This cohort study assesses the association between proximity to parental symptom onset and amyloid-β burden in asymptomatic individuals with a parental history of sporadic Alzheimer disease.
Key Points
Question
Can proximity to parental symptom onset be used to help estimate amyloid-β burden in preclinical sporadic Alzheimer disease?
Findings
In this study of 101 cognitively normal individuals with a parental history of sporadic Alzheimer disease, amyloid-β burden increased as individuals approached their parent’s age at symptom onset.
Meaning
This study indicates that, as in autosomal dominant Alzheimer disease, an individual’s proximity to parent’s age at symptom onset may help estimate the advancement of preclinical disease.
Abstract
Importance
Alzheimer disease (AD) develops during several decades. Presymptomatic individuals might be the best candidates for clinical trials, but their identification is challenging because they have no symptoms.
Objective
To assess whether a sporadic parental estimated years to symptom onset calculation could be used to identify information about amyloid-β (Aβ) levels in asymptomatic individuals with a parental history of AD dementia.
Design, Setting, and Participants
This cohort study analyzed Aβ1-42 in cerebrospinal fluid (CSF) specimens from 101 cognitively normal individuals who had a lumbar puncture as part of the Presymptomatic Evaluation of Novel or Experimental Treatments for Alzheimer Disease (PREVENT-AD) cohort from September 1, 2011, through November 30, 2016 (374 participants were enrolled in the cohort during this period). The study estimated each participant’s proximity to his/her parent’s symptom onset by subtracting the index relative’s onset age from his/her current age. The association between proximity to parental symptom onset and Aβ levels was then assessed using apolipoprotein E ε4 (APOE4) status and sex as interactive terms. These analyses were performed again in 2 independent cohorts using CSF and Pittsburgh compound B carbon 11–labeled positron emission tomography (PIB-PET) Aβ biomarkers: the Adult Children Study (ACS) and the Wisconsin Registry for Alzheimer Prevention (WRAP) cohorts.
Main Outcomes and Measures
The association between proximity to parental symptom onset and Aβ burden in asymptomatic individuals with a parental history of sporadic AD.
Results
The present analysis included a subset of 101 PREVENT-AD individuals (mean [SD] age, 61.8 [5.1] years; 30 [29.7%] male), 128 ACS participants (112 participants underwent CSF measurement: mean [SD] age, 63.4 [5.1] years; 31 [27.7%] male; and 107 underwent PIB-PET: mean [SD] age, 64.6 [5.3] years; 27 [25.2%] male), and 135 WRAP participants (85 participants underwent CSF measurement: mean [SD] age, 59.9 [6.0] years; 27 [31.8%] male; and 135 underwent PIB-PET: mean [SD] age, 59.6 [6.1] years; 43 [31.9%] male). In the PREVENT-AD cohort, individuals approaching their parent’s onset age had lower CSF Aβ1-42 levels (range, 402-1597; B = −9.09, P = .04). This association was stronger in APOE4 carriers (B = −17.9, P = .03) and women (B = −19.8, P = .02). In the ACS cohort, the main association was replicated using PIB-PET data, and the sex interaction was replicated using CSF and PIB-PET data. In the WRAP cohort, the results were not replicated using cross-sectional data, but the main association and the APOE interaction were replicated using PIB-PET longitudinal data.
Conclusions and Relevance
These results suggest that proximity to parental symptom onset may help estimate Aβ biomarker changes in women or APOE4 carrier asymptomatic individuals with a parental history of sporadic AD.
Introduction
No treatment currently exists to stop or prevent Alzheimer disease (AD). The best time window to prevent AD is likely when individuals are still asymptomatic, before extensive neuronal degeneration has occurred. Identifying asymptomatic individuals is challenging and expensive, posing significant difficulties for the current generation of clinical trials.
In autosomal dominant AD (ADAD), symptom onset is determinable across generations. It is therefore possible to calculate the estimated years to symptom onset (EYO) score in individuals with ADAD by subtracting their parent’s onset age from their current age.1 With this approach, ADAD findings suggest that a decrease in cerebrospinal fluid (CSF) amyloid-β1-42 (Aβ1-42) concentrations can be detected 25 years before expected symptom onset and brain Aβ aggregation can be detected 15 years before expected symptom onset.1 The EYO score can therefore be used in ADAD to estimate AD biomarker abnormalities and potentially guide the optimal timing of treatments.
The heritability of sporadic AD dementia is estimated at approximately 70%,2,3 with age at onset heritability being 67% to 87% in early-onset AD.3 Whether the parent’s age at onset can help determine biomarker abnormalities in AD is not known. The aim of the current study was to test whether a sporadic parental EYO calculation could be used to identify information about Aβ levels in asymptomatic individuals with a parental history of AD dementia. Given the heterogeneity of AD and because apolipoprotein E ε4 (APOE4)4,5 and female sex6,7 increase the risk of AD, we further assessed whether the association between sporadic parental EYO score and Aβ levels is influenced by these risk factors. The current study was performed using CSF Aβ1-42 data from the Presymptomatic Evaluation of Novel or Experimental Treatments for Alzheimer Disease (PREVENT-AD) cohort.8 We repeated the analysis in the Adult Children Study (ACS)9,10 and the Wisconsin Registry for Alzheimer Prevention (WRAP) cohort11 using CSF and Pittsburgh compound B carbon 11–labeled positron emission tomography (PIB-PET) Aβ biomarkers.
Methods
Participants and Study Design
All individuals included in the current study had a parental history of AD and Aβ quantification (CSF and/or PIB-PET). The Table gives the participants’ characteristics. All specified procedures were approved by the local ethics committees of each respective cohort (institutional review board of McGill University Faculty of Medicine, Washington University Human Research Protection Office, and University of Wisconsin Institutional Review Board), and all participants provided signed informed consent forms before participation. Data were deidentified.
Table. Demographic Characteristics of the Study Participantsa.
| Characteristic | PREVENT-AD (n = 101) | ACS (n = 128) | WRAP (n = 135) | Main Effects P Value |
|---|---|---|---|---|
| Age, y | ||||
| CSF | 61.8 (5.1) [55 to 78] | 63.4 (5.1) [55 to 76] | 59.9 (6.0) [48 to 70] | <.001b,c,d |
| PIB-PET | NA | 64.6 (5.3) [55 to 76] | 59.6 (6.1) [46 to 71] | <.001 |
| Male, No. (%) | ||||
| CSF | 30 (29.7) | 31 (27.7)b | 27 (31.8) | .82 |
| PIB-PET | NA | 27 (25.2)c | 43 (31.9) | .26 |
| Educational level | ||||
| CSF | 15.0 (3.0) [10 to 27] | 15.9 (2.3) [11 to 20] | 16.5 (2.5) [12 to 25] | .002b,c |
| PIB-PET | NA | 16.0 (2.2) [11 to 20] | 16.3 (2.6) [12 to 25] | .32 |
| APOE4, No. (%) | ||||
| CSF | 39 (38.6) | 49 (43.8)b | 36 (42.4) | .70 |
| PIB-PET | NA | 50 (46.7)c | 63 (46.7) | .99 |
| CDR of 0, No. (%) | ||||
| CSF | 101 (100) | 128 (100) | 49 (71.0)e | NA |
| PIB-PET | NA | 100 | 82 (75.3)f | NA |
| MMSE score | ||||
| CSF | NA | 29.0 (1.3) [24 to 30] | 29.2 (1.2) [24 to 30] | .30 |
| PIB-PET | NA | 29.0 (1.3) [24 to 30] | 29.2 (1.2) [23 to 30] | .25 |
| MOCA for the CSF group | 27.9 (1.5) [24 to 30] | NA | NA | NA |
| Age of parent at disease onset, y | ||||
| CSF | 74.3 (7.4) [55 to 90] | 73.6 (7.6) [51 to 93] | 73.8 (8.0) [55 to 91] | .80 |
| PIB-PET | NA | 74.1 (7.6) [51 to 93] | 73.2 (8.3) [45 to 91] | .41 |
| Sporadic parental EYO scoreg | ||||
| CSF | −12.5 (6.7) [−29 to 7] | −10.2 (8.7) [−33 to 13] | −13.9 (7.4) [−28 to 6] | .004b,d |
| PIB-PET | NA | −9.5 (9.2) [−36 to 13] | −13.6 (8.0) [−34 to 13] | <.001d |
| CSF Aβ1-42 | 1056 (284) [402 to 1597] | 665 (252) [202 to 1615] | 750 (235) [280 to 1763] | NAh |
| PIB-PET indexi | NA | 0.28 (0.34) [0.02 to 1.57] | 1.18 (–0.18) [1.00 to 2.07] | NAh |
| PIB-PET rate of change | NA | 0.033 (0.041) [−0.03 to 0.16] | 0.005 (0.029) [−0.07 to 0.10] | NAh |
Abbreviations: Aβ, amyloid-β; ACS, Adult Children Study; APOE, apolipoprotein E; CDR, Clinical Dementia Rating; CSF, cerebrospinal fluid; EYO, estimated years to symptom onset; MMSE, Mini-Mental State Examination; MOCA, Montreal Cognitive Assessment; NA, not applicable; PIB-PET, Pittsburgh compound B carbon 11–labeled positron emission tomography; PREVENT-AD, Presymptomatic Evaluation of Novel or Experimental Treatments for Alzheimer Disease; WRAP, Wisconsin Registry for Alzheimer Prevention.
Data are expressed as mean (SD) [range] unless otherwise indicated. In the ACS cohort, n = 112 in the CSF group and n = 107 in the PIB-PET group. In the WRAP cohort, n = 85 in the CSF group and n = 135 in the PIB-PET group.
Difference between PREVENT-AD and ACS is significant.
Difference between PREVENT-AD and WRAP is significant.
Difference between ACS and WRAP is significant.
Information missing for 16 individuals.
Information missing for 26 individuals.
Calculated as the age of the participant at assessment minus the age of the parent at symptom onset.
Distribution of Aβ values (CSF and PIB-PET) cannot be statistically compared among the 3 cohorts because of differences in quantification methods.
Mean cortical potential binding was computed for the ACS cohort, whereas distributed volume ratios were computed for the WRAP cohort (eMethods in the Supplement).
PREVENT-AD Cohort
Recruitment for the PREVENT-AD cohort was begun in 2011 with enrollment of individuals from the greater Montreal metropolitan area (details in the eMethods in the Supplement). The PREVENT-AD study participants must satisfy the following conditions: (1) a family history of AD-like dementia, (2) 60 years or older at study entry or 55 to 59 but less than 15 years from their siblings’ or parents’ age at symptom onset, and (3) normal cognition and no other major neurologic disease.8 Data from 374 individuals were collected from September 1, 2011, through November 30, 2016, and archived in the PREVENT-AD internal data release 3.0. The present analysis included a subset of 101 PREVENT-AD individuals with a parental history of AD who underwent lumbar puncture (LP) to assess CSF Aβ1-42 concentration. Four individuals included in the current study were suspected to have mild cognitive impairment based on an extensive neuropsychological battery. All 101 individuals had a Clinical Dementia Rating (CDR) of 012 and a score above 23 on the Montreal Cognitive Assessment.13
To assess the robustness of our results, we conducted similar analyses in 2 independent cohorts of individuals with a parental history of AD: the ACS9,10 and WRAP.11 Because these 2 studies included PIB-PET and CSF Aβ1-42 assessments, we included the participants who had either or both of these assessments. Longitudinal PIB-PET data were available for both cohorts; thus, we also explored the association between sporadic parental EYO score and annual rate of change in PIB-PET indexes.
ACS Cohort
The ACS is a longitudinal study enrolling cognitively normal individuals that was conducted at the Knight Alzheimer Disease Research Center at Washington University School of Medicine in St Louis, Missouri.9,10 The ACS eligibility criteria include age at entry between 45 and 75 years and normal cognition, defined as a CDR of 0. A total of 128 participants 55 years and older were included in the study, among whom 112 underwent LP and 107 underwent PIB-PET. Fifty-nine individuals underwent PIB-PET a second time. The mean (SD) delay between the 2 scans was 3 years (mean [SD], 36.1 [17.3] months; median, 36 months; range, 13.0-106.3 months).
WRAP Cohort
WRAP is a longitudinal study conducted by the Wisconsin Alzheimer Institute at the University of Wisconsin School of Medicine and Public Health that enrolls individuals free of dementia.11,14 Participants are aged 40 to 65 years at entry and were free of dementia based on a neuropsychological evaluation with expert review. The cohort therefore included cognitively normal individuals (CDR, 0) and individuals with mild cognitive impairment (CDR, 0.5). A total of 135 participants 48 years and older were included in the study. The PIB-PET scans were available for all participants. Among them, 85 also underwent LP. An overlapping 92 participants had undergone PIB-PET a second time after an interval of approximately 2 years (mean [SD], 26.7 [3.5] months; median, 26.2 months; range, 20.5-40.7 months).
Aβ Assessments
The CSF from the PREVENT-AD cohort was collected by LP in the morning under fasting conditions. Measurement of Aβ1-42 was determined by enzyme-linked immunosorbent assay (INNOTEST; Fujirebio, formerly Innogenetics). In the ACS and WRAP cohorts, the CSF samples were acquired and assayed using previously published procedures (details are provided in the eMethods in the Supplement).15,16
In the ACS and WRAP cohorts, PIB-PET acquisition and processing were performed using in-house procedures that have been described previously.9,16 Accordingly, mean cortical binding potential values corrected for regional spread function are presented for the ACS cohort, and distribution to volume ratio data are presented for the WRAP cohort (eMethods in the Supplement).
Additional analyses were performed to assess whether proximity to parental onset was associated with the annual rate of brain Aβ accumulation in the ACS (n = 59) and WRAP (n = 92) cohorts. To do so, the PIB-PET annual rate of change was calculated as follows: amyloid scores at follow-up minus amyloid scores at baseline divided by the interval (in years) between the 2 scans.
Sporadic Parental EYO Score Calculation
The sporadic parental EYO score was calculated as the age of the participant at assessment minus the age of the parent at symptom onset.1 If an individual had 2 parents with a history of AD dementia, the age of the parent with the earliest onset was used to calculate the sporadic parental EYO score. The age of the parent at symptom onset corresponds to the age at which the family observed significant cognitive or memory changes in the PREVENT-AD and WRAP cohorts, whereas it corresponds to the age at which dementia symptoms began in the ACS cohort (eMethods in the Supplement).
APOE Genotype
APOE genotype in the PREVENT-AD cohort has previously been described (eMethods in Supplement).17 For statistical analysis, participants were classified as APOE4 carriers (1 or 2 ε4 alleles) or noncarriers (no ε4 allele). A similar approach was used in the 2 other cohorts.
Statistical Analysis
We examined differences among the 3 cohorts using linear regression analyses for continuous variables and Pearson χ2 tests for categorical variables. In the PREVENT-AD cohort, independent linear regression analyses were first used to determine CSF Aβ1-42 level (dependent variable) by age while controlling for sex and educational level and CSF Aβ1-42 level (dependent variable) by parent’s age at symptom onset while controlling for sex and educational level. A step-by-step hierarchical linear regression analysis was then used to determine CSF Aβ1-42 level by age while controlling for sex and educational level (model A, step 1, same as previous analysis); CSF Aβ1-42 level by sporadic parental EYO score while controlling for age, sex, and educational level (model A, step 2); and the interaction between sporadic parental EYO score and sex (sporadic parental EYO score × sex) on CSF Aβ1-42 level while including age, sex, educational level, and sporadic parental EYO score as covariates (model A, step 3). A similar hierarchical analysis (model B) was used to assess the interaction between sporadic parental EYO score and APOE (sporadic parental EYO score × APOE status). APOE status was included in this second and independent model. Exploratory analyses also assessed parental sex as a possible interactive term.
Models A and B were replicated in the 2 validation cohorts (ACS and WRAP) using baseline CSF Aβ1-42 values and PIB-PET values. Annual rate of change in PIB-PET binding was also investigated in the ACS and WRAP cohorts. One ACS participant and 1 WRAP participant with high baseline PIB-PET uptake were removed from the longitudinal analysis because they had a decrease in PIB-PET binding over time, which suggests that they could no longer accumulate additional brain Aβ.18,19
All analyses were completed using SPSS software, version 20 (IBM Inc). A 2-sided P < .05 was considered to be significant.
Results
The present analysis included a subset of 101 PREVENT-AD individuals (mean [SD] age, 61.8 [5.1] years; 30 [29.7%] male), 128 ACS participants (112 participants underwent CSF measurement: mean [SD] age, 63.4 [5.1] years; 31 [27.7%] male; and 107 underwent PIB-PET: mean [SD] age, 64.6 [5.3] years; 27 [25.2%] male), and 135 WRAP participants (85 participants underwent CSF measurement: mean [SD] age, 59.9 [6.0] years; 27 [31.8%] male; and 135 underwent PIB-PET: mean [SD] age, 59.6 [6.1] years; 43 [31.9%] male).
Association Between Sporadic Parental EYO Score and Aβ Burden
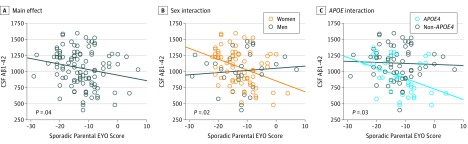
Cross-sectional analyses from the PREVENT-AD cohort demonstrated no association between age and CSF Aβ1-42 level or between parent’s age at symptom onset and CSF Aβ1-42 level but found a reduction in CSF Aβ1-42 levels as participants approached the parent’s age at onset (eFigure in the Supplement). Adding sporadic parental EYO score as a covariate into the model that already included age increased the explained variance (step 1 with age: r2 = 0.034, F change = 1.135; step 2 with age and sporadic parental EYO score: r2 = 0.077, F change = 4.441; difference between step 1 and step 2: P = .04; effect of sporadic parental EYO: B = −9.09, P = .04). An interaction was found between sporadic parental EYO score and sex (B = −19.8, P = .02), suggesting that the association between sporadic parental EYO score and Aβ1-42 level was stronger among women than among men (Figure 1 and eTable 1 in the Supplement). An interaction was also found between sporadic parental EYO score and APOE status (B = −17.9, P = .03), revealing a stronger association in APOE4 carriers than in noncarriers.
Figure 1. Proximity to Parental Symptom Onset and Amyloid-β (Aβ) Burden in the Presymptomatic Evaluation of Novel or Experimental Treatments for Alzheimer Disease (PREVENT-AD) Cohort.
P values were obtained with linear regression models controlled for age, sex, and educational level. Analyses that include the sporadic parental estimated years to symptom onset (EYO) score (calculated as the age of the participant at assessment minus the age of the parent at symptom onset) × APOE status interaction were also controlled for the main effect of APOE status. Unadjusted (raw) data are plotted. APOE4 indicates apolipoprotein E ɛ4; CSF, cerebrospinal fluid.
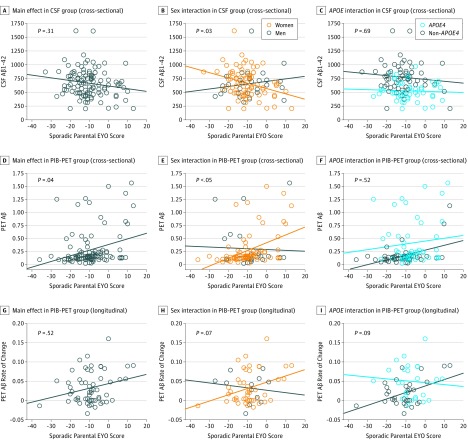
In the ACS cohort, age was associated with CSF Aβ1-42 and PIB-PET data, but these associations were no longer significant when sporadic parental EYO score was included in the models (eTable 2 in the Supplement). The association between sporadic parental EYO score and Aβ burden was replicated in the members of the ACS cohort who underwent PIB-PET, with an increase of brain Aβ deposition as individuals approached their parent’s age at symptom onset (Figure 2 and eTable 2 in the Supplement). The interaction between sporadic parental EYO score and sex was also replicated using CSF Aβ1-42 and PIB-PET data. In addition, longitudinal ACS PIB-PET data further suggest that women tend to accumulate brain Aβ at a faster rate than men as they approach the age at their parent’s onset. The sporadic parental EYO score × APOE interaction was not replicated in the ACS cohort using cross-sectional or longitudinal data. No other association reached significance.
Figure 2. Proximity to Parental Symptom Onset and Amyloid-β (Aβ) Burden in the Adult Children Study (ACS) Cohort .
P values were obtained with linear regression models controlled for age, sex, and educational level. Analyses that include the sporadic parental estimated years to symptom onset (EYO) score (calculated as the age of the participant at assessment minus the age of the parent at symptom onset) × APOE status interaction were also controlled for the main effect of APOE status. Unadjusted (raw) data are plotted. APOE4 indicates apolipoprotein E ɛ4; CSF, cerebrospinal fluid; and PIB-PET, Pittsburgh compound B carbon 11–labeled positron emission tomography.
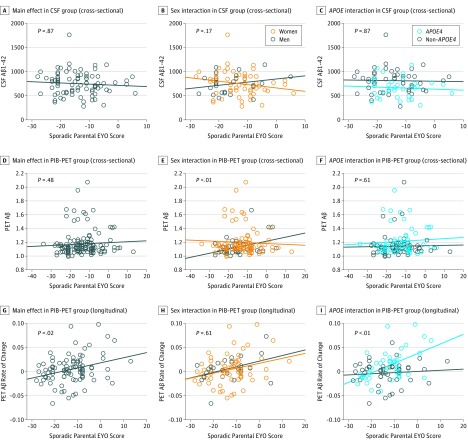
In the WRAP cohort, age alone was associated with cross-sectional CSF Aβ1-42 and PIB-PET data, and the association between age and PIB-PET was still significant when including sporadic parental EYO score in the model (eTable 3 in the Supplement). We did not replicate the PREVENT-AD cross-sectional findings (Figure 3 and eTable 3 in the Supplement). However, as individuals approached their parent’s age at symptom onset, they demonstrated a faster rate of brain Aβ accumulation (longitudinal data). The sporadic parental EYO score × APOE interaction further suggests that this association was stronger in APOE4 carriers than in APOE4 noncarriers. We did not find a sporadic parental EYO score × sex interaction for the rate of brain Aβ accumulation. Significant results from the main analyses are summarized in Figure 4 and eTable 4 in the Supplement.
Figure 3. Proximity to Parental Symptom Onset and Amyloid-β (Aβ) Burden in the Wisconsin Registry for Alzheimer Prevention (WRAP) Cohort .
P values were obtained with linear regression models controlled for age, sex, and educational level. Analyses that include the sporadic parental estimated years to symptom onset (EYO) score (calculated as the age of the participant at assessment minus the age of the parent at symptom onset) score × APOE status interaction were also controlled for the main effect of APOE status. Unadjusted (raw) data are plotted. APOE4 indicates apolipoprotein E ɛ4; CSF, cerebrospinal fluid; and PET, positron emission tomography.
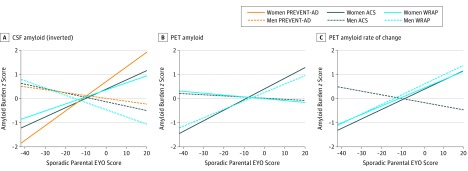
Figure 4. Normalized Amyloid-β (Aβ) Burden Levels as a Function of Sporadic Parental Estimated Years to Symptom Onset (EYO) Score in Men and Women in the Presymptomatic Evaluation of Novel or Experimental Treatments for Alzheimer Disease (PREVENT-AD), Adult Children Study (ACS), Wisconsin Registry for Alzheimer Prevention (WRAP) Cohorts .
Regression lines were obtained by calculating z scores in each cohort and Aβ measurement. Values were inverted for cerebrospinal fluid (CSF) so that higher z scores correspond to an increased level of pathologic findings PET indicates positron emission tomography.
Exploratory Analysis on Parental Sex
The association between Aβ accumulation and sporadic parental EYO score was not associated with parental sex in any of the cohorts (B = −10.7, P = 0.45 for the PREVENT-AD; B = 4.2, P = .49 for the ACS individuals with CSF; B = 0.01, P = .41 for the ACS individuals with PIB-PET; B = −0.001, P = .49 for the ACS individuals with longitudinal PIB-PET; B = 13.2, P = .13 for the WRAP individuals with CSF; B = −0.002, P = .62 for the WRAP individuals with PIB-PET; B = 0.0003, P = .78 for the WRAP individuals with longitudinal PIB-PET).
Discussion
Disappointing outcomes from clinical trials performed in individuals with AD dementia and increasing experimental research performed in the preclinical phase of AD suggest that asymptomatic individuals in the preclinical stages of AD might be the best candidates for disease modification therapies. An important challenge facing AD prevention trials is the identification of asymptomatic individuals who could benefit from such therapies.
Our results suggest that proximity to parental symptom onset (or sporadic parental EYO score)2 might help estimate Aβ biomarker advancement in individuals with a parental history of AD. Specifically, asymptomatic individuals with such a family history were more likely to demonstrate abnormal CSF (PREVENT-AD findings) (Figure 1) and brain (ACS findings) (Figure 2) Aβ biomarkers as they approached their parent’s age at onset irrespective of their own age. Individuals who were closer to their parent’s age at onset also accumulated brain Aβ deposition at a faster rate (WRAP findings) (Figure 3). Of importance, this association seemed to be independent of age-related changes in Aβ burden. Because Aβ burden is known to increase with age20 and sporadic parental EYO score is based (in part) on individuals’ age, it is reasonable to hypothesize that age itself could have driven our results. However, in the PREVENT-AD, we did not find any association between CSF Aβ1-42 and age, and adjusting for age did not alter the significant association between sporadic parental EYO score and CSF Aβ1-42 (model A, step 1) (eTable 1 in the Supplement). Thus, in our sample, the sporadic parental EYO score appeared to be a better indicator of Aβ variability than age. In addition, although increasing age was associated with increasing Aβ burden in the 2 replication cohorts, this association with sporadic parental EYO score was still present after age adjustment (eTable 2 and eTable 3 in the Supplement). The relatively low mean age of the 3 cohorts (approximately 62 years) might partially explain this age-independent finding. The sporadic parental EYO score may be a better predictor of Aβ burden in late middle-age individuals, who are less likely to have age-related Aβ, compared with elderly individuals, who have a higher prevalence of Aβ deposition.20
Not all individuals with a parental history of AD will develop the disease within their lifespan; thus, we assessed whether the association between sporadic parental EYO score and Aβ pathologic findings is influenced by sex or APOE4 status, 2 important risk factors for AD.4,5,6,7 Of interest, findings from the PREVENT-AD cohort indicated that asymptomatic women had a stronger decrease in concentration of CSF Aβ1-42 than men as they approach their parent’s age at symptom onset (Figure 1 and Figure 4). This interaction of sporadic parental EYO score with sex was replicated in the ACS cohort members with CSF data, where it was additionally supported by PIB-PET imaging findings comparing women and men (Figure 2 and Figure 4). In the ACS cohort members with PIB-PET data, we further found a marginal interaction with sex while looking at the annual rate of Aβ accumulation, suggesting that women might also present a faster rate of Aβ accumulation when approaching their parent’s age at onset. In addition, analyses in the PREVENT-AD cohort highlighted that the association between sporadic parental EYO score and CSF Aβ1-42 was stronger in APOE4 carriers (Figure 1). Consistently, APOE4 carriers in the WRAP cohort accumulated Aβ deposition faster than did the APOE4 noncarriers (Figure 3). Altogether, these results suggest that the association between sporadic parental EYO score and Aβ burden was increased in individuals with a higher risk of developing AD.
These latter results regarding the sporadic parental EYO score × APOE4 status interaction do not make clear whether APOE4 status directly influences the association between Aβ burden and sporadic parental EYO score or whether we found a stronger association between sporadic parental EYO score and Aβ burden in APOE4 carriers only because most noncarriers did not have significant Aβ deposition in these limited age groups. The population prevalence of Aβ positivity in APOE4 noncarriers without dementia at approximately 60 years of age has been found to be only approximately 10%.20 An increasing literature suggests that the effect of APOE4 on AD risk is stronger in women than in men.6,21 Although our data did not permit us to directly test this 3-way interaction, most APOE4 carriers with low Aβ1-42 and/or high brain Aβ burden were women in the PREVENT-AD and the ACS and WRAP cohorts.
Our finding of an association between proximity to parental symptom onset and Aβ in 3 independent cohorts at approximately 50 to 70 years of age supports the well-accepted idea that brain changes start many years before clinical symptoms. This finding also supports the perspective that AD shares common pathophysiologic features with ADAD,22 emphasizing that AD is a strongly heritable disorder. In the PREVENT-AD cohort, for instance, we found no association between the age of a person and CSF Aβ1-42 concentration when the age of the parent at symptom onset was not subtracted from the age of the person. We also did not find an association between the parent’s age at symptom onset and CSF Aβ1-42 concentration, which implies that, in the PREVENT-AD cohort, it was really the proximity to the parent’s age at onset that provided information about Aβ levels.
Strengths and Limitations
Considering the relatively small sample size of each cohort and that not all individuals with a family history of AD will develop dementia, the sporadic parental EYO score was associated with Aβ burden in all 3 cohorts. Each association was replicated in at least 1 cohort despite the 3 cohorts differing in inclusion criteria (ie, the WRAP cohort included individuals with mild cognitive impairment), the participants being recruited in different regions and countries, and different amyloidosis mechanisms being assessed (ie, decrease of Aβ levels in the CSF, Aβ deposition detection and accumulation over time using in vivo PET) (Figure 4 and eTable 4 in the Supplement).
In this article, we used the proposed term sporadic parental EYO score.1 We acknowledge, however, that this term might not be appropriate for sporadic AD because not all individuals with a family history of sporadic AD will develop AD dementia. Furthermore, although the heritability of age at onset has been estimated to be 67% to 87% in AD,2,3 longitudinal studies will be needed to test whether a score of −10 means that a person is truly 10 years away from symptom onset of AD. That some individuals (particularly in the ACS cohort) passed the age at onset of their parents, even when exhibiting abnormal biomarkers, might suggest that some individuals have an age at onset that is delayed with regard to their parent’s age at onset. Compared with ADAD, sporadic AD is hypothesized to be multifactorial,23,24,25 and better lifestyle habits and increased cognitive and brain reserve could play a role in delaying symptom onset.26,27,28
Conclusions
Overall, the sporadic parental EYO score may help the identification of asymptomatic candidates for clinical trials. For instance, the sporadic parental EYO score could be applied as an inexpensive prescreening method that could enrich trial candidate populations for Aβ positivity. Although the sporadic parental EYO score will never replace in vivo Aβ assessment, limiting PET or CSF screening to individuals who are within a 10-year time window of their parent’s symptom onset could significantly reduce the number of LP or PET procedures performed.
eMethods. Participants, Study Design, and Parental Age at Onset Assessment
eTable 1. Proximity to Parental Symptom Onset and Amyloid Burden in the PREVENT-AD Cohort
eTable 2. Proximity to Parental Symptom Onset and Amyloid Burden in the ACS Cohort
eTable 3. Proximity to Parental Symptom Onset and Amyloid Burden in the WRAP Cohort
eTable 4. Age of Parental Symptom Onset and Amyloid Burden in the ACS and WRAP Cohorts
eFigure. Cross-sectional Analyses of Cognitively Normal Individuals From the PREVENT-AD Cohort Demonstrated No Relationship Between Age and CSF Aβ1-42 (A) or Between Age of Parent’s Symptom Onset and CSF Aβ1-42 (B), but Individuals Showed Reductions in CSF Aβ1-42 Levels as They Approached the Age at Their Parent’s Onset (C)
eReferences
References
- 1.Bateman RJ, Xiong C, Benzinger TL, et al. ; Dominantly Inherited Alzheimer Network . Clinical and biomarker changes in dominantly inherited Alzheimer’s disease. N Engl J Med. 2012;367(9):795-804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gatz M, Reynolds CA, Fratiglioni L, et al. Role of genes and environments for explaining Alzheimer disease. Arch Gen Psychiatry. 2006;63(2):168-174. [DOI] [PubMed] [Google Scholar]
- 3.Wingo TS, Lah JJ, Levey AI, Cutler DJ. Autosomal recessive causes likely in early-onset Alzheimer disease. Arch Neurol. 2012;69(1):59-64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Corder EH, Saunders AM, Strittmatter WJ, et al. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer’s disease in late onset families. Science. 1993;261(5123):921-923. [DOI] [PubMed] [Google Scholar]
- 5.Poirier J, Davignon J, Bouthillier D, Kogan S, Bertrand P, Gauthier S. Apolipoprotein E polymorphism and Alzheimer’s disease. Lancet. 1993;342(8873):697-699. [DOI] [PubMed] [Google Scholar]
- 6.Altmann A, Tian L, Henderson VW, Greicius MD; Alzheimer’s Disease Neuroimaging Initiative Investigators . Sex modifies the APOE-related risk of developing Alzheimer disease. Ann Neurol. 2014;75(4):563-573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Snyder HM, Asthana S, Bain L, et al. Sex biology contributions to vulnerability to Alzheimer’s disease: a think tank convened by the Women’s Alzheimer’s Research Initiative. Alzheimers Dement. 2016;12(11):1186-1196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Breitner JCS, Poirier J, Etienne PE, Leoutsakos JM. Rationale and structure for a new center forStudies on Prevention of Alzheimer's Disease (StoP-AD). J Prev Alzheimers Dis. 2016;3(4):236-242. [DOI] [PubMed] [Google Scholar]
- 9.Xiong C, Roe CM, Buckles V, et al. Role of family history for Alzheimer biomarker abnormalities in the adult children study. Arch Neurol. 2011;68(10):1313-1319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Coats M, Morris JC. Antecedent biomarkers of Alzheimer’s disease: the Adult Children Study. J Geriatr Psychiatry Neurol. 2005;18(4):242-244. [DOI] [PubMed] [Google Scholar]
- 11.Sager MA, Hermann B, La Rue A. Middle-aged children of persons with Alzheimer’s disease: APOE genotypes and cognitive function in the Wisconsin Registry for Alzheimer’s Prevention. J Geriatr Psychiatry Neurol. 2005;18(4):245-249. [DOI] [PubMed] [Google Scholar]
- 12.Morris JC. The Clinical Dementia Rating (CDR): current version and scoring rules. Neurology. 1993;43(11):2412-2414. [DOI] [PubMed] [Google Scholar]
- 13.Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MOCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53(4):695-699. [DOI] [PubMed] [Google Scholar]
- 14.Bendlin BB, Ries ML, Canu E, et al. White matter is altered with parental family history of Alzheimer’s disease. Alzheimers Dement. 2010;6(5):394-403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fagan AM, Roe CM, Xiong C, Mintun MA, Morris JC, Holtzman DM. Cerebrospinal fluidtau/β-amyloid42 ratio as a prediction of cognitive decline in nondemented older adults. Arch Neurol. 2007;64(3):343-349. [DOI] [PubMed] [Google Scholar]
- 16.Racine AM, Koscik RL, Berman SE, et al. Biomarker clusters are differentially associated with longitudinal cognitive decline in late midlife. Brain. 2016;139(pt 8):2261-2274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gosselin N, De Beaumont L, Gagnon K, et al. BDNFVal66Met polymorphism interacts with sleep consolidation to predict ability to create new declarative memories. J Neurosci. 2016;36(32):8390-8398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Villemagne VL, Burnham S, Bourgeat P, et al. ; Australian Imaging Biomarkers and Lifestyle (AIBL) Research Group . Amyloid β deposition, neurodegeneration, and cognitive decline in sporadic Alzheimer’s disease: a prospective cohort study. Lancet Neurol. 2013;12(4):357-367. [DOI] [PubMed] [Google Scholar]
- 19.Jack CR Jr, Wiste HJ, Lesnick TG, et al. Brain β-amyloid load approaches a plateau. Neurology. 2013;80(10):890-896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jansen WJ, Ossenkoppele R, Knol DL, et al. ; Amyloid Biomarker Study Group . Prevalence of cerebral amyloid pathology in persons without dementia: a meta-analysis. JAMA. 2015;313(19):1924-1938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Damoiseaux JS, Seeley WW, Zhou J, et al. ; Alzheimer’s Disease Neuroimaging Initiative . Gender modulates the APOE ε4 effect in healthy older adults: convergent evidence from functional brain connectivity and spinal fluid tau levels. J Neurosci. 2012;32(24):8254-8262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bateman RJ, Aisen PS, De Strooper B, et al. Autosomal-dominant Alzheimer’s disease: a review and proposal for the prevention of Alzheimer’s disease. Alzheimers Res Ther. 2011;3(1):1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Chételat G. Alzheimer disease: Aβ-independent processes-rethinking preclinical AD. Nat Rev Neurol. 2013;9(3):123-124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Small SA, Duff K. Linking Aβ and τ in late-onset Alzheimer’s disease: a dual pathway hypothesis. Neuron. 2008;60(4):534-542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Villeneuve S, Wirth M, La Joie R. Are AD-typical regions the convergence point of multiple pathologies? Front Aging Neurosci. 2015;7:42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Craik FI, Bialystok E, Freedman M. Delaying the onset of Alzheimer disease: bilingualism as a form of cognitive reserve. Neurology. 2010;75(19):1726-1729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Villeneuve S, Reed BR, Madison CM, et al. Vascular risk and Aβ interact to reduce cortical thickness in AD vulnerable brain regions. Neurology. 2014;83(1):40-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Wirth M, Villeneuve S, La Joie R, Marks SM, Jagust WJ. Gene-environment interactions: lifetime cognitive activity, APOE genotype, and β-amyloid burden. J Neurosci. 2014;34(25):8612-8617. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
eMethods. Participants, Study Design, and Parental Age at Onset Assessment
eTable 1. Proximity to Parental Symptom Onset and Amyloid Burden in the PREVENT-AD Cohort
eTable 2. Proximity to Parental Symptom Onset and Amyloid Burden in the ACS Cohort
eTable 3. Proximity to Parental Symptom Onset and Amyloid Burden in the WRAP Cohort
eTable 4. Age of Parental Symptom Onset and Amyloid Burden in the ACS and WRAP Cohorts
eFigure. Cross-sectional Analyses of Cognitively Normal Individuals From the PREVENT-AD Cohort Demonstrated No Relationship Between Age and CSF Aβ1-42 (A) or Between Age of Parent’s Symptom Onset and CSF Aβ1-42 (B), but Individuals Showed Reductions in CSF Aβ1-42 Levels as They Approached the Age at Their Parent’s Onset (C)
eReferences