What is 4P Medicine?
The current era of medicine is often named “4P Medicine”. Four P represents the acronym of Personalized, Predictive, Preventive and Participative (Fig. 1). In 2011 Hood and Friend [1] developed the concept of 4P Medicine, comprising not only the idea of a personalized medicine based on a well-defined molecular predictive medicine, but also a health concept in which disease prevention and citizen articipation in the management of health should become the medicine of the future, following a systems medicine approach. The backbone of 4P medicine is translational research aiming to define molecular pathways that are selectively involved in each type of cancer, allowing for targeted therapy on a personalized basis.
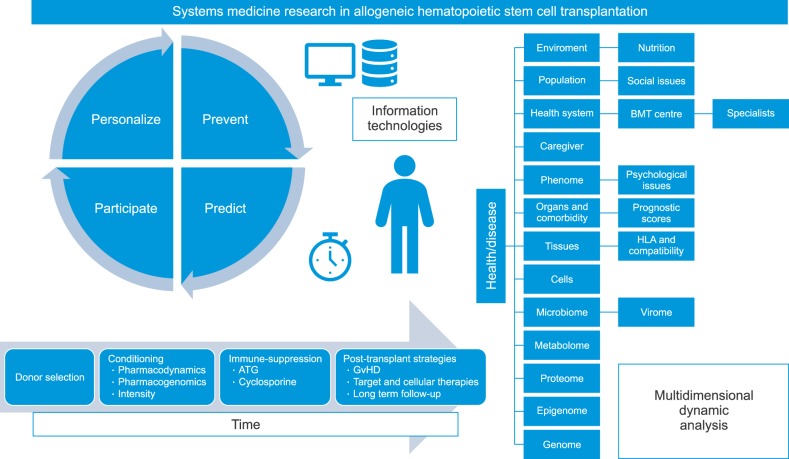
Fig. 1. Systems medicine research studied health and diseases through the investigation of several layers of knowledge (biological, psychological, social and environmental, the so called “Omics”) and could be interpreted as an informational science in which the interpretation of a multilayer analysis could analyze the dynamic of the pathological process over time. New technologies for large-scale data analysis and a definition of a multidimensional minimal data set for everyone are under investigation to override a simple mechanistic interpretation. For these reasons “Omics” are part of the dynamic study of allogeneic transplanted patients and should be integrated with the interpretation of the connections between individual and family, society, environment and health system. This process is crucial for the personalization of the transplanted patient's care. The interaction of the several inter- and intra-individual layers over time will contribute to a new sets of complex phenotypes typical for the transplanted patient. In a few years, billions of data concerning complex phenotypes will be available for everyone. Personalization, prevention, prediction and participation are thus cornerstones of the care for such complex patients and Information Technologies will be part of this research process.
The “magic bullet”
Systems medicine developed after the first studies on molecular pathways involved in carcinogenesis defined possible targets to switch off tumor growth with the aim of generating a “magic bullet” for each type of cancer [2]. In fact, the most relevant model of neoplastic disease susceptible to a true “targeted therapy” is represented by the Philadelphia-positive chronic myeloid leukemia (CML), which, between 1999 and 2000, started to be treated with tyrosine-kinase inhibitors, namely imatinib [3].
Ph+ CML is a type of leukemia driven by a single, specific, chromosome translocation (t9;22) leading to a new, hybrid, leukemia-specific gene (BCR-ABL1) that encodes for a deregulated tyrosine-kinase protein (p210) driving the leukemic transformation of hematopoietic stem cells and the progression from the chronic phase (CP) to the accelerated-blastic phase (ABP), which fatally concludes the course of the disease.
The introduction of Imatinib significantly changed the fate of CML patients, transforming the disease from fatal to chronic [4]. Due to these exciting results, from 2000 onwards the number of CML patients who have undergone allogeneic hematopoietic stem cell transplantation (allo-HSCT) has decreased dramatically and within a few years, CML has gone from being the first to being the last indication of allo-HSCT [5].
The “death” of allogeneic HSCT?
Looking at the results of imatinib, many hematologists believe that the advent of “targeted” therapy would have brought about the “death” of allo-HSCT for the treatment of many hematological diseases, including CML. Almost 20 years later, however, we can say that this prediction did not come true. The reasons for this are mainly two: firstly, the progress in the knowledge of the pathways of leukemogenesis did not really translate into decisive advances in the outcome of leukemias and cancers, except for CML.
On the other hand, a “rough” therapy such as allo-HSCT was confirmed to be the most powerful treatment for high-risk hematological malignancies, representing one of the best answer to patients' need of cure. Analyzing the causes of the partial disappointment on the results of targeted therapies and growing satisfaction with allo-HSCT results, we want also to point out that attention needs to be paid to the relevance of psychological and social aspects related to these approaches, and that they should be considered in the era of 4P medicine.
What is the role of allogeneic HSCT in the era of targeted therapies and 4P medicine?
Allo-HSCT currently represents the most powerful treatment for hematologic malignancies. It can be used to cure patients with high-risk disease in complete remission at the time of transplant (e.g. acute myeloid leukemia). Moreover, it is the most curative option for refractory diseases, despite its highly reduced effectiveness in comparison to diseases in complete remission at the time of transplant.
In fact, the numbers of allo-HSCT continue to increase over time, reaching half a million procedures in 2013. About 32,000 allo-HSCT were performed worldwide in 2012 [6], in comparison to 21,000 in 2008 [7]. These procedures increase over time not only in the industrialized world, but also in developing countries, for various reasons.
In industrialized countries, the increase of allo-HSCT is related to the expansion of indications due to better selection of the donors and better supportive care, allowing for transplantations up to advanced ages and phases of disease; on the other hand, the increment in transplants in developing countries is related to the reduction of allo-HSCT costs, in comparison to more expensive targeted therapies.
A second reason is the great improvement in the transplant procedure. Improvements in the conditioning regimens (more active but less toxic) and in supportive and anti-infective therapies have brought about a significant reduction in transplant-related mortality (TRM) and in the incidence of graft failure. In addition, the introduction of high-resolution HLA typing and the amelioration of prophylaxis and treatment of GvHD have contributed to the reduction of the incidence and severity of GvHD, which remains one of the most important causes of morbidity and mortality. These facts have really contributed to making transplantation a safer procedure and to spread the use of alternative sources of hematopoietic stem cells, including haploidentical and cord blood stem cells.
In a systems medicine approach, allo-HSCT represents the best example of 4P medicine integrating and balancing prediction, personalization, prevention and participation of patients in the management of health. Firstly, allo-HSCT is curative for almost all hematological cancers, even in the case of heavily pre-treated patients in an advanced stage. Despite comorbidity, nowadays, allo-HSCT could be offered to a wider number of patients due to the better supportive care and better management of the whole transplant procedure (donor, conditioning, immune-suppressive agents and caregiver selection) [8]. On the other hand, the development of chronic GvHD or late side effects of cancer treatment are sufficient reasons to define long term strategies, in order to prevent morbidity and mortality due to the procedure and to ensure the best quality of life for each patient [9].
Allo-HSCT is personalized: an individualized approach could also include personalized conditioning according to pharmacogenetic and pharmacokinetic data and different risk-stratified post-transplant procedures [10,11]. The personalized management of the underlying disease is, to date, molecular-driven, and MRD monitoring [12] predicts relapse and permits to act with cellular [13], targeted [14] or manipulated cellular therapies.
Allo-HSCT is predictive: the optimization of comorbidity indexes is crucial for the selection and personalization of conditioning in order to reduce TRM and to improve survival [15,16]. These scores could be improved by a systems medicine approach, using big data analysis and integration with social and supportive factors.
Allo-HSCT is preventive: prevention strategies are implemented and warranted in all transplant center programs, to reduce the risk of infections and long term complications [17]. Furthermore, monitoring of minimal residual disease (MRD) can prevent relapse after transplantation, with cellular or target therapies, as mentioned above.
Last but not least, allo-HSCT is participative: according to a chronic care model [18], participation of patients in the management of long term follow-up is crucial for greater empowerment, increasing self-esteem and quality of life. Long term follow-up clinics should include patients in the definition of needs, and efforts towards participative medicine are warranted. A critical issue is the use of information technology in the management of care delivery and validation of patient-reported outcomes. Self-reporting of chronic GvHD, for example, is superimposable to NIH scores [19] and could be screened with the help of Information Technologies (NMDP BMT application). The use of telemedicine, medical applications and other web-based suites could help transplanted patients in the management of their chronicity, ensuring constant contact with the transplant center [20,21].
Conclusions
Allogeneic HSCT is not an alternative to targeted therapies, but the constant increase in the number of transplanted patients worldwide suggests that allo-HSCT could represent a winning systemic health-model: predictive, personalized, preventive and participative. More studies are warranted to better elucidate the complexity of allotransplanted patients (“omics”, social, and psychological studies).
Footnotes
Authors' Disclosures of Potential Conflicts of Interest: No potential conflicts of interest relevant to this article were reported.
References
- 1.Hood L, Friend SH. Predictive, personalized, preventive, participatory (P4) cancer medicine. Nat Rev Clin Oncol. 2011;8:184–187. doi: 10.1038/nrclinonc.2010.227. [DOI] [PubMed] [Google Scholar]
- 2.Strebhardt K, Ullrich A. Paul Ehrlich's magic bullet concept: 100 years of progress. Nat Rev Cancer. 2008;8:473–480. doi: 10.1038/nrc2394. [DOI] [PubMed] [Google Scholar]
- 3.Schindler T, Bornmann W, Pellicena P, Miller WT, Clarkson B, Kuriyan J. Structural mechanism for STI-571 inhibition of abelson tyrosine kinase. Science. 2000;289:1938–1942. doi: 10.1126/science.289.5486.1938. [DOI] [PubMed] [Google Scholar]
- 4.O'Brien SG, Guilhot F, Larson RA, et al. Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med. 2003;348:994–1004. doi: 10.1056/NEJMoa022457. [DOI] [PubMed] [Google Scholar]
- 5.Innes AJ, Milojkovic D, Apperley JF. Allogeneic transplantation for CML in the TKI era: striking the right balance. Nat Rev Clin Oncol. 2016;13:79–91. doi: 10.1038/nrclinonc.2015.193. [DOI] [PubMed] [Google Scholar]
- 6.Niederwieser D, Baldomero H, Szer J, et al. Hematopoietic stem cell transplantation activity worldwide in 2012 and a SWOT analysis of the Worldwide Network for Blood and Marrow Transplantation Group including the global survey. Bone Marrow Transplant. 2016;51:778–785. doi: 10.1038/bmt.2016.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gratwohl A, Baldomero H, Aljurf M, et al. Hematopoietic stem cell transplantation: a global perspective. JAMA. 2010;303:1617–1624. doi: 10.1001/jama.2010.491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Passweg JR, Baldomero H, Bader P, et al. Use of haploidentical stem cell transplantation continues to increase: the 2015 European Society for Blood and Marrow Transplant activity survey report. Bone Marrow Transplant. 2017;52:811–817. doi: 10.1038/bmt.2017.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.D'Souza A, Pasquini M, Spellecy R. Is 'informed consent' an 'understood consent' in hematopoietic cell transplantation? Bone Marrow Transplant. 2015;50:10–14. doi: 10.1038/bmt.2014.207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.ten Brink MH, Zwaveling J, Swen JJ, Bredius RG, Lankester AC, Guchelaar HJ. Personalized busulfan and treosulfan conditioning for pediatric stem cell transplantation: the role of pharmacogenetics and pharmacokinetics. Drug Discov Today. 2014;19:1572–1586. doi: 10.1016/j.drudis.2014.04.005. [DOI] [PubMed] [Google Scholar]
- 11.El Cheikh J, Otrock ZK, Qannus AA, Kharfan-Dabaja MA, Bazarbachi A. Risk-adapted approach to HLA-matched sibling hematopoietic cell allografting: impact of adjusting conditioning intensity and integrating post-transplant therapeutic interventions. Clin Lymphoma Myeloma Leuk. 2016;16:304–310. doi: 10.1016/j.clml.2016.01.005. [DOI] [PubMed] [Google Scholar]
- 12.Malagola M, Skert C, Borlenghi E, et al. Postremission sequential monitoring of minimal residual disease by WT1 Q-PCR and multiparametric flow cytometry assessment predicts relapse and may help to address risk-adapted therapy in acute myeloid leukemia patients. Cancer Med. 2016;5:265–274. doi: 10.1002/cam4.593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Tsirigotis P, Byrne M, Schmid C, et al. Relapse of AML after hematopoietic stem cell transplantation: methods of monitoring and preventive strategies. A review from the ALWP of the EBMT. Bone Marrow Transplant. 2016;51:1431–1438. doi: 10.1038/bmt.2016.167. [DOI] [PubMed] [Google Scholar]
- 14.Giebel S, Czyz A, Ottmann O, et al. Use of tyrosine kinase inhibitors to prevent relapse after allogeneic hematopoietic stem cell transplantation for patients with Philadelphia chromosomepositive acute lymphoblastic leukemia: A position statement of the Acute Leukemia Working Party of the European Society for Blood and Marrow Transplantation. Cancer. 2016;122:2941–2951. doi: 10.1002/cncr.30130. [DOI] [PubMed] [Google Scholar]
- 15.Sorror ML, Maris MB, Storb R, et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood. 2005;106:2912–2919. doi: 10.1182/blood-2005-05-2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.DeFor TE, Le C, Smith AR, Warlick ED, Bejanyan N, Weisdorf DJ. Validation of a modified comorbidity index for allogeneic hematopoietic cell transplant. Blood. 2016;128(Suppl):abst 1167. (ASH Annual Meeting Abstracts) [Google Scholar]
- 17.Hashmi S, Carpenter P, Khera N, Tichelli A, Savani BN. Lost in transition: the essential need for long-term follow-up clinic for blood and marrow transplantation survivors. Biol Blood Marrow Transplant. 2015;21:225–232. doi: 10.1016/j.bbmt.2014.06.035. [DOI] [PubMed] [Google Scholar]
- 18.Wagner EH, Davis C, Schaefer J, Von Korff M, Austin B. A survey of leading chronic disease management programs: are they consistent with the literature. Manag Care Q. 1999;7:56–66. [PubMed] [Google Scholar]
- 19.Jacobsohn DA, Kurland BF, Pidala J, et al. Correlation between NIH composite skin score, patient-reported skin score, and outcome: results from the Chronic GVHD Consortium. Blood. 2012;120:2545–2552. doi: 10.1182/blood-2012-04-424135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rioth MJ, Warner J, Savani BN, Jagasia M. Next-generation long-term transplant clinics: improving resource utilization and the quality of care through health information technology. Bone Marrow Transplant. 2016;51:34–40. doi: 10.1038/bmt.2015.210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dyer G, Gilroy N, Brown L, et al. What they want: inclusion of blood and marrow transplantation survivor preference in the development of models of care for long-term health in Sydney, Australia. Biol Blood Marrow Transplant. 2016;22:731–743. doi: 10.1016/j.bbmt.2015.12.019. [DOI] [PubMed] [Google Scholar]