Summary
Background
Testicular adrenal rest tumours (TARTs) are benign testicular masses that are found in inadequately treated patients with congenital adrenal hyperplasia (CAH). Recognizing this association and identifying characteristic ultrasound features of TARTs is important so as to avoid misdiagnosing them as malignancies, which can lead to unnecessary interventions.
Case Report
We describe a case of a 9-year-old boy, with a diagnosis of CAH and precocious puberty, who was referred to our department for an ultrasound evaluation of the abdomen and scrotum. On ultrasound, there were well-defined, heterogeneous, predominantly hypoechoic, round-to-oval masses in both testes. Taking into account the presence of CAH and a typical sonographic appearance of bilateral testicular masses, a diagnosis of testicular adrenal rest tumour was made; biopsy was deferred and hormonal treatment was modified.
Conclusions
Prompt diagnosis of testicular adrenal rest tumours is essential, as it only indicates inadequate hormonal control. Moreover, it can prevent unnecessary biopsies and orchidectomies, and can maintain fertility. TARTs have a typical imaging appearance that every radiologist must be aware of.
Keywords: Adrenal Hyperplasia, Congenital; Testicular Neoplasms; Ultrasonography
Background
Congenital adrenal hyperplasia (CAH) is an autosomal recessive disorder which is characterized by a deficiency of enzymes involved in the synthesis of glucocorticoids. Glucocorticoid deficiency leads to an increase in ACTH secretion which often results in adrenal hyperplasia. It also leads to overstimulation of aberrant adrenal rests in the testes, which leads to formation of testicular adrenal rest tumours (TARTs). TARTs are benign masses that usually respond to medical therapy [1]. However, as most intratesticular masses are considered malignant, identifying characteristics features of TARTs on ultrasound (the most common modality used for assessment of testes) is of utmost importance. This can prevent unnecessary biopsies and radical orchidectomies. Ultrasound helps differentiate TARTs from malignant testicular tumours (like Leydig cell tumours), and is also helpful in monitoring regression of these tumours. Thus, every radiologist must be aware of the sonographic features of TART’s. Therefore, we decided to describe the ultrasound appearance of a testicular adrenal rest tumour in a patient with congenital adrenal hyperplasia.
Case Report
A 9-year-old boy, diagnosed with CAH at the age of 1 year following an adrenal crisis, was poorly controlled due to lack of compliance. He presented with precocious puberty, short stature, and aggressive behaviour. The cortisol level was low, and levels of 17 alpha-hydroxyprogesterone, plasma corticotrophin (ACTH), and testosterone were high. Scrotal ultrasound revealed well-defined, heterogeneous, predominantly hypoechoic, round-to-oval masses with posterior acoustic shadowing in both testes, in the medial aspects near the mediastinum testis. The right testicular mass measured 1.3×1.1 cm, and the left testicular mass measured 1.5×1.2cm (Figures 1, 2). On colour Doppler, the masses had minimal internal vascularity (Figure 3). The rest of the testes showed normal flow. The epididymis and cord structures were normal. There was no evidence of hydrocoele on both sides. Testicular tumour markers were negative. Abdominal ultrasound did not reveal any mass lesion in the suprarenal region, ruling out adrenal gland enlargement (Figure 4). In a patient with CAH and bilateral testicular masses that had a typical sonographic appearance, a diagnosis of testicular adrenal rest tumour was made, and biopsy was deferred. Hormonal treatment was intensified, and the testicular masses are followed up. The patient improved clinically, which was associated with a decrease in testosterone and 17 alpha-hydroxyprogesterone levels; however, the testicular masses did not significantly change in size on follow up ultrasound performed after 3 and 6 months. Currently, the patient is advised to undergo follow-up ultrasound examinations every 3 months.
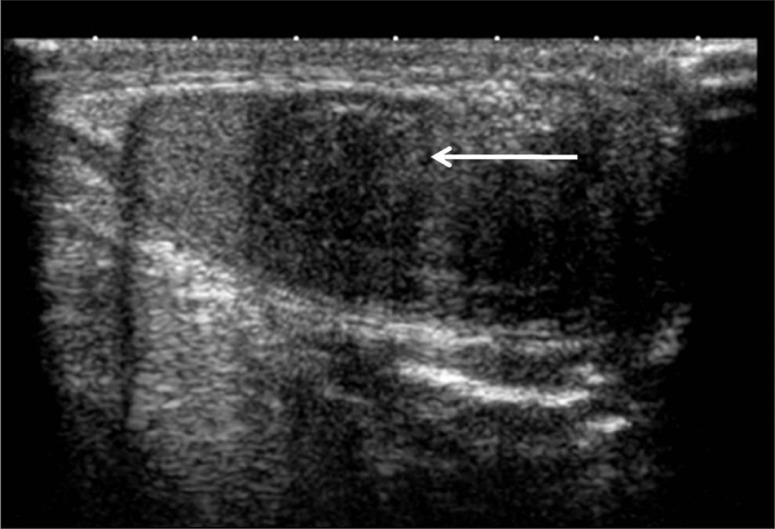
Figure 1.
There is a well-defined, heterogeneous, predominantly hypoechoic, round-to-oval lesion, with posterior acoustic shadowing in the medial aspect of the right testis, near the mediastinum testis, measuring approximately 1.3×1.1 cm.
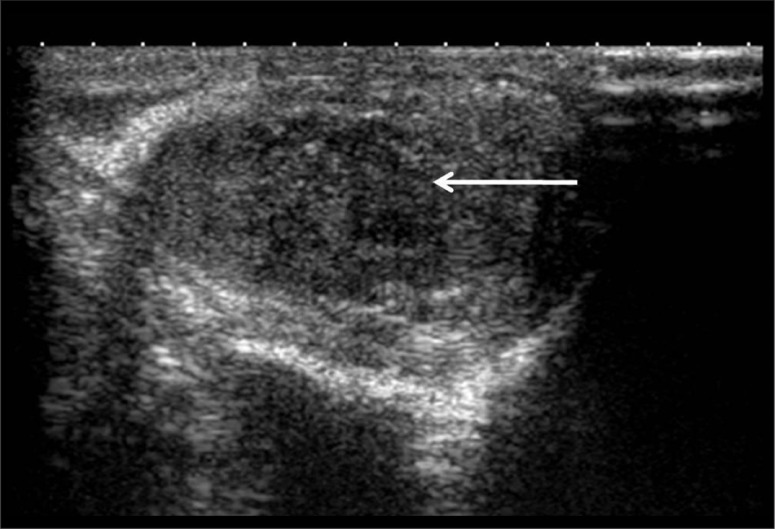
Figure 2.
There is a well-defined, heterogeneous, predominantly hypoechoic round-to-oval lesion, with posterior acoustic shadowing, in the medial aspect of the left testis, near the mediastinum testis, measuring approximately 1.5×1.2 cm.
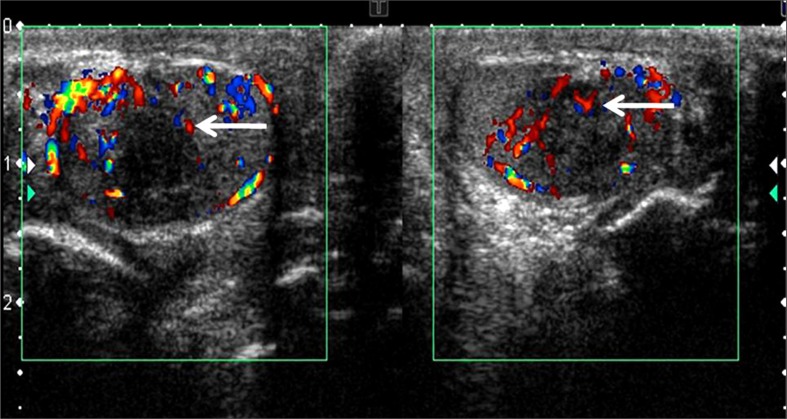
Figure 3.
The lesions in both testes revealed minimal internal vascularity. The rest of the testis showed normal flow.
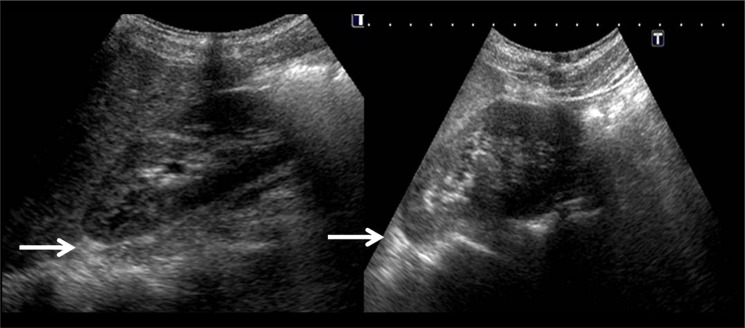
Figure 4.
Abdominal ultrasound did not reveal any mass lesion in the suprarenal region, ruling out adrenal gland enlargement.
Discussion
Benign intratesticular masses are rare. Testicular adrenal rest tumours are benign testicular masses that are found in approximately 29% percent of patients with CAH, usually in adolescents or young adults aged between 20–40 years [2]. TARTs were first described in 1940 by Wilkins et al. [3]. As regards the pathogenesis of TARTs, the following hypotheses have been proposed:
Testicular, hilar pluripotent cells proliferate due to increased ACTH levels.
An embryological hypothesis states that aberrant adrenal cortical tissue, which adhered to the gonads and descended during prenatal life with the testes or ovaries, proliferates due to ACTH stimulation [4].
Foci of ectopic adrenal rest tissue have been found in neonates in the retroperitoneum, broad ligament, testes, ovaries, an inguinal canal. These foci usually regress in adulthood; however, in patients with CAH, testicular adrenal rest tumours can occur, with prevalence rates of up to 94% [4]. These lesions are bilateral and usually situated within the rete testis; hence, they are not palpable unless larger than 2 cm. Ultrasound assessment, being cheap and easily available, is a good screening modality for patients with CAH. If not detected early, TARTs can expand and destroy normal parenchyma, inhibit spermatogenesis, and lead to infertility.
On ultrasound, TARTs present as lobular, elongated, predominantly hypoechoic masses with sharp margins; the size ranges from 4 to 38 mm [5]. Larger lesions (>2 cm) can have areas of increased echogenicity and appear heterogeneous. They are usually bilateral and located inside or close to the testicular hilus [6]. Cystic areas and calcifications are not seen. These features were found in our patient. TARTs can present as single nodules, as in our patient, but also as multiple nodules (in 16% of patients in a study by Kang et al. [7]). In the study by Wang et al., colour Doppler revealed that 76.7% (23/30) of TARTs exhibited a rich blood supply, whereas 23.3% (7/30) exhibited a scarce blood supply [8]. In the study by Delfino et al., colour Doppler sonography showed perilesional flow in 2 patients, perilesional and intralesional flow in 1, and no flow in the remaining 8 patients. Thus, there have been mixed reports regarding the vascularity of these tumours. In our study, the lesion showed minimal internal vascularity. Avila et al. determined that ultrasound is as sensitive as MRI in detection of adrenal rests tumours [9]. Thus, ultrasound is the modality of choice, given its easy availability and low cost, as compared to MRI. MRI-based assessment is preferred only prior to testis sparing surgery, in order to map the exact size and extent of the tumour. On MRI, TARTs appear iso- to hyperintense on T1-weighted images and hypointense on T2-weighted images, relative to the normal parenchyma.
The major differential diagnoses include synchronous malignant testicular tumours; however, they are rare and occur in 1–2% of patients [10].Ultrasound also helps differentiate TARTs from other testicular tumours that are found in a similar age group and in patients with precocious puberty (like Leydig cell tumours). These tumours are usually unilateral but can be bilateral in 3% of patients, whereas TARTs are bilateral in more than 80% cases [11]. Other tumours can occur anywhere in the testis, as compared to the specific location in the rete testis for TART’s, and show internal vascularity. Another differential diagnosis is Leydig cell hyperplasia which is usually solitary; distinction is made based on its size (less than 5 mm). However, sometimes testicular tumours are indistinguishable, based on solely on radiological findings. Therefore, clinical and hormonal features should be taken into consideration. Lymphomas and leukaemias can present as bilateral testicular masses; however, they tend to occur in older patients and are not typically included in the differential diagnosis of testicular masses in young people.
Ultrasound also can be employed for monitoring TARTs. Lesions increase or decrease in size, depending on hormonal status. TARTs regress in size and decrease in number in patients with good hormonal control and increase in patients with poor hormonal control. Claahsen-van der Grinten et al. proposed that screening for TARTs in male patients with CAH should start at the age of 8 years [12]. Therefore, scrotal ultrasound screening must be performed at least before puberty and regularly during adulthood in male patients with CAH in order to prevent infertility. Zehra Aycan et al. recommended that regular screening by scrotal ultrasound should be performed at least every 2 years in early childhood and annually in the peripubertal period [13,14].
Conclusions
Sonographic characteristics of TARTs include bilateral growth, location adjacent to the testicular mediastinum, presence of well-defined lobulated hypoechoic masses, and vascular flow on colour Doppler. Also, early testicular imaging is important in patients with congenital adrenal hyperplasia and should be performed at puberty, as these tumours can enlarge, destroy normal testicular tissue, and lead to infertility. Most importantly, radiologists must be aware of the above-mentioned characteristics of these benign tumours in order to avoid unnecessary biopsies and orchidectomies in patients with CAH.
Conflicts of interest
None.
References
- 1.Olpin JD, Witt B. Testicular adrenal rest tumors in a patient with congenital adrenal hyperplasia. J Radiol Case Rep. 2014;8(2):46–53. doi: 10.3941/jrcr.v8i2.1489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Proto G, DiDonna A, Grimaldi F, Mazzolini A. Bilateral testicular adrenal rest tissue in congenital adrenal hyperplasia: US and MR features. J Endocrinol Invest. 2001;24:529–31. doi: 10.1007/BF03343887. [DOI] [PubMed] [Google Scholar]
- 3.Wilkins L, Fleishmann W, Howard JE. Macrogenitosomia precox associated with hyperplasia of the androgenic tissue of the adrenal and death from corticoadrenal insufficiency. Endocrinology. 1940;26:385–95. [Google Scholar]
- 4.Stikkelbroeck NM, Otten BJ, Pasic A, et al. High prevalence of testicular adrenal rest tumors, impaired spermatogenesis, and Leydig cell failure in adolescent and adult males with congenital adrenal hyperplasia. J Clin Endocrinol Metab. 2001;86:5721–28. doi: 10.1210/jcem.86.12.8090. [DOI] [PubMed] [Google Scholar]
- 5.Delfino M, Elia J, Imbrogno N, et al. Testicular adrenal rest tumors in patients with congenital adrenal hyperplasia: Prevalence and sonographic, hormonal, and seminal characteristics. J Ultrasound Med. 2012;31(3):383–88. doi: 10.7863/jum.2012.31.3.383. [DOI] [PubMed] [Google Scholar]
- 6.Dogra V, Nathan J, Bhatt S. Sonographic appearance of testicular adrenal rest tissue in congenital adrenal hyperplasia. J Ultrasound Med. 2004;23:979–81. doi: 10.7863/jum.2004.23.7.979. [DOI] [PubMed] [Google Scholar]
- 7.Kang MJ, Kim JH, Lee SH, et al. The prevalence of testicular adrenal rest tumors and associated factors in postpubertal patients with congenital adrenal hyperplasia caused by 21-hydroxylase deficiency. Endocr J. 2011;58:501–8. doi: 10.1507/endocrj.k11e-034. [DOI] [PubMed] [Google Scholar]
- 8.Wang Z, Yang Z, et al. Diagnosis of testicular adrenal rest tumors on ultrasound. Medicine (Baltimore) 2015;94(36):e1471. doi: 10.1097/MD.0000000000001471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Avila NA, Premkumar A, Shawker TH, et al. Testicular adrenal rest tissue in congenital adrenal hyperplasia: findings at gray-scale and color Doppler US. Radiology. 1996;198:99–104. doi: 10.1148/radiology.198.1.8539414. [DOI] [PubMed] [Google Scholar]
- 10.Dieckmann KP, Boeckmann W, Brosig W, et al. Bilateral testicular germ cell tumors: report of 9 cases and review of the literature. Cancer. 1986;57:1254–58. doi: 10.1002/1097-0142(19860315)57:6<1254::aid-cncr2820570632>3.0.co;2-2. [DOI] [PubMed] [Google Scholar]
- 11.Claahsen-van der Grinten HL, Otten BJ, Stikkelbroeck MM, et al. Testicular adrenal rest tumours in congenital adrenal hyperplasia. Best Pract Res Clin Endocrinol Metab. 2009;23:209–20. doi: 10.1016/j.beem.2008.09.007. [DOI] [PubMed] [Google Scholar]
- 12.Claahsen-van der Grinten HL, Hermus AR, Otten BJ. Testicular adrenal rest tumours in congenital adrenal hyperplasia. Int J Pediatr Endocrinol. 2009;2009:624823. doi: 10.1155/2009/624823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yu MK, Jung MK, Kim KE, et al. Clinical manifestations of testicular adrenal rest tumor in males with congenital adrenal hyperplasia. Ann Pediatr Endocrinol Metab. 2015;20(3):155–61. doi: 10.6065/apem.2015.20.3.155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Aycan Z, Bas VN, Cetinkaya S, et al. Prevalence and long-term follow-up outcomes of testicular adrenal rest tumours in children and adolescent males with congenital adrenal hyperplasia. Clin Endocrinol (Oxf) 2013;78(5):667–72. doi: 10.1111/cen.12033. [DOI] [PubMed] [Google Scholar]