI] [PubMed] [Google Scholar]
- 95.Doody GM, Justement LB, Delibrias CC, et al. A role in B cell activation for CD22 and the protein tyrosine phosphatase SHP. Science. 1995;269:242–244. doi: 10.1126/science.7618087. [DOI] [PubMed] [Google Scholar]
- 96.Campbell MA, Klinman NR. Phosphotyrosine-dependent association between CD22 and protein tyrosine phosphatase 1C. Eur J Immunol. 1995;25:1573–1579. doi: 10.1002/eji.1830250616. [DOI] [PubMed] [Google Scholar]
- 97.Law CL, Sidorenko SP, Chandran KA, et al. CD22 associates with protein tyrosine phosphatase 1C, Syk, and phospholipase C-gamma(1) upon B cell activation. J Exp Med. 1996;183:547–560. doi: 10.1084/jem.183.2.547. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.O’Keefe TL, Williams GT, Davies SL, Neuberger MS. Hyperresponsive B cells in CD22-deficient mice. Science. 1996;274:798–801. doi: 10.1126/science.274.5288.798. [DOI] [PubMed] [Google Scholar]
- 99.Sato S, Miller AS, Inaoki M, et al. CD22 is both a positive and negative regulator of B lymphocyte antigen receptor signal transduction: Altered signaling in CD22-deficient mice. Immunity. 1996;5:551–562. doi: 10.1016/s1074-7613(00)80270-8. [DOI] [PubMed] [Google Scholar]
- 100.Pezzutto A, Rabinovitch PS, Dörken B, Moldenhauer G, Clark EA. Role of the CD22 human B cell antigen in B cell triggering by anti-immunoglobulin. J Immunol. 1988;140:1791–1795. [PubMed] [Google Scholar]
- 101.Nitschke L, Carsetti R, Ocker B, Köhler G, Lamers MC. CD22 is a negative regulator of B-cell receptor signalling. Curr Biol. 1997;7:133–143. doi: 10.1016/s0960-9822(06)00057-1. [DOI] [PubMed] [Google Scholar]
- 102.Otipoby KL, Draves KE, Clark EA. CD22 regulates B cell receptor-mediated signals via two domains that independently recruit Grb2 and SHP-1. J Biol Chem. 2001;276:44315–44322. doi: 10.1074/jbc.M105446200. [DOI] [PubMed] [Google Scholar]
- 103.Smith KG, Tarlinton DM, Doody GM, Hibbs ML, Fearon DT. Inhibition of the B cell by CD22: A requirement for Lyn. J Exp Med. 1998;187:807–811. doi: 10.1084/jem.187.5.807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Chan VW, Lowell CA, DeFranco AL. Defective negative regulation of antigen receptor signaling in Lyn-deficient B lymphocytes. Curr Biol. 1998;8:545–553. doi: 10.1016/s0960-9822(98)70223-4. [DOI] [PubMed] [Google Scholar]
- 105.Cornall RJ, Cyster JG, Hibbs ML, et al. Polygenic autoimmune traits: Lyn, CD22, and SHP-1 are limiting elements of a biochemical pathway regulating BCR signaling and selection. Immunity. 1998;8:497–508. doi: 10.1016/s1074-7613(00)80554-3. [DOI] [PubMed] [Google Scholar]
- 106.Aruffo A, Kanner SB, Sgroi D, Ledbetter JA, Stamenkovic I. CD22-mediated stimulation of T cells regulates T-cell receptor/CD3-induced signaling. Proc Natl Acad Sci USA. 1992;89:10242–10246. doi: 10.1073/pnas.89.21.10242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Sjoberg ER, Powell LD, Klein A, Varki A. Natural ligands of the B cell adhesion molecule CD22 beta can be masked by 9-O-acetylation of sialic acids. J Cell Biol. 1994;126:549–562. doi: 10.1083/jcb.126.2.549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Sgroi D, Varki A, Braesch-Andersen S, Stamenkovic I. CD22, a B cell-specific immunoglobulin superfamily member, is a sialic acid-binding lectin. J Biol Chem. 1993;268:7011–7018. [PubMed] [Google Scholar]
- 109.Powell LD, Sgroi D, Sjoberg ER, Stamenkovic I, Varki A. Natural ligands of the B cell adhesion molecule CD22 beta carry N-linked oligosaccharides with alpha-2,6-linked sialic acids that are required for recognition. J Biol Chem. 1993;268:7019–7027. [PubMed] [Google Scholar]
- 110.van der Merwe PA, Crocker PR, Vinson M, Barclay AN, Schauer R, Kelm S. Localization of the putative sialic acid-binding site on the immunoglobulin superfamily cell-surface molecule CD22. J Biol Chem. 1996;271:9273–9280. [PubMed] [Google Scholar]
- 111.Sgroi D, Nocks A, Stamenkovic I. A single N-linked glycosylation site is implicated in the regulation of ligand recognition by the I-type lectins CD22 and CD33. J Biol Chem. 1996;271:18803–18809. doi: 10.1074/jbc.271.31.18803. [DOI] [PubMed] [Google Scholar]
- 112.Hanasaki K, Varki A, Powell LD. CD22-mediated cell adhesion to cytokine-activated human endothelial cells. Positive and negative regulation by alpha 2-6-sialylation of cellular glycoproteins. J Biol Chem. 1995;270:7533–7542. doi: 10.1074/jbc.270.13.7533. [DOI] [PubMed] [Google Scholar]
- 113.Braesch-Andersen S, Stamenkovic I. Sialylation of the B lymphocyte molecule CD22 by alpha 2,6-sialyltransferase is implicated in the regulation of CD22-mediated adhesion. J Biol Chem. 1994;269:11783–11786. [PubMed] [Google Scholar]
- 114.Tuscano J, Engel P, Tedder TF, Kehrl JH. Engagement of the adhesion receptor CD22 triggers a potent stimulatory signal for B cells and blocking CD22/CD22L interactions impairs T-cell proliferation. Blood. 1996;87:4723–4730. [PubMed] [Google Scholar]
- 115.Shan D, Press OW. Constitutive endocytosis and degradation of CD22 by human B cells. J Immunol. 1995;154:4466–4475. [PubMed] [Google Scholar]
- 116.Engel P, Nojima Y, Rothstein D, et al. The same epitope on CD22 of B lymphocytes mediates the adhesion of erythrocytes, T and B lymphocytes, neutrophils, and monocytes. J Immunol. 1993;150:4719–4732. [PubMed] [Google Scholar]
- 117.Stamenkovic I, Seed B. The B-cell antigen CD22 mediates monocyte and erythrocyte adhesion. Nature. 1990;345:74–77. doi: 10.1038/345074a0. [DOI] [PubMed] [Google Scholar]
- 118.Nitschke L, Floyd H, Ferguson DJ, Crocker PR. Identification of CD22 ligands on bone marrow sinusoidal endothelium implicated in CD22-dependent homing of recirculating B cells. J Exp Med. 1999;189:1513–1518. doi: 10.1084/jem.189.9.1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Floyd H, Nitschke L, Crocker PR. A novel subset of murine B cells that expresses unmasked forms of CD22 is enriched in the bone marrow: Implications for B-cell homing to the bone marrow. Immunology. 2000;101:342–347. doi: 10.1046/j.1365-2567.2000.00103.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.O’Keefe TL, Williams GT, Batista FD, Neuberger MS. Deficiency in CD22, a B cell-specific inhibitory receptor, is sufficient to predispose to development of high affinity autoantibodies. J Exp Med. 1999;189:1307–1313. doi: 10.1084/jem.189.8.1307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Andreu P, Johansson M, Affara NI, et al. FcRgamma activation regulates inflammation-associated squamous carcinogenesis. Cancer Cell. 2010;17:121–134. doi: 10.1016/j.ccr.2009.12.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.de Visser KE, Korets LV, Coussens LM. De novo carcinogenesis promoted by chronic inflammation is B lymphocyte dependent. Cancer Cell. 2005;7:411. doi: 10.1016/j.ccr.2005.04.014. [DOI] [PubMed] [Google Scholar]
- 123.Hogarth PM, Pietersz GA. Fc receptor-targeted therapies for the treatment of inflammation, cancer and beyond. Nat Rev Drug Discov. 2012;11:311–331. doi: 10.1038/nrd2909. [DOI] [PubMed] [Google Scholar]
- 124.Yuen GJ, Demissie E, Pillai S. B lymphocytes and Cancer: A love-hate relationship. Trends Cancer. 2016;2:747–757. doi: 10.1016/j.trecan.2016.10.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Jabbour E, O’Brien S, Ravandi F, Kantarjian H. Monoclonal antibodies in acute lymphoblastic leukemia. Blood. 2015;125:4010–4016. doi: 10.1182/blood-2014-08-596403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Kantarjian HM, DeAngelo DJ, Stelljes M, et al. Inotuzumab ozogamicin versus standard therapy for acute lymphoblastic leukemia. N Engl J Med. 2016;375:740–753. doi: 10.1056/NEJMoa1509277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Daniels MA, Hogquist KA, Jameson SC. Sweet “n” sour: The impact of differential glycosylation on T cell responses. Nat Immunol. 2002;3:903–910. doi: 10.1038/ni1002-903. [DOI] [PubMed] [Google Scholar]
- 128.Perdicchio M, Ilarregui JM, Verstege MI, et al. Sialic acid-modified antigens impose tolerance via inhibition of T-cell proliferation and de novo induction of regulatory T cells. Proc Natl Acad Sci USA. 2016;113:3329–3334. doi: 10.1073/pnas.1507706113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Redelinghuys P, Antonopoulos A, Liu Y, et al. Early murine T-lymphocyte activation is accompanied by a switch from N-Glycolyl-to N-acetyl-neuraminic acid and generation of ligands for siglec-E. J Biol Chem. 2011;286:34522–34532. doi: 10.1074/jbc.M111.243410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Takagi H, Fukaya T, Eizumi K, et al. Plasmacytoid dendritic cells are crucial for the initiation of inflammation and T cell immunity in vivo. Immunity. 2011;35:958–971. doi: 10.1016/j.immuni.2011.10.014. [DOI] [PubMed] [Google Scholar]
- 131.Bandala-Sanchez E, Zhang Y, Reinwald S, et al. T cell regulation mediated by interaction of soluble CD52 with the inhibitory receptor Siglec-10. Nat Immunol. 2013;14:741–748. doi: 10.1038/ni.2610. [DOI] [PubMed] [Google Scholar]
- 132.Welte K, Miller G, Chapman PB, et al. Stimulation of T lymphocyte proliferation by monoclonal antibodies against GD3 ganglioside. J Immunol. 1987;139:1763–1771. [PubMed] [Google Scholar]
- 133.Wu C, Rauch U, Korpos E, et al. Sialoadhesin-positive macrophages bind regulatory T cells, negatively controlling their expansion and autoimmune disease progression. J Immunol. 2009;182:6508–6516. doi: 10.4049/jimmunol.0804247. [DOI] [PMC free article] [PubMed] [Google Scholar]


