Abstract
Background
Percutaneous coronary intervention (PCI) had become the major therapeutic procedure for coronary artery disease (CAD), but the high rate of in-stent restenosis (ISR) still remained an unsolved clinical problem in clinical practice. Increasing evidences suggested that diabetes mellitus (DM) was a major risk factor for ISR, but the risk predictors of ISR in CAD patients with DM had not been well characterized. The aim of this study was to investigate the clinical and angiographic characteristic predictors significantly associated with the occurrence of ISR in diabetic patients following coronary stenting with drug-eluting stent (DES).
Methods
A total of 920 patients with diabetes who diagnosed CAD and underwent coronary DES implantation at Beijing Anzhen Hospital in China were consecutively enrolled from January 2012 to December 2012. Of these, 440 patients underwent the second angiography within ≥ 6 months due to the progression of treated target lesions. Finally, 368 of these patients who met the inclusion and exclusion criteria were followed up by angiography after baseline PCI. According to whether ISR was detected at follow-up angiography, patients were divided into the ISR group (n = 74) and the non-ISR group (n = 294). The independent predictors of ISR in patients with DM were explored by multivariate Cox's proportional hazards regression models.
Results
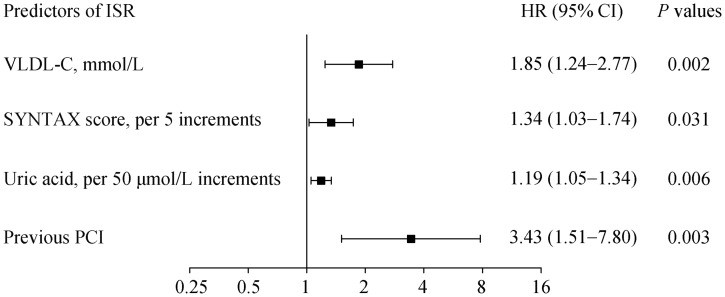
A total of 368 patients (260 women and 108 men) with a mean ages of 58.71 ± 10.25 years were finally enrolled in this study. Of these, ISR occurred in 74/368 diabetic patients (20.11%) by follow-up angiography. Univariate analysis showed that most baseline characteristics of the ISR and non-ISR group were similar. Patients in the ISR group had significantly higher serum very low density lipoprotein cholesterol (VLDL-C), triglyceride (TG) and uric acid (UA) levels, more numbers of target vessel lesions, higher prevalence of multi-vessel disease, higher SYNTAX score, higher rate of previous but lower rate of drinking compared with patients in the non-ISR group. The independent predictors of ISR in patients with DM after DES implantation included VLDL-C (HR = 1.85, 95% CI: 1.24–2.77, P = 0.002), UA (per 50 µmol/L increments, HR = 1.19, 95% CI: 1.05–1.34, P = 0.006), SYNTAX score (per 5 increments, HR = 1.34, 95% CI: 1.03–1.74, P = 0.031) and the history of PCI (HR = 3.43, 95% CI: 1.57–7.80, P = 0.003) by the multivariate Cox's proportional hazards regression analysis.
Conclusions
The increased serum VLDL-C and UA level, higher SYNTAX score and the history of previous PCI were independent predictors of ISR in patients with DM after coronary DES implantation. It provided new evidence for physicians to take measures to lower the risk of ISR for the better management of diabetic patients after PCI.
Keywords: Diabetes mellitus, In-stent restenosis, Uric acid, SYNTAX score, Very low density lipoprotein cholesterol
1. Introduction
Percutaneous coronary intervention (PCI) has been the major therapeutic procedure for coronary artery disease (CAD) for about three decades.[1],[2] However, several large-scale clinical trials had confirmed that even in the drug-eluting stent (DES) ear, in-stent restenosis (ISR) occurred in 3% to 20% of patients after coronary stenting implantation,[3] which still remained an unsolved clinical problem in clinical practice. Therefore, exploring the reliable risk factors to predict ISR and defining the subgroups of patients at increased risk for ISR would be of massive utility for patients risk assessment and stratification.[4],[5]
Accumulating evidences suggested that diabetes mellitus (DM) played a pivotal role in ISR development.[6] Diabetic patients appeared to have a 2–4 times higher risk of developing ISR after PCI compared to non-diabetic patients.[7] Patients with DM have more complex coronary lesion anatomy with small and diffusely diseased vessels.[8] Furthermore, patients with DM often have hypertriglyceridemia and systemic prothrombotic conditions related to the activation of the platelet aggregation and coagulation systems.[9],[10] Altogether, these made diabetic patients a challenging subpopulation to give more attention and treatment.
To the best of our knowledge, so far, most previous related studies focus on exploring the risk factor of ISR among general CAD patients,[11]–[13] the risk predictors of ISR in CAD patients with DM had not been well characterized. Additionally, China had over 92.4 million diabetic patients (9.7% of the adult population), which ranked at top one with the numbers of DM patients and bore a higher diabetes-related burden than other countries.[14]
Therefore, the aim of our study was to investigate the clinical and angiographic characteristic predictors for functionally significant in occurrence of ISR in diabetic patients after coronary stenting with DES for further understanding the underlying molecular and cellular mechanisms of ISR. This study will provide new evidence for better treatment and prevention of ISR in CAD patients with DM.
2. Methods
2.1. Study patients
A total of 920 patients with diabetes who diagnosed CAD and underwent coronary DES implantation at Beijing Anzhen Hospital (Beijing, China) were consecutively enrolled from Junuary 2012 to December 2012. The inclusion criteria were post PCI patients who had undergone follow-up angiography within ≥ 6 months. Of 920 patients, 480 patients underwent the second angiography due to the progression of untreated nontarget lesions, remaining 440 patients were further selected from the study if they met the exclusion rules.
A total of 440 patients with diabetes who diagnosed coronary heart disease (CHD) and underwent coronary drug-eluting stent (DES) implantation at Beijing Anzhen Hospital (Beijing, China) were consecutively enrolled from January 2012 to December 2012. The inclusion criteria were post PCI patients who had undergone follow-up angiography within ≥ 6 months. Patients were excluded from the study if they met the exclusion rules: (1) patients who died in-hospital after baseline PCI (n = 8); (2) patients with myocardial infarction (MI) within one month of baseline PCI (to exclude potential subacute stent thrombosis of the intervened arterial segment) (n = 12); (3) patients without sufficient clinical and angiographic data at baseline and follow up (n = 52). During the study period, 368 of these patients who met the inclusion and exclusion criteria were followed up by angiography after baseline PCI. Of these, 74 patients with 126 lesions were treated with repeat DES (re-DES) implantation after the occurrence of ISR in the target vessels.
This study was approved by the Clinical Research Ethics Committee of Beijing Anzhen Hospital, Capital Medical University, and all patients provided written informed consent for participation in this study.
2.2. Stent implantation
All patients received DES implantation in our catheterization center. Stent implantation was performed according to current practice guidelines, and stents were selected by experienced interventional cardiologists. During the procedure, patients received a bolus of 100 IU/kg heparin, with a repeated bolus of 2000 IU heparin to maintain the activated clotting time of ≥ 300 s. All patients received aspirin (100 mg/day was administered) and clopidogrel (300 mg loading dose, followed by 75 mg/day for at least 12 months). When ISR was diagnosed, patients were treated with re-DES implantation. Procedural success was defined as reduction of stenosis to less than 10% residual narrowing, thrombolysis in myocardial infarction (TIMI) flow grade III, with improvement in ischemic symptoms, and without major procedure related complications.[15]
2.3. Data collection
A standard case report form was used to collect patients' clinical and demographic characteristics including age, sex, smoking, drinking, CAD risk factors, family history, life style, medical history and coronary angiographic information at baseline PCI and follow-up angiography. During a physical examination, anthropometric indices such as weight, height and blood pressure (BP) were measured. Body mass index (BMI) was calculated as weight in kilograms divided by the square of the height in meters.
Coronary angiograms data like the minimal stent diameter, average stent length and the percent diameter stenosis were also recorded by two experienced investigators at baseline and follow-up for coronary angiography analysis.
2.4. Laboratory analysis
Venous blood samples were collected after an overnight for testing the lipid profiles, fasting blood glucose (FBG), high-sensitivity C-reactive protein (hs-CRP) and uric acid (UA) level using standard laboratory method at baseline PCI and follow-up angiography. The total cholesterol (TC), TG, FBG and UA were determined according to enzymatic methods. The low density lipoprotein cholesterol (LDL-C) and high density lipoprotein cholesterol (HDL-C) levels were measured by homogeneous assays (Daiichi, Tokyo, Japan). The Non-high density lipoprotein cholesterol (Non-HDL-C) level was calculated by TC minus HDL-C, and very low density lipoprotein cholesterol (VLDL-C) level was calculated as TC minus LDL-C and HDL-C according to the recommendation of lipid guidelines.[16],[17]
2.5. Disease definitions
The primary end point of the study was the occurrence of ISR. ISR was defined as a diameter stenosis of ≥ 50% occurring in the segment inside the stent or a 5 mm proximal or distal to the stent at follow-up angiography.[18],[19] According to whether ISR was detected, patients were classified into two groups: the ISR group and the non-ISR group. Target lesion was considered the most severe narrowing, identified by angiographic appearance with Electrocardiograph (ECG) changes. Multivessel disease (MVD) was defined as a diameter stenosis of ≥ 50% occurring in two or more vessels.
Diabetes mellitus was defined as either a previous diagnosis of DM treated with diet, oral agents or insulin or a new diagnosis of DM if FBG ≥ 7.0mmol/L on two occasions during hospitalization.[20] Hypertension was defined by systolic blood pressure (SBP) ≥ 140 mmHg and/or diastolic blood pressure (DBP) ≥ 90 mmHg, and/or the use of antihypertensive treatment in the past two weeks.[21] Low levels of HDL-C were defined as < 1.04 mmol/L.[22] The severity of coronary artery lesions was quantified with Synergy Between PCI With Taxus and Cardiac Surgery (SYNTAX) score, which was calculated using the online calculator for SYNTAX score.[23]
2.6. Statistical analysis
Continuous variables were expressed as mean ± SD in case of normal distribution and differences between two groups were determined by two-sided t-test. Data were expressed as medians (interquartile ranges, P25, P75) in case of skewed distribution and compared between two groups using the Mann-Whitney test. Categorical variables are presented as counts (percentages) and compared by Chi-square test.
Univariate Cox's proportional hazards regression modeling was performed to identify determinants of ISR in diabetic patients firstly. Baseline variables were selected if they had either a clinically plausible relation with the ISR or appeared to be imbalanced between ISR and non-ISR patients with a P-value < 0.2. The potential variables were entered into multivariate Cox's proportional hazards regression modeling using the stepwise method (entry, 0.05; removal, 0.05) to determine their independent risk associated with ISR in diabetes. The variables included traditional risk factors (age, gender, BMI, current smoking, drinking, SBP), lipid profiles (LDL-C, HDL-C, VLDL-C, TG), angiographic factors (minimal stent diameter, average stent length), SYNTAX score, medical history, and other biomarkers (UA, CRP). The hazard ratio (HR) and 95% confidence intervals (95% CI) were calculated to estimate the adjusted risk of ISR in diabetic patients. The predictive value of the Cox's regression model was evaluated using the area under the receiver operating characteristics curve (AUC).
Statistical analyses were performed using SPSS software for Windows (version 20.0, SPSS Inc., Chicago, IL). Two tailed P < 0.05 was considered statistically significant in all analyses.
3. Results
A total of 368 patients (260 women and 108 men) with mean ages of 58.71 ± 10.25 years were enrolled in this study. Repeat angiography after baseline PCI was conducted with a mean time of 15.85 ± 9.18 months follow-up. Angiography result showed that ISR occurred in 74/368 diabetic patients (20.11%). According to whether ISR was detected, patients were divided into an ISR group (n = 74) and Non-ISR group (n = 294).
3.1. Baseline clinical characteristics
The baseline clinical characteristics were displayed in Table 1. No significant differences were found between the ISR and Non-ISR group in terms of age, gender, BMI, smoking, history of hypertension, myocardial infarction (MI), stroke, and the family history of CAD. Patients with ISR had lower drinking rates (2.7% vs. 17.0%, P = 0.026), whereas the percentage of patients with prior PCI was higher in the ISR group than that in the Non-ISR group (24.3% vs. 8.8%, P = 0.021). In addition, for the laboratory results, patients with ISR had increased serum VLDL-C and TG levels compared with those without ISR (0.64 vs. 0.51 mmol/L, P = 0.014; 1.83 vs. 1.69 mmol/L, P = 0.027), the levels of UA were also higher in ISR group than non-ISR group (364.67 vs. 317.66 mmol/L, P = 0.036). But the level of TC, LDL-C, HDL-C, GLU, CRP, total bilirubin and red cell distribution width (RDW) were similar between the two groups. The use of drugs was also showed no significant difference.
Table 1. Baseline clinical characteristics of study population.
| Characteristics | ISR (n = 74) | Non-ISR (n = 294) | P values |
| Age, yrs | 56.61 ± 10.37 | 59.37 ± 10.05 | 0.127 |
| Male | 56 (75.7%) | 204 (69.4%) | 0.453 |
| BMI, kg/m2 | 25.98 ± 3.26 | 26.72 ± 3.12 | 0.201 |
| SBP, mmHg | 128.14 ± 18.86 | 131.77 ± 18.59 | 0.291 |
| DBP, mmHg | 75.78 ± 11.48 | 78.12 ± 10.23 | 0.227 |
| Smoking | 26 (35.1%) | 128 (43.5%) | 0.354 |
| Drinking | 2 (2.7%) | 50 (17.0%) | 0.026 |
| Medical history | |||
| Hypertension | 48 (64.9%) | 192 (65.3%) | 0.960 |
| Hyperlipidemia | 40 (54.1%) | 152 (51.7%) | 0.798 |
| History of MI | 6 (8.1%) | 24 (8.2%) | 1.000 |
| History of stroke | 2 (2.7%) | 40 (13.6%) | 0.115 |
| Previous PCI | 18 (24.3%) | 26 (8.8%) | 0.021 |
| Previous CABG | 2 (2.7%) | 2 (0.7%) | 0.862 |
| Family history of CAD | 14 (18.9%) | 54 (18.4%) | 0.938 |
| Laboratory results | |||
| TG, mmol/L | 1.83 (1.28, 3.08) | 1.69 (1.17, 2.47) | 0.027 |
| TC, mmol/L | 4.55 ± 0.95 | 4.52 ± 1.14 | 0.885 |
| LDL-C, mmol/L | 2.71 ± 0.81 | 2.93 ± 1.06 | 0.233 |
| HDL-C, mmol/L | 0.91 ± 0.18 | 0.96 ± 0.25 | 0.285 |
| VLDL-C, mmol/L | 0.64 (0.35, 1.18) | 0.51 (0.26, 0.80) | 0.014 |
| FBG, mmol/L | 7.65 ± 2.38 | 7.65 ± 2.26 | 0.992 |
| HbA1c | 7.67% ± 1.47% | 7.29% ± 1.33% | 0.128 |
| CRP, mg/L | 2.31 (1.05, 3.72) | 2.34 (0.90, 4.25) | 0.933 |
| Creatinine, µmol/L | 73.35 ± 18.50 | 75.89 ± 18.15 | 0.449 |
| GFR, mL/min | 110.66 ± 32.25 | 102.17 ± 28.96 | 0.121 |
| UA, µmol/L | 364.67 ± 133.15 | 317.66 ± 124.17 | 0.036 |
| TB, µmol/L | 13.01 (8.38, 15.60) | 13.24 (9.31, 17.44) | 0.093 |
| DB, µmol/L | 3.46 (2.03, 4.63) | 3.67 (2.64,4.73) | 0.359 |
| RDW | 12.49% ± 0.77% | 12.49% ± 0.80% | 0.977 |
| Platelet count, ×109/L | 205.10 ± 57.17 | 203.20 ± 64.65 | 0.874 |
| LVEF | 61.30% ± 7.76% | 62.01% ± 89.12% | 0.664 |
| Medical treatment | |||
| Statins | 66 (89.2%) | 274 (93.2%) | 0.635 |
| Asprin | 74 (100%) | 288 (98.0%) | 0.881 |
| β-Blocker | 58 (78.4%) | 220 (74.8%) | 0.654 |
| Clopidogrel | 72 (97.3%) | 292 (99.3%) | 0.862 |
| Insulin | 18 (24.3%) | 66 (22.4%) | 0.808 |
| ACEI | 28 (37.8%) | 112 (38.1%) | 0.977 |
| ARB | 6 (8.1%) | 46 (15.8%) | 0.234 |
Continuous variables are expressed as mean ± SD in case of normal distribution or as median (interquartile ranges) in case of skewed distribution. Categorical variables are presented as n (%). ACEI: angiotensin converting enzyme inhibitor; AGB: angiotensin receptor blocker; BMI: body mass index; CABG: coronary artery bypass grafting; CAD: coronary artery disease; CRP: C-reactive protein; DB: direct bilirubin; DBP: diastolic blood pressure; FBG: fasting blood glucose; GFR: glomerular filtration rate; HDL-C: high density lipoprotein cholesterol; ISR: in-stent restenosis; LDL-C: low density lipoprotein cholesterol; LVEF: left ventricular ejection fraction; MI: myocardial infraction; PCI: Percutaneous coronary intervention; RDW: red cell distribution width; SBP: systolic blood pressure; TB: total bilirubin; TC: total cholesterol; TG: triglyceride; UA: uric acid; VLDL-C: very low density lipoprotein cholesterol.
3.2. Baseline angiographic characteristics
Baseline angiographic and procedural characteristics in 368 patients with 550 lesions were shown in Table 2. Patients in the ISR group had more numbers of target vessel lesions compared with non-ISR group (1.70 vs. 1.45, P = 0.021). The prevalence of multi-vessel disease (≥ 2 vessels) was significantly higher in the ISR group than in the non-ISR group (59.5% vs. 40.8%, P = 0.041). The SYNTAX score was also significantly higher in the ISR group than in the non-ISR group (15.0 vs. 11.0, P = 0.022). The other basic angiographic information like target vessels, minimal stent diameter and stent length showed no real difference between the two groups.
Table 2. Baseline angiographic characteristics of study population.
| Characteristics | ISR (n =74) | Non-ISR (n = 294) | P values |
| Number of target vessels | 1.70 ± 0.66 | 1.45 ± 0.58 | 0.021 |
| One | 30 (40.5%) | 174 (59.2%) | 0.041 |
| Two | 36 (48.6%) | 108 (36.7%) | 0.184 |
| Three | 8 (10.8%) | 12 (4.1%) | 0.227 |
| Multivessel disease | 44 (59.5%) | 120 (40.8%) | 0.041 |
| Target vessels | |||
| LM | 0 | 18 (6.1%) | 0.264 |
| LAD | 52 (70.3%) | 172 (58.5%) | 0.190 |
| LCX | 34 (45.9%) | 114 (38.8%) | 0.427 |
| RCA | 40 (54.1%) | 120 (40.8%) | 0.147 |
| SYNTAX score | 15.00 (9.00,18.50) | 11.00 (8.00,19.00) | 0.022 |
| Minimal stent diameter, mm | 2.81 ± 0.41 | 2.83 ± 0.45 | 0.849 |
| Stent length, mm | 22.41 ± 4.37 | 21.32 ± 5.62 | 0.272 |
Continuous variables are expressed as mean ± SD in case of normal distribution or as median (interquartile ranges) in case of skewed distribution. Categorical variables are presented as n (%). ISR: in-stent restenosis; LAD: left anterior descending; LCX: left circumflex artery; LM: left main; RCA: right coronary artery; SYNTAX: Synergy Between PCI With Taxus and Cardiac Surgery.
3.3. Predictors of ISR in diabetes
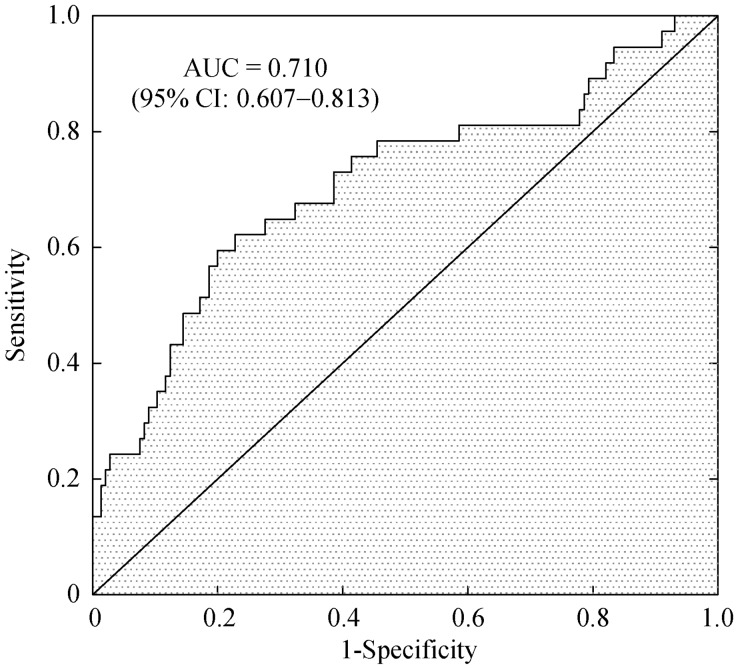
In the multivariate Cox's proportional hazards regression, after adjusting for traditional risk factors, lipid profiles, angiographic factors, medical history, and other biomarkers, the VLDL-C level, the UA level, SYNTAX score and previous PCI were identified as the independent predictors associated with ISR in diabetic patients (Figure 1). The HR for the occurrence of ISR associated with VLDL-C level was 1.85 (95% CI: 1.24–2.77, P = 0.002). For SYNTAX score (per 5 increments) and UA level (per 50 µmol/L increments), the HR was 1.34 (95% CI: 1.03–1.74, P = 0.031) and 1.19 (95% CI: 1.05–1.34, P = 0.006), respectively. A significantly higher proportion of patients had ISR in patients with the history of PCI (HR = 3.43, 95% CI: 1.57–7.80, P = 0.003). The ROC curve was used to measure the predictive values of the Cox's regression model, and the AUC was 0.710 (95% CI: 0.607–0.813, P < 0.001) which showed a good predictive accuracy for the occurrence of ISR in diabetic patients (Figure 2).
Firgue 1. Independent predictors of ISR in patients with diabetes mellitus after PCI by multivariate Cox's proportional hazards regression model.
ISR: in-stent restenosis; PCI: percutaneous coronary intervention; SYNTAX: Synergy between PCI with Taxus and Cardiac Surgery; VLDL-C: very low density lipoprotein cholesterol.
Figure 2. Receiver operating characteristics curve analysis for the prediction of the Cox's regression model.
AUC: area under the receiver operating characteristics curve.
4. Discussion
The results of this study showed that the occurrence rate of ISR reached up to 20% in diabetic patients after undergoing DES implantation. Patients with ISR had a significant increase of VLDL-C, UA levels and higher SYNTAX score compared with non-ISR patients. Multivariable analysis showed that even after adjustment for potential clinical variables and angiographic factors, the elevated baseline level of VLDL-C and UA, higher SYNTAX score and the history of previous PCI were identified to be independent predictors of ISR in patients with DM after coronary DES implantation.
Diabetes had been considered as a coronary disease equivalent condition. Patients with DM commonly had an increased risk of CAD with more severe disease phenotypes and had a poorer prognosis and higher mortality than non-diabetic patients.[24],[25] Increasing evidences suggested that DM was a major risk factor for ISR.[6],[7],[25],[26] A recent meta-analysis including 9578 total patients and 2667 DM patients showed that there was significant association between DM and ISR (OR = 1.70, 95% CI: 1.53–1.89).[7] As the clinical situation of CAD patients combined with DM was worse than ordinary CAD patients, it became significant to find reliable factors to predict the risk of ISR, especially in the subpopulation of DM patients.
A recent multi-center study showed that the prevalence of dyslipidemia had reached up to 67.1% in DM patients in China.[10] It is uniquely manifested by high level of TG and low level of HDL-C in DM patients, while the LDL-C level was not usually raised.[27] Among the various components of serum lipids, VLDL-C was synthesized and secreted by hepatic cell,[28] and it was the major lipoprotein of triglycerides-rich lipoprotein (TGRL), which was rich in 55% TG and 20% cholesterol.[29],[30] Additionally, current guidelines increasingly focused on reducing Non-HDL-C as the primary target of lipid-lowering therapy, which was mainly including LDL-C and VLDL-C.[16],[17] As a result, more and more researchers paid attention to the potential atherogenic effect of VLDL-C.[31] Basic medicine experiments have shown that VLDL could penetrate the arterial intima and be taken up by macrophages, and then eventually resulted in cholesterol accumulation, which would promote the occurrence and development of atherosclerosis.[32],[33] Clinical observation studies in humans also indicated that the elevated levels of VLDL-C could increase the risk of CHD and have been regarded as an independent risk factor of CHD.[34],[35] For diabetic patients after PCI, most previous studies showed that there was no significant difference between the ISR and Non-ISR group in terms of the levels of TC, LDL-C and HDL-C.[15] However, these studies always ignored the potential difference of VLDL-C level in diabetic patients. Similarly, no significant differences were observed between the ISR and non-ISR group in terms of the levels of TC, LDL-C and HDL-C in our study, but patients with ISR had higher VLDL-C and TG levels compared with those without ISR. Considering VLDL-C level was highly positive in correlation with TG, they were together entered into the multivariate regression model using the stepwise method after adjusting for traditional risk factors and other lipid profiles, and VLDL-C was identified as an independent predictor associated with ISR in diabetic patients but not TG (VLDL-C, HR = 1.85, 95% CI: 1.24–2.77, P = 0.002). This suggested that VLDL-C might be the most major lipid profiles in promoting the occurrence and development of ISR among diabetic patients who usually had high level of TG. The American Diabetes Association and the American College of Cardiology recommended that non-HDL-C target goals should be < 130 mg/dL for diabetic patients with overt CVD, as well as those without overt CVD.[36],[37] This is due to the fact that LDL-C was not usually elevated in diabetes, thus leaving VLDL-C the major therapeutic goal in the lipid management among patients with DM after PCI.
Hyperuricemia was not only critical to gout, but also found to be linked with diabetes and associated with metabolic syndrome.[38] The elevated baseline serum UA was in fact an independent risk factor in the development of insulin resistance and subsequent diabetes.[39] Previous study had found that high preprocedural UA level was a powerful and independent predictor of bare metal stent restenosis in patients with stable or unstable angina pectoris (OR = 1.07, 95% CI: 1.03–1.12, P = 0.003).[40] Similarly, our study indicated that the preprocedural UA level (per 50 µmol/L increments) was significantly associated with further ISR in diabetes who underwent successful DES implantation (HR = 1.19, 95% CI: 1.05–1.34, P = 0.006). Findings of experimental and clinical studies had proved the proinflammatory properties of UA, which was positively related with the level of TNF-α and IL-6 and it promoted the secretion of CRP in human vascular cells.[41] UA could also stimulate vascular smooth muscle cell (SMC) proliferation and the neointimal formation in the surface of the stent.[42],[43] Persistence of inflammatory stimuli and subsequent cellular proliferation within vulnerable plaque was considered to play an important role in the occurrence of ISR after PCI.[44] Based on existing evidences, it was speculated that the UA might increase the risk for ISR through increasing proinflammatory status and proliferation of vascular SMC. Therefore, the level of UA could serve as a meaningful marker in PCI patients especially in diabetics.
SYNTAX score was an angiographic scoring system to objectively grade the complexity of the coronary anatomy, which was initially introduced in 2005 and validated by a stage III clinical trial.[23] Subsequent reports had demonstrated that the SYNTAX score could be able to stratify patients with complex coronary diseases and had a prognostic value to predict the risk of short- and long-term major ischemic events in patients undergoing PCI.[45]–[47] Considering the diabetic patients had more sever coronary atherosclerosis with small and diffusely lesions,[8] our findings indicated that SYNTAX score (per 5 increments) was independently associated with ISR of target lesions (HR = 1.34, 95% CI: 1.03–1.74, P = 0.031) in patients with DM, suggesting that greater anatomic complexity of CAD conferred a higher risk of progression of atherosclerosis and ISR after PCI. Previous studies have found that the SYNTAX score was an independent predictor of target vessel revascularization (TVR) (OR = 1.03, 95% CI: 1.02–1.05, P < 0.0001)[47] and any revascularization (OR = 1.04, 95% CI: 1.03–1.05, P < 0.0001)[48] in patients treated with DES, even after correcting for clinical variables. Those present findings were consistent with a report indicating that lesion progression occurred more rapidly in diabetic patients with complex lesions than in diabetic patients with simple lesions.[49] In this way, as a surrogate marker of disease burden before PCI based on the location and characteristics of coronary lesions, SYNTAX score might provide new incremental value to assess the risk of ISR in diabetic patients.
Recent reports confirmed that the history of previous PCI was identified as an independent predictor of TVR (OR = 1.90, 95% CI: 1.49–2.41, P < 0.0001) and any revascularization (OR = 1.64, 95% CI: 1.28–2.11, P < 0.001),[48] suggesting that the preoperative factors affecting coronary atherosclerosis were still working in patients who underwent PCI. Our Study also demonstrated that the diabetic patients with a history of PCI had a higher risk of ISR after DES implantation (HR = 3.43, 95% CI: 1.57–7.80, P = 0.003), which was consistent with those of the PROSPECT (Providing Regional Observations to Study Predictors of Events in the Coronary Tree) study findings with respect to the impact of diabetes and previous PCI on any revascularization (HR = 2.03, 95% CI: 1.15–3.59, P = 0.02).[50]
Some limitations of the present study had to be acknowledged. First, the study was only a single-center study with a small sample size, which would have weakened the statistical power of the conclusions. Although the required number of observations had been a priori calculated by power analysis, additional large-scale prospective cohort studies at multiple centers were needed to confirm our results in additional patient cohorts before any clinical conclusions could be drawn. Second, in the present study, 92% patients had taken statin medication, which might have effect on the lipid profile levels, such as TC, LDL-C and HDL-C. However, to the best of our knowledge, this was the first report to demonstrate the level of VLDL-C was significantly associated with the risk ISR in diabetic patients even when other lipid profiles like TC, LDL-C were controlled by statin medication. This suggested that VLDL-C might play a profound role in the development of ISR and should be given more attention as a new target goal for lipid-lowering treatment among patients with DM. Third, duration of DM had been associated to poor clinical outcomes of CAD. The present study did not record patient-reported duration of DM, so it was not possible to investigate the influence of the DM duration on the development of ISR. Forth, our study only included patients who underwent the second angiography due to the progression of treated target lesions rather than untreated non-target lesions, which made our results more applicable for the specific subpopulation for better prevention of ISR.
In summary, our study provided evidence that the increased serum VLDL-C and UA level, higher SYNTAX score and the history of previous PCI were strong independent predictors of ISR in patients with DM after coronary DES implantation. Predicting the risk of ISR in diabetic patients after PCI allowed physicians to proactively intervene in ways that would lower the risk for the management of these patients, such as prescribing aggressive lipid-lowering and UA-lowering medication to reduce the circulating level of VLDL-C and UA. SYNTAX score could also be well-used to comprehensively evaluate the degree of coronary artery lesions and the risk to develop ISR in the subpopulation of DM patients.
Acknowledgments
This work were supported by the grant from National Key Research and Development Program of China (2017YFC0908800), the Beijing Municipal Administration of Hospitals Clinical Medicine Development of Special Funding Support (ZYLX201303), the National Key Clinical speciality Construction Project (2013–2014), and the “Beijing Municipal Administration of Hospitals” Ascent Plan (DFL20150601).
References
- 1.Liu Y, Xu C, Fu P, et al. Family history of myocardial infarction as a strong independent predictor of in-stent restenosis in the era of drug eluting stents. Int J Cardiol. 2015;179:55–57. doi: 10.1016/j.ijcard.2014.10.082. [DOI] [PubMed] [Google Scholar]
- 2.Demyanets S, Tentzeris I, Jarai R, et al. An increase of interleukin-33 serum levels after coronary stent implantation is associated with coronary in-stent restenosis. Cytokine. 2014;67:65–70. doi: 10.1016/j.cyto.2014.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dangas GD, Claessen BE, Caixeta A, et al. In-stent restenosis in the drug-eluting stent era. J Am Coll Cardiol. 2010;56:1897–1907. doi: 10.1016/j.jacc.2010.07.028. [DOI] [PubMed] [Google Scholar]
- 4.Jukema JW, Verschuren JJ, Ahmed TA, Quax PH. Restenosis after PCI. Part 1: pathophysiology and risk factors. Nat Rev Cardiol. 2012;9:53–62. doi: 10.1038/nrcardio.2011.132. [DOI] [PubMed] [Google Scholar]
- 5.Niccoli G, Montone RA, Ferrante G, Crea F. The evolving role of inflammatory biomarkers in risk assessment after stent implantation. J Am Coll Cardiol. 2010;56:1783–1793. doi: 10.1016/j.jacc.2010.06.045. [DOI] [PubMed] [Google Scholar]
- 6.Daemen J, Wenaweser P, Tsuchida K, et al. Early and late coronary stent thrombosis of sirolimus-eluting and paclitaxel-eluting stents in routine clinical practice: data from a large two-institutional cohort study. Lancet. 2007;369:667–678. doi: 10.1016/S0140-6736(07)60314-6. [DOI] [PubMed] [Google Scholar]
- 7.Qin SY, Zhou Y, Jiang HX, et al. The association of diabetes mellitus with clinical outcomes after coronary stenting: a meta-analysis. PLoS One. 2013;8:e72710. doi: 10.1371/journal.pone.0072710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Barry A, Wilkinson I, Halford V, et al. A clinical study and the national service framework for diabetes. J Tissue Viability. 2004;14 doi: 10.1016/s0965-206x(04)44002-9. [DOI] [PubMed] [Google Scholar]
- 9.Yonetsu T, Kato K, Kim SJ, et al. Predictors for neoatherosclerosis: a retrospective observational study from the optical coherence tomography registry. Circ Cardiovasc Imaging. 2012;5:660–666. doi: 10.1161/CIRCIMAGING.112.976167. [DOI] [PubMed] [Google Scholar]
- 10.Yan L, Xu MT, Yuan L, et al. Prevalence of dyslipidemia and its control in type 2 diabetes: A multicenter study in endocrinology clinics of China. J Clin Lipidol. 2016;10:150–160. doi: 10.1016/j.jacl.2015.10.009. [DOI] [PubMed] [Google Scholar]
- 11.Wihanda D, Alwi I, Yamin M, et al. Factors associated with in-stent restenosis in patients following percutaneous coronary intervention. Acta Med Indones. 2015;47:209–215. [PubMed] [Google Scholar]
- 12.Kang SJ, Cho YR, Park GM, et al. Predictors for functionally significant in-stent restenosis: an integrated analysis using coronary angiography, IVUS, and myocardial perfusion imaging. JACC Cardiovasc Imaging. 2013;6:1183–1190. doi: 10.1016/j.jcmg.2013.09.006. [DOI] [PubMed] [Google Scholar]
- 13.Park CB, Park HK. Predictors of diffuse-type in-stent restenosis following drug-eluting stent implantation. Exp Ther Med. 2013;5:1486–1490. doi: 10.3892/etm.2013.1024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yang W, Lu J, Weng J, et al. Prevalence of diabetes among men and women in China. N Engl J Med. 2010;362:1090–1101. doi: 10.1056/NEJMoa0908292. [DOI] [PubMed] [Google Scholar]
- 15.Zhao K, Li YJ, Gao S. Role of red blood cell distribution in predicting drug-eluting stent restenosis in patients with stable angina pectoris after coronary stenting. Coron Artery Dis. 2015;26:220–224. doi: 10.1097/MCA.0000000000000221. [DOI] [PubMed] [Google Scholar]
- 16.Jacobson TA, Ito MK, Maki KC, et al. National lipid association recommendations for patient-centered management of dyslipidemia: part 1--full report. J Clin Lipidol. 2015;9:129–169. doi: 10.1016/j.jacl.2015.02.003. [DOI] [PubMed] [Google Scholar]
- 17.An International Atherosclerosis Society Position Paper: global recommendations for the management of dyslipidemia--full report. J Clin Lipidol. 2014;8:29–60. doi: 10.1016/j.jacl.2013.12.005. [DOI] [PubMed] [Google Scholar]
- 18.Pan J, Lu Z, Zhang J, et al. Angiographic patterns of in-stent restenosis classified by computed tomography in patients with drug-eluting stents: correlation with invasive coronary angiography. Eur Radiol. 2013;23:101–107. doi: 10.1007/s00330-012-2559-2. [DOI] [PubMed] [Google Scholar]
- 19.Alfonso F, Byrne RA, Rivero F, Kastrati A. Current treatment of in-stent restenosis. J Am Coll Cardiol. 2014;63:2659–2673. doi: 10.1016/j.jacc.2014.02.545. [DOI] [PubMed] [Google Scholar]
- 20.Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care. 2003;26(Suppl 1):S5–S20. doi: 10.2337/diacare.26.2007.s5. [DOI] [PubMed] [Google Scholar]
- 21.Mansia G, De Backer G, Dominiczak A, et al. 2007 ESH-ESC Guidelines for the management of arterial hypertension: the task force for the management of arterial hypertension of the European Society of Hypertension (ESH) and of the European Society of Cardiology (ESC) Blood Press. 2007;16:135–232. doi: 10.1080/08037050701461084. [DOI] [PubMed] [Google Scholar]
- 22.Grundy SM, Cleeman JI, Merz CN, et al. Implications of recent clinical trials for the National Cholesterol Education Program Adult Treatment Panel III Guidelines. J Am Coll Cardiol. 2004;44:720–732. doi: 10.1016/j.jacc.2004.07.001. [DOI] [PubMed] [Google Scholar]
- 23.Sianos G, Morel MA, Kappetein AP, et al. The SYNTAX Score: an angiographic tool grading the complexity of coronary artery disease. EuroIntervention. 2005;1:219–227. [PubMed] [Google Scholar]
- 24.Groot MW, Head SJ, Bogers AJ, Kappetein AP. Coronary revascularization in diabetic patients. A focus on the 3-year SYNTAX trial outcomes. Herz. 2012;37:281–286. doi: 10.1007/s00059-012-3602-4. [DOI] [PubMed] [Google Scholar]
- 25.Carson JL, Scholz PM, Chen AY, et al. Diabetes mellitus increases short-term mortality and morbidity in patients undergoing coronary artery bypass graft surgery. J Am Coll Cardiol. 2002;40:418–423. doi: 10.1016/s0735-1097(02)01969-1. [DOI] [PubMed] [Google Scholar]
- 26.Ma S, Yang D, Zhang X, et al. Comparison of restenosis rate with sirolimus-eluting stent in STEMI patients with and without diabetes at 6-month angiographic follow-up. Acta Cardiol. 2011;66:603–606. doi: 10.1080/ac.66.5.2131085. [DOI] [PubMed] [Google Scholar]
- 27.Kumar A, Singh V. Atherogenic dyslipidemia and diabetes mellitus: what's new in the management arena. Vasc Health Risk Manag. 2010;6:665–669. doi: 10.2147/vhrm.s5686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Shelness GS, Sellers JA. Very-low-density lipoprotein assembly and secretion. Curr Opin Lipidol. 2001;12:151–157. doi: 10.1097/00041433-200104000-00008. [DOI] [PubMed] [Google Scholar]
- 29.Rosenson RS, Davidson MH, Hirsh BJ, et al. Genetics and causality of triglyceride-rich lipoproteins in atherosclerotic cardiovascular disease. J Am Coll Cardiol. 2014;64:2525–2540. doi: 10.1016/j.jacc.2014.09.042. [DOI] [PubMed] [Google Scholar]
- 30.Nielsen S, Karpe F. Determinants of VLDL-triglycerides production. Curr Opin Lipidol. 2012;23:321–326. doi: 10.1097/MOL.0b013e3283544956. [DOI] [PubMed] [Google Scholar]
- 31.Khetarpal SA, Rader DJ. Triglyceride-rich lipoproteins and coronary artery disease risk: new insights from human genetics. Arterioscler Thromb Vasc Biol. 2015;35:e3–e9. doi: 10.1161/ATVBAHA.114.305172. [DOI] [PubMed] [Google Scholar]
- 32.Nordestgaard BG, Wootton R, Lewis B. Selective retention of VLDL, IDL, and LDL in the arterial intima of genetically hyperlipidemic rabbits in vivo. Molecular size as a determinant of fractional loss from the intima-inner media. Arterioscler Thromb Vasc Biol. 1995;15:534–542. doi: 10.1161/01.atv.15.4.534. [DOI] [PubMed] [Google Scholar]
- 33.Rapp JH, Lespine A, Hamilton RL, et al. Triglyceride-rich lipoproteins isolated by selected-affinity anti-apolipoprotein B immunosorption from human atherosclerotic plaque. Arterioscler Thromb. 1994;14:1767–1774. doi: 10.1161/01.atv.14.11.1767. [DOI] [PubMed] [Google Scholar]
- 34.Ren J, Grundy SM, Liu J, et al. Long-term coronary heart disease risk associated with very-low-density lipoprotein cholesterol in Chinese: the results of a 15-Year Chinese Multi-Provincial Cohort Study (CMCS) Atherosclerosis. 2010;211:327–332. doi: 10.1016/j.atherosclerosis.2010.02.020. [DOI] [PubMed] [Google Scholar]
- 35.Varbo A, Benn M, Nordestgaard BG. Remnant cholesterol as a cause of ischemic heart disease: evidence, definition, measurement, atherogenicity, high risk patients, and present and future treatment. Pharmacol Ther. 2014;141:358–367. doi: 10.1016/j.pharmthera.2013.11.008. [DOI] [PubMed] [Google Scholar]
- 36.Brunzell JD, Davidson M, Furberg CD, et al. Lipoprotein management in patients with cardiometabolic risk: consensus statement from the American Diabetes Association and the American College of Cardiology Foundation. Diabetes Care. 2008;31:811–822. doi: 10.2337/dc08-9018. [DOI] [PubMed] [Google Scholar]
- 37.Boronat M, García-Cantón C, López-Ríos L, et al. Potential implications of the choice among three alternative treatment targets for apolipoprotein B100 in the management of patients with type 2 diabetes and chronic kidney disease. Diab Vasc Dis Res. 2014;11:53–59. doi: 10.1177/1479164113511834. [DOI] [PubMed] [Google Scholar]
- 38.Sluijs I, Beulens JW, van der A DL, et al. Plasma uric acid is associated with increased risk of type 2 diabetes independent of diet and metabolic risk factors. J Nutr. 2013;143:80–85. doi: 10.3945/jn.112.167221. [DOI] [PubMed] [Google Scholar]
- 39.Krishnan E, Pandya BJ, Chung L, et al. Hyperuricemia in young adults and risk of insulin resistance, prediabetes, and diabetes: a 15-year follow-up study. Am J Epidemiol. 2012;176:108–116. doi: 10.1093/aje/kws002. [DOI] [PubMed] [Google Scholar]
- 40.Turak O, Canpolat U, Özcan F, et al. Usefulness of preprocedural serum uric acid level to predict restenosis of bare metal stents. Am J Cardiol. 2014;113:197–202. doi: 10.1016/j.amjcard.2013.09.004. [DOI] [PubMed] [Google Scholar]
- 41.Kang DH, Park SK, Lee IK, Johnson RJ. Uric acid-induced C-reactive protein expression: implication on cell proliferation and nitric oxide production of human vascular cells. J Am Soc Nephrol. 2005;16:3553–3562. doi: 10.1681/ASN.2005050572. [DOI] [PubMed] [Google Scholar]
- 42.Kaya EB, Yorgun H, Canpolat U, et al. Serum uric acid levels predict the severity and morphology of coronary atherosclerosis detected by multidetector computed tomography. Atherosclerosis. 2010;213:178–183. doi: 10.1016/j.atherosclerosis.2010.08.077. [DOI] [PubMed] [Google Scholar]
- 43.Kanellis J, Watanabe S, Li JH, et al. Uric acid stimulates monocyte chemoattractant protein-1 production in vascular smooth muscle cells via mitogen-activated protein kinase and cyclooxygenase-2. Hypertension. 2003;41:1287–1293. doi: 10.1161/01.HYP.0000072820.07472.3B. [DOI] [PubMed] [Google Scholar]
- 44.Misaki K, Uchiyama N, Mohri M, Hayashi Y, Ueda F, Nakada M. Prediction of carotid artery in-stent restenosis by quantitative assessment of vulnerable plaque using computed tomography. J Neuroradiol. 2016;43:18–24. doi: 10.1016/j.neurad.2015.09.002. [DOI] [PubMed] [Google Scholar]
- 45.Magro M, Nauta S, Simsek C, et al. Value of the SYNTAX score in patients treated by primary percutaneous coronary intervention for acute ST-elevation myocardial infarction: The MI SYNTAXscore study. Am Heart J. 2011;161:771–781. doi: 10.1016/j.ahj.2011.01.004. [DOI] [PubMed] [Google Scholar]
- 46.Palmerini T, Genereux P, Caixeta A, et al. Prognostic value of the SYNTAX score in patients with acute coronary syndromes undergoing percutaneous coronary intervention: analysis from the ACUITY (Acute Catheterization and Urgent Intervention Triage StrategY) trial. J Am Coll Cardiol. 2011;57:2389–2397. doi: 10.1016/j.jacc.2011.02.032. [DOI] [PubMed] [Google Scholar]
- 47.Yang CH, Hsieh MJ, Chen CC, et al. The prognostic significance of SYNTAX score after early percutaneous transluminal coronary angioplasty for acute ST elevation myocardial infarction. Heart Lung Circ. 2013;22:341–345. doi: 10.1016/j.hlc.2012.12.003. [DOI] [PubMed] [Google Scholar]
- 48.Taniwaki M, Stefanini GG, Silber S, et al. Four-year clinical outcomes and predictors of repeat revascularization in patients treated with new-generation drug-eluting stents: a report from the RESOLUTE All-Comers trial (A Randomized Comparison of a Zotarolimus-Eluting Stent With an Everolimus-Eluting Stent for Percutaneous Coronary Intervention) J Am Coll Cardiol. 2014;63:1617–1625. doi: 10.1016/j.jacc.2013.12.036. [DOI] [PubMed] [Google Scholar]
- 49.Kedhi E, Généreux P, Palmerini T, et al. Impact of coronary lesion complexity on drug-eluting stent outcomes in patients with and without diabetes mellitus: analysis from 18 pooled randomized trials. J Am Coll Cardiol. 2014;63:2111–2118. doi: 10.1016/j.jacc.2014.01.064. [DOI] [PubMed] [Google Scholar]
- 50.Stone GW, Maehara A, Lansky AJ, et al. A prospective natu-ral-history study of coronary atherosclerosis. N Engl J Med. 2011;364:226–235. doi: 10.1056/NEJMoa1002358. [DOI] [PubMed] [Google Scholar]