Abstract
Serotonin is perhaps best known as a neurotransmitter that modulates neural activity and a wide range of neuropsychological processes, and drugs that target serotonin receptors are used widely in psychiatry and neurology. However, most serotonin is found outside the central nervous system, and virtually all of the 15 serotonin receptors are expressed outside as well as within the brain. Serotonin regulates numerous biological processes including cardiovascular function, bowel motility, ejaculatory latency, and bladder control. Additionally, new work suggests that serotonin may regulate some processes, including platelet aggregation, by receptor-independent, transglutaminase-dependent covalent linkage to cellular proteins. We review this new “expanded serotonin biology” and discuss how drugs targeting specific serotonin receptors are beginning to help treat a wide range of diseases.
Keywords: 5-HT, receptor, SSRI, behavior, cardiovascular, gastrointestinal
INTRODUCTION
Although serotonin (5-hydroxytryptamine, 5-HT) was discovered 60 years ago (1), the study of serotonin and its receptors continues to yield new biological insights of medical relevance in virtually all major organ systems, including the cardiovascular, pulmonary, gastrointestinal (GI), and genitourinary systems as well as the central nervous system (CNS) (2). Serotonin and serotonin receptors are important in the regulation of virtually all brain functions, and dysregulation of the serotonergic system has been implicated in the pathogenesis of many psychiatric and neurological disorders (3, 4).
A greater understanding of serotonin function has emerged during the last two decades with the cloning of at least 15 serotonin receptors, which are grouped into seven families based on signaling mechanisms (5). Other important advances have included the subsequent development of receptor-specific knockout mice, and the development of receptor subtype-selective drugs. These advances have also shown us that serotonin has critically important functions in many human organ systems outside the CNS, including the regulation of energy balance and food intake, GI and endocrine function, and cardiovascular and pulmonary physiology. These findings may help explain the diverse side effects of serotonergic drugs—from diabetes and metabolic syndrome to valvular heart disease (6, 7). These recent findings also imply that developers of new serotonin receptor subtype-selective drugs will need to consider the roles of a given receptor subtype in the physiology of multiple organ systems. In time, these advances may lead to therapies with improved efficacy and side-effect profiles, and will enhance our understanding of a variety of neuropsychiatric and medical disorders.
Here we review how serotonin and its cognate receptors regulate the function of multiple human organ systems and disease processes. We also highlight specific settings where new serotonergic drugs may be introduced to medical practice in the future.
FROM BRAIN TO BEHAVIOR
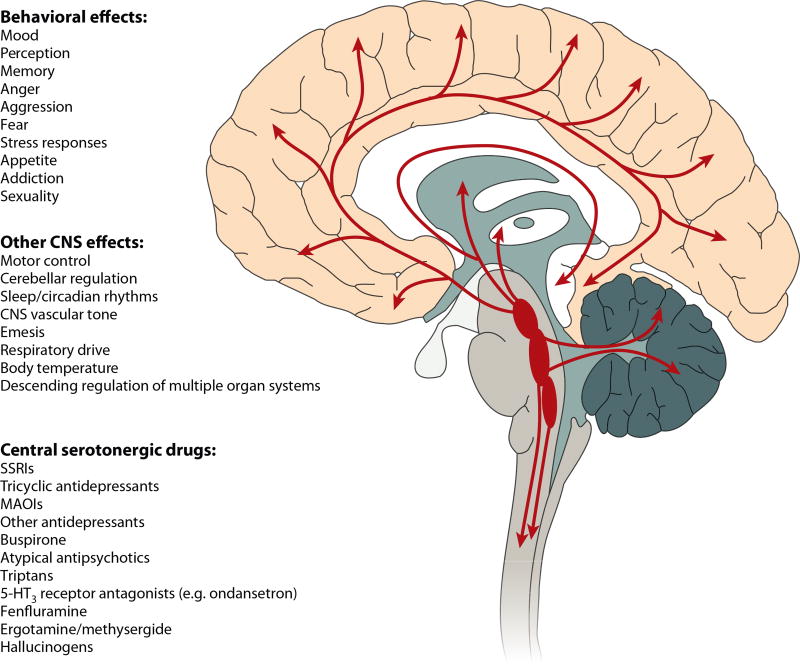
Serotonin modulates virtually all human behavioral processes. This finding may seem surprising given that less than one in a million CNS neurons produce serotonin and the vast majority of total body serotonin is found outside the CNS (8). However, brainstem serotonin neurons send ascending projections that terminate in a defined and organized manner in cortical, limbic, midbrain, and hindbrain regions (Figure 1). Indeed, all brain regions express multiple serotonin receptors in a receptor subtype-specific fashion (9). Additionally, individual neurons may express multiple serotonin receptors. For instance, Layer V pyramidal neurons express 5-HT1A and 5-HT2A receptors, which exert opposing effects on pyramidal neuron firing (10). CNS serotonin neurons are thus ideally positioned to modulate the activity of a wide variety of human brain circuits, which explains, in part, the pleiotropic behavioral effects of brain serotonin (9).
Figure 1.
Central serotonergic pathways, effects, and drugs. In the central nervous system (CNS), serotonin is almost exclusively produced in neurons originating in the raphe nuclei located in the midline of the brainstem. These serotonin-producing neurons form the largest and most complex efferent system in the human brain. The most caudal raphe innervate the spinal cord, while the more rostral raphe, the dorsal raphe nucleus and the medial raphe nucleus, innervate much of the rest of the CNS by diffuse projections. Indeed, virtually every cell in the brain is close to a serotonergic fiber, and nearly all behaviors as well as many other brain functions are regulated by serotonin. Not surprisingly, serotonin receptors and transporters are a major focus of CNS drug development, and many current medications modulate serotonin neurotransmission. 5-HT, serotonin; MAOI, monoamine oxidase inhibitor; SSRI, selective serotonin reuptake inhibitor.
The behavioral and neuropsychological processes modulated by serotonin include mood, perception, reward, anger, aggression, appetite, memory, sexuality, and attention, among others. Indeed, it is difficult to find a human behavior that is not regulated by serotonin. (A full discussion of how serotonin modulates the neural circuitry of behavior and emotion is beyond the scope of this review; see, e.g., References 11–13.) Although the neural circuitry responsible for each of these behavioral processes is still being elucidated, in many cases there is at least one specific brain region or nucleus that is critical for a given behavior. The expression pattern of each serotonin receptor within the human CNS is also known (9). Thus, the question of how serotonin modulates each behavioral process can usually be framed in terms of how specific serotonin receptors modulate the specific brain region(s)/nuclei involved in producing the behavioral output.
Just as each behavior is regulated by multiple serotonin receptors, each serotonin receptor is expressed in multiple brain regions and likely contributes to the modulation of multiple behavioral processes. For example, anxiety-like behavior is regulated primarily by 5-HT1A and 5-HT2C receptors, among others (14, 15), but the 5-HT2C receptor regulates not only anxiety but also reward processing, locomotion, appetite, and energy balance (16). This principle explains why drugs targeting a specific serotonin receptor nonetheless have effects on multiple behavioral processes (16).
Despite this lack of a one-to-one correspondence between specific serotonin receptors and individual behavioral processes, various drugs targeting central serotonergic activity and serotonergic receptors are currently used clinically or are in clinical development for the treatment of nearly every neuropsychiatric disorder (17, 18).
SEROTONIN AND VASCULAR BIOLOGY
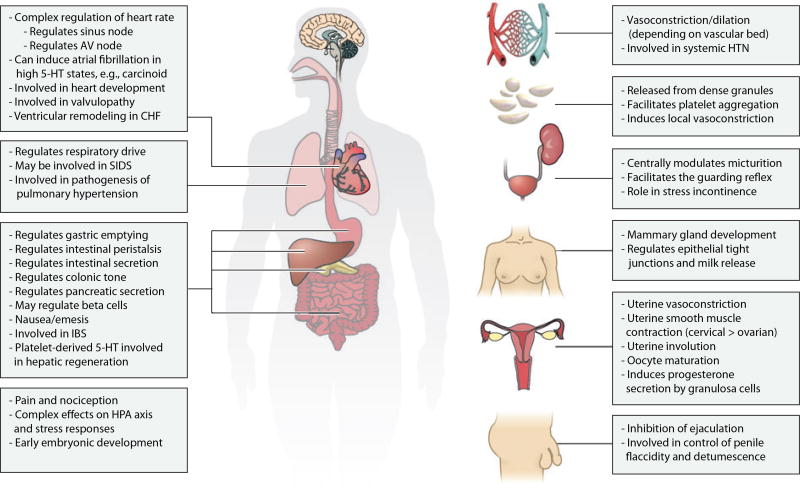
Both within the CNS and throughout the body, serotonin plays a number of roles in vascular biology, ranging from the control of vascular resistance and blood pressure to the control of hemostasis and platelet function (Figure 2). Serotonin causes vasoconstriction or vasodilation in different vascular beds depending on the particular receptors that are expressed in each vessel wall and surrounding smooth muscle tissue (19). Indeed, activation of 5-HT1B receptors on cerebral blood vessels causes vasodilation, which may partly explain the analgesic effects of the triptan antimigraine drugs (20).
Figure 2.
Myriad effects of serotonin outside the central nervous system. 5-HT, serotonin; AV, atrioventricular; CHF, congestive heart failure; HPA, hypothalamic-pituitary-adrenal; HTN, hypertension; IBS, irritable bowel syndrome; SIDS, sudden infant death syndrome.
Platelets have significant vesicular serotonin stores but lack the enzymes to synthesize serotonin (21); instead, they take up serotonin from the plasma via the serotonin transporter. Serotonin is then secreted by the platelet dense granules during platelet activation and plays a role in promoting platelet aggregation and vasoconstriction of surrounding blood vessels, facilitating hemostasis. Selective serotonin reuptake inhibitors (SSRIs) can increase bleeding time by inhibiting the uptake and storage of platelet serotonin, so caution should be used in patients at high risk for bleeding or on anticoagulants. Indeed, platelets from individuals treated with SSRIs, as well as platelets from serotonin transporter knockout mice, show decreased aggregation responses (22).
Accumulating data suggest that SSRI treatment may decrease myocardial infarction (MI) risk. Several case-control studies have observed lower MI rates among depressed patients taking SSRIs versus controls, but not among patients taking tricyclic antidepressants (23). A retrospective secondary analysis of post-MI patients randomized to receive SSRIs or placebo showed decreases in recurrent MIs and all-cause mortality among patients on SSRIs (24). Although depression is also an independent risk factor for MIs that may be related to altered serotonin biology (25), the idea that SSRIs may actually reduce MIs is intriguing and still awaits prospective testing (26).
Interestingly, recent studies suggest that intracellular serotonin may also play a role in platelet activation through covalent linkage to small G proteins via tissue transglutaminase. This modification constitutively activates G protein–dependent signaling pathways and stimulates platelet aggregation (27). In addition, serotonin is covalently cross-linked to a variety of adhesion proteins and clotting factors on the platelet cell surface (28), a process essential for the activation of a subset of platelets. These findings have expanded the classical paradigm, in which serotonin works by noncovalent interactions with membrane-bound receptors, and creates many new questions about whether serotonin can exert biological activity by covalently attaching to cellular proteins in other organ systems, including the brain.
SEROTONIN AND CARDIAC FUNCTION
Serotonin regulates several different aspects of cardiac function, ranging from electrical conduction to valvular closure to post–MI remodeling (Figure 2). Studies of cardiac abnormalities in patients with serotonin-producing carcinoid tumors provided early evidence that serotonin modulates heart function. High serotonin levels in these patients can cause atrial fibrillation (29), an effect that may be mediated by cardiac 5-HT4 receptors (30).
Aside from this role in the atria, 5-HT4 receptor expression increases in failing cardiac ventricles (31), and this may play a role in ventricular remodeling. Animal studies suggest that 5-HT4 antagonists may help improve cardiac function and block pathological remodeling in congestive heart failure (32), a possibility that awaits clinical trials in humans. Meanwhile, 5-HT2A antagonists may have utility in treating vasospastic angina and ischemic heart disease, and 5-HT3 antagonists have been reported to be useful in treating post-MI pain (33).
Serotonin also plays a pathological role in the cardiac valvulopathy caused by the appetite suppressant fenfluramine (see sidebar, Serotonergic Drugs andValvular Heart Disease) (7, 34). 5-HT2B receptor activation on valvular interstitial cells is mitogenic (35), increasing valve leaflet area and causing the poor valve closure seen in patients exposed to these drugs. It has been suggested that the valvulopathy and fibrosis resulting from carcinoid syndrome may have a similar etiology (7). Mice lacking the 5- HT2B receptor either die of cardiac defects or develop dilated cardiomyopathy in adulthood (36). Thus, the 5-HT2B receptor plays an important role in cardiac development as well as adult cardiac valvular function.
SEROTONIN, BREATHING, AND PULMONARY ARTERY HYPERTENSION
Serotonin helps control breathing and respiratory drive through effects on brainstem respiratory control centers as well as on the pulmonary vasculature (Figure 2). In pulmonary artery hypertension (PAH), hypoxia elevates plasma serotonin levels (37) and likely increases mitogenic 5-HT2B receptor signaling on pulmonary artery endothelial cells. Increased 5-HT2B receptor signaling increases vascular resistance (38) and is necessary for the development of PAH, since mice deficient for 5-HT2B do not develop PAH (39). One patient with a heterozygous activating 5-HT2B receptor mutation developed PAH (40), suggesting that increased 5-HT2B receptor signaling may be sufficient to cause PAH. Indeed, 5-HT2B antagonists can prevent the development of increased pulmonary vascular resistance, suggesting that these drugs could be useful clinically in treating early PAH (38).
Serotonin may also induce pulmonary artery remodeling by direct covalent attachment to intracellular signaling proteins in vascular smooth muscle cells (41), similar to the way serotonin influences platelet function by receptor-independent covalent attachment to proteins. Indeed, acute blockade of the serotonin transporter by SSRIs can block the development of PAH and subsequent right ventricular hypertrophy in animal models (42), suggesting that intracellular rather than extracellular serotonin may be an etiological factor in PAH.
Serotonin also modulates the activity of rhythm-generating respiratory neurons in the brainstem pre-Boetzinger complex via the 5-HT4 receptor. Opioid analgesic drugs cause respiratory depression by suppressing the activity of these cells via mu opioid receptors, whereas activation of the 5-HT4 receptor is excitatory (43). These findings have led to the intriguing idea that 5-HT4 agonists could be used to block opioid-induced respiratory depression while still leaving opioid-induced analgesia intact (44).
Serotonergic abnormalities have also been found in roughly 50% of infants who have died from sudden infant death syndrome (SIDS) (45). These infants have significantly more medullary serotonin neurons but significantly less expression of serotonin transporter and 5-HT1A receptor in these cells (46). Medullary serotonin neurons are highly pH sensitive and serve as central chemoreceptors (47), suggesting that these abnormalities may cause a defective respiratory response to hypercapnia that may underlie SIDS. Mice with defective development of serotonergic neurons have a SIDS-like syndrome (48).
SEROTONIN, ENDOCRINOLOGY, AND METABOLISM
The functions of serotonin in the endocrine system and metabolism range from the central control of energy balance and central modulation of the hypothalamic-pituitary-adrenal (HPA) axis to the direct regulation of mammary gland development (Figure 2). Several lines of evidence suggest an important role for hypothalamic 5-HT2C receptors in regulating energy balance and modulating glucose homeostasis (49). In particular, hypothalamic 5-HT2C and 5-HT1B receptors act by modulating melanocortin pathways, and serotonin release into the hypothalamus stimulates sympathetic nerves that innervate brown adipose tissue (50). Because of these mechanisms, 5-HT2C receptor agonists may be useful for treating obesity and diabetes (51). Serotonin also plays a role in setting overall metabolic rate and temperature control. Serotonin-deficient mice show a rapid hypothermic response when placed in a cold environment (52), a response that may be mediated by hypothalamic 5-HT1A and 5-HT7 receptors (53). Serotonin regulates the HPA axis at multiple levels (54) and thus has complex effects on the overall stress response.
Serotonin has also been implicated in the development and regeneration of metabolic and endocrine organs. For example, serotonin is synthesized within the developing mammary gland, where it is part of an autocrine-paracrine loop that is essential for mammary gland development (55). In the adult mammary gland, serotonin regulates epithelial tight junctions and milk release (56). In the liver, serotonin is important in regeneration following transection or volume loss. In particular, platelet-derived serotonin signals through 5-HT2A and 5-HT2B receptors to promote liver regeneration (57).
SEROTONIN AND THE GASTROINTESTINAL SYSTEM
Serotonin regulates digestion at multiple levels within the human GI system and throughout the phylogenetic spectrum (Figure 2) (58). Roughly95%of total body serotonin is released into the gut by intestinal enterochromaffin cells (8), but serotonin is involved at the very moment that food enters the body. Activation of taste-bud cells on the tongue causes serotonin release onto sensory afferent nerves (59) that transmit taste information to the CNS. Once food enters the GI tract, it is propelled along by peristaltic waves; these waves, as well as intestinal motility and secretion, are modulated by serotonin (reviewed extensively in Reference 8). For example, intestinal serotonin regulates pancreatic enzyme secretion (60), a mechanism by which the gut may communicate exocrine enzyme needs to the pancreas based on GI contents.
Altered serotonin signaling has been implicated in functional bowel disorders, including irritable bowel syndrome (IBS) (8). Drugs targeting both the 5-HT3 and 5-HT4 receptors have been used to treat IBS. In addition, excessive GI serotonin release can activate 5-HT3 receptors on afferent vagal nerves that innervate brainstem vomiting centers (61), which may partly explain why 5-HT3 antagonists such as ondansetron are effective antiemetics. SEROTONIN AND PAIN CONTROL, ANESTHESIA, AND SPINAL NOCICEPTION Serotonin modulates pain perception and nociceptive processing at multiple levels within the central and peripheral nervous systems (Figure 2). Within locally inflamed tissue, serotonin release sensitizes peripheral nerve fibers that carry nociceptive information to the CNS (62). Brainstem serotonin neurons send descending projections into the spinal cord that modulate incoming nociceptive information (63). Finally, brainstem raphe serotonin neurons send ascending projections to cortical and limbic regions that may modulate the psychological perception of pain (64).
Serotonergic abnormalities have been reported in patients with mood disorders, and altered serotonergic modulation of pain processing at these multiple levels may explain increased pain perception in these patients (64). The multiple levels at which serotonin modulates nociceptive processing and pain perception may also explain the efficacy of serotonergic drugs in treating pain disorders. For example, triptan drugs are thought to relieve migraine symptoms by activating thalamic 5-HT1B and 5-HT1D receptors (20), whereas tricyclic antidepressants and combined serotonin/ norepinephrine reuptake inhibitors such as duloxetine may work via the serotonergic modulation of incoming nociceptive information in the spinal cord (64).
The serotonin system also plays an important role in anesthesia response. Inhalational anesthetic agents may work in part by suppressing serotonin release, and patients taking serotonergic antidepressants may require increased dosage of these agents (65).
Patients taking serotonergic antidepressants are also at increased risk of developing serotonin syndrome, a poorly understood syndrome involving altered mental status, autonomic instability, and neuromuscular rigidity, when exposed to multiple drugs in an inpatient setting (66).
SEROTONIN AND GENITOURINARY FUNCTION
Serotonin plays a variety of roles in the central and peripheral control of genitourinary function within both the brain and the spinal cord (Figure 2). Serotonin increases ejaculatory latency and delays orgasm through 5-HT2C and 5-HT1B receptors but also decreases ejaculatory latency through the 5-HT1A receptor (67). The net effect of serotonin is to prolong ejaculatory latency and to delay orgasm, and thus SSRIs are often prescribed off-label to treat premature ejaculation (68). Although all SSRIs prolong ejaculatory latency to some degree, this effect is most pronounced with paroxetine, and there is no direct correlation between antidepressant efficacy and antiejaculatory latency.
Serotonin modulates micturition in a similar fashion as it does ejaculation. It controls urinary function via actions in the brain and spinal cord, and it regulates parasympathetic neural input to the bladder and somatic input to the external urinary sphincter. 5-HT2C receptors prevent urination whereas 5-HT1A receptors promote urination (69). The net effect of serotonin on urination is likely inhibitory, since combined serotonin-norepinephrine reuptake inhibitors are used clinically to treat stress incontinence (70).
SEROTONIN, REPRODUCTIVE FUNCTION, AND PREGNANCY
Increased serotonin levels are found in the serum of pregnant women and may play a role in the altered vascular physiology of pregnancy (71). Roughly tenfold increases in serotonin have been observed in the serum of preeclamptic women, and serotonin levels correlate with the severity of preeclampsia. These findings led some to propose nearly 50 years ago that elevated serotonin may cause preeclampsia (reviewed in Reference 71). The elevated serotonin in preeclamptic patients likely stems from increased platelet activation and aggregation as well as decreased metabolism by monoamine oxidase. The altered vascular tone seen in preeclampsia in high-serotonin states may be mediated by 5-HT1B and 5-HT1D receptors (72); if so, blocking these serotonin receptors or blocking platelet serotonin accumulation with SSRIs could help treat preeclampsia (73). Interestingly, although SSRIs can increase hemorrhage risk, recent data show that pregnant women taking SSRIs are not at increased risk of postpartum hemorrhage (74). However, exposure to SSRIs during pregnancy may increase the newborn’s risk of developing persistent pulmonary hypertension (75).
Serotonin also regulates uterine contraction through 5-HT2A receptors (Figure 2) (76). 5-HT2A receptor–induced uterine contractions favor the cervical end of the uterus, suggesting a role in promoting sperm transport toward the oviduct (77). In addition, serotonin has been shown to induce uterine collagenase expression after delivery, which promotes uterine involution (78).
CONCLUSIONS
As these examples show, serotonin is much more than a neurotransmitter essential for the modulation of mood. It regulates a wide range of physiologic and pathophysiologic processes in most human organs. This explains why serotonergic drugs modulate phenomena ranging from ejaculatory latency to hemostatic function in addition to their more commonly appreciated effects on mood and cognition.
Serotonin typically regulates a given physiologic process (such as digestion, pain perception, or energy balance) at multiple steps through different and frequently opposing mechanisms. For example, serotonin is not simply pro- or antianalgesic; instead, it both potentiates and inhibits nociceptive processing at various levels of the nervous system.
Why is serotonergic modulation of these processes so complex? One possibility is that this complexity allows wide modulatory activity of biological processes while preserving homeostasis even under diverse environmental conditions. For example, depleting serotonin acutely has little effect on mood in normal individuals (79), even though serotonin receptors modulate a number of neural pathways that subserve mood. At the same time, serotonergic dysregulation is seen in multiple disease processes ranging from IBS to depression, and correction of specific serotonin receptor signaling abnormalities can often help treat the disease in question.
The richness and complexity of serotonergic modulation of physiologic and pathophysiologic processes discussed here provide both a pharmacologic opportunity and challenge. On the one hand, the involvement of specific serotonin receptors in a given process provides an opportunity to pharmacologically target these specific receptors in a related disease state. On the other hand, the fact that each individual serotonin receptor is involved in multiple physiologic processes also presents a challenge, since even a drug targeting a single serotonin receptor is likely to have effects on multiple body systems. For example, although 5-HT4 agonists may have rapid-onset antidepressant activity within the CNS, their clinical use may be tempered by their effects on other organ systems, such as on GI motility and on cardiac hypertrophy in heart failure.
As research on the role of specific serotonin receptors in human physiology progresses, the difficulty of this challenge will become clear. In the process, we will likely gain new serotonergic drugs and disease treatments as well as a deeper understanding of the beauty and complexity of human biology.
SEROTONERGIC DRUGS AND VALVULAR HEART DISEASE.
The drug combination of fenfluramine/phentermine was exceptionally effective for inducing weight loss in obese individuals (80), although this advantage was offset by the induction of valvular heart disease by fenfluramine (81), which led to the withdrawal of fenfluramine. In 2000, we and others discovered that a metabolite of fenfluramine—norfenfluramine, a potent activator of 5-HT2B receptors—was likely responsible for the valvulopathy induced by fenfluramine and the “fen/phen” combination (34, 82). We subsequently screened a small library of available drugs and identified certain ergolines (pergolide and cabergoline) and amphetamine derivatives [methylenedioxymethamphetamine (MDMA; “Ecstasy”)] as potent 5-HT2B agonists (35).We predicted that they too would be associated with valvular heart disease with chronic use (for review see References 7 and 83). Subsequent clinical studies have indicated that pergolide, cabergoline, and MDMA use are associated with valvular heart disease indistinguishable from that induced by fenfluramine (84–86). Based on these findings, we have recommended that all approved and candidate medications be screened at 5-HT2B receptors for agonist actions prior to use in humans (7, 34).
Acknowledgments
We thank Laurence Tecott for helpful discussions. Work on 5-HT receptor pharmacology is supported by RO1MH61887 and U19MH82441 and by the National Institute of Mental Health Psychoactive Drug Screening Program (all grants to B.L. Roth).
Footnotes
DISCLOSURE STATEMENT
M. Berger personally owns stock in Arena Pharmaceuticals. J.A. Gray has no conflicts. B.L. Roth, since 2006, has consulted with the following pharmaceutical companies on 5-HT receptor pharmacology: Merck, Johnson & Johnson, GlaxoSmithKline, AMRI, Supernus, Labopharm, Wyeth-Solvay Alliance, Roche, Mediavation, and DaiNipponSumitomo. Dr. Roth is listed as coinventor on several patents related to the use of 5-HT receptor subtype-selective drugs in treating human disease.
LITERATURE CITED
- 1.Rapport MM, Green AA, Page IH. Serum vasoconstrictor, serotonin; isolation and characterization. J. Biol. Chem. 1948;176:1243–51. [PubMed] [Google Scholar]
- 2.Roth BL, editor. The Serotonin Receptors: From Molecular Pharmacology to Human Therapeutics. Totowa, NJ: Humana; 2007. [Google Scholar]
- 3.Roth BL. Multiple serotonin receptors: clinical and experimental aspects. Ann. Clin. Psychiatry. 1994;6:67–78. doi: 10.3109/10401239409148985. [DOI] [PubMed] [Google Scholar]
- 4.Roth BL, Xia Z. Molecular and cellular mechanisms for the polarized sorting of serotonin receptors: relevance for genesis and treatment of psychosis. Crit. Rev. Neurobiol. 2004;16:229–36. doi: 10.1615/critrevneurobiol.v16.i4.10. [DOI] [PubMed] [Google Scholar]
- 5.Kroeze WK, Kristiansen K, Roth BL. Molecular biology of serotonin receptors: structure and function at the molecular level. Curr. Top. Med. Chem. 2002;2:507–28. doi: 10.2174/1568026023393796. [DOI] [PubMed] [Google Scholar]
- 6.Kroeze WK, Hufeisen SJ, Popadak BA, et al. H1-histamine receptor affinity predicts short-term weight gain for typical and atypical antipsychotic drugs. Neuropsychopharmacology. 2003;28:519–26. doi: 10.1038/sj.npp.1300027. [DOI] [PubMed] [Google Scholar]
- 7.Roth BL. Drugs and valvular heart disease. N. Engl. J. Med. 2007;356:6–9. doi: 10.1056/NEJMp068265. [DOI] [PubMed] [Google Scholar]
- 8.Gershon MD, Tack J. The serotonin signaling system: from basic understanding to drug development for functional GI disorders. Gastroenterology. 2007;132:397–414. doi: 10.1053/j.gastro.2006.11.002. [DOI] [PubMed] [Google Scholar]
- 9.Mengod G, Vilaro MT, Cortes R, et al. Chemical neuroanatomy of 5-HT receptor subtypes in the mammalian brain. In: Roth BL, editor. The Serotonin Receptors: From Molecular Pharmacology to Human Therapeutics. Totowa, NJ: Humana; 2007. pp. 319–64. [Google Scholar]
- 10.Araneda R, Andrade R. 5-Hydroxytryptamine 2 and 5-hydroxytryptamine 1A receptors mediate opposing responses on membrane excitability in rat association cortex. Neuroscience. 1991;40:399–412. doi: 10.1016/0306-4522(91)90128-b. [DOI] [PubMed] [Google Scholar]
- 11.Airan RD, Meltzer LA, Roy M, et al. High-speed imaging reveals neurophysiological links to behavior in an animal model of depression. Science. 2007;317:819–23. doi: 10.1126/science.1144400. [DOI] [PubMed] [Google Scholar]
- 12.Canli T, Lesch KP. Long story short: the serotonin transporter in emotion regulation and social cognition. Nat. Neurosci. 2007;10:1103–9. doi: 10.1038/nn1964. [DOI] [PubMed] [Google Scholar]
- 13.Roth BL, Hanizavareh SM, Blum AE. Serotonin receptors represent highly favorable molecular targets for cognitive enhancement in schizophrenia and other disorders. Psychopharmacology (Berl.) 2004;174:17–24. doi: 10.1007/s00213-003-1683-8. [DOI] [PubMed] [Google Scholar]
- 14.Gross C, Hen R. The developmental origins of anxiety. Nat. Rev. Neurosci. 2004;5:545–52. doi: 10.1038/nrn1429. [DOI] [PubMed] [Google Scholar]
- 15.Lesch KP. Serotonergic gene inactivation in mice: models for anxiety and aggression? Novartis Found. Symp. 2005;268:111–40. [PubMed] [Google Scholar]
- 16.Giorgetti M, Tecott LH. Contributions of 5-HT2C receptors to multiple actions of central serotonin systems. Eur. J. Pharmacol. 2004;488:1–9. doi: 10.1016/j.ejphar.2004.01.036. [DOI] [PubMed] [Google Scholar]
- 17.Gray JA, Roth BL. The pipeline and future of drug development in schizophrenia. Mol. Psychiatry. 2007;12:904–22. doi: 10.1038/sj.mp.4002062. [DOI] [PubMed] [Google Scholar]
- 18.Schechter LE, Ring RH, Beyer CE, et al. Innovative approaches for the development of antidepressant drugs: current and future strategies. NeuroRx. 2005;2:590–611. doi: 10.1602/neurorx.2.4.590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kaumann AJ, Levy FO. 5-hydroxytryptamine receptors in the human cardiovascular system. Pharmacol. Ther. 2006;111:674–706. doi: 10.1016/j.pharmthera.2005.12.004. [DOI] [PubMed] [Google Scholar]
- 20.Hamel E. Serotonin and migraine: biology and clinical implications. Cephalalgia. 2007;27:1293–300. doi: 10.1111/j.1468-2982.2007.01476.x. [DOI] [PubMed] [Google Scholar]
- 21.Ni W, Watts SW. 5-hydroxytryptamine in the cardiovascular system: focus on the serotonin transporter (SERT) Clin. Exp. Pharmacol. Physiol. 2006;33:575–83. doi: 10.1111/j.1440-1681.2006.04410.x. [DOI] [PubMed] [Google Scholar]
- 22.Carneiro AM, Cook EH, Murphy DL, Blakely RD. Interactions between integrin αIIbβ3 and the serotonin transporter regulate serotonin transport and platelet aggregation in mice and humans. J. Clin. Invest. 2008;118:1544–52. doi: 10.1172/JCI33374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sauer WH, Berlin JA, Kimmel SE. Effect of antidepressants and their relative affinity for the serotonin transporter on the risk of myocardial infarction. Circulation. 2003;108:32–36. doi: 10.1161/01.CIR.0000079172.43229.CD. [DOI] [PubMed] [Google Scholar]
- 24.Taylor CB, Youngblood ME, Catellier D, et al. Effects of antidepressant medication on morbidity and mortality in depressed patients after myocardial infarction. Arch. Gen. Psychiatry. 2005;62:792–98. doi: 10.1001/archpsyc.62.7.792. [DOI] [PubMed] [Google Scholar]
- 25.Glassman AH. Does treating postmyocardial infarction depression reduce medical mortality? Arch. Gen. Psychiatry. 2005;62:711–12. doi: 10.1001/archpsyc.62.7.711. [DOI] [PubMed] [Google Scholar]
- 26.Somberg TC, Arora RR. Depression and heart disease: therapeutic implications. Cardiology. 2008;111:75–81. doi: 10.1159/000119692. [DOI] [PubMed] [Google Scholar]
- 27.Walther DJ, Peter JU, Winter S, et al. Serotonylation of small GTPases is a signal transduction pathway that triggers platelet alpha-granule release. Cell. 2003;115:851–62. doi: 10.1016/s0092-8674(03)01014-6. [DOI] [PubMed] [Google Scholar]
- 28.Dale GL. Coated-platelets: an emerging component of the procoagulant response. J. Thromb. Haemost. 2005;3:2185–92. doi: 10.1111/j.1538-7836.2005.01274.x. [DOI] [PubMed] [Google Scholar]
- 29.Langer C, Piper C, Vogt J, et al. Atrial fibrillation in carcinoid heart disease: the role of serotonin. A review of the literature. Clin. Res. Cardiol. 2007;96:114–18. doi: 10.1007/s00392-006-0463-y. [DOI] [PubMed] [Google Scholar]
- 30.Singh S. Trials of new antiarrhythmic drugs for maintenance of sinus rhythm in patients with atrial fibrillation. J. Interv. Cardiol. Electrophysiol. 2004;10(Suppl. 1):71–76. doi: 10.1023/B:JICE.0000011348.05608.e5. [DOI] [PubMed] [Google Scholar]
- 31.Brattelid T, Qvigstad E, Lynham JA, et al. Functional serotonin 5-HT4 receptors in porcine and human ventricular myocardium with increased 5-HT4mRNAin heart failure. Naunyn Schmiedebergs Arch. Pharmacol. 2004;370:157–66. doi: 10.1007/s00210-004-0963-0. [DOI] [PubMed] [Google Scholar]
- 32.Birkeland JA, Sjaastad I, Brattelid T, et al. Effects of treatment with a 5-HT4 receptor antagonist in heart failure. Br. J. Pharmacol. 2007;150:143–52. doi: 10.1038/sj.bjp.0706966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Doggrell SA. The role of 5-HT on the cardiovascular and renal systems and the clinical potential of 5-HT modulation. Expert Opin. Investig. Drugs. 2003;12:805–23. doi: 10.1517/13543784.12.5.805. [DOI] [PubMed] [Google Scholar]
- 34.Rothman RB, Baumann MH, Savage JE, et al. Evidence for possible involvement of 5-HT2B receptors in the cardiac valvulopathy associated with fenfluramine and other serotonergic medications. Circulation. 2000;102:2836–41. doi: 10.1161/01.cir.102.23.2836. [DOI] [PubMed] [Google Scholar]
- 35.Setola V, Hufeisen SJ, Grande-Allen KJ, et al. 3,4-methylenedioxymethamphetamine (MDMA, “Ecstasy”) induces fenfluramine-like proliferative actions on human cardiac valvular interstitial cells in vitro. Mol. Pharmacol. 2003;63:1223–29. doi: 10.1124/mol.63.6.1223. [DOI] [PubMed] [Google Scholar]
- 36.Nebigil CG, Maroteaux L. Functional consequence of serotonin/5-HT2B receptor signaling in heart: role of mitochondria in transition between hypertrophy and heart failure? Circulation. 2003;108:902–8. doi: 10.1161/01.CIR.0000081520.25714.D9. [DOI] [PubMed] [Google Scholar]
- 37.Kereveur A, Callebert J, Humbert M, et al. High plasma serotonin levels in primary pulmonary hypertension. Effect of long-term epoprostenol (prostacyclin) therapy. Arterioscler. Thromb. Vasc. Biol. 2000;20:2233–39. doi: 10.1161/01.atv.20.10.2233. [DOI] [PubMed] [Google Scholar]
- 38.Esteve JM, Launay JM, Kellermann O, Maroteaux L. Functions of serotonin in hypoxic pulmonary vascular remodeling. Cell. Biochem. Biophys. 2007;47:33–44. doi: 10.1385/cbb:47:1:33. [DOI] [PubMed] [Google Scholar]
- 39.Launay JM, Herve P, Peoc’h K, et al. Function of the serotonin 5-hydroxytryptamine 2B receptor in pulmonary hypertension. Nat. Med. 2002;8:1129–35. doi: 10.1038/nm764. [DOI] [PubMed] [Google Scholar]
- 40.Deraet M, Manivet P, Janoshazi A, et al. The natural mutation encoding a C terminus–truncated 5-hydroxytryptamine 2B receptor is a gain of proliferative functions. Mol. Pharmacol. 2005;67:983–91. doi: 10.1124/mol.104.008268. [DOI] [PubMed] [Google Scholar]
- 41.Guilluy C, Rolli-Derkinderen M, Tharaux PL, et al. Transglutaminase-dependent RhoA activation and depletion by serotonin in vascular smooth muscle cells. J. Biol. Chem. 2007;282:2918–28. doi: 10.1074/jbc.M604195200. [DOI] [PubMed] [Google Scholar]
- 42.Marcos E, Adnot S, Pham MH, et al. Serotonin transporter inhibitors protect against hypoxic pulmonary hypertension. Am. J. Respir. Crit. Care Med. 2003;168:487–93. doi: 10.1164/rccm.200210-1212OC. [DOI] [PubMed] [Google Scholar]
- 43.Eilers H, Schumacher MA. Opioid-induced respiratory depression: Are 5-HT4a receptor agonists the cure? Mol. Interv. 2004;4:197–99. doi: 10.1124/mi.4.4.3. [DOI] [PubMed] [Google Scholar]
- 44.Manzke T, Guenther U, Ponimaskin EG, et al. 5-HT4a receptors avert opioid-induced breathing depression without loss of analgesia. Science. 2003;301:226–29. doi: 10.1126/science.1084674. [DOI] [PubMed] [Google Scholar]
- 45.Kinney HC, Filiano JJ, White WF. Medullary serotonergic network deficiency in the sudden infant death syndrome: review of a 15-year study of a single dataset. J. Neuropathol. Exp. Neurol. 2001;60:228–47. doi: 10.1093/jnen/60.3.228. [DOI] [PubMed] [Google Scholar]
- 46.Paterson DS, Trachtenberg FL, Thompson EG, et al. Multiple serotonergic brainstem abnormalities in sudden infant death syndrome. JAMA. 2006;296:2124–32. doi: 10.1001/jama.296.17.2124. [DOI] [PubMed] [Google Scholar]
- 47.Richerson GB, Wang W, Tiwari J, Bradley SR. Chemosensitivity of serotonergic neurons in the rostral ventral medulla. Respir. Physiol. 2001;129:175–89. doi: 10.1016/s0034-5687(01)00289-4. [DOI] [PubMed] [Google Scholar]
- 48.Erickson JT, Shafer G, Rossetti MD, et al. Arrest of 5HT neuron differentiation delays respiratory maturation and impairs neonatal homeostatic responses to environmental challenges. Respir. Physiol. Neurobiol. 2007;159:85–101. doi: 10.1016/j.resp.2007.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Tecott LH, Abdallah L. Mouse genetic approaches to feeding regulation: serotonin 5-HT2C receptor mutant mice. CNS Spectr. 2003;8:584–88. [PubMed] [Google Scholar]
- 50.Lam DD, Heisler LK. Serotonin and energy balance: molecular mechanisms and implications for type 2 diabetes. Expert Rev. Mol. Med. 2007;9:1–24. doi: 10.1017/S1462399407000245. [DOI] [PubMed] [Google Scholar]
- 51.Lam DD, Przydzial MJ, Ridley SH, et al. Serotonin 5-HT2C receptor agonist promotes hypophagia via downstream activation of melanocortin 4 receptors. Endocrinology. 2008;149:1323–28. doi: 10.1210/en.2007-1321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hodges MR, Tattersall GJ, Harris MB, et al. Defects in breathing and thermoregulation in mice with near-complete absence of central serotonin neurons. J. Neurosci. 2008;28:2495–505. doi: 10.1523/JNEUROSCI.4729-07.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hedlund PB, Kelly L, Mazur C, et al. 8-OH-DPAT acts on both 5-HT1A and 5-HT7 receptors to induce hypothermia in rodents. Eur. J. Pharmacol. 2004;487:125–32. doi: 10.1016/j.ejphar.2004.01.031. [DOI] [PubMed] [Google Scholar]
- 54.Carrasco GA, Van de Kar LD. Neuroendocrine pharmacology of stress. Eur. J. Pharmacol. 2003;463:235–72. doi: 10.1016/s0014-2999(03)01285-8. [DOI] [PubMed] [Google Scholar]
- 55.Matsuda M, Imaoka T, Vomachka AJ, et al. Serotonin regulates mammary gland development via an autocrine-paracrine loop. Dev. Cell. 2004;6:193–203. doi: 10.1016/s1534-5807(04)00022-x. [DOI] [PubMed] [Google Scholar]
- 56.Stull MA, Pai V, Vomachka AJ, et al. Mammary gland homeostasis employs serotonergic regulation of epithelial tight junctions. Proc. Natl. Acad. Sci. USA. 2007;104:16708–13. doi: 10.1073/pnas.0708136104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Lesurtel M, Graf R, Aleil B, et al. Platelet-derived serotonin mediates liver regeneration. Science. 2006;312:104–7. doi: 10.1126/science.1123842. [DOI] [PubMed] [Google Scholar]
- 58.Tecott LH. Serotonin and the orchestration of energy balance. Cell. Metab. 2007;6:352–61. doi: 10.1016/j.cmet.2007.09.012. [DOI] [PubMed] [Google Scholar]
- 59.Roper SD. Cell communication in taste buds. Cell. Mol. Life Sci. 2006;63:1494–500. doi: 10.1007/s00018-006-6112-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Suzuki A, Naruse S, Kitagawa M, et al. 5-hydroxytryptamine strongly inhibits fluid secretion in guinea pig pancreatic duct cells. J. Clin. Invest. 2001;108:749–56. doi: 10.1172/JCI12312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Minami M, Endo T, Hirafuji M, et al. Pharmacological aspects of anticancer drug–induced emesis with emphasis on serotonin release and vagal nerve activity. Pharmacol. Ther. 2003;99:149–65. doi: 10.1016/s0163-7258(03)00057-3. [DOI] [PubMed] [Google Scholar]
- 62.Sommer C. Serotonin in pain and analgesia: actions in the periphery. Mol. Neurobiol. 2004;30:117–25. doi: 10.1385/MN:30:2:117. [DOI] [PubMed] [Google Scholar]
- 63.Braz JM, Basbaum AI. Genetically expressed transneuronal tracer reveals direct and indirect serotonergic descending control circuits. J. Comp. Neurol. 2008;507:1990–2003. doi: 10.1002/cne.21665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Jann MW, Slade JH. Antidepressant agents for the treatment of chronic pain and depression. Pharmacotherapy. 2007;27:1571–87. doi: 10.1592/phco.27.11.1571. [DOI] [PubMed] [Google Scholar]
- 65.Mukaida K, Shichino T, Koyanagi S, et al. Activity of the serotonergic system during isoflurane anesthesia. Anesth. Analg. 2007;104:836–39. doi: 10.1213/01.ane.0000255200.42574.22. [DOI] [PubMed] [Google Scholar]
- 66.Isbister GK, Buckley NA, Whyte IM. Serotonin toxicity: a practical approach to diagnosis and treatment. Med. J. Aust. 2007;187:361–65. doi: 10.5694/j.1326-5377.2007.tb01282.x. [DOI] [PubMed] [Google Scholar]
- 67.de Jong TR, Veening JG, Waldinger MD, et al. Serotonin and the neurobiology of the ejaculatory threshold. Neurosci. Biobehav. Rev. 2006;30:893–907. doi: 10.1016/j.neubiorev.2006.01.001. [DOI] [PubMed] [Google Scholar]
- 68.Giuliano F. 5-Hydroxytryptamine in premature ejaculation: opportunities for therapeutic intervention. Trends Neurosci. 2007;30:79–84. doi: 10.1016/j.tins.2006.12.002. [DOI] [PubMed] [Google Scholar]
- 69.Ramage AG. The role of central 5-hydroxytryptamine (5-HT, serotonin) receptors in the control of micturition. Br. J. Pharmacol. 2006;147(Suppl. 2):S120–31. doi: 10.1038/sj.bjp.0706504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Basu M, Duckett J. The treatment of urinary incontinence with duloxetine. J. Obstet. Gynaecol. 2008;28:166–69. doi: 10.1080/01443610801912931. [DOI] [PubMed] [Google Scholar]
- 71.Bolte AC, van Geijn HP, Dekker GA. Pathophysiology of preeclampsia and the role of serotonin. Eur. J. Obstet. Gynecol. Reprod. Biol. 2001;95:12–21. doi: 10.1016/s0301-2115(00)00367-5. [DOI] [PubMed] [Google Scholar]
- 72.Gupta S, Hanff LM, Visser W, et al. Functional reactivity of 5-HT receptors in human umbilical cord and maternal subcutaneous fat arteries after normotensive or pre-eclamptic pregnancy. J. Hypertens. 2006;24:1345–53. doi: 10.1097/01.hjh.0000234115.40648.88. [DOI] [PubMed] [Google Scholar]
- 73.Dawes SD. Can SSRIs reduce the risk of preeclampsia in pregnant, depressed patients? Med. Hypotheses. 2005;64:33–36. doi: 10.1016/j.mehy.2003.10.038. [DOI] [PubMed] [Google Scholar]
- 74.Salkeld E, Ferris LE, Juurlink DN. The risk of postpartum hemorrhage with selective serotonin reuptake inhibitors and other antidepressants. J. Clin. Psychopharmacol. 2008;28:230–34. doi: 10.1097/JCP.0b013e318166c52e. [DOI] [PubMed] [Google Scholar]
- 75.Chambers CD, Hernandez-Diaz S, Van Marter LJ, et al. Selective serotonin-reuptake inhibitors and risk of persistent pulmonary hypertension of the newborn. N. Engl. J. Med. 2006;354:579–87. doi: 10.1056/NEJMoa052744. [DOI] [PubMed] [Google Scholar]
- 76.Minosyan TY, Lu R, Eghbali M, et al. Increased 5-HT contractile response in late pregnant rat myometrium is associated with a higher density of 5-HT2A receptors. J. Physiol. 2007;581:91–97. doi: 10.1113/jphysiol.2007.129726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Oropeza MV, Ponce Monter H, Reynoso Isla M, Campos MG. The ovarian and cervical regions of the rat uterus display a different contractile response to serotonin and prostaglandin F2αI. The estrous cycle. Life Sci. 2000;66:PL345–51. doi: 10.1016/s0024-3205(00)80010-7. [DOI] [PubMed] [Google Scholar]
- 78.Jeffrey JJ, Ehlich LS, Roswit WT. Serotonin: an inducer of collagenase in myometrial smooth muscle cells. J. Cell Physiol. 1991;146:399–406. doi: 10.1002/jcp.1041460310. [DOI] [PubMed] [Google Scholar]
- 79.Ruhe HG, Mason NS, Schene AH. Mood is indirectly related to serotonin, norepinephrine and dopamine levels in humans: a meta-analysis of monoamine depletion studies. Mol. Psychiatry. 2007;12:331–59. doi: 10.1038/sj.mp.4001949. [DOI] [PubMed] [Google Scholar]
- 80.Weintraub M, Hasday JD, Mushlin AI, Lockwood DH. A double-blind clinical trial in weight control. Use of fenfluramine and phentermine alone and in combination. Arch. Intern. Med. 1984;144:1143–48. [PubMed] [Google Scholar]
- 81.Connolly HM, Crary JL, McGoon MD, et al. Valvular heart disease associated with fenfluraminephentermine. N. Engl. J. Med. 1997;337:581–88. doi: 10.1056/NEJM199708283370901. [DOI] [PubMed] [Google Scholar]
- 82.Fitzgerald LW, Burn TC, Brown BS, et al. Possible role of valvular serotonin 5-HT2B receptors in the cardiopathy associated with fenfluramine. Mol. Pharmacol. 2000;57:75–81. [PubMed] [Google Scholar]
- 83.O’Connor KA, Roth BL. Finding new tricks for old drugs: an efficient route for public-sector drug discovery. Nat. Rev. Drug Discov. 2005;4:1005–14. doi: 10.1038/nrd1900. [DOI] [PubMed] [Google Scholar]
- 84.Droogmans S, Cosyns B, D’Haenen H, et al. Possible association between 3,4-methylenedioxymethamphetamine abuse and valvular heart disease. Am. J. Cardiol. 2007;100:1442–45. doi: 10.1016/j.amjcard.2007.06.045. [DOI] [PubMed] [Google Scholar]
- 85.Schade R, Andersohn F, Suissa S, et al. Dopamine agonists and the risk of cardiac-valve regurgitation. N. Engl. J. Med. 2007;356:29–38. doi: 10.1056/NEJMoa062222. [DOI] [PubMed] [Google Scholar]
- 86.Zanettini R, Antonini A, Gatto G, et al. Valvular heart disease and the use of dopamine agonists for Parkinson’s disease. N. Engl. J. Med. 2007;356:39–46. doi: 10.1056/NEJMoa054830. [DOI] [PubMed] [Google Scholar]