Abstract
OBJECTIVE
Improved blood pressure control and use of renin-angiotensin-aldosterone system blockers have altered the clinical presentation or phenotype of chronic kidney disease (CKD) in U.S. adults with diabetes. These changes may influence mortality.
RESEARCH DESIGN AND METHODS
Data from the National Health and Nutrition Examination Surveys (NHANES) 1988–2006 were used to examine mortality trends in adults with diabetes, defined as physician diagnosis, fasting glucose ≥126 mg/dL, HbA1c >6.5% (48 mmol/mol), or use of glucose-lowering medications. Mortality trends by CKD phenotype (estimated glomerular filtration rate [eGFR] and urine albumin-to-creatinine ratio [ACR] level) were obtained via linkage with the National Death Index through 31 December 2011 while accounting for the complex survey design.
RESULTS
From 1988 to 2006, adults with an eGFR <60 mL/min/1.73 m2 and an ACR <30 mg/g increased from ∼0.9 million (95% CI 0.7, 1.1) or 6.6% of the total population with diabetes during years 1988–1994 to 2.4 million (95% CI 1.9, 2.9) or 10.1% of the total population with diabetes during years 2007–2010. Mortality rates generally trended downward for adults with diabetes and an ACR ≥30 mg/g but increased in those with eGFR <60 mL/min/1.73 m2 and an ACR <30 mg/g from 35 deaths per 1,000 person-years (95% CI 22, 55) during years 1988–1994 to 51 deaths per 1,000 person-years (95% CI 33, 83) during years 2003–2006.
CONCLUSIONS
ACR values are decreasing in U.S. adults with diabetes, but optimal management strategies are needed to reduce mortality in those with a low eGFR and an ACR <30 mg/g.
Introduction
Diabetes now affects more than 10 million U.S. adults, and prevalence is expected to increase during the next two decades due to the ongoing obesity epidemic and the aging of the U.S. population (1). Diabetes increases the risk for cardiovascular disease and mortality and now accounts for almost 12% of all deaths in the U.S. adult population, making diabetes the third leading cause of death after heart disease and cancer (2). However, previous studies have shown that the excess mortality risk associated with diabetes is mainly noted in those with chronic kidney disease (CKD) (3–5), defined as an estimated glomerular filtration rate (eGFR) <60 mL/min/1.73 m2 and/or increased urine albumin excretion (random urine albumin-to-creatinine ratio [ACR] ≥30 mg/g). Thus, population interventions that address CKD prevention may help mitigate diabetes-associated mortality.
Historically, the progression of CKD attributed to diabetes was presumed to follow a linear path, with transitions from normal urine albumin excretion (ACR <30 mg/g) to moderately increased (ACR 30–300 mg/g), followed by severely increased urine albumin excretion (ACR >300 mg/g) based on studies conducted before the routine use of renin-angiotensin-aldosterone system (RAAS) inhibitors. Only after urine albumin excretion becomes severely increased would eGFR then decline (6,7). However, as medical management of diabetes improved during the past three decades with increased use of RAAS inhibitors and better blood pressure and glucose control, the development of CKD in the setting of diabetes appeared more heterogeneous (8). Concurrent with temporal changes in diabetes management, the frequency of increased urine albumin excretion has declined while low eGFR in the U.S. population with diabetes has increased and low GFR in the absence of increased urine albumin excretion has become more prevalent (9). These dynamics in CKD clinical presentations or phenotypes, as defined by the eGFR and ACR in the U.S. population, are important because rates of cardiovascular disease, end-stage renal disease (ESRD), and mortality differ by these CKD phenotypes (10–12).
Using data from the National Health and Nutrition Examination Surveys (NHANES), we examined temporal trends in the total deaths over 5 years by CKD phenotypes in the U.S. population with diabetes during years 1988–2006. We hypothesized that the proportion of deaths in the U.S. population with diabetes occurring in the setting of low GFR (eGFR <60 mL/min/1.73 m2) without increased urine albumin excretion (ACR <30 mg/g) has increased over time. Information on the distribution of mortality in the U.S. population with diabetes may help guide population interventions for reducing morbidity and mortality in populations facing a rapid increase in diabetes incidence.
Research Design and Methods
The NHANES are a multistage stratified probability sample of noninstitutionalized U.S. adults conducted by the National Center for Health Statistics to determine the health and nutritional status of the noninstitutionalized U.S. population. The NHANES were originally completed in 6-year periods, with the last 6-year survey completed during years 1988–1994. Starting in 1999, NHANES began completing surveys in 2-year cycles. The current study population consisted of adults aged 20 years or older who participated in NHANES conducted during years 1988–1994 (n = 18,825), 1999–2002 (n = 10,291), 2003–2006 (n = 10,020), and 2007–2010 (n = 12,153). The analysis was then limited to NHANES participants with diabetes, defined as a self-reported physician diagnosis, use of oral hypoglycemic medications or insulin, fasting plasma glucose ≥126 mg/dL, or HbA1c ≥6.5% (n = 2,284 for NHANES 1988–1994, n = 1,287 for 1999–2002, n = 1,341 for 2003–2006, and n = 2,039 for 2007–2010).
After individuals with missing data on serum creatinine, urine ACRs, and mortality status, and individuals with ESRD (use of dialysis within the past 12 months) were excluded, a total of 1,834, 1,067, 1,160, and 1,781 sampled persons with diabetes were included in the analyses of NHANES for years 1988–1994, 1999–2002, 2003–2006, and 2007–2010, respectively. The National Center for Health Statistics Institutional Review Board approved each NHANES cycle, and all participants provided written informed consent.
In each NHANES cycle, data were collected via a medical evaluation and participant interviews. The participant interview collected self-reported data on age, race/ethnicity, sex, smoking status, history of a diagnosis of diabetes, heart failure, hypertension, myocardial infarction, angina, coronary heart disease, or stroke, as well as receipt of dialysis in the past 12 months or the use of antihypertensive or glucose-lowering medications.
CKD Phenotypes
NHANES participants completed standardized study visits conducted by trained study personnel following standardized protocols (13). Blood and urine samples were collected, processed, and transported to central laboratories following standardized protocols. Serum creatinine was calibrated according to published recommendations (14). The Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation based on serum creatinine was used to calculate eGFR (15), and low eGFR was defined as <60 mL/min/1.73 m2. Urine albumin and creatinine were measured with a fluorescent immunoassay and Jaffe rate reaction, respectively. The eGFR (≥90, 60–89, and <60 mL/min/1.73 m2) and ACR (<30, 30–300, and >300 mg/g) values were used to define nine eGFR-ACR groups or CKD phenotypes (16).
All-Cause Mortality
The NHANES data are linked with the National Death Index using probabilistic matching based on 12 identifiers for each participant. NHANES data from 1988 to 2006 were linked with the National Death Index to determine 5-year mortality. Mortality follow-up is available through 31 December 2011. The follow-up period for each participant was calculated as the interval between their NHANES evaluation and the data of death or 31 December 2011 for participants who did not die. As a result of the limited follow-up period, mortality rates were not examined for the 2007–2010 NHANES population.
Statistical Analyses
We used the svy suite of commands in Stata 14 software (StataCorp, College Station, TX) to account for the complex probability sample design and followed the NHANES analytical guidelines (17). Hence, all reported results take survey weights into account. Mortality rates were calculated using Stata’s stptime function (using survey weights), which calculates person-time and incidence rates over groups. Population estimates of 5-year deaths in the U.S. population with diabetes and by CKD phenotype were determined by multiplying the observed mortality risk by the population estimate for that particular group. The proportion of deaths was calculated using the proportion function in Stata’s svy suite. Age-standardized cumulative incidence of mortality over a 5-year period was determined by standardizing the age distributions of each time period to the 2000 U.S. population. Cox proportional hazards models were used to model mortality rates by time period for the population with diabetes, eGFR <60 mL/min/1.73 m2, and ACR <30 mg/g. Unadjusted models and models adjusted for age, sex, and race (model 1), comorbid conditions (model 2), and use of RAAS inhibitors and statins (model 3) were examined with the time period 1988–1994 as the referent.
Results
The prevalence of diabetes in the U.S. population increased during the past three decades from 8.0% during years 1988–1994 to 11.8% during years 2007–2010 (47.5% relative increase). As a result of the growth of the U.S. population, the absolute number of U.S. adults with diabetes increased from 13.2 million during years 1988–1994 to 23.8 million during years 2007–2010 (80.3% relative increase). Table 1 summarizes the characteristics of the U.S. population with diabetes by time period. Overall, use of RAAS inhibitors and statin medications increased and systolic and diastolic blood pressures and serum LDL-cholesterol levels decreased over time, similar to findings in the general U.S. population. The mean age and waist circumference and the prevalence of hypertension, heart failure, and cancer have increased over time in the total U.S. population with diabetes (Table 1).
Table 1.
Characteristics of U.S. population with diabetes* by time period
| 1988–1994 population estimate = 13.2 million; | 1999–2002 population estimate = 16.0 million; | 2003–2006 population estimate = 19.7 million; | 2007–2010 population estimate = 23.8 million; | |
|---|---|---|---|---|
| sampled persons = 1,834 | sampled persons = 1,067 | sampled persons = 1,160 | sampled persons = 1,781 | |
| Age (years) | 58.0 ± 20.5 | 58.5 ± 13.5 | 58.5 ± 12.7 | 59.2 ± 13.7 |
| Male# (%) | 46.9 | 53.0 | 49.5 | 53.0 |
| African American race (%) | 15.8 | 14.4 | 14.7 | 14.8 |
| BMIΔ (kg/m2) | 30.4 ± 8.8 | 32.3 ± 7.2 | 32.4 ± 6.5 | 32.9 ± 7.2 |
| Waist circumferenceΔ (cm) | 104.0 ± 19.9 | 108.6 ± 15.7 | 109.3 ± 14.3 | 110.7 ± 15.8 |
| Hypertension# (%) | 62.0 | 66.1 | 70.3 | 69.2 |
| Coronary heart disease (%) | 11.4 | 10.8 | 11.1 | 9.9 |
| Congestive heart failure (%) | 7.6 | 6.6 | 9.1 | 8.3 |
| Stroke (%) | 6.6 | 6.5 | 8.3 | 7.8 |
| Lung disease (%) | 13.4 | 10.1 | 11.1 | 12.3 |
| CancerΔ (%) | 5.8 | 12.3 | 13.6 | 16.4 |
| Current smoking (%) | 20.4 | 20.8 | 19.0 | 17.4 |
| ACE-I or ARB useΔ (%) | 15.8 | 33.9 | 46.0 | 52.1 |
| Statin useΔ (%) | 4.6 | 24.8 | 38.4 | 45.6 |
| Blood pressure (mmHg) | ||||
| SystolicΔ | 134.6 ± 26.7 | 134.2 ± 19.9 | 132.2 ± 18.6 | 129.0 ± 20.1 |
| DiastolicΔ | 75.9 ± 13.9 | 70.9 ± 14.9 | 69.5 ± 13.7 | 68.3 ± 14.2 |
| Cholesterol (mg/dL | ||||
| LDLΔ | 130.2 ± 61.4 | 118.9 ± 30.1 | 111.0 ± 33.7 | 104.7 ± 34.5 |
| HDLΔ | 45.6 ± 20.6 | 45.3 ± 12.3 | 50.1 ± 13.0 | 46.6 ± 13.8 |
| eGFRΔ (mL/min/1.73 m2) | 87.5 ± 32.3 | 84.3 ± 23.2 | 83.4 ± 20.9 | 84.0 ± 23.8 |
| Urine ACR# (μg/mg) | 12.8 (6.0, 41.8) | 12.9 (6.2, 49.3) | 11.9 (6.4, 36.5) | 11.1 (6.0, 31.0) |
Survey weight-adjusted means ± SDs, medians (interquartile range), or proportions are presented.
*Defined as a fasting glucose ≥126 mg/dL, HbA1c ≥6.5% (48 mmol/mol), and/or use of glucose-lowering medications.
#P < 0.05.
ΔP < 0.001.
Supplementary Tables 1 and 2 provide the population estimates of diabetes and the CKD phenotypes within the U.S. population with diabetes by time period and their overall prevalence. From 1988 to 2010, the number of adults with diabetes and low eGFR (<60 mL/min/1.73 m2), regardless of ACR status, increased by 134% from ∼1.73 million during years 1988–1994 to 4.05 million during years 2007–2010. The total estimated adults with diabetes and low eGFR with ACR <30 mg/g increased from ∼0.9 million (95% CI 0.7–1.1) or 6.7% of the total adult population with diabetes during years 1988–1994 to 2.4 million (95% CI 1.9–2.9) or 10.1% of the total adult population with diabetes during years 2007–2010 (Table 1).
Supplementary Fig. 1A–C shows the temporal changes in the distribution of ACR groups in the total U.S. adult population with diabetes by eGFR ≥90, 60–89, and <60 mL/min/1.73 m2. Prevalence of ACR groups ≥30 mg/g has overall declined over time with the exception of ACR ≥300 mg/g in the group with eGFR 60–80 mL/min/1.73 m2. Among adults with diabetes and low eGFR, prevalence of ACR <30 mg/g has increased over time from 50.4% during years 1988–1994 to 59.4% during years 2007–2010 (Supplementary Fig. 1C).
Table 2 summarizes the characteristics of adults with diabetes and low eGFR and ACR <30 mg/g by time period. The presence of comorbid conditions, such as heart failure and cancer, and use of RAAS inhibitors and statin medications have all increased over time in this group.
Table 2.
Characteristics of U.S. adults with diabetes and an eGFR <60 mL/min/1.73 m2 and ACR <30 mg/g by time period
| 1988–1994 population estimate = 0.9 million; | 1999–2002 population estimate = 1.3 million; | 2003–2006 population estimate = 2.0 million; | 2007–2010 population estimate = 2.4 million; | |
|---|---|---|---|---|
| sampled persons = 135 | sampled persons = 95 | sampled persons = 131 | sampled persons = 191 | |
| Age# (years) | 74.2 ± 10.8 | 73.7 ± 10.0 | 71.3 ± 8.5 | 71.3 ± 9.0 |
| Male (%) | 42.3 | 38.0 | 44.7 | 40.7 |
| African American race (%) | 13.8 | 11.1 | 12.2 | 14.0 |
| BMIΔ (kg/m2) | 28.5 ± 7.6 | 30.1 ± 7.0 | 31.4 ± 6.5 | 33.0 ± 7.6 |
| Waist circumferenceΔ (cm) | 102.0 ± 15.9 | 104.9 ± 14.6 | 107.1 ± 15.0 | 109.6 ± 16.3 |
| Hypertension (%) | 80.5 | 86.0 | 85.4 | 82.1 |
| Coronary heart disease (%) | 20.2 | 27.7 | 17.7 | 10.5 |
| Congestive heart failure (%) | 11.1 | 19.2 | 20.9 | 15.8 |
| Stroke (%) | 12.9 | 9.9 | 15.4 | 16.3 |
| Lung disease (%) | 16.5 | 9.9 | 15.6 | 14.3 |
| Cancer# (%) | 6.7 | 19.3 | 17.0 | 28.9 |
| Current smoking (%) | 10.5 | 4.3 | 14.2 | 7.1 |
| ACE-I or ARB useΔ (%) | 13.3 | 46.6 | 70.3 | 72.0 |
| Statin useΔ (%) | 2.2 | 41.1 | 51.4 | 59.4 |
| Blood pressure (mmHg) | ||||
| SystolicΔ | 139.6 ± 24.7 | 136.4 ± 22.4 | 134.1 ± 21.1 | 125.2 ± 16.9 |
| DiastolicΔ | 72.9 ± 12.9 | 61.0 ± 14.9 | 57.2 ± 17.6 | 58.4 ± 13.8 |
| Cholesterol (mg/dL) | ||||
| LDLΔ | 133.1 ± 55.9 | 112.8 ± 29.9 | 106.8 ± 40.1 | 94.7 ± 26.2 |
| HDL# | 45.6 ± 22.2 | 47.0 ± 14.0 | 52.2 ± 14.8 | 48.5 ± 15.0 |
| HbA1c (%) | 7.02 ± 1.81 | 6.69 ± 1.32 | 6.71 ± 0.89 | 6.69 ± 1.02 |
| eGFR (mL/min/1.73 m2) | 49.5 ± 11.7 | 46.6 ± 9.7 | 48.9 ± 8.6 | 49.0 ± 9.8 |
| Urine ACR (μg/mg) | 10.0 (6.5, 19.8) | 8.6 (4.8, 15.0) | 9.9 (5.8, 16.5) | 7.4 (4.5, 14.5) |
Survey weight-adjusted means ± SDs, medians (interquartile range), or proportions are presented.
ACE-I, ACE inhibitor; ARB, angiotensin receptor blocker.
#P < 0.05.
ΔP < 0.001.
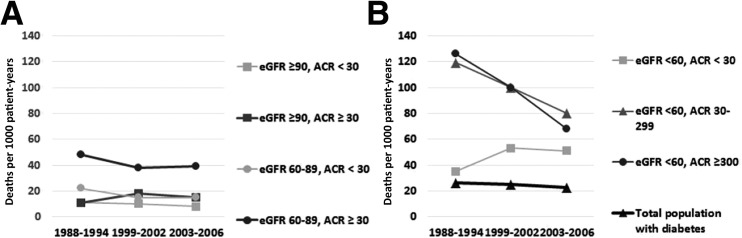
The crude and age-standardized 5-year mortality risks by CKD phenotype and by time period are reported in Table 3. Temporal increases in the crude and age-standardized mortality risk were noted for adults with diabetes and low eGFR with ACR <30 mg/g, whereas mortality risk generally decreased over time for those with low eGFR and ACR ≥30 mg/g. Figure 1 demonstrates the temporal changes in 5-year mortality rates in the total U.S. adult population with diabetes and in adults with diabetes and low eGFR by ACR groups. Mortality rates have overall declined for the total population of U.S. adults with diabetes and for those with diabetes and low eGFR with ACR ≥30 mg/g. However, the mortality rate in the CKD group with low eGFR and ACR <30 mg/g increased from 35 deaths per 1,000 person-years (95% CI 23, 55) during years 1988–1994 to 51 deaths per 1,000 person-years (95% CI 33, 83) during years 2003–2006.
Table 3.
Crude and age-standardized 5-year mortality risk in the U.S. adult population with diabetes by time period and by CKD phenotype
| 1988–1994 population estimate = 13.2 million | 1999–2002 population estimate = 16.0 million | 2003–2006 population estimate = 19.7 million | |
|---|---|---|---|
| Crude mortality risk (95% CI)+ | |||
| eGFR ≥90 mL/min/1.73 m2 | |||
| ACR (mg/g) | |||
| <30 | 5.6 (2.9, 8.3) | 5.0 (0.9, 9.1) | 3.8 (1.3, 6.3) |
| 30–299 | 5.5 (0.4, 10.6) | 10.1 (1.7, 18.5) | 7.0 (3.7, 12.3) |
| ≥300 | 4.9++ | 2.3++ | 8.8++ |
| eGFR 60–89 mL/min/1.73 m2 | |||
| ACR (mg/g) | |||
| <30 | 11.8 (8.7, 14.9) | 7.5 (4.0, 11.0) | 7.2 (3.7, 10.7) |
| 30–299 | 19.1 (11.1, 27.1) | 14.2 (5.0, 23.4) | 18.6 (11.2, 26.0) |
| ≥300 | 34.6 (21.2, 47.9) | 28.9 (4.4, 53.4) | 14.4 (0.7, 27.7) |
| eGFR <60 mL/min/1.73 m2 | |||
| ACR (mg/g) | |||
| <30 | 16.3 (9.4, 23.2) | 23.2 (12.4, 34.0) | 22.8 (13.2, 32.4) |
| 30–299 | 46.4 (33.9,58.9) | 40.0 (23.9, 56.1) | 33.5 (20.2, 46.8) |
| ≥300 | 44.7 (25.1, 64.3) | 43.7 (27.6, 62.7) | 29.8 (18.4, 41.2) |
| Age-standardized mortality risk (95% CI)* | |||
| eGFR ≥90 mL/min/1.73 m2 | |||
| ACR (mg/g) | |||
| <30 | 10.0 (6.3, 13.7) | 12.2 (5.9, 18.5) | 11.2 (7.5, 14.9) |
| 30–299 | 5.4 (1.9, 8.9) | 18.4 (13.3, 23.5) | 10.8 (7.5, 14.1) |
| ≥300 | 12.0 (4.9, 19.1) | 7.7++ | 10.0 (7.1, 12.9) |
| eGFR 60–89 mL/min/1.73 m2 | |||
| ACR (mg/g) | |||
| <30 | 9.6 (7.1, 12.1) | 8.2 (3.9, 12.5) | 6.8 (3.9, 9.7) |
| 30–299 | 13.3 (8.8, 17.8) | 15.9 (6.9, 24.9) | 11.9 (7.0, 16.8) |
| ≥300 | 29.4 (19.6, 39.2) | 21.2 (10.6, 31.8) | 25.9 (16.9, 34.9) |
| eGFR <60 mL/min/1.73 m2 | |||
| ACR (mg/g) | |||
| <30 | 6.2 (3.7, 8.7) | 18.3 (6.9, 29.7) | 20.9 (10.5, 26.2) |
| 30–299 | 37.0 (25.0, 49.0) | 39.6 (27.6, 51.6) | 23.8 (13.0, 34.6) |
| ≥300 | 43.1 (29.8, 56.4) | 51.1 (38.4, 63.8) | 28.9 (16.4, 41.4) |
The CKD-EPI equation was used to calculate eGFR (15).
+Percentage who died within 5 years.
*Populations age standardized to U.S. population in 2000.
++Sample size was too small to determine stable estimate of deaths.
Figure 1.
Mortality rates in the U.S. adult population with diabetes and an eGFR ≥60 (A) and <60 (B) mL/min/1.73 m2 by ACR groups (in mg/g) and by time period. Owing to the limited number of adults with ACR ≥300 mg/g in those with eGFR ≥60 mL/min/1.73 m2, the ACR group ≥300 was pooled with ACR ≥30–299. Due to the small number of individuals with ACR ≥300 mg/g with eGFR ≥60 mL/min/1.73 m2, ACR groups were categorized as <30 and ≥30 mg/g in panel A. B: Mortality rates in the total U.S. adult population with diabetes by time period are also shown (15). Mortality rates are number of deaths per 1,000 person-years.
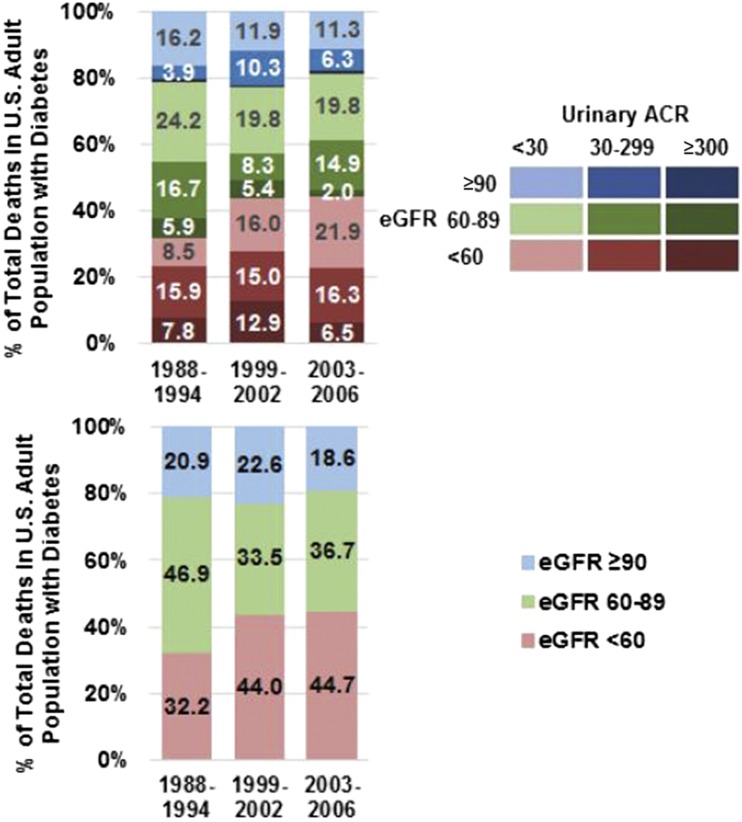
Figure 2 shows the distribution of deaths in the U.S. adult population with diabetes by CKD phenotypes by time period. The proportion of deaths occurring in the setting of low eGFR increased from 32.2% during years 1988–1994 to 44.7% during years 2003–2006. Some of this increase in percentage of total deaths occurring in the U.S. population with diabetes and low eGFR was due to the increasing proportion of deaths among adults with low eGFR and ACR <30 mg/g, which increased from 8.5% during years 1988–1994 to 16.0% during years 1999–2002 to 21.9% during years 2003–2006.
Figure 2.
Distribution of deaths in the U.S. population with diabetes by time period and by CKD phenotypes. The top panel shows the distribution of ACR groups within eGFR groups by time period, and the bottom panel shows the distribution of eGFR groups by time period (15).
In unadjusted models, no significant association was noted between time period and mortality among U.S. adults with diabetes and low eGFR with ACR <30 mg/g (see Supplementary Table 3). Compared with years 1988–1994, hazard rates of mortality were higher for all later time periods for the adults with diabetes and low eGFR with ACR <30 mg/g after adjustment for demographics and comorbidities, but differences did not meet statistical significance. Use of RAAS inhibitors and statin drugs attenuated these differences.
Conclusions
As a result of the increasing prevalence of low eGFR and decreasing prevalence of increased urine albumin excretion, the combined prevalence of low eGFR without increased urine albumin excretion in the U.S. population with diabetes has been increasing, as previously documented (9). Low eGFR, defined as <60 mL/min/1.73 m2, now affects nearly one in six U.S. adults with diabetes, or ∼4.0 million, and more than half of adults with diabetes and low eGFR do not have increased urine albumin excretion. Our study demonstrates the potential effect that the CKD phenotype dynamics may have on mortality. Currently, more than one of every five deaths in the U.S. population with diabetes occurs in the setting of low eGFR with an ACR <30 mg/g. Although mortality rates have trended downward for adults with diabetes and low eGFR with ACR ≥30 mg/g, mortality rates appear higher for those with eGFR <60 mL/min/1.73 m2 and ACR <30 mg/g compared with rates during years 1988–1994. Consequently, the proportion of deaths in the U.S. population with diabetes associated with low eGFR and ACR <30 mg/g has increased over time.
The reduction in albuminuria in diabetes is probably a reflection of the improved glycemic control and blood pressure control and the increased use of RAAS inhibitors during the past three decades (18). Indeed, the use of RAAS inhibitors is dramatically higher and systolic and diastolic blood pressures are significantly lower in the later years in adults with diabetes and low eGFR with urinary ACR <30 mg/g. These results are consistent with the earlier observations that the mortality and incidence of ESRD in diabetes (19) have been declining in recent years. With decreased mortality and decreased progression to ESRD, the prevalence of less advanced kidney disease in diabetes would be expected to increase, and the results of this study support that premise.
Over time, mortality rates have declined for most adults with diabetes who have increased urine albumin excretion. In contrast, mortality rates have not trended downward for individuals with low eGFR in the absence of increased urine albumin excretion, and these findings hold strong public health implications. Most clinical trials examining interventions to slow CKD progression in the setting of diabetes are limited to individuals with increased urine albumin excretion. Currently, we have few interventions that slow CKD progression in adults with diabetes in the absence of increased urine albumin excretion. The newer glucose-lowering agents, such as sodium–glucose cotransporter 2 inhibitors and the glucagon like peptide 1 agonists, have demonstrated both renal and cardiovascular benefits in adults with diabetes and moderate CKD, and clinical trials included individuals without increased urine albumin excretion (20–22). Use of these agents in the setting of moderate CKD (eGFR 59–30 mL/min/1.73 m2) may not only slow eGFR decline but also reduce cardiovascular mortality.
This study also showed increases in the use of RAAS inhibitors and progressive lowering of average systolic and diastolic blood pressures during the past two decades among adults with diabetes. Some of these individuals with reduced eGFR in the absence of increased urine albumin excretion possibly have CKD due to reduced renal perfusion pressure from aggressive blood pressure reduction. These individuals with reduced eGFR in the absence of increased urine albumin excretion may reflect sicker individuals who would have had increased urine albumin excretion in the absence of aggressive blood pressure lowering and use of RAAS inhibitors.
A limitation of the study is that diabetic kidney disease is based on eGFR and ACR values. In some individuals, low eGFR in the absence of increased urine albumin excretion may be due to nondiabetes disease processes. However, studies have demonstrated the presence of classical pathologic lesions associated with diabetes in kidneys of adults with low eGFR in the absence of increased urine albumin excretion (23–25). That low eGFR in the absence of increased urine albumin excretion may be due to tubulointerstitial disease caused by diabetes has also been hypothesized (26). Thus, nondiabetic kidney diseases are not likely the cause of low eGFR in most adults with diabetes. Our study included a fasting glucose ≥126 mg/dL to define the presence of diabetes in addition to an elevated HbA1c and/or use of glucose-lowering medications, whereas a previous analysis of diabetes in NHANES did not (9). Differences in definition of diabetes may lead to small differences in prevalence estimates for CKD phenotypes defined by eGFR and ACR values. Our analysis focused on three eGFR groups and could not specifically focus on adults with more advanced CKD due to sample size limitations. Our analyses may also be inadequately powered to detect significant differences in mortality in the CKD group with eGFR <60 mL/min/1.73 m2 and ACR <30 mg/g by time period due to the small number of sample participants. NHANES are multiple cross-sectional samples reflecting snap shots of the noninstitutionalized U.S. population at that given time period. Longitudinal follow-up data are not available in NHANES. Thus, we cannot determine whether the natural history of CKD development and progression differed within a given individual across time. However, we did show that the higher hazard rate for mortality in the earlier time period in this CKD group was attenuated after adjusting for differences in comorbid conditions and use of RAAS inhibitors during later time periods.
In summary, the phenotype or clinical presentation of CKD in the U.S. population with diabetes has evolved over time, and this change appears to have influenced mortality rates. The most common phenotype of kidney disease in diabetes is now low eGFR with ACR <30 mg/g. Apart from targeting CKD progression in those with severely increased urinary albumin excretion (ACR >300 mg/g), clinical interventions and public health policies should also address the high mortality rates in adults with diabetes and low eGFR in the absence of increased urine albumin excretion.
Supplementary Material
Article Information
Funding. This work is supported by grants from the National Institute of Diabetes and Digestive and Kidney Diseases (R01-DK-091437 and R21-DK-106574) and the University of Utah Study Design and Biostatistics Center (funded in part from the Public Health Services research grant numbers UL1-RR-025764 and C06-RR-11234 from the National Center for Research Resources).
Duality of Interest. No potential conflicts of interest relevant to this article were reported.
Author Contributions. H.K. developed hypotheses, analyzed data, and wrote and revised the manuscript. R.E.B. performed data analysis, analyzed data, and revised the manuscript. D.L. revised the manuscript. L.F. helped develop hypotheses and revised the manuscript. G.W. and G.C. assisted with data analyses and manuscript revisions. T.G. assisted with statistical analyses, study design, and manuscript revision. S.E.R. and R.C. helped revise the manuscript. S.B. developed hypotheses, analyzed data, and revised the manuscript. H.K. and S.B. are the guarantors of this work and, as such, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Footnotes
This article contains Supplementary Data online at http://care.diabetesjournals.org/lookup/suppl/doi:10.2337/dc17-1954/-/DC1.
References
- 1.Centers for Disease Control and Prevention (CDC) Increasing prevalence of diagnosed diabetes–United States and Puerto Rico, 1995-2010. MMWR Morb Mortal Wkly Rep 2012;61:918–921 [PubMed] [Google Scholar]
- 2.Stokes A, Preston SH. Deaths attributable to diabetes in the United States: comparison of data sources and estimation approaches. PLoS One 2017;12:e0170219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Groop PH, Thomas MC, Moran JL, et al.; FinnDiane Study Group . The presence and severity of chronic kidney disease predicts all-cause mortality in type 1 diabetes. Diabetes 2009;58:1651–1658 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Orchard TJ, Secrest AM, Miller RG, Costacou T. In the absence of renal disease, 20 year mortality risk in type 1 diabetes is comparable to that of the general population: a report from the Pittsburgh Epidemiology of Diabetes Complications Study. Diabetologia 2010;53:2312–2319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Afkarian M, Sachs MC, Kestenbaum B, et al. . Kidney disease and increased mortality risk in type 2 diabetes. J Am Soc Nephrol 2013;24:302–308 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Adler AI, Stevens RJ, Manley SE, Bilous RW, Cull CA, Holman RR; UKPDS GROUP . Development and progression of nephropathy in type 2 diabetes: the United Kingdom Prospective Diabetes Study (UKPDS 64). Kidney Int 2003;63:225–232 [DOI] [PubMed] [Google Scholar]
- 7.Mogensen CE, Christensen CK, Vittinghus E. The stages in diabetic renal disease: with emphasis on the stage of incipient diabetic nephropathy. Diabetes 1983;32(Suppl. 2):64–78 [DOI] [PubMed] [Google Scholar]
- 8.Kramer HJ, Nguyen QD, Curhan G, Hsu CY. Renal insufficiency in the absence of albuminuria and retinopathy among adults with type 2 diabetes mellitus. JAMA 2003;289:3273–3277 [DOI] [PubMed] [Google Scholar]
- 9.Afkarian M, Zelnick LR, Hall YN, et al. . Clinical manifestations of kidney disease among US adults with diabetes, 1988-2014. JAMA 2016;316:602–610 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Matsushita K, Coresh J, Sang Y, et al.; CKD Prognosis Consortium . Estimated glomerular filtration rate and albuminuria for prediction of cardiovascular outcomes: a collaborative meta-analysis of individual participant data. Lancet Diabetes Endocrinol 2015;3:514–525 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Astor BC, Matsushita K, Gansevoort RT, et al.; Chronic Kidney Disease Prognosis Consortium . Lower estimated glomerular filtration rate and higher albuminuria are associated with mortality and end-stage renal disease. A collaborative meta-analysis of kidney disease population cohorts. Kidney Int 2011;79:1331–1340 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Coresh J, Turin TC, Matsushita K, et al.; CKD Prognosis Consortium . Decline in estimated glomerular filtration rate and subsequent risk of end-stage renal disease and mortality. JAMA 2014;311:2518–2531 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Centers for Disease Control and Prevention (CDC); National Center for Health Statistics (NCHS). National Health and Nutrition Examination Survey Data [Internet], 1988–2010. Hyattsville, MD, U.S. Department of Health and Human Services, Centers for Disease Control and Prevention. Available from http://www.cdc.gov.archer.luhs.org/nchas/nhanes.htm. Accessed 1 October 2016
- 14.Selvin E, Manzi J, Stevens LA, et al. . Calibration of serum creatinine in the National Health and Nutrition Examination Surveys (NHANES) 1988-1994, 1999-2004. Am J Kidney Dis 2007;50:918–926 [DOI] [PubMed] [Google Scholar]
- 15.Inker LA, Schmid CH, Tighiouart H, et al.; CKD-EPI Investigators . Estimating glomerular filtration rate from serum creatinine and cystatin C [published corrections appear in N Engl J Med 2012;367:681 and 2060]. N Engl J Med 2012;367:20–29 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kidney Disease: Improving Global Outcomes (KDIGO) CKD Work Group. KDIGO 2012 clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int Suppl 2013;3:1–150 [Google Scholar]
- 17.Centers for Disease Control and Prevention, National Center for Health Statistics. National Health and Nutrition Examination Survey Methods and Analytic Guidelines [article online]. Hyattsville, MD, U.S. Department of Health and Human Services, Centers for Disease Control and Prevention. Available from https://wwwn.cdc.gov/nchs/nhanes/analyticguidelines. Accessed 1 October 2016
- 18.Tuttle KR, Bakris GL, Bilous RW, et al. . Diabetic kidney disease: a report from an ADA Consensus Conference. Am J Kidney Dis 2014;64:510–533 [DOI] [PubMed] [Google Scholar]
- 19.Burrows NR, Li Y, Geiss LS. Incidence of treatment for end-stage renal disease among individuals with diabetes in the U.S. continues to decline. Diabetes Care 2010;33:73–77 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hiramatsu T, Ozeki A, Ishikawa H, Furuta S. Long term effects of liraglutide in Japanese patients with type 2 diabetes among the subgroups with different renal functions: results of 2-year prospective study. Drug Res (Stuttg) 2017;67:640–646 [DOI] [PubMed] [Google Scholar]
- 21.Mann JF, Ørsted DD, Brown-Frandsen K, et al.; LEADER Steering Committee and Investigators . Liraglutide and renal outcomes in type 2 diabetes. N Engl J Med 2017;377:839–848 [DOI] [PubMed] [Google Scholar]
- 22.Wanner C, Inzucchi SE, Lachin JM, et al.; EMPA-REG OUTCOME Investigators . Empagliflozin and progression of kidney disease in type 2 diabetes. N Engl J Med 2016;375:323–334 [DOI] [PubMed] [Google Scholar]
- 23.Ekinci EI, Jerums G, Skene A, et al. . Renal structure in normoalbuminuric and albuminuric patients with type 2 diabetes and impaired renal function. Diabetes Care 2013;36:3620–3626 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Klessens CQ, Woutman TD, Veraar KA, et al. . An autopsy study suggests that diabetic nephropathy is underdiagnosed. Kidney Int 2016;90:149–156 [DOI] [PubMed] [Google Scholar]
- 25.Caramori ML, Fioretto P, Mauer M. Low glomerular filtration rate in normoalbuminuric type 1 diabetic patients: an indicator of more advanced glomerular lesions. Diabetes 2003;52:1036–1040 [DOI] [PubMed] [Google Scholar]
- 26.Chawla V, Roshan B.. Non-proteinuric diabetic nephropathy. Curr Diab Rep 2014;14:529. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.