Abstract
Neoadjuvant chemotherapy (NAC) is a widely-used treatment for breast cancer, as it may render unresectable breast tumors to become resectable. In addition, NAC provides the unique opportunity to assess response to treatments within months rather than years of follow-up. However, predictive markers of tumor response to NAC are lacking. Therefore, the present study aimed to investigate the expression of endoglin, a marker of angiogenesis, and its association with pathologic responses to NAC. Samples from 34 breast cancer patients were obtained prior to and following NAC treatment. Immunohistochemical staining for endoglin and the mechanistic target of rapamycin (mTOR) was performed, and the correlation between the expression of these markers and pathologic response was examined. The overall response rate to NAC of these 34 patients was 67.6%. A mean microvascular density value of 14 served as a threshold score for the increased expression of endoglin. Increased expression of endoglin in primary tumors prior to NAC correlated with improved response in primary tumors (P=0.019) or in primary tumors and regional lymph nodes (P=0.014), when compared with reduced expression of endoglin. Increased expression of mTOR following NAC was additionally correlated with improved response to NAC. The results of the present study demonstrated that the expression of endoglin in breast tumor samples prior to NAC may be a predictor of treatment response. Long-term follow-up of clinical outcome is required to explain the elevation of mTOR expression levels following NAC treatment in responsive tumors.
Keywords: endoglin, breast cancer, neoadjuvant chemotherapy, predictive marker
Introduction
Breast cancer (BC) is one of the most common types of cancer and is the leading cause of cancer mortality among females worldwide, accounting for 25% of all cancer cases and 15% of all cancer-associated mortality among females (1). A number of adjuvant therapies for early BC have been developed; however, the recurrence rate remains as high as 20–30%. Robust clinical and pathological markers determine treatment options, including pathological evaluation of the size of the primary tumor, number of metastatic lymph nodes, lymphovascular invasion, as well as the expression of the estrogen receptor (ER), progesterone receptor (PR), the Ki-67 marker of proliferation and human epidermal growth factor receptor 2 (HER-2) are all established markers (2). Aside from histologic prognostic factors, a number of molecular classifications of BC have been identified, involving the mammaprint test or the OncotypeDX™21-Gene Recurrence Score assay, which may be used to predict the requirement for adjuvant chemotherapy treatment (3).
Neoadjuvant chemotherapy (NAC) describes therapeutic intervention prior to surgery. The aim of NAC in BC is to reduce the size of unresectable tumors in locally advanced or inflammatory BC, thus allowing surgery to be performed (4). For operable tumors, the aim is to downstage the tumor for improved loco-regional control and to increase the conservative surgery rate. As NAC provides the unique opportunity to assess response to chemotherapy within months rather than years of follow-up, it provides the opportunity to assess the efficacy of therapy and to change to an alternative treatment if appropriate. NAC has recently become a popular treatment option, which is widely used to treat patients that fulfill the criteria for adjuvant chemotherapy following surgery, and is considered to be a platform for testing novel therapies. Clinical studies have made use of the in vivo response to conduct sequential tissue biopsies and assess a range of biomarkers of resistance and sensitivity to neoadjuvant treatment (5).
Response to NAC is an excellent indicator of disease outcome (4). Achieving a pathologic complete response (pCR), which is defined as the absence of residual invasive cancer in the breast and ipsilateral axilla lymph nodes, following NAC treatment is associated with improved disease-free survival (DFS) in patients with luminal B/HER-2 positive tumors (6). The US Food and Drug Administration (FDA) has recommended pCR as an end point for the approval of novel agents for neoadjuvant treatment of early-stage BC (7).
Angiogenesis is an essential process in the progression of malignant tumors, as solid tumors cannot grow beyond 1–2 mm in diameter without the formation of new vessels (8). In BC, extensive neovascularization and lymphovascular invasion have been reported to be poor indicators of prognosis (9). Microvascular density (MVD) has become the morphological gold standard to assess angiogenesis in human tumors. Numerous studies have demonstrated that the MVD of BC predicts tumor progression and metastasis, and thus predicts prognosis (10).
Endoglin [also known as cluster of differentiation (CD) 105], is a co-receptor of transforming growth factor-β (TGF-β)-1 and −2, and is expressed on the endothelial cells of peri- and intra-tumoral blood vessels and tumor stromal components (11). Endoglin has demonstrated to be superior to CD34 and CD31 in the evaluation of angiogenesis, as demonstrates greater affinity for the angiogenic endothelium, whereas CD34 and CD31 react nonspecifically with the endothelium of healthy and pathological vessels (12).
The phosphatidylinositol 3-kinase (PI3K)-protein kinase B (Akt)-mechanistic target of rapamycin (mTOR) signaling pathway is an important pathway that is involved in hormone therapy and trastuzumab resistance (13,14). Treatment with an mTOR inhibitor may reverse resistance in advanced BCs (15,16).
The present study examined the association between the therapeutic effects of NAC and the expression of numerous markers, including endoglin and mTOR, in locally advanced BC, in order to determine whether determination of endoglin expression prior to NAC may be used as a predictive marker of treatment response.
Materials and methods
BC tissues
The Ethics Committee of Kaohsiung Chang Gung Memorial Hospital (Niao-Song, Taiwan) approved this study. With permission from the Institutional Review Board of Kaohsiung Chang Gung Memorial Hospital (Kaohsiung, Taiwan), clinical information and archived tissue specimens were collected from 34 patients with BC that were diagnosed and treated in Kaohsiung Chang Gung Memorial Hospital between 2012 and 2014. Biopsy specimens prior to NAC treatment and tumor specimens following NAC treatment were collected for diagnosis and the analysis of the expression of markers. Patients provided written informed consent for the use of their tissue samples for research purposes. In addition, all data were analyzed anonymously. Clinical data including age at diagnosis, clinical and pathological stage, and pathological features, including ER, PR, Ki-67 and HER-2 expression and responses to NAC were obtained from a combined review of clinical and pathological records. Patients routinely underwent computed tomography examination prior to and following NAC treatment. Evaluation of clinical response was measured according to the Response Evaluation Criteria In Solid Tumors guidelines (version 1.1) (17). Resected specimens were sent for pathological examination by pathologists at Kaohsiung Chang Gung Memorial Hospital (Kaohsiung, Taiwan).
Tissue microarray (TMA)
Tumor specimens (following treatment) from archived specimens were collected for TMA blocks. TMA blocks were constructed using the TMA Grand Master system (3DHISTECH Ltd., Budapest, Hungary). Target regions for the array (areas with BC) were identified by marking the areas on hematoxylin and eosin-stained sections from each paraffin-embedded sample. A total of 3 tissue cores with a diameter of 3 mm were transferred from each donor block to the recipient TMA block. Liver or skeletal muscle tissues were placed in the first lane core of the three upper right cores of the TMA block to ensure correct orientation.
Immunohistochemical (IHC) staining
IHC staining procedures were followed as previously described (18). Biopsy specimens and TMA blocks constructed from formalin-fixed paraffin-embedded human BC tissue were sectioned at 3-µm thickness and dried overnight at 37°C. Slides were deparaffinized in xylene and rehydrated through a graded alcohol series to water. For antigen retrieval, the slides were incubated with an anti-endoglin primary antibody (cat. no. NCL-CD105; 1:50; Novocastra™; Leica Biosystems, Ltd., Milton Keynes, UK) for 3 h at room temperature and anti-mTOR primary antibody (cat. no. ab2732; 1:100; Abcam, Cambridge, MA, USA) 1:100 for 2 h at room temperature. Following a wash step with PBS, the UltraVision™ Quanto Detection system HRP (cat. no. TL-125-QHL; 1:10; Thermo Fisher Scientific, Inc., Waltham, MA, USA) for 10 min at room temperature was added. The slides were then analyzed using the Dako Liquid DAB+ Substrate Chromogen system (cat. no. K3468; Dako; Agilent Technologies GmbH, Waldbronn, Germany), followed by counterstaining with hematoxylin 1:1 at room temperature for 1 min and mounting onto coverslips using Entellan® New Mounting Medium (cat. no. 107961; Merck Millipore, Darmstadt, Germany). Incubation of slides without the primary antibody was used as a negative control. Slides were scanned using the Pannoramic SCAN scanner (3DHISTECH Ltd.) for analysis. The staining intensity of these markers was determined by two independent pathologists, and classified as low or high.
Evaluation of MVD
MVD was evaluated as described previously (18). Endoglin positive single cells or clusters of cells clearly in the vessel lumen were considered to be an individual vessel. Areas of inflammation, fibrosis or necrosis, and vessels with a muscle wall were excluded. The sections were scanned (magnification, ×100) by two observers simultaneously to select the hotspots (regions with the highest endoglin staining) of the three tissue array spots for every patient. The number of microvessels in each hotspot were counted (magnification level, ×200) and their density was expressed as the mean number/high-power field. Mean values of endoglin staining were calculated for each individual tumor and used for further analysis.
Statistical analysis
The association between IHC findings and clinical features, including alterations of tumor stage, pathological stage and expression markers were analyzed by Spearman's rank correlation coefficient. SPSS software (version, 10.0; SPSS Inc., Chicago, IL, USA) was used for all calculations. P<0.05 was considered to indicate a statistically significant association.
Results
Tumor responses to NAC
A total of 34 paired specimens were available for staining and analysis from 34 patients. The mean age of the patients was 55 years. All tumors were invasive ductal carcinoma. All patients received NAC including anthracycline and taxane, either administered sequentially or in combination. For breast tumors with HER-2 overexpression, trastuzumab was added into the treatment regimen. The overall response rate of primary tumors was 67.6% (Table I); only 5 (14.7%) patients exhibited disease progression during NAC treatment.
Table I.
Clinical information of patients.
| Clinical information | No. of patients |
|---|---|
| Mean age at time of diagnosis (years) | 55 (33–72) |
| Clinical stage prior to NAC | |
| Stage II | 13 |
| Stage III | 18 |
| Stage IV | 3 |
| Subtype | |
| Luminal A | 9 |
| Luminal B | 9 |
| HER-2 enriched | 8 |
| Triple negative | 8 |
| NAC regimen | |
| EC ×4 | 8 |
| ET ×6 | 18 |
| ETH ×6 | 8 |
| Response to NAC | |
| Progressive disease | 5 |
| Stable disease | 6 |
| Partial response | 18 |
| Complete response | 5 |
NAC, neoadjuvant chemotherapy; EC, epirubicin and cyclophosphamide; EC ×4, EC every 3 weeks for 4 cycles; ET, epirubicin and docetaxel; ETx6: ET every 3 weeks for 6 cycles; ETH, epirubicin, docetaxel and trastuzumab; ETH ×6, ETH every 3 weeks for 6 cycles; HER-2, human epidermal growth factor receptor 2.
High endoglin expression correlates with improved response to NAC
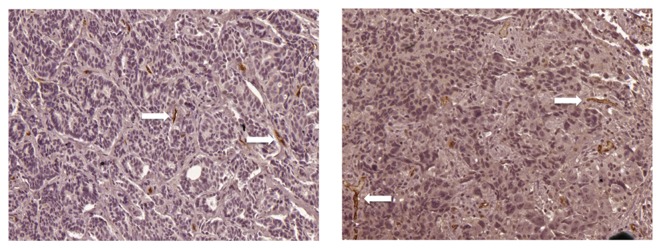
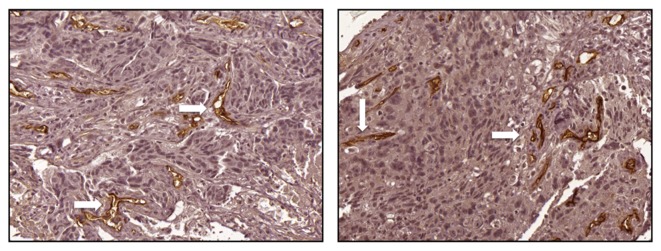
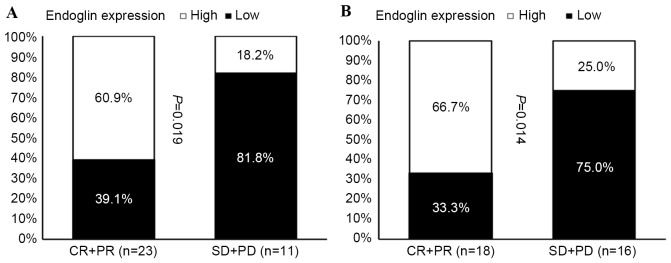
The expression of endoglin was assessed by immunohistochemistry to evaluate the MVD of tumors. A mean value of 14 was selected as the cut-off point for MVD; a value of ≤14 was considered as low expression (Fig. 1) and a value of>14 was considered as high expression (Fig. 2). A high MVD score in the tumor biopsy samples obtained prior to NAC was significantly associated with an improved patient response rate of primary tumors (Fig. 3A), and of primary tumors with regional lymph node involvement (Fig. 3B).
Figure 1.
Representative immunohistochemistry images of reduced expression of endoglin in breast tumor samples following neoadjuvant chemotherapy from patients (magnification, ×200). Positive endoglin staining (white arrows) was observed as thin, linear deposits in the membrane and cytoplasm of endothelial cells in the microvessels.
Figure 2.
Representative immunohistochemistry images of increased expression of endoglin in breast tumor samples following neoadjuvant chemotherapy from patients (magnification, ×200). Positive endoglin staining (white arrows) was observed as thin, linear deposits in the membrane and cytoplasm of endothelial cells within the microvessels.
Figure 3.
Correlation analysis of patient response and endoglin expression in breast tumor tissue samples prior to neoadjuvant chemotherapy. Correlation of endoglin expression and patient response for (A) primary tumors and (B) primary tumors with regional lymph node involvement. CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease.
Increased mTOR expression is linked to improved NAC response
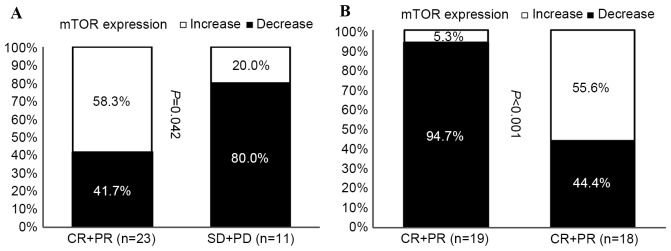
The expression of mTOR in tumors removed prior to NAC and in residual tumors following NAC were compared in primary tumors (Fig. 4A), and in primary tumors with regional lymph node involvement (Fig. 4B). The expression of mTOR was increased in tumors with improved responses to NAC treatment, while decreased mTOR expression was observed in tumors that progressed following NAC treatment (Fig. 4A).
Figure 4.
Correlation analysis of patient response and the altered expression levels of mTOR in breast tumor tissue samples prior to and following neoadjuvant chemotherapy. Correlation of mTOR expression and patient response for (A) primary tumors and (B) primary tumors with regional lymph node involvement. CR, complete response; PR, partial response; SD, stable disease; PD, progressive disease; mTOR, mechanistic target of rapamycin
Discussion
BC is currently one of the most common types of cancer among females in Taiwan (19). Although the first step to determine treatment for BC depends on the Tumor Node Metastasis stage (20), there are a variety of biomarkers that have been reported for prognostic and predictive purposes. The most common biomarkers used are hormone receptors, HER-2 expression status, and the Ki-67 index, which classifies BC types into luminal A, luminal B, HER-2-enriched and basal triple-negative breast cancers (21). These classifications are associated with different prognoses and treatment options. Identification of novel markers has led to a greater understanding of the importance of existing biomarkers, and a more definitive insight into tumor biology.
NAC is the current standard of treatment for patients with locally advanced BC, and is frequently used for patients with operable BC, with the aim of downstaging the tumor and improving the success rate of breast-conserving surgery (22). Response to NAC may predict a reduction in the micrometastatic burden, and allow for the individualization of systemic treatments. Traditionally, patients are administered with adjuvant chemotherapy following resection of primary tumors. However, determining the effectiveness of adjuvant chemotherapy is difficult, as there is no evaluable lesion following surgery. With NAC treatment, responses to drug activity are rapidly available, and valuable information may be collected from proof-of-concept studies that involve a relatively small number of patients.
It is the general consensus that the absence of residual invasive cancer in the breast and lymph nodes is the preferred definition of pCR that provides the best indicator of clinical outcome (23).
For breast tumors overexpressing HER-2, HER-2-targeted agents have been reported to improve the pCR rate and DFS. A novel anti-HER-2 antibody, known as pertuzumab, obtained FDA approval for the treatment of HER-2-positive BC due to the significant improvement in pCR when combined with trastuzumab as part of the NAC regimen (24). Patients with triple-negative breast cancer (TNBC) demonstrate a worse prognosis once pCR is not achieved; however, they display an increased probability of obtaining pCR when compared with non-TNBC patients. Once TNBC patients attain pCR following NAC treatment, they demonstrate an excellent survival rate (25).
Angiogenesis is important for cancer cells to proliferate, grow and metastasize. Inhibiting angiogenesis therefore leads to inhibition of these characteristics, which is may be detrimental to tumor growth and survival (8). Currently, there are numerous agents that target the neovascularization pathway; however, a predictive marker of response remains to be detected (26). Although numerous types of malignancies are hypervascular tumors, it is uncertain to what extent the angiogenesis signaling pathway is involved, as anti-angiogenetic agents only moderately prolong the overall survival rate of these cancers (27–29). The plasma levels of vascular growth factor-A (VEGFA) prior to treatment have been evaluated retrospectively in a previous study (30). Until recently, elevated levels of VEGFA have been demonstrated to be an indicator of poor prognosis; however, it is unable to predict response to anti-angiogenic therapies, including bevacizumab (31). MVD has become the pathological gold standard for assessing angiogenesis in solid tumors. Studies have demonstrated that the angiogenic potential of BC, as assessed by MVD, correlates with the potential of tumor progression and metastasis, and therefore may be used to predict clinical outcome (8,32).
Traditionally, pan endothelial markers, including CD31, CD34 and Von Willebrand factor are used to assess MVD histologically (33,34). However, endoglin is reportedly a more effective marker than CD34 and CD31 in the evaluation of neovascularization of tumors, as it demonstrates a greater affinity for endothelial cells in tumor tissues, whereas CD34 and CD31 react nonspecifically with pathological and healthy vessels (12).
A previously study demonstrated that MVD, as determined by endoglin staining, was correlated positively with HER-2 expression, and negatively with hormone receptor expression. The importance of MVD on overall survival is greater for early stage BCs (18). The present study revealed that MVD, as evaluated by endoglin staining, correlated with tumor response to NAC treatment. Therefore, endoglin may be a suitable predictor for patient response following NAC treatment.
Endoglin is an accessory receptor for TGF and is upregulated during hypoxia via induction of hypoxia-inducible factor 1α. Therefore, its expression is elevated in the actively proliferating endothelium (35,36). A clear positive correlation has been reported between several markers of cell proliferation, including cyclin-A and Ki-67, and endoglin expression levels. Therefore, endoglin has been suggested as an appropriate marker for tumor-associated neovascularization (36). Additionally, endoglin has been demonstrated to be a potential marker for tumor diagnosis and prognosis in a previous study (36).
As high endoglin expression correlates with the rapid proliferation of tumor cells, and as chemotherapy effective for inhibiting the growth of rapidly proliferating tumor cells (37), this may explain why high endoglin expression correlated with an improved response rate of primary tumors to NAC treatment in the present study. However, whether this response translates to improved DFS or overall survival remains to be elucidated.
The present study additionally demonstrated that mTOR expression levels were elevated in tumors responsive to NAC. Endocrine therapy inhibits the growth-promoting effects of estrogen via ERs, and may therefore be considered as the cornerstone of direct target therapy. Approximately 70–75% of BCs express ERs, indicating a high level of dependence on estrogen for tumor growth (38). Although endocrine therapy continues to be an effective treatment for ER-positive (ER+) BC, many patients with advanced ER + BC develop de novo or acquired resistance and require more intensive and toxic treatments, such as chemotherapy. Novel approaches which augment the benefit of existing endocrine therapies, by prolonging time to disease progression, preventing or overcoming resistance, and delaying the use of chemotherapy are required. The PI3K/Akt/mTOR signaling pathway is a key intracellular signaling system that induces cellular growth and survival. A previous study demonstrated that the hyper activation of this signaling pathway is implicated in the tumorigenesis of ER+ BC and resistance to endocrine therapy. Furthermore, a previous study reported that inhibition of the PI3K/AKT/mTOR signaling pathway may augment the benefit of endocrine therapy in ER+ BC (39). AspCR is considered to be the best response for cancer cells that survive following NAC, elevated mTOR expression may serve as an alternative signaling pathway for residual tumors. Further follow-up is required to assess the outcome of these patients.
The limitations of the present study included the use of insufficient samples for further analysis. This was due to the fact that patients and doctors in Taiwan remain hesitant to accept NAC, even though NAC is used in routine clinical practice in numerous Western countries. Furthermore, a number of patients achieved pCR; therefore, there were no residual tumors for the assessment of mTOR expression levels. Additionally, there was a limited duration of follow-up; therefore, it could not be confirmed whether response to NAC or the expression of endoglin prior to NAC correlated with improved DFS or overall survival.
In conclusion, the present study demonstrated that the expression of endoglin in BC tissue samples prior to NAC may be a useful predictor of treatment response. Long-term follow-up of clinical outcome is required to explain the elevation of mTOR expression levels following NAC treatment in responsive but non-pCR tumors.
Acknowledgements
The authors would like to thank Mr. Hsiao JK, MS (Kaohsiung, Taiwan), for his technical support, the Tissue Bank and Biobank Laboratory (grant no. CLRPG8B00330), Chang Gung Medical Foundation (grant no. CMRPG890112) and the Kaohsiung Chang Gung Memorial Hospital for providing the materials.
References
- 1.Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108. doi: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 2.Dai X, Xiang L, Li T, Bai Z. Cancer hallmarks, biomarkers and breast cancer molecular subtypes. J Cancer. 2016;7:1281–1294. doi: 10.7150/jca.13141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cianfrocca M, Gradishar W. New molecular classifications of breast cancer. CA Cancer J Clin. 2009;59:303–313. doi: 10.3322/caac.20029. [DOI] [PubMed] [Google Scholar]
- 4.Gralow JR, Burstein HJ, Wood W, Hortobagyi GN, Gianni L, von Minckwitz G, Buzdar AU, Smith IE, Symmans WF, Singh B, Winer EP. Preoperative therapy in invasive breast cancer: Pathologic assessment and systemic therapy issues in operable disease. J Clin Oncol. 2008;26:814–819. doi: 10.1200/JCO.2007.15.3510. [DOI] [PubMed] [Google Scholar]
- 5.von Minckwitz G, Untch M, Blohmer JU, Costa SD, Eidtmann H, Fasching PA, Gerber B, Eiermann W, Hilfrich J, Huober J, et al. Definition and impact of pathologic complete response on prognosis after neoadjuvant chemotherapy in various intrinsic subtypes. J Clin Oncol. 2012;30:1796–1804. doi: 10.1200/JCO.2011.38.8595. [DOI] [PubMed] [Google Scholar]
- 6.Cortazar P, Zhang L, Untch M, Mehta K, Costantino JP, Wolmark N, Bonnefoi H, Cameron D, Gianni L, Valagussa P, et al. Pathological complete response and long-term clinical benefit in breast cancer: The CTNeoBC pooled analysis. Lancet. 2014;384:164–172. doi: 10.1016/S0140-6736(13)62422-8. [DOI] [PubMed] [Google Scholar]
- 7.Prowell TM, Pazdur R. Pathological complete response and accelerated drug approval in early breast cancer. N Engl J Med. 2012;366:2438–2441. doi: 10.1056/NEJMp1205737. [DOI] [PubMed] [Google Scholar]
- 8.Folkman J, Watson K, Ingber D, Hanahan D. Induction of angiogenesis during the transition from hyperplasia to neoplasia. Nature. 1989;339:58–61. doi: 10.1038/339058a0. [DOI] [PubMed] [Google Scholar]
- 9.Gasparini G. Clinical significance of the determination of angiogenesis in human breast cancer: Update of the biological background and overview of the Vicenza studies. Eur J Cancer. 1996;32A:1–2493. doi: 10.1016/s0959-8049(96)00376-0. [DOI] [PubMed] [Google Scholar]
- 10.Uzzan B, Nicolas P, Cucherat M, Perret GY. Microvessel density as a prognostic factor in women with breast cancer: A systematic review of the literature and meta-analysis. Cancer Res. 2004;64:2941–2955. doi: 10.1158/0008-5472.CAN-03-1957. [DOI] [PubMed] [Google Scholar]
- 11.Li C, Hampson IN, Hampson L, Kumar P, Bernabeu C, Kumar S. CD105 antagonizes the inhibitory signaling of transforming growth factor beta1 on human vascular endothelial cells. FASEB J. 2000;14:55–64. doi: 10.1096/fasebj.14.1.55. [DOI] [PubMed] [Google Scholar]
- 12.Tanaka F, Otake Y, Yanagihara K, Kawano Y, Miyahara R, Li M, Yamada T, Hanaoka N, Inui K, Wada H. Evaluation of angiogenesis in non-small cell lung cancer: Comparison between anti-CD34 antibody and anti-CD105 antibody. Clin Cancer Res. 2001;7:3410–3415. [PubMed] [Google Scholar]
- 13.Schiff R, Massarweh SA, Shou J, Bharwani L, Mohsin SK, Osborne CK. Cross-talk between estrogen receptor and growth factor pathways as a molecular target for overcoming endocrine resistance. Clin Cancer Res. 2004;10(Suppl):331S–336S. doi: 10.1158/1078-0432.CCR-031212. [DOI] [PubMed] [Google Scholar]
- 14.Berns K, Horlings HM, Hennessy BT, Madiredjo M, Hijmans EM, Beelen K, Linn SC, Gonzalez-Angulo AM, Stemke-Hale K, Hauptmann M, et al. A functional genetic approach identifies the PI3K pathway as a major determinant of trastuzumab resistance in breast cancer. Cancer Cell. 2007;12:395–402. doi: 10.1016/j.ccr.2007.08.030. [DOI] [PubMed] [Google Scholar]
- 15.Baselga J, Campone M, Piccart M, Burris HA, III, Rugo HS, Sahmoud T, Noguchi S, Gnant M, Pritchard KI, Lebrun F, et al. Everolimus in postmenopausal hormone-receptor-positive advanced breast cancer. N Engl J Med. 2012;366:520–529. doi: 10.1056/NEJMoa1109653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Andre F, O'Regan R, Ozguroglu M, Toi M, Xu B, Jerusalem G, Masuda N, Wilks S, Arena F, Isaacs C, et al. Everolimus for women with trastuzumab-resistant, HER2-positive, advanced breast cancer (BOLERO-3): A randomised, double-blind, placebo-controlled phase 3 trial. Lancet Oncol. 2014;15:580–591. doi: 10.1016/S1470-2045(14)70138-X. [DOI] [PubMed] [Google Scholar]
- 17.Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1) Eur J Cancer. 2009;45:228–247. doi: 10.1016/j.ejca.2008.10.026. [DOI] [PubMed] [Google Scholar]
- 18.Rau KM, Huang CC, Chiu TJ, Chen YY, Lu CC, Liu CT, Pei SN, Wei YC. Neovascularization evaluated by CD105 correlates well with prognostic factors in breast cancers. Exp Ther Med. 2012;4:231–236. doi: 10.3892/etm.2012.594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Health Promotion Administration, Ministry of Health and Welfare, Taiwan, corp-author. Taiwan Cancer Registry annual report. Taiwan, Taipei: 2013. 2016. [Jun 14;2017 ]. [Google Scholar]
- 20.American Joint Committee on Cancer, corp-author. https://cancerstaging.org/references-tools/deskreferences/Pages/default.aspx. [Sep 9;2017 ]; [Google Scholar]
- 21.Sorlie T, Perou CM, Tibshirani R, Aas T, Geisler S, Johnsen H, Hastie T, Eisen MB, van de Rijn M, Jeffrey SS, et al. Gene expression patterns of breast carcinomas distinguish tumor subclasses with clinical implications; Proc Natl Acad Sci USA; 2001; pp. 10869–10874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Berruti A, Brizzi MP, Generali D, Ardine M, Dogliotti L, Bruzzi P, Bottini A. Presurgical systemic treatment of nonmetastatic breast cancer: Facts and open questions. Oncologist. 2008;13:1137–1148. doi: 10.1634/theoncologist.2008-0162. [DOI] [PubMed] [Google Scholar]
- 23.von Minckwitz G, Untch M, Blohmer JU, Costa SD, Eidtmann H, Fasching PA, Gerber B, Eiermann W, Hilfrich J, Huober J, et al. Definition and impact of pathologic complete response on prognosis after neoadjuvant chemotherapy in various intrinsic breast cancer subtypes. J Clin Oncol. 2012;30:1796–1804. doi: 10.1200/JCO.2011.38.8595. [DOI] [PubMed] [Google Scholar]
- 24.Gianni L, Pienkowski T, Im YH, Roman L, Tseng LM, Liu MC, Lluch A, Staroslawska E, de la Haba-Rodriguez J, Im SA, et al. Efficacy and safety of neoadjuvant pertuzumab and trastuzumab in women with locally advanced, inflammatory, or early HER2-positive breast cancer (NeoSphere): A randomised multicentre, open-label, phase 2 trial. Lancet Oncol. 2012;13:25–32. doi: 10.1016/S1470-2045(11)70336-9. [DOI] [PubMed] [Google Scholar]
- 25.Liedtke C, Mazouni C, Hess KR, André F, Tordai A, Mejia JA, Symmans WF, Gonzalez-Angulo AM, Hennessy B, Green M, et al. Response to neoadjuvant therapy and long-term survival in patients with triple-negative breast cancer. J Clin Oncol. 2008;26:1275–1281. doi: 10.1200/JCO.2007.14.4147. [DOI] [PubMed] [Google Scholar]
- 26.Schneider BP, Shen F, Miller KD. Pharmacogenetic biomarkers for the prediction of response to antiangiogenic treatment. Lancet Oncol. 2012;13:e427–e436. doi: 10.1016/S1470-2045(12)70275-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pang R, Poon RT. Angiogenesis and antiangiogenic therapy in hepatocellular carcinoma. Cancer Lett. 2006;242:151–167. doi: 10.1016/j.canlet.2006.01.008. [DOI] [PubMed] [Google Scholar]
- 28.Webber K, Cooper A, Kleiven H, Yip D, Goldstein D. Management of metastatic renal cell carcinoma in the era of targeted therapies. Intern Med J. 2011;41:594–605. doi: 10.1111/j.1445-5994.2011.02540.x. [DOI] [PubMed] [Google Scholar]
- 29.Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378–390. doi: 10.1056/NEJMoa0708857. [DOI] [PubMed] [Google Scholar]
- 30.Fakhrejahani E, Toi M. Antiangiogenesis therapy for breast cancer: An update and perspectives from clinical trials. Jpn J Clin Oncol. 2014;44:197–207. doi: 10.1093/jjco/hyt201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Burstein HJ, Chen YH, Parker LM, Savoie J, Younger J, Kuter I, Ryan PD, Garber JE, Chen H, Campos SM, et al. VEGF as a marker for outcome among advanced breast cancer patients receiving anti-VEGF therapy with bevacizumab and vinorelbine chemotherapy. Clin Cancer Res. 2008;14:7871–7877. doi: 10.1158/1078-0432.CCR-08-0593. [DOI] [PubMed] [Google Scholar]
- 32.Kraby MR, Krüger K, Opdahl S, Vatten LJ, Akslen LA, Boflin AM. Microvascular proliferation in luminal A and basal-like breast cancer subtypes. J Clin Pathol. 2015;68:891–897. doi: 10.1136/jclinpath-2015-203037. [DOI] [PubMed] [Google Scholar]
- 33.Fox SB, Gasparini G, Harris AL. Angiogenesis: Pathological, prognostic, and growth-factor pathways and their link to trial design and anticancer drugs. Lancet Oncol. 2001;2:278–289. doi: 10.1016/S1470-2045(00)00323-5. [DOI] [PubMed] [Google Scholar]
- 34.Mineo TC, Ambrogi V, Baldi A, Rabitti C, Bollero P, Vincenzi B, Tonini G. Prognostic impact of VEGF, CD31, CD34, and CD105 expression and tumour vessel invasion after radical surgery for IB-IIA non-small cell lung cancer. J Clin Pathol. 2004;57:591–537. doi: 10.1136/jcp.2003.013508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Miller DW, Graulich W, Karges B, Stahl S, Ernst M, Ramaswamy A, Sedlacek HH, Müller R, Adamkiewicz J. Elevated expression of endoglin, a component of the TGF-beta-receptor complex, correlates with proliferation of tumor endothelial cells. Int J Cancer. 1999;81:568–572. doi: 10.1002/(SICI)1097-0215(19990517)81:4<568::AID-IJC11>3.0.CO;2-X. [DOI] [PubMed] [Google Scholar]
- 36.Nassiri F, Cusimano MD, Scheithauer BW, Rotondo F, Fazio A, Yousef GM, Syro LV, Kovacs K, Lloyd RV. Endoglin (CD105): A review of its role in angiogenesis and tumor diagnosis, progression and therapy. Anticancer Res. 2011;31:2283–2290. [PubMed] [Google Scholar]
- 37.Klijn JG, Berns EM, Foekens JA. Prognostic factors and response to therapy in breast cancer. Cancer Surv. 1993;18:165–198. [PubMed] [Google Scholar]
- 38.Systemic treatment of early breast cancer by hormonal, cytotoxic, or immune therapy. 133 randomised trials involving 31,000 recurrences and 24,000 deaths among 75,000 women. Early Breast Cancer Trialists' Collaborative Group. Lancet. 1992;339:71–85. [PubMed] [Google Scholar]
- 39.Ciruelos Gil EM. Targeting the PI3K/AKT/mTOR pathway in estrogen receptor-positive breast cancer. Cancer Treat Rev. 2014;40:862–871. doi: 10.1016/j.ctrv.2014.03.004. [DOI] [PubMed] [Google Scholar]