Abstract
Introduction:
Identification of Extended spectrum beta lactamases (ESBL), AmpC production and carbapenemase production among isolates of Escherichia coli, helps clinician to rationalize the choice of antibiotics. However, there is a lack of simple and effective method for simultaneous identification of these beta lactamases.
Aim:
To determine the concurrent production of beta lactamases using twelve disc method on E. coli isolates.
Materials and Methods:
A total of 200 multidrug resistant E. coli were screened using twelve disc method. The isolates of ESBL were confirmed by ceftazidime/clavulanic acid and cefotaxime/clavulanic acid method. Metallo-beta-lactamases (MBL) were confirmed by imipenem EDTA combined disc method.
Results:
Among the 200 isolates, 42.5% were ESBL producers, 9% were MBL and 6.5% were Klebsiella pneumoniae carbapenemase (KPC) and AmpC each respectively. Coproduction was seen in 54 (27%). A significant difference in sensitivity was seen in cefuroxime, aztreonam, cefoxitin and ceftriaxone among inpatient and outpatients.
Conclusion:
The present study highlights burden of ESBL, AmpC, KPC and MBL along with their coproduction in a tertiary care hospital. In-house antibiotic policy, infection control and epidemiological surveys will help us in controlling these resistant bugs. We believe, the twelve disc method is a simple, inexpensive screening method for beta lactamase production.
Key words: AmpC, carbapenemases, extended spectrum beta lactamases, twelve disc method
INTRODUCTION
Emergence of multidrug resistant (MDR) organisms are a global threat. Extended spectrum beta lactamases (ESBL) and AmpC production among Escherichia coli often leads to failure of beta lactam therapy. Carbapenems are the treatment options for ESBL and AmpC producers. Resistance to carbapenems by production of carbapenemases leaves us with potentially toxic drugs, like polymyxin and colistin.[1] Co-production of multiple beta lactamases in a single isolate limits the treatment options further.[2] Hence the present study was designed to determine the concurrent production of beta-lactamases using a simple twelve disc method on E. coli isolates.
MATERIALS AND METHODS
This cross-sectional study was conducted in tertiary care teaching hospital. Data was collected from February 2014 to January 2015, for a period of 1 year.
A total of 200 consecutive, clinical isolates of E. coli which were resistant to third generation cephalosporin or multidrug resistant isolates from various clinical samples (urine, blood, pus, stool, sputum, body fluids, throat swab, high vaginal swabs and cerebrospinal fluid.) were included in the study. The isolates were processed by twelve disc method.[3]
Twelve disc method
On a 150 mm Mueller Hinton agar petridishes on a lawn culture of the isolate, 12 antibiotic disc was placed. The antibiotics were aztreonam (30 μg), ceftazidime (30 μg), ceftazidime/clavulanic acid (30/10 μg), cefotaxime (30 μg), cefotaxime/clavulanate (30/10 μg), cefoxitin (30 μg), cefotetan (30 μg), ceftriaxone (30 μg), cefepime (30 μg), ertapenem (10 μg), imipenem (10 μg) and meropenem (10 μg), in the specific sequence. Interpretation was as follows:
ESBL production was considered positive if zone diameter of inhibition around disc of ceftazidime clavulanic acid and cefotaxime clavulanic acid ≥5 mm than the ceftazidime or cefotaxime disc alone.
AmpC production was suggested when the isolate was resistant to cefoxitin (Zone diameter ≤14 mm) but susceptible to cefepime (Zone diameter ≥25 mm)
MBL production was suggested if the strain was resistant to all carbapenems (Zone diameter of imipenem ≤19 mm, meropenem ≤19 mm and ertapenem ≤18 mm). This was confirmed by Modified Hodge test.[4]
Klebsiella pneumoniae carbapenemase (KPC) was suggested if strain was imipenem sensitive (Zone diameter ≥23 mm) and ertapenem resistant (Zone diameter ≤18 mm). This was confirmed by Modified Hodge test[4] [Figure 1].
Figure 1.
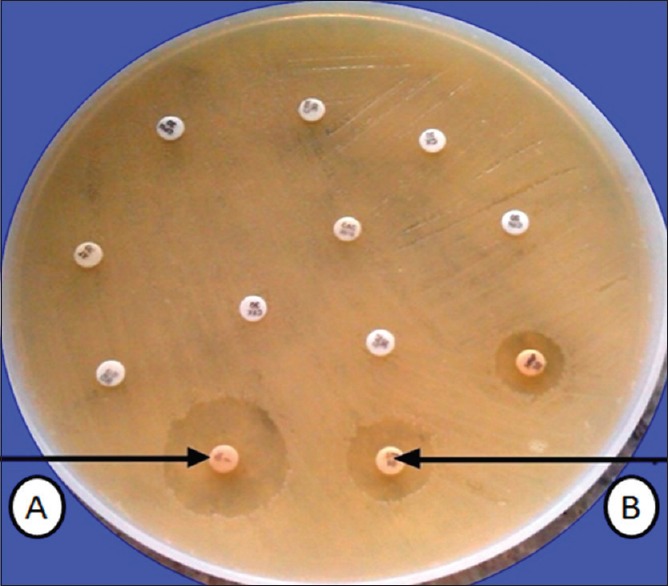
Twelve disc method showing Klebsiella pneumoniae carbapenemase beta lactamase production. (A) indicates imipenem sensitive and (B) indicates ertapenem resistance
All the isolates suggesting MBL production were further tested for inhibition by EDTA using the Imipenem EDTA combined disc test as per Yong et al.[5]
Statistical analysis
All the data was entered in Microsoft excel and analyzed using SPSS 15.0 Version (SPSS Inc. Chicago IL, United States of America). All categorical variables were expressed as number and proportion. Categorical variables was compared between the two groups using Chi-square test. P < 0.05 was considered significant.
RESULTS
The study involved a total of 200 consecutive, multidrug resistant isolates of E. coli from various clinical samples over a period of 1 year. Among the 200 isolates, 138 (69%) were from male patients with the male to female ratio of 2.2:1. Almost 60% of the patients were in the age group of 21–40 years. Infection were detected in urine samples (64%), followed by blood (15%), stool (7%), pus (5%), body fluids (5%) and high vaginal swab (3%) in the present study. A total of 95 (47.5%) isolates were from outpatient, 77 (38.5%) from inpatient and 28 (14%) from intensive unit cases.
Susceptibility pattern of the isolates
The susceptibility pattern of the isolates and among inpatients and outpatients is mentioned in Table 1. Among the E. coli isolates high sensitivity was found for carbapenems (ertapenem 85%, imipenem 78% and meropenem 71.5%). Sixty one and fifty six percentage of isolates were sensitive for ofloxacin and levofloxacin respectively. Sensitivity to cefuroxime and aztreonam was more significantly associated with outpatient than inpatients (P < 0.05). Sensitivity to cefoxitin and ceftriaxone was more among inpatients than outpatients and was found to be statistically significant.
Table 1.
Antimicrobial susceptibility pattern of the isolates among inpatients and outpatients
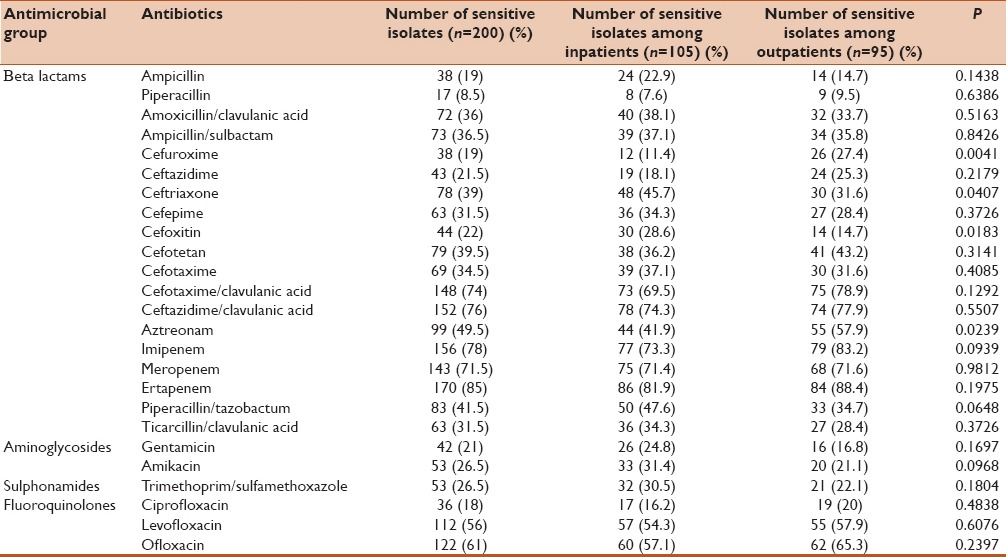
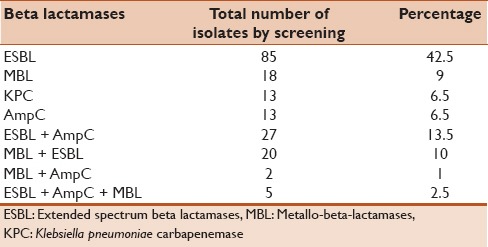
Table 2 shows the coproduction of various beta lactamases. Out of the 200 E. coli 42.5% showed confirmed ESBL production. Among the 13 KPC screened by 12 disc method only 5 isolates were confirmed KPCs by the Modified Hodge test. Thirteen isolates of the MBL screened, were confirmed as MBL producer using imipenem EDTA combined disc test.
Table 2.
Coproduction of various beta lactamases
DISCUSSION
The present study found that 42.5% were ESBL producers, 9% were MBL and 6.5% were KPC and AmpC each respectively among the strains of multidrug resistant E. coli. The study also highlights that coproduction of beta-lactamase was observed in 54 (27%). A significant difference in sensitivity was seen in cefuroxime, aztreonam, cefoxitin and ceftriaxone among inpatient and outpatients. This emerging trend of multidrug resistant E. coli among clinical samples raises a major concern for the clinician.
E. coli, a member of family enterobacteriaceae, is most common cause of nosocomial infection.[6] Beta lactam antibiotics are considered efficacious with broad spectrum coverage and minimal side effect for treatment of bacterial infection.[7,8] Antibiotic resistance is a major problem. Risk factors for antibiotic resistance are long term and inappropriate use of antibiotics, severe illness, comorbidities, long term hospital stay, poor sanitation and instrumentation or catheterization.[1,9] Infection caused by these MDR organisms have a heterogenous expression leading to long term hospital stay and high mortality.[10]
ESBL, AmpC and carbapenemases like MBL and KPC leaves us with limited treatment options. Further, MBL just like ESBL and AmpC, can be transferred between species by plasmids.[6] Genes encoding for MBL are often present on class 1 integrons. Antibiotics like aminoglycosides and fluoroquinolones also have their gene cassette in the integrons, thus leading to cross resistance. Transposons, highly transmissible genetic element responsible for drug resistance have integrons embedded within them leading to transfer of resistance.[11] Hence, timely detection prevents the spread of infection.
In the current study resistance to ampicillin, ampicillin/sulbactum, meropenem and ofloxacin was 81%, 63.5%, 28.5% and 39%. The resistance pattern was comparable to study by Ansari et al., who had shown a rate of 74%, 57%, 37% and 47% respectively.[12] Resistance to levofloxacin, cefuroxime and ceftazidime was 35.9%, 82% and 65.4% in a study by Nisha et al., which are comparable to the present study.[13] Cotrimoxazole resistance of 73.5% in present study is comparable with 84% in study conducted by Sharma et al.[14]
Table 3 compares the percentage of ESBL, AmpC, MBL and KPC production among the various studies.
Table 3.
Comparison of various studies for beta lactamase production and coproduction
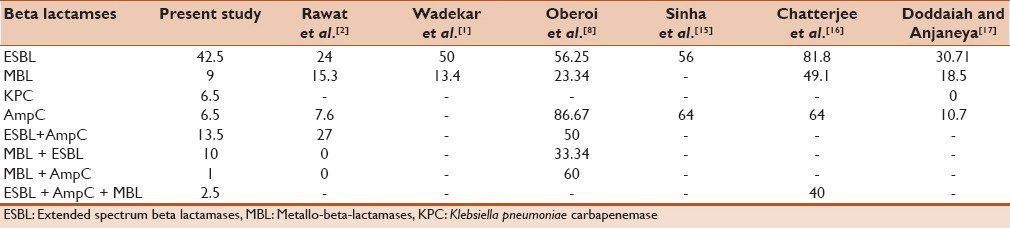
The first ESBL was disovered as early as 1980.[7] In India rate of ESBL production is 66%, which is comparable to studies in Turkey which shows 54.7%–61%, 41% in United Arab Emirates, 31.7% in Kuwait and 72.1% in Iran.[9] In the present study we have done ESBL detection using both ceftazidime as well as cefotaxime. it is known that ceftazidime is best indicator of TEM and SHV and cefotaxime is a good indicator of CTX-M type.[7] The present study had a ESBL detection rate of 42.5%. The yield of ESBL varies from as low as 24% to as high as 81%. This could be as a result of varied geographical region and method used for detection.[1,2,8,15,16,17]
AmpC Betalactamases are plasmid mediated beta lactamases that hydrolyse all cephalosporins except cefepime and the carbapenems. In AmpC, the inducible chromosomal genes becomes mobilized as plamids.[18] In the present study the rate of AmpC detection was 6.5%. The yield of AmpC in our hospital setting is less compared to other studies in India.[1,2,8,15,16,17]
Carbepenems are the main stay of treatment of isolates resistant to penicillin and cephalosporins. Carbapenem resistance due to carbapenamase production was first discovered in the year 1988.[19] This leads to very few treatment options. In the present study the rate of MBL detection was 9% and this is in concordant with studies done by Rawat et al. and Wadekar et al. and discordant with studies done by Oberoi et al. and Chatterjee et al.[1,2,8,16] KPC production in the present study was 6.5%. As carbapenemase production in the present study is low compared to other studies, we still have to adhere to the concept of reserve drugs and minimize the misuse of available antimicrobial to preserve it for future generations.
Coproduction in the present study was 54 (27%). This is low when compared to other studies.[2,8,15,16,17] The 12 disc method is a simple, easy, inexpensive, single plate method for the screening of various beta lactamases. However the disadvantage of this method is it can be used only for screening purpose and the results need to be confirmed with a confirmatory test. Molecular methods for detection of beta lactamases are gold standard, but due to cost constrains it could not be done in the present study, which could be a limitation of the study.
CONCLUSION
The present study highlights burden of ESBL, MBL, KPC, AmpC and coproduction of these carbapenamase among multidrug resistant isolates of E. coli in a North Indian tertiary care centre. We believe 12 disc method is simple and effective mean for rapid and simultaneous identification of carbapenamase production among E. coli culture isolates. An integrated system of action of clinicians and microbiologist in deciding the antibiotic treatment, maintaining proper sanitation, antimicrobial policy and epidemiological surveys will help in controlling and preventing the spread of these resistant bugs in the hospital environment.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Wadekar MD, Anuradha K, Venkatesha D. Phenotypic detection of ESBL and MBL in clinical isolates of Enterobacteriaceae. Int J Curr Res Acad Rev. 2013;1:89–5. [Google Scholar]
- 2.Rawat V, Singhai M, Verma PK. Detection of different β-lactamases and their co-existence by using various discs combination methods in clinical isolates of Enterobacteriaceae and Pseudomonas spp. J Lab Physicians. 2013;5:21–5. doi: 10.4103/0974-2727.115918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Schreckenberger P, Rekasius V. Detecting Resistance to Beta Lactams in Gram-Negative Bacilli. [Last accessed on 2018 Feb 17]. Available from: http://www.hardydiagnostics.com/articles/antibiotic-resistance.pdf .
- 4.CLSI. CLSI Document M100-S25. 25th ed. Wayne, Pennysylvani: Clinical and Laboratory Standards Institute; 2015. Performance standards for antimicrobial susceptibility test. Approved Standard. [Google Scholar]
- 5.Yong D, Lee K, Yum JH, Shin HB, Rossolini GM, Chong Y, et al. Imipenem-EDTA disk method for differentiation of metallo-beta-lactamase-producing clinical isolates of Pseudomonas spp. and Acinetobacter spp. J Clin Microbiol. 2002;40:3798–801. doi: 10.1128/JCM.40.10.3798-3801.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Khajuria A, Praharaj AK, Kumar M, Grover N. Emergence of Escherichia coli, co-producing NDM-1 and OXA-48 carbapenemases, in urinary isolates, at a tertiary care centre at central India. J Clin Diagn Res. 2014;8:DC01–4. doi: 10.7860/JCDR/2014/7952.4413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Puri JS, Kulkarni S, Jaywant A, Khare AS. Prevalence of extended spectrum β-lactamases in E. coli and Klebsiella spp. in a tertiary care hospital. Int J Curr Microbiol Appl Sci. 2014;3:474–8. [Google Scholar]
- 8.Oberoi L, Singh N, Sharma P, Aggarwal A. ESBL, MBL and AmpC β lactamases producing superbugs – Havoc in the Intensive care Units of Punjab India. J Clin Diagn Res. 2013;7:70–3. doi: 10.7860/JCDR/2012/5016.2673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sujatha R, Kumar A, Mishra V. A study by double disc diffusion (DDDT) method to compare ceftazidime + clavulanic acid and cefotaxime + clavulanic acid for the detection of extended spectrum β-lactamases among Escherichia coli and Klebsiella pneumoniae in urinary isolates. Int J Curr Microbiol Appl Sci. 2017;6:411–5. [Google Scholar]
- 10.Upadhyay S, Sen MR, Bhattacharjee A. Identification and characterization of carbapenem hydrolyzing β lactamases-KPC among Enterobacteriaceae: A report from North India. Asian J Med Sci. 2012;3:11–5. [Google Scholar]
- 11.Deshmukh DG, Damle AS, Bajaj JK, Bhakre JB, Patwardhan NS. Metallo-β-lactamase-producing clinical isolates from patients of a tertiary care hospital. J Lab Physicians. 2011;3:93–7. doi: 10.4103/0974-2727.86841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ansari S, Nepal HP, Gautam R, Shrestha S, Neopane P, Gurung G, et al. Community acquired multi-drug resistant clinical isolates of Escherichia coli in a tertiary care center of Nepal. Antimicrob Resist Infect Control. 2015;4:15. doi: 10.1186/s13756-015-0059-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nisha KV, Veena SA, Rathika SD, Vijaya SM, Avinash SK. Antimicrobial susceptibility, risk factors and prevalence of bla cefotaximase, temoneira, and sulfhydryl variable genes among Escherichia coli in community-acquired pediatric urinary tract infection. J Lab Physicians. 2017;9:156–62. doi: 10.4103/0974-2727.208262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Sharma S, Kaur N, Malhotra S, Madan P, Ahmad W, Hans C, et al. Serotyping and antimicrobial susceptibility pattern of Escherichia coli isolates from urinary tract infections in pediatric population in a tertiary care hospital. J Pathog 2016. 2016:2548517. doi: 10.1155/2016/2548517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sinha P, Goyal P, Sharma R, Vyas A, Maheshwari RK. Evaluation of a 12 disc test for phenotypic detection of β-lactamases resistance in gram negative Bacilli. Int J Curr Microbiol Appl Sci. 2016;5:105–14. [Google Scholar]
- 16.Chatterjee SS, Karmacharya R, Madhup SK, Gautam V, Das A, Ray P, et al. High prevalence of co-expression of newer beta-lactamases (ESBLs, AmpC β lactamases, and metallo-beta-lactamases) in gram-negative Bacilli. Indian J Med Microbiol. 2010;28:267–8. doi: 10.4103/0255-0857.66479. [DOI] [PubMed] [Google Scholar]
- 17.Doddaiah V, Anjaneya D. Prevalence of ESBL, Amp C and carbapenemase among gram negative Bacilli isolated from clinical specimen. Am J Life Sci. 2014;2:76–81. [Google Scholar]
- 18.Manoharan A, Sugumar M, Kumar A, Jose H, Mathai D, Khilnani GC, et al. Phenotypic and molecular characterization of AmpC β lactamases among Escherichia coli, Klebsiella. spp and Enterobacter spp. From five Indian medical centers. Indian J Med Res. 2012;135:359–64. [PMC free article] [PubMed] [Google Scholar]
- 19.Zavascki AP, Barth AL, Gonçalves AL, Moro AL, Fernandes JF, Martins AF, et al. The influence of metallo-beta-lactamase production on mortality in nosocomial Pseudomonas aeruginosa infections. J Antimicrob Chemother. 2006;58:387–92. doi: 10.1093/jac/dkl239. [DOI] [PubMed] [Google Scholar]


