Abstract
An organic compound known as 5-hydroxymethylfurfural (HMF) is formed from reducing sugars in honey and various processed foods in acidic environments when they are heated through the Maillard reaction. In addition to processing, storage conditions affect the formation HMF, and HMF has become a suitable indicator of honey quality. HMF is easily absorbed from food through the gastrointestinal tract and, upon being metabolized into different derivatives, is excreted via urine. In addition to exerting detrimental effects (mutagenic, genotoxic, organotoxic and enzyme inhibitory), HMF, which is converted to a non-excretable, genotoxic compound called 5-sulfoxymethylfurfural, is beneficial to human health by providing antioxidative, anti-allergic, anti-inflammatory, anti-hypoxic, anti-sickling, and anti-hyperuricemic effects. Therefore, HMF is a neo-forming contaminant that draws great attention from scientists. This review compiles updated information regarding HMF formation, detection procedures, mitigation strategies and effects of HMF on honey bees and human health.
Keywords: 5-Hydroxymethylfurfural, Mutagen, Carcinogen, Antioxidant, Anti-allergen
Introduction
Honey is a sweet natural product produced by honeybees (Apis mellifera), which gather nectar from flowers before converting it to nutritious food. Honey is mainly composed of water (15–20%) and two sugars (dextrose and levulose), with the presence of small amounts of at least 22 other more complex sugars (80–85%, w/w) [41, 179]. Honey has also been reported to contain an intricate mixture of nitrogenous compounds, lactone, proteins, antibiotic-rich inhibine, enzymes, phenol antioxidants, aroma compounds, amino and organic acids, gluconic acid, phenolic acids, flavonoids, minerals, vitamins, 5-hydroxymethylfurfural (HMF) and other phytochemicals [20, 56, 181, 183]. Honey composition varies depending on its floral, geographical and entomological sources [13, 56]. In addition, external features such as seasonal and environmental factors, honey processing, and storage time and conditions have crucial effects on honey’s composition [58, 76, 104].
Honey is considered both nutritional and medicinal, although the presence of certain constituents, for example, heavy metals (even in trace amounts), some alkaloids, and HMF and its derivatives may contribute to honey’s toxicity [77, 152]. HMF is a cyclic aldehyde produced by sugar degradation through the Maillard reaction (a non-enzymatic browning reaction) during food processing or long storage of honey [102]. The presence of simple sugars (glucose and fructose) and many acids, as well as minerals, in honey can further enhance the production of this substance [92]. HMF concentration is widely recognized as a parameter affecting honey freshness because it is typically absent (or is present in only very small amounts in fresh honeys), while its concentration tends to rise during processing and/or because of aging. Previous studies have reported that honey stored at low temperatures and/or under fresh conditions has low or minimal HMF concentrations, while aged and/or honey stored at comparatively higher or medium temperature has high HMF concentrations. In addition to storage conditions, the use of metallic containers and honey floral sources are critical factors affecting HMF levels (Table 1). Hence, higher HMF concentration is indicative of poor storage conditions and/or excess heating of honey [48, 81]. Therefore, the Codex Alimentarius Standard commission has set the maximum limit for HMF in honey at 40 mg/kg (with a higher limit of 80 mg/kg for honeys originating from tropical regions) to ensure that the product has not undergone extensive heating during processing and is safe for consumption [6].
Table 1.
Variation in HMF concentration in honey samples based on their storage time and geographical sources
| Country | Storage time | Storage temperature (°C) | HMF concentration (mg/kg) | References |
|---|---|---|---|---|
| Asia | ||||
| Bangladesh | > 1.5 years | 20–25 | 3.18–703.10 | [76] |
| India | Fresh | – | 0.15–1.70 | [160] |
| Malaysia (year) | < 1 | 4–5 | 0.26–68.99 | [108] |
| > 1 | 25–30 | 206.06–383.39 | [81] | |
| > 2 | 25–30 | 986.57–1131.76 | ||
| Nepal | – | 25–29 | 30.36–56.10 | [131] |
| Pakistan | – | 4–5 | ND–6.00 | [3] |
| – | – | 23.18–27.37 | [74] | |
| Iran | – | – | 0.04–17.20 | [104] |
| Turkey | Fresh | – | 0.00–11.50 | [188] |
| < 6 months | – | 19.20–28.6 | [91] | |
| 1 year | 20 ± 5 | 8.60–39.00 | [188] | |
| Europe | ||||
| Italy | Fresh | – | 1.23–5.95 | [47] |
| Croatia | – | – | 0.00–23.69 | [173] |
| Czech Republic | 4 years | 20 ± 2 | 10.30–44.20 | [80] |
| Poland | – | – | 0.70–3.50 | [89] |
| Portugal | – | Room temperature | 18.00–94.00 | [66] |
| Spain | < 3 years | – | 0.00–21.39 | [116] |
| Northwest Spain | < 2 years | − 30 | 0.00–1.60 | [120] |
| Switzerland | < 7 years | 4 | 0.00–112.00 | [148] |
| Africa | ||||
| Algeria | < 1 year | 4–6 | 1.73–480.00 | [36, 82] |
| Burkina Faso | < 1 year | 0–4 | 3.00–27.50 | [103] |
| Ethiopia | < 6 months | – | 0.68–6.56 | [25, 85] |
| Kenya | < 1 year | 25 ± 2 | 3.70–389.36 | [112] |
| Morocco | – | 4 | 3.20–52.60 | [169] |
| Nigeria | < 6 months | – | 0.66–1.43 | [16] |
| Tanzania | – | – | 5.00–26.40 | [115] |
| South America | ||||
| Argentina | – | 23 ± 2 | 0.05–2.94 | [21] |
| Within 2 months | – | 1.48–34.08 | [34] | |
| Argentinean Patagonia | > 3 years | – | 0.00–14.70 | [8] |
| Brazil | – | – | 1.50–115.20 | [5, 162] |
| North America | ||||
| Cuba | < 1 year | 4 | 3.30–15.90 | [9] |
| Mexico | – | Room temperature | 9.01–21.96 | [106] |
| Australia | ||||
| Australia | 2 years | − 18 | 1.30–12.40 | [71] |
ND not detected, HMF 5-hydroxymethylfurfural
HMF is not only present in honey; it is nearly ubiquitous in our daily heat-processed, sugar-containing foodstuffs, from our breakfast cereals, breads, dairy products, and fruit juices to liquors at different concentrations [17, 37, 114, 137, 155, 170]. Therefore, HMF is considered one of the main quality indexes of different commercial whey proteins, molasses and many other products [40]. In most previous studies, HMF has been reported to have negative effects on human health, such as cytotoxicity toward mucous membranes, the skin and the upper respiratory tract; mutagenicity; chromosomal aberrations; and carcinogenicity toward humans and animals [59, 94, 107]. However, in more recent extensive studies, HMF has been proved to have a wide range of positive effects, such as antioxidative [193], anti-allergic [187], anti-inflammatory [86], anti-hypoxic [96], anti-sickling [1], anti-hyperuricemic effects [97].
It has been reported that humans may ingest between 30 and 150 mg HMF daily via various food products; however, safe levels of HMF consumption are not well clarified. The reason is that HMF’s metabolism, biotransformation and excretion and thus clearance rate from the body depend on the organ function of an individual [176], which have not been considered. The aim of this review is to describe the effects of HMF present in honey, which, if broadly analyzed, can be used to promote more widespread application of honey with special medicinal implications. Upon highlighting the HMF content in honey, a general discussion of the HMF content in other foods and HMF’s detection, optimized formation, mitigation and eradication from food is provided. For this purpose, all related articles relevant to the topic of “honey and other foods, HMF: its toxicity and therapeutic effects” were included, regardless of the time of publication. To our knowledge, this is the first study to extensively report on HMF in honey from all over the world and to provide a general overview to explore its effects on both bee and human health.
Formation of HMF in honey
HMF is a six-carbon heterocyclic organic compound containing both aldehyde and alcohol (hydroxymethyl) functional groups. The ring of the structure is centered on furan moieties, whereas the two functional groups, i.e., formyl and hydroxy-methyl groups, are linked at the second and fifth positions, respectively (Fig. 1). HMF is a solid, yellow substance that has a low melting point but is highly soluble in water [142].
Fig. 1.
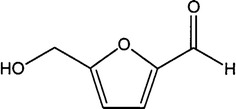
Chemical structure of HMF
HMF is considered the most important intermediate product formed during two reactions (i) acid-catalyzed degradation of hexose and (ii) decomposition of 3-deoxyosone in the Maillard reaction (Fig. 2) [46]. HMF formation is correlated with chemical characteristics such as pH, free acid content, total acidity, lactone content and mineral content, which in turn are related to the floral source of collected honey samples. The presence of simple sugars such as glucose and fructose and of many acids has been reported to be favorable for honey production [47].
Fig. 2.
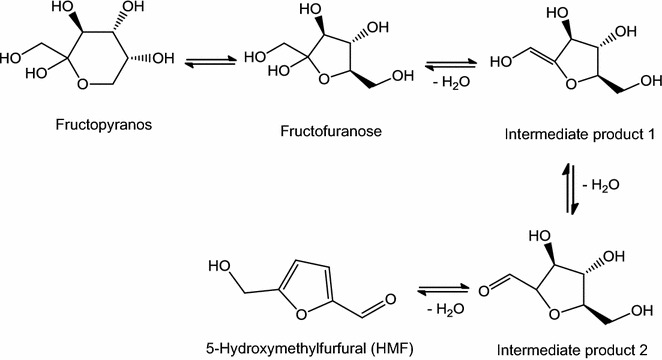
Formation of HMF in honey
Similarly to honey, which is rich in glucose and fructose, most sugar-containing foods also contain HMF [71]. Nevertheless, although HMF occurs at very low concentrations and can even be absent in both fresh honey and food products, heat treatment and/or prolonged storage conditions can enhance further HMF production. Moniruzzaman et al. [108] reported the mean HMF concentration in Malaysian honeys stored for 2 months at 4–5 °C to be 35.98 mg/kg. In contrast, Khalil et al. [81] found that HMF concentrations in Malaysian honey stored at 25–30 °C for more than a year could reach very high levels (118.47–1139.95 mg/kg). [76] also observed high HMF levels (3.18–703.10 mg/kg) in honey samples from Bangladesh stored for more than a year at room temperature (20–25 °C). Therefore, HMF level is not only indicative of honey freshness but also of storage duration and conditions (Table 1).
Factors affecting HMF formation in honey
In addition to being directly produced when heating sugar from the degradation of hexoses under acidic conditions at high temperatures and/or during the Maillard reaction [15], HMF is produced from the oligo- and polysaccharides that can yield hexoses upon hydrolysis. However, HMF appears to be more selectively produced from keto-hexoses such as fructose [141]. Interestingly, there are two reasons why higher yields of HMF are obtained from fructose (ketose) than from glucose (aldose). First, the reactivity of glucose is lower than that of fructose, with a lower enolization rate [92]. Enolization is believed to be the rate-determining step of HMF production. Second, fructose forms an equilibrium mixture of difructoses and dianhydrides and thus internally blocks most reactive groups, leading to the formation of certain by-products. In contrast, glucose forms true oligosaccharides that still contain reactive reducing groups, posing a higher risk for cross-polymerization with the reactive intermediates, including HMF [92].
Honey has been reported to contain many different types of sugars. In fact, a recent review by Solayman et al. [163] indicated that honey contains approximately 39.44% fructose, 28.15% glucose and 3.19% sucrose. The cyclic aldehyde HMF is also produced in honey by the degradation of related sugars [81]. Heating of honey during its processing reduces its viscosity, which can prevent crystallization or fermentation [161]. In addition to heating, several other factors influence the formation of HMF in honey, such as honey’s physicochemical properties (pH, free acid content, total acidity, lactone content and mineral content), water activity (aw), the use of metallic containers [182], and thermal and photochemical stress [164].
HMF is easily formed at low temperatures in the presence of low-pH or acidic conditions [93], while high temperature and long storage duration increase its concentration to a large extent. Nevertheless, a different pathway is proposed in dry and pyrolytic conditions under which HMF is formed from fructose and sucrose. In addition to temperature and pH, the rate of HMF formation in honey is also dependent on honey’s moisture content [62, 63]. Therefore, many steps are taken to maintain low moisture content in honey samples, including gamma irradiation and heat treatment to inhibit HMF formation.
The rate of HMF formation is also dependent on the fructose:glucose ratio and the type of sugars formed because it has been reported that at pH 4.6, fructose has five times more reactivity than glucose, and a high fructose:glucose ratio will accelerate the reaction [93]. Turhan [175] showed that temperature and duration of heat treatment may both affect HMF formation in honey samples. Moreover, it was shown that heating honey samples collected from Anatolia in Turkey at 135 °C for 100 s produced similar amounts of HMF as that yielded by heating samples to 150 °C for 40 s. According to [150], there is a logarithmic relationship between the storage time and HMF levels in honey. In their study on Malaysian honey samples [81], showed that honey samples stored for 3–6 months had HMF values below the International Honey Commission (IHC) limit for tropical honey (< 80 g/kg); however, samples stored for 12–24 months had HMF concentrations above the recommended level.
Khalil et al. [81] investigated the correlation between the physicochemical properties of honey and HMF formation and found that there was a strong correlation between free acids and total acidity, while there was only a moderate correlation between pH and lactones. In another study conducted on four honey samples (eucalyptus, sulla orange and chestnut), Fellico et al. [47] reported a relationship between HMF concentration and heating time, acidity and pH. A similar relationship was indicated in another study [30], which reported a proportional increase in HMF formation with an increase in temperature by one degree, moisture content and water activity. In addition, the concentrations of metallic ions such as manganese, zinc, magnesium, and iron(II) present in honey have also been reported to have a positive effect on HMF formation [122].
HMF in foods correlates with urine metabolites
Following oral or intravenous administration, HMF is mainly metabolized into three major metabolites: 5-hydroxymethylfuroic acid (HMFA), 2,5-furandicarboxylic acid (FDCA) and 5-(hydroxymethyl)-2-furoyl glycine (HMFG), with the possible formation of a fourth metabolic product termed 5-sulfoxymethylfurfural (SMF) [67]. However, studies designed to investigate the distribution of radio-labeled [U-14C]-HMF and its metabolites by whole-body autoradiography have revealed that neither HMF nor its metabolites are accumulated. It is estimated that 80–100% of the total amount of radioactivity is released within the first 24 h following its administration [55, 61]. HMF is first oxidized to carboxylic acid and is conjugated with glycine, leading to the formation of HMFG. The concentration of free glycine is the rate-limiting step in this pathway. Subsequently, HMF undergoes further oxidation to yield FDCA.
In an in vivo study, [61] reported the presence of three metabolites in urine following oral administration of labeled HMF (10–500 mg/kg), where the relative amounts of FDCA, HMFA, and HMFG excreted were 2–6, 78–85 and 5–8%, respectively (depending on the species and the doses). In humans, HMF is completely cleared following its oral administration (using the juice of dried plum) [130]. Similarly, in another clinical study by Hardt-Stremayr et al. [67], HMF was shown to be completely excreted through urine within 48 h following oral administration at 240 mg/day, while SMF was not detected in urine, as also reported in other studies.
HMF in various food products
In addition to being found in honey, HMF is also present in dried fruits (> 1 g/kg), products containing caramel, instant coffee (up to 6.2 g/kg), apple juice, citrus juices, beer, brandy, milk, breakfast cereal, baked foods, and tomato products, and HMF is released from sugar and carbohydrates after home cooking, indicating that HMF is ubiquitous in the diet.
We consume several different food products, including bakery goods, milk, fruit juice, cereals, coffee, chocolate, soft drinks, vinegar, wine, nuts and grilled meat in our daily lives. The majority of these produces undergo thermal treatments prior to consumption, such as boiling, baking, extrusion cooking, roasting, pasteurization and other processing. These processes are performed not only to make the products more edible but also 1) for preservation (by means of reducing microbial load and/or eliminating enzymatic activities) and 2) to generate more desirable sensory (color, aroma and taste) and texture properties. Nevertheless, extensive processing produces adverse effects by introducing undesirable, harmful and non-nutritive compounds or by reducing nutritive value, fresh appearance and taste. During thermal processing and preservation, the Maillard reaction or non-enzymatic browning may also occur, where HMF is a common product whose extent of formation depends on the processing and preservation conditions [87].
Sugar
Sugar is a disaccharide composed of fructose and glucose. It is produced exclusively from sugar beet and sugar cane. Although the extraction and purification processes are very simple, HMF is formed as a result of heat. Polovková and Šimko [128] analyzed brown (n = 25) and white sugars (n = 13) from local markets in the Bratislava territory, the capital city of the Slovak Republic. Upon preparing the sugar samples, HMF levels were determined by high-performance liquid chromatography coupled with a diode array detector (HPLC-DAD) at 284 nm. Surprisingly, white sugar was free of HMF, but brown sugar was found to contain HMF (0.17–6.45 mg/kg). The presence of HMF in brown sugar may be due to the addition of treacle to the preparation preserved at 50 °C to maintain its liquidity. Similarly, using HPLC, Risner et al. [139] found similar ranges of HMF concentration (11.9–16.4 and 12.3–23.3 mg/kg) for light and dark-brown sugars, respectively.
Cereals
According to Norwegian and German researchers, cereals and cereal products, including bread, are some of the most prominent sources of human exposure to HMF [2, 73]. The extent of HMF formation in cereal products is heavily dependent on many factors, including temperature, dough fermentation process, water activity and the presence of fruits, grains and other flavorings or additives (such as cocoa, malt, sucrose, glucose, salt, and honey).
Mańkowska et al. [101] investigated the HMF concentrations in 41 food products. Wheat bread with cranberries was reported to contain the highest amount of HMF (210 mg/kg), followed by breakfast cereals, i.e., honey wheat loops (85.09 mg/kg). The lowest amount of HMF was reported to occur in gluten-free sponge cakes and whole-grain oatmeal. Sweetened breakfast cereals contained HMF at 25.55 mg/kg, which was higher than the mean HMF concentration (18.40 mg/kg) in bakery products. In another study, the mean HMF concentrations in cereals, i.e., mixed grains (240 mg/kg), cornflakes (7–114 mg/kg) and wheat-based cereals (6–132 mg/kg), were also determined to be high [144, 145]. The products for which the dough fermentation process and the types of flour were specifically affected were again reported to be bread and other bakery products, including cookies. Additionally, HMF concentrations in different types of flours, including wheat, whole wheat, bread flour and rye flour, were investigated. The HMF concentration in bread made with rye flour was determined to be the highest (mean 26.88 mg/kg), perhaps due to the high amino acid content.
Different types of dried fruits (raisins, cranberries, palms, strawberries, red currants and apple) are added to cereal products, which normally contribute to high HMF concentrations. In fitness and rice-wheat flakes with raisins added, the HMF concentrations were reported to be 6.78 and 11.70 mg/kg, respectively. The lowest (6.06 mg/kg) HMF concentration was reported to occur in crunchy products containing raisins and plums. On the other hand, red fruits such as apples, strawberries and red currants confer some of the highest concentrations of HMF to food products, including oatmeal (47.62 mg/kg) [101]. Similarly, bread containing dried fruits tend to have higher HMF concentrations than does white bread [137, 156], indicating that the main contributor of HMF is dried fruits.
Coffee
Coffee is one of the most common drinks reported to contain HMF. The HMF concentration in coffee depends on the brewing processes or types of coffee (mocha, espresso, filtered coffee or plunger-brewed coffees) used, as well as the amount of sugar added to it. Mortas et al. [111] investigated HMF concentrations in Turkish coffees (either prepared traditionally or of the instant variety) using HPLC coupled with a diode array detector. The authors reported that before brewing, instant and traditional Turkish coffee samples contained HMF over ranges of 336.03–362.05 and 213.02–238.99 mg/kg, respectively. However, following brewing, the HMF concentration increased by 32.29–55.83% (in instant coffee) and 74.12–224.75% (traditional coffee), respectively.
Arribas-Lorenzo and Morales [15] used reversed-phase HPLC coupled with UV detection to detect HMF concentrations in three types of ground coffee consumed by the Spanish and found significant differences in the concentrations observed. HMF levels were 110 mg/kg (natural coffee: produced by traditional roasting of coffee beans), 625 mg/kg (torrefecto coffee: obtained by adding sucrose prior to the roasting process) and 1734 mg/kg (blended coffee: a combination of natural and torrefacto ground coffee in various proportions). However, soluble coffee contained the highest level (2480 mg/kg). The authors concluded that the daily HMF intake of a high coffee consumer in Spain is approximately 122.42 μg/kg, indicating that heat contributes to HMF formation.
Dairy products
HMF is formed via side reactions during heat sterilization and browning processes. Albalá-Hurtado et al. [4] investigated the formation of HMF as a result of exposure to different storage temperatures (20, 30, 37 °C) and storage duration (up to 9 months) in liquid as well as in powdered infant milk. They found that HMF formation follows a zero-order kinetics profile independent of storage temperature and milk type. A powdered infant milk sample stored at 37 °C for 12 months contained higher amounts of HMF and furfural compounds (31.5 µmol/L) than those present in liquid milk (2.5 µmol/L).
In another study on ultra-high temperature processed (UHT) milk, no significant variation in HMF levels was observed for samples stored at 4 and 8 °C. However, storage at room temperature led to a two-fold increased formation of HMF [33]. In the case of traditional Indian dairy products, there was a strong positive correlation between HMF concentration and the products’ their flavors, colors and textures [70]. Morales and Jiménez-Pérez [110] used micellar electrokinetic capillary chromatography and reported a mean HMF concentration of 29.5 µg/kg for many infant milk-based formulas.
Fruits and vegetables
Due to their rich content of sugars and amino acids, fruits and vegetables contain high HMF levels. In a study involving jam products (prepared commercially and under laboratory conditions) stored at 20 and 35 °C for 12 months, a temperature-dependent relationship was established between HMF formation and storage duration [133]. Additionally, a positive correlation between storage time and temperature with HMF formation has been observed for two varieties of apple juice (Golden amasya and delicious) [32]. Ordóñez-Santos et al. [121], who investigated changes in HMF levels in bottled tomato puree stored at 20 °C for 180 days, reported a negative correlation between HMF formation and the content of organic acids such as ascorbic, citric and mallic acids.
Murkovic and Pichler [114] analyzed HMF concentrations in dried apricot, peach, pear, fig, date, apple and pineapple products. HMF concentrations were highest in dates (1000 mg/kg) and plums (1100–2200 mg/kg). The mean range of HMF concentrations in other dried fruits was 1–780 mg/kg [114]. On the other hand, in a study by Rufían-Henares et al. [146] on commercially dehydrated vegetables, HMF was not detected, except in cabbage, tomato and artichoke (58.60, 18.20 and 6.97 mg/kg respectively).
Oil concentration in products may also affect HMF formation. To investigate this hypothesis, Fallico et al. [45] roasted defatted and ground hazelnuts containing different amounts of hazelnut oils or oil containing saccharose and/or hexanol. The defatted hazelnuts roasted in high oil concentrations exhibited increased HMF formation. Non-defatted hazelnuts with saccharose contained the highest HMF concentration (372 mg/kg), while defatted hazelnuts with saccharose contained the lowest (33.5 mg/kg). Because the HMF concentration increased from 66.5 to 144.0 mg/kg in non-defatted samples upon prolonged roasting (from 30 to 60 min respectively), the authors also concluded that HMF levels increased with the duration of the heat treatment applied to the products.
Other miscellaneous food products
It is concluded that there are no heat-processed food products that are free of HMF. The HMF concentrations in several food products are listed in Table 2.
Table 2.
HMF concentrations in different food products
| Food type | HMF concentrations (mg/kg) | Detection method | Country | References |
|---|---|---|---|---|
| Must syrup | 3500–11,000 | GC–MS | Spain | [147] |
| Molasses | 100 | |||
| Sugarcane syrup | 100–300 | |||
| Palm syrup | < 3 | |||
| Prunes | 237 | HPLC | Norway | [73] |
| Dark bear | 13.3 | |||
| Canned peaches | 5.8 | |||
| Raisins | 5.0 | |||
| Balsamic vinegar | 316–3250 | HPLC | Italy | [171] |
GC–MS gas chromatography–mass spectrometry, HPLC high performance liquid chromatography
Comparison of HMF levels in honey and different food products
HMF is not present naturally in food products. The compound is formed upon thermal treatment and in combination with other factors. As it is a product of the non-enzymatic Maillard reaction, there is no fixed concentration of HMF in different food items. The baking temperature, rate of saccharose degradation, concentration of reducing sugars, type of sugar (glucose, fructose or others), water activity, the addition of other food additives such as HMF-containing sweeteners, coloring agents, caramelization, storage time and temperature, type of metallic storage and processing container vary widely among different food items. Therefore, HMF content varies among food items, even among those of the same type. Nevertheless, honey is a safer food item than other processed foodstuffs with respect to its HMF concentration. Turhan et al. [174] showed that the initial heating temperature and time are not directly correlated with HMF concentration in honey. Nectar honey and honeydew honey processed at 95 °C for 90 min and 90 °C for 75 min showed HMF levels lower than 40 mg/kg. Storage temperature and storage duration in particular directly influence the development of HMF in stored honey [44, 81]. Unlike for honey, in the processing of other foodstuffs, comparatively higher temperatures (during baking, roasting), longer duration times, and different additives are required, which profoundly affect the HMF content in the foods. For example, cookies baked at high temperature contain 10–100 times more HMF (167.4–1100.1 mg/kg) than cookies baked at 200 °C (9.9–39.6 mg/kg) [10]. Fresh cookies baked at 300 °C and with saccharose added during processing have been reported to contain as much as 1100 mg/kg HMF. Even ammonium bicarbonate addition may dramatically increase the HMF content (above 3500 mg/kg) of saccharose-containing cookies baked at 220 °C. In investigating 22 coffee samples, Murkovic and Pichler [114] reported HMF concentrations ranging from 300 to 1900 mg/kg. Murkovic and Bornik [113] found that HMF formation was dramatically increased in coffee roasted at 240 °C within the first 3 min, with concentrations reaching up to 900 mg/kg. In the case of bread, HMF values vary widely, from 3.4 to 176.1 mg/kg, depending on the fermentation conditions, the leavening agents added, the thickness of the crust and crumb, and the type of bread (oat bread, white bread or wheat bread) [137]. According to Raimirez-Jimenez et al. [137], the HMF content of biscuits (15.6 mg/kg) is higher than that of confectionaries such as doughnuts and croissants (9.5 mg/kg). Thus, the formation of HMF is inevitable, and comparing or ranking food items with respect to HMF concentration cannot be performed precisely.
Detection methods of HMF
Accurate quantitative analysis of HMF is of great importance because HMF is a marker of quality deterioration, thermal processing and other adulteration practices. HMF is also important in clinical research and therapeutics. The IHC has recommended three main methods for HMF determination: two spectrophotometric methods by White and Winkler and a reversed-phase high-performance liquid chromatography (HPLC) method [191].
Prior to the availability of the spectrophotometric methods, both optical and chemical methods were used. In his newer spectrophotometric method, White utilized a clarified honey sample containing 0.1% sodium bisulfate as a reference and as honey solution without sodium bisulfate as a sample [180]. On the other hand, the Winkler method utilizes honey solutions with p-toluidine and barbituric acid [184]. Nevertheless, although these methods are fast, they lack good sensitivity and specificity. Moreover, the method described by Winkler requires the use of p-toluidine, which is carcinogenic. Although the HPLC method is relatively more expensive, it is advantageous with respect to both labor and time. In addition, the method is deemed as an automated and sensitive method that can exclude many interferences from other related compounds [185]. Nevertheless, although HPLC is a sophisticated technique, the method is still not satisfactory to some who have recommended further development and modification of the method [13].
Reyes-Salas et al. [138] reported an electrochemical approach for HMF detection. In this method, a single and sharp reduction signal was created at − 1100 mV versus argentum or argentum chloride, while borate was used as a supporting electrolyte. Another method is the ion exchange liquid chromatography–photodiode array detection technique described by Yuan and Chen [189], which is consistent with Winkler’s method. Another method involves automated flow injection, as reported by Iglesia et al. [38], which is based on the operating principle of the Winkler method and provides a detection range of 5–40 ppm. Micellar electrokinetic capillary chromatography (MEKC) is another rapid method that uses caffeine as a standard. The technique is suitable for rapid quantification of HMF, particularly in honey samples, without requiring sample pretreatment [140]. A unique and efficient rapid screening technique is direct analysis in real time (DART) coupled with time-of-flight mass spectrometry (TOF-MS), which has been reported to yield a chromatogram with high resolution [134]. The method can quantitatively analyze HMF concentrations in a more precise manner than other methods and requires no (or very little) sample pretreatment.
Optimization of HMF formation in different foods
HMF is formed unavoidably in most foods containing monosaccharides (such as glucose, galactose, or fructose) during processing as a function of temperature and storage period. The process of HMF formation in any food is multifactorial. Factors affecting the rate of HMF formation include temperature, pH [62], type of sugar [93], concentration of divalent cations [64] and water activity [63, 90] in the medium. Hence, to control and optimize the level of HMF formation is a troublesome task. Therefore, food processors must know the effects of different factors so that necessary precautions can be adopted previously to limit and/or optimize the synthesis of HMF in food. To understand the conditions under which HMF would be formed only at desirable levels, a more fundamental and systematic approach must be adopted. The possible effects of these factors must be studied simultaneously and in combination. Toker et al. [172] recommended central composite face-centered design (CCFD) of response surface methodology (RSM) as a useful experimental technique for this purpose. According to Hunter [72], this versatile and effective systematic tool can be used to determine the optimal levels of the contributing factors for the parameters concerned. The mathematical expression derived by this methodology can be employed to develop predictive models upon setting the levels of the various influencing factors to reach the optimum HMF concentration in food.
Hazard posed by HMF to honey bees
The productivity of apiculture is highly dependent on the health, survival and quality of honey bees. Therefore, improving the health quality of bee food is an important priority. Normally, sources of carbohydrates for honey bees are the nectar and honeydew collected from plants, as processed by foragers. However, in the case of commercial honey production, the food and nutrients of cultured bees are depleted following the collection of the autumn yield from hives. Furthermore, honey bees’ natural resources tend to wane during winter. Therefore, bee keepers must use alternative sources to replenish the carbohydrate demand of bees, such as sucrose, high fructose corn syrup (HFCS), various fruit sugars and invert sugars [117]. Among these, however, compared with sucrose, HFCS is considered a dynamic source due to its practical storage, transportation facility and liquidity as opposed to the crystallization method. HFCS is produced commercially from corn starch by using enzymes such as alpha amylase, amylo-glucosidase and invertase [26, 29], whereas sucrose is produced by acid hydrolysis alone [18]. Regardless, these sources are not safe because HMF and its daughter products (formic and levulinic acids) are formed, thus posing a potential threat to honey bees (Fig. 3). Beet sugar, which is also used to feed bees, has been reported to contain up to 475 mg/kg HMF, depending on the processing and storage conditions [177]. In another study, a sugar solution containing 30–150 mg/kg of HMF was used to feed honey bees and was found to cause 15–58.7% of deaths of caged bees within 20 days [78]. Krainer et al. [88] reported that the LD50 values of HMF for bee larvae fed for more than 6 days were 4280 and 2424 mg/kg on days 7 and 22, respectively. Interestingly, they found that 7-day larvae emerging after being fed HMF for 6 days were more sensitive than were adult bees, but the emerging adult bees at day 22 were less sensitive. Moreover, the authors concluded that HMF used as a supplement in food does compromise colony fitness but may not cause great losses of brood. It is hypothesized that HMF causes bees to experience dysentery-like symptoms and ulcers in the gastrointestinal intestinal tract [18], leading to their death.
Fig. 3.
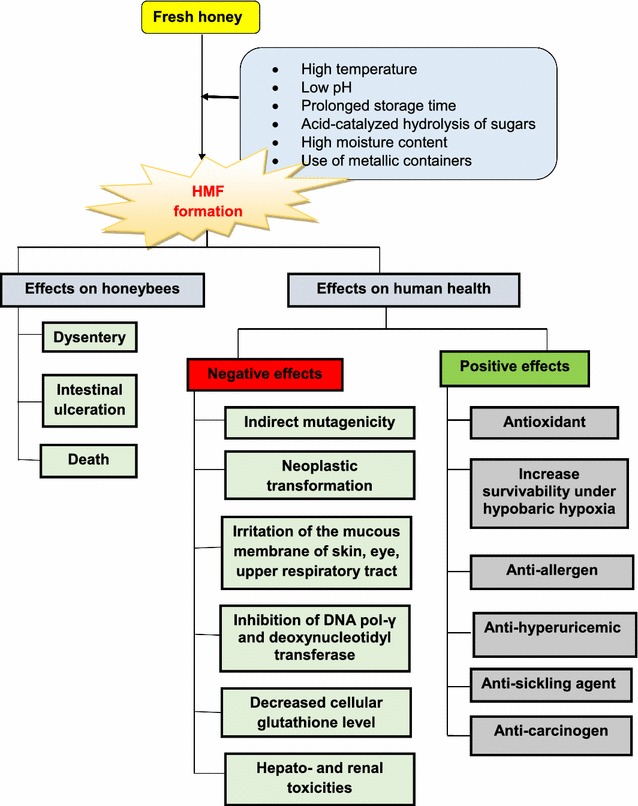
HMF effects on honey bee and human health
Effects of HMF on human health
HMF and other congeners exert both detrimental and positive effects on human health (Fig. 3).
Adverse effects on human health
HMF and its derivatives have been confirmed to confer genotoxic, mutagenic, carcinogenic, DNA-damaging, organotoxic and enzyme inhibitory effects.
HMF as an indirect mutagen
Florin et al. [51] investigated the mutagenic activity of certain compounds, including HMF, towards four mutant strains of Salmonella typhimurium. Liver extracts from methylcholanthrene-induced rats were used for metabolic activation of the investigated compounds, and it was confirmed that HMF is not a mutagen. In contrast, Lee et al. [94] demonstrated that HMF is an indirect mutagen because it is converted to an active metabolite, the sulfuric acid ester 5-sulfo-oxymethylfurfural (SMF), with mutagenicity towards S. typhimurium TA104. HMF is enzymatically activated to SMF by sulfotransferases (SULT) contained in rat liver extracts enriched with the sulfo group donor 3′-phosphoadenosine-5′-phosphosulfate (PAPS). This finding is further supported by another study in which HMF, 2,5-bishydroxymethylfuran (metabolite of HMF), furfuryl alcohol (FFA) and 5-methyl-FFA showed mutagenicity towards S. typhimurium TA100 expressing human SULT1C2 but not towards the parental strains [60].
Another study using FVB/N (FVB) mice expressing hSULT1A1/1A2 revealed that HMF has a genotoxic effect. Mice orally administered single doses of 900 or 1300 mg/kg showed significant DNA damage of their renal cells, as detected by an alkaline single cell gel electrophoresis assay. Another furan derivative, 2,5 dimethylfuran (DMF), also causes DNA damage in the kidney and the colon [69]. In an in vitro erythropoietic micronucleus assay, rat bone marrow cells were exposed to DMF at a concentration of 0.1 mM for 1 h. The study indicated that DMF exhibits genotoxicity toward hematopoietic cells [53].
In another study conducted on five cell lines possessing different levels of SULT1A1 activity (mouse L5178Y, no activity; Chinese hamster: V79-Hp-PST, high activity; V79, negligible activity; human: HEK293, higher activity; and Caco-2, low activity), HMF exerted DNA damage when exposed at a concentration of 100 mM for 3 h [42]. However, it was also determined that HMF poses DNA-damaging effects irrespective of the SULT1A1 activity of the cell lines. In addition, 5-HMF was determined to cause chromosomal aberrations in a Chinese hamster V79-derived cell line constitutively expressing human sulfotransferase SULT1A1 and CYP2E1. In fact, 5-HMF is a potent inducer of sister-chromatid exchange at a concentration of 19.8–3808.0 µM in cells exposed for 32 h [59]. Nishi et al. [118] confirmed that 5-HMF causes chromosomal aberrations of V79 cells at 15.8 mM. In both their pre-clinical and clinical studies, Pastoriza et al. [125] indicated that orally administered HMF is converted to reactive SMF after being absorbed through the gastrointestinal tract. SMF forms a DNA-SMF adduct in mice’s kidney, leukocytes and liver cells, as well as in leukocytes of pre-adolescent populations. In addition, SMF is not properly excreted through urine due to renal reabsorption, thus allowing SMF to accumulate in the plasma, making it available to react with cellular proteins and DNA.
HMF as a dual player in carcinogenesis
HMF and its derivative SMF have been confirmed to be potent carcinogens in several studies at the preclinical level. The compounds cause neoplastic transformations in several organs, including the colon and the skin. Colon cancer involves the development of a multistep stage in which micro-adenomas and aberrant crypt foci (ACF) are the morphological markers [14]. HMF acts as an initiator as well as a promoter of ACF [28]. A study was conducted on multiple intestinal neoplasia mice that were subcutaneously administered with a single dose of HMF (500 mg/kg) or SMF (25 mg/kg) within 3–6 days after birth. After 12 weeks, administration of both HMF and SMF increased the number of small intestinal adenomas and flat dysplastic lesions (flat ACF) in the large intestine [168]. The mice had a mutant copy of the tumor suppressor gene adenomatous polyposis coli (APC). Mutation in the APC gene leads to the development of adenomas in the small and large intestines, analogously to human familial adenomatous polyposis syndrome (FAP) [127, 129].
In a study by Zhang et al. [192], 45% of F344 female rats administered with HMF at 250 mg/kg twice per day by oral gavaging were found to develop a large intestinal ACF on day 30. HMF increases both the number and size of ACF. In contrast, an extensive study (with two mouse models: wild-type FVB/N and genetically engineered mice having several copies of human SULT1A1 and SULT1A2 at chromosome 9), Florian et al. [50] yielded opposite findings (i.e., neither HMF nor its metabolite SMF induces ACF and colonic tumors). HMF also induces skin papilloma. Upon topical application of sulfoxymethyl and chloromethyl derivatives of HMF, the mice were found to develop papillomas on their skin.
Another derivative of HMF, 5-chloromethyfurfural, has been found to induce hepatocarcinoma in B6C3F1 male rats at a very early age [166]. Schoental et al. [153] showed that rats subcutaneously administered with HMF (200 mg/kg), developed renal lipomatous tumors. In contrast to these studies, a study by Zhao et al. [194] using the A375 cell line indicated that HMF can induce apoptosis and G0/G1 arrest in DNA-damaged cells via the reactive oxygen species (ROS)-mediated signal transduction pathway. Thus, it is concluded that HMF is a potent anti-carcinogen.
HMF as an organotoxic agent
HMF has cytotoxic effects at high concentrations, as the compound causes irritation in the mucous membranes, skin, eyes and upper respiratory tract [109]. SMF was revealed to be a strong nephrotoxic agent in an in vivo study in which male FVB/N mice were intraperitoneally administered with SMF (250 mg/kg). After 5–11 days post-treatment, the mice either died or became morbid, possibly due to liver damage or more severe renal damage, particularly at the proximal tubules. A histopathological study revealed extensive proteinaceous casts and necrosis in the affected area [22]. The organic anion transporters type 1 and 2 (OAT1 and OAT2) are highly conserved in different species, including rat and human, and are usually expressed on the cellular basolateral membrane of the proximal tubules. The transporters mediate the uptake of different organic anions, including SMF, from the blood stream as their substrates and are concentrated into tubular cells, which mostly affect the proximal tubules [31]. Another study using human embryonic kidney cells (HEK293) that constitutively express OAT1 and OAT2 transporters supports this mechanism [19]. Glutathione is an important endogenous antioxidant in the body. An ex vivo study involving two mammalian cell lines, V79 and Caco-2, indicated that HMF decreased cellular glutathione levels in a concentration-dependent manner at 50 mM and 120 mM, respectively [79].
HMF as an enzyme inhibitor
The human genome encodes 16 DNA-dependent DNA polymerases. Among them, three are involved in nuclear DNA replication, while the remaining are involved in the repair system [24, 52]. Human polymerase γ is a multifunctional enzyme with DNA polymerase, terminal transferase, 5′-deoxyribose phosphate (dRP) lyase and polynucleotide synthetase activities. The primary structure of the polymerase includes a nuclear localization signal, a BRCA1 carboxy terminus domain, a proline-rich region, a pol β-like region and a pol X region [27, 135, 136]. DNA pol γ shares a sequence homology with the terminal deoxynucleotidyl transferase (TdT) that catalyzes the addition of deoxyribonucleotide to the 3’ end of dsDNA or ssDNA in a template-independent manner [124, 143]. HMF competitively inhibits DNA pol γ and TdT with respect to the deoxynucleotide substrate and DNA template primer with 50% minimum inhibitory concentration (IC50) values of 26.1 and 5.5 µM, respectively [105].
Positive effects of HMF on human health
HMF as an antioxidant
ROS are produced as toxic by-products of the body’s aerobic metabolism. The species oxidize cellular macromolecules such as proteins, membrane lipids, and DNA and cause cellular damage. The consequences range from stress to metabolic defects, neurodegenerative diseases or even neoplastic transformations [159]. In a study by Zhao et al. [193], HMF showed a dose-dependent (0.8–6.4 mM) free-radical scavenging capacity. HMF also has significant protective effects on erythrocytes against ROS-induced damage. To investigate the protective effect and oxidative stress induced by 2,2′-azobis (2-amidinopropane) dihydrochloride (AAPH), the levels of ROS and malondialdehyde (MDA: indirect determinant of lipid peroxidation) production, the activity of the antioxidant enzymes glutathione peroxidase (GPx), superoxide dismutase (SOD), and catalase (CAT) in erythrocytes pre-treated with HMF were determined. It was revealed that the contents of ROS and MDA were reduced in HMF-treated cells, while the activities of these enzymes were elevated relative to those of negative control cells [193]. The ROS scavenging activity of HMF is imparted by its structure, which features functional reactive groups such as an aldehyde oxygen, double bonds and another oxygen in its furan ring. These features can attract electrons easily and quench ROS [176].
HMF also has a protective effect at the morphological and biochemical levels on hepatocytes damaged by hydrogen peroxide (H2O2)-induced oxidative stress, under which cells undergo morphological changes such as wrinkling, condensation of chromatin and splitting of nuclei—hallmarks of apoptotic and necrotic cells. However, in a H2O2-stressed human liver cell line (L02), cells treated with HMF (0.79 µM) could preserve their morphology better than non-treated cells could. HMF also reduces the levels of caspases 3 and 9 (executioner of apoptosis) in these cells [178]. Ding et al. [39] hypothesized the underlying protective biochemical mechanism, which may be due to inhibition of apoptosis by accelerating the transition of cells in the S phase to the G2 or M phase, as well as decreased levels of nitric oxide and caspase 3.
HMF against hypoxic injury
Oxygen is required for cell survival. Deficiency of oxygen (hypoxic condition) has numerous detrimental and even life-changing effects on health. Hypoxia may be induced by many factors, including altitude and self-related conditions such as ischemia, atherosclerosis and cancer [126]. Several cellular mechanisms are triggered and can ameliorate hypoxic conditions, among which extracellular signal-regulated kinase(ERK)-mediated transactivation of the transcription factor and hypoxia-inducible factors (HIF) are believed to participate [151]. The mitochondrial membrane potential is also reduced and negatively affects hypoxic cells [75]. In their in vitro study of the cell line ECV304 (human umbilical cord vein endothelial cell), Li et al. [96] showed that cells pre-treated with HMF (200 µg/ml for 1 h) before being exposed to hypoxic conditions (0.3% oxygen for 24 h) exhibited increased mitochondrial membrane potential and decreased phosphorylated ERK levels. The number of apoptotic and necrotic cells also significantly declined. In their further study with a Kunming mice model, the authors showed that pre-exposure to HMF (100 µg/ml, 1 h) significantly attenuates the extent of hypobaric hypoxia-induced permeability of the blood–brain barrier (BBB). Pre-exposure also decreases the extent of neuronal damage in the CA1 region of the hippocampus. Thus, because HMF increases survivability under hypobaric hypoxic conditions, it may be a potent therapeutic agent against acute mountain sickness (AMS), high-altitude cerebral edema (HACE) and high-altitude pulmonary edema (HAPE) [95].
HMF as an anti-allergen
Basophils and mast cells participate in the pathogenesis and manifestations of allergic reactions such as asthma, atopic dermatitis and allergic rhinitis. RBL-2H3 cells are mast cells located in the mucosal layer. These cells express immunoglobulin Fc epsilon receptor type I (FcεRI) on their surface. The crosslinking of IgE with specific protein antigens and the binding of crosslinked IgE to FcεRI trigger intracellular signal transduction cascades. These events lead to Ca2+ influx and the release of mediators by degranulation, MAPK phosphorylation, upregulation of cytokine gene expression and increased ROS generation [23, 83, 84, 154]. Yamada et al. [187] showed that HMF acts at different stages to inhibit degranulation at doses of 0.01–0.30 µg/ml. In addition, HMF interferes with antigen-antibody crosslinking and antibody-receptor binding. HMF also blocks calcium (Ca2+) influx into IgE-sensitized bovine serum albumin-stimulated RBL-2H3 cells. Nicotinamide adenine dinucleotide phosphate (NADPH) oxidase plays a major role in the production of ROS in IgE-mediated RBL-2H3 cells. H2O2 and NO, two major ROS, are known to regulate degranulation and Ca2+ signaling in mast cells [83, 84, 186]. A significant inverse association exists between the release of histamine and Ca2+ from intracellular stores and the superoxide anion or DPPH scavenging activities. The anti-allergen effect of HMF on cells is due to the blocking of histamine release and Ca2+ signaling through the compound’s free-radical scavenging activity [98, 167].
In another study using ovalbumin (OVA)-immunized BALB/c mice, HMF decreased the levels of total IgE and OVA-specific IgE. The study also showed that HMF-treated immunized mice exhibited lower levels of IFNγ (Interferon gamma) and IL-4 (Interleukin 4) than those of untreated mice. Therefore, HMF may be a potent anti-allergic compound [7].
The use of HMF for other pathologic conditions
Uric acid is the end product of purine catabolism. The final two steps of the purine catabolic pathway are catalyzed by a critical enzyme, xanthine oxidase (XO). Uric acid is mainly excreted via urine. High levels of uric acid in the blood lead to the development of hyperuricemia [123], which is the main cause of gout. In addition, many other pathological states are associated with hyperuricemia, including metabolic syndrome, heart failure, pulmonary disorder and type 2 diabetes mellitus [68]. Increased XO activity downregulates the anti-inflammatory transcription factor peroxisome proliferator-activated receptor-γ (PPARγ) and accelerates inflammatory action [57]. XO is also an endogenous producer of superoxide, a potent activator of nuclear factor kappa B (NFκB) [99]. NFκB acts as a transcription factor and upregulates the expression of nitric oxide synthase 2 (NOS2) and interleukin 8 (IL-8). HMF exerts an anti-inflammatory effect by downregulating NFκB [86] and inhibits the activity of XO [97].
HMF as an anti-sickling agent
Hemoglobinopathies such as sickle cell disease are life-threatening. The underlying mechanism is polymerization of abnormal hemoglobin (sickle hemoglobin, HbS) under hypoxic conditions. The pathophysiology is manifested by the deformation of red blood cells, loss of resilience and occlusion of tiny blood capillaries, leading to morbidity [65, 100, 165]. Although there are many anti-sickling agents, these agents are not free of side effects. Many agents also exhibit reduced bioavailability in minimal doses and react to non-target proteins. In addition, HMF can act as an effective anti-sickling agent. Abdulmalik et al. [1] showed that HMF orally administered in low concentrations is absorbed into the blood stream from the gastrointestinal tract in transgenic (Tg) sickle mice. Subsequently, HMF can penetrate the erythrocytes and form stable a Schiff-base adduct with the N terminal αV11 nitrogen of HbS in a symmetrical fashion. Following the formation of such an adduct, HMF allosterically shifts the oxygen equilibrium curve to the left and prevents the sickling of erythrocytes. In fact, the authors also reported that Tg sickle mice pre-treated with HMF survived longer under hypoxic conditions than did untreated mice, indicating the potential of HMF as an anti-sickling agent.
Tolerable daily intake (TDI) of HMF
Many studies have revealed that different cells respond to 5-HM-induced cytotoxicity differently. The susceptibility of cells to HMF depends on the presence and expression levels of receptors [119], metabolism [157], structure [149] and the enzyme activity of HMF [158]. At the preclinical level, no toxic effects have been observed at daily doses ranging from 80 to 100 mg/kg body weight [2]. Zaitzev et al. [190] established the TDI for HMF as 132 mg/day using a 40-fold safety margin. The European Food Safety Authority (EFSA) [43] established a threshold of concern of 0.54 mg/day for the intake of furan derivatives used as flavoring agents in Europe. However, in their study involving 268 Spanish school children, Pastoriza et al. [125] found that although the students’ daily HMF intake, well-distributed throughout the day, was 10–70 mg, a statistically significant level of SMF, a potent toxic metabolite of HMF, was found in their plasma. In addition, most experiments concerning the health effects of HMF have been carried out in vitro, ex vivo and at the experimental animal level. Thus, based on the data available to date, it is not possible to ascertain a TDI. In addition, further research, particularly at the clinical level, paving the way for recommending a TDI for HMF is worth considering and appreciating.
Mitigation strategies
There is no particular strategy for mitigating the formation of HMF in honey due to the numerous precursors of HMF and the types of reaction orders involved [35]. HMF is formed following a zero-order kinetic process in an exponential manner [54]. Various mechanistic strategies may be adapted to mitigate the formation of HMF. There is a positive relation between a high temperature-time profile and acidic pH for the formation of HMF, which can be modified [62]. In fact, the reduction of thermal input can significantly decrease the formation of HMF, which can be achieved by various means, for example, by adjusting the temperature of the processing oven when the moisture content is high or, conversely, by decreasing the temperature when the moisture content is low. Reduced thermal input can also be achieved by heating food to a low temperature for a prolonged period by vacuum frying or by dielectric heating (microwave and radiofrequency). In the case of dielectric heating, an external alternating current is applied, causing the dielectric molecules in food products to undergo rotation and friction, thus ensuring the rapid generation of uniform heat. In addition, because water molecules are the main target of this mechanism, heat is generated in the portion of food containing water [195]. Because long storage periods lead to high HMF formation in honey [81], shortening the storage period can reduce the formation of HMF.
Strategy for removing HMF and other furans
To remove already formed HMF, a post-process vacuum technology can be exploited. In this technology, depending on the physical and chemical properties of the compounds to be removed, the temperature, pressure and time can be adjusted. The effectiveness of this technology depends on food composition, moisture content and the nature of the compound to be removed [12, 132]. Viscosity minimizes the diffusion of molecules through the matrix and thus compromises the effect of the process in dry foods [49]. Because honey is liquid or semi-liquid, the process can effectively remove HMF from honey. The technology can be applied to any finished product without any modification of any of the process parameters [11].
Conclusion and future directions
Multiple factors affect HMF production in honey and other food products. Therefore, determining how to prevent or reduce the formation of HMF remains a challenge. In addition, the determination of the compound’s concentration and the low efficiency of removal methods make it difficult to establish a complete database of HMF concentrations in different monofloral and multifloral honeys. As a constituent of processed foods, HMF has both profoundly adverse and beneficial effects on human and bee health. Some effects of HMF on human health and its carcinogenic as well as anti-carcinogenic properties remain inconclusive, with many studies conducted only at preclinical levels.
Authors’ contributions
UMS, MS and MIK participated in designing the overall idea and discussions of views represented in the paper. UMS and MS were involved in searching the literature and preparing the draft manuscript. NA, MIK and SHG provided the overall concept and critically edited and approved the final version of the manuscript. All authors read and approved the final manuscript.
Competing interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
Not applicable.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Abbreviation
- AAPH
2,2′-azobis (2-amidinopropane) dihydrochloride
- ACF
aberrant crypt foci
- AMS
acute mountain sickness
- APC
adenomatous polyposis coli
- BBB
blood–brain barrier
- CAT
catalase
- CCFD
central composite face-centered design
- CYP2E1
cytochrome P450 2E1
- DART
direct analysis in real time
- DMF
2,5-dimethylfuran
- DPPH
2,2-diphenyl-1-picrylhydrazyl
- DRP
deoxyribose phosphate
- EFSA
European Food Safety Authority
- ERK
extracellular signal-regulated kinase
- FAP
familial adenomatous polyposis syndrome
- FcεRI
express immunoglobulin Fc epsilon receptor type I
- FDCA
2,5-furandicarboxylic acid
- FFA
furfuryl alcohol
- GPx
glutathione peroxidase
- IFNγ
interferon gamma
- IgE
immunoglobulin E
- IHC
International Honey Commission
- IL-4
interleukin 4
- H2O2
hydrogen peroxide
- HACE
high-altitude cerebral edema
- HAPE
high-altitude pulmonary edema
- HbS
sickle hemoglobin
- HFCS
high fructose corn syrup
- HIF
hypoxia inducible factors
- HMF
5-hydroxymethylfurfural
- HMFA
5-hydroxymethylfuroic acid
- HMFG
5-(hydroxymethyl)-2-furoyl glycine
- HPLC-DAD
high-performance liquid chromatography coupled with a diode array detector
- MAPK
mitogen activated protein kinase
- MAD
malondialdehyde
- MEKC
micellar electrokinetic capillary chromatography
- NADPH
nicotinamide adenine dinucleotide phosphate
- NFκB
nuclear factor kappa B
- NOS2
nitric oxide synthase 2
- OAT1 and OAT2
organic anion transporters type 1 and 2
- OVA
ovalbumin
- PAPS
3′-phosphoadenosine-5′-phosphosulfate
- PPARγ
peroxisome proliferator-activated receptor γ
- ROS
reactive oxygen species
- RSM
response surface methodology
- SMF
5-sulfoxymethylfurfural
- SOD
superoxide dismutase
- TDI
tolerable daily intake
- SULT
sulfotransferase
- TdT
terminal deoxynucleotidyl transferase
- TOF-MS
time-of-flight mass spectrometry
- UHT
ultra-high temperature processing
- XO
xanthine oxidase
Footnotes
Ummay Mahfuza Shapla and Md. Solayman contributed equally to this work
Contributor Information
Ummay Mahfuza Shapla, Email: shapla.ar@gmail.com.
Md. Solayman, Email: naeem40thju@gmail.com
Nadia Alam, Email: najui.bd@gmail.com.
Md. Ibrahim Khalil, Email: drmikhalil@gmail.com.
Siew Hua Gan, Email: gan.siewhua@monash.edu.
References
- 1.Abdulmalik O, et al. 5-hydroxymethyl-2-furfural modifies intracellular sickle haemoglobin and inhibits sickling of red blood cells†. Br J Haematol. 2005;128:552–561. doi: 10.1111/j.1365-2141.2004.05332.x. [DOI] [PubMed] [Google Scholar]
- 2.Abraham K, Gürtler R, Berg K, Heinemeyer G, Lampen A, Appel KE. Toxicology and risk assessment of 5-Hydroxymethylfurfural in food. Mol Nutr Food Res. 2011;55:667–678. doi: 10.1002/mnfr.201000564. [DOI] [PubMed] [Google Scholar]
- 3.Akhtar S, Ali J, Javed B, Hassan S, Abbas S, Siddique M. Comparative physiochemical analysis of imported and locally produced Khyber Pakhtunkhwa honey. Global J Biotechnol Biochem. 2014;9:55–59. [Google Scholar]
- 4.Albalá-Hurtado S, Veciana-Nogués MT, Mariné-Font A, Vidal-Carou MC. Changes in furfural compounds during storage of infant milks. J Agric Food Chem. 1998;46:2998–3003. doi: 10.1021/jf980079f. [DOI] [PubMed] [Google Scholar]
- 5.Alfredo ASC, da Silva CG, Marchini SLC. Características físico-químicas de amostras de mel de Melipona asilvai (Hymenoptera: Apidae) Ciência Rural. 2004;34:644–650. [Google Scholar]
- 6.Alimentarius C. Revised codex standard for honey. Codex Stan. 2001;12:1982. [Google Scholar]
- 7.Alizadeh M, Khodaei H, Mesgari Abbasi M, Saleh-Ghadimi S. Assessing the effect of 5-hydroxymethylfurfural on selected components of immune responses in mice immunised with ovalbumin. J Sci Food Agric. 2017;97:3979–3984. doi: 10.1002/jsfa.8261. [DOI] [PubMed] [Google Scholar]
- 8.Aloisi PV. Determination of quality chemical parameters of honey from Chubut (Argentinean Patagonia) Chilean. J Agric Res. 2010;70:640–645. [Google Scholar]
- 9.Alvarez-Suarez JM, et al. Antioxidant and antimicrobial capacity of several monofloral Cuban honeys and their correlation with color, polyphenol content and other chemical compounds. Food Chem Toxicol. 2010;48:2490–2499. doi: 10.1016/j.fct.2010.06.021. [DOI] [PubMed] [Google Scholar]
- 10.Ameur LA, Mathieu O, Lalanne V, Trystram G, Birlouez-Aragon I. Comparison of the effects of sucrose and hexose on furfural formation and browning in cookies baked at different temperatures. Food Chem. 2007;101:1407–1416. doi: 10.1016/j.foodchem.2006.03.049. [DOI] [Google Scholar]
- 11.Anese M, Manzocco L, Calligaris S, Nicoli MC. Industrially applicable strategies for mitigating acrylamide, furan, and 5-hydroxymethylfurfural in food. J Agric Food Chem. 2013;61:10209–10214. doi: 10.1021/jf305085r. [DOI] [PubMed] [Google Scholar]
- 12.Anese M, Suman M, Nicoli MC. Acrylamide removal from heated foods. Food Chem. 2010;119:791–794. doi: 10.1016/j.foodchem.2009.06.043. [DOI] [Google Scholar]
- 13.Anklam E. A review of the analytical methods to determine the geographical and botanical origin of honey. Food Chem. 1998;63:549–562. doi: 10.1016/S0308-8146(98)00057-0. [DOI] [Google Scholar]
- 14.Archer MC, et al. Aberrant crypt foci and microadenoma as markers for colon cancer. Environ Health Perspect. 1992;98:195. doi: 10.1289/ehp.9298195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Arribas-Lorenzo G, Morales FJ. Estimation of dietary intake of 5-hydroxymethylfurfural and related substances from coffee to Spanish population. Food Chem Toxicol. 2010;48:644–649. doi: 10.1016/j.fct.2009.11.046. [DOI] [PubMed] [Google Scholar]
- 16.Ayansola AA, Banjo AD. Physico-chemical Evaluation of the Authenticity of Honey Marketed in Southwestern Nigeria. J Basic Appl Sci Res. 2011;1:3339–3344. [Google Scholar]
- 17.Bachmann S, Meier M, Kanzig A. 5-Hydroxymethyl-2-furfural (HMF) in lebensmitteln. Lebensmittelchemie. 1997;51:49–50. [Google Scholar]
- 18.Bailey L. The effect of acid-hydrolysed sucrose on honeybees. J Apic Res. 1966;5:127–136. doi: 10.1080/00218839.1966.11100146. [DOI] [Google Scholar]
- 19.Bakhiya N, Monien B, Frank H, Seidel A, Glatt H. Renal organic anion transporters OAT1 and OAT3 mediate the cellular accumulation of 5-sulfooxymethylfurfural, a reactive, nephrotoxic metabolite of the Maillard product 5-hydroxymethylfurfural. Biochem Pharmacol. 2009;78:414–419. doi: 10.1016/j.bcp.2009.04.017. [DOI] [PubMed] [Google Scholar]
- 20.Ball DW. The chemical composition of honey. J Chem Educ. 2007;84:1643. doi: 10.1021/ed084p1643. [DOI] [Google Scholar]
- 21.Baroni MV, Arrua C, Nores ML, Fayé P, del Pilar Díaz M, Chiabrando GA, Wunderlin DA. Composition of honey from Córdoba (Argentina): assessment of North/South provenance by chemometrics. Food Chem. 2009;114:727–733. doi: 10.1016/j.foodchem.2008.10.018. [DOI] [Google Scholar]
- 22.Bauer-Marinovic M, Taugner F, Florian S, Glatt H. Toxicity studies with 5-hydroxymethylfurfural and its metabolite 5-sulphooxymethylfurfural in wild-type mice and transgenic mice expressing human sulphotransferases 1A1 and 1A2. Arch Toxicol. 2012;86:701–711. doi: 10.1007/s00204-012-0807-5. [DOI] [PubMed] [Google Scholar]
- 23.Beaven MA, Metzger H. Signal transduction by Fc receptors: the Fc&RI case. Immunol Today. 1993;14:222–226. doi: 10.1016/0167-5699(93)90167-J. [DOI] [PubMed] [Google Scholar]
- 24.Bebenek K, Kunkel T. DNA repair and replication. Adv Protein Chem. 2004;69:137–165. doi: 10.1016/S0065-3233(04)69005-X. [DOI] [PubMed] [Google Scholar]
- 25.Belay A, Solomon W, Bultossa G, Adgaba N, Melaku S. Physicochemical properties of the Harenna forest honey, Bale, Ethiopia. Food Chem. 2013;141:3386–3392. doi: 10.1016/j.foodchem.2013.06.035. [DOI] [PubMed] [Google Scholar]
- 26.Blanchard P, Geiger E. Production of high fructose corn syrup in the USA. A review article. Sugar Technol Rev. 1984;11:1–94. [Google Scholar]
- 27.Bork P, Hofmann K, Bucher P, Neuwald A, Altschul S, Koonin E. A superfamily of conserved domains in DNA damage-responsive cell cycle checkpoint proteins. FASEB J. 1997;11:68–76. doi: 10.1096/fasebj.11.1.9034168. [DOI] [PubMed] [Google Scholar]
- 28.Bruce WR, et al. Diet, aberrant crypt foci and colorectal cancer. Mutat Res. 1993;290:111–118. doi: 10.1016/0027-5107(93)90038-H. [DOI] [PubMed] [Google Scholar]
- 29.Buck AW. High fructose corn syrup food science and technology. New York: Marcel Dekker; 2001. pp. 391–412. [Google Scholar]
- 30.Bulut L, Kilic M. Kinetics of hydroxymethylfurfural accumulation and color change in honey during storage in relation to moisture content. J Food Process Preserv. 2009;33:22–32. doi: 10.1111/j.1745-4549.2008.00233.x. [DOI] [Google Scholar]
- 31.Burckhardt B, Burckhardt G. Transport of organic anions across the basolateral membrane of proximal tubule cells. In: Reviews of physiology, biochemistry and pharmacology. Berlin: Springer; 2003. pp. 95–158. [DOI] [PubMed] [Google Scholar]
- 32.Burdurlu HS, Karadeniz F. Effect of storage on nonenzymatic browning of apple juice concentrates. Food Chem. 2003;80:91–97. doi: 10.1016/S0308-8146(02)00245-5. [DOI] [Google Scholar]
- 33.Cais-Sokolińska D, Pikul J, Danków R. Measurement of colour parameters as index of the hidroxymethylfurfural content in the UHT sterilised milk during its storage. Electron J Pol Agric Univ. 2004;7:1–3. [Google Scholar]
- 34.Cantarelli M, Pellerano R, Marchevsky E, Camiña J. Quality of honey from Argentina: Study of chemical composittion and trace elements. J Argent Chem Soc. 2008;96:33–41. [Google Scholar]
- 35.Capuano E, Ferrigno A, Acampa I, Ait-Ameur L, Fogliano V. Characterization of the Maillard reaction in bread crisps. Eur Food Res Technol. 2008;228:311–319. doi: 10.1007/s00217-008-0936-5. [DOI] [Google Scholar]
- 36.Chefrour C, Draiaia R, Tahar A, Kaki YA, Bennadja S, Battesti M. Physicochemical characteristics and pollen spectrum of some north-east algerian honeys. Afr J Food Agric Nutr Dev. 2009;9:1276–1293. [Google Scholar]
- 37.Claeys WL, Van Loey AM, Hendrickx ME. Influence of seasonal variation on kinetics of time temperature integrators for thermally processed milk. J Dairy Res. 2003;70:217–225. doi: 10.1017/S0022029903006137. [DOI] [PubMed] [Google Scholar]
- 38.De la Iglesia F, Lázaro F, Puchades R, Maquieira A. Automatic determination of 5-hydroxymethylfurfural (5-HMF) by a flow injection method. Food Chem. 1997;60:245–250. doi: 10.1016/S0308-8146(96)00329-9. [DOI] [Google Scholar]
- 39.Ding X, Wang M-Y, Yao Y-X, Li G-Y, Cai B-C. Protective effect of 5-hydroxymethylfurfural derived from processed Fructus Corni on human hepatocyte LO2 injured by hydrogen peroxide and its mechanism. J Ethnopharmacol. 2010;128:373–376. doi: 10.1016/j.jep.2010.01.043. [DOI] [PubMed] [Google Scholar]
- 40.Dogan M, Sienkiewicz T, Oral RA. Hydroxymethylfurfural content of some commercial whey protein concentrates. Milchwissenschaft. 2005;60:309–311. [Google Scholar]
- 41.Doner LW. The sugars of honey—a review. J Sci Food Agric. 1977;28:443–456. doi: 10.1002/jsfa.2740280508. [DOI] [PubMed] [Google Scholar]
- 42.Durling LJ, Busk L, Hellman BE. Evaluation of the DNA damaging effect of the heat-induced food toxicant 5-hydroxymethylfurfural (HMF) in various cell lines with different activities of sulfotransferases. Food Chem Toxicol. 2009;47:880–884. doi: 10.1016/j.fct.2009.01.022. [DOI] [PubMed] [Google Scholar]
- 43.EFSA Opinion of the scientific panel on food additives, flavourings, processing aids and materials in contact with food (AFC) on a request from the commission related to flavouring group evaluation 13: furfuryl and furan derivatives with and without additional side-chain substituents and heteroatoms from chemical group 14. EFSA J. 2005;215:1–73. [Google Scholar]
- 44.Escriche I, Visquert M, Carot JM, Domenech E, Fito P. Effect of honey thermal conditions on hydroxymethylfurfural content prior to pasteurization. Food Sci Technol Int. 2008;14:29. doi: 10.1177/1082013208094580. [DOI] [Google Scholar]
- 45.Fallico B, Arena E, Zappala M. Roasting of hazelnuts. Role of oil in colour development and hydroxymethylfurfural formation. Food Chem. 2003;81:569–573. doi: 10.1016/S0308-8146(02)00497-1. [DOI] [Google Scholar]
- 46.Fallico B, Arena E, Zappala M. Degradation of 5-hydroxymethylfurfural in honey. J Food Sci. 2008;73:C625–C631. doi: 10.1111/j.1750-3841.2008.00946.x. [DOI] [PubMed] [Google Scholar]
- 47.Fallico B, Zappala M, Arena E, Verzera A. Effects of conditioning on HMF content in unifloral honeys. Food Chem. 2004;85:305–313. doi: 10.1016/j.foodchem.2003.07.010. [DOI] [Google Scholar]
- 48.Fallico B, Zappalà M, Arena E, Verzera A. Effects of heating process on chemical composition and HMF levels in Sicilian monofloral honeys. Food Chem. 2004;85:305–313. doi: 10.1016/j.foodchem.2003.07.010. [DOI] [Google Scholar]
- 49.Flink J, Karel M. Retention of organic volatiles in freeze-dried solutions of carbohydrates. J Agric Food Chem. 1970;18:295–297. doi: 10.1021/jf60168a014. [DOI] [Google Scholar]
- 50.Florian S, Bauer-Marinovic M, Taugner F, Dobbernack G, Monien BH, Meinl W, Glatt H. Study of 5-hydroxymethylfurfural and its metabolite 5-sulfooxymethylfurfural on induction of colonic aberrant crypt foci in wild-type mice and transgenic mice expressing human sulfotransferases 1A1 and 1A2. Mol Nutr Food Res. 2012;56:593–600. doi: 10.1002/mnfr.201100574. [DOI] [PubMed] [Google Scholar]
- 51.Florin I, Rutberg L, Curvall M, Enzell CR. Screening of tabacco smoke constituents for mutagenicity using the Ames’ test. Toxicology. 1980;15:219–232. doi: 10.1016/0300-483X(80)90055-4. [DOI] [PubMed] [Google Scholar]
- 52.Friedberg EC, Feaver WJ, Gerlach VL. The many faces of DNA polymerases: strategies for mutagenesis and for mutational avoidance. Proc Natl Acad Sci. 2000;97:5681–5683. doi: 10.1073/pnas.120152397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Fromowitz M, et al. Bone marrow genotoxicity of 2, 5-dimethylfuran, a green biofuel candidate. Environ Mol Mutagen. 2012;53:488–491. doi: 10.1002/em.21707. [DOI] [PubMed] [Google Scholar]
- 54.Gentry TS, Roberts JS. Formation kinetics and application of 5-hydroxymethylfurfural as a time–temperature indicator of lethality for continuous pasteurization of apple cider. Innov Food Sci Emerg Technol. 2004;5:327–333. doi: 10.1016/j.ifset.2004.03.005. [DOI] [Google Scholar]
- 55.Germond JE, Philippossian G, Richli U, Bracco I, Arnaud MJ. Rapid and complete urinary elimination of [14C]-5-hydroxymethyl-2-furaldehyde administered orally or intravenously to rats. J Toxicol Environ Health. 1987;22:79–89. doi: 10.1080/15287398709531052. [DOI] [PubMed] [Google Scholar]
- 56.Gheldof N, Wang X-H, Engeseth NJ. Identification and quantification of antioxidant components of honeys from various floral sources. J Agric Food Chem. 2002;50:5870–5877. doi: 10.1021/jf0256135. [DOI] [PubMed] [Google Scholar]
- 57.Gibbings S, et al. Xanthine oxidoreductase promotes the inflammatory state of mononuclear phagocytes through effects on chemokine expression, peroxisome proliferator-activated receptor-γ sumoylation, and HIF-1α. J Biol Chem. 2011;286:961–975. doi: 10.1074/jbc.M110.150847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Gidamis AB, Chove BE, Shayo NB, Nnko SA, Bangu NT. Quality evaluation of honey harvested from selected areas in Tanzania with special emphasis on hydroxymethyl furfural (HMF) levels. Plant Foods Hum Nutr. 2004;59:129–132. doi: 10.1007/s11130-004-0020-7. [DOI] [PubMed] [Google Scholar]
- 59.Glatt H, Schneider H, Liu Y. V79-hCYP2E1-hSULT1A1, a cell line for the sensitive detection of genotoxic effects induced by carbohydrate pyrolysis products and other food-borne chemicals. Mutat Res Genet Toxicol Environ Mutagen. 2005;580:41–52. doi: 10.1016/j.mrgentox.2004.11.005. [DOI] [PubMed] [Google Scholar]
- 60.Glatt H, Schneider H, Murkovic M, Monien BH, Meinl W. Hydroxymethyl-substituted furans: mutagenicity in Salmonella typhimurium strains engineered for expression of various human and rodent sulphotransferases. Mutagenesis. 2011;27:41–48. doi: 10.1093/mutage/ger054. [DOI] [PubMed] [Google Scholar]
- 61.Godfrey VB, Chen L-J, Griffin RJ, Lebetkin EH, Burka LT. Distribution and metabolism of (5-hydroxymethyl) furfural in male F344 rats and B6C3F1 mice after oral administration. J Toxicol Environ Health Part A. 1999;57:199–210. doi: 10.1080/009841099157764. [DOI] [PubMed] [Google Scholar]
- 62.Gökmen V, Açar ÖÇ, Köksel H, Acar J. Effects of dough formula and baking conditions on acrylamide and hydroxymethylfurfural formation in cookies. Food Chem. 2007;104:1136–1142. doi: 10.1016/j.foodchem.2007.01.008. [DOI] [Google Scholar]
- 63.Gökmen V, Açar ÖÇ, Serpen A, Morales FJ. Effect of leavening agents and sugars on the formation of hydroxymethylfurfural in cookies during baking. Eur Food Res Technol. 2008;226:1031–1037. doi: 10.1007/s00217-007-0628-6. [DOI] [Google Scholar]
- 64.Gökmen V, Şenyuva HZ. Effects of some cations on the formation of acrylamide and furfurals in glucose–asparagine model system. Eur Food Res Technol. 2006;225:815–820. doi: 10.1007/s00217-006-0486-7. [DOI] [Google Scholar]
- 65.Goldberg MA, Brugnara C, Dover GJ, Schapira L, Lacroix L, Bunn HF. Hydroxyurea and erythropoietin therapy in sickle cell anemia. Seminars in oncology. Amsterdam: Elsevier; 1992. pp. 74–81. [PubMed] [Google Scholar]
- 66.Gomes S, Dias LG, Moreira LL, Rodrigues P, Estevinho L. Physicochemical, microbiological and antimicrobial properties of commercial honeys from Portugal. Food Chem Toxicol. 2010;48:544–548. doi: 10.1016/j.fct.2009.11.029. [DOI] [PubMed] [Google Scholar]
- 67.Hardt-Stremayr M, Mattioli S, Greilberger J, Stiegler P, Matzi V, Schmid MG, Wintersteiger R. Determination of metabolites of 5-hydroxymethylfurfural in human urine after oral application. J Sep Sci. 2013;36:670–676. doi: 10.1002/jssc.201200768. [DOI] [PubMed] [Google Scholar]
- 68.Hayden MR, Tyagi SC. Uric acid: a new look at an old risk marker for cardiovascular disease, metabolic syndrome, and type 2 diabetes mellitus: The urate redox shuttle. Nutr Metabol. 2004;1:10. doi: 10.1186/1743-7075-1-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Høie AH, Svendsen C, Brunborg G, Glatt H, Alexander J, Meinl W, Husøy T. Genotoxicity of three food processing contaminants in transgenic mice expressing human sulfotransferases 1A1 and 1A2 as assessed by the in vivo alkaline single cell gel electrophoresis assay. Environ Mol Mutagen. 2015;56:709–714. doi: 10.1002/em.21963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Hossain SA, Pal PK, Sarkar PK, Patil GR. Sensory characteristics of dudh churpi in relation to its chemical composition. Zeitschrift für Lebensmitteluntersuchung und-Forschung A. 1999;208:178–182. doi: 10.1007/s002170050398. [DOI] [Google Scholar]
- 71.Huber GW, Iborra S, Corma A. Synthesis of transportation fuels from biomass: chemistry, catalysts, and engineering. Chem Rev. 2006;106:4044–4098. doi: 10.1021/cr068360d. [DOI] [PubMed] [Google Scholar]
- 72.Hunter JS. Determination of optimum operating conditions by experimental methods. Ind Qual Control. 1959;15:6–11. [Google Scholar]
- 73.Husøy T, et al. Dietary exposure to 5-hydroxymethylfurfural from Norwegian food and correlations with urine metabolites of short-term exposure. Food Chem Toxicol. 2008;46:3697–3702. doi: 10.1016/j.fct.2008.09.048. [DOI] [PubMed] [Google Scholar]
- 74.Iftikhar F, Masood MA, Waghchoure ES. Comparison of Apis cerana, Apis dorsata, Apis florea and Apis mellifera honey from different areas of Pakistan. Asian J Exp Biol Sci. 2011;2:399–403. [Google Scholar]
- 75.Iijima T. Mitochondrial membrane potential and ischemic neuronal death. Neurosci Res. 2006;55:234–243. doi: 10.1016/j.neures.2006.04.005. [DOI] [PubMed] [Google Scholar]
- 76.Islam A, Khalil I, Islam N, Moniruzzaman M, Mottalib A, Sulaiman SA, Gan SH. Physicochemical and antioxidant properties of Bangladeshi honeys stored for more than 1 year. BMC Complement Altern Med. 2012;12:177. doi: 10.1186/1472-6882-12-177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Islam M, Khalil M, Gan SH. Toxic compounds in honey. J Appl Toxicol. 2014;34:733–742. doi: 10.1002/jat.2952. [DOI] [PubMed] [Google Scholar]
- 78.Jachimowicz T, El Sherbiny G. Zur Problematik der Verwendung von Invertzucker für die. Bienenfütterung Apidologie. 1975;6:121–143. doi: 10.1051/apido:19750203. [DOI] [Google Scholar]
- 79.Janzowski C, Glaab V, Samimi E, Schlatter J, Eisenbrand G. 5-Hydroxymethylfurfural: assessment of mutagenicity, DNA-damaging potential and reactivity towards cellular glutathione. Food Chem Toxicol. 2000;38:801–809. doi: 10.1016/S0278-6915(00)00070-3. [DOI] [PubMed] [Google Scholar]
- 80.Kalabova K, Vorlova L, Borkovcova I, Smutna M, Vecerek V. Hydroxymethylfurfural in Czech honeys. Czech J Anim Sci. 2003;48:551–557. [Google Scholar]
- 81.Khalil M, Sulaiman S, Gan S. High 5-hydroxymethylfurfural concentrations are found in Malaysian honey samples stored for more than 1 year. Food Chem Toxicol. 2010;48:2388–2392. doi: 10.1016/j.fct.2010.05.076. [DOI] [PubMed] [Google Scholar]
- 82.Khalil MI, et al. Physicochemical and antioxidant properties of algerian honey. Molecules. 2012;17:11199–11215. doi: 10.3390/molecules170911199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Kim K, Kim Y, Kim HY, Ro JY, Jeoung D. Inhibitory mechanism of anti-allergic peptides in RBL2H3 cells. Eur J Pharmacol. 2008;581:191–203. doi: 10.1016/j.ejphar.2007.11.033. [DOI] [PubMed] [Google Scholar]
- 84.Kim Y, Lee YS, Hahn JH, Choe J, Kwon HJ, Ro JY, Jeoung D. Hyaluronic acid targets CD44 and inhibits FcɛRI signaling involving PKCδ, Rac1, ROS, and MAPK to exert anti-allergic effect. Mol Immunol. 2008;45:2537–2547. doi: 10.1016/j.molimm.2008.01.008. [DOI] [PubMed] [Google Scholar]
- 85.Kinati C, Tolemariam T, Debele K. Quality evaluation of honey produced in Gomma Woreda of South Western Ethiopia Livestock. Res Rural Dev. 2011;23:190. [Google Scholar]
- 86.Kitts DD, Chen X-M, Jing H. Demonstration of antioxidant and anti-inflammatory bioactivities from sugar–amino acid Maillard reaction products. J Agric Food Chem. 2012;60:6718–6727. doi: 10.1021/jf2044636. [DOI] [PubMed] [Google Scholar]
- 87.Kowalski S, Lukasiewicz M, Duda-Chodak A, Zięć G. 5-Hydroxymethyl-2-furfural (HMF)—heat-induced formation, occurrence in food and biotransformation–a review. Pol J Food Nutr Sci. 2013;63:207–225. [Google Scholar]
- 88.Krainer S, Brodschneider R, Vollmann J, Crailsheim K, Riessberger-Gallé U. Effect of hydroxymethylfurfural (HMF) on mortality of artificially reared honey bee larvae (Apis mellifera carnica) Ecotoxicology. 2016;25:320–328. doi: 10.1007/s10646-015-1590-x. [DOI] [PubMed] [Google Scholar]
- 89.Krauze A, Zalewski RI. Classification of honeys by principal component analysis on the basis of chemical and physical parameters Zeitschrift fuer Lebensmittel-Untersuchung und. Forschung. 1991;192:19–23. [Google Scholar]
- 90.Kroh LW. Caramelisation in food and beverages. Food Chem. 1994;51:373–379. doi: 10.1016/0308-8146(94)90188-0. [DOI] [Google Scholar]
- 91.Küçük M, Kolaylı S, Karaoğlu Ş, Ulusoy E, Baltacı C, Candan F. Biological activities and chemical composition of three honeys of different types from Anatolia. Food Chem. 2007;100:526–534. doi: 10.1016/j.foodchem.2005.10.010. [DOI] [Google Scholar]
- 92.Kuster B. 5-Hydroxymethylfurfural (HMF). A review focussing on its manufacture. Starch Stärke. 1990;42:314–321. doi: 10.1002/star.19900420808. [DOI] [Google Scholar]
- 93.Lee HS, Nagy S. Relative reactivities of sugars in the formation of 5-hydroxymethylfurfural in sugar-catalyst model systems1. J Food Process Preserv. 1990;14:171–178. doi: 10.1111/j.1745-4549.1990.tb00126.x. [DOI] [Google Scholar]
- 94.Lee Y-C, Shlyankevich M, Jeong H-K, Douglas JS, Surh Y-J. Bioactivation of 5-hydroxymethyl-2-furaldehyde to an electrophilic and mutagenic allylic sulfuric acid ester. Biochem Biophys Res Commun. 1995;209:996–1002. doi: 10.1006/bbrc.1995.1596. [DOI] [PubMed] [Google Scholar]
- 95.Li M-M, Wu L-Y, Zhao T, Wu K-W, Xiong L, Zhu L-L, Fan M. The protective role of 5-hydroxymethyl-2-furfural (5-HMF) against acute hypobaric hypoxia. Cell Stress Chaperones. 2011;16:529. doi: 10.1007/s12192-011-0264-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Li MM, et al. The protective role of 5-HMF against hypoxic injury. Cell Stress Chaperones. 2011;16:267–273. doi: 10.1007/s12192-010-0238-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Lin S-M, Wu J-Y, Su C, Ferng S, Lo C-Y, Chiou RY-Y. Identification and mode of action of 5-hydroxymethyl-2-furfural (5-HMF) and 1-methyl-1,2,3,4-tetrahydro-β-carboline-3-carboxylic acid (MTCA) as potent xanthine oxidase inhibitors in vinegars. J Agric Food Chem. 2012;60:9856–9862. doi: 10.1021/jf302711e. [DOI] [PubMed] [Google Scholar]
- 98.Li YX, Li Y, Qian ZJ, Kim MM, Kim SK. In vitro antioxidant activity of 5-HMF isolated from marine red alga Laurencia undulata in free-radical-mediated oxidative systems. J Microbiol Biotechnol. 2009;19:1319–1327. doi: 10.4014/jmb.0901.00004. [DOI] [PubMed] [Google Scholar]
- 99.Lorne E, et al. Role of extracellular superoxide in neutrophil activation: interactions between xanthine oxidase and TLR4 induce proinflammatory cytokine production. Am J Physiol Cell Physiol. 2008;294:C985–C993. doi: 10.1152/ajpcell.00454.2007. [DOI] [PubMed] [Google Scholar]
- 100.Magdoff-Fairchild B, Swerdlow PH, Bertles JF. Intermolecular organization of deoxygenated sickle haemoglobin determined by X-ray diffraction. Nature. 1972;239:217–219. doi: 10.1038/239217a0. [DOI] [PubMed] [Google Scholar]
- 101.Mańkowska D, Majak I, Bartos A, Słowianek M, Łącka A, Leszczyńska J (2017) 5-hydroxymethylfurfural content in selected gluten-and gluten-free cereal food products
- 102.Markowicz DB, Monaro E, Siguemoto E, Séfora M, Valdez B. Maillard reaction products in processed foods: pros and cons. In: Valdez B, editor. Food industrial processes-methods and equipment. 1. Rijeka: InTech; 2012. pp. 281–300. [Google Scholar]
- 103.Meda A, Lamien C, Millogo J, Romito M, Nacoulma O. Physiochemical Analyses of Burkina Fasan Honey. Acta Vet Brno. 2005;74:147–152. doi: 10.2754/avb200574010147. [DOI] [Google Scholar]
- 104.Mehryar L, Esmaiili M, Hassanzadeh A. Evaluation of some physicochemical and rheological properties of iranian honeys and the effect of temperature on its viscosity. Am Eurasian J Agric Environ Sci. 2013;13:807–819. [Google Scholar]
- 105.Mizushina Y, et al. 5-(Hydroxymethyl)-2-furfural: a selective inhibitor of DNA polymerase λ and terminal deoxynucleotidyltransferase. Arch Biochem Biophys. 2006;446:69–76. doi: 10.1016/j.abb.2005.11.015. [DOI] [PubMed] [Google Scholar]
- 106.Mondragón-Cortez P, Ulloa J, Rosas-Ulloa P, Rodríguez-Rodríguez R, Resendiz Vázquez J. Physicochemical characterization of honey from the West region of México. CyTA J Food. 2013;11:7–13. doi: 10.1080/19476337.2012.673175. [DOI] [Google Scholar]
- 107.Monien BH, Engst W, Barknowitz G, Seidel A, Glatt H. Mutagenicity of 5-hydroxymethylfurfural in V79 cells expressing human SULT1A1: identification and mass spectrometric quantification of DNA adducts formed. Chem Res Toxicol. 2012;25:1484–1492. doi: 10.1021/tx300150n. [DOI] [PubMed] [Google Scholar]
- 108.Moniruzzaman M, Khalil MI, Sulaiman SA, Gan SH. Physicochemical and antioxidant properties of Malaysian honeys produced by Apis cerana, Apis dorsata and Apis mellifera. BMC Complement Altern Med. 2013;13:43. doi: 10.1186/1472-6882-13-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Morales FJ. Hydroxymethylfurfural (HMF) and related compounds process-induced food toxicants: occurrence, formation, mitigation, and health risks. New Jersey: Wiley; 2008. pp. 135–174. [Google Scholar]
- 110.Morales FJ, Jiménez-Pérez S. Hydroxymethylfurfural determination in infant milk-based formulas by micellar electrokinetic capillary chromatography. Food Chem. 2001;72:525–531. doi: 10.1016/S0308-8146(00)00284-3. [DOI] [Google Scholar]
- 111.Mortas M, Gul O, Yazici F, Dervisoğlu M. Effect of brewing process and sugar content on 5-hydroxymethylfurfural and related substances from Turkish coffee. Int J Food Prop. 2017;20:1866–1875. doi: 10.1080/10942912.2016.1222587. [DOI] [Google Scholar]
- 112.Muli E, Munguti A, Raina S. Quality of honey harvested and processed using traditional methods in rural areas of Kenya. Acta Vet Brno. 2007;76:315–320. doi: 10.2754/avb200776020315. [DOI] [Google Scholar]
- 113.Murkovic M, Bornik MA. Formation of 5-hydroxymethyl-2-furfural (HMF) and 5-hydroxymethyl-2-furoic acid during roasting of coffee. Mol Nutr Food Res. 2007;51:390–394. doi: 10.1002/mnfr.200600251. [DOI] [PubMed] [Google Scholar]
- 114.Murkovic M, Pichler N. Analysis of 5-hydroxymethylfurfual in coffee, dried fruits and urine. Mol Nutr Food Res. 2006;50:842–846. doi: 10.1002/mnfr.200500262. [DOI] [PubMed] [Google Scholar]
- 115.Muruke MH. Assessment of quality of Tanzanian honey based on physicochemical properties. Food Sci Qual Manag. 2014;33:61–72. [Google Scholar]
- 116.Nalda MN, Yagüe JB, Calva JD, Gómez MM. Classifying honeys from the Soria Province of Spain via multivariate analysis. Anal Bioanal Chem. 2005;382:311–319. doi: 10.1007/s00216-005-3161-0. [DOI] [PubMed] [Google Scholar]
- 117.Neupane K, Thapa R. Alternative to off-season sugar supplement feeding of honeybees. J Inst Agric Anim Sci. 2005;26:77–81. [Google Scholar]
- 118.Nishi Y, Miyakawa Y, Kato K. Chromosome aberrations induced by pyrolysates of carbohydrates in Chinese hamster V79 cells. Mutat Res Lett. 1989;227:117–123. doi: 10.1016/0165-7992(89)90007-9. [DOI] [PubMed] [Google Scholar]
- 119.Ohmi K, Kiyokawa N, Takeda T, Fujimoto J. Human microvascular endothelial cells are strongly sensitive to Shiga toxins. Biochem Biophys Res Commun. 1998;251:137–141. doi: 10.1006/bbrc.1998.9417. [DOI] [PubMed] [Google Scholar]
- 120.Olga E, María F-G, Carmen SM. Differentiation of blossom honey and honeydew honey from Northwest Spain. Agriculture. 2012;2:25–37. doi: 10.3390/agriculture2010025. [DOI] [Google Scholar]
- 121.Ordóñez-Santos LE, Vázquez-Odériz L, Arbones-Maciñeira E, Romero-Rodríguez MÁ. The influence of storage time on micronutrients in bottled tomato pulp. Food Chem. 2009;112:146–149. doi: 10.1016/j.foodchem.2008.05.051. [DOI] [Google Scholar]
- 122.OumaáAnam O. Influence of metal ions on hydroxymethylfurfural formation in honey. In: Analytical proceedings including analytical communications. London: Royal Society of Chemistry; 1995. pp. 515–517. [Google Scholar]
- 123.Pacher P, Nivorozhkin A, Szabó C. Therapeutic effects of xanthine oxidase inhibitors: renaissance half a century after the discovery of allopurinol. Pharmacol Rev. 2006;58:87–114. doi: 10.1124/pr.58.1.6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Pandey V, Modak MJ. Biochemistry of terminal deoxynucleotidyltransferase (TdT): characterization and mechanism of inhibition of TdT by P1, P5-bis (5’-adenosyl) pentaphosphate. Biochemistry. 1987;26:2033–2038. doi: 10.1021/bi00381a036. [DOI] [PubMed] [Google Scholar]
- 125.Pastoriza de la Cueva S, Álvarez J, Végvári Á, Montilla-Gómez J, Cruz-López O, Delgado-Andrade C, Rufián-Henares JA. Relationship between HMF intake and SMF formation in vivo: an animal and human study. Mol Nutr Food Res. 2017;61:1600773. doi: 10.1002/mnfr.201600773. [DOI] [PubMed] [Google Scholar]
- 126.Pattinson KT, Sutherland AI, Smith TG, Dorrington KL, Wright AD. Acute mountain sickness, vitamin C, free radicals, and HIF-1α. Wilderness Environ Med. 2005;16:172–175. doi: 10.1580/1080-6032(2005)16[172:AMSVCF]2.0.CO;2. [DOI] [PubMed] [Google Scholar]
- 127.Paulsen JE, Løberg EM, Ølstørn HB, Knutsen H, Steffensen I-L, Alexander J. Flat dysplastic aberrant crypt foci are related to tumorigenesis in the colon of azoxymethane-treated rat. Can Res. 2005;65:121–129. [PubMed] [Google Scholar]
- 128.Polovková M, Šimko P. Determination and occurrence of 5-hydroxymethyl-2-furaldehyde in white and brown sugar by high performance liquid chromatography. Food Control. 2017;78:183–186. doi: 10.1016/j.foodcont.2017.02.059. [DOI] [Google Scholar]
- 129.Preston S, et al. The development of duodenal microadenomas in FAP patients: the human correlate of the Min mouse. J Pathol. 2008;214:294–301. doi: 10.1002/path.2294. [DOI] [PubMed] [Google Scholar]
- 130.Prior RL, Wu X, Gu L. Identification and urinary excretion of metabolites of 5-(hydroxymethyl)-2-furfural in human subjects following consumption of dried plums or dried plum juice. J Agric Food Chem. 2006;54:3744–3749. doi: 10.1021/jf0601113. [DOI] [PubMed] [Google Scholar]
- 131.Qamer S, Ahmad F, Latif F, Ali SS, Shakoori AR. Physicochemical analysis of Apis dorsata honey from terai forests, Nepal. Pak J Zool. 2008;40:53–58. [Google Scholar]
- 132.Quarta B, Anese M. Furfurals removal from roasted coffee powder by vacuum treatment. Food Chem. 2012;130:610–614. doi: 10.1016/j.foodchem.2011.07.083. [DOI] [Google Scholar]
- 133.Rada-Mendoza M, Sanz M, Olano A, Villamiel M. Formation of hydroxymethylfurfural and furosine during the storage of jams and fruit-based infant foods. Food Chem. 2004;85:605–609. doi: 10.1016/j.foodchem.2003.07.002. [DOI] [Google Scholar]
- 134.Rajchl A, Drgová L, Grégrová A, Čížková H, Ševčík R, Voldřich M. Rapid determination of 5-hydroxymethylfurfural by DART ionization with time-of-flight mass spectrometry. Anal Bioanal Chem. 2013;405:4737–4745. doi: 10.1007/s00216-013-6875-4. [DOI] [PubMed] [Google Scholar]
- 135.Ramadan K, Maga G, Shevelev IV, Villani G, Blanco L, Hübscher U. Human DNA polymerase λ possesses terminal deoxyribonucleotidyl transferase activity and can elongate RNA primers: implications for novel functions. J Mol Biol. 2003;328:63–72. doi: 10.1016/S0022-2836(03)00265-1. [DOI] [PubMed] [Google Scholar]
- 136.Ramadan K, Shevelev IV, Maga G, Hübscher U. De novo DNA synthesis by human DNA polymerase λ, DNA polymerase μ and terminal deoxyribonucleotidyl transferase. J Mol Biol. 2004;339:395–404. doi: 10.1016/j.jmb.2004.03.056. [DOI] [PubMed] [Google Scholar]
- 137.Ramirez-Jimenez A, Guerra-Hernández E, García-Villanova B. Browning indicators in bread. J Agric Food Chem. 2000;48:4176–4181. doi: 10.1021/jf9907687. [DOI] [PubMed] [Google Scholar]
- 138.Reyes-Salas EO, Manzanilla-Cano JA, Barceló-Quintal MH, Juárez-Mendoza D, Reyes-Salas M. Direct electrochemical determination of hydroxymethylfurfural (HMF) and its application to honey samples. Anal Lett. 2006;39:161–171. doi: 10.1080/00032710500423476. [DOI] [Google Scholar]
- 139.Risner CH, Kiser MJ, Dube MF. An aqueous high-performance liquid chromatographic procedure for the determination of 5-hydroxymethylfurfural in honey and other sugar-containing materials. J Food Sci. 2006;71:C179–C184. doi: 10.1111/j.1365-2621.2006.tb15614.x. [DOI] [Google Scholar]
- 140.Rizelio VM, Gonzaga LV, Borges GdSC, Micke GA, Fett R, Costa ACO. Development of a fast MECK method for determination of 5-HMF in honey samples. Food Chem. 2012;133:1640–1645. doi: 10.1016/j.foodchem.2011.11.058. [DOI] [Google Scholar]
- 141.Román-Leshkov Y, Chheda JN, Dumesic JA. Phase modifiers promote efficient production of hydroxymethylfurfural from fructose. Science. 2006;312:1933–1937. doi: 10.1126/science.1126337. [DOI] [PubMed] [Google Scholar]
- 142.Rosatella AA, Simeonov SP, Frade RF, Afonso CA. 5-Hydroxymethylfurfural (HMF) as a building block platform: biological properties, synthesis and synthetic applications. Green Chem. 2011;13:754–793. doi: 10.1039/c0gc00401d. [DOI] [Google Scholar]
- 143.Roychoudhury R, Jay E, Wu R. Terminal labeling and addition of homopolymer tracts to duplex DNA fragments by terminal deoxynudeotidyl transferase. Nucleic Acids Res. 1976;3:101–116. doi: 10.1093/nar/3.1.101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Rufian-Henares J, De la Cueva S. Assessment of hydroxymethylfurfural intake in the Spanish diet. Food Addit Contam. 2008;25:1306–1312. doi: 10.1080/02652030802163406. [DOI] [PubMed] [Google Scholar]
- 145.Rufián-Henares J, Delgado-Andrade C, Morales F. Analysis of heat-damage indices in breakfast cereals: influence of composition. J Cereal Sci. 2006;43:63–69. doi: 10.1016/j.jcs.2005.05.001. [DOI] [Google Scholar]
- 146.Rufián-Henares JA, García-Villanova B, Guerra-Hernández E. Occurrence of furosine and hydroxymethylfurfural as markers of thermal damage in dehydrated vegetables. Eur Food Res Technol. 2008;228:249–256. doi: 10.1007/s00217-008-0929-4. [DOI] [Google Scholar]
- 147.Ruiz-Matute A, Soria A, Sanz M, Martínez-Castro I. Characterization of traditional Spanish edible plant syrups based on carbohydrate GC–MS analysis. J Food Compos Anal. 2010;23:260–263. doi: 10.1016/j.jfca.2009.08.017. [DOI] [Google Scholar]
- 148.Ruoff K, et al. Quantitative determination of physical and chemical measurands in honey by near-infrared spectrometry. Eur Food Res Technol. 2007;225:415–423. doi: 10.1007/s00217-006-0432-8. [DOI] [Google Scholar]
- 149.Safo MK, Abdulmalik O, Danso-Danquah R, Burnett JC, Nokuri S, Joshi GS, Musayev FN, Asakura T, Abraham DJ. Structural basis for the potent antisickling effect of a novel class of five-membered heterocyclic aldehydic compounds. J Med Chem. 2004;47:4665–4676. doi: 10.1021/jm0498001. [DOI] [PubMed] [Google Scholar]
- 150.Sancho MT, Muniategui S, Huidobro JF, Simal Lozano J. Aging of honey. J Agric Food Chem. 1992;40:134–138. doi: 10.1021/jf00013a026. [DOI] [Google Scholar]
- 151.Sang N, Stiehl DP, Bohensky J, Leshchinsky I, Srinivas V, Caro J. MAPK signaling up-regulates the activity of hypoxia-inducible factors by its effects on p300. J Biol Chem. 2003;278:14013–14019. doi: 10.1074/jbc.M209702200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Sanna G, Pilo MI, Piu PC, Tapparo A, Seeber R. Determination of heavy metals in honey by anodic stripping voltammetry at microelectrodes. Anal Chim Acta. 2000;415:165–173. doi: 10.1016/S0003-2670(00)00864-3. [DOI] [Google Scholar]
- 153.Schoental R, Hard G, Gibbard S. Histopathology of renal lipomatous tumors in rats treated with the “natural” products, pyrrolizidine alkaloids and α, β-unsaturated aldehydes. J Natl Cancer Inst. 1971;47:1037–1044. [PubMed] [Google Scholar]
- 154.Schroeder J, Kagey-Sobotka A, Lichtenstein L. The role of the basophil in allergic inflammation. Allergy. 1995;50:463–472. doi: 10.1111/j.1398-9995.1995.tb01180.x. [DOI] [PubMed] [Google Scholar]
- 155.Schultheiss J, Jensen D, Galensa R. Hydroxymethylfurfural und furfural in kaffeeproben: HPLC-biosensor-kopplung mit supressionstechnik. Lebensmittelchemie. 1999;53:159. [Google Scholar]
- 156.Serpen A, Gökmen V, Mogol BA. Effects of different grain mixtures on Maillard reaction products and total antioxidant capacities of breads. J Food Compos Anal. 2012;26:160–168. doi: 10.1016/j.jfca.2012.02.001. [DOI] [Google Scholar]
- 157.Severin I, Dumont C, Jondeau-Cabaton A, Graillot V, Chagnon MC. Genotoxic activities of the food contaminant 5-hydroxymethylfurfural using different in vitro bioassays. Toxicol Lett. 2010;192:189–194. doi: 10.1016/j.toxlet.2009.10.022. [DOI] [PubMed] [Google Scholar]
- 158.Shinohara K, Kong ZL, Miwa M, Tsushida T, Kurogi M, Kitamura Y, Murakami H. Effect of mutagens on the viability and some enzymes of a serum-free cultured human histiocytic lymphoma cell line, U-937. Agric Biol Chem. 1990;54:373–380. [Google Scholar]
- 159.Sies H. Oxidative stress: introductory remarks. In: Sies H, editor. Oxidative stress. London: Academic Press; 1985. pp. 1–8. [Google Scholar]
- 160.Singh N, Bath PK. Quality evaluation of different types of Indian honey. Food Chem. 1997;58:129–133. doi: 10.1016/S0308-8146(96)00231-2. [DOI] [Google Scholar]
- 161.Singh N, Singh S, Bawa A, Sekhon K. Honey—its food uses. Indian Food Packer. 1988;42:15–25. [Google Scholar]
- 162.Sodré GdS, Marchini LC, Moreti ACdCC, Otsuk IP, Carvalho CALd. Physico–chemical characteristics of honey produced by Apis mellifera in the Picos region, state of Piauí. Brazil Revista Brasileira de Zootecnia. 2011;40:1837–1843. doi: 10.1590/S1516-35982011000800030. [DOI] [Google Scholar]
- 163.Solayman M, Islam M, Paul S, Ali Y, Khalil M, Alam N, Gan SH. Physicochemical properties, minerals, trace elements, and heavy metals in honey of different origins: a comprehensive review. Compr Rev Food Sci Food Saf. 2015;5:219–233. doi: 10.1111/1541-4337.12182. [DOI] [PubMed] [Google Scholar]
- 164.Spano N, et al. An RP-HPLC determination of 5-hydroxymethylfurfural in honey: the case of strawberry tree honey. Talanta. 2006;68:1390–1395. doi: 10.1016/j.talanta.2005.08.003. [DOI] [PubMed] [Google Scholar]
- 165.Stetson CA., Jr The state of hemoglobin in sickled erythrocytes. J Exp Med. 1966;123:341. doi: 10.1084/jem.123.2.341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Surh Y-J, Liem A, Miller JA, Tannenbaum SR. 5-Sulfooxymethylfurfural as a possible ultimate mutagenic and carcinogenic metabolite of the Maillard reaction product, 5-hydroxymethylfurfural. Carcinogenesis. 1994;15:2375–2377. doi: 10.1093/carcin/15.10.2375. [DOI] [PubMed] [Google Scholar]
- 167.Suzuki M, Nakamura T, Iyoki S, Fujiwara A, Watanabe Y, Mohri K, Isobe K, Ono K, Yano S. Elucidation of anti-allergic activities of curcumin-related compounds with a special reference to their anti-oxidative activities. Biol Pharm Bull. 2005;28:1438–1443. doi: 10.1248/bpb.28.1438. [DOI] [PubMed] [Google Scholar]
- 168.Svendsen C, HUSØY T, Glatt H, Paulsen JE, Alexander J. 5-Hydroxymethylfurfural and 5-sulfooxymethylfurfural increase adenoma and flat ACF number in the intestine of Min/+ mice. Anticancer Res. 2009;29:1921–1926. [PubMed] [Google Scholar]
- 169.Terrab A, González AG, Díez MJ, Heredia FJ. Characterisation of moroccan unifloral honeys using multivariate analysis. Eur Food Res Technol. 2003;218:88–95. doi: 10.1007/s00217-003-0797-x. [DOI] [Google Scholar]
- 170.Teixidó E, Santos F, Puignou L, Galceran MT. Analysis of 5-hydroxymethylfurfural in foods by gas chromatography–mass spectrometry. J Chromatogr A. 2006;1135:85–90. doi: 10.1016/j.chroma.2006.09.023. [DOI] [PubMed] [Google Scholar]
- 171.Theobald A, Müller A, Anklam E. Determination of 5-hydroxymethylfurfural in vinegar samples by HPLC. J Agric Food Chem. 1998;46:1850–1854. doi: 10.1021/jf970912t. [DOI] [Google Scholar]
- 172.Toker OS, Dogan M, Ersöz NB, Yilmaz MT. Optimization of the content of 5-hydroxymethylfurfural (HMF) formed in some molasses types: HPLC-DAD analysis to determine effect of different storage time and temperature levels. Ind Crops Prod. 2013;50:137–144. doi: 10.1016/j.indcrop.2013.05.030. [DOI] [Google Scholar]
- 173.Tucak Z, Periškić M, Škrivanko M, Konjarević A. The influence of the botanic origin of honey plants on the quality of honey. Agriculture. 2007;13:234–236. [Google Scholar]
- 174.Turhan I, Tetik N, Karhan M, Gurel F, Tavukcuoglu HR. Quality of honeys influenced by thermal treatment LWT-Food. Sci Technol. 2008;41:1396–1399. [Google Scholar]
- 175.Turhan K. Effects of thermal treatment and storage on hydroxymethylfurfural (HMF) content and diastase activity of honeys collected from middle Anatolia in Turkey. In: Innovations in chemical biology. Berlin: Springer; 2009. pp. 233–239. [Google Scholar]
- 176.Ulbricht RJ, Northup SJ, Thomas JA. A review of 5-hydroxymethylfurfural (HMF) in parenteral solutions. Toxicol Sci. 1984;4:843–853. doi: 10.1093/toxsci/4.5.843. [DOI] [PubMed] [Google Scholar]
- 177.Van der Zee R, Pisa L. Bijensterfte 2009–10 en toxische invertsuikersiroop. NCB Rapp. 2010;2:1–15. [Google Scholar]
- 178.Wang M-Y, et al. Investigation on the morphological protective effect of 5-hydroxymethylfurfural extracted from wine-processed Fructus corni on human L02 hepatocytes. J Ethnopharmacol. 2010;130:424–428. doi: 10.1016/j.jep.2010.05.024. [DOI] [PubMed] [Google Scholar]
- 179.White J, Doner LW. Honey composition and properties. Beekeep US Agric Handb. 1980;335:82–91. [Google Scholar]
- 180.White J., Jr Spectrophotometric method for hydroxymethylfurfural in honey. J Assoc Off Anal Chem. 1979;62:509–514. [PubMed] [Google Scholar]
- 181.White JW, Jr, Kushnir I. Composition of honey. VII. J Apic Res. 1967;6:163–178. doi: 10.1080/00218839.1967.11100177. [DOI] [Google Scholar]
- 182.White Júnior J. Honey. Adv Food Res. 1978;22:287–374. doi: 10.1016/S0065-2628(08)60160-3. [DOI] [PubMed] [Google Scholar]
- 183.White JW. Composition of honey. Honey: a comprehensive survey. London: Heinemann; 1975. pp. 157–206. [Google Scholar]
- 184.Winkler O. Beitrag zum Nachweis und zur Bestimmung von Oxymethylfurfurol in Honig und Kunsthonig. Zeitschrift für Lebensmitteluntersuchung und-Forschung A. 1955;102:161–167. doi: 10.1007/BF01683776. [DOI] [Google Scholar]
- 185.Wootton M, Ryall L. A comparison of Codex Alimentarius Commission and HPLC methods for 5-hydroxymethyl-2-furaldehyde determination in honey. J Apic Res. 1985;24:120–124. doi: 10.1080/00218839.1985.11100659. [DOI] [Google Scholar]
- 186.Xiao PG, Xing ST, Wang LW. Immunological aspects of Chinese medicinal plants as antiageing drugs. J Ethnopharmacol. 1993;38:159–165. doi: 10.1016/0378-8741(93)90012-T. [DOI] [PubMed] [Google Scholar]
- 187.Yamada P, Nemoto M, Shigemori H, Yokota S, Isoda H. Isolation of 5-(hydroxymethyl) furfural from lycium chinense and its inhibitory effect on the chemical mediator release by basophilic cells. Planta Med. 2011;77:434–440. doi: 10.1055/s-0030-1250402. [DOI] [PubMed] [Google Scholar]
- 188.Yilmaz H, Küfrevioğlu Öİ. Composition of honeys collected from Eastern and South-Eastern Anatolia and effect of storage on hydroxymethylfurfural content and diastase activity. Turk J Agric For. 2001;25:347–349. [Google Scholar]
- 189.Yuan J-P, Chen F. Separation and identification of furanic compounds in fruit juices and drinks by high-performance liquid chromatography photodiode array detection. J Agric Food Chem. 1998;46:1286–1291. doi: 10.1021/jf970894f. [DOI] [Google Scholar]
- 190.Zaitzev AN, Simonian TA, Pozdniakov AL. Hygienic standards for hydroxymethylfurfural in food products. Vopr Pitan. 1975;1:52–55. [PubMed] [Google Scholar]
- 191.Zappala M, Fallico B, Arena E, Verzera A. Methods for the determination of HMF in honey: a comparison. Food Control. 2005;16:273–277. doi: 10.1016/j.foodcont.2004.03.006. [DOI] [Google Scholar]
- 192.Zhang X-M, Chan CC, Stamp D, Minkin S, Archer MC, Bruce WR. Initiation and promotion of colonic aberrant crypt foci in rats by 5-hydroxymethy1-2-furaldehyde in thermolyzed sucrose. Carcinogenesis. 1993;14:773–775. doi: 10.1093/carcin/14.4.773. [DOI] [PubMed] [Google Scholar]
- 193.Zhao L, et al. In vitro antioxidant and antiproliferative activities of 5-hydroxymethylfurfural. J Agric Food Chem. 2013;61:10604–10611. doi: 10.1021/jf403098y. [DOI] [PubMed] [Google Scholar]
- 194.Zhao L, Su J, Li L, Chen J, Hu S, Zhang X, Chen T. Mechanistic elucidation of apoptosis and cell cycle arrest induced by 5-hydroxymethylfurfural, the important role of ROS-mediated signaling pathways. Food Res Int. 2014;66:186–196. doi: 10.1016/j.foodres.2014.08.051. [DOI] [Google Scholar]
- 195.Zhao Y, Flugstad B, Kolbe E, Park JW, Wells JH. Using capacitive (radio frequency) dielectric heating in food processing and preservation–a review. J Food Process Eng. 2000;23:25–55. doi: 10.1111/j.1745-4530.2000.tb00502.x. [DOI] [Google Scholar]


