Abstract
Cellular neurothekeomas (CNs) are distinctive benign tumors of uncertain histogenesis, with predilection for the skin of the head and neck region. We describe the first case of multiple desmoplastic CNs (DCNs) affecting the oral cavity in a 9-year-old girl. Histopathologic evaluation showed a proliferation of spindle and epithelioid cells, forming nests and bundles, supported by exuberant fibrous stroma, as well as scattered multinucleated floret-like giant cells. The tumor cells were immunopositive for vimentin, CD63, CD56, whereas AE1/AE3, S100, CD34, α-SMA, GFAP, EMA, CD57 and NSE were negative. Ki-67 was <2%. Multiple DCNs should be considered in the differential diagnosis of oral nodular lesions.
Keywords: Oral cavity, Cellular neurothekeoma, Desmoplastic cellular neurothekeoma, Immunohistochemistry, Child
Introduction
Cellular neurothekeoma (CN) is an uncommon benign tumor of uncertain histogenesis, being more frequently observed in subcutaneous tissue of the upper limbs, head, neck and trunk. Clinically, CN presents as a solitary non-ulcerated nodule or papule, measuring <2.0 cm in diameter, usually affecting young adults with a slight female predominance [1]. Histologically, this neoplasm is poorly circumscribed occupying the thickness of the dermis, but sometimes it can extend to the subcutis. At higher magnifications, it is noticed the presence of nests and cords of epithelioid to spindle cells with slightly granular eosinophilic cytoplasm surrounding by dense fibrotic stroma. Uncommonly, areas exhibiting plexiform growth pattern or focal cellular atypia can also be visualized [1, 2]. CN behaves in a benign fashion and rarely recur (remarkably, in some incompletely excised facial lesions). Large tumor size (>2 cm) and histologic features such as mitotic figures, cellular pleomorphism and infiltration of fat or skeletal muscle, seem to have no clinical significance [1].
Oral CN is extremely rare [3]. To the best or our knowledge, only 15 oral cases diagnosed as CN and/or nerve sheath myxoma (NSM) have been reported in the English-language literature (Table 1). In this concern, according to relevant studies, the main differential diagnosis for CN is NSM. Unlike NSM, CN rarely occurs on the hands and fingers. Microscopically, CNs show irregular, infiltrative margins, while NSM exhibits lobules well demarcated. NSM is consistently positive for S100 and negative for NKI-C3, whereas CN shows an opposite immunostaining pattern [1, 2, 4].
Table 1.
Cases of cellular neurothekeoma affecting the oral mucosa in children and adults
| Author | Age (years)/Gender | Site | Size (cm) | Duration before excision |
Clinical diagnosis | Original diagnosis |
Diagnosis after reclassificationa |
Growth rate/Follow-up |
|---|---|---|---|---|---|---|---|---|
| Breuer et al. [24] | 12/F | Tongue | 2 | Birth/12 years | NI | NTK | Probable CNc | Rapid in the interval between two operations |
| Penarrocha et al. [25] | Newborn/F | Tongue | 3 × 2.5 | At birth | NI | NSM (NTK) | Probable CNc | NI |
| Barret and Suhr [16] | 29/M | Buccal vestibular sulcus | 1 | NI | Scar tissue | CN | CN | NI |
| Kim et al. [26] | 15/F | Tongue | 2.1 | 7 months | NI | NTK | Probable CNc | Slow |
| Nishioka et al. [17] | 53/M | Buccal mucosa | 4.0 × 2.0 | NI | Benign tumor | NSM (NTK) | NSMb | NI |
| 2/F | Buccal region | 0.7 × 0.8 | 9 months | Fibro-epithelial polyp | NSM (NTK) | NSM | NI | |
| 52/F | Lower lip | 0.7 × 0.7 | 3 years | Benign tumor | NSM (NTK) | NSM | NI | |
| Plaza et al. [27] | 64/F | Mouth | NI | NI | NI | CN | CN | NI |
| 36/F | Cheek | NI | NI | CN | CN | NI | ||
| Safadi et al. [28] | 32/F | Gingiva | 0.8 × 0.6 | NI | NI | NSM (NTK) | Probable CNc | NI |
| Vered et al. [3] | 12/M | Buccal gingiva | 1 | 1 year | Fibroma, giant cell lesion | Classic NTK | NSM | NI |
| 25/M | Buccal gingiva | 0.3 | >1 year | Fibroma | CN | CN | NI | |
| 31/F | Maxillary vestibule | 0.7 | 4 months | Mucocele | Classic NTK | NSM | NI | |
| 35/F | Palatal gingiva | 0.6 | 20 years | Fibroma | Classic NTK | NSM | NI | |
| Pan et al. [29] | 6 months/ M | Upper lip | 1 | NI | Epidermoid cyst | CN | Probable CNc | NI |
| Emami et al. [30] | 15/F | Floor of the mouth | 0.8 | NI | NI | CN | CN | No recurrence after 8 months |
| Ishikawa et al. [31] | 35/F | Upper Lip | 0.35 | 4 years | NI | CN | Probable CNc | NI |
| Present case | 9/F | Buccal mucosa/upper lip (internal mucosa) | 1.5 × 1.5 | 3 months | Neurofibroma Schwannoma Myofibroma |
CN | CN | No recurrence after 3 years |
CN cellular neurothekeoma, F female, M male, NTK neurothekeoma, NSM nerve sheath myxoma, NI not informed
aDiagnosis according to Fetsch et al. [2], Fetsch et al. [4] and Hornick & Fletcher [1] studies
bThis case showed histopathological features mimicking spindle cell lipoma
cProbable CN: incomplete immunohistochemical panel
Clinically, oral CN manifests itself as a nodular lesion, firm consistency, covered by normal-appearing oral mucosa, asymptomatic or mildly painful [3]. The clinical differential diagnosis includes benign mesenchymal tumors like neurofibroma, schwannoma, leiomyoma, myofibroma and, eventually, fibrous hyperplasia [1, 3].
Interestingly, several CN cases containing marked stromal hyalinization have been reported, being referred as desmoplastic CN (DCN) [1, 5–8]. Moreover, multiple CNs in the head and neck region affecting a 30-year-old male [9], and multiple DCNs localized to the face of a 16-year-old boy [10], have been reported. To the best of our knowledge, to date, there is no published case showing oral cavity involvement by DCN (Table 2).
Table 2.
Cases of desmoplastic cellular neurothekeoma (DCN) collected in Medline database
| Author | Age (years)/gender | Site | Size (cm) | Duration | Clinical diagnosis | Follow-up without recurrence (months) |
|---|---|---|---|---|---|---|
| D’ Antonio et al. [7] | 44/M | Thigh | NI | NI | NI | 3 |
| Garcia- Gutierrez et al. [10] | 16/M | Face | 2–3 | 2 years | Angiofibromas, adnexal neoplasms, xanthogranulomas | NI |
| Weng et al. [8]a | 29/F | Nose | 0.5 | 2 months | Basal cell carcinoma, epidermoid cyst, follicular neoplasm, adnexal tumor, dermal melanocytic nevus | NI |
| Zedek et al. [5] | 26/F | Ulnar wrist | 0.8 | NI | Papule | 22 |
| 11/M | Posterior scalp | NI | NI | Painful nodule | 42 | |
| 43/F | Postauricular region | NI | NI | Gray nodule | 42 | |
| 41/F | Eyebrow | NI | NI | Calcified cyst, pilomatrixoma | 31 | |
| 3/F | Sternum | NI | NI | NI | NI | |
| 16/F | Buttock | NI | NI | NI | Lost to follow-up | |
| 44/M | Second finger | 0.5 | 3 months | Papule | 11 | |
| 55/F | Breast | NI | NI | Fibrous histiocytoma | 24 | |
| 14/M | Thigh | NI | NI | Plaque with central papule | 22 | |
| 7/F | Arm | 1.25 | NI | Nodule, pilomatrixoma | 13 | |
| 52/F | Arm | NI | NI | Keloyd | 18 | |
| 48/F | Thigh | NI | NI | Cyst | 14 | |
| Present case | 9/F | Buccal mucosa/upper lip (internal mucosa) | 1.5 | 3 months | Neurofibroma Schwannoma Myofibroma |
36 |
We present herein the first case of a 9-year-old girl with multiple DCNs affecting the buccal mucosa and upper lip which appeared over a period of 3 months.
Case Report
A 9-year-old girl presented to a Stomatology Service (UFVJM), Diamantina, Brazil, accompanied by his father, with multiple, slightly symptomatic, oral nodules affecting the buccal mucosa, which arose over the course of 3 months. During anamnesis, the child reported exacerbation of symptoms during palpation. Moreover, she does not report trauma or previous surgery in the lesional area.
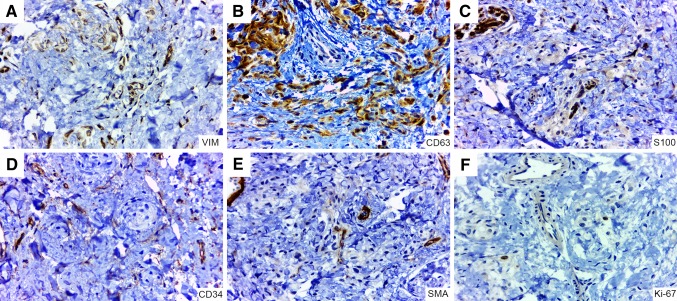
The clinical examination revealed the presence of two submucosal nodular lesions, mobile, painful on palpation, measuring approximately 1.5 × 1.5 cm in diameter, as well as other small, ill-defined lesions, located on the left side of buccal mucosa and upper lip (Fig. 1). The clinical differential diagnoses were neurofibroma, schwannoma or myofibroma. After incisional biopsy of the lesion, the microscopical analysis revealed a diffuse proliferation of spindle cells supported by exuberant connective tissue stroma, permeating bundles of skeletal muscle and adipose tissue in the deeper part. Moreover, a vague micronodular architecture was evidenced. In high-power view, the lesion was composed of epithelioid to spindled cells with palely granular eosinophilic cytoplasm forming nests and bundles, separated by desmoplastic stroma. Moreover, scattered multinucleated floret-like giant cells were visualized (Fig. 2a–c). The cytoarchitectural organization and desmoplastic stroma were highlighted by Masson’s Trichrome (Fig. 2d). By immunohistochemical analysis, the tumor cells were positive for vimentin, CD63 (NKI-C3), focally for CD56, whereas AE1/AE3, S100, CD34, α-SMA, GFAP, EMA, CD57 and NSE were negative. Ki-67 labelling index was <2% (Fig. 3).
Fig. 1.
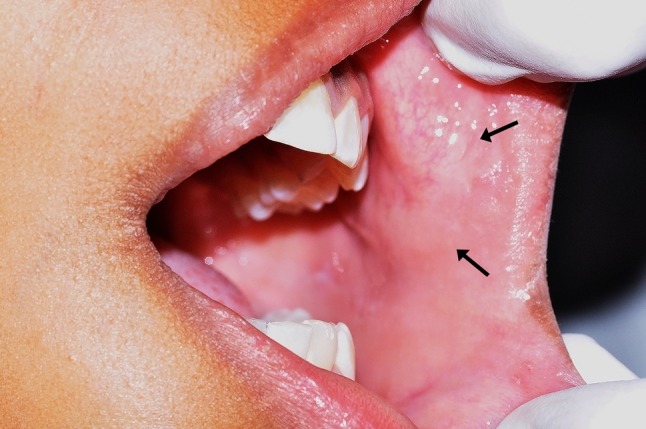
Clinical features of the desmoplastic cellular neurothekeoma affecting the buccal mucosa and upper lip of the left side in a child patient
Fig. 2.
a Diffuse proliferation of spindle cells supported by exuberant connective tissue stroma (H&E, ×10). Notice sparse multinucleated floret-like giant cells (inset, ×40). b The tumor cells permeate bundles of skeletal muscle and adipose tissue (H&E ×10). c In high-power view, visualize epithelioid cells with granular eosinophilic cytoplasm forming nests adjacent to neurovascular bundle (H&E, ×40). d The cytoarchitectural organization and desmoplastic stroma are highlighted by Masson’s Trichrome (×40)
Fig. 3.
Immunohistochemical analysis of desmoplastic cellular neurothekeoma. a The tumor cells were positive for vimentin and, b CD63 (NKI-C3). c S100 (nerves serve as a positive internal control), d CD34 (endothelial cells serve as a positive internal control) and e α-SMA (smooth muscle cells serve as a positive internal control), were negative. f The Ki-67 labelling index was <2% (IHQ, ×40). Notice the cytoarchitectural organization of the tumor cells (nests and bundles) highlighted in all photomicrographs
Given the morphological and immunohistochemical findings, was established the diagnosis of oral DCN. Since the patient has other lesional areas, surgical removal of them all, was performed. The material was sent for histopathological analysis that newly confirmed the diagnosis of DCN. After 3 years of follow-up, the patient is well, without recurrence or alteration.
Discussion
The term neurothekeoma was introduced for the first time by Gallager and Helwig in 1980 [11] to describe a superficial skin neoplasm, of probably nerve sheath derivation, predominantly observed in children and adolescents, whereas the term CN was firstly used by Rosati et al. in 1986 [12].
Initially, through a histomorphological analysis, neurothekeoma was classified as cellular, mixed or myxoid, depending on the amount of myxoid matrix [4, 13]. Thus, because of myxoid stroma, some authors initially suggested that neurothekeoma and NSM represented lesions within a morphologic spectrum [13], whereas others questioned this relationship [14, 15]. Actually, there is consensus that NSM is a true nerve sheath tumor regarding their immunohistochemical profile and ultrastructural findings, which is consistent with Schwannian differentiation [2], whereas the nosologic status of neurothekeoma continues to be debated. Thus, on the basis of the results of a large series of NSMs [2] and neurothekeomas [1, 4], it seems that these tumors are unrelated and that the so-called mixed or myxoid neurothekeoma can in fact represent CN with myxoid stroma [1, 2, 4].
A review of the literature was conducted on the information collected from the Medline database (http://www.ncbi.nim.nih.gov) up to December 2016, with cross-referencing using the terms “cellular neurothekeoma and oral mucosa”; “nerve sheath myxoma and oral mucosa”. The search resulted in a total of 11 articles [3, 16–24], which reported a total de 17 cases diagnosed as neurothekeoma, CN, NSM/Neurothekeoma and classic neurothekeoma of the oral cavity [3, 16, 17, 24–31] (Table 1). However, after careful examination by using strict criteria [1, 2, 4], only five CNs were identified; of them, a single case in a pediatric patient [30]. Thus, the current case appears to be the second pediatric case of CN affecting the oral mucosa.
Moreover, a review of the DCN cases was also performed. The information was collected from the Medline database (http://www.ncbi.nim.nih.gov) up to December 2016, with cross-referencing using the terms “desmoplastic neurothekeoma”, “desmoplastic cellular neurothekeoma” and “desmoplastic nerve sheath myxoma”. The search results in a total of 15 cases of DCN in cutaneous localization [5, 7, 8, 10], of which six cases affected pediatric patients, being found a single case with involvement of the face [10]. Thus, the current case appears to be the first report of DCN with involvement of the oral cavity. Furthermore, as we detected clinically two nodular masses admixed with other small, ill-defined lesions on the left buccal mucosa and upper lip, which by histopathological and immunohistochemical analysis proved to be DCN, the final diagnosis was multiple DCNs of the oral cavity. In fact, similar to clinicopathological features of the current case, multiple CNs in the head and neck region affecting a 30-year-old male [9] and multiple DCNs localized to the face of a 16-year-old boy [10] have been reported.
As previously mentioned, the main differential diagnosis for NSM is CN. NSM is a peripheral nerve sheath tumor, with a peak incidence in the fourth decade of life, without gender preference. Notably, NSM have a strong predilection for the extremities, particularly the fingers. Approximately 15% of NSMs show involvement of the trunk or head and neck regions. Microscopically, NSMs exhibit delimited myxoid, multinodular masses, containing spindled and epithelioid Schwann cells, which are S-100, GFAP, NSE and CD57 positive. When treated by simple local excision, NSM shows a relatively high local recurrence rate. Thus, complete local excision with a margin of normal tissue is recommended [2].
The histopathologic differential diagnosis of CN includes plexiform fibrohistiocytic tumor, neurofibroma, schwannoma, Spitz nevus, leiomyoma, benign fibrohistiocytoma and, eventually, melanoma. The DCN should be differentiated from desmoplastic fibroblastoma, desmoplastic nevi and sclerosing perineurioma, including desmoplastic melanoma and desmoplastic squamous cell carcinoma. All these lesions were excluded through of a detailed clinical, histopathological and immunohistochemical evaluation [10, 32].
Interestingly, giant cells (osteoclastic, Touton and/or tumor) were seen in 20 (15%) CN cases [1]. However, differently, in the current case we have observed scattered multinucleated floret-like giant cells (Fig. 1, inset). To the best of our knowledge, this is the first report showing this giant cell type in CN/DCN, which is similar to those observed in giant cell fibroblastoma, pleomorphic lipoma, multinucleate cell angiohistiocytoma, giant cell collagenoma, pleomorphic fibroma, and remarkably, neurofibroma. Moreover, multinucleated floret-like giant cells have been documented in normal tissues such as lower female genital tract, testis, urinary bladder, anus, breast and skin. The histogenesis of multinucleated floret-like giant cells is unknown; however, some studies indicate that native dermal or interstitial CD34+ fibroblasts/dendritic cells can acquire a reactive multinucleated and floret-like appearance in response to unknown stimuli and/or due to interactions with mast cells [33, 34].
Although DCN is a benign tumor, and knowing that it can present microscopically irregular and infiltrative margins, is recommended the complete surgical excision of the lesion, especially in facial lesions, since it occasionally recurs, many times within a short period following removal [17, 24]. In this way, the current patient is being kept under observation as recommended, and after 3 years of follow-up is well without showing recurrence or alteration.
The current case appears to be the first report showing oral cavity involvement by DCN, which affected a pediatric patient. Microscopically, DCN shows irregular and infiltrative margins and can be misinterpreted as an aggressive lesion, which may result in inadequate treatment. Complete surgical excision of the DCN lesions is recommended, since it can recur, many times within a short period following removal of the lesion. Multiple DCNs, such as shown in the current case, should be considered in the differential diagnosis of oral nodular lesions in pediatric patients.
Acknowledgements
The authors would like to thank the parents and child who agreed to participate in this study.
References
- 1.Hornick JL, Fletcher CD. Cellular neurothekeoma: detailed characterization in a series of 133 cases. Am J Surg Pathol. 2007;31(3):329–340. doi: 10.1097/01.pas.0000213360.03133.89. [DOI] [PubMed] [Google Scholar]
- 2.Fetsch JF, Laskin WB, Miettinen M. Nerve sheath myxoma: a clinicopathologic and immunohistochemical analysis of 57 morphologically distinctive, S-100 protein- and GFAP-positive, myxoid peripheral nerve sheath tumors with a predilection for the extremities and a high local recurrence rate. Am J Surg Pathol. 2005;29(12):1615–1624. doi: 10.1097/01.pas.0000173025.87476.a4. [DOI] [PubMed] [Google Scholar]
- 3.Vered M, Fridman E, Carpenter WM, Buchner A. Classic neurothekeoma (nerve sheath myxoma) and cellular neurothekeoma of the oral mucosa: immunohistochemical profiles. J Oral Pathol Med. 2011;40(2):174–180. doi: 10.1111/j.1600-0714.2010.00952.x. [DOI] [PubMed] [Google Scholar]
- 4.Fetsch JF, Laskin WB, Hallman JR, Lupton GP, Miettinen M. Neurothekeoma: an analysis of 178 tumors with detailed immunohistochemical data and long-term patient follow-up information. Am J Surg Pathol. 2007;31(7):1103–1114. doi: 10.1097/PAS.0b013e31802d96af. [DOI] [PubMed] [Google Scholar]
- 5.Zedek DC, White WL, McCalmont TH. Desmoplastic cellular neurothekeoma: clinicopathological analysis of twelve cases. J Cutan Pathol. 2009;36(11):1185–1190. doi: 10.1111/j.1600-0560.2009.01263.x. [DOI] [PubMed] [Google Scholar]
- 6.Barnhill RL, Mihm MC., Jr Cellular neurothekeoma. A distinctive variant of neurothekeoma mimicking nevomelanocytic tumors. Am J Surg Pathol. 1990;14(2):113–120. doi: 10.1097/00000478-199002000-00002. [DOI] [PubMed] [Google Scholar]
- 7.D’Antonio A, Cuomo R, Angrisani B, Memoli D, Angrisani P. Desmoplastic cellular neurothekeoma mimicking a desmoplastic melanocytic tumor. Am Acad Dermatol. 2011;65(2):e57–e58. doi: 10.1016/j.jaad.2010.12.029. [DOI] [PubMed] [Google Scholar]
- 8.Weng YC, Shen JL, Yang CS, Li WH. Desmoplastic cellular neurothekeoma mimicking basalcell carcinoma. Dtsch Dermatol Ges. 2015;13(3):243–245. doi: 10.1111/ddg.12523. [DOI] [PubMed] [Google Scholar]
- 9.Mahalingam M, Alter JN, Bhawan J. Multiple cellular neurothekeomas—a case report and review on the role of immunohistochemistry as a histologic adjunct. J Cutan Pathol. 2006;33(11):51–56. doi: 10.1111/j.0303-6987.2006.00400.x. [DOI] [PubMed] [Google Scholar]
- 10.Garcia-Gutierrez M, Toussaint-Caire S, Gonzalez-Sanchez P, Ortiz-Hidalgo C. Multiple desmoplastic cellular neurothekeomas localized to the face of a 16-year-old boy. Am J Dermatopathol. 2010;32(8):509–513. doi: 10.1097/DAD.0b013e3181c98f89. [DOI] [PubMed] [Google Scholar]
- 11.Gallager RL, Helwig EB. Neurothekeoma—a benign cutaneous tumor of neural origin. Am J Clin Pathol. 1980;74(6):759–764. doi: 10.1093/ajcp/74.6.759. [DOI] [PubMed] [Google Scholar]
- 12.Rosati LA, Fratamico FC, Eusebi V. Cellular neurothekeoma. Appl Pathol. 1986;4(3):186–191. [PubMed] [Google Scholar]
- 13.Husain S, Silvers DN, Halperin AJ, McNutt NS. Histologic spectrum of neurothekeoma and the value of immunoperoxidase staining for S-100 protein in distinguishing it from melanoma. Am J Dermatopathol. 1994;16(5):496–503. doi: 10.1097/00000372-199410000-00004. [DOI] [PubMed] [Google Scholar]
- 14.Barnhill RL, Dickersin GR, Nickeleit V, Bhan AK, Muhlbauer JE, Phillips ME, et al. Studies on the cellular origin of neurothekeoma: clinical, light microscopic, immunohistochemical, and ultrastructural observations. Am Acad Dermatol. 1991;25(1 ):80–88. doi: 10.1016/0190-9622(91)70177-4. [DOI] [PubMed] [Google Scholar]
- 15.Laskin WB, Fetsch JF, Miettinen M. The “neurothekeoma”: immunohistochemical analysis distinguishes the true nerve sheath myxoma from its mimics. Hum Pathol. 2000;31(10):1230–1241. doi: 10.1053/hupa.2000.18474. [DOI] [PubMed] [Google Scholar]
- 16.Barrett AW, Suhr M. Cellular neurothekeoma of the oral mucosa. Oral Oncol. 2001;37(8):660–664. doi: 10.1016/S1368-8375(00)00117-2. [DOI] [PubMed] [Google Scholar]
- 17.Nishioka M, Aguirre RL, Ishikawa A, Nagumo K, Wang LH, Okada N. Nerve sheath myxoma (neurothekeoma) arising in the oral cavity: histological and immunohistochemical features of 3 cases. Oral Surg Oral Med Oral Pathol Oral Radiol Endodontology. 2009;107(5):e28–e33. doi: 10.1016/j.tripleo.2009.01.018. [DOI] [PubMed] [Google Scholar]
- 18.Prado JD, Andrade RG, Silva-Sousa YT, Andrade MF, Soares FA, Perez DE. Nerve sheath myxoma of the gingiva: report of a rare case and review of the literature. J Periodontol. 2007;78(8):1639–1643. doi: 10.1902/jop.2007.060425. [DOI] [PubMed] [Google Scholar]
- 19.Rodriguez-Peralto JL, el-Naggar AK. Neurothekeoma of the oral cavity: case report and review of the literature. J Oral Maxillofac Surg. 1992;50(11):1224–1226. doi: 10.1016/0278-2391(92)90159-W. [DOI] [PubMed] [Google Scholar]
- 20.Rozza-de-Menezes RE, Andrade RM, Israel MS, Goncalves Cunha KS. Intraoral nerve sheath myxoma: case report and systematic review of the literature. Head Neck. 2013;35(12):E397–E404. doi: 10.1002/hed.23361. [DOI] [PubMed] [Google Scholar]
- 21.Sist TC, Jr, Greene GW., Jr Benign nerve sheath myxoma: light and electron microscopic features of two cases. Oral Surg Oral Med Oral Pathol. 1979;47(5):441–444. doi: 10.1016/0030-4220(79)90126-9. [DOI] [PubMed] [Google Scholar]
- 22.Tiffee JC, Pulitzer DR. Nerve sheath myxoma of the oral cavity: case report and review. Oral Surg Oral Med Oral Pathol Oral Radiol Endodontology. 1996;82(4):423–425. doi: 10.1016/S1079-2104(96)80308-8. [DOI] [PubMed] [Google Scholar]
- 23.Spadari F, Guzzi G, Bombeccari GP, Mariani U, Gianatti A, Ruffoni D, et al. Nerve sheath myxoma of the tongue. Acta Dermatovenerol Croat. 2014;22(1):52–56. [PubMed] [Google Scholar]
- 24.Breuer T, Koester M, Weidenbecher M, Steininger H. Neurothekeoma, a rare tumour of the tongue. J Otorhinolaryngol Relat Spec. 1999;61(3):161–164. doi: 10.1159/000027663. [DOI] [PubMed] [Google Scholar]
- 25.Penarrocha M, Bonet J, Minguez JM, Vera F. Nerve sheath myxoma (neurothekeoma) in the tongue of a newborn. Oral Surg Oral Med Oral Pathol Oral Radiol Endodontology. 2000;90(1):74–77. doi: 10.1067/moe.2000.106335. [DOI] [PubMed] [Google Scholar]
- 26.Kim HJ, Baek CH, Ko YH, Choi JY. Neurothekeoma of the tongue: CT, MR, and FDG PET imaging findings. Am J Neuroradiol. 2006;27(9):1823–1825. [PMC free article] [PubMed] [Google Scholar]
- 27.Plaza JA, Torres-Cabala C, Evans H, Diwan AH, Prieto VG. Immunohistochemical expression of S100A6 in cellular neurothekeoma: clinicopathologic and immunohistochemical analysis of 31 cases. Am J Dermatopathol. 2009;31(5):419–422. doi: 10.1097/DAD.0b013e3181a13afc. [DOI] [PubMed] [Google Scholar]
- 28.Safadi RA, Hellstein JW, Diab MM, Hammad HM. Nerve sheath myxoma (neurothekeoma) of the gingiva, a case report and review of the literature. Head Neck Pathol. 2010;4(3):242–245. doi: 10.1007/s12105-010-0183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Pan HY, Tseng SH, Weng CC, Chen Y. Cellular neurothekeoma of the upper lip in an infant. Pediatr Neonatol. 2014;55(1):71–74. doi: 10.1016/j.pedneo.2012.12.004. [DOI] [PubMed] [Google Scholar]
- 30.Emami N, Zawawi F, Ywakim R, Nahal A, Daniel SJ. Oral cellular neurothekeoma. Case Rep Otolaryngol. 2013;2013:935435. doi: 10.1155/2013/935435. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ishikawa M, Yamamoto T, Izumi M. Cellular Neurothekeoma on the Lip. Indian J Dermatol. 2016;61(1):122. doi: 10.4103/0019-5154.174138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Gonzalez-Arriagada WA, Leon JE, Vargas PA, Paes de Almeida O, Lopes MA. Intraoral sclerosing perineurioma: a case report and review of the literature. Oral Surg Oral Med Oral Pathol Oral Radiol Endodontology. 2010;109(5):e46–e52. doi: 10.1016/j.tripleo.2010.01.021. [DOI] [PubMed] [Google Scholar]
- 33.Magro G, Amico P, Vecchio GM, Caltabiano R, Castaing M, Kacerovska D, et al. Multinucleated floret-like giant cells in sporadic and NF1-associated neurofibromas: a clinicopathologic study of 94 cases. Virchows Arch. 2010;456(1):71–76. doi: 10.1007/s00428-009-0859-y. [DOI] [PubMed] [Google Scholar]
- 34.de Andrade CR, Lopes MA, de Almeida OP, Leon JE, Mistro F, Kignel S. Giant cell angiofibroma of the oral cavity: a case report and review of the literature. Med Oral Patol Oral Cir Bucal. 2008;13(9):E540–E543. [PubMed] [Google Scholar]