Abstract
Most research on cannabis pharmacokinetics has evaluated inhaled cannabis, but oral (“edible”) preparations comprise an increasing segment of the cannabis market. To assess oral cannabis pharmacokinetics and pharmacodynamics, healthy adults (N = 6 per dose) were administered cannabis brownies containing 10, 25 or 50 mg 9-tetrahydrocannabinol (THC). Whole blood and oral fluid specimens were obtained at baseline and then for 9 days post-exposure; 6 days in a residential research setting and 3 days as outpatients. Measures of subjective, cardiovascular and performance effects were obtained at baseline and for 8 h post-ingestion. The mean Cmax for THC in whole blood was 1, 3.5 and 3.3 ng/mL for the 10, 25 and 50 mg THC doses, respectively. The mean maximum concentration (Cmax) and mean time to maximum concentration (Tmax) of 11-OH-THC in whole blood were similar to THC. Cmax blood concentrations of THCCOOH were generally higher than THC and had longer Tmax values. The mean Tmax for THC in oral fluid occurred immediately following oral dose administration, and appear to reflect local topical residue rather than systemic bioavailbility. Mean Cmax oral fluid concentrations of THCCOOH were lower than THC, erratic over time and mean Tmax occurred at longer times than THC. The window of THC detection ranged from 0 to 22 h for whole blood (limit of quantitation (LOQ) = 0.5 ng/mL) and 1.9 to 22 h for oral fluid (LOQ = 1.0 ng/mL). Subjective drug and cognitive performance effects were generally dose dependent, peaked at 1.5–3 h post-administration, and lasted 6–8 h. Whole blood cannabinoid concentrations were significantly correlated with subjective drug effects. Correlations between blood cannabinoids and cognitive performance measures, and between oral fluid and all pharmacodynamic outcomes were either non-significant or not orderly by dose. Quantitative levels of cannabinoids in whole blood and oral fluid were low compared with levels observed following inhalation of cannabis. The route of administration is important for interpretation of cannabinoid toxicology.
Introduction
Legal use of cannabis (marijuana) for medical and non-medical purposes is expanding worldwide. At present, medical use of cannabis has been legalized in 23 US states and the District of Columbia, and in multiple countries including Australia, Canada, Israel and the Netherlands. Legal non-medical (aka “recreational”) use of cannabis is also allowed in four US states and Uruguay, and cannabis remains the most used illicit drug in the majority of countries where its use is prohibited (1).
Expansion of legal cannabis markets has substantially increased the type and variety of cannabis/cannabinoid products commercially available. In addition to traditional preparations of dried cannabis plant flowers and cannabis resin (“hashish”), cannabis products available for retail purchase now include a large number of “edible” food and drink products, oils and tinctures, highly concentrated extracts, and transdermal products. Edible cannabis products predominantly include baked goods (e.g., brownies, cookies), beverages (e.g., coffee, tea, juice and soda), candies (e.g., chocolate, hard and soft candies), cooking ingredients (e.g., butter, oils and honey) containing cannabis or cannabis oil, or capsules containing cannabis extract to be swallowed whole. Surveys of medical cannabis users indicate that 16–26% of medical cannabis patients use edible cannabis products (2, 3), and edible products comprised an estimated 40% of medical and non-medical retail cannabis sales in Colorado in 2014 (4). Edible administration may be more attractive than smoking to some individuals, due to longer time course of drug effects, lack of exposure to harmful byproducts produced by combustion of plant material, and because use of edible cannabis products may be more discreet than smoking. Thought to be safer than smoked cannabis in some ways (e.g., reduced carcinogen exposure), titration of dose through oral administration, unlike smoking, is difficult and may result in stronger and more frequent adverse side effects (e.g., panic, paranoia and performance impairment) (5, 6). This risk stems from slow and unpredictable absorption of orally administered cannabis and because edible product labeling of delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD) content may be inaccurate (7).
The increased availability and use of cannabis products designed for routes of administration other than inhalation present a number of scientific and regulatory challenges. Most research on the pharmacokinetic and pharmacodynamic effects of cannabis has been conducted via acute administration of inhaled cannabis. As a result, regulations that define biological cutoffs for detecting cannabis intoxication (e.g., roadside and workplace drug testing), and precedents for interpreting forensic toxicology results related to cannabis use are based on data that may not be applicable to other routes of administration. To our knowledge, only three published studies have performed controlled evaluations of acute oral cannabis exposure. In one laboratory study, five participants were administered cannabis-containing brownies with THC doses of 0, 22 and 45 mg using a within-subjects crossover dosing procedure (8). Dose-related increases in subjective drug effects were observed and 11-nor-9-carboxy-THC (THCCOOH), a major metabolite of THC, was detectable in urine for 3–14 days post-exposure (LOQ = 2 ng/mL). In a series of three small experiments, Niedbala et al. (9) allowed participants to self-administer cannabis containing ~20–25 mg THC via either smoking (cannabis cigarette) or ingesting orally (cannabis-containing brownie). Quantitative levels of THC in oral fluid were substantially lower following oral administration compared with smoking, but the disposition of THCCOOH in urine was generally similar across both routes of administration. Recently, Huestis and colleagues reported oral fluid pharmacokinetics in frequent and infrequent cannabis users administered approximately 50 mg oral cannabis along with acute smoked and vaporized cannabis in a within-subjects crossover study (10). They found evidence of cannabinoid contamination in the oral mucosa immediately following oral, smoked and vaporized cannabis. Compared with the two inhaled routes of administration, Cmax for THC in oral fluid was reduced and Tmax for THCCOOH in oral fluid was delayed.
The results of the prior studies must be considered in light of two important limitations. First, participants in both studies were allowed to eat and/or drink ad libitum, which may have introduced variability in cannabinoid absorption. Second, the published studies to date have not examined the cognitive/psychomotor effects of oral cannabis administration. As a result, there are no data on relations between biological markers of oral cannabis administration and levels of cognitive/psychomotor impairment to inform standards for biological verification of cannabis intoxication. Additional data that circumvents the limitations of prior studies would be valuable with regard to informing workplace and roadside drug testing guidelines for oral cannabis and for defining an appropriate unit dose for edible cannabis products sold in legal cannabis markets. The current study was conducted to extend prior research on the pharmacokinetics and pharmacodynamics of oral cannabis administration. We report the results of whole blood and oral fluid analyses of specimens obtained before and after exposure to three doses of cannabis under controlled laboratory conditions. Quantitative testing was conducted to measure levels of THC and its metabolites across biological matrices and to evaluate their respective windows of detection via qualitative enzyme-linked immunosorbent assay (ELISA) and quantitative liquid chromatography–tandem mass spectrometry (LC–MS-MS) test methods. This report provides a detailed description of the experimental methods, initial screening and confirmatory data derived from whole blood and oral fluid specimens, and outcomes from concurrently collected pharmacodynamic assessments.
Experimental Methods
Participants
Study volunteers were recruited through media advertisements and word-of-mouth. Participants were healthy adults between the ages of 18 and 45 years (ascertained via medical history, electrocardiogram (EKG), and routine blood chemistry, hematology, serology analysis), who had a history of lifetime cannabis exposure, but had not used cannabis or other illicit substances within the previous 3 months (ascertained via self-report, and urine screening for cannabis, amphetamines, benzodiazepines, cocaine, 3,4-methylenedioxy-methamphetamine (MDMA), opioids and phencyclidine (PCP) using rapid enzyme immunoassay test kits) at the screening visit and upon admission to the research unit for each experimental session.
A total of 18 participants (9 males and 9 females) completed the study. They had a mean (standard deviation (SD), range) age of 26 (4, 20–33) years, weighed a mean of 71 (17, 44–102) kg, and had a mean body mass index (BMI) of 23 (3, 19–30). Participants self-identified their race/ethnicity as follows: 13 Caucasian, non-Hispanic, 2 Caucasian, Hispanic, 1 African American, non-Hispanic, 1 Asian, non-Hispanic and 1 American Indian, Hispanic.
Written informed consent was obtained prior to study participation. The study was approved by the Johns Hopkins Medicine Institutional Review Board and conducted in accordance with the ethical standards of the Helsinki Declaration. All participants were compensated for completing study procedures.
Study design and procedures
The primary aim of the study was to characterize the pharmacokinetics of cannabis following oral administration. A between-subjects design was used to evaluate three doses: 10, 25 and 50 mg of THC. Six participants were randomly assigned to receive one dose of oral cannabis, counterbalanced by sex such that three males and three females participants received each dose. Data collection was conducted over 9 consecutive days. The first 6 days were completed in a residential research unit. Days 7–9 were completed on an outpatient basis with one laboratory assessment each day. Tobacco use was not permitted during the study. Participants were allowed a single caffeinated beverage each morning to mitigate the effects of caffeine withdrawal.
On Day 1 of the study, participants arrived to the Johns Hopkins Behavioral Pharmacology Research Unit (BPRU) in Baltimore, MD at ~07:30 a.m. Urine drug and breath alcohol tests were administered to confirm that participants were not under the influence of alcohol or drugs, and to ensure no recent use of cannabis had occurred. Urine pregnancy tests were administered for female participants. Participants were fed a standard low-fat breakfast and were not allowed snacks for 30 min post-dosing to minimize differences in drug absorption across participants. Nursing staff placed an intravenous catheter in the non-dominant arm of each participant for repeated blood sampling. Baseline assessments were completed, including biological specimen collection, self-report ratings, vitals and performance assessments. Next, study participants were given a single, weighed cannabis brownie to eat. Dosing was double blind, and participants consumed the entire brownie under staff observation within 5 min. Water was allowed to drink as needed. After the brownie had been consumed, urine, blood and oral fluid were collected over the next 9 days, and pharmacodynamic assessments were completed for 8 h. Hair samples were obtained at baseline and on Day 9. Results of urine and hair testing are not reported here.
Study drug
Cannabis containing ~11% total THC; 0.1% CBD and 0.8% cannabinol (CBN) was obtained from the National Institute on Drug Abuse (NIDA) Drug Supply Program. The cannabis was baked into brownies at the Johns Hopkins BPRU Pharmacy. To prepare the brownies, cannabis was first ground into a powder using a food processor. The plant material was baked at 250°F (121°C) for 30 min to facilitate decarboxylation of tetrahydrocannabinolic acid (THC-A) to THC. Each brownie was prepared individually and contained measured amounts of cannabis in order to produce target THC doses of 10, 25 or 50 mg per serving. Cannabis was stirred into an individual baking tray containing commercial chocolate brownie mix and the recommended mix of vegetable oil, egg and water provided on the recipe for the brownie mix. Brownies were made 24–48 h before administration. Sample brownies containing each dose were analyzed by ElSohly Laboratories, Inc. (Oxford, MS) and found to have a THC content of 9.4, 23.6 and 48.5 mg THC, respectively. Each contained <1 mg THC-A, indicating complete decarboxylation of THC-A to THC during preparation. The deviation of measured THC doses to target THC doses (10, 25 and 50 mg) is within the margin of error taking into account testing the cannabis potency and THC content of each brownie.
Study measures
Participant screening
Locally developed questionnaires were administered to collect background demographic data, medical history and history of licit and illicit substance use. The Time-line Follow-Back (TLFB) method (11) was used to obtain a detailed account of substance use for the 90 days prior to the screening interview. A physical examination was performed to assess the health of each participant, including assessment of major organ systems, including head, eyes, ears, nose and throat (HEENT); cardiovascular system; lungs; abdomen (liver/spleen); extremities; skin; central nervous system; musculoskeletal system; and general appearance. A 12-lead EKG was conducted to ascertain cardiovascular health. Blood, urine and breath specimens were obtained and tested for routine clinical chemistry, hematology, serology, serum pregnancy (females only) and for evidence of recent alcohol and illicit drug use.
Blood specimens
Whole blood specimens (10 mL) were obtained from indwelling venous catheters using gray-top vacutainer collection tubes at baseline and 0.17, 0.5, 1, 1.5, 2, 3, 4, 5, 6, 8, 12, 22, 26, 30, 34, 46, 50, 54, 58, 70, 74, 78, 82, 94, 98, 102, 106, 118, 122, 126 and 130 h after oral cannabis administration. The specimens were mixed by inversion, then two 5 mL aliquots were transferred to plastic cryotubes, labeled and stored frozen at −60°C until shipped frozen on dry ice for analysis.
Blood specimens were analyzed by ELISA and LC–MS-MS at Immunalysis Corporation (Pomona, CA) upon receipt at the laboratory. The Cannabinoids Direct ELISA Kit (THC-A/C-THC) was tested according to the manufacturer's procedure at a cutoff concentration for THCCOOH of 10 ng/mL. Cross-reactivities for this assay, as listed in the manufacturer's brochure, are: THCCOOH (100%); 11-nor-9-carboxy-Δ8-THC (110%); Δ9-THC (21%); Δ8-THC (45%); 11-OH-THC (<5%); 8-11-dihydroxy- Δ9-THC (<5%); CBN (<5%); and CBD (<5%). The cross-reactivity of THCCOOH-glucuronide was not listed in the manufacturer's brochure, but was communicated by the manufacturer to be 5% cross-reactivity to THCCOOH at a 10 ng/mL cutoff concentration (personal communication, Christine Moore).
LC–MS-MS analyses for THC, 11-hydroxy-Δ9-tetrahydrocannabinol (11-OH-THC) and THCCOOH were conducted according to the methods described by Coulter et al. (12). Specimens were not hydrolyzed prior to analyses. Consequently, blood concentrations of analytes are reported as “free” concentrations. The limit of quantitation (LOQ) and upper limit of linearity (ULOL), respectively, for analyses of blood were: THC, 0.5, 100 ng/mL; THCCOOH, 0.5, 100 ng/mL; and 11-OH-THC, 0.5, 100 ng/mL. Control samples were prepared from Cerillant (Round Rock, TX) solutions. The inter-day ranges (5 days; n = 6/day) of percent deviation from the target concentration of control samples prepared for whole blood analyses were: THC, 7.5 ng/mL (n = 30), −21.3 to −4.00%; 11-OH-THC, 7.5 ng/mL (n = 30), −21.3 to −6.7%; and THCCOOH, 7.5 ng/mL (n = 30), −13.3 to 16.0%. The intra-run precisions for blood control samples were: THC, 7.5 ng/mL (n = 6), 3.8%; 11-OH-THC, 7.5 ng/mL (n = 6), 3.0%; and THCCOOH, 7.5 ng/mL (n = 6), 5.8%.
Oral fluid specimens
Oral fluid specimens (5 mL) were obtained by expectoration into 8 mL glass screw cap culture tubes (Thermo Fisher Scientific, Waltham, MA, 16 × 100 mm, #14-959-35AA) at Baseline and 0.17, 0.5, 1, 1.5, 2, 3, 4, 5, 6, 8, 12, 22, 26, 30, 34, 46, 50, 54, 58, 70, 74, 78, 82, 94, 98, 102, 106, 118, 122, 126 and 130 h after ingestion of the cannabis-containing brownie. Three additional specimens were obtained during the outpatient visits on Days 7, 8 and 9 after ingestion; the exact timing of collection on these days varied across participants based on their availability. Prior to collection, the inner surface of the collection tubes was silanized with Sylon-CT™ (Sigma-Aldrich, St. Louis, MO, #33065-U), rinsed with methanol and dried. After expectoration was completed the culture tubes were sealed with a plastic screw cap containing a PTFE-liner (Thermo Fischer, #4506615), wrapped with para-film and stored refrigerated. Within 1 week of collection, specimens were shipped overnight in refrigerated containers for analysis.
Oral fluid specimens were analyzed by Immunalysis Corporation (Pomona, CA) using ELISA and LC–MS-MS testing methods. Analyses included two ELISA kits and LC–MS-MS upon receipt at the laboratory. The Saliva/Oral Fluids Cannabinoids ELISA Kit was tested according to the manufacturer's recommended procedure at a cutoff concentration for THC of 4 ng/mL. Cross-reactivities for this assay, as listed in the manufacturer's brochure, were: THC (100%); Δ8-THC (66.7%); CBN (4%); CBD (50%); and conjugated-THC (25%). The Ultra-Sensitive Cannabinoids ELISA Kit was tested according to the manufacturer's procedure at a cutoff concentration for THCCOOH of 0.05 ng/mL. Cross-reactivities for this assay, as listed in the manufacturer's brochure, were: THCCOOH (100%); 11-nor-9-carboxy- Δ8-THC (125%); Δ9-THC (10%); 11-OH-THC (33%); CBN ( <0.25%); and CBD ( <0.25%).
LC–MS-MS analyses for THC and THCCOH were conducted according to the methods described by Coulter et al. (9). The limit of quantitation (LOQ) and upper limit of linearity (ULOL), respectively, for oral fluid analyses were: THC, 0.5, 100 ng/mL; and THCCOOH, 0.02, 0.1 ng/mL. Control samples were prepared from Cerillant (Round Rock, TX) solutions. The inter-day ranges (5 days; n = 6/day) of percent deviation from the target concentration of control samples prepared for oral fluid analyses were: THC, 12 ng/mL (n = 30), −5.8 to 10.0%; and THCCOOH, 0.075 ng/mL (n = 30), −4.0 to 2.7%. The intra-run precisions for oral fluid control samples were: THC, 12 ng/mL (n = 6), 0.9%; and THCCOOH, 0.075 ng/mL (n = 6), 1.6%.
Physiological assessments
Vital signs (heart rate, systolic blood pressure (SBP), diastolic blood pressure (DBP)) were measured using automated monitors with participants in the seated position at Baseline, and at 0.17, 0.5, 1, 1.5, 2, 3, 4, 5, 6, 8, 12, 22, 26, 30, 34, 46, 50, 54, 58, 70, 74, 78, 82, 94, 98, 102, 106, 118, 122, 126 and 130 h after oral cannabis administration.
Subjective drug effect assessments
Participant-rated drug effects were measured using a 15-item Drug Effect Questionnaire (DEQ). Individual items on the DEQ included three ratings of drug effects (“Do you feel a drug effect?,” “Do you feel a pleasant drug effect?,” “Do you feel an unpleasant drug effect?”) and 12 ratings of behavioral/mood states often associated with cannabis intoxication (“sick”, “heart racing”, “anxious”, “relaxed”, “paranoid”, “tired”, “alert”, “irritable”, “vigorous”, “restless”, “hungry/have munchies”), and a question of whether they were “craving” cannabis. Each item was rated using a unipolar 100 mm visual analog scale (VAS) anchored with “not at all” on one end and “extremely” on the other. The DEQ was administered at Baseline, and at 0.5, 1, 1.5, 2, 3, 4, 5, 6 and 8 h after oral cannabis administration. Measurement stopped at the 8-hour time point because subjective drug effects following a single acute dose were not expected to persist beyond that time.
Cognitive/psychomotor performance assessments
Participants completed three different computerized behavioral tasks to assess aspects of psychomotor/cognitive performance known to be sensitive to the acute effects of smoked cannabis and relevant to functioning in the workplace and/or operating a motor vehicle or heavy machinery (13–15). Participants completed each task under the supervision of study staff three times during the screening visit in order to ensure proper understanding and to minimize the influence of practice effects on task performance during the study. Tasks included a computerized version of the Digit Symbol Substitution Task (DSST; 16), Divided Attention Task (DAT; 17), and a computerized version of the Paced Auditory Serial Addition Task (PASAT; 18). For the DSST, participants replicated the shape of patterns presented to them on a computer screen using a computer keyboard. The duration of this task was 90 s. Primary outcomes are the number of patterns attempted, number correct and accuracy within the allotted time. The DAT required participants simultaneously to perform a central motor tracking task (tracking a horizontally moving stimulus with the computer cursor) and to respond to visual stimuli presented in the periphery of the computer screen (clicking the computer mouse button each time a number one corner of the computer monitor matched a target number presented at the bottom of the computer monitor). Primary outcomes are the mean distance of the cursor from the central target stimulus during the task, the number of peripheral stimuli correctly identified, and the response time for peripheral stimulus recognition. For the PASAT, participants viewed a string of single digit integers on a computer screen. They were instructed to sum the 2 integers most recently presented and respond by selecting the answer using the computer mouse on the screen. The primary outcome for the task is the total number of correct trials during the task. Performance assessments were completed as a single test battery at baseline and 1, 1.5, 2, 3, 4, 5, 6 and 8 h after oral cannabis administration. The order of the tasks was constant and reflects the order in which they are presented in this report.
Data presentation and analysis
Participant demographics and biological specimen test results are presented using descriptive statistics. Agreement between ELISA testing and LC–MS-MS confirmation testing was conducted for blood THC results and oral fluid THC and THCCOOH test results. For whole blood, a THC metabolite screening cutoff of 10 ng/mL and confirmatory test cutoff concentration of 1 ng/mL were used. For oral fluid, a screening cutoff of 4 ng/mL was used for THC and THCCOOH, and confirmatory test cutoff concentration of 1 ng/mL was used for THC, and a 0.050 ng/mL cutoff was used for THCCOOH. Individual tests were designated as true-positive (TP; ELISA response ≥ cutoff concentration and LC–MS-MS positive), true-negative (TN; ELISA response < cutoff concentration and LC–MS-MS negative), false-positive (FP; ELISA response ≥ cutoff concentration and LC–MS-MS negative) or false-negative (FN; ELISA response < cutoff concentration and LC–MS-MS positive) based on screening and confirmation test outcomes. Sensitivity (100 × [TP/(TP + FN)]), specificity (100 × [TN/(TN + FP)]) and agreement (100 × [(TP + TN)/(TP + TN + FP + FN)]) were then calculated to summarize individual outcomes.
Cardiovascular, subjective and cognitive/psychomotor performance outcomes were analyzed using repeated-measures regressions with a compound symmetry covariance structure. Main effects of oral cannabis were assessed by comparing baseline values with peak values obtained after drug administration. Main effects of dose (10, 25 and 50 mg THC) were assessed by evaluating peak effects across each dose condition. This analytic approach was selected to maximize the likelihood of detecting drug and dose effects on variables of interest given the small sample size, between-subjects design and absence of a placebo condition. Correlations were conducted between quantitative blood and oral fluid cannabinoid (THC, 11-OH-THC and THCCOOH) levels and subjective ratings of Drug Effect, DSST Percent Correct, PASAT Number Correct and Divided Attention Mean Distance from Target at each dose. A multiple regression procedure that allows for calculation of correlation coefficients with repeated within-subjects observations was used (19), and change from baseline scores were utilized for cognitive task outcomes due to baseline variability in performance. Analyses were conducted in SAS PROC MIXED, version 9.4. and SPSS version 23.
Results
Pharmacokinetics outcomes
Prior to each session, all baseline whole blood and oral fluid specimens tested negative for cannabinoids by ELISA and LC–MS-MS, indicating compliance with the pre-study cannabis abstinence requirements. Complete quantitative and qualitative test results to the last positive (for any measure) for whole blood and oral fluid analyses for each participant are presented in Table I. Aggregate data on THC, 11-OH-THC and THCCOOH concentrations over time by cannabis dose in whole blood and oral fluid are provided in Figures 1–4. Mean maximum concentration (Cmax) and mean time to maximum concentration (Tmax) of THC, 11-OH-THC and THCCOOH (free) in whole blood and THC and THCCOOH (hydrolyzed) in oral fluid are summarized in Table II.
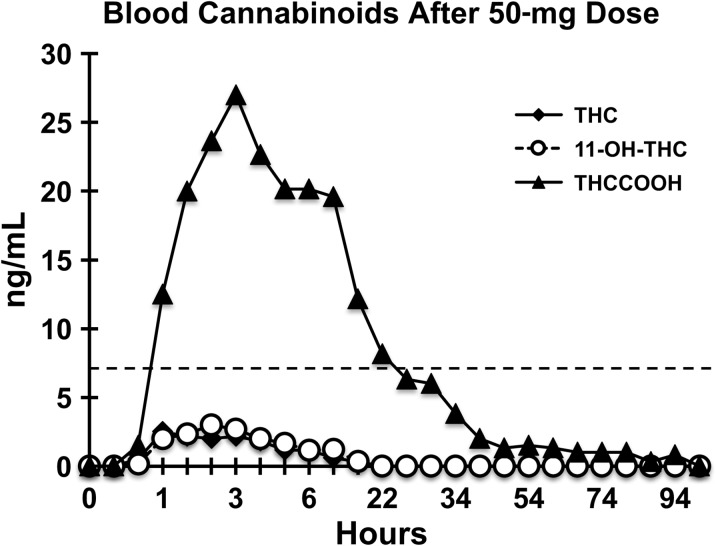
Figure 2.
Cannabinoid profile in whole blood after oral administration of 50 mg THC.
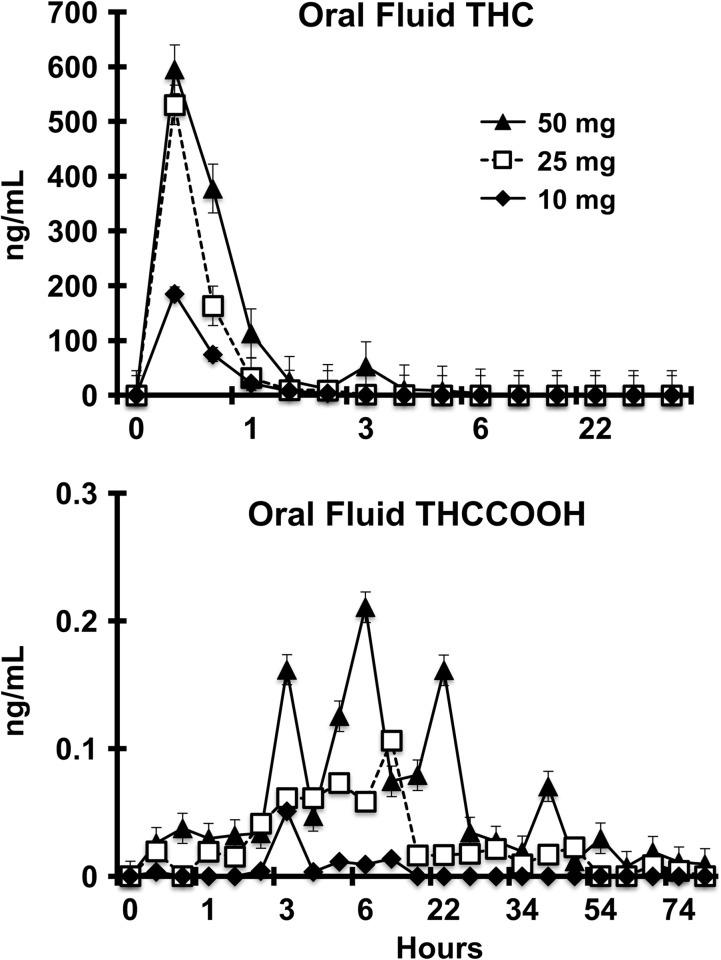
Figure 3.
Quantitative THC and THCCOOH in oral fluid.
Table I.
Analyses of blood and oral fluid specimens following oral consumption of cannabis brownies
| Subject # | Time (h)a | THC dose (mg) | Blood THCCOOH ELISA (cutoff = 10 ng/mL)b | Blood THC LC–MS-MS (ng/mL) | Blood 11-OH-THC LC–MS-MS (ng/mL) | Blood THCCOOH LC–MS-MS(ng/mL) | Oral fluid THC ELISA (cutoff = 4 ng/mL) | Oral fluid THCCOOH ELISA (cutoff = 0.050 ng/mL) | Oral fluid THC LC–MS-MS (ng/mL) | Oral fluid THCCOOH LC–MS-MS (ng/mL) |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | −1 | 10 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 1 | 0.17 | 10 | NEG | 0 | 0 | 0 | POS | POS | 144 | 0 |
| 1 | 0.5 | 10 | NEG | 0 | 0 | 0 | POS | POS | 18 | 0 |
| 1 | 1 | 10 | NEG | 0 | 0 | 0 | POS | NEG | 2 | 0 |
| 1 | 1.5 | 10 | POS | 0 | 0 | 0 | POS | NEG | 2 | 0 |
| 1 | 2 | 10 | POS | 0 | 0 | 4 | NEG | NEG | 0 | 0 |
| 1 | 3 | 10 | POS | 0 | 0 | 5 | NEG | NEG | 0 | 0 |
| 1 | 4 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 1 | 5 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 1 | 6 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 1 | 8 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 1 | 12 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 1 | 22 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 1 | 26 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 1 | 30 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 1 | 34 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 6 | −1 | 10 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 6 | 0.17 | 10 | NEG | 0 | 0 | 0 | POS | POS | 263 | 0 |
| 6 | 0.5 | 10 | NEG | 0 | 0 | 0 | POS | NEG | 80 | 0 |
| 6 | 1 | 10 | POS | 1 | 1 | 6 | POS | POS | 28 | 0 |
| 6 | 1.5 | 10 | POS | 1 | 1 | 8 | POS | NEG | 14 | 0 |
| 6 | 2 | 10 | POS | 1 | 0 | 6 | POS | NEG | 5 | 0 |
| 6 | 3 | 10 | POS | 1 | 1 | 10 | NEG | NEG | 2 | 0.231 |
| 6 | 4 | 10 | POS | 1 | 1 | 14 | NEG | NEG | 0 | 0 |
| 6 | 5 | 10 | POS | 1 | 0 | 8 | NEG | NEG | 0 | 0.045 |
| 6 | 6 | 10 | POS | 0 | 0 | 5 | NEG | NEG | 0 | 0.029 |
| 6 | 8 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0.037 |
| 6 | 12 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 6 | 22 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 6 | 26 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 6 | 30 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 12 | −1 | 10 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 12 | 0.17 | 10 | NEG | 0 | 0 | 0 | POS | POS | 47 | 0 |
| 12 | 0.5 | 10 | NEG | 0 | 0 | 0 | POS | NEG | 3 | 0 |
| 12 | 1 | 10 | NEG | 1 | 1 | 0 | POS | NEG | 2 | 0 |
| 12 | 1.5 | 10 | POS | 2 | 1 | 0 | POS | NEG | 4 | 0 |
| 12 | 2 | 10 | POS | 3 | 2 | 0 | NEG | NEG | 0 | 0 |
| 12 | 3 | 10 | POS | 3 | 2 | 5 | NEG | NEG | 0 | 0 |
| 12 | 4 | 10 | POS | 0.8 | 1 | 0 | NEG | NEG | 0 | 0 |
| 12 | 5 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 12 | 6 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 12 | 8 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 12 | 12 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 12 | 22 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 12 | 26 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 12 | 30 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 12 | 34 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 15 | −1 | 10 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 15 | 0.17 | 10 | NEG | 0 | 0 | 0 | POS | POS | 412 | 0.021 |
| 15 | 0.5 | 10 | NEG | 0 | 0 | 0 | POS | NEG | 139 | 0 |
| 15 | 1 | 10 | NEG | 0 | 1 | 3 | POS | NEG | 52 | 0 |
| 15 | 1.5 | 10 | NEG | 1 | 1 | 4 | POS | NEG | 8 | 0 |
| 15 | 2 | 10 | POS | 1 | 1 | 5 | NEG | NEG | 2 | 0.025 |
| 15 | 3 | 10 | POS | 0 | 1 | 5 | NEG | NEG | 0 | 0.074 |
| 15 | 4 | 10 | POS | 0 | 0 | 4 | NEG | NEG | 0 | 0.02 |
| 15 | 5 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0.024 |
| 15 | 6 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0.026 |
| 15 | 8 | 10 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0.044 |
| 15 | 12 | 10 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 15 | 22 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 20 | −1 | 10 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 20 | 0.17 | 10 | NEG | 0 | 0 | 0 | POS | POS | 161 | 0 |
| 20 | 0.5 | 10 | NEG | 0 | 0 | 0 | POS | POS | 199 | 0 |
| 20 | 1 | 10 | POS | 0.8 | 1 | 5 | POS | POS | 48 | 0 |
| 20 | 1.5 | 10 | POS | 0 | 1 | 6 | POS | NEG | 15 | 0 |
| 20 | 2 | 10 | POS | 0 | 0 | 6 | POS | POS | 15 | 0 |
| 20 | 3 | 10 | POS | 0 | 0 | 8 | NEG | NEG | 0 | 0 |
| 20 | 4 | 10 | POS | 0 | 0 | 5 | NEG | NEG | 0 | 0 |
| 20 | 5 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 20 | 6 | 10 | POS | 0 | 0 | 5 | NEG | NEG | 0 | 0 |
| 20 | 8 | 10 | POS | 0 | 0 | 4 | NEG | NEG | 0 | 0 |
| 20 | 12 | 10 | POS | 0 | 0 | 3 | NEG | NEG | 0 | 0 |
| 20 | 22 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 20 | 26 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 20 | 30 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 20 | 34 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 20 | 46 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 20 | 50 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 20 | 54 | 10 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 22 | −1 | 10 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 22 | 0.17 | 10 | NEG | 0 | 0 | 0 | POS | POS | 84 | 0 |
| 22 | 0.5 | 10 | NEG | 0 | 0 | 0 | POS | NEG | 8 | 0 |
| 22 | 1 | 10 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 22 | 1.5 | 10 | NEG | 0 | 0 | 0 | NEG | NEG | 2 | 0 |
| 22 | 2 | 10 | POS | 0 | 0 | 3 | NEG | NEG | 0 | 0 |
| 22 | 3 | 10 | POS | 0 | 1 | 5 | NEG | NEG | 0 | 0 |
| 22 | 4 | 10 | POS | 0 | 0 | 6 | NEG | NEG | 0 | 0 |
| 22 | 5 | 10 | POS | 0 | 0 | 4 | NEG | NEG | 0 | 0 |
| 22 | 6 | 10 | POS | 0 | 0 | 3 | NEG | NEG | 0 | 0 |
| 22 | 8 | 10 | POS | 0 | 0 | 3 | NEG | NEG | 0 | 0 |
| 22 | 12 | 10 | NEG | 0 | 0 | 2 | NEG | NEG | 0 | 0 |
| 5 | −1 | 25 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 5 | 0.17 | 25 | NEG | 0 | 0 | 0 | POS | POS | 1128 | 0.098 |
| 5 | 0.5 | 25 | NEG | 0 | 0 | 0 | POS | POS | 310 | 0 |
| 5 | 1 | 25 | POS | 1 | 0 | 7 | POS | NEG | 49 | 0 |
| 5 | 1.5 | 25 | POS | 1 | 1 | 12 | POS | NEG | 30 | 0 |
| 5 | 2 | 25 | POS | 2 | 1 | 15 | POS | POS | 19 | 0.135 |
| 5 | 3 | 25 | POS | 1 | 1 | 20 | POS | POS | 3 | 0.251 |
| 5 | 4 | 25 | POS | 3 | 2 | 20 | NEG | POS | 0 | 0 |
| 5 | 5 | 25 | POS | 1 | 0 | 14 | NEG | POS | 0 | 0.217 |
| 5 | 6 | 25 | POS | 1 | 0 | 16 | NEG | POS | 0 | 0.153 |
| 5 | 8 | 25 | POS | 0 | 0 | 5 | NEG | NEG | 0 | 0.081 |
| 5 | 12 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 5 | 22 | 25 | POS | 0 | 0 | 0 | NEG | POS | 0 | 0 |
| 5 | 26 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 5 | 30 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0.027 |
| 5 | 34 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 5 | 46 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 5 | 50 | 25 | POS | 0 | 0 | 0 | NEG | POS | 0 | 0.096 |
| 5 | 54 | 25 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 5 | 58 | 25 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 5 | 70 | 25 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0.053 |
| 5 | 74 | 25 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0.026 |
| 8 | −1 | 25 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 8 | 0.17 | 25 | NEG | 0 | 0 | 0 | NS | NS | NS | NS |
| 8 | 0.5 | 25 | NEG | 2 | 2 | 7 | POS | POS | 213 | 0 |
| 8 | 1 | 25 | POS | 2 | 3 | 21 | NS | NS | NS | NS |
| 8 | 1.5 | 25 | MS | NS | NS | NS | NS | NS | NS | NS |
| 8 | 2 | 25 | POS | 1 | 2 | 27 | POS | NEG | 23 | 0.115 |
| 8 | 3 | 25 | POS | 4 | 5 | 39 | POS | NEG | 4 | 0.092 |
| 8 | 4 | 25 | POS | 1 | 2 | 22 | POS | NEG | 0 | 0.091 |
| 8 | 5 | 25 | POS | 2 | 3 | 28 | NEG | NEG | 0 | 0.079 |
| 8 | 6 | 25 | POS | 0 | 4 | 39 | NEG | POS | 2 | 0.209 |
| 8 | 8 | 25 | POS | 0 | 2 | 25 | NEG | NEG | 0 | 0.061 |
| 8 | 12 | 25 | POS | 0 | 0 | 11 | NEG | NEG | 0 | 0.038 |
| 8 | 22 | 25 | POS | 0 | 0 | 12 | NEG | NEG | 0 | 0.204 |
| 8 | 26 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 8 | 30 | 25 | POS | 0 | 0 | 8 | NEG | NEG | 0 | 0 |
| 8 | 34 | 25 | POS | 0 | 0 | 8 | NEG | NEG | 0 | 0 |
| 8 | 46 | 25 | POS | 0 | 0 | 9 | NEG | NEG | 0 | 0 |
| 8 | 50 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 8 | 54 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 8 | 58 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 9 | −1 | 25 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 9 | 0.17 | 25 | NEG | 0 | 0 | 0 | POS | POS | 70 | 0 |
| 9 | 0.5 | 25 | POS | 1 | 1 | 3 | POS | POS | 51 | 0 |
| 9 | 1 | 25 | POS | 2 | 1 | 9 | POS | NEG | 12 | 0 |
| 9 | 1.5 | 25 | POS | 2 | 1 | 9 | POS | NEG | 2 | 0 |
| 9 | 2 | 25 | POS | 3 | 1 | 14 | POS | NEG | 2 | 0 |
| 9 | 3 | 25 | POS | 2 | 1 | 15 | NEG | NEG | 0 | 0 |
| 9 | 4 | 25 | POS | 2 | 2 | 16 | NEG | NEG | 0 | 0.032 |
| 9 | 5 | 25 | POS | 0 | 1 | 11 | NEG | NEG | 0 | 0 |
| 9 | 6 | 25 | POS | 0 | 0 | 10 | NEG | NEG | 0 | 0 |
| 9 | 8 | 25 | POS | 0 | 0 | 8 | NEG | POS | 0 | 0.101 |
| 9 | 12 | 25 | POS | 0 | 0 | 8 | NEG | NEG | 0 | 0 |
| 9 | 22 | 25 | POS | 0 | 0 | 5 | NEG | NEG | 0 | 0 |
| 9 | 26 | 25 | POS | 0 | 0 | 6 | NEG | NEG | 0 | 0 |
| 9 | 30 | 25 | POS | 0 | 0 | 2 | NEG | NEG | 0 | 0 |
| 9 | 34 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 9 | 46 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 9 | 50 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 11 | −1 | 25 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 11 | 0.17 | 25 | NEG | 0 | 0 | 0 | POS | POS | 400 | 0 |
| 11 | 0.5 | 25 | POS | 0 | 2 | 0 | POS | POS | 125 | 0 |
| 11 | 1 | 25 | POS | 4 | 4 | 17 | NS | NS | 47 | 0 |
| 11 | 1.5 | 25 | POS | 2 | 5 | 23 | POS | NEG | 4 | 0.021 |
| 11 | 2 | 25 | POS | 1 | 2 | 19 | POS | NEG | 6 | 0.037 |
| 11 | 3 | 25 | POS | 1 | 2 | 13 | NEG | NEG | 0 | 0.036 |
| 11 | 4 | 25 | POS | 0 | 1 | 12 | NEG | NEG | 0 | 0.055 |
| 11 | 5 | 25 | POS | 0 | 0 | 12 | NEG | NEG | 0 | 0.086 |
| 11 | 6 | 25 | POS | 0 | 0 | 10 | NEG | NEG | 0 | 0.103 |
| 11 | 8 | 25 | POS | 1 | 1 | 9 | NS | NS | NS | NS |
| 11 | 12 | 25 | POS | 0 | 0 | 7 | NEG | NEG | 0 | 0.057 |
| 11 | 22 | 25 | POS | 0 | 0 | 4 | NEG | POS | 0 | 0.098 |
| 11 | 26 | 25 | POS | 0 | 0 | 4 | NEG | POS | 0 | 0.1 |
| 11 | 30 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0.106 |
| 11 | 34 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0.102 |
| 11 | 46 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0.057 |
| 11 | 50 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0.102 |
| 11 | 54 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0.041 |
| 11 | 58 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 11 | 70 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 11 | 74 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 25 | −1 | 25 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 25 | 0.17 | 25 | NEG | 0 | 0 | 0 | POS | POS | 110 | 0 |
| 25 | 0.5 | 25 | NEG | 0 | 0 | 0 | POS | POS | 57 | 0 |
| 25 | 1 | 25 | POS | 0 | 0 | 0 | POS | POS | 36 | 0 |
| 25 | 1.5 | 25 | POS | 0 | 0 | 0 | NS | NS | 18 | 0 |
| 25 | 2 | 25 | POS | 1 | 1 | 8 | NS | NS | 15 | 0 |
| 25 | 3 | 25 | POS | 3 | 2 | 16 | NEG | NEG | 3 | 0 |
| 25 | 4 | 25 | POS | 4 | 3 | 18 | NEG | NEG | 0 | 0.067 |
| 25 | 5 | 25 | POS | 2 | 2 | 15 | NEG | NEG | 0 | 0.033 |
| 25 | 6 | 25 | POS | 0 | 0 | 2 | NEG | NEG | 0 | 0.073 |
| 25 | 8 | 25 | POS | 0 | 0 | 5 | NEG | POS | 0 | 0.148 |
| 25 | 12 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 25 | 22 | 25 | POS | 1 | 0 | 5 | NEG | NEG | 0 | 0 |
| 25 | 26 | 25 | POS | 0 | 0 | 8 | NEG | NEG | 0 | 0 |
| 25 | 30 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 25 | 34 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 27 | −1 | 25 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 27 | 0.17 | 25 | NEG | 0 | 0 | 0 | POS | POS | 944 | 0 |
| 27 | 0.5 | 25 | NEG | 0 | 0 | 0 | POS | POS | 224 | 0 |
| 27 | 1 | 25 | POS | 0 | 0 | 0 | POS | NEG | 22 | 0 |
| 27 | 1.5 | 25 | POS | 3 | 3 | 12 | POS | NEG | 0 | 0 |
| 27 | 2 | 25 | POS | 3 | 2 | 12 | POS | NEG | 10 | 0 |
| 27 | 3 | 25 | POS | 0 | 2 | 10 | NEG | NEG | 0 | 0 |
| 27 | 4 | 25 | POS | 0 | 0 | 8 | NEG | NEG | 0 | 0.023 |
| 27 | 5 | 25 | POS | 0 | 0 | 7 | NEG | NEG | 0 | 0 |
| 27 | 6 | 25 | POS | 0 | 0 | 8 | NEG | NEG | 0 | 0 |
| 27 | 8 | 25 | POS | 0 | 0 | 5 | NEG | NEG | 0 | 0 |
| 27 | 12 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 27 | 22 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 27 | 26 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 27 | 30 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 27 | 34 | 25 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 4 | −1 | 50 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0.062 |
| 4 | 0.17 | 50 | NEG | 0 | 0 | 0 | POS | POS | 479 | 0.122 |
| 4 | 0.5 | 50 | NEG | 0 | 0 | 0 | POS | POS | 492 | NS |
| 4 | 1 | 50 | POS | 1 | 2 | 12 | NS | NS | NS | NS |
| 4 | 1.5 | 50 | POS | 0 | 1 | 10 | NS | NS | NS | 0.067 |
| 4 | 2 | 50 | POS | 1 | 2 | 16 | POS | POS | 29 | 0 |
| 4 | 3 | 50 | POS | 1 | 2 | 11 | POS | POS | 168 | 0.052 |
| 4 | 4 | 50 | POS | 0 | 1 | 10 | POS | NEG | 24 | 0 |
| 4 | 5 | 50 | POS | 0 | 1 | 8 | POS | NEG | 3 | 0 |
| 4 | 6 | 50 | POS | 0 | 0 | 11 | POS | NEG | 2 | 0 |
| 4 | 8 | 50 | NS | NS | NS | NS | POS | NEG | 2 | 0 |
| 4 | 12 | 50 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 4 | 22 | 50 | POS | 0 | 0 | 0 | POS | NEG | 0 | 0 |
| 7 | −1 | 50 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 7 | 0.17 | 50 | NEG | 0 | 0 | 0 | POS | POS | 636 | 0 |
| 7 | 0.5 | 50 | NEG | 0 | 0 | 0 | POS | POS | 225 | 0 |
| 7 | 1 | 50 | POS | 4 | 1 | 9 | POS | POS | 162 | 0 |
| 7 | 1.5 | 50 | POS | 5 | 2 | 18 | POS | POS | 10 | 0.037 |
| 7 | 2 | 50 | POS | 5 | 3 | 27 | POS | POS | 3 | 0.08 |
| 7 | 3 | 50 | POS | 0 | 2 | 23 | POS | POS | 8 | 0.048 |
| 7 | 4 | 50 | POS | 2 | 1 | 17 | NEG | NEG | 0 | 0.048 |
| 7 | 5 | 50 | POS | 1 | 0 | 12 | NEG | NEG | 0 | 0.063 |
| 7 | 6 | 50 | POS | 0 | 0 | 11 | NEG | NEG | 0 | 0.062 |
| 7 | 8 | 50 | POS | 0 | 0 | 11 | NEG | NEG | 0 | 0.114 |
| 7 | 12 | 50 | POS | 2 | 0 | 17 | NEG | NEG | 0 | 0.096 |
| 7 | 22 | 50 | POS | 0 | 0 | 12 | NEG | NEG | 0 | 0.12 |
| 7 | 26 | 50 | POS | 0 | 0 | 9 | NEG | POS | 0 | 0.114 |
| 7 | 30 | 50 | POS | 0 | 0 | 8 | NEG | POS | 0 | 0.073 |
| 7 | 34 | 50 | POS | 0 | 0 | 7 | NEG | NEG | 0 | 0.087 |
| 7 | 46 | 50 | POS | 0 | 0 | 6 | NEG | NEG | 0 | 0.071 |
| 7 | 50 | 50 | POS | 0 | 0 | 4 | NEG | POS | 0 | 0.137 |
| 7 | 54 | 50 | POS | 0 | 0 | 5 | NEG | POS | 0 | 0.045 |
| 7 | 58 | 50 | POS | 0 | 0 | 4 | NEG | NEG | 0 | 0.096 |
| 7 | 70 | 50 | POS | 0 | 0 | 4 | NEG | POS | 0 | 0.028 |
| 7 | 74 | 50 | POS | 0 | 0 | 3 | NEG | NEG | 0 | 0.058 |
| 7 | 78 | 50 | POS | 0 | 0 | 3 | NEG | NEG | 0 | 0.042 |
| 7 | 82 | 50 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0.032 |
| 7 | 94 | 50 | POS | 0 | 0 | 3 | NEG | NEG | 0 | 0.021 |
| 7 | 98 | 50 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0.02 |
| 7 | 102 | 50 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 7 | 106 | 50 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0.04 |
| 7 | 118 | 50 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0.042 |
| 7 | 122 | 50 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0.04 |
| 7 | 126 | 50 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 17 | −1 | 50 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 17 | 0.17 | 50 | NEG | 0 | 0 | 0 | POS | POS | 378 | 0 |
| 17 | 0.5 | 50 | NEG | 0 | 0 | 0 | POS | POS | 160 | NS |
| 17 | 1 | 50 | POS | 1 | 2 | 11 | NS | NS | NS | NS |
| 17 | 1.5 | 50 | POS | 1 | 3 | 22 | NS | NS | NS | NS |
| 17 | 2 | 50 | POS | 1 | 4 | 27 | NS | NS | NS | NS |
| 17 | 3 | 50 | POS | 1 | 3 | 34 | POS | POS | NS | NS |
| 17 | 4 | 50 | POS | 1 | 2 | 30 | NS | NS | NS | 0 |
| 17 | 5 | 50 | POS | 1 | 3 | 31 | POS | POS | 16 | 0 |
| 17 | 6 | 50 | POS | 0 | 2 | 26 | POS | POS | 6 | 0 |
| 17 | 8 | 50 | POS | 0 | 1 | 20 | NEG | POS | 0 | 0 |
| 17 | 12 | 50 | POS | 0 | 1 | 18 | NEG | NEG | 0 | 0 |
| 17 | 22 | 50 | POS | 0 | 0 | 12 | NEG | NEG | 0 | 0 |
| 17 | 26 | 50 | POS | 0 | 0 | 8 | NEG | NEG | 0 | 0 |
| 17 | 30 | 50 | POS | 0 | 0 | 14 | NEG | NEG | 0 | 0 |
| 17 | 34 | 50 | POS | 0 | 0 | 4 | NEG | NEG | 0 | 0 |
| 17 | 46 | 50 | POS | 0 | 0 | 3 | NEG | NEG | 0 | 0 |
| 17 | 50 | 50 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 17 | 54 | 50 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 17 | 58 | 50 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 17 | 70 | 50 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 17 | 74 | 50 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 19 | −1 | 50 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0.051 |
| 19 | 0.17 | 50 | NEG | 0 | 0 | 0 | POS | POS | 719 | 0.04 |
| 19 | 0.5 | 50 | NEG | 1 | 0 | 0 | POS | POS | 349 | 0.089 |
| 19 | 1 | 50 | POS | 4 | 3 | 10 | NS | NS | 75 | 0.129 |
| 19 | 1.5 | 50 | POS | 2 | 2 | 10 | NS | NS | 80 | NS |
| 19 | 2 | 50 | POS | 3 | 3 | 13 | NEG | NEG | NS | 0.451 |
| 19 | 3 | 50 | POS | 4 | 4 | 22 | POS | POS | 24 | NS |
| 19 | 4 | 50 | POS | 3 | 4 | 19 | NS | NS | NS | 0.367 |
| 19 | 5 | 50 | POS | 3 | 3 | 13 | POS | POS | 5 | 0.359 |
| 19 | 6 | 50 | POS | 3 | 3 | 14 | POS | POS | 2 | 0.269 |
| 19 | 8 | 50 | POS | 1 | 2 | 9 | POS | POS | 2 | 0.102 |
| 19 | 12 | 50 | POS | 0 | 0 | 8 | NEG | NEG | 0 | 0.642 |
| 19 | 22 | 50 | POS | 0 | 0 | 8 | POS | POS | 2 | 0.125 |
| 19 | 26 | 50 | POS | 0 | 0 | 6 | NEG | POS | 0 | NS |
| 19 | 30 | 50 | POS | 0 | 0 | 5 | NEG | NEG | 0 | 0.044 |
| 19 | 34 | 50 | POS | 0 | 0 | 4 | NEG | NEG | 0 | 0.264 |
| 19 | 46 | 50 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 19 | 50 | 50 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0.041 |
| 19 | 54 | 50 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 19 | 58 | 50 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0.18 |
| 19 | 70 | 50 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0.038 |
| 19 | 74 | 50 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 23 | 0 | 50 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0.044 |
| 23 | 0.17 | 50 | NEG | 0 | 0 | 0 | POS | POS | 1010 | 0.063 |
| 23 | 0.5 | 50 | POS | 1 | 0 | 5 | POS | POS | 851 | 0.029 |
| 23 | 1 | 50 | POS | 2 | 1 | 16 | POS | POS | 196 | 0 |
| 23 | 1.5 | 50 | POS | 3 | 2 | 33 | POS | NEG | 4 | 0 |
| 23 | 2 | 50 | POS | 0 | 2 | 29 | POS | POS | 2 | 0.116 |
| 23 | 3 | 50 | POS | 5 | 2 | 32 | POS | POS | 11 | 0 |
| 23 | 4 | 50 | POS | 2 | 1 | 23 | POS | POS | 20 | 0 |
| 23 | 5 | 50 | POS | 1 | 1 | 26 | POS | POS | 24 | 0.02 |
| 23 | 6 | 50 | POS | 0 | 0 | 22 | NEG | NEG | 0 | 0.02 |
| 23 | 8 | 50 | POS | 0 | 0 | 14 | NEG | NEG | 0 | 0.099 |
| 23 | 12 | 50 | POS | 0 | 0 | 13 | NEG | POS | 2 | 0.163 |
| 23 | 22 | 50 | POS | 0 | 0 | 10 | NEG | POS | 0 | 0.052 |
| 23 | 26 | 50 | POS | 0 | 0 | 9 | NEG | NEG | 0 | 0 |
| 23 | 30 | 50 | POS | 0 | 0 | 5 | NEG | NEG | 0 | 0 |
| 23 | 34 | 50 | POS | 0 | 0 | 3 | NEG | NEG | 0 | 0 |
| 23 | 46 | 50 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 23 | 50 | 50 | POS | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 24 | −1 | 50 | NEG | 0 | 0 | 0 | NEG | NEG | 0 | 0 |
| 24 | 0.17 | 50 | NEG | 0 | 0 | 0 | POS | POS | 350 | 0 |
| 24 | 0.5 | 50 | NEG | 1 | 1 | 4 | POS | POS | 187 | 0 |
| 24 | 1 | 50 | POS | 3 | 3 | 17 | POS | NEG | 18 | 0 |
| 24 | 1.5 | 50 | POS | 2 | 4 | 27 | POS | NEG | 10 | 0.031 |
| 24 | 2 | 50 | POS | 2 | 4 | 30 | POS | NEG | 10 | NS |
| 24 | 3 | 50 | POS | 2 | 3 | 40 | NS | NS | NS | 0.089 |
| 24 | 4 | 50 | NEG | 3 | 3 | 37 | NEG | NEG | 2 | 0.337 |
| 24 | 5 | 50 | POS | 1 | 2 | 31 | NS | NS | 3 | 0.822 |
| 24 | 6 | 50 | POS | 4 | 2 | 37 | NS | NS | 7 | 0.095 |
| 24 | 8 | 50 | POS | 2 | 3 | 44 | NEG | NEG | 0 | 0.16 |
| 24 | 12 | 50 | POS | 0 | 1 | 17 | NEG | NEG | 0 | 0.067 |
| 24 | 22 | 50 | POS | 0 | 0 | 7 | NEG | POS | 0 | 0 |
| 24 | 26 | 50 | POS | 0 | 0 | 6 | NEG | NEG | 0 | 0.05 |
| 24 | 30 | 50 | POS | 0 | 0 | 4 | NEG | NEG | 0 | 0 |
| 24 | 34 | 50 | POS | 0 | 0 | 5 | NEG | NEG | 0 | 0.071 |
| 24 | 46 | 50 | POS | 0 | 0 | 3 | NEG | NEG | 0 | 0 |
| 24 | 50 | 50 | POS | 0 | 0 | 4 | NEG | NEG | 0 | 0 |
| 24 | 54 | 50 | POS | 0 | 0 | 4 | NEG | NEG | 0 | 0 |
| 24 | 58 | 50 | POS | 0 | 0 | 4 | NEG | NEG | 0 | 0 |
| 24 | 70 | 50 | NEG | 0 | 0 | 2 | NEG | NEG | 0 | 0 |
| 24 | 74 | 50 | POS | 0 | 0 | 3 | NEG | NEG | 0 | 0 |
| 24 | 78 | 50 | POS | 0 | 0 | 3 | NEG | NEG | 0 | 0 |
| 24 | 82 | 50 | NEG | 0 | 0 | 2 | NEG | NEG | 0 | 0 |
| 24 | 94 | 50 | NEG | 0 | 0 | 2 | NEG | NEG | 0 | 0 |
aData are tabulated over time to the last specimen that a positive ELISA response was obtained or a measurable quantity of a THC analyte was recorded.
bPOS = positive; NEG = negative; NS = no sample.
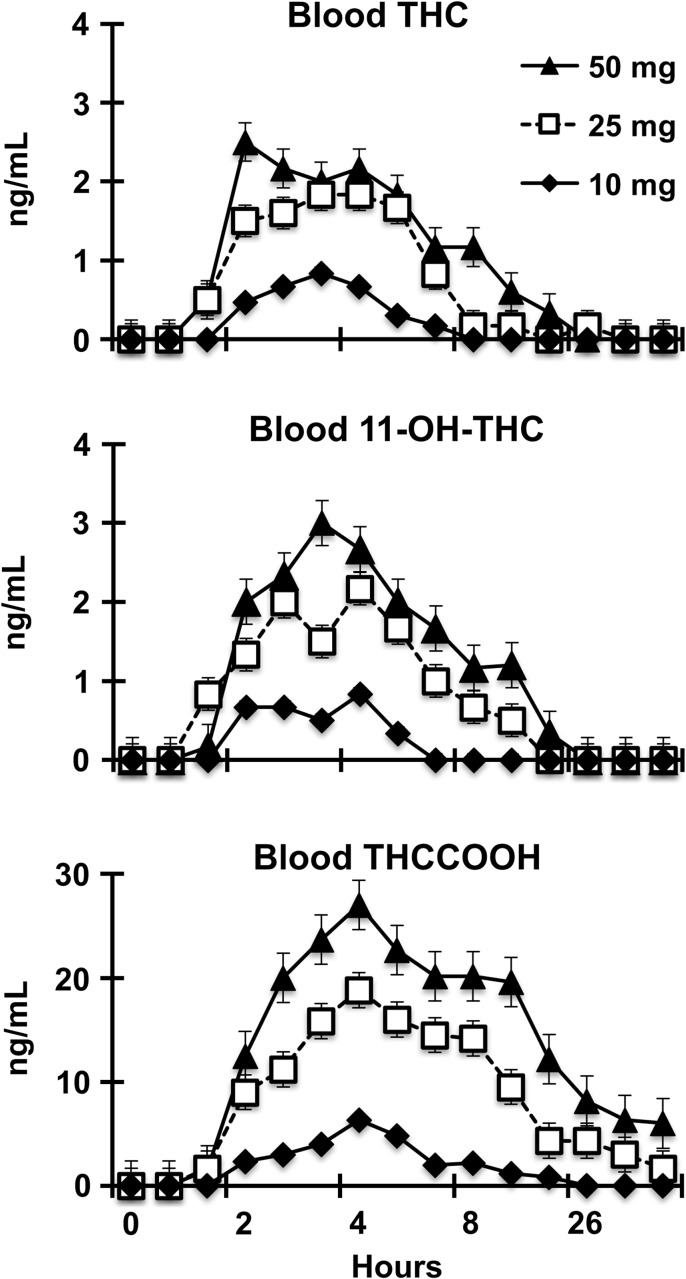
Figure 1.
Quantitative THC, 11-OH-THC and THCCOOH in whole blood.
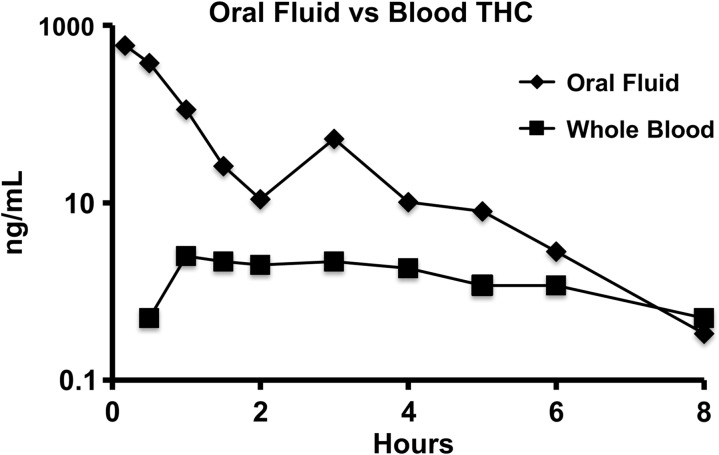
Figure 4.
THC in oral fluid versus whole blood following 50 mg dose.
Table II.
Average THC, 11-OH-THC, and THCCOOH maximum concentrations, times and individual ranges in blood and oral fluid following oral consumption of cannabis brownies
| Dose (mg) | THC Cmax (ng/mL) | THC Tmax (h) | 11-OH-THC Cmax (ng/mL) | 11-OH-THC Tmax (h) | THCCOOH Cmax (ng/mL) | THCCOOH Tmax (h) |
|---|---|---|---|---|---|---|
| Blood | ||||||
| 10 | 1.0 (0–3) | 0.9 (0–2) | 1.0 (0–2) | 1.3 (0–3) | 7.2 (5–14) | 3.2 (2–4) |
| 25 | 3.5 (3.0–4) | 2.6 (1.0–4) | 3.3 (2–5) | 3.0 (1.5–4) | 21.3 (12–39) | 3.3 (1.5–6) |
| 50 | 3.3 (1.0–5) | 2.3 (1.0–6) | 3.2 (2–4) | 1.8 (1–3) | 29.3 (16–44) | 3.3 (1.5–6) |
| Oral fluid | ||||||
| 10 | 191.5 (47–412) | 0.2 (0.2–0.5) | NT | NT | 0.051 (0–0.231) | 1.0 (0–3) |
| 25 | 477.5 (70–1128) | 0.2 (0.2–0.5) | NT | NT | 0.140 (0.023–0.251) | 9.8 (3–30) |
| 50 | 597.5 (350–1010) | 0.2 (0.2–0.5) | NT | NT | 0.314 (0–0.822) | 17.4 (0–54) |
THC generally became detectable in whole blood in the range of 0.5–2 h and declined thereafter over 2–12 h. This is consistent with the onset of self-reported drug effects and changes in behavioral task performance. Two participants completed the study with no detectable blood THC at any time point (10 mg dose; Participants #1 and #22), and five participants had blood THC concentrations that never exceeded 1 ng/mL (10 mg dose: Participants #6, #15 and #20; and 50 mg dose: Participants #4 and #17). Cmax blood concentration and Tmax of 11-OH-THC were generally similar to those observed for THC. Appearance of THCCOOH in blood tended to be slightly delayed relative to THC and persisted longer. Cmax blood concentrations of THCCOOH were generally higher than THC and had longer Tmax values.
In contrast to whole blood, THC in oral fluid was generally detectable in highest concentration immediately after cannabis consumption and declined thereafter over the next 2–22 h. Mean Cmax oral fluid concentrations of THCCOOH were considerably lower than THC and mean Tmax were observed at longer times than THC. The appearance of THCCOOH in oral fluid was erratic and not present in some participants (10 mg dose: Participants #1, #12, #20 and #22; and 50 mg dose: Participant #17), but was detectable for a longer time than THC.
Mean detection time windows and individual ranges to first positive and last positive across matrices for THC and metabolites are listed in Table III. The time to the first positive for THC and metabolites in whole blood were generally in the range of 0.5–2 h. Initial detection of THC in oral fluid was almost immediate (10 or 30 min collection), whereas the appearance of THCCOOH was generally delayed by 1–2 h.
Table III.
Average detection times of THC, metabolites and individual ranges in blood and oral fluid following oral consumption of cannabis brownies
| THC dose (mg) | Blood THCCOOH ELISA (h) (cutoff = 10 ng/mL | Blood THC LC–MS-MS (h) (LOQ = 0.5 ng/mL) | Blood 11-OH-THC LC–MS-MS (h) (LOQ = 0.5 ng/mL) | Blood THCCOOH LC–MS-MS (h) (LOQ = 0.5 ng/mL) | Oral fluid THC ELISA (h) (cutoff = 4 ng/mL) | Oral fluid THCCOOH ELISA (h) (cutoff = 4 ng/mL) | Oral fluid THC LC–MS-MS (h) (LOQ = 1 ng/mL) | Oral fluid THCCOOH LC–MS-MS (h) (cutoff = 0.050 ng/mL) |
|---|---|---|---|---|---|---|---|---|
| Detection time (h) to first positive, mean (range) | ||||||||
| 10 | 1.5 (1.0–2.0) | 1.1 (1.0–1.5) | 1.4 (1.0–3.0) | 1.7 (1.0–3.0) | 0.17 (0.2–0.2) | 0.17 (0.2–0.2) | 0.17 (0.2–0.2) | 3.0 (3.0–3.0) |
| 25 | 0.8 (0.5–1.0) | 1.1 (0.5–2.0) | 1.1 (0.5–2.0) | 1.1 (0.5–2.0) | 0.23 (0.2–0.5) | 0.23 (0.2–0.5) | 0.23 (0.2–0.5) | 3.6 (0.2–8.0) |
| 50 | 0.9 (0.5–1.0) | 0.8 (0.5–1.0) | 0.9 (0.5–1.0) | 0.8 (0.5–1.0) | 0.17 (0.2–0.2) | 0.17 (0.2–0.2) | 0.17 (0.2–0.2) | 1.6 (0.2–4.0) |
| Detection time (h) to last positive, mean (range) | ||||||||
| 10 | 30.3 (8–54) | 2.0 (0–5) | 2.6 (0–4) | 6.7 (3–12) | 1.5 (1,2) | 0.7 (0–2) | 1.9 (2,3) | 1.0 (0–3) |
| 25 | 50.0 (34–74) | 7.8 (4–22) | 5.5 (3–8) | 24.0 (8–46) | 2.3 (1–4) | 16.4 (1–50) | 3.0 (2–6) | 26.3 (0–70) |
| 50 | 66.7 (22–122) | 6.8 (3–12) | 7.7 (4–12) | 51.3 (6–94) | 10.0 (2–22) | 25.2 (3–70) | 9.5 (3–22) | 37.3 (0–78) |
Time to the last positive specimen were highly variable by assay type, matrix, analyte and by individual. Somewhat longer detection times were observed in whole blood with the ELISA THCCOOH (10 ng/mL cutoff) assay compared to measurement of THCCOOH (non-hydrolyzed) by LC–MS-MS (LOQ = 0.5 ng/mL), presumably because of ELISA cross-reactivity with conjugated THCCOOH. In contrast, detection times with the oral fluid ELISA THC (4 ng/mL cutoff) were generally comparable to measurement of THC by LC–MS-MS (LOQ = 1.0 ng/mL). Similar results were also observed with the oral fluid ELISA THCCOOH assay (0.05 ng/mL cutoff) compared to measurement of THCCOOH (hydrolyzed) by LC–MS-MS (LOQ = 0.02 ng/mL).
Mean detection time window (time from first positive to last positive) by LC–MS-MS for THC in blood were relatively short (2–8 h), but comparable to THC in oral fluid. Detection time window by LC–MS-MS for THCCOOH in blood were longer (7–51 h) than for THC, and were similar to those observed for THCCOOH in oral fluid. The time to last positive for THCCOOH in oral fluid for four participants was more than 2 days post-exposure, but in these cases not all preceding specimens were continuously positive indicating irregularity in the pharmacokinetic profile of this metabolite. All four of these participants received either 25 or 50 mg THC doses.
The disparity between the detection of THC and THCCOOH in oral fluid is shown in Table IV. Of the 148 specimens that tested positive for THC and/or THCCOOH, only 15.5% of the specimens collected during the first 8 h were positive for both analytes. During the same period, approximately one-half of the specimens were positive for THC and negative for THCCOOH and 12.2% were negative for THC and positive for THCCOOH. After 8 h, only 1.4% were jointly positive and 20.3% were negative for THC and positive for THCCOOH.
Table IV.
Comparison of positive/negative detection rates by LC–MS-MS for THC (2 ng/mL) and THCCOOH (0.050 ng/mL) in oral fluid in the first 8 h and thereafter following cannabis consumption (n = 148 specimens)
| Positive THC:Positive THCCOOH, N (%) | Positive THC:Negative THCCOOH, N (%) | Negative THC:Positive THCCOOH, N (%) | |
|---|---|---|---|
| 0–8 h | 23 (15.5) | 75 (50.7) | 18 (12.2) |
| >8 h | 2 (1.4) | 0 (0.0) | 30 (20.3) |
A summary of screening and confirmation testing agreement is provided in Table V. For whole blood, ELISA (10 ng/mL) and LC–MS-MS (0.5 ng/mL) were compared for 574 samples. The sensitivity, specificity and agreement of ELISA relative to LC–MS-MS was 95, 78 and 84%, respectively. For oral fluid, ELISA screening test (4 ng/mL) results were compared with THC (1 ng/mL) and THCCOOH (0.050 ng/mL) confirmation results (LC–MS-MS). The relative sensitivity, specificity and agreement of ELISA relative to mass spectrometry tests were 93, 99 and 98% with confirmation of THC at 1 ng/mL, and 46, 92 and 87% with confirmation of THCCOOH at 0.050 ng/mL.
Table V.
Comparisons of immunoassay responses to confirmation analyses in blood and oral fluid specimens following oral cannabis ingestion
| Blood THCCOOH ELISA (cutoff = 10 ng/mL) vs THCCOOH LC–MS-MS (cutoff = 2 ng/mL) | Oral Fluid THC ELISA (cutoff = 4 ng/mL) vs THC LC–MS-MS (cutoff = 2 ng/mL) | Oral Fluid THCCOOH ELISA (cutoff = 4 ng/mL) vs THCCOOH LC–MS-MS (cutoff = 0.050 ng/mL) | |
|---|---|---|---|
| #True positive (%) | 173 (30.1) | 87 (14.3) | 32 (5.3) |
| #True negative (%) | 307 (53.5) | 508 (88.5) | 495 (81.5) |
| #False positive (%) | 85 (14.8) | 5 (0.9) | 43 (7.1) |
| #False negative (%) | 9 (1.6) | 7 (1.2) | 37 (6.1) |
| N | 574 | 607 | 607 |
| % Sensitivity | 95.1 | 92.6 | 46.4 |
| % Specificity | 78.3 | 99.0 | 92.0 |
| % Agreement | 83.6 | 98.0 | 86.8 |
Pharmacodynamic outcomes
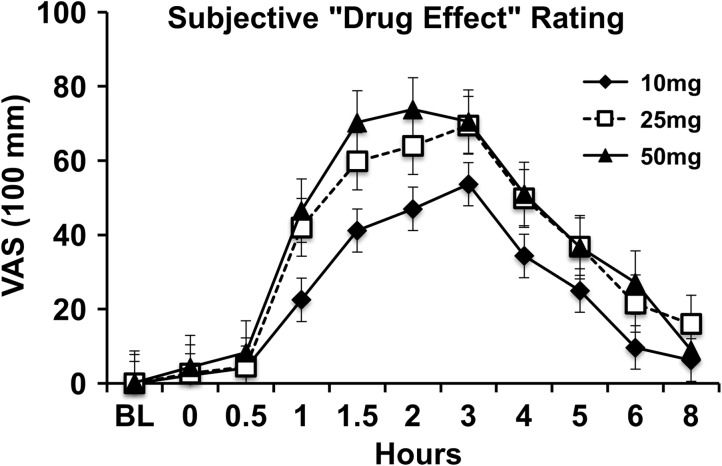
Significant drug effects (peak change from baseline) were observed on most pharmacodynamic study outcome variables. Mean subjective ratings of drug effect (F = 167.36, P < 0.001), unpleasant drug effect (F = 22.54, P < 0.001), good drug effect (F = 93.31, P < 0.001), sick (F = 10.03, P < 0.01), heart racing (F = 15.31, P < 0.001), anxious/nervous (F = 17.20, P < 0.001), relaxed (F = 10.08, P < 0.01), paranoid (F = 12.61, P < 0.01), sleepy/tired (F = 57.83, P < 0.001), irritable (F = 8.77, P < 0.01), restless (F = 11.41, P < 0.01), and hungry/have munchies (F = 43.45, P < 0.001) significantly increased from baseline after oral cannabis administration. Subjective ratings of alert (F = 27.80, P < 0.001) and vigorous (F = 12.17, P < 0.01) significantly decreased. Qualitatively, dose orderly differences were observed, with higher doses resulting in larger self-reported drug effects, however, the differences between doses were not statistically significant.
Subjective drug effects onset occurred 30–60 min after administration, had a sustained peak effect period from 90 to 180 min after administration, and gradually decreased in severity until the 8-h time point. Subjective ratings of “Drug Effect” by dose and time are illustrated in Figure 5.
Figure 5.
Subjective rated “Drug Effect” by dose.
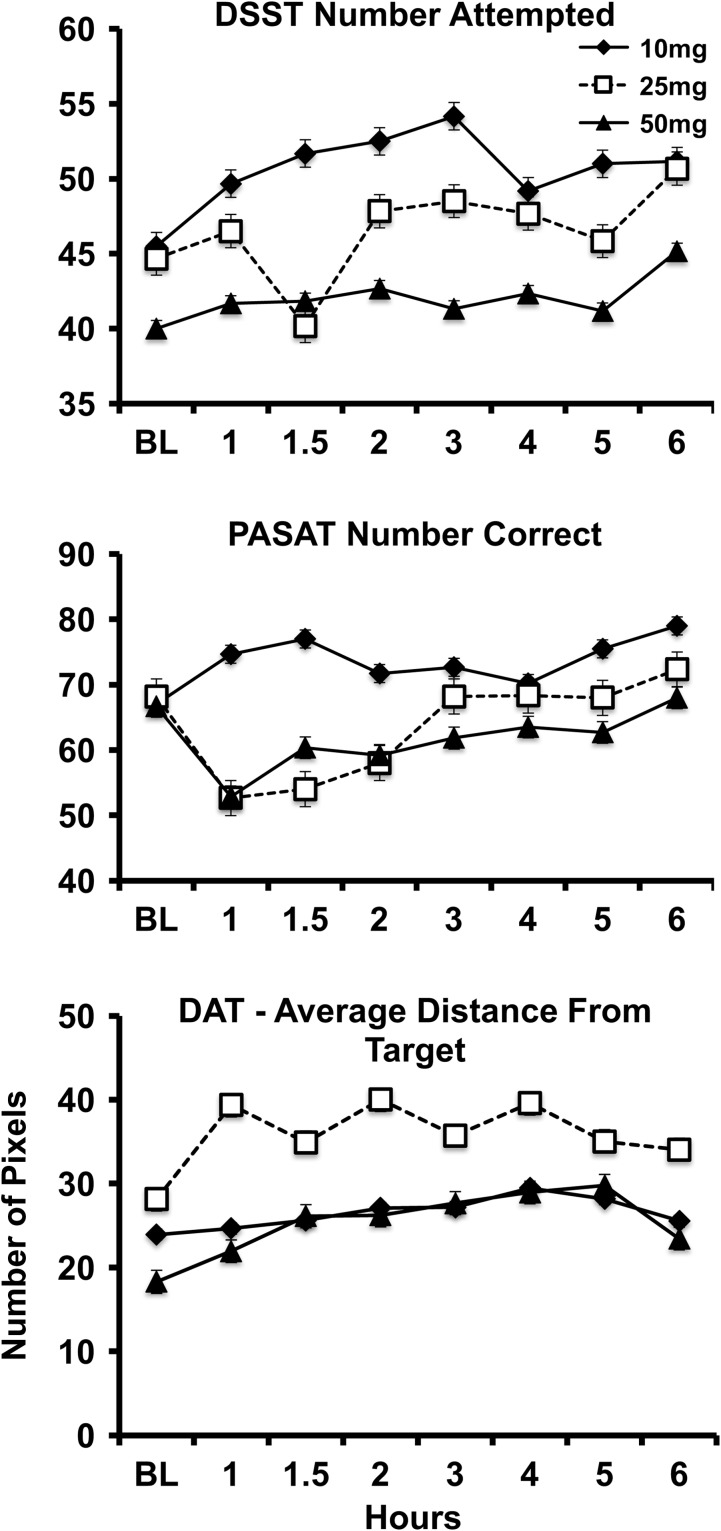
Significant drug effects were observed for several performance outcomes including: DSST % Correct (F = 19.64, P < 0.001), DAT mean distance from central tracking stimulus (F = 25.25, P < 0.001), DAT peripheral targets correct (F = 11.83, P < 0.01), DAT mean reaction time to peripheral targets (F = 26.18, P < 0.001), and total correct on the PASAT (F = 7.04, P < 0.05). For each of these variables, impairment relative to baseline was observed at the 25 and 50 mg doses, but not the 10 mg dose (Figure 6), and the time course of these performance effects was similar to the subjective drug effects. Comparisons of performance outcomes by dose were not statistically significant.
Figure 6.
Effects of oral cannabis on DSST (number attempted) and PASAT (total correct) performance outcomes.
Correlations between pharmacokinetic and pharmacodynamic outcomes
Despite low levels of blood cannabinoids observed in the study, self-reported subjective ratings of “Drug Effect” were positively correlated with whole blood THC, 11-OH-THC and THCCOOH levels (summary of correlations provided in Table VI). On the other hand, Drug Effect ratings were not significantly correlated with oral fluid THC and THCCOOH. Significant negative correlations were observed between DSST percent correct and whole blood THC levels after 10 mg and 25 mg THC doses, whole blood 11-OH-THC at the 10 mg THC dose, and oral fluid THCCOOH at the 25 mg dose. PASAT number correct had a significant negative correlation with whole blood THCCOOH at the 10 mg dose. There were no significant correlations between divided attention mean distance from target and whole blood or oral fluid biomarkers.
Table VI.
Correlations (Pearson's r) between blood and oral fluid cannabinoids and pharmacodynamic outcomes
| Whole blood THC | Whole blood 11-OH-THC | Whole blood THCCOOH | Oral fluid THC | Oral fluid THCCOOH | |
|---|---|---|---|---|---|
| 10 mg | |||||
| VAS: drug effect | 0.57* | 0.48* | 0.50* | −0.15 | 0.22 |
| DSST: % correct | −0.46* | −0.31* | −0.17 | 0.19 | −0.08 |
| PASAT: # correct | 0.18 | 0.11 | −0.47* | −0.27 | −0.23 |
| Divided attention:distance from target | 0.04 | 0.01 | −0.12 | −0.23 | −0.21 |
| 25 mg | |||||
| VAS: drug effect | 0.54* | 0.66* | 0.65* | −0.25 | 0.19 |
| DSST: % correct | −0.36* | −0.19 | −0.40 | 0.10 | −0.39* |
| PASAT: # correct | −0.17 | −0.19 | −0.18 | −0.01 | −0.22 |
| Divided attention:distance from target | 0.04 | 0.05 | 0.00 | −0.22 | −0.24 |
| 50 mg | |||||
| VAS: drug effect | 0.41* | 0.72* | 0.62* | −0.24 | 0.06 |
| DSST: % correct | 0.26 | −0.33 | −0.06 | 0.16 | 0.17 |
| PASAT: # correct | −0.11 | −0.32 | 0.03 | 0.08 | −0.02 |
| Divided attention: distance from target | −0.06 | −0.02 | 0.29 | −0.33 | −0.04 |
*Correlation is significant at the 0.01 level (two-tailed).
Description of adverse events
Two study participants vomited ~3 h after administration of the 50 mg dose. Emesis was transient and both participants reported feeling relief afterwards. One participant experienced a significant period of anxiety ~2 h after administration of the 25 mg dose. The participant was shaky, could not complete study assessments, and was uncomfortable communicating with study staff. The participant lay down alone in a dark room for ~40 min and assessments for one time point were missed. The anxiety gradually dissipated over the remainder of the session.
Discussion
Understanding the pharmacokinetics of oral cannabis administration and comparative pharmacodynamic effects is important for a number of reasons. First, the pharmacokinetic profile of cannabis is essential to understanding and interpreting toxicology testing for detection of recent use. Such testing is important for workplace and roadside drug testing programs as well as evaluation of outcomes in clinical trials of treatments for Cannabis Use Disorder (CUD). In particular, there is interest in determining whether there are concentrations of THC or its metabolites in various biological matrices that are predictive of intoxication/impairment, or that can distinguish between acute/recent cannabis use versus residual cannabinoids that remain long into abstinence.
This study suggests there are significant differences in blood and oral fluid cannabinoid pharmacokinetics following oral administration compared with inhalation. Given the increasing popularity of oral cannabis products, these data must be considered with respect to regulations related to workplace and roadside testing for cannabis use and/or intoxication, interpretation of forensic data in cases where cannabis use is considered important, and approaches for biochemical detection of cannabis abstinence for patients in treatment for CUD.
In the present study, we administered three doses of THC (10, 25 and 50 mg) to healthy adults who were not current cannabis users (last use was >3 months prior to study participation), allowing us to evaluate a single acute exposure of orally administered cannabis without interference from the residual effects of prior cannabis use. The low dose (10 mg THC) matches the current unit dose adopted by the State of Colorado for “edibles” that are sold for non-medical adult consumption. The maximum dose (50 mg THC) approximates the median dose (54 mg THC) found in 75 edible products recently obtained from medical cannabis dispensaries in California and Washington (7). Thus, the range of doses examined is relevant for individuals who may be exposed to cannabis for medical or non-medical use.
Following inhaled cannabis (smoked or vaporized), peak whole blood THC levels are dose dependent, with Cmax concentrations in controlled laboratory studies ranging from 15 to 192 ng/mL, and median Cmax ranges of 42–87 ng/mL across recent studies (20–22). In those studies of inhaled cannabis, blood THC levels peaked within 10 min of exposure and sharply decreased thereafter with a return to baseline within 3–6 h. In contrast, maximum blood levels of THC observed in the present study never exceeded 5 ng/mL (reached by 2 of 18 participants), a cutoff concentration that is commonly used to determine cannabis intoxication in roadside drug testing. Also, for two participants, no blood cannabinoids were detected after administration of the 10 mg THC dose. Oral dosing had a slower onset of peak effect (Tmax = 2–3 h) relative to reports of inhaled cannabis and return to baseline did not occur until 6–20 h post-exposure.
The low levels of blood cannabinoids observed in this study are consistent with prior reports of oral cannabis/THC administration (23, 24). Participants in the study by Ménétrey et al. (24) reported strong subjective drug effects and showed impairment on cognitive tasks, but the mean Cmax of blood THC was only 8.4 ng/mL. Favrat et al. (23) described two research participants who had adverse reactions to 20 mg oral dronabinol (synthetic THC). Both participants experienced periods of significant anxiety, performance impairment and psychotomimetic symptoms, and, at the time of peak effects for these individuals, whole blood THC levels were 1.8 and 6.2 ng/mL. Thus, the magnitude and time course of blood cannabinoid levels observed in studies of inhaled cannabis are substantially different compared with controlled studies of oral cannabis/THC. This is consistent with differences in plasma cannabinoid concentrations previously illustrated by Hollister and colleagues (25).
Though quantitative levels of THC and the psychoactive metabolite 11-OH-THC were very low, participants reported significant subjective drug effects at all three doses tested, and showed evidence of significant cognitive/psychomotor impairment after the 25 mg and 50 mg doses. Interestingly, blood levels of THC, 11-OH-THC and THCCOOH were moderate to highly correlated (Pearson's r = 0.41–0.72, P < 0.01) with subjective drug effect ratings at all three doses. The strength of correlation between THC and subjective drug effects decreased with increasing THC dose, but strength of correlations between 11-OH-THC increased with increasing THC dose administered suggesting that 11-OH-THC may be a more valid analyte for predicting stronger drug effects. Significant correlations were also observed between psychoactive blood cannabinoids and DSST percent correct, but these correlations were highest at the 10 mg THC dose when there was no impairment relative to baseline task performance. The use of blood cannabinoid levels as a predictive biomarker of intoxication or impairment following oral cannabis exposure is further complicated by the fact that the levels observed in this study are comparable to those likely to be measured in frequent cannabis users during periods of extended abstinence when no intoxication or impairment is evident (26, 27).
The subjective drug effect ratings and impairment observed on measures of psychomotor ability, working memory and divided attention (cognitive domains typically associated with acute cannabis intoxication) demonstrates the relevance of the doses administered in the present study. The time course of effects on these measures was also different than what is typically observed following inhaled cannabis. In the present study, the onset of drug effects typically occurred 30–60 min after ingestion, with a sustained peak effect occurring 90–180 min after administration and a gradual return to baseline 6–8 h post-exposure. In contrast, peak subjective drug effects and cognitive performance impairment following inhaled cannabis typically peaks immediately after exposure and returns to baseline in 3–4 h (25, 28). Participants in the present study were not regular cannabis users, and reported strong drug effects following the 25 and 50 mg doses including increased ratings of adverse effects such as sick, paranoid, nervousness/anxiety, irritability and feeling sleepy/tired.
A common side effect of cannabis use is dry mouth. In this study, participants were not allowed to drink or eat anything for 10 min prior to oral fluid specimen collection to avoid sample contamination or dilution. In total, 15 of 576 (2.6%) planned oral fluid specimens were missed due to participant inability to expectorate saliva. Though production of oral fluid was slow and challenging in many cases, collection was feasible most of the time. Detectible levels of THC were present in oral fluid immediately in the first (10 min) or second collection (30 min). However, concentrations declined rapidly over the subsequent 2 h in most cases. This suggests that the THC was directly deposited in the oral cavity as a result of consuming the cannabis brownies, and suggests that THC would not likely be detectable if encapsulated oral cannabis products were consumed whole (e.g., cannabis extract capsules). This is consistent with a previous study that showed no increase in oral fluid THC following oral administration of encapsulated dronabinol (29). Tmax for THCCOOH in oral fluid occurred a period of 0–54 h after ingestion of brownies.
Additional concerns with oral fluid testing for oral cannabis exposure include that five participants had no detectable THCCOOH in oral fluid (four participants following the 10 mg THC dose and one participant following the 50 mg THC dose). In addition, at the higher doses there was often variability in detection for consecutive samples; and there was frequent disagreement between THC and THCCOOH test results and between ELISA and LC–MS-MS testing results. Indeed, within the first 8 h after exposure (time course for subjective drug effect ratings) 148 oral fluid samples were positive for either THC or THCCOOH. Of those samples, 23 samples were positive for both THC and THCCOOH, 75 samples were positive for THC, but not THCCOOH, and 18 samples were positive for THCCOOH, but not THC. After 8 h, there were only two samples that were positive for both THC and THCCOOH, 30 samples were positive for THCCOOH, but not THC, and no samples were positive for THC, but not THCCOOH. Thus, detection of THC in oral fluid appears to be a somewhat more reliable test for recent oral cannabis use than THCCOOH. However, for most participants, the time to last THC detection in oral fluid preceded the time to last reported subjective drug effect.
Study limitations
Several limitations of the present study are worth noting to help interpret the generality of the findings. First, this study was limited to a small number of healthy adults without recent use of cannabis. The pharmacokinetic and pharmacodynamic effects of oral cannabis administration may vary in other populations. Most notably, individuals who are frequent cannabis users would likely have residual baseline blood and oral fluid cannabinoid levels and exhibit tolerance to the pharmacodynamic effects of these doses. Absorption and metabolism of oral cannabis may vary based on recent food consumption, use of medications that affect THC metabolism, and in children or older adults. Also, the small sample size, between-subjects design and lack of a placebo dose condition limit interpretation of the pharmacodynamic data. Inclusion of a placebo drug condition in the present protocol was not fiscally feasible given the focus on pharmacokinetic outcomes and the cost of a 9-day study, 6 days of which were residential. Future studies evaluating the pharmacodynamic effects of oral cannabis using a placebo-controlled design are needed. Lastly, the cannabis used in the present study was high in THC content (~11%) and low in CBD (<1%). It is unknown whether variation in the chemical profile of cannabis (i.e., ratio of THC to minor cannabinoids or non-cannabinoid compounds in the plant) would impact the pharmacokinetic or pharmacodynamic outcomes measured in this study, which warrants additional research consideration.
Conclusion
Consumption of a single oral dose of cannabis baked in a brownie produced measurable THC and related metabolites in healthy adults at most dose levels tested. Oral cannabis administration increased subjective drug effect ratings at all three doses tested, but impairment on cognitive performance tasks was only observed at the higher doses (25 and 50 mg THC). Substantial variability was observed in the window of detection by target analyte and across biological matrices. The window of THC detection ranged from 0 to 22 h for whole blood (LOQ = 0.5 ng/mL), and 1.9–22 h for oral fluid (LOQ = 1.0 ng/mL). Subjective drug and cognitive performance effects were generally dose dependent, peaked 1.5–3 h post-exposure, and lasted 6–8 h. Compared with inhalation, quantitative levels of cannabinoids in whole blood and oral fluid following oral cannabis are low, variable across participants, and generally did not parallel periods of self-reported intoxication or performance impairment. Future studies are needed to examine whether other biomarkers not tested in this study could be identified to better predict impairment and to evaluate the pharmacodynamic effects of oral cannabis using a placebo-controlled study design.
Acknowledgments
Dr Cone is a consultant to the Division of Workplace Programs, Substance Abuse and Mental Health Services Administration (SAMHSA) and has an Adjunct Professor appointment with Johns Hopkins University School of Medicine, Baltimore, MD. Drs Vandrey and Bigelow are faculty at Johns Hopkins School of Medicine, Baltimore, MD. Dr Herrmann is faculty at Columbia University and New York State Psychiatric Institute. Dr Mitchell is an employee of RTI International and Mr LoDico and Mr Flegel are employees of SAMHSA. We thank the support staff of the Johns Hopkins University Behavioral Pharmacology Research Unit and Clinical Research Unit for outstanding contributions to the implementation of this study. We also thank Dr. Christine Moore and Ms. Cynthia Coulter at Immunalysis Inc.
Funding
This study was funded by the Substance Abuse and Mental Health Services Administration (SAMHSA). The National Institute on Drug Abuse (NIDA) provided material support for the study (cannabis) and financial support for the participation of Dr Herrmann (T32-DA07209). We also obtained clinical services support from Grant UL1-RR025005 from the National Center for Research Resources (NCRR), a component of the National Institutes of Health (NIH), and NIH Roadmap for Medical Research.
Conflicts of Interest
Dr Vandrey has served as a consultant or received honoraria from Zynerba Pharmaceuticals, Insys Therapeutics, and CW Botanicals. There are no other conflicts of interest to declare.
References
- 1. WHO, W. H. O World Drug Report. World Health Organization: Vienna, Austria, 2015. [Google Scholar]
- 2. Grella C. E., Rodriguez L., Kim T. (2014) Patterns of medical marijuana use among individuals sampled from medical marijuana dispensaries in Los Angeles. Journal of Psychoactive Drugs, 46, 263–272. [DOI] [PubMed] [Google Scholar]
- 3. Walsh Z., Callaway R., Belle-Isle L., Capler R., Kay R., Lucas P. et al. (2013) Cannabis for therapeutic purposes: patient characteristics, access, and reasons for use. International Journal of Drug Policy, 24, 511–6. [DOI] [PubMed] [Google Scholar]
- 4. Weiss S. (2015) Edibles: for experts only. State Legislatures Magazine. [PubMed]
- 5. Hudak M., Severn D., Nordstrom K. (2015) Edible cannabis–induced psychosis: intoxication and beyond. American Journal of Psychiatry, 172, 911–912. [DOI] [PubMed] [Google Scholar]
- 6. Favrat B., Menetrey A., Augsburger M., Rothuizen L. E., Appenzeller M., Buclin T. et al. (2005) Two cases of “cannabis acute psychosis” following the administration of oral cannabis. BMC Psychiatry, 5, 17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Vandrey R., Raber J. C., Raber M. E., Douglass B., Miller C., Bonn-Miller M. O. (2015) Cannabinoid dose and label accuracy in edible medical cannabis products. Journal of the American Medical Association, 313, 2491–2493. [DOI] [PubMed] [Google Scholar]
- 8. Cone E. J., Johnson R. E., Paul B. D., Mell L. D., Mitchell J. (1988) Marijuana-laced brownies: behavioral effects, physiological effects, and urinalysis in humans following ingestion. Journal of Analytical Toxicology, 12, 169–175. [DOI] [PubMed] [Google Scholar]
- 9. Niedbala R. S., Kardos K. W., Fritch D. F., Kardos S., Fries T., Waga J. (2001) Detection of marijuana use by oral fluid and urine analysis following single-dose administration of smoked and oral marijuana. Journal of Analytical Toxicology, 25, 289–303. [DOI] [PubMed] [Google Scholar]
- 10. Swortwood M. J., Newmeyer M. N., Andersson M., Abulseoud O. A., Scheidweiler K. B., Huestis M. A. (2016). Cannabinoid disposition in oral fluid after controlled smoked, vaporized, and oral cannabis administration Drug testing and analysis. doi:10.1002/dta.2092 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Sobell L. C., Sobell M. B.. Timeline follow-back: a technique for assessing self-reported alcohol consumption In: Allen J. P., Litten R. Z. (eds).. Measuring Alcohol Consumption: Psychosocial and Biochemical Methods. Human Press: Totowa, NJ, 1992; pp. 41–72. [Google Scholar]
- 12. Coulter C., Miller E., Crompton K., Moore C. (2008) Tetrahydrocannabinol and two of its metabolites in whole blood using liquid chromatography-tandem mass spectrometry. Journal of Analytical Toxicology, 32, 653–658. [DOI] [PubMed] [Google Scholar]
- 13. Hooker W. D., Jones R. T. (1987) Increased susceptibility to memory intrusions and the Stroop interference effect during acute marijuana intoxication. Psychophamacologia, 91, 20–24. [DOI] [PubMed] [Google Scholar]
- 14. Wilson W. H., Ellinwood E. H., Mathew R. J., Johnson K. (1994) Effects of marijuana on performance of a computerized cognitive-neuromotor test battery. Psychiatry Research, 51, 115–125. [DOI] [PubMed] [Google Scholar]
- 15. Vandrey R. G., Mintzer M. Z.. Performance and cognitive alterations In: Cohen L., Collins F. L., Young A. M., McChargue D. E., Leffingwell T. R. (eds).. Pharmacology and Treatment of Substance Abuse Using Evidence and Outcomes Based Perspectives. Lawrence Erlbaum Associates, Inc.: Mahwah, 2009. [Google Scholar]
- 16. McLeod D. R., Griffiths R. R., Bigelow G. E., Yingling J. (1982) An automated version of the digit symbol substitution test (DSST). Behavior Research Methods and Instrumentation, 14, 463–466. [Google Scholar]
- 17. Kleykamp B. A., Griffiths R. R., Mintzer M. Z. (2010) Dose effects of triazolam and alcohol on cognitive performance in healthy volunteers. Experimental and Clinical Psychopharmacology, 18, 1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Herrmann E. S., Cone E. J., Mitchell J. M., Bigelow G. E., LoDico C., Flegel R. et al. (2015) Non-smoker exposure to secondhand cannabis smoke II: effect of room ventilation on the physiological, subjective, and behavioral/cognitive effects. Drug and Alcohol Dependence, 151, 194–202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Bland J. M., Altman D. G. (1995) Calculating correlation coefficients with repeated observations: Part 1—correlation within subjects. British Medical Journal, 310, 446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Hartman R. L., Huestis M. A. (2013) Cannabis effects on driving skills. Clinical Chemistry, 59, 478–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Fabritius M., Chtioui H., Battistella G., Annoni J. M., Dao K., Favrat B. et al. (2013) Comparison of cannabinoid concentrations in oral fluid and whole blood between occasional and regular cannabis smokers prior to and after smoking a cannabis joint. Analytical and Bioanalytical Chemistry, 405, 9791–803. [DOI] [PubMed] [Google Scholar]
- 22. Schwope D. M., Bosker W. M., Ramaekers J. G., Gorelick D. A., Huestis M. A. (2012) Psychomotor performance, subjective and physiological effects and whole blood Delta(9)-tetrahydrocannabinol concentrations in heavy, chronic cannabis smokers following acute smoked cannabis. Journal of Analytical Toxicology, 36, 405–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Favrat B., Menetrey A., Augsburger M., Rothuizen L., Appenzeller M., Buclin T. et al. (2005) Two cases of “cannabis acute psychosis” following the administration of oral cannabis. BMC Psychiatry, 5, 17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Menetrey A., Augsburger M., Favrat B., Pin M. A., Rothuizen L. E., Appenzeller M. et al. (2005) Assessment of driving capability through the use of clinical and psychomotor tests in relation to blood cannabinoids levels following oral administration of 20 mg dronabinol or of a cannabis decoction made with 20 or 60 mg Delta9-THC. Journal of Analytical Toxicology, 29, 327–338. [DOI] [PubMed] [Google Scholar]
- 25. Hollister L. E., Gillespie H. K., Ohlsson A., Lindgren J. E., Wahlen A., Agurell S. (1981) Do plasma concentrations of delta 9-tetrahydrocannabinol reflect the degree of intoxication. Journal of Clinical Pharmacology, 21, 171S–177S. [DOI] [PubMed] [Google Scholar]
- 26. Milman G., Bergamaschi M. M., Lee D., Mendu D. R., Barnes A. J., Vandrey R. et al. (2014) Plasma cannabinoid concentrations during dronabinol pharmacotherapy for cannabis dependence. Therapeutic Drug Monitoring, 36, 218–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Fabritius M., Favrat B., Chtioui H., Battistella G., Annoni J. M., Appenzeller M. et al. (2014) THCCOOH concentrations in whole blood: are they useful in discriminating occasional from heavy smokers. Drug Testing and Analysis, 6, 155–163. [DOI] [PubMed] [Google Scholar]
- 28. Hunault C. C., Mensinga T. T., de Vries I., Kelholt-Dijkman H. H., Hoek J., Kruidenier M. et al. (2008) Delta-9-tetrahydrocannabinol (THC) serum concentrations and pharmacological effects in males after smoking a combination of tobacco and cannabis containing up to 69 mg THC. Psychopharmacology, 201, 171–181. [DOI] [PubMed] [Google Scholar]
- 29. Lee D., Karschner E. L., Milman G., Barnes A. J., Goodwin R. S., Huestis M. A. (2013) Can oral fluid cannabinoid testing monitor medication compliance and/or cannabis smoking during oral THC and oromucosal Sativex administration. Drug and Alcohol Dependence, 130, 68–76. [DOI] [PMC free article] [PubMed] [Google Scholar]