Abstract
The aim of the present study was to examine the role of Wnt signaling in lipopolysaccharide (LPS)-induced acute respiratory distress syndrome (ARDS). ARDS was induced by LPS and compared in mice treated with either glycogen synthase kinase-3β inhibitor (GSKI) or PBS. The protein expression levels of interleukin (IL)-6, IL-8, tumor necrosis factor (TNF)-α, IL-17, IL-18 and IL-1β in the bronchoalveolar lavage fluid (BALF) were examined using murine cytokine-specific enzyme-linked immunosorbent assays. The accumulation of neutrophils and macrophages in the BALF were detected using flow cytometry. The extent of pathological lesions was evaluated using an immunohistochemical assay. The differentiation of mesenchymal stem cells (MSCs) into type II alveolar (ATII) epithelial cells was analyzed using immunofluorescence staining. Treatment with GSKI led to maintained body weights and survival in mice with LPS-induced ARDS. Treatment with GSKI effectively reduced the levels of total protein, albumin, IgM and keratinocyte growth factor in the BALF. Smith scores showed that GSKI significantly alleviated LPS-induced lung injury. GSKI also functioned to reduce inflammatory cell accumulation and pro-inflammatory cytokine secretion. Finally, it was found that GSKI promoted the differentiation of MSCs into ATII epithelial cells in vivo. Taken together, the GSKI-treated mice exhibited reduced acute lung injury through inhibited intra-fluid inflammatory cell infiltration and decreased expression of pro-inflammatory cytokines, and GSKI increased the differentiation of MSCs into ATII epithelial cells.
Keywords: Wnt signaling, inflammation, neutrophil, cytokine, respiratory distress syndrome
Introduction
Acute respiratory distress syndrome (ARDS), a leading cause of morbidity and mortality in patients in critical care (1–5), may be caused by several virulent substances, however, the severe inflammation, which accompanies the condition is most frequently linked to bacteria, including lipopolysaccharide (LPS) and lipoteichoic acid (6). LPS has been previously administered in experimental animals, causing continuous sepsis and concomitant ARDS-like lung injuries, including polymorphonuclear neutrophil sequestration and lung edema (7). Although recombinant human activated protein C has been developed for use in treating human sepsis (8), it was found not to demonstrate a significant effect in the Prowess-Shock trial (9). Therefore, additional sepsis targets have been established, including complement C5a (10) and its receptor (11), macrophage migration inhibitory factor (12), high-mobility group box 1 protein (13) and histones (14). Pharmaceuticals are being developed against each of these targets; however, no drugs have achieved successful treatment of sepsis. Therefore, further investigations to identify novel therapeutic targets are required.
Wnt pathways, which are crucial for regulating the mechanisms required for the proliferation, development, and differentiation of cells and organisms, can be further characterized as canonical and noncanonical Wnt pathway branches. The canonical Wnt signaling pathway is key in cell proliferation and motility, cell fate decisions, and cell polarity during embryonic development and adult tissue homeostasis (15). Wnts and their downstream canonical signaling pathways also have critical effects on the self-renewal and differentiation of mesenchymal stem cells (MSCs) (16), which possess multipotency and immunoregulatory properties, have been shown to differentiate into alveolar epithelial cells, promote re-epithelialization, alleviate inflammation, improve pathological impairment, and reduce mortality rates in ARDS models (17–19). However, due to the low engraftment of MSCs and the differentiation rates in lung tissues of these models, they have limited therapeutic effect (20,21). Therefore, clarification of the mechanisms underlying the Wnt pathway in mediating MSCs or other cell functions in ARDS may lead to improved cellular retention in injured lung tissue.
The present study focused on the use of Wnt signaling to activate SB216763, a glycogen synthase kinase-3β inhibitor (GSKI), to alleviate the inflammation and sepsis caused by Wnt signaling on ARDS. An LPS-induced ARDS model was used to investigate the condition and possible associated mechanisms. The aim of the present study was to provide a novel perspective and more detailed understanding of the etiology of ARDS, and provide evidence for a potential novel therapeutic strategy.
Materials and methods
Reagents and antibodies
Murine interleukin (IL)-6, IL-8, tumor necrosis factor (TNF)-α, IL-17, IL-18 and IL-1β enzyme-linked immunosorbent assay (ELISA) kits were purchased from Invitrogen Life Technologies; Thermo Fisher Scientific, Inc. (Waltham, MA, USA). Rat anti-mouse-Ly-6 G (cat. no. 551495) monoclonal antibodies (mAbs) were purchased from BD Pharmingen (San Diego, CA, USA). Rat anti-mouse F4/80 (clone A3-1) mAb (cat. no. MCA497GA) was acquired from Serotec (Bio-Rad Laboratories, Inc., Hercules, CA, USA). FITC-conjugated sheep anti-rat IgG mAb (cat. no. PA1-28638) was purchased from Pierce (Thermo Fisher Scientific, Inc.). Rabbit anti mouse SP-C polyclonal Abs (cat. no. bs-10067R) was acquired from Bioss Inc. (Woburn, MA, USA). Total protein, albumin and keratinocyte growth factor (KGF) ELISA kits and Texas Red-conjugated goat anti-rabbit IgG (H + L) (cat. no. ABIN287315) were acquired from Antibodies-Online GmbH (Aachen, Germany).
Preparation of experimental animals
A total of 144 specific pathogen-free 7–8-week-old male C57/B6 mice weighing 20–25 g were obtained from Shanghai SLAC Laboratory Animal Co., Ltd. (Shanghai, China), and were housed in the animal facility of the First Affiliated Hospital of Soochow University (Suzhou, China) under specific pathogen-free conditions. A 12-h light/dark cycle and 19–21°C ambient temperature were maintained during the entire course of the investigation. The animals were housed in groups of 5, and fed regular laboratory chow and water ad libitum. All animal experiments performed in the present study conformed to the Guide for the Care and Use of Laboratory Animals (22) and were approved by the Institutional Animal Care and Use Committee of Soochow University (Suzhou, China).
Murine model of LPS-induced ARDS
The murine model of LPS-induced ARDS was established as previously reported (23). Briefly, the mice were first anesthetized with an intraperitoneal injection of 1.8% (v/v) Avertin (Sigma-Aldrich; Merck KGaA, Darmstadt, Germany) at a dose of 0.20 ml/10 g body weight, and received a single dose of LPS (100 µg intratracheally) from Escherichia coli serotype 0111:B4 (Sigma-Aldrich; Merck KGaA) in 50 µl sterile normal saline (24). The mice were then allowed to recover in a 100% oxygen chamber until fully awake. The control mice received 0.9% PBS instead of LPS. The mice were then sacrificed humanely at indicated time points of day 3 and day 14 following LPS challenge to collect tissues for analysis. The initial experiment showed that 20 mg/kg of the GSKI (SB216763; Selleck, Houston, TX, USA) was effective at significantly inhibiting the effect of GSK-3β and activating WNT signaling. Therefore, 20 mg/kg of GSKI SB216763 was used for the inhibition experiments in the present study.
Cytokine and protein measurements in bronchoalveolar lavage fluid (BALF)
According to a previously described method (23), BALF was collected by flushing 1 ml ice-cold PBS back and forth three times through a tracheal cannula, and then centrifuged at 1,000 × g at 4°C for 10 min. The protein concentrations of IL-6 (cat. no. BMS603-2), IL-8 (cat. no. EMCXCL15), TNF-α (cat. no. BMS607-3), IL-17 (cat. no. BMS6001), IL-18 (cat. no. BMS618-3) and IL-1β (cat. no. EM2IL1B2) in the supernatant were measured using murine cytokine-specific ELISA kits (Invitrogen Life Technologies; Thermo Fisher Scientific, Inc.) in strict accordance with the manufacturer's protocol. The quantities of total protein (cat. no. ABIN996404), albumin (cat. no. ABIN2756308) and KGF (cat. no. ABIN2703018) in the BALF were measured as markers of epithelial permeability using ELISA kits (Antibodies-Online GmbH).
Evaluation of lung edema
Lung edema was evaluated according to the ratio of lung wet weight to body weight (LWW/BW) measured, as previously described (25). Briefly, the whole lung was removed and cleared of all extrapulmonary tissues, and the LWW/BW was calculated based on the values of the respective weights (mg/g).
Determination of neutrophils and macrophages
According to a previously described method (22), BALF was obtained by instilling 0.9% NaCl, containing 0.6 mmol/l ethylenediaminetetraacetic acid, in two separate 0.5 ml aliquots. The fluid was recovered by gentle suction and placed on ice for immediate processing. An aliquot of the BALF was processed immediately for total and differential cell counts. The remainder of the BALF was centrifuged at 1,200 × g at 4°C for 10 min, following which the supernatant was removed aseptically and stored in individual aliquots at −80°C. The numbers of neutrophils and macrophages were calculated as the percentage of neutrophils and macrophages multiplied by the total number of cells in the BALF sample using flow cytometry (FCM). All analyses were performed in a blinded-manner.
Histopathology
Lung tissues were fixed in 4% paraformaldehyde at 37°C for 1 h, embedded in paraffin and cut into 5-µm thick sections. The tissue sections were stained with hematoxylin and eosin, and images were captured (magnification, ×200) using a fluorescence microscope (MZ16; Leica Microsystems GmbH, Wetzlar, Germany). An investigator, who was blinded to the identity of the slides, evaluated the images and lung injury scores were assigned, as previously described (23,26). In brief, the extent of the pathological lesions was graded between 0 and 3 as shown in Table I. The score for each animal was calculated by dividing the total score for the number of sections observed.
Table I.
Smith scores of the extent of pathological lesions.
| Score | Alveolar hemorrhage | Extent of fibrin | Alveolar infiltration/field |
|---|---|---|---|
| 0 | No hemorrhage | No fibrin in alveolar | <5 cells |
| 1 | >5 erythrocytes per alveolus in 1–5 alveoli | Fibrin occupation <1/3 of field | 5–10 cells |
| 2 | >5 erythrocytes per alveolus in 5–10 alveoli | Fibrin occupation <2/3 but >1/3 of field | 10–20 cells |
| 3 | >5 erythrocytes per alveolus in >10 alveoli | Fibrin occupation >2/3 of field | >20 cells |
Statistical analysis
The data are presented as the mean ± standard deviation. Statistical analyses were performed using SPSS 18.0 software (SPSS, Inc., Chicago, IL, USA). Comparisons among multiple groups were performed using one-way analysis of variance followed by Bonferroni's post hoc test if the data were normally distributed. P<0.05 was considered to indicate a statistically significant difference.
Results
GSKI leads to maintained body weight and improved survival of mice with LPS-induced ARDS
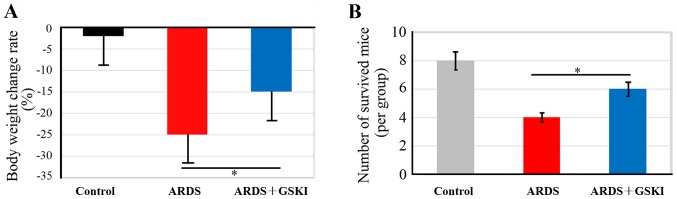
The present study evaluated the effect of GSKI treatment on body weight maintenance and survival of mice with LPS-induced ARDS, which had lost ~25% of their body weight (Fig. 1A). GSKI consequently abrogated the weight loss of the mice with LPS-induced ARDS (P<0.05). In addition, the mortality rate was ~40% in the LPS-induced ARDS mice, however, with GSKI treatment, the mice had an increased survival rate, up to 60% at 40 h intervals (Fig. 1B; P<0.05), which suggested that the Wnt signaling pathway was effective in preventing the development of ARDS.
Figure 1.
GSKI treatment maintains body weight and increases survival rates of ARDS mice. (A) Body weight was measured 3 days following LPS challenge. (B) Mice were examined for survival. All values are presented as the mean ± standard deviation (n=6–8 animals). *P<0.05. LPS, lipopolysaccharide; ARDS, acute respiratory distress syndrome.
GSKI reduces LPS-induced lung permeability
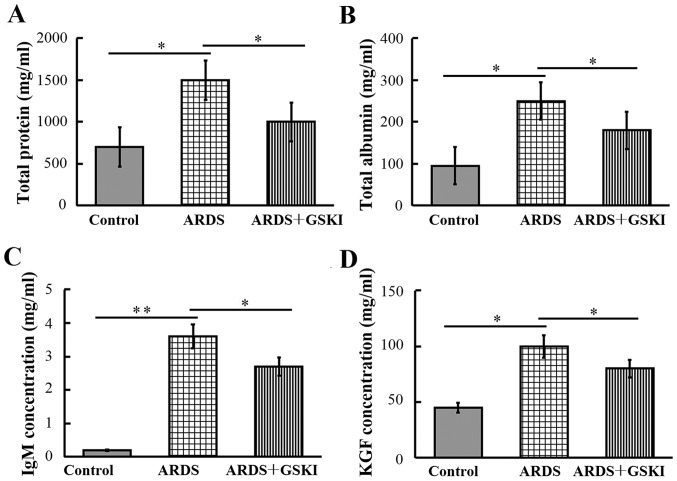
The present study subsequently examined the concentrations of total protein, albumin, IgM and KGF in BALF, to evaluate the integrity of the alveolar-capillary membrane barrier and to assess pulmonary vascular leakage, the latter of which is a marker for ARDS. The results revealed that the levels were significantly increased in LPS-challenged mice compared with those in the control mice (P<0.05 and P<0.01). Following GSKI treatment, the levels of total protein, albumin, IgM and KGF were reduced (P<0.05; Fig. 2).
Figure 2.
Treatment with GSKI reduces LPS-induced lung permeability. The levels of (A) total protein, (B) total albumin, (C) IgM and (D) KGF in bronchoalveolar lavage fluid were determined 3 days following LPS challenge. All values are presented as the mean ± standard deviation (n=6–8 animals). *P<0.05 and **P<0.01. GSKI, glycogen synthase kinase-3β inhibitor; LPS, lipopolysaccharide; ARDS, acute respiratory distress syndrome; KGF, keratinocyte growth factor.
GSKI alleviates histopathological characteristics of mice with LPS-induced ARDS
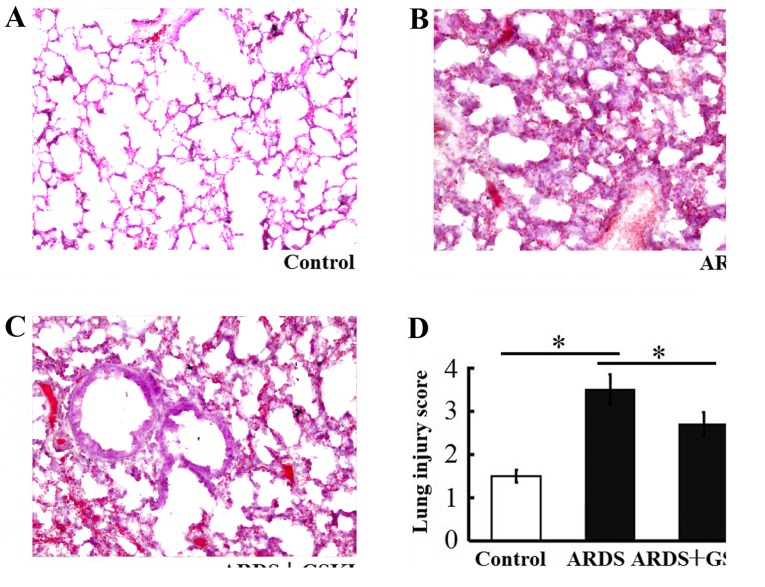
The present study found increased thickening of the alveolar wall, alveolar and interstitial inflammatory cell infiltration, hemorrhaging alveolar exudates, and edema in the lung tissue of mice following LPS-induced lung injury. The Smith score for quantifying lung injury was also increased. By contrast, the histopathological characteristics and the Smith score were reduced in the LPS+GSKI group, compared with those in the LPS group (P<0.05; Fig. 3A-D).
Figure 3.
Effect of GSKI on the histopathology of LPS-induced lung injury and survival over 14 days. Histopathological analysis of lung tissues from mice in the (A) control, (B) ARDS and (C) ARDS+GSKI groups were performed at indicated times following LPS challenge (hematoxylin and eosin staining; magnification, ×200) (D) Quantification of lung injury showed a significant reduction in the severity of lung injury in the LPS+GSKI group. The change was more marked in the LPS group, compared with that in the LPS+GSKI group. All values are presented as the mean ± standard deviation (n=6–8 animals). *P<0.05. GSKI, glycogen synthase kinase-3β inhibitor; LPS, lipopolysaccharide; ARDS, acute respiratory distress syndrome.
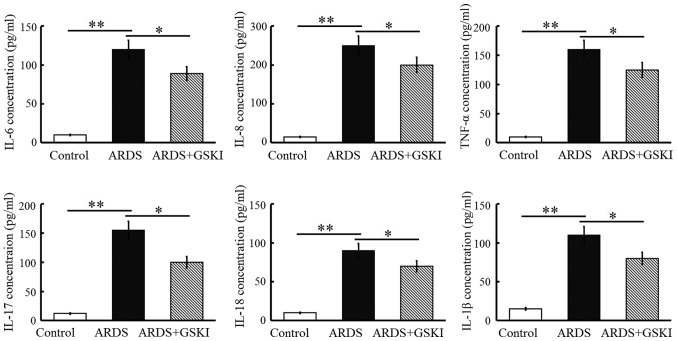
GSKI attenuates acute LPS-induced pulmonary inflammation
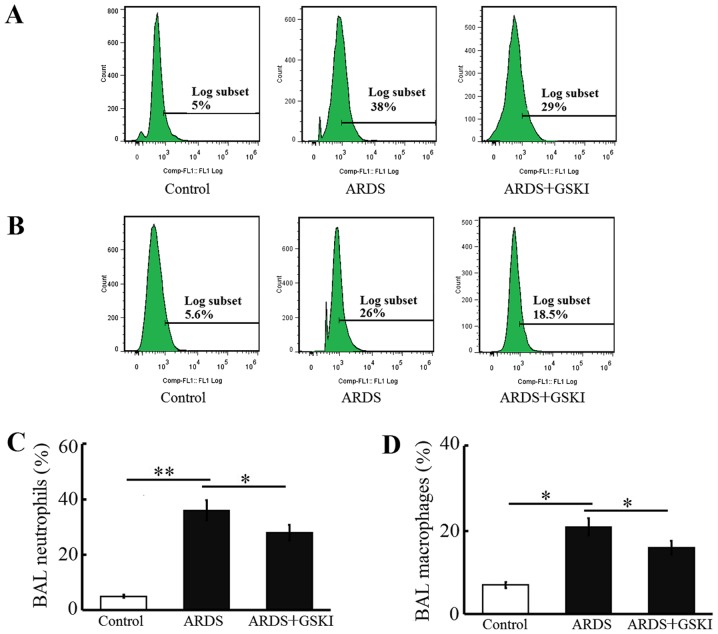
The present study examined inflammatory cell (neutrophil and macrophage) counts, pro-inflammatory cytokines and chemokines in BALF to further assess the anti-inflammatory effect of Wnt signaling in LPS-induced ARDS. The neutrophil and macrophage counts were increased (P<0.05 and P<0.01; Fig. 4) and the levels of pro-inflammatory cytokines, including IL-6, IL-8, TNF-α, IL-17, IL-18, and IL-1β, were all significantly elevated in response to the LPS challenge (P<0.05 and P<0.01; Fig. 5). GSKI administration was effective in decreasing the inflammatory cell counts, and the levels of pro-inflammatory cytokines and chemokines (P<0.05; Figs. 4 and 5).
Figure 4.
Treatment with GSKI reduces LPS-induced pulmonary inflammatory cell accumulation. Groups of mice were challenged with LPS and treated with GSKI 30 min later. (A) Neutrophils; (B) macrophages; (C) ratios of Ly6G-positive neutrophils in control (open bars), LPS-treated and LPS+GSKI-treated (black bars) mice using FCM; (D) ratios of F4/80-positive macrophages in control (open bars), LPS-treated and LPS+GSKI-treated (black bars) mice using FCM. Data are presented as the mean ± standard deviation of one experiment consisting of three replicates. Experiments were performed in triplicate. *P<0.05 and **P<0.01. GSKI, glycogen synthase kinase-3β inhibitor; LPS, lipopolysaccharide; ARDS, acute respiratory distress syndrome; FCM, flow cytometry.
Figure 5.
GSKI treatment attenuates LPS-induced pulmonary inflammation. LPS-challenged mice were treated with GSKI 30 min following LPS challenge, and pro-inflammatory cytokines and chemokines in bronchoalveolar lavage fluid were determined on day 3. Data are presented as the mean ± standard deviation of one experiment consisting of three replicates. *P<0.05; and **P<0.01. GSKI, glycogen synthase kinase-3β inhibitor; LPS, lipopolysaccharide; ARDS, acute respiratory distress syndrome; IL, interleukin; TNF-α, tumor necrosis factor-α.
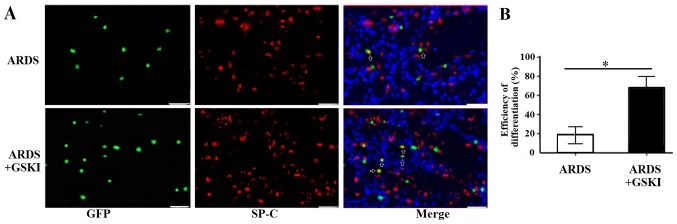
GSKI promotes the differentiation of MSCs into ATII epithelial cells in vivo
The present study also evaluated the differentiation of MSCs into ATII cells 14 days following treatment with GSKI by analyzing the expression of the ATII cell marker, SP-C, through immunofluorescence staining. The SP-C (red) and MSCs (green) co-localized in the lung tissue (yellow) of the LPS and LPS+GSKI groups; however, the rate of differentiation was higher following GSKI treatment, compared with group without GSKI treatment (P<0.05; Fig. 6A and B).
Figure 6.
Effect of GSKI on retention of MSCs in the lungs 14 days following LPS challenge in the LPS control and LPS+GSKI groups. (A) Immunofluorescent staining to detect MSC engraftment in lung tissues. White arrows indicate green fluorescent protein-positive cells; nuclei were stained blue with DAPI (magnification, ×200; scale bar, 20 µm). (B) All values are presented as the mean ± standard deviation (n=6–8 animals). *P<0.05. ARDS, acute respiratory distress syndrome; GSKI, glycogen synthase kinase-3β inhibitor; MSCs, mesenchymal stem cells.
Discussion
The canonical Wnt pathway is key in the development, differentiation, and physiological functions of cells and organisms (27). Canonical Wnt ligands bind to Frizzled co-receptors and low-density lipoprotein receptor-related proteins 5 or 6, which results in GSK-3β inhibition and the accumulation of β-catenin translocating into the nucleus, ultimately regulating target gene expression (28). This process makes β-catenin the pivotal signaling regulator of the canonical Wnt pathway. Due to its importance in the pathological process of numerous diseases, the present study examined the effect of Wnt signaling in experimental ARDS.
GSKI has been applied as a canonical Wnt signaling activator in previous studies. Troussard et al (29) reported that the phosphorylation of integrin-linked kinases inhibited GSK-3I activity, but activated AP-1 activity, resulting in regulation of the survival, proliferation, differentiation and migration of cells. In addition, the reduction of cAMP response element binding protein activity has been shown to facilitate apoptosis (30,31). Due its efficiency and stability, the present study used GSKI as treatment reagent administered to LPS-induced ARDS mice. The results in vivo revealed that GSKI functionally alleviated LPS-induced acute lung injury, and abrogated inflammatory cell infiltration into the BALF and pro-inflammatory cytokine secretion. In addition, GSKI was shown to induce the differentiation of MSCs into ATII epithelial cells, which is a positive outcome in ARDS rehabilitation. Therefore, the present study revealed a novel mechanism underlying the mediation of experimental ARDS by Wnt signaling. Although further investigations are required, GSKI was shown to be a prospective candidate as a chemopreventive agent in clinical settings for therapy for ARDS.
Although it was determined that GSKI attenuated lung injury, as shown by its effect on body weight and the survival of the mice, and reduction of lung permeability (Figs. 1 and 2), there are several signal transduction processes involved in the pathogenesis of ARDS, making it complex to investigate. Investigations have focused on Wnt signaling, which is necessary for the proliferation and migration of cells, expression of multiple cytokines, and inflammatory responses (32).
To the best of our knowledge, inflammatory responses are the primary cause of LPS-induced lung injury. During the acute pathological process, inflammatory cells, including neotrophils and macrophages, are recruited to lung lesions, and they are induced to secrete pro-inflammatory cytokines to further accelerate inflammatory responses and deteriorate ARDS. Thus, effective inhibition of inflammatory responses is fundamental treatment strategy for ARDS. It is known that the canonical Wnt pathway is a preventive factor in the ARDS pathological process. Consistently, the activation of Wnt signaling by GSKI in the present study retarded inflammatory cell infiltration and suppressed the expression of pro-inflammatory cytokines. It was concluded that the alleviation of experimental ARDS was associated with the effects of Wnt signaling on the cellular and protein regulation of the inflammatory response.
MSCs have been shown to migrate to and engraft in injured lungs, and to differentiate into lung epithelial cells in vivo, indicating their potential in the treatment of ARDS (33–36). Therefore, promoting MSCs to differentiate into lung epithelial cells may be another route to facilitate ARDS restoration. The present study examined whether Wnt signaling exhibits protective functions via promoting MSC differentiation into lung epithelial cells. As expected, it was found that activation of the canonical Wnt/β-catenin pathway by GSKI promoted the differentiation of MSCs into ATII cells and migration to injured lung tissues, as detected using immunofluorescence assay, which was consistent with previous reports (37).
In conclusion, the results of the present study revealed that GSKI was an efficient activator for Wnt signaling in LPS-induced ARDS, and this activating effect may facilitate MSC differentiation into lung epithelial cells, reduce inflammatory responses and thus alleviate lung injury. These results confirmed GSKI as a prospective candidate chemopreventive agent in clinical settings for ARDS therapy.
Acknowledgements
This study was supported by the National Natural Science Foundation of China (grant no. 31270940), Jiangsu Provincial Medical Youth Talent (grant no. QNRC2016718), the Clinical Medical Center of Suzhou (grant no. Szzx201502) and the Clinical Key Specialty Project of China and Suzhou Municipal Natural Science Foundation (grant nos. SYS201448 and SYSD2015107).
References
- 1.Ware LB, Matthay MA. The acute respiratory distress syndrome. N Engl J Med. 2000;342:1334–1349. doi: 10.1056/NEJM200005043421806. [DOI] [PubMed] [Google Scholar]
- 2.Goss CH, Brower RG, Hudson LD, Rubenfeld GD. ARDS Network: Incidence of acute lung injury in the United States. Crit Care Med. 2003;31:1607–1611. doi: 10.1097/01.CCM.0000063475.65751.1D. [DOI] [PubMed] [Google Scholar]
- 3.Mendez JL, Hubmayr RD. New insights into the pathology of acute respiratory failure. Curr Opin Crit Care. 2005;11:29–36. doi: 10.1097/00075198-200502000-00005. [DOI] [PubMed] [Google Scholar]
- 4.Rubenfeld GD, Caldwell E, Peabody E, Weaver J, Martin DP, Neff M, Stern EJ, Hudson LD. Incidence and outcomes of acute lung injury. N Engl J Med. 2005;353:1685–1693. doi: 10.1056/NEJMoa050333. [DOI] [PubMed] [Google Scholar]
- 5.Quílez ME, López-Aguilar J, Blanch L. Organ crosstalk during acute lung injury, acute respiratory distress syndrome, and mechanical ventilation. Curr Opin Crit Care. 2012;18:23–28. doi: 10.1097/MCC.0b013e32834ef3ea. [DOI] [PubMed] [Google Scholar]
- 6.Middelveld RJ, Alving K. Synergistic septicemic action of the gram-positive bacterial cell wall components peptidoglycan and lipoteichonic acid in the pig in vivo. Shock. 2000;13:297–306. doi: 10.1097/00024382-200004000-00008. [DOI] [PubMed] [Google Scholar]
- 7.Yoshinari D, Takeyoshi I, Koibuchi Y, Matsumoto K, Kawashima Y, Koyama T, Ohwada S, Morishita Y. Effects of a dual inhibitor of tumor necrosis factor-alpha and interleukin-1 on lipopolysaccharide-induced lung injury in rats: Involvement of the p38 mitogen-activated protein kinase pathway. Crit Care Med. 2001;29:628–634. doi: 10.1097/00003246-200103000-00029. [DOI] [PubMed] [Google Scholar]
- 8.Tanaka S, Nishiumi S, Nishida M, Mizushina Y, Kobayashi K, Masuda A, Fujita T, Morita Y, Mizuno S, Kutsumi H, et al. Vitamin K3 attenuates lipopolysaccharide-induced acute lung injury through inhibition of nuclear factor-kappaB activationce. Clin Exp Immunol. 2010;160:283–292. doi: 10.1111/j.1365-2249.2009.04083.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ranieri VM, Thompson BT, Barie PS, Dhainaut JF, Douglas IS, Finfer S, Gårdlund B, Marshall JC, Rhodes A, Artigas A, et al. Drotrecogin alfa (activated) in adults with septic shock. N Engl J Med. 2012;366:2055–2064. doi: 10.1056/NEJMoa1202290. [DOI] [PubMed] [Google Scholar]
- 10.Huber-Lang MS, Younkin EM, Sarma JV, McGuire SR, Lu KT, Guo RF, Padgaonkar VA, Curnutte JT, Erickson R, Ward PA. Complement-induced impairment of innate immunity during sepsis. J Immunol. 2002;169:3223–3231. doi: 10.4049/jimmunol.169.6.3223. [DOI] [PubMed] [Google Scholar]
- 11.Riedemann NC, Guo RF, Neff TA, Laudes IJ, Keller KA, Sarma VJ, Markiewski MM, Mastellos D, Strey CW, Pierson CL, et al. Increased C5a receptor expression in sepsis. J Clin Invest. 2002;110:101–108. doi: 10.1172/JCI0215409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lehmann LE, Novender U, Schroeder S, Pietsch T, von Spiegel T, Putensen C, Hoeft A, Stüber F. Plasma levels of macrophage migration inhibitory factor are elevated in patients with severe sepsis. Intensive Care Med. 2001;27:1412–1415. doi: 10.1007/s001340101022. [DOI] [PubMed] [Google Scholar]
- 13.Ulloa L, Ochani M, Yang H, Tanovic M, Halperin D, Yang R, Czura CJ, Fink MP, Tracey KJ. Ethyl pyruvate prevents lethality in mice with established lethal sepsis and systemic inflammation; Proc Natl Acad Sci; 2002; USA: pp. 12351–12356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wang Y, Li M, Stadler S, Correll S, Li P, Wang D, Hayama R, Leonelli L, Han H, Grigoryev SA, et al. Histone hypercitrullination mediates chromatin decondensation and neutrophil extracellular trap formation. J Cell Biol. 2009;184:205–213. doi: 10.1083/jcb.200806072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bouldin CM, Manning AJ, Peng YH, Farr GH, III, Hung KL, Dong A, Kimelman D. Wnt signaling and tbx16 form a bistable switch to commit bipotential progenitors to mesoderm. Development. 2015;142:2499–2507. doi: 10.1242/dev.124024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Etheridge SL, Spencer GJ, Heath DJ, Genever PG. Expression profiling and functional analysis of wnt signaling mechanisms in mesenchymal stem cells. Stem Cells. 2004;22:849–860. doi: 10.1634/stemcells.22-5-849. [DOI] [PubMed] [Google Scholar]
- 17.Ortiz LA, Gambelli F, McBride C, Gaupp D, Baddoo M, Kaminski N, Phinney DG. Mesenchymal stem cell engraftment in lung is enhanced in response to bleomycin exposure and ameliorates its fibrotic effects; Proc Natl Acad Sci USA; 2003; pp. 8407–8411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yamada M, Kubo H, Kobayashi S, Ishizawa K, Numasaki M, Ueda S, Suzuki T, Sasaki H. Bone marrow-derived progenitor cells are important for lung repair after lipopolysaccharide-induced lung injury. J Immunol. 2004;172:1266–1272. doi: 10.4049/jimmunol.172.2.1266. [DOI] [PubMed] [Google Scholar]
- 19.Kotton DN, Ma BY, Cardoso WV, Sanderson EA, Summer RS, Williams MC, Fine A. Bone marrow-derived cells as progenitors of lung alveolar epithelium. Development. 2001;128:5181–5188. doi: 10.1242/dev.128.24.5181. [DOI] [PubMed] [Google Scholar]
- 20.Gupta N, Su X, Popov B, Lee JW, Serikov V, Matthay MA. Intrapulmonary delivery of bone marrow-derived mesenchymal stem cells improves survival and attenuates endotoxin-induced acute lung injury in mice. J Immunol. 2007;179:1855–1863. doi: 10.4049/jimmunol.179.3.1855. [DOI] [PubMed] [Google Scholar]
- 21.Geburek F, Mundle K, Conrad S, Hellige M, Walliser U, van Schie HT, van Weeren R, Skutella T, Stadler PM. Tracking of autologous adipose tissue-derived mesenchymal stromal cells with in vivo magnetic resonance imaging and histology after intralesional treatment of artificial equine tendon lesions - a pilot study. Stem Cell Res Ther. 2016;7:21. doi: 10.1186/s13287-016-0281-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Takaoka Y, Goto S, Nakano T, Tseng HP, Yang SM, Kawamoto S, Ono K, Chen CL. Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) prevents lipopolysaccharide (LPS)-induced, sepsis-related severe acute lung injury in mice. Sci Rep. 2014;4:5204. doi: 10.1038/srep05204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Mei SH, McCarter SD, Deng Y, Parker CH, Liles WC, Stewart DJ. Prevention of LPS-induced acute lung injury in mice by mesenchymal stem cells overexpressing angiopoietin 1. PLoS Med. 2007;4:e269. doi: 10.1371/journal.pmed.0040269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fang WF, Cho JH, He Q, Lin MC, Wu CC, Voelkel NF, Douglas IS. Lipid A fraction of LPS induces a discrete MAPK activation in acute lung injury. Am J Physiol Lung Cell Mol Physiol. 2007;293:L336–L344. doi: 10.1152/ajplung.00011.2007. [DOI] [PubMed] [Google Scholar]
- 25.Dong L, He HL, Lu XM, Yang Y, Qiu HB. Modulation of FLT3 signaling targets conventional dendritic cells to attenuate acute lung injury. APMIS. 2012;120:808–818. doi: 10.1111/j.1600-0463.2012.02906.x. [DOI] [PubMed] [Google Scholar]
- 26.Matute-Bello G, Winn RK, Jonas M, Chi EY, Martin TR, Liles WC. Fas (CD95) induces alveolar epithelial cell apoptosis in vivo: Implications for acute pulmonary inflammation. Am J Pathol. 2001;158:153–161. doi: 10.1016/S0002-9440(10)63953-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Logan CY, Nusse R. The Wnt signaling pathway in development and disease. Annu Rev Cell Dev Biol. 2004;20:781–810. doi: 10.1146/annurev.cellbio.20.010403.113126. [DOI] [PubMed] [Google Scholar]
- 28.Gordon MD, Nusse R. Wnt signaling: Multiple pathways, multiple receptors, and multiple transcription factors. J Biol Chem. 2006;281:22429–22433. doi: 10.1074/jbc.R600015200. [DOI] [PubMed] [Google Scholar]
- 29.Troussard AA, Tan C, Yoganathan TN, Dedhar S. Cell-extracellular matrix interactions stimulate the AP-1 transcription factor in an integrin-linked kinase- and glycogen synthase kinase 3-dependent manner. Mol Cell Biol. 1999;19:7420–7427. doi: 10.1128/MCB.19.11.7420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jean D, Harbison M, McConkey DJ, Ronai Z, Bar-Eli M. CREB and its associated proteins act as survival factors for human melanoma cells. J Biol Chem. 1998;273:24884–24890. doi: 10.1074/jbc.273.38.24884. [DOI] [PubMed] [Google Scholar]
- 31.Walton M, Woodgate AM, Muravlev A, Xu R, During MJ, Dragunow M. CREB phosphorylation promotes nerve cell survival. J Neurochem. 1999;73:1836–1842. [PubMed] [Google Scholar]
- 32.Kurosaki T. Checks and balances on developing B cells. Nat Immunol. 2003;4:13–15. doi: 10.1038/ni0103-13. [DOI] [PubMed] [Google Scholar]
- 33.Masterson C, Jerkic M, Curley GF, Laffey JG. Mesenchymal stromal cell therapies: Potential and pitfalls for ARDS. Minerva Anestesiol. 2015;81:179–194. [PubMed] [Google Scholar]
- 34.Li J, Huang S, Wu Y, Gu C, Gao D, Feng C, Wu X, Fu X. Paracrine factors from mesenchymal stem cells: A proposed therapeutic tool for acute lung injury and acute respiratory distress syndrome. Int Wound J. 2014;11:114–121. doi: 10.1111/iwj.12202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Simonson OE, Mougiakakos D, Heldring N, Bassi G, Johansson HJ, Dalén M, Jitschin R, Rodin S, Corbascio M, El Andaloussi S, et al. In vivo effects of mesenchymal stromal cells in two patients with severe acute respiratory distress syndrome. Stem Cells Transl Med. 2015;4:1199–1213. doi: 10.5966/sctm.2015-0021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Grove JE, Lutzko C, Priller J, Henegariu O, Theise ND, Kohn DB, Krause DS. Marrow-derived cells as vehicles for delivery of gene therapy to pulmonary epithelium. Am J Respir Cell Mol Biol. 2002;27:645–651. doi: 10.1165/rcmb.2002-0056RC. [DOI] [PubMed] [Google Scholar]
- 37.Liu AR, Liu L, Chen S, Yang Y, Zhao HJ, Liu L, Guo FM, Lu XM, Qiu HB. Activation of canonical wnt pathway promotes differentiation of mouse bone marrow-derived MSCs into type II alveolar epithelial cells, confers resistance to oxidative stress, and promotes their migration to injured lung tissue in vitro. J Cell Physiol. 2013;228:1270–1283. doi: 10.1002/jcp.24282. [DOI] [PubMed] [Google Scholar]