Abstract
Introduction
Tuberculosis is a highly infectious disease affecting humans and animals. It is caused by the Mycobacterium tuberculosis complex (MTBC) - Mycobacterium bovis and Mycobacterium caprae, which are aetiological factors of bovine tuberculosis (bTB). In Poland, the bTB eradication programme exists. Animals diagnosed with tuberculosis are in the majority of cases not treated, but removed from their herd and then sanitary slaughtered.
Material and Methods
In total, 134 MTBC strains isolated from cattle in Poland were subjected to microbiological analysis. The resistance phenotype was tested for first-line antimycobacterial drugs used in tuberculosis treatment in humans: streptomycin, isoniazid, rifampicin, ethambutol, and pyrazinamide. The strains were isolated from tissues collected post mortem, so the test for drug resistance fulfilled only epidemiological criterion.
Results
The analysis of drug-resistance of MTBC strains revealed that strains classified as M. bovis were susceptible to 4 antimycobacterial drugs: isoniazid, rifampicin, streptomycin, and ethambutol, and resistant to pyrazynamide. The strains classified as M. caprae were sensitive to all tested drugs.
Conclusion
The results indicate that despite enormously dynamic changes in mycobacterial phenotype, Polish strains of MTBC isolated from cattle have not acquired environmental resistance. The strains classified as M. bovis are characterised by natural resistance to pyrazinamide, which is typical for this species.
Keywords: cattle, bovine tuberculosis, Mycobacterium tuberculosis complex, antimycobacterial drugs, Poland
Introduction
Tuberculosis is a highly infectious disease, affecting both humans and animals, caused by the Mycobacterium tuberculosis complex (MTBC) (29) - Mycobacterium bovis and Mycobacterium caprae, which are aetiological factors of bovine tuberculosis (bTB, bovine TB) (1, 15). According to the classification of the World Health Organisation (WHO), bTB is rated as direct zoonosis, involving direct transmission of an infectious agent from an infected animal to a sensitive animal (human), without participation of an indirect host. Due to the direction of infectious agent transmission, bovine TB can be also rated as reverse zoonosis.
Poland has an established national programme of eradication of bTB, functioning in accordance with the EU Directive 64/432 (10). Presently, the obligation to eradicate this zoonosis is imposed in the “Act on Veterinary Inspection” and the “Act on animal health protection and infectious diseases eradication”. The methods of control of bTB in Poland and the procedures in case of suspicion, verification, and stamping out of a bTB outbreak are described in the Regulation of the Minister of Agriculture and Rural Development from 23 November 2004. In 2009, Poland gained the status of the country free from this zoonosis (8). However, in the past five years 12 - 18 outbreaks of bovine TB have been reported (17). Animals diagnosed with tuberculosis are in most cases not treated, but removed from their herd and then sanitary slaughtered. The only described case of tuberculosis treatment in animals in Poland regarded a 10-year-old male giraffe kept in the Silesian Zoological Garden in Chorzów (20).
First results of the research on resistance to streptomycin (SM), isoniazid (INH), rifampicin (RMP), and ethambutol (EMB) - S.I.R.E, and pyrazinamide (PZA) of MTBC strains isolated from tissues of different animal species were published in Poland in 2011. This study is a continuation of research on MTBC strains isolated from cattle.
Material and Methods
MTBC isolates
Research material, consisting of 134 MTBC strains isolated from domestic cattle (Bos taurus), was collected from 10 provinces: West Pomeranian, Masovian, Podlasie, Lesser Poland, Pomeranian, Lower Silesian, Silesian Łödź, Holy Cross, and Greater Poland. The strains were isolated from animal tissues retrieved post mortem in such way that estimation of drug resistance in this task met only the epidemiological criterion. The isolates were cultured on the Stonebrink medium. Affiliation to MTBC was confirmed by immunochromatographic test for detection of MPT64 protein fraction produced by MTBC cells within the culture (Identification Test MGIT TBC®, Becton and Dickinson, USA). Species identification was conducted with GenoType MTBC test (Hain Lifescience, Germany), which allowed to classify 103 strains as Mycobacterium bovis and 31 strains as Mycobacterium caprae.
The Bactec method for determination of S.I.R.E
Test for resistance to S.I.R.E. was done with the use of Bactec MGIT 960 and Middlebrook 7H10 Agar (Bactec MIGIT 960 SIRE Kits, Becton, Dickinson, USA) at the concentration limit of 1.0 ug/mL for SM, 0.1 ug/mL for INH, 1.0 ug/mL for RMP, and 5.0 ug/mL for EMB. Analysis of fluorescence in test tubes containing drugs compared with fluorescence in control test tubes allowed to define sensitivity.
The Bactec method for determination of PZA
The estimation of drug resistance to PZA was carried out on the isotopic Bactec 960-Tb system (27), Middlebrook's liquid culture medium 7H12 containing palmitic acid 14C, at pH 5.9-6.0, with PZA concentration limit of 100 ug/mL (Bactec MIGIT 960 PZA Kit, Becton, Dickinson, USA). Growing bacteria metabolise the substrate present in the culture medium up to 14CO2. The amount of produced 14CO2 is directly proportional to the intensity of population growth in culture medium. The addition of PZA to culture media results in cessation of M. caprae growth. This is reflected by a decrease in amount of 14CO2 in relation to the control, in which the amount of 14CO2 and growth index increase systematically. In the case of resistant strains there is little or no difference in the growth index between the control and an analysed strain (26).
Results
In the group of 103 bovine bacillus strains identified as M. bovis, all strains demonstrated susceptibility to four basic antimycobacterial drugs: INH, RMP, SM, and EMB, and were resistant to PZA (Fig. 1). In the group of 31 strains identified as M. caprae, all strains demonstrated susceptibility to PZA and to INH, RMP, SM, and EMB. These results indicating no drug resistance except for PZA were identical to our findings of 2013 when 58 cattle isolates were examined (19).
Fig. 1.
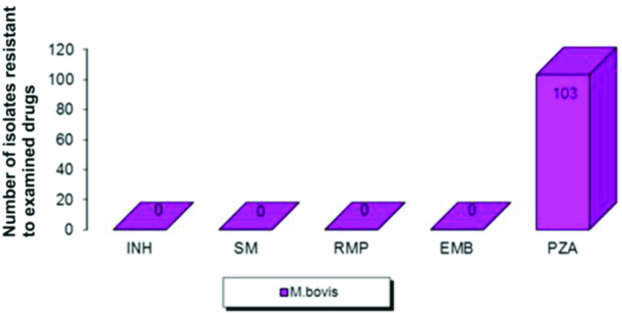
The susceptibility of M. bovis strains to S.I.R.E. and PZA
Discussion
All confirmed M. caprae strains were susceptible to all examined drugs. Data regarding drug susceptibility of M. caprae TB in humans and animals is limited. Strains classified as M. bovis are marked by a feature which is unique for this type, namely their natural resistance to PZA, which was proved in the study. These results indicating no drug resistance, except to PZA, were identical with research by Daly et al. (12) and Fitzgerald et al. (13). The material for their study consisted of 100 strains of M. bovis: 91 from deer, 4 from coyotes, 3 from cattle, and 2 from raccoons. The obtained results are similar due to the lack of antimycobacterial therapy which, if applied to either cattle or wildlife species, would cause them to develop environmental resistance. Resistance of cattle M. bovis strains to INH and RMP was reported in Northern Italy (7). Fifteen out of 61 tested strains were resistant to either INH or RMP, or both drugs, showing multidrug resistance (MDR). Detection of resistant cattle M. bovis strains appears of great interest.
The literature describes also MDR M. bovis strains from human clinical samples (33). This type of resistance is mainly observed in countries with a high prevalence of HIV infection (4). The emergence of drug-resistant M. bovis is an important public health problem that jeopardises the success of human TB control programmes.
The incidence rate of bovine TB among people in Europe is higher than in last few years. That is why establishing the phenotype of resistant MTBC strains retrieved from animals on anti-tubercle bacilli and TB treatment in humans seems to be justified in the public health aspect. Data pertaining to incidence of bovine tuberculosis among humans in 2014, which were presented in the EFSA report, were gathered from 27 EU members (excluding France) and Iceland, Norway, and Switzerland. As many as 134 incidents of M. bovis were confirmed, among which the highest number occurred in Germany - 45 people, next in UK - 29, and in Spain - 25 (28).
The WHO estimates that in the countries where programmes against TB were insufficiently monitored, 1% of TB incidence is caused by tubercle bacilli. So far, there have been two incidents of bovine TB among people in Poland, specifically in the south of the country (16). According to the EFSA report, the main source of M. bovis in humans is the milk originating from infected cows.
It should be emphasised that wildlife, especially infected wild animals living in the vicinity of households (9), is considered one of the main reservoirs of these microorganisms which may be transmitted directly and indirectly to livestock (common pasture) and even humans. Although the biggest natural reservoir of M. bovis in Poland is sick cattle (17), the main source of tuberculosis in Spain is the wild boar (3), and the reservoir of M. bovis in England is the population of badgers (14). There was one incident noted in the area of Cornwall (England) where a veterinary nurse had a pulmonary M. bovis TB infection. Prior to that, the patient assisted in tuberculin tests of cattle herds and had contact with badgers in the past (reservoir of M. bovis). Four months later, the dog in the patient's household displayed pulmonary symptoms of bTB. The genotype of isolated M. bovis strains was identical, which allowed to establish the common source of infection (31).
M. bovis can cause tuberculosis in other livestock species as well. It is of high virulence under natural conditions among goats (11), pigs (2, 5), sheep (25), and cats (23), whereas it is lower in horses (24) and dogs (32).
Cattle demonstrates low susceptibility to tubercle bacillus, but some incidents of M. tuberculosis among cattle were reported as well (18).
The most essential components of modern programmes that help to reduce tuberculosis incidence are primarily its early diagnosis (21, 30) and appropriately selected anti-tuberculosis treatment (22). The biggest obstacle in carrying out programmes against tuberculosis all over the world is the ever-increasing occurrence of the bacillus drug resistance phenomenon as well as the absence of new drugs (7) since the invention of RMP method in 1965. Drug-resistant tuberculosis, especially its specific MDR strains and extensively drug resistant (XDR) strains, is a disease of great mortality, with the risk especially increased in people diagnosed with HIV (21, 34).
Footnotes
Conflict of Interests Statement: The authors declare that there is no conflict of interests regarding the publication of this article.
Financial Disclosure Statement: The research was financed from the statutory funds of the National Veterinary Research Institute in Pulawy, Task No. S/188, and the National Tuberculosis and Lung Diseases Research Institute, Task No 1.23.
Animal Rights Statement: None required.
References
- 1.Aranaz A., Cousins D., Mateos A., Domínguez L.. Elevation of Mycobacterium tuberculosis subsp. caprae Aranaz et al. 1999 to species rank as Mycobacterium caprae comb. nov., sp. nov. Int J Syst Evol Microbiol. 2003;53:1785–1789. doi: 10.1099/ijs.0.02532-0. [DOI] [PubMed] [Google Scholar]
- 2.Bailey S.S., Crawshaw T.R., Smith N.H., Palgrave C.J.. Mycobacterium bovis infection in domestic pigs in Great Britain. Vet J. 2013;198:391–397. doi: 10.1016/j.tvjl.2013.08.035. [DOI] [PubMed] [Google Scholar]
- 3.Beltrán-Beck B., Ballestros C., Vicente J., de la Fuente J., Gortäzar C.. Progress in oral vaccination against tuberculosis in its main wildlife resevoir in Iberia, the Eurasian wild boar. Vet Med Int. 2012 doi: 10.1155/2012/978501">10.1155/2012/978501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bobadilla-del Valle M., Torres-Gonzälez P., Cervera-Hernändez M.E., Martínez-Gamboa A., Crabtree-Ramirez B., Chävez-Mazari B., Ortiz-Conchi N., Rodríguez-Cruz L., Cervantes-Sänchez A., Gudiño-Enríquez T., Cinta-Severo C., Sifuentes-Osornio J., Ponce de Leön A.. Trends of Mycobacterium bovis isolation and first-line anti-tuberculosis drug susceptibility profile: A fifteen-year laboratory-based surveillance. PLoS Negl Trop Dis. 2015 doi: 10.1371/journal.pntd.0004124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Campbell T.A., Long D.B., Bazan L.R., Thomsen B.V., Robbe-Austerman S., Davey R.B., Soliz L.A., Swafford S.R., Ver Cauteren K.C.. Absence of Mycobacterium bovis in feral swine (Sus scrofa) from the southern Texas border region. J Wildl Dis. 2011;47:974–978. doi: 10.7589/0090-3558-47.4.974. [DOI] [PubMed] [Google Scholar]
- 6.Cavirani S., Fanti F., Benecchi M., Calderaro A., Taddei S., Arcangeletti C., Medici M.C., Dettori G., Chezzi C.. Evaluation of susceptibility of Mycobacterium bovis to antituberculous drugs by radiometric BACTEC 460TB system. New Microbiol. 2003;26:181–186. [PubMed] [Google Scholar]
- 7.Chiu Chang K., Wai Yew W., Chiu R., Chan Y.. Rapid assays for fluoroquinolone resistance in Mycobacterium tuberculosis: a systematic review and meta-analysis. J Antimicrob Chemother. 2010;65:1551–1561. doi: 10.1093/jac/dkq202. [DOI] [PubMed] [Google Scholar]
- 8.Commission Decision 2009/342/EC as regards the declaration that certain administrative regions of Poland are officially free of zoonotic - bovine - leukosis and that Poland and Slovenia are officially free of bovine tuberculosis. Oj L. 2009 Apr 24;104:51–56. [Google Scholar]
- 9.Corner L.A.. The role of wild animal populations in the epidemiology of tuberculosis in domestic animals: how to assess the risk. Vet Microbiol. 2006;112:303–312. doi: 10.1016/j.vetmic.2005.11.015. [DOI] [PubMed] [Google Scholar]
- 10.Council Directive 64/432/EWG of 26 June 1964 on animal health problems affecting intra-Community trade in bovine animals and swine. OJ L. 1964 Jul 29;121:1977–2012. [Google Scholar]
- 11.Crawshaw T., Daniel R., Clifton-Hadley R., Clark J., Evans H., Rolfe S., de la Rua-Domenech R.. TB in goats caused by Mycobacterium bovis. Vet Rec. 2008;163:127. doi: 10.1136/vr.163.4.127. [DOI] [PubMed] [Google Scholar]
- 12.Daly M., Diegel K.L., Fitzgerald S.D., Schooley A., Berry D.E., Kaneene J.B.. Patterns of antimicrobial susceptibility in Michigan wildlife and bovine isolates of Mycobacterium bovis. J Vet Diagn Invest. 2006;18:401–404. doi: 10.1177/104063870601800416. [DOI] [PubMed] [Google Scholar]
- 13.Fitzgerald S.D., Schooley A.M., Berry D.E., Kaneene J.B.. Antimicrobial susceptibility testing of Mycobacterium bovis isolates from Michigan white-tailed deer during the 2009 hunting season. Vet Med Int. 2011 doi: 10.4061/2011/903683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gallagher J., Clifton-Hadley R.S.. Tuberculosis in badgers; a review of the disease and its significance for other animals. Res Vet Sci. 2000;69:203–217. doi: 10.1053/rvsc.2000.0422. [DOI] [PubMed] [Google Scholar]
- 15.Karlson A.G., Lessel E.F.. Mycobacterium bovis nom. Nov Int J Syst Bacteriol. 1970;20:273–282. [Google Scholar]
- 16.Kozińska M., Zientek J., Augustynowicz-Kopeć E., Zwolska Z., Kozielski J.. Transmission of tuberculosis among people living in the border areas of Poland, the Czech Republic, and Slovakia. Pol Arch Med Wewn. 2016;126:32–40. doi: 10.20452/pamw.3263. [DOI] [PubMed] [Google Scholar]
- 17.Krajewska M., Kozińska M., Kubajka M., Weiner M., Augustynowicz-Kopeć E., Bełkot Z., Lipiec M., Szulowski K.. Tuberculosis in humans and in animals - current epidemiological data. Życie Wet. 2015;90:647–651. [Google Scholar]
- 18.Krajewska M., Kozińska M., Zwolska Z., Lipiec M., Augustynowicz-Kopeć E., Szulowski K.. Human as a source of tuberculosis for cattle. First evidence of transmission in Poland. Vet Microbiol. 2012;159:269–271. doi: 10.1016/j.vetmic.2012.04.001. [DOI] [PubMed] [Google Scholar]
- 19.Krajewska M., Napiörkowska A., Augustynowicz-Kopeć E.. Pyrazinamide resistance among Mycobacterium bovis strains isolated from animals in Poland. Życie Wet. 2013;88:575–577. [Google Scholar]
- 20.Krajewska M., Załuski M.. Tuberculosis in giraffe - attempt to treatment. Abstract of 2nd Conference Pharmacology Department of University of Life Sciences in Lublin: Pharmacological and environmental aspects of rational therapy. Krynica Görska, Poland. 2012:20. [Google Scholar]
- 21.Kruczak K., Niżankowska-Mogilnicka E.. Multidrug resistance tuberculosis - current problems. Pneumol Alergol Pol. 2009;77:276–283. [PubMed] [Google Scholar]
- 22.Michałowska-Mitczuk D.. Pharmacotherapy of tuberculosis. Post Farmakoterapii. 2009;65:51–58. [Google Scholar]
- 23.Monies R.J., Cranwell M.P., Palmer N., Inwald J., Hewinson R.G., Rule B.. Bovine tuberculosis in domestic cats. Vet Rec. 2000;146:407–408. doi: 10.1136/vr.146.14.407. [DOI] [PubMed] [Google Scholar]
- 24.Monreal L., Segura D., Segalés J., Garrido J.M., Prades M.. Diagnosis of Mycobacterium bovis infection in a mare. Vet Rec. 2001;149:712–714. [PubMed] [Google Scholar]
- 25.Muñoz Mendoza M., Juan L.D., Menéndez S., Ocampo A., Mourelo J., Säez J.L., Dominguez L., Gortäzar C., García Marin J.F., Balseiro A.. Tuberculosis due to Mycobacterium bovis and Mycobacterium caprae in sheep. Vet J. 2012;191:267–269. doi: 10.1016/j.tvjl.2011.05.006. [DOI] [PubMed] [Google Scholar]
- 26.Napiörkowska A., Zwolska Z., Augustynowicz-Kopeć E.. Resistance to pyrazinamide among Mycobacterium tuberculosis strains isolated from patients from the Mazowian Voivodeship in 2008 - 2010 years. Post Nauk Med. 2011;24:819–823. [Google Scholar]
- 27.Olesiejuk R., Maciak L., Milanowski J.. Meaning of the radiometric method Bactec 460 TB for the application of antituberculosis therapy. Pneumol Alergol Pol. 2002;70:5–11. [PubMed] [Google Scholar]
- 28.Osek J., Wieczorek K.. Prevalence of zoonoses in animals and zoonotic agents in food in Europe in 2013. Życie Wet. 2015;90:210–216. [Google Scholar]
- 29.Rodriguez-Campos S., Smith N.H., Boniotti M.B., Aranaz A.. Overview and phylogeny of Mycobacterium tuberculosis complex organisms: implication for diagnostics and legislation of bovine tuberculosis. Res Vet Sci. 2014;97:5–19. doi: 10.1016/j.rvsc.2014.02.009. [DOI] [PubMed] [Google Scholar]
- 30.Safinowska A., Walkiewicz R., Nejman-Gryz P., Grubek-Jaworska H.. Two selected commercially based nucleic acid amplification tests for the diagnosis of tuberculosis. Pneumonol Alergol Pol. 2012;80:6–12. [PubMed] [Google Scholar]
- 31.Shrikrishana D., de la Rua-Domenech R., Smith N.H., Colloff A., Coutts I.. Human and canine pulmonary Mycobacterium bovis infections in the same household: re-emergence of an old zoonotic threat? Thorax. 2009;64:89–91. doi: 10.1136/thx.2008.106302. [DOI] [PubMed] [Google Scholar]
- 32.van der Burgt G.M., Crawshaw T., Foster A.P., Denny D.J., Schock A.. Mycobacterium bovis infection in dogs. Vet Rec. 2009;165:634. doi: 10.1136/vr.165.21.634. [DOI] [PubMed] [Google Scholar]
- 33.Vazquez-Chacon C.A., Martínez-Guarneros A., Couvin D., Gonzälez-Y-Merchand J.A., Rivera-Gutierrez S., Escobar-Gutierrez A., De-la-Cruz Löpez J.J., Gomez-Bustamante A., Gonzalez-Macal G.A., Gonçalves Rossi L.M., Muñiz-Salazar R., Rastogi N., Vaughan G.. Human multidrug-resistant Mycobacterium bovis infection in Mexico. Tuberculosis (Edinb) 2015 doi: 10.1016/j.tube.2015.07.010. [DOI] [PubMed] [Google Scholar]
- 34.World Health Organization. TB and HIV Programme. http://www.who.int/tb/areas-of-work/tb-hiv/en/


