Abstract
Objective
Arterial stiffness has been associated with evidence of cerebral small vessel disease (cSVD) and fibrillar β-amyloid (Aβ) deposition in the brain. These complex relationships have not been examined in racially and cognitively diverse cohorts.
Methods
The Atherosclerosis Risk in Communities (ARIC)–Neurocognitive Study collected detailed cognitive testing for adjudication of dementia and mild cognitive impairment (MCI), brain MRI, and arterial stiffness by pulse wave velocity (PWV, carotid-femoral [cfPWV] and heart-carotid [hcPWV]). The ARIC-PET ancillary study added Aβ imaging using florbetapir ([18F]-AV-45) to obtain standardized uptake volume ratios and defined global Aβ-positivity as standardized uptake volume ratio >1.2. One-SD increase in PWV was related to brain volume, MRI-defined cSVD (e.g., cerebral microbleeds and white matter hyperintensity), and cortical Aβ deposition adjusted for age, body mass index, sex, race, and APOE ε4 status. We examined the cross-sectional relationships including interactions by race, APOE ε4 status, and cognition.
Results
Among the 320 ARIC-PET participants (76 [5] years, 45% black, 27% MCI), greater central stiffness (hcPWV) was associated with greater Aβ deposition (odds ratio [OR] = 1.31, 95% confidence interval [CI] 1.01–1.71). Greater central stiffness (cfPWV) was significantly associated with having lower brain volumes in Alzheimer disease–susceptible regions (in mm3, β = −1.5 [0.7 SD], p = 0.03) and high white matter hyperintensity burden (OR = 1.6, 95% CI 1.2–2.1). Furthermore, cfPWV was associated with a higher odds of concomitant high white matter hyperintensity and Aβ-positive scans (OR = 1.4, 95% CI 1.1–2.1). These associations were strongest among individuals with MCI and did not differ by race or APOE ε4 status.
Conclusions
Arterial stiffness, measured by PWV, is an emerging risk factor for dementia through its repeated relationships with cognition, cSVD, and Aβ deposition.
Vascular disease is an important modifiable risk factor for clinically diagnosed Alzheimer disease (AD) dementia and related dementias.1,2 A greater burden of midlife vascular risk factors is associated with a greater burden of β-amyloid (Aβ) deposition in the brain in late life.3 It remains unclear what mechanisms relate vascular and AD risk factors and whether these processes are additive or interactive in influencing dementia pathology. A growing body of research shows that high blood pressure (BP) and the extent of underlying arterial stiffness are associated with cognitive dysfunction4,5 and evidence of dementia-related pathology, including cerebral small vessel disease (cSVD)6–8 and the extent of Aβ deposition in the brain.9–13 Greater central arterial stiffness is associated with the extent of white matter disease and accumulation of Aβ in the brain over time in elderly adults without dementia, independent of BP.11 Furthermore, individuals with greater arterial stiffness tend to have greater risk of concomitant white matter disease and Aβ deposition.11
Arterial stiffness may be one underlying mechanism linking hypertension to both evidence of cSVD and Aβ deposition in the brain. The overlap of cSVD and Aβ deposition in the brain is proposed to lower the threshold at which individual dementia pathology is expressed as cognitive symptoms and signs.14 To date, only our previous work has examined the potential contribution of arterial stiffness to the overlap of cSVD and Aβ deposition in the brain in a modest sample of very elderly white adults.11,13 It is unclear whether these observed associations differ between racial/ethnic groups. This is especially important for black older adults who are at a greater risk of AD and are twice as likely to have elevated levels of Aβ deposition in the brain compared to white adults.15 Black older adults are also at a higher risk of hypertension, greater arterial stiffness,16–19 stroke, and AD.20 In this study, we examined the individual and joint associations for arterial stiffness with evidence of specific dementia pathologies in black and white older adults in the Atherosclerosis Risk in Communities (ARIC) Study. We hypothesized that arterial stiffness is associated with evidence of both cSVD (e.g., white matter disease and cerebral microbleeds) and Aβ deposition, in both white and black older adults.
Methods
Participants
Participants were recruited to the ARIC-PET ancillary study from the ARIC–Neurocognitive Study (ARIC-NCS), a major ancillary to the ARIC study, as previously described.21 The parent ARIC study began in 1987–1989 with 15,792 individuals from 4 US communities. At the fifth visit (2011–2013), 6,538 surviving ARIC-NCS participants received an extensive neuropsychological battery,22 with informant interview in a subset, and expert classification of cognitive status, including dementia and mild cognitive impairment (MCI).23 A subset of ARIC-NCS participants who received MRI also received Aβ-PET imaging at 3 ARIC sites (Jackson, MS; Washington County, MD; and Forsyth County, NC), as previously described.21
Standard protocol approvals, registrations, and patient consents
This study was approved by each institution's institutional review board. All participants provided written informed consent. The institutional review board at each ARIC site approved the conduct of this study.
Brain MRI and PET
MRI scans were performed on 3-tesla MRI scanners at each site; magnetization-prepared rapid-acquisition gradient echo (MPRAGE) was used for coregistration of PET images. Details about MRI analysis, completed at the ARIC MRI Reading Center (Mayo Clinic), were previously reported.22 Briefly, white matter hyperintensity (WMH) volume (in cubic millimeters) was quantified from fluid-attenuated inversion recovery sequences, using an in-house algorithm,24 and total intracranial volume (cubic millimeters) was measured on MPRAGE using FreeSurfer version 5.1. Using the FreeSurfer atlas, ARIC investigators prespecified regions of interest (ROIs) based on relevance to cognition, including an AD signature volume in cubic millimeters from ROIs from both right and left hemispheres: hippocampus, parahippocampal gyrus, entorhinal cortex, inferior parietal lobule, precuneus, and cuneus.22 Cortical and subcortical brain infarcts, lacunar infarcts, and cerebral microbleeds were identified, counted, and measured by a trained imaging technician and confirmed by radiologists, as previously described.25
PET scans were performed at each site, within 1 year of the brain MRI. The florbetapir isotope was injected through a butterfly needle, with images acquired from 50 to 70 minutes for a 20-minute (4 × 5 minutes) uptake scan. Images were transferred to the PET image analysis center (Johns Hopkins) where they were reviewed qualitatively for incidental findings and image quality, and quantified for standardized uptake value ratios (SUVRs). Images were coregistered to the MRI, spatially normalized, and 34 total ROIs were manually drawn and applied to the SUVR images. Global cortical measure of Aβ deposition was calculated as a weighted average of the gray matter uptake.
Arterial dynamics
Arterial stiffness was measured by pulse wave velocity (PWV) using a noninvasive and automated waveform analyzer (VP1000 plus; Omron Co., Komaki, Japan) at visit 5 (2011–2013). Participants were asked not to consume food or drinks and to refrain from tobacco and vigorous physical activity after midnight or for at least 8 hours before the visit. The instrument also calculates central aortic pressures: systolic BP (SBP) and pulse pressure. All measures were performed under standardized conditions as previously described.26 PWV was measured across 3 vascular beds: central (carotid-femoral [cfPWV], heart-femoral PWV, heart-carotid [hcPWV]); peripheral (femoral-ankle [faPWV]); and mixed (brachial-ankle [baPWV]). PWV was calculated as the distance in centimeters between arterial sites of interest over time (in seconds) that the pressure waveforms traveled from the heart to the respective arterial sites. The average of 2 runs was calculated to determine average PWV. For baPWV, heart-femoral PWV, and faPWV, the right side measures were utilized in the analysis. Reproducibility of PWV measures obtained from this instrument in this cohort was accepted as high based on intraclass correlation coefficient (ICC): baPWV (ICC = 0.84), faPWV (ICC = 0.69), and cfPWV (ICC = 0.70).26
Demographic and covariate information
The primary demographic variables obtained from visit 5 (2011–2013) were included, as covariates in models: age at the time of PET scan (in years), body mass index (kg/m2), sex, race, brachial SBP, and APOE ε4 status (by TaqMan assay; Applied Biosystems, Foster City, CA). Seated brachial SBP and diastolic BP (DBP) were measured twice with the participants in the seated position using an oscillometric automated sphygmomanometer, and the average of each measurement was used for analyses.
Statistical analysis
Associations among Aβ status (SUVR > 1.2), demographic factors, and cSVD were assessed by χ2 tests and t tests and then adjusted for age using logistic regression. Associations between vascular measures and Aβ deposition were assessed first for Aβ status using logistic regression and then with log-transformed continuous measures of total cortical Aβ deposition using general linear models, both adjusted for covariates. Associations between vascular measures and MRI volumes for log-transformed WMH and AD signature volumes were assessed by general linear models adjusted for covariates and total intracranial volume.
Associations between vascular measures and cSVD burden were assessed first for each marker of cSVD individually and then as a composite of cSVD burden, which was created by adding together dichotomized (presence/absence = 0/1) definitions of cerebral microbleeds, lacunes, and high WMH burden to create an index from 0 to 3. For this composite, WMH was dichotomized (high/low) by median split (11,000 mm3) adjusted for total intracranial volume. Because WMH is the most common form of cSVD and showed the strongest associations with both arterial stiffness and Aβ deposition in this and other cohorts, we simplified the overlap between cerebral Aβ deposition and cSVD into 4 groups by combining Aβ status (0/1) and WMH (0/1 by median split). Associations between overlapping high WMH and Aβ with vascular measures were assessed by multinomial logistic regression with low burden of WMH and low Aβ held as referent.
This study focuses on central measures of arterial stiffness (hcPWV and cfPWV, which is the gold standard for measuring central arterial stiffness)27 and includes central pressures (central SBP and central pulse pressure) and how they relate to brain health. Results for peripheral vascular stiffness (baPWV and faPWV) are included in tables e-1 to e-6 (links.lww.com/WNL/A317). Analyses were conducted in SAS version 9.4 (SAS Institute, Cary, NC) for all vascular measures shown per 1 SD. All multivariable models included the following covariates: age, body mass index, sex, race, and APOE ε4. Measures of arterial stiffness that remained significant after the above covariate adjustment were then fitted to a model that included covariates plus seated brachial SBP. Interaction terms were evaluated for potential effect modification by race, APOE ε4, and cognitive status for each model (significance level set at p < 0.15).
Results
Of the 346 participants with Aβ-PET imaging, 321 participants had detailed assessments of arterial stiffness and structural brain abnormalities (table 1). Table 2 describes the demographics of this sample by Aβ status (44% black race and 43% women). As previously reported,15 the proportion of Aβ-positive participants was 53% (n = 169), which was more frequently seen among men, black participants, older age participants, and those with higher seated brachial SBP and DBP (table 2).
Table 1.
Characteristics of dementia-related biomarkers in ARIC participants by age in this sample

Table 2.
Participant characteristics and cortical Aβ deposition

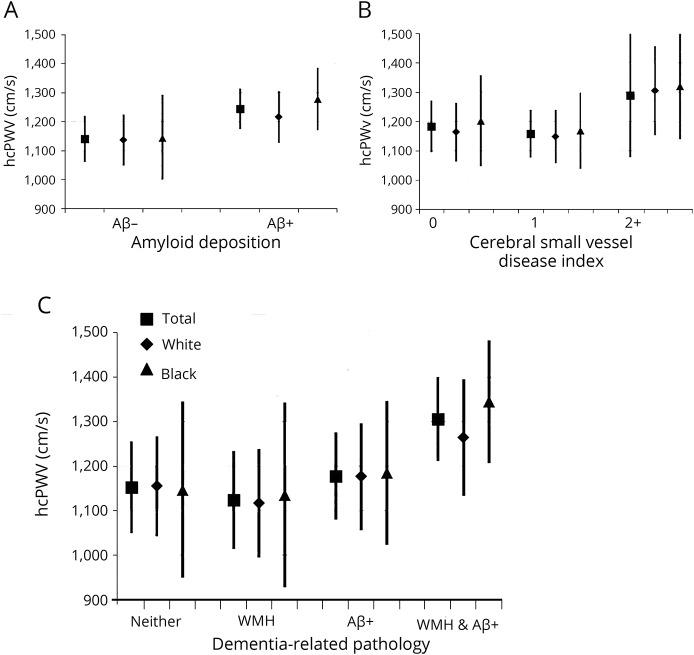
Arterial stiffness measures were correlated with SBP (with the exception of hcPWV; table e-1, links.lww.com/WNL/A317) and were similar between races (with the exception of measures of femoral segments; table e-2). Greater arterial stiffness measured from the heart to the common carotid artery (hcPWV) was significantly associated with approximately a 30% greater odds of being Aβ-positive per 1 SD increase in hcPWV (table 3; figure, A). This was only slightly attenuated after additional adjustment for brachial SBP (odds ratio [OR] = 1.28, 95% confidence interval [CI] 0.99–1.68). This relationship was further evidenced by strong linear associations between hcPWV and the log-transformed extent of total cortical Aβ deposition (β = 0.04, SE = 0.01, p < 0.01) independent of covariates and brachial SBP (table 4). Associations between hcPWV and Aβ status appear to be modified by cognitive status (p interaction = 0.075) with a graded effect in cognitively normal participants (OR = 1.19, 95% CI 0.89–1.59) and most pronounced in MCI (OR = 3.59, 95% CI 1.15–9.20). Significant interactions were not observed for the central measures.
Table 3.
Associations among central arterial stiffness, hemodynamics, and brain Aβ deposition

Figure. hcPWV as it relates to dementia-related pathology by race.
hcPWV (measured in centimeters per second) relates to (A) Aβ deposition (Aβ status, positive or negative, based on SUVR > 1.2) measured by PET; (B) the burden of cerebral small vessel disease (0, 1, and 2 or more forms); and (C) the overlap of common dementia-related pathology, Aβ status, and high WMH volume (high vs low, based on median split) measured by MRI. Shown as means and 95% confidence interval adjusted for age, sex, race, and APOE ε4 status. Aβ = β-amyloid; hcPWV = heart-carotid pulse wave velocity; SUVR = standardized uptake value ratio; WMH = white matter hyperintensity.
Table 4.
Relationships for continuous measures of arterial stiffness with brain β-amyloid deposition and MRI regions

In support of the observed associations between central PWV measures and Aβ status, higher central aortic SBP was associated with nearly a 6-fold-higher odds of being Aβ-positive (OR = 6.08, 95% CI 1.40–26.21, adjusted for covariates; table 3). Central aortic SBP was highly correlated with brachial SBP, which precluded further adjustment for brachial SBP (table e-1, links.lww.com/WNL/A317). Central arterial stiffness was also associated with smaller brain volumes in areas affected by AD, where cfPWV was significantly associated with smaller AD signature regional volumes (in mm3, β = −528, SE = 239, p value = 0.03), adjusted for covariates and intracranial volume (table 4), primarily driven by smaller volumes in the precuneus. Other measures of arterial stiffness and central pressures were not significantly associated with Aβ deposition (tables 3 and e-3).
Among this sample of ARIC participants, greater central arterial stiffness was also associated with a greater burden of cSVD as measured with a cumulative cSVD burden (2 or more forms of cSVD [n = 77], OR = 1.80, 95% CI 1.26–2.59; 1 form of cSVD [n = 154], OR = 1.27, 95% CI 0.93–1.74; compared with no evidence of cSVD [n = 115]), which was independent of covariates and SBP (figure, B). Associations between central arterial stiffness and evidence of cSVD were primarily driven by evidence of high WMH (median split OR = 1.43, 95% CI 1.09–1.88) and total cerebral microbleeds (80/321 [25% prevalence], OR = 1.46, 95% CI 1.09–1.95; data not shown). No interactions among arterial stiffness, APOE ε4, or race were observed for other dementia-related outcomes (p interaction all >0.15; figure, A–C).
Arterial stiffness is associated with concomitant cSVD and Aβ deposition
Brain Aβ deposition was significantly associated with evidence of cSVD, specifically high WMH and deep microbleeds (table e-4, links.lww.com/WNL/A317). Greater arterial stiffness was associated with the overlapping burden of high WMH and Aβ deposition (92/321, 29%), where each SD increase in cfPWV was associated with a 44% higher odds of having both forms of dementia-related pathology (OR = 1.44, 95% CI 1.01–2.06) when compared to the individuals with little evidence of either pathology (tables 5 and e-5; figure, C).
Table 5.
Central arterial stiffness and pressure as they relate to the overlap between white matter disease and Aβ deposition

Discussion
In this biracial cohort of older adults in the ARIC Study, we observed that arterial stiffness of the central and carotid segments was associated with the burden of multiple forms of dementia-related pathology, including cSVD, Aβ deposition, and smaller brain volumes in AD-prone regions of the brain. These relationships were similar for black and white older adults and the strongest among individuals with MCI. Greater arterial stiffness, measured by higher hcPWV, corresponds to the faster speed at which the pulse travels from the heart to the carotid artery and was most strongly associated with greater Aβ burden in this study. This marked increased pulsatile flow velocity and central pressure observed among Aβ-positive individuals was equivalent to a 103 cm/s faster PWV and a 25 mm Hg higher central aortic SBP relative to Aβ-negative individuals (figure, A–C). Greater arterial stiffness was strongly associated with the overlap of dementia-related pathology (WMH, cerebral microbleeds, and Aβ deposition) and lower brain volumes in regions susceptible to AD and Aβ deposition. The observed findings replicate our previous results relating arterial stiffness to both white matter disease and the extent of Aβ deposition in white older adults11,13 and extend them to a younger cohort of black and white older adults to show no differences by race and stronger associations with MCI.
This work contributes to a growing literature showing that arterial stiffness and elevated central pulsatility are important modifiable risk factors for dementia pathology in older adults. Arterial stiffness increases monotonically with age across multiple vascular beds and is accelerated by cardiometabolic disorders.28–32 Arterial stiffening is proposed to be a key hemodynamic process that results in the transmission of excessive pulsatile pressure from the aorta to the distal vascular beds, which damages the microvascular structure of the brain and kidneys, evidenced by white matter disease, other forms of cSVD, and renal disease.32 This study provides consistent evidence that greater arterial stiffening may contribute to dementia through its associations with white matter disease, cerebral microbleeds, and Aβ deposition, pathology that is consistent with both cerebral amyloid angiopathy and AD.
We have already shown that greater arterial stiffness is also associated with a higher rate of Aβ accumulation over time in older adults,13 but it remains unclear how arterial stiffening may contribute directly to Aβ deposition in the brain.33 It is possible that these 2 common age-related processes occur in parallel and do not contribute directly to the progression of the other. There is considerable ongoing research and debate as to whether carotid or intracerebral stiffness, distensibility, and pulsatility are more important for brain health than central aortic stiffness.34 Given the associations between arterial stiffness and greater intracerebral vascular pulsatility35 and cSVD, it is likely that excess pulsatile pressure delivered by stiffer arteries damages the cerebral microvasculature of the basil ganglia while delivering lower cortical cerebral blood flow to the cortex and subcortical white matter. The key locus of this injury is the arterioles and perivascular spaces within the cerebral cortex: alterations in the penetrating arterioles can be hypothesized to disrupt intramural and lymphatic transport of Aβ from the brain.35 The lack of consistent associations between cSVD measures and Aβ burden presented here and in other studies suggests that these forms of dementia pathology are largely unrelated.14 Thus, arterial stiffness may link the progression of these independent forms of dementia pathology.
The associations among central arterial stiffness, WMH, and brain Aβ deposition have been documented in very elderly adults aged 80+ years11 and here in a younger, biracial cohort of older adults aged 70+ years. These data suggest that life course factors that promote arterial stiffness would also be associated with Aβ deposition in the brain. This remains to be investigated in cohorts specifically with hypertension and metabolic disorders that contribute to the faster progression of central arterial stiffness with age.31,36,37 Greater midlife and antemortem pulse pressure are associated with a greater burden of Aβ plaque and neurofibrillary tangles in the brain.38,39 While elevated brachial SBP, DBP, and hypertension have been associated with greater Aβ deposition in the brain,9,10,12 here we show that associations between arterial stiffness and Aβ largely remain independent of brachial pressure. Taken together, this study adds to a growing literature linking cardiometabolic disorders to accelerated arterial stiffness37 and from arterial stiffness to multiple aspects of dementia pathology.40 Arterial stiffness may serve as an important intermediary linking cardiometabolic disorders and dementia pathology in both white and black older adults.
There are some important limitations to this study. First, this study is cross-sectional in nature, but consistent with our previous longitudinal work.13 Second, it is possible that these results may still be biased by unmeasured confounding, especially in the absence of microvascular measures and tau pathology that may fall within the causal pathway. Specifically, future work would benefit from more direct measures of intracranial pulsatility and blood flow as well as novel microinfarcts visible on MRI.41 Additional cerebral hemodynamic measures may mediate the relationships between arterial stiffness and cerebral pathology and provide mechanistic insights into causal pathways. It is important to note that these modest associations are observed in older adults free from dementia, who are by selection less likely to show signs of later-stage disease processes, including severe amyloid deposition, neurodegeneration, and cognitive and functional disabilities. Participants in this study are on average 10 years younger than those in our previous work and show less arterial stiffness.11,13 Selection bias may mitigate the relationships between arterial stiffness and brain structural abnormalities; however, we still detect associations between arterial stiffness and dementia-related pathology in the brain. Future longitudinal studies assessing changes in arterial stiffness, brain structural abnormalities, cognitive decline, and conversion to dementia are ongoing.
These findings show that greater arterial stiffness as measured by PWV is associated with greater evidence of multiple dementia-related pathologies in the brain, adding to a growing literature showing that arterial stiffness is a risk factor for age-related dementias. The mechanical transference of greater pulsatile pressure waveforms along stiff arteries is proposed to result in mechanical damage to the cerebral small vessels, which may contribute to cSVD and accelerated Aβ accumulation in older adults, through yet unknown means. These 2 forms of dementia pathology are common in older adults and appear to exacerbate brain atrophy and cognitive decline. Interventions aimed at reducing cerebral pulsatility by improving central vascular function may affect dementia risk by preventing dementia-related pathology.
Acknowledgment
The authors thank the staff and participants of the ARIC Study for their important contributions. Avid Radiopharmaceuticals provided the florbetapir isotope for the study, but had no role in the study design or interpretation of results.
Glossary
- Aβ
β-amyloid
- AD
Alzheimer disease
- ARIC
Atherosclerosis Risk in Communities
- ARIC-NCS
Atherosclerosis Risk in Communities–Neurocognitive Study
- baPWV
brachial-ankle pulse wave velocity
- BP
blood pressure
- cfPWV
carotid-femoral pulse wave velocity
- CI
confidence interval
- cSVD
cerebral small vessel disease
- DBP
diastolic blood pressure
- faPWV
femoral-ankle pulse wave velocity
- hcPWV
heart-carotid pulse wave velocity
- ICC
intraclass correlation coefficient
- MCI
mild cognitive impairment
- MPRAGE
magnetization-prepared rapid-acquisition gradient echo
- OR
odds ratio
- PWV
pulse wave velocity
- ROI
region of interest
- SBP
systolic blood pressure
- SUVR
standardized uptake value ratio
- WMH
white matter hyperintensity
Footnotes
Editorial, page 635
Author contributions
Timothy M. Hughes: drafting/revising the manuscript, study concept or design, analysis or interpretation of data, accepts responsibility for conduct of research and will give final approval, statistical analysis. Lynne E. Wagenknecht: drafting/revising the manuscript, study concept or design, accepts responsibility for conduct of research and will give final approval, acquisition of data, study supervision, obtaining funding. Suzanne Craft: drafting/revising the manuscript, analysis or interpretation of data, accepts responsibility for conduct of research and will give final approval. Akiva Mintz: study concept or design, analysis or interpretation of data, accepts responsibility for conduct of research and will give final approval, contribution of vital reagents/tools/patients, acquisition of data, study supervision. Gerardo Heiss: drafting/revising the manuscript, accepts responsibility for conduct of research and will give final approval, obtaining funding. Priya Palta: drafting/revising the manuscript, accepts responsibility for conduct of research and will give final approval. Dean F. Wong: drafting/revising the manuscript, accepts responsibility for conduct of research and will give final approval. Yun Zhou: drafting/revising the manuscript, analysis or interpretation of data, accepts responsibility for conduct of research and will give final approval. David S. Knopman: drafting/revising the manuscript, accepts responsibility for conduct of research and will give final approval. Thomas H. Mosley: drafting/revising the manuscript, accepts responsibility for conduct of research and will give final approval, acquisition of data, study supervision. Rebecca F. Gottesman: drafting/revising the manuscript, study concept or design, analysis or interpretation of data, accepts responsibility for conduct of research and will give final approval, study supervision, obtaining funding.
Study funding
The Atherosclerosis Risk in Communities Study is performed as a collaborative study supported by National Heart, Lung, and Blood Institute (NHLBI) contracts (HHSN268201100005C, HHSN268201100006C, HHSN268201100007C, HHSN268201100008C, HHSN268201100009C, HHSN268201100010C, HHSN268201100011C, and HHSN268201100012C). Neurocognitive data are collected by U01 2U01HL096812, 2U01HL096814, 2U01HL096899, 2U01HL096902, and 2U01HL096917 from the NIH (NHLBI, NINDS, NIA, and NIDCD), and with previous brain MRI examinations funded by R01-HL70825 from the NHLBI and analyses funded by R01-AG053938. The ARIC-PET Study is funded by the National Institute on Aging (R01AG040282).
Disclosure
The authors report no disclosures relevant to the manuscript. Go to Neurology.org/N for full disclosures.
References
- 1.Gorelick PB, Scuteri A, Black SE, et al. Vascular contributions to cognitive impairment and dementia: a statement for healthcare professionals from the American Heart Association/American Stroke Association. Stroke 2011;42:2672–2713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Montine TJ, Koroshetz WJ, Babcock D, et al. Recommendations of the Alzheimer's Disease–Related Dementias Conference. Neurology 2014;83:851–860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Gottesman RF, Schneider AL, Zhou Y, et al. Association between midlife vascular risk factors and estimated brain amyloid deposition. JAMA 2017;317:1443–1450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rabkin SW. Arterial stiffness: detection and consequences in cognitive impairment and dementia of the elderly. J Alzheimers Dis 2012;32:541–549. [DOI] [PubMed] [Google Scholar]
- 5.Cooper LL, Woodard T, Sigurdsson S, et al. Cerebrovascular damage mediates relations between aortic stiffness and memory. Hypertension 2016;67:176–182. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rosano C, Watson N, Chang Y, et al. Aortic pulse wave velocity predicts focal white matter hyperintensities in a biracial cohort of older adults. Hypertension 2013;61:160–165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Henskens LH, Kroon AA, van Oostenbrugge RJ, et al. Increased aortic pulse wave velocity is associated with silent cerebral small-vessel disease in hypertensive patients. Hypertension 2008;52:1120–1126. [DOI] [PubMed] [Google Scholar]
- 8.van Sloten TT, Protogerou AD, Henry RM, Schram MT, Launer LJ, Stehouwer CD. Association between arterial stiffness, cerebral small vessel disease and cognitive impairment: a systematic review and meta-analysis. Neurosci Biobehav Rev 2015;53:121–130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Langbaum JB, Chen K, Launer LJ, et al. Blood pressure is associated with higher brain amyloid burden and lower glucose metabolism in healthy late middle-age persons. Neurobiol Aging 2012;33:827.e11–829.e11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Toledo JB, Toledo E, Weiner MW, et al. Cardiovascular risk factors, cortisol, and amyloid-beta deposition in Alzheimer's Disease Neuroimaging Initiative. Alzheimers Dement 2012;8:483–489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hughes TM, Kuller LH, Barinas-Mitchell EJ, et al. Pulse wave velocity is associated with β-amyloid deposition in the brains of very elderly adults. Neurology 2013;81:1711–1718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Rodrigue KM, Rieck JR, Kennedy KM, Devous MD Sr, Diaz-Arrastia R, Park DC. Risk factors for β-amyloid deposition in healthy aging: vascular and genetic effects. JAMA Neurol 2013;70:600–606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hughes TM, Kuller LH, Barinas-Mitchell EJ, et al. Arterial stiffness and beta-amyloid progression in nondemented elderly adults. JAMA Neurol 2014;71:562–568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Provenzano FA, Muraskin J, Tosto G, et al. White matter hyperintensities and cerebral amyloidosis: necessary and sufficient for clinical expression of Alzheimer disease? JAMA Neurol 2013;70:455–461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gottesman RF, Schneider AL, Zhou Y, et al. The ARIC-PET amyloid imaging study: brain amyloid differences by age, race, sex, and APOE. Neurology 2016;87:473–480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Shen J, Poole JC, Topel ML, et al. Subclinical vascular dysfunction associated with metabolic syndrome in African Americans and whites. J Clin Endocrinol Metab 2015;100:4231–4239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Meyer ML, Tanaka H, Palta P, et al. Correlates of segmental pulse wave velocity in older adults: the Atherosclerosis Risk in Communities (ARIC) Study. Am J Hypertens 2016;29:114–122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Din-Dzietham R, Couper D, Evans G, Arnett DK, Jones DW. Arterial stiffness is greater in African Americans than in whites: evidence from the Forsyth County, North Carolina, ARIC cohort. Am J Hypertens 2004;17:304–313. [DOI] [PubMed] [Google Scholar]
- 19.Goel A, Maroules CD, Mitchell GF, et al. Ethnic difference in proximal aortic stiffness: an observation from the Dallas Heart Study. JACC Cardiovasc Imaging 2016;10:54–61. [DOI] [PubMed] [Google Scholar]
- 20.Barnes LL, Bennett DA. Alzheimer's disease in African Americans: risk factors and challenges for the future. Health Aff 2014;33:580–586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Rawlings AM, Sharrett AR, Schneider AL, et al. Diabetes in midlife and cognitive change over 20 years: a cohort study. Ann Intern Med 2014;161:785–793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Knopman DS, Griswold ME, Lirette ST, et al. Vascular imaging abnormalities and cognition: mediation by cortical volume in nondemented individuals: Atherosclerosis Risk in Communities–Neurocognitive Study. Stroke 2015;46:433–440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Knopman DS, Gottesman RF, Sharrett AR, et al. Mild cognitive impairment and dementia prevalence: the Atherosclerosis Risk in Communities Neurocognitive Study (ARIC-NCS). Alzheimers Dement 2016;2:1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Jack CR Jr, O'Brien PC, Rettman DW, et al. FLAIR histogram segmentation for measurement of leukoaraiosis volume. J Magn Reson Imaging 2001;14:668–676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Kantarci K, Weigand SD, Przybelski SA, et al. Risk of dementia in MCI: combined effect of cerebrovascular disease, volumetric MRI, and 1H MRS. Neurology 2009;72:1519–1525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Meyer ML, Tanaka H, Palta P, et al. Repeatability of central and peripheral pulse wave velocity measures: the Atherosclerosis Risk in Communities (ARIC) Study. Am J Hypertens 2016;29:470–475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Laurent S, Cockcroft J, Van Bortel L, et al. Expert consensus document on arterial stiffness: methodological issues and clinical applications. Eur Heart J 2006;27:2588–2605. [DOI] [PubMed] [Google Scholar]
- 28.Lakatta EG, Levy D. Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises: Part I: aging arteries: a “set up” for vascular disease. Circulation 2003;107:139–146. [DOI] [PubMed] [Google Scholar]
- 29.AlGhatrif M, Strait JB, Morrell CH, et al. Longitudinal trajectories of arterial stiffness and the role of blood pressure: the Baltimore longitudinal study of aging. Hypertension 2013;62:934–941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Najjar SS, Scuteri A, Shetty V, et al. Pulse wave velocity is an independent predictor of the longitudinal increase in systolic blood pressure and of incident hypertension in the Baltimore Longitudinal Study of Aging. J Am Coll Cardiol 2008;51:1377–1383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Safar ME, Balkau B, Lange C, et al. Hypertension and vascular dynamics in men and women with metabolic syndrome. J Am Coll Cardiol 2013;61:12–19. [DOI] [PubMed] [Google Scholar]
- 32.Mitchell GF. Effects of central arterial aging on the structure and function of the peripheral vasculature: implications for end-organ damage. J Appl Physiol 2008;105:1652–1660. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gangoda SVS, Avadhanam B, Jufri NF, Sohn EH, Butlin M, Gupta V, Chung R, Avolio AP. Pulsatile stretch as a novel modulator of amyloid precursor protein processing and associated inflammatory markers in human cerebral endothelial cells. Sci Rep. 2018;8:1689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Geijselaers SL, Sep SJ, Schram MT, et al. Carotid stiffness is associated with impairment of cognitive performance in individuals with and without type 2 diabetes. The Maastricht Study. Atherosclerosis 2016;253:186–193. [DOI] [PubMed] [Google Scholar]
- 35.Webb AJ, Simoni M, Mazzucco S, Kuker W, Schulz U, Rothwell PM. Increased cerebral arterial pulsatility in patients with leukoaraiosis: arterial stiffness enhances transmission of aortic pulsatility. Stroke 2012;43:2631–2636. [DOI] [PubMed] [Google Scholar]
- 36.Hughes TM, Craft S. The role of insulin in the vascular contributions to age-related dementia. Biochim Biophys Acta 2016;1862:983–991. [DOI] [PubMed] [Google Scholar]
- 37.Smulyan H, Lieber A, Safar ME. Hypertension, diabetes type II, and their association: role of arterial stiffness. Am J Hypertens 2016;29:5–13. [DOI] [PubMed] [Google Scholar]
- 38.Petrovitch H, White LR, Izmirilian G, et al. Midlife blood pressure and neuritic plaques, neurofibrillary tangles, and brain weight at death: the HAAS. Honolulu-Asia Aging Study. Neurobiol Aging 2000;21:57–62. [DOI] [PubMed] [Google Scholar]
- 39.Nation DA, Delano-Wood L, Bangen KJ, et al. Antemortem pulse pressure elevation predicts cerebrovascular disease in autopsy-confirmed Alzheimer's disease. J Alzheimers Dis 2012;30:595–603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Hughes TM, Craft S, Lopez OL. Review of 'the potential role of arterial stiffness in the pathogenesis of Alzheimer's disease.' Neurodegener Dis Manag 2015;5:121–135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.van Veluw SJ, Hilal S, Kuijf HJ, et al. Cortical microinfarcts on 3T MRI: clinical correlates in memory-clinic patients. Alzheimers Dement 2015;11:1500–1509. [DOI] [PubMed] [Google Scholar]