Abstract
The increased incidence of cancer and its high treatment costs have encouraged the search for new compounds to be used in adjuvant therapies for this disease. This study discloses the synthesis of (Z)-4-((1,5-dimethyl-3-oxo-2-phenyl-2,3dihydro-1H-pyrazol-4-yl) amino)-4-oxobut-2-enoic acid (IR-01) and evaluates not only the action of this compound on genetic integrity, increase in splenic phagocytosis and induction of cell death but also its effects in combination with the commercial chemotherapeutic agents doxorubicin, cisplatin and cyclophosphamide. IR-01 was designed and synthesized based on two multifunctionalyzed structural fragments: 4-aminoantipyrine, an active dipyrone metabolite, described as an antioxidant and anti-inflammatory agent; and the pharmacophore fragment 1,4-dioxo-2-butenyl, a cytotoxic agent. The results indicated that IR-01 is an effective chemoprotector because it can prevent clastogenic and/or aneugenic damage, has good potential to prevent genomic damage, can increase splenic phagocytosis and lymphocyte frequency and induces cell death. However, its use as an adjuvant in combination with chemotherapy is discouraged since IR-01 interferes in the effectiveness of the tested chemotherapeutic agents. This is a pioneer study as it demonstrates the chemopreventive effects of IR-01, which may be associated with the higher antioxidant activity of the precursor structure of 4-aminoantipyrine over the effects of the 1,4-dioxo-2-butenyl fragment.
Keywords: Splenic phagocytosis, comet assay, micronucleus test, cell death, chemoprevention
Introduction
Cancer comprises a group of diseases characterized by the progressive accumulation of mutations in the genome of a cell. These mutations lead to the altered expression or function of genes important for the maintenance of homeostasis, causing the loss of cell proliferation control (Steward and Brown, 2013). The genesis of cancer can occur via mutations (Ames et al., 1973); therefore, the chemopreventive and chemotherapeutic potential of synthetic compounds that are able to reduce or increase the frequency of DNA damage has been explored, yielding good models for genetic toxicology (de Araújo et al., 2017).
Nearly 2000 natural and synthetic compounds, including anti-inflammatory and antioxidant chemicals, have shown chemoprotective activity in preclinical trials, with good results also achieved in chemoprevention studies (Kim et al., 2002).
The class of pyrazolones and its derivatives, such as antipyrines, aminoantipyrines and dipyrones, comprises compounds with antioxidant activity (Pisoschi and Pop, 2015). This group also includes 4-aminoantipyrine, one of the active metabolites of dipyrone (Hedenmaln and Spigset, 2002), an anti-inflammatory, antipyretic and analgesic nonsteroidal drug (Salgado et al., 2015).
In another line of research involving the development of anticancer drugs, the pharmacophore fragment 1,4-dioxo-2-butenyl stands out because of its cytotoxic activity and ability to reduce cell proliferation (Jha et al., 2010). These are desirable characteristics in chemotherapeutic agents because they can be associated with good regulators of the cell cycle and cause the elimination of cells with DNA damage, such as tumor cells.
The possibility of success is enhanced by the ability of these structural fragments to interfere in early stages of carcinogenesis, acting on molecular and/or cellular targets specific to inflammatory and proliferation processes (Pathak et al., 2003).
To produce a compound that would have all the above characteristics, we conducted the synthesis of (Z)-4-((1,5-dimethyl-3-oxo-2-phenyl-2,3dihydro-1H-pyrazol-4-yl) amino)-4-oxobut-2-enoic acid (IR-01), which has the pharmacophore group 1,4-dioxy-2-butenyl as its structure base and contains the fragment 4-aminoantipyrine (an active dipyrone metabolite). Our aim was to develop a molecule with specific and effective therapeutic applications in the prevention and/or treatment of cancer.
In addition to the biological properties, the synthetic design took into account the low cytotoxicity of N-aryl-maleamic acids, which is attributable to the interaction between the two carboxyl groups and the olefinic fragment that may hinder the passage of the compounds through the cell membrane. The hypothesis proposed in the literature (Jha et al., 2010) proposes that the cytotoxic capacity of the compounds containing these fragments is primarily controlled by the olefinic and aryl groups and the spatial arrangement between these fragments, which can directly affect the compound’s access through the lipid bilayer to the interior of the cell. The polarity balance may facilitate both the passage of the compound through the cell membrane (which would potentiate its biological effects) and the excretion of the compound from the body (after exerting its biological effects).
This study reports the synthesis of IR-01, taking into consideration the structural characteristics described above and the evaluation of IR-01 regarding genetic integrity, splenic phagocytosis evaluation and the induction of cell death. Furthermore, the study describes the effects of IR-01 in combination with the commercial chemotherapeutic agents doxorubicin, cisplatin and cyclophosphamide.
Material and Methods
Chemistry
Synthesis of IR-01
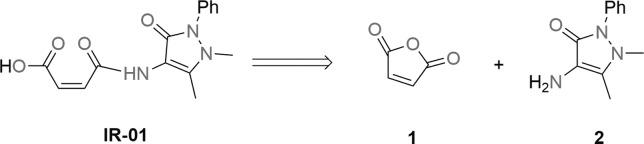
Starting with low-cost materials and a one-pot procedure, the reaction of maleic anhydride with the corresponding amine was performed using a microwave reactor but no solvent, for a cleaner methodology with high reproducibility and good yield, further lending validity to the method (Figure 1).
Figure 1. Retrosynthetic analysis for (Z)-4-((1,5-dimethyl-3-oxo-2-phenyl-2,3dihydro-1H-pyrazol-4-yl) amino)-4-oxobut-2-enoic acid.
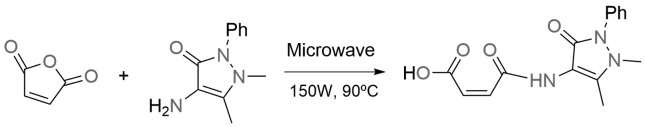
The formation of the synthetic target occurs after the attack of the 4-aminoantipyrine nitrogen on the carbonyl carbon of maleic anhydride, which provides ring opening and the subsequent formation of the acid of interest with good yield (Figure 2).
Figure 2. Synthesis of (Z)-4-((1,5-dimethyl-3-oxo-2-phenyl-2,3dihydro-1H-pyrazol-4-yl) amino)-4-oxobut-2-enoic acid.
Reagents and techniques
All reagents and spectrograde solvents for synthesis and NMR measurements were purchased commercially and used without further purification.
The melting point was determined on a Quimis dry melting point apparatus, model Q340S23, and used as uncorrected data. The microwave procedure was performed in a CEM/Discover microwave reactor with sealed tube.
1H and 13C NMR spectra were recorded at room temperature on a Bruker 300 spectrometer (10% in deuterated dimethylsulfoxide (DMSO-d6) solutions at 298 K) operating at 300.132 and 75.476 MHz, respectively. Data processing was conducted on a Solaris workstation. The 1H and 13C chemical shifts are reported on the δ scale (ppm) and referenced to internal DMSO-d6; coupling constants J are reported in hertz (Hz). The abbreviations s, d and m represent simplet, douplet and multiplet, respectively.
Synthesis process
In a sealed tube, 4-aminoantipyrine (2.0 g, 10 mmol) and maleic anhydride (1.0 g, 10 mmol) were subjected to microwave irradiation (150 W) at 90 °C for 10 s. The solid was washed with ethyl acetate and filtered. The remaining yellow solid was recrystallized from CH3Cl, giving IR-01 (2.79 g, 93%). 1H NMR (DMSO-d6, 300 MHz) δ (ppm): 2.13 (s, 3H), 3.02 (s, 3H), 6.26 (d, 1H, J cis = 12.3 Hz), 6.48 (d, 1H, J cis = 12.3 Hz), 7.30 (m, 3H), 7.46 (m, 2H). 13C NMR (CDCl3, 75 MHz) δ (ppm): 11.71 (CH3), 36.24 (CH3), 106.62 (C), 124.22 (CH), 126.94 (CH), 129.60 (CH), 131.54 (CH), 131.60 (CH), 135.25 (C), 152.56 (C), 161.73 (C=O), 164.58 (C=O), 167.03 (C=O). Melting point: 178.1-179.8 °C.
Chemical agents, animals and experimental design
The DNA-damage-inducing agents (commercial chemotherapeutic agents) used in this study were the following: doxorubicin (Glenmark Pharmaceuticals Ltd., Argentina. MS Reg. No. 1.1013.0232.002-4, Lot #21130040) at a dose of 16 mg/kg body weight (b.w.) intraperitoneally (ip) cisplatin (Accord Pharmaceuticals Ltd., UK. MS Reg. No. 1.5537.0002.003-7, Lot #88549) at a dose of 6 mg/kg (b.w., ip), and cyclophosphamide (Genuxal®, Baxter Ltda., Germany. MS Reg. No. 1.00683.0168.003-1, Lot #F728) at a dose of 100 mg/kg (b.w., ip). Doxorubicin and cyclophosphamide were diluted in distilled water.
IR-01 was first diluted in 5% DMSO and then in glycated serum before the drug was administered at doses of 12, 24 and 48 mg/kg (b.w., ip).
Eighty Swiss female mice (with a mean weight of 30 g, 6-8 weeks old) were randomly distributed into 16 experimental groups (n = 5).
The animals were housed in individual cages lined with wood shavings on a ventilated rack (Alesco®) and provided commercial feed (Nuvital®) and filtered water ad libitum. The experimental conditions were controlled, with a 12-hour light:12-hour dark photoperiod, mean temperature of 22 2 °C and mean humidity of 55 ± 10%. The experiment was approved by the Animal Ethics Committee of the Federal University of Mato Grosso do Sul (Universidade Federal de Mato Grosso do Sul - UFMS) under protocol no. 399/2012 and performed according to the Declaration of Animal Rights.
For the evaluation of the IR-01 effects, the following experimental groups were established:
In lot 1, a negative control group comprised animals that received a dose of distilled water and another of 5% DMSO in glycated serum, both at 0.1 mL/10 g (b.w., ip). The IR-01 groups in lot 1 comprised animals treated with IR-01 at concentrations of 12, 24 and 48 mg/kg (b.w., ip) and with a dose of distilled water at 0.1 mL/10 g (b.w., ip).
To assess the effects of combining IR-01 with the commercial chemotherapeutic agents, the following experimental groups were established.
In lot 2, a doxorubicin group (DOX) comprised animals treated with doxorubicin at a dose of 16 mg/kg (b.w., ip) and with 5% DMSO in glycated serum at a dose of 0.1 mL/10 g (b.w., ip). The DOX + IR-01 groups in lot 2 comprised animals that were treated with doxorubicin at a dose of 16 mg/kg (b.w., ip) and IR-01 at doses of 12, 24 and 48 mg/kg (b.w., ip).
The animals in lots 3 and 4 were treated as described in lot 2 except that doxorubicin was replaced by cisplatin (CIS and CIS + IR-01) and cyclophosphamide (CPP and CPP + IR-01) at doses of 6 and 100 mg/kg (b.w., ip), respectively.
At 24 (T1), 48 (T2) and 72 (T3) hours after the treatments, 20 μL of peripheral blood was collected to perform a micronucleus assay. Additionally, 20 μL of peripheral blood was collected at T1 to perform a comet assay. A new aliquot was collected at T3 for a differential blood cell count. At the end of the experiment, at T3, the animals were euthanized by cervical dislocation to collect the spleen for a phagocytosis test and the kidney and liver for cell death analysis.
Biological assays
Peripheral blood Comet assay
The comet assay was performed according to the protocol of Singh et al. (1988), with modifications by Oliveira et al. (2015a). The material was analyzed using an epifluorescence microscope (Bioval®, model L 2000A) with a 40 objective, a 420-490 nm excitation filter and a 520 nm barrier filter. As described by Kobayashi et al. (1995), a total of 100 cells per treatment were inspected visually and the comets were classified as: class 0, undamaged cells showing no tail; class 1, cells with a tail size smaller than the diameter of the nucleoid; class 2, cells with a tail size 1- 2 times the diameter of the nucleoid; class 3, cells with a tail size greater than two times the diameter of the nucleoid. Apoptotic cells that showed a totally fragmented nucleus were not scored. The total score was calculated as the sum of the number of cells scored for each class times that class value.
Peripheral blood Micronucleus assay
The micronucleus assay in peripheral blood was performed according to Hayashi et al. (1990), with modifications by Oliveira et al. (2015a). A 20 μL peripheral blood aliquot was covered with a cover slip after its deposition on a slide precoated with 20 μL of acridine orange (1.0 mg/mL). The slide was stored in a freezer (-20 °C) for at least seven days. The analysis was performed under an epifluorescence microscope with a 40 objective (Bioval®, model L 2000A) along with a 420-490 nm excitation filter and a 520 nm barrier filter. Two thousand cells were analyzed per animal.
Cell death assay
One hundred microliters of a macerated liver or kidney solution was placed on a slide. Next, the slide was fixed in Carnoy’s solution for 5 min and was then subjected to a decreasing series of ethanol concentrations (95-25%), washed with McIlvaine’s buffer for 5 min, stained with 0.01% acridine orange for 5 min and washed again with buffer. Dying cells were identified through an analysis of the DNA fragmentation patterns, according to Carvalho et al. (2015) and Navarro et al. (2014).
Splenic phagocytosis assay
The spleen was macerated in saline solution. One hundred microliters of cell suspension was covered with a coverslip after its placement on a slide previously coated with 20 μL of acridine orange (1.0 mg/mL). The slides were stored in a freezer until their analysis, which was performed with a fluorescence microscope (Bioval®, model L 2000A) using a 40 objective along with a 420-490 nm filter and a 520 nm barrier filter. Two hundred cells were analyzed per animal. The presence or absence of phagocytosis was determined based on the descriptions of Carvalho et al. (2015) and Hayashi et al. (1990).
Differential blood cell count
A 20 μL aliquot of peripheral blood was used to prepare blood smears on glass slides. These slides were air dried and stained with a panoptic kit for 10 min. The cells were visualized under bright field microscopy using a 100 objective. A total of 100 cells per animal were analyzed and classified as lymphocytes, neutrophils, monocytes, eosinophils and basophils (Ishii et al., 2011).
Calculation of percent damage reduction (%DR) and percent damage increase (%DI)
Manoharan and Banerjee (1985) and Waters et al. (1990) proposed the calculation of percent damage reduction to assess the chemopreventive ability of a substance when it is associated with a substance known to be mutagenic, such as the commercial chemotherapeutic agents used as positive control (PC). According to Oliveira et al. (2015a) and Navarro et al. (2014), the same calculation can be used to estimate the increase in DNA damage. Thus, for the present study, both the percent DNA damage reduction and percent DNA damage increase were calculated using the same formula:
Statistical Analysis
Data are reported as the mean ± standard error of the mean (SEM) and analyzed using Student’s t-test or the Mann-Whitney test, depending on whether the data distribution was parametric or nonparametric, respectively, using GraphPad InStat Demo version 3.6 (GraphPad Software Inc., San Diego, CA, USA). The significance level adopted was p < 0.05.
Results
Synthesis
The product formed was characterized by 1H and 13C NMR, and the results described below demonstrated chemical shifts and integrations consistent with IR-01.
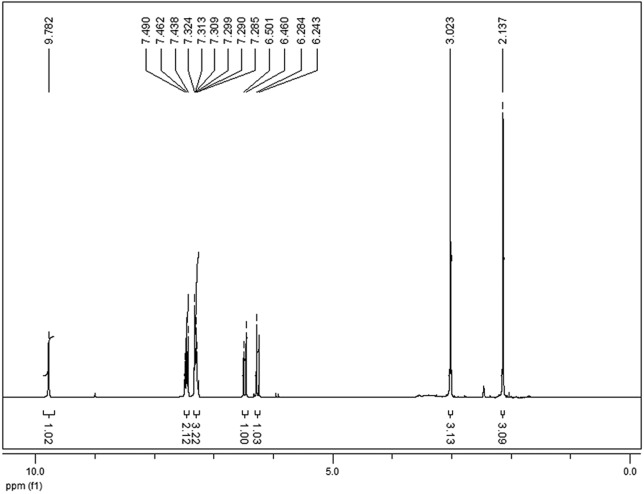
In the 1H NMR spectra (Figure 3), two signals at the 6.26 and 6.48 ppm regions (J = 12.3 Hz), representing the two olefinic hydrogen doublets of the 1,4-dioxo-butenyl fragment, indicate the Z configuration of the compound obtained; the signal at 9.78 ppm refers to the amidic hydrogen.
Figure 3. 1H RMN spectra of IR-01 in DMSO-d6 at 300 Mhz.
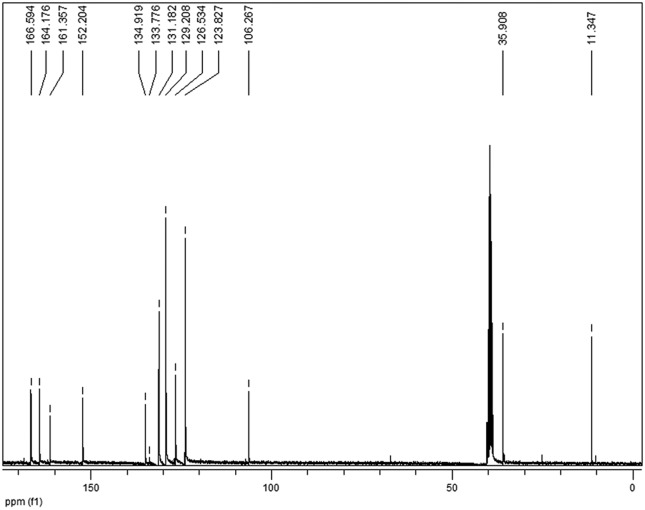
In the 13C NMR spectra (Figure 4), the three signals observed between 161.35 and 166.59 ppm, representing the IR-01 carbonyls, confirm the formation of the synthetic target.
Figure 4. 13C RMN spectra of IR-01 in DMSO-d6 at 75 MHz.
All other 1H and 13C NMR signals are in agreement with the data reported in the literature for the same compound (Cunha et al., 2005).
Biological assays
Assessment of genetic integrity and effects of IR-01 on DNA damage caused by commercial chemotherapeutic agents
The genetic integrity assessment indicated that IR-01 can cause genomic damage (comet assay) but is unable to cause chromosomal damage (micronucleus assay). The treatments with the test compound caused an increase (p < 0.05) in the frequency of DNA damage by 2.37 and 4.44 and in the score by 2.3 and 4.05 for the 24 and 48 mg/kg doses, respectively (Table 1). The micronucleus frequency ranged from 0.6 ± 0.24 to 3.0 ± 0.44 in the control group and from 3.0 ± 0.31 to 5.2 ± 0.66 in the IR-01-treated groups (Table 2).
Table 1. Results of the comet assay showing the ability of IR-01 to cause or prevent genomic damage.
| Experimental groups | Damaged cells | Damage classes | Score | %DR | |||
|---|---|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | ||||
| LOT 1 | |||||||
| NC | 16 ± 1.77 | 81 ± 2.43 | 12 ± 1.43 | 2.6 ± 0.24 | 1.0 ± 0.31 | 20 ± 2.35a | - |
| IR-01 12 mg/kg | 21 ± 1.71a | 78 ± 1.71 | 15 ± 0.67 | 4.2 ± 0.86 | 2.0 ± 0.44 | 30 ± 3.39ª | - |
| IR-01 24 mg/kg | 38 ± 0.86a* | 61 ± 0.86 | 33 ± 0.96 | 3.8 ± 1.02 | 1.80.58 | 46 ± 2.95a* | - |
| IR-01 48 mg/kg | 71 ± 0.37a* | 28 ± 0.37 | 64 ± 1.56 | 5.4 ± 1.20 | 2.0 ± 0.83 | 81 ± 2.47a* | - |
| LOT 2 | |||||||
| DOX | 88 ± 0.50a | 11 ± 0.50 | 56 ± 1.12 | 18 ± 1.03 | 13 ± 0.67 | 134 ± 2.48a | - |
| +IR-01 12 mg/kg | 62 ± 2.61b | 37 ± 2.61 | 47 ± 1.24 | 13. ± 1.24 | 1.20.58 | 77 ± 4.30b | 36.11 |
| +IR-01 24 mg/kg | 73 ± 2.47b | 26 ± 2.47 | 49 ± 2.70 | 20 ± 1.64 | 4.6 ± 1.16 | 103 ± 4.83b | 20.83 |
| +IR-01 48 mg/kg | 85 ± 0.86b | 14 ± 0.86 | 55 ± 3.95 | 252.51 | 4.4 ± 1.63 | 120 ± 4.05b | 4.17 |
| LOT 3 | |||||||
| CIS | 27 ± 1.37a | 72 ± 1.31 | 22 ± 1.77 | 4.4 ± 0.40 | 0.4 ± 0.40 | 35 ± 3.16a | - |
| +IR-01 12 mg/kg | 19 ± 1.24c | 80 ± 1.24 | 19 ± 1.18 | 0.2 ± 0.20 | 0.0 ± 0.0 | 19 ± 1.32c | 72.73 |
| +IR-01 24 mg/kg | 46 ± 4.77c | 53 ± 4.77 | 33 ± 5.62 | 8.8 ± 1.39 | 2.2 ± 0.96 | 59 ± 3.57c | -172.73 |
| +IR-01 48 mg/kg | 56 ± 1.65c | 43 ± 1.65 | 44 ± 1.67 | 10 ± 0.87 | 1.6 ± 0.40 | 70 ± 1.94c | -263.64 |
| LOT 4 | |||||||
| CPP | 74 ± 2.01a | 252.01 | 64 ± 2.21 | 10 ± 0.87 | 0.0 ± 0.0 | 85 ± 2.09a | - |
| +IR-01 12 mg/kg | 89 ± 0.92d | 10 ± 0.92 | 73 ± 1.63 | 16 ± 1.67 | 0.0 ± 0.0 | 101 ± 3.53d | -25.86 |
| +IR-01 24 mg/kg | 19 ± 2.52d | 80 ± 2.65 | 14 ± 1.88 | 3.0 ± 0.70 | 1.8 ± 0.73 | 26 ± 3.81d | 94.83 |
| +IR-01 48 mg/kg | 63 ± 3.52d | 3522.8 | 57 ± 2.80 | 7.2 ± 0.86 | 0.0 ± 0.0 | 72 ± 1.22d | 18.97 |
Data is represented as mean values ± standard error of the mean. %DR: Percent damage reduction; NC: Negative control group; DOX: Doxorubicin group; CIS: Cisplatin group; CPP: Cyclophosphamide group. (a)Statistically compared to the NC group; (b)Statistically compared to the DOX group; (c)Statistically compared to the CIS group; (d)Statistically compared to the CPP group; *statistically different (p < 0.05; Mann-Whitney test).
Table 2. Results of the micronucleus assay related to the ability of IR-01 to cause or prevent chromosomal damage.
| Experimental groups | Mean ± SE | %DR | |||||
|---|---|---|---|---|---|---|---|
| 24 h | 48 h | 72 h | 24 h | 48 h | 72 h | ||
| LOT 1 | |||||||
| NC | 3.0 ± 0.44 | 1.8 ± 0.20 | 0.6 ± 0.24 | - | - | - | |
| IR-01 12 mg/kg | 3.8 ± 0.37a | 3.0 ± 0.31ª* | 4.2 ± 0.37ª* | - | - | - | |
| IR-01 24 mg/kg | 4.2 ± 0.20a | 4.6 ± 0.60ª* | 4.8 ± 0.37ª* | - | - | - | |
| IR-01 48 mg/kg | 4.8 ± 0.37ª* | 3.4 ± 0.50ª* | 5.2 ± 0.66ª* | - | - | - | |
| LOT 2 | |||||||
| DOX | 52 ± 2.56a* | 36 ± 1.72a* | 26 ± 1.16ª* | - | - | - | |
| + IR-01 12 mg/kg | 7.20.58b* | 5.2 ± 0.37b* | 7.2 ± 0.86b* | 91.43 | 90.06 | 74.01 | |
| + IR-01 24 mg/kg | 12 ± 0.70b* | 6.4 ± 0.50b* | 7.2 ± 0.37b* | 81.63 | 86.55 | 74.01 | |
| + IR-01 48 mg/kg | 13 ± 1.06b* | 9.4 ± 0.40b* | 9.4 ± 0.60b* | 79.59 | 77.78 | 65.35 | |
| LOT 3 | |||||||
| CIS | 27 ± 0.50ª* | 20 ± 0.55a* | 15 ± 0.50ª* | - | - | - | |
| + IR-01 12 mg/kg | 6.2 ± 0.37c* | 5.6 ± 0.24c* | 4.2 ± 0.20c* | 86.67 | 79.12 | 75.00 | |
| + IR-01 24 mg/kg | 7.0 ± 0.37c* | 7.6 ± 0.50c* | 6 ± 0.31c* | 83.33 | 68.14 | 62.50 | |
| + IR-01 48 mg/kg | 8.4 ± 0.50c* | 7.4 ± 0.60c* | 5.8 ± 0.37c* | 77.50 | 69.23 | 63.89 | |
| LOT 4 | |||||||
| CPP | 41 ± 2.17a* | 50 ± 2.34a* | 28 ± 2.80a* | - | - | - | |
| + IR-01 12 mg/kg | 33 ± 2.71d | 63 ± 3.75d | 24 ± 2.16d | 21.05 | -26.97 | 14.60 | |
| + IR-01 24 mg/kg | 55 ± 3.63d* | 77 ± 2.95d* | 42 ± 3.88d* | -36.84 | -56.02 | -51.10 | |
| + IR-01 48 mg/kg | 42 ± 3.85d | 71 ± 2.70d | 28 ± 2.10d | -2.63 | -43.57 | 00.00 | |
SE: Standard error of the mean; %DR: Percent damage reduction; NC: Negative control group; DOX: Doxorubicin group; CIS: Cisplatin group; CPP: Cyclophosphamide group. (a)Statistically compared to the NC group; (b)Statistically compared to the DOX group; (c)Statistically compared to the CIS group; (d)Statistically compared to the CPP group; *statistically different (p < 0.05; Mann-Whitney test).
When associated with the commercial chemotherapeutic agents, IR-01 showed chemopreventive activity for most associations, except for the two highest doses when administered with cisplatin and evaluated in the comet assay (Table 1) and the intermediate dose in combination with cyclophosphamide in the icronucleus assay (Table 2).
The percent damage reduction by IR-01 in the Comet assay ranged between 4.17 and 36.11% in combination with doxorubicin and between 18.87 and 94.83% in combination with cyclophosphamide and was 72.73% for the lowest IR-01 dose in combination with cisplatin. An increase in percent damage was observed for the two highest IR-01 doses in combination with cisplatin (172.73 and 263.64% for 24 and 48 mg/kg, respectively) and for the lowest IR-01 dose in combination with cyclophosphamide (25.86%) (Table 1).
In the micronucleus assay, the percent damage reduction ranged from 65.35 to 91.43% for the combination with doxorubicin, from 62.50 to 86.67% for the combination with cisplatin and from 0 to 14.60% for the cyclophosphamide combination. For the latter, an increase in DNA damage that reached 56.02% was observed at the intermediate dose (Table 2).
Evaluation of the splenic phagocytosis potential and effects of IR-01 in combination with commercial chemotherapeutic agents
When administered alone, the two highest doses of IR-01, 24 and 48 mg/kg, increased (p < 0.05) the rate of splenic phagocytosis by 1.43 and 1.67, respectively (Table 3).
Table 3. Results related to splenic phagocytosis evaluation.
| Experimental groups | Phagocytosis | |
|---|---|---|
| Absolute values | Mean ± SE | |
| LOT 1 | ||
| NC | 221 | 44.2 ± 0.66 |
| IR-01 12 mg/kg | 228 | 45.6 ± 1.77ª |
| IR-01 24 mg/kg | 316 | 63.2 ± 1.28ª* |
| IR-01 48 mg/kg | 369 | 73.8 ± 1.15ª* |
| LOT 2 | ||
| DOX | 670 | 134.0 ± 1.37ª* |
| +IR-01 12 mg/kg | 321 | 64.2 ± 1.59b* |
| +IR-01 24 mg/kg | 405 | 81.0 ± 1.30b* |
| +IR-01 48 mg/kg | 373 | 74.6 ± 1.43b* |
| LOT 3 | ||
| CIS | 269 | 53.8 ± 1.35ª* |
| +IR-01 12 mg/kg | 106 | 21.2 ± 1.35c* |
| +IR-01 24 mg/kg | 74 | 14.8 ± 1.02c* |
| +IR-0148 mg/kg | 54 | 10.8 ± 0.86c* |
| LOT 4 | ||
| CPP | 515 | 103.0 ± 2.00ª* |
| +IR-01 12 mg/kg | 275 | 55.0 ± 3.46d* |
| +IR-01 24 mg/kg | 316 | 63.2 ± 2.57d* |
| +IR-01 48 mg/kg | 534 | 106.8 ± 3.13d |
SE: Standard error of the mean; NC: Negative control group; DOX: Doxorubicin group; CIS: Cisplatin group; CPP: Cyclophosphamide group. (a)Statistically compared to the NC group; (b)Statistically compared to the DOX group; (c)Statistically compared to the CIS group; (d)Statistically compared to the CPP group; *statistically different (p < 0.05; Student’s t-test).
In combination with doxorubicin, IR-01 reduced (p < 0.05) the frequency of phagocytosis by 47.91, 60.45 and 55.67% at the 12, 24 and 48 mg/kg doses, respectively. In combination with cisplatin, the respective reductions were 39.40, 27.51 and 20.07%. In combination with cyclophosphamide, the reductions were 53.40 and 61.36% for the 12 and 24 mg/kg doses, respectively (Table 3).
The differential blood cell count showed that at the three doses tested, IR-01 administered alone can increase (p < 0.05) the frequency of lymphocytes and reduce (p < 0.05) neutrophil and monocyte counts. Additionally, in the treatment with the commercial chemotherapeutic agents, an increase (p < 0.05) in the frequency of lymphocytes and a reduction in that of monocytes occurred for doxorubicin, cisplatin and cyclophosphamide, with a reduction (p < 0.05) in neutrophils also occurring for doxorubicin (Table 4).
Table 4. Reference values and results related to the differential blood cell count.
| Experimental Groups | Cell types | ||||
|---|---|---|---|---|---|
| 55-95% | 10-40% | 0.0-0.3% | 0.0-0.4% | 0.1-3.5% | |
| Lymphocytes2 | Neutrophils2 | Basophils1 | Eosinophils1 | Monocytes2 | |
| LOT 1 | |||||
| NC | 67 ± 0.74 | 22.0 ± 0.81 | 0 ± 0.001 | 0 ± 0.00 | 10.0 ± 0.40 |
| IR-01 12 mg/kg | 78 ± 1.48a* | 11.0 ± 0.67a* | 0 ± 0.00a | 0 ± 0.00a | 9.6 ± 0.50a* |
| IR-01 24 mg/kg | 82 ± 1.00a* | 8.0 ± 0.63ª* | 0 ± 0.00a | 0 ± 0.00a | 6.8 ± 1.31a* |
| IR-01 48 mg/kg | 86 ± 0.81a* | 9.0 ± 0.87a* | 0 ± 0.00a | 0 ± 0.00a | 4.6 ± 1.74a* |
| LOT 2 | |||||
| DOX | 76 ± 2.46ª* | 12 ± 0.67ª* | 0 ± 0.00a | 2.81.31a | 9.4 ± 1.83a* |
| +IR-01 12 mg/kg | 85 ± 1.53b* | 8.4 ± 1.03b* | 0 ± 0.00b | 1.80.58b | 3.6 ± 0.92b* |
| +IR-01 24 mg/kg | 85 ± 1.68b* | 11.00.58b | 0 ± 0.00 b | 1.4 ± 0.92b | 1.4 ± 0.50b* |
| +IR-01 48 mg/kg | 891.58b* | 7.0 ± 0.47b* | 0 ± 0.00b | 2.5 ± 1.50b | 1.2 ± 0.62b* |
| LOT 3 | |||||
| CIS | 78 ± 0.73ª* | 17 ± 1.88a | 0 ± 0.00a | 0 ± 0.00a | 3.4 ± 1.32a* |
| +IR-01 12 mg/kg | 83 ± 2.62c | 10 ± 1.20c* | 0 ± 0.00c | 0.2 ± 0.20c | 6.4 ± 0.00c |
| +IR-01 24 mg/kg | 85 ± 1.36c* | 10 ± 0.96c* | 0 ± 0.00c | 0 ± 0.00c | 4.2 ± 0.76c |
| +IR-01 48 mg/kg | 85 ± 1.00c* | 10 ± 0.50c* | 0 ± 0.00c | 0 ± 0.00c | 4.4 ± 0.67c |
| LOT 4 | |||||
| CPP | 85 ± 3.49ª* | 11 ± 3.53a | 0 ± 0.00ª | 0 ± 0.00a | 2.81.06a* |
| +IR-01 12 mg/kg | 85 ± 3.92d | 10 ± 2.64d | 0 ± 0.00d | 0 ± 0.00d | 4.2 ± 1.35d |
| +IR-01 24 mg/kg | 92 ± 2.31d | 4.6 ± 1.60d | 0 ± 0.00d | 0 ± 0.00d | 3.0 ± 1.00d |
| +IR-01 48 mg/kg | 86 ± 0.48d | 11 ± 0.50d | 0 ± 0.00d | 0 ± 0.00d | 1.8 ± 0.86d |
Data is represented as mean values ± standard error of the mean. Statistical tests: (1)Student’s t-test(p < 0.05) and (2)Mann-Whitney test (p < 0.05). NC: Negative control group; (a)Statistically compared to the NC group; (b)Statistically compared to the DOX group; (c)Statistically compared to the CIS group; (d)Statistically compared to the CPP group; *statistically different.
The following results were observed for the combinations of chemotherapeutic agents with IR-01: (I) for doxorubicin, an increase (p < 0.05) in the frequency of lymphocytes and a reduction (p < 0.05) in that of monocytes for all the doses tested and a reduction (p < 0.05) in neutrophil frequency for the lowest and highest doses; (II) for cisplatin, an increase (p < 0.05) in the frequency of lymphocytes for the two higher doses and a reduction (p < 0.05) in that of neutrophils for all the doses; and (III) for cyclophosphamide, no statistically significant change (Table 4).
Neutropenia was observed in the groups treated with the two highest doses of IR-01, in DOX + IR-01 at the lowest and highest doses and in CPP + IR-01 at the intermediate dose. Eosinophilia was observed in the animals treated with cisplatin when combined with all doses of IR-01, and monocytosis occurred in the control groups treated with IR-01, in DOX, in DOX + IR-01 at the lowest dose, in CIS + IR-01 at all three doses, and in CPP + IR-01 at the lowest dose tested (Table 4).
Evaluation of cell death induction and effects of IR-01 in combination with commercial chemotherapeutic agents
The administration of IR-01 increased (p < 0.05) the frequency of dead cells in the liver by 1.69, 2.44 and 3.17 and in the kidneys by 2.05, 3.11 and 3.89 at the 12, 24 and 48 mg/kg doses, respectively (Table 5).
Table 5. Cell death evaluation on mice kidneys and liver.
| Liver | Kidneys | |||
|---|---|---|---|---|
| Experimental Groups | Number of dead cells | Mean ± SE | Number of dead cells | Mean ± SE |
| LOT 1 | ||||
| NC | 88 | 17.6 ± 1.03 | 63 | 12.6 ± 0.81 |
| IR-01 12 mg/kg | 149 | 29.8 ± 1.93a* | 129 | 25.8 ± 1.24a* |
| IR-01 24 mg/kg | 215 | 43.0 ± 1.93a | 196 | 39.20.58a* |
| IR-01 48 mg/kg | 279 | 55.8 ± 0.86a* | 245 | 49.0 ± 1.04a* |
| LOT 2 | ||||
| DOX | 499 | 99.8 ± 2.55a* | 176 | 35.2 ± 2.57a* |
| +IR-01 12 mg/kg | 779 | 1552.57b* | 358 | 71.6 ± 3.40b* |
| +IR-01 24 mg/kg | 610 | 122 ± 1.84b* | 291 | 58.2 ± 1.39b* |
| +IR-01 48 mg/kg | 565 | 113 ± 2.12b* | 246 | 49.2 ± 2.28b* |
| LOT 3 | ||||
| CIS | 622 | 124.4 ± 6.03a* | 528 | 1051.72a* |
| +IR-01 12 mg/kg | 402 | 80.4 ± 5.92c* | 297 | 59.4 ± 1.03c* |
| +IR-01 24 mg/kg | 247 | 49.4 ± 3.80c* | 221 | 44.2 ± 2.08c* |
| +IR-01 48 mg/kg | 143 | 28.6 ± 2.42c* | 197 | 39.4 ± 2.92c* |
| LOT 4 | ||||
| CPP | 614 | 122 ± 3.36a* | 588 | 117 ± 1.16a* |
| +IR-01 12 mg/kg | 307 | 61.4 ± 3.95d* | 522 | 104 ± 1.93d* |
| +IR-01 24 mg/kg | 362 | 72.4 ± 4.63d* | 381 | 76.2 ± 3.35d* |
| +IR-01 48 mg/kg | 197 | 39.4 ± 5.47d* | 377 | 75.4 ± 2.08d* |
SE: Standard error of the mean; NC: Negative control group; DOX: Doxorubicin group; CIS: Cisplatin group; CPP: Cyclophosphamide group. (a)Statistically compared to the NC group; (b)Statistically compared to the DOX group; (c)Statistically compared to the CIS group; (d)Statistically compared to the CPP group; *statistically different (p < 0.05; Student’s t-test).
The commercial chemotherapeutic agents doxorubicin, cisplatin and cyclophosphamide caused an increase in dead cells frequency of 5.67, 7.07 and 6.98 in the liver and 2.79, 8.38 and 9.33 in the kidneys, respectively (Table 5).
The following results were observed for the chemotherapeutic agents tested in combination with IR-01: (I) for doxorubicin, the potentiation of cell death (p < 0.05) by 155.31% in the liver and 203.41% in the kidneys for the lowest IR-01 dose tested; (II) for cisplatin, a reduction of dead cells (p < 0.05) by up to 22.99% in the liver and 37.52% in the kidneys for the highest IR-01 dose; and (III) for cyclophosphamide, reductions of 32.29 and 64.44% in the liver and kidneys, respectively, also for the highest IR-01 dose (Table 5).
Discussion
The increase in cancer incidence and the high cost of treatments motivate the search for new strategies to prevent and manage this disease (Mauro et al., 2011). An approach with great potential is chemoprevention, which involves the use of natural and/or synthetic agents to suppress, inhibit or reverse the process of carcinogenesis in its early stages (Friedman and Rasooly, 2013).
Organic synthesis has gained prominence in the search to develop more potent and less toxic molecules, and the redesign and structural modification of previously known compounds or radicals allow important advances in defining biological activities and in structure-activity studies. Under this perspective, our research group designed and synthesized IR-01 (Z)-4-((1,5-dimethyl-3-oxo-2-phenyl-2,3dihydro-1H-pyrazol-4-yl) amino)-4-oxobut-2-enoic acid using 4-aminoantipyrine associated to the structural fragment 1,4-dioxo-2-butenyl, observing the influence of the position of the phenyl ring, the distance between fragments, the increased number of heteroatoms and the increased number of olefins, as indicated in the literature (Jha et al., 2010).
Often, the synthesis of a biologically effective compound can use reagents and/or approaches of considerable environmental impact. Thus, compared with the efficient but environmently agressive synthetic procedure described in the literature (Cunha et al., 2005), our one-pot method assisted by microwave irradiation can be a similarly efficient but cleaner approach for the synthesis of IR-01, with a very good yield and a reaction time of only 1% compared to that reported in the literature.
The results of the biological studies on IR-01, prepared using this new synthesis method, suggest that it can cause DNA damage. However, this damage does not become fixed in the cell genome, a hypothesis that is reinforced by the fact that the damage evaluated by the comet assay did not result in a significant increase in micronucleus frequency. According to Rundell et al. (2003), genotoxic damage is likely to undergo repair, whereas mutagenic damage is not, with changes becoming fixed in the genetic material as mutations. Such occurrence is not uncommon in preclinical chemoprevention experiments, both with natural products (Synder and Gillies, 2002; Cunha et al., 2005; Rodeiro et al., 2006; Hoshina et al., 2013; Mendanha da Cunha et al., 2013; Luo et al., 2015) and with synthetic compounds (Zhan et al., 2008; Cao et al., 2015; Frolova et al., 2015; de Araújo et al., 2017). Although there are significant differences in the frequency of micronuclei between the negative control group and the groups treated with IR-01, Vaz et al. (2016) describes that this isolated fact does not necessarily imply in toxicogenetic damage. Also, according to the results observed in other experiments from our research group (Oliveira et al., 2009a, 2013; Mauro et al., 2010; Pesarini et al., 2014; Navarro et al., 2015), the baseline frequency of micronuclei can be greater than the frequency observed for the animals treated with IR-01 in the present study. For example, according to Oliveira et al. (2015b), the baseline frequency of micronuclei in Swiss female mice may reach 11.00 ± 3.16.
Based on these data, IR-01 is considered to cause genomic damage but is unable to cause chromosomal damage. The genotoxic activity may occur because of the pharmacophore 1,4-dioxo-2-butenyl, which has already been described as an effective cytotoxic agent in the tumor cell lines Molt4/C8 and CEM L1210 (Jha et al., 2010). However, the addition of 4-aminoantipyrine may have modified this property, which is required in anticancer agents. It is important to highlight that chemotherapeutic agents generally include in their mechanism of action the induction of DNA damage that causes cell death, especially that of tumor cells (Elsendoorn et al., 2001; Nadin et al., 2005).
The chemopreventive action described may be explained by IR-01, which originated from 4-aminoantipyrine, retaining the antioxidant activity of its precursor. This hypothesis is consistent with the data from the present study because the best capacity for preventing genomic damage were observed for doxorubicin and cyclophosphamide, chemotherapeutic agents that are capable of generating free radicals that cause rather extensive genotoxic damage (Almeida et al., 2005), triggering cell death and thereby exerting their anticancer action. The lack of a pattern in the chemopreventive response of the three studied commercial chemotherapeutic agents suggests that their mechanisms of action may interfere with the response to DNA damage in the presence of IR-01.
Cisplatin is also a chemotherapeutic agent that generates free radicals (Antunes and Bianchi, 2004), and this needs to be considered in its antitumor action. However, when this drug was combined with IR-01, the rate of DNA damage reduction increased, contrary to what was observed for doxorubicin and cyclophosphamide. Thus, despite the existence of the 4-aminoantipyrine radical, the antioxidant activity was undetectable, and this increase in DNA damage reduction could be attributed to the pharmacophore 1,4-dioxo-2-butenyl. However, further studies are needed to clarify this potentiation of the toxicogenic effects. A hypothesis for discussion is the reduction caused by cisplatin in the activity of antioxidant enzymes, such as superoxide dismutase, catalase, GSH peroxidase and GSH reductase (Hyppolito and Oliveira, 2005). Thus, the oxidative and consequently genotoxic capacity of cisplatin is more intense than that of the previously cited antineoplastic agents, which may have led to the increased genomic damage.
Regarding the micronucleus assay, the pattern of response to doxorubicin resembled that shown for the comet assay, i.e., the percent damage reduction decreased with higher dose, showing an inverse correlation. Note that over time the chemopreventive activity decreased, but the inversely proportional response pattern was maintained. This pattern was expected, and the decrease in the chemopreventive activity was perhaps due mainly to the metabolization and elimination of IR-01. Metabolization and secretion were also observed for doxorubicin because the capacity for DNA damage induction decreased.
For cisplatin, a high percentage of damage reduction was observed at 24 hours. However, the percentages decreased over the three time points. These data suggest that the increased DNA damage observed in the comet assay was not fixed into the genetic material because no chromosomal damage occurred. This finding, in turn, suggests that the repair mechanism was effective in preventing that genomic damage would be fixed as chromosomal damage.
For cyclophosphamide, increased toxicogenic activity was observed over the 72 hours of the study. It occurred at the intermediate test dose despite its efficient capacity to prevent genomic damage, with reduced chemopreventive activity at the other two doses.
In addition to the ability to reduce commercial chemotherapeutics effects, IR-01 also has a pharmacophoric radical in its structure, which could increase the antioxidant defenses in non-injured cells. This hypothesis is supported by Bianchi and Antunes (1999), Albertini and Ruiz (2001), Antunes and Bianchi (2004), and Oliveira et al. (2013), who reported that cells that have DNA lesions are deficient in antioxidant defenses. Therefore, these cells are more prone to suffer cytotoxic damage and undergo cell death more easily when exposed to certain cytotoxic and/or genotoxic agents. On the other hand, when normal cells with adequate antioxidant enzyme activity are in contact with another antioxidant agent, there is less of a chance that DNA damage is caused by free radicals.
The splenic phagocytosis test revealed that the same toxicogenetic doses also stimulated splenic phagocytosis. Thus, cells with DNA damage were efficiently removed from the bloodstream. Other studies have shonw that the spleen has the ability to remove tumor cells and/or DNA-damaged cells that are prone to carcinogenesis from the bloodstream (Cruvinel et al., 2010; Navarro et al., 2014; Carvalho et al., 2015). In addition, splenic phagocytosis is also associated with the biomonitoring of blood cell viability, and it promotes the removal of senescent leukocytes, platelets and erythrocytes (Freitas et al., 2009), as well as of apoptotic bodies and pathogens (Huysentruyt and Seyfried, 2010).
The splenic phagocytosis analysis also showed that all the chemotherapeutic agents were able to increase splenic activity, which was expected because these agents cause DNA damage and these damaged cells tend to be sequestered. When the agents were associated with IR-01, a decrease in phagocytosis occurred in all the experimental groups. This result suggests that the absence of cells with chromosomal damage did not stimulate the spleen to increase splenic phagocytosis.
Considering the alterations in both leukometry and splenic phagocytosis caused by IR-01, interestingly, it was observed that this molecule was able to increase the number of circulating lymphocytes and to cause a reduction in neutrophil numbers. These data suggest that the increase in phagocytic activity may be characterized by neutrophils exiting the blood and migrating into the spleen to sequester cells with DNA damage. Similar findings have been reported in other studies that also identified immunostimulatory compounds through this association (Lee et al., 2003; Leung et al., 2005; Ishii et al., 2011; Sang et al., 2013).
The combination of IR-01 with doxorubicin and cisplatin also showed an increase in the number of leukocytes and a reduction in the number of neutrophils. These findings corroborate the increase in splenic phagocytosis observed and discussed for the previous assay.
No variation in the frequency of blood cells was observed for cyclophosphamide. However, this does not contradict the splenic phagocytosis observed for the two lower doses. According to Oliveira et al. (2015a), splenic phagocytosis can occur efficiently even in the absence of a change in blood cell counts.
The present study also assessed cell death, given that unrepaired cells with DNA damage tend to disrupt the cell cycle and enter apoptosis (Zhou and Elledge, 2000). The results showed that IR-01 is capable of increasing the frequency of dead cells, from now on considered apoptotic cells, in the liver and kidneys. This is an important issue because IR-01 can stimulate splenic phagocytosis, increase the number of lymphocytes, and induce cell death, despite that it can cause genomic damage without causing chromosomal damage. Such important biological activities may be required in chemopreventive compounds (De Flora and Ferguson, 2005).
Nevertheless, although IR-01 is capable of increasing apoptosis when administered alone, in combination with doxorubicin, cisplatin and cyclophosphamide, IR-01 it generally reduced the frequency of apoptosis caused by these agents. The potentiation of apoptosis could be a good indicator of its adjuvant action in chemotherapy. However, such potentiation occurred only for doxorubicin. Therefore, the results do not encourage the use of IR-01 in combination with chemotherapeutic agents in anticancer therapy.
Given the above data, IR-01 has properties that render it sufficient to be classified as a chemopreventive agent, such as the inability of causing chromosomal damage, antigenotoxic potential, and the ability to alter leukometry, increase phagocytosis and induce cell death. These properties are possibly correlated with the 4-aminoantipyrine radical, an important antioxidant moiety present in the IR-01 molecule and described as having anti-inflammatory, analgesic, and antipyretic properties (Burdulene et al., 1999; Turan-Zitouni et al., 2001; Pisoschi and Pop, 2015).
According to Fedel-Miyasato et al. (2014) and Rocha et al. (2015) a good correlation exists between effective anti-inflammatory and immunostimulatory actions and chemopreventive effects. As reported in the literature, antioxidants that block carcinogenesis can exert their chemopreventive function via two different lines of organic defense: (I) by preventing the formation of free radicals that interact with and degrade DNA and (II) by intercepting existing or newly formed free radicals in cells, thereby causing a delay or inhibition of oxidation rates (Maxwell, 1995). The latter is correlated with the desmutagenic mode of action of substances reported as potential chemopreventive agents (Sato et al., 1984; Ferrara et al., 2000; Pesarini et al., 2013; Navarro et al., 2015).
If these lines of defense are still insufficient, the body may also facilitate the excretion of xenobiotics through detoxification enzymes, making them more water soluble and, thus, assisting their elimination by the kidneys (Cordon-Cardo et al., 1989; Hooiveld et al., 2001), while modulating the DNA repair system. This last line of defense is associated with bioantimutagenesis, in which enzymes are modulated by test compounds, thus favoring the correction and integrity of the genetic material, reducing the probability of developing cancer (Oliveira et al., 2009b; Nakamura et al., 1999; Di Giacomo et al., 2014; Leite et al., 2015).
All of the actions that are attributed to IR-01 discourage its use in combination with chemotherapeutic agents because of the maintenance of the antioxidant activity of its precursor, 4-aminoantipyrine. This use is discouraged because despite the fact that IR-01 can cause genomic damage, can increase splenic phagocytosis, lymphocyte number and the frequency of cell death, all these being properties required for chemotherapeutic agents, IR-01 can interfere negatively when associated with drugs already used extensively in anticancer therapy. Such interference prevents DNA damage and apoptosis, which are the main pathways for the elimination of tumor cells.
Thus, we consider that IR-01 is not indicated for use as an adjuvant in anticancer therapy in combination with doxorubicin, cisplatin or cyclophosphamide. In this case, the properties derived from 4-aminoantipyrine, even when in combination with the 1,4-dioxo-butenyl fragment, recognized as cytotoxic (Jha et al., 2010), largely overrode the ability to induce cell death. Corroborating this, the study by Berno et al. (2016) states that 4-aminoantipyrine, a dipyrone metabolite, reduces DNA damage, apoptosis induction and phagocytosi when administered in combination with doxorubicin, cisplatin or cyclophosphamide.
The present study is the first to propose a new synthetic methodology to efficiently and cleanly produce IR-01 and the first to demonstrate the chemopreventive effects of this molecule. In addition, we contraindicate the use of IR-01 as an adjuvant in anticancer therapies in combination with doxorubicin, cisplatin and cyclophosphamide because of its ability to reduce important effects of these agents.
Acknowledgments
This project was funded by Fundação de Apoio ao Desenvolvimento do Ensino, Ciência e Tecnologia do Estado de Mato Grosso do Sul (FUNDECT), Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES).
Footnotes
Associate Editor: Daisy Maria Fávero Salvadori
References
- Albertini SM, Ruiz MA. O papel da glutamina na terapia nutricional do transplante de medula óssea. Rev Bras Hematol Hemoter. 2001;23:41–47. [Google Scholar]
- Almeida VL, Leitão A, Reina LDCB, Montanari CA, Donnici CL, Lopes MTP. Cancer and cell cicle-specific and cell cicle nonspecific anticancer DNA-interactive agents: an introduction. Quim Nova. 2005;28:118–129. [Google Scholar]
- Ames BN, Durston WE, Yamasaki E, Lee FD. Carcinogens are mutagens: A simple test system combining liver homogenates for activation and bacteria for detection. Proc Natl Acad Sci U S A. 1973;70:2281–2285. doi: 10.1073/pnas.70.8.2281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Antunes LMG, Bianchi MLP. Dietary antioxidants as inhibitors of cisplatin-induced nephrotoxicity. Rev Nutr. 2004;17:89–96. [Google Scholar]
- Berno CR, Rós BT, da Silveira IO, Coelho HR, Antoniolli AC, Beatriz A, de Lima DP, Monreal AC, Sousa FG, da Silva Gomes R, et al. 4-Aminoantipyrine reduces toxic and genotoxic effects of doxorubicin, cisplatin, and cyclophosphamide in male mice. Mutat Res Genet Toxicol Environ Mutagen. 2016;805:19–24. doi: 10.1016/j.mrgentox.2016.05.009. [DOI] [PubMed] [Google Scholar]
- Bianchi MLP, Antunes LMG. Radicais livres e os principais antioxidantes da dieta. Rev Nutr. 1999;12:123–130. [Google Scholar]
- Burdulene D, Palaima A, Stumbryavichyute Z, Talaikite Z. Synthesis and antiinflammatory activity of 4-aminoantipyrine derivatives of succinamides. Pharm Chem J. 1999;33:191–193. [Google Scholar]
- Cao M, Onyango EO, Williams CR, Royce DB, Gribble GW, Sporn MB, Liby KT. Novel synthetic pyridyl analogues of CDDO-Imidazolide are useful new tools in cancer prevention. Pharmacol Res. 2015;100:135–147. doi: 10.1016/j.phrs.2015.07.024. [DOI] [PubMed] [Google Scholar]
- Carvalho PC, Santos EA, Schneider BU, Matuo R, Pesarini JR, Cunha-Laura AL, Monreal AC, Lima DP, Antoniolli AC, Oliveira RJ. Diaryl sulfide analogs of combretastatin A-4: Toxicogenetic, immunomodulatory and apoptotic evaluations and prospects for use as a new chemotherapeutic drug. Environ Toxicol Pharmacol. 2015;40:715–721. doi: 10.1016/j.etap.2015.08.028. [DOI] [PubMed] [Google Scholar]
- Cordon-Cardo C, O’Brien JP, Casals D, Rittman-Grauer L, Biedler JL, Melamed MR, Bertino JR. Multidrug-resistance gene (P-glycoprotein) is expressed by endothelial cells at blood-brain barrier sites. Proc Natl Acad Sci U S A. 1989;86:695–698. doi: 10.1073/pnas.86.2.695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cruvinel WM, Mesquita D, Jr, Araújo JAP, Catelan TT, de Souza AW, da Silva NP, Andrade LE. Immune system: Part I. Fundamentals of innate immunity with emphasis on molecular and cellular mechanisms of inflammatory response. Rev Bras Reumatol. 2010;50:434–461. [PubMed] [Google Scholar]
- Cunha S, Oliveira SH, Rodrigues MT, Jr, Bastos RM, Ferrari J, de Oliveira CMA, Kato L, Napolitano HB, Vencato I, Lariucci C. Structural studies of 4-aminoantipyrine derivatives. J Mol Struct. 2005;752:32–39. [Google Scholar]
- de Araújo FH, de Figueiredo D, Auharek S, Pesarini JR, Meza A, Gomes R, Monreal ACD, de Lima D, Antoniolli-Silva ACMB, Kassuya CA, et al. In vivo chemotherapeutic insight of a novel isocoumarin (3-hexyl-5,7-dimethoxy-isochromen-1-one): Genotoxicity, cell death induction, leukometry and phagocytic evaluation. Genet Mol Biol. 2017;40:665–675. doi: 10.1590/1678-4685-GMB-2016-0316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Flora S, Ferguson LR. Overview of mechanisms of cancer chemopreventive agents. Mutat Res. 2005;591:8–15. doi: 10.1016/j.mrfmmm.2005.02.029. [DOI] [PubMed] [Google Scholar]
- Di Giacomo S, Mazzanti G, Sarpietro MG, Di Sotto A. α-Hexylcinnamaldehyde inhibits the genotoxicity of environmental pollutants in the bacterial reverse mutation assay. J Nat Prod. 2014;77:2664–2670. doi: 10.1021/np500567d. [DOI] [PubMed] [Google Scholar]
- Elsendoorn TJ, Weijl NI, Mithoe S, Zwinderman AH, Van Dam F, De Zwart FA, Tates AD, Osanto S. Chemotherapy induced chromosomal damage in peripheral blood lymphocytes of cancer patients supplemented with antioxidants or placebo. Mutat Res. 2001;498:145–158. doi: 10.1016/s1383-5718(01)00278-9. [DOI] [PubMed] [Google Scholar]
- Fedel-Miyasato LES, Formagio ASN, Auharek SA, Kassuya CA, Navarro SD, Cunha-Laura AL, Monreal AC, Vieira MC, Oliveira RJ. Antigenotoxic and antimutagenic effects of Schinus terebinthifolius Raddi in Allium cepa and Swiss mice: A comparative study. Genet Mol Res. 2014;13:3411–3425. doi: 10.4238/2014.April.30.2. [DOI] [PubMed] [Google Scholar]
- Ferrara G, Loffredo E, Simeone R, Senesi N. Evaluation of antimutagenic and desmutagenic effects of humic and fulvic acids on root tips of Vicia faba . Environ Toxicol. 2000;15:513–517. [Google Scholar]
- Freitas SH, Evêncio JN, Dória RGS, Mendonça FS, Simões MJ, Camargo LM, Sebe AA. Morphologic, morphometric, and ultrastructural aspects of the spleen of rats after hepatic pedicle total clamping. Arq Bras Med Vet Zootec. 2009;61:1314–1321. [Google Scholar]
- Friedman M, Rasooly R. Review of inhibition of biological activities of food-related. Toxins. 2013;5:743–775. doi: 10.3390/toxins5040743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frolova TS, Kukina TP, Sinitsyna OI. Genotoxic and mutagenic properties of synthetic betulinic acid and betulonic acid. Bioorg Khim. 2015;41:462–467. doi: 10.1134/s1068162015040056. [DOI] [PubMed] [Google Scholar]
- Hayashi M, Morita T, Kodama Y, Sofuny T, Ishidate M., Jr. The micronucleus assay with mouse peripheral blood reticulocytes using acridine orange-coated slides. Mutat Res. 1990;245:245–249. doi: 10.1016/0165-7992(90)90153-b. [DOI] [PubMed] [Google Scholar]
- Hedenmaln K, Spigset O. Agranulocytosis and other blood dyscrasias associated with dipyrone (metamizol) Eur J Clin Pharmacol. 2002;58:265–274. doi: 10.1007/s00228-002-0465-2. [DOI] [PubMed] [Google Scholar]
- Hooiveld GJEJ, Vam-Montfoort JE, Meijer DKF, Müller M. Function and regulation of ATP-binding cassette transport proteins involved in hepatobiliary transport. Eur J Pharm Sci. 2001;12:525–543. doi: 10.1016/s0928-0987(01)00101-4. [DOI] [PubMed] [Google Scholar]
- Hoshina MM, Santos LD, Palma MS. Cytotoxic, genotoxic/antigenotoxic and mutagenic/antimutagenic effects of the venom of the wasp Polybia paulista . Toxicon. 2013;72:64–70. doi: 10.1016/j.toxicon.2013.06.007. [DOI] [PubMed] [Google Scholar]
- Huysentruyt LC, Seyfried TN. Perspectives on the mesenchymal origin of metastatic cancer. Cancer Metastasis Rev. 2010;29:695–707. doi: 10.1007/s10555-010-9254-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hyppolito MA, Oliveira JÁA. Ototoxycity, otoprotection and self defense of the coclear outer hair cells. Medicina (Ribeirão Preto) 2005;38:279–289. [Google Scholar]
- Ishii PL, Prado CK, Mauro M de O, Carreira CM, Mantovani MS, Ribeiro LR, Dichi JB, Oliveira RJ. Evaluation of Agaricus blazei in vivo for antigenotoxic, anticarcinogenic, phagocytic and immunomodulatory activities. Regul Toxicol Pharmacol. 2011;59:412–422. doi: 10.1016/j.yrtph.2011.01.004. [DOI] [PubMed] [Google Scholar]
- Jha A, Mukherjee C, Prasad AK, Parmar VS, Vadaparti M, Das U, De Clercq E, Balzarini J, Stables JP, Shrivastav A, et al. Derivatives of aryl amines containing the cytotoxic 1,4-dioxo-2-butenyl pharmacophore. Bioorg Med Chem Lett. 2010;20:1510–1515. doi: 10.1016/j.bmcl.2010.01.098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim ES, Khuri FR, Hong WK. Bertino JR. Encyclopedia of Cancer. Academic Press; Orlando: 2002. Chemoprevention trials; pp. 457–472. [Google Scholar]
- Kobayashi H, Sugiyama C, Morikawa Y, Hayashi M, Sofuny T. A comparison between manual microscopic analysis and computerized image analysis in the single cell gel electrophoresis assay. MMS Commun. 1995;3:103–115. [Google Scholar]
- Lee YS, Han OK, Park CW, Suh SI, Shin SW, Yang CH, Jeon TW, Lee ES, Kim KJ, Kim SH, et al. Immunomodulatory effects of aqueous-extracted Astragali radix in methotrexate-treated mouse spleen cells. J Ethnopharmacol. 2003;84:193–198. doi: 10.1016/s0378-8741(02)00298-2. [DOI] [PubMed] [Google Scholar]
- Leite AS, Dantas AF, Oliveira GL, Gomes AL, Júnior, de Lima SG, Citó AM, de Freitas RM, Melo-Cavalcante AA, Dantas Lopes JA. Evaluation of toxic, cytotoxic, mutagenic, and antimutagenic activities of natural and technical cashew nut shell liquids using the Allium cepa and Artemia salina bioassays. Biomed Res Int. 2015;2015:626835. doi: 10.1155/2015/626835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leung KN, Leung PY, Kong LP, Leung PK. Immunomodulatory effects of esculetin (6,7-dihydroxycoumarin) on murine lymphocytes and peritoneal macrophages. Cell Mol Immunol. 2005;2:181–188. [PubMed] [Google Scholar]
- Luo X, Yu X, Liu S, Deng Q, Liu X, Peng S, Li H, Liu J, Cao Y. The role of targeting kinase activity by natural products in cancer chemoprevention and chemotherapy. Oncol Rep. 2015;34:547–554. doi: 10.3892/or.2015.4029. [DOI] [PubMed] [Google Scholar]
- Manoharan K, Banerjee MR. Beta-carotene reduces sister chromatid exchanges induced by chemical carcinogens in mouse mammary cells in organ culture. Cell Biol Int Rep. 1985;9:783–789. doi: 10.1016/0309-1651(85)90096-7. [DOI] [PubMed] [Google Scholar]
- Mauro MO, Pesarini JR, Ishii PL, da Silva AF, Oliveira RJ. Chemopreventive activity of phenylalanine against damage mutagenic prompted by the acute administration of cyclophosphamide in pregnant and non-pregnant mice using the micronucleus test. Rev Bras Farmacogn. 2010;20:334–339. [Google Scholar]
- Mauro MO, Sartori D, Oliveira RJ, Ishii PL, Mantovani MS, Ribeiro LR. Activity of selenium on cell proliferation, cytotoxicity, and apoptosis and on the expression of CASP9, BCL-XL and APC in intestinal adenocarcinoma cells. Mutat Res. 2011;715:7–12. doi: 10.1016/j.mrfmmm.2011.06.015. [DOI] [PubMed] [Google Scholar]
- Maxwell SR. Prospects for the use of antioxidant therapies. Drugs. 1995;49:345–361. doi: 10.2165/00003495-199549030-00003. [DOI] [PubMed] [Google Scholar]
- Mendanha da Cunha CR, Mendanha SA, Neto, Carlos SC, Cortez AP, Gomes MN, Martins FI, Alonso A, Rezende KR, Menegatti R, de Magalhães MT, et al. 4-Nerolidylcatechol and its synthetic analogues: Antioxidant activity and toxicity evaluation. Eur J Med Chem. 2013;2013 62:371–378. doi: 10.1016/j.ejmech.2012.12.028. [DOI] [PubMed] [Google Scholar]
- Nadin SB, Vargas-Roig LM, Drago G, Ibarra J, Ciocca DR. DNA damage and repair in peripheral blood lymphocytes from health individuals and cancer patients: A pilot study on the implications in the clinical response to chemotherapy. Cancer Lett. 2005;21:11–14. doi: 10.1016/j.canlet.2005.07.025. [DOI] [PubMed] [Google Scholar]
- Nakamura Y, Suganuma E, Matsuo T, Okamoto S, Sato K, Ohtsuki K. 2,4-Nonadienal and benzaldehyde bioantimutagens in Fushimi Sweet Pepper (Fushimi-Togarashi) J Agric Food Chem. 1999;47:544–549. doi: 10.1021/jf980653n. [DOI] [PubMed] [Google Scholar]
- Navarro SD, Beatriz A, Meza A, Pesarini JR, Gomes RS, Karaziack CB, Cunha-Laura AL, Monreal AC, Romão W, Lacerda V, Júnior, et al. A new synthetic resorcinolic lipid 3-heptyl-3,4,6-trimethoxy-3H-isobenzofuran-1-one: Evaluation of toxicology and ability to potentiate the mutagenic and apoptotic effects of cyclophosphamide. Eur J Med Chem. 2014;75:132–142. doi: 10.1016/j.ejmech.2014.01.057. [DOI] [PubMed] [Google Scholar]
- Navarro SD, Mauro MO, Pesarini JR, Ogo FM, Oliveira RJ. Resistant starch: A functional food that prevents DNA damage and chemical carcinogenesis. Genet Mol Res. 2015;14:1679–1691. doi: 10.4238/2015.March.6.14. [DOI] [PubMed] [Google Scholar]
- Oliveira RJ, Salles MJ, Da Silva AF, Kanno TY, Lourenço AC, Freiria GA, Matiazi HJ, Ribeiro LR, Mantovani MS. Effects of the polysaccharide b-glucan on clastogenicity and teratogenicity caused by acute exposure to cyclophosphamide in mice. Regul Toxicol Pharmacol. 2009a;53:164–173. doi: 10.1016/j.yrtph.2008.12.007. [DOI] [PubMed] [Google Scholar]
- Oliveira RJ, Baise E, Mauro MO, Pesarini JR, Matuo R, Silva AF, Ribeiro LR, Mantovani MS. Evaluation of chemopreventive activity of glutamine by the comet and the micronucleus assay in mice’s peripheral blood. Environ Toxicol Pharmacol. 2009b;28:120–124. doi: 10.1016/j.etap.2009.03.008. [DOI] [PubMed] [Google Scholar]
- Oliveira RJ, Sassaki ES, Monreal AC, Monreal MT, Pesarini JR, Mauro MO, Matuo R, Silva AF, Zobiole NN, Siqueira JM, et al. Pre-treatment with glutamine reduces genetic damage due to cancer treatment with cisplatin. Genet Mol Res. 2013;12:6040–6051. doi: 10.4238/2013.December.2.2. [DOI] [PubMed] [Google Scholar]
- Oliveira RJ, Navarro SD, Lima DP, Mauro MO, da Silva AF, Souza TR, Ribeiro LR. A novel cytosporone 3-Heptyl-4,6-dihydroxy-3H-isobenzofuran-1-one: Synthesis; toxicological, apoptotic and immunomodulatory properties; and potentiation of mutagenic damage. BMC Cancer. 2015a;15:561. doi: 10.1186/s12885-015-1532-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oliveira RJ, Mantovani MS, Pesarini JR, Mauro MO, da Silva AF, Souza TR, Ribeiro LR. 6-Dimethylaminopurine and cyclohexamide are mutagenic and alter reproductive performance and intrauterine development in vivo. Genet Mol Res. 2015b;14:834–849. doi: 10.4238/2015.February.2.8. [DOI] [PubMed] [Google Scholar]
- Pathak SK, Sharma RA, Mellon JK. Chemoprevention of prostate cancer by diet-derived antioxidant agents and hormonal manipulation. Int J Oncol. 2003;22:5–13. [PubMed] [Google Scholar]
- Pesarini JR, Zaninetti PT, Mauro MO, Carreira CM, Dichi JB, Ribeiro LR, Mantovani MS, Oliveira RJ. Antimutagenic and anticarcinogenic effects of wheat bran in vivo. Genet Mol Res. 2013;12:1646–1659. doi: 10.4238/2013.May.14.5. [DOI] [PubMed] [Google Scholar]
- Pesarini JR, Victorelli SG, Vicentini AP, Ferreira LK, Mauro MO, Matuo R, Oliveira JR, Antoniolli AC, Mantovani MS, Oliveira RJ. Antigenotoxic and antimutagenic effects of glutamine supplementation on mice treated with cisplatin. Genet Mol Res. 2014;13:4820–4830. doi: 10.4238/2014.July.2.11. [DOI] [PubMed] [Google Scholar]
- Pisoschi AM, Pop A. The role of antioxidants in the chemistry of oxidative stress: A Review. Eur J Med Chem. 2015;15:55–74. doi: 10.1016/j.ejmech.2015.04.040. [DOI] [PubMed] [Google Scholar]
- Rocha RS, Kassuya CAL, Formagio ASN, Mauro MO, Andrade-Silva M, Monreal AC, Cunha-Laura AL, Vieira MC, Oliveira RJ. Analysis of the anti-inflammatory and chemopreventive potential and description of the antimutagenic mode of action of the Annona crassiflora methanolic extract. Pharm Biol. 2015;54:35–47. doi: 10.3109/13880209.2015.1014567. [DOI] [PubMed] [Google Scholar]
- Rodeiro I, Cancino L, González JE. Evaluation of the genotoxic potential of Mangifera indica L. extract (Vimang), a new natural product with antioxidant activity. Food Chem Toxicol. 2006;44:1707–1713. doi: 10.1016/j.fct.2006.05.009. [DOI] [PubMed] [Google Scholar]
- Rundell MS, Wagner ED, Plewa MJ. The comet assay: Genotoxic damage or nuclear fragmentation? Environ Mol Mutagen. 2003;42:61–67. doi: 10.1002/em.10175. [DOI] [PubMed] [Google Scholar]
- Salgado P, Suarez-de-La-Rica A, Maseda E, Maggi G, Hernández-Gancedo C, Lopez-Tofiño A, Palacios E, Ruiz E, Gilsanz F. Severe Mucor necrotizing fasciitis associated to dipyrone-induced agranulocytosis. Rev Esp Quimioter. 2015;28:58–60. [PubMed] [Google Scholar]
- Sang X, Fei M, Sheng L, Zhao X, Yu X, Hong J, Ze Y, Gui S, Sun Q, Ze X, et al. Immunomodulatory effects in the spleen-injured mice following exposure to titanium dioxide nanoparticles. J Biomed Mater Res A. 2013;102:3562–3572. doi: 10.1002/jbm.a.35034. [DOI] [PubMed] [Google Scholar]
- Sato T, Suzuki Y, Ose Y, Ishikawa T. Desmutagenic substance in water extract of grass-wrack pondweed (Potamogeton oxyphylus Miquel) Mutat Res. 1984;129:33–38. doi: 10.1016/0027-5107(84)90120-9. [DOI] [PubMed] [Google Scholar]
- Singh NP, McCoy MT, Tice RR, Schneider EL. A simple technique for quantitation of low levels of DNA damage in individual cells. Exp Cell Res. 1988;175:184–191. doi: 10.1016/0014-4827(88)90265-0. [DOI] [PubMed] [Google Scholar]
- Steward WP, Brown K. Cancer chemoprevention: A rapidly evolving field. Br J Cancer. 2013;109:1–7. doi: 10.1038/bjc.2013.280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Synder D, Gillies PJ. Evaluation of the clastogenic DNA intercalative and topoisomerase II interactive properties of bioflavonoids in Chinese hamster V79 cells. Environ Mol Mutagen. 2002;40:266–276. doi: 10.1002/em.10121. [DOI] [PubMed] [Google Scholar]
- Turan-Zitouni G, Sivaci M, Kiliç FS, Erol K. Synthesis of some triazolyl-antipyrine derivatives and investigation of analgesic activity. Eur J Med Chem Chemistry. 2001;36:685–689. doi: 10.1016/s0223-5234(01)01252-1. [DOI] [PubMed] [Google Scholar]
- Vaz MS, Vaz da Silva MS, Oliveira RJ, da Silva Mota J, Brait DR, de Carvalho LN, Vani JM, Berno CR, Araújo FH, de Barros ME. Evaluation of the toxicokinetics and apoptotic potential of ethanol extract from Echinodorus macrophyllus leaves in vivo . Regul Toxicol Pharmacol. 2016;82:32–38. doi: 10.1016/j.yrtph.2016.10.017. [DOI] [PubMed] [Google Scholar]
- Waters MD, Brady AL, Stack HF, Brockman HE. Antimutagenicity profiles for some model compounds. Mutat Res. 1990;238:57–85. doi: 10.1016/0165-1110(90)90039-e. [DOI] [PubMed] [Google Scholar]
- Zhan Y, Du X, Chen H, Liu J, Zhao B, Huang D, Li G, Xu Q, Zhang M, Weimer BC, et al. Cytosporone B is an agonist for nuclear orphan receptor Nur77. Nat Chem Biol. 2008;4:548–556. doi: 10.1038/nchembio.106. [DOI] [PubMed] [Google Scholar]
- Zhou BB, Elledge SJ. The DNA damage response: putting checkpoints in perspective. Nature. 2000;408:433–439. doi: 10.1038/35044005. [DOI] [PubMed] [Google Scholar]