Abstract
There were three epoch making events in therapy of varicose veins. The first one is that the endovascular heat ablation (EVHA) using diode laser was authorized by the Ministry of Health Labor and Welfare in January 2011. The second one is that Subfascial Endoscopic Perforator Surgery (SEPS) was also authorized in April 2014. All of the therapies which were covered by the national insurance system had been the procedures for superficial veins but the SEPS is procedure for the perforating veins. The third one is that the foam usage of Polidocasklerol was listed formally at the medical package insert in September 2016. Moreover stub avulsion was introduced as figure-related improvement method with a smaller operation wound instead of conventional varicectomy and the other existing therapies are progressing every day. Therefore, by this education seminar, I lecture mainly on the EVHA, SEPS and foam sclerotherapy in varicose vein treatment. Finally I show one case which you should remember. (This is a translation of Jpn J Vasc Surg 2017; 26: 225–230.)
Keywords: varicose vein, laser ablation, SEPS, sclerotherapy
Introduction
Sclerotherapy for varicose veins gained popularity in the 1990s and many sclerotherapy agents, such as osmotic agents, hypertonic saline, and hypertonic glucose solution, and detergent sclerosing agents, such as hospital preparations, have since then been developed and used. In 2006, the Ministry of Health, Labour and Welfare approved the currently widely used Polidocasklerol. Following Western countries, the preferred formulation of Polidocasklerol rapidly shifted from liquid to foam in the 2000s,1–7) but information on Polidocasklerol use as foam was only added to package inserts in September 2016. Foamed sclerotherapy agent injected intravenously requires attention in terms of techniques of injection, position, and compression, which differ from those for the conventional liquid sclerotherapy agent. With regard to endovascular heat ablation (EVHA), laser therapy and radiofrequency ablation are rapidly gaining popularity nationwide as treatments for varicose veins under the management of the Japanese Committee for Endovenous Laser Treatment. With the spread of this surgical technique arose new heat-related complications such as neuropathy and deep vein thrombosis, which differ from the complications of stripping that has been conventionally selected as a procedure for the treatment of the main trunk of the saphenous vein. Furthermore, the subfascial endoscopic perforator surgery (SEPS), in which perforating vein reflux located in parts of the legs affected by stasis dermatitis and ulceration is dissected endoscopically, has been covered by national insurance since April 2014. All treatments that have been conventionally covered by insurance are methods to treat superficial venous valve insufficiencies, whereas SEPS is a treatment for the incompetent perforating vein (IPV); thus, a facility must be certified to perform this procedure under national insurance coverage (Table 1).
Table 1 Surgical procedures for treatment of venous valve insufficiency.
| 〈Superficial vein system〉 |
| ・Stripping, High Ligation, Varicectomy |
| ・Sclerotherapy |
| ・Endovascular Heat Ablation (Diode Laser 980 nm/1470 nm, Radio Frequency Ablation) |
| 〈Deep vein system〉 |
| ・Valvuloplasty, valvuloconstruction, valve transplantation, venous transposition |
| 〈Perforating vein system〉 |
| ・SEPS (Subfascial Endoscopic Perforator Surgery), Linton’s operation, PAPs (Percutaneous Ablation of Perforators) |
The methods shown by underlines are covered by the national insurance system. Conventionally, all procedures authorized by the Ministry of Health Labor and Welfare were for the insufficiency of superficial venous system, whereas SEPS is chosen for the incompetency of perforating vein valve function.
Besides these treatments, continuous progress is seen in conventional surgical methods as well. For example, the stub avulsion, which has improved esthetic outcomes by making a smaller surgical wound than conventional varicectomy, has been developed. Furthermore, tumescent local anesthesia (TLA) introduced for attenuating pain and bleeding accompanying the extraction of the main trunk of the saphenous vein in the stripping procedure has become essential in EVHA. Developments in TLA and various compression therapies have made day surgeries possible for the treatment of varicose veins today. Here, we have described foamed sclerotherapy compared with liquid sclerotherapy, complications specific to EVHA, and current status of SEPS.
Surgical Therapy
EVHA
In 2011, EVHA using a 980 nm wavelength diode laser was approved for coverage by national insurance, followed by the additional approval of a 1,470 nm wavelength diode laser and radiofrequency ablation (RFA) in 2014. Differences in diode laser wavelengths primarily affect whether the energy of diode laser is absorbed by blood (hemoglobin) or water. Given that 980 nm laser waves are absorbed by hemoglobin, the blood in the blood vessels is heated by the laser waves, and this heat damages the inner wall of nearby blood vessels. On the contrary, laser wavelengths ≥1,000 nm are easily absorbed by water; thus, the heat generated by the laser acts directly on the water component of the vascular walls and constricts and obliterates the vascular wall. Methods of irradiation have also improved with changes in laser wavelengths. Laser at 980 nm wavelengths uses bare-tip fibers in which the laser is emitted only forward in a straight line from the laser fiber tip, making it difficult to irradiate the inner wall of the vein evenly. In contrast, 1,470 nm laser fiber, also known as a radial ring fiber, enables even irradiation of the inner wall of the vein with a laser beam that irradiates radially from the fiber tip. Compared with the 980 nm laser, venous wall perforations decreased in frequency using the 1,470 nm laser, which also reduced subdermal bleeding and postoperative pain associated with ablation8) (Fig. 1). The surgical method using RFA is similar to laser ablation. It is essential to ensure sufficient contact between the heat-generating catheter tip and vascular wall and to squeeze the vascular lumen by echography-guided TLA before ablation and to exert pressure on the vessel with the echography probe during ablation to adhere the heat-generating part of the catheter to the venous wall. The starting point of ablation, as indicated in the EVHA training textbook, is directly under the vena epigastrica superficialis branch or 1–2 cm peripheral to the saphenofemoral junction (SFJ) considering surgical methods using the bare-tip fiber. A point closer to the SFJ is considered adequate for ablation with the radial ring fiber, which is frequently used in current laser treatments. With changes in the equipment used, guidelines must be modified accordingly.
Fig. 1 Bare tip fiber and radial ring fiber. Bare tip fiber: A laser is irradiated only forward. The direct contact between the unevenly placed heated tip and the vessel wall results in heterogeneous heating of the vein and perforation. Radial (2) ring fiber: A laser is irradiated in entire circumference via prism, and uniform irradiation prevent the perforation of vein wall. The bottom figure is called radial 2 ring fiber with structure to irradiate from two places, but there is 1 ring fiber to irradiate from one place. About the direction of the irradiation light, there are right angle for fiber and slant ahead too.
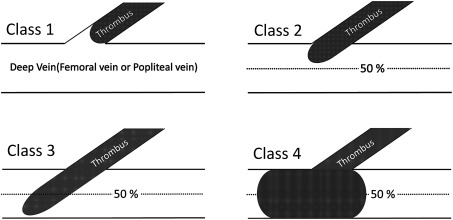
Deep vein thrombosis seen in EVHA is known as endovenous heat-induced thrombosis (EHIT) that results from progression of thrombosis in the femoral vein or popliteal vein secondary to thrombosis in SFJ or the saphenopopliteal junction (SPJ), which is rare but can cause fatal pulmonary thromboembolism. Therefore, when an EHIT ≥Class 3 is identified, drug therapy is recommended.9–14) Anti-Xa inhibitors known as direct oral anticoagulants are also covered by insurance for the treatment of deep vein thrombosis since 2014, permitting their use in outpatient treatment of cases for which antithrombotic therapy during hospitalization was previously the only treatment available15) (Fig. 2).
Fig. 2 Endovenous heat-induced thrombosis (EHIT) classification. Class 1: Venous thrombosis to superficial deep junction (SFJ of SPJ), but not extending into deep system. Class 2: Non-occlusive venous thrombosis, with an extension into deep system of a cross sectional area less than 50%. Class 3: Non-occlusive venous thrombosis, with an extension into deep system of a cross sectional area greater than 50%. Class 4: Occlusive deep vein thrombosis of common femoral/popliteal vein.
A video of ultrasound findings has demonstrated potential neuropathy as another point of caution. Extraction of the main trunk of the great saphenous vein during its stripping can cause sensory disturbance in the medial side of the lower thigh because of saphenous nerve disorder and that sensory disturbance of the lateral side of the lower thigh can be caused by sural nerve disorder during extraction of the main trunk of the small saphenous vein. Both occur because of injury to the saphenous or sural nerves, which run along the main trunk of the saphenous vein. Heat from ablation during EVHA can also cause neuropathy. It is possible to identify the saphenous or sural nerves intraoperatively by ultrasound scanning, so nerve injury can be prevented by injecting the fluid to separate the wall of the target vein for ablation from the nerve fibers during TLA. Many anatomical variables of the influx site of SPJ, which is the junction between the small saphenous and popliteal veins, are well known, but what must be noted during EVA is that the tibial nerve runs close to SPJ. The tibial nerve controls plantar flexion of the ankle and sensations of the lateral malleolus and lateral aspect and sole of the foot; therefore, injury to this nerve can cause not only sensory disturbance of the lower thigh but also talipes calcaneus, a motor dysfunction of plantar flexion of the ankle. Tibial nerve dysfunction can be prevented by setting the starting point of ablation at a distance from SPJ, but if the veins close to SPJ are to be treated, intraoperative ultrasound scanning must be conducted to visualize SPJ and the tibial nerve bundle, and sufficient amounts of TLA must be administered to separate the nerve bundle from the small saphenous vein trunk (Table 2).
Table 2 Nerve injury during endovenous heat ablation.
| ・Short saphenous vein: |
| Tibial nerve injury (motor and sensory nerve) |
| Sural nerve injury (sensory nerve) |
| ・Great saphenous vein: |
| Saphenous nerve injury (sensory nerve) |
Of the treatments for lower extremity varicose veins, stripping has been selected for many cases as a treatment for the main trunk of the saphenous vein. In addition, several studies on long-term prognosis of 10 years or more for determining surgical outcomes were conducted. However, reports on long-term prognosis of EVHA are few.16,17) When stripping and high ligation are selected for treating the main branch of the great saphenous vein, the standard surgical method comprises resecting the entire SFJ side branch, including the superficial epigastric vein. In contrast, standard surgical methods of EVHA maintain blood flow of the superficial epigastric vein while treating SFJ given that one of its purposes is also EHIT prevention. Therefore, reports on long-term outcomes of EVHA, including the influences of the prevention of the superficial epigastric vein are anticipated.
SEPS
SEPS has been covered by insurance since April 2014. When varicose veins are left untreated for a long time period, progression of dilation of lower extremity veins, particularly veins in the thighs, can occur. This can further progress to dilation of intradermal capillaries peripheral to the ankle or stasis dermatitis and ulceration near the medial malleolus (C4b–C6) and eventually lead to rupture of the varicose vein. Therefore, some cases in which dermal lesions have progressed to C4b–C6 are accompanied by IPV of the site of the dermal lesion. When reflux occurs directly in the venous system superior to deep veins, intractable ulcer formation or bleeding from varicose vein rupture of the legs can occur.
Varicose veins that have progressed to C4b–C6 are at risk factors for new ulcer formation. Therefore, performing direct vein treatment at the site of the leg dermal lesion is difficult. This led to the development of SEPS, in which IPV is resected without surgical invasion to the skin lesion.18–20) Detailed description of the operative method will be omitted and supplemented by an illustration (Fig. 3). In brief, in this method, endoscopy is used to gain access through the normal skin for resection of IPV under the affected skin lesion. The surgical method illustrated in Fig. 3 is called the two-port system SEPS because the access ports are inserted from two points in this method. In addition, the one-port system SEPS is performed by inserting an SEPS-specialized device from one incision to the skin.21,22) The Japanese SEPS Study Group (JSEPS: http://jseps.sakura.ne.jp/db/), established in 2002, has been involved in spreading the use of this surgical method. “SEPS” is the title of the book published under supervision of JSEPS in February 2016 (Tokyo Houki Shuppan). JSEPS has been providing opportunities to observe operations and assistance in surgeries for the safe spread of this technique. This is a therapeutic option that should be learned and adopted by all facilities that specialize in varicose veins, but to perform SEPS under insurance-covered care, the facility must obtain certification from the Ministry of Health, Labour and Welfare.
Fig. 3 Schema of TPS-SEPS (two port system subfascial endoscopic perforator surgery). One port is for an endoscope, the other is for dissecting forceps and the ultrasonic coagulation and cutting device. All two ports are inserted at the normal skin without stasis dermatitis. We do SEPS without air tourniquet nor dissecting balloon catheter. The all devices we use are originally designed for laparoscopic surgery. UCCD: Ultrasonic Coagulation & Cutting Device; IPV: incompetent perforating vein.
Foamed sclerotherapy
Conventionally, undiluted sclerotherapy agent was injected after ejecting the blood from the target superficial veins. Methods of ejecting this blood include raising the affected limb, direct compression of the varicose vein, or air injection. In contrast, in foamed sclerotherapy, the sclerotherapy agent is mixed with a gas (air or medical carbon dioxide gas) and then injected as foam. Hence, blood is expulsed by the gas during the procedure so that the undiluted sclerotherapy agent comes into direct contact with the inner vascular epithelium to permit sufficient endothelial damage with low concentrations and low quantities of the sclerotherapy agent; this method provides the benefit of slow migration of the sclerosing agent so that it can be compressed relatively slowly following injection.1,3,4,6,7) The currently frequently used sclerosing agent is Polidocasklerol, which is available in 0.5%, 1%, and 3% concentrations that contain 10, 20, or 60 mg of polidocanol in each 2 mL ampule solution. Solutions of low concentration of both liquid and foamed Polidocasklerol are used for varicose veins of small diameters. Table 3 summarizes the warnings and characteristics of the liquid and foam preparations written in the package insert. Although there are no particular directions pertaining to the position for injection in using the sclerosing agent in liquid form, its use in foamed form requires elevation of the affected limb and slow administration of the sclerosing agent. Furthermore, compression and lower leg exercise are initiated immediately after administration of the sclerosing agent in liquid form, whereas compression immediately after administration is not recommended, and 2–5 min of rest with the elevated affected limb is recommended rather than exercising the leg immediately after administration of the foam. Moreover, there is a report on neuropathy involving paradoxical embolism when Polidocasklerol is used as foam, and a basic warning is given to confirm the presence or absence of patent foramen ovale prior to performing foamed sclerotherapy.
Table 3 Comparison between liquid sclerotherapy and foam sclerotherapy (citation from the medical package insert of Polidocasklerol with partial modification).
| Liquid sclerotherapy | Foam sclerotherapy |
|---|---|
| Inject Polidocasklerol slowly. (There are no instructions about leg position.) | Slowly inject the foam sclerosant by elevating the lower extremities. |
| After injecting Polidocasklerol, immediately compress the injection site using compression material, etc., and then apply elastic bandage or elastic stocking to achieve vascular endothelial adhesion. | After injecting the foam sclerosant, cover the injection site and avoid the exercise of lower extremities for 2 to 5 minutes and not to do Valsalva maneuver or muscle activities. |
| When compressing the injection site, avoid immediate compression and compress the injection site about 10 minutes after the injection in the treatment of great and small saphenous veins or about 5 minutes after the injection in the treatment of branch varicose veins, recurrent varicose veins or perforating veins using compression material, etc. | |
| If foam sclerotherapy is performed in patients with a history of severe cerebrovascular attack, pulmonary hypertension, or migraine with aura, confirm the presence of foramen ovale, etc., before performing foam sclerotherapy. | |
| Maximum daily dose: 1%; 60 kg 12.0 mL | Maximum daily dose: 1%; 60 kg 12.0 mL (<2 mg/kg) |
| 【warning】 1) Do not inject Polidocasklerol into an artery. There is no description about usage during an operation. | 【warning】 1) Do not inject Polidocasklerol into an artery. There is no description about usage during an operation. |
Here, it is important to take caution since 0.5% Polidocasklerol is not approved for use as foam, and the conventional liquid form is the only choice available for use on reticular and spider varicose veins with diameters <1 mm.
Treatments similar to sclerotherapy include “glue therapies,” which are in clinical use on varicose veins in Western countries. Glue therapies consist of obliteration of the main trunk of the saphenous vein using n-butyl cyanoacrylate (NBCA) or octyl cyanoacrylate (OCA), which are derived from cyanoacrylate superglue for medical use. These are introduced as new varicose vein treatments involving less pain for lower extremities.23, 24) Varicose vein therapies continue to be a topic of interest to study.
Case Presentation of a Memorable Young Patient with Varicose Veins
Case:
34-year-old man
Occupation:
Gasoline station worker
Medical history:
Multiple laparotomies for small bowel obstruction as a newborn.
History of the present illness:
Underwent full-length stripping on both greater saphenous veins for bilateral varicose veins diagnosed at the age of 25 years. Ulceration in the bilateral medial gaiter areas occurred at age 31. He underwent varicectomy and sclerotherapy at 10 medical institutions, but the ulcers could not be cured, and he was examined at our hospital on August 2014 (Fig. 4). Magnetic resonance venography revealed extensive occlusion of the deep vein system below the inferior vena cava, and venous reflux was observed through several collateral circulations of the veins of both legs. No vascular insufficiency in arteries of both legs was found (Fig. 5).
Fig. 4 Both leg findings at initial investigation. He had multiple former operation skin scars and ulcers with stasis dermatitis on his both legs. Pseudomonas aeruginosa and MSSA were detected at the ulcers by the bacterial culture.
Fig. 5 MR Venography findings & CT Angiogram findings. MR Venography (right side) showed the extensive occlude lesions of IVC, both iliac veins, femoral veins, popliteal veins, anterior & posterior tibial veins and fibular veins. CT Angiogram (left side) showed normal arterial flow to both legs.
Therapeutic course:
SEPS was performed in December 2014. The ulcers were cured, and use of compression therapy with elastic stockings has maintained the cured ulceration.
Discussion and conclusion:
Patients with history of surgery in infancy or childhood may have undergone venous “cut-down,” wherein the transfusion access is gained by exposing the femoral or axillary veins in the perioperative period, which may have undergone repeated puncture and insertion of venous catheters. In these cases, the deep venous systems of the limbs can be occluded and deteriorated, thereby causing secondary varicose veins. When varicose veins are found in young patients, it is important to run compulsory tests of the deep venous system, and surgical treatments must be selected with caution.
Conclusion
Combinations of various therapeutic methods are practiced in different institutions to achieve maximum outcomes with minimally invasive methods. However, the facility’s approval must be obtained to use SEPS in insurance-covered treatment, and there are limited options of treatment and facilities where these treatments can be performed for varicose vein cases that have progressed to stasis dermatitis and ulceration (C4b–C6).
Disclosure Statement
The author of this article has no conflicts of interest to declare.
This is a translation of Jpn J Vasc Surg 2017; 26: 225–230.
Additional Remarks
The content of this study was presented at the 25th Japanese Society of Vascular Surgery Educational Seminar held at the 1st floor auditorium of Phoenix Hall at the International Conference Center Hiroshima on April 21, 2017. Informed consent from the patients described in the case reports has been obtained.
References
- 1).Tessari L, Cavezzi A, Frullini A. Preliminary experience with a new sclerosing foam in the treatment of varicose veins. Dermatol Surg 2001; 27: 58-60. [PubMed] [Google Scholar]
- 2).Frullini A, Cavezzi A. Sclerosing foam in the treatment of varicose veins and telangiectases: history and analysis of safety and complications. Dermatol Surg 2002; 28: 11-5. [DOI] [PubMed] [Google Scholar]
- 3).Hamel-Desnos C, Desnos P, Wollmann JC, et al. Evaluation of the efficacy of polidocanol in the form of foam compared with liquid form in sclerotherapy of the greater saphenous vein: initial results. Dermatol Surg 2003; 29: 1170-5; discussion, 1175. [DOI] [PubMed] [Google Scholar]
- 4).Yamaki T, Nozaki M, Iwasaka S. Comparative study of duplex-guided foam sclerotherapy and duplex-guided liquid sclerotherapy for the treatment of superficial venous insufficiency. Dermatol Surg 2004; 30: 718-22; discussion, 722. [DOI] [PubMed] [Google Scholar]
- 5).Yamaki T. Efficacy of polidocanol foam compared with polidocanol liquid in sclerotherapy of venous insufficiency. In: Bergan J, Cheng L eds. Foam Sclerotherapy: A Textbook. London: The Royal Society of Medicine Press Ltd., 2008: 177-82.
- 6).Yamaki T, Nozaki M, Sakurai H, et al. Prospective randomized efficacy of ultrasound-guided foam sclerotherapy compared with ultrasound-guided liquid sclerotherapy in the treatment of symptomatic venous malformations. J Vasc Surg 2008; 47: 578-84. [DOI] [PubMed] [Google Scholar]
- 7).Rabe E, Otto J, Schliephake D, et al. Efficacy and safety of great saphenous vein sclerotherapy using standardised polidocanol foam (ESAF): a randomised controlled multicenter clinical trial. Eur J Vasc Endovasc Surg 2008; 35: 238-45. [DOI] [PubMed] [Google Scholar]
- 8).Yamamoto T, Sakata M. Influence of fibers and wavelengths on the mechanism of action of endovenous laser ablation. J Vasc Surg Venous Lymphat Disord 2014; 2: 61-9. [DOI] [PubMed] [Google Scholar]
- 9) Kabnick S, Ombrellino M, Agis H, et al. Endovenous heat induced thrombus (EHIT) at the superficial-deep venous junction: a new post-treatment clinical entity, classification and potential treatment strategies. 18th Annual Meeting of the American Venous Forum, Miami, Florida; 2006.
- 10).Mozes G, Kalra M, Carmo M, et al. Extension of saphenous thrombus into the femoral vein: a potential complication of new endovenous ablation techniques. J Vasc Surg 2005; 41: 130-5. [DOI] [PubMed] [Google Scholar]
- 11).Puggioni A, Kalra M, Carmo M, et al. Endovenous laser therapy and radiofrequency ablation of the great saphenous vein: analysis of early efficacy and complications. J Vasc Surg 2005; 42: 488-93. [DOI] [PubMed] [Google Scholar]
- 12).Rhee SJ, Cantelmo NL, Conrad MF, et al. Factors influencing the incidence of endovenous heat-induced thrombosis (EHIT). Vasc Endovascular Surg 2013; 47: 207-12. [DOI] [PubMed] [Google Scholar]
- 13).Marsh P, Price A, Holdstock J, et al. Deep vein thrombosis (DVT) after venous thermoablation techniques: rates of endovenous heat-induced thrombosis (EHIT) and classical DVT after radiofrequency and endovenous laser ablation in a single centre. Eur J Vasc Endovasc Surg 2010; 40: 521-7. [DOI] [PubMed] [Google Scholar]
- 14).Kurihara N, Hirokawa M, Yamamoto T. Postoperative venous thromboembolism in patients undergoing endovenous laser and radiofrequency ablation of the saphenous vein. Ann Vasc Dis 2016; 9: 259-66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15).Uthoff H, Holtz D, Broz P, et al. Rivaroxaban for thrombosis prophylaxis in endovenous laser ablation with and without phlebectomy. J Vasc Surg Venous Lymphat Disord 2017; 5: 515-23. [DOI] [PubMed] [Google Scholar]
- 16).Min RJ, Khilnani N, Zimmet SE. Endovenous laser treatment of saphenous vein reflux: long-term results. J Vasc Interv Radiol 2003; 14: 991-6. [DOI] [PubMed] [Google Scholar]
- 17).Theivacumar NS, Dellagrammaticas D, Beale RJ, et al. Fate and clinical significance of saphenofemoral junction tributaries following endovenous laser ablation of great saphenous vein. Br J Surg 2007; 94: 722-5. [DOI] [PubMed] [Google Scholar]
- 18).Kusagawa H, Shomura S, Komada T, et al. Subfascial endoscopic perforator surgery using screw-type ports is a very useful component of a comprehensive treatment program for chronic venous insufficiency. Ann Vasc Dis 2012; 5: 357-63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19).Gloviczki P, Bergan J, Menawat S, et al. Safety, feasibility, and early efficacy of subfascial endoscopic perforator surgery: a preliminary report from the North American registry. J Vasc Surg 1997; 25: 94-105. [DOI] [PubMed] [Google Scholar]
- 20).Nelzén O, Fransson I. True long-term healing and recurrence of venous leg ulcers following SEPS combined with superficial venous surgery: a prospective study. Eur J Vasc Endovasc Surg 2007; 34: 605-12. [DOI] [PubMed] [Google Scholar]
- 21).Haruta N, Shinhara R. Subfascial Endoscopic Perforating Veins Surgery (SEPS): Two Port System Subfascial Endoscopic Perforating Veins Surgery (TPS-SEPS) with the use of EndoTIP® cannula. Jpn J Phlebol 2011; 22: 63-7. (In Japanese) [Google Scholar]
- 22).Haruta N, Shinhara R, Uchida K, et al. One hundred one experiences of Subfascial Endoscopic Perforating Veins Surgery (SEPS) in patients with stasis ulcerations: how to get the knack of simplified SEPS for beginners. Jpn J Phlebol 2010; 21: 333-8. (In Japanese) [Google Scholar]
- 23).Chan YC, Law Y, Cheung GC, et al. Cyanoacrylate glue used to treat great saphenous reflux: measures of outcome. Phlebology 2017; 32: 99-106. [DOI] [PubMed] [Google Scholar]
- 24).Bozkurt AK, Yılmaz MF. A prospective comparison of a new cyanoacrylate glue and laser ablation for the treatment of venous insufficiency. Phlebology 2016; 31 Suppl: 106-13. [DOI] [PubMed] [Google Scholar]