Abstract
Osteoarthritis (OA) is the leading form of arthritis in the elderly, causing pain, disability, and immobility. OA has been associated with accumulation of senescent cells in or near joints. However, evidence for a causal link between OA and cellular senescence is lacking. Here, we present a novel senescent cell transplantation model involving injection of small numbers of senescent or nonsenescent cells from the ear cartilage of luciferase-expressing mice into the knee joint area of wild-type mice. By using bioluminescence and 18FDG PET imaging, we could track the injected cells in vivo for more than 10 days. Transplanting senescent cells into the knee region caused leg pain, impaired mobility, and radiographic and histological changes suggestive of OA. Transplanting nonsenescent cells had less of these effects. Thus, senescent cells can induce an OA-like state and targeting senescent cells could be a promising strategy for treating OA.
Keywords: Senolytics, Cell transplantation, Bioluminescence imaging, Fluorodeoxyglucose, Mechanical allodynia
Osteoarthritis (OA) is one of the leading causes of pain and disability worldwide (1). It can greatly increase health care costs and reduce quality of life. The key characteristics of age-related OA in humans include damage of articular cartilage with joint space narrowing, osteophyte formation, subchondral bone alteration, and degeneration of soft tissues (2). Age is the leading predictor for developing OA. Other risk factors include increased body weight, female sex, diet, genetics, race, and injuries (3). Injury-induced OA can be modeled in mice by damaging joints surgically (2). However, modeling age- or senescence-associated OA, which may be distinct from injury-related OA (2), in mice has been challenging. So far, no disease-modifying drug has been approved to treat OA other than pain reducers, partly because etiological mechanisms of age-related OA have been poorly understood to date (2). Potential cellular mechanisms contributing to the development of OA include low-grade inflammation (4), chondrocyte alteration (5), mitochondrial dysfunction (6), loss of glycosaminoglycans (7), and dysregulated energy metabolism (8,9). In addition, a potential contribution by senescent cells has been suggested (10,11).
Cellular senescence refers to a state of stable arrest of cell proliferation in replication-competent but apoptosis-resistant cells (12,13). Senescent cells accumulate with aging in various tissues (14), including the articular cartilage (15). A variety of stimuli and stresses can cause senescence, including telomere shortening, genomic DNA damage, oncogenic insults, metabolic stresses, epigenetic changes, and mitochondrial dysfunction (12,16). One key feature of senescent cells is secretion of an array of pro-inflammatory cytokines, chemokines, and growth factors, termed the senescence-associated secretory phenotype (SASP) (17,18). The SASP is observed across a number of senescent cell types, including fibroblasts and mesenchymal stem cells (17,19). Although mounting evidence suggests that cellular senescence is associated with OA (10,11,20–22), whether this link is causal remains to be determined.
We transplanted senescent cells into the knee joint region of mice and found that these cells induced a phenotype with features resembling OA, including articular cartilage erosion, increased pain, and impaired function. To our knowledge, this is the first evidence suggesting that cellular senescence can actually cause OA. Our findings also imply that targeting senescent cells is a promising approach for preventing or treating OA.
Materials and Methods
Details about methods are included in Supplementary Information.
Results
Establishing a Senescent Cell Transplantation Model
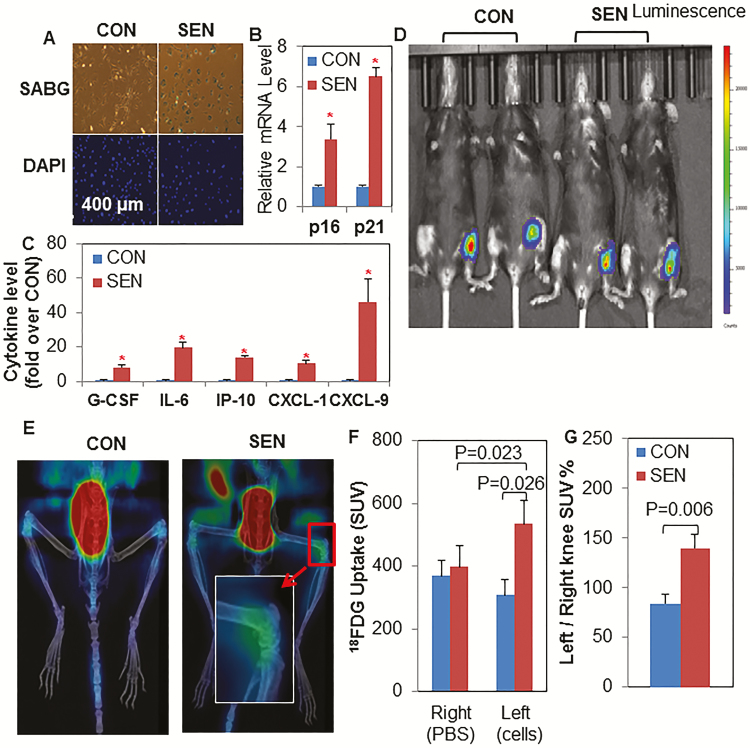
To test if senescent cells cause an OA-like arthropathy, we injected either senescent or control nonsenescent fibroblasts into the knee joint region of mice. We transplanted seven mice with control cells and seven with senescent cells. We isolated mouse fibroblasts from ear clips of CAG-luc mice and induced cellular senescence with 10 Gy radiation. Twenty days after radiation, more than 95% of fibroblasts were senescence-associated β-galactosidase (SABG) positive, whereas less than 5% of nonradiated parallel control fibroblasts were SABG positive (Figure 1A). p16Ink4a and p21Cip1 transcript levels, two key cellular senescence markers, were significantly increased in the radiated fibroblasts (Figure 1B). To assess the SASP, we collected conditioned medium from radiated and control fibroblasts for 24 hours. Twenty-three cytokine and chemokine proteins were measured in conditioned medium using an addressable laser bead-based multiplex assay. We found that the secreted protein levels of 19 SASP components were significantly increased in conditioned medium collected from radiated fibroblasts compared with nonradiated control cells (Supplementary Table 1). The most highly induced SASP components were: granulocyte-colony stimulating factor (G-CSF, 8-fold), interleukin-6 (IL-6, 20-fold), IFN-γ–induced protein 10 (IP-10, 14-fold), chemokine (C-X-C motif) ligand 1 (CXCL-1, 10-fold), and CXCL-9 (46-fold) (Figure 1C). These results suggested that radiation-induced senescent ear cartilage fibroblasts exhibited phenotypes typical of cellular senescence.
Figure 1.
Establishing a senescent cell transplantation model. Primary mouse ear cartilage fibroblasts were isolated from CAG-luc transgenic mice. (A) Control nonsenescent (CON) and radiation-induced senescent fibroblasts (SEN) were assayed for cellular senescence-associated β-galactosidase (SABG) activity and stained with DAPI. (B) The relative mRNA abundance of p16Ink4a and p21Cip1 is shown. Results (N = 6) are expressed as mean ± SEM. *p < .05. (C) Conditioned medium (CM) was collected from CON and SEN fibroblasts. Cytokine protein levels in CM were measured by multiplex assay and are expressed as a function of cell number. Selected cytokine levels are shown as the fold change relative to the average level in the CON group. Results (N = 6) are expressed as mean ± SEM. *p < .05. Right knees from 7-month-old C57BL/6 female mice were injected with PBS and left knees were injected with 2 × 105 senescent or nonsenescent control CAG-luc mouse primary ear fibroblasts in PBS. (D) One day later, bioluminescence was assayed using a Xenogen Ivis 200 Imaging System. Representative images are shown. (E) A further 3 days later, mice were injected with 18fluorodeoxyglucose (18FDG) and images focused on knee region were acquired by PET/CT scanning. Representative images are shown. 18FDG standard uptake value (F) and uptake ratios of the left to right knees within the same mice (G) are shown as mean ± SEM (N = 7).
Because both the senescent and nonsenescent control cells expressed luciferase, we were able to track these injected cells in vivo by bioluminescence imaging (BLI) after transplantation. One day after injection, we detected a strong bioluminescent signal in and around the left knee joint region in mice in which either senescent or nonsenescent fibroblasts had been injected (Figure 1D). These signals persisted for more than 10 days (Supplementary Figure 1), indicating that the transplanted cells were viable in the knee area for at least 10 days. We did not observe detectable bioluminescent signal in any of the mice in the right knee region, where buffer without cells had been injected. Increased glucose utilization is a feature of senescent cells (23,24). We tested whether we could detect the transplanted senescent cells using 18fluorodeoxyglucose positron emission tomography (FDG PET) imaging, a labeled, nonmetabolized glucose analog, in our model. Three days after transplantation, we found that the 18FDG PET standard uptake value (F-SUV) was significantly higher in the left knee joint region injected with senescent cells compared with the animals injected with control nonsenescent cells or buffer (Figure 1E and F). The F-SUV ratios of the left to the right knees within the same mice were also about 50% higher in the senescent compared with control nonsenescent cell-transplanted groups (Figure 1G). These results suggest it is feasible to detect senescent cells in vivo and to distinguish them from nonsenescent cells using glucose analog PET imaging.
Senescent Cell Transplantation Induces an OA-Like Phenotype and Impairs Function
Three months after cell injection, senescent and nonsenescent cell-injected knees were evaluated histologically and radiologically to assess articular cartilage and overall joint structure (Figure 2A and B). The senescent cell-injected knees exhibited severe articular cartilage damage at the lateral and medial tibial plateaus, as well as the femoral condyles (Figure 2A). Reduced Safranin O staining (cartilage content), erosion of articular cartilage, and delamination of the articular surface were evident (Figure 2A). Senescent cell-injected knees displayed significant erosion within the medial joint spaces down to the growth plate (histology score = 16.8±3.1) and erosion of the lateral femoral condyles, which was also observed radiologically (Figure 2C). Moreover, senescent cell-injected knees exhibited osteophyte formation, subchondral bone structure alteration, and damaged menisci, hallmarks of OA (Figure 2C, arrows). We did observe some damage to the nonsenescent cell-injected knees (Figure 2B) within the medial joint spaces (histology score = 5.7±3.3); however, this damage was significantly less than in the senescent cell-transplanted knees (Figure 2B). We postulate that some of the damage to the control nonsenescent cell-injected joint resulted from the relatively large volume (50 μL) injected into the knee area.
Figure 2.
Senescent cell transplantation induces osteoarthritis-like phenotypes and impairs function. Seven-month-old C57BL/6 female mice were subjected to control nonsenescent (CON) or senescent (SEN) primary ear fibroblast transplantation in the knee region. (A) Safranin O/Fast Green staining was performed 3 months after transplantation. Representative images (5× magnification of the knee joint) are shown. (B) Histology scores for the knee joints are shown as mean ± SEM (N = 5). (C) Representative radiographs are shown. (D and E) The von Frey filament assay was performed 3 months after transplantation. Paw withdrawal frequencies using both 0.16g (D) and 0.4g filaments (E) are shown as mean ± SEM (N = 7). (F) Rotarod assay was performed before and 1 month after transplantation. Results are shown as percent change in time to falling relative to baseline. Results (N = 7) are expressed as mean ± SEM. (G and H) Locomotor activity during 20 minutes of evaluation was monitored 3 months after transplantation. (G) Stationary time (ST) and active time (AT) are shown as mean ± SEM (N = 7). (H) Distance traveled is shown as mean ± SEM (N = 7).
OA-associated pain is a key, disabling complaint of patients suffering from OA. We examined whether transplantation of senescent cells increased pain by performing the von Frey filament assay. Senescent cell-injected left knees showed a trend to increased paw withdrawal (p = .061) when using 0.16g monofilaments (Figure 2D). With greater fiber bowing weight (0.4g), mice with the senescent cell-injected left knees had significantly increased mechanical hypersensitivity compared with the mice with control nonsenescent cell-injected left knees (Figure 2E). Pain sensitivity in senescent cell-injected left knees was also significantly higher than buffer-injected right knees within the same animals, but there was no significant difference between nonsenescent cell-injected and buffer-injected knees (Figure 2E). These data indicate that injection of senescent cells in and around the joint space of the knee causes pain.
We next tested whether senescent cell transplantation into knees impaired locomotor function. We found that Rotarod performance was significantly decreased in the mice injected with senescent cells compared with animals injected with control nonsenescent cells or those that were not injected (Figure 2F). In addition, we found that mice injected with senescent cells moved less and traveled shorter distances than mice injected with control nonsenescent cells (Figure 2F and G). Thus, senescent cell transplantation causes reduced locomotor function in mice.
Discussion
OA is a major cause of pain, immobility, and disability in the elderly (1). No disease-modifying interventions are now in clinical use, with current treatments focused on pain control, mobility aides such as canes, braces, or walkers, or surgical joint replacement (25). Impeding progress in developing disease-modifying treatments is a lack of understanding of the initial pathogenesis of age-related OA and a lack of mouse models of this condition, as opposed to the surgical damage-induced OA in mice that models injury-associated OA. Here, we provide evidence that accumulation of senescent cells in and around previously healthy joints can actually cause an OA-like arthropathy in mice. This both provides a new model of OA and implies that clearing senescent cells with senolytics or interfering with their pro-inflammatory SASP could be a disease-modifying therapeutic option. A next step will be to test such interventions in our senescent cell-transplanted model.
One of the potential mechanisms by which senescent cells could induce an OA-like phenotype is through the SASP. OA is linked to inflammation (4) and immune cells have been found in early stage OA (26). IL-6, one of the key SASP components, is highly associated with OA progression (27,28). We found that the senescent cells we transplanted secreted 20 times more IL-6 than nonsenescent cells (Figure 1C and Supplementary Table 1). We previously reported that senescent cells increase immune cell chemoattraction (19). Furthermore, immune cell chemokines, such as the macrophage chemoattractant, MCP-1, were secreted by the senescent cells we transplanted (Supplementary Table 1). Thus, senescent cell-provoked immune cell infiltration and the inflammation that they, in turn, induce might contribute to the OA-like phenotype we observed. In addition, senescent cells can directly impair progenitor function through the SASP (29) and spread senescence to nearby cells (30,31), both of which might contribute to dysfunction of chondrocytes and therefore to OA. Moreover, transplanted senescent cells might persist longer than nonsenescent cells in vivo due to the fact that they are resistant to apoptosis (13), which could allow them more time to damage the knee.
In our model, we transplanted 2×105 ear fibroblasts into the knee joint region. It will be important to ascertain the minimum number of injected senescent cells sufficient to induce the OA-like phenotype. In addition, extensive studies comparing effects of different transplanted senescent cell types (such as chondrocytes or mesenchymal stem cells), dose responses, effects of gender, effects of recipient age, use of other species such as rats, persistence of different types of transplanted cells, use of different drugs such as senolytics and SASP inhibitors after transplantation, additive effects of stressing the joints around which senescent cells have been transplanted, time course of histological changes to the knee following senescent cell transplantation, and many other analyses remain to be done to fully characterize this model.
One of the limitations of our model is that the injection itself might cause minor damage to the joint, which we did observe in some of the nonsenescent cell-injected joints (Figure 2B). We speculate that some of the damage to the control nonsenescent cell-injected joints resulted from the injection itself or the fact that some of nonsenescent cells possibly become senescent in vivo after injection. However, the damage to the senescent cell-injected joints was significantly more severe (Figure 2B). Additionally, only senescent cell-injected knees exhibited mechanical allodynia (Figure 2E), indicating that senescent cells contribute to the pain that is a consequence of OA. Advantages of our model are that it is relatively simple and it does not require breeding of genetically modified mice. Also, animals can be followed in real time in vivo during development of OA or before and after administering interventions by functional and pain testing and luciferase and PET imaging. Specifically, senescent cell abundance can be followed. Moreover, our model is clinically relevant because senescent cells accumulate within the joints in both OA patients (22) and aged individuals (15).
The finding that cellular senescence can drive development of an OA-like state is consistent with the geroscience hypothesis—that fundamental aging mechanisms, of which cellular senescence is one, predispose to age-related disabilities and chronic diseases, such as OA. If correct, this would imply that senescent cell accumulation may not only predispose to OA, but to multiple other age-related conditions, as is increasingly appearing to be the case (19,29,32–34). The fact that we were able to detect senescent cells in vivo using 18FDG PET imaging without any genetic modification (Figure 1E) might open a potential avenue to assess senescent cell burden in humans with a variety of conditions involving local accumulation of senescent cells, such as in idiopathic pulmonary fibrosis or at sites subjected to therapeutic radiation.
A variety of anti-inflammatory agents have been used for OA, mainly for pain control. They have not proven to be effective in modifying disease progression (2). Furthermore, these agents have side effects and toxicity due to continuous administration (35). Therefore, a novel therapeutic strategy is needed. Our findings increase the likelihood that senolytic agents or SASP inhibitors will be disease modifying. Based on a hypothesis-driven drug discovery approach, we recently identified agents that eliminate senescent cells selectively—senolytic drugs (33,36). We predict that senolytics or SASP inhibitors such as ruxolitinib, which decreases IL-6 secretion and effects by senescent cells and also alleviates the senescent cell-induced stem cell dysfunction caused by TGFβ-related SASP factors (19,29), will delay, prevent, or alleviate OA. Consistent with this possibility, we found that senolytics attenuate age-related loss of glycosaminoglycans, a contributor to developing OA (7), from the intervertebral discs of progeroid mice (33). Moreover, senolytics are effective when administered periodically (33), likely because senescent cells do not of course divide and may be slow to re-accumulate once cleared in the absence of a strong continuing insult. We predict that senolytics may have fewer side effects than the anti-inflammatory agents currently used for controlling pain.
In summary, our study indicates that senescent cells can directly contribute to the pathogenesis of OA. We also present a novel model of OA, which is clinically relevant and allows us and others to test whether clearing senescent cells or interfering with the SASP alleviates or delays OA. Thus, our model could be useful for the OA research field in conjunction with current surgical injury-based models. Targeting senescent cells might be a promising way to delay, prevent, or treat OA.
Supplementary Material
Supplementary data is available at The Journals of Gerontology, Series A: Biomedical Sciences and Medical Sciences online.
Funding
This work was supported by R01 AG013925 (J.L.K.), R21 AG047984 (J.L.K. and V.L.), K01AR065397 (E.W.B.), R01AR068103 (J.J.W.), MRM2015 6272 (E.W.B. and J.J.W.), the Orthopedic Research and Education Foundation in collaboration with the Howard Hughes Medical Institute (SMH), the Connor Group, and the Noaber Foundation.
Conflict of Interest
JK, TT, TP and Mayo Clinic have a financial interest related to this research. This research has been reviewed by the Mayo Clinic Conflict of Interest Review Board and is being conducted in compliance with Mayo Clinic conflict of interest policies.
Supplementary Material
Acknowledgments
We appreciate Jacqueline Armstrong’s editorial assistance.
References
- 1. Vos T, Flaxman AD, Naghavi M, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380:2163–2196. doi:10.1016/S0140-6736(12)61729-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Fang H, Beier F. Mouse models of osteoarthritis: modelling risk factors and assessing outcomes. Nat Rev Rheumatol. 2014;10:413–421. doi:10.1038/nrrheum.2014.46 [DOI] [PubMed] [Google Scholar]
- 3. Johnson VL, Hunter DJ. The epidemiology of osteoarthritis. Best Pract Res Clin Rheumatol. 2014;28:5–15. doi:10.1016/j.berh.2014.01.004 [DOI] [PubMed] [Google Scholar]
- 4. Attur M, Krasnokutsky S, Statnikov A, et al. Low-grade inflammation in symptomatic knee osteoarthritis: prognostic value of inflammatory plasma lipids and peripheral blood leukocyte biomarkers. Arthritis Rheumatol. 2015;67:2905–2915. doi:10.1002/art.39279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Rose J, Söder S, Skhirtladze C, et al. DNA damage, discoordinated gene expression and cellular senescence in osteoarthritic chondrocytes. Osteoarthritis Cartilage. 2012;20:1020–1028. doi:10.1016/j.joca.2012.05.009 [DOI] [PubMed] [Google Scholar]
- 6. Blanco FJ, Rego I, Ruiz-Romero C. The role of mitochondria in osteoarthritis. Nat Rev Rheumatol. 2011;7:161–169. doi:10.1038/nrrheum.2010.213 [DOI] [PubMed] [Google Scholar]
- 7. Plaas AH, West LA, Wong-Palms S, Nelson FR. Glycosaminoglycan sulfation in human osteoarthritis. Disease-related alterations at the non-reducing termini of chondroitin and dermatan sulfate. J Biol Chem. 1998;273:12642–12649. doi:10.1074/jbc.273.20.12642 [DOI] [PubMed] [Google Scholar]
- 8. Terkeltaub R, Yang B, Lotz M, Liu-Bryan R. Chondrocyte AMP-activated protein kinase activity suppresses matrix degradation responses to proinflammatory cytokines interleukin-1beta and tumor necrosis factor alpha. Arthritis Rheum. 2011;63:1928–1937. doi:10.1002/art.30333 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Liu-Bryan R, Terkeltaub R. Emerging regulators of the inflammatory process in osteoarthritis. Nat Rev Rheumatol. 2015;11:35–44. doi:10.1038/nrrheum.2014.162 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Loeser RF, Collins JA, Diekman BO. Ageing and the pathogenesis of osteoarthritis. Nat Rev Rheumatol. 2016;12:412–420. doi:10.1038/nrrheum.2016.65 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Loeser RF. Aging and osteoarthritis: the role of chondrocyte senescence and aging changes in the cartilage matrix. Osteoarthritis Cartilage. 2009;17:971–979. doi:10.1016/j.joca.2009.03.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Tchkonia T, Zhu Y, van Deursen J, Campisi J, Kirkland JL. Cellular senescence and the senescent secretory phenotype: therapeutic opportunities. J Clin Invest. 2013;123:966–972. doi:10.1172/JCI64098 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Wang E. Senescent human fibroblasts resist programmed cell death, and failure to suppress bcl2 is involved. Cancer Res. 1995;55:2284–2292. [PubMed] [Google Scholar]
- 14. Wang C, Jurk D, Maddick M, Nelson G, Martin-Ruiz C, von Zglinicki T. DNA damage response and cellular senescence in tissues of aging mice. Aging Cell. 2009;8:311–323. doi:10.1111/j.1474-9726.2009.00481.x [DOI] [PubMed] [Google Scholar]
- 15. Martin JA, Buckwalter JA. Telomere erosion and senescence in human articular cartilage chondrocytes. J Gerontol A Biol Sci Med Sci. 2001;56:B172–B179. doi:10.1093/gerona/56.4.B172 [DOI] [PubMed] [Google Scholar]
- 16. Wiley CD, Velarde MC, Lecot P, et al. Mitochondrial dysfunction induces senescence with a distinct secretory phenotype. Cell Metab. 2016;23:303–314. doi:10.1016/j.cmet.2015.11.011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Coppé JP, Patil CK, Rodier F, et al. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 2008;6:2853–2868. doi:10.1371/journal.pbio.0060301 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Kuilman T, Michaloglou C, Vredeveld LC, et al. Oncogene-induced senescence relayed by an interleukin-dependent inflammatory network. Cell. 2008;133:1019–1031. doi:10.1016/j.cell.2008.03.039 [DOI] [PubMed] [Google Scholar]
- 19. Xu M, Tchkonia T, Ding H, et al. JAK inhibition alleviates the cellular senescence-associated secretory phenotype and frailty in old age. Proc Natl Acad Sci U S A. 2015;112:E6301–E6310. doi:10.1073/pnas.1515386112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Mobasheri A, Matta C, Zákány R, Musumeci G. Chondrosenescence: definition, hallmarks and potential role in the pathogenesis of osteoarthritis. Maturitas. 2015;80:237–244. doi:10.1016/j.maturitas.2014.12.003 [DOI] [PubMed] [Google Scholar]
- 21. Philipot D, Guérit D, Platano D, et al. p16INK4a and its regulator miR-24 link senescence and chondrocyte terminal differentiation-associated matrix remodeling in osteoarthritis. Arthritis Res Ther. 2014;16:R58. doi:10.1186/ar4494 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Price JS, Waters JG, Darrah C, et al. The role of chondrocyte senescence in osteoarthritis. Aging Cell. 2002;1:57–65. doi:10.1046/j.1474-9728.2002.00008.x [DOI] [PubMed] [Google Scholar]
- 23. Dörr JR, Yu Y, Milanovic M, et al. Synthetic lethal metabolic targeting of cellular senescence in cancer therapy. Nature. 2013;501:421–425. doi:10.1038/nature12437 [DOI] [PubMed] [Google Scholar]
- 24. James EL, Michalek RD, Pitiyage GN, et al. Senescent human fibroblasts show increased glycolysis and redox homeostasis with extracellular metabolomes that overlap with those of irreparable DNA damage, aging, and disease. J Proteome Res. 2015;14:1854–1871. doi:10.1021/pr501221g [DOI] [PubMed] [Google Scholar]
- 25. Carr AJ, Robertsson O, Graves S, et al. Knee replacement. Lancet. 2012;379:1331–1340. doi:10.1016/S0140-6736(11)60752-6 [DOI] [PubMed] [Google Scholar]
- 26. Benito MJ, Veale DJ, FitzGerald O, van den Berg WB, Bresnihan B. Synovial tissue inflammation in early and late osteoarthritis. Ann Rheum Dis. 2005;64:1263–1267. doi:10.1136/ard.2004.025270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Livshits G, Zhai G, Hart DJ, et al. Interleukin-6 is a significant predictor of radiographic knee osteoarthritis: the Chingford Study. Arthritis Rheum. 2009;60:2037–2045. doi:10.1002/art.24598 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Tsuchida AI, Beekhuizen M, ‘t Hart MC, et al. Cytokine profiles in the joint depend on pathology, but are different between synovial fluid, cartilage tissue and cultured chondrocytes. Arthritis Res Ther. 2014;16:441. doi:10.1186/s13075-014-0441-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Xu M, Palmer AK, Ding H, et al. Targeting senescent cells enhances adipogenesis and metabolic function in old age. Elife. 2015;4:e12997. doi:10.7554/eLife.12997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Acosta JC, Banito A, Wuestefeld T, et al. A complex secretory program orchestrated by the inflammasome controls paracrine senescence. Nat Cell Biol. 2013;15:978–990. doi:10.1038/ncb2784 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Nelson G, Wordsworth J, Wang C, et al. A senescent cell bystander effect: senescence-induced senescence. Aging Cell. 2012;11:345–349. doi:10.1111/j.1474-9726.2012.00795.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Baker DJ, Wijshake T, Tchkonia T, et al. Clearance of p16Ink4a-positive senescent cells delays ageing-associated disorders. Nature. 2011;479:232–236. doi:10.1038/nature10600 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Zhu Y, Tchkonia T, Pirtskhalava T, et al. The Achilles’ heel of senescent cells: from transcriptome to senolytic drugs. Aging Cell. 2015;14:644–658. doi:10.1111/acel.12344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Roos CM, Zhang B, Palmer AK, et al. Chronic senolytic treatment alleviates established vasomotor dysfunction in aged or atherosclerotic mice. Aging Cell. 2016. February 10 [Epub ahead of print]. doi:10.1111/acel.12458 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Richette P, Latourte A, Frazier A. Safety and efficacy of paracetamol and NSAIDs in osteoarthritis: which drug to recommend? Expert Opin Drug Saf. 2015;14:1259–1268. doi:10.1517/14740338.2015.1056776 [DOI] [PubMed] [Google Scholar]
- 36. Zhu Y, Tchkonia T, Fuhrmann-Stroissnigg H, et al. Identification of a novel senolytic agent, navitoclax, targeting the Bcl-2 family of anti-apoptotic factors. Aging Cell. 2016;15:428–435. doi:10.1111/acel.12445 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Jurk D, Wilson C, Passos JF, et al. Chronic inflammation induces telomere dysfunction and accelerates ageing in mice. Nat Commun. 2014;2:4172. doi:10.1038/ncomms5172 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Bradley EW, Carpio LR, McGee-Lawrence ME, et al. Phlpp1 facilitates post-traumatic osteoarthritis and is induced by inflammation and promoter demethylation in human osteoarthritis. Osteoarthritis Cartilage. 2016;24:1021–1028. doi:10.1016/j.joca.2015.12.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Chaplan SR, Bach FW, Pogrel JW, Chung JM, Yaksh TL. Quantitative assessment of tactile allodynia in the rat paw. J Neurosci Methods. 1994;53:55–63. doi:10.1016/0165-0270(94)90144-9 [DOI] [PubMed] [Google Scholar]
- 40. Pritzker KP, Gay S, Jimenez SA, et al. Osteoarthritis cartilage histopathology: grading and staging. Osteoarthritis Cartilage. 2006;14:13–29. doi:10.1016/j.joca.2005.07.014 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.