Abstract
Objective:
The is to report immunohistochemical observations of aortic α-smooth muscle actin (SMA) expressions in patients with aortic aneurysm, acute aortic dissection, and coronary artery disease and to discuss phenotypic switching of smooth muscle cells (SMCs) of these lesions.
Methods:
Forty-nine consecutive patients scheduled for surgical treatment for acute type A aortic dissection (20 patients), aortic aneurysm (9 patients), and coronary artery disease (20 patients) were included. Surgical specimens of the aorta were obtained and prepared for hematoxylin and eosin and immunohistochemical stainings.
Results:
A comparison of aortic structural changes between the three groups showed that patients with coronary artery disease had the least severe aorta degeneration with the most intense α-SMA positivity. Aortic structural impairment was the most severe in patients with aortic dissection, whereas α-SMA positivity was more intense in patients with aortic dissection than in those with aortic aneurysm.
Conclusion:
Disparities in α-SMA expressions in the aortic tissues of the three groups represent the extent of SMC degenerations or a phenotypic switching between contractile and synthetic SMCs. The results imply severe SMC degenerations in patients with aortic aneurysm, which may be beneficial because of the production of extracellular matrix necessary for healing of the vascular wall, but severe disruptions in elastic fibers in patients with aortic dissection. Patients with coronary artery disease show slight SMC degeneration and phenotypic switching among the three groups. The possible apoptotic and genetic mechanisms of aortic structural impairments warrant further elaborations.
Keywords: actin, aorta, thoracic, immunohistochemistry
Introduction
α-smooth muscle actin (SMA), encoded by the acyl-coenzyme A (CoA):cholesterol acyltransferase 2 (ACTA2) gene, is an isoform of smooth muscle cells (SMCs). It is typically expressed in vascular SMCs. Mutations in the ACTA2 gene lead to various vascular disorders, such as aortic aneurysms and dissections, and ischemic or occlusive vascular diseases. Disruptions in α-SMA may result in SMC hyperplasia via diverse cellular signaling pathways, including focal adhesion kinase, p53, and platelet-derived growth factor receptor-α pathways, leading to aortic disease or vascular occlusive disorders in patients with ACTA2 missense mutations (1).
Immunohistochemistry is a sensitive method for detecting α-SMA expressions in the aortic tissue of human and experimental animals (2). Aortic α-SMA expressions of patients with abdominal aortic aneurysm, coarctation of the aorta, atherosclerosis, and thoracic aorta dissection determined by immunohistochemistry have been reported (3-7). However, a comparative study on aortic α-SMA expressions between aortopathies and coronary artery disease using an immunostaining method is limited. Moreover, the role that phenotypic switching plays in this process of aortic wall remodeling in the context of aortopathies and aortic atherosclerosis remain unclear. Here the results of an immunohistochemical study on aortic α-SMA expressions in patients with aortic aneurysm, acute aortic dissection, and coronary artery disease are reported and possible mechanisms regarding phenotypic switching are discussed.
Methods
Forty-nine consecutive patients scheduled for surgical treatment of acute type A aortic dissection (20 patients), aortic aneurysm (9 patients), and coronary artery disease (20 patients) were randomly included in this study. The diameter of the thoracic aortic aneurysms was 6.2±0.8 (range, 5.1–7.1; median, 6.4) cm (n=9). In total, 25 (51.0%) patients (20 patients from the aortic dissection group and 5 patients from the coronary artery disease group) were operated on an urgent basis and the remaining 24 (49%) patients underwent an elective operation. The patients’ information are shown in Table 1.
Table 1.
Demographic data of the patients
| Group | Aortic dissection | Aortic aneurysm | CAD |
|---|---|---|---|
| Case, n | 20 | 9 | 21 |
| Gender, male/female, n | 18/2 | 6/3 | 19/2 |
| Age, year | 53.8±9.7 | 46.2±11.2 | 60.3±4.9 |
| Hypertension, n | 16 | 6 | 18 |
| Diabetes mellitus, n | 2 | 0 | 9 |
| Renal failure, n | 2 | 0 | 0 |
| Operation, n | Aorta replacement with or without aortic valve replacement (20) | Ascending aorta replacement with or without aortic valve replacement (5); Bentall operation (1); arch replacement (1); thoracic and abdominal aorta replacement (1); descending aorta replacement (1) | Off-pump coronary artery bypass (15); on-pump artery bypass (5); beating heart coronary artery revascularization (1) |
| Survival rate, % | 100 | 85 | 100 |
No statistical analysis was used. CAD - coronary artery disease
Patients’ age was expressed in mean±SD
Surgical specimens of aortic tissues were obtained immediately after they were severed during the surgeries for replacement of the aorta in patients with aortic dissection or aortic aneurysm. In patients receiving coronary artery bypass grafting, the tiny aortic tissues were obtained when holes were punched for the proximal anastomosis on the anterior wall of the ascending aorta. The aortic tissues were for immunohistochemical staining of α-SMA. Three to four histological sections per specimen were evaluated. Tissue slides were analyzed using a four-point semi-quantitative scale for determining nuclear and cytoplasmic optical staining intensity [graded as 3+ (strong), 2+ (moderate), 1+(weak), and 0 (no staining)] and determining the percentage of positively stained cells (0: no staining, 1: <10% of the cells, 2–11: 50% of the cells, and 3: >50% of the cells) (8).
Quantitative data are expressed in mean±standard deviation with range and median values and were compared by independent sample t-test. A p<0.05 was considered to be statistically significant.
Hematoxylin and eosin and immonuhistochemistry stainings
Aortic tissue samples were fixed in 10% methanol solution in 1 mm3 blocks, embedded in paraffin wax, processed routinely, and stained with hematoxylin and eosin on 4-µm sections (9). Immunohistochemical staining of α-SMA was performed using EnVision method (9). A primary antibody α-SMA sc-58671 (1:100) purchased from Santa Cruz Biotechnology, Inc., CA, USA, was used.
Informed consent was obtained from each patient. This study conforms to the Declaration of Helsinki and was approved by the institutional research Ethics Committee board.
Results
Hematoxylin and eosin staining
In the aorta of patients with aortic dissection, extensive disarrangement and disruption in elastic fibers of the medial layer were observed. Close to the dissected lesion, the medial walls showed severe necrosis. Loss of elastic fibers and infiltration of neutrophils was present in the adventitia, and intimal fibrous thickening was apparent (Fig. 1a).
Figure 1.
Hematoxylin and eosin staining: (a) aortic aneurysm: The aortic wall seems to be thickened and uneven. There are ruptures in elastic fibers in the aortic media (arrows), which are replaced with fibrous scar tissues. Local congestion and hemorrhage and filtrations with a few chronic inflammatory cells can be observed in the aortic wall; (b) aortic dissection: Degeneration, necrosis, rupture, dissolution, and loss of elastic and smooth muscle fibers (arrows) as well as focal lymphoid and mononuclear cell infiltrations can be observed in the aortic media. Focal lymphoid and mononuclear cell infiltrations and fibroplastic proliferation in the adventitia are seen; and (c) coronary artery disease: A hyaline fibrous tissue degeneration in the intima and media, endocardial deposition of atheromas, disruptions in elastic fiber (arrows), and loss and calcification of the tunica media are the major pathological changes. Hematoxylin and eosin x200
In the aortic tissue of patients with aortic aneurysm, there was focal disruption and patchy necrosis of elastic fibers of the medial layers. Adventitial structures were loose with scattered inflammatory cell infiltrations. The intimas were thickened (Fig. 1b).
In the aortic tissues of patients with coronary artery disease, structural disruptions of the three layers were milder than those in the other two patient groups. Scattered inflammation infiltration was observed in the adventitial layers (Fig. 1c).
Immunohistochemistry
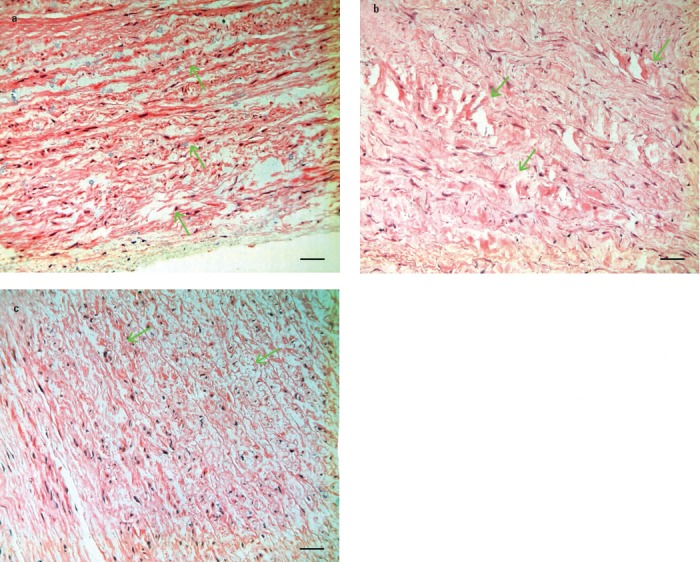
Immunohistochemistry showed mild-to-moderate positivity of α-SMA expressed in the aortic media of patients with aortic aneurysm; more intense positivity in the aortic media of patients with acute type A aortic dissection; and most intense positivity in the aorta of patients with coronary artery disease (Table 2, Fig. 2).
Table 2.
Semi-quantitative results of immunostaining
| Location | Aortic aneurysm | Aortic dissection | CAD |
|---|---|---|---|
| Staining intensity | |||
| Media | |||
| Cytoplasm, n | 1+~2+(9) | 2+~3++(20) | 3+(21) |
| Nucleus, n | 0~1+(9) | 1+~2+(20) | 3+(21) |
| Interstitium, n | 0~1+(8) | 0~1+(18) | 3+(20) |
| Intima, n | 1+~2+(9) | 1+~2+(21) | 1+~2+(20) |
| Adventitia, n | 0~1+(9) | 1+~2+(20) | 3+(21) |
| Staining extent | |||
| Score | 2 | 3 | 3 |
| Percentage | 52.9±5.9 | 61.7±6.1* | 94.0±5.5** |
P=0.0011 in comparison to Aortic Aneurysm Group by independent t-test; CAD-coronary artery disease
P<0.0001 in comparison to Aortic Aneurysm Group and to Aortic Dissection Group by independent t-test.
Figure 2.

Immunohistochemistry of α-smooth muscle actin of the aortic wall of patients with (a) ascending aortic aneurysm, (b) acute type A aortic dissection, and (c) coronary artery disease showing incremental positivity in aortic media and intima from aortic aneurysm to aortic dissection to coronary artery disease. Arrows indicated the positive reaction of α-smooth muscle actin in the cytoplasma and nucleus. EnVision x200
Discussion
The aorta is composed of three layers: intima, media, and adventitia. The tensile strength and elasticity of the aorta rely on media, which is composed of concentrically arranged elastic fibers and SMCs (10). In patients with ACTA2 missense mutations, aortic dilation and aortic wall thickening are observed often (11). In patients with thoracic aortic aneurysm and dissection, there are signs of medial degeneration, such as fragmentation of elastic fibers, loss of SMCs, and accumulation of proteoglycans (10). However, it is worthwhile to mention that elastin does not stain quantitatively on routine hematoxylin and eosin staining. Therefore, it often appears partially unstained.
Aortic aneurysm formation is caused by aortic adaptation to aortic flow and wall shear stress (12), whereas aortic dissection can be caused by acute high-energy trauma, and pulsatile flow and high blood pressure contribute to the propagation of aortic dissection (13). Thoracic aneurysms, on the other hand, are relatively rare and exhibit a strong heritable pattern (14). Mutations in SMC-specific α-myosin and α-actin predispose individuals to the development of thoracic aortic aneurysm. Based on this theory, maintenance of the contractile phenotype of SMC might preserve the aortic structure and prevent the formation of aortic aneurysm (14).
In an immunohistochemical staining study, it was confirmed that α-SMA expressions in aortic SMCs of patients with abdominal aortic aneurysm were significantly reduced. The metrological analysis showed that, compared with the normal control, the SMC density decreased by 74.5%, while no significant difference was found between the aorta of patients with arteriosclerosis obliterans and individuals with a normal aorta (3). Fukui et al. (4) compared the medial layer of patients with abdominal aortic aneurysm with that of those with occlusive aortas in their studies. They found that significant α-SMA expressions in the media of the occlusive aorta and aneurysmal neck, but SMCs in the aneurysmal dilated region were disrupted and disorganized. Tanaskovic et al. (5) found by an immunohistochemical study that α-SMA and vimentin were positively stained in SMCs of the media and in the margins of the plaques. Hou et al. (6) used western blotting and immunohistochemical studies to investigate α-SMA expression in the dissected thoracic aorta and found that α-SMA expressions were significantly downregulated in medial SMCs compared with those in a normal aorta (6). Surgical specimens of coarctation segments from younger patients were examined by the immunocytochemical methods, which revealed that SMCs were dedifferentiated and expressed α-SMA and vimentin, but lacked desmin and myosin heavy chains (7). In addition, reduced cell numbers in the intima and media and apoptotic SMCs in the inner media were observed, and the results suggested an apoptotic mechanism is responsible for the aortic pathology (7).
Diabetes is associated with increased synthesis and decreased degradation of the extracellular matrix and promotion of SMC proliferation. Patients with diabetes show an altered SMC phenotype with a disrupted α-SMA cytoskeleton. Antidiabetic treatments with angiotensin-converting enzyme inhibitors, metformin, or rosiglitazone may prevent aortic expansion and rupture by reducing vascular inflammation, increasing elastin deposition, and inhibiting matrix metalloproteinase secretion (15). Thiazolidinediones are shown to have superior effects than other antidiabetic agents, such as sulfonylureas and biguanides, in inhibiting abnormal arterial SMC proliferation associated with atherosclerosis (16).
Normally, SMCs of the aortic wall are in a well-differentiated, contractile state; whereas in atherosclerosis, SMCs shift toward the synthetic phenotype. Vascular SMCs in the normal arterial media express a range of SMC markers, conventionally including SMC myosin heavy chain and α-actin. SMCs of the atherosclerotic vessels have reduced expression of these markers and increased capacity for cell proliferation, migration, and generation of extracellular matrix proteins (17). The process is regulated by a switch of vascular SMCs to the synthetic phenotype in atherosclerotic lesions (18). The expressions of actin in the aortic media were in a close inverse proportion to the depth of the microvessel invasion into the media; the deeper the invasion of the microvessels, the less actin expressions and the less contractile phenotypes are in the media (19).
Aortic wall remodeling is typically associated with an altered proportion of degradation and synthesis of the matrix, and increased collagenase and elastase activities have been demonstrated during rapid progression of aneurysms (20). During the progression of abdominal aortic aneurysm, SMCs became less abundant in terms of plasminogen activator inhibitor type 1 expressions (21). As a contractile phenotype protein, α-SMA has been extensively investigated in vascular pathologies. The present study implied disparities in the extent of phenotypic switching between the three groups of patients by showing different aortic α-SMA positivities. The weaker aortic α-SMA positivity in patients with aortic aneurysm demonstrated a severe SMC degeneration or a switch between the contractile and synthetic phenotype of SMCs in aortic aneurysm, which may be even beneficial because of the extracellular matrix dependent phenotypic balance necessary for healing of the vascular wall. This result was consistent with the prevailing synthetic phenotype protein vimentin in the vascular SMCs of patients with abdominal aortic aneurysm reported by Houdek et al. (22). Moreover, Eberlová et al. (23) revealed an inverse relationship between actin positivity in the aortic intima and media and the size of abdominal aortic aneurysm, which might be, at least in part, an explanation for our observation of a weak aortic α-SMA positivity in the aorta of patients with thoracic aneurysm.
Although severe disruptions in elastic fiber were present, more intense α-SMA positivity were observed in patients with dissected aorta. Such a condition displayed a more severely impaired aortic contractile profile and plasticity; however, with an SMC phenotype switching in the dissected media. Similarly, Zhang et al. (24) noted a reduced aortic α-SMA positivity to 60.1% in the patients with dissected media compared with that in a control without dissected media.
Study limitations
A smaller patient population and independent immunocytochemical observations of aortic α-SMA constitute the major drawbacks of this study. The synthetic phenotype protein, such as vimentin, and alternative contractile phenotype protein, desmin, should also be investigated in the future.
Conclusion
Disparities in α-SMA expressions in the aortic tissues of the three patient groups represent the extent of SMC degeneration other than those of the elastic fibers. The results imply severe SMC degeneration or a switch between the contractile and synthetic phenotype of SMCs in patients with aortic aneurysm, which may be even beneficial because of the production of an extracellular matrix that is necessary for healing of the vascular wall, but severe disruptions in elastic fiber in patients with aortic dissection. Patients with coronary artery disease show slight SMC degeneration and phenotypic switch among the three patient groups. The possible apoptotic and genetic mechanisms of aortic structural impairments warrant further elaborations. Increase in the number of patients enrolled with incorporation of molecular biological studies on aortic α-SMA in the near future would ensure more precise results and reliable conclusions.
Footnotes
Funding: None declared.
Conflict of interest: None declared.
Peer-review: Externally peer-reviewed.
Authorship contributions: Concept – S.M.Y.; Design – S.M.Y.; Supervision – S.M.Y.; Funding – S.M.Y.; Materials – S.M.Y., N.W.; Data collection &/or processing – S.M.Y., N.W.; Analysis &/or interpretation – S.M.Y., N.W.; Literature search – S.M.Y.; Writing – S.M.Y.; Critical review – S.M.Y., N.W.
References
- 1.Yuan SM. α-smooth muscle actin and ACTA2 gene expressions in vasculopathies. Braz J Cardiovasc Surg. 2015;30:644–9. doi: 10.5935/1678-9741.20150081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Azuma K, Ichimura K, Mita T, Nakayama S, Jin WL, Hirose T, et al. Presence of alpha-smooth muscle actin-positive endothelial cells in the luminal surface of adult aorta. Biochem Biophys Res Commun. 2009;380:620–6. doi: 10.1016/j.bbrc.2009.01.135. [DOI] [PubMed] [Google Scholar]
- 3.López-Candales A, Holmes DR, Liao S, Scott MJ, Wickline SA, Thompson RW. Decreased vascular smooth muscle cell density in medial degeneration of human abdominal aortic aneurysms. Am J Pathol. 1997;150:993–1007. [PMC free article] [PubMed] [Google Scholar]
- 4.Fukui D, Miyagawa S, Soeda J, Tanaka K, Urayama H, Kawasaki S. Overexpression of transforming growth factor β1 in smooth muscle cells of human abdominal aortic aneurysm. Eur J Vasc Endovasc Surg. 2003;25:540–5. doi: 10.1053/ejvs.2002.1857. [DOI] [PubMed] [Google Scholar]
- 5.Tanaskovic I, Mladenovic-Mihailovic A, Usaj-Knezevic S, Stankovic V, Aleksic A, Kastratovic T, et al. Histochemical and immunohistochemical analysis of ruptured atherosclerotic abdominal aortic aneurysm wall. Vojnosanit Pregl. 2010;67:959–64. doi: 10.2298/vsp1012959t. [DOI] [PubMed] [Google Scholar]
- 6.Hou LW, Liao MF, Weng JF, Yang L, Zou SL, Jing ZP. Expression of α-smooth muscle actin in human's thoracic aorta dissection. Prog Mod Biomed. 2010;10:113–4. [Google Scholar]
- 7.Vukovic I, Lackovic V, Todorivic V, Kanjuh V, Ilic S. Cytohistologic and immunohistochemical characteristics of the aortic intima and media in coarctation of the aorta of the adult type. Srp Arh Celok Lek. 2004;132(Suppl 1):66–71. doi: 10.2298/sarh04s1066v. [DOI] [PubMed] [Google Scholar]
- 8.Kostianets O, Antoniuk S, Filonenko V, Kiyamova R. Immunohistochemical analysis of medullary breast carcinoma autoantigens in different histological types of breast carcinomas. Diagn Pathol. 2012;7:161. doi: 10.1186/1746-1596-7-161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yuan SM, Wang YQ, Shen Y, Jing H. Transforming growth factor-βin graft vessels:histology and immunohistochemistry. Clinics (Sao Paulo) 2011;66:895–901. doi: 10.1590/S1807-59322011000500029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Milewicz DM, Guo DC, Tran-Fadulu V, Lafont AL, Papke CL, Inamoto S, et al. Genetic basis of thoracic aortic aneurysms and dissections:focus on smooth muscle cell contractile dysfunction. Annu Rev Genomics Hum Genet. 2008;9:283–302. doi: 10.1146/annurev.genom.8.080706.092303. [DOI] [PubMed] [Google Scholar]
- 11.Guo DC, Papke CL, Tran-Fadulu V, Regalado ES, Avidan N, Johnson RJ, et al. Mutations in smooth muscle alpha-actin (ACTA2) cause coronary artery disease, stroke, and Moyamoya disease, along with thoracic aortic disease. Am J Hum Genet. 2009;84:617–27. doi: 10.1016/j.ajhg.2009.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Humphrey JD, Taylor CA. Intracranial and abdominal aortic aneurysms:similarities, differences, and need for a new class of computational models. Annu Rev Biomed Eng. 2008;10:221–46. doi: 10.1146/annurev.bioeng.10.061807.160439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gao F, Watanabe M, Matsuzawa T. Stress analysis in a layered aortic arch model under pulsatile blood flow. Biomed Eng Online. 2006;5:25. doi: 10.1186/1475-925X-5-25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kim HW, Stansfield BK. Genetic and epigenetic regulation of aortic aneurysms. Biomed Res Int. 2017;2017:7268521. doi: 10.1155/2017/7268521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Shantikumar S, Ajjan R, Porter KE, Scott DJ. Diabetes and the abdominal aortic aneurysm. Eur J Vasc Endovasc Surg. 2010;39:200–7. doi: 10.1016/j.ejvs.2009.10.014. [DOI] [PubMed] [Google Scholar]
- 16.Peuler JD, Phare SM, Iannucci AR, Hodorek MJ. Differential inhibitory effects of antidiabetic drugs on arterial smooth muscle cell proliferation. Am J Hypertens. 1996;9:188–92. doi: 10.1016/0895-7061(95)00393-2. [DOI] [PubMed] [Google Scholar]
- 17.Bennett MR, Sinha S, Owens GK. Vascular smooth muscle cells in atherosclerosis. Circ Res. 2016;118:692–702. doi: 10.1161/CIRCRESAHA.115.306361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tonar Z, Kubíková T, Prior C, Demjén E, Liska V, Králícková M, et al. Segmental and age differences in the elastin network, collagen, and smooth muscle phenotype in the tunica media of the porcine aorta. Ann Anat. 2015;201:79–90. doi: 10.1016/j.aanat.2015.05.005. [DOI] [PubMed] [Google Scholar]
- 19.Witter K, Tonar Z, Matejka VM, Martinca T, Jonák M, Rokosny S, et al. Tissue reaction to three different types of tissue glues in an experimental aorta dissection model:a quantitative approach. Histochem Cell Biol. 2010;133:241–59. doi: 10.1007/s00418-009-0656-3. [DOI] [PubMed] [Google Scholar]
- 20.Stary HC, Chandler AB, Dinsmore RE, Fuster V, Glagov S, Insull W, Jr, et al. A definition of advanced types of atherosclerotic lesions and a histological classification of atherosclerosis. A report from the Committee on Vascular Lesions of the Council on Arteriosclerosis, American Heart Association. Circulation. 1995;92:1355–74. doi: 10.1161/01.cir.92.5.1355. [DOI] [PubMed] [Google Scholar]
- 21.Kríková V, Korabecná M, Kocová J, Treska V, Molácek J, Tonar Z, et al. Quantification of plasminogen activator inhibitor type 1 in the aortic wall. Int Angiol. 2009;28:44–9. [PubMed] [Google Scholar]
- 22.Houdek K, Molácek J, Treska V, Krízková V, Eberlová L, Boudová L, et al. Focal histopathological progression of porcine experimental abdominal aortic aneurysm is mitigated by atorvastatin. Int Angiol. 2013;32:291–306. [PubMed] [Google Scholar]
- 23.Eberlová L, Tonar Z, Witter K, Krízková V, Nedorost L, Korabecná M, et al. Asymptomatic abdominal aortic aneurysms show histological signs of progression:a quantitative histochemical analysis. Pathobiology. 2013;80:11–23. doi: 10.1159/000339304. [DOI] [PubMed] [Google Scholar]
- 24.Zhang J, Wang L, Fu W, Wang C, Guo D, Jiang J, Wang Y. Smooth muscle cell phenotypic diversity between dissected and unaffected thoracic aortic media. J Cardiovasc Surg (Torino) 2013;54:511–21. [PubMed] [Google Scholar]


