Abstract
We have an operant rat model of upper extremity reaching and grasping in which we examined the impact of performing a high force high repetition (High-ForceHR) versus a low force low repetition (Low-ForceHR) task for 18 weeks on the radius and ulna, compared to age-matched controls. High-ForceHR rats performed at 4 reaches/min and 50% of their maximum voluntary pulling force for 2 hours/day, 3 days/week. Low-ForceHR rats performed at 6% maximum voluntary pulling force. High-ForceHR rats showed decreased trabecular bone volume in the distal metaphyseal radius, decreased anabolic indices in this same bone region (e.g., decreased osteoblasts and bone formation rate), and increased catabolic indices (e.g., microcracks, increased osteocyte apoptosis, secreted sclerostin, RANKL, and osteoclast numbers), compared to controls. Distal metaphyseal trabeculae in the ulna of High-ForceHR rats showed a non-significant decrease in bone volume, some catabolic indices (e.g., decreased trabecular numbers) yet also some anabolic indices (e.g., increased osteoblasts and trabecular thickness). In contrast, the mid-diaphyseal region of High-ForceHR rats’ radial and ulnar bones showed few to no microarchitecture differences and no changes in apoptosis, sclerostin or RANKL levels, compared to controls. In further contrast, Low-ForceHR rats showed increased trabecular bone volume in the radius in the distal metaphysis and increased cortical bone area its mid-diaphysis. These changes were accompanied by increased anabolic indices, no microcracks or osteocyte apoptosis, and decreased RANKL in each region, compared to controls. Ulnar bones of Low-ForceHR rats also showed increased anabolic indices, although fewer than in the adjacent radius. Thus, prolonged performance of an upper extremity reaching and grasping task is loading-, region-, and bone-dependent, with high force loads at high repetition rates inducing region-specific increases in bone degradative changes that were most prominent in distal radial trabeculae, while low force task loads at high repetition rates induced adaptive bone responses.
Keywords: Overuse injury, mechanical loading, osteoprotegerin, osteoactivin, sclerostin, RANKL, RANK
Graphical Abstract

INTRODUCTION
Most types of physical activity are considered beneficial, whether sports, planned exercise, household work or occupational tasks (Clark, Goodship et al. 1975, Blair, Kohl et al. 1992). Yet, work activities are not considered exercise; the former typically lacking any cardiovascular benefits (Kivimaki, Jokela et al. 2015). Involvement in heavy manual occupations is linked to higher incidence of musculoskeletal injuries than nonmanual and light manual occupations (Villotte, Castex et al. 2010, Bureau of Labor Statistics 2016). The negative effects of repetitive and high compressive forces on lumbar spine structures when performing occupational jobs are well established (Vanharanta, Heliovaara et al. 1987, Seidler, Bergmann et al. 2009, Hlavkova, Lebeda et al. 2016). A small number of studies showing increased incidence of hand/wrist osteoarthritis and reduced bone mass in the hand and wrist in individuals with heavy or one-sided hand workloads (Solovieva, Vehmas et al. 2005, Vehmas, Solovieva et al. 2005, Ding, Solovieva et al. 2010, Dale, Ryan et al. 2015). This is a concern because incidence of distal radial bone fractures is increasing in the U.S. and worldwide (Nellans, Kowalski et al. 2012, Karl, Olson et al. 2015). There is also a need for etiologic research examining mechanisms underlying tissue degradative changes occurring with high demand upper extremity occupational tasks (NIOSH 2011).
Although not yet investigated with overuse injuries occurring as a consequence of occupational tasks, RANKL (Receptor activator of nuclear factor kappa-B ligand) is expressed by osteoblasts and osteocytes (Findlay and Atkins 2011), and stimulates differentiation and activation of osteoclasts by binding to its receptor, RANK (Matsuo 2012, Kennedy, Laudier et al. 2014). Osteocytes show metabolic responsiveness to bone loading or unloading (Skerry, Bitensky et al. 1989, Robling, Niziolek et al. 2008), and RANKL is one protein released by apoptotic osteocytes following loading-induced microdamage (Al-Dujaili, Lau et al. 2011, Kennedy, Laudier et al. 2014). Such microdamage has a catabolic effect on bones by promoting osteoclast activity in a RANKL-dependent manner (Wijenayaka, Kogawa et al. 2011). Osteoprotegerin (OPG) is a RANK decoy receptor that binds RANKL, segregating the ligand and preventing activation of the RANKL/RANK pathway (Boyce and Xing 2007). While osteocyte apoptosis and their subsequent release of RANKL is essential for bone remodeling, a prolonged shift in the RANKL and OPG ratio towards more RANKL enhances osteoclastogenesis and net bone resorption (Kennedy, Laudier et al. 2014).
Sclerostin is another protein expressed by osteocytes (Nakashima, Hayashi et al. 2011) that has yet to be examined with overuse occupational injuries. It is the product of the SOST gene and a negative regulator of osteoblast differentiation and function, making it a potent inhibitor of bone formation (Brunkow, Gardner et al. 2001, Mabilleau, Mieczkowska et al. 2010). Although the exact mechanisms by which sclerostin inhibits bone formation are still unclear, it is known that sclerostin selectively inhibits Wnt proteins from binding to low-density lipoprotein receptor-related protein 5/6 (LPR5/6) receptors, a change that antagonizes Wnt signaling (Lin, Jiang et al. 2009). The Wnt pathway is essential for loading-induced osteogenesis (Bonewald 2007), and its inhibition suppresses osteoblast activity and reduces osteoblast and osteocyte viability (Lin, Jiang et al. 2009). Applied mechanical loads typically suppress sclerostin production, a change that releases the break on Wnt signaling and allows for bone formation (Robling, Niziolek et al. 2008). However, loading-induced matrix microdamage in which osteocyte apoptosis occurs is associated with an enhanced release of both sclerostin and RANKL (Mabilleau, Mieczkowska et al. 2010), changes that promote bone catabolism.
We have developed an operant and clinically relevant rat model of upper extremity WMSDs in which rats learn a reaching and lever bar pulling task for a food reward (Barbe, Gallagher et al. 2013). Rats reach forward using their whole forearm to pull on a lever bar located outside of the chamber (which is attached to a force transducer) at learned and defined reach rates and target forces for a food reward. Young adult rats performing the task at low force loads and high repetition rates (Low-ForceHR) for 12 weeks show adaptive bone changes, including increased trabecular bone volume in the distal metaphysis of the radius (Barbe, Gallagher et al. 2013, Massicotte, Frara et al. 2015). This is consistent with studies reporting bone anabolism in response to loading (Christen, Ito et al. 2014). In contrast, rats performing a high force high repetition (High-ForceHR) task for 12 weeks show significant trabecular bone loss in the distal metaphysis (Barbe, Gallagher et al. 2013, Jain, Barr-Gillespie et al. 2014). Initially, an underlying inflammatory mechanism contributed to this overuse-induced trabecular bone loss, a mechanism confirmed with findings that anti-inflammatory treatments reduced this loss when provided in task weeks 4 through 12 since the inflammatory changes peaked in task week 6 (Rani, Barbe et al. 2010, Jain, Barr-Gillespie et al. 2014). These inflammatory responses were resolved in serum and muscles in 18 week High-ForceHR task rats (Fisher, Zhao et al. 2015). However, we hypothesized that trabecular bone catabolism would persist in distal radial metaphysis of 18 week High-ForceHR rats in association with increased microdamage, osteocyte apoptosis, sclerostin and RANKL. These findings would be consistent with a Damage-Repair Theory for bone in which persistent high loading causes an accumulation of damage in bone if the loading is so high that self-repair mechanisms cannot keep pace with the level of damage or overload-induced resorption (Frost 1994, Prendergast and Taylor 1994, Li, Li et al. 2007, Li, Li et al. 2011). That said, as stated in a recent review: “the contributions of varied, but unique, stimuli generated by muscle to bone remains to be established” (Avin, Bloomfield et al. 2015).
To explore this hypothesis, we compared radial and ulnar bone microarchitecture changes in rats performing a High-ForceHR reaching and lever pulling task for 18 weeks to those occurring in rats performing a Low-ForceHR task for 18 weeks (extending our past 12 week studies), versus resting control (C) rats. We examined for indices of bone microdamage and changes in proteins known to modulate loading-induced bone resorption versus formation (e.g., RANKL, RANK, OPG and sclerostin), proteins not yet examined in our operant repetitive loading model or in other models of chronic muscle-contraction loading of bone.
MATERIALS AND METHODS
Animals and Overview
The Temple University Institutional Animal Care and Use Committee approved all experiments in compliance with NIH guidelines for the care and use of laboratory animals. A total of 70 young (2.5 months of age at onset) female Sprague-Dawley rats were used (one rat was excluded after onset of experiments reducing the total number of rats used to 69; Fig 1A). All rats were housed in a central animal facility in separate cages with a 12 hour light:dark cycle, with free access to water and environment enrichment toys. All rats were initially food-restricted for 7 days to no more than 10–15% less than their naive weight to encourage interest in 45 mg food reward pellets (a 1:1 mix of purified grain and banana flavored pellets; Bioserve, NJ, USA). Then, the food-restricted rats were given extra rat chow and allowed to gain weight thereafter, to no more than 5% less than age-matched normal control rats (used for weight comparison only). Randomly chosen rats first learned to pull the lever bar at high force during a 6-wk shaping period of 10–15 min/day for 5 days/wk, before performing a high force high repetition (High-ForceHR) task that involved reaching and grasping of a lever bar for 2 hr/day, 3 days/wk for 18 wks (n=20) (Fig. 1A). As indicated above, one High-ForceHR rat was removed from the study when a tumor was detected, reducing the total number in this group to 19 (Fig. 1A). A second cohort initially shaped to learn to pull the lever bar at low force, before performing a low force high repetition (Low-ForceHR) task at similar timing (n=23). The remaining food restricted rats did not undergo shaping or task performance and were considered control (C) rats (n=27). The individuals carrying out all tissue analyses were blinded to treatment.
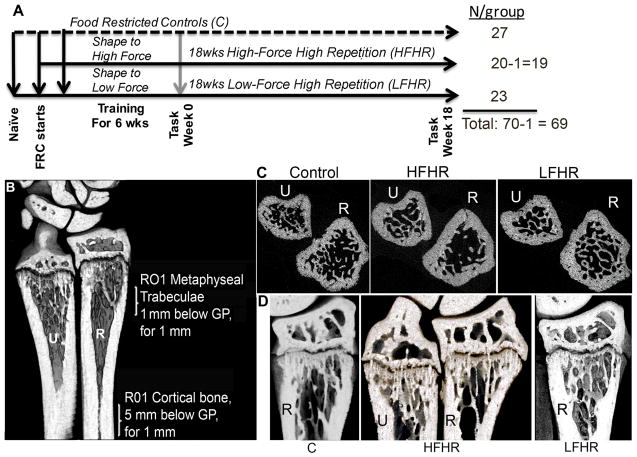
Figure 1.
Design and microCT images. (A) Young adult (2.5 months at onset) Sprague-Dawley rats were randomly assigned as food-restricted control (C) rats, rats that performed a high force high repetition task (High-ForceHR) for 18 weeks, or rats that performed a low force high repetition task (Low-ForceHR) for 18 weeks. Numbers per group shown at right. (B) Regions of interest (ROI) in radius (R) and ulnar (U) analyzed using micro-computerized tomography. (C) Upper row: Representative transaxial 2D image slices of distal ulnar and radial metaphyses. Lower row: Representative longitudinal 3D images of the distal radius. The ulna is also shown for a HFHR rat.
Task Apparatuses, Shaping and Task Paradigm
Operant behavioral apparatuses were as previously described (Barbe, Gallagher et al. 2013). Briefly, rats reached through a shoulder height portal and pulled on a vertical 1.5 mm metal bar, positioned 2.5 cm outside of the chamber wall, attached to a load cell (Futek Advanced Sensor Technology, Irvine, CA) (See Supplemental Movie). The load cell was interfaced with custom written Force-Lever software that allowed us to choose a required force level for provision of a food reward (Med Associates, St. Albans, VT). Light and auditory indicators (Med Associates) lasting 500 milliseconds (msec) cued the animal to attempt a reach every 15 sec.
Rats were first shaped to learn a reaching and lever-pulling task during a 6-wk period for 10 min/day, 5 days/wk, in which they ramped upwards from naïve towards high force or low force pulling (Fig. 1A), as previously described (Barbe, Gallagher et al. 2013, Jain, Barr-Gillespie et al. 2014). Briefly, rats learned to reach for a lever bar at increasing pulling forces, without any specified repetition rate, for a food reward. By the end of shaping week 6, rats randomly chosen to be High-ForceHR task rats were required to pull at 130 grams of force (gf; 1.27 Newtons), estimated to be 50% of their maximum pulling force, without any specified repetition rate, for a food reward. They then moved on to the High-ForceHR task. Rats randomly chosen to be Low-ForceHR task rats were required to pull at 17 grams of force (0.17 Newtons; 6% of their maximum pulling force), without any specified repetition rate, for a food reward, by the end of shaping period. They then moved on to the Low-ForceHR task.
The mean maximum voluntary pulling force of rats was determined on the last day of shaping by requiring the rat to pull at increasing levels for a food reward until their maximum level was reached. The maximum voluntary pulling force was 260 ± 7.02 (mean ± SEM) grams, which is 2.45 ± 0.07 Newtons.
The High-ForceHR task consisted of pulling the lever bar at a rate of 4 reaches/min, and at a graded force effort of 50% ± 5% (mean ± SEM) of their maximum pulling force, for 2 hr/day, 3 days/wk for 18 weeks. The Low-ForceHR task had the same reach rate and work/rest hrs, but had to pull on the lever bar at a graded force effort of 6% ± 5% of their maximum pulling force. Each daily task was divided into four 30 min sessions separated by 1.5 hrs each, to avoid satiation. If the above criteria were met within a 5 sec cueing period, a 45 mg food pellet reward was dispensed into a trough located at floor height for the animal to lick up. Rats were not prevented from reaching at a higher or lower force than their target force; however, if they either undershot the minimum criterion, or overshot the maximum criterion, no food reward was delivered (Supplemental Fig. 1). Rats were allowed to use their preferred limb to reach.
Determination of Reach Performance Behaviors in task rats
Force lever data were recorded continuously during each task session for later calculation of the dependent variables (mean reaches/session, reaches/min, reaches/day, reach force and grasp time) via a custom written and automated script (MATLAB, MathWorks®, Natick, MA) that translated the Force Lever Program data into Excel spreadsheets. Reach force was the mean recordable reach force (in gf) applied to the force lever bar for all reaches across a given day. Grasp time was the mean average time (in msec) that the rats spent grasping and pulling on the lever bar for all recordable reaches. Mean reach force and grasp time were calculated over the interval that ended when the force fell below 2.5% of baseline. Mean reach impulse was calculated for each rat by multiplying the mean reach force in Newton’s by the mean duration of grasp in msec. The percent duty cycle (the percent of time the rats were engaged in the reaching efforts) was calculated, as suggested for ergonomic reaching and grasping studies (Ciriello, Snook et al. 2001, Potvin 2012): ((number of reaches/cycle × grasp time/reach)/60 sec per cycle) × 100. Data for each variable was calculated on the last day of task weeks 1, 3, 6, 12 and 18 for 15 Low-ForceHR rats and 12 High-ForceHR rats. The total number of reaches, total volume of grasping the lever bar, and the total volume of loading (total reaches × total msec of grasping × mean force per grasp) across all weeks were estimated from these times points. These data could not be generated for control rats, as they did not perform the task.
Tissue collection
Animals were anesthetized with 5% isoflurane using oxygen as a carrier, and euthanized by cardiac puncture for blood collection using an 18-gauge needle at 36 hr after their last task session. Blood was kept on ice for 30 min, and allowed to clot before being centrifuged. Serum was harvested and frozen at −80°C until use. Thereafter, subcohorts of rats per group were perfused transcardially with 0.9% saline and then fixed with 4% paraformaldehyde in 0.1M PO4 buffer, pH 7.4, for serum analyses (n=8–15/group), forelimb bone (radius and ulna) micro-computerized tomography (microCT, n=5–14/group), en bloc basic fucshin staining and microcrack analysis (3 control and 3 High-ForceHR rat bones), bone static histomorphometry (n=6–12/group) and/or TUNEL staining (n=4–7/group), bone immunohistochemistry (n=5–7/group), muscle histomorphometry (n=5–6/group), or void analysis on radial and ulnar bones (n=6–9/group). Bones, forelimb flexor muscles and tendons were collected from other subcohorts of rats, flash frozen and used for mRNA (n=4–6/group) or protein assays (n=4–6/group).
MicroCT Imaging and Analysis
After fixation, as described above, forelimb bones were collected, cleaned of soft tissues, and stored in phosphate buffered saline (PBS) with sodium azide until microCT analysis of the radius (n=5–14/group) using a Skyscan 1172, 12 megapixel, high-resolution cone-beam microCT scanner (Bruker, Kontich, Belgium), using the following settings: a pixel resolution size of 5.89 μm, x-ray source spot size of 300 nm, Al 0.5mm filter, voltage of 59 kV, current of 167 μA, rotation step of 0.40°, frame averaging of 5. Skyscan volume rendering software (CTVox) and analysis software (CTAn) was used to render 3D models of longitudinal sections (Fig. 1C). MicroCT analysis was performed, as previously described (Massicotte, Frara et al. 2015) in metaphyseal and diaphyseal regions of the radius and ulna (Fig. 1B).
Histomorphometry of bone and muscle
Bones of rats assayed for microCT and from additional rats (in order to reach n=6–12/group) were used for histomorphometry using published guidelines (Parfitt, Drezner et al. 1987, Dempster, Compston et al. 2013). Calcein (i.p.,10 mg/kg body weight) had been previously injected at 9 and 2 days before euthanasia. Subcohorts of bones (n=4–5/gp) were processed and embedded in methyl methacrylate resin (MMA, Osteo-Bed Bone Embedding Kit, Polysciences, Warrington, PA), and sectioned into 5 μm longitudinal sections and placed on charged slides. Unstained plasticized longitudinal sections were used to measure the fluorochrome label. Histomorphometry for dynamic parameters of trabecular bone formation was then performed in the distal metaphyseal radius, as previously described (Massicotte, Frara et al. 2015). Sections were then stained with Masson’s Trichrome or TRAP5b and used for histomorphometry for static parameters of trabecular bone microarchitecture, as previously described (Massicotte, Frara et al. 2015) using an image analysis program (Osteo II, Bioquant, Nashville, TN). The remaining forelimb bones were paraffin-embedded (n=5–7/group), after decalcification in a 14% acid free EDTA solution. The paraffin-embedded sections were used for TUNEL and immunohistochemical staining (described below). The percentage of empty lacunae was determined in H&E stained bones by counting the number of empty osteocyte lacunae and then dividing by the total number of lacunae with or without evidence of cells, in 3 sections per rat radius (same metaphyseal and mid-diaphyseal regions as assayed with microCT). Void areas within the marrow areas of all bones were also qualitatively assessed during this scoring.
Flexor digitorum muscles were dissected off the radial and ulnar bones prior to microCT analyses, equilibrated in 30% sucrose in phosphate buffer (pH 7.4; n=5–6/group), and then cryosectioned at the mid-region of the muscle (at its widest point) into cross-sections of 12 μm thick each. These sections were mounted on slides, dried and stained with hematoxylin and eosin. An image analysis program was used (Bioquant) to measure the cross-sectional area (CSA) of the entire flexor digitorum muscle belly in mm2.
TUNEL staining in radius
Paraffin-embedded bone sections were deparaffinized with xylene (three times, for 45 min at 37°C, each), rehydrated in alcohol washes, and then washed in a 0.9% NaCl solution, followed by a PBS wash. Sections were permeabilized with a proteinase K buffer, followed by a rTdT enzyme, nucleotide mix and equilibration buffer solution incubation for 60 min at 37°C protected from light (G3250, Promega DeadEnd Fluorometric, Madison, WI, USA). Sections were batched stained to reduce variability. Positive control staining was achieved by pre-treating one tissue section with DNAse to generate DNA fragments. For negative controls, the rTdT enzyme was omitted during incubation. Slides were coverslipped with Vectashield + DAPI (4′,6-diamidino-2-phenylindole, H-1200, Vector Lab, Burlingame, CA, USA). The number of TUNEL-positive and DAPI-stained osteocytes was counted in metaphyseal trabecular and mid-diaphyseal cortical regions of the radius. Two sections were counted in the preferred reach limbs of 4–7 rats/group. Percent of TUNEL positive osteocytes per area was determined by dividing the number of TUNEL-positive osteocytes by the total number of DAPI-stained osteocytes, and multiplying by 100.
Immunohistochemistry
Immunohistochemistry for sclerostin and RANKL was performed on paraffin-embedded bone sections (n=5–7/group), after deparaffinization with xylene and rehydration through a series of alcohol washes, 0.5% pepsin antigen retrieval and blocking steps (4% goat serum in PBS, 30 min). Primary antibodies used were anti-Sclerostin (SostDC1, ab99340, Abcam, Cambridge, MA) and anti-RANKL (sc-7628, Santa Cruz Biotechnology, Santa Cruz, CA), at 1:200 and 1:400 dilutions, respectively, in PBS, for 24 hr at room temperature. Appropriate secondary antibodies (Jackson Immuno Research Labs, West Chester, PA) tagged with horseradish peroxidase were used (1:100 dilution in PBS for 2 hr at room temperature) and visualized using diaminobenzidine (SigmaFast D0426, Sigma-Aldrich, St. Louis, MO). Sections were batched stained to reduce variability. Preabsorption controls were performed to demonstrate specificity of RANKL and sclerostin antibodies using recombinant RANKL protein (ab9958, Abcam) and SOSTDC1 peptide (ab119134, Abcam). A two-fold excess of purified protein or peptide was pre-incubated with the matching antibody overnight at 4°C, the mixture centrifuged, and the pre-absorbed antibody supernatant then incubated with tissues (after pepsin and goat serum treatment) for two hr, before washing and incubation with secondary antibodies; no labeling was observed (data not shown). Negative control staining was performed by omitting either the primary or secondary antibody; no labeling was observed in any case (data not shown). Sclerostin and RANKL immunoexpression was quantified as percent area of tissue with immunostaining per area, using previously described methods (Al-Shatti, Barr et al. 2005), using a Nikon microscops (E800) linked to a digital camera (Q-Imaging Retiga camera) and a computer with image analysis software (Osteo II, Bioquant). Cells within the marrow area were excluded from these analyses. At least 3 adjacent trabecular regions (at 1.5 mm below the center of the epiphyseal plate) and mid-diaphyseal cortical regions were quantified per rat bone.
Microcrack Staining and Analysis
Bones of six additional rats (3 contols and 3 High-ForceHR rats) were used for en bloc basic fuchsin staining, using previously described methods (Colopy, Benz-Dean et al. 2004, Muir, Sample et al. 2007). Specimens of the radius and ulna (still attached via their interosseous membrane) were placed in 70% ethanol, then bulk stained in 1% basic fuchsin in a graded series of alcohols (80, 90 and 100%) under vacuum of 20 mm Hg for 24 hr before being embedded without decalcification in MMA. Specimens were cut into 80 micrometer transverse sections using a diamond blade saw at the level of the metaphyseal regions (trabeculae and cortical bone included) and mid-diaphysis regions. Cross-section were mounted on slides and linear microcracks were counted at 400 × magnification using a light microscope (Nikon E600) interfaced with a Nikon camera (DS-Ri2) and NIS-Elements. In vivo microcracks were defined as open cracks that were intensely stained with basic fuchsin, having a certain depth of field, and a surrounding halo of increased basic fucshin stain (Wenzel, Schaffler et al. 1996). Linear microcrack density (Cr.Dn = Cr.N/B.Ar, #/mm2) was then measured. Data from several sections of radial and ulnar bones were summarized.
qPCR analysis
Forelimb bones (radius and ulnar together) were collected from 6 Controls and 4 High-ForceHR rats (without flushing the marrow area), divided into distal (metaphysis and epiphysis combined) and mid-diaphysis using sharp scissors, flash frozen separately and stored at −80°C until use. After thawing on ice, the bones regions were powdered, keeping distal and mid-diaphyseal regions separate, using liquid nitrogen and a mortar and pestle, and RNA isolated using Trizol (15596-026, Life Technologies, Grand Island, NY, USA) and chloroform, before precipitating with 2-propanol. RNA was quantified using a Beckman spectrophotometer (Beckman Coulter, Jersey City, NJ, USA). cDNA was then synthesized using a cDNA kit (A-B1453/A, Thermo Scientific Verso, Pittsburgh, PA, USA) and a GeneAmp PCR 9700 system (Applied Biosystem, Grand Island, NY, USA). Quantification was achieved by Real Time PCR using SYBR Green PCR Master Mix (4309155, Applied Biosystems), ran in duplicate using a Real-Time PCR 7500 Instrument and analyzed with sequence detection software (V:1.2.3) from Applied Biosystems. Beta-actin was selected as the housekeeping gene. Primer’s sequences used are provided in Supplemental Table 1. Values reported are fold difference compared to Beta-actin using the 2ΔΔCT method.
ELISAs
Serum was collected from n=8–15/group and assayed using commercially available ELISA kits for levels of osteocalcin (AC-12F1, Rat-MID Osteocalcin EIA, Immunodiagnostic Systems, Tyne & Wear, UK), CTX-1 (AC-06F1, RatLaps EIA, Immunodiagnostic Systems), RANKL (RRNKLMAG-31K-01, Millipore Corporation, Billerica, MA), and sclerostin (MSST00, R&D Systems) following manufacturer protocols. For the RANKL assay, each sample was run in duplicate and analyzed using a Bio-Rad Bio-Plex System with a Bioplex manager 4.0 software (Hercules, CA). Forelimb bones (radius and ulnar together) of subsets of rats (n=4–6/group) were collected and divided into distal metaphyseal and mid-cortical regions, stored at −80°C until homogenized individually, as previously described (Barbe, Elliott et al. 2008), and analyzed for RANKL and sclerostin using the same ELISAs kits as for serum. Bones were also analyzed for osteoactivin using a commercially available DuoSet ELISA Development kit (DY2330, R&D Systems) onto which an anti-osteoactivin antibody was added (1:350, custom made anti-chicken OA; produced and verified for specificity, as previously described (Abdelmagid, Barbe et al. 2007)). Flexor muscles of the forelimb (flexor digitorum, flexor carpi ulnaris and flexor carpi radialis) and flexor digitorum tendons were also collected from 4–6 rats/group and analyzed for osteoactivin levels using ELISA. Results were analyzed using a VersaMax microplate reader and SoftmaxPro Version 5 software (Molecular Devices LLC, Sunnyvale, CA). ELISA data (pg cytokine protein) were normalized to μg total protein, which was determined using a bicinchoninic acid (3225, Pierce BCA Protein assay, Thermo Fisher Scientific, Waltham, MA). Each sample was run in duplicate in a blinded manner.
Western blot analysis
Bone homogenates from above were also assayed using 4–12% Tris-Glycine gels (Novex, XP04122BOX, Thermo Fisher Scientific) and then Western Blot analysis methods for TNFalpha levels using a specific anti-TNFalpha antibody (1:1000 dilution, ab6671, Abcam) and visualized using Licor imaging methods. GAPDH (glyceraldehyde-3-phosphate dehydrogenase) and/or Beta-actin were used as loading controls and were detected using specific antibodies (anti-GAPDH, 1:500 dilution, sc32233, Santa Cruz; anti-Beta-actin, 1:300 dilution, A1978, Sigma). Densitometry was performed using myImageAnalysis version 1.1 (Thermo Fisher Scientific). Western blots were repeated at least three times.
Statistical Analyses
Graphpad Prism 7 was used for the statistical analyses. P values of < 0.05 were considered as statistically significant. Data are expressed as mean ± standard error of the mean (SEM). Two-way ANOVAs were used to determine: 1) differences in task parameters between High-ForceHR versus Low-ForceHR rats using the factors group and time (week of task performance); 2) differences in microCT and histomorphometry outcomes using the factors group and bone (radius versus ulna); 3) differences in mRNA expression levels, and sclerostin and RANKL protein levels that were assayed via ELISA, using the factors group and region (distal versus proximal); and 4) differences in sclerostin and RANKL protein levels that were assayed immunohistochemically, using the factors group and bone (radius versus ulna). One-way ANOVAs were used to determine differences in serum protein levels between the three groups (High-ForceHR, Low-ForceHR and control rats). ANOVAs were followed by Tukey posthoc tests (for one-way ANOVAs) or Sidak posthoc tests (for two-way ANOVAs) to determine differences. Two-tailed t-tests were used when only two groups were compared.
RESULTS
Reach performance across the 18 weeks
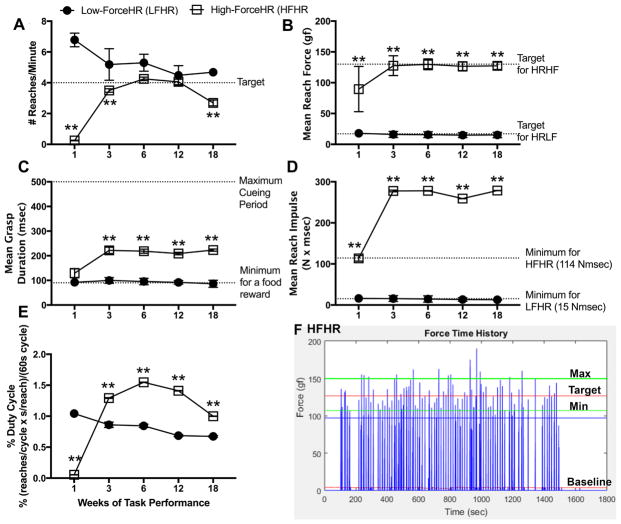
We first investigated the consistency of performance of the operant High-ForceHR and Low-ForceHR tasks by the task rats. Each group learned to perform their respective task appropriately by task week 3 (Fig. 2A–D). The High-ForceHR rats performed the task at or near 4 reaches/min in task weeks 3 through 12, although below target levels in weeks 1 and 18 (Fig. 2A), matching past findings of symptoms of forepaw discomfort in High-ForceHR 18 week rats (Fisher, Zhao et al. 2015). The Low-ForceHR rats had a tendency to overreach, particularly in task week 1, yet neared target levels in weeks 12 and 18. Each task group met their target mean reach force consistently in weeks 3 through 18 (Fig. 2B). The High-ForceHR rats had higher, yet consistent mean grasp durations and reach impulses levels than Low-ForceHR rats, in weeks 3 through 18 (Fig. 2C,D), and worked at a significantly higher percent duty cycle than Low-ForceHR rats in weeks 3–18 (Fig. 2E). The mean % duty cycle of the High-ForceHR rats was 0.98%, and 0.79% for the Low-ForceHR rats calculated from the formula as suggested by Potvin, 2012, for repetitive upper extremity tasks (Potvin 2012) (see methods). During individual sessions, High-ForceHR rats had ranging grasp force levels around their target level, with many reaches within maximum and minimum range levels, but a few reaches above or below required force thresholds, for which they received no food reward (Fig. 2F; Supplemental Fig. 1). Thus, despite the voluntary nature of the task, the rats performed each task fairly consistently, while still maintaining some dynamic properties of pulling and tissue loading. Low-ForceHF rats showed similar dynamic loading within sessions (data not shown).
Figure 2.
Reach parameters in High-ForceHR (HFHR) and Low-ForceHR (LFLR) rats across weeks of task performance (n=12 and 15/gp, respectively). (A) Number of reaches per minute; the target was 4 reaches/minute (dashed line). (B) Mean reach force on the lever bar (grams force, gf). Target for HFHR rats was 130 grams of force (gf); target for LFLR rats was 17 gf. (C) Mean duration of grasping the lever bar (msec) per reach. The minimum duration required for a food reward was 90 msec. (D) Mean Reach Impulse (Nmsec), defined as mean reach force (N) multiplied by grasp duration (msec). (E) Percent Duty Cycle, defined as reaches/cycle multiplied by grasp duration (in seconds (s)) on the lever bar per reach, divided the cycle time (60 s), and then multiplied by 100. (F) Representative graph of force time history of one 30 minute session (1800 seconds) by a High-ForceHR rat, extracted from Force Lever program files using a custom-written MATLAB program. The vertical blue lines indicate a pull on the force lever bar; the horizontal red line indicates the target of 130 gf; the green lines indicate the minimum (110 gf) and maximum (150 gf) thresholds of force. Baseline as indicated. Mean ± SEM is shown; ** p<0.01, compared to LFLR rats.
The Low-ForceHR rats had a higher total number of reaches across the 18 weeks of task performance than High-ForceHR rats, due to the former’s tendency to overreach (Table 1). Yet, the total grasp time and the total volume of lever bar grasping and loading across the 18 weeks (total reaches × total msec of grasping × mean force per grasp) were higher in High-ForceHR rats than in Low-ForceHR rats (Table 1).
Table 1.
Estimated total volume of loading across 18 weeks
| Reach Parameter | HFHR (n=12) Mean ± SEM |
LFHR (n=9) Mean ± SEM |
|---|---|---|
| Total volume of reaches (total reaches) | 19,105 ± 795* | 34,252 ± 3,724 |
| Total grasp time of lever bar (msec) | 3,605 ± 178 ** | 1,676 ± 213 |
| Mean Grasp Force (N) | 1.18 ± 0.08 ** | 0.16 ± 0.01 |
| Total volume of loading (total reaches × total msec grasping × mean force per grasp) | 81,266,234 ± 11,333 ** | 9,187,665 ± 7,938 |
and
: p<0.05 and p<0.01, compared to LFHR
The rats also showed variations in the style of pulling with continued task performance. The Low-ForceHR rats maintained a fairly isometric style of lever pull across weeks of task performance, although the number of digits used to pull might vary (Supplemental Fig. 2A,B). High-ForceHR showed increasing tendencies towards supinated or twisted forelimb and forepaw positions, as show in Supplemental Fig. 2C, and as previously reported (Fisher, Zhao et al. 2015, Bove, Harris et al. 2016).
Distal metaphyseal trabecular bone
We next examined the impact of performance of the tasks for 18 weeks on trabecular bone microarchitecture in the distal metaphyseal region of the radius and ulna using microCT (Table 2). Figure 1C shows representative two-dimensional (2D) and 3D images of each group. Two-way ANOVAs showed several significant differences by group and between the radius and ulna bones. Distal metaphyseal trabeculae in the radius showed reduced % BV/TV and Tb.N in High-ForceHR rats, compared to controls, as well as increased Tb.Sp and degree of anisotropy (DA). In contrast, trabeculae in this same region in Low-ForceHR rats showed increased BV/TV and Tb.Th, and a decrease in DA, compared to controls. Microarchitectural changes induced by the tasks differed in the ulna, with High-ForceHR rats showing decreases in their ulnar metaphyseal trabecular % BV/TV that did not reach significance, compared to controls, although they did have significantly decreased Tb.N and increased Tb.Sp and Tb.Th (the latter an adaptive change that apparently prevented the loss of overall trabecular bone volume in High-ForceHR rats’ ulnas). Low-ForceHR rats’ ulnas showed only an increase in metaphyseal Tb.Th, compared to controls.
Table 2.
MicroCT results for distal metaphyseal trabeculae in radius and ulna in response to 18 weeks of High-Force High Repetition (HFHR) or Low-Force High Repetition (LFHR) task, compared to age-matched control rats (C).
| Radial distal metaphysis | C | HFHR | HFHR/C | LFHR | LFH R/C | Two-Way ANOVAs | |
|---|---|---|---|---|---|---|---|
| Trabecular Bone | n=14 | n=8 Reach Limbs | % Diff a | n=7 Reach Limbs | % Diff a | Radius vs Ulna b | |
| 45.16 ± | Gp p=0.005, Bone p<0.0001 | ||||||
| Bone volume fraction | BV/TV (%) | 37.35 ± 2.26 | 32.30 ± 1.29 * | 86% | 1219 % | ||
| Trabecular number | Tb.N (1/mm) | 5.27 ± 0.23 | 4.58 ± 0.29 * | 87% | 6.08 ± 0.35† | 115 % | Gp p=0.002, Bone p<0.0001 |
| Trabecular thickness | Tb.Th (mm) | 0.07 ± 0.001 | 0.07 ± 0.001 | 100% | 0.08 ± 0.004 * | 114 % | Interaction p=0.04 |
| Trabecular separation | Tb.Sp (mm) | 0.12 ± 0.006 | 0.15 ± 0.01 * | 125% | 0.12 ± 0.009 | 100 % | n.s. |
| Degree of anisotropy, 0=isotropic | DA | 0.47 ± 0.03 | 0.56 ± 0.03 * | 119% | 0.34 ± 0.04 **,†† | 72% | n.s. |
|
| |||||||
| Ulnar distal metaphysis | C | HFHR | HFHR/C | LFHR | LFH R/C | ||
| Trabecular Bone | n=8 | n=7 Reach Limbs | % Diff a | n=5 Reach Limbs | % Diff a | ||
|
| |||||||
| Bone volume fraction | BV/TV (%) | 27.62 ± 2.65 | 24.23 ± 1.57 | 88% | 29.1 ± | 104 % | |
| Trabecular number | Tb.N (1/mm) | 3.76 ± 0.30 | 2.50 ± 0.18 ** | 67% | 3.34 ± 0.42 | 88% | |
| Trabecular thickness | Tb.Th (mm) | 0.08 ± 0.001 | 0.09 ± 0.002** | 112% | 0.09 ± 0.002** | 112 % | |
| Trabecular separation | Tb.Sp (mm) | 0.18 ± 0.01 | 0.24 ± 0.02* | 133% | 0.22 ± 0.02 | 122 % | |
| Degree of anisotropy | DA | 0.49 ± 0.02 | 0.46 ± 0.06 | 94% | 0.45 ± 0.06 | 92% | |
and
p < 0.05 and p<0.01, compared to age-matched C rats;
and
p < 0.05 and p<0.01, compared to HFHR rats
% Difference between means; means ± SEM shown.
Two way ANOVA results using the factors group (gp) and bone. n.s. = not significant.
Mid-diaphyseal cortical bone
We then examined the mid-diaphyseal cortical bone of the radius and ulna using microCT (Table 3). Two-way ANOVAs showed several significant differences by group and between the radius and ulna bones. Specifically, analyses of mid-diaphyseal cortical bone of the radius in High-ForceHR rats revealed only a small increase in cortical area fraction, compared to controls. In contrast, the Low-ForceHR rats showed increased total cross-sectional thickness and cortical area, and decreased cortical area fraction in the mid-diaphysis of the radius, compared to controls. In further contrast, there were no significant changes in the ulna’s mid-diaphyseal cortical bone in High-ForceHR rats, and Low-ForceHR rats only showed an increased total cross-sectional area in this same region of the ulna, compared to controls.
Table 3.
MicroCT results for mid-diaphyseal cortical bone in radius and ulna in response to 18 weeks of High-Force High Repetition (HFHR) or Low-Force High Repetition (LFHR) task, compared to age-matched control rats (C).
| Radial mid-diaphyseal | C | HFHR | HFHR/C | LFH R | LFHR/C | Two-Way ANOVA s | |
|---|---|---|---|---|---|---|---|
| Cortical Bone | n=14 | n=8 Reach Limbs | % | n=6 Reach Limb s | % Diff a | Radius vs Ulna b | |
| Total cross-sectional area | Tt.Ar (mm2) | 1.56 ± 0.06 | 1.50 ± 0.03 | 96% | 1.92 ± 0.002 **,†† | 123% | Gp p=0.009, Bone p<0.0001 |
| Cortical bone area | Ct.Ar (mm2) | 1.46 ± 0.06 | 1.45 ± 0.02 | 99% | 1.76 ± 0.02 **,†† | 121% | Gp p=0.009, Bone p<0.0001 |
| Cortical area fraction | Ct.Ar/Tt.Ar (%) | 94.86 ± 1.21 | 96.66 ± 0.30 * | 102% | 91.62 ± 0.83 *,†† | 96% | Interaction p=0.03 |
| Marrow area | Ma.Ar (mm2) | 0.10 ± 0.02 | 0.12 ± 0.45 | 120% | 0.15 ± 0.02 | 150% | n.s. |
| Mean cortical thickness | Ct.Th (mm) | 0.17 ± 0.02 | 0.17 ± 0.01 | 100% | 0.18 ± 0.03 | 106% | n.s. |
| Closed Cortical Porosity | Po(cl) (%) | 0.83 ± 0.17 | 0.69 ± 0.14 | 83% | 0.70 ± 0.19 | 84% | n.s. |
|
| |||||||
| Ulnar mid-diaphyseal | C | HFHR | HFHR/C | LFHR n=5 | LFHR/C % Diff | ||
| Cortical Bone | n=7 | n=8 Reach Limbs | % | Reach Limbs | a | ||
|
| |||||||
| Total cross-sectional area | Tt.Ar (mm2) | 1.19 ± 0.04 | 1.27 ± 0.13 | 114% | 1.45 ± 0.07 * | 122% | |
| Cortical bone area | Ct.Ar (mm2) | 1.19 ± 0.04 | 1.28 ± 0.13 | 108% | 1.38 ± 0.04 | 116% | |
| Cortical area fraction | Ct.Ar/Tt.Ar (%) | 99.81 ± 0.20 | 99.72 ± 0.71 | 100% | 99.78 ± 0.30 | 96% | |
| Marrow area | Ma.Ar (mm2) | 0.002 ± 0.0001 | 0.003 ± 0.0002 | 150% | 0.003 ± 0.000 | 150% | |
| Mean cortical thickness | Ct.Th (mm) | 0.17 ± 0.01 | 0.18 ± 0.005 | 106% | 50.19 ± 0.004 | 112% | |
| Cortical Porosity | Ct.Po (%) | 0.34 ± 0.05 | 0.39 ± 0.12 | 114% | 0.33 ± 0.05 | 97% | |
and
p < 0.05 and p<0.01, compared to age-matched C rats;
and
: p < 0.05 and p<0.01, compared to HFHR rats
% Difference between means; means ± SEM shown.
Two way ANOVA results using the factors group (gp) and bone. n.s. = not significant.
Histomorphometry of distal metaphyseal trabeculae
Static histomorphometry of the distal metaphyseal trabeculae showed several significant differences by group and between the radius and ulna bones (Table 4). Specifically, in this region of the radius, there were decreased numbers of osteoblasts per bone surface (N.Ob/BS) in High-ForceHR rats, yet increased N.Ob/BS and osteoid volume per bone volume (% OV/BV) in Low-ForceHR rats, compared to controls. The percent osteoid surface (% OS/BS) was also increased in Low-ForceHR rats, compared to High-ForceHR rats. Also in this region of the radius, the number of osteoclasts per bone perimeter (N.Oc/B.Pm) and percent osteoclast surface to total bone surface (Oc.S/BS) were considerably increased in High-ForceHR rats, compared to control and Low-ForceHR rats. Most of these attributes were unchanged in this same region of the ulna of both High-ForceHR and Low-ForceHR rats, compared to control rats, with the exception of increases in both osteoblast and osteoclast numbers in High-ForceHR rats. Void areas were also assessed in all stained bone sections; no differences were seen when comparing any group or bone.
Table 4.
Static histomorphometry of metaphyseal trabecular bone in radius and ulna in response to 18 weeks of High-Force High Repetition (HFHR) or Low-Force High Repetition (LFHR) task, compared to age-matched control rats (C).
| Radial distal metaphysis | C | HFHR | HFHR/C | LFHR | LFH R/C | Two-Way ANOVAs | |
|---|---|---|---|---|---|---|---|
| Trabecular Bone | n=7 | n=8 Reach Limbs | % | n=6 Reach Limbs | % Diff a | Radius vs Ulna b | |
| Osteoblast surface | N.Ob/BS, ( mm−1) | 2.25 ± 0.66 | 0.99 ± 0.19 * | 44% | 4.63 ± 0.64 *,†† | 206 % | Interaction p=0.02 |
| Osteoid volume | OV/BV (%) | 0.007 ± 0.002 | 0.003 ± 0.0007 | 43% | 0.05 ± 0.006 *,† | 714 % | Group p=0.049 |
| Osteoid surface | OS/BS (%) | 0.04 ± 0.01 | 0.02 ± 0.005 | 50% | 0.05 ± 0.009 † | 96% | n.s. |
| Osteoclast Number | N.Oc/BS ( mm−1) | 0.33 ± 0.05 | 1.62 ± 0.21** | 490% | 0.62 ± 0.25 †† | 188 % | Group p<0.0001 |
| Osteoclast Surface | Oc.S/BS (%) | 0.02 ± 0.005 | 0.14 ± 0.02** | 700% | 0.04 ± 0.02 †† | 200 % | Interaction p=0.04 |
|
| |||||||
| Ulnar distal metaphysis | C | HFHR | HFHR/C | LFHR | LFH R/C | ||
| Trabecular Bone | n=5 | n=5 Reach Limbs | % | n=5 Reach Limbs | % Diff a | ||
|
| |||||||
| Osteoblast surface | N.Ob/BS, ( mm−1) | 3.04 ± 0.54 | 3.80 ± 0.95 * | 125% | 3.26 ± 0.90 | 107 % | |
| Osteoid volume | OV/BV (%) | 0.007 ± 0.001 | 0.004 ± 0.0007 | 57% | 0.005 ± 0.001 | 72% | |
| Osteoid surface | OS/BS (%) | 0.05 ± 0.007 | 0.05 ± 0.01 | 100% | 0.05 ± 0.01 | 100 % | |
| Osteoclast Number | N.Oc/BS ( mm−1) | 0.21 ± 0.17 | 1.16 ± 0.28 * | 552% | 0.36 ± 0.12 | 171 % | |
| Osteoclast surface | Oc.S/BS (%) | 0.02 ± 0.006 | 0.06 ± 0.01 | 300% | 0.03 ± 0.02 | 150 % | |
and
p < 0.05 and p<0.01, compared to age-matched C rats;
and
: p < 0.05 and p<0.01, compared to HFHR rats
% Difference between means; means ± SEM shown.
Two way ANOVA results using the factors group (gp) and bone. n.s. = not significant.
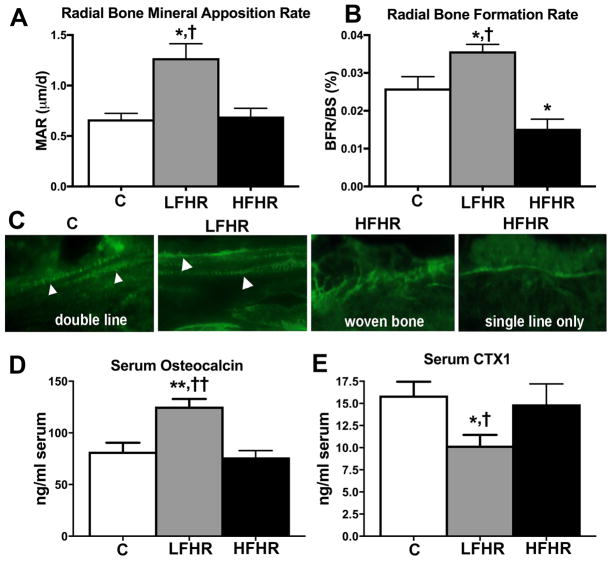
Based on the above, dynamic histomorphometry of distal metaphyseal radial trabeculae was examined and showed a reduced bone formation rate (BFR) in High-ForceHR rats, compared to controls, yet increases in Low-ForceHR rats (as well an increased mineral apposition rate, MAR), compared to both control and High-ForceHR rats (Fig. 3A,B). The distal radial trabeculae of control rats showed the presence of double-labeled calcein lines, as did Low-ForceHR rats (Fig. 3C). In contrast, distal radial trabeculae of High-ForceHR rats showed more sites with woven bone and single-labeled calcein lines (Fig. 3G).
Figure 3.
Dynamic histomorphometry in distal radial metaphyseal trabeculae (n=6–12/gp) and serum biomarkers of bone formation and turnover (n=8–12/gp). (A) Mineral apposition rate (MAR). (B) Bone formation rate (% BFR/BS). (C) Calcein incorporation in C and LFLR rats was observed as double-labeled lines (arrowheads) in distal radial trabeculae, while calcein incorporation in HFHR rats was observed only as woven bone or single-labeled lines. (D) Serum levels of osteocalcin (a biomarker of bone formation). (E) Serum levels of CTX1 (a biomarker of bone degradation). Magnification 400X. Mean + SEM shown. * and **: p<0.05 and p<0.01, compared to control rats; † and ††: p<0.05 and p<0.01, compared to High-ForceHR rats.
Serum osteocalcin, a marker of bone formation throughout the body, was increased only in Low-ForceHR rats compared to the other groups (Fig. 3D). Serum CTX1, a biomarker of bone degradation, was altered only in Low-ForceHR rats (decreased), compared to control and High-ForceHR rats (Fig. 3E).
Apoptosis in distal metaphyseal trabeculae of the radius
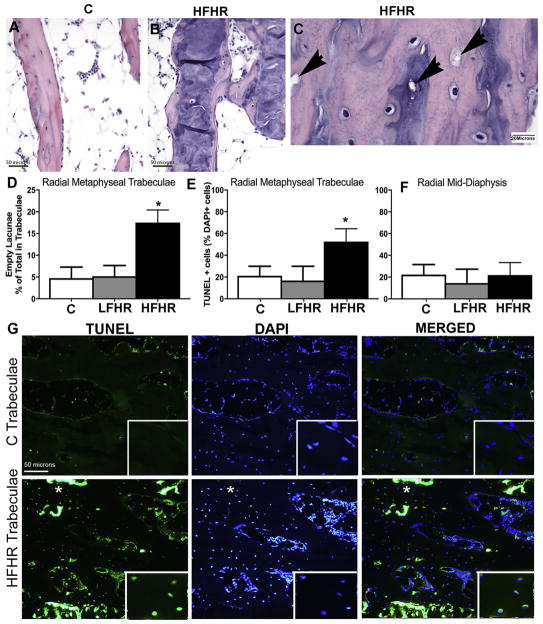
When examining radial bone sections, we noticed that High-ForceHR rats had more empty osteocyte lacunae in distal metaphyseal trabeculae than controls (Fig. 4A–C). Histomorphometry confirmed this finding (Fig. 4D). In contrast, the mid-diaphyseal radial cortical bone did not show such increases (See Figure 6H for an example), nor did any region of the radius in Low-ForceHR rats, compared to controls (Fig. 4D; mid-diaphyseal data not shown). Both regions of the ulna in each group also lacked this increase in empty osteocyte lacunae (data not shown).
Figure 4.
Increased empty lacunae and TUNEL stained cells in distal radial metaphyseal trabeculae (n=4–7/gp). (A–C) Representative distal radial trabeculae stained with H&E showing disrupted organization and increased empty lacunae (arrows in B and C) in High-ForceHR (HFHR) rats, relative to a Control rat (C). (D) Percentage of empty lacunae to all lacunae. (E and F) Percentage of TUNEL positive cells as a ratio of all DAPI stained cells in radial metaphyseal trabeculae and mid-diaphyseal cortical bone. (G–L) TUNEL stained cells (green), DAPI stained cells (blue) and merged representative images in C and HFHR rats. Asterisks indicate an area in panels J–L that is enlarged in the insets, showing examples of cells double-labeled with both TUNEL and DAPI. Mean ± SEM shown. *: p<0.05, compared to C rats.
Figure 6.
Sclerostin expression in forelimb bones. (A) Sclerostin protein levels in forelimb bones assayed using ELISA (n=4–6/gp). Distal region included radial and ulnar metaphyses and epiphyses; mid-diaphyseal region included the radial and ulnar diaphyses. (B) Quantification of sclerostin immunoexpression in distal metaphyseal trabeculae of the radius and ulna (n=4–7/gp). (C and D) Representative low power images of sclerostin immunostaining (brown) in osteocytes and matrix (asterisks) of a control (C) and a High-ForceHR (HFHR) rat, respectively. (E and F) Representative higher power images showing sclerostin immunostaining in a C rat’s trabecular and mid-diaphyseal cortical bone, respectively. (G and H) Representative higher power images showing sclerostin immunostaining in a HFHR rat’s trabecular and mid-diaphyseal cortical bone, respectively. Small arrows indicate empty lacunae observed in HFHR 18W rats’ trabecular bone. Asterisks indicate an area in panel G enlarged in the inset. Bones in panels C–H were counterstained lightly with H&E. Mean + SEM shown. * p<0.05, compared to C rats.
To determine if the High-ForceHR task induced osteocyte apoptosis, we performed TUNEL staining and quantification. High-ForceHR had increased numbers of TUNEL-positive osteocytes in distal metaphyseal trabeculae of the radius, compared to controls (Fig. 4E). Such increases were neither observed in Low-ForceHR rat bones, nor in the mid-diaphysis of either group of task rats (Fig. 4F). Representative images of TUNEL stained distal radial trabeculae are shown in Figure 4G–L.
Microdamage analysis
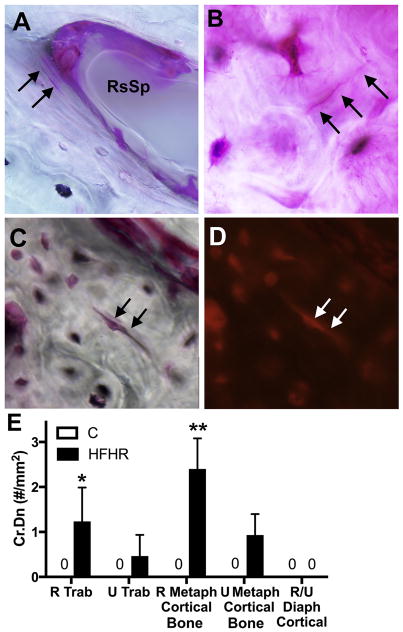
Since apoptosis of osteocytes has been associated with presence of microdamage (Mabilleau, Mieczkowska et al. 2010), we next assayed radial and ulnar bones of High-ForceHR and control rats for presence of linear microcracks (Fig. 5A–E). Linear microcracks were visible in distal metaphyseal trabeculae (some in association with resorptive spaces; Fig. 5A), and distal metaphyseal interstitial cortical bone regions of both bones in High-ForceHR rats (Fig. 5B–D). Quantification of Cr.Dn revealed higher numbers in metaphyseal trabecular and metaphyseal cortical bone regions of the radius in High-ForceHR rats, compared to controls (Fig. 5E). The number of microcracks in the ulna did not reach significance, and no microcracks were found in mid-diaphyseal bone regions of either bone in High-ForceHR or control rats.
Figure 5.
Microcracks in radius bones. (A) Small linear microcracks in a trabeculae adjacent to a resorptive space (RsSp). (B–D) Examples of microcracks in interstitial bone of the metaphyseal radius cortical shell. Panel D shows same crack as in panel C using orange fluorescence. (E) Crack density (Cr.Dn) quantification, n=3/gp. 400× magnification. Mean ± SEM shown. * and **: p<0.05 and 0.01, compared to same region in control rats.
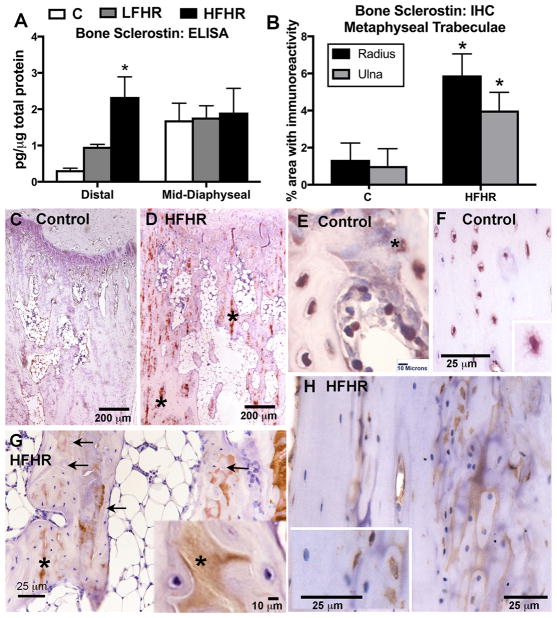
Sclerostin protein levels
Since apoptosing osteocytes have been shown to increase their release of sclerostin (Mabilleau, Mieczkowska et al. 2010), we next examined sclerostin protein levels in distal versus mid-diaphyseal regions of the radius and ulna (distal epiphyseal and metaphyseal regions of the radius and ulna were homogenized together, as were diaphyseal regions of the radius and ulna). ELISA revealed increased levels of sclerostin protein in distal regions of these forelimb bones in High-ForceHR rats, compared to control rats, but not in mid-diaphyseal regions, and not in either region in Low-ForceHR rats (Fig 6A). Immunohistochemistry confirmed the increase in sclerostin protein in distal regions of the radius and ulna of High-ForceHR rats, compared to control rats, although a slightly higher level in the distal radius than distal ulna of High-ForceHR rats (Fig. 6B). Sclerostin immunostaining was localized to osteocytes in trabecular and cortical bone regions of control rats (in both the radius and ulna), yet there was only low to no immunoexpression in the bone matrix in these regions in control rat bones (Fig. 6C,E,F). Small rounded cells adjacent to the trabecular bone also showed immunostaining for sclerostin (Fig. 6E); closer examination showed that these cells were not osteoblasts or osteoclasts. In contrast, High-ForceHR rats showed clear increases in sclerostin immunostaining within radial bone matrices (Fig. 6D,G,H). The High-ForceHR rats typically had no stained osteocytes in distal metaphyseal trabeculae of the radius (Fig. 6D,G), yet a mix of stained and unstained osteocytes in mid-diaphyseal cortical bone of the radius (Fig. 6H). Bone lining cells also showed increased sclerostin immunostaining in High-Force HR rats (Fig. 6H). Serum levels of sclerostin showed no significant differences (p=0.10) between the groups (FRC: 348 ± 22.5, n=15; High-ForceHR: 285 ± 16.51, n=9; Low-ForceHR: 386 ± 36.36, n=9).
RANKL levels
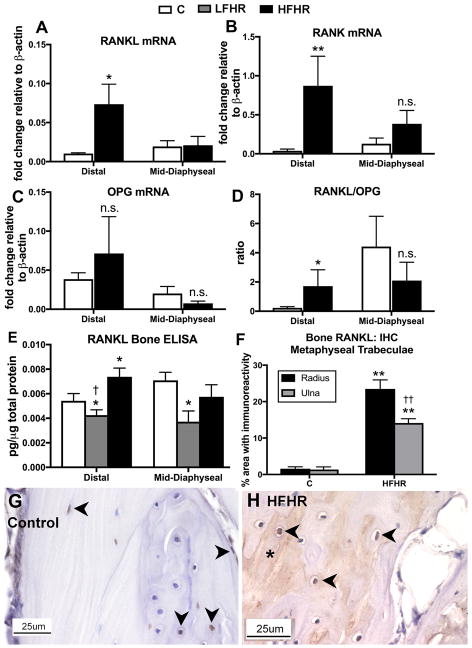
Since RANKL is also a signal released by apoptotic osteocytes following loading-induced microdamage (Kennedy, Laudier et al. 2014), and known to promote osteoclast activity (Wijenayaka, Kogawa et al. 2011), we next examined RANKL levels using qPCR and ELISA methods (for each subcohort of bones, distal epiphyseal and metaphyseal regions of the radius and ulna were homogenized together, as were diaphyseal regions of the radius and ulna). Increased mRNA expression of RANKL was observed in distal radius and ulna regions of High-ForceHR rats, compared to controls (Fig. 7A; Low-ForceHR bones were not examined using qPCR), as was mRNA expression of its receptor, RANK (Fig 7B). Osteoprotegerin (OPG) mRNA levels did not differ significantly by region or group (Fig. 7C). However, the ratio of RANKL mRNA expression to OPG mRNA expression increased in distal regions of the radius and ulna in High-ForceHR rats, compared to controls (Fig. 7D). No significant increases in mRNA expression of any analyte were observed in the mid-diaphysis regions (Fig. 7A–D).
Figure 7.
RANKL, RANK and OPG analyses. (A–C) qPCR quantification of RANKL, RANK and OPG mRNA in forelimb bones (bones and regions as described in Figure 7 legend). Fold changes compared to housekeeping gene beta-actin are reported. (D) The ratio of RANKL mRNA levels to OPG mRNA levels. (E and F) RANKL protein levels in forelimb bones, assayed using ELISA and immunohistochemical methods (n=4–7/gp). (G and H) Representative images showing RANKL immunostaining (brown staining) in distal radial trabeculae of control (C) and High-ForceHR (HFHR) rats, respectively. Arrowheads indicate RANKL immunostained cells; asterisk indicates RANKL immunoreactive matrix. Sections were counterstained lightly with hematoxylin. Mean + SEM shown. Symbols as in Figure 3’s legend; n.s. = not significant.
Increased RANKL mRNA expression was matched by increased RANKL protein levels in the distal radius and ulna regions of High-ForceHR, compared to control rats, but not in the mid-diaphyseal regions (Fig. 7E). In contrast, RANKL protein levels were decreased in both of the radius and ulna in Low-ForceHR rats, compared to controls (Fig. 7E), and distally compared to High-ForceHR rats. Quantification of RANKL immunoexpression in distal metaphyseal trabeculae showed increased levels of RANKL in High-ForceHR rat radial bones, compared to control rat radii and High-ForceHR rat ulna bones, although these latter bones also showed increased RANKL immunoexpression, compared to control rat ulna bones (Fig. 7G). Immunohistochemistry was also used to identify RANKL producing cells and location and showed RANKL immunoexpression in osteocytes and bone lining cells, but not in osteoclasts, of controls (Fig. 7G). In High-ForceHR rats, the relative number of osteocytes expressing RANKL appeared higher than in controls, and the bone matrix of the distal radial trabeculae showed increased RANKL immunostaining (Fig. 7H). Matrix RANKL staining was not present in control rats (Fig. 7G) or Low-ForceHR rats (data not shown). Serum levels of RANKL showed no significant differences (p=0.15) between the groups (FRC: 10.26 ± 2.63, n=8; High-ForceHR: 16.61 ± 5.18, n=9; Low-ForceHR: 14.43 ± 1.96, n=12).
Investigation of other possible contributors of observed bone differences
Radius and ulna bone levels of osteoactivin, a bone anabolic growth factor (Abdelmagid, Barbe et al. 2007), were assayed using ELISA and showed decreased levels (p=0.02) in High-ForceHR rats (0.14 ± 0.001, n=7), compared to controls (0.19 ± 0.01, n=7) Since osteoactivin is also considered a potential “myometabokine” (a substance secreted by skeletal muscles that effects metabolism in other tissues including bone) (Tagliaferri, Wittrant et al. 2015), we also examined its levels in flexor muscles and flexor digitorum tendons. Using ELISA, we observed decreased levels (p=0.03) in flexor forearm muscles (flexor digitorum, flexor carpi ulnaris and flexor carpi radialis muscles combined) of High-ForceHR rats (0.18 ± 0.02, n=6), compared to controls (0.26 ± 0.02, n=5). No group differences were observed in the flexor digitorum tendons (0.13 ± 0.009 in controls, versus 0.14 ± 0.006 in High-ForceHR rats, p=0.48).
We also examined TNFalpha protein levels in distal regions of the radius and ulna (homogenized together) since it can stimulate osteoclastogenesis (Guo, Yamashita et al. 2008) and may serve as a mediator of increased sclerostin expression (Kim, Bae et al. 2012). We have also observed increased TNFalpha levels in 6- and 12-week High-ForceHR rat forelimb bones in association with increased osteoclast numbers (Rani, Barbe et al. 2010, Barbe, Gallagher et al. 2013). We observed no differences in TNFalpha levels in distal regions of control versus 18-week High-ForceHR rat radial and ulnar bones, using Western blot analysis (Supplemental Fig. 3), matching prior results of resolution of inflammatory responses in forearm muscles and serum of 18-week High-ForceHR rats (Fisher, Zhao et al. 2015).
To make sure that muscle atrophy was not contributing to bone catabolism, we examined for changes in the cross-sectional area of the flexor digitorum muscle, a key muscle involved in performing the High-ForceHR task and therefore bone loading. The cross-sectional area of the flexor digitorum muscle was 50.84 ± 3.12 mm2 (mean ± SEM) in controls (n=6), 49.73 ± 2.10 mm2 in Low-ForceHR rats (n=12), and 41.99 ± 2.76 mm2 in High-ForceHR rats (n=6). Although being lower in High-ForceHR rats, these differences were not significant.
Lastly, we tracked body weight across time and investigated serum estrogen levels at the time of euthanasia. No significant differences were observed between the groups, matching past findings for body weights and estrogen levels for 12- and 18-week Low-ForceHR rats, and 12-week High-ForceHR rats (Gao, Fisher et al. 2013, Jain, Barr-Gillespie et al. 2014, Massicotte, Frara et al. 2015).
Thus, weight changes, differences in estrogen levels, potential muscle atrophy, or inflammatory responses at the time of tissue collection were ruled out as possible contributors to the observed bone changes. However, muscle changes in osteoactivin levels and past inflammatory changes (Rani, Barbe et al. 2010, Barbe, Gallagher et al. 2013) may be potential contributors.
DISCUSSION
This is the first study, to our knowledge, to show clear support of the Damage Repair Theory response in trabecular bone when loaded at high force levels, rather than just positive adaptive changes in response to prolonged muscle contraction-induced loading. Prolonged high force loading was a key factor driving radial bone microarchitecture and cellular responses. Rats performing a High-ForceHR task for 18 weeks showed several catabolic indices trabeculae in their distal radial metaphyses, including decreased trabecular bone volume and osteoblasts, increased osteoclasts, microcracks and osteocyte apoptosis, as well as increased sclerostin and RANKL protein levels, compared to control rats. The mid-diaphyseal cortical bone of the radius showed few microarchitecture differences in High-ForceHR rats, compared to control rats, and unchanged apoptosis, sclerostin and RANKL protein levels. In contrast, the 18-week Low-ForceHR task induced many indices of positive adaptation in both distal metaphyseal trabeculae and mid-diaphyseal cortical bone of the radius, as well as decreased RANKL levels, and no apoptosis or changes in sclerostin levels, compared to control rats. Interestingly, in general, changes in the ulna bone of each task group were significantly less than in the radius, indicating that this reaching and grasping of a lever bar task results in more loading of the radial bone than the ulna. Thus, prolonged performance of an upper extremity reaching and grasping task is loading-, region-, and bone-dependent, as discussed further below.
In each task group, we observed small variations in the within session force-time history that are plausible reasons for the continued response of trabecular bone to either task since dynamic loads are thought to determine the osteogenic response (Avin, Bloomfield et al. 2015). We also observed that both task groups were able to maintain their target maximum voluntary pulling force and reach impulse levels in task weeks 3–18, meaning that each group’s forelimb bones were loaded fairly consistently across time, according to their target requirements. Thus, as intended, the High-ForceHR rats maintained higher reach force, reach impulse levels and duty cycles than Low-ForceHR rats (despite the higher reaches/minute in the latter group).
Duty cycle is the frequency of effort duration (percent time) an individual worker is engaged in a repetitive task (Ciriello, Snook et al. 2001, Potvin 2012, Potvin 2012). Demands of a particular task are specific with regards to required grasp, load distributions on tissues, force requirements, and so on (Potvin, Calder et al. 2006). Maximum acceptable efforts (MAE) and maximum acceptable forces (MAF) of a job task on tissues are better predicted by force and duty cycle, than force and repetition, although all three are important (Potvin 2012). If we first calculate the MAE for our two tasks, performed voluntarily by rats according to the expectations for psychophysical methodology (Snook and Irvine 1968), using an equation suggested by Potvin (MAE = 1 − DC0.24) (Potvin 2012), we can predict that the MAE for High-ForceHR rats is 0.67, and 0.69 for Low-ForceHR rats. Calculation of the MAF by multiplying MAE by the rats’ mean maximum voluntary force (2.45N; see methods for determination) (Potvin 2012), shows that the predicted MAF is 1.64N and 1.69N for the High-ForceHR and Low-ForceHR tasks, respectively. The target grasp force of the High-ForceHR rats was 50% of their maximum voluntary force (1.27N), easily below the predicted MAF. Yet, they developed bone microcracks and small muscle tears (reported previously (Fisher, Zhao et al. 2015)). We suggest that these equations over estimate the MAF on these tissues by 1.3 fold (1.64/1.27). These equations were developed from 69 short-term psychophysical data tasks (each < 4 weeks in duration) and using physiological data from 6 studies demonstrating muscle fatigue after only 1 hour (Potvin 2012). As acknowledged by their creator, corrections are needed to accommodate longer endurance times, changes in worker performance, and very low duty cycles as limited data was available for validation. Since we observed increased radial trabecular bone volume in Low-ForceHR rats in this 18 week study, and previously in young adult rats performing at 15% of their maximum voluntary force and a similar duty cycle of 0.79% for 12 weeks (Barbe, Gallagher et al. 2013, Massicotte, Frara et al. 2015), perhaps 15% is the MAF for prolonged performance of this task when performed at 4 reaches/minute. Lowering force levels from 50% to 15% of maximum in task weeks 5–12, as a type of ergonomic intervention, rescues the loss of radial trabecular bone volume (Barbe, Jain et al. 2015), further supporting high force as a key risk factor. Rest allowances, e.g., lowering duty cycles, may increase MAF and enhance tissue recovery (Rohmert 1973, Potvin 2012). Previously, we reduced the duty cycle to 0.66% by lowering the repetition rate to 2 reaches/minute, yet maintained the same high force requirement (50% of maximum) in order to examine the effects of a 12-week High-Force Low-Repetition task; no significant losses were observed in radial trabecular bone volume, yet also no gains (Barbe, Gallagher et al. 2013). Increasing periods of complete relaxation between work cycles can also reduce health risks (Sjogaard, Jorgensen et al. 2000, Sjogaard, Lundberg et al. 2000). For the tasks reported here, periods of complete relaxation included the 1.5 hours between the 30-minute work sessions, and the time between the Monday, Wednesday and Friday workdays. We are currently exploring the effectiveness of longer rest periods in our model.
The observed reductions in distal metaphyseal trabecular BV/TV and Tb.N, increased Tb.Sp, and woven bone seen in radii of 18-week High-ForceHR rats are indicators of reduced trabecular bone volume and quality in this bone region (Burr, Forwood et al. 1997, Turner 2002, Jiang, Zhao et al. 2005, Borah, Dufresne et al. 2010). These changes could make the trabeculae at this site more brittle, increasing fracture risk (Turner 2002, Jiang, Zhao et al. 2005). The increase in degree of anisotropy (DA) in distal metaphyseal trabeculae of High-ForceHR radial bones may have also contributed to the microcracks and osteocyte apoptosis in this bone region, since a disorganized trabecular pattern would increase stress in the trabeculae (Ding, Odgaard et al. 2003). These results are consistent with prior studies from our lab in which 12-week High-ForceHR rats showed similar losses in bone volume in distal radial trabeculae, although empty osteocyte lacunae were not observed in those shorter studies (Barbe, Gallagher et al. 2013, Jain, Barr-Gillespie et al. 2014, Barbe, Jain et al. 2015). The loss of trabecular bone volume in the distal metaphysis of the radius in High-ForceHR rats indicates that a pure mechanostat theory in which bones adapt to mechanical loads (Frost 1994, Klein-Nulend, Bacabac et al. 2012) does not apply to prolonged High-ForceHR loading of trabeculae. The various rest periods (discussed above) were apparently not often enough or long enough to allow for sufficient removal of the damaged bone and repair. This would be consistent with the Damage Repair Theory suggested previously for cortical bone (Prendergast and Taylor 1994, Li, Li et al. 2007) and with the Fatigue-Failure Theory for musculoskeletal disorder injuries (Barbe, Gallagher et al. 2013, Gallagher and Schall 2017).
In contrast to the distal trabeculae, the mid-diaphyseal cortical bones of both the radius and ulna remained relatively unchanged in High-ForceHR rats. There were no significant changes of mediators of either bone formation or resorption, and no induction of osteocyte apoptosis, in the mid-diaphyses. This finding of fewer changes in mid-diaphyseal regions of the radius and ulna than in their distal regions is consistent with prior findings from young adult rats performing either a negligible force repetitive reaching task for 8 weeks or the Low-ForceHR task for 12 weeks (Barr, Safadi et al. 2003, Massicotte, Frara et al. 2015). Our findings also match those by Tu et al showing the cortical bone exhibits lower remodeling rates than trabecular bone (Tu, Rhee et al. 2012). It is also likely the interosseous membrane, which is attached to both the radius and ulna, equalized the peak strain magnitudes by the mid-diaphysis level, as shown previously using a mouse radius-ulna compression loading model (Thiagarajan, Lu et al. 2014).
Our data differs from those of ulna end-loading compression models, in which the ulnar bone shows more involvement than the radius and significant changes in the mid-diaphysis region (Hsieh and Silva 2002, Kotha, Hsieh et al. 2004, Matsuzaki, Wohl et al. 2007, Thiagarajan, Lu et al. 2014). We postulate that this is because our model is an operant reach and grasping model in which the forelimb is used as a freelimb. The increased degradative changes in distal metaphyseal trabeculae of the radius versus ulna of High-ForceHR rats is likely a result of repeated high force compressive forces from muscles involved in performing this lever pulling task (Laurent, Dubois et al. 2016). Enthesis attachments to bones (both fibrocartilagenous and fibrous muscle-to-tendon-to-bone insertions) are known to drive changes in the underlying bone, including trabecular bone (Benjamin, Toumi et al. 2006, Benjamin, Toumi et al. 2007, Apostolakos, Durant et al. 2014). In our model, such entheses would include the brachioradialis muscle inserting on the distal epiphyseal radius, the pronator quadratus muscle inserting on the distal metaphyseal radius, and the pronator teres and supinator muscles inserting on proximal-to-mid regions of the radius. The ulna bone lacks such muscle insertion entheses, which may also contribute to fewer changes in the ulna than the radius. A number of static and dynamic loading experiments using human wrists/forearms in cadaveric studies, or models, have examined force distributions across the wrist joint to forelimb bones using sensors (Hara, Horii et al. 1992, Werner, Palmer et al. 1996, Rikli, Honigmann et al. 2007, Fischer, Johnson et al. 2011, Erhart, Schmoelz et al. 2012, Johnson, McIff et al. 2014, Marquez-Florez, Vergara-Amador et al. 2015). Results show that the wrist is extended and ulnarly deviated when in a neutral position, and that applied muscle-tendon loading while in this neutral position leads to force transmissions across carpal joints to the radius than through the triangular cartilage to the ulna (Erhart et al reported a ratio of force transmission of 77:23, radius versus ulna, for example) (Hara, Horii et al. 1992, Rikli, Honigmann et al. 2007, Erhart, Schmoelz et al. 2012,
Johnson, McIff et al. 2014, Marquez-Florez, Vergara-Amador et al. 2015). During power gripping, there is even a greater ulnar deviation that rotates the proximal row of carpal bones dorsally, and extends the lunate and translates it radially, so that the force-transmission increases even further to the radius and decreases to the ulna (Hara, Horii et al. 1992, Erhart, Schmoelz et al. 2012). Both Low-ForceHR and High-ForceHR rats used isometric-like pulls (Barbe, Gallagher et al. 2013), although increasing incidence supinated/twisted power grip pulls in the High-ForceHR rats with continued task performance (See Supplemental Fig. 2 and previously reported results for High-ForceHR rats (Fisher, Zhao et al. 2015, Bove, Harris et al. 2016)). These findings combined also help explain the greater changes in radial versus ulnar architecture in the 18 week High-ForceHR rats.
Findings in the Low-ForceHR rats differ considerably from those in High-ForceHR rats. The increased anabolic indices in metaphyseal trabecular and mid-diaphyseal cortical bone in 18-week Low-ForceHR rats is consistent with studies showing that physical activity increases bone adaptation in response to loading (Frost 1994, Laurent, Dubois et al. 2016). Furthermore, the continued osteogenic response of the radius to Low-ForceHR loading is consistent with current evidence in human bones suggesting that there is no “lazy zone”, and that bone can continue to adapt to imposed loads (Christen, Ito et al. 2014). Note though, that these results pertain only to young adult rats. Aged rats performing this same Low-ForceHR for 12 weeks developed many signs of trabecular and cortical bone degradation that appeared to be due to increased systemic and tissue inflammatory cytokine responses occurring as consequences of both aging and continued loading, as well as reduced osteoblastic responses (Xin, Harris et al. 2011, Massicotte, Frara et al. 2015).
To return to the responses in High-ForceHR rats, increased empty lacunae were observed, as were increased numbers of TUNEL-positive osteocytes in distal radial bone trabeculae, compared to control rats. Findings of microcracks and apoptotic osteocytes in High-ForceHR metaphyseal trabeculae are suggestive of loading-induced microdamage in the bone matrix of the trabeculae through which the osteocytes extend their canaliculi (Wenzel, Schaffler et al. 1996, Colopy, Benz-Dean et al. 2004, Noble 2005, Robling, Bellido et al. 2006, Kennedy, Herman et al. 2012). Osteocyte apoptosis after microcrack damage is known to enhance the release of sclerostin and RANKL (Mabilleau, Mieczkowska et al. 2010), which then increase bone catabolism and osteoclast activity (Al-Dujaili, Lau et al. 2011, Hoshi, Kawaki et al. 2014).
Sclerostin production typically decreases with physiological bone loading and increases with bone unloading (Robling, Niziolek et al. 2008, Papanicolaou, Phipps et al. 2009, Spatz, Ellman et al. 2012, Tu, Rhee et al. 2012, Nguyen, Tang et al. 2013). Although no decrease in sclerostin levels was seen in either task group, only prolonged performance of the High-ForceHR task was associated with significantly increased sclerostin bone protein levels, compared to control rats. Immunohistochemistry also showed enhanced sclerostin production in the remaining osteocytes surrounding empty lacunae and deposition in the radial bone trabeculae of High-ForceHR rats, findings not present in control or Low-ForceHR rats (data not shown for the latter group). A proposed mathematical model using an inhibitory protein mechanism suggests that a reduction in osteocyte numbers by apoptosis should result in a decline in sclerostin production (van Oers, van Rietbergen et al. 2011). Instead, our results are similar to those reporting that osteocyte apoptosis increases sclerostin secretion into the bone matrix (Mabilleau, Mieczkowska et al. 2010). The increase of sclerostin in distal bones in this current study was paralleled by decreased bone formation, which is interesting in light of studies indicating that sclerostin is a negative regulator of bone formation (Poole, van Bezooijen et al. 2005, Robling, Kang et al. 2016). Recombinant sclerostin upregulates RANKL production and increases osteoclast formation (Wijenayaka, Kogawa et al. 2011), changes that enhance bone resorption. A key point to make is that the bones in this study were loaded continuously for 18 weeks after the shaping period, in which there was also increasing loading with each week (thus, 24 total weeks). No prior experimental study has examined sclerostin in bones after this length of loading time. Lastly, serum levels of sclerostin were unchanged from control levels in High-ForceHR rats, matching the mid-diaphyseal results (and likely levels in other bones throughout the body) rather than the distal metaphyseal results. This is consistent with a recent study indicating that serum levels of sclerostin may not reflect local levels (Delgado-Calle, Sato et al. 2017).
RANKL immunoexpression was primarily seen in osteocytes in this study, matching recent reports that osteocytes are the primary source of RANKL post-embryogenesis (Nakashima, Hayashi et al. 2011, Matsuo 2012, Xiong and O’Brien 2012). It has been suggested that production and activity of RANKL and OPG, its receptor decoy, is dictated by the severity of the damage (i.e., microcracks, stress fractures), disrupting the osteocytic network (Mulcahy, Taylor et al. 2011). Osteocytes in proximity to the damage site undergo apoptosis, while neighboring cells increase their production of RANKL, a response believed to encourage removal of the damaged matrix by osteoclasts (Al-Dujaili, Lau et al. 2011, Matsuo 2012, O’Brien, Nakashima et al. 2012, Kennedy, Laudier et al. 2014). The increase in RANKL mRNA and protein expression in distal forelimb bone regions of High-ForceHR rats, combined with the presence of microcracks, is suggestive of loading-induced matrix microdamage that was disruptive to the osteocytes, leading to their apoptosis, and triggering an increase in RANKL production by neighboring cells and then osteoclastogenesis (Colopy, Benz-Dean et al. 2004, Mulcahy, Taylor et al. 2011, Kennedy, Herman et al. 2012, O’Brien, Nakashima et al. 2012, Kennedy, Laudier et al. 2014).
We also attempted to investigate a muscle-bone interaction by examining osteoactivin levels in multiple forelimb tissues. Osteoactivin is an osteoblast-related glycoprotein and bone anabolic growth factor (Abdelmagid, Barbe et al. 2007, Moussa, Hisijara et al. 2014, Frara, Abdelmagid et al. 2016), as well as a potential “myometabokine” that can affect metabolism in bone (Tagliaferri, Wittrant et al. 2015). The decreased osteoactivin protein levels in both forelimb flexor muscles and bones of High-ForceHR rats could be contributing to the decreased osteoblast activity. We have reported an increase of osteoactivin in flexor forelimb muscles of rats that had performed a related Low-ForceHR task for 8 weeks (Frara, Abdelmagid et al. 2016). Although more research is needed on this topic, perhaps the increased muscle osteoactivin contributed to bone anabolism in Low-ForceHR rats, and muscle reductions contributed to lowered metabolic responses in the underlying bone of High-ForceHR rats, as suggested in the literature for myometabokines (Baskin, Winders et al. 2015, Tagliaferri, Wittrant et al. 2015, Laurent, Dubois et al. 2016).
Regarding possible contributions from underlying inflammatory responses, we have not yet assessed inflammatory cytokine responses occurring between 12 and 18 weeks of task performance in either task group (Gao, Fisher et al. 2013, Fisher, Zhao et al. 2015). However, we have reported that inflammatory cytokine levels in serum and forelimb muscles were highest immediately after training to high force levels or in task week 6 of High-ForceHR rats, and then declined progressively and significantly in task weeks 12 and 18, compared to post-training or task week 6 levels (Fisher, Zhao et al. 2015). By week 18, serum and muscle inflammation appears resolved in serum and muscle of High-ForceHR rats (Fisher, Zhao et al. 2015), findings further confirmed here in bone. That said, the prior inflammatory cytokine responses likely contributed to the bone microarchitecture changes observed by week 18, since we have previously found that reducing this inflammation using ibuprofen beginning in task week 5 and continuing to week 12, prevented High-ForceHR induced trabecular bone loss.
We have several limitations in this study, including that serum levels of osteocalcin, CTX-1, RANKL and sclerostin are representative of changes occurring in all bones involved in performing the task, such as the humerus and scapula. The serum changes should be interpreted with that in mind. For mRNA and bone protein level analyses, we collected and homogenized both ulna and radial bones together after separating them into distal and mid-diaphyseal regions. Therefore, these latter data are representative of changes in distal versus mid-diaphyseal regions of both bones. Quantification of immunohistochemical expression of these proteins in radial versus ulna bones was used to reduce this limitation. Lastly, although we were able to rule out flexor muscle mass, weight and circulating estrogen level differences as potential contributors, median nerve pathology in the form of both irritative (inflammatory) and compressive injury (increased collagen deposition) is present in High-ForceHR rats that performed the task for 12 weeks (Clark, Al-Shatti et al. 2004, Jain, Barr-Gillespie et al. 2014). Such median nerve pathology may be contributing to radial bone loss in High-ForceHR rats, although it would be more likely to affect carpal or phalangeal bones than the radius since the latter receives its primary input from branches of the radial nerve (Tageldin, Alrashid et al. 2015). Future studies should attempt to determine if median nerve pathology is causing a general permissive effect on bone turnover, as suggested in a recent review (Laurent, Dubois et al. 2016).
CONCLUSION
It is commonly assumed that bone mass increases with vigorous exercise and decreases as a result of unloading or reduced mechanical stimulation. We observed different remodeling responses when using a similar repetition rate yet two different stress magnitudes (Low-Force versus High-Force). Performance of a Low-ForceHR task for 18 weeks was matched by positive bone adaptation in both distal trabecular and mid-diaphyseal cortical bone of the radius (and somewhat in the ulna of these rats), matching most theories of loading-induced bone anabolism. In contrast, 18 weeks of performance of a High-ForceHR task for 2 hours/day, in 30 minute intervals separated by 1.5 hour rest periods, 3 days/week, for 18 weeks, was demanding enough to increase many indices of bone degradation consistent with damage repair and fatigue in the radius. The ulna bone showed fewer loading effects, in line with other studies showing that in muscular loading of forelimb bones in both neutral and ulnar deviated positions (such as used in power grip) that the radius is the primary bone receiving force transmission (Hara, Horii et al. 1992, Erhart, Schmoelz et al. 2012). Perhaps high demand repetitive tasks or movements are contributing to the increasing prevalence of distal radial fractures in pediatric and elderly populations (Nellans, Kowalski et al. 2012). Upon diagnosis of long-term upper extremity overuse injuries perhaps bone scans of the distal forelimb should become part of clinical testing. We also suggest from these data that prolonged performance of highly repetitive upper extremity tasks (> 2 reaches/min) (Silverstein, Fine et al. 1986, Nelson, Park et al. 1992) should not be performed at force loads greater than 15% of the maximum voluntary force for this particular task, and/or that future studies should investigate the effectiveness of longer rest/recovery periods when performing such tasks.
Supplementary Material
Supplemental Movie. A rat performing the High-ForceHR lever pulling task for a food reward. A radio is playing the background to provide white noise for the rats.
Supplemental Figure 1. Diagram showing reach impulse as the area under the force curve. Reaches receiving a food reward had to occur between the maximum and minimum force thresholds (upper and lower green lines); the target force is indicated by the red line. Reaches occurring over the maximum force threshold were not rewarded.
Supplemental Figure 2. Various pulling styles by three different rats. (A) Typical pull in which all digits are used to grasp the lever bar. (B) Another typical pull in which a rat uses only digits 2 through 4 to pull on the lever bar. (C) A bilateral pull with the primary grasping limb in a supinated position. This type of pull occurred with more frequency with prolonged task performance. Each style lead to a successful pull in which a food reward was delivered.
Supplemental Figure 3. TNFalpha levels in control and High-ForceHR distal forelimb bones. A representative Western blot of distal forelimb bone (radius and ulnar metaphyses and epiphyses combined) homogenates from a control (C) and 18-week High-ForceHR rat, probed with anti-TNFalpha (at 17 kDa). GAPDH and beta Actin were used as loading controls (at 42 and 37 kDa as indicated). Blots were repeated at least three times.
HIGHLIGHTS.
Performance of a high force high repetition reaching and grasping task by young adult rats for 18 weeks was associated with bone loss, microcracks and increased catabolic indices in distal metaphyseal trabeculae.
Prolonged performance of a low force high repetition task by young adult rats for 18 weeks induced adaptive bone remodeling in the distal metaphysis of the radius.
Fewer changes are induced in the ulnar bone by either reaching and grasping task.
Prolonged performance of a high force high repetition reaching and grasping task induces distal radial bone catabolic changes that may increase fracture risk.
Acknowledgments
Research reported in this publication was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases of the National Institutes of Health under Award Number AR056019 to MFB. This study utilized services offered by core facilities of the Center for Substance Abuse Research (Grant Number DA013429), the MicroCT and Imaging Core at Temple University School of Medicine, and Bioquant Image Analysis Corporation. We would also like to thank Vicki Kalscheur at the School of Veterinary Medicine, University of Wisconsin-Madison, for her help with part of the basic fuchsin staining and sectioning. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Footnotes
COMPETING INTERESTS
None of the authors have any competing interests to declare.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Abdelmagid SM, Barbe MF, Arango-Hisijara I, Owen TA, Popoff SN, Safadi FF. Osteoactivin acts as downstream mediator of BMP-2 effects on osteoblast function. J Cell Physiol. 2007;210(1):26–37. doi: 10.1002/jcp.20841. [DOI] [PubMed] [Google Scholar]
- Al-Dujaili SA, Lau E, Al-Dujaili H, Tsang K, Guenther A, You L. Apoptotic osteocytes regulate osteoclast precursor recruitment and differentiation in vitro. J Cell Biochem. 2011;112(9):2412–2423. doi: 10.1002/jcb.23164. [DOI] [PubMed] [Google Scholar]
- Al-Shatti T, Barr AE, Safadi FF, Amin M, Barbe MF. Increase in inflammatory cytokines in median nerves in a rat model of repetitive motion injury. J Neuroimmunol. 2005;167(1–2):13–22. doi: 10.1016/j.jneuroim.2005.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Apostolakos J, Durant TJ, Dwyer CR, Russell RP, Weinreb JH, Alaee F, Beitzel K, McCarthy MB, Cote MP, Mazzocca AD. The enthesis: a review of the tendon-to-bone insertion. Muscles Ligaments Tendons J. 2014;4(3):333–342. [PMC free article] [PubMed] [Google Scholar]
- Avin KG, Bloomfield SA, Gross TS, Warden SJ. Biomechanical aspects of the muscle-bone interaction. Curr Osteoporos Rep. 2015;13(1):1–8. doi: 10.1007/s11914-014-0244-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barbe MF, Elliott MB, Abdelmagid SM, Amin M, Popoff SN, Safadi FF, Barr AE. Serum and tissue cytokines and chemokines increase with repetitive upper extremity tasks. Journal of orthopaedic research : official publication of the Orthopaedic Research Society. 2008;26(10):1320–1326. doi: 10.1002/jor.20674. [DOI] [PubMed] [Google Scholar]
- Barbe MF, Gallagher S, Massicotte VS, Tytell M, Popoff SN, Barr-Gillespie AE. The interaction of force and repetition on musculoskeletal and neural tissue responses and sensorimotor behavior in a rat model of work-related musculoskeletal disorders. BMC Musculoskelet Disord. 2013;14(1):303. doi: 10.1186/1471-2474-14-303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barbe MF, Jain NX, Massicotte VS, Popoff SN, Barr-Gillespie AE. Ergonomic task reduction prevents bone osteopenia in a rat model of upper extremity overuse. Ind Health. 2015;53(3):206–221. doi: 10.2486/indhealth.2014-0159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barr AE, Safadi FF, Gorzelany I, Amin M, Popoff SN, Barbe MF. Repetitive, negligible force reaching in rats induces pathological overloading of upper extremity bones. J Bone Miner Res. 2003;18(11):2023–2032. doi: 10.1359/jbmr.2003.18.11.2023. [DOI] [PubMed] [Google Scholar]
- Baskin KK, Winders BR, Olson EN. Muscle as a “mediator” of systemic metabolism. Cell Metab. 2015;21(2):237–248. doi: 10.1016/j.cmet.2014.12.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benjamin M, Toumi H, Ralphs JR, Bydder G, Best TM, Milz S. Where tendons and ligaments meet bone: attachment sites (‘entheses’) in relation to exercise and/or mechanical load. J Anat. 2006;208(4):471–490. doi: 10.1111/j.1469-7580.2006.00540.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benjamin M, Toumi H, Suzuki D, Redman S, Emery P, McGonagle D. Microdamage and altered vascularity at the enthesis-bone interface provides an anatomic explanation for bone involvement in the HLA-B27-associated spondylarthritides and allied disorders. Arthritis Rheum. 2007;56(1):224–233. doi: 10.1002/art.22290. [DOI] [PubMed] [Google Scholar]
- Blair SN, Kohl HW, Gordon NF, Paffenbarger RS., Jr How much physical activity is good for health? Annu Rev Public Health. 1992;13:99–126. doi: 10.1146/annurev.pu.13.050192.000531. [DOI] [PubMed] [Google Scholar]
- Bonewald LF. Osteocyte messages from a bony tomb. Cell Metab. 2007;5(6):410–411. doi: 10.1016/j.cmet.2007.05.008. [DOI] [PubMed] [Google Scholar]
- Borah B, Dufresne T, Nurre J, Phipps R, Chmielewski P, Wagner L, Lundy M, Bouxsein M, Zebaze R, Seeman E. Risedronate reduces intracortical porosity in women with osteoporosis. J Bone Miner Res. 2010;25(1):41–47. doi: 10.1359/jbmr.090711. [DOI] [PubMed] [Google Scholar]
- Bove GM, Harris MY, Zhao H, Barbe MF. Manual therapy as an effective treatment for fibrosis in a rat model of upper extremity overuse injury. J Neurol Sci. 2016;361:168–180. doi: 10.1016/j.jns.2015.12.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boyce BF, Xing L. Biology of RANK, RANKL, and osteoprotegerin. Arthritis Res Ther. 2007;9(Suppl 1):S1. doi: 10.1186/ar2165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brunkow ME, Gardner JC, Van Ness J, Paeper BW, Kovacevich BR, Proll S, Skonier JE, Zhao L, Sabo PJ, Fu Y, Alisch RS, Gillett L, Colbert T, Tacconi P, Galas D, Hamersma H, Beighton P, Mulligan J. Bone dysplasia sclerosteosis results from loss of the SOST gene product, a novel cystine knot-containing protein. Am J Hum Genet. 2001;68(3):577–589. doi: 10.1086/318811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bureau of Labor Statistics. Nonfatal Occupational Injuries and Illnesses Requiring Days Away From Work, 2015. U. S. D. o. Labor; 2016. https://www.bls.gov/news.release/pdf/osh2.pdf. www.bls.gov/iif/oshcdnew.htm, iifstaff@bls.gov. [Google Scholar]
- Burr DB, Forwood MR, Fyhrie DP, Martin RB, Schaffler MB, Turner CH. Bone microdamage and skeletal fragility in osteoporotic and stress fractures. J Bone Miner Res. 1997;12(1):6–15. doi: 10.1359/jbmr.1997.12.1.6. [DOI] [PubMed] [Google Scholar]
- Christen P, Ito K, Ellouz R, Boutroy S, Sornay-Rendu E, Chapurlat RD, van Rietbergen B. Bone remodelling in humans is load-driven but not lazy. Nat Commun. 2014;5:4855. doi: 10.1038/ncomms5855. [DOI] [PubMed] [Google Scholar]
- Ciriello VM, Snook SH, Webster BS, Dempsey P. Psychophysical study of six hand movements. Ergonomics. 2001;44(10):922–936. doi: 10.1080/00140130110068924. [DOI] [PubMed] [Google Scholar]
- Clark BD, Al-Shatti TA, Barr AE, Amin M, Barbe MF. Performance of a high-repetition, high-force task induces carpal tunnel syndrome in rats. J Orthop Sports Phys Ther. 2004;34(5):244–253. doi: 10.2519/jospt.2004.34.5.244. [DOI] [PubMed] [Google Scholar]
- Clark EA, Goodship AE, Lanyon LE. Locomotor bone strain as the stimulus for bone’s mechanical adaptability. J Physiol. 1975;245(2):57P. [PubMed] [Google Scholar]
- Colopy SA, Benz-Dean J, Barrett JG, Sample SJ, Lu Y, Danova NA, Kalscheur VL, Vanderby R, Jr, Markel MD, Muir P. Response of the osteocyte syncytium adjacent to and distant from linear microcracks during adaptation to cyclic fatigue loading. Bone. 2004;35(4):881–891. doi: 10.1016/j.bone.2004.05.024. [DOI] [PubMed] [Google Scholar]
- Dale AM, Ryan D, Welch L, Olsen MA, Buchholz B, Evanoff B. Comparison of musculoskeletal disorder health claims between construction floor layers and a general working population. Occup Environ Med. 2015;72(1):15–20. doi: 10.1136/oemed-2014-102313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delgado-Calle J, Sato AY, Bellido T. Role and mechanism of action of sclerostin in bone. Bone. 2017;96:29–37. doi: 10.1016/j.bone.2016.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dempster DW, Compston JE, Drezner MK, Glorieux FH, Kanis JA, Malluche H, Meunier PJ, Ott SM, Recker RR, Parfitt AM. Standardized nomenclature, symbols, and units for bone histomorphometry: a 2012 update of the report of the ASBMR Histomorphometry Nomenclature Committee. J Bone Miner Res. 2013;28(1):2–17. doi: 10.1002/jbmr.1805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ding H, Solovieva S, Vehmas T, Takala EP, Leino-Arjas P. Hand osteoarthritis and pinch grip strength among middle-aged female dentists and teachers. Scand J Rheumatol. 2010;39(1):84–87. doi: 10.3109/03009740903201834. [DOI] [PubMed] [Google Scholar]
- Ding M, Odgaard A, Hvid I. Changes in the three-dimensional microstructure of human tibial cancellous bone in early osteoarthritis. J Bone Joint Surg Br. 2003;85(6):906–912. [PubMed] [Google Scholar]
- Erhart S, Schmoelz W, Arora R, Lutz M. The biomechanical effects of a deepened articular cavity during dynamic motion of the wrist joint. Clin Biomech (Bristol, Avon) 2012;27(6):557–561. doi: 10.1016/j.clinbiomech.2012.01.003. [DOI] [PubMed] [Google Scholar]
- Findlay DM, Atkins GJ. Relationship between serum RANKL and RANKL in bone. Osteoporos Int. 2011;22(10):2597–2602. doi: 10.1007/s00198-011-1740-9. [DOI] [PubMed] [Google Scholar]
- Fischer KJ, Johnson JE, Waller AJ, McIff TE, Toby EB, Bilgen M. MRI-based modeling for radiocarpal joint mechanics: validation criteria and results for four specimen-specific models. J Biomech Eng. 2011;133(10):101004. doi: 10.1115/1.4005171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fisher PW, Zhao Y, Rico MC, Massicotte VS, Wade CK, Litvin J, Bove GM, Popoff SN, Barbe MF. Increased CCN2, substance P and tissue fibrosis are associated with sensorimotor declines in a rat model of repetitive overuse injury. J Cell Commun Signal. 2015;9(1):37–54. doi: 10.1007/s12079-015-0263-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frara N, Abdelmagid SM, Sondag GR, Moussa FM, Yingling VR, Owen TA, Popoff SN, Barbe MF, Safadi FF. Transgenic Expression of Osteoactivin/gpnmb Enhances Bone Formation In Vivo and Osteoprogenitor Differentiation Ex Vivo. J Cell Physiol. 2016;231(1):72–83. doi: 10.1002/jcp.25020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frara N, Abdelmagid SM, Tytell M, Amin M, Popoff SN, Safadi FF, Barbe MF. Growth and repair factors, osteoactivin, matrix metalloproteinase and heat shock protein 72, increase with resolution of inflammation in musculotendinous tissues in a rat model of repetitive grasping. BMC Musculoskelet Disord. 2016;17:34. doi: 10.1186/s12891-016-0892-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frost HM. Wolff’s Law and bone’s structural adaptations to mechanical usage: an overview for clinicians. Angle Orthod. 1994;64(3):175–188. doi: 10.1043/0003-3219(1994)064<0175:WLABSA>2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- Gallagher S, Schall MC., Jr Musculoskeletal disorders as a fatigue failure process: evidence, implications and research needs. Ergonomics. 2017;60(2):255–269. doi: 10.1080/00140139.2016.1208848. [DOI] [PubMed] [Google Scholar]
- Gao HG, Fisher PW, Lambi AG, Wade CK, Barr-Gillespie AE, Popoff SN, Barbe MF. Increased serum and musculotendinous fibrogenic proteins following persistent low-grade inflammation in a rat model of long-term upper extremity overuse. PLoS One. 2013;8(8):e71875. doi: 10.1371/journal.pone.0071875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo R, Yamashita M, Zhang Q, Zhou Q, Chen D, Reynolds DG, Awad HA, Yanoso L, Zhao L, Schwarz EM, Zhang YE, Boyce BF, Xing L. Ubiquitin ligase Smurf1 mediates tumor necrosis factor-induced systemic bone loss by promoting proteasomal degradation of bone morphogenetic signaling proteins. J Biol Chem. 2008;283(34):23084–23092. doi: 10.1074/jbc.M709848200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hara T, Horii E, An KN, Cooney WP, Linscheid RL, Chao EY. Force distribution across wrist joint: application of pressure-sensitive conductive rubber. J Hand Surg Am. 1992;17(2):339–347. doi: 10.1016/0363-5023(92)90417-n. [DOI] [PubMed] [Google Scholar]
- Hlavkova J, Lebeda T, Tichy T, Gadourek P, Urban P, Nakladalova M, Lastovkova A, Fenclova Z, Ridzon P, Ehler E, Richter M, Pesakova L, Pelclova D. Evaluation of Lumbar Spine Load by Computational Method in Order to Acknowledge Low-back Disorders as Occupational Diseases. Cent Eur J Public Health. 2016;24(1):58–67. doi: 10.21101/cejph.a4332. [DOI] [PubMed] [Google Scholar]
- Hoshi K, Kawaki H, Takahashi I, Takeshita N, Seiryu M, Murshid SA, Masuda T, Anada T, Kato R, Kitaura H, Suzuki O, Takano-Yamamoto T. Compressive force-produced CCN2 induces osteocyte apoptosis through ERK1/2 pathway. J Bone Miner Res. 2014;29(5):1244–1257. doi: 10.1002/jbmr.2115. [DOI] [PubMed] [Google Scholar]
- Hsieh YF, Silva MJ. In vivo fatigue loading of the rat ulna induces both bone formation and resorption and leads to time-related changes in bone mechanical properties and density. J Orthop Res. 2002;20(4):764–771. doi: 10.1016/S0736-0266(01)00161-9. [DOI] [PubMed] [Google Scholar]
- Jain NX, Barr-Gillespie AE, Clark BD, Kietrys DM, Wade CK, Litvin J, Popoff SN, Barbe MF. Bone loss from high repetitive high force loading is prevented by ibuprofen treatment. J Musculoskelet Neuronal Interact. 2014;14(1):78–94. [PMC free article] [PubMed] [Google Scholar]
- Jiang Y, Zhao J, Liao EY, Dai RC, Wu XP, Genant HK. Application of micro-CT assessment of 3-D bone microstructure in preclinical and clinical studies. J Bone Miner Metab. 2005;23(Suppl):122–131. doi: 10.1007/BF03026336. [DOI] [PubMed] [Google Scholar]
- Johnson JE, McIff TE, Lee P, Toby EB, Fischer KJ. Validation of radiocarpal joint contact models based on images from a clinical MRI scanner. Comput Methods Biomech Biomed Engin. 2014;17(4):378–387. doi: 10.1080/10255842.2012.684446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karl JW, Olson PR, Rosenwasser MP. The Epidemiology of Upper Extremity Fractures in the United States, 2009. J Orthop Trauma. 2015;29(8):e242–244. doi: 10.1097/BOT.0000000000000312. [DOI] [PubMed] [Google Scholar]
- Kennedy OD, Herman BC, Laudier DM, Majeska RJ, Sun HB, Schaffler MB. Activation of resorption in fatigue-loaded bone involves both apoptosis and active pro-osteoclastogenic signaling by distinct osteocyte populations. Bone. 2012;50(5):1115–1122. doi: 10.1016/j.bone.2012.01.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kennedy OD, Laudier DM, Majeska RJ, Sun HB, Schaffler MB. Osteocyte apoptosis is required for production of osteoclastogenic signals following bone fatigue in vivo. Bone. 2014;64:132–137. doi: 10.1016/j.bone.2014.03.049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim BJ, Bae SJ, Lee SY, Lee YS, Baek JE, Park SY, Lee SH, Koh JM, Kim GS. TNF-alpha mediates the stimulation of sclerostin expression in an estrogen-deficient condition. Biochem Biophys Res Commun. 2012;424(1):170–175. doi: 10.1016/j.bbrc.2012.06.100. [DOI] [PubMed] [Google Scholar]
- Kivimaki M, Jokela M, Nyberg ST, Singh-Manoux A, Fransson EI, Alfredsson L, Bjorner JB, Borritz M, Burr H, Casini A, Clays E, De Bacquer D, Dragano N, Erbel R, Geuskens GA, Hamer M, Hooftman WE, Houtman IL, Jockel KH, Kittel F, Knutsson A, Koskenvuo M, Lunau T, Madsen IE, Nielsen ML, Nordin M, Oksanen T, Pejtersen JH, Pentti J, Rugulies R, Salo P, Shipley MJ, Siegrist J, Steptoe A, Suominen SB, Theorell T, Vahtera J, Westerholm PJ, Westerlund H, O’Reilly D, Kumari M, Batty GD, Ferrie JE, Virtanen M IP-W Consortium. Long working hours and risk of coronary heart disease and stroke: a systematic review and meta-analysis of published and unpublished data for 603,838 individuals. Lancet. 2015;386(10005):1739–1746. doi: 10.1016/S0140-6736(15)60295-1. [DOI] [PubMed] [Google Scholar]
- Klein-Nulend J, Bacabac RG, Bakker AD. Mechanical loading and how it affects bone cells: the role of the osteocyte cytoskeleton in maintaining our skeleton. Eur Cell Mater. 2012;24:278–291. doi: 10.22203/ecm.v024a20. [DOI] [PubMed] [Google Scholar]
- Kotha SP, Hsieh YF, Strigel RM, Muller R, Silva MJ. Experimental and finite element analysis of the rat ulnar loading model-correlations between strain and bone formation following fatigue loading. J Biomech. 2004;37(4):541–548. doi: 10.1016/j.jbiomech.2003.08.009. [DOI] [PubMed] [Google Scholar]
- Laurent MR, Dubois V, Claessens F, Verschueren SM, Vanderschueren D, Gielen E, Jardi F. Muscle-bone interactions: From experimental models to the clinic? A critical update. Mol Cell Endocrinol. 2016;432:14–36. doi: 10.1016/j.mce.2015.10.017. [DOI] [PubMed] [Google Scholar]
- Li H, Li J, Zou Z, Fok AS. Fracture simulation of restored teeth using a continuum damage mechanics failure model. Dent Mater. 2011;27(7):e125–133. doi: 10.1016/j.dental.2011.03.006. [DOI] [PubMed] [Google Scholar]
- Li J, Li H, Shi L, Fok AS, Ucer C, Devlin H, Horner K, Silikas N. A mathematical model for simulating the bone remodeling process under mechanical stimulus. Dent Mater. 2007;23(9):1073–1078. doi: 10.1016/j.dental.2006.10.004. [DOI] [PubMed] [Google Scholar]
- Lin C, Jiang X, Dai Z, Guo X, Weng T, Wang J, Li Y, Feng G, Gao X, He L. Sclerostin mediates bone response to mechanical unloading through antagonizing Wnt/beta-catenin signaling. J Bone Miner Res. 2009;24(10):1651–1661. doi: 10.1359/jbmr.090411. [DOI] [PubMed] [Google Scholar]
- Mabilleau G, Mieczkowska A, Edmonds ME. Thiazolidinediones induce osteocyte apoptosis and increase sclerostin expression. Diabet Med. 2010;27(8):925–932. doi: 10.1111/j.1464-5491.2010.03048.x. [DOI] [PubMed] [Google Scholar]
- Marquez-Florez K, Vergara-Amador E, de Las Casas EB, Garzon-Alvarado DA. Theoretical distribution of load in the radius and ulna carpal joint. Comput Biol Med. 2015;60:100–106. doi: 10.1016/j.compbiomed.2015.02.016. [DOI] [PubMed] [Google Scholar]
- Massicotte VS, Frara N, Harris MY, Amin M, Wade CK, Popoff SN, Barbe MF. Prolonged performance of a high repetition low force task induces bone adaptation in young adult rats, but loss in mature rats. Exp Gerontol. 2015;72:204–217. doi: 10.1016/j.exger.2015.10.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuo K. Osteocytes communicate with osteoclast lineage cells via RANKL. IBMS BoneKEy. 2012;9 (February 2012) [Google Scholar]
- Matsuzaki H, Wohl GR, Novack DV, Lynch JA, Silva MJ. Damaging fatigue loading stimulates increases in periosteal vascularity at sites of bone formation in the rat ulna. Calcif Tissue Int. 2007;80(6):391–399. doi: 10.1007/s00223-007-9031-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moussa FM, Hisijara IA, Sondag GR, Scott EM, Frara N, Abdelmagid SM, Safadi FF. Osteoactivin promotes osteoblast adhesion through HSPG and alphavbeta1 integrin. J Cell Biochem. 2014;115(7):1243–1253. doi: 10.1002/jcb.24760. [DOI] [PubMed] [Google Scholar]
- Muir P, Sample SJ, Barrett JG, McCarthy J, Vanderby R, Jr, Markel MD, Prokuski LJ, Kalscheur VL. Effect of fatigue loading and associated matrix microdamage on bone blood flow and interstitial fluid flow. Bone. 2007;40(4):948–956. doi: 10.1016/j.bone.2006.11.012. [DOI] [PubMed] [Google Scholar]
- Mulcahy LE, Taylor D, Lee TC, Duffy GP. RANKL and OPG activity is regulated by injury size in networks of osteocyte-like cells. Bone. 2011;48(2):182–188. doi: 10.1016/j.bone.2010.09.014. [DOI] [PubMed] [Google Scholar]
- Nakashima T, Hayashi M, Fukunaga T, Kurata K, Oh-Hora M, Feng JQ, Bonewald LF, Kodama T, Wutz A, Wagner EF, Penninger JM, Takayanagi H. Evidence for osteocyte regulation of bone homeostasis through RANKL expression. Nat Med. 2011;17(10):1231–1234. doi: 10.1038/nm.2452. [DOI] [PubMed] [Google Scholar]
- Nellans KW, Kowalski E, Chung KC. The epidemiology of distal radius fractures. Hand Clin. 2012;28(2):113–125. doi: 10.1016/j.hcl.2012.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson NA, Park RM, Silverstein MA, Mirer FE. Cumulative trauma disorders of the hand and wrist in the auto industry. Am J Public Health. 1992;82(11):1550–1552. doi: 10.2105/ajph.82.11.1550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nguyen J, Tang SY, Nguyen D, Alliston T. Load Regulates Bone Formation and Sclerostin Expression through a TGFbeta-Dependent Mechanism. PLoS ONE. 2013;8(1):e53813. doi: 10.1371/journal.pone.0053813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- NIOSH. National Manufacturing Agenda - June 2010. 2011 Retrieved October 23, 2011, from http://www.cdc.gov/niosh/nora/comment/agendas/manuf.
- Noble B. Microdamage and apoptosis. Eur J Morphol. 2005;42(1–2):91–98. doi: 10.1080/09243860500096248. [DOI] [PubMed] [Google Scholar]
- O’Brien CA, Nakashima T, Takayanagi H. Osteocyte control of osteoclastogenesis. Bone. 2012 doi: 10.1016/j.bone.2012.08.121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Papanicolaou SE, Phipps RJ, Fyhrie DP, Genetos DC. Modulation of sclerostin expression by mechanical loading and bone morphogenetic proteins in osteogenic cells. Biorheology. 2009;46(5):389–399. doi: 10.3233/BIR-2009-0550. [DOI] [PubMed] [Google Scholar]
- Parfitt AM, Drezner MK, Glorieux FH, Kanis JA, Malluche H, Meunier PJ, Ott SM, Recker RR. Bone histomorphometry: standardization of nomenclature, symbols, and units. Report of the ASBMR Histomorphometry Nomenclature Committee. J Bone Miner Res. 1987;2(6):595–610. doi: 10.1002/jbmr.5650020617. [DOI] [PubMed] [Google Scholar]
- Poole KE, van Bezooijen RL, Loveridge N, Hamersma H, Papapoulos SE, Lowik CW, Reeve J. Sclerostin is a delayed secreted product of osteocytes that inhibits bone formation. FASEB J. 2005;19(13):1842–1844. doi: 10.1096/fj.05-4221fje. [DOI] [PubMed] [Google Scholar]
- Potvin JR. An equation to predict maximum acceptable loads for repetitive tasks based on duty cycle: evaluation with lifting and lowering tasks. Work. 2012;41(Suppl 1):397–400. doi: 10.3233/WOR-2012-0189-397. [DOI] [PubMed] [Google Scholar]
- Potvin JR. Predicting maximum acceptable efforts for repetitive tasks: an equation based on duty cycle. Hum Factors. 2012;54(2):175–188. doi: 10.1177/0018720811424269. [DOI] [PubMed] [Google Scholar]
- Potvin JR, Calder IC, Cort J, Agnew MJ, Stephens A. Maximal acceptable forces for manual insertions using a pulp pinch, oblique grasp and finger press. Journal of Industrial Ergonomics. 2006;36:779–787. [Google Scholar]
- Prendergast PJ, Taylor D. Prediction of bone adaptation using damage accumulation. J Biomech. 1994;27(8):1067–1076. doi: 10.1016/0021-9290(94)90223-2. [DOI] [PubMed] [Google Scholar]
- Rani S, Barbe MF, Barr AE, Litivn J. Role of TNF alpha and PLF in bone remodeling in a rat model of repetitive reaching and grasping. J Cell Physiol. 2010;225(1):152–167. doi: 10.1002/jcp.22208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rikli DA, Honigmann P, Babst R, Cristalli A, Morlock MM, Mittlmeier T. Intra-articular pressure measurement in the radioulnocarpal joint using a novel sensor: in vitro and in vivo results. J Hand Surg Am. 2007;32(1):67–75. doi: 10.1016/j.jhsa.2006.10.007. [DOI] [PubMed] [Google Scholar]
- Robling AG, Bellido T, Turner CH. Mechanical stimulation in vivo reduces osteocyte expression of sclerostin. J Musculoskelet Neuronal Interact. 2006;6(4):354. [PubMed] [Google Scholar]
- Robling AG, Kang KS, Bullock WA, Foster WH, Murugesh D, Loots GG, Genetos DC. Sost, independent of the non-coding enhancer ECR5, is required for bone mechanoadaptation. Bone. 2016;92:180–188. doi: 10.1016/j.bone.2016.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robling AG, Niziolek PJ, Baldridge LA, Condon KW, Allen MR, Alam I, Mantila SM, Gluhak-Heinrich J, Bellido TM, Harris SE, Turner CH. Mechanical stimulation of bone in vivo reduces osteocyte expression of Sost/sclerostin. J Biol Chem. 2008;283(9):5866–5875. doi: 10.1074/jbc.M705092200. [DOI] [PubMed] [Google Scholar]
- Rohmert W. Problems in determining rest allowances Part 1: use of modern methods to evaluate stress and strain in static muscular work. Appl Ergon. 1973;4(2):91–95. doi: 10.1016/0003-6870(73)90082-3. [DOI] [PubMed] [Google Scholar]
- Seidler A, Bergmann A, Jager M, Ellegast R, Ditchen D, Elsner G, Grifka J, Haerting J, Hofmann F, Linhardt O, Luttmann A, Michaelis M, Petereit-Haack G, Schumann B, Bolm-Audorff U. Cumulative occupational lumbar load and lumbar disc disease--results of a German multi-center case-control study (EPILIFT) BMC Musculoskelet Disord. 2009;10:48. doi: 10.1186/1471-2474-10-48. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Silverstein BA, Fine LJ, Armstrong TJ. Hand wrist cumulative trauma disorders in industry. Br J Ind Med. 1986;43(11):779–784. doi: 10.1136/oem.43.11.779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sjogaard G, Jorgensen LV, Ekner D, Sogaard K. Muscle involvement during intermittent contraction patterns with different target force feedback modes. Clin Biomech (Bristol, Avon) 2000;15(Suppl 1):S25–29. doi: 10.1016/s0268-0033(00)00056-5. [DOI] [PubMed] [Google Scholar]
- Sjogaard G, Lundberg U, Kadefors R. The role of muscle activity and mental load in the development of pain and degenerative processes at the muscle cell level during computer work. Eur J Appl Physiol. 2000;83(2–3):99–105. doi: 10.1007/s004210000285. [DOI] [PubMed] [Google Scholar]
- Skerry TM, Bitensky L, Chayen J, Lanyon LE. Early strain-related changes in enzyme activity in osteocytes following bone loading in vivo. J Bone Miner Res. 1989;4(5):783–788. doi: 10.1002/jbmr.5650040519. [DOI] [PubMed] [Google Scholar]
- Snook SH, Irvine CH. Maximum frequency of lift acceptable to male industrial workers. Am Ind Hyg Assoc J. 1968;29(6):531–536. doi: 10.1080/00028896809343050. [DOI] [PubMed] [Google Scholar]
- Solovieva S, Vehmas T, Riihimaki H, Luoma K, Leino-Arjas P. Hand use and patterns of joint involvement in osteoarthritis. A comparison of female dentists and teachers. Rheumatology (Oxford) 2005;44(4):521–528. doi: 10.1093/rheumatology/keh534. [DOI] [PubMed] [Google Scholar]
- Spatz J, Ellman R, Cloutier A, Louis L, van Vliet M, Suva L, Dwyer D, Stolina M, Ke H, Bouxsein M. Sclerostin antibody inhibits skeletal deterioration due to reduced mechanical loading. J Bone Miner Res. 2012 doi: 10.1002/jbmr.1807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tageldin ME, Alrashid M, Khoriati AA, Gadikoppula S, Atkinson HD. Periosteal nerve blocks for distal radius and ulna fracture manipulation--the technique and early results. J Orthop Surg Res. 2015;10:134. doi: 10.1186/s13018-015-0277-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tagliaferri C, Wittrant Y, Davicco MJ, Walrand S, Coxam V. Muscle and bone, two interconnected tissues. Ageing Res Rev. 2015;21:55–70. doi: 10.1016/j.arr.2015.03.002. [DOI] [PubMed] [Google Scholar]
- Thiagarajan G, Lu Y, Dallas M, Johnson ML. Experimental and finite element analysis of dynamic loading of the mouse forearm. J Orthop Res. 2014;32(12):1580–1588. doi: 10.1002/jor.22720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tu X, Rhee Y, Condon KW, Bivi N, Allen MR, Dwyer D, Stolina M, Turner CH, Robling AG, Plotkin LI, Bellido T. Sost downregulation and local Wnt signaling are required for the osteogenic response to mechanical loading. Bone. 2012;50(1):209–217. doi: 10.1016/j.bone.2011.10.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turner CH. Biomechanics of bone: determinants of skeletal fragility and bone quality. Osteoporos Int. 2002;13(2):97–104. doi: 10.1007/s001980200000. [DOI] [PubMed] [Google Scholar]
- van Oers RF, van Rietbergen B, Ito K, Hilbers PA, Huiskes R. A sclerostin-based theory for strain-induced bone formation. Biomech Model Mechanobiol. 2011;10(5):663–670. doi: 10.1007/s10237-010-0264-0. [DOI] [PubMed] [Google Scholar]
- Vanharanta H, Heliovaara M, Korpi J, Troup JD. Occupation, work load and the size and shape of lumbar vertebral canals. Scand J Work Environ Health. 1987;13(2):146–149. doi: 10.5271/sjweh.2076. [DOI] [PubMed] [Google Scholar]
- Vehmas T, Solovieva S, Riihimaki H, Luoma K, Leino-Arjas P. Hand workload and the metacarpal cortical index. a study of middle-aged teachers and dentists. Osteoporos Int. 2005;16(6):672–680. doi: 10.1007/s00198-004-1742-y. [DOI] [PubMed] [Google Scholar]
- Villotte S, Castex D, Couallier V, Dutour O, Knusel CJ, Henry-Gambier D. Enthesopathies as occupational stress markers: evidence from the upper limb. Am J Phys Anthropol. 2010;142(2):224–234. doi: 10.1002/ajpa.21217. [DOI] [PubMed] [Google Scholar]
- Wenzel TE, Schaffler MB, Fyhrie DP. In vivo trabecular microcracks in human vertebral bone. Bone. 1996;19(2):89–95. doi: 10.1016/8756-3282(96)88871-5. [DOI] [PubMed] [Google Scholar]
- Werner FW, Palmer AK, Somerset JH, Tong JJ, Gillison DB, Fortino MD, Short WH. Wrist joint motion simulator. J Orthop Res. 1996;14(4):639–646. doi: 10.1002/jor.1100140420. [DOI] [PubMed] [Google Scholar]
- Wijenayaka AR, Kogawa M, Lim HP, Bonewald LF, Findlay DM, Atkins GJ. Sclerostin stimulates osteocyte support of osteoclast activity by a RANKL-dependent pathway. PLoS One. 2011;6(10):e25900. doi: 10.1371/journal.pone.0025900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xin DL, Harris MY, Wade CK, Amin M, Barr AE, Barbe MF. Aging enhances serum cytokine response but not task-induced grip strength declines in a rat model of work-related musculoskeletal disorders. BMC Musculoskelet Disord. 2011;12:63. doi: 10.1186/1471-2474-12-63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiong J, O’Brien CA. Osteocyte RANKL: new insights into the control of bone remodeling. J Bone Miner Res. 2012;27(3):499–505. doi: 10.1002/jbmr.1547. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Movie. A rat performing the High-ForceHR lever pulling task for a food reward. A radio is playing the background to provide white noise for the rats.
Supplemental Figure 1. Diagram showing reach impulse as the area under the force curve. Reaches receiving a food reward had to occur between the maximum and minimum force thresholds (upper and lower green lines); the target force is indicated by the red line. Reaches occurring over the maximum force threshold were not rewarded.
Supplemental Figure 2. Various pulling styles by three different rats. (A) Typical pull in which all digits are used to grasp the lever bar. (B) Another typical pull in which a rat uses only digits 2 through 4 to pull on the lever bar. (C) A bilateral pull with the primary grasping limb in a supinated position. This type of pull occurred with more frequency with prolonged task performance. Each style lead to a successful pull in which a food reward was delivered.
Supplemental Figure 3. TNFalpha levels in control and High-ForceHR distal forelimb bones. A representative Western blot of distal forelimb bone (radius and ulnar metaphyses and epiphyses combined) homogenates from a control (C) and 18-week High-ForceHR rat, probed with anti-TNFalpha (at 17 kDa). GAPDH and beta Actin were used as loading controls (at 42 and 37 kDa as indicated). Blots were repeated at least three times.