Abstract
Background & objectives:
Development of antibacterial resistance and its association with antibiotic overuse makes it necessary to identify a specific and sensitive biomarker for the diagnosis of bacterial infection and guiding antibiotic therapy. Procalcitonin (PCT), as a sepsis biomarker, may play a role in guiding antibiotics treatment in hospital settings. The aim of the current meta-analysis was to analyze the utility of PCT on various outcomes of interest in inpatients.
Methods:
Different databases were searched for randomized controlled trials comparing PCT-guided therapy with standard therapy in admitted patients with bacterial infections. Twenty six articles were found suitable for full text search and of these, 16 studies were considered finally for data extraction.
Results:
There were no significant differences found in total mortality [pooled odds ratio (OR) 1.04, 95% confidence interval (CI) 0.89-1.22, P=0.63], 28-day mortality (pooled OR 0.97, 95% CI 0.80-1.19, P=0.79), need of Intensive Care Unit admission (OR=0.80, 95% CI 0.59-1.09, P=0.16) and duration of stay in hospital (pooled mean difference −0.01, 95% CI −0.50-0.49, P=0.98) between treatment and control groups. PCT-guided treatment significantly decreased the duration of antibiotic treatment (pooled mean difference −2.79, 95% CI −3.52-−2.06, P<0.00001).
Interpretation & conclusions:
PCT-guided therapy significantly decreased antibiotics exposure and thus treatment cost. However, the hard endpoints did not demonstrate any significant benefits, possibly due to low power to detect differences and/or the presence of comorbidities.
Keywords: Antibiotics, emergency, Intensive Care Unit, meta-analysis, procalcitonin, ward setting
In-hospital settings such as Intensive Care Units (ICUs), inpatient wards and emergency room witness heterogeneous group of infections of different aetiologies comprising bacterial, viral, fungal or combined infections. Antibiotic prescriptions face several challenges such as deciding the duration of treatment, choice of empiric antibiotics and de-escalation as important considerations. The importance of this decision-making is more paramount keeping in mind the variable severity of presentations in different settings within a hospital. Overprescription or overuse of antibiotics leading to increased cost to patients’ treatment and care, ineffectiveness of antibacterial due to emergence of resistance, toxicity and adverse effects are frequently encountered1. Hence, use of biomarkers to guide antibiotic therapy in the management of nosocomial infections was considered. Previously used biomarker such as C-reactive protein, erythrocyte sedimentation rate (ESR) and total leucocyte count (TLC) lack specificity. Procalcitonin (PCT) has been found to be a promising biomarkers with the high sensitivity (85%) and specificity (91%) for differentiating patients with infective aetiology from those with non-infective inflammatory or autoimmune diseases2.
PCT is a peptide precursor of calcitonin (CALC-1) secreted from C-cells of thyroid and the parenchymal cells of lung, liver, kidney, adipocytes and muscles. In case of infection, PCT level correlates directly with the level of the microbial toxins (e.g. endotoxins) or indirectly to the host immune response and level of different cytokines [interleukin (IL)-6, IL-1β, tumour-necrosis-factor-α (TNF-α)]3. Bacterial infections induce an increase of CALC-1 gene expression, which surges PCT in the serum within 3-6 h of exposure of the pathogen3. In vivo half-life of PCT is 20-24 h with high stability in serum or plasma ex-vivo4. In the healthy individuals, PCT level is usually <0.05 ng/ml5. Cut-off values have been described ranging from 0.5 to 2 ng/ml and these are dependent on several factors6. Hence, an informed decision with respect to severity of illness, patient population and comorbidities must be made for considering an appropriate cut-off. Numerous randomized control trials (RCTs) have been carried out to determine the role of PCT in early assessment and prediction of the severity of infection to guide effective antibiotic treatment and management. In view of the variable conclusion of these trials, we aimed at reviewing the literature systematically with meta-analytic approach to evaluate the effect of PCT-based antibiotic administration in the management of infections, only in the inpatient settings. Most of the reviews done earlier are based on particular diagnosis (acute respiratory tract infections)7,8 infections in autoimmune disease5 or in febrile neutropenic episodes in cancer patients9 or have considered certain group of population (age-specific10 or ICU patients11). Our objective was to know the clinical utility of PCT-guided therapy particularly in inpatient management.
Material & Methods
Search strategy: MEDLINE, EMBASE, Cochrane Library and Ovid database were searched from their inception to July 2013 to identify suitable RCTs. MeSH terms used for searches were ‘PCT’, ‘Pro-CT’, ‘procalcitonin’, ‘PCT precursor’, ‘antibiotic therapy’, ‘antimicrobial therapy’, separately and in combination. Studies considered to be eligible were RCTs comparing PCT-guided therapy with standard therapy in admitted patients with bacterial infections. No language restriction was applied. Searches were limited to human studies. The titles and abstract were screened for studies. Potentially relevant studies were retrieved and reviewed by two reviewers. A final list of selected studies was obtained.
Inclusion and exclusion criteria: All RCTs based on PCT-guided therapy and done in ICU, wards and emergency settings were included in this analysis.
Data extraction: Two of the researchers independently searched and assessed all RCTs related to PCT-guided management and standard of care treatment. Full-text articles were retrieved and examined by two of them separately, and then, data extraction was performed; Tables were constructed, in which characteristics of the studies were evaluated based on randomization, allocation concealment, blinding, intention to treat analysis, lost to follow up, inclusion criteria, control group (antibiotic usages as per the standard guideline in that setting) and PCT group (test arm), and outcomes. Pooled data were reviewed by the third researcher to resolve all discrepancies.
Quality assessment: The quality of the study was evaluated by the methods described in Cochrane Handbook for Systematic Reviews12. This was assessed under different aspects: random sequence generation (selection bias), allocation concealment (selection bias), blinding of participants and personnel (performance bias), blinding of outcome assessment (detection bias), incomplete outcome data (attrition bias), selective reporting (reporting bias) and other bias as well.
Results
Various outcomes evaluated were total mortality, 28-day mortality, need for stay in ICU, duration of stay in hospital, proportion of patients treated with antibiotics, duration of antibiotic treatment and antibiotic use per 1000 day of follow up. The comparison was between PCT-guided treatment and standard of care (without PCT observation) in inpatients. In keeping with the objectives of the study, three subgroups were considered important - ICU, ward and emergency.
Analysis: For each individual study, the data were expressed as ‘n’ and percentage (%) for dichotomous variables, and for continuous variables, mean and standard deviations were obtained. If parameters were not reported in the required format, then those were derived if possible. For example, standard deviation was derived using standard error and sample size. The data were pooled by random effects model in case of significant heterogeneity; otherwise, fixed-effect model was used. Heterogeneity was calculated using Chi-square statistics. Pooled odds ratio (OR) with 95 per cent confidence interval (CI) and mean difference with 95 per cent CI were the pooled outcome for dichotomous and continuous variables, respectively. Mantel-Haenzel (M-H) method was used for calculation of pooled OR and P<0.05 was considered significant. For investigational and publication bias, funnel plot was generated.
Outcomes: A total of 145 hits were obtained after combining search of all selected databases; 75 articles remained after excluding the duplicate articles. After abstracts and searching of cross-references, 26 studies were found suitable for full-text search. Of these, 16 studies13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28 were considered for data extraction and quality assessment. Ten studies were excluded for the following reasons: one was pilot study29, five studies did not fulfil the inclusion criteria6,30,31,32,33, two were study protocols34,35 and in two studies, randomization was not done36,37 (Fig. 1).
Fig. 1.
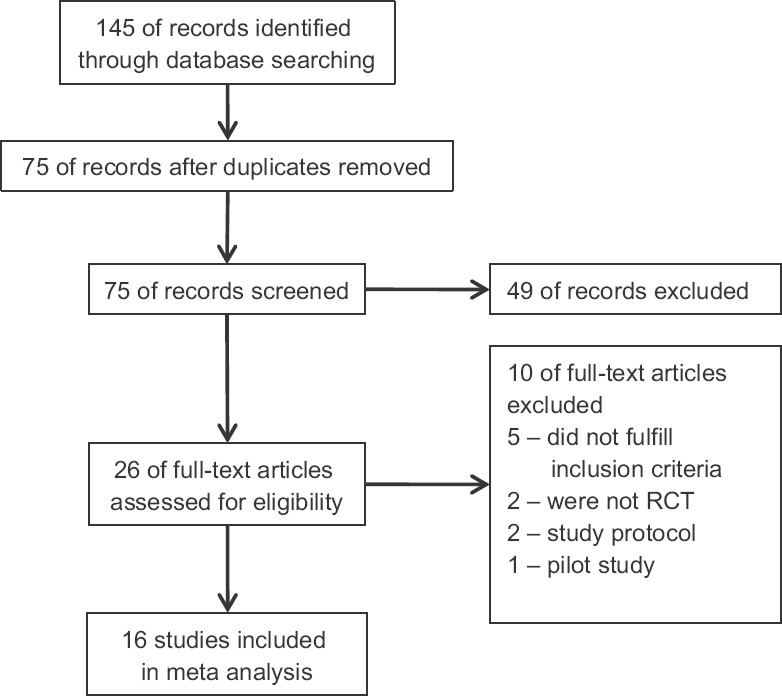
Flow chart showing selection process of the studies.
Quality of study: The studies largely had low risk of bias. Appropriate performance of randomization was reported in 16 studies13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28, allocation concealment (investigator blinding) was reported in 10 studies13,14,15,17,21,23,24,25,26,28, blinding of both participants and personnel was performed in nine trials13,14,15,17,21,24,25,26,28, with blinded outcome assessment observed in one trial15 and attrition was reported in 12 studies13,14,15,16,17,20,21,23,24,26,27,28.
Outcomes:
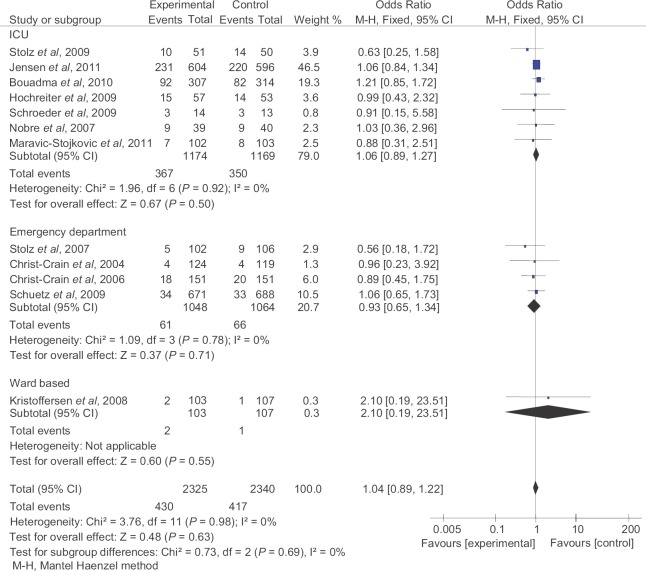
Total mortality: Twelve studies13,14,15,16,17,18,20,21,22,23,26,27 including 2325 patients in PCT-guided and 2340 patients in control groups were included for this analysis. There was no significant difference in incidence of total mortality between PCT-guided and control group (pooled OR 1.04, 95% CI 0.89-1.22, P=0.63). Similar results were found on subgroup analysis (Fig. 2).
Fig. 2.
Total mortality in patients and controls. M-H, Mantel Haenzel method.
28-day mortality: Five ICU-based studies14,15,17,26,27 including 1095 patients in PCT-guided and 1100 patients in control groups were included for analysis. There was no significant difference in incidence of 28-day mortality between PCT-guided and control group (pooled OR 0.97, 95% CI 0.80-1.19, P=0.79).
Need of ICU admission: Five studies13,16,20,21,23 including 1151 patients in PCT-guided and 1171 patients in control groups were included for analysis. There was no significant difference between PCT-guided and control group (pooled OR 0.80, 95% CI 0.59-1.09, P=0.16).
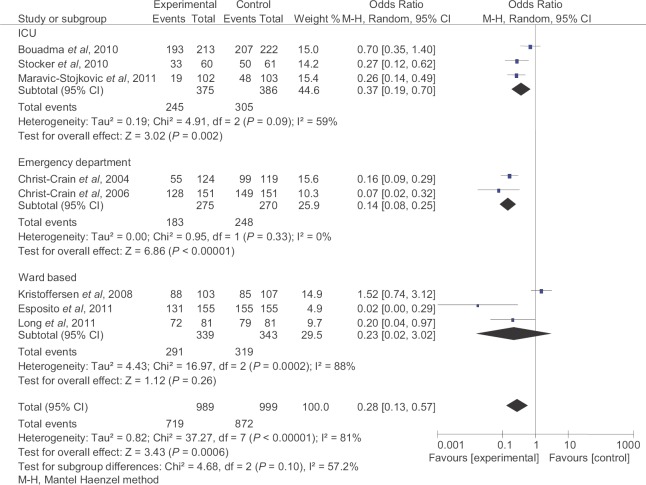
Proportion of patients treated with antibiotics: Eight studies16,17,19,20,21,25,27,28 including 989 patients in PCT-guided group and 999 patients in control group were included for analysis. The PCT-guided patients had a lower antibiotic exposure overall (pooled OR 0.28, 95% CI 0.13-0.57, P<0.0006). All the three subgroups of ICU, emergency departments and ward settings showed a significant reduction in incidence of antibiotic use (Fig. 3).
Fig. 3.
Proportion of patients treated with antibiotics. M-H, Mantel Haenzel method.
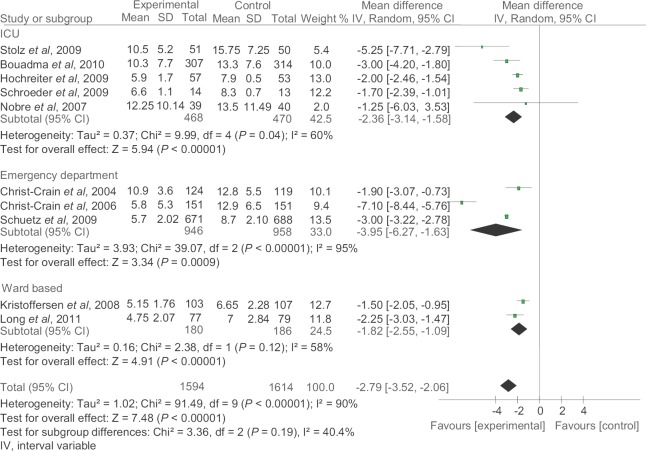
Duration of antibiotic treatment: Ten studies14,16,17,18,20,21,22,23,26,28 including 1594 patients in PCT-guided group and 1614 patients in control group were included for analysis. There was significant decrease in duration of antibiotic treatment in PCT-guided group as compared to control group (pooled mean difference −2.79, 95% CI −3.52-−2.06, P<0.00001) (Fig. 4).
Fig. 4.
Duration of antibiotic treatment. IV, intervariable
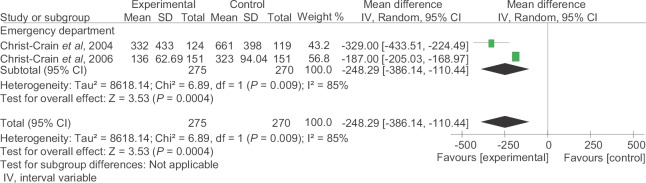
Antibiotic use per 1000 days of follow up: Two emergency-based studies20,21 including 275 patients in PCT-guided group and 270 patients in control group were included for analysis. The pooled mean difference of antibiotic use per 1000 days of follow up was −248.29 (95% CI −386.14-−110.44, P<0.00004), which was significant in favour of PCT-guided group (Fig. 5).
Fig. 5.
Antibiotic use per 1000 days of follow up. IV, intervariable
Duration of stay in hospital: Five studies16,20,21,23,27 including 1151 patients in PCT-guided group and 1168 patients in control group were included for this analysis. There was no significant difference in duration of hospital stay between PCT-guided group and control group (pooled mean difference −0.01, 95% CI −0.50-0.49, P=0.98). Similar results were observed on subgroup analysis.
Discussion
The present meta-analysis analyzed the utility of PCT on various outcomes of interest in inpatients. Inpatient setting (ICU, ward and emergency) was chosen for utilization of PCT-guided antibiotic therapy. The results of our meta-analysis indicated that the hard endpoints of mortality and need for ICU admissions were unaffected by the use of PCT-guided therapy. This would be expected since both the groups were given antibiotics as necessary. Further, these hard endpoints would be determined by several other factors such as disease severity, admission criteria, associated comorbid conditions such as diabetes, hypertension and immunocompromised state.
Furthermore, different protocols for PCT estimation were used in different trials7,20,21,27. For example, in emergency settings, Schuetz et al7 used a protocol of encouraging initiation or continuation of antibiotic treatment at PCT levels >0.5 μg/l, encouraged use of antibiotics when PCT levels were >0.25 μg/l and strongly discouraged its use when PCT levels were <0.1 μg/l. Two studies20,21 considered different protocol and considered PCT concentration 0.1 μg/l or less as indicative of the absence of infection; 0.1-0.25 μg/l as unlikely to have bacterial infection and discouraged use of antibiotics, and 0.25-0.5 μg/l was treated as possible infection and antibacterial treatment was started. On the contrary, the study by Maravic-Stojkovic et al27 used a protocol based on PCT of 0.5 ng/ml or less as an indicative of the absence of bacterial infection and antibiotic usage was discouraged. Further, in this study, a final decision to initiate antibacterial therapy was left at the discretion of the doctor in-charge. Hence, there was no standardization in serum PCT levels for initiation or discontinuation of antibiotic therapy, which could lead to inconsistencies in results.
A significant decrease in antibiotic prescription was demonstrated in PCT group as compared to the control group13,20,21,25. This is an important outcome considering the fact that antibiotics constitute an important cost-driver of treatment in in-hospital setting38. While applying measurement of serum PCT at a tertiary care hospital in ICU, wards or emergency settings, certain factors should be taken into account. First is the evaluation of cost-effectiveness. This parameter was not evaluated in our meta-analysis as the perspective differs considerably in setups in developing countries. For instance, in India, the majority of payment is out-of-pocket money. Second, decreased use of antibacterials has favourable implications in retarding development of resistance. Practical issues such as measuring serum PCT levels could be possible only in tertiary care hospitals and further time taken for reports to be available and start of antibiotic therapy have to be taken into account. Jensen et al15 reported increased usage of antibiotic in PCT-guided therapy group as compared to the control group. The most likely reason was inclusion of severely ill patients with chronic infections and underlying bloodstream infections. In this study, on the basis of serum PCT, antibiotics were started even before the actual culture reports were available. Another study done in severely ill patients showed no significant increase in antibiotic usage17.
In the emergency setting trials13,20,21,23, all trials included in our analysis reported reduced antibiotic usage as compared to the control group. Reduction in the number of days (mean difference=3.95) exceeded the duration of stay in most emergencies. Despite a favourable outcome of this aspect, an important consideration would be evaluation of readmission rates, referrals for admissions, which was not undertaken.
In ward-based trials, Kristoffersen et al16 reported increased antibiotic usage in PCT group as compared to control group. The reason was that antibiotics were still prescribed despite levels of PCT were <0.25 μg/l which were in variance with the study design. A meta-analysis conducted by Schuetz et al7 in patients with lower respiratory tract infections favoured PCT therapy in terms of decreased antibiotic prescription and duration of the drug use.
Antibiotic use per 1000 days of follow up favoured significantly PCT group as compared to the control group. This result was based on the two trials conducted in emergency department20,21 in which patients with respiratory tract infections were evaluated. Pooled analysis also showed significant heterogeneity pointing towards the variations in the diseases ranging from community-acquired pneumonia to asthma to bronchitis in these patients. This would imply its application to various respiratory conditions, for which patients report in emergency setting.
Duration of the stay in all the three settings, i.e. ICU, emergency and ward, did not show significant difference between the PCT-guided therapy and standard treatment. While PCT-guided therapy did not add any extra day for patient stay, it also did not lead to reduction in the number of days. A hospital stay could be an important factor in determining newer and more likely hospital-acquired infection. A significant effect on this outcome if proven would be of immense benefit.
Limitation of our meta-analysis included that not all the trials included were blinded in allocating treatment16,18,19,22,27. Another important limitation was the discretion of doctor in-charge to the final decision to initiate antibiotic therapy, which would imply that a different set of doctors may not necessarily replicate the findings. Further, potential publication bias could not be ruled out. PCT represents a novel approach as a diagnostics for bacterial infection but is also limited by both false-negative19 and false-positive results like, after trauma and major surgeries39,40. Hence, highly sensitive assays would be needed and also there is a need to standardize the disease-specific cut-off values.
In conclusion, application of PCT-guided antibiotic therapy should be based on analysis of cost and logistics of investigation when weighed against the benefits offered. Further, it is important to evaluate the data of the strategy when applied to real-life setting. Importantly, data regarding its effect on antibiotic resistance would need to be looked into before making it a usual practice. Though PCT-guided therapy has the potential for reducing antibiotic exposure and treatment cost, its usefulness on hard endpoints such as total mortality, 28-day mortality, need of ICU stay and resistance pattern will need to be seen. The real usefulness of this strategy would come if the test becomes available as point of care test.
Acknowledgment
Authors thank Dr Narottam Sharma, MA (Eng), formerly at Regional Institute of English Chandigarh (UT) for the valuable suggestions and proofreading of the manuscript.
Footnotes
Conflicts of Interest: None.
References
- 1.Simon L, Gauvin F, Amre DK, Saint-Louis P, Lacroix J. Serum procalcitonin and C-reactive protein levels as markers of bacterial infection: A systematic review and meta-analysis. Clin Infect Dis. 2004;39:206–17. doi: 10.1086/421997. [DOI] [PubMed] [Google Scholar]
- 2.Balcl C, Sungurtekin H, Gürses E, Sungurtekin U, Kaptanoglu B. Usefulness of procalcitonin for diagnosis of sepsis in the Intensive Care Unit. Crit Care. 2003;7:85–90. doi: 10.1186/cc1843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Becker KL, Snider R, Nylen ES. Procalcitonin in sepsis and systemic inflammation: A harmful biomarker and a therapeutic target. Br J Pharmacol. 2010;159:253–64. doi: 10.1111/j.1476-5381.2009.00433.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Meisner M, Schmidt J, Hüttner H, Tschaikowsky K. The natural elimination rate of procalcitonin in patients with normal and impaired renal function. Intensive Care Med. 2000;26(Suppl 2):S212–6. doi: 10.1007/BF02900740. [DOI] [PubMed] [Google Scholar]
- 5.Wu JY, Lee SH, Shen CJ, Hsieh YC, Yo PH, Cheng HY, et al. Use of serum procalcitonin to detect bacterial infection in patients with autoimmune diseases: A systematic review and meta-analysis. Arthritis Rheum. 2012;64:3034–42. doi: 10.1002/art.34512. [DOI] [PubMed] [Google Scholar]
- 6.Burkhardt O, Ewig S, Haagen U, Giersdorf S, Hartmann O, Wegscheider K, et al. Procalcitonin guidance and reduction of antibiotic use in acute respiratory tract infection. Eur Respir J. 2010;36:601–7. doi: 10.1183/09031936.00163309. [DOI] [PubMed] [Google Scholar]
- 7.Schuetz P, Briel M, Christ-Crain M, Stolz D, Bouadma L, Wolff M, et al. Procalcitonin to guide initiation and duration of antibiotic treatment in acute respiratory infections: An individual patient data meta-analysis. Clin Infect Dis. 2012;55:651–62. doi: 10.1093/cid/cis464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Li H, Luo YF, Blackwell TS, Xie CM. Meta-analysis and systematic review of procalcitonin-guided therapy in respiratory tract infections. Antimicrob Agents Chemother. 2011;55:5900–6. doi: 10.1128/AAC.00335-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Phillips RS, Wade R, Lehrnbecher T, Stewart LA, Sutton AJ. Systematic review and meta-analysis of the value of initial biomarkers in predicting adverse outcome in febrile neutropenic episodes in children and young people with cancer. BMC Med. 2012;10:6. doi: 10.1186/1741-7015-10-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Soni NJ, Samson DJ, Galaydick JL, Vats V, Huang ES, Aronson N, et al. Procalcitonin-guided antibiotic therapy: A systematic review and meta-analysis. J Hosp Med. 2013;8:530–40. doi: 10.1002/jhm.2067. [DOI] [PubMed] [Google Scholar]
- 11.Agarwal R, Schwartz DN. Procalcitonin to guide duration of antimicrobial therapy in Intensive Care Units: A systematic review. Clin Infect Dis. 2011;53:379–87. doi: 10.1093/cid/cir408. [DOI] [PubMed] [Google Scholar]
- 12.Higgins PTJ, Green S, editors. Cochrane Handbook for Systematic Reviews of Interventions. Version 510. London: The Cochrane Collaboration; 2011. [accessed on August 18, 2017]. updated March 2011. Available from: http://handbook-5-1.cochrane.org/ [Google Scholar]
- 13.Stolz D, Christ-Crain M, Bingisser R, Leuppi J, Miedinger D, Müller C, et al. Antibiotic treatment of exacerbations of COPD: A randomized, controlled trial comparing procalcitonin-guidance with standard therapy. Chest. 2007;131:9–19. doi: 10.1378/chest.06-1500. [DOI] [PubMed] [Google Scholar]
- 14.Stolz D, Smyrnios N, Eggimann P, Pargger H, Thakkar N, Siegemund M, et al. Procalcitonin for reduced antibiotic exposure in ventilator-associated pneumonia: A randomised study. Eur Respir J. 2009;34:1364–75. doi: 10.1183/09031936.00053209. [DOI] [PubMed] [Google Scholar]
- 15.Jensen JU, Hein L, Lundgren B, Bestle MH, Mohr TT, Andersen MH, et al. Procalcitonin-guided interventions against infections to increase early appropriate antibiotics and improve survival in the Intensive Care Unit: A randomized trial. Crit Care Med. 2011;39:2048–58. doi: 10.1097/CCM.0b013e31821e8791. [DOI] [PubMed] [Google Scholar]
- 16.Kristoffersen KB, Søgaard OS, Wejse C, Black FT, Greve T, Tarp B, et al. Antibiotic treatment interruption of suspected lower respiratory tract infections based on a single procalcitonin measurement at hospital admission - a randomized trial. Clin Microbiol Infect. 2009;15:481–7. doi: 10.1111/j.1469-0691.2009.02709.x. [DOI] [PubMed] [Google Scholar]
- 17.Bouadma L, Luyt CE, Tubach F, Cracco C, Alvarez A, Schwebel C, et al. Use of procalcitonin to reduce patients’ exposure to antibiotics in Intensive Care Units (PRORATA trial): A multicentre randomised controlled trial. Lancet. 2010;375:463–74. doi: 10.1016/S0140-6736(09)61879-1. [DOI] [PubMed] [Google Scholar]
- 18.Hochreiter M, Köhler T, Schweiger AM, Keck FS, Bein B, von Spiegel T, et al. Procalcitonin to guide duration of antibiotic therapy in intensive care patients: A randomized prospective controlled trial. Crit Care. 2009;13:R83. doi: 10.1186/cc7903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Stocker M, Fontana M, El Helou S, Wegscheider K, Berger TM. Use of procalcitonin-guided decision-making to shorten antibiotic therapy in suspected neonatal early-onset sepsis: Prospective randomized intervention trial. Neonatology. 2010;97:165–74. doi: 10.1159/000241296. [DOI] [PubMed] [Google Scholar]
- 20.Christ-Crain M, Jaccard-Stolz D, Bingisser R, Gencay MM, Huber PR, Tamm M, et al. Effect of procalcitonin-guided treatment on antibiotic use and outcome in lower respiratory tract infections: Cluster-randomised, single-blinded intervention trial. Lancet. 2004;363:600–7. doi: 10.1016/S0140-6736(04)15591-8. [DOI] [PubMed] [Google Scholar]
- 21.Christ-Crain M, Stolz D, Bingisser R, Müller C, Miedinger D, Huber PR, et al. Procalcitonin guidance of antibiotic therapy in community-acquired pneumonia: A randomized trial. Am J Respir Crit Care Med. 2006;174:84–93. doi: 10.1164/rccm.200512-1922OC. [DOI] [PubMed] [Google Scholar]
- 22.Schroeder S, Hochreiter M, Koehler T, Schweiger AM, Bein B, Keck FS, et al. Procalcitonin (PCT)-guided algorithm reduces length of antibiotic treatment in surgical intensive care patients with severe sepsis: Results of a prospective randomized study. Langenbecks Arch Surg. 2009;394:221–6. doi: 10.1007/s00423-008-0432-1. [DOI] [PubMed] [Google Scholar]
- 23.Schuetz P, Christ-Crain M, Thomann R, Falconnier C, Wolbers M, Widmer I, et al. Effect of procalcitonin-based guidelines vs standard guidelines on antibiotic use in lower respiratory tract infections: The ProHOSP randomized controlled trial. JAMA. 2009;302:1059–66. doi: 10.1001/jama.2009.1297. [DOI] [PubMed] [Google Scholar]
- 24.Manzano S, Bailey B, Girodias JB, Galetto-Lacour A, Cousineau J, Delvin E. Impact of procalcitonin on the management of children aged 1 to 36 months presenting with fever without source: A randomized controlled trial. Am J Emerg Med. 2010;28:647–53. doi: 10.1016/j.ajem.2009.02.022. [DOI] [PubMed] [Google Scholar]
- 25.Esposito S, Tagliabue C, Picciolli I, Semino M, Sabatini C, Consolo S, et al. Procalcitonin measurements for guiding antibiotic treatment in pediatric pneumonia. Respir Med. 2011;105:1939–45. doi: 10.1016/j.rmed.2011.09.003. [DOI] [PubMed] [Google Scholar]
- 26.Nobre V, Harbarth S, Graf JD, Rohner P, Pugin J. Use of procalcitonin to shorten antibiotic treatment duration in septic patients: A randomized trial. Am J Respir Crit Care Med. 2008;177:498–505. doi: 10.1164/rccm.200708-1238OC. [DOI] [PubMed] [Google Scholar]
- 27.Maravic-Stojkovic V, Lausevic-Vuk L, Jovic M, Rankovic A, Borzanovic M, Marinkovic J. Procalcitonin-based therapeutic strategy to reduce antibiotic use in patients after cardiac surgery: A randomized controlled trial. Srp Arh Celok Lek. 2011;139:736–42. [PubMed] [Google Scholar]
- 28.Long W, Deng X, Zhang Y, Lu G, Xie J, Tang J. Procalcitonin guidance for reduction of antibiotic use in low-risk outpatients with community-acquired pneumonia. Respirology. 2011;16:819–24. doi: 10.1111/j.1440-1843.2011.01978.x. [DOI] [PubMed] [Google Scholar]
- 29.Chromik AM, Endter F, Uhl W, Thiede A, Reith HB, Mittelkötter U. Pre-emptive antibiotic treatment vs.'standard’ treatment in patients with elevated serum procalcitonin levels after elective colorectal surgery: A prospective randomised pilot study. Langenbecks Arch Surg. 2006;391:187–94. doi: 10.1007/s00423-005-0009-1. [DOI] [PubMed] [Google Scholar]
- 30.Engle WD, Jackson GL, Sendelbach DM, Stehel EK, Ford DM, McHugh KM, et al. Pneumonia in term neonates: Laboratory studies and duration of antibiotic therapy. J Perinatol. 2003;23:372–7. doi: 10.1038/sj.jp.7210949. [DOI] [PubMed] [Google Scholar]
- 31.Qu R, Ji Y, Ling Y, Ye CY, Yang SM, Liu YY, et al. Procalcitonin is a good tool to guide duration of antibiotic therapy in patients with severe acute pancreatitis. A randomized prospective single-center controlled trial. Saudi Med J. 2012;33:382–7. [PubMed] [Google Scholar]
- 32.Briel M, Schuetz P, Mueller B, Young J, Schild U, Nusbaumer C, et al. Procalcitonin-guided antibiotic use vs. a standard approach for acute respiratory tract infections in primary care. Arch Intern Med. 2008;168:2000–7. doi: 10.1001/archinte.168.18.2000. [DOI] [PubMed] [Google Scholar]
- 33.Long W, Deng XQ, Tang JG, Xie J, Zhang YC, Zhang Y, et al. The value of serum procalcitonin in treatment of community acquired pneumonia in outpatient. Zhonghua Nei Ke Za Zhi. 2009;48:216–9. [PubMed] [Google Scholar]
- 34.Stocker M, Hop WC, van Rossum AM. Neonatal Procalcitonin Intervention Study (NeoPInS): Effect of Procalcitonin-guided decision making on duration of antibiotic therapy in suspected neonatal early-onset sepsis: A multi-centre randomized superiority and non-inferiority Intervention Study. BMC Pediatr. 2010;10:89. doi: 10.1186/1471-2431-10-89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Schuetz P, Christ-Crain M, Wolbers M, Schild U, Thomann R, Falconnier C, et al. Procalcitonin guided antibiotic therapy and hospitalization in patients with lower respiratory tract infections: A prospective, multicenter, randomized controlled trial. BMC Health Serv Res. 2007;7:102. doi: 10.1186/1472-6963-7-102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Louw JJ, Toelen J, Proesmans M, Vermeulen F, Billen J, de Boeck K. Serum procalcitonin is not an early marker of pulmonary exacerbation in children with cystic fibrosis. Eur J Pediatr. 2012;171:139–42. doi: 10.1007/s00431-011-1502-x. [DOI] [PubMed] [Google Scholar]
- 37.Layios N, Lambermont B, Canivet JL, Morimont P, Preiser JC, Garweg C, et al. Procalcitonin usefulness for the initiation of antibiotic treatment in Intensive Care Unit patients. Crit Care Med. 2012;40:2304–9. doi: 10.1097/CCM.0b013e318251517a. [DOI] [PubMed] [Google Scholar]
- 38.Kaur S, Rajagopalan S, Kaur N, Shafiq N, Bhalla A, Pandhi P, et al. Drug utilization study in medical emergency unit of a tertiary care hospital in North India. Emerg Med Int. 2014;2014:973578. doi: 10.1155/2014/973578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Meisner M, Tschaikowsky K, Hutzler A, Schick C, Schüttler J. Postoperative plasma concentrations of procalcitonin after different types of surgery. Intensive Care Med. 1998;24:680–4. doi: 10.1007/s001340050644. [DOI] [PubMed] [Google Scholar]
- 40.Mimoz O, Benoist JF, Edouard AR, Assicot M, Bohuon C, Samii K. Procalcitonin and C-reactive protein during the early posttraumatic systemic inflammatory response syndrome. Intensive Care Med. 1998;24:185–8. doi: 10.1007/s001340050543. [DOI] [PubMed] [Google Scholar]