Keywords: nerve regeneration, glycogen synthase kinase 3 beta inhibitor, SB216763, myelination, myotube differentiation, denervated muscle atrophy, Wnt/β-catenin, Schwann cell, muscle cells, peripheral nerve injury, neural regeneration
Abstract
Delay of axon regeneration after peripheral nerve injury usually leads to progressive muscle atrophy and poor functional recovery. The Wnt/β-catenin signaling pathway is considered to be one of the main molecular mechanisms that lead to skeletal muscle atrophy in the elderly. We hold the hypothesis that the innervation of target muscle can be promoted by accelerating axon regeneration and decelerating muscle cell degeneration so as to improve functional recovery of skeletal muscle following peripheral nerve injury. This process may be associated with the Wnt/β-catenin signaling pathway. Our study designed in vitro cell models to simulate myelin regeneration and muscle atrophy. We investigated the effects of SB216763, a glycogen synthase kinase 3 beta inhibitor, on the two major murine cell lines RSC96 and C2C12 derived from Schwann cells and muscle satellite cells. The results showed that SB216763 stimulated the Schwann cell migration and myotube contraction. Quantitative polymerase chain reaction results demonstrated that myelin related genes, myelin associated glycoprotein and cyclin-D1, muscle related gene myogenin and endplate-associated gene nicotinic acetylcholine receptors levels were stimulated by SB216763. Immunocytochemical staining revealed that the expressions of β-catenin in the RSC96 and C2C12 cytosolic and nuclear compartments were increased in the SB216763-treated cells. These findings confirm that the glycogen synthase kinase 3 beta inhibitor, SB216763, promoted the myelination and myotube differentiation through the Wnt/β-catenin signaling pathway and contributed to nerve remyelination and reduced denervated muscle atrophy after peripheral nerve injury.
Introduction
Peripheral nerve injury results in developmental atrophy of the target skeletal muscle and also poor functional recovery when surgery is delayed (Gigo-Benato et al., 2010; Gu et al., 2010; Willand et al., 2014; Zhang et al., 2017). The peripheral nervous system is different from the central nervous system as it has the capacity to regenerate after injury (David and Aguayo, 1985; Schmitt et al., 2003; Hall, 2005; Höke and Brushart, 2010). However, regeneration of the peripheral nerve after injury is a slow process (1–3 mm/d). Axons can take more than 3 months to regenerate into the distal target organs or tissues, meanwhile the distal nerve stump and skeletal muscle usually atrophy (Aydin et al., 2004). To improve the functional recovery of target muscle after peripheral nerve injury, it is essential to reduce the reinnervation time and reduce the atrophy of the denervated muscle (Moimas et al., 2013).
Previous studies have revealed that the Wallerian degeneration occurs at the lesion site after peripheral nerve injury (Wagner et al., 1998; Pesini et al., 1999; Wang et al., 2013; Xin et al., 2013). Promoting the migration and proliferation of the Schwann cells has benefits for the improvement of myelination and nerve regeneration (Le et al., 1988; Wang et al., 2016; Wen et al., 2017). Muscle regeneration also depends on resident satellite cells, which are located between the sarcolemma and basement membrane of muscle fibers (Montarras et al., 2005; Le and Rudnicki, 2007; Lepper et al., 2011). Restoration of myotube and myogenesis differentiation has been linked to a reduction of muscle atrophy (Sorci et al., 2003; Johnson et al., 2013; Lee et al., 2017). We hypothesize a therapy that has a positive effect on both Schwann cells and muscle cells, leading to a short reinnervation interval and good muscular function after peripheral nerve injury. In recent years, studies on the peripheral nerve injury have increasingly focused on the role of various signaling pathways, including the Wnt signaling pathway.
The discovery of the wingless gene found by Sharma in 1973 was the basis of the important Wnt signaling pathway (Sharma and Chopra, 1976). Since then other studies have shown that the Wnt/β-catenin signaling pathway has a direct role in myelin gene expression (Chew et al., 2011; Tawk et al., 2011; Meffre et al., 2015; Hichor et al., 2017). Wnt/β-catenin signals act as positive regulators during remyelination (Fancy et al., 2009; Makoukji et al., 2011). Recently, the Wnt/β-catenin signaling pathway has been considered the main molecular mechanism in age-related skeletal muscle atrophy (Rajasekaran et al., 2017). Wnt/β-catenin was involved in impaired muscle repair, such as loss of satellite cell number, muscle cell dysfunction, decreased myoblast proliferation and attenuated differentiation (Carlson et al., 2009). 3-(2,4-Dichlorophenyl)-4-(1-methyl-1H-indol-3-yl)-1H-pyrrole-2,5-dione (SB216763) is a typical inhibitor of glycogen synthase kinase 3 beta (GSK3β), which stimulates canonical Wnt/β-catenin signal (Li et al., 2012). Our previous study demonstrated that SB216763 can promote remyelination and myelin protein production (Chen et al., 2016). SB216763 reduces skeletal muscle atrophy (Litwiniuk et al., 2016). Pretreatment of muscle cell cultures with SB216763 prevented loss of myogenic differentiation and myogenesis induced by TNF-α (Verhees et al., 2013).
Considering the importance of the Wnt signaling pathway in nerve and muscle regeneration, we investigated the effect of the GSK3β inhibitor on both Schwann cells and muscle cells. Our in vitro model system mimics the progress of myelination and muscle atrophy, and evaluates the effect of SB216763 on RSC96 cells and C2C12 myotubes.
Materials and Methods
Cells culture and SB216763 treatment
Murine RSC96 Schwann cell and C2C12 myoblast cell lines, passages 1–3, were obtained from American Type Culture Collection (Manassas, VA, USA). Cells were cultured in growth media: RSC96 cells with Dulbecco's modified Eagle's medium (DMEM)/F12 containing 10% fetal bovine serum (Gibco, Grand Island, NY, USA), 100 units/mL penicillin and 100 µg/mL streptomycin (Sigma-Aldrich, St. Louis, MO, USA); C2C12 cells with high-glucose DMEM (Sigma-Aldrich) containing 5 mM glucose, 10% fetal bovine serum and antibiotics., When cells reached 80–90% confluence, the medium was replaced by a differentiation medium (high-glucose DMEM containing 2% calf serum (Gibco) and antibiotics) to induce C2C12 cell myotube formation and differentiation. The differentiation medium was changed daily for 6 days. SB216763 (Apexbio, Houston, TX, USA) was prepared as a 20 mM stock in dimethyl sulfoxide. RSC96 and C2C12 cells were exposed to 10 µM of SB216763 for 48 hours as described previously (Chen et al., 2016). Following these treatments, cells were washed with phosphate-buffered saline (PBS; Gibco) to remove any free drug prior to the sequence experiments.
Scratch wound assay
RSC96 cells were plated in 6-well plates 24 hours before the scratch. A scratch length, approximately 1 mm, was made in the middle area of the plate using a 200 µL pipette tip. All wells were washed with PBS to remove cellular debris. Cells were incubated with growth media as mentioned above. The scratch was photographed at 0, 24 and 48 hours using an inverted fluorescence microscope equipped with microscope camera (BM CAM, Shanghai, China) and ToupView software (ToupTek, Hangzhou, China). Five photographs were taken of the scratched region of each group at each time point. Relative area of the gap was calculated as follows: Relative area in the gap of wound = (area in the gap at 24 or 48 hours/area in the gap at 0 hours) × 100%.
Index of C2C12 myotube contraction
On day 6 of differentiation, cells were stimulated with 100 µM carbachol (Apexbio). Five high-quality images of cells were taken sequentially at 1-minute intervals during carbachol stimulation (Niu et al., 2011). The difference in integrated optical density (IOD) between the first and subsequent images is mainly due to the myotube contraction. Thus, the average IOD of differential image represents the index of contraction. Calculated as below, index of contraction = {∑(| IOD5–IOD4|+|IOD4–IOD3|+|IOD3–IOD2|+|IOD2–IOD1|)}/4. IOD1 to IOD5 stand for the mean IOD value of the first to fifth images. IOD was analyzed using Image-Pro Plus software (Media Cybernetics, Rockville, MD, USA).
Cell proliferation assay
RSC96 and C2C12 cells were seeded into 96-well plates at a density of 10,000 cells/well. After incubation for 48 hours with or without SB216763, cell viability was evaluated by Cell Counting Kit-8 (CCK-8) assay. Four hours before detection, 10 µL of CCK-8 solution (Sigma-Aldrich) was added into each well, followed by a further 1-hour incubation. The optical density (OD) values were measured at 450 nm with a microplate reader (Bio-Rad, Hercules, CA, USA).
Quantitative real-time polymerase chain reaction (PCR)
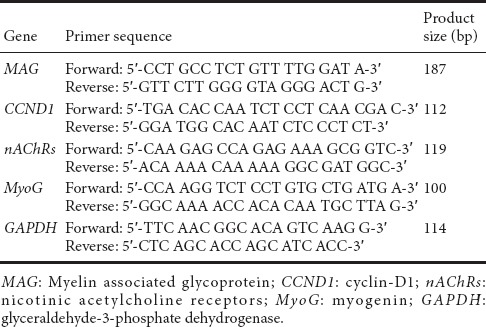
Total RNA was isolated from RSC96 and C2C12 cells using TRIzol reagent (Thermo Fisher, Waltham, MA, USA). cDNA was synthesized using M-MLV reverse transcriptase (Promega, Madison, WI, USA). The amplification was completed by two steps at each target gene annealing temperature. The specificity of the amplified product was determined according to the fusion curve, and the 2-ΔΔCt method was used to determine the mRNA expression. The primers used for PCR are given in Table 1.
Table 1.
Real-time fluorescent quantitative polymerase chain reaction primer sequences
Immunocytochemical staining
RSC96 and C2C12 cells with or without SB216763 treatment were washed three times with PBS and fixed for 15 minutes in 4% paraformaldehyde and 20 minutes in 0.5% Triton X-100 (Amresco, Solon, OH, USA), then deactivated with 3% H2O2 for 30 minutes. Subsequently, cell slices were blocked in PBS supplemented with 5% goat serum (Gibco) at room temperature for 20 minutes. Cells were incubated with anti-β-catenin antibody (1:300; Abcam, Cambridge, MA, USA) at 37°C for 1 hour, washed in PBS and incubated with anti-mouse IgG-horseradish peroxidase secondary antibody (1:1,000; GTVision, Shanghai, China) for 30 minutes at room temperature. Following washing with PBS, horseradish peroxidase activity was detected using a diaminobenzidine substrate kit (Vector Labs, Burlingame, CA, USA) according to the manufacturer's instructions. Five frames per slide were analyzed. Images were collected with a Leica DFC-550 connected to an Olympus IX70 inverted microscope (Olympus, Tokyo, Japan) and analyzed using the software Leica Application Suite Advanced Fluorescence (Leica, Wetzlar, Germany). The ratio of nuclear localization cells was calculated as follows: The positive ratio was equal to the number of cells with positively stained nuclei/the number of cells with negatively stained nuclei × 100%.
Statistical analysis
Statistical analysis was performed using SPSS 21.0 for Windows software (IBM, Armonk, NY, USA). All data are presented as the mean ± SD and analyzed with Student's t-test. A value of P less than 0.05 was considered statistically significant.
Results
GSK3β inhibitor accelerated migration of RSC96 cells
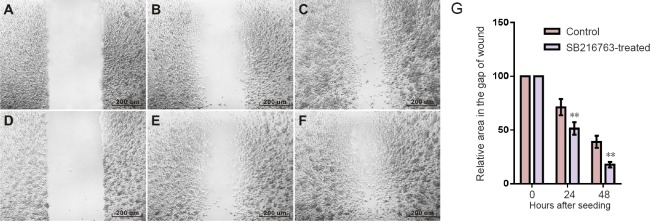
One therapeutic aspect of a GSK-3 inhibitor for the treatment of peripheral nerve injury was the migration ability of Schwann cells as Wallerian degeneration progressed. To compare the motility of RSC96 cells with or without SB216763 treatment, scratch assay was used to detect cell migration in the area of a “wound”, which was scratched into a confluent cell monolayer. The relative area of the gap of wound was measured by the advancement of cells over 24 and 48 hours. The scratch assay demonstrated that the relative area of the gap of wound in control group decreased to 71.2 ± 7.6% at 24 hours and 39.0 ± 5.6% at 48 hours, while that in the SB216763-treated group decreased to 51.4 ± 5.8% at 24 hours and 17.8 ± 2.5% at 48 hours. The results showed that there were more cells in the gap and the area of the gap was smaller in the SB216763-treated group compared with the control group (P < 0.01; Figure 1).
Figure 1.
SB216763 promoted gap closure in RSC96 cells.
(A–C) Control cells at 0, 24 and 48 hours. (D–F) SB216763-treated cells at 0, 24 and 48 hours. Scale bars: 200 μm. (G) Relative area in the gap of wound at each time. At 24 hours after seeding, confluent RSC96 cells were treated with SB216763 to conduct the scratch assay. Relative area in the gap of wound was determined at 24 and 48 hours after scratching. As a result, SB216763 significantly promoted gap closure as compared with the control (**P < 0.01, vs. control group; mean ± SD, n = 3, Student's t-test). Three independent experiments were performed.
GSK3β inhibitor stimulated contraction of C2C12 myotubes
Six days after differentiation, approximately 50% C2C12 cells had fused together to form the typical multinucleated myotubes. After exposure to 100 µM carbachol, vigorous contraction of C2C12 myotubes was readily observed. Examples at the first two times are shown in Figure 2A–D. Measurement of the index of contraction is described above, in the Methods. Analysis of the results revealed that the C2C12 myotubes in SB216763-treated group showed significantly higher contractile activity compared with the control group (Figure 2E).
Figure 2.
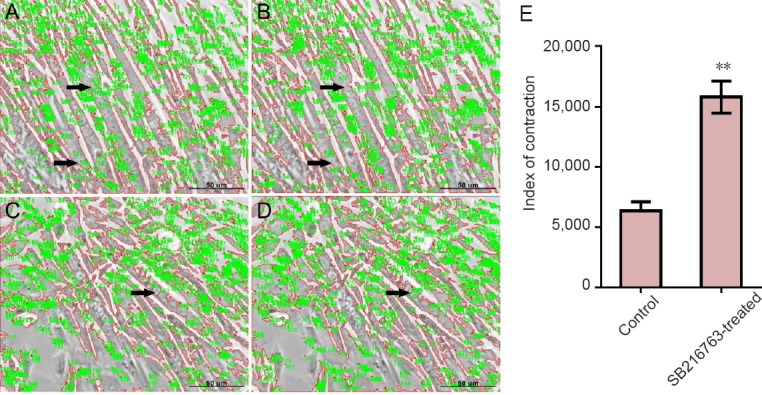
Significant differences in the contraction in C2C12 myotubes after carbachol treatment in each group.
(A, B) The first and second images of control cells. (C, D) The first and second images of SB216763-treated cells. Scale bars: 50 μm. C2C12 cells were differentiated into multinuclear myotubes by incubation with differentiation medium for 6 days. Images of contraction were taken sequentially at 1-minute intervals during carbachol exposure and images were observed as described in Materials and Methods sections (two of five images in each group were shown, black arrows reflected the integrated optical density changes of myotube). (E) Index of contraction: Higher index of SB216763-treated group reflected higher contractile activity (**P < 0.01, vs. control group; mean ± SD, n = 5, Student's t-test). Five independent experiments were performed.
GSK3β inhibitor induced proliferation of both Schwann cells and muscle cells
CCK-8 assay was used to determine the effects of 48 hours of treatment with SB216763 on the proliferation of RSC96 and C2C12 cells. The results showed that treatment with SB216763 significantly stimulated the growth of Schwann cells and muscle cells (Figure 3).
Figure 3.
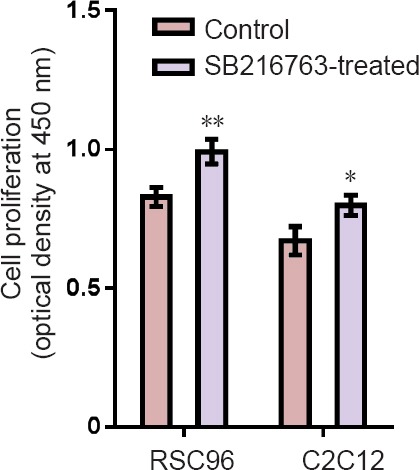
SB216763 promotes the proliferation of RSC96 and C2C12 cells (Cell Counting Kit-8 assay).
From the day after seeding, RSC96 and C2C12 cells were continuously exposed to SB216763 for 48 hours.There was significant difference in cell activity in the SB216763-treated group compared with the untreatedcontrol group (*P < 0.05, **P < 0.01; mean ± SD, n = 3, Student's t-test). Three independent experiments were performed.
GSK3β inhibitor treatment resulted in regulation of related genes
The mRNA expression of myelin associated glycoprotein (MAG), cyclin-D1 (CCND1), myogenin (MyoG) and nicotinic acetylcholine receptors (nAChRs) were detected by quantitative PCR. The mRNA expression of both MAG and CCND1 was significantly greater in the SB216763-treated than the control RSC96 group. Similarly, the mRNA expression of MyoG and nAChRs was significantly higher (P < 0.01) in SB216763-treated than in the control C2C12 group (Figure 4).
Figure 4.
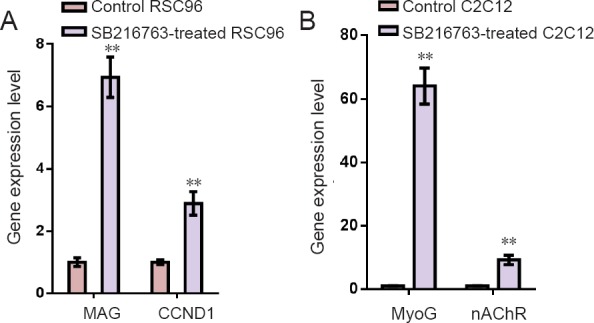
Quantitative polymerase chain reaction of mRNA expression in RSC96 and C2C12 cells in control and SB216763 groups.
Expression levels of myelin related gene MAG and CCND1, muscle related gene MyoG and endplate-associated gene nAChRs were all upregulated in the SB216763-treated cells compared with the control cells (**P < 0.01, vs. control group; mean ± SD, n = 3, Student's t-test). Three independent experiments were performed. MAG: Myelin associated glycoprotein; CCND1: cyclin-D1; MyoG: myogenin; nAChRs: nicotinic acetylcholine receptors.
GSK3β inhibitor promoted β-catenin expression and nuclear localization
Immunocytochemical staining was performed to detect β-catenin expression. Quantification of β-catenin nuclear localization was analyzed by counting the average positive cell ratio from five frames per slide. After SB216763 treatment, the overall β-catenin expression was greatly upregulated and the nuclear concentration of β-catenin was elevated in RSC96 cells. Beta-catenin expression level was higher and greater nuclear localization was shown in C2C12 cells of SB216763-treated group than in the control group (Figure 5). Our results demonstrated that the GSK3β inhibitor enhanced β-catenin expression and nuclear localization and possibly exerted its efficacy via Wnt/β-catenin signaling pathway.
Figure 5.
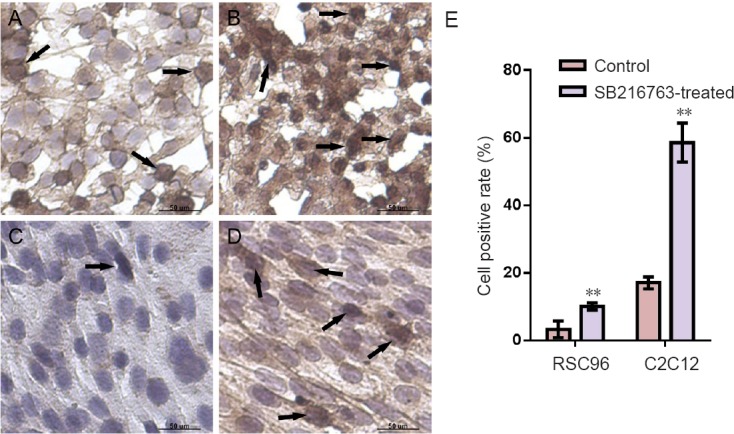
Immunocytochemical staining of β-catenin in RSC96 and C2C12 cells from SB216763-treated group and control group.
(A) Control RSC96 cells. (B) SB216763- treated RSC96 cells. (C) Control C2C12 cells. (D) SB216763-treated RSC96 cells. β-Catenin expression increased and nuclear localization was visible in the cells of the SB216763-treated group (black arrows). (E) The quantification of positive nuclear localization cells showed significant differences (**P < 0.01; mean ± SD, n = 3, Student's t-test). Three independent experiments were performed.
Discussion
Peripheral nerve injury is a common disease and has been a focus in neuroscience research for decades. In a clinical setting of peripheral nerve damage, both nerve and muscles undergo degeneration processes. The identification of the molecular mechanism that fosters nerve regeneration, as well as to protect skeletal muscle from atrophy, is thus required. Enhancing the efficacy of remyelination is one of the main areas of research for the treatment of peripheral nerve injury (Torii and Yamauchi, 2016; Yi et al., 2017). Remyelination failure usually leads to irreversible axonal loss and functional impairment (Brück, 2005; Frohman et al., 2006). For successful axon myelin formation, it is critical to increase the proliferation of Schwann cells to support the injured axon. As reported, any delay of interaction between axon and distal target may lead to axonal degeneration, resulting in neuronal apoptosis (Ohnishi et al., 1985; Scheib and Höke, 2013). In turn, peripheral nerve injury could result in the atrophy of the target skeletal muscle. This denervation process induces some complex changes in the expression of genes involved in muscle atrophy (Durbeej et al., 2003). However, the mechanisms underlying the degeneration and regeneration of Schwann cells and muscle cells remain unclear. In our in vitro model, the GSK3β inhibitor, SB216763, plays a role in regulating both myelin formation and myogenesis. The preliminary data have shown that a GSK3β inhibitor may promote myelination and also reverse muscle cell atrophy through regulating the Wnt/β-catenin signaling pathway.
SB216763 directly inhibits GSK3β, leading to β-catenin stabilization and nuclear translocation, thereby activating the Wnt/β-catenin signaling pathway (Lenox and Wang, 2003). In the peripheral nervous system, GSK3β inhibitor treatment promotes remyelination of sciatic and facial nerves after crush (Makoukji et al., 2012). Previously we found that up-regulating Wnt/β-catenin signaling by SB216763 induced the clearance of myelin debris and the expression of myelin-related genes (Chen et al., 2016). Others have found that activation of Wnt signaling is important for muscle regeneration (Polesskaya et al., 2003; Brack et al., 2009). Wnt signaling also enhances the proliferation of muscle satellite cells (Otto et al., 2008).
In this study, two kinds of in vitro cellular models of peripheral nerve injury were used to study the effect of SB216763 on myelination and myotube differentiation. In the two cell lines, RSC96 and C2C12, tested, the beneficial effects of SB216763 on the progression of peripheral nerve repair are not limited to the local injured site. A previous study showed that migration of Schwann cell is essential to create a permissive environment to promote axon sprouting and also inhibit nerve scar formation (Chehrehasa et al., 2010). In our RSC96 model, SB216763 treatment also accelerated the migratory capacities of the cells. We obtained other comparable beneficial results in other assays. SB216763 treatment increased RSC96 cell proliferation, and enhanced myelin-related gene expression (MAG and CCND1). CCND1 is a β-catenin target gene for an important protein in the cell cycle and can promote the proliferation of cells (Zhao et al., 2016). MAG is a myelin-specific marker which plays a key role in the early stage of myelination and the maintenance of stable axonal myelin interaction (Shim and Ming, 2010). These encouraging results revealed that the treatment with a GSK3β inhibitor can increase myelin formation.
Positive effects of SB216763 on C2C12 muscle cell line, in vitro, were also found. SB216763 stimulated C2C12 myotube contraction, which is caused by acetylcholine receptor activation. CCK-8 assay showed that SB216763 induced C2C12 cell proliferation. Quantitative PCR data indicated that SB216763 up-regulated the expression of MyoG and nAChRs of C2C12 cells. MyoG is a muscle-specific transcription factor that is essential for the development of skeletal muscle, and atrophy of denervated muscle could be prevented by high MyoG expression (Teraoka et al., 2012). nAChR acts as an activator of genes encoding endplate-associated proteins (Méjat et al., 2005). These data revealed that GSK3β inhibitor has the potential to restore muscular function as well as accelerate the myelination.
The mechanism by which GSK3β inhibitor exerts its efficacy was explored by immunocytochemical staining. Exposure to SB216763 increased the nuclear localization and expression of β-catenin, the landmark molecular event of the Wnt signaling pathway. All results indicated that SB216763, as a GSK3β inhibitor, may exert its positive effects on Schwann cells and muscle cells via the Wnt/β-catenin signaling pathway.
In summary, this in vitro study supports the hypothesis that this GSK3β inhibitor generates a permissive environment for promoting nerve regeneration and decelerating muscle atrophy in the peripheral nervous system. Our results suggest that a GSK3β inhibitor can be considered as an important therapeutic target in myelination and myotube differentiation. This research in vitro provides the basis for the hitherto absent in vivo animal model studies and further investigations into the underlying mechanism.54
Additional file (130.7KB, pdf) : Open peer review reports 1 and 2.
Footnotes
Funding: This study was funded by the National Basic Research Program of China (973 Program), No. 2014CB542201; the National High Technology Research and Development Program of China (863 Program), No. SS2015AA020501; the National Natural Science Foundation of China (General Program), No. 31571235, 31771322, 31671248, 31571236, 31271284, 31171150, 81171146, 31471144, 30971526, 31100860, 31040043, 31371210, and 81372044.
Conflicts of interest: There are no competing interests to declare.
Financial support: This study was supported by the National Basic Research Program of China (973 Program), No. 2014CB542201; the National High Technology Research and Development (863 Program), No. SS2015AA020501; the National Natural Science Foundation of China (General Program), No. 31571235, 31771322, 31671248, 31571236, 31271284, 31171150, 81171146, 31471144, 30971526, 31100860, 31040043, 31371210, and 81372044. The funding sources had no role in the design, execution, and data collection, analysis, and interpretation or decision to submit results.
Data sharing statement: Datasets analyzed during the current study are available from the corresponding author on reasonable request.
Plagiarism check: Checked twice by iThenticate.
Peer review: Externally peer reviewed.
Open peer reviewer: Yiren Hu, University of California San Diego, USA; Dirk Montag, Head Neurogenetics Special Laboratory, Leibniz Institute for Neurobiology, Neurogenetics Special Laboratory, Germany.
(Copyedited by Wang J, Li CH, Qiu Y, Song LP, Zhao M)
References
- 1.Aydin MA, Mackinnon SE, Gu XM, Kobayashi J, Kuzon WM. Force deficits in skeletal muscle after delayed reinnervation. Plast Reconstr Surg. 2004;113:1712–1718. doi: 10.1097/01.prs.0000118049.93654.ca. [DOI] [PubMed] [Google Scholar]
- 2.Brack AS, Murphy-Seiler F, Hanifi J, Deka J, Eyckerman S, Keller C, Aguet M, Rando TA. BCL9 is an essential component of canonical Wnt signaling that mediates the differentiation of myogenic progenitors during muscle regeneration. Dev Biol. 2009;335:93–105. doi: 10.1016/j.ydbio.2009.08.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brück W. The pathology of multiple sclerosis is the result of focal inflammatory demyelination with axonal damage. J Neurol. 2005;252(Suppl 5):3–9. doi: 10.1007/s00415-005-5002-7. [DOI] [PubMed] [Google Scholar]
- 4.Carlson ME, Suetta C, Conboy MJ, Aagaard P, Mackey A, Kjaer M, Conboy I. Molecular aging and rejuvenation of human muscle stem cells. EMBO Mol Med. 2009;1:381–391. doi: 10.1002/emmm.200900045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chehrehasa F, Windus LC, Ekberg JA, Scott SE, Amaya D, Mackay-Sim A, St JJA. 2010.Olfactory glia enhance neonatal axon regeneration. Mol Cell Neurosci. 45:277–288. doi: 10.1016/j.mcn.2010.07.002. [DOI] [PubMed] [Google Scholar]
- 6.Chen Y, Weng J, Han D, Chen B, Ma M, Yu Y, Li M, Liu Z, Zhang P, Jiang B. GSK3β inhibition accelerates axon debris clearance and new axon remyelination. Am J Transl Res. 2016;8:5410–5420. [PMC free article] [PubMed] [Google Scholar]
- 7.Chew LJ, Shen W, Ming X, Senatorov VV, Chen HL, Cheng Y, Hong E, Knoblach S, Gallo V. SRY-box containing gene 17 regulates the Wnt/β-catenin signaling pathway in oligodendrocyte progenitor cells. J Neurosci. 2011;31:13921–13935. doi: 10.1523/JNEUROSCI.3343-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.David S, Aguayo AJ. Axonal regeneration after crush injury of rat central nervous system fibres innervating peripheral nerve grafts. J Neurocytol. 1985;14:1–12. doi: 10.1007/BF01150259. [DOI] [PubMed] [Google Scholar]
- 9.Durbeej M, Sawatzki SM, Barresi R, Schmainda KM, Allamand V, Michele DE, Campbell KP. Gene transfer establishes primacy of striated vs. smooth muscle sarcoglycan complex in limb-girdle muscular dystrophy. Proc Natl Acad Sci U S A. 2003;100:8910–8915. doi: 10.1073/pnas.1537554100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fancy SP, Baranzini SE, Zhao C, Yuk DI, Irvine KA, Kaing S, Sanai N, Franklin RJ, Rowitch DH. Dysregulation of the Wnt pathway inhibits timely myelination and remyelination in the mammalian CNS. Genes Dev. 2009;23:1571–1585. doi: 10.1101/gad.1806309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Frohman E, Costello F, Zivadinov R, Stuve O, Conger A, Winslow H, Trip A, Frohman T, Balcer L. Optical coherence tomography in multiple sclerosis. Lancet Neurol. 2006;5:853–863. doi: 10.1016/S1474-4422(06)70573-7. [DOI] [PubMed] [Google Scholar]
- 12.Gigo-Benato D, Russo TL, Geuna S, Domingues NR, Salvini TF, Parizotto NA. Electrical stimulation impairs early functional recovery and accentuates skeletal muscle atrophy after sciatic nerve crush injury in rats. Muscle Nerve. 2010;41:685–693. doi: 10.1002/mus.21549. [DOI] [PubMed] [Google Scholar]
- 13.Gu S, Shen Y, Xu W, Xu L, Li X, Zhou G, Gu Y, Xu J. Application of fetal neural stem cells transplantation in delaying denervated muscle atrophy in rats with peripheral nerve injury. Microsurgery. 2010;30:266–274. doi: 10.1002/micr.20722. [DOI] [PubMed] [Google Scholar]
- 14.Hall S. 2005.The response to injury in the peripheral nervous system. J Bone Joint Surg Br. 87:1309–1319. doi: 10.1302/0301-620X.87B10.16700. [DOI] [PubMed] [Google Scholar]
- 15.Hichor M, Sampathkumar NK, Montanaro J, Borderie D, Petit PX, Gorgievski V, Tzavara ET, Eid AA, Charbonnier F, Grenier J, Massaad C. Paraquat induces peripheral myelin disruption and locomotor defects: crosstalk with LXR and Wnt pathways. Antioxid Redox Signal. 2017;27:168–183. doi: 10.1089/ars.2016.6711. [DOI] [PubMed] [Google Scholar]
- 16.Höke A, Brushart T. 2010.Introduction to special issue: challenges and opportunities for regeneration in the peripheral nervous system. Exp Neurol. 223:1–4. doi: 10.1016/j.expneurol.2009.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Johnson AN, Mokalled MH, Valera JM, Poss KD, Olson EN. Post-transcriptional regulation of myotube elongation and myogenesis by Hoi Polloi. Development. 2013;140:3645–3656. doi: 10.1242/dev.095596. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Le BJ, LaCorbiere M, Powell HC, Ellisman MH, Schubert D. Extracellular fluid conditioned during peripheral nerve regeneration stimulates Schwann cell adhesion, migration and proliferation. Brain Res. 1988;459:93–104. doi: 10.1016/0006-8993(88)90289-2. [DOI] [PubMed] [Google Scholar]
- 19.Le GF, Rudnicki M. Satellite and stem cells in muscle growth and repair. Development. 2007;134:3953–3957. doi: 10.1242/dev.005934. [DOI] [PubMed] [Google Scholar]
- 20.Lee H, Lee SJ, Bae GU, Baek NI, Ryu JH. Canadine from Corydalis turtschaninovii stimulates myoblast differentiation and protects against myotube atrophy. Int J Mol Sci. 2017;18:e2748. doi: 10.3390/ijms18122748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lenox RH, Wang L. Molecular basis of lithium action: integration of lithium-responsive signaling and gene expression networks. Mol Psychiatry. 2003;8:135–144. doi: 10.1038/sj.mp.4001306. [DOI] [PubMed] [Google Scholar]
- 22.Lepper C, Partridge TA, Fan CM. An absolute requirement for Pax7-positive satellite cells in acute injury-induced skeletal muscle regeneration. Development. 2011;138:3639–3646. doi: 10.1242/dev.067595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Li Y, Gao Q, Yin G, Ding X, Hao J. WNT/β-catenin-signaling pathway stimulates the proliferation of cultured adult human Sertoli cells via upregulation of C-myc expression. Reprod Sci. 2012;19:1232–1240. doi: 10.1177/1933719112447126. [DOI] [PubMed] [Google Scholar]
- 24.Litwiniuk A, Pijet B, Pijet-Kucicka M, Gajewska M, Pająk B, Orzechowski A. FOXO1 and GSK-3β are main targets of insulin-mediated myogenesis in c2c12 muscle cells. PLoS One. 2016;11:e0146726. doi: 10.1371/journal.pone.0146726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Makoukji J, Belle M, Meffre D, Stassart R, Grenier J, Shackleford G, Fledrich R, Fonte C, Branchu J, Goulard M, de Waele C, Charbonnier F, Sereda MW, Baulieu EE, Schumacher M, Bernard S, Massaad C. Lithium enhances remyelination of peripheral nerves. Proc Natl Acad Sci U S A. 2012;109:3973–3978. doi: 10.1073/pnas.1121367109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Makoukji J, Shackleford G, Meffre D, Grenier J, Liere P, Lobaccaro JM, Schumacher M, Massaad C. Interplay between LXR and Wnt/β-catenin signaling in the negative regulation of peripheral myelin genes by oxysterols. J Neurosci. 2011;31:9620–9629. doi: 10.1523/JNEUROSCI.0761-11.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Meffre D, Massaad C, Grenier J. Lithium chloride stimulates PLP and MBP expression in oligodendrocytes via Wnt/β-catenin and Akt/CREB pathways. Neuroscience. 2015;284:962–971. doi: 10.1016/j.neuroscience.2014.10.064. [DOI] [PubMed] [Google Scholar]
- 28.Moimas S, Novati F, Ronchi G, Zacchigna S, Fregnan F, Zentilin L, Papa G, Giacca M, Geuna S, Perroteau I, Arnež ZM, Raimondo S. Effect of vascular endothelial growth factor gene therapy on post-traumatic peripheral nerve regeneration and denervation-related muscle atrophy. Gene Ther. 2013;20:1014–1021. doi: 10.1038/gt.2013.26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Montarras D, Morgan J, Collins C, Relaix F, Zaffran S, Cumano A, Partridge T, Buckingham M. Direct isolation of satellite cells for skeletal muscle regeneration. Science. 2005;309:2064–2067. doi: 10.1126/science.1114758. [DOI] [PubMed] [Google Scholar]
- 30.Méjat A, Ramond F, Bassel-Duby R, Khochbin S, Olson EN, Schaeffer L. Histone deacetylase 9 couples neuronal activity to muscle chromatin acetylation and gene expression. Nat Neurosci. 2005;8:313–321. doi: 10.1038/nn1408. [DOI] [PubMed] [Google Scholar]
- 31.Niu W, Bilan PJ, Yu J, Gao J, Boguslavsky S, Schertzer JD, Chu G, Yao Z, Klip A. PKCε regulates contraction-stimulated GLUT4 traffic in skeletal muscle cells. J Cell Physiol. 2011;226:173–180. doi: 10.1002/jcp.22320. [DOI] [PubMed] [Google Scholar]
- 32.Ohnishi A, Chua CL, Kuroiwa Y. Axonal degeneration distal to the site of accumulation of vesicular profiles in the myelinated fiber axon in experimental isoniazid neuropathy. Acta Neuropathol. 1985;67:195–200. doi: 10.1007/BF00687801. [DOI] [PubMed] [Google Scholar]
- 33.Otto A, Schmidt C, Luke G, Allen S, Valasek P, Muntoni F, Lawrence-Watt D, Patel K. Canonical Wnt signalling induces satellite-cell proliferation during adult skeletal muscle regeneration. J Cell Sci. 2008;121:2939–2950. doi: 10.1242/jcs.026534. [DOI] [PubMed] [Google Scholar]
- 34.Pesini P, Kopp J, Wong H, Walsh JH, Grant G, Hökfelt T. An immunohistochemical marker for Wallerian degeneration of fibers in the central and peripheral nervous system. Brain Res. 1999;828:41–59. doi: 10.1016/s0006-8993(99)01283-4. [DOI] [PubMed] [Google Scholar]
- 35.Polesskaya A, Seale P, Rudnicki MA. Wnt signaling induces the myogenic specification of resident CD45+ adult stem cells during muscle regeneration. Cell. 2003;113:841–852. doi: 10.1016/s0092-8674(03)00437-9. [DOI] [PubMed] [Google Scholar]
- 36.Rajasekaran MR, Kanoo S, Fu J, Nguyen ML, Bhargava V, Mittal RK. Age-related external anal sphincter muscle dysfunction and fibrosis: possible role of Wnt/β-catenin signaling pathways. Am J Physiol Gastrointest Liver Physiol. 2017;313:G581–588. doi: 10.1152/ajpgi.00209.2017. [DOI] [PubMed] [Google Scholar]
- 37.Scheib J, Höke A. Advances in peripheral nerve regeneration. Nat Rev Neurol. 2013;9:668–676. doi: 10.1038/nrneurol.2013.227. [DOI] [PubMed] [Google Scholar]
- 38.Schmitt AB, Breuer S, Liman J, Buss A, Schlangen C, Pech K, Hol EM, Brook GA, Noth J, Schwaiger FW. Identification of regeneration-associated genes after central and peripheral nerve injury in the adult rat. BMC Neurosci. 2003;4:8. doi: 10.1186/1471-2202-4-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sharma RP, Chopra VL. Effect of the Wingless (wg1) mutation on wing and haltere development in Drosophila melanogaster. Dev Biol. 1976;48:461–465. doi: 10.1016/0012-1606(76)90108-1. [DOI] [PubMed] [Google Scholar]
- 40.Shim S, Ming GL. Roles of channels and receptors in the growth cone during PNS axonal regeneration. Exp Neurol. 2010;223:38–44. doi: 10.1016/j.expneurol.2009.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Sorci G, Riuzzi F, Agneletti AL, Marchetti C, Donato R. S100B inhibits myogenic differentiation and myotube formation in a RAGE-independent manner. Mol Cell Biol. 2003;23:4870–4881. doi: 10.1128/MCB.23.14.4870-4881.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tawk M, Makoukji J, Belle M, Fonte C, Trousson A, Hawkins T, Li H, Ghandour S, Schumacher M, Massaad C. Wnt/beta-catenin signaling is an essential and direct driver of myelin gene expression and myelinogenesis. J Neurosci. 2011;31:3729–3742. doi: 10.1523/JNEUROSCI.4270-10.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Teraoka M, Hato N, Takahashi H, Komobuchi H, Sawai N, Okada M, Hakuba N. Myogenin expression in facial muscle following damage to the facial nerve. Acta Otolaryngol. 2012;132:783–787. doi: 10.3109/00016489.2011.653671. [DOI] [PubMed] [Google Scholar]
- 44.Torii T, Yamauchi J. Gas6-Tyro3 signaling is required for Schwann cell myelination and possible remyelination. Neural Regen Res. 2016;11:215–216. doi: 10.4103/1673-5374.177714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Verhees KJ, Pansters NA, Baarsma HA, Remels AH, Haegens A, de Theije CC, Schols AM, Gosens R, Langen RC. Pharmacological inhibition of GSK-3 in a guinea pig model of LPS-induced pulmonary inflammation: II. Effects on skeletal muscle atrophy. Respir Res. 2013;14:117. doi: 10.1186/1465-9921-14-117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Wagner R, Heckman HM, Myers RR. Wallerian degeneration and hyperalgesia after peripheral nerve injury are glutathione-dependent. Pain. 1998;77:173–179. doi: 10.1016/S0304-3959(98)00091-8. [DOI] [PubMed] [Google Scholar]
- 47.Wang Y, Zhao Y, Sun C, Hu W, Zhao J, Li G, Zhang L, Liu M, Liu Y, Ding F, Yang Y, Gu X. Chitosan degradation products promote nerve regeneration by stimulating schwann cell proliferation via miR-27a/FOXO1 axis. Mol Neurobiol. 2016;53:28–39. doi: 10.1007/s12035-014-8968-2. [DOI] [PubMed] [Google Scholar]
- 48.Wang Z, Zhang P, Kou Y, Yin X, Han N, Jiang B. Hedysari extract improves regeneration after peripheral nerve injury by enhancing the amplification effect. PLoS One. 2013;8:e67921. doi: 10.1371/journal.pone.0067921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Wen J, Qian C, Pan M, Wang X, Li Y, Lu Y, Zhou Z, Yan Q, Li L, Liu Z, Wu W, Guo J. Lentivirus-mediated rna interference targeting RhoA slacks the migration, proliferation, and myelin formation of schwann cells. Mol Neurobiol. 2017;54:1229–1239. doi: 10.1007/s12035-016-9733-5. [DOI] [PubMed] [Google Scholar]
- 50.Willand MP, Holmes M, Bain JR, de Bruin H, Fahnestock M. Sensory nerve cross-anastomosis and electrical muscle stimulation synergistically enhance functional recovery of chronically denervated muscle. Plast Reconstr Surg. 2014;134:736e, 745e. doi: 10.1097/PRS.0000000000000599. [DOI] [PubMed] [Google Scholar]
- 51.Xin ZT, Liu XZ, Zhang MJ, Li W, Chen BH, Ma XX. Exogenous cardiotrophin-1:The possibility to protect PC12 cells and Schwann cells. Zhongguo Zuzhi Gongcheng Yanjiu. 2013;17:7265–7271. [Google Scholar]
- 52.Yi S, Wang QH, Zhao LL, Qin J, Wang YX, Yu B, Zhou SL. miR-30c promotes Schwann cell remyelination following peripheral nerve injury. Neural Regen Res. 2017;12:1708–1715. doi: 10.4103/1673-5374.217351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zhang W, Fang X, Zhang C, Li W, Wong WM, Xu Y, Wu W, Lin J. Transplantation of embryonic spinal cord neurons to the injured distal nerve promotes axonal regeneration after delayed nerve repair. Eur J Neurosci. 2017;45:750–762. doi: 10.1111/ejn.13495. [DOI] [PubMed] [Google Scholar]
- 54.Zhao A, Yang L, Ma K, Sun M, Li L, Huang J, Li Y, Zhang C, Li H, Fu X. Overexpression of cyclin D1 induces the reprogramming of differentiated epidermal cells into stem cell-like cells. Cell Cycle. 2016;15:644–653. doi: 10.1080/15384101.2016.1146838. [DOI] [PMC free article] [PubMed] [Google Scholar]