Northern wheatears—small migratory songbirds that weigh about the same as an AA battery—fly 30,000 km every year from Alaska via Asia to Kenya and back again.1 Their ability to do so depends crucially on having an internal magnetic compass with which to sense the Earth’s magnetic field. It appears that all avian migrants have such a light-dependent compass. Although it is clear that the primary magnetoreceptors are located in the birds’ retinas, the chemical identity of the sensors and the biophysical sensory mechanism remain something of a mystery.2 Now, Jacqueline Barton and colleagues at the California Institute of Technology, collaborating with Dongping Zhong at Ohio State University, have suggested, intriguingly, that a DNA repair enzyme could be responsible.3
The enzyme in question is photolyase, a photoactive protein containing flavin adenine dinucleotide (FAD) as its principal chromophore. Photolyases repair DNA lesions, for example by splitting the cyclobutane pyrimidine dimers (CPDs) that can be formed by adjacent thymine bases after absorption of ultraviolet light (Figure 1). The repair mechanism involves blue-light photoexcitation of FADH–, the fully reduced state of the cofactor, followed by transfer of an electron from the flavin to the damaged DNA to form a pair of radicals, with one unpaired electron on the CPD and the other on the flavin (FADH•). The cyclobutane bonds that link the two thymines then break sequentially, and the electron is passed back to the flavin. The whole process is complete within a nanosecond.4
Figure 1.
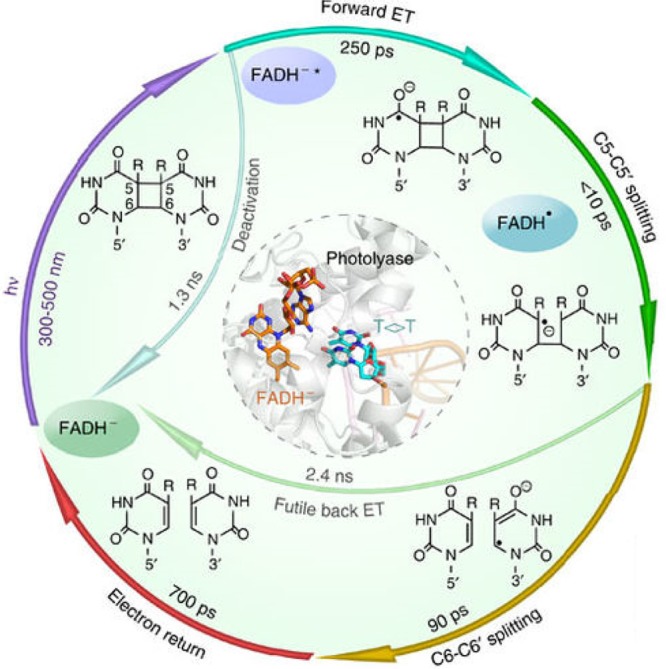
Mechanism of FAD mediated photolyase DNA repair. Reprinted with permission from ref (11). Copyright 2015 Macmillan Publishers Limited.
The new study by Zwang et al.3 uses a clever electrochemical method to monitor the repair of CPD lesions in monolayers of 29-base-pair DNA duplexes bound to Escherichia coli photolyase. Remarkably, the rate at which the repair proceeds depends on the intensity and direction of the external magnetic field provided by a nearby Nd–Fe–B magnet. Even magnetic fields weaker than the Earth’s (∼40 μT in Barton’s lab) appear to have a measurable effect on the efficiency of repair.
Individual molecules are so weakly magnetic that their interaction with a 40 μT magnetic field would normally be completely overwhelmed by their chaotic thermal motions whose energy is larger by a factor of a million. However, radical pairs have unique properties: they can exist in long-lived coherent spin states and undergo spin-selective chemical reactions. These features make them sensitive to tiny magnetic interactions.2 Indeed, the currently favored hypothesis for the mechanism of compass magnetoreception is based on light-induced radical pairs in a closely related protein, cryptochrome.5 Cryptochromes occur widely in the natural world, including birds’ retinas,6 have a range of functions, and are generally believed to be the evolutionary progeny of photolyases. They too contain a FAD chromophore, but they form magnetically sensitive radical pairs by a different light-dependent route: electron transfer from aromatic amino acid residues to the fully oxidized state of the FAD.7 For the most part, cryptochromes do not have DNA repair functions.
Barton’s study is the first to report magnetic field effects on photolyase-mediated DNA repair, and the results seem to be consistent with a radical pair mechanism. The magnetic field inhibits DNA repair, as would be expected for a radical pair created in a singlet state, and the effect saturates at a plausible field strength (∼3 mT). Also as expected, the change in the repair rate is unaffected by exact inversion of the magnetic field but is otherwise sensitive to the angle between the field vector and the alignment axis of the protein–DNA complexes. Importantly for a potential compass sensor, Barton’s magnetic field effects are substantially bigger than anything previously reported for cryptochromes where effects have only been seen for magnetic fields ≥1 mT.7
However, there are some puzzling aspects. Considering the lifetime and the separation of the radicals in photolyase, it is astonishing that a magnetic field effect can be observed at all. All radicals disappear within 1 ns, and in particular the radical pair most likely to be magnetically sensitive—the one containing the FAD radical and a CPD molecule in which the C5–C5′ bond, but not the C6–C6′, bond, has been broken—persists for less than 100 ps according to ultrafast spectroscopic measurements.8 To respond significantly to a 40 μT magnetic field, a radical pair would have to last for at least 1 μs.9
Moreover, the two radicals are quite close together: the edge of the isoalloxazine ring of the FAD is ∼0.8 nm away from the 5′ side of the CPD and only ∼0.43 nm from the 3′ side.8 At these distances, one can anticipate a strong electron exchange interaction that would lock the radical pair in its initial singlet spin state and block any effect of the much weaker interactions that give rise to the magnetic field dependence. This issue becomes even more severe if, as proposed,3 the radical pair is confined to the pyrimidine dimer. The 1,4 biradical that would result from homolytic cleavage of the C5–C5′ bond (Figure 5 of Zwang et al.3), for example, would have an even larger exchange interaction than the more conventional FADH• + CPD•– pair.
One way to resolve some of these uncertainties might be to use Zhong’s femtosecond techniques to look for magnetic field effects on both the ∼100 ps lifetime of the FADH• + [T–T]•– state and the branching ratio of its forward (→ FADH• + T + T•–) and backward (→ FADH– + T□T) reaction steps. Additionally, if it is not already known, it would probably be worthwhile to establish the occurrence and distribution of photolyases in the retinas of migratory songbirds.
It is now 18 years since cryptochrome was first proposed as a potential radical pair magnetoreceptor,10 and we are still not close to a “killer experiment” that has the power to confirm the hypothesis. Proving the existence of a magnetic sensory system with a requirement for UV-induced DNA damage could prove to be equally challenging.
References
- Bairlein F.; Norris D. R.; Nagel R.; Bulte M.; Voigt C. C.; Fox J. W.; Hussell D. J. T.; Schmaljohann H. Cross-hemisphere migration of a 25 g songbird. Biol. Lett. 2012, 8, 505–507. 10.1098/rsbl.2011.1223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hore P. J.; Mouritsen H. The radical pair mechanism of magnetoreception. Annu. Rev. Biophys. 2016, 45, 299–344. 10.1146/annurev-biophys-032116-094545. [DOI] [PubMed] [Google Scholar]
- Zwang T. J.; Tse E. C. N.; Zhong D. P.; Barton J. K. A compass at weak magnetic fields using thymine dimer repair. ACS Cent. Sci. 2018, 10.1021/acscentsci.8b00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang M.; Wang L. J.; Zhong D. P. Photolyase: dynamics and electron-transfer mechanisms of DNA repair. Arch. Biochem. Biophys. 2017, 632, 158–174. 10.1016/j.abb.2017.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J.; Du X. L.; Pan W. S.; Wang X. J.; Wu W. J. Photoactivation of the cryptochrome/photolyase superfamily. J. Photochem. Photobiol., C 2015, 22, 84–102. 10.1016/j.jphotochemrev.2014.12.001. [DOI] [Google Scholar]
- Günther A.; Einwich A.; Sjulstok E.; Feederle R.; Bolte P.; Koch K. W.; Solov’yov A. V.; Mouritsen H. Double-cone localization and seasonal expression pattern suggest a role in magnetoreception for European robin cryptochrome 4. Curr. Biol. 2018, 28, 211–223. 10.1016/j.cub.2017.12.003. [DOI] [PubMed] [Google Scholar]
- Maeda K.; Robinson A. J.; Henbest K. B.; Hogben H. J.; Biskup T.; Ahmad M.; Schleicher E.; Weber S.; Timmel C. R.; Hore P. J. Magnetically sensitive light-induced reactions in cryptochrome are consistent with its proposed role as a magnetoreceptor. Proc. Natl. Acad. Sci. U. S. A. 2012, 109, 4774–4779. 10.1073/pnas.1118959109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhong D. P. Electron transfer mechanisms of DNA repair by photolyase. Annu. Rev. Phys. Chem. 2015, 66, 691–715. 10.1146/annurev-physchem-040513-103631. [DOI] [PubMed] [Google Scholar]
- Maeda K.; Henbest K. B.; Cintolesi F.; Kuprov I.; Rodgers C. T.; Liddell P. A.; Gust D.; Timmel C. R.; Hore P. J. Chemical compass model of avian magnetoreception. Nature 2008, 453, 387–390. 10.1038/nature06834. [DOI] [PubMed] [Google Scholar]
- Ritz T.; Adem S.; Schulten K. A model for photoreceptor-based magnetoreception in birds. Biophys. J. 2000, 78, 707–718. 10.1016/S0006-3495(00)76629-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tan C.; Liu Z.; Li J.; Guo X.; Wang L.; Sancar A.; Zhong D. The molecular origin of high DNA-repair efficiency by photolyase. Nat. Commun. 2015, 6, 7302. 10.1038/ncomms8302. [DOI] [PubMed] [Google Scholar]


