Abstract
Community-acquired pneumonia represents the most frequent bacterial infection in patients with HIV/AIDS.
Purpose
We aimed to assess variables associated with lower respiratory tract infection (LRTI) among HIV-infected adults using ART.
Methods
A cohort study of HIV-infected patients aged ≥18 years, enrolled from 2000 to 2015, on ART for at least 60 days, with primary outcome as the 1st episode of LRTI during follow-up. The independent variables included were sex at birth, age, race/skin color, educational level, tobacco smoking, alcohol use, cocaine use, diabetes mellitus, CD4 count, HIV viral load, influenza and pneumococcal vaccination. Extended Cox proportional hazards models accounting for time-updated variables were fitted to assess LRTI predictors.
Results
2669 patients were included; median follow-up was 3.9 years per patient. LRTI was diagnosed in 384 patients; incidence rate was 30.7/1000 PY. In the unadjusted Cox extended models, non-white race [crude hazard ratio (cHR) 1.28, p = 0.020], cocaine use (cHR 2.01, p < 0.001), tobacco smoking (cHR 1.34, p value 0.007), and HIV viral load ≥400 copies/mL (cHR 3.40, p < 0.001) increased the risk of LRTI. Lower risk of LRTI was seen with higher educational level (cHR 0.61, p < 0.001), rise in CD4 counts (cHR 0.81, p < 0.001, per 100 cells/mm3 increase), influenza (cHR 0.60, p = 0.002) and pneumococcal vaccination (cHR 0.57, p < 0.001). In the adjusted model, aHR for CD4 count was 0.86, for cocaine use 1.47 and for viral load ≥400 copies 2.20.
Conclusions
LRTI has a high incidence in HIV-infected adults using ART. Higher CD4 counts and undetectable viral loads were protective, as were pneumococcal and influenza vaccines.
Keywords: Community-acquired pneumonia, HIV, AIDS, Antiretroviral therapy, Vaccination
Introduction
Pneumonia is the fourth most common cause of death globally, after ischaemic heart disease, stroke and chronic obstructive pulmonary disease (COPD), and is the second most frequent reason for years of life lost [1]. Risk factors for community-acquired pneumonia (CAP) in adults include age greater than 65 years, chronic respiratory disease, HIV infection, smoking, alcohol abuse, poor dental condition and other comorbidities such as chronic heart failure, cerebrovascular disease (including dementia), and chronic liver or kidney disease [2–4].
Among HIV infected individuals, antiretroviral therapy (ART) has significantly impacted on the incidence of opportunistic infections and bacterial diseases both in high and middle-low income countries [5–7]. However, bacterial infections are still the main cause of hospitalization and a major cause of death in this population [8–11].
CAP represents the most frequent bacterial infection in patients with HIV/AIDS [8, 12–14], and although its incidence has been reduced after ART scale-up, it remains frequent even in patients on ART and with high CD4 counts [8, 14]. An observational study that included 9101 hospitalized HIV-infected patients in the United States (from 2004 to 2008) found that non-recurrent bacterial pneumonia was the second most common cause of death accounting for 20% of the deaths, behind sepsis which accounted for 38% of the observed deaths [13]. Similarly, in Mexico (during the years 2010–2011), in a study conducted with patients hospitalized due to a respiratory condition, CAP represented 18.6% of all hospitalizations and 50% of deaths in HIV infected patients [15].
Besides ART, pneumococcal and influenza vaccinations are indicated as adjuvant preventive strategies to reduce the burden of CAP among HIV infected individuals [14]. The effectiveness of pneumococcal and influenza vaccinations on preventing CAP have been shown among the elderly population, and among other specific high risk population.
HIV infection is independently associated with increased risk of CAP [12, 14, 16]. Despite its high incidence and mortality, few studies have evaluated factors associated with CAP in HIV-infected individuals.
The aim of our study was to assess variables contemporarily associated with lower respiratory tract infection (LRTI)among HIV-infected adults using antiretroviral therapy.
Methods
Study site and population
Instituto Nacional de Infectologia Evandro Chagas (INI, formerly known as Instituto de Pesquisa Clínica Evandro Chagas/IPEC) is a referral center for research care of HIV-infected patients, in Rio de Janeiro, Brazil, since 1986. A longitudinal database including sociodemographic, clinical, laboratory and therapeutic information abstracted from medical charts is maintained and periodically updated for all HIV infected patients followed at INI. Cohort procedures and results have been described elsewhere [7]. Patients included in the cohort follow Brazilian guidelines for HIV treatment which includes the recommendation of vaccination with pneumococcal polysaccharide vaccine (PPSV23) (with a second dose after 5 years) and annual influenza vaccination for all asymptomatic HIV infected patients with CD4 counts above 200 cells/mm3 [17].
For this study, we included HIV-infected patients aged ≥18 years at cohort entry, enrolled from January 2000 to October 2015, with no previous report of antiretroviral therapy (ART) use, who were followed for a period of at least 60 days (definition of active care) and who started ART after cohort enrollment. The primary outcome was the occurrence of the first episode of LRTI during follow-up. Since we aimed to study predictors of LRTI among HIV infected patients, we considered the start of follow up to be the first ART initiation day plus 60 days. End of follow-up was defined as the date of the first LRTI episode, for those who developed LRTI during follow-up. For those with no LRTI during follow-up, the follow-up ended at the date of death, or the date of the last clinical visit or study closure (31st December 2015), whichever occurred first. Patients whose date of the last clinical visit was prior to 1st January 2015 were deemed lost to follow-up. Exclusion criteria were: not have used ART for a minimum period of 60 days before end of follow-up (n = 35) and not having a CD4 measure during follow-up (n = 5).
Community-acquired pneumonia and lower respiratory tract infection definitions
Probable pneumonia was defined as at least two of the following signs and symptoms (1) fever and/or cough; (2) new purulent sputum; (3) dyspnea or tachypnea and (4) pleuritic chest pain AND no radiological confirmation AND adequate antibiotic treatment given. Definite pneumonia was defined as the clinical syndrome as in probable pneumonia AND antibiotic treatment AND a new pulmonary infiltrate on chest radiographs or CT scans, with or without bacteriological confirmation. Probable and definite pneumonias were grouped as lower respiratory tract infections.
For this study, we used the first diagnosis of LRTI after cohort enrollment for each patient.
Statistical analysis and independent variables
The independent variables included in the models were sex at birth, age, race/skin color, educational level, tobacco smoking (ever, as a dichotomic yes or no variable), alcohol use (ever, yes or no), cocaine inhalation, smoking or injection drug use (ever, yes or no), diabetes mellitus, CD4 count, HIV viral load (within the last year of follow up), influenza vaccination and PPSV23 vaccination. The protective effects of vaccination were defined to start at the 14th day after the vaccination, and to last 1 year for influenza vaccine and for 5 years for PPSV23. Demographic and clinical characteristics were compared between patients who had LRTI and those who did not by using the Wilcoxon Rank-Sum test for continuous variables and Chisquared for categorical variables.
Extended Cox proportional hazards models accounting for time-updated variables were fitted to assess LRTI predictors among HIV infected patients on ART. Patients’ follow-ups were split annually until the end of follow-up. For each patient’s period a CD4 count was assigned (the closest CD4 result available within the year before that period’s final date), to account for the immunologic recovery experienced by the patients after ART initiation. Besides CD4, age and influenza and PPSV23 vaccination status were included as time dependent variables. For 5% of all patients’ follow-up periods, there were no CD4 counts available, and simple interpolation of consecutives CD4 results within the same patient was performed. Independent variables with a p value below 0.10 in the unadjusted models were included in the adjusted model, in order to explore their effects after adjusting for confounding variables. Age and gender were forced into the final model. Proportional hazards’ assumption was tested using Schoenfeld residuals, and no variable violated this assumption. R (version 3.0.3) and libraries “survival” and “rms” were used for the analyses.
Ethical considerations
This study was approved by the ethics committee of INI Evandro Chagas, Fiocruz (CAAE 0032.0.009.000-10) and was conducted according to the principles expressed in the Declaration of Helsinki. All patient records/information were de-identified prior to analysis.
Results
Overall, 2709 HIV infected ART-naïve patients aged 18 years or more, enrolled at the INI cohort between 1st January 2000 and 31st October 2015, who were followed-up for at least 60 days and started ART after enrollment were eligible for this study. Of those, 35 were excluded because they used ART for less than 60 days before end of follow-up and 5 because they had no CD4 count result during follow-up. The final study population included 2669 patients, accounting for a total follow-up of 12,505 person-years PY (median follow-up of 3.9 years per patient). Three hundred and eighty-four patients had an episode of LRTI during follow-up, yielding an incidence rate of 30.7/1000 PY [95% confidence interval (CI) 27.8–33.9/1000PY] and 112 (4.2%) were deemed lost to follow-up (Fig. 1). According to the definitions used, of the 384 cases of pneumonia, 83.6% had probable and 16.4% had a definite diagnosis. Comparison of patients with definite and probable pneumonia is provided in supplementary Table 1.
Fig. 1.
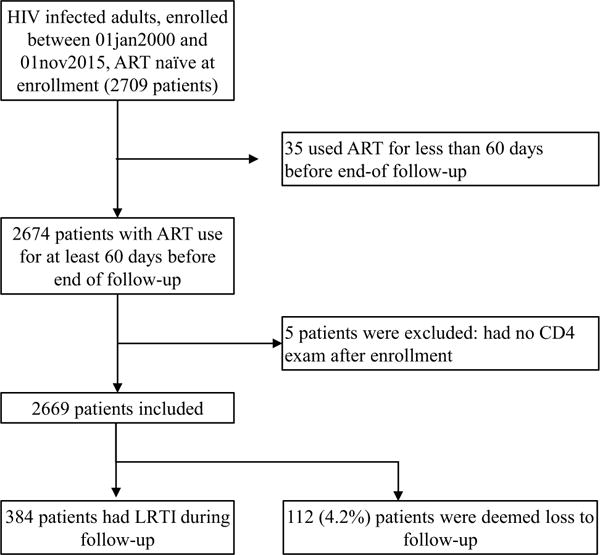
Inclusion and follow up of HIV-infected adults for the outcome lower respiratory tract infection, INI cohort, 2000–2015
Characteristics of the study population are shown in Table 1. Patients who had LRTI were more likely to have lower educational level (62 vs. 44.6%, p value <0.001), diabetes mellitus (13.3 vs. 8.7%, p value 0.06), cocaine use (16.4 vs. 6.5%, p value <0.001), tobacco smoking (63 vs. 56.8%, p value 0.027), lower CD4 counts (nadir: median 103 vs. 220 cells/mm3, p value <0.001) and within last year of follow up: 385 vs. 595 cells/mm3, p value <0.001) and HIV viral load ≥400 copies/mL within the last year of follow-up (27.3% vs. 10.3%, p value <0.001) than those with no LRTI.
Table 1.
Sociodemographic and clinical features of the study population according to diagnosis of community-acquired lower respiratory tract infection, INI cohort, 2000–2015
| No LRTI during FU (n = 2285) | Had LRTI during FU(n = 384) | Total study population (n = 2669) | p value | |
|---|---|---|---|---|
| Sex at birth (%) | 0.186 | |||
| Male | 1615 (70.7) | 258 (67.2) | 1873 (70.2) | |
| Female | 670 (29.3) | 126 (32.8) | 796 (29.8) | |
| Age at end of FU in years, median (IQR) | 40.6 (33.5,48.8) | 39.6 (33,48.5) | 40.4 (33.5,48.7) | 0.603 |
| <30 (%) | 302 (13.2) | 56 (14.6) | 358 (13.4) | 0.223 |
| 30–39 (%) | 794 (34.7) | 143 (37.2) | 937 (35.1) | |
| 40–49 (%) | 688 (30.1) | 101 (26.3) | 789 (29.6) | |
| 50–59 (%) | 379 (16.6) | 56 (14.6) | 435 (16.3) | |
| 60+ (%) | 122 (5.3) | 28 (7.3) | 150 (5.6) | |
| Race/skin color (%) | 0.759 | |||
| White | 1082 (47.4) | 178 (46.4) | 1260 (47.2) | |
| Non-white | 1203 (52.6) | 206 (53.6) | 1409 (52.8) | |
| Educational level (%) | <0.001 | |||
| Up to 9 years | 1019 (44.6) | 238 (62) | 1257 (47.1) | |
| More than years | 1266 (55.4) | 146 (38) | 1412 (52.9) | |
| Diabetes mellitus (%) | ||||
| No | 2086 (91.3) | 333 (86.7) | 2419 (90.6) | 0.006 |
| Yes | 199 (8.7) | 51 (13.3) | 250 (9.4) | |
| Cocaine usea (%) | <0.001 | |||
| No | 2136 (93.5) | 321 (83.6) | 2457 (92.1) | |
| Yes | 149 (6.5) | 63 (16.4) | 212 (7.9) | |
| Tobacco smoking ever | ||||
| No | 986 (43.2) | 142 (37) | 1128 (42.3) | 0.027 |
| Yes | 1299 (56.8) | 242 (63) | 1541 (57.7) | |
| Alcohol use report | 0.219 | |||
| No | 564 (24.7) | 82 (21.4) | 646 (24.2) | |
| Yes | 1721 (75.3) | 302 (78.6) | 2023 (75.8) | |
| Hepatitis C | <0.001 | |||
| No | 2159 (94.5) | 345 (89.8) | 2504 (93.8) | |
| Yes | 126 (5.5) | 39 (10.2) | 165 (6.2) | |
| CD4 nadir (cells/mm3), median(IQR) | 220 (89,329) | 103 (32,233) | 205 (75,318) | <0.001 |
| ≥350 (%) | 496 (21.7) | 27 (7) | 523 (19.6) | <0.001 |
| 200–350 (%) | 745 (32.6) | 92 (24) | 837 (31.4) | |
| 50–199 (%) | 658 (28.8) | 145 (37.8) | 803 (30.1) | |
| <50 (%) | 386 (16.9) | 120 (31.2) | 506 (19) | |
| Last year CD4 countª (cells/mm3), median(IQR) | 595 (408,800) | 385 (219,601) | 562 (367,781) | <0.001 |
| >500 (%) | 1133 (49.6) | 123 (32) | 1256 (47.1) | <0.001 |
| 500–350 (%) | 329 (14.4) | 72 (18.8) | 401 (15) | |
| <350 (%) | 344 (15.1) | 157 (40.9) | 501 (18.8) | |
| Missing (%) | 479 (21) | 32 (8.3) | 511 (19.1) | |
| Last year HIV viral loadª (copies/mL) | <0.001 | |||
| <400 (%) | 1838 (80.4) | 241 (62.8) | 2079 (77.9) | |
| ≥400 (%) | 236 (10.3) | 105 (27.3) | 341 (12.8) | |
| Missing (%) | 211 (9.2) | 38 (9.9) | 249 (9.3) | |
| Vaccination | ||||
| Influenza (%)b | 341 (14.9) | 68 (17.7) | 409 (15.3) | 0.185 |
| PPVS23 (%)c | 1187 (51.9) | 121 (31.5) | 1308 (49) | <0.001 |
| Vaccination combined | ||||
| No vaccination | 1012 (44.3) | 241 (62.8) | 1253 (46.9) | <0.001 |
| Influenza + PPVS23 | 255 (11.2) | 46 (12) | 301 (11.3) | |
| Only PPVS23 | 932 (40.8) | 75 (19.5) | 1007 (37.7) | |
| Only Influenza | 86 (3.8) | 22 (5.7) | 108 (4) |
INI Instituto Nacional de Infectologia Evandro Chagas, Rio de Janeiro, Brazil, LRTI lower respiratory tract infection,FU follow-up, PPSV23 Pneumococcal polysaccharide vaccine
The last result within 12 months of before follow-up
Received Influenza vaccine within the last year and 14 days before end of follow-up
Received PPSV23 within the last five years and 14 days before end of follow-up
Within 1 year before ART start and 60 days after
Report of cocaine or injection drug (n = 23) ever use
Overall, influenza and PPSV23 vaccination covertures at the last year of FU were low (15.3 and 49%). Only 11.3% of the patients were vaccinated with both influenza and PPSV23. Regarding influenza vaccination coverture, the difference between patients who had LRTI and those who did not was not significant. On the other hand, a higher proportion of PPSV23 vaccination was observed among those who did not have LRTI (51.9 vs. 31.5%, p value <0.001). Additionally, there was a relevant overlap of both vaccines coverture in the study population. Regarding the last year of follow-up, amongst the 414 patients vaccinated against influenza, 73% were also vaccinated with PPSV23.
Table 2 shows the Cox extended models. In the unadjusted Cox extended models, non-white race/skin color, [crude hazard ratio (cHR) 1.28, p value = 0.016], report of cocaine use (cHR 2.01, p value <0.001), report of tobacco smoking (cHR 1.34, p value 0.007), hepatitis C co-infection (cHR 1.51, p value = 0.013) and having HIV viral load ≥400 copies/mL within the last year of follow-up (cHR 3.40, p value <0.001) increased the risk of LRTI. On the other hand, higher educational level (cHR 0.61, p value <0.001), rise in CD4 counts over the years (cHR 0.81, p value <0.001, per 100 cells/mm3 increase), having been vaccinated for influenza (cHR 0.60, p value = 0.002) and for PPVS23 (cHR 0.57, p value <0.001) were associated with lower risk of LRTI. The Cox extended model for patients with definite pneumonia is provided in supplementary Table 2.
Table 2.
Time dependent Cox extended models for the risk of having lower respiratory tract infection during follow-up, INI cohort, 2000–2015
| Unadjusted HR (95% CI) | Adjusted HR (95% CI) | |
|---|---|---|
| Sex | ||
| Male | Ref. | Ref. |
| Female | 1.04 (0.83, 1.29) | 1.07 (0.85, 1.34) |
| Age category (in years)a | ||
| <30 | Ref. | Ref. |
| 30–39 | 0.92 (0.68, 1,26) | 0.92 (0.67, 1.25) |
| 40–49 | 0.73 (0.52, 1.01) | 0.73 (0.53, 1.02) |
| 50–59 | 0.94 (0.65, 1.37) | 0.96 (0.66, 1.41) |
| 60+ | 1.40 (0.90, 2.18) | 1.44 (0.92, 2.26) |
| Race/skin color | ||
| White | Ref. | |
| Non-white | 1.28 (1.05, 1.58) | 1.14 (0.93, 1.41) |
| Educational level | ||
| Up to 9 years | Ref. | |
| More than 9 years | 0.61 (0.49, 0.75) | 0.84 (0.67, 1.06) |
| Cocaine use | ||
| No | Ref. | Ref |
| Yes | 2.01 (1.52, 2.65) | 1.47 (1.07, 2.02) |
| Tobacco smoking (ever) | ||
| No | Ref. | Ref. |
| Yes | 1.30 (1.04, 1.62) | 1.23 (0.99, 1.53) |
| Alcohol drinking | ||
| No | Ref. | |
| Yes | 1.16 (0.90, 1.49) | |
| Diabetes mellitus | ||
| No | Ref. | |
| Yes | 1.07 (0.79, 1.46) | |
| Hepatitis C | ||
| No | Ref. | Ref. |
| Yes | 1.51 (1.09, 2.10) | 1.14 (0.80, 1.63) |
| CD4 counta (per 100cells/mm3 increase) | 0.81 (0.77, 0.85) | 0.86 (0.82, 0.91) |
| Last year HIV viral load (copies/mL) | ||
| <400 | Ref. | Ref. |
| ≥400 | 3.40 (2.67, 4.33) | 2.20 (1.70, 2.86) |
| Missing | 1.21 (0.86, 1.71) | 0.99 (0.69, 1.41) |
| Influenza vaccinationa | ||
| No | Ref. | Ref. |
| Yes | 0.60 (0.46, 0.79) | 0.81 (0.61, 1.08) |
| PPVS23 vaccinationa | ||
| No | Ref. | Ref. |
| Yes | 0.57 (0.45, 0.70) | 0.81 (0.64, 1.03) |
Bold values indicate statistical significance
INI Instituto Nacional de Infectologia Evandro Chagas, Rio de Janeiro, Brazil, HR hazard ratio, CI confidence interval, PPSV23 pneumococcal polysaccharide
Variables included in the model as time-dependent
On the final adjusted model, report of cocaine use (aHR 1.47, p value = 0.018) and having HIV viral load ≥400 copies/mL within the last year of follow-up (aHR 2.20, p value <0.001) were still significantly associated with increased risk of LRTI. Rise in CD4 counts protected against LRTI (aHR 0.86 per 100 cells/mm3 increase, p value <0.001). The effect of the other independent variables was attenuated in the adjusted model.
Hospitalization occurred on the same day of the diagnosis of LRTI for 74 (19.3%) patients and within 72 h of diagnosis for an additional 5(79, 20.6%).
Mortality within 30 days of the date of diagnosis of LRTI was 10/384 (2.6%). The underlying cause given on the death certificates were AIDS for 7 patients, COPD for 1, alcoholism for 1 and oral cancer for 1.
Discussion
Incidence of LRTI
Diagnosis of pneumonia in the outpatient real life scenario is often limited by the lack of radiographic and microbiological confirmation, as was the case in our study. Therefore, we used the term lower respiratory tract infection to include cases of definite and probable pneumonia. Although all our patients were on ART, the incidence rate of LRTI was 30.7/1000 person-years(PY), higher than that encountered for CAP in HIV infected patients reported in high-income settings. This may be due to the fact ours is a middle-income scenario or that tracheobronchitis cases were included as well as CAP. In France, a cohort study of 3336 HIV infected patients (65% of them were on ART), followed between 2000 and 2007, estimated an incidence of CAP of 12/1000 PY [18]. In the US, in a cohort of 3707 HIV-infected patients (65% of them were on ART) from 1999 to 2007, incidence rate of CAP was 28.0/1000 PY among HIV-infected patients [19]. In Spain, in 186 episodes of LRTI in 161 patients; wherein “any antiretroviral treatment” had been given to 45% of the patients, incidence rates of CAP were 31 and 24 in the years 2000 and 2005, respectively [20]. In a large, predominantly European study, the incidence rate of CAP was 5.4 per 1000 PY in 10,851 HIV infected patients included from 2006 to the first event; 84% of this cohort was on ART and the median of follow up of 3.9 years [8]. To our knowledge, incidence rates of CAP or of LRTI among HIV infected individuals living in low- and middle-income countries have not been reported in the modern, post ART era.
Most important predictors for LRTI
The most important predictors for LRTI were viral load and CD4 cell counts. Uncontrolled infection (detectable viral load) doubled the risk for LRTI, while time updated increase in CD4 counts were protective for LRTI.
The most recent (within the last year before end of follow-up) median CD4 count in patients who did not develop LRTI was 595 cells/mm3, while it was 385 in those who did. These values are higher than the ones previously reported among HIV infected patients with CAP, but it is worth mentioning that all these reports focused on hospitalized patients, and therefore the sickest and/or the most vulnerable socially [12, 15, 16, 19]. The median CD4 count was 56 (IQR 23-187) in hospitalized patients with respiratory conditions in Mexico [15], 88 (IQR 28-231) in hospitalized patients with non-AIDS related infections in West African countries [12]; 264 (IQR 108-451) in American patients with pulmonary diseases, mostly bacterial pneumonia and COPD [19], and 313 (100-460) in Danish patients hospitalized for bacterial pneumonia [16].
In our study, we showed that increases in CD4 count (included in the model as a time-updated variable) protected against LRTI with an aHR of 0.86 (0.82, 0.91) per 100 cells/mm3 increase. This finding is in accordance with those from a North American cohort of HIV-infected women, wherein increments of 50 CD4+ cells/mm3 decreased the risk of CAP, with an aHR of 0.88 (0.86–0.93) [21], a similar finding to ours. In a study on severe bacterial non-AIDS infection [8], when an adjusted model of pneumonia alone was used, CD4 counts as high as 350–499 cells/mL, compared with CD4 counts greater than 500 cells/mL, were associated with increased risk of infection (adjusted IRR 1.58, 95% CI 1.03–2.43).
In our cohort, patients of non-white race had a higher risk of LRTI. In Brazil, as in other countries in the Americas, non-white groups are still in social disadvantage when compared to whites, for historical reasons. This gap is narrowing, but is still present. In parallel, a higher educational level (>9 years of formal education) was protective for LRTI. We used years of formal education as a proxy for socioeconomic level. The influence of socio-economic status on incidence of LRTI has not been reported on HIV positive patients. Paradoxically, however, socio-economic status did not affect pneumonia hospitalization outcomes such as death and ICU admission, but did increase length of hospital stay and readmission rates in studies in developed countries [22–24]. This has not been reported in HIV positive patients with pneumonia.
Our study showed a borderline increased risk of LRTI associated with age >60 years; age above 60 years has been shown to be associated with a higher risk for CAP in HIV infected patients [18] as well as in the general population [2].
Cocaine
Another independent variable associated with LRTI was cocaine use, with an aHR 1.47 (1.07, 2.02). Use of cocaine included any mode of administration such as smoking (freebase crack cocaine), inhalation of powder cocaine, or, infrequently, intravenous injection. Respiratory symptoms are common after cocaine exposure, particularly after exposure to the combustion products of crack cocaine; furthermore, smoking crack cocaine alters alveolar macrophage function and cytokine production, severely limiting their ability to kill bacteria [25]. A previous study has shown that smoking illicit drugs (marijuana, cocaine, or crack) increased the risk of CAP in HIV positive individuals [26]; in multivariate analysis smoking illicit drugs (OR 2.24, 95% CI 1.03–4.89) remained significantly associated with bacterial pneumonia. More recently, it has been demonstrated that cocaine enhances the rate of HIV gene expression and replication by activating various signal transduction pathways and downstream transcription factors [27], and epidemiological associations between cocaine use and progression to AIDS [28] may have this pathophysiological underlying role. However, it must be emphasized that abusing drugs is well known to lead to poor compliance with antiretroviral therapy. A study by Nacher et al. in a large cohort of HIV positive individuals in the French Guiana showed that untreated patients and those taking HARRT, HIV-1 viral load was significantly higher among patients addicted to cocaine, after adjusting for age, sex, and CD4 [29]. Patients addicted to crack cocaine were also more likely to progress to AIDS than those not using crack cocaine, controlling for age, sex, nationality, CD4, CD8, HAART use, and alcohol addiction [29]. Crack cocaine users were more likely than non-users to develop a number of AIDS related illnesses, including bacterial pneumonia.
Vaccines
Importantly, a protective effect for 23-valent pneumococcal vaccine and influenza vaccines were seen regarding acquisition of LRTI, although overall coverage of these vaccines was only 49% and 15% respectively in our cohort. Having been vaccinated for influenza (cHR 0.60, p value = 0.002) and for PPVS23 (cHR 0.57, p value <0.001) were associated with a lower risk for LRTI.
Vaccination of HIV positive individuals with the 23-valent pneumococcal has long been recommended [30], but its effectiveness is debated [31, 32]. There are currently two types of pneumococcal vaccines—the PPV23, available since 1983, and the conjugate pneumococcal vaccine (PCV), containing 13 serotypes, available since 2010. In Brazil, the only vaccine available in the public health system is the PPV23, which is recommended at HIV diagnosis and 5 years after the first dose. Influenza vaccination is recommended annually. A recent meta-analysis confirmed that vaccination among HIV patients reduces both influenza-like illness and lab-confirmed influenza [33].
Limitations and strengths
Our study has some limitations. Firstly, our database does not reliably provide information on the intensity of tobacco use and alcohol consumption or on comorbidities such as COPD and heart failure, all of which have been associated with pneumonia in adults. Also, information on cotrimoxazole prophylaxis was inconsistent, and it may have provided a protective effect on LRTI. Lastly, our cohort population is non-probabilistic such that our findings may lack generalizability.
The strengths of our study are that it has a large number of patients (n = 2669) that have been enrolled and followed as outpatients for a fair amount of time (3.9 years median). All patients were on ART so that its effect could be measured. Adherence to ART was objectively assessed by CD4 counts and viral loads, which were regularly verified in the outpatient clinic. Furthermore, data on vaccination status was available for analysis.
Conclusions
LRTI has a high incidence in HIV infected adults using antiretroviral therapy.
Higher CD4 counts and undetectable viral loads were protective, reinforcing the importance of ART.
A protective effect for 23-valent pneumococcal vaccine and influenza vaccines was seen, despite low coverage for both.
Cocaine use was a strong predictor of LRTI in this population.
These data provide substrata for recommendations to curb the burden of LRTI in HIV positive adults. In the short term, early start of ART is mandatory, as is complying with the pneumococcal and flu vaccination guidelines. In the short and medium term, addressing the issue of cocaine drug abuse is very important. And in the medium to longer term, providing education to our population is crucial.
Supplementary Material
Acknowledgments
We thank Dr. Michael Bruce Macrae for his assistance with language editing. This work was supported in part by the NIH-funded Caribbean, Central and South America network for HIV epidemiology (CCASAnet), a member cohort of the International Epidemiologic Databases to Evaluate AIDS (leDEA) (U01AI069923). This award is funded by the following institutes: Eunice Kennedy Shriver National Institute of Child Health & Human Development (NICHD), National Cancer Institute (NCI), National Institute Of Allergy And Infectious Diseases (NIAID), National Institute Of Mental Health (NIMH), and the Office Of The Director, National Institutes Of Health (OD).”.
Footnotes
Electronic supplementary material The online version of this article (doi:10.1007/s15010-017-1041-0) contains supplementary material, which is available to authorized users.
Compliance with ethical standards
Conflict of interest: The authors declare that they have no competing interests.
References
- 1.Lozano R, Naghavi M, Foreman K, et al. Global and regional mortality from 235 causes of death for 20 age groups in 1990 and 2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet. 2012;380:2095–128. doi: 10.1016/S0140-6736(12)61728-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Torres A, Peetermans WE, Viegi G, Blasi F. Risk factors for community-acquired pneumonia in adults in Europe: a literature review. Thorax. 2013;68:1057–65. doi: 10.1136/thoraxjnl-2013-204282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mullerova H, Chigbo C, Hagan GW, et al. The natural history of community-acquired pneumonia in COPD patients: a population database analysis. Respir Med. 2012;106:1124–33. doi: 10.1016/j.rmed.2012.04.008. [DOI] [PubMed] [Google Scholar]
- 4.Kornum JB, Due KM, Norgaard M, et al. Alcohol drinking and risk of subsequent hospitalisation with pneumonia. Eur Respir J. 2012;39:149–55. doi: 10.1183/09031936.00000611. [DOI] [PubMed] [Google Scholar]
- 5.Schwarcz L, Chen MJ, Vittinghoff E, Hsu L, Schwarcz S. Declining incidence of AIDS-defining opportunistic illnesses: results from 16 years of population-based AIDS surveillance. Aids. 2013;27:597–605. doi: 10.1097/QAD.0b013e32835b0fa2. [DOI] [PubMed] [Google Scholar]
- 6.Lima VD, Lourenço L, Yip B, Hogg RS, Phillips P, Montaner JS. AIDS incidence and AIDS-related mortality in British Columbia, Canada, between 1981 and 2013: a retrospective study. Lancet HIV. 2015;2:e92–7. doi: 10.1016/S2352-3018(15)00017-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Coelho L, Cardoso SW, Amancio RT, et al. Trends in AIDS-defining opportunistic illnesses incidence over 25 years in Rio de Janeiro, Brazil. PLoS One. 2014;9:e98666. doi: 10.1371/journal.pone.0098666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Søgaard OS, Reekie J, Ristola M, et al. Severe bacterial non-aids infections in HIV-positive persons: incidence rates and risk factors. J Infect. 2013;66:439–46. doi: 10.1016/j.jinf.2012.12.012. [DOI] [PubMed] [Google Scholar]
- 9.Ribeiro SR, Luz PM, Campos DP, et al. Incidence and determinants of severe morbidity among HIV-infected patients from Rio de Janeiro, Brazil, 2000–2010. Antivir Ther. 2014;19:387–97. doi: 10.3851/IMP2716. [DOI] [PubMed] [Google Scholar]
- 10.Grinsztejn B, Luz PM, Pacheco AG, et al. Changing mortality profile among HIV-infected patients in Rio de Janeiro, Brazil: shifting from AIDS to non-AIDS related conditions in the HAART era. PLoS One. 2013;8:e59768. doi: 10.1371/journal.pone.0059768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ford N, Shubber Z, Meintjes G, et al. Causes of hospital admission among people living with HIV worldwide: a systematic review and meta-analysis. Lancet HIV. 2015;2:e438–44. doi: 10.1016/S2352-3018(15)00137-X. [DOI] [PubMed] [Google Scholar]
- 12.Lewden C, Drabo YJ, Zannou DM, et al. Disease patterns and causes of death of hospitalized HIV-positive adults in West Africa: a multicountry survey in the antiretroviral treatment era. J Int AIDS Soc. 2014;7:18797. doi: 10.7448/IAS.17.1.18797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kim JH, Psevdos G, Jr, Gonzalez E, Singh S, Kilayko MC, Sharp V. All-cause mortality in hospitalized HIV-infected patients at an acute tertiary care hospital with a comprehensive outpatient HIV care program in New York City in the era of highly active antiretroviral therapy (HAART) Infection. 2013;41:545–51. doi: 10.1007/s15010-012-0386-7. [DOI] [PubMed] [Google Scholar]
- 14.Feikin DR, Feldman C, Schuchat A, et al. Global strategies to prevent bacterial pneumonia in adults with HIV disease. Lancet Infect Dis. 2004;4:445–55. doi: 10.1016/S1473-3099(04)01060-6. [DOI] [PubMed] [Google Scholar]
- 15.Báez-Saldaña R, Villafuerte-García A, Cruz-Hervert P, et al. Association between highly active antiretroviral therapy and type of infectious respiratory disease and all-cause in-hospital mortality in patients with HIV/AIDS: a case series. PLoS One. 2015;10:e0138115. doi: 10.1371/journal.pone.0138115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sogaard OS, Lohse N, Gerstoft J, et al. Hospitalization for pneumonia among individuals with and without HIV infection, 1995–2007: a Danish population-based, nationwide cohort study. Clin Infect Dis. 2008;47:1345–53. doi: 10.1086/592692. [DOI] [PubMed] [Google Scholar]
- 17.BRASIL. Ministério da Saúde/Secretaria de Vigilância em Saúde. Departamento de DST, Aids e Hepatites Virais. Protocolo clínico e diretrizes terapêuticas para manejo da infecção pelo HIV em adultos. Brasília 2013. 2015 [Google Scholar]
- 18.Benard A, Mercie P, Alioum A, et al. Bacterial pneumonia among HIV-infected patients: decreased risk after tobacco smoking cessation. ANRS CO3 Aquitaine Cohort, 2000–2007. PLoS ONE. 2010;5:e8896. doi: 10.1371/journal.pone.0008896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Crothers K, Huang L, Goulet JL, et al. HIV infection and risk for incident pulmonary diseases in the combination antiretroviral therapy era. Am J Respir Crit Care Med. 2011;183:388–95. doi: 10.1164/rccm.201006-0836OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Curran A, Falco V, Crespo M, et al. Bacterial pneumonia in HIV-infected patients: use of the pneumonia severity index and impact of current management on incidence, aetiology and outcome. HIV Med. 2008;9:609–15. doi: 10.1111/j.1468-1293.2008.00603.x. [DOI] [PubMed] [Google Scholar]
- 21.Kohli R, Lo Y, Homel P, Flanigan TP, Gardner LI, Howard AA, et al. Bacterial pneumonia, HIV therapy, and disease progression among HIV-infected women in the HIV epidemiologic research (HER) study. Clin Infect Dis. 2006;43:90e8. doi: 10.1086/504871. [DOI] [PubMed] [Google Scholar]
- 22.Izquierdo C, Oviedo M, Ruiz L, Sintes X, Vera I, Nebot M, et al. Influence of socioeconomic status on community-acquired pneumonia outcomes in elderly patients requiring hospitalization: a multicenter observational study. BMC Public Health. 2010;15:421. doi: 10.1186/1471-2458-10-421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Jasti H, Mortensen EM, Obrosky DS, Kapoor WN, Fine MJ. Causes and risk factors for re-hospitalization of patients hospitalized with community-acquired pneumonia. Clin Infect Dis. 2008;46:550–6. doi: 10.1086/526526. [DOI] [PubMed] [Google Scholar]
- 24.Stelianides S, Golmard JL, Carbon C, Fantin B. Influence of socioeconomic status on features and outcome of community-acquired pneumonia. Eur J Clin Microbiol Infect Dis. 1999;18:704–8. doi: 10.1007/s100960050382. [DOI] [PubMed] [Google Scholar]
- 25.Baldwin GC, Tashkin DP, Buckley DM, Park AN, Dubinett SM, Roth MD. Marijuana and cocaine impair alveolar macrophage function and cytokine production. Am J Respir Crit Care Med. 1997;156:1606–13. doi: 10.1164/ajrccm.156.5.9704146. [DOI] [PubMed] [Google Scholar]
- 26.Caiaffa WT, Vlahov D, Graham NM, et al. Drug smoking, Pneumocystis carinii pneumonia, and immunosuppression increase risk of bacterial pneumonia in human immunodeficiency virus-sero-positive injection drug users. Am J Respir Crit Care Med. 1994;150:1493–8. doi: 10.1164/ajrccm.150.6.7952605. [DOI] [PubMed] [Google Scholar]
- 27.Tyagi M, Weber J, Bukrinsky M, Simon GL. The effects of cocaine on HIV transcription. J Neurovirol. 2016;22:261–74. doi: 10.1007/s13365-015-0398-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cook JA. Associations between use of crack cocaine and HIV-1 disease progression: research findings and implications for mother-to-infant transmission. Life Sci. 2011;88:931–9. doi: 10.1016/j.lfs.2011.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Nacher M, Adenis A, Hanf M, et al. Crack cocaine use increases the incidence of AIDS-defining events in French Guiana. AIDS. 2009;23:2223–6. doi: 10.1097/QAD.0b013e32833147c2. [DOI] [PubMed] [Google Scholar]
- 30.Centers for Disease Control (CDC) Recommendations of the Immunizations Practices Advisory Committee: pneumococcal polysaccharide vaccine. MMWR. 1989;38:73–6. [PubMed] [Google Scholar]
- 31.Barry PM, Zetola N, Keruly JC, Moore RD, Gebo KA, Lucas GM. Invasive pneumococcal disease in a cohort of HIV-infected adults: incidence and risk factors, 1990–2003. AIDS (London, England) 2006;20:437e44. doi: 10.1097/01.aids.0000206507.54901.84. [DOI] [PubMed] [Google Scholar]
- 32.Pedersen R, Lohse N, Ostergaard L, Sogaard O. The effectiveness of pneumococcal polysaccharide vaccination in HIV-infected adults: a systematic review. HIV Med. 2011;12:323e33. doi: 10.1111/j.1468-1293.2010.00892.x. [DOI] [PubMed] [Google Scholar]
- 33.Beck CR, McKenzie BC, Hashim AB, Harris RC, University of Nottingha1m Influenza and the Immuno-Compromised (UNIIC) Study Group. Nguyen-Van-Tam JS. Influenza vaccination for immunocompromised patients: systematic review and meta-analysis by etiology. J Infect Dis. 2012;206:1250–9. doi: 10.1093/infdis/jis487. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


