Abstract
Context
Naloxone distribution has historically been implemented in a community-based, expanded public health model; however, there is now a need to further explore primary care clinic-based naloxone delivery to effectively address the nationwide opioid epidemic.
Objective
To create a general medicine infrastructure to identify patients with high-risk opioid use and provide 25% of this population with naloxone autoinjector prescription and training within a 6-month period.
Design
The quality improvement study was conducted at an outpatient clinic serving 1238 marginally housed veterans with high rates of comorbid substance use and mental health disorders. Patients at high risk of opioid-related adverse events were identified using the Stratification Tool for Opioid Risk Management and were contacted to participate in a one-on-one, 15-minute, hands-on naloxone training led by nursing staff.
Main Outcome Measures
The number of patients identified at high risk and rates of naloxone training/distribution.
Results
There were 67 patients identified as having high-risk opioid use. None of these patients had been prescribed naloxone at baseline. At the end of the intervention, 61 patients (91%) had been trained in the use of naloxone. Naloxone was primarily distributed by licensed vocational nurses (42/61, 69%).
Conclusion
This study demonstrates the feasibility of high-risk patient identification and of a primary care-based and nursing-championed naloxone distribution model. This delivery model has the potential to provide access to naloxone to a population of patients with opioid use who may not be engaged in mental health or specialty care.
INTRODUCTION
Deaths caused by opioid overdose— both heroin and prescription opioids—have tripled in the US since 2010, and opioid use has now formally been recognized as a national epidemic.1,2 Veterans have high rates of pain and mental health disorders, such as posttraumatic stress disorder, as well as concomitant benzodiazepine and opioid use, placing them at particularly high risk of opioid-related accidents and overdose.3,4 As such, veterans are estimated to die of opioid overdose at twice the national average rate.5
Naloxone is an opioid antagonist previously used primarily in hospital settings to reverse fatal respiratory depression owing to opiate overdose. The escalation of opioid overdoses has led community organizations to distribute naloxone widely among individuals with high-risk use.6–8 In 2014, the Department of Veterans Affairs (VA) implemented the Opioid Overdose Education and Naloxone Distribution (OEND) program, which aims to provide veterans with naloxone and trains them to administer it properly. Despite the implementation of this program, no formal workflow procedures existed in our VA Community-Based Outpatient Clinic for the distribution of naloxone kits, and no kits had been distributed to high-risk patients in our clinic before October 2015.
With the input of key clinic stakeholders, a multidisciplinary team composed of internal medicine residents, a psychology trainee, registered nurses (RNs), and licensed vocational nurses (LVNs) set the goal of increasing both the distribution of naloxone autoinjectors and training in their appropriate use to our high-risk veteran population. The primary aim of the project was to develop the infrastructure to provide naloxone kits and training to 25% of patients at high-risk of opioid-related adverse events within 6 months. The objective was selected with consideration of attainability given the finite period of the resident quality improvement program.
METHODS
Setting
The study was conducted at an outpatient clinic engaging marginally housed veterans (defined as those who are without housing or are living in transitional living programs or in single-room occupancy units) who have high rates of comorbid substance use and mental health disorders. The clinic provides comprehensive services, including medical, mental health, social work, and substance use treatment, as well as housing referrals for veterans. The medical staff includes five primary care practitioners (PCPs), six internal medicine residents, three RNs, and three LVNs.
Planning the Intervention
The planning phase of this work began in October 2015. At that time, no patients with high-risk opioid use had been prescribed naloxone. This quality intervention was deemed exempt from the need for institutional review board approval.
The implementation phase took place from November 2015 to March 2016. There were three key elements of the planned intervention. The first was the identification of patients with high-risk opiate use. This identification was completed using a national VA dashboard known as the Stratification Tool for Opioid Risk Management (STORM) in which real-time, patient-specific data regarding comorbidities, mental health, substance use, and opioid/sedating medication use are utilized to determine three-year risk of opioid-related adverse outcomes.9
The second phase of this intervention was the education of clinic staff (LVNs, RNs, and physicians) about counseling patients on naloxone use. This was initially done through 30- to 60-minute, in-person training sessions led by a medical center pharmacist dedicated to the OEND project.10,11 There were no formal posttraining assessments performed to evaluate competency; some nursing staff did elect to be directly observed by a physician during their first patient counseling experience. Reinforcement education was done through online educational videos.
The final element of this intervention was the distribution of naloxone to patients. In this phase, high-risk patients were contacted using a tiered and multifaceted approach. A champion LVN attempted contact with identified patients first by telephone and then by letter, inviting them to the clinic for safety education regarding their opioid medication or opioid recreational use. For patients unable to be engaged by these methods, in-person contact was attempted while the patient was at the clinic for either medical or psychosocial services. To identify when these patients would be physically present in the clinic, a portion of the medical team “huddle” each morning was dedicated to reviewing the clinic schedule for high-risk patients.
The patient training consisted of 15-minute verbal discussion of signs of opioid overdose, administration of naloxone, and the importance of contacting emergency services.10 If additional training was needed, patients were referred to a training video.10 Primary supports for patients were also invited to attend; however, given the social isolation of this patient population, it was rare for an individual other than the patient to participate.
The initial Plan-Do-Study-Act (PDSA) cycle included the quantitative (time/session, use of media) and qualitative (subjective assessment of educator comfort, patient receptiveness) evaluation of several patient education encounters. From this study, we found it easier to engage patients in the conversation about naloxone if we normalized the use of naloxone as a medication (ie, comparison to epinephrine injection [EpiPen]) and emphasized its role in aiding other social contacts such friends or family members who use opioids (ie, service to others). This content was then added to the outline of the naloxone training discussions. To aid our LVN staff in providing comprehensive education, we developed an automated clinic note template, which was completed by the trainer during the session.
To minimize prescriber/physician effort, the staff member providing the training was able to order the naloxone kit under the supervising prescriber’s name, thereby allowing the provision of the naloxone autoinjector to the patient at the time of training.
Method of Evaluation
We tabulated the number of patients identified via the STORM tool as high-risk users at baseline and subsequently tracked the number of patients prescribed naloxone on a monthly basis. These data were provided to frontline staff as well as the ambulatory clinic leadership in the form of e-mail and in verbal huddle communication. We additionally tracked the type of provider (eg, physician, RN, LVN) who distributed each naloxone kit. Finally, we documented the patients who were not prescribed naloxone as well as the underlying reason (eg, failure to contact, patient refusal of naloxone).
RESULTS
There were 67 patients identified as having high-risk opioid use, and none of these patients had been prescribed naloxone at the beginning of the intervention (0%). At the end of the intervention period, 61 patients (91%) had been provided with and trained in the use of naloxone (Figure 1). The naloxone was primarily distributed to patients by LVNs (42/61, 69%), with the remainder distributed by RNs (16/61, 26%) and physicians (3/61, 5%).
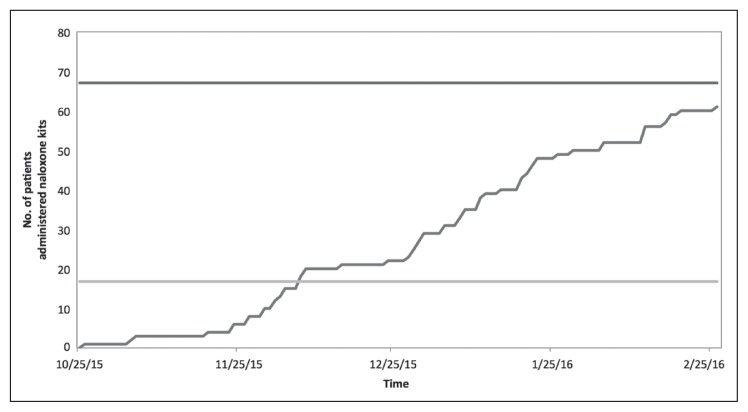
Figure 1.
Naloxone distribution over time. Demonstrates the number of patients who received naloxone training and prescription during the course of the intervention (November 2015-March 2016). There were 67 patients identified at high risk (upper black line). The goal was to administer naloxone to at least 25% of these patients (n = 17), as demonstrated by the lower gray line. This goal was achieved in November 2015, and 61 patients (91%) were prescribed naloxone at the end of the intervention in March 2016.
Of the 6 patients (9%) who were not prescribed naloxone, 3 patients declined the medication/training for undisclosed reasons, 2 patients were no longer receiving their care at our clinic, and 1 could not be contacted by telephone or letter and had not yet returned to the clinic.
DISCUSSION
Naloxone training and distribution has historically been implemented in a community-based expanded public health model, in which naloxone is distributed to those who use opioids as well as to potential bystanders (defined as those who may be present for an overdose event but not personally experiencing such).12,13 Although this model has shown efficacy, the reach and impact of naloxone training and distribution could be extended substantially by involving primary care clinics and clinicians. This general practice model of distribution is uncommon and little explored because of, in part, the inherent challenges of conducting naloxone training and distribution in the primary care setting. Barriers that have been cited include lack of practitioner time, lack of practitioner knowledge, lack of generalist ownership (ie, belief that specialists should manage substance-use disorder), and stigma surrounding opioid use.14,15
We describe an innovative, primary care-based quality improvement initiative that increased naloxone distribution and training to a veteran population at high risk of opioid-related adverse events. There were several unique features of this intervention that addressed previously cited barriers and contributed to its success. The first of these barriers was practitioner time, which was addressed both in the patient identification and patient training process. This study demonstrated the feasibility of using electronic medical record tools to identify and prioritize patients at high risk of adverse events related to opioid use. We also demonstrated that a patient identification and naloxone training process need not be championed by a physician but, rather, could be effectively implemented by nursing or pharmacy members of clinic teams after appropriate training. A previous qualitative study of general practitioners demonstrated a significant portion that preferred a model of naloxone delivery that engages nonphysician members of the multidisciplinary team, as our study did.14
This intervention also addressed the perceived barrier of practitioner knowledge. Our clinical team endorsed comfort with naloxone training, as measured by binary assessment of willingness to provide naloxone counseling, after a single 30-minute, pharmacist-led session with supplemental online educational videos. Given the brevity of this clinician training, it is feasible to implement during routine all-clinic team meetings and does not require additional time away from clinic.
Finally, although there are substance use treatment services accessible at our Community-Based Outpatient Clinic, there was no involvement by mental health or substance use specialists. This demonstrates the feasibility of primary care-based programming and addresses the historical lack of generalist ownership.
Sustainability
The Centers for Disease Control and Prevention now recommends that naloxone be prescribed to patients with a history of substance-use disorder, prior overdose, high-dose opioid prescriptions, and/or concurrent benzodiazepine prescription.16 To adhere to this recommendation, clinics must develop a process for prescribing naloxone to patients.
Although the workflow we have described was effective in providing naloxone training to existing patients with opioid use, separate and sustainable processes were concurrently developed for patients who are newly prescribed opioids. To ensure the sustainability of naloxone prescribing, our VA medical center now embeds naloxone orders within opioid medication electronic order sets. Practitioners are also prompted to prescribe naloxone and provide counseling when initiating opiate therapy of any kind and for any duration. This model is supported by a recent nonrandomized intervention in safety-net primary care clinics demonstrating its feasibility and association with a 63% reduction in opioid-related Emergency Department visits at 1 year.17
Limitations
Our project demonstrates the ability to engage patients in naloxone training but studied only the process measure of naloxone kit distribution. Although we may extrapolate the benefit of this medication from other literature, outcome measures such as patient knowledge and skills with naloxone administration and the number of opioid-related adverse events, Emergency Department visits, and deaths among those administered a naloxone kit were not measured. The generalizability of our findings may also be limited because of our clinic’s concentration on marginally housed veterans with high rates of comorbid substance use and mental health disorders and who have a close connection with their primary care home given the comprehensive services provided. Additionally, the STORM tool used to identify high-risk patients requires an electronic medical record and is validated for veterans but not for other patients.
CONCLUSION
It is feasible to distribute to and to educate high-risk patients on naloxone use in primary care clinics to reduce opioid-related adverse events. When clinical pharmacists and nurses deliver patient naloxone counseling, it requires minimal physician time. Ultimately, this type of delivery model can increase the number of patients receiving naloxone who are not engaged in mental health or other specialty care.
Acknowledgments
This work was funded by the US Department of Veterans Affairs (VA), Office of Academic Affiliations, as a VA Center of Excellence in Primary Care Education.
The authors would like to thank the San Francisco Downtown Clinic staff as well as Medical Director Amy Noack, MD, for their commitment to the naloxone distribution efforts.
Kathleen Louden, ELS, of Louden Health Communications provided editorial assistance.
Footnotes
Disclosure Statement
The author(s) have no conflicts of interest to disclose.
References
- 1.Jones CM, Logan J, Gladden RM, Bohm MK. Vital Signs: Demographic and substance use trends among heroin users—United States, 2002–2013. MMWR Morb Mortal Wkly Rep. 2015 Jul 10;64(26):719–25. [PMC free article] [PubMed] [Google Scholar]
- 2.Bohnert AS, Valenstein M, Bair MJ, et al. Association between opioid prescribing patterns and opioid overdose-related deaths. JAMA. 2011 Apr 6;305(13):1315–21. doi: 10.1001/jama.2011.370. DOI: https://doi.org/10.1001/jama.2011.370. [DOI] [PubMed] [Google Scholar]
- 3.Seal KH, Shi Y, Cohen G, et al. Association of mental health disorders with prescription opioids and high-risk opioid use in US veterans of Iraq and Afghanistan. JAMA. 2012 Mar 7;307(9):940–7. doi: 10.1001/jama.2012.234. DOI: https://doi.org/10.1001/jama.2012.234. [DOI] [PubMed] [Google Scholar]
- 4.Park TW, Saitz R, Ganoczy D, Ilgen MA, Bohnert AS. Benzodiazepine prescribing patterns and deaths from drug overdose among US veterans receiving opioid analgesics: Case-cohort study. BMJ. 2015 Jun 10;350:h2698. doi: 10.1136/bmj.h2698. DOI: https://doi.org/10.1136/bmj.h2698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bohnert AS, Ilgen MA, Galea S, McCarthy JF, Blow FC. Accidental poisoning mortality among patients in the Department of Veterans Affairs Health System. Med Care. 2011 Apr;49(4):393–6. doi: 10.1097/MLR.0b013e318202aa27. DOI: https://doi.org/10.1097/mlr.0b013e318202aa27. [DOI] [PubMed] [Google Scholar]
- 6.Clark AK, Wilder CM, Winstanley EL. A systematic review of community opioid overdose prevention and naloxone distribution programs. J Addict Med. 2014 May-Jun;8(3):153–63. doi: 10.1097/ADM.0000000000000034. DOI: https://doi.org/10.1097/adm.0000000000000034. [DOI] [PubMed] [Google Scholar]
- 7.Enteen L, Bauer J, McLean R, et al. Overdose prevention and naloxone prescription for opioid users in San Francisco. J Urban Health. 2010 Dec;87(6):931–41. doi: 10.1007/s11524-010-9495-8. DOI: https://doi.org/10.1007/s11524-010-9495-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Galea S, Worthington N, Piper TM, Nandi VV, Curtis M, Rosenthal DM. Provision of naloxone to injection drug users as an overdose prevention strategy: Early evidence from a pilot study in New York City. Addict Behav. 2006 May;31(5):907–12. doi: 10.1016/j.addbeh.2005.07.020. DOI: https://doi.org/10.1016/j.addbeh.2005.07.020. [DOI] [PubMed] [Google Scholar]
- 9.Oliva EM, Bowe T, Tavakoli S, et al. Development and application of the Veterans Health Administration’s Stratification Tool for Opioid Risk Mitigation (STORM) to improve opioid safety and prevent overdose and suicide. Psychol Serv. 2017 Feb;14(1):34–49. doi: 10.1037/ser0000099. DOI: https://doi.org/10.1037/ser0000099. [DOI] [PubMed] [Google Scholar]
- 10.Pharmacy Benefits Management Services. Academic detailing service—opioid overdose education & naloxone distribution (OEND) [Internet] Washington, DC: US Department of Veterans Affairs; updated 2017 Feb 9 [cited 2017 Sept 13]. Available from: www.pbm.va.gov/AcademicDetailingService/Opioid_Overdose_Education_and_Naloxone_Distribution.asp. [Google Scholar]
- 11.Oliva EM. Opioid overdose education and naloxone distribution (OEND): Preventing and responding to an opioid overdose [presentation] [Internet] Palo Alto, CA: Veterans Affairs Program Evaluation and Resource Center; 2014. Sep 2, [cited 2018 Jan 26]. Available from: www.hsrd.research.va.gov/for_researchers/cyber_seminars/archives/868-notes.pdf. [Google Scholar]
- 12.Centers for Disease Control and Prevention (CDC) Community-based opioid overdose prevention programs providing naloxone—United States, 2010. MMWR Morb Mortal Wkly Rep. 2012 Feb 17;61(6):101–5. [PMC free article] [PubMed] [Google Scholar]
- 13.Walley AY, Xuan Z, Hackman HH, et al. Opioid overdose rates and implementation of overdose education and nasal naloxone distribution in Massachusetts: Interrupted time series analysis. BMJ. 2013 Jan 30;346:f174. doi: 10.1136/bmj.f174. DOI: https://doi.org/10.1136/bmj.f174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Matheson C, Pflanz-Sinclair C, Aucott L, et al. Reducing drug related deaths: A pre-implementation assessment of knowledge, barriers and enablers for naloxone distribution through general practice. BMC Fam Pract. 2014 Jan 15;15:12. doi: 10.1186/1471-2296-15-12. DOI: https://doi.org/10.1186/1471-2296-15-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Winstanley EL, Clark A, Feinberg J, Wilder CM. Barriers to implementation of opioid overdose prevention programs in Ohio. Subst Abus. 2016;37(1):42–6. doi: 10.1080/08897077.2015.1132294. DOI: https://doi.org/10.1080/08897077.2015.1132294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain—United States, 2016. JAMA. 2016 Apr 19;315(15):1624–45. doi: 10.1001/jama.2016.1464. DOI: https://doi.org/10.1001/jama.2016.1464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Coffin PO, Behar E, Rowe C, et al. Nonrandomized intervention study of naloxone coprescription for primary care patients receiving long-term opioid therapy for pain. Ann Intern Med. 2016 Aug 16;165(4):245–52. doi: 10.7326/M15-2771. DOI: https://doi.org/10.7326/m15-2771. [DOI] [PMC free article] [PubMed] [Google Scholar]