Abstract
Purpose
The purpose of this study was to characterize whether induced pluripotent stem cells (iPSCs) affect survival of grafted retinal ganglion cells (RGCs) after transplantation.
Methods
For in vitro studies, human iPSCs were either directly cocultured with mouse RGCs or plated in hanging inserts in RGC cultures for 1 week. For ex vivo studies, RGCs and iPSCs were seeded onto the inner surface of an adult rat retina explant and cultured for 1 week. For in vivo studies, RGCs and iPSCs were intravitreally coinjected into an adult rat eye 1 week before examining retinas by explant and immunostaining.
Results
A dose-dependent increase in RGC survival was observed in RGC-iPSC direct cocultures, and RGC-iPSC indirect cocultures showed a similar RGC protective effect, but to a lesser extent than in direct coculture. Enhanced RGC survival was also identified in RGC-iPSC cotransplantations to adult retinas ex vivo and in vivo. In addition, RGCs with iPSC cotransplantation extended significantly longer neurites than RGC-only transplants.
Conclusions
Human iPSCs promote transplanted RGC survival and neurite extension. This effect may be mediated at least partially through secretion of diffusible neuroprotective factors.
Keywords: cell transplantation, retinal ganglion cell, induced pluripotent stem cell, cell survival, neuroprotection
Central nervous system (CNS) neuron loss is observed in many neurodegenerative diseases such as glaucoma,1,2 and those neurons are generally not replaced from endogenous repair. Neuron transplantation is a promising approach to restore neurons lost in degenerative diseases in the CNS. Our previous studies demonstrated that retinal ganglion cells (RGCs) could be successfully transplanted onto an adult retina.3 Transplanted RGCs survived and integrated into the host retina, displaying both morphologically and electrophysiologically mature phenotypes, with some extent of axonal and dendritic regeneration in vivo.3,4 However, RGC transplant efficiency was low, possibly because most transplanted cells remained in the vitreous cavity, an environment low in neurotrophic survival signals.
Recently, various stem cells have been proposed as sources of neuroprotection of endogenous host neurons in degenerative disease. For example, mesenchymal stem cells (MSCs) protect neurons throughout the CNS from numerous types of injuries including traumatic, ischemic, and inflammatory.5,6 In particular, MSCs promoted endogenous RGC survival in an experimental glaucoma model.7 Like MSCs, induced pluripotent stem cells (iPSCs) are multipotent, proliferative, and capable of differentiating into many cell types; however, their neuroprotective abilities have not been well studied. Could stem cells provide neuroprotective signaling to mature neurons undergoing transplant into the adult CNS? The present study was aimed to assess whether iPSCs promote RGC survival and neurite growth after transplant to the adult retina.
Materials and Methods
Animals and Stem Cells
All use of animals conformed to the ARVO Statement for the Use of Animals in Research and was approved by the Institutional Animal Care and Use Committee (IACUC) and the Institutional Biosafety Committee of Stanford University. GFP+ mice, both male and female, were bred from the C57BL/6-Tg(CAG-EGFP)1Osb/J strain obtained from Jackson Laboratories (Bar Harbor, ME, USA). Ten- to 12-week-old Sprague-Dawley (SD) rats, both male and female, were obtained from Charles River (Portage, MI, USA). Human iPSC line 297 (iPSC/297), derived from blood cells with Sendai virus transfection (Cytotune 2.0 kit; Life Technologies Inc., Gaithersburg, MD, USA) were kindly provided by the Stem Cell Core, Stanford University Department of Genetics (Stanford, CA, USA).
RGC Purification
RGCs were purified from 2- to 4-day postnatal GFP+ mice by sequential immunopanning with the CD90 (Thy1.2; AbD Serotec, Düsseldorf, Germany) antibody as previously described,8,9 yielding 99.5% pure RGCs. Freshly extracted retinas were dissociated with papain (65 units; Worthington, Freehold, NJ, USA), followed by removal of contaminating macrophages and endothelial cells by immunopanning using an anti-macrophage antibody (AIA31240; Accurate Chemical and Scientific Corporation, Westbury, NY, USA). RGCs were selected from the macrophage-depleted cell suspension immunologically, using an anti-CD90 antibody.
RGC-iPSC Coculture
iPSCs were maintained on reconstituted basement membrane matrix (Matrigel; Corning, Kennebunk, ME, USA) coated on six-well tissue culture plates in serum-free human iPSC growth medium (StemMACS iPS-Brew XF, human; Miltenyi Biotec, Bergisch Gladbach, Germany) containing 1× of antibiotic-antimycotic (Life Technologies; 15240062). After the wells were at least 80% confluent, cells were detached using 0.02% EDTA (Sigma-Aldrich Corp., St. Louis, MO, USA) and resuspended with 2 mL iPSC growth medium. Volumes from 10 to 40 μL of the suspension were taken out for coculture.
RGCs for culture were stained with a fluorescent tracking dye (CellTracker Deep Red Dye, 1 μM working concentration; Life Technologies) for 30 minutes at the end of the immunopanning procedure. Immediately after the purification, RGCs were plated on Matrigel-coated 24-well tissue culture plates (Falcon; Corning, Acton, MA, USA) at a concentration of 20,000 cells per well with or without iPSCs in 400 μL serum-free RGC growth medium plus 200 μL iPSC growth medium. For direct-indirect culture comparison, RGCs were plated on plates with hanging inserts (Costar; Corning), and iPSCs were plated either directly with RGCs (direct coculture) or on Matrigel-coated hanging inserts (6.5 mm diameter, 0.4 μm pore size; indirect coculture). RGC growth medium was prepared with Neurobasal (Life Technologies) supplemented with insulin (5 mg/mL; Sigma-Aldrich Corp.), sodium pyruvate (1 mM; Sigma-Aldrich Corp.), l-glutamine (1 mM; Sigma-Aldrich Corp.), triiodothyronine (T3; 40 ng/mL; Sigma-Aldrich Corp.), N-acetyl cysteine (5 mg/mL; Sigma-Aldrich Corp.), GS21 (1:100; GlobalStem, Gaithersburg, MD, USA), brain-derived neurotrophic factor (BDMF; 50 ng/mL; Peprotech, Rocky Hill, NJ, USA), ciliary neurotrophic factor (CTNF; 10 ng/mL; Peprotech), and forskolin (5 mM; Sigma-Aldrich Corp.) as described.8,9 RGCs were cultured or cocultured for 7 days before proceeding to viability tests.
In Vitro RGC Viability Assays
RGC viability was measured with Hoechst 3334 (4 μM)/calcein acetoxymethyl (AM; 2 μM) and SYTOX orange (1 μM) combination staining. A 3 × 3-tile 5× microscopy image was captured for each well using an inverted fluorescent microscope (Zeiss Germany, Oberkochen, Germany) 10 minutes after staining. All Hoechst+/Calcein+/SYTOX−/CellTracker+ cells were counted as live RGCs, and all Hoechst+/SYTOX+/CellTracker+ cells were counted as dead RGCs. Cell numbers were calculated using the automated counting function on FIJI ImageJ software (National Institutes of Health, Bethesda, MD, USA). In addition, to verify iPSC growth in the indirect coculture, cells in all hanging inserts were also stained with the same protocol and imaged under a Stereo and Zoom Microscope (Zeiss Germany).
Ex Vivo RGC-iPSC Cotransplantation
Culture of retinal explants was performed following our previous protocol.3,10 Briefly, fresh retinas were dissected from SD rats and plated on Millicell chamber filters (Millipore, Billerica, MA, USA; diameter, 30 mm; pore size, 0.4 um), keeping the ganglion cell layer upward. GFP+ RGCs (40,000 cells per explant in 40 μL RGC medium), with or without human iPSCs (40 μL/retina out of 4 mL suspension), were directly transplanted on the top of the retinal explants and allowed to settle on the inner retinal surface by gravity. Twenty minutes after transplantation, the chamber was transferred to a six-well culture plate (Falcon; VWR, Radnor, PA, USA) containing 1.2 mL RGC medium plus 0.6 mL iPSC medium as above. The explants were cultured for 7 days before fixation.
In Vivo RGC-iPSC Cotransplantation
GFP+ RGCs, with or without iPSCs, were transplanted intravitreally into rats, as previously described.3,4 Both retinas from a given rat were used, one assigned to the transplant and the other to the cotransplant group. For cotransplantation, after iPSCs were detached and resuspended, they were centrifuged at 300g for 5 minutes and again resuspended with 100 μL iPSC medium. After that, 2 μL from the 100 μL cell suspension or vehicle (stem cell medium) was mixed with 40,000 RGCs in 2 μL RGC medium. The 4-μL mixture was then injected into the vitreous of anaesthetized rats using a 33-gauge Hamilton syringe. The rats were placed on a heating pad until awake and were killed 7 days after transplantation.
Immunostaining and Imaging
The retinas from ex vivo transplantation were fixed in 4% paraformaldehyde (PFA; Sigma-Aldrich Corp.) for 3 hours and then placed on glass slides. After in vivo transplantation, rats were killed and perfused with PBS followed by 4% PFA. Eyes were dissected and fixed in 4% PFA for 3 hours, and then retinas were dissected and flat-mounted on glass slides. The flat mounted samples, either from the ex vivo or in vivo procedure, were permeabilized with 0.3% Triton X-100 (Sigma-Aldrich Corp.) for 20 minutes, blocked with 5% normal goat serum (Invitrogen, San Diego, CA, USA) in PBS for 1 hour, and then incubated with a mouse monoclonal anti-human nucleus primary antibody (1:200; Millipore, Schwalbach, Germany) overnight at 4°C. Retina samples were rinsed three times with PBS and incubated with rabbit polyclonal Alexa Fluor 488–tagged anti-GFP antibody (1:500; Life Technologies) and Alexa Fluor 647–conjugated secondary antibody (1:500; Life Technologies) overnight. The explants were then rinsed twice, stained with Hoechst (1:500 in PBS) for 15 minutes, rinsed twice again, and sealed under 1.5-mm coverslips with anti-fade mounting medium (ProLong Gold; Life Technologies) before imaging via confocal microscopy.
Neurite Length Measurement
Neurites from GFP+ RGCs on retinal explants were measured using the Simple Neurite Tracer plugin in FIJI. Average neurite length was calculated as overall neurite length divided by live RGC number on the explant.
Statistical Analysis
Paired t-tests were conducted to identify RGC survival rate differences among control and transplanted groups in vitro, ex vivo, and in vivo, as well as the average neurite length differences between control and variable groups in vivo. If more than two variants were compared, we performed repeated-measures ANOVA, followed by Bonferroni-corrected paired t-tests. P < 0.05 was considered significant.
Results
Human iPSCs Enhance RGC Viability In Vitro
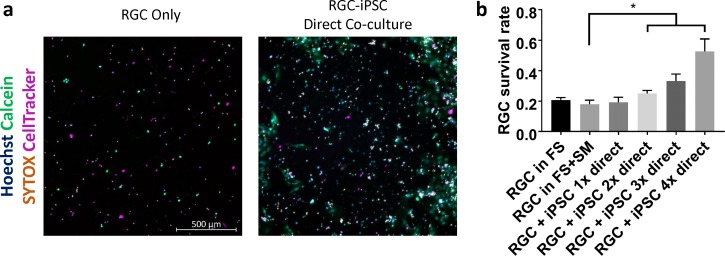
We first asked whether coculture with iPSCs increased RGC survival in vitro, using a combination of staining for Hoechst, Calcein, SYTOX, and CellTracker (Fig. 1a). After one week of RGC-iPSC direct culture, a significant iPSC dose-dependent increase in RGC viability was observed (Fig. 1b; P < 0.05; N = 3 experiments; n > 1000 cells/experiment). The coculture with highest iPSC dose demonstrated almost 2.5 times the RGC survival rate as the control.
Figure 1.
RGC-iPSC direct coculture promotes RGC survival. (a) RGCs and iPSCs were cocultured for 1 week. Different numbers of iPSCs were initially added into RGC culture, and RGCs were stained with CellTracker Deep Red. (b) After 1 week, an iPSC dose-dependent increase in RGC survival was found (*P < 0.05; paired t-test. N = 3 experiments; n > 1000 cells/experiment). FS, full sato (RGC growth medium); SM, stemMACS (iPSC growth medium). Error bar denotes SD.
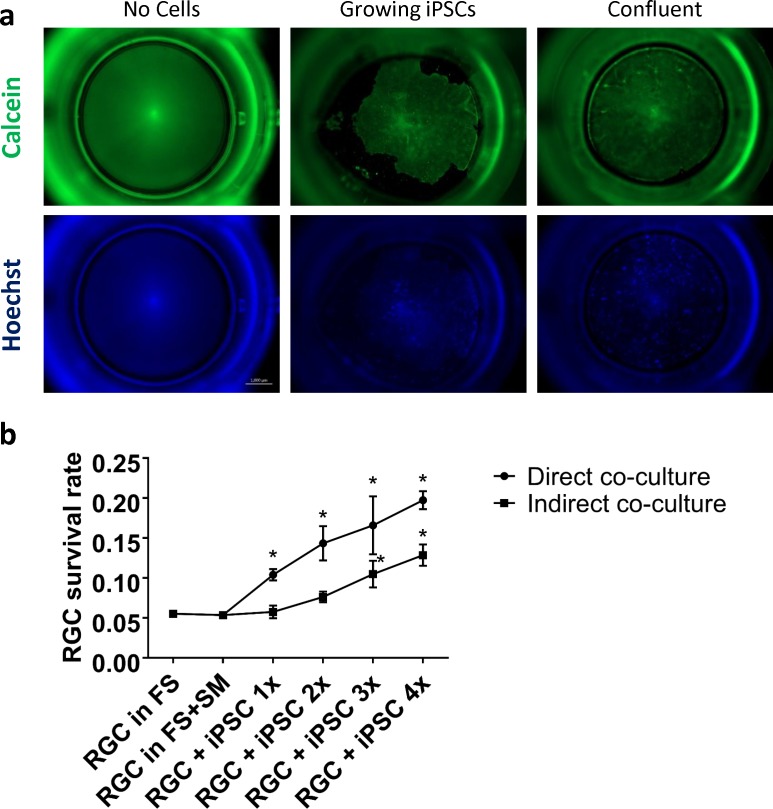
We next examined whether indirect coculture, in which the iPSCs are kept in hanging inserts away from direct RGC contact (Fig. 2a), would have a similar effect. Indirect RGC-iPSC cocultures also displayed a similar dose-dependent increase in RGC survival, but to a lesser extent than the direct coculture, as shown by live/dead assays (Fig. 2b; P < 0.05; N = 3 experiments; n > 1000 cells/experiment). Thus, RGC-iPSC indirect coculture also promotes RGC survival, but less than observed in direct coculture, suggesting the hypothesis that at least part of the survival effect is mediated by soluble factors released by the iPSCs. In addition, we found that RGCs showed slightly poorer survival in culture plates with hanging inserts, even if no cells were added into the inserts. This finding was further validated through head-to-head tests (results not shown).
Figure 2.
RGC-iPSC indirect coculture promotes RGC survival, but with lower yield than direct coculture. (a) For indirect coculture, iPSCs were plated in hanging inserts. (b) Live/dead assays showed that both direct and indirect coculture promoted RGC survival. However, direct coculture showed better rates of RGC survival rate (*P < 0.05; Bonferroni-corrected paired t-test; N = 3 experiments; n > 1000 cells/experiment). FS, full sato (RGC growth medium); SM, stemMACS (iPSC growth medium). Error bar denotes SD.
iPSCs Promote Transplanted RGC Survival After Ex Vivo and In Vivo Transplant to Adult Retina
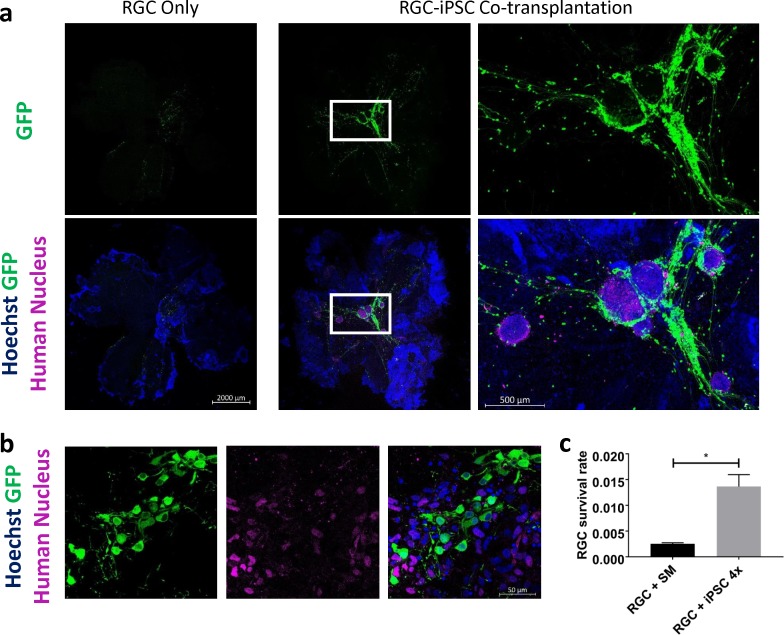
We next used an ex vivo retinal explant model of RGC transplant3 to examine whether iPSCs would influence RGC survival after transplant. Transplanted RGCs were identified by GFP expression; transplanted iPSCs were identified by expression of a human nuclear antigen detected by immunofluorescence staining (Figs. 3a, 3b). Most live iPSCs were detected in oval-shaped or irregular colonies, whereas others were widely scattered; most live transplanted RGCs, as well as their neurites, were found in close proximity to iPSCs (Figs. 3a, 3b). We quantified transplanted RGC survival with and without cotransplanted iPSCs and found significantly improved transplanted RGC survival after RGC-iPSC cotransplant to adult retinas ex vivo (Fig. 3c, P < 0.001; N = 3 experiments; n = 40,000 cells/experiments).
Figure 3.
RGC-iPSC 1-week ex vivo coculture promotes transplanted RGC survival. (a) Human iPSCs significantly increased RGC survival, especially the RGCs surrounding iPSC colonies. (b) No transplanted RGC-iPSC cytoplasmic exchange (GFP+/Human Nucleus+) was detected. (c) Difference between transplanted RGC survival rates with and without cotransplanted iPSCs was quantified (*P < 0.001, paired t-test; N = 3 experiments; n = 40,000 cells/experiment). SM, stemMACS (iPSC growth medium). Error bar denotes SD.
We also asked whether the GFP+ RGCs were “real” transplanted cells, because of increasing concerns about cell-cell cytoplasmic exchange with which the results would be illusive. To address whether cytoplasmic exchange occurred between the two types of transplanted cells, we screened all the retinal explants under high magnification (40×) for the presence of GFP+/human nucleus+ (i.e. double-positive) cells but did not find any (e.g., inset magnified in Fig. 3b).
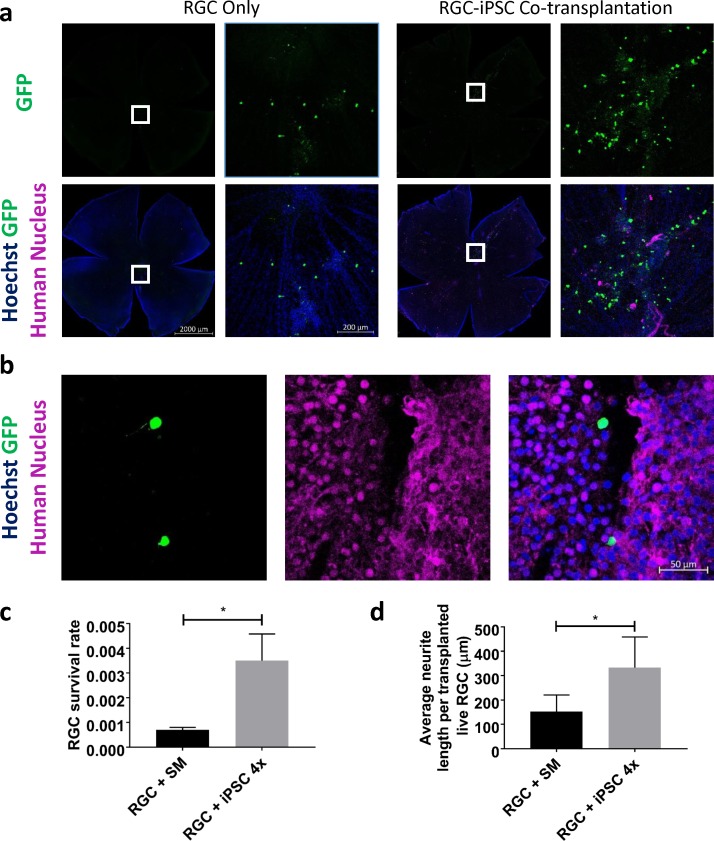
Finally, we asked whether cotransplant of iPSCs would increase RGC survival after transplant in vivo, injecting transplanted cells intravitreally and then harvesting retinas for immunostaining and quantification 7 days later. Twenty percent of these transplantation experiments showed retinal engraftment after intravitreal delivery, similar to our prior in vivo transplant experiments,4 and in the successful 20%, transplanted cells typically distributed on approximately one-fifth to one-third of the host retina. In contrast to the explanted retina model, transplanted RGCs found at the retina after in vivo transplant were still close to but not restricted to the proximity of iPSCs, which themselves were less likely to grow in colonies after in vivo transplant (Figs. 4a, 4b). Again, no cytoplasmic exchange between two transplanted cells was observed (Fig. 4b). Similar to our ex vivo transplantation results, RGCs transplanted with iPSCs in vivo demonstrated a significantly higher survival (Fig. 4c; P < 0.001; N = 3 experiments; n = 40,000 cells/experiment). Cotransplantation also promoted a significantly higher average neurite length of transplanted RGCs compared with RGC-only transplantation (Fig. 4d; P < 0.001; N = 3 experiments; n > 26 cells/experiment). Thus, RGC-iPSC cotransplantation promotes transplanted RGC survival and neurite extension in vivo.
Figure 4.
RGC-iPSC in vivo cotransplantation promotes transplanted RGC survival and neurite extension. (a) RGCs transplanted with iPSCs showed significantly higher survival rates, compared with RGC-only transplant 1 week after transplantation in vivo. (b) No transplanted RGC-iPSC cytoplasmic exchange (GFP+/Human Nucleus+) was detected. (c) Difference between transplanted RGC survival rates with and without cotransplanted iPSCs was quantified (*P < 0.001, paired t-test, N = 3 experiments; n = 40,000 cells/experiment). (d) Donor RGCs with cotransplanted iPSCs grew significantly longer neurites than those from RGC-only transplantation in vivo (*P < 0.001, paired t-test; N = 3 experiments; n > 26 cells/experiment). SM, stemMACS (iPSC growth medium). Error bar denotes SD.
Discussion
The present study demonstrates that human iPSCs possess a potent neuroprotective capacity capable of enhancing mouse donor RGC survival on adult rat retinas after transplant ex vivo and in vivo, and this may be attributable at least in part to iPSCs' secretion of one or more neuroprotective compounds. These data extend previous findings on stem cell transplantation for endogenous (host) RGC neuroprotection. For example, MSCs have been shown to promote endogenous RGC survival in experimental glaucoma models,5,7 and a similar RGC neuroprotection in a rat optic nerve crush model was observed with human umbilical cord blood stem cells transplanted intravitreally.11 Our study indicates that iPSCs also can confer neuroprotection for RGCs in vitro and in vivo, although future experiments could directly compare iPSCs' and MSCs' or other transplanted cells' capacity in animal models of glaucoma or trauma.
The survival rates of transplanted RGCs in this study were lower than in some of our previous data,3 potentially due to the differences between the cell medium, syringes and needles being used. We made these modifications in this study, as in our recent publication,4 because we found the current protocol enhanced the proportion of successful transplants into the retina, although the survival and neurite growth for example may have suffered in return. Also, the mixture of the RGC- and stem cell–optimized media is hypothesized to improve the injection milieu for both RGCs and iPSCs, although it might affect RGC survival and integration on the retina after injection.
Recently, cell-to-cell cytoplasmic exchange has been widely discussed in interpreting cell transplant experiments,12,13 as it could confound interpretation of what is a transplanted cell. Here we studied this question by looking for coimmunostaining of GFP marking RGCs and human nuclear antigen marking iPSCs both ex vivo and in vivo and found no evidence for cytoplasmic exchange. Although we did not specifically stain for cytoplasmic exchange between donor RGCs and recipient host cells, we did not find any GFP+ RGCs that resembled typical native RGCs in terms of small round-shaped cell morphology or axon targeting to the optic nerve head. Although this could have occurred in rare instances, such cytoplasmic exchange has not yet been documented between postmitotic neurons. Another interesting finding is that, although most transplanted iPSCs retained typical stem cell–like morphologies, some of them, especially those that were dissociated, transformed into different morphologies, suggesting at least some measure of differentiation after transplant. Whether and into what those iPSCs differentiated presents another question to be studied in future experiments, ideally using lines that express cell type–specific markers after differentiation.14
These data also suggest that iPSCs may promote transplanted RGC survival through secretion of neuroprotective compounds, consistent with previous MSC studies.5,7 MSCs secrete a number of neuroprotective proteins, with platelet-derived factor regarded as the strongest one.15 Other studies have found that some stem cells, including MSCs and neural stem cells, secrete exosomes that carry neuroprotective compounds.16 However, the mechanisms of iPSC's neuroprotective capacity are still not clear. In addition, our data showed that, although RGC-iPSC indirect coculture enhanced RGC survival, it was less potent than direct coculture, suggesting that cell–cell interaction might also confer additional promotion of RGC survival.
To our knowledge, this study is the first cell-level approach for protecting primary grafted CNS neurons. Transplanted RGCs, unlike endogenous RGCs, are exposed in the vitreous to a hostile environment and receive no trophic support from other retinal cells before they integrate into host retina, and perhaps form synapses with host cells.4 Limited survival rates likely limit retinal integration after transplant.3 We also noted that the transplanted RGCs showed much less axon targeting reaching the optic nerve head by 1 week, in contrast to a few RGCs that successfully grew axons to the optic nerve head at longer survival times in our previous findings,4 highlighting the need in future work to extend these time courses. Here our study demonstrates that human iPSCs enhance donor RGC survival as well as neurite growth of donor RGCs after transplant. On the other hand, many more questions need to be addressed with regard to potential therapeutic application (e.g., whether cotransplanted iPSCs enhance other key measures of RGC including integration synapse formation, electrophysiologic response to light, neurite growth and orientation toward the optic nerve, and what level of transplant efficiency is needed to protect or restore function in normal and disease model recipient retinas), which should be examined in future studies.
Acknowledgments
Supported by the China Scholarship Council (201606100222), the BrightFocus Foundation, the National Eye Institute (P30-EY026877), and Research to Prevent Blindness, Inc.
Disclosure: S. Wu, None; K.-C. Chang, None; M. Nahmou, None; J.L. Goldberg, None
References
- 1. Sulzer D. . Multiple hit hypotheses for dopamine neuron loss in Parkinson's disease. Trends Neurosci. 2007; 30: 244– 250. [DOI] [PubMed] [Google Scholar]
- 2. Cohen LP, Pasquale LR. . Clinical characteristics and current treatment of glaucoma. Cold Spring Harbor Perspect Med. 2014; 4: 1– 16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Hertz J, Qu B, Hu Y, Patel RD, Valenzuela DA, Goldberg JL. . Survival and integration of developing and progenitor-derived retinal ganglion cells following transplantation. Cell Transplant. 2014; 23: 855– 872. [DOI] [PubMed] [Google Scholar]
- 4. Venugopalan P, Wang Y, Nguyen T, Huang A, Müller KJ, Goldberg JL. . Transplanted neurons integrate into adult retinas and respond to light. Nature Commun. 2016; 7: 10472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Johnson TV, Martin KR. . Cell transplantation approaches to retinal ganglion cell neuroprotection in glaucoma. Curr Opin Pharmacol. 2013; 13: 78– 82. [DOI] [PubMed] [Google Scholar]
- 6. Pearson C, Martin K. . Stem cell approaches to glaucoma: from aqueous outflow modulation to retinal neuroprotection. Progr Brain Res. 2015; 220: 241– 256. [DOI] [PubMed] [Google Scholar]
- 7. Johnson TV, Bull ND, Hunt DP, Marina N, Tomarev SI, Martin KR. . Neuroprotective effects of intravitreal mesenchymal stem cell transplantation in experimental glaucoma. Invest Ophthalmol Vis Sci. 2010; 51: 2051– 2059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Barres BA, Silverstein BE, Corey DP, Chun LL. . Immunological, morphological, and electrophysiological variation among retinal ganglion cells purified by panning. Neuron. 1988; 1: 791– 803. [DOI] [PubMed] [Google Scholar]
- 9. Meyer-Franke A, Kaplan MR, Pfrieger FW, Barres BA. . Characterization of the signaling interactions that promote the survival and growth of developing retinal ganglion cells in culture. Neuron. 1995; 15: 805– 819. [DOI] [PubMed] [Google Scholar]
- 10. Suzuki T, Akimoto M, Imai H,et al. Chondroitinase ABC treatment enhances synaptogenesis between transplant and host neurons in model of retinal degeneration. Cell Transplant. 2007; 16: 493– 503. [DOI] [PubMed] [Google Scholar]
- 11. Zhao T, Li Y, Tang L, Li Y, Fan F, Jiang B. . Protective effects of human umbilical cord blood stem cell intravitreal transplantation against optic nerve injury in rats. Graefe's Arch Clin Exp Ophthalmol. 2011; 249: 1021– 1028. [DOI] [PubMed] [Google Scholar]
- 12. Pearson RA, Gonzalez-Cordero A, West EL,et al. Donor and host photoreceptors engage in material transfer following transplantation of post-mitotic photoreceptor precursors. Nature Commun. 2016; 7: 13029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Santos-Ferreira T, Llonch S, Borsch O, Postel K, Haas J, Ader M. . Retinal transplantation of photoreceptors results in donor-host cytoplasmic exchange. Nature Commun. 2016; 7: 13028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Sluch VM, Chamling X, Liu MM,et al. Enhanced stem cell differentiation and immunopurification of genome engineered human retinal ganglion cells. Stem Cells Translational Med. 2017; 6: 1972– 1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Johnson TV, DeKorver NW, Levasseur VA,et al. Identification of retinal ganglion cell neuroprotection conferred by platelet-derived growth factor through analysis of the mesenchymal stem cell secretome. Brain. 2014; 137: 503– 519. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Phinney DG, Pittenger MF. . Concise review: MSC-derived exosomes for cell-free therapy. Stem Cells. 2017; 35: 851– 858. [DOI] [PubMed] [Google Scholar]