Abstract
Polybrominated diphenyl ethers (PBDEs) may affect male reproductive function. 4-bromodiphenyl ether (BDE-3), the photodegradation products of higher brominated PBDEs, is the most fundamental mono-BDE in environment but is less studied. The purpose of this study was to investigate the reproductive toxicity induced by BDE-3 and explore the mechanism by metabolomics approach. In this study, mice were treated intragastrically with BDE-3 for consecutive six weeks at the dosages of 0.0015, 1.5, 10 and 30 mg/kg. The reproductive toxicity was evaluated by sperm analysis and histopathology examinations. UPLC-Q-TOF/MS was applied to profile the metabolites of testis tissue, urine and serum samples in the control and BDE-3 treated mice. Results showed the sperm count was dose-dependently decreased and percentage of abnormal sperms increased by the treatment of BDE-3. Histopathology examination also revealed changes in seminiferous tubules and epididymides in BDE-3 treated mice. Metabolomics analysis revealed that different BDE-3 groups showed metabolic disturbances to varying degrees. We identified 76, 38 and 31 differential metabolites in testis tissue, urine and serum respectively. Pathway analysis revealed several pathways including Tyrosine metabolism, Purine metabolism and Riboflavin metabolism, which may give a possible explanation for the toxic mechanism of BDE-3. This study indicates that UHPLC-Q-TOFMS-based metabolomics approach provided a better understanding of PBDEs-induced toxicity dynamically.
Introduction
Polybrominated diphenyl ethers (PBDEs) are a class of brominated flame retardants that have emerged as a major environmental pollutant. In 2009, PBDEs were officially identified as a new class of persistent organic pollutants (POPs) by the United Nations Environment Programs (UNEP)1. Higher brominated diphenyl ethers can degrade in the environment to PBDEs with fewer bromines, the mono-BDE exhibits the longest photolysis lifetime among all the PBDEs in the air2. 4-bromodiphenyl ether (BDE-3) is the most fundamental mono-BDE in the environment and the most abundant photodegradation products of higher brominated PBDEs3–6. BDE-3 can be generated and accumulate in fish (Carassius auratus) by biotransformation of PBDEs7.
The health risk of PBDEs has recently raised global concern. PBDEs may disrupt the endocrine system, have neurodevelopment toxicity, teratogenicity and potential carcinogenicity8,9. PBDEs could affect the male reproductive function, exposure level of PBDEs in human serum is negatively associated with sperm mobility and concentration10,11. Exposure to 2,2′,4,4′-tetrabromodiphenyl ether (BDE-47) decreased the rate of sperm capacitation, altered sperm motility parameters and increased germ cell loss and apoptosis in both mice and rat12,13. Previous studies have shown that PBDEs with fewer bromines are more volatile and bioaccumulative8,9 and hence may be more harmful to human health14. Mono-BDE may induce genetic recombination in mammalian cells15. Our previous studies in C. elegans also demonstrated BDE-3 could induce reproductive dysfunction and germ cell apoptosis by induction of ROS and DNA damage16. By far, however, few study in mono-BDE toxicity have been reported in rodents17.
Metabolomics protocols are used to comprehensively characterize the metabolite content of biological samples by exploiting cutting-edge analytical platforms. Liquid chromatography-mass spectrometry technology platform is the most commonly used analytical technique for metabolomics research in recent years.
Nowadays, non-targeted metabolomics has gained widely attention as a method of profiling endogenous metabolites because of its high sensitivity, high throughput, and unbiasedness. Therefore, Metabolomics techniques were gradually applied to many aspects, such as evaluating and monitoring drugs and even atmospheric pollutant toxicity18. The analysis also showed enormous potential for the detection of health effect of POPs, e.g. metabolomic-based profiling revealed plasma responses to dioxin-associated dietary contaminant exposure19; identificated changed metabolites as potencial hepatotoxicity biomarker of Polychlorinated Biphenyls (PCBs) and 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD)20; determinated toxic effects of PCBs and explained the mechanism of metabolic disturbance in obesity21; revealed the mechanisms of complex pesticide mixtures22 exposure increase oxidative stress and disturb energy metabolism.
In this study, we examined the effects of BDE-3 on the reproductive function in mice, with the lowest dose of 0.0015 mg/kg/day, which equal to the highest ingestion concentration of PBDEs in human (141 ng/kg/day) by the U.S. Environmental Protection Agency (EPA), 1.5 mg/kg/day, an equal dose to the lowest effective dose of its congener of BDE-4717, middle dose of 10 mg/kg/day and high dose of 30 mg/kg/day, respectively. We then performed metabolomics analyses for the testis, urine and serum samples after BDE-3 treatment using UPLC-Q-TOF/MS. The differential metabolites were identified by the pattern recognition using PLS-DA and univariate analysis for the control and BDE-3 treated mice. Our results may provide more evidence to better understand the mechanism of PBDEs-induced reproductive toxicity.
Result
Body, tissue weight and clinical observation
During the experimental period, animals were weighted once a week and no significant differences were observed in the body weight or clinical observation of the animals (Fig. S1 and Table S1). Testis and epididymis weight of each animal was measured on the day of the anatomy. No statistical differences in the testis weight or epididymis weight were found between the solvent group and BDE-3 groups after 6 weeks of BDE-3 treatment (Fig. S2).
Sperm count, vitality and morphology
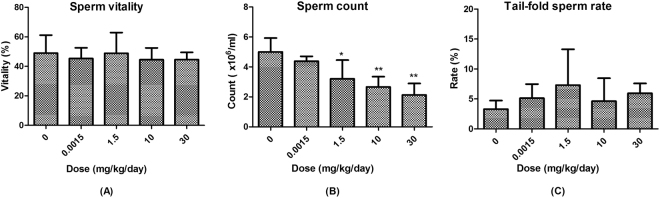
BDE-3 treated mice showed no change in sperm vitality compare with vehicle control group, (Fig. 1A). However, the sperm count was dose-dependently decreased by the treatment of BDE-3 (Fig. 1B). The average counts of sperm at 1.5, 10 and 30 mg/kg dose groups were 3.2, 2.7, 2.1 × 106/mL, and were significantly decreased by 36%, 47% and 57% compared to the vehicle control group, respectively. Moreover, as shown in Fig. 1C, in the sperm morphology analysis, the rate of tail folding sperm in BDE-3 treated mice were increased 1.4 to 2.2-fold higher compared to the vehicle group although without statistical difference. No significant differences were observed in the banana gate, enlarged-headed, amorphous, double-headed and double-tailed (Fig. S3).
Figure 1.
(A) Percentage of sperm vitality at mice treated with different concentration of BDE-3, mean ± SD (n = 6). (B) Count of sperm at mice treated with different concentration of BDE-3, mean ± SD (n = 6). *p < 0.05 versus solvent group, **p < 0.01 versus solvent group. (C) Percentage of tail-fold sperm at mice treated with different concentration of BDE-3, mean ± SD (n = 6).
Testis and epididymis histopathology
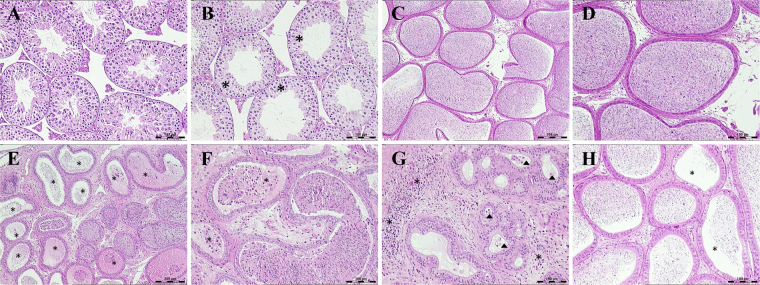
Compared with vehicle control group, minimal to mild germ cells decrease were found in seminiferous tubules in four males (4/6) of 30 mg/kg BDE-3 treated group as shown in Fig. 2A,B. Mature sperm decrease was revealed in epididymides in three males at 30 mg/kg BDE-3 treated group with 1 animal showed cellular debris in lumen and inflammatory cell infiltration in epididymal interstitium (Fig. 2C–H). No microscopically change was found in 0.0015, 1.5 and 10 mg/kg treated mice.
Figure 2.
Photomicrographs of testes and epididymides. (A) Normal structure of seminiferous tubules from vehicle control group. (B) Mild germ cells decrease in seminiferous tubules from 30 mg/kg BDE-3 treated mice. (C,D) Normal structure of epididymides from vehicle control group. (E) Oligospermiain epididymal duct (*) from 30 mg/kg BDE-3 treated mice. (F) Cellular debris in epididymal duct (*) from 30 mg/kg BDE-3 treated mice. (G) Inflammatory cell infiltration in epididymal interstitium (*) and cellular debris in epididymal duct (∆) from 30 mg/kg BDE-3 treated mice. (H) Oligospermia in epididymal duct (*) from 30 mg/kg BDE-3 treated mice.
Metabolomics profiling analysis
Metabolomic data of the testis, urine and serum samples were acquired using the abovementioned optimal conditions of UPLC-Q-TOF/MS. The representative total ion chromatograms (TICs) in both positive and negative ion modes of testis, urine and serum from the control and BDE-3 groups are shown in Fig. 3.
Figure 3.
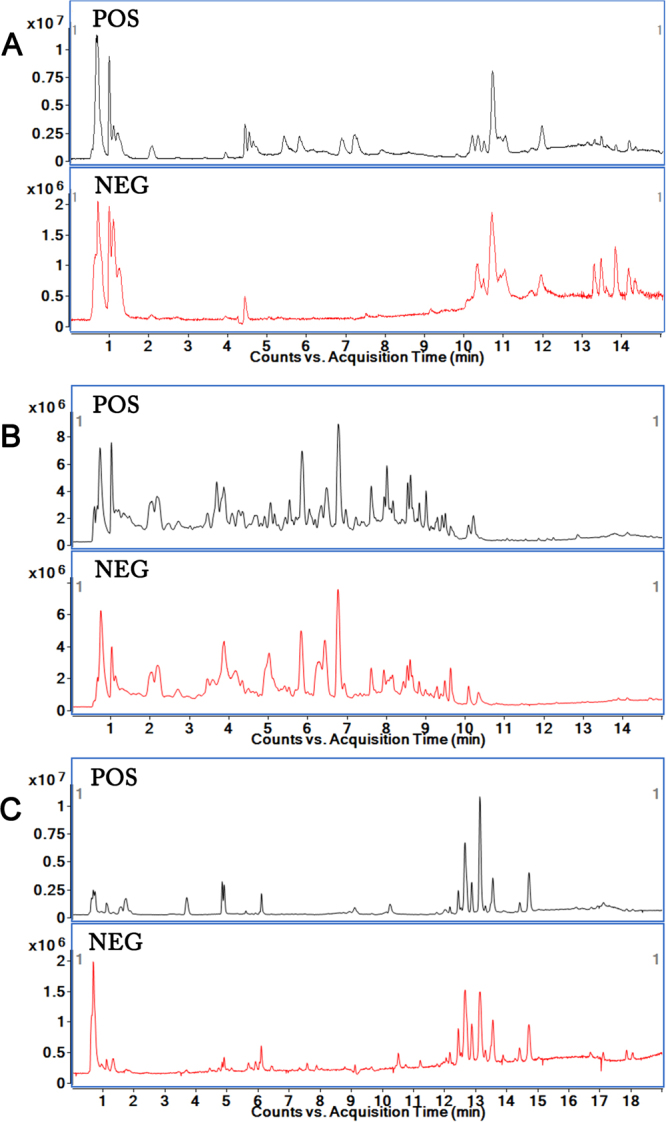
Representative total ion chromatograms (TICs) of samples based on UHPLC-Q-TOF/MS in electrospray ionization (ESI) positive and negative mode. (A) TICs of testis sample. (B) TICs of urine sample. (C) TICs of serum sample.
Initially, unsupervised PCA was used to observe the stability of the assay in the sequence between QC samples and other testis, urine and serum samples in different groups. Score plots from the PCA model have shown that the QC samples clustered together well in positive and negative mode, respectively, indicating the stability of the system was satisfying (Fig. S4).
In order to fully differentiate the testis, urine and serum metabolites between the control and the BDE-3 groups, the PLS-DA model was applied. PLS-DA is an efficient method to identify the ions that contribute to the clustering of samples. It also helps to eliminate non-correlated variations within the data set.
Score plots from the PLS-DA model have shown that the testicular samples from control group and dose groups clustered together respectively, and were clearly separated from each other (Fig. 4). Besides, the different BDE-3 groups exhibited an obvious separation trend, indicating that BDE-3 resulted in difference indeed among groups. The similar separation trend can also be observed in the score plots from the PLS-DA model of urine and serum samples (Figs S5 and S6).
Figure 4.
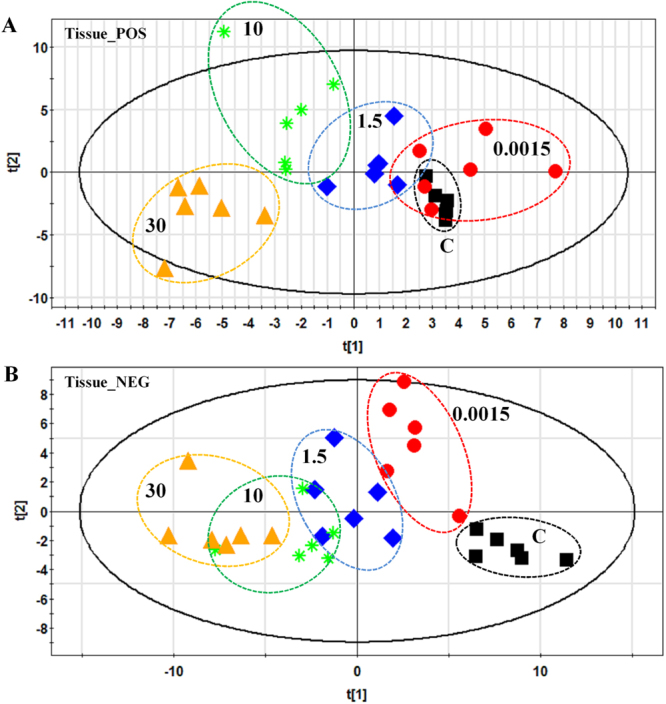
PLS-DA score plots of testis samples in the control group and BDE-3 groups at different dosages by RPLC-MS methods in positive (A) and negative (B) mode. (A) Testis samples of control group and all BDE-3 groups based on RP-MS methods in positive mode. (B) Testis samples of control group and all BDE-3 groups based on RP-MS methods in negative mode.
In order to further differentiate the testicular metabolites, we applied PLS-DA model to characterize the differences between the control and each different BDE-3 groups. There was a distinct clustering between the control group and each BDE-3 groups at different dosages by UPLC-Q-TOF/MS method (Figs S7 and S8). The detailed model validation parameters (R2X, R2Y, and Q2) were shown on the Figs S7–S12. That showed good degree of fitting and predictive ability to screen the differential variables between groups.
The corresponding S-plot showed the contribution of different variables for the difference between the control and the BDE-3 groups. Each point in the S-plot represents an ion. Ions far away from the origin are significantly important to the differences between groups and have greater VIP values. Independent sample t-test was performed to assess the statistical significance. The important ions differentiating the BDE-3 groups from the control group at different dosages (VIP > 1, p < 0.05) were identified according our previous methods. Finally, 76 differentiated metabolites were identified in testis and listed in Table 1. Furthermore, we applied the PLS-DA model to characterize the differences among urine and serum samples in each group. Score plots from the PLS-DA model have shown that dose groups clustered together respectively, and were clearly separated from the control group (Figs S5 and S6). There was also a distinct separation of control group and each BDE-3 groups at different dosages by UPLC-Q-TOF/MS method (Figs S9–S12). We used T-Test method to screen the significantly changed metabolites between the control group and each BDE-3 groups at different dosages. Using the P < 0.05 and VIP > 1 as cut-off, 38 and 31 differentiated metabolites were identified in urine and serum respectively in positive mode and negative mode eventually (See Tables 2 and 3).
Table 1.
Significantly differential metabolites in testicular tissue of BDE-3 treated mice.
| No. | M/Z | RT(min) | Ion | VIP | HMDB | Metabolites | Formula | aFC(A) | FC(B) | FC(C) | FC(D) | Pathway | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.0015 | 1.5 | 10 | 30 | ||||||||||||
| 1 | 184.073 | 0.717718 | [M+H]+ | 1.08 | 2.03 | 2.57 | 0.53 | HMDB0001565 | Phosphocholine | C5H14NO4P | — | — | 1.33‡ | — | Sphingolipid metabolism |
| 2 | 215.034 | 0.668722 | [M+Cl]− | 0.78 | 0.39 | 0.37 | 1.16 | HMDB0000122 | D-Glucose | C6H12O6 | — | — | — | 0.85* | Glycolysis |
| 3 | 162.112 | 0.677051 | [M+H]+ | 1.10 | 1.30 | 1.75 | 1.20 | HMDB0000062 | L-Carnitine | C7H15NO3 | — | — | 1.33* | — | Lysine degradation |
| 4 | 90.055 | 0.698048 | [M+H]+ | 0.31 | 0.85 | 1.56 | 0.19 | HMDB0000161 | Alanine | C3H7NO2 | — | — | 1.19† | — | Phenylalanine, tyrosine and tryptophan biosynthesis |
| 5 | 132.077 | 0.700012 | [M+H]+ | 4.13 | 9.42 | 11.54 | 1.57 | HMDB0000064 | Creatine | C4H9N3O2 | — | — | 1.22‡ | — | Arginine and proline metabolism |
| 6 | 116.07 | 0.712477 | [M+H]+ | 2.00 | 1.15 | 1.45 | 1.39 | HMDB0000162 | L-Proline | C5H9NO2 | 1.14* | — | 1.15* | — | Arginine and proline metabolism |
| 7 | 346.047 | 0.729041 | [M+H]+ | 0.13 | 1.55 | 1.28 | 1.01 | HMDB0001314 | cGMP | C10H12N5O7P | — | 1.11* | 1.15† | 1.13† | cGMP-PKG signaling pathway |
| 8 | 330.073 | 0.732617 | [M+Na]+ | 0.74 | 1.35 | 1.00 | 0.91 | HMDB0000125 | Glutathione | C10H17N3O6S | 1.08* | 1.16‡ | 1.2‡ | 1.17‡ | Glutathione metabolism |
| 9 | 259.023 | 0.733917 | [M−H]− | 0.65 | 0.43 | 1.37 | 0.56 | HMDB0001401 | D-Glucose 6-phosphate | C6H13O9P | — | — | 1.29‡ | — | Glycolysis |
| 10 | 171.007 | 0.7364 | [M−H]− | 0.30 | 0.98 | 1.55 | 0.86 | — | D-Glycerol 1-phosphate | C3H9O6P | — | — | 1.83‡ | 1.4† | |
| 11 | 175.025 | 0.868175 | [M−H]− | 0.90 | 2.36 | 4.07 | 3.67 | HMDB0000044 | L-Ascorbic acid | C6H8O6 | — | — | — | 0.6* | |
| 12 | 204.123 | 0.869535 | [M+H]+ | 3.89 | 1.19 | 2.99 | 0.25 | HMDB0000201 | Acetylcarnitine | C9H17NO4 | — | — | 1.38* | — | Insulin resistance |
| 13 | 565.048 | 0.87205 | [M−H]− | 1.21 | 0.88 | 1.67 | 1.18 | HMDB0000286 | UDP-glucose | C15H24N2O17P2 | — | — | 1.17† | 1.15* | Pyrimidine metabolism |
| 14 | 606.074 | 0.875378 | [M−H]− | 1.41 | 0.80 | 0.82 | 0.94 | HMDB0000304 | UDP-N-acetyl-D-galactosamine | C17H27N3O17P2 | 1.17* | — | — | 1.19* | Amino sugar and nucleotide sugar metabolism |
| 15 | 150.058 | 1.00794 | [M+H]+ | 3.03 | 1.07 | 0.49 | 1.96 | HMDB0000696 | L-Methionine | C5H11NO2S | 1.21‡ | — | — | 0.84† | Cysteine and methionine metabolism |
| 16 | 137.046 | 1.01139 | [M+H]+ | 2.26 | 3.94 | 2.61 | 4.59 | HMDB0000157 | Hypoxanthine | C5H4N4O | — | — | — | 0.86‡ | Purine metabolism |
| 17 | 151.026 | 1.03714 | [M−H]− | 0.93 | 1.46 | 0.36 | 1.45 | HMDB0000292 | Xanthine | C5H4N4O2 | — | 0.88† | — | 0.82‡ | Purine metabolism |
| 18 | 180.067 | 1.11158 | [M−H]− | 1.13 | 0.25 | 0.64 | 0.50 | HMDB0000158 | L-Tyrosine | C9H11NO3 | 1.22† | — | 1.17* | — | Tyrosine metabolism |
| 19 | 268.104 | 1.18454 | [M+H]+ | 2.15 | 1.67 | 2.05 | 0.83 | HMDB0000050 | Adenosine | C10H13N5O4 | 0.79* | — | 0.71† | — | Purine metabolism |
| 20 | 132.102 | 1.20861 | [M+H]+ | 9.50 | 4.85 | 0.75 | 3.67 | HMDB0000687 | L-Leucine | C6H13NO2 | 1.23‡ | — | — | 0.91* | Valine, leucine and isoleucine degradation |
| 21 | 269.088 | 1.27399 | [M+H]+ | 0.32 | 1.94 | 2.01 | 2.24 | HMDB0000195 | Inosine | C10H12N4O5 | — | — | 1.13† | 1.13† | Purine metabolism |
| 22 | 164.072 | 2.0733 | [M−H]− | 1.44 | 1.01 | 1.15 | 1.26 | HMDB0000159 | L-Phenylalanine | C9H11NO2 | 1.11* | — | 0.88* | 0.84† | Phenylalanine, tyrosine and tryptophan biosynthesis |
| 23 | 357.089 | 2.45789 | [M−H]− | 0.45 | 0.45 | 0.98 | 1.37 | HMDB0001416 | D-Pantetheine 4′-phosphate | C11H23N2O7PS | — | — | 1.12† | 1.26‡ | |
| 24 | 220.118 | 2.69811 | [M+H]+ | 0.58 | 0.22 | 0.89 | 1.11 | HMDB0000210 | Pantothenic Acid | C9H17NO5 | — | — | 0.92* | 0.9† | Vitamin digestion and absorption |
| 25 | 203.083 | 3.9441 | [M−H]− | 0.78 | 1.00 | 1.08 | 1.07 | HMDB0000929 | L-Tryptophan | C11H12N2O2 | — | 0.92* | 0.87† | 0.83‡ | Tryptophan metabolism/Aminoacyl-tRNA biosynthesis/Phenylalanine, tyrosine and tryptophan biosynthesis |
| 26 | 119.073 | 4.44353 | [M+H]+ | 0.51 | 0.88 | 1.05 | 0.20 | HMDB0040735 | Ethyl lactate | C5H10O3 | — | — | 1.21† | — | Pyruvate metabolism |
| 27 | 790.208 | 4.59486 | [M+H]+ | 1.45 | 0.13 | 0.26 | 0.47 | HMDB0001197 | Reduced flavine adenine dinucleotide (FADH2) | C27H37N9O15P2 | 1.6‡ | — | — | — | Riboflavin metabolism |
| 28 | 677.484 | 4.59681 | [M+H]+ | 1.34 | 0.19 | 0.28 | 0.56 | HMDB0011204 | PG(P-16:0/14:1) | C36H69O9P | 1.52† | — | — | — | |
| 29 | 131.072 | 5.05562 | [M−H]− | 0.57 | 1.05 | 0.88 | 0.85 | HMDB0000665 | Leucic acid | C6H12O3 | 0.88* | 0.7‡ | 0.68‡ | 0.61‡ | |
| 30 | 779.581 | 5.82323 | [M+H]+ | 0.26 | 1.09 | 1.29 | 2.26 | HMDB0010602 | PG(18:0/18:0) | C42H83O10P | — | — | 1.3‡ | 0.24‡ | |
| 31 | 957.628 | 6.86155 | [M+H]+ | 0.44 | 1.05 | 0.93 | 0.72 | HMDB0009817 | PI(21:0/22:4) | C52H93O13P | — | 1.47‡ | 1.81‡ | 0.5‡ | |
| 32 | 157.061 | 7.51482 | [M+Na]+ | 0.98 | 1.90 | 0.72 | 0.76 | HMDB0012251 | L-Canaline | C4H10N2O3 | 1.62 | 2.67‡ | 1.72 | 1.86 | |
| 33 | 621.303 | 10.0596 | [M+H]+ | 1.44 | 1.86 | 1.43 | 1.80 | HMDB0009897 | PI(20:4/0:0) | C29H49O12P | 0.81‡ | 0.73‡ | 0.66‡ | 0.46‡ | |
| 34 | 478.293 | 10.2657 | [M+H]+ | 0.22 | 1.08 | 0.25 | 0.35 | HMDB0011477 | LysoPE(0:0/18:2) | C23H44NO7P | — | 1.24† | — | — | |
| 35 | 524.278 | 10.2884 | [M−H]− | 2.32 | 2.21 | 2.02 | 2.04 | HMDB0011496 | LysoPE(0:0/22:6) | C27H44NO7P | 0.77‡ | 0.76‡ | 0.73‡ | 0.64‡ | |
| 36 | 1001.56 | 10.3001 | [M+Cl]− | 1.49 | 1.49 | 1.49 | 1.34 | HMDB0009791 | PI(22:2/22:4) | C53H91O13P | 0.64‡ | 0.57‡ | 0.45‡ | 0.39‡ | |
| 37 | 520.34 | 10.304 | [M+H]+ | 0.68 | 3.06 | 0.55 | 0.56 | HMDB0010386 | LysoPC(18:2) | C26H50NO7P | — | 1.26† | — | — | |
| 38 | 500.279 | 10.3084 | [M−H]− | 4.38 | 4.44 | 4.23 | 4.19 | HMDB0011487 | LysoPE(0:0/20:4) | C25H44NO7P | 0.79‡ | 0.75‡ | 0.71‡ | 0.61‡ | |
| 39 | 568.34 | 10.3287 | [M+H]+ | 3.44 | 1.46 | 2.39 | 1.93 | HMDB0010404 | LysoPC(22:6) | C30H50NO7P | 0.83† | — | 0.84† | 0.87* | |
| 40 | 1043.61 | 10.3316 | [M+Cl]− | 1.50 | 1.43 | 1.57 | 1.43 | HMDB0004888 | Ganglioside GA2 (d18:1/12:0) | C50H92N2O18 | 0.73‡ | 0.69‡ | 0.56‡ | 0.5‡ | |
| 41 | 566.321 | 10.3614 | [M+Na]+ | 0.72 | 0.67 | 1.09 | 0.25 | HMDB0010396 | LysoPC(20:4) | C28H50NO7P | — | — | 1.22† | — | |
| 42 | 546.355 | 10.5766 | [M+H]+ | 3.82 | 4.56 | 3.63 | 4.11 | HMDB0010393 | LysoPC(20:3) | C28H52NO7P | 0.69‡ | 0.59‡ | 0.48‡ | 0.31‡ | |
| 43 | 448.342 | 10.5995 | [M+H]+ | 1.07 | 0.88 | 1.02 | 1.04 | HMDB0062343 | N-stearoyl tyrosine | C27H45NO4 | 1.5* | — | 1.73‡ | 1.76‡ | |
| 44 | 454.293 | 10.64 | [M+H]+ | 1.82 | 4.81 | 4.33 | 5.75 | HMDB0011473 | LysoPE(0:0/16:0) | C21H44NO7P | — | 0.91† | 0.85‡ | 0.75‡ | |
| 45 | 571.289 | 10.6421 | [M−H]− | 1.03 | 1.27 | 0.64 | 1.53 | — | PI(16:0/0:0) | C25H49O12P | — | — | — | 0.89† | |
| 46 | 496.268 | 10.6473 | [M−H]− | 1.31 | 1.07 | 1.01 | 0.93 | — | PS(16:0/0:0) | C22H44NO9P | — | — | 0.92* | — | |
| 47 | 518.321 | 10.7014 | [M+Na]+ | 1.73 | 2.28 | 2.22 | 1.66 | HMDB0010382 | LysoPC(16:0) | C24H50NO7P | — | 1.19* | 1.41‡ | 1.21* | |
| 48 | 528.308 | 10.8622 | [M+H]+ | 3.82 | 2.46 | 2.61 | 2.63 | HMDB0011495 | LysoPE(0:0/22:5) | C27H46NO7P | 0.77‡ | 0.87* | 0.8‡ | 0.77‡ | |
| 49 | 1095.64 | 10.8841 | [M−H]− | 1.53 | 1.33 | 1.32 | 1.07 | HMDB0004842 | Ganglioside GM3 (d18:1/12:0) | C53H96N2O21 | 0.52‡ | 0.55‡ | 0.43‡ | 0.48‡ | |
| 50 | 570.355 | 10.9047 | [M+H]+ | 6.34 | 1.51 | 3.77 | 1.44 | HMDB0010403 | LysoPC(22:5) | C30H52NO7P | 0.81† | 0.95 | 0.85* | 0.97 | |
| 51 | 438.299 | 10.9115 | [M−H]− | 1.51 | 0.97 | 1.70 | 0.50 | HMDB0005780 | PE(O-16:0/0:0) | C21H46NO6P | 1.32‡ | 1.21* | 1.63‡ | 0.86 | |
| 52 | 480.309 | 10.9772 | [M+H]+ | 0.95 | 1.92 | 1.99 | 3.33 | HMDB0011476 | LysoPE(0:0/18:1) | C23H46NO7P | — | — | 0.87† | 0.74‡ | |
| 53 | 438.298 | 11.0137 | [M+H]+ | 1.72 | 1.67 | 1.03 | 1.24 | HMDB0011152 | PE(P-16:0/0:0) | C21H44NO6P | 1.11* | 1.09* | 1.09* | 0.89* | |
| 54 | 528.309 | 11.1544 | [M−H]− | 0.81 | 1.17 | 1.13 | 1.12 | HMDB0011493 | LysoPE(0:0/22:4) | C27H48NO7P | 0.88† | 0.76‡ | 0.69‡ | 0.59‡ | |
| 55 | 508.375 | 11.2894 | [M+H]+ | 0.92 | 1.14 | 1.19 | 0.19 | — | PC(O-18:1(9E)/0:0) | C26H54NO6P | — | 1.24* | 1.47‡ | 1.02 | |
| 56 | 483.273 | 11.6194 | [M−H]− | 1.55 | 1.47 | 1.92 | 1.04 | — | PG(16:0/0:0) | C22H45O9P | 0.92* | 0.88† | 0.76‡ | 0.89† | |
| 57 | 556.339 | 11.6446 | [M+Na]+ | 1.05 | 0.88 | 0.98 | 1.11 | HMDB0011492 | LysoPE(0:0/22:2) | C27H52NO7P | 0.85‡ | 0.87† | 0.74‡ | 0.67‡ | |
| 58 | 504.306 | 11.8836 | [M+H]+ | 0.22 | 0.44 | 0.51 | 1.04 | HMDB0011485 | LysoPE(0:0/20:3) | C25H46NO7P | — | — | 0.89* | 0.65‡ | |
| 59 | 480.31 | 11.8854 | [M−H]− | 1.25 | 1.40 | 2.05 | 3.41 | HMDB0011129 | LysoPE(0:0/18:0) | C23H48NO7P | — | — | 0.87† | 0.61‡ | |
| 60 | 524.371 | 11.947 | [M+H]+ | 5.50 | 4.13 | 4.66 | 8.38 | HMDB0010384 | LysoPC(18:0) | C26H54NO7P | 1.08* | — | 0.87† | 0.71‡ | |
| 61 | 1047.73 | 11.9471 | [M+Na]+ | 1.15 | 3.94 | 3.58 | 4.31 | HMDB0055889 | TG(22:5/22:6/22:6) | C69H100O6 | — | 0.76† | 0.56‡ | 0.43‡ | |
| 62 | 546.353 | 11.9579 | [M+H]+ | 1.10 | 1.33 | 1.30 | 2.27 | — | PS(20:4/0:0) | C26H44NO9P | — | — | 0.9* | 0.78‡ | |
| 63 | 600.402 | 12.1688 | [M+Na]+ | 0.23 | 1.10 | 1.25 | 0.95 | HMDB0010399 | LysoPC(22:1) | C30H60NO7P | — | — | 0.75‡ | 0.85† | |
| 64 | 524.299 | 12.2169 | [M−H]− | 0.45 | 1.14 | 1.07 | 1.56 | — | PS(18:0/0:0) | C24H48NO9P | — | 0.92* | 0.88† | 0.75‡ | |
| 65 | 466.329 | 12.2984 | [M+H]+ | 0.18 | 0.23 | 0.19 | 1.02 | — | PE(P-18:0/0:0) | C26H43NO6 | — | — | — | 0.63‡ | |
| 66 | 300.289 | 12.6925 | [M+H]+ | 1.53 | 1.88 | 1.76 | 2.32 | HMDB0000252 | Sphingosine | C18H37NO2 | 0.8‡ | 0.74‡ | 0.53‡ | 0.7‡ | Sphingolipid metabolism |
| 67 | 756.551 | 12.9843 | [M+H]+ | 0.59 | 1.87 | 2.42 | 1.41 | HMDB0008166 | PC(18:3/16:0) | C42H78NO8P | — | — | 5.14‡ | — | |
| 68 | 303.233 | 13.4473 | [M−H]− | 3.87 | 4.80 | 4.13 | 4.31 | HMDB0001043 | Arachidonic Acid (peroxide free) | C20H32O2 | 0.89‡ | 0.83‡ | 0.82‡ | 0.75‡ | Biosynthesis of unsaturated fatty acids |
| 69 | 788.554 | 13.4499 | [M+H]+ | 1.10 | 1.25 | 1.40 | 1.50 | HMDB0012390 | PS(18:1/18:1) | C42H78NO10P | 0.68 | 0.67* | 0.41† | 0.34‡ | |
| 70 | 353.266 | 13.5693 | [M+Na]+ | 0.54 | 1.12 | 1.01 | 0.83 | — | 1-Monopalmitin | C19H38O4 | — | 0.58† | 0.31‡ | 0.47‡ | |
| 71 | 279.233 | 13.5874 | [M−H]− | 1.62 | 1.07 | 1.54 | 1.27 | HMDB0000673 | Linoleic acid | C18H32O2 | 0.76† | 0.85* | 0.68‡ | 0.68‡ | Biosynthesis of unsaturated fatty acids |
| 72 | 754.536 | 14.1461 | [M+H]+ | 1.28 | 0.88 | 0.54 | 0.74 | HMDB0007974 | PC(16:0/18:4) | C42H76NO8P | 0.79* | — | — | 0.79* | |
| 73 | 804.551 | 14.1466 | [M+H]+ | 2.98 | 4.50 | 3.73 | 4.10 | HMDB0008023 | PC(16:1/22:6) | C46H78NO8P | 0.84† | 0.75‡ | 0.65‡ | 0.57‡ | |
| 74 | 805.554 | 14.1495 | [M+H]+ | 1.62 | 1.28 | 1.40 | 2.42 | HMDB0115266 | PA(22:0/22:6) | C47H81O8P | — | — | — | 0.81* | |
| 75 | 255.233 | 14.1514 | [M−H]− | 2.04 | 1.39 | 1.42 | 1.39 | HMDB0000220 | Palmitic acid | C16H32O2 | 0.72‡ | 0.82‡ | 0.78‡ | 0.71‡ | Biosynthesis of unsaturated fatty acids/Fatty acid metabolism |
| 76 | 381.174 | 14.307 | [M+Cl]- | 1.23 | 1.98 | 2.07 | 2.02 | HMDB0001547 | corticosterone | C21H30O4 | — | 0.86‡ | 0.8‡ | 0.74‡ | Steroid hormone biosynthesis |
aFC, fold changes at different dosages, compared to the control mice. *p < 0.05, †p < 0.01, ‡p < 0.001, “—” no statistically significant changes.
Table 2.
Significantly differential metabolites in urine of BDE-3 treated mice.
| NO. | M/Z | Ion | VIP | HMDB | Metabolite | Formula | aFC(A) | FC(B) | FC(C) | FC(D) | Pathway | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.0015 | 1.5 | 10 | 30 | |||||||||||
| 1 | 128.036 | [M−H]− | 0.77 | 0.60 | 1.42 | 0.89 | HMDB0000267 | Pyroglutamic acid | C5H7NO3 | — | — | 2.24† | — | Glutathione metabolism |
| 2 | 129.021 | [M−H]− | 1.29 | 2.33 | 2.24 | 3.81 | — | Acetylpyruvate | C5H6O4 | — | — | — | 0.62* | |
| 3 | 130.052 | [M−H]− | 0.59 | 0.23 | 1.30 | 0.22 | HMDB0002113 | 3-Hydroxy-L-proline | C5H9NO3 | — | — | — | 0.78* | |
| 4 | 132.077 | [M+H]+ | 0.96 | 0.96 | 3.79 | 2.62 | HMDB0000064 | Creatine | C4H9N3O2 | — | 0.33* | — | — | Arginine and proline metabolism |
| 5 | 132.101 | [M+H]+ | 2.22 | 2.22 | 2.09 | 1.45 | HMDB0000687 | L-Leucine | C6H13NO2 | 0.79* | 0.73* | — | — | Valine, leucine and isoleucine degradation |
| 6 | 136.039 | [M+H]+ | 1.99 | 1.99 | 1.28 | 1.32 | HMDB0000742 | L-Homocysteine | C4H9NO2S | 1.88† | 2.24† | — | — | Biosynthesis of amino acids |
| 7 | 137.071 | [M+Na]+ | 0.31 | 0.31 | 1.60 | 1.28 | — | L-Prolinamide | C5H10N2O | — | 2.08* | — | — | |
| 8 | 139.051 | [M+H]+ | 1.07 | 1.07 | 1.71 | 1.77 | HMDB0002730 | Nicotinamide N-oxide | C6H6N2O2 | — | — | 1.73† | — | |
| 9 | 145.016 | [M+FA−H]− | 0.40 | 1.21 | 0.69 | 2.32 | HMDB0032523 | Succinic anhydride | C4H4O3 | 0.78* | — | — | — | |
| 10 | 162.079 | [M+H]+ | 1.11 | 1.11 | 2.65 | 1.74 | — | Alanopine | C6H11NO4 | 0.68‡ | 0.51‡ | — | — | |
| 11 | 170.043 | [M+Na]+ | 0.94 | 0.94 | 0.96 | 0.59 | — | L-Glutamate | C5H9NO4 | 1.61* | 1.83† | — | — | D-Glutamine and D-glutamate metabolism |
| 12 | 171.067 | [M+Na]+ | 1.19 | 1.19 | 1.03 | 0.59 | HMDB0000227 | Mevalonic acid | C6H12O4 | — | — | — | 1.35* | Pantothenate and CoA biosynthesis |
| 13 | 176.039 | [M−H]− | 1.32 | 0.99 | 2.39 | 1.54 | HMDB0001015 | N-Formylmethionine | C6H11NO3S | — | — | 1.61* | — | Cysteine and methionine metabolism |
| 14 | 189.098 | [M−H]− | 1.45 | 1.43 | 0.26 | 0.38 | — | beta-Hydroxyarginine | C6H14N4O3 | — | — | — | 0.65* | Arginine and proline metabolism |
| 15 | 190.107 | [M+H]+ | 0.94 | 0.94 | 0.48 | 0.70 | — | Alexine | C8H15NO4 | — | — | 1.72* | 1.89† | |
| 16 | 204.069 | [M+Na]+ | 1.57 | 1.57 | 0.89 | 0.65 | HMDB0000158 | L-Tyrosine | C9H11NO3 | — | 0.51* | — | — | Tyrosine metabolism |
| 17 | 206.044 | [M+H]+ | 3.33 | 3.33 | 5.59 | 4.68 | HMDB0000881 | Xanthurenic acid | C10H7NO4 | — | — | 0.15* | — | Tryptophan metabolism |
| 18 | 226.143 | [M+H]+ | 1.23 | 1.23 | 1.62 | 0.80 | — | Prenalterol | C12H19NO3 | — | — | — | 1.45* | |
| 19 | 232.117 | [M+H]+ | 1.68 | 1.68 | 2.86 | 3.68 | HMDB0000953 | Suberylglycine | C10H17NO5 | — | — | — | 1.29* | |
| 20 | 238.022 | [M+FA−H]− | 0.54 | 0.56 | 1.29 | 1.68 | HMDB0041624 | Phosphocreatinine | C4H8N3O4P | — | — | 1.37* | 1.45* | |
| 21 | 241.093 | [M+Na]+ | 0.73 | 0.73 | 0.99 | 1.19 | HMDB0001238 | N-Acetylserotonin | C12H14N2O2 | — | — | 1.81‡ | — | Tryptophan metabolism |
| 22 | 245.011 | [M−H]− | 2.68 | 1.28 | 0.86 | 4.32 | — | Phosphatidyl glycerol | C6H15O8P | — | — | 2.97‡ | 3.45† | Glycerophospholipid metabolism |
| 23 | 245.095 | [M+H]+ | 0.44 | 0.44 | 0.88 | 1.89 | HMDB0000030 | D-Biotin | C10H16N2O3S | 0.66* | — | — | — | Biotin metabolism |
| 24 | 248.113 | [M+H]+ | 1.60 | 1.60 | 2.00 | 1.89 | HMDB0002095 | Malonylcarnitine | C10H17NO6 | — | — | 1.38* | 1.39* | |
| 25 | 258.104 | [M+H]+ | 1.18 | 1.18 | 0.67 | 0.60 | HMDB0000982 | 5-Methylcytidine | C10H15N3O5 | — | — | 1.34* | — | |
| 26 | 268.104 | [M+H]+ | 2.81 | 2.81 | 2.53 | 0.57 | HMDB0000050 | Adenosine | C10H13N5O4 | — | — | 1.30* | — | Purine metabolism |
| 27 | 274.11 | [M+H]+ | 0.32 | 0.32 | 0.33 | 0.71 | HMDB0000667 | L-Thyronine | C15H15NO4 | — | 0.42† | — | — | |
| 28 | 283.114 | [M+H]+ | 0.23 | 0.23 | 1.03 | 1.14 | — | 2-Aminoadenosine | C10H14N6O4 | — | — | 1.28* | — | |
| 29 | 319.184 | [M+H]+ | 1.07 | 1.07 | 1.46 | 1.28 | HMDB0006709 | Ubiquinone (Q2) | C19H26O4 | — | — | 1.76* | — | Oxidative phosphorylation |
| 30 | 321.044 | [M−H]− | 4.12 | 3.84 | 1.42 | 1.64 | — | dTMP | C10H15N2O8P | 1.33* | 1.36* | — | — | Pyrimidine metabolism |
| 31 | 348.088 | [M−H]− | 1.13 | 1.09 | 0.16 | 0.91 | HMDB0060507 | S-(Formylmethyl)glutathione | C12H19N3O7S | — | 1.59† | 1.80† | 1.91† | Metabolism of xenobiotics by cytochrome P450 |
| 32 | 375.131 | [M−H]− | 1.18 | 1.30 | 0.56 | 0.69 | HMDB0000244 | Riboflavin (Vitamin B2) | C17H20N4O6 | — | — | — | 0.54* | Riboflavin metabolism |
| 33 | 377.145 | [M−H]− | 1.32 | 1.36 | 0.82 | 0.84 | HMDB0001577 | Reduced riboflavin | C17H22N4O6 | 0.87* | — | 0.84* | — | Riboflavin metabolism |
| 34 | 397.235 | [M+FA-H]- | 2.84 | 2.52 | 0.42 | 0.90 | HMDB0001220 | PGE2 | C20H32O5 | — | — | 0.63* | — | Arachidonic acid metabolism |
| 35 | 482.361 | [M+H]+ | 0.11 | 0.11 | 1.21 | 0.91 | — | Lyso-PAF C-16 | C24H52NO6P | — | — | 0.68* | — | Ether lipid metabolism |
| 36 | 496.34 | [M+H]+ | 1.22 | 1.22 | 2.04 | 0.97 | HMDB0010382 | LysoPC(16:0) | C24H50NO7P | — | — | — | 1.20* | Glycerophospholipid metabolism |
| 37 | 569.333 | [M+FA-H]- | 0.91 | 1.82 | 0.68 | 0.43 | — | PG(P-20:0/0:0) | C26H53O8P | — | 0.75* | 0.58‡ | 0.72† | |
| 38 | 605.309 | [M+FA-H]- | 0.62 | 1.44 | 0.48 | 0.32 | — | PG(22:4/0:0) | C28H49O9P | 0.80* | 0.61† | 0.77* | — | |
aFC, fold changes at different dosages, compared to the control mice. *p < 0.05, †p < 0.01, ‡p < 0.001, “—” no statistically significant changes.
Table 3.
Significantly differential metabolites in serum of BDE-3 treated mice.
| NO. | m/z | Ion | VIP | HMDB | Metabolite | Formula | aFC(0.0015) | FC(1.5) | FC(10) | FC(30) | Pathway | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0.0015 | 1.5 | 10 | 30 | |||||||||||
| 1 | 116.071 | [M+H]+ | 1.85 | 1.62 | 1.34 | 1.37 | HMDB0000162 | L-Proline | C5H9NO2 | 1.46* | 1.65† | — | 1.51* | Arginine and proline metabolism |
| 2 | 118.086 | [M+H]+ | 2.43 | 2.58 | 1.93 | 2.05 | HMDB0000883 | L-Valine | C5H11NO2 | — | 1.51† | 1.41* | 1.47* | Valine, leucine and isoleucine degradation |
| 3 | 129.056 | [M−H]− | 0.81 | 1.38 | 1.46 | 1.86 | HMDB0000695 | Ketoleucine | C6H10O3 | — | 1.71* | 1.57* | 1.82† | Valine, leucine and isoleucine degradation |
| 4 | 130.087 | [M−H]− | 0.96 | 1.13 | 1.08 | 0.99 | HMDB0000687 | L-Leucine | C6H13NO2 | — | 1.58† | 1.46† | 1.39* | Valine, leucine and isoleucine degradation |
| 5 | 136.076 | [M+H]+ | 1.50 | 0.84 | 1.41 | 0.85 | HMDB0010715 | 2-Phenylacetamide | C8H9NO | 1.42† | 1.35* | 1.76† | 1.33* | Phenylalanine metabolism |
| 6 | 150.058 | [M+H]+ | 2.17 | 1.44 | 1.42 | 1.17 | HMDB0000696 | L-Methionine | C5H11NO2S | 1.34* | 1.33* | — | — | Cysteine and methionine metabolism |
| 7 | 157.061 | [M+H]+ | 1.28 | 0.89 | 0.56 | 1.22 | HMDB0001014 | 4-Imidazolone-5-propionic acid | C6H8N2O3 | 1.53‡ | 1.50* | 1.56† | 1.45† | Histidine metabolism |
| 8 | 162.112 | [M+H]+ | 1.55 | 0.61 | 1.01 | 1.57 | HMDB0000062 | L-Carnitine | C7H15NO3 | — | — | — | 1.34* | Lysine degradation |
| 9 | 164.071 | [M−H]− | 1.18 | 1.29 | 1.24 | 1.01 | HMDB0000159 | L-Phenylalanine | C9H11NO2 | 1.26* | 1.36† | 1.34† | 1.25* | Phenylalanine, tyrosine and tryptophan biosynthesis |
| 10 | 165.055 | [M+H]+ | 1.44 | 0.83 | 1.41 | 0.82 | — | L-Tyrosine fragment | C9H8O3 | 1.40† | 1.34* | 1.77† | 1.31* | |
| 11 | 172.991 | [M−H]− | 1.89 | 1.27 | 0.24 | 1.38 | — | Phenyl sulfate | C6H6O4S | 2.61* | 1.73* | — | 2.11* | |
| 12 | 182.081 | [M+H]+ | 3.61 | 1.93 | 3.34 | 1.94 | HMDB0000158 | L-Tyrosine | C9H11NO3 | 1.42† | 1.33* | 1.75† | 1.31* | Tyrosine metabolism |
| 13 | 185.154 | [M+H]+ | 1.45 | 1.34 | 1.39 | 1.50 | — | 2-hendecenoic acid | C11H20O2 | 1.92‡ | 2.28‡ | 2.29‡ | 2.44‡ | |
| 14 | 188.071 | [M+H]+ | 3.30 | 1.57 | 2.80 | 2.31 | — | L-Tryptophan fragment | C11H9NO2 | 1.25† | — | 1.32‡ | 1.24† | |
| 15 | 188.986 | [M−H]− | 1.21 | 1.35 | 0.32 | 1.16 | HMDB0059724 | Pyrocatechol sulfate | C6H6O5S | 1.87* | 2.10* | — | 1.91* | |
| 16 | 203.082 | [M−H]− | 1.68 | 0.74 | 1.46 | 1.05 | HMDB0000929 | L-Tryptophan | C11H12N2O2 | 1.19† | — | 1.20† | 1.12* | Phenylalanine, tyrosine and tryptophan biosynthesis |
| 17 | 212.002 | [M−H]− | 2.74 | 3.14 | 1.23 | 2.28 | — | Indoxylsulfuric acid | C8H7NO4S | 2.08† | 2.22† | — | 1.95* | |
| 18 | 225.123 | [M+NH4]+ | 1.62 | 1.13 | 0.68 | 1.56 | HMDB0000512 | N-Acetyl-L-phenylalanine | C11H13NO3 | 0.50† | 0.43† | — | 0.35‡ | Phenylalanine metabolism |
| 19 | 231.159 | [M+H]+ | 1.22 | 1.11 | 1.11 | 1.23 | HMDB0000623 | Dodecanedioic acid | C12H22O4 | 1.95‡ | 2.30‡ | 2.24‡ | 2.46‡ | |
| 20 | 267.073 | [M−H]− | 0.73 | 0.44 | 2.18 | 0.58 | HMDB0000195 | Inosine | C10H12N4O5 | — | — | 3.26‡ | — | Purine metabolism |
| 21 | 454.294 | [M+H]+ | 1.40 | 1.19 | 1.10 | 1.04 | HMDB0011473 | LysoPE(0:0/16:0) | C21H44NO7P | 1.27* | 1.44† | 1.32* | 1.30* | |
| 22 | 478.293 | [M−H]− | 0.63 | 1.31 | 1.10 | 0.72 | HMDB0011505 | LysoPE(18:1/0:0) | C23H46NO7P | — | 1.73† | 1.43† | — | Pentose and glucuronate interconversions |
| 23 | 478.294 | [M+H]+ | 0.16 | 2.91 | 2.18 | 0.58 | HMDB0011507 | LysoPE(18:2/0:0) | C23H44NO7P | — | 1.92‡ | 1.51† | — | |
| 24 | 494.325 | [M+H]+ | 4.34 | 2.99 | 3.84 | 2.74 | HMDB0010383 | LysoPC(16:1) | C24H48NO7P | 1.81‡ | 2.06* | 2.14‡ | 1.69† | Glycerophospholipid metabolism |
| 25 | 518.323 | [M+H]+ | 1.59 | 1.53 | 1.56 | 1.47 | HMDB0010387 | LysoPC(18:3) | C26H48NO7P | 1.09* | 1.16‡ | 1.15‡ | 1.14† | Pentose and glucuronate interconversions |
| 26 | 522.357 | [M+H]+ | 8.72 | 7.06 | 8.03 | 6.31 | HMDB0010385 | LysoPC(18:1) | C26H52NO7P | 1.49† | 1.74† | 1.76† | 1.52* | |
| 27 | 542.325 | [M+H]+ | 2.14 | 2.06 | 2.51 | 1.87 | HMDB0010397 | LysoPC(20:5/0:0) | C28H48NO7P | 1.44‡ | 1.94† | 1.97‡ | 1.63‡ | |
| 28 | 544.341 | [M+H]+ | 0.81 | 1.71 | 2.55 | 1.20 | HMDB0010396 | LysoPC(20:4) | C28H50NO7P | — | — | 1.20* | — | Glycerophospholipid metabolism |
| 29 | 546.357 | [M+H]+ | 2.17 | 1.86 | 3.03 | 2.53 | HMDB0010393 | LysoPC(20:3) | C28H52NO7P | — | 1.45* | 1.80† | 1.53† | Pentose and glucuronate interconversions |
| 30 | 570.356 | [M+H]+ | 1.14 | 1.05 | 1.39 | 1.04 | HMDB0010403 | LysoPC(22:5) | C30H52NO7P | — | 2.01† | 2.03† | — | Pentose and glucuronate interconversions |
| 31 | 619.288 | [M+Cl]- | 0.37 | 1.64 | 1.76 | 0.81 | HMDB0002577 | Cholic acid glucuronide | C30H48O11 | — | 1.89† | 1.84† | — | Pentose and glucuronate interconversions |
aFC, fold changes at different dosages, compared to the control mice. *p < 0.05, †p < 0.01, ‡p < 0.001, “—” no statistically significant changes.
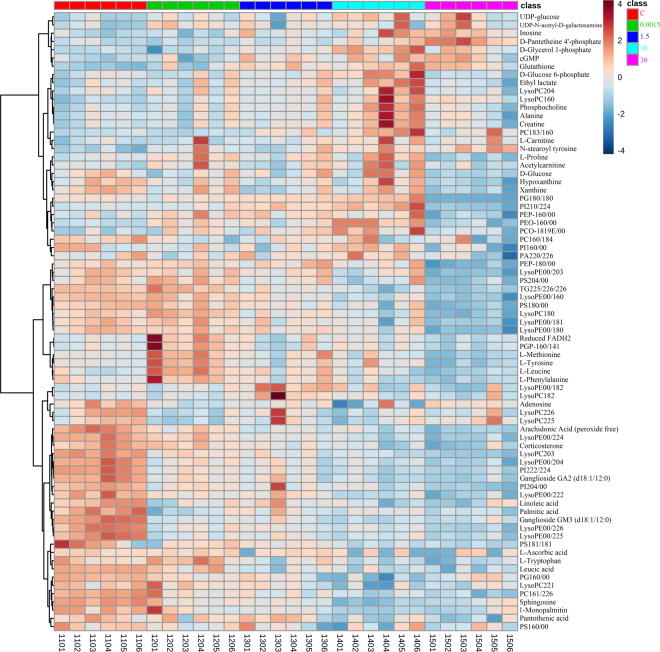
Heat maps can visually display the gradient changes of the differential metabolites between the control and BDE-3 groups. The heat map was constructed (Fig. 5) based on the normalized data set of the differential metabolites in testis, urine and serum. According to our results, in testicular tissue, Phosphocholine, L-Carnitine, Alanine, Creatine, L-Proline, cGMP, Glutathione, D-Glucose 6-phosphate, D-Glycerol 1-phosphate, Acetylcarnitine, UDP-glucose, UDP-N-acetyl-D-galactosamine, L-Tyrosine, Inosine, D-Pantetheine 4′-phosphate, Reduced flavine adenine dinucleotide (FADH2), N-stearoyl tyrosine and PG(P-16:0/14:1) were increased in BDE-3 treated groups. Leucic acid, Ganglioside GA2 (d18:1/12:0), Ganglioside GM3 (d18:1/12:0), Arachidonic Acid (peroxide free), Palmitic acid, corticosterone and amount of lipids metabolites were obviously decreased in the BDE-3 groups compared with the control group.
Figure 5.
The clustering heat map of the control (C) and BDE-3 mice across different dosages (0.0015, 1.5, 10, 30) based on the 76 differentially metabolites in testicular tissue. Each column is labeled with different colors according to the sample type.
In urine (Fig. S13A), 2-Aminoadenosine, 5-Methylcytidine, Adenosine, Alexine, dTMP, L-Glutamate, L-Homocysteine, L-Prolinamide, LysoPC(16:0), Malonylcarnitine, Mevalonic acid, N-Acetylserotonin, N-Formylmethionine, Nicotinamide N-oxide, Phosphatidyl glycerol, Phosphocreatinine, Prenalterol, Pyroglutamic acid, S-(Formylmethyl)glutathione, Suberylglycine, Ubiquinone (Q2)significantly increased in the BDE-3 groups compared with the control group. However, the metabolites including 3-Hydroxy-L-proline, Acetylpyruvate, Alanopine, beta-Hydroxyarginine, Creatine, D-Biotin, L-Leucine, L-Thyronine, L-Tyrosine, Lyso-PAF C-16, PG(22:4/0:0), PG(P-20:0/0:0), PGE2, Reduced riboflavin, Riboflavin (Vitamin B2), Succinic anhydride, Xanthurenic acid were down-regulated. In serum (Fig. S13B), N-Acetyl-L-phenylalanine was down-regulated in BDE-3 groups. However, most metabolites were up-regulated including L-Proline, L-Valine, Ketoleucine, L-Leucine, 2-Phenylacetamide, L-Methionine, 4-Imidazolone-5-propionic acid, L-Carnitine, L-Phenylalanine, L-Tyrosine fragment, Phenyl sulfate, L-Tyrosine, 2-hendecenoic acid, L-Tryptophan fragment, Pyrocatechol sulfate, L-Tryptophan, Indoxylsulfuric acid, Dodecanedioic acid, Inosine, LysoPE(0:0/16:0), LysoPE(18:1/0:0), LysoPE(18:2/0:0), LysoPC(16:1), LysoPC(18:3), LysoPC(18:1), LysoPC(20:5/0:0), LysoPC(20:4), LysoPC(20:3), LysoPC(22:5) and Cholic acid glucuronide.
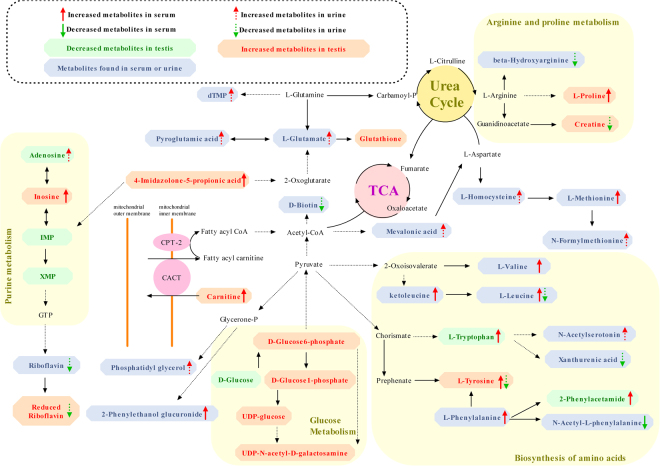
The method applied to identify the differential metabolites was described above. Using this method, we identified a total of 76 differential metabolites in testis, 38 differential metabolites in urine and 31 differential metabolites in serum. The detailed information is shown in Table 1. Using the KEGG pathway database (http://www.genome.jp/kegg/)23–25, we constructed the metabolic network (Fig. 6) that clearly showed that BDE-3-induced toxicity was related to the alterations in the Arginine and proline metabolism, Valine, leucine and isoleucine degradation, Phenylalanine metabolism, Cysteine and methionine metabolism, Tyrosine metabolism, Purine metabolism, Pentose and glucuronate interconversions, Tryptophan metabolism, Biotin metabolism, Oxidative phosphorylation, Riboflavin metabolism, and Glycerophospholipid metabolism.
Figure 6.
Metabolic pathways related to the differential metabolites identified in the BDE-3 groups.
Discussion
BDE-3 is the most fundamental mono-BDE which could be degraded from PBDEs in environment, although its bioaccumulation properties and potential healthy risk have raised concern, the effect on the adult male reproductive system are currently unclear. To our knowledge, it is the first report on assessment of male reproductive toxicity induced by mono-BDE in rodent. In this study, we found that short-term exposure to low-dose BDE-3 may have adverse effects on spermatogenesis in adult male mice. The sperm count decreased after the treatment of BDE-3 for 6 weeks at a very low dose (0.0015 mg/kg/day, a dose based on environmental exposure), and at the dose of ≥1.5 mg/kg/day, the sperm count decreased with statistical significance, and microscopical changes of germ cell loss in seminiferous tubules and epididymides were clearly observed at the dose of 30 mg/kg. Our previous studies on C. Elegans revealed that BDE-3 could induce ROS and DNA damage in germ cell16, which might be involved in the male reproductive toxicity that observed in mice.
Metabolomics has been applied in toxicity studies of chemicals as a newly emerging technology, especially in risk assessment of low dose exposure of environmental pollutants26–29, it may lead to critical insights because of its high sensitivity. Although several studies on toxicological effects of PBDEs using metabolomics analysis have been reported30–32, studies in rodent are limited and few data of mono-BDE is available. In this study, to obtain a better understanding of the toxic mechanism of BDE-3, we applied the UPLC-Q-TOF/MS profile to the metabolites of testicular tissue, urine and serum in the control and BDE-3 treated mice at different dosages.
In our study, we analyzed the testis tissue, serum and urine of BDE-3 treated mice. The differential metabolites from biological samples at different levels were screened and analyzed. As mentioned above, testis is susceptible to BDE-3 induced toxicity, so the metabolic alterations of testis tissue could, to some extent, reflect BDE-3 induced reproductive toxicity. Based on the pathway enrichment analysis on differential metabolites screened in testis tissue, we could preliminarily reveal that BDE-3 induced toxicity disturbed organism metabolic regulation network. Meanwhile, the metabolic alterations would spread through blood circulation and urine directly or indirectly. So pathway enrichment analysis and correlation analysis on differential metabolites screened in serum and urine could further clarify and verify the BDE-3 induced metabolic alterations. Besides, body fluid sample like serum and urine could reflect the systematic metabolic state of the body, so the differential metabolites which were highly correlated with BDE-3 induced toxicity screened in body fluid were expected to be potential toxic biomarker, improving the efficiency to detect the toxicity of PBDEs. In total, 76 differential metabolites in testicular tissue, 38 differential metabolites in urine and 31 differential metabolites in serum were identified. According to the score plots from the PLS-DA model of tissue and urinary samples, we can observed that the different BDE-3 groups showed a consistent separation trend from the control group. While as for serum samples, it had a minor pullback in the last BDE-3 group (30 mg/kg/day), and some metabolites corresponded with this variation trend, such as Inosine, L-Phenylalanine and some lipids metabolites (Fig. S14). We thought the possible reason is that the mice had developed resistance to the BDE-3 induced toxicity and self-healing capacity. Furthermore, the metabolic changes in urine was secondary to the changes in serum and target organs injury of BDE-3. That may be why the urine samples did not show a similar pullback change in the highest dose group.
Nucleotides metabolism
Nucleotides play a key role as precursors of DNA and RNA, as activated intermediates in many biosynthetic processes, and as metabolic regulators. In the testis, nucleosides and nucleobases are important substrates of the salvage pathway for nucleotide biosynthesis33. We observed several purine and pyrimidine nucleobases metabolites in this study, such as Adenosine, Xanthine, Hypoxanthine and UDP-glucose, which significantly decreased in testicular tissue of BDE-treated mice compared with the control ones. Indicating that there will not be enough substrates for DNA and RNA biosynthesis.
In the mammalian spermatogenic pathway, Sertoli cells can provide nutrients and metabolic precursors to spermatogenic cells located within the blood-testis barrier (BTB)34. Because large amounts of nucleotides are required for spermatogenesis, less substrates will decelerate spermatogenesis. Accordingly, BDE-3 might affect the transportation of nucleobases and nucleosides across the BTB, or disturb the uptake of substrates by Sertoli cells. On the other hand, we observed several purine nucleobases metabolites, such as Adenosine and Inosine, significantly increased in serum and urine of BDE-treated mice compared with the control ones, which was potentially related to the reduction of purine and pyrimidine nucleobases metabolites level in the testis. Adenosine is an important endogenous nucleoside. Extracellular adenosine has the potential to influence the target cell metabolism in many aspects35. Adenosine can be phosphorylated to AMP, which can further generate Inosine by adenosine deaminase and IMP by AMP deaminase. The previous study had determined that Adenosine is also an important signaling molecule which is released under inflammatory conditions and there are four distinct subtypes are known, termed A1, A2A, A2B and A336. It usually shows its endocrine effects including insulin and glucagon secretion interference by A1 receptor in mice37. Besides, Hiroyasu Nakata presented the first purification of a peripheral A1 adenosine receptor of rat testis membranes38. In this study, we observed reduction of nucleobases and nucleosides level in testis and the accumulation in serum and urine accordingly. Less nucleobases and nucleosides in testis will affect spermatogenesis and the extra purine nucleobases metabolites in serum will disturb the internal environment homeostasis, which was consistent with previous studies, indicating that the BDE-3 possibly disturbed the above purine and pyrimidine metabolism, and induced reproductive toxicity.
Lipids metabolism
Lipids are abundant in testicles, which play an important role in membrane structure and function, energy storage and cell signaling39. Linoleic acid (LA) (18:2n-6) is the major Polyunsaturated fatty acid (PUFA) in vegetable oils and is a metabolic precursor to arachidonic acid (AA) (20:4n-6)40. A Previous report has indicated that LA family PUFA plays a criticle role in testicular function41. AA (20:4n-6) is a compound of important potent bioactivity, which mainly exists in phospholipid and LA mainly exists in phospholipid and triglyceride41. AA affects testis as a membrane component and providing energy storage, and a signal molecule in regulating steroidogenesis in Leydig cells as well42. In this study, decreased levels of LA and AA were found in BDE-3 exposed groups. Similarly, the increasing consumption of AA and LA level and corresponding ascending oxidative stress were found in testis after Bisphenol A exposure43. PUFA composition change observed in this metabolomics study was consistent with decreased spermatogenesis in this mice model. Therefore, testicular AA and LA alterations might be involved in BDE-3 testicular toxicity. Meanwhile, PUFA is very susceptible to peroxidation44. Thus, we speculated that the PUFA composition alterations indicated testicular oxidative stress.
We found significant increased GSH and L-carnitine levels in BDE-3 exposed group. The efficacy of l-carnitine and GSH as antioxidant substance has been confirmed both in humans and mice45. This kind of anti-oxidative composition might biologically adapt to oxidative stress46, thus the alterations was consistent with the hypothesis that testicular oxidative stress was increased in the BDE-3 exposed group. Oxidative stress could lead to both membrane lipid peroxidation and DNA fragmentation in testes47. It is reported that spermatogenesis48 and Leydig cell steroidogenisis49 are both vulnerable to oxidative stress43. There are clinical studies demonstrating that male infertility patients showed higher oxidative stress50. Additionally, oxidative stress and disturbed equilibrium of oxidant/antioxidant has been considered as a major mechanism of reproductive toxicity51. In present study, a possible explanation of decreased spermatogenesis of the mice model is oxidant/antioxidant imbalance, which has been well supported by population and animal studies52,53. Besides, in our study, a series of metabolites related to lipids metabolism such as lysophosphatidylcholine (LPC) and lysophosphatidyl ethanolamine (LysoPE) were detected, especially LPE significantly decreased in testicular tissue of the BDE-treated mice compared with the control group. LPE is an amphiphilic metabolite that is produced from membrane-phospholipids via the activation of phospholipase A2 (PLA2)54, and it also can synthesize PE under the action of LPCAT3 enzyme55. On the one hand, LysoPEs can stimulate invariant natural killer T cell activation through self-antigenicity, suggesting a possible role in innate immunity56, so we hypothesized that decreased LysoPEs in testicular tissue may cause its own immune disorder. On the other hand, it is essential that lysophospholipases (LysPLAs) and other phospholipases regulate phospholipid levels to maintain membrane homeostasis, flexibility and permeability57,58. This is critical to cell maintenance. In our study, the increase of various lipids metabolites potentially contributed to the BDE-induced toxicity.
Other metabolic pathway
Riboflavin (vitamin B-2) belongs to micronutrients that play important roles in carbohydrate energy metabolism59. There are studies that suggest that the supplement of Ribovlavin, can significantly reduce the embryolethal action of methophenazine60. On the contrary, other studies suggest a syndrome, which included various limb, brain, orofacial, gastrointestinal, and miscellaneous malformations, can be induced by an intense riboflavin deficiency61. Besides, cytoflavin is proven to reduce the manifestations of mitochondrial dysfunction and the cell damage degree because of the antioxidant and membrane-protective properties62. In this study, the reduction of riboflavin in the BDE-3 groups, played an important role in the BDE-3 induced reproductive toxicity.
Several amino acids and their metabolites significantly changed in both blood and urine of the BDE-treated mice. They are mainly involved in arginine and proline metabolism, valine, leucine and isoleucine degradation, cysteine and methionine metabolism, phenylalanine metabolism, tyrosine metabolism, tryptophan metabolism, glycine, serine and threonine metabolism, D-Glutamine and D-glutamate metabolism, cysteine and methionine metabolism, especially valine, leucine and isoleucine biosynthesis. Arginine, one of the most metabolically versatile amino acids, is a semiessential or conditionally essential amino acid in humans and serves as a precursor for the synthesis of urea, nitric oxide, polyamines, proline, glutamate, creatine, and agmatine. Arginine is metabolized through a complex and highly regulated set of pathways63. Besides, the present studies found that the arginine-rich dipeptides, in particular Proline-Arginine (PR), are potently neurotoxic. The Proline-Arginine dipeptide (PR), is potently neurotoxic when expressed in vivo and neurons with nuclear PR aggregates have a much higher risk to undergo degeneration64. In this study, L-Proline is increased in serum of BDE-treated mice. Beta-Hydroxyarginine and creatine were decreased in urine. The BDE-induced toxicity was possibly related to the disturbed arginine and proline metabolism.
In addition, we observed that L-Tyrosine, L-Leucine and L-Isoleucine, significantly decreased in the urine, while they increased in the blood, in the BDE-treated mice. These results suggested that the thiamine metabolism, valine, leucine and isoleucine degradation, and especially the polarity and clearance of the above metabolites are disturbed during the formation of HLP-induced toxicity, thus resulting in the abnormalities of body’s reproductive system metabolism.
Materials and Methods
Chemicals and reagents
Methanol (HPLC grade), acetonitrile (HPLC grade), formic acid (HPLC grade), and 2-chloro-L-phenylalanine (as an internal standard) were purchased from Sigma. (Saint Louis, USA). Ultrapure water was produced using the Milli-Q system (Millipore, Bedford, MA, USA). Hematoxylin-eosin staining kit was purchased from Shanghai Ailex Technology Co., Ltd.BDE-3 (CAS 101-55-3, 99% purity) was purchased from Sigma-Aldrich (St. Louis, MO).
Animals and treatment
The C57BL/6J gpt delta mice were kindly provided by Dr. Nohmi of the National Institutes of Health Sciences, Japan. Animals were acclimatized in Specific Pathogen Free rooms with the temperature at 20–26 °C, the humidity at 30–70% and a 12-h light/dark cycle for at least 1 week. Regular laboratory chow and filtered tap water were allowed adlibitum. All animal experiments were approved by the National Institutes of Health Guide for the Care and Use of Laboratory Animals with the approval (SYXK‐2013‐0050) of the Scientific Investigation Board of Shanghai Jiao Tong University School of Medicine, Shanghai, China. All animal experiments were approved (A-2016–004) by Department of Lab Animal Science and The Animal Care and Welfare Committee of Shanghai Jiao Tong University School of Medicine, Shanghai, China. All experiments were performed in accordance with Standard operation procedure and relevant guidelines and regulations of Shanghai Jiao Tong University School of Medicine, Shanghai, China.
In this study, we examined the effects of BDE-3 on the reproductive function in mice, with the lowest dose of 0.0015 mg/kg/day, which equal to the highest ingestion concentration of PBDEs in human (141 ng/kg/day) by the U.S. Environmental Protection Agency (EPA), 1.5 mg/kg/day, an equal dose to the lowest effective dose of its congener of BDE-471, middle dose of 10 mg/kg/day and high dose of 30 mg/kg/day, respectively.
Fifteen-weeks-old male mice were randomly allocated to 5 groups (n = 6 per group) and treated intragastrically with BDE-3 dissolved in corn oil 6 days/week for consecutive six weeks at different dosages of 0.0015, 1.5, 10 and 30 mg/kg. The vehicle control group received corn oil at the same manner. Mice were forced into metabolismcage for 24 hours once every two weeks to facilitate the collection of urine, the collections were immediately centrifuged at 4000 rpm for 5 min, and the supernatant was stored at −80 °C until being analyzed. Twenty-four hours after the last treatment, the mice were anesthetized by chloral hydrate. The blood samples were collected via eyeball and clotted at 4 °C for 40–60 minutes and then centrifuged at 4000 rpm for 5 minutes to obtain serum. The serum samples were stored at −80 °C until analysis. After blood collection, the epididymis and testis were dissected and weighed (wet weight). We collected the urine at different time points, but to match our serum samples (collected at 6th week), we did metabolomics profiling analysis only using the urine which were collected at 6th week. The remaining urine samples might be used for some deeper researches in the future.
Sperm count, vitality and morphology analysis
For sperm count analysis, one epididymis from each individual was put in a 1.5 mL tube with 37 °C pre-warmed M199 (containing 0.5% Fetal Bovine Serum) and cut into pieces for the preparation of sperm suspension. A drop of sperm suspension was put on a counting plate to measure the sperm count and sperm vitality. For sperm vitality examination, at least 200 sperm were checked and divided into 4 categories: I-fast straight forward movement, II-slow forward movement or rotation, III-in situ fibrillation or spinning, IV-no movement. Rate of sperm vitality % = (I + II + III)/(I + II + III + IV) × 100%. For morphological examination, 1–2 drops of sperm suspension were smeared on a slide. After drying, the slides were fixed in methanol for 5 minutes and stained with eosin for about 60 minutes before examined. At least 500 intact sperm per individual were examined under the microscope to calculate the ratio of abnormality. The abnormality was divided for 6 types: banana gate, enlarged-headed, amorphous, tail fold, double-headed and double-tailed. Rate of sperm abnormality (%) = total deformed sperm/total observed sperm × 100%.
Histopathology
One testis from each animal was fixed in modified Davidson solution and one epididymis of each animal was fixed in 10% neutral formalin fixative. After being well fixed, the tissues were sampled, dehydrated in automatic tissue hydroextractor (Pathcentre, Thremo) and embedded in paraffin embedding machine (Shandon Histocentre 2, Thremo) and then cut into 3 µm paraffin section in microtome (Shandon Finesse E+, Thremo). The sections were stained with hematoxylin and eosin. All tissues were analysised by manual check and scanned by slice scanning system (Scan ScopeXT, Aperio).
UHPLC-Q-TOF/MS metabolomics analysis
The frozen urine and serum samples were thawed at room temperature. 300 μL methanol solution that contained 5 μg/mL 2-Chloro-L-phenylalanine as the internal standard was added into 100 μL urine or serum samples. The mixed samples were homogenized by vortex for 5 min and then were placed at room temperature for 10 min and centrifuged at 13,000 rpm, 4 °C for 15 min. 150 μL of supernatant were added into the sample vials.
The frozen testicular tissue samples were thawed at room temperature. Then they were weighed, and homogenized in 900 μl 80% methanol solution that contained 5 μg/mL 2-chloro-l-phenylalanine as an internal standard. Then the mixture were centrifuged at 13,000 rpm, 4 ◦C for 15 min. 150 μl of supernatant were added into the sample vials.
A quality control sample (QC) was prepared by mixing the same volume from all supernatant samples (10 μL from each sample). The QC sample was run six times prior to the start of the analytical run to “condition” the system and analyzed after every 9 samples to check the system stability65.
UHPLC-Q-TOF/MS analysis was performed using an Agilent 1290 Infinity LC system coupled to an Agilent 6538 Accurate-Mass Quadrupole Time-of-Flight (Q-TOF) mass spectrometer (Agilent, USA). Chromatographic separations were performed at 25 °C using a Waters XSelect® HSS T3 analytical column (2.1 mm × 100 mm, 2.5 μm, Waters, Milford, MA). The flow rate was 0.4 mL/min and the injection volume was 3 μL. The mobile phase consisted of 0.1% formic acid (A) and ACN modified with 0.1% formic acid (B). The gradient elution conditions for urinary samples were 5% B at 0–1 min, 5–20% B at 1–6 min, 20–50% B at 6–9 min, 50–95% B at 9–13 min, 95% B at 13–15 min and followed by re-equilibrated step of 5 min. As for serum samples, the following gradient program was used: 5%B at 0–2 min, 5–95%B at 2–17 min, 95%B at 17–19 min. The gradient elution conditions for tissue samples were 5% B at 0–2 min, 5–95% B at 2–13 min, 95% B at 13–15 min. The capillary voltage was 4 kV in positive mode and 3.5 kV in negative mode, the drying gas flow was 11 L/min, and the gas temperature was 350 °C. The nebulizer pressure was set at 45 psig. The fragmentor voltage was set at 120 V and skimmer voltage was set at 60 V. The reference ions were 121.0508 and 922.0098 in positive mode, 119.03632 and 966.000725 in negative mode. Data were collected in a centroid mode and the mass range was set at m/z 50–1100 using an extended dynamic range. MS/MS analysis was carried out to study the structure of the potential biomarkers and the collision energy was range from 10 to 40 eV.
Data processing and statistical analysis
In the analysis of body weight, testis and epididymis weight, sperm count, vitality and morphology, the Dunnett-t test was used to determine significant differences between BDE-3 exposure groups and the vehicle control group after the one-way ANOVA was performed. A value of P < 0.05 was considered significant. SPSS version 17.0 (IBM, USA) was used for statistical analysis.
The raw UHPLC–MS data were transformed into a common data file format (.mzdata) using MassHunter Qualitative software. The interferences of the isotopes were excluded and the absolute peak height was set at 300 counts. In the R software platform, the XCMS program was applied for peak extraction, peak alignment, and automatic integration66. The 80% rule was used to filter the ions and the remaining ions were then normalized to internal standard peak area. The Retention time (Rt)-m/z pairs, observations, and relative ion intensities were imported into SIMCA-P 11.0 software package (Umetrics, Umea, Sweden) for the multivariate statistical analysis. Prior to multivariate analysis, the resultant data matrices were mean-centered and scaled to Pareto variance. Unsupervised principal component analysis (PCA) and partial least squares discriminate analysis (PLS-DA) were applied to observe the stability of the assay in the sequence, the separating trends, and further identify the metabolite candidates contributing to the clustering of samples. We applied variable importance plot (VIP) representing the confirmation of the importance or power of the selected candidates to select metabolite candidates with the threshold value of 1. In addition, the model of PLS-DA was evaluated according to the cross-validation of R2, Q2 value and permutation test. An independent sample t-test was performed for the statistical analysis using SPSS version 17.0 (IBM, USA) and p < 0.05 was considered statistically significant.
Pathway enrichment analysis
We applied the Pathway Analysis module on Metaboanalyst website (http://www.metaboanalyst.ca/). We firstly import the differential metabolites of testis tissue, serum, and urine to this website, matching the HMDB, PUBCHEM and KEGG database (The mass error tolerance was “<15ppm”), and the metabolites were categorized based on the link from KEGG. Select mouse as species to do pathway enrichment analysis. The results were shown on Fig. S15. Then manually construct the pathway network based on the KEGG pathways we have got, and some important pathways included were shown on Fig. S16.
Conclusion
In this study, we successfully observed the reproductive toxicity induced by BDE-3 in mice via clinical observation, Sperm count and vitality, Sperm morphology and Histopathology examination. A new scientific metabolomic method based on UPLC-Q-TOF/MS was applied to profile the differential metabolites in testis, urine and serum in the control and BDE-3 treated mice at different dosages. In total, 76 differential metabolites in testicular tissue, 38 differential metabolites in urine and 31 differential metabolites in serum were identified. Pathway analysis revealed that several pathways were potentially related to BDE-3 induced toxicity including Arginine and proline metabolism, Valine, leucine and isoleucine degradation, Phenylalanine metabolism, Cysteine and methionine metabolism, Tyrosine metabolism, Purine metabolism, Pentose and glucuronate interconversions, Tryptophan metabolism, Biotin metabolism, Oxidative phosphorylation, Riboflavin metabolism, and Glycerophospholipid metabolism. These findings demonstrated the BDE-3 induced reproductive toxicity in mice. Our study may be helpful to arouse our awareness of the toxicity of PBDEs and to give a possible explanation for the mechanism, which indicates that UHPLC-Q-TOFMS-based metabolomics approach allowed a better understanding of PBDEs-induced toxicity dynamically.
Data Availability
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
Electronic supplementary material
Acknowledgements
This study was supported in part by the grants from the National Natural Science Foundation of China (21477078), TCM Supported Project (15401900800) from Science and Technology Commission of Shanghai Municipality. We thank Dr. Takehiko Nohmi (NIHS, Japan) for providing the C57BL/6J gpt delta mice.
Author Contributions
X.D., Y.L. initiated and supervised the study. Z.H.W., J.X. and S.Y.G. planned the experiments. Y.L. and X.D. provided the experimental background guidance. S.Y.G., Z.H.W. performed the data process, Z.H.W., J.X. wrote the manuscript. J.X., Y.Y.C. and L.P.W. performed the animal experiment and collected the samples. N.L. and S.Y.G. conducted the UPLC-Q-TOF/MS analysis and pretreatment of the samples. J.X., Y.Y.C. and L.P.W. performed the toxicology experiments. All authors reviewed the draft manuscript and approved the final manuscript.
Competing Interests
The authors declare no competing interests.
Footnotes
Ziheng Wei, Jing Xi and Songyan Gao contributed equally to this work.
Electronic supplementary material
Supplementary information accompanies this paper at 10.1038/s41598-018-23484-2.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Yang Luan, Email: yluan@sjtu.edu.cn.
Xin Dong, Email: dongxinsmmu@126.com.
References
- 1.Stockholm Convention. Stockholm Convention on Persistent Organic Pollutants (POPs) (2009).
- 2.Raff JD, Hites RA. Deposition versus photochemical removal of PBDEs from Lake Superior air. Environmental science & technology. 2007;41:6725–6731. doi: 10.1021/es070789e. [DOI] [PubMed] [Google Scholar]
- 3.Li X, et al. Photodegradation of 2,2′,4,4′-tetrabromodiphenyl ether in nonionic surfactant solutions. Chemosphere. 2008;73:1594–1601. doi: 10.1016/j.chemosphere.2008.08.031. [DOI] [PubMed] [Google Scholar]
- 4.Zhuang Y, Ahn S, Luthy RG. Debromination of polybrominated diphenyl ethers by nanoscale zerovalent iron: pathways, kinetics, and reactivity. Environmental science & technology. 2010;44:8236–8242. doi: 10.1021/es101601s. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Zhuang Y, Jin L, Luthy RG. Kinetics and pathways for the debromination of polybrominated diphenyl ethers by bimetallic and nanoscale zerovalent iron: effects of particle properties and catalyst. Chemosphere. 2012;89:426–432. doi: 10.1016/j.chemosphere.2012.05.078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Huang K, et al. Photodegradation of 4,4′-dibrominated diphenyl ether in Triton X-100 micellar solution. Chemosphere. 2017;180:423–429. doi: 10.1016/j.chemosphere.2017.04.056. [DOI] [PubMed] [Google Scholar]
- 7.Cheng, J. Bioaccumulation and Biotransformation of Polybrominated Diphenyl Ethers (BDE 15, BDE 47) by Crucian Carp. Ph.D. Dissertation, Nanjing University (2011).
- 8.Gill S, Hou Y, Li N, Pulido O, Bowers W. Developmental neurotoxicity of polybrominated diphenyl ethers mixture de71 in Sprague-Dawley rats. Journal of toxicology and environmental health. Part A. 2016;79:482–493. doi: 10.1080/15287394.2016.1182001. [DOI] [PubMed] [Google Scholar]
- 9.Yogui GT, Sericano JL. Polybrominated diphenyl ether flame retardants in the U.S. marine environment: a review. Environment international. 2009;35:655–666. doi: 10.1016/j.envint.2008.11.001. [DOI] [PubMed] [Google Scholar]
- 10.Abdelouahab N, Ainmelk Y, Takser L. Polybrominated diphenyl ethers and sperm quality. Reproductive toxicology. 2011;31:546–550. doi: 10.1016/j.reprotox.2011.02.005. [DOI] [PubMed] [Google Scholar]
- 11.Akutsu K, et al. Polybrominated diphenyl ethers in human serum and sperm quality. Bulletin of environmental contamination and toxicology. 2008;80:345–350. doi: 10.1007/s00128-008-9370-4. [DOI] [PubMed] [Google Scholar]
- 12.Wang Y, et al. Adverse effects of 2,2′,4,4′-tetrabromodiphenyl ether on semen quality and spermatogenesis in male mice. Bulletin of environmental contamination and toxicology. 2013;90:51–54. doi: 10.1007/s00128-012-0867-5. [DOI] [PubMed] [Google Scholar]
- 13.Zhang Z, et al. Cytochrome P450 3A1 mediates 2,2′,4,4′-tetrabromodiphenyl ether-induced reduction of spermatogenesis in adult rats. PloS one. 2013;8:e66301. doi: 10.1371/journal.pone.0066301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.O’Driscoll K, et al. The environmental fate of polybrominated diphenyl ethers (PBDEs) in western Taiwan and coastal waters: evaluation with a fugacity-based model. Environmental science and pollution research international. 2016;23:13222–13234. doi: 10.1007/s11356-016-6428-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Helleday T, Tuominen KL, Bergman A, Jenssen D. Brominated flame retardants induce intragenic recombination in mammalian cells. Mutation research. 1999;439:137–147. doi: 10.1016/S1383-5718(98)00186-7. [DOI] [PubMed] [Google Scholar]
- 16.You X, Xi J, Cao Y, Zhang J, Luan Y. 4-Bromodiphenyl Ether Induces Germ Cell Apoptosis by Induction of ROS and DNA Damage in Caenorhabditis elegans. Toxicological sciences: an official journal of the Society of Toxicology. 2017;157:510–518. doi: 10.1093/toxsci/kfx064. [DOI] [PubMed] [Google Scholar]
- 17.Maranghi F, et al. Dietary exposure of juvenile female mice to polyhalogenated seafood contaminants (HBCD, BDE-47, PCB-153, TCDD): comparative assessment of effects in potential target tissues. Food and chemical toxicology: an international journal published for the British Industrial Biological Research Association. 2013;56:443–449. doi: 10.1016/j.fct.2013.02.056. [DOI] [PubMed] [Google Scholar]
- 18.Surowiec I, et al. Multi-platform metabolomics assays for human lung lavage fluids in an air pollution exposure study. Analytical and bioanalytical chemistry. 2016;408:4751–4764. doi: 10.1007/s00216-016-9566-0. [DOI] [PubMed] [Google Scholar]
- 19.O’Kane AA, Chevallier OP, Graham SF, Elliott CT, Mooney MH. Metabolomic profiling of in vivo plasma responses to dioxin-associated dietary contaminant exposure in rats: implications for identification of sources of animal and human exposure. Environmental science & technology. 2013;47:5409–5418. doi: 10.1021/es305345u. [DOI] [PubMed] [Google Scholar]
- 20.Lu C, et al. NMR-based metabonomic analysis of the hepatotoxicity induced by combined exposure to PCBs and TCDD in rats. Toxicology and applied pharmacology. 2010;248:178–184. doi: 10.1016/j.taap.2010.07.020. [DOI] [PubMed] [Google Scholar]
- 21.Shi X, et al. Metabolomic analysis of the effects of polychlorinated biphenyls in nonalcoholic fatty liver disease. Journal of proteome research. 2012;11:3805–3815. doi: 10.1021/pr300297z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bonvallot N, et al. Metabolomics tools for describing complex pesticide exposure in pregnant women in Brittany (France) PloS one. 2013;8:e64433. doi: 10.1371/journal.pone.0064433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kanehisa M, Sato Y, Kawashima M, Furumichi M, Tanabe M. KEGG as a reference resource for gene and protein annotation. Nucleic acids research. 2016;44:D457–462. doi: 10.1093/nar/gkv1070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kanehisa M, Goto S. KEGG: kyoto encyclopedia of genes and genomes. Nucleic acids research. 2000;28:27–30. doi: 10.1093/nar/28.1.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kanehisa M, Furumichi M, Tanabe M, Sato Y, Morishima K. KEGG: new perspectives on genomes, pathways, diseases and drugs. Nucleic acids research. 2017;45:D353–d361. doi: 10.1093/nar/gkw1092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Chen M, et al. Metabolomic analysis reveals metabolic changes caused by bisphenol A in rats. Toxicological sciences: an official journal of the Society of Toxicology. 2014;138:256–267. doi: 10.1093/toxsci/kfu016. [DOI] [PubMed] [Google Scholar]
- 27.Nagato EG, Simpson AJ, Simpson MJ. Metabolomics reveals energetic impairments in Daphnia magna exposed to diazinon, malathion and bisphenol-A. Aquatic toxicology (Amsterdam, Netherlands) 2016;170:175–186. doi: 10.1016/j.aquatox.2015.11.023. [DOI] [PubMed] [Google Scholar]
- 28.Zhang J, et al. Urinary metabolomics revealed arsenic internal dose-related metabolic alterations: a proof-of-concept study in a Chinese male cohort. Environmental science & technology. 2014;48:12265–12274. doi: 10.1021/es503659w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lu K, et al. Arsenic exposure perturbs the gut microbiome and its metabolic profile in mice: an integrated metagenomics and metabolomics analysis. Environmental health perspectives. 2014;122:284–291. doi: 10.1289/ehp.1307429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ji C, Wu H, Wei L, Zhao J, Yu J. Proteomic and metabolomic analysis reveal gender-specific responses of mussel Mytilus galloprovincialis to 2,2′,4,4′-tetrabromodiphenyl ether (BDE 47) Aquatic toxicology (Amsterdam, Netherlands) 2013;140-141:449–457. doi: 10.1016/j.aquatox.2013.07.009. [DOI] [PubMed] [Google Scholar]
- 31.Jung YS, Lee J, Seo J, Hwang GS. Metabolite profiling study on the toxicological effects of polybrominated diphenyl ether in a rat model. Environmental toxicology. 2017;32:1262–1272. doi: 10.1002/tox.22322. [DOI] [PubMed] [Google Scholar]
- 32.Eguchi A, Miyaso H, Mori C. The effects of early postnatal exposure to a low dose of decabromodiphenyl ether (BDE-209) on serum metabolites in male mice. The Journal of toxicological sciences. 2016;41:667–675. doi: 10.2131/jts.41.667. [DOI] [PubMed] [Google Scholar]
- 33.Kato R, Maeda T, Akaike T, Tamai I. Characterization of novel Na+-dependent nucleobase transport systems at the blood-testis barrier. American journal of physiology. Endocrinology and metabolism. 2006;290:E968–975. doi: 10.1152/ajpendo.00160.2005. [DOI] [PubMed] [Google Scholar]
- 34.Hogue DL, Ling V. A human nucleobase transporter-like cDNA (SLC23A1): member of a transporter family conserved from bacteria to mammals. Genomics. 1999;59:18–23. doi: 10.1006/geno.1999.5847. [DOI] [PubMed] [Google Scholar]
- 35.Blackburn MR, et al. Adenosine levels in the postimplantation mouse uterus: quantitation by HPLC-fluorometric detection and spatiotemporal regulation by 5′-nucleotidase and adenosine deaminase. Developmental dynamics: an official publication of the American Association of Anatomists. 1992;194:155–168. doi: 10.1002/aja.1001940208. [DOI] [PubMed] [Google Scholar]
- 36.Akkari R, Burbiel JC, Hockemeyer J, Muller CE. Recent progress in the development of adenosine receptor ligands as antiinflammatory drugs. Current topics in medicinal chemistry. 2006;6:1375–1399. doi: 10.2174/15680266106061375. [DOI] [PubMed] [Google Scholar]
- 37.Johansson SM, et al. A1 receptor deficiency causes increased insulin and glucagon secretion in mice. Biochemical pharmacology. 2007;74:1628–1635. doi: 10.1016/j.bcp.2007.08.006. [DOI] [PubMed] [Google Scholar]
- 38.Nakata H. A1 adenosine receptor of rat testis membranes. Purification and partial characterization. The Journal of biological chemistry. 1990;265:671–677. [PubMed] [Google Scholar]
- 39.Oresic M, Hanninen VA, Vidal-Puig A. Lipidomics: a new window to biomedical frontiers. Trends in biotechnology. 2008;26:647–652. doi: 10.1016/j.tibtech.2008.09.001. [DOI] [PubMed] [Google Scholar]
- 40.Lawton LJ, Donaldson WE. Lead-induced tissue fatty acid alterations and lipid peroxidation. Biological trace element research. 1991;28:83–97. doi: 10.1007/BF02863075. [DOI] [PubMed] [Google Scholar]
- 41.Davis JT, Bridges RB, Coniglio JG. Changes in lipid composition of the maturing rat testis. The Biochemical journal. 1966;98:342–346. doi: 10.1042/bj0980342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wang X, Walsh LP, Reinhart AJ, Stocco DM. The role of arachidonic acid in steroidogenesis and steroidogenic acute regulatory (StAR) gene and protein expression. The Journal of biological chemistry. 2000;275:20204–20209. doi: 10.1074/jbc.M003113200. [DOI] [PubMed] [Google Scholar]
- 43.Chen M, et al. Bisphenol A alters n-6 fatty acid composition and decreases antioxidant enzyme levels in rat testes: a LC-QTOF-based metabolomics study. PloS one. 2012;7:e44754. doi: 10.1371/journal.pone.0044754. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Jordan RA, Schenkman JB. Relationship between malondialdehyde production and arachidonate consumption during NADPH-supported microsomal lipid peroxidation. Biochemical pharmacology. 1982;31:1393–1400. doi: 10.1016/0006-2952(82)90034-X. [DOI] [PubMed] [Google Scholar]
- 45.Cabral, R. E. L., Mendes, T. B., Vendramini, V. & Miraglia, S. M. Carnitine partially improves oxidative stress, acrosome integrity, and reproductive competence in doxorubicin-treated rats. Andrology, 10.1111/andr.12426 (2017). [DOI] [PubMed]
- 46.Burczynski JM, Southard SJ, Hayes JR, Longhurst PA, Colby HD. Changes in mitochondrial and microsomal lipid peroxidation and fatty acid profiles in adrenal glands, testes, and livers from alpha-tocopherol-deficient rats. Free radical biology & medicine. 2001;30:1029–1035. doi: 10.1016/S0891-5849(01)00497-X. [DOI] [PubMed] [Google Scholar]
- 47.Aitken RJ, Roman SD. Antioxidant systems and oxidative stress in the testes. Advances in experimental medicine and biology. 2008;636:154–171. doi: 10.1007/978-0-387-09597-4_9. [DOI] [PubMed] [Google Scholar]
- 48.Naughton CK, Nangia AK, Agarwal A. Pathophysiology of varicoceles in male infertility. Human reproduction update. 2001;7:473–481. doi: 10.1093/humupd/7.5.473. [DOI] [PubMed] [Google Scholar]
- 49.Hales DB, et al. Mitochondrial function in Leydig cell steroidogenesis. Annals of the New York Academy of Sciences. 2005;1061:120–134. doi: 10.1196/annals.1336.014. [DOI] [PubMed] [Google Scholar]
- 50.Sikka SC. Relative impact of oxidative stress on male reproductive function. Current medicinal chemistry. 2001;8:851–862. doi: 10.2174/0929867013373039. [DOI] [PubMed] [Google Scholar]
- 51.Mathur PP, D’Cruz SC. The effect of environmental contaminants on testicular function. Asian journal of andrology. 2011;13:585–591. doi: 10.1038/aja.2011.40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Appasamy M, et al. Relationship between male reproductive hormones, sperm DNA damage and markers of oxidative stress in infertility. Reproductive biomedicine online. 2007;14:159–165. doi: 10.1016/S1472-6483(10)60783-3. [DOI] [PubMed] [Google Scholar]
- 53.Aly HA, Domenech O, Abdel-Naim AB. Aroclor 1254 impairs spermatogenesis and induces oxidative stress in rat testicular mitochondria. Food and chemical toxicology: an international journal published for the British Industrial Biological Research Association. 2009;47:1733–1738. doi: 10.1016/j.fct.2009.03.019. [DOI] [PubMed] [Google Scholar]
- 54.Tepperman BL, Soper BD. The role of phospholipase A2 in calcium-ionophore-mediated injury to rat gastric mucosal cells. Digestive diseases and sciences. 1999;44:494–502. doi: 10.1023/A:1026688819939. [DOI] [PubMed] [Google Scholar]
- 55.Li Z, et al. Deficiency in lysophosphatidylcholine acyltransferase 3 reduces plasma levels of lipids by reducing lipid absorption in mice. Gastroenterology. 2015;149:1519–1529. doi: 10.1053/j.gastro.2015.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Facciotti F, et al. Peroxisome-derived lipids are self antigens that stimulate invariant natural killer T cells in the thymus. Nature immunology. 2012;13:474–480. doi: 10.1038/ni.2245. [DOI] [PubMed] [Google Scholar]
- 57.Selle H, Chapman BE, Kuchel PW. Glycerophosphocholine release in human erythrocytes. 1 H spin-echo and 31P-NMR evidence for lysophospholipase. European Journal of Biochemistry. 1993;212:411. doi: 10.1111/j.1432-1033.1993.tb17676.x. [DOI] [PubMed] [Google Scholar]
- 58.Wang A, Dennis EA. Mammalian lysophospholipases. Biochimica Et Biophysica Acta. 1999;1439:1. doi: 10.1016/S1388-1981(99)00063-3. [DOI] [PubMed] [Google Scholar]
- 59.Diet, nutrition and the prevention of chronic diseases. World Health Organization technical report series916, i–viii, 1–149, backcover (2003). [PubMed]
- 60.Horvath C, Druga A. Action of the phenothiazine derivative methophenazine on prenatal development in rats. Teratology. 1975;11:325–329. doi: 10.1002/tera.1420110312. [DOI] [PubMed] [Google Scholar]
- 61.Kalter H. Analysis of the syndrome of congenital malformations induced in genetically defined mice by acute riboflavin deficiency. Teratogenesis, carcinogenesis, and mutagenesis. 1990;10:385–397. doi: 10.1002/tcm.1770100504. [DOI] [PubMed] [Google Scholar]
- 62.Forgione MA, et al. Heterozygous cellular glutathione peroxidase deficiency in the mouse: abnormalities in vascular and cardiac function and structure. Circulation. 2002;106:1154–1158. doi: 10.1161/01.CIR.0000026820.87824.6A. [DOI] [PubMed] [Google Scholar]
- 63.Morris SM., Jr. Arginine: beyond protein. The American journal of clinical nutrition. 2006;83:508s–512s. doi: 10.1093/ajcn/83.2.508S. [DOI] [PubMed] [Google Scholar]
- 64.Wen X, et al. Antisense proline-arginine RAN dipeptides linked to C9ORF72-ALS/FTD form toxic nuclear aggregates that initiate in vitro and in vivo neuronal death. Neuron. 2014;84:1213–1225. doi: 10.1016/j.neuron.2014.12.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Gao S, et al. Metabolomics analysis for hydroxy-L-proline-induced calcium oxalate nephrolithiasis in rats based on ultra-high performance liquid chromatography quadrupole time-of-flight mass spectrometry. Scientific reports. 2016;6:30142. doi: 10.1038/srep30142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Smith CA, Want EJ, O’Maille G, Abagyan R, Siuzdak G. XCMS: processing mass spectrometry data for metabolite profiling using nonlinear peak alignment, matching, and identification. Analytical chemistry. 2006;78:779–787. doi: 10.1021/ac051437y. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.