Abstract
Background
The sexual experience is shaped by sensory function; with aging, sensory dysfunction may interfere with sexuality and sexual behavior between partners. Specifically, older adults with age-related sensory dysfunction may have less sexual activity than those with better sensory function. In addition, since sexual desire and attraction rests in part upon sensory function, sensory dysfunction may also be associated with less sexual motivation.
Aim
To test the association between sexual activity and motivation in older adults and their sensory dysfunction.
Methods
Sensory dysfunction was measured both by global sensory impairment (GSI, a validated measure of dysfunction shared among the five classical senses—olfaction, vision, taste, touch, hearing) and by total sensory burden (cumulative sensory loss). Sexual activity was quantified by frequency and type of sexual behavior. Sexual motivation was measured by the frequency of sexual ideation and the importance of sex to the respondent. We used cross-sectional data from a nationally representative sample of community-dwelling older adults (aged 57-85) in the United States (National Social Life, Health, and Aging Project, N=3,005) in logistic regression analyses.
Outcomes
Self-reports of sexual activity, sexual motivation, and satisfaction with the sexual relationship.
Results
Older adults with sensory dysfunction were less likely to be sexually active – an association that persisted when accounting for other factors that also affected sexual activity (age, gender, partnered status, mental and physical health, and relationship satisfaction). Nonetheless, sensory dysfunction did not impair sexual motivation, nor affect the physical and emotional satisfaction with the sexual relationship. Among currently sexually active older adults, sensory dysfunction did not affect the frequency of sex or the type of sexual activity (foreplay, vaginal intercourse, or oral sex). These results were the same for two different measures of sensory dysfunction.
Clinical Implications
Medical and health care interventions that can reduce the burden of sensory dysfunction may improve older adults’ sexual experience.
Strengths and Limitations
This is the first nationally representative study of sexuality and multisensory dysfunction in community-dwelling older adults. Four of the five classic senses were measured with objective tests, and hearing was rated by interviewers in the context of their conversation.
Conclusion
Sensory dysfunction is associated with sexual inactivity, but not with sexual motivation. Among those who are sexually active, sensory dysfunction did not interfere with sexual expression. Improving the sexual experience of older adults requires a focus on sensory dysfunction as an impediment to sexual activity given that older adults remain sexually motivated.
Keywords: Sensory Dysfunction, Sensory Loss, Aging, Sexual Activity, Sexual Motivation
Introduction
Older adults continue to enjoy regular sex [1], and for many, sexual activity remains an important component of their lives [2–3]. However, increasing age is associated with less sexual activity and more sexual dysfunctions [1, 4–5]. It is widely accepted that poor physical and mental health, chronic illness, lack of an available sexual partner, and relationship unhappiness are all associated with sexual inactivity and sexual dysfunction among older adults [1, 4–10], but little is known about how age-related decline across all of the classical senses is associated with sexuality even though multisensory loss is prevalent among older adults in the US [11] and sensory information is important for human sexuality [12–18].
Previous empirical studies have demonstrated that sensory information from each of the 5 classical senses (olfaction, vision, taste, touch, and hearing) shapes sexual motivation [12–15] as well as sexual activity [10,16]. In addition, sensory impairment can negatively affect quality of life [19–21], as well as marital quality and a partner’s wellbeing [22]; sensory impairment is also associated with more depressive symptoms [23]. All of these factors can lead to sexual inactivity. Indeed, a large survey of a retirement community in Florida found that both hearing loss and vision loss were associated with sexual inactivity [10], while in a study of clinical patients with olfactory dysfunction, Gudziol and colleagues suggested that depression from olfactory loss may be associated with sexual inactivity [16].
However, these empirical studies have only looked at the role or function of one or at most two sensory modalities at a time and have not considered the effects of multisensory loss on sexual motivation and sexual activity. Studies that focus on the impairment of one type of sensory modality fail to recognize that sexuality is shaped by stimuli from each of the senses [12–15], that there are gender differences with respect to the use of sensory information [12], that individuals may vary in their responsiveness to different types of sexual stimuli [18, 24–26], and that the combination of sensory information can synergistically enhance sexual desire and arousal [27]. Therefore, it is important to consider how multisensory dysfunction (impairment of more than one of the 5 classical senses) matters for human sexuality.
A significant proportion of older adults in the US have multisensory loss, which is likely driven by a shared mechanism of physiological and neurological decline [11]. Given that sensory information undergirds human sexuality and that sensory function declines with age, it is important to understand whether there is an association between comprehensive sensory dysfunction and multiple aspects of sexuality to mitigate the effects of sensory burden on older adults’ sexuality.
We hypothesize that multisensory dysfunction is associated with sexual inactivity and decreased sexual motivation among older adults. To test our hypotheses, we use publicly available data from the National Social Life, Health, and Aging Project (NSHAP) [28].
Methods
Study Population
NSHAP is a nationally representative study of the health and social relationships of older adults in the US. In 2005-2006, trained interviewers conducted in-home interviews with community-dwelling older adults (aged 57-85) that resulted in a sample of 3,005 respondents. The design of the sampling frame, oversampling of smaller demographic groups, and the establishing of the characteristics of both respondents and non-respondents enabled algorithms to weight the raw data (the weights account for differential probability of selection and nonresponse by age, gender, and race/ethnicity), and thereby describe characteristics of the US population born between 1920 and 1947 [29]. The interviewer administered an in-home questionnaire that collected demographic, social, psychological, and biological measures that also included objective measures of sensory function [29–30].
The institutional review boards of the National Opinion Research Center and University of Chicago approved the study, and all respondents provided written informed consent.
Data
Sensory Variables
NSHAP tested olfactory, gustatory, tactile, and visual function; auditory function was rated by the interviewer after the two hour interview [30]. Olfactory function was measured by testing odor identification using felt-tip pens impregnated with odorants [31]. Filter papers with sour, bitter, sweet, and salty taste were administered to respondents to measure gustatory function [30]. Tactile function was assessed using the 2-point discrimination test to measure the finger’s sensitivity to touch [30]. Visual acuity was assessed using a distance chart [30]. Trained interviewers were asked to rate the respondents’ auditory function during the two-hour interview in the home [30]. All five measures were used to construct our multisensory dysfunction variables.
The two independent variables in our study were the global sensory impairment (GSI) score and the total sensory burden score. Correia and colleagues have demonstrated that GSI can be considered a measure of a mechanism of sensory decline shared among the 5 classical senses [11]. Using this measure allows us to test the possibility that a shared neural or physiological process underlying all sensory decline shapes sexuality within older adults.
The GSI score is based on a single-factor model fit to observed measures of dysfunction across all five senses. The GSI score was derived from this model using empirical Bayes mean predictions of the latent variable [11]. Higher GSI scores indicate greater sensory impairment; scores on the high end of the scale indicate that older adults have poor function on all 5 senses, while scores on the low end of the scale indicate that older adults have good function on all 5 senses.
For robustness, we also measured multisensory dysfunction in the form of total sensory burden, which was a measure of the combined amount of sensory dysfunction totaled across all 5 senses. For each of the five senses, points were assigned for having poor (2), fair (1), or good (0) function; these were summed (range 0-10). Higher scores indicate greater sensory burden. This measure allows us to look at the total effect of sensory dysfunction on sexuality within older adults.
Sexuality Variables
The key dependent variables were sexual activity, sexual frequency, sexual motivation, and the emotional and physical satisfaction with the sexual relationship. During the in-home interviews, sex and sexual activity were defined as: any mutually voluntary activity with another person that involves sexual contact, whether or not intercourse or orgasm occurs [1].
All respondents were asked about their sexual partnership history, and those who reported any sexual activity within the past year were considered sexually active, while those who did not engage in sexual activity within the past year were considered sexually inactive [32–33]. Sexual motivation was determined by asking all respondents how important sex was (extremely important, very important, moderately important, somewhat important, not at all important), and the frequency with which they thought about sex (never, less than once a month, one to a few times a month, one to a few times a week, everyday, several times a day) [32–33].
Respondents who identified a partner in the sexual partnership history were asked how emotionally satisfying and physically pleasurable the relationship was (extremely, very, moderately, slightly, not at all) [32–33].
Sexually active respondents were asked how frequently they engaged in sexual activity (once a day or more, 3-6 times a week, once or twice a week, 2 to 3 times a month, once a month or less), as well as the frequency with which they participated in foreplay, oral, and vaginal intercourse when they engaged in sexual activity (always, usually, sometimes, rarely, never) [32–33].
Demographic, health, and social variables
We considered whether age, race, gender, partnership status, relationship happiness, physical and mental health could affect sexual activity and motivation or mediate the relationship between sensory dysfunction and sexuality.
Partnership status was a dichotomous measure of “partnered” and “unpartnered”. Respondents were considered partnered if they reported being in a marriage or in a cohabiting relationship with a romantic partner. Respondents who were separated, widowed, divorced, or never married were asked if they had a current sexual or romantic partner; if these respondents reported having a sexual or romantic partner, they were also coded as “partnered”. The “unpartnered” respondents were those who were separated, widowed, divorced, or never married and did not have a current sexual or romantic partner. Relationship happiness was measured on a 7-point scale and reverse coded (1 = very happy; 7 = very unhappy).
Physical health was assessed using the modified Charlson Comorbidity Index [34] totaling chronic conditions (range 0-10.5). Functional health was quantified by number of difficulties with activities of daily living: walking a block, walking across a room, getting in or out of bed, dressing, eating, bathing, using the toilet (range 0-7) [35].
Frequency of 11 depressive symptoms was measured with NSHAP’s modified Iowa CES-D, standardized by following Payne and colleagues’ recommendation to combine never/rarely (0), some of the time (1) and occasionally/most of the time (2), range 0-22 [36].
Since cognition is associated with sensory decline [37], we ran a sensitivity analysis by excluding respondents with poor cognition, assessed with a modified version of the Short Portable Mental Status Questionnaire (SPMSQ; range 0-10) [38].
Table 1.
Characteristics of US Adults 57–85 years of age
| Demographics & Health Variables | Percentages |
|---|---|
| Female, N=3,005 | 51.5% |
| Race, N=2,993 | |
| White | 80.6% |
| Black | 10.0% |
| Hispanic | 6.8% |
| Other | 2.5% |
| Partnered Status, N=3,005 | 72.8 % |
| Median (Range) | |
| Age, N=3,005 | 67.0 (57 - 85) |
| GSI, N=2,968 | −.14 (−.85 - 2.16) |
| Total Sensory Burden, N=2,968 | 3.0 (0 - 10) |
| Charlson Comorbidity Index, N=3,005 | 1.0 (0 - 10.5) |
| Depressive Symptoms, N=2,955 | 4.0 (0 - 22) |
| Difficulties with Activities of Daily Living, N=3,004 | 0.0 (0 - 7) |
Statistical Methods
We used logistic regression to test whether GSI and total sensory burden were associated with sexual inactivity. In Model 1, we used bivariate statistics to demonstrate the association between sensory dysfunction and sexual inactivity. We added control variables (age, gender, partnership status, relationship happiness, physical and mental health) to Model 2 to test whether the relationship between multisensory dysfunction and sexual inactivity persist even when accounting for other factors associated with sexual inactivity.
We used ordinal logistic regression to test the association between GSI and total sensory burden and sexual motivation, frequency of sexual activity, and the emotional and physical satisfaction with the sexual relationship. In Model 1, we used bivariate statistics to test the association between multisensory dysfunction and the sexuality variables. In Model 2, we added control variables that could mediate these relationships.
We tested GSI and total sensory burden separately on the measures of sexuality because GSI and total sensory burden are different measures of multisensory loss. With GSI, we are testing whether a common mechanism of physiological decline among the senses is associated with sexuality, while with total sensory burden we are testing whether cumulative sensory loss is associated with sexuality. We estimated all models in Stata 14 (StataCorp., College Station, TX) using logistic and ologit commands along with the survey weights provided with the data.
Results
Sexual Inactivity
Older US adults with poor sensory function shared across all 5 senses (GSI), were less likely to have engaged in sexual activity in the past year (OR=.48, p ≤ 0.01). In particular, those in the fourth quartile of GSI scores (poorest sensory functioning) were less likely to have engaged in sexual activity in the past year (40%) in comparison to those in the first quartile of GSI scores (65%; Figure 1A). Adjusting for covariates attenuated but did not remove the effect of GSI (OR=.78, p-value=0.027, Table 2).
Figure 1.
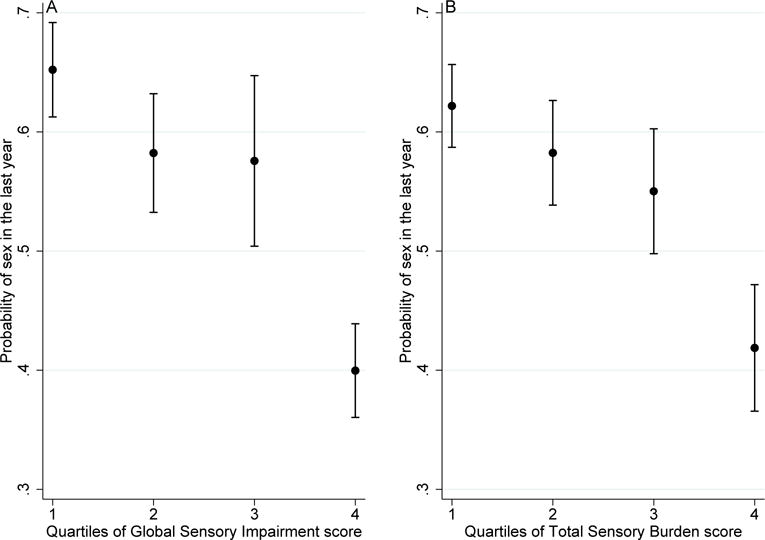
Global sensory impairment (A) and total sensory burden (B): association with probability of sexual activity
Table 2.
GSI and Total Sensory Burden: Odds Ratio (95% CI) of Sexual Activity, N=2,678
| Global Sensory Impairment |
.78* (.62–.97) | Total Sensory Burden |
.92* (.86–.98) |
|---|---|---|---|
| Age (per year) | .90*** (.88–.92) | .90*** (.88–.91) | |
| Female | .46*** (.34–.62) | .45*** (.34–.60) | |
| Race/Ethnicity (vs White) | |||
| Black | 1.38 (.96–1.98) | 1.36 (.95–1.94) | |
| Hispanic | 1.09 (.76–1.55) | 1.09 (.76–1.56) | |
| Other | 1.01 (.55–1.85) | 1.00 (.54–1.86) | |
| Unpartnered | .05*** (.03–.07) | .05*** (.03–.07) | |
| Unhappiness with relationship | .86** (.77–.96) | .86** (.77–.95) | |
| Depressive Symptoms | .96*** (.93–.98) | .96*** (.93–.98) | |
| Charlson Comorbidity Index | .86*** (.79–.93) | .85*** (.79–.92) | |
| Difficulties with Activities of Daily Living | .85** (.77–.94) | .85** (.77–.94) |
p<0.001,
p<0.01,
p<0.05
These results indicate that a shared physiological decline among the senses is associated with sexual inactivity among older adults, particularly because the model controlled for other significant factors associated with sexual activity. Sexual inactivity was also associated independently with being a woman, increasing age, being unpartnered, having more depressive symptoms, being unhappy with the relationship, having more comorbidities, and experiencing more difficulty with activities of daily living, consistent with previous studies. Race/ethnicity was not associated with sexual inactivity (Table 2).
Total sensory burden was also associated with sexual inactivity. Older adults with a larger sensory burden had reduced odds of sexual activity (OR=.85 p ≤ 0.01). Those in the fourth quartile of total sensory burden scores were less likely to have engaged in sexual activity in the past year (42%) in comparison to those in the first quartile of scores (62%; Figure 1B). We confirmed this relationship by including the covariates known to affect sexuality; again, the effect was attenuated but not removed (OR=.92, p=0.012, Table 2).
Poor sensory functioning may reflect diminished brain function and is a known associate of poor cognitive functioning [37]. Therefore, we were concerned that the observed associations between multiple sensory dysfunction and sexual inactivity might be driven solely by people with large cognitive deficits. To test this hypothesis, we ran a sensitivity analysis by excluding respondents with poor cognition (SPMSQ<8). Even with these extreme cases excluded, both GSI (p=0.01) and total sensory burden (p=0.004) were still associated with sexual inactivity (Table A1).
Sexual Motivation
In contrast, sensory dysfunction was not associated with sexual motivation (importance of sex and frequency of thinking about sex), even though sensory dysfunction was found to be associated with sexual inactivity (Table A2). These results suggest that while older adults remain sexually motivated, they may not be able to engage in sexual activity because of the physiological decline of their sensory systems.
Physical and Emotional Satisfaction with Relationship
Since sensory dysfunction impairs communication between partners, decreases marital quality, and negatively affects a partner’s wellbeing [22], sensory dysfunction may also decrease physical and emotional satisfaction with the relationship. Contrary to our expectation, we found that sensory loss was not associated with emotional and physical satisfaction with the relationship (Table A2).
Types of Sexual Activity
Although sensory impairment reduced the odds of having engaged in sexual activity within the past year, among those who are currently sexually active, sensory loss was not associated with the frequency of sex, nor with the various types of sexual activity (e.g. foreplay, oral sex, and vaginal intercourse) (Table A2).
Discussion
Previous studies have dispelled the myth that older adults do not engage in sexual activity. Lindau and colleagues have shown that many remain sexually active well into their eighth and ninth decade [1]. In addition, recent qualitative studies have elicited the meanings that older adults attach to sexual relationships and show that, while the meanings associated with sexual activity have changed over the life course, older adults still ascribe significance to sexual activity and to the pleasures and intimacy that it offers [2–3]. Given the continued centrality of sexuality in the lives of older adults, it is necessary to understand the impediments to sexual activity within this population in order to support healthy aging.
Similar to previous studies, we found that poor physical and mental health, lack of an available sexual partner, and relationship unhappiness were all associated with sexual inactivity. While sensory function is an important component of sexuality, only a handful of studies have examined the relationship between sensory dysfunction and sexual inactivity. The few studies that have explored this relationship suggest that there is an association between sensory impairment and sexual inactivity [10, 16–17]. However, these studies have only looked at the loss of one or at most two types of sensory modalities, even though each of the five classical senses are individually important for sexuality.
Using two different measures of multisensory dysfunction, we found that a physiological decline shared among the 5 senses (GSI) and a higher sensory burden (the sum of sensory impairment severity across the five senses) were each associated with sexual inactivity in older adults. However, among sexually active older adults, sensory dysfunction was not associated with the frequency of sex, nor with engaging in different types of sexual activity. This suggests that sensory function may be important for initiating sexual activity, but sustaining a sexual relationship may rest upon factors other than sensory function. It appears that sensory function matters for whether or not older adults have a sex life, but for those who are sexually active, sensory function does not shape the contours of sexual expression.
We also found that sensory dysfunction was not associated with sexual motivation nor with physical and emotional satisfaction with the relationship. Given that sensory dysfunction shapes sexual activity but not sexual motivation, sexually motivated older adults may be facing barriers to engaging in sexual activity because of sensory decline. Since sensory dysfunction was not associated with physical and emotional satisfaction with the relationship, it may mean that even if sensory dysfunction is straining the relationship, older adults are nevertheless finding ways to cope and to enjoy the relationship.
Limitations
First, the survey data did not include the physiological and neurological data that are needed to determine mechanisms by which multisensory dysfunction affects sexual inactivity. Further laboratory-based studies are needed.
Second, we used cross-sectional data so we could not establish mediating effects that might explain the relationship between multisensory dysfunction and sexual inactivity. However, we controlled for age, gender, partnership status, relationship happiness, physical health, and mental health, all of which are known to be associated with sexual inactivity. Even when controlling for these confounders, our measures of sensory dysfunction remained significant. Further study using longitudinal data is necessary to establish potential mediating effects between multisensory dysfunction and sexual inactivity.
Third, our sexuality variables did not incorporate the broader aspects of sexual activity such as affection and intimacy. However, we controlled for relationship happiness, which may indirectly capture affection within the sexual relationship.
Despite these limitations, we have demonstrated the need for studies of sexuality to take into account multisensory dysfunction. While prior literature has separately studied sensory function and either sexual motivation or sexual activity, we measured both of these aspects of sexuality in the same people. In doing so, we found that sensory dysfunction shapes sexual activity but not sexual motivation. Thus, sensory dysfunction may have a detrimental effect on sexual behavior without reducing sexual motivation. This highlights the need to consider how sensory function differentially shapes the various components of human sexuality. Moreover, this finding suggests that sensory dysfunction poses an obstacle to engaging in sexual activity, especially since individuals remain sexually motivated. Thus, understanding how sensory impairment is related to sexual inactivity can help us mitigate the effects of sensory loss on older adults who remain sexually motivated. Sexuality is important for successful aging, and older adults continue to attach importance to sexual relationships, so understanding sensory loss and reducing its harmful effects will improve sexual function and enhance elders’ quality of life.
Acknowledgments
Funding: The National Social Life, Health and Aging Project is supported by the National Institute on Aging and the National Institutes of Health (R01AG043538; R01AG048511). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Table A1.
Global Sensory Impairment and Total Sensory Burden: Odds Ratio (95% CI) of Sexual Activity Excluding Those with Poor Cognition (SPMSQ<8), N=2,475
| Global Sensory Impairment |
.75* (.60–.93) | Total Sensory Burden |
.91** (.85–.97) |
|---|---|---|---|
| Age (per year) | .90*** (.88–.91) | .89*** (.88–.91) | |
| Female | .44*** (.31–.60) | .42*** (.31–.58) | |
| Race/Ethnicity (vs White) | |||
| Black | 1.32 (.86–2.0) | 1.31 (.86–2.0) | |
| Hispanic | 1.09 (.70–1.67) | 1.09 (.70–1.69) | |
| Other | 1.03 (.48–2.20) | 1.02 (.47–2.20) | |
| Unpartnered | .05*** (.03–.07) | .05*** (.03–.07) | |
| Unhappiness with relationship | .82*** (.74–.90) | .82*** (.74–.90) | |
| Depressive Symptoms | .96** (.93–.98) | .95** (.93–.98) | |
| Charlson Comorbidity Index | .86** (.79–.94) | .86** (.79–.94) | |
| Difficulties with Activities of Daily Living | .85** (.77–.94) | .85** (.77–.95) |
p<0.001,
p<0.01,
p<0.05
Table A2.
Global Sensory Impairment and Total Sensory Burden: Odds Ratio (95% CI) of Sexual Motivation, Relationship Quality, and Types of Sexual Activity
| Global Sensory Impairment | Total Sensory Burden | |
|---|---|---|
| Sexual Motivation | ||
| Think about Sex | .99 (.81–1.20) | 1.00 (.95–1.05) |
| Sex Important | .97 (.78–1.19) | 1.00 (.95–1.06) |
| Relationship Quality | ||
| Relationship Physically Pleasurable | 1.09 (.93–1.28) | 1.01 (.96–1.06) |
| Relationship Emotionally Satisfying | 1.02 (.88–1.18) | .99 (.95–1.04) |
| Sexual Activities Among Sexually Active | ||
| Frequency of Sexa | 1.24 (.96–1.61) | 1.04 (.97–1.12) |
| Frequency of Foreplayb | 1.17(.86–1.59) | 1.02 (.94–1.11) |
| Frequency of Vaginal Sexb | 1.25 (.97–1.62) | 1.03 (.96–1.10) |
| Frequency of Giving Oral Sexb | 1.14 (.85–1.52) | 1.03 (.94–1.12) |
| Frequency of Receiving Oral Sexb | 1.10 (.84–1.43) | 1.02 (.94–1.11) |
Note: Covariates include: age, race, gender, partnership status, relationship happiness, Charlson comorbidity index, depressive symptoms, and difficulties with activities of daily living
Sexually active respondents were asked how frequently they engaged in sexual activity (once a day or more, 3–6 times a week, once or twice a week, 2 to 3 times a month, once a month or less),
Sexually active respondents were asked how frequently they participated in foreplay, oral sex, and vaginal intercourse when they engaged in sexual activity (always, usually, sometimes, rarely, never)
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
Conflicts of Interest: The authors report no conflicts of interest.
References
- 1.Lindau ST, Schumm LP, Laumann EO, Levinson W, O’muircheartaigh CA, Waite LJ. A study of sexuality and health among older adults in the United States. N Engl J Med. 2007;357:762–74. doi: 10.1056/NEJMoa067423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fileborn B, Hinchliff S, Lyons A, Heywood W, Miniciello V, Brown G, et al. Importance of sex and the meaning of sex and sexual pleasure for men aged 60 and older who engage in heterosexual relationships: Findings from a qualitative interview study. Arch Sex Behav. 2017;46:2097–110. doi: 10.1007/s10508-016-0918-9. [DOI] [PubMed] [Google Scholar]
- 3.Watson WK, Stelle C, Bell N. Older women in new romantic relationships: Understanding the meaning and importance of sex in later life. Int J Aging Hum Dev. 2017;85:33–43. doi: 10.1177/0091415016680067. [DOI] [PubMed] [Google Scholar]
- 4.Laumann EO, West S, Glasser D, Carson C, Rosen R, Kang JH. Prevalence and correlates of erectile dysfunction by race and ethnicity among men aged 40 or older in the United States: From the male attitudes regarding sexual health survey. J Sex Med. 2007;4:57–65. doi: 10.1111/j.1743-6109.2006.00340.x. [DOI] [PubMed] [Google Scholar]
- 5.Laumann EO, Aniruddha D, Waite LJ. Sexual dysfunction among older adults: Prevalence and risk factors from a nationally representative US probability sample of men and women 57–85 years of age. J Sex Med. 2008;5:2300–11. doi: 10.1111/j.1743-6109.2008.00974.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jackson G. Sexual dysfunction and diabetes. Int J Clin Pract. 2004;58:358–62. doi: 10.1111/j.1368-5031.2004.00180.x. [DOI] [PubMed] [Google Scholar]
- 7.Call V, Sprecher S, Schwartz P. The incidence and frequency of marital sex in a national sample. J Marriage Fam. 1995;57:639–52. [Google Scholar]
- 8.DeLamater J. Sexual expression in later life: A review and synthesis. J Sex Res. 2012;49:125–41. doi: 10.1080/00224499.2011.603168. [DOI] [PubMed] [Google Scholar]
- 9.Karraker A, DeLamater J. Past-year sexual inactivity among older married persons and their partners. J Marriage Fam. 2013;75:142–63. [Google Scholar]
- 10.Bach LE, Mortimer JA, VandeWeerd C, Corvin J. The association of physical and mental health with sexual activity in older adults in a retirement community. J Sex Med. 2013;10:2671–8. doi: 10.1111/jsm.12308. [DOI] [PubMed] [Google Scholar]
- 11.Correia C, Lopez KJ, Wroblewski KE, Huisingh-Scheetz M, Kern DW, Chen RC, et al. Global sensory impairment in older adults in the United States. J Am Geriatr Soc. 2016;64:306–13. doi: 10.1111/jgs.13955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Herz RS, Cahill ED. Differential use of sensory information in sexual behavior as a function of gender. Hum Nat. 1997;8:275–86. doi: 10.1007/BF02912495. [DOI] [PubMed] [Google Scholar]
- 13.Ren D, Tan K, Arriaga XB, Chan KQ. Sweet love: The effects of sweet taste experience on romantic perceptions. J Soc Pers Relat. 2015;32:905–21. [Google Scholar]
- 14.Rowland DL, Greenleaf W, Mas M, Myers L, Davidson JM. Penile and finger sensory thresholds in young, aging, and diabetic males. Arch Sex Behav. 1989;18:1–12. doi: 10.1007/BF01579287. 1989. [DOI] [PubMed] [Google Scholar]
- 15.Frohlich PF, Meston CM. Tactile sensitivity in women with sexual arousal disorder. Arch Sex Behav. 2005;34:207–17. doi: 10.1007/s10508-005-1798-6. [DOI] [PubMed] [Google Scholar]
- 16.Gudziol V, Wolff-Stephan S, Aschenbrenner K, Joraschky P, Hummel T. Depression resulting from olfactory dysfunction is associated with reduced sexual appetite – A cross-sectional cohort study. J Sex Med. 2009;6:1924–9. doi: 10.1111/j.1743-6109.2009.01266.x. [DOI] [PubMed] [Google Scholar]
- 17.Lehane CM, Dammeyer J, Hovaldt HB, Elsass P. Sexuality and well-being among couples living with acquired deafblindness. Sex Disabil. 2017;35:135–146. [Google Scholar]
- 18.Whalen RE. Sexual motivation. Psychol Rev. 1966;73:151–63. doi: 10.1037/h0023026. [DOI] [PubMed] [Google Scholar]
- 19.Dalton DS, Cruickshanks KJ, Klein BE, Klein R, Wiley TL, Nondahl DM. The impact of hearing loss on quality of life in older adults. Gerontologist. 2003;43:661–8. doi: 10.1093/geront/43.5.661. [DOI] [PubMed] [Google Scholar]
- 20.Fischer ME, Cruickshanks KJ, Klein BE, Klein R, Schubert CR, Wiley TL. Multiple sensory impairment and quality of life. Ophthalmic Epidemiol. 2009;16:346–53. doi: 10.3109/09286580903312236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Khil L, Wellmann J, Berger K. Impact of combined sensory impairments on health-related quality of life. Qual Life Res. 2015;24:2099–103. doi: 10.1007/s11136-015-0941-7. [DOI] [PubMed] [Google Scholar]
- 22.Strawbridge WJ, Wallhagen MI, Shema SJ. Impact of spouse vision impairment on partner health and well-being: A longitudinal analysis of couples. J Gerontol B Psychol Sci Soc Sci. 2007;62:S315–22. doi: 10.1093/geronb/62.5.s315. [DOI] [PubMed] [Google Scholar]
- 23.Capella-McDonall ME. The effects of single and dual sensory loss on symptoms of depression in the elderly. Geriatric Psychiatry. 2005;20:855–61. doi: 10.1002/gps.1368. [DOI] [PubMed] [Google Scholar]
- 24.Kippin TE, Pfaus JG. The development of olfactory conditioned ejaculatory preferences in the male rat: I. Nature of the unconditioned stimulus. Physiol Behav. 2001;73:457–69. doi: 10.1016/s0031-9384(01)00484-x. [DOI] [PubMed] [Google Scholar]
- 25.Kippin TE, Taliankis S, Schattman L, Bartholomew S, Pfaus JG. Olfactory conditioning of sexual behavior in the male rat (Rattus norvegicus) J Comp Psychol. 1998;112:389–99. [Google Scholar]
- 26.Hoffman H, Janssen E, Turner S. Classical conditioning of sexual arousal in women and men: Effects of varying awareness and biological relevance of the conditioned stimulus. Arch Sex Behav. 2004;33:43–53. doi: 10.1023/B:ASEB.0000007461.59019.d3. [DOI] [PubMed] [Google Scholar]
- 27.Partan S, Yelda S, Price V, Shimizu T. Female pigeons, Columba livia, respond to multisensory audio/video playbacks of male courtship behavior. Anim Behav. 2005;70:957–66. [Google Scholar]
- 28.Waite LJ, Laumann EO, Levinson W, Lindau ST, O’Muircheartaigh CA. National Social Life, Health, and Aging Project (NSHAP): Wave 1. ICPSR20541-v6. Ann Arbor, MI: Inter-university Consortium for Political and Social Research [distributor]; 2014-04-30. [DOI] [Google Scholar]
- 29.O’Muircheartaigh C, Eckman S, Smith S. Statistical design and estimation for the National Social Life, Health, and Aging Project. J Gerontol B Psychol Sci Soc Sci. 2009;64B:i12–9. doi: 10.1093/geronb/gbp045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Schumm LP, McClintock M, Williams S, Leitsch S, Lundstrom J, Hummel T, et al. Assessment of sensory function in the National Social Life, Health, and Aging Project. J Gerontol B Psychol Sci Soc Sci. 2009;64B:i76–85. doi: 10.1093/geronb/gbp048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kern DW, Wroblewski KE, Schumm LP. Field survey measures of olfaction: The olfactory function field exam (OFFE) Field Method. 2014;26:421–434. doi: 10.1177/1525822X14547499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Waite LJ, Laumann EO, Das A, Schumm LP. Sexuality: Measures of partnerships, practices, attitudes, and problems in the National Social Life, Health, and Aging Study. Gerontol B Psychol Sci Soc Sci. 2009;64B:i56–i66. doi: 10.1093/geronb/gbp038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Galinksy AM, McClintock MK, Waite LJ. Sexuality and physical contact in National Social Life, Health, and Aging Project Wave 2. Gerontol B Psychol Sci Soc Sci. 2014;69:S83–S98. doi: 10.1093/geronb/gbu072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Katz JN, Chang LC, Sangha O, Fossel AH, Bates DW. Can comorbidity be measured by questionnaire rather than medical record review? Med Care. 1996;34:73–84. doi: 10.1097/00005650-199601000-00006. [DOI] [PubMed] [Google Scholar]
- 35.Huisingh-Scheetz M, Kocherginsky M, Schumm LP, Engelmann M, McClintock MK, Dale W, et al. Geriatric syndromes and functional status in NSHAP: Rationale, measurement, and preliminary findings. Gerontol B Psychol Sci Soc Sci. 2014;69:S177–S190. doi: 10.1093/geronb/gbu091. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Payne C, Hedberg EC, Kozloski M, Dale W, McClintock MK. Using and interpreting mental health measures in the National Social Life, Health, and Aging Project. J Gerontol B Psychol Sci Soc Sci. 2014;69:S99–116. doi: 10.1093/geronb/gbu100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Li KZH, Lindenberger U. Relations between aging sensory/sensorimotor and cognitive functions. Neurosci Biobehav Rev. 2002;26:777–783. doi: 10.1016/s0149-7634(02)00073-8. [DOI] [PubMed] [Google Scholar]
- 38.Pfeiffer E. A short portable mental status questionnaire for the assessment of organic brain deficit in elderly patients. J Am Geriatr Soc. 1975;23:433–441. doi: 10.1111/j.1532-5415.1975.tb00927.x. [DOI] [PubMed] [Google Scholar]


