Abstract
A study of the bat diversity was conducted in Hulu Terengganu dipterocarp forest and Setiu Wetland Beach Ridges Interspersed with Swales (BRIS) forest in Terengganu, to study the species diversity, composition and stratification of fruit bats from the understorey to the forest canopy. Mist nets were set up at the understorey, sub-canopy and canopy layer while harp traps were set up at the understorey layer. We recorded 170 individuals from six families’ compromised 21 species from Hulu Terengganu dipterocarp forests and four species from Setiu Wetland BRIS forests throughout the sampling period. Megaerops ecaudatus and Cynopterus brachyotis were the most dominant species in Hulu Terengganu dipterocarp forest and Setiu Wetland BRIS forests. Our study also recorded two species with new distributional records for the east coast of Peninsular Malaysia, namely, Rhinolophus chiewkweeae and Chaerephon johorensis in Hulu Terengganu dipterocarp forests. Potential factors that might influence the results were in terms of the canopy covers, the structural complexity of canopy, food availability and spatial characteristics. This study was able to increase the knowledge on the species diversity and composition of bats in Hulu Terengganu dipterocarp forest and Setiu Wetland BRIS forest, thus, further aid in the effort of bat conservation in both areas.
Keywords: Bat, Diversity, Stratification, Dipterocarp Forest, BRIS Forest
Abstract
Satu kajian kepelbagaian kelawar telah dijalankan di hutan dipterokarp Hulu Terengganu dan hutan Beach Ridges Interspersed with Swales (BRIS) tanah lembap Setiu di Terengganu untuk mengkaji kepelbagaian spesies, komposisi dan stratifikasi kelawar dari bawah hingga ke kanopi hutan. Jaring kelawar telah di pasang di bawah kanopi, sub-kanopi dan kanopi, manakala perangkap harp telah diletak pada lapisan bawah kanopi. Kami merekodkan 170 individu daripada enam keluarga dan 21 spesies dari hutan dipterokarp Hulu Terengganu dan lima spesies dari hutan BRIS tanah lembap Setiu sepanjang tempoh pensampelan. Megaerops ecaudatus dan Cynopterus brachyotis adalah spesies yang paling dominan dalam hutan dipterocarp Hulu Terengganu dan hutan BRIS tanah lembap Setiu. Kajian kami juga telah mencatatkan dua spesies yang mempunyai rekod penyebaran baru di pantai timur Malaysia, iaitu Rhinolophus chiewkweeae dan Chaerephon johorensis daripada hutan dipterokarp Hulu Terengganu. Faktor-faktor yang berpotensi mempengaruhi keputusan adalah dari segi litupan kanopi, kepelbagaian struktur kanopi, ketersediaan makanan dan ciri-ciri ruang. Kajian ini dapat meningkatkan pengetahuan mengenai kepelbagaian spesies dan komposisi kelawar di hutan dipterokarp Hulu Terengganu dan hutan BRIS tanah lembap Setiu. Dengan itu, kajian lanjut diperlukan dalam usaha pemuliharaan kelawar di kedua-dua kawasan tersebut.
INTRODUCTION
Bats performed many significant ecological services and often regarded as a keystone species. In agricultural and natural ecosystems, they deliver some ecological regulating services such as pest suppression, seed dispersal and pollination that benefits human economically (Kunz et al. 2011). Globally, bats are the second most diverse mammals on earth after with 1,116 species currently, have been described (Kunz & Parsons 2009). Currently, in Malaysia, there are 133 species of bats in Malaysia (Kingston et al. 2012). In Peninsular Malaysia, the study on bats has been studied very well in Krau Wildlife Reserve, Peninsular Malaysian east-coast state of Pahang and also in Peninsular Malaysian west-coast states. However, the study of bats in Peninsular Malaysian east-coast despite being studied very well in Pahang, in other states namely Kelantan and Terengganu is still understudied. We also studied the vertical stratification of Pteropodid bats to increase the bat’s species checklist that forages in a higher level. The vertical stratification study of bats in Malaysia has been conducted by Francis (1994), Zubaid (1994), Abdullah and Hall (1997), and Hodgkison et al. (2004). The study on bats ecology in Malaysia has been concentrated at ground level due to easy deployments of mist nets and harp traps. The study on vertical stratification on bats is still understudied in Malaysia with only five known published information.
This study has been conducted in Hulu Terengganu district which is part of Central Forest Spine (CFS) Project and Setiu Wetland in Setiu district. The habitat and forest types in these two study sites are different and contrasting where Hulu Terengganu is a dipterocarp forest and Setiu Wetland consisting of various forest types. Currently, there are some established forest reserves (FR) in Terengganu, namely, Lata Belatan FR, Lata Tembakah FR, Lata Paying FR, Bukit Kesing FR, Sekayu FR, Chemerong FR, Jambu Bongkok FR, Rimba Bandar Bukit Bauk FR, Air Menderu FR, Rasau Kertih FR, and Besul FR. Other than that, dipterocarp forests in Hulu Terengganu also serve as a part of the CFS, which is the seventh, out of 17 linkages in Peninsular Malaysia. The CFS is one of the government efforts to create a broad adjoining forest corridor through the combination of interconnecting forests in 17 habitat linkages (Hedges et al. 2013). Due to the discovery of unique floral and faunal composition in Setiu Wetland (Tamblyn et al. 2006; Pesiu et al. 2015), this area is proposed to be the first state park of Terengganu along with the motion of establishing Tasik Kenyir in Hulu Terengganu and its surrounding forests as a Geopark (Che Aziz et al. 2015). Perhaps the information from this study will assist the government and non-governmental agencies in conservation planning and decision making.
MATERIALS AND METHODS
Description of the Study Area and Sampling Site
The sampling period was done from January to March 2015. The sampling sites and sampling duration within those study areas are as shown in Figure 1 and described in Table 1.
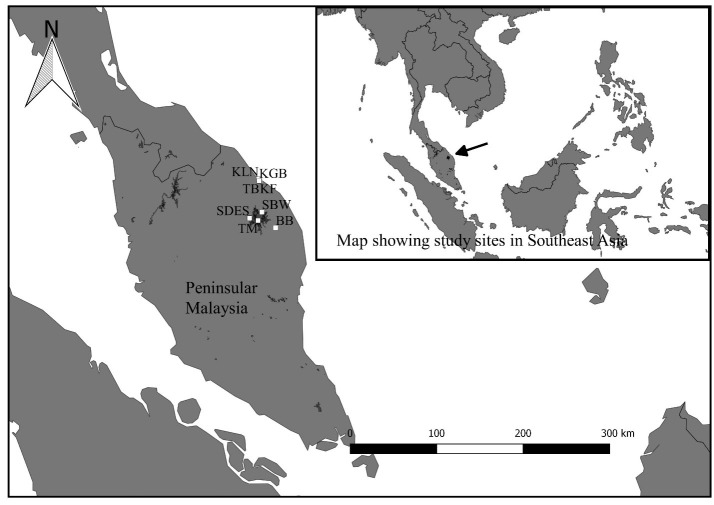
Figure 1.
Study sites of bats in Terengganu. Hulu Terengganu dipterocarp forest: SBW-Sungai Buweh Waterfall, SDES- Sungai Deka Elephant Sanctuary, TM- Tanjung Mentong and BB- Belukar Bukit. Setiu Wetland BRIS forest: KLN- Kampung Limau Nipis, KF-Kampung Fikri, KGB- Kampung Gong Batu and TB- Tasik Berombak.
Table 1.
List of study areas, forest types, date of studies and locations.
| Study Areas | Forest Types | Date | Locations |
|---|---|---|---|
| Hulu Terengganu dipterocarp forest | |||
| Sungai Buweh Waterfall | Lowland riparian forest | 19 to 23 January 2015 | N 05° 08′52″ E 102° 46′00″ |
| Sungai Deka Elephant Sanctuary | Lowland and old secondary forest | 9 to 12 February 2015 | N 05° 02′58″ E 102° 33′57″ |
| Tanjung Mentong | Hill dipterocarp forest | 7 to 13 March 2015 | N 05° 00′39″ E 102° 42′13″ |
| Belukar Bukit | Old secondary forest | 15 to 21 May 2015 | N 04° 53′25″ E 102° 59′ 33″ |
| Setiu Wetland BRIS forest | |||
| Kampung Limau Nipis | BRIS forest | 21 to 27 February 2015 | N 05° 40′58″ E 102° 42′58″ |
| Kampung Fikri | BRIS forest | 23 to 29 March 2015 | N 05° 39′19″ E 102° 44′08″ |
| Kampung Gong Batu | BRIS forest | 30 March 5 April 2015 | N 05° 39′23″ E 102° 43′18″ |
| Tasik Berombak | BRIS forest | 7 to 13 March 2015 | N 05° 39′19″ E 102° 43′37″ |
BRIS = Beach Ridges Interspersed with Swales
A vegetation survey was done following Soepadmo (1971) on the plant diversity in the study areas. The vegetation surveys were done in Hulu Terengganu dipterocarp forests (Appendix A) and Setiu Wetland BRIS forests (Figure 1; Appendix B). The most of the common tree species observed were recorded. Descriptions of the habitat types and their coordinates are listed as in Table 1.
Field Methods
Due to the limited resources and difficulty of canopy access and deployment, only eight mist nets and three four-banks harp traps were deployed in the understorey level whereby two to five mist nets were set up at the canopy layer for six consecutive nights. Each mist nets and harp traps below the canopy layer were set up at various locations, such as, across trails and water streams which were believed to be the flyways for bats (Kunz & Kurta 1988; Khan et al. 2007). The understorey mist nets were relocated after two days to cover more area and increase the chances of catching more individuals and species (Mohd-Hanif et al. 2015) and to prevent the “net shyness” (Kunz & Kurta 1988). The canopy mist nets were deployed by using two methods; sets of scaffolding towers with an approximate of 12 m height from the ground floor level and another using rope and pulley systems. All of the mist nets and harp traps were checked regularly with an interval of 15–30 minutes after dusk until 9.00 pm and were continued in the following mornings from 5.00 am to 6.30 am.
The stratification of Hulu Terengganu dipterocarp forest followed the method by Abdullah and Hall (1997) as; forest floor (S1: <7 m), understorey level (S2: 7 to 15 m), canopy level (S3: 15 to 20 m) and emergent (S4: >20 m; Figure 2). Meanwhile, the stratification of Setiu Wetland BRIS forest can be generalised into ground level (T1: <5 m) and canopy level (T2: 5 to 10 m; Figure 2) due to the relatively lower height as compared to Hulu Terengganu dipterocarp forest.
Figure 2.
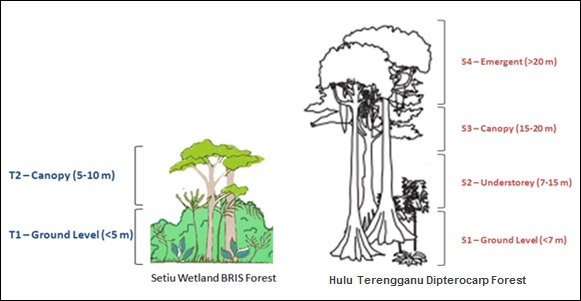
The canopy stratification level in Hulu Terengganu dipterocarp forest and Setiu Wetland BRIS forest.
The species identification followed the keys by Francis (2008) and Kingston et al. (2009). The morphological measurements were taken using digital vernier calliper. The weight for each individual was weighted using a spring balance and selected species were photographed for future references. Representative species (maximum of five individuals for each species) were preserved in 70% ethanol solution and deposited in the Museum of Zoology Kenyir (MZK), Universiti Malaysia Terengganu (UMT) for future references.
Paleontological Statistic (PAST) v2.17 software (Hammer et al. 2012) was used to calculate Shannon’s diversity index. To perform diversity comparison with unequal sampling effort among sampling sites, we used rarefaction curve constructed by using EcoSim v1.2d software (Entsminger 2014) and Microsoft Excel. We also constructed species accumulation curve using EcoSim software. In addition, a taxonomic list of bats was compared with previous studies done within Hulu Terengganu dipterocarp forest and Setiu Wetland BRIS forest.
RESULTS
Vegetation Survey
Following the classification by Saw (2010), several characteristics of the forest formations and vegetation were gathered in order to describe the forests of Hulu Terengganu. Generally, Hulu Terengganu is occupied by lowland evergreen rain forest with per-humid (a seasonal) climate having elevations ranging from sea level to 1,200 m above sea level. The lowland evergreen rainforest has three tree layer characteristics, mainly upper layer with emergent trees as tall as or greater than 70 m, the main stratum at about 24 m until 36 m and a lower layer with smaller, shade-tolerant trees and immature trees. Meanwhile, the forest floor or ground vegetation often sparsely occupied by small trees, shrubs, herbs and understorey palms (Saw 2010).
In Peninsular Malaysia, the lowland evergreen rainforests can be described further into three sub-forest types based on the changes in species composition that occur with altitude (Symington 1943; Wyatt-Smith 1963; Saw 2010). The three sub-forest types, namely lowland dipterocarp forest (<300 m), hill dipterocarp forests (300–750 m) and upper hill dipterocarp forests (750–1200 m) and these can be found in Hulu Terengganu. However, in this research, more evidence was found on the lowland dipterocarp forest giving an indication that the study site is located within the extensive stands of lowland dipterocarp forests. Moving into the species compositions, the common tree species in lowland dipterocarp forests of Hulu Terengganu include Hopea dyeri, Shorea leprosula, and Dipterocarpus grandiflorus from the major timber family, Dipterocarpaceae. The other tree species include Baccaurea parviflora and Croton laevifolius (Euphorbiaceae); Pometia pinnata (Sapindaceae), Monocarpia marginalis (Annonaceae), and Saraca thaipingensis (Caesalpiniaceae) (Pesiu et al. 2015). This description is also consistent to the distributions of the tree species by Turner (1995).
Setiu Wetland is well-known for its unique soil formation called Beach Ridges Intersped with Swales (BRIS). Recent studies by Jamilah et al. (2013) on the natural vegetation of BRIS soil ecosystem on the coastal dune of Terengganu has described three distinct natural vegetation formations on the BRIS soil ecosystem as lowland mixed dipterocarp forest, melaleuca swamp and heath vegetation. Our extensive study has been done in some parts of Setiu Wetland revealing similar vegetation types that made up the land. Small patches of peat swamp forest, mangrove forests, and beach vegetation occupied the land of Setiu. The mangrove forests and beach vegetation generally made up the coastal framework of Setiu Wetland with small patches of beach vegetation interval with the mangrove forests decorating the continuous coastal region along Setiu Wetland. The mangrove forest generally dominated by species from family Rhizophoraceae. Common species that can be found in mangrove forest are Rhizophora apiculata (Rhizophoraceae), Heritiera littoralis (Malvaceae), Pouteria obovata (Sapotaceae), Excoecaria agallocha (Euphorbiaceae), and Hibiscus tiliaceus (Malvaceae) (Pesiu et al. 2015).
Meanwhile, predominant species found with beach vegetation was Casuarina equisetifolia (Casuarinaceae). Other tree species, including Terminalia catappa (Combretaceae), Pandanus odoratissimus (Pandanaceae) and Cycas litoralis were commonly found. Other vegetation types that can be found in Setiu is the peat swamp forest. This type of forest is not too notable due to its adaptations on permanently waterlogged site; often hidden by the large scale of heath vegetation and Melaleuca swamp forests. However, the tree species within this forests are distinctive and easily identified. Some of the frequently encountered species were Alstonia spatula (Apocyaceae), Campnosperma sp. (Anacardiaceae), Macaranga hypoleuca (Euphorbiaceae), Cratoxylum arborescens (Hypericaceae) and Ilex cymosa (Aquifoliaceae) found to be scattered throughout the forests (Pesiu et al. 2015).
Chiropteran Survey
A total of 170 individuals of chiropterans representing 21 species from six families were recorded in Hulu Terengganu dipterocarp forests and Setiu Wetland BRIS forests (Appendix C). From the total of species captured, eight species were frugivorous and the rest were insectivorous. We identified Sungai Buweh Waterfall as sampling site with the highest recorded species (14 species), whereas Kampung Limau Nipis site with the least recorded species (one species). The highest number of individuals recorded was at Sungai Buweh Waterfall (45 individuals) and the least, was at Kampung Fikri (three individuals). Cynopterus brachyotis was encountered in all study sites and were the most abundant in Setiu Wetland BRIS forest. Moreover, Megaerops ecaudatus was the dominant species in Hulu Terengganu dipterocarp forest. Our study also highlighted the two new distributional record species in Sungai Buweh Waterfall and Belukar Bukit, which were Rhinolophus chiewkweeae (Morni et al. 2016) and Chaerephon johorensis (Roslan et al. 2016).
The taxonomic diversity of chiropterans based on stratification level shows that pteropodid bats utilised each forest stratum in Hulu Terengganu dipterocarp forests and Setiu Wetland BRIS forests. In Hulu Terengganu dipterocarp forest, we netted five species in the canopy layer of Belukar Bukit forest, namely, Penthetor lucasi (20.34 m height) M. ecaudatus, C. johorensis and Megaderma spasma (20.65 m); and Balionycteris maculata (22.65 m). Only C. brachyotis was netted at canopy level (T2) in Setiu Wetland forests at 12 m from the ground floor in Tasik Berombak. The rest of the species dominantly occurred on a much lower elevation in both study areas.
Figure 3 shows unique and shared species between two contrasting ecosystems; Hulu Terengganu dipterocarp forest and Setiu Wetland forest. The insectivorous bats were captured in ground level except for C. johorensis. Within the Family Rhinolophidae, R. affinis, R. chiewkweeae, and R. luctus are confined to the dipterocarp forest while R. lepidus is found in both ecosystems. Meanwhile, C. brachyotis was observed to be present in all sampling sites of two contrasting ecosystems in Terengganu.
Figure 3.
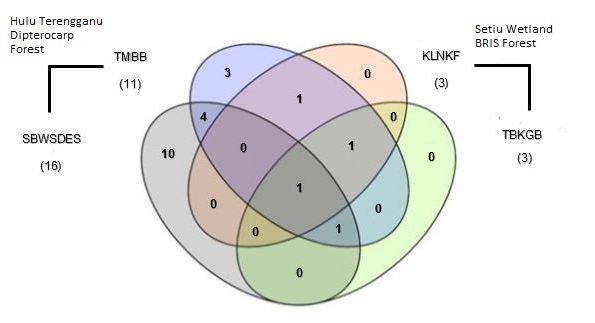
Venn diagram showing numbers of unique and shared species between Hulu Terengganu dipterocarp forest and Setiu Wetland BRIS forest. Sampling sites were pooled to represent each ecosystem of dipterocarp and BRIS forest. The number of species is shown in each of the subsets and the total number of species for each sampling sites are given in parentheses. SBWSDES is combination of Sungai Buweh Waterfall and Sungai Deka Elephant Sanctuary; TMBB- combination of Tanjong Mentong and Belukar Bukit; KLNKF- combination of Kampung Limau Nipis and Kampung Fikri; TBKGB- combination of Kampung Gong Batu and Tasik Berombak.
Figure 4 shows unique and shared species between four forest strata in both Hulu Terengganu dipterocarp forest and Setiu Wetland BRIS forest. The understorey layer recorded the highest number of species followed by the canopy layer and sub-canopy layer. Species that are shared between the understorey and canopy layer showed that those species, namely, B. maculata, C. brachyotis, P. lucasi and M. spasma can be found in both lower and upper elevations. Meanwhile, C. brachyotis was found in the understorey, sub-canopy and canopy layer.
Figure 4.
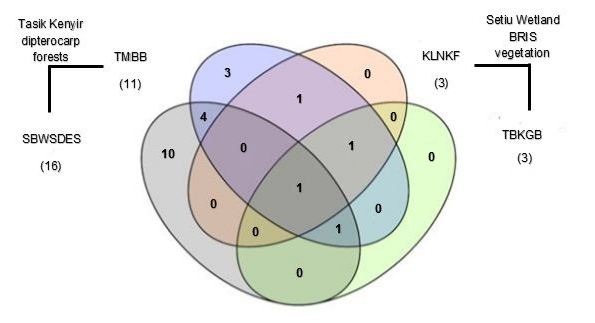
Venn diagram showing numbers of unique and shared species between forest strata; understorey, sub-canopy, canopy and emergent layer in Hulu Terengganu dipterocarp forest and Setiu Wetland BRIS forest. The number of species is shown in each of the subsets and the total number of species for each sampling sites are given in parentheses.
Sungai Buweh Waterfall has the highest diversity compared to the other sites with the highest number of Shannon index is 2.123 (Table 2). Meanwhile, Kampung Limau Nipis has the lowest diversity (Shannon index = 0.000). Overall, Hulu Terengganu dipterocarp forest hold the highest diversity of bats (Shannon index = 2.619) compared to Setiu Wetland BRIS forest (Shannon index = 0.314; Table 3). The individual rarefaction curve of bats in Hulu Terengganu dipterocarp forest and Setiu Wetland BRIS forests is shown in Figure 5. The 95% confidence intervals showed that the species richness between the two areas is significantly different.
Table 2.
Diversity and species richness of chiropteran in each study sites.
| Station | Dipterocarp Forest | Setiu Wetland BRIS Forest | ||||||
|---|---|---|---|---|---|---|---|---|
|
| ||||||||
| SBW | SDES | TM | BB | KLN | KF | KGB | TB | |
| n | 45 | 21 | 5 | 39 | 40 | 3 | 9 | 9 |
| Shannon Index | 2.123 | 0.887 | 0.500 | 1.817 | 0.000 | 0.637 | 0.349 | 0.562 |
| Species Richness | 3.415 | 0.985 | 0.621 | 2.730 | 0.000 | 0.910 | 0.455 | 0.481 |
SBW: Sungai Bewah Waterfall; SDES: Sungai Deka Elephant Sanctuary; TM: Tanjung Mentong; BB: Belukar Bukit; KLN: Kampung Limau Nipis; KF: Kampung Fikri; KGB: Kampung Gong Batu; TB: Tasik Berombak
Table 3.
Diversity and species richness of bats in Hulu Terengganu dipterocarp forest and Setiu Wetland BRIS forest.
| Station | Hulu Terengganu dipterocarp forest | Setiu Wetland BRIS forest |
|---|---|---|
| n | 110 | 60 |
| Shannon Index | 2.619 | 0.314 |
n = number of individuals
Figure 5.
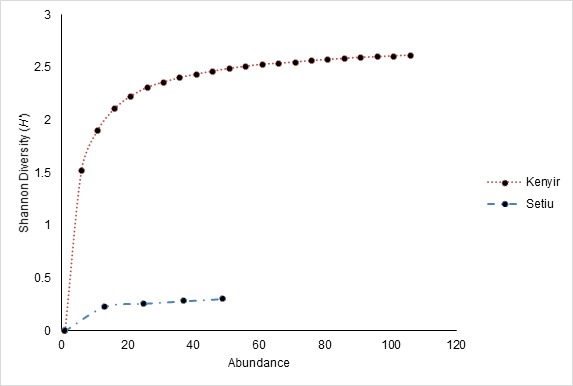
The individual rarefaction curve of bats in Hulu Terengganu dipterocarp forest and Setiu Wetland BRIS forest. The highest diversity of both sites after rarefaction curve analysis 2.46 in Hulu Terengganu dipterocarp forest.
DISCUSSION
From the result, the bat diversity of Hulu Terengganu dipterocarp forest is higher than Setiu Wetland BRIS forest. There were many factors that contributed to the higher number of bats in Hulu Terengganu dipterocarp forest. Dipterocarp forest holds up more niches for higher numbers of unique species compositions (Hill & Hill 2001; Bruhl et al. 2003; Okuda et al. 2003; Podong & Poolsiri 2013) due to a denser forest area with higher structural diversity, and higher food availability (Molles 2005). Meanwhile, Setiu Wetland BRIS forest recorded the lower diversity of bats due to the forest clearance by anthropogenic activities such as human settlements, agriculture, aquaculture, and land conversion into palm oil plantations that caused forest fragmentation (Tamblyn et al. 2006; Mohd Azmi 2014; Suratman et al. 2014). Fragmentation followed by deforestation had caused serious impact towards the loss of bats habitats in Southeast Asia (Lane et al. 2006; Sodhi & Brook 2006). Fukuda et al. (2009) reported that chiropterans were one of the most susceptible taxa affected by anthropogenic activities. However, the high abundance of C. brachyotis in Setiu wetland forest shows that this species is able to thrive in most areas including those in the disturbed vicinity. This species is a strong flyer and is cosmopolitan species (Abdullah, 2003). This species has cryptic forms, the small-sized and the large-sized where the small-sized C. brachyotis inhabit the forested area whereas the large-sized C. brachyotis preferring the open areas (Abdullah et al. 2010; Abdullah, 2003).
The results of this study were compared with previous studies conducted in various habitats in Hulu Terengganu dipterocarp forest and Setiu Wetland BRIS Forest and Jambu Bongkok FR BRIS Forest (Appendix D). The present study done in Hulu Terengganu recorded the higher number of bat species compared to Norfarhana et al. (2015);Shahrul Anuar & Mohd Hifni (2001), Nor Zalipah et al. (2010) and Nor Zalipah et al. (2015) due to the longer sampling periods, covering more areas, and the usage of canopy mist nets. The effective usage of canopy mist nets resulting more capture on species that commonly forage in a higher level such as R. amplexicaudatus, E. spelaea, and M. ecaudatus. Francis (1994) noted that the capture rates for the bats above the ground level are more than 100 times. This study also recorded the new distributional records namely R. chiewkweeae and C. johorensis. The R. chiewkweeae were captured in Sungai Buweh Waterfall forest marked the seventh records in Malaysia (Morni et al. 2016) after Lubuk Sembilang, Langkawi Island, and Ulu Muda FR in Kedah, Asahan FR in Melaka, Gunung Ledang and Labis FR in Johor, and Wang Kelian State Park in Perlis (Yoshiyuki & Lim 2005; Jayaraj et al. 2011). Meanwhile, C. johorensis was the first record in Terengganu in Belukar Bukit forest and forth record in Malaysia (Roslan et al. 2016) after Hutan Lipur Air Terjun Batu Hampar, Kedah (Jayaraj et al. 2013) and Krau Wildlife Reserve, Pahang, Malaysia (Francis 2008).
Meanwhile, for the vertical stratification of fruit bats, the abundance of fruit bats in sub canopy level and canopy level in Hulu Terengganu dipterocarp forest is comparable with the study done by Francis (1994); Zubaid (1994); Abdullah & Hall (1997) and Hodgkison et al. (2004). High distribution of species from family Pteropodidae at the canopy layer shows that higher food source is available and the tropical rain forests’ canopy layers provide more fruits to animals such as bats (Harrison 1962; Francis 1994; Abdullah & Hall 1997; Mohd-Azlan et al. 2000). Although we used mist nets to capture fruit bats, we also managed to capture insectivorous bats in canopy level namely C. johorensis. This species is known to forage at the canopy level (Francis 2008). In Setiu Wetland BRIS forest, there was only C. brachyotis captured at the height of more than 10 m, the other species were captured at the height range of less than 10 m. This is because the forest in Setiu Wetland BRIS forest has been logged therefore many species that were generally foraging in canopy such as E. spelaea can be found at ground level.
CONCLUSION
The result of the bat diversity in contrasting ecosystems shows that the species diversity of bats in Terengganu is still understudied. There are still many areas need to be explored to obtain comprehensive bat data. More species recorded in Hulu Terengganu dipterocarp forest were due to the higher structural complexity of the vegetation that fulfills the needs of the bat’s niche compared to the less complex composition of Setiu Wetland BRIS forest. Particular species that were found and dominated certain forest strata implies that the species adaptations complemented the structure and features of the certain strata they inhabit. For improvement in doing further research, sampling period and the number of nets deployed particularly at canopy levels should be increased to get more recorded species and number of bats. Lunar phase needs to be avoided during the sampling period because the nocturnal animals are passive during the full moon phase. The bat detector and camera traps can be deployed at canopy level to detect more bat species.
Figure 6.
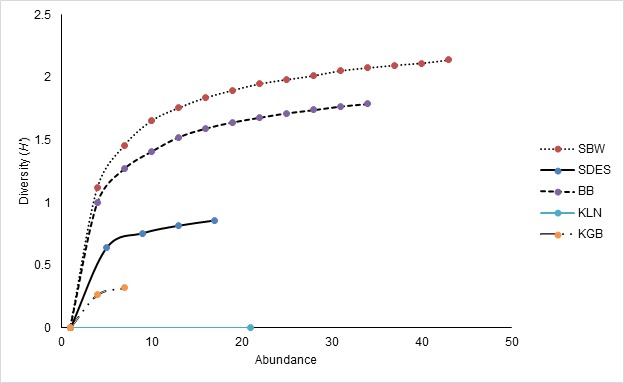
The individual rarefaction curve of each study sites at Hulu Terengganu dipterocarp forest and Setiu Wetland BRIS forests. Study sites at and Tanjung Mentong (Hulu Terengganu dipterocarp forest), and Kampung Fikri and Tasik Berombak (Setiu Wetland BRIS forest) are excluded due to the low number of species captured. The highest species diversity was observed in Sungai Buweh Waterfall. SBW- Sungai Buweh Waterfall, SDES- Sungai Deka Elephant Sanctuary, BB- Belukar Bukit, KLN- Kampung Limau Nipis, KGB- Kampung Gong Batu.
Figure 7.
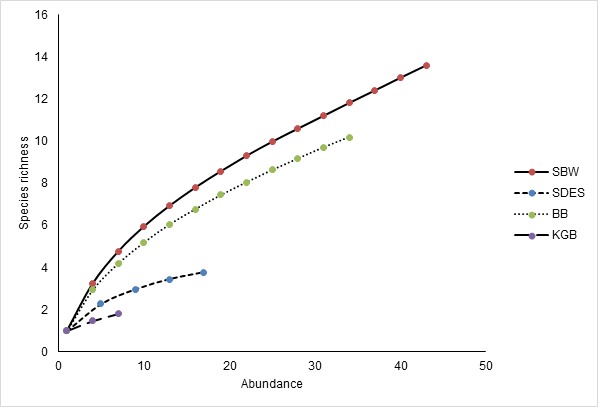
Species accumulation curve showing the species increasing trend. Study sites at Tanjung Mentong (Hulu Terengganu dipterocarp forest) and, Kampung Limau Nipis, Kampung Fikri and Tasik Berombak (Setiu Wetland BRIS forest) are excluded due to the low number of species captured. SBW- Sungai Buweh Waterfall, SDES- Sungai Deka Elephant Sanctuary, BB- Belukar Bukit and KGB- Kampung Gong Batu.
ACKNOWLEDGEMENTS
We would like to express our appreciation to the former Director of Kenyir Research Institute (now Institute of Tropical Biodiversity and Sustainable Development; ITBSuD), Universiti Malaysia Terengganu (UMT), Professor Emeritus Dr Faizah Shahrom for her permission to conduct the research. In addition, we would like to thank the Department of Wildlife and National Parks (DWNP) for their logistical supports during the research. We also like to thank staffs and post-graduate students of ITBSuD, and Mr Tarmizi Abdullah for their help throughout the field works. Our study was partially funded by Geran Galakan Penyelidikan (GGP/UMT/68007/2014/127) and Niche Research Grant Scheme (NRGS/2014/53131) awarded to M. T. Abdullah and colleagues.
APPENDICES
Appendix A
Photographs of the habitats in Tasik Kenyir. (a) Sungai Buweh Waterfall, (b) Belukar Bukit, (c) Sungai Deka Elephant Sanctuary, (d) Tanjung Mentong.
Appendix B
Photographs of the habitats in Setiu Wetland. (e) Kampung Fikri, (f) Kampung Gong Batu, (g) Tasik Berombak, (h) Kampung Limau Nipis.
Appendix C
Taxonomic compositions, stratification and relative abundance in parentheses of bats in Hulu Terengganu dipterocarp forest and Setiu Wetland BRIS forest in this study.
| Family | Hulu Terengganu dipterocarp forests | Setiu Wetland (BRIS Forest) | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
|||||||||||||||||
| Species | SBW | SDES | TM | BB | KLN | KF | KGB | TB | |||||||||
| n (RA) | SC | n (RA) | SC | n (RA) | sc | n (RA) | SC | n (RA) | SC | n (RA) | SC | n (RA) | SC | n (RA) | SC | ||
| Pteropodidae | |||||||||||||||||
| 1 | Balionycteris maculata | 0 | - | 0 | - | 0 | - | 4 (0.024) | S1 | 0 | - | 0 | - | 0 | - | 2 (0.012) | T1 |
| 2 | Cynopterus brachyotis | 1 (0.006) | S1 | 3 (0.018) | S1.S2 | 1 (0.006) | S2 | 7(0.041) | S1.S3 | 40 (0.235) | T1.T2 | 2(0.012) | T1 | 8 (0.047) | T1.T2 | 6 (0.035) | T1.T2 |
| 3 | Cynopterus horsfieldii | 0 | - | 0 | - | 0 | - | 1 (0.006) | S1 | 0 | - | 0 | - | 0 | - | 0 | - |
| 4 | Eonycteris spelaea | 0 | - | 0 | - | 0 | - | 1 (0.006) | S1 | 0 | - | 1 (0.006) | T1 | 0 | - | 0 | - |
| 5 | Macroglossus minimus | 1 (0.006) | S1 | 0 | - | 0 | - | 1 (0.006) | S1 | 0 | - | 0 | - | 0 | - | 0 | - |
| 6 | Macroglossus sobrinus | 0 | - | 0 | - | 0 | - | 0 | S1 | 0 | - | 0 | - | 0 | - | 0 | - |
| 7 | Megaerops ecaudatus | 1 (0.006) | S3 | 0 | - | 0 | - | 2 (0.012) | S1 | 0 | - | 0 | - | 0 | - | 0 | - |
| 8 | Penthetor lucasi | 1 (0.006) | S1 | 0 | - | 0 | - | 17 (0.100) | S3 | 0 | - | 0 | - | 0 | - | 0 | - |
| Megadermatidae | S3 | ||||||||||||||||
| 9 | Megaderma spasma | 0 | - | 1 (0.006) | S1 | 4(0.024) | S1 | 1 (0.006) | S1 | 0 | - | 0 | - | 0 | - | 0 | - |
| Rhinolophidae | |||||||||||||||||
| 10 | Rhinolophus affinis | 4 (0.024) | S1 | 0 | - | 0 | - | 3 (0.018) | S1 | 0 | - | 0 | - | 0 | - | 0 | - |
| 11 | Rhinolophus chiewkweeae | 4 (0.024) | S1 | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - |
| 12 | Rhinolophus lepidus | 1 (0.006) | S1 | 0 | - | 0 | - | 1 (0.006) | S1 | 0 | - | 0 | - | 1 (0.006) | T1 | 0 | - |
| 13 | Rhinolophus luctus | 1 (0.006) | S1 | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - |
| Hipposideridae | |||||||||||||||||
| 14 | Hipposideros bicolor | 1 (0.006) | S1 | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - |
| 15 | Hipposideros cervinus | 10 (0.059) | S1 | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - |
| 16 | Hipposideros diadema | 1 (0.006) | S1 | 2 (0.012) | S1 | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - |
| 17 | Hipposideros dyacorum | 6 (0.035) | S1 | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - |
| 18 | Hipposideros larvatus | 12 (0.071) | S1 | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - | 0 | |
| Vespertillionidae | |||||||||||||||||
| 19 | Kerivoula pellucida | 1 (0.006) | S1 | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - |
| 20 | Myotis ridleyi | 0 | - | 15 (0.088) | S1 | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - | 0 | - |
| Molossidae | |||||||||||||||||
| 21 | Chaerephon johorensis | 0 | 0 | - | 0 | - | 1 (0.006) | S3 | 0 | - | 0 | - | 0 | - | 0 | - | |
|
| |||||||||||||||||
| Number of individuals | 45 | 21 | 5 | 39 | 40 | 3 | 9 | 8 | |||||||||
| Number of species | 14 | 4 | 2 | 11 | 1 | 2 | 2 | 3 | |||||||||
| Number of families | 4 | 4 | 2 | 4 | 1 | 1 | 2 | 2 | |||||||||
| Sampling effort | 52 | 32 | 40 | 78 | 48 | 40 | 40 | 40 | |||||||||
| Capture rate | 0.865 | 0.656 | 0.125 | 0.5 | 0.833 | 0.075 | 0.225 | 0.2 | |||||||||
| Diversity index | 2.147 | 0.887 | 0.673 | 1.817 | 0 | 0.637 | 0.349 | 0.562 | |||||||||
n = number; RA = relative abundance; SBW = Sungai Buweh Waterfall, SDES = Sungai Deka Elephant Sanctuary, TM = Tanjong Mentong, BB = Belukar Bukit, KLN = Kampung Limau Nipis, KF = Kampung Fikri, KGB = Kampung Gong Batu,TB = Tasik Berombak; SC = Strata Class; S1: <7 m, S2: 7–15 m, S3:15–30 m; S4: >30 m (Abdullah and Hall, 1997); T1: <10 m and T2:10–16 m
Appendix D
Comparison of taxonomic list of species recorded in this study with previous studies in Hulu Terengganu dipterocarp forest and Setiu Wetland BRIS forest.
| No. | Family | Hulu Terengganu | Setiu Wetland | Jambu Bongkok FR | IUCN 2013 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|||||||||
| Dipterocarp forest | BRIS forest | |||||||||
|
|
|
|||||||||
| Species | This study | Norfarhana et al. (2015) | Shahrul Anuar & Mohd Hifi (2001) | Jayaraj et al. (2015) | This study | Tamblyn et al. (2006) | Nor-Zalipah et al (2015) | Nor-Zalipah et al. (2010) | ||
| Pteropodidae | ||||||||||
| 1 | Rousettus amplexicaudatus | − | + | − | − | − | − | + | − | LC |
| 2 | Cynopterus brachyotis | + | + | + | + | + | + | + | − | LC |
| 3 | Cynopterus horsfieldii | + | − | + | − | − | − | + | − | LC |
| 4 | Penthetor lucasi | + | + | + | − | − | + | − | − | LC |
| 5 | Megaerops ecaudatus | + | + | − | − | − | − | − | − | LC |
| 6 | Dyacopterus spadiceus | − | − | − | − | − | + | − | − | NT |
| 7 | Chironax melanocephalus | − | − | − | − | − | + | − | − | LC |
| 8 | Balionycteris maculata | + | + | + | − | + | + | − | − | LC |
| 9 | Eonycteris spelaea | + | + | + | − | + | + | + | − | LC |
| 10 | Maoroglossus minimus | + | + | + | − | − | − | − | − | LC |
| 11 | Macroglossus sobrinus | + | − | − | + | − | − | LC | ||
| Nycteridae | − | |||||||||
| 12 | Nycteris tragata | − | + | |||||||
| Megadermatidae | − | |||||||||
| 13 | Megaderma lyra | − | + | − | − | − | − | − | − | LC |
| 14 | Megaderma spasma | + | + | − | − | − | + | − | − | LC |
| Rhinolophidae | − | |||||||||
| 15 | Rhinolophus trifoliatus | − | − | − | − | − | − | − | + | LC |
| 16 | Rhinolophus sedulus | − | + | − | − | − | − | − | − | NT |
| 17 | Rhinolophus luctus | + | − | − | − | − | − | − | − | LC |
| 18 | Rhinolophus marshalli | − | − | − | − | − | + | − | − | LC |
| 19 | Rhinolophus acuminatus | − | − | − | − | − | − | − | − | LC |
| 20 | Rhinolophus lepidus | + | − | − | − | + | − | − | + | LC |
| 21 | Rhinolophus affinis | + | − | − | + | − | − | − | − | LC |
| 22 | Rhinolophus robinsoni | − | − | − | + | − | − | − | − | NT |
| 23 | Rhinolophus stheno | − | − | − | + | − | − | − | − | LC |
| 24 | Rhinolophus creaghii | − | − | − | − | − | + | − | − | LC |
| 25 | Rhinolophus chiewkweeae | + | − | − | − | − | − | − | − | NE |
| Hipposideridae | ||||||||||
| 26 | Hipposideros bicolor | + | − | − | + | − | − | − | − | LC |
| 27 | Hipposideros armiger | − | − | − | − | − | − | − | − | LC |
| 28 | Hipposideros cineraceus | − | + | − | + | − | − | − | − | LC |
| 29 | Hipposideros dyacorum | + | − | + | − | − | − | − | LC | |
| 30 | Hipposideros cervinus | + | − | − | + | − | + | − | − | LC |
| 31 | Hipposideros galeritus | − | + | LC | ||||||
| 32 | Hipposideros larvatus | + | + | − | + | − | − | − | − | LC |
| 33 | Hipposideros diadema | + | + | − | + | − | − | − | − | LC |
| 34 | Hipposideros armiger | − | + | − | − | − | − | LC | ||
| Vespertilionidae | ||||||||||
| 35 | Myotis muricola | − | − | − | − | − | − | − | + | LC |
| 36 | Myotis ridleyi | + | + | − | − | − | − | − | − | NT |
| 37 | Myotis horsfieldii | − | + | − | + | − | − | − | − | LC |
| 38 | Myotis siligorensis | − | + | − | − | − | − | − | − | LC |
| 39 | Scotophilus kuhlii | − | − | − | − | − | + | − | + | LC |
| 40 | Tylonycteris pachypus | − | + | − | − | − | − | − | − | LC |
| 41 | Munna aenea | − | − | − | − | − | − | − | + | VU |
| 42 | Kerivoula minuta | − | + | NT | ||||||
| 43 | Kerivoula intermedia | − | − | − | − | − | − | − | + | NT |
| 44 | Kerivoula pellucida | + | − | − | − | − | − | − | − | LC |
| 45 | Miniopterus magnater | − | − | − | − | − | + | − | − | LC |
| 46 | Miniopterus medius | − | − | − | − | − | − | − | + | LC |
| Mollosidae | − | |||||||||
| 47 | Chaerephon johorensis | + | − | − | − | − | − | − | − | VU |
|
| ||||||||||
| Number of families | 6 | 5 | 1 | 5 | 2 | 6 | 1 | 2 | ||
| Number of species** | 21 | 17 | 6 | 15 | 4 | 14 | 4 | 7 | ||
| Number of individuals** | 110 | 56 | n.a. | 30 | 60 | 43 | 221 | 27 | ||
| Sampling effort/nights | 312 | n.a. | n.a. | n.a. | 312 | n.a. | n.a. | 33 | ||
| Capture rate | 0.35 | n.a. | n.a. | n.a. | 0.19 | n.a. | n.a. | 0.82 | ||
The Unidentified species were excuded from the taxonomic list; IUCN = International Union for Conservation of Nature; NE = Not Evaluated, LC = Least Concern, VU = Valnerable, NT = Near Thrated; n.a = data not available
REFERENCES
- Abdullah MT, Hall LS. Abundance and distribution of fruit bats and other mammals in the tropical forest canopy in Borneo. Sarawak Museum Journal. 1997;72:63–74. [Google Scholar]
- Abdullah MT. PhD Thesis. The University of Queensland; St Lucia: 2003. Biogeography and variation of Cynopterus brachyotis in Southeast Asia. [Google Scholar]
- Abdullah MT, Wong SF, Ketol B. Catalogue of mammals of UNIMAS zoological museum. Kota Samarahan: Universiti Malaysia Sarawak; 2010. [Google Scholar]
- Bruhl CA, Eltz T, Linsemair KE. Size does matter-effects of tropical rainforest fragmentation on the leaf litter ant community in Sabah, Malaysia. Biodiversity and Conservation. 2003;12:1371–1389. https://doi.org/10.1023/A:1023621609102. [Google Scholar]
- Che Aziz A, Hamlee I, Razidi SAK, Norzuhairil Z. Geopark Tasik Kenyir: Keistimewaan dan cadangan membangunkan geopark kebangsaan. Paper presented at National Geoscience Conference 2015; 31 July–1 August 2015; Kota Bharu, Kelantan. 2015. [Google Scholar]
- Entsminger GL. EcoSim professional: Null modeling software for ecologist. Version 1. Montrose: Acquired Intelligenc Kesey-Bear & Pinyon Publishing; 2014. [Google Scholar]
- Francis CM. Vertical stratification of fruit bats (Pteropodidae) in lowland dipterocarp rainforest in Malaysia. Journal of Tropical Ecology. 1994;10:523–530. https://doi.org/10.1017/S0266467400008191. [Google Scholar]
- Fukuda D, Taisen O, Momose K, Sakai S. Bat diversity in the vegetation mosaic around a lowland dipterocarp forest of Borneo. Raffles Bulletin of Zoology. 2009;57:213–221. [Google Scholar]
- Hammer Ø, Harper DAT, Ryan PD. PAST: Paleontological statistics software package for education and data analysis. Palaeontologia Electronica. 2012;4(1):9. [Google Scholar]
- Harrison JL. The distribution of feeding habits among animals in a tropical rain forest. Journal Animal Ecology. 1962;31:53–63. https://doi.org/10.2307/2332. [Google Scholar]
- Hedges L, Clements GR, Aziz SA, Yap W, Laurance S, Goosem M, Laurance WF. Small carnivore records from a threatened habitat linkage in Terengganu, Peninsular Malaysia. Small Carnivore Conservation. 2013;49:9–14. [Google Scholar]
- Hill JL, Hill RA. Why are tropical rain forests so species rich? Classifying, reviewing and evaluating theories. Progress in Physical Geography. 2001;25(3):326–354. https://doi.org/10.1177/030913330102500302. [Google Scholar]
- Hodgkison R, Balding ST, Zubaid A, Kunz TH. Habitat structure, wing morphology, and the vertical stratification of Malaysian fruit bats (Megachiroptera: Pteropodidae) Journal of Tropical Ecology. 2004;20:667–663. https://doi.org/10.1017/S0266467404001737. [Google Scholar]
- Jamilah MS, Umairah H, Nur-Hanani AJ, Risnayati L, Razali MS. Natural vegetation of BRIS soil ecosystem on coastal dune of Terengganu. Paper presented at the Seminar Kebangsaan Pemuliharaan Hutan Pesisir Pantai Negara; Universiti Malaysia Terengganu, Malaysia. 2013. [Google Scholar]
- Jayaraj VK, Ketol B, Marni W, Sait I, Mohamad-Jalani M, Khan FAA, Fong PH, Hall LS, Abdullah MT. Comparative distribution and diversity of bats from selected localities in Sarawak. Borneo Journal of Resource Science and Technology. 2011;1:1–13. [Google Scholar]
- Jayaraj VK, Azhar MI, Daud SHM. A new record of Chaerephon johorensis at Mount Jerai, Kedah, Malaysia. Malayan Nature Journal. 2013;65(1):233–235. [Google Scholar]
- Khan FAK, Sazali SN, Zaini MK, Ketol B, Ryan JR, Julaihi AM, Hall LS, Abdullah MT. Bats of Bako National Park, Sarawak, Malaysian Borneo. Sarawak Museum Journal. 2007;84:267–300. [Google Scholar]
- Kingston T, Lim BL, Zubaid A. Bats of Krau Wildlife Reserve. Bangi, Selangor: Penerbit Universiti Kebangsaan Malaysia; 2009. [Google Scholar]
- Kingston T, Juliana S, Nurul-Ain E, Hashim R, Zubaid A. The Malaysian bat conservation research unit: From a national model to an international network. Malaysian Applied Biology. 2012;41:1–10. [Google Scholar]
- Kunz TH, Kurta A. Capture methods and holding devices. In: Kunz TH, editor. Ecological and behavioral methods for the study of bats. Washington: Smithsonian Institution Press; 1988. [Google Scholar]
- Kunz TH, Parsons S. Ecological and behavioral methods for the study of bats. 2nd ed. Baltimore, Maryland: Johns Hopkins University Press; 2009. [Google Scholar]
- Kunz TH, Torrez EBD, Bauer D, Lobova T, Fleming TH. Ecosystem services provided by bats. Annals of the New York Academy of Sciences. 2011;1223(2011):1–38. doi: 10.1111/j.1749-6632.2011.06004.x. https://doi.org/10.1111/j.1749-6632.2011.06004.x. [DOI] [PubMed] [Google Scholar]
- Lane DJW, Kingston T, Lee BP. Dramatic decline in bat species richness in Singapore, with implications for Southeast Asia. Biological Conservation. 2006;131:584–593. https://doi.org/10.1016/j.biocon.2006.03.005. [Google Scholar]
- Mohd-Azlan J, Sharma RSK, Zakaria M. Species diversity and abundance of understorey bats at Air Hitam Forest Reserve, Selangor, Malaysia. Malayan Nature Journal. 2000;54(1):69–75. [Google Scholar]
- Mohd Azmi MI. Valuing the potential economic value of mangrove resources in Setiu Wetlands, Terengganu, Malaysia: A preliminary findings. International Journal of Education and Research. 2014;12(4):487–504. [Google Scholar]
- Mohd-Hanif RMD, Nur-Aida MT, Zahirunisa AR, Mohd-Ridwan AR, Abdullah MT. Contribution of regenerated forest in conservation of bats in Peninsular Malaysia. Journal of Tropical Forest Science. 2015;27(4):506–516. [Google Scholar]
- Molles MC. Ecology: Concepts and applications. 3rd edition. New York: McGraw-Hill Companies; 2005. [Google Scholar]
- Morni MA, Tahir NFDA, Rosli QS, Dee JW, Azhar I, Roslan A, Zahidin MA, Abdullah MT, Khan FAA. New record of Rhinolophus chiewkweeae (Chiroptera: Rhinolophidae) from the east coast of Peninsular Malaysia with new information on their echolocation calls, genetics and their taxonomy. Raffles Bulletin of Zoology. 2016;64:242–249. [Google Scholar]
- Norfarhana M, Chin-Fang T, Mohamad-Abid K, Madinah A, Abdullah MT. Preliminary survey of small mammals in Bukit Taat, Tasik Kenyir, Hulu Terengganu, Malaysia. Borneo Journal of Resource Science and Technology. 2015;5(2):79–83. [Google Scholar]
- NorZalipah M, Hussain Mohd-Lokman, Sulong I. Pollination importance of bats in mangrove communities of Setiu Wetlands, Terengganu. In: Faridah M, Jamilah MS, Jarina MS, Rohani S, editors. Setiu Wetlands: Species, Ecosystems and Livelihoods. Kuala Terengganu, Malaysia: Penerbit Universiti Malaysia Terengganu (UMT); 2015. pp. 73–83. [Google Scholar]
- NorZalipah M, Siti-Tafzil MSAK, Ho WC, Embong M, Mazrul AM. A brief survey of birds and bats at Jambu Bongkok Forest Reserve, Dungun, Terengganu. In: Jamilah MS, Faridah M, editors. A biological assessment of Jambu Bongkok Forest Reserve, Terengganu and nearby ecosystem. Kuala Terengganu, Malaysia: Universiti Malaysia Terengganu; 2010. pp. 30–35. [Google Scholar]
- Okuda T, Suzuki M, Adachi N, Quah ES, Hussein NA, Manokaran N. Effect of selective logging on canopy and stand structure and tree species composition in a lowland dipterocarp forest in Peninsular Malaysia. Forest Ecology and Management. 2003;175:297–320. https://doi.org/10.1016/S0378-1127(02)00137-8. [Google Scholar]
- Pesiu E, Abdullah MT, Jamilah MS, Clements GR. Comparative forest phenology in Kenyir Dipterocarp Species and Setiu Wetland, Terengganu. 2015. Unpublished information. [Google Scholar]
- Podong C, Poolsiri R. Forest structure and species diversity of secondary forest after cultivation in relation to various sources at lower northern Thailand. Proceedings of the International Academy of Ecology and Environmental Sciences. 2013;3(3):208–218. [Google Scholar]
- Rapala-Kozik M, Kowalska E, Ostrowska K. Modulation of thiamine metabolism in Zea mays seedlings under conditions of abiotic stress. Journal of Experimental Botany. 2008;59(15):4133–4143. doi: 10.1093/jxb/ern253. https://doi.org/10.1093/jxb/ern253. [DOI] [PubMed] [Google Scholar]
- Rapala-Kozik M, Olczak M, Ostrowska K, Starosta A, Kozik A. Molecular characterization of the thi3 gene involved in thiamine biosynthesis in Zea mays: cDNA sequence and enzymatic and structural properties of the recombinant bifunctional protein with 4-amino-5-hydroxymethyl-2-methylpyrimidine (phosphate) kinase and thiamine monophosphate synthase activities. Biochemistry Journal. 2007;408:149–159. doi: 10.1042/BJ20070677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roslan A, David G, Muhamad-Aidil Z, Fathihi-Hakimi R, Nur Izzah IA, Abdullah MT. A new distributional record of Chaerephon johorensis (Chiroptera: Mollosidae) at Belukar Bukit, Hulu Terengganu, Malaysia. Journal of Wildlife and Parks. 2016;31:65–70. [Google Scholar]
- Saw GL. Vegetation of Peninsular Malaysia. In: Kiew R, Chung RCK, Saw LG, Soepadmo E, editors. Flora of Peninsular Malaysia. Series II: Seed Plants. Kuala Lumpur, Malaysia: Malayan Forest Records, Forest Research Institute Malaysia; 2010. pp. 21–45. [Google Scholar]
- Shahrul Anuar MS, Mohd Hifni B. Conservation of mammal and bird fauna at Hulu Kenyir, Terengganu. Proceeding of the International Conference on Insitu and Ex-situ Biodiversity Conservation in the New Millenium; Sabah, Malaysia. 20–22 June 2000; 2001. pp. 279–292. [Google Scholar]
- Sodhi NS, Brook BW. Southeast Asian biodiversity in crisis. Cambridge: University of Cambridge Press; 2006. [Google Scholar]
- Soepadmo E. Plants and vegetation along the paths from Kuala Tahan to Gunong Tahan. Malayan Nature Journal. 1971;24:118–124. [Google Scholar]
- Suratman S, Hussein ANAR, Latif MT, Weston K. Reassesment of physicchemical water quality in Setiu Wetland, Malaysia. Sains Malaysiana. 2014;43(8):1127–1131. [Google Scholar]
- Suyanto A, Struebig MJ. Bats of the Sangkulirang limestone karst formations, East Kalimantan-a priority region for Bornean bat conservation. Acta Chiropterologica. 2007;9:67–95. https://doi.org/10.3161/1733-5329(2007)9[67:BOTSLK]2.0.CO;2. [Google Scholar]
- Symington CF. Foresters’ manual of dipterocarps. Kuala Lumpur: University of Malaya Press; 1943. (Malayan Forest Records. No. 16). [Google Scholar]
- Tamblyn A, Turner C, Raines P. Malaysia tropical forest conservation project: A collaborative project between the Department of Wildlife and National Parks, Malaysia (PERHILITAN) and Coral Cay Conservation. Jaquelin Fisher Associates Ltd; 2006. pp. 1–97. [Google Scholar]
- Turner IM. A catalogue of the vascular plants on Malaya. Garden’s Bulletin Singapore. 1995;47(1):1–757. [Google Scholar]
- Wyatt-Smith J. Malayan Forest Records No. 23. Vol. 1. Kuala Lumpur: Forest Research Institute; 1963. Manual of Malayan silviculture for inland forest. [Google Scholar]
- Yoshiyuki M, Lim BL. A new horseshoe bat, Rhinolophus chiewkweeae (Chiroptera, Rhinolophidae) from Malaysia. Bulletin of the National Science Museum Tokyo, Series A. 2005;31:29–36. [Google Scholar]
- Zubaid A. Vertical stratification pteropodid bats in a Malaysian lowland rainforest. Mammalia. 1994;58:309–311. [Google Scholar]