Abstract
Flowering time regulation has significant effects on the agricultural and horticultural industries. Plants respond to changing environments and produce appropriate floral inducers (florigens) or inhibitors (anti-florigens) that determine flowering time. Recent studies have demonstrated that members of two homologous proteins, FLOWERING LOCUS T (FT) and TERMINAL FLOWER 1 (TFL1), act as florigen and anti-florigen, respectively. Studies in diverse plant species have revealed universal but diverse roles of the FT/TFL1 gene family in many developmental processes. Recent studies in several crop species have revealed that modification of flowering responses, either due to mutations in the florigen/anti-florigen gene itself, or by modulation of the regulatory pathway, is crucial for crop domestication. The FT/TFL1 gene family could be an important potential breeding target in many crop species.
Keywords: anti-florigen, chrysanthemum, florigen, FLOWERING LOCUS T (FT), photoperiod, TERMINAL FLOWER 1 (TFL1)
Introduction
Many plants utilize fluctuations in day-length (photoperiod) as the most reliable indicator of seasonal progression to determine when to initiate flowering. This phenomenon, called photoperiodism, enables plants to set seeds at favorable conditions and maximize their chance of survival. Photoperiodism was first described in detail by Garner and Allard (1920). They investigated the flowering response of a late-flowering tobacco cultivar ‘Maryland Mammoth’ and a soybean cultivar ‘Biloxi’, and found that these plants flower in response to changes in day-length, not light intensity, temperature, or nutrient availability. They categorized plants into three types based on their photoperiodic responses; short-day plants (SDPs), long-day plants (LDPs), and day-neutral plants (DNPs). Flowering in SDPs occurs or is accelerated when the night length is greater than a critical minimum, whereas flowering in LDP occurs or is promoted when the day becomes longer, and DNPs flower regardless of day-length. Chailakhyan (1936) proposed the concept of the flowering hormone “florigen”, which is synthesized in the leaves and transmitted to the shoot apex to induce flowering. Recent molecular genetics approaches have demonstrated that homologs of the FLOWERING LOCUS T (FT) protein of Arabidopsis act as florigens in several plant species (Corbesier et al. 2007, Lifschitz et al. 2006, Lin et al. 2007, Tamaki et al. 2007). In Arabidopsis, the FT protein is induced under flower-inductive long day (LD) photoperiod in leaves, whereas it forms a complex with a bZIP type transcription factor FD at the shoot apical meristem (SAM) to induce floral meristem-identity genes, such as APETALA1 (AP1) and FRUITFULL (FUL) (Abe et al. 2005, Kardailsky et al. 1999, Kobayashi et al. 1999, Wigge et al. 2005). The long-distance transmission of the FT protein and its rice homolog Heading date 3a (Hd3a) from the leaves to the shoot apex via the phloem was further determined (Corbesier et al. 2007, Tamaki et al. 2007). The FT/Hd3a family protein acts as the universal flowering hormone “florigen” in many plant species (Matsoukas 2015, Wickland and Hanzawa 2015) (Table 1).
Table 1.
List of FT/TFL1 family genes in representative species
| Species | Gene name | Effect on flowering | Other function | Regulatory input | Site of expression | References |
|---|---|---|---|---|---|---|
| Arabidopsis | FT | Induction | LD | Leaf |
Bradley et al. 1997 Kobayashi et al. 1999 Kardailsky et al. 1999 Yamaguchi et al. 2005 Yoo et al. 2010 Xi et al. 2010 Huang et al. 2012 Ryu et al. 2014 |
|
| TSF | Induction | LD | Leaf | |||
| TFL1 | Repression | Inflorescence development | Shoot apex Root |
|||
| BFT | Repression | Inflorescence development | LD, Salinity stress | Leaf | ||
| ATC | Repression | SD | Vascular tissue | |||
| MFT | Induction | Seed germination | Seed | |||
| Rice | Hd3a | Induction | SD | Leaf |
Kojima et al. 2002 Izawa et al. 2002 Nakagawa et al. 2002 Tamaki et al. 2007 Komiya et al. 2009 |
|
| RFT1 | Induction | LD | Leaf | |||
| RCN1 | Repression | Shoot apex | ||||
| RCN2 | Repression | Shoot apex | ||||
| Chrysanthemum | CsFTL1 | Induction | LD, NB | Leaf |
Oda et al. 2012 Higuchi et al. 2013 Nakano et al. 2013 Higuchi and Hisamatsu 2015 Nakano et al. 2015a Sun et al. 2017 |
|
| CsFTL2 | Induction | LD, Sucrose | Leaf | |||
| CsFTL3 | Induction | SD, Heat | Leaf | |||
| CsTFL1 | Repression | Shoot apex Root |
||||
| CsAFT | Repression | LD, NB | Leaf | |||
| Strawberry | FvTFL1 | Repression | LD, Cool temperature | Shoot apex |
Koskela et al. 2012 Mouhu et al. 2013 Nakano et al. 2015b Rantanen et al. 2015 Koskela et al. 2016 |
|
| FvFT1 | Induction | LD | Leaf | |||
| FaFT3 | – | SD | Shoot apex | |||
| Tomato | SFT | Induction | Matured leaf |
Lifschitz et al. 2006 Shalit et al. 2009 Cao et al. 2016 Soyk et al. 2017 |
||
| SP | Repression | Inflorescence development | Young leaf, Shoot apex | |||
| SP5G | Repression | LD | Cotyledon, Leaf | |||
| SP5G2 | Repression | SD | Cotyledon, Leaf | |||
| SP5G3 | Repression | SD | Cotyledon, Leaf | |||
| Sugar beet | BvFT1 | Repression | SD, Vernalization | Leaf | Pin et al. 2010 | |
| BvFT2 | Induction | LD, Vernalization | Leaf | |||
| Rose | RoKSN | Repression | Shoot apex |
Iwata et al. 2012 Randoux et al. 2013 Otagaki et al. 2015 |
||
| RoFT | Induction | Shoot (reproductive) | ||||
| Morning glory | PnFT1 | Induction | SD | Cotyledon, Leaf |
Hayama et al. 2007 Wada et al. 2010 |
|
| PnFT2 | – | SD, Stress | Cotyledon, Leaf | |||
| Sunflower | HaFT1 | Repression | Shoot apex | Blackman et al. 2010 | ||
| HaFT4 | Induction | LD | Leaf | |||
| Potato | StSP3D | Induction | Leaf |
Navarro et al. 2011 Abelenda et al. 2016 |
||
| StSP6A | Induction | Induction of tuberization | SD | Leaf, Stolon | ||
| StSP5G | Repression | Inhibition of tuberization | LD | Leaf | ||
| Onion | AcFT2 | Induction | Vernalization | Central bud Leaf |
Lee et al. 2013 | |
| AcFT1 | Induction | Induction of bulb formation | LD | Leaf | ||
| AcFT4 | Repression | Inhibition of bulb formation | SD | Leaf |
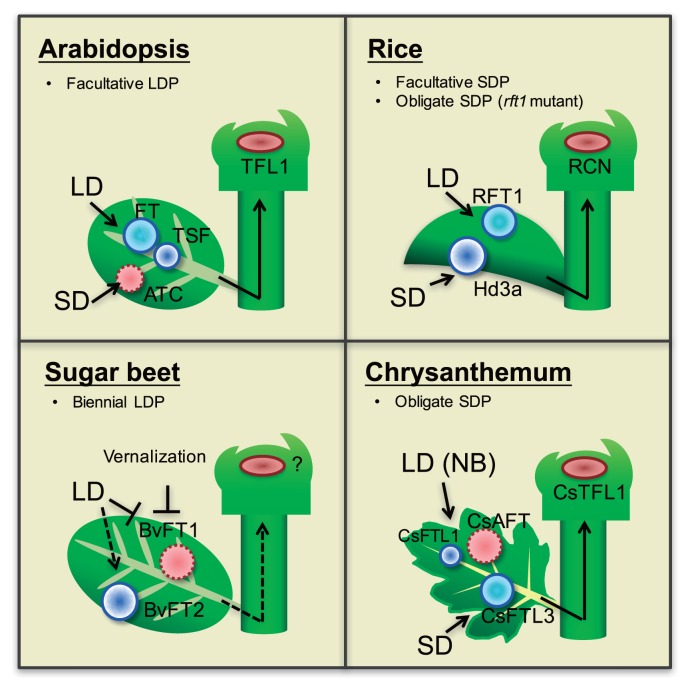
In addition to the floral inducer florigen, the systemic floral inhibitor produced in non-induced leaves inhibit flowering. The concept of a floral repressor (anti-florigen) was proposed almost at the same time with that of the florigen (Lang and Melchers 1943). Many physiological observations in Hyoscyamus, strawberry, Lolium, chrysanthemum, tobacco, and Pharbitis suggested the existence of the systemic floral inhibitor (Evans 1960, Guttridge 1959, Lang and Melchers 1943, Lang et al. 1977, Ogawa and King 1990, Tanaka 1967). A grafting experiment in tobacco plants with different photoperiodic responses strongly supported this hypothesis; a floral inhibitor produced in the leaves of LD tobacco under non-inductive short day (SD) systemically inhibited the flowering of the grafted day-neutral (DN) tobacco plants (Lang et al. 1977). Since the 1990s, molecular genetic studies in Arabidopsis revealed that the TERMINAL FLOWER 1 (TFL1), a member of the phosphatidylethanolamine-binding protein (PEBP) family protein, acts to suppress flowering (Bradley et al. 1997). The TFL1 is expressed in the SAM and maintains an indeterminate inflorescence (Conti and Bradley 2007, Jaeger et al. 2013, Ratcliffe et al. 1999). TFL1 also forms a complex with FD, an interacting partner of FT, and acts to suppress flowering by antagonizing the florigenic activity of the FT-FD complex (Abe et al. 2005). Although the TFL1 acts as a floral inhibitor, it only moves over short distances within the meristematic zone (Conti and Bradley 2007). In Arabidopsis, it was reported that another TFL1-like gene, Arabidopsis thaliana CENTRORADIALIS homolog (ATC), is expressed in the vasculature tissues under non-inductive SD photoperiod, and acts systemically to suppress flowering (Huang et al. 2012). In sugar beet (Beta vulgaris), two FT homologs (BvFT1 and BvFT2) with antagonistic function have been reported (Pin et al. 2010). BvFT1 suppresses flowering under SDs and before vernalization by repressing the expression of the floral promoter BvFT2. A recent study in a wild diploid chrysanthemum (Chrysanthemum seticuspe) identified a floral inhibitor, Anti-florigenic FT/TFL1 family protein (CsAFT), which moves over long distances (Higuchi et al. 2013). The CsAFT was induced in leaves under non-inductive LD or night-break (NB) photoperiods. CsAFT proteins move long distances from leaves to the shoot apex, and inhibit flowering by directly antagonizing the florigen complex activity. These findings suggest that the balance between floral inducers (florigens) and inhibitors (anti-florigens) determine flowering time variations in many plant species (Fig. 1).
Fig. 1.
Flowering time regulation by florigen and anti-florigen in Arabidopsis, rice, sugar beet, and chrysanthemums. The blue (solid) circles indicate systemic floral inducers, while red (dotted) circles indicate systemic floral inhibitors. TFL1 homologs suppress flowering at the shoot apex.
Molecular mechanisms of the FT/TFL1 function
The FT/TFL1 gene encodes a small protein similar to the PEBP. In Arabidopsis, there are six members of the PEBP gene family. FT and TWIN SISTER OF FT (TSF) act as floral activators, whereas TFL1, ATC, and BROTHER OF FT AND TFL1 ( BFT) act as floral repressors (Bradley et al. 1997, Kardailsky et al. 1999, Kobayashi et al. 1999, Mimida et al. 2001, Yamaguchi et al. 2005, Yoo et al. 2010). The MOTHER OF FT AND TFL1 (MFT) has a weak floral-inducer activity and is involved in seed germination (Xi et al. 2010, Yoo et al. 2004). FT/TFL1-like proteins control flowering probably through transcriptional regulation of target genes, because these proteins form a complex with a transcription factor FD (Abe et al. 2005, Hanano and Goto 2011, Huang et al. 2012, Jang et al. 2009, Ryu et al. 2014, Wigge et al. 2005). In rice, the Hd3a forms a complex with the 14-3-3 adaptor proteins and OsFD1, known as the florigen activation complex (FAC), and then induces a rice AP1 homolog OsMADS15 (Taoka et al. 2011). Plant PEBP family proteins can be classified into three major clades, FT-like, TFL1-like, and MFT-like. FT-like and TFL1-like proteins have similar structures, but they have opposing roles in regulating flowering. A critical region or amino acid residues that convert a floral inducer into a repressor have been identified previously. In Arabidopsis, the segment B in the fourth exon encoding an external loop structure of PEBP is particularly important (Ahn et al. 2006). Specific mutations at Tyr-85, Glu-109, Tyr-134, Trp-138, Gln-140, and Asn-152 residues could convert FT into a TFL1-like repressor (Ahn et al. 2006, Hanzawa et al. 2005, Ho and Weigel 2014). In sugar beet, substitution of 3 amino acid residues in the external loop of BvFT1 (including Tyr-134 and Trp-138) was sufficient to convert its repressing activity to a promoting activity (Pin et al. 2010). The surface charges of FT and TFL1 are thought to be critical for recruitment of yet unidentified transcriptional coactivators or corepressors (Ho and Weigel 2014).
Photoperiodic flowering in Arabidopsis and rice
In Arabidopsis, induction of FT under LD photoperiod is dependent on the interaction of the endogenous biological clock and the external light inputs (Golembeski and Imaizumi 2015). The circadian rhythm entrained by light/dark cycles sets the expression of CONSTANS (CO), a positive regulator of FT, to occur in the evening. When high CO expression coincides with the light signal perceived by photoreceptors, a CO protein is stabilized and it induces FT (Valverde et al. 2004, Yanovsky and Kay 2002). In rice, a facultative SDP, expression of a CO homolog, Heading date 1 ( Hd1), is regulated by a circadian clock peaking in the evening. The coincidence of Hd1 with the phytochrome signal under LD evenings suppresses flowering by negatively regulating the expression of Hd3a (Hayama et al. 2003, Izawa et al. 2002). Rice contains unique pathways that function independent of CO ( Hd1). Early heading date 1 (Ehd1), encoding a B-type response regulator, promotes flowering by up-regulating Hd3a expression independent of Hd1 (Doi et al. 2004). Grain number, plant height, and heading date 7 (Ghd7), a CCT domain protein, is induced under LDs and suppresses flowering by down-regulating Ehd1 expression (Xue et al. 2008). Interestingly, induction of both Ehd1 and Ghd7 by light is limited to a specific time of day (the photo-sensitive phase or the “gate”) by a circadian clock action. The gate for Ehd1 induction always opens around dawn, but the gate for Ghd7 induction with red light opens at different times depending on day length. Acute induction of Hd3a in response to critical day-length is achieved by the interaction of these two gating mechanisms (Itoh et al. 2010). In addition to Hd3a, rice has another florigen gene, RICE FLOWERING LOCUS T1 ( RFT1), that functions under LD photoperiods (Komiya et al. 2009). Loss-of-function of RFT1 results in extremely late flowering under LD, which is similar to the flowering response of absolute SDPs (Ogiso-Tanaka et al. 2013) (Fig. 1).
Flowering time regulation in chrysanthemum
Chrysanthemum (C. morifolium) is one of the most important floricultural crops around the world. The autumn-flowering chrysanthemum cultivars are categorized as absolute SDP that require repeated SD photoperiod for successful flowering, but their vegetative growth can be strictly maintained under LD or NB conditions. Soon after the discovery of photoperiodism by Garner and Allard, methods for day-length manipulation to control chrysanthemum flowering were established (Laurie 1930, Poesch 1936, Post 1931). Chrysanthemum growers use blackouts or artificial lighting (day-length extension or NB) to meet the demand for marketable flowers throughout the year. Thus, chrysanthemum is the most successful example of the use of day-length manipulation to benefit commercial crop production. Although the light sensitive flowering behavior made this plant a major floricultural crop, molecular mechanisms of photoperiodic flowering in chrysanthemums is largely unknown. The cultivated chrysanthemums are complex hybrids derived from several different species (Klie et al. 2014). The complex hybridity and polyploidy in this species make it difficult to conduct molecular-genetic modifications. To overcome this issue, a wild diploid species C. seticuspe has recently been used as an alternative model of chrysanthemum cultivars.
FT/TFL1-like genes in chrysanthemum
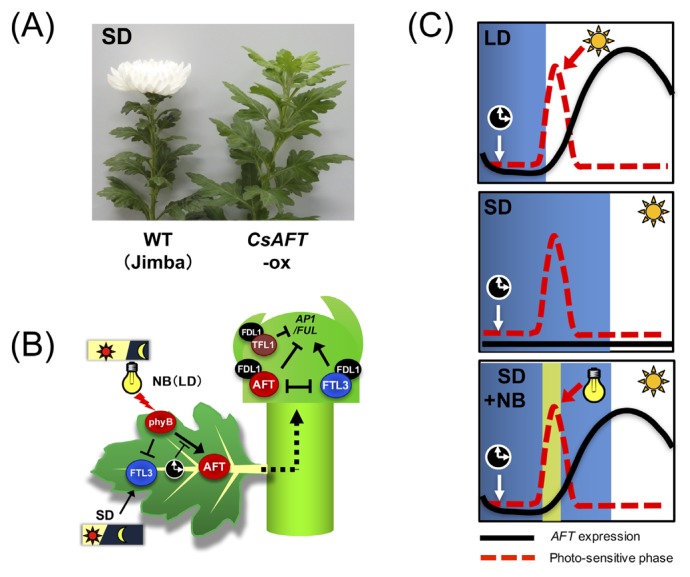
Oda et al. (2012) identified three FT-like genes from C. seticuspe: CsFTL1, CsFTL2, and CsFTL3. Among these, only CsFTL3 is up-regulated in the leaves under flowering-inducible SD photoperiod. CsFTL1 is up-regulated under LD or NB photoperiods, inhibiting flowering, but has weak florigenic activity (Higuchi et al. 2013, Higuchi and Hisamatsu 2015). CsFTL2 is expressed at very low levels in the leaves, but transient expression in protoplasts suggests that it also has weak florigenic activity (Higuchi and Hisamatsu 2015). More recently, CmFTL2 was suggested to be involved in sucrose-induced promotion of flowering in a photo-insensitive C. morifolium cultivar (Sun et al. 2017). Constitutive expression of CsFTL3 in C. morifolium resulted in photoperiod-insensitive flowering, and this flower-inducing effect was graft-transmissible. These results suggest that CsFTL3 encodes a systemic floral inducer florigen in chrysanthemums (Oda et al. 2012). Unlike Arabidopsis and Pharbitis, chrysanthemums require repeated cycles of SD photoperiod for floral initiation and successful anthesis (Corbesier et al. 2007, Hayama et al. 2007, Oda et al. 2012). Consistent with this requirement, CsFTL3 expression was not immediately induced by shifting plants from LD to SD photoperiod, but gradually increased with repetitive SD cycles (Nakano et al. 2013). Moreover, another floral inducer CsFTL1 is expressed under non-inductive LD or NB, but flowering is strictly suppressed under those conditions. From the screening of highly expressed genes in leaves under NB compared to SD photoperiod, one TFL1-like gene (CsAFT) has been identified. CsAFT was induced in leaves under non-inductive LD or NB photoperiods and it rapidly decreased after a shift to SD photoperiod (Higuchi et al. 2013). Constitutive expression of CsAFT in C. seticuspe and C. morifolium (CsAFT-ox) resulted in extremely late flowering under SD conditions, indicating that CsAFT has a strong floral-repressor activity (Fig. 2A). Furthermore, the knockdown of CsAFT by RNAi resulted in reduced sensitivity to NB and promoted flowering. Grafting experiment using CsAFT-ox plants clearly demonstrated that CsAFT proteins can move long distances across the grafting union and act as a systemic floral inhibitor. Transient gene expression assay revealed that both CsFTL3 and CsAFT interact with the C. seticuspe homolog of FD (CsFDL1), suggesting that CsAFT suppresses flowering by directly antagonizing the flower inducibility of CsFTL3-CsFDL1 (Higuchi et al. 2013). In addition to a systemic floral inhibitor CsAFT, a TFL1 homolog (CsTFL1) is constitutively expressed in shoot tips regardless of the photoperiods and shows strong floral inhibitor activity (Higuchi and Hisamatsu 2015). CsTFL1 also interacts with CsFDL1, suggesting that it suppresses flowering by directly interfering with the CsFTL3-CsFDL1 complex formation. Thus, in chrysanthemums, strict maintenance of a vegetative state under non-inducible photoperiod is achieved by a dual inhibitory system; one is a systemic floral inhibitor produced in non-inducible leaves (AFT), and another is a local inhibitor constitutively expressed at the shoot apex (TFL1) (Higuchi and Hisamatsu 2015) (Fig. 2B).
Fig. 2.
Photoperiodic regulation of flowering in chrysanthemums. (A) Over-expression of AFT in Chrysanthemum morifolium results in extremely late flowering under short day (SD) photoperiod. (B) Under SD, FTL3 is produced in leaves to systemically induce flowering. Under non-inductive long day (LD) or night-break (NB), AFT is induced in leaves to systemically inhibit flowering. The red light signal perceived by phyB induces AFT but suppresses FTL3 expression. Induction of AFT by phyB is gated by the circadian clock. TFL1 acts as a constitutive local repressor of flowering. (C) Model for induction of AFT under LD, SD, and NB conditions. The gate for AFT induction opens at a constant time after dusk regardless of the photoperiod. Under LD, the photo-sensitive phase (dotted line) of AFT interacts with red light in the morning and induces AFT expression (solid line) to inhibit flowering. Under NB, illumination at midnight coincides with the photo-sensitive phase of AFT.
Photo-perception and light sensitive time of day
Light quality affects chrysanthemum flowering. NB with red light effectively inhibits flowering, which is partially reversed by subsequent exposure to far-red (FR) light, suggesting the involvement of phytochromes in this response (Cathey and Borthwick 1957, Sumitomo et al. 2012). Interestingly, NB with blue light and FR light are both effective in inhibiting flowering when the plants are grown under a daily photoperiod with monochromatic blue light, but not white (blue + red) light (Higuchi et al. 2012). This suggests that light quality during the daily photoperiod affects the sensitivity to NB at midnight, and at least two distinct phy-mediated regulation systems might exist. In rice, it has been reported that phyB acts as a primary photoreceptor mediating NB response (Ishikawa et al. 2005). In C. seticuspe, the knock-down of CsPHYB by RNAi resulted in reduced sensitivity to NB with red light and flowered extremely early. In CsPHYB-RNAi plants, CsFTL3 was up-regulated, whereas CsAFT was down-regulated under NB conditions. These results indicated that CsPHYB acts as a primary photoreceptor mediating NB response and inhibits flowering by repressing CsFTL3 and inducing CsAFT (Higuchi et al. 2013).
In C. seticuspe, the most sensitive time for NB occurs 8 to 11 h after dusk. Consistent with this, CsAFT expression was strongly induced by red light from 8 to 10 h after dusk under both SD and LD conditions. Moreover, if long nights (14 h) were given, flowering was successfully induced, even under non-24-h light/dark cycles. Thus, induction of CsAFT by phy signaling is gated by an endogenous clock, and the gate for induction of CsAFT opens fully at a constant time after dusk, regardless of the photoperiod conditions provided (Fig. 2C). Therefore, day-length recognition by chrysanthemums is dependent on the absolute duration of darkness. Chrysanthemums detect the length of night by a timekeeping component, which is initiated by the dusk signal. Although the time-keeping mechanisms of chrysanthemums is still unclear, a recent study suggested the involvement of a core clock-component LATE ELONGATED HYPOCOTYL homolog of C. seticuspe (CsLHY). Constitutive expression of CsLHY fused with the transcriptional repressor domain (CsLHY-SRDX) showed photoperiod-insensitive floral transition, but further development of the capitulum was arrested (Oda et al. 2017). SRDX is a short transcriptional repression domain that turns a transcriptional activator into a strong repressor (Hiratsu et al. 2003). Thus, the plant expressing chimeric repressor shows phenotype similar to the loss-of-function mutation of the original transcription factor. In CsLHY-SRDX plants, diurnal and circadian expression of clock-related genes was not drastically attenuated, but expression of both CsFTL3 and CsAFT was downregulated. To date, homologs of circadian clock components and clock-controlled flowering time genes have been isolated in several species in the genus Chrysanthemum (Fu et al. 2014, 2015, Higuchi et al. 2012, Ren et al. 2016, Yang et al. 2014). It is of great interest to test whether these genes are involved in dark-time measurement and dark-dominant flowering of chrysanthemums.
Currently in Japan, chrysanthemum growers regulate flowering time by applying NB of 4–5 h at the middle of night. In C. seticuspe, systemic anti-florigen CsAFT was maximally induced by red light provided at a consistent time after dusk, but not during the middle of the darkness period (Higuchi et al. 2013). Consistent with this finding, the maximum sensitivity to NB (NB-max) appeared at a constant time after dusk in cultivated chrysanthemum varieties (Hakuzan and Kooriyama 2013). From these findings, by reconsidering the NB-max, which gradually changes depending on the season, it is possible to develop more efficient and energy-saving methods of artificial lighting.
Flowering retardation by high temperature
The growth temperature also affects chrysanthemum flowering. The flowering of autumn-flowering cultivars is severely delayed by high temperature during summer and early autumn in Japan. Recent studies have shown that heat-induced delay of flowering is mainly caused by inhibition of capitulum development (Nakano et al. 2013). Under optimum growth temperature, expression of FTL3 continues to increase under repeated SD cycles, which leads to successful development of capitulum. However, up-regulation of FTL3 in leaves was inhibited under high temperature (Nakano et al. 2013). In contrast to FTL3, expression of AFT was not affected by high temperature. Interestingly, high temperature from midnight to dawn was the most effective condition delaying flowering (Nakano et al. 2015a), suggesting that some common time-keeping mechanisms may operate to set both heat-sensitive and light-sensitive time-of-day in chrysanthemums. Transcriptional regulation of FTL3 is more sensitive to high temperature compared to that of AFT.
Regulation of flowering time and inflorescence architecture in tomato
Tomato (Solanum lycopersicum) is one of the most important vegetable crops around the world. The flowering time of tomato is regulated by the balance between the flowering inducer SINGLE FLOWER TRUSS (SFT) and the repressor SELF PRUNING (SP). SFT, a tomato ortholog of FT, is expressed in expanded mature leaves and systemically promotes flowering (Lifschitz et al. 2006, Shalit et al. 2009). In contrast, SP, a tomato homolog of TFL1, is expressed in young leaves and the shoot apex, and suppresses flowering (Shalit et al. 2009). The sp mutant flower earlier and gradually results in diminished sympodial growth, resulting in a more compact, determinate plant with nearly synchronized fruit ripening. Introduction of recessive sp mutation into modern tomato cultivars facilitated once-over mechanical harvesting, resulting in a major expansion of the tomato processing sector (Pnueli et al. 1998). The balance between SFT and SP regulates flowering and determinate or indeterminate shoot architecture. In the sp mutant background, the combination of weak alleles of SFT and mutations in SUPPRESSOR OF SP (SSP, FD homolog) weakened the activity of the florigen activation complex (FAC), resulting in a partially determinate architecture that provided maximum yield (Park et al. 2014).
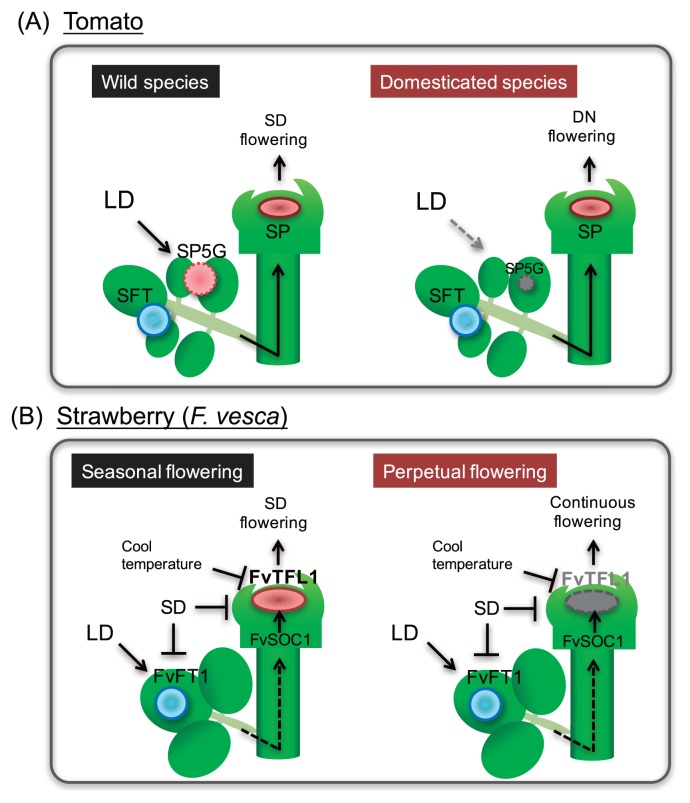
A BLAST survey against tomato whole genome-database revealed the presence of at least 13 PEBP genes, of which six (SP3D/SFT, SP6A, SP5G, SP5G1, SP5G2, and SP5G3) were FT-like genes (Cao et al. 2016). Among these, only SP3D/SFT had floral inducer activities whereas SP5G, SP5G2, and SP5G3 had floral inhibitor activities. The cultivated modern tomato species are categorized as DNPs, which do not respond to change in day-length, but the flowering of the wild species is promoted under SD conditions. A recent study reported that one of the SFT paralogs, SELF PRUNING 5G (SP5G), is highly induced in leaves grown under LD conditions in wild species, but not in cultivated species (Soyk et al. 2017). The loss-of-function mutation of SP5G induced by CRISPR/Cas9 system resulted in rapid flowering and early yield. Thus, SP5G acts as anti-florigen to suppress flowering under non-inductive LD photoperiod in a wild tomato species (Soyk et al. 2017). Mutations in the cis-regulatory region of cultivated tomato species reduced LD-induction of the floral repressor SP5G, resulting in early flowering under LD photoperiod, which enabled cultivation of tomato in high latitude areas (Fig. 3A). Thus, day-length sensitivity is essentially lost in domesticated tomato. Recently, it has been reported that domesticated tomato cultivars show longer period lengths in circadian rhythms (Müller et al. 2016). In cultivated tomato, allelic variation in the homolog of Arabidopsis, EMPFINDLICHER IM DUNKELROTEN LICHT 1 (EID1), which encodes the F-box protein, is responsible for the deceleration of the circadian clock. The EID1 allele of cultivated tomato enhances photosynthetic performance specifically under LD photoperiod (Müller et al. 2016).
Fig. 3.
The model for flowering time regulation in two major horticultural crops. (A) Flowering regulation in tomato. In wild species, a systemic anti-florigen SP5G is induced under long days (LD) to inhibit flowering. In day neutral (DN) domesticated species, the induction pathway of SP5G is attenuated. (B) Flowering regulation in wild strawberry (F. vesca). In seasonal flowering of short day (SD) cultivars, a strong floral repressor TFL1 suppresses flowering under LD. In perpetual flowering cultivars in which repressor activity of TFL1 is absent, FT1 and SOC1, act to promote flowering under LD.
Flowering time regulation in strawberry
Strawberry (Fragaria × ananassa) is a perennial plant that belongs to the Rosaceae family, in which flowering is induced by low temperature and SD photoperiod (Heide et al. 2013). They normally initiate the flower bud in response to SD and cool temperature in autumn, but further development of the flower bud (blooming) is promoted under LD photoperiod in the following spring. Recent studies in rose and diploid woodland strawberry (F. vesca) revealed that the loss-of-function mutation in TFL1 homolog is the principal cause of the continuous flowering phenotype of ever bearing cultivars (Iwata et al. 2012, Koskela et al. 2012). In seasonal flowering F. vesca, FvTFL1 mRNA expression is induced by LD photoperiod in shoot tips, but SD photoperiod suppresses it. However, in ever bearing (continuous flowering) cultivars, loss-of-function of a strong floral repressor, FvTFL1, resulted in the de-repression of flowering under LD photoperiod and reversed photoperiodic response (Koskela et al. 2012). Interestingly, a homolog of FT (FvFT1) is up-regulated in mature leaves specifically under LD conditions, and SOC1 ( FvSOC1) was similarly upregulated under LD conditions in the shoot tips to activate expression of FvTFL1 (Mouhu et al. 2013, Rantanen et al. 2014). Thus, FvFT1 and FvSOC1 act to suppress flowering under LD conditions through the activation of the strong floral repressor FvTFL1, but they act to promote flowering in continuous flowering accessions that lack functional FvTFL1. Moreover, FvTFL1 was regulated by a temperature-dependent pathway, independent of the regulation of FvFT1-FvSOC1 by photoperiod (Rantanen et al. 2015) (Fig. 3B). Molecular mechanisms of flowering regulation in cultivated octoploid species (F. × ananassa) are likely to be more complex. Genetic studies have suggested that continuous flowering of cultivated strawberry is controlled by a single dominant locus (Gaston et al. 2013, Morishita et al. 2012), whereas the continuous flowering trait of F. vesca is recessive. Recent studies have reported that flowering of F. × ananassa is strongly dependent on FaTFL1 regulation by day-length and temperature (Koskela et al. 2016, Nakano et al. 2015b), but the factors involved in its promotion remain unclear. One of the FT paralogs, FaFT3, was upregulated in the shoot tip under SD photoperiod and/or low temperature, in accordance with the promotion of flowering in F. × ananassa (Nakano et al. 2015b). It is of great interest to know whether the flowering of strawberry can be explained only by the reduced activity of a strong floral repressor or by the up-regulation of yet an unidentified floral promoter. Comparing the molecular mechanisms of flowering between F. × ananassa and F. vesca could provide further information.
Control of storage organ formation by florigens
The timing of vegetative reproduction events, such as the formation of the underground storage organ, is regulated by photoperiod. In potato (Solanum tuberosum), over-expression of the rice florigen gene Hd3a induced tuberization even under the non-inductive LD photoperiod (Navarro et al. 2011). One of the FT paralogs, StSP6A, is up-regulated in response to the inductive SD photoperiod in leaves, and acts systemically to induce tuberization. StSP6A expression is regulated by auto-regulatory mechanisms that amplify this signal in the stolons (Navarro et al. 2011). On the other hand, another FT paralog, StSP5G, is induced in leaves under non-inductive LD photoperiod in a StPHYB- and StCOL1-dependent manner and acts as a repressor of tuberization by preventing StSP6A expression (Abelenda et al. 2016). In onion (Allium cepa), different FT-like genes were shown to be involved in flowering and bulb formation (Lee et al. 2013). Onion is a biennial crop that forms bulbs under LDs of late spring to summer, and the overwintered bulbs flower in the next season. AcFT2 expression is correlated with flowering response, suggesting that this gene encodes a florigen. AcFT1 is induced in leaves under LD photoperiod and promotes bulb formation. On the other hand, AcFT4 is induced under SD photoperiod and inhibits bulb formation by preventing up-regulation of AcFT1 (Lee et al. 2013).
Conclusions and future perspectives
Recent studies in diverse plant species have suggested that modifications in the floral inducer or inhibitor activity could lead to variations in flowering responses. As reported in Arabidopsis and sugar beet, a small number of amino acid substitutions could cause conversion of the floral inducer to repressor activity (Ho and Weigel 2014, Pin et al. 2010). Studies in rose and strawberry indicated that the loss-of-function of a single strong floral repressor, TFL1, can convert seasonal flowering to a perpetual flowering habit (Iwata et al. 2012, Koskela et al. 2012). In domesticated tomato, mutations in the cis-regulatory region of a systemic anti-florigen, SP5G, reduced its expression under non-inducible LD photoperiod, resulting in rapid flowering and early yield (Soyk et al. 2017) (Fig. 3). These results suggest that manipulating the gene structure or the expression levels of florigens/anti-florigens through conventional breeding or biotechnological approaches could greatly accelerate the development of new cultivars with desirable flowering characteristics. These findings suggest that the FT/TFL1 gene family could be one of the most important breeding targets in many crop species. In addition to crop breeding, artificial control of flowering by manipulating the light and/or temperature conditions is particularly important for horticultural industries. Investigating the detailed expression profile of florigen/anti-florigen genes in response to various environmental stimuli could help us to develop efficient and energy-saving methods to control flowering. Due to the rapid spread of next generation sequencing technologies, it has become much easier to obtain genomic sequences of individual horticultural crops. By utilizing the various genetic resources and their genomic information, it will become possible to supply various marketable flowers, vegetables, and fruits year-round in the future.
Literature Cited
- Abe, M., Kobayashi, Y., Yamamoto, S., Daimon, Y., Yamaguchi, A., Ikeda, Y., Ichinoki, H., Notaguchi, M., Goto, K. and Araki, T. (2005) FD, a bZIP protein mediating signals from the floral pathway integrator FT at the shoot apex. Science 309: 1052–1056. [DOI] [PubMed] [Google Scholar]
- Abelenda, J.A., Cruz-Oró, E., Franco-Zorrilla, J.M. and Prat, S. (2016) Potato StCONSTANS-like1 suppresses storage organ formation by directly activating the FT-like StSP5G repressor. Curr. Biol. 26: 872–881. [DOI] [PubMed] [Google Scholar]
- Ahn, J.H., Miller, D., Winter, V.J., Banfield, M.J., Lee, J.H., Yoo, S.Y., Henz, S.R., Brady, R.L. and Weigel, D. (2006) A divergent external loop confers antagonistic activity on floral regulators FT and TFL1. EMBO J. 25: 605–614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blackman, B.K., Strasburg, J.L., Raduski, A.R., Michaels, S.D. and Rieseberg, L.H. (2010) The role of recently derived FT paralogs in sunflower domestication. Curr. Biol. 20: 629–635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradley, D., Ratcliffe, O., Vincent, C., Carpenter, R. and Coen, E. (1997) Inflorescence commitment and architecture in Arabidopsis. Science 275: 80–83. [DOI] [PubMed] [Google Scholar]
- Cao, K., Cui, L., Zhou, X., Ye, L., Zou, Z. and Deng, S. (2016) Four tomato FLOWERING LOCUS T-like proteins act antagonistically to regulate floral initiation. Front. Plant Sci. 6: 1213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cathey, H. and Borthwick, H. (1957) Photoreversibility of floral initiation in Chrysanthemum. Bot. Gaz. 119: 71–76. [Google Scholar]
- Chailakhyan, M. (1936) New facts in support of the hormonal theory of plant development. Dokl. Akad. Nauk. SSSR 13: 79–83. [Google Scholar]
- Conti, L. and Bradley, D. (2007) TERMINAL FLOWER1 is a mobile signal controlling Arabidopsis architecture. Plant Cell 19: 767–778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corbesier, L., Vincent, C., Jang, S., Fornara, F., Fan, Q., Searle, I., Giakountis, A., Farrona, S., Gissot, L., Turnbull, C. et al. (2007) FT protein movement contributes to long-distance signaling in floral induction of Arabidopsis. Science 316: 1030–1033. [DOI] [PubMed] [Google Scholar]
- Doi, K., Izawa, T., Fuse, T., Yamanouchi, U., Kubo, T., Shimatani, Z., Yano, M. and Yoshimura, A. (2004) Ehd1, a B-type response regulator in rice, confers short-day promotion of flowering and controls FT-like gene expression independently of Hd1. Genes Dev. 18: 926–936. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans, L. (1960) Inflorescence initiation in Lolium temulentum L. II. Evidence for inhibitory and promotive photoperiodic processes involving transmissible products. Aust. J. Biol. Sci. 13: 429–440. [Google Scholar]
- Fu, J., Yang, L. and Dai, S. (2014) Conservation of Arabidopsis thaliana circadian clock genes in Chrysanthemum lavandulifolium. Plant Physiol. Biochem. 80: 337–347. [DOI] [PubMed] [Google Scholar]
- Fu, J., Yang, L. and Dai, S. (2015) Identification and characterization of the CONSTANS-like gene family in the short-day plant Chrysanthemum lavandulifolium. Mol. Genet. Genomics 290: 1039–1054. [DOI] [PubMed] [Google Scholar]
- Garner, W. and Allard, H. (1920) Effect of the relative length of day and night and other factors of the environment on growth and reproduction in plants. J. Agric. Res. 18: 553–606. [Google Scholar]
- Gaston, A., Perrotte, J., Lerceteau-Köhler, E., Rousseau-Gueutin, M., Petit, A., Hernould, M., Rothan, C. and Denoyes, B. (2013) PFRU, a single dominant locus regulates the balance between sexual and asexual plant reproduction in cultivated strawberry. J. Exp. Bot. 64: 1837–1848. [DOI] [PubMed] [Google Scholar]
- Golembeski, G.S. and Imaizumi, T. (2015) Photoperiodic regulation of florigen function in Arabidopsis thaliana. Arabidopsis Book 13: e0178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guttridge, C. (1959) Further evidence for a growth-promoting and flower-inhibiting hormone in strawberry. Ann. Bot. 23: 612–621. [Google Scholar]
- Hakuzan, R. and Kooriyama, K. (2013) Effect of night-break treatment at different times on inhibition of flower bud initiation in chrysanthemum. Hort. Res. (Japan) 12: 427–432. [Google Scholar]
- Hanano, S. and Goto, K. (2011) Arabidopsis TERMINAL FLOWER1 is involved in the regulation of flowering time and inflorescence development through transcriptional repression. Plant Cell 23: 3172–3184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanzawa, Y., Money, T. and Bradley, D. (2005) A single amino acid converts a repressor to an activator of flowering. Proc. Natl. Acad. Sci. USA 102: 7748–7753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayama, R., Yokoi, S., Tamaki, S., Yano, M. and Shimamoto, K. (2003) Adaptation of photoperiodic control pathways produces short-day flowering in rice. Nature 422: 719–722. [DOI] [PubMed] [Google Scholar]
- Hayama, R., Agashe, B., Luley, E., King, R. and Coupland, G. (2007) A circadian rhythm set by dusk determines the expression of FT homologs and the short-day photoperiodic flowering response in Pharbtis. Plant Cell 19: 2988–3000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heide, O., Stavang, J. and Sønsteby, A. (2013) Physiology and genetics of flowering in cultivated and wild strawberries. J. Hortic. Sci. Biotechnol. 88: 1–18. [Google Scholar]
- Higuchi, Y., Sumitomo, K., Oda, A., Shimizu, H. and Hisamatsu, T. (2012) Day light quality affects the night-break response in the short-day plant chrysanthemum, suggesting differential phytochrome-mediated regulation of flowering. J. Plant Physiol. 169: 1789–1796. [DOI] [PubMed] [Google Scholar]
- Higuchi, Y., Narumi, T., Oda, A., Nakano, Y., Sumitomo, K., Fukai, S. and Hisamatsu, T. (2013) The gated induction system of a systemic floral inhibitor, antiflorigen, determines obligate short-day flowering in chrysanthemums. Proc. Natl. Acad. Sci. USA 110: 17137–17142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Higuchi, Y. and Hisamatsu, T. (2015) CsTFL1, a constitutive local repressor of flowering, modulates floral initiation by antagonising florigen complex activity in chrysanthemum. Plant Sci. 237: 1–7. [DOI] [PubMed] [Google Scholar]
- Hiratsu, K., Matsui, K., Koyama, T. and Ohme-Takagi, M. (2003) Dominant repression of target genes by chmeric repressors that include the EAR motif, a repression domain, in Arabidopsis. Plant J. 34: 733–739. [DOI] [PubMed] [Google Scholar]
- Ho, W.W.H. and Weigel, D. (2014) Structural features determining flower-promoting activity of Arabidopsis FLOWERING LOCUS T. Plant Cell 26: 552–564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang, N.C., Jane, W.N., Chen, J. and Yu, T.S. (2012) Arabidopsis thaliana CENTRORADIALIS homologue (ATC) acts systemically to inhibit floral initiation in Arabidopsis. Plant J. 72: 175–184. [DOI] [PubMed] [Google Scholar]
- Ishikawa, R., Tamaki, S., Yokoi, S., Inagaki, N., Shinomura, T., Takano, M. and Shimamoto, K. (2005) Suppression of the floral activator Hd3a is the principal cause of the night break effect in rice. Plant Cell 17: 3326–3336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Itoh, H., Nonoue, Y., Yano, M. and Izawa, T. (2010) A pair of floral regulators sets critical day length for Hd3a florigen expression in rice. Nat. Genet. 42: 635–638. [DOI] [PubMed] [Google Scholar]
- Iwata, H., Gaston, A., Remay, A., Thouroude, T., Jeauffre, J., Kawamura, K., Oyant, L.H.-S., Araki, T., Denoyes, B. and Foucher, F. (2012) The TFL1 homologue KSN is a regulator of continuous flowering in rose and strawberry. Plant J. 69: 116–125. [DOI] [PubMed] [Google Scholar]
- Izawa, T., Oikawa, T., Sugiyama, N., Tanisaka, T., Yano, M. and Shimamoto, K. (2002) Phytochrome mediates the external light signal to repress FT orthologs in photoperiodic flowering of rice. Genes Dev. 16: 2006–2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaeger, K.E., Pullen, N., Lamzin, S., Morris, R.J. and Wigge, P.A. (2013) Interlocking feedback loops govern the dynamic behavior of the floral transition in Arabidopsis. Plant Cell 25: 820–833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jang, S., Torti, S. and Coupland, G. (2009) Genetic and spatial interactions between FT, TSF and SVP during the early stages of floral induction in Arabidopsis. Plant J. 60: 614–625. [DOI] [PubMed] [Google Scholar]
- Kardailsky, I., Shukla, V.K., Ahn, J.H., Dagenais, N., Christensen, S.K., Nguyen, J.T., Chory, J., Harrison, M.J. and Weigel, D. (1999) Activation tagging of the floral inducer FT. Science 286: 1962–1965. [DOI] [PubMed] [Google Scholar]
- Klie, M., Schie, S., Linde, M. and Debener, T. (2014) The type of ploidy of chrysanthemum is not black or white: a comparison of a molecular approach to published cytological methods. Front. Plant Sci. 5: 479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi, Y., Kaya, H., Goto, K., Iwabuchi, M. and Araki, T. (1999) A pair of related genes with antagonistic roles in mediating flowering signals. Science 286: 1960–1962. [DOI] [PubMed] [Google Scholar]
- Kojima, S., Takahashi, Y., Kobayashi, Y., Monna, L., Sasaki, T., Araki, T. and Yano, M. (2002) Hd3a, a rice ortholog of the Arabidopsis FT gene, promotes transition to flowering downstream of Hd1 under short-day conditions. Plant Cell Physiol. 43: 1096–1105. [DOI] [PubMed] [Google Scholar]
- Komiya, R., Yokoi, S. and Shimamoto, K. (2009) A gene network for long-day flowering activates RFT1 encoding a mobile flowering signal in rice. Development 136: 3443–3450. [DOI] [PubMed] [Google Scholar]
- Koskela, E.A., Mouhu, K., Albani, M.C., Kurokura, T., Rantanen, M., Sargent, D.J., Battey, N.H., Coupland, G., Elomaa, P. and Hytönen, T. (2012) Mutation in TERMINAL FLOWER1 reverses the photoperiodic requirement for flowering in the wild strawberry Fragaria vesca. Plant Physiol. 159: 1043–1054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koskela, E.A., Sønsteby, A., Flachowsky, H., Heide, O.M., Hanke, M.V., Elomaa, P. and Hytönen, T. (2016) TERMINAL FLOWER1 is a breeding target for a novel everbearing trait and tailored flowering responses in cultivated strawberry (Fragaria × ananassa Duch.). Plant Biotechnol. J. 14: 1852–1861. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lang, A. and Melchers, G. (1943) Die photoperiodische reaktion von Hyoscyamus niger. Planta 33: 653–702. [Google Scholar]
- Lang, A., Chailakhyan, M.K. and Frolova, I. (1977) Promotion and inhibition of flower formation in a dayneutral plant in grafts with a short-day plant and a long-day plant. Proc. Natl. Acad. Sci. USA 74: 2412–2416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laurie, A. (1930) Photoperiodism—practical application to greenhouse culture. Proc. Am. Soc. Hortic. Sci. 27: 319–322. [Google Scholar]
- Lee, R., Baldwin, S., Kenel, F., McCallum, J. and Macknight, R. (2013) FLOWERING LOCUS T genes control onion bulb formation and flowering. Nat. Commun. 4: 2884. [DOI] [PubMed] [Google Scholar]
- Lifschitz, E., Eviatar, T., Rozman, A., Shalit, A., Goldshmidt, A., Amsellem, Z., Alvarez, J.P. and Eshed, Y. (2006) The tomato FT ortholog triggers systemic signals that regulate growth and flowering and substitute for diverse environmental stimuli. Proc. Natl. Acad. Sci. USA 103: 6398–6403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin, M.K., Belanger, H., Lee, Y.J., Varkonyi-Gasic, E., Taoka, K., Miura, E., Xoconostle-Cázares, B., Gendler, K., Jorgensen, R.A., Phinney, B. et al. (2007) FLOWERING LOCUS T protein may act as the long-distance florigenic signal in the cucurbits. Plant Cell 19: 1488–1506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsoukas, I. (2015) Florigens and antiflorigens: a molecular genetic understanding. Essays Biochem. 58: 133–149. [DOI] [PubMed] [Google Scholar]
- Mimida, N., Goto, K., Kobayashi, Y., Araki, T., Ahn, J.H., Weigel, D., Murata, M., Motoyoshi, F. and Sakamoto, W. (2001) Functional divergence of the TFL1-like gene family in Arabidopsis revealed by characterization of a novel homologue. Genes Cells 6: 327–336. [DOI] [PubMed] [Google Scholar]
- Morishita, M., Honjo, M., Hamano, M., Yamazaki, H. and Yano, T. (2012) Genetic analysis of the everbearing habit in strawberry cultivars under 24-hour daylength condition. Hort. Res. (Japan) 11: 301–307. [Google Scholar]
- Mouhu, K., Kurokura, T., Koskela, E.A., Albert, V.A., Elomaa, P. and Hytönen, T. (2013) The Fragaria vesca homolog of SUPPRESSOR OF OVEREXPRESSION OF CONSTANS1 represses flowering and promotes vegetative growth. Plant Cell 25: 3296–3310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Müller, N.A., Wijnen, C.L., Srinivasan, A., Ryngajllo, M., Ofner, I., Lin, T., Ranjan, A., West, D., Maloof, J.N., Sinha, N.R. et al. (2016) Domestication selected for deceleration of the circadian clock in cultivated tomato. Nat. Genet. 48: 89–93. [DOI] [PubMed] [Google Scholar]
- Nakagawa, M., Shimamoto, K. and Kyozuka, J. (2002) Overexpression of RCN1 and RCN2, rice TERMINAL FLOWER 1/CENTRORADIALIS homologs, confers delay of phase transition and altered panicle morphology in rice. Plant J. 29: 743–750. [DOI] [PubMed] [Google Scholar]
- Nakano, Y., Higuchi, Y., Sumitomo, K. and Hisamatsu, T. (2013) Flowering retardation by high temperature in chrysanthemums: involvement of FLOWERING LOCUS T-like 3 gene expression. J. Exp. Bot. 64: 909–920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakano, Y., Higuchi, Y., Sumitomo, K., Oda, A. and Hisamatsu, T. (2015a) Delay of flowering by high temperature in chrysanthemum: heat-sensitive time-of-day and heat effects on CsFTL3 and CsAFT gene expression. J. Hortic. Sci. Biotechnol. 90: 143–149. [Google Scholar]
- Nakano, Y., Higuchi, Y., Yoshida, Y. and Hisamatsu, T. (2015b) Environmental responses of the FT/TFL1 gene family and their involvement in flower induction in Fragaria × ananassa. J. Plant. Physiol. 177: 60–66. [DOI] [PubMed] [Google Scholar]
- Navarro, C., Abelenda, J.A., Cruz-Oró, E., Cuéllar, C.A., Tamaki, S., Silva, J., Shimamoto, K. and Prat, S. (2011) Control of flowering and storage organ formation in potato by FLOWERING LOCUS T. Nature 478: 119–122. [DOI] [PubMed] [Google Scholar]
- Oda, A., Narumi, T., Li, T., Kando, T., Higuchi, Y., Sumitomo, K., Fukai, S. and Hisamatsu, T. (2012) CsFTL3, a chrysanthemum FLOWERING LOCUS T-like gene, is a key regulator of photoperiodic flowering in chrysanthemums. J. Exp. Bot. 63: 1461–1477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oda, A., Higuchi, Y. and Hisamatsu, T. (2017) Photoperiod-insensitive floral transition in chrysanthemum induced by constitutive expression of chimeric repressor CsLHY-SRDX. Plant Sci. 259: 86–93. [DOI] [PubMed] [Google Scholar]
- Ogawa, Y. and King, R. (1990) The inhibition of flowering by non-induced cotyledons of Pharbitis nil. Plant Cell Physiol. 31: 129–135. [Google Scholar]
- Ogiso-Tanaka, E., Matsubara, K., Yamamoto, S., Nonoue, Y., Wu, J., Fujisawa, H., Ishikubo, H., Tanaka, T., Ando, T., Matsumoto, T. et al. (2013) Natural variation of the RICE FLOWERING LOCUS T 1 contributes to flowering time divergence in rice. PLoS ONE 8: e75959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Otagaki, S., Ogawa, Y., Oyant, L.H.-S., Foucher, F., Kawamura, K., Horibe, T. and Matsumoto, S. (2015) Genotype of FLOWERING LOCUS T homologue contributes to flowering time differences in wild and cultivated roses. Plant Biol. (Stuttg) 17: 808–815. [DOI] [PubMed] [Google Scholar]
- Park, S.J., Jiang, K., Tal, L., Yichie, Y., Gar, O., Zamir, D., Eshed, Y. and Lippman, Z.B. (2014) Optimization of crop productivity in tomato using induced mutations in the florigen pathway. Nat. Genet. 46: 1337–1342. [DOI] [PubMed] [Google Scholar]
- Pin, P.A., Benlloch, R., Bonnet, D., Wremerth-Weich, E., Kraft, T., Gielen, J.J.L. and Nilsson, O. (2010) An antagonistic pair of FT homologs mediates the control of flowering time in sugar beet. Science 330: 1397–1400. [DOI] [PubMed] [Google Scholar]
- Pnueli, L., Carmel-Goren, L., Hareven, D., Gutfinger, T., Alvarez, J.P., Ganal, M., Zamir, D. and Lifschitz, E. (1998) The SELF-PRUNING gene of tomato regulates vegetative to reproductive switching of sympodial meristems and is the ortholog of CEN and TFL1. Development 125: 1979–1989. [DOI] [PubMed] [Google Scholar]
- Poesch, G.H. (1936) Prolonging the flowering period of chrysanthemums with the use of supplementary illumination. Proc. Am. Soc. Hortic. Sci. 34: 624–626. [Google Scholar]
- Post, K. (1931) Reducing the day length of chrysanthemums for the production of early blooms by the use of black sateen cloth. Proc. Am. Soc. Hortic. Sci. 28: 382–388. [Google Scholar]
- Randoux, M., Davière, J.-M., Jeauffre, J., Thouroude, T., Pierre, S., Toualbia, Y., Perrotte, J., Reynoird, J.-P., Jammes, M.-J., Oyant, L.H.-S. et al. (2013) RoKSN, a floral repressor, forms protein complexes with RoFD and RoFT to regulate vegetative and reproductive development in rose. New Phytol. 202: 161–173. [DOI] [PubMed] [Google Scholar]
- Rantanen, M., Kurokura, T., Mouhu, K., Pinho, P., Tetri, E., Halonen, L., Palonen, P., Elomaa, P. and Hytönen, T. (2014) Light quality regulates flowering in FvFT1/FvTFL1 dependent manner in the woodland strawberry Fragaria vesca. Front. Plant Sci. 5: 271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rantanen, M., Kurokura, T., Jiang, P., Mouhu, K. and Hytönen, T. (2015) Strawberry homologue of TERMINAL FLOWER 1 integrates photoperiod and temperature signals to inhibit flowering. Plant J. 82: 163–173. [DOI] [PubMed] [Google Scholar]
- Ratcliffe, O., Bradley, D. and Coen, E. (1999) Separation of shoot and floral identity in Arabidopsis. Development 126: 1109–1120. [DOI] [PubMed] [Google Scholar]
- Ren, L., Liu, T., Cheng, Y., Sun, J., Gao, J., Dong, B., Chen, S., Chen, F. and Jiang, J. (2016) Transcriptomic analysis of differentially expressed genes in the floral transition of the summer flowering chrysanthemum. BMC Genomics 17: 673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryu, J.Y., Lee, H.J., Seo, P.J., Jung, J.H., Ahn, J.H. and Park, C.M. (2014) The Arabidopsis floral repressor BFT delays flowering by competing with FT for FD binding under high salinity. Mol. Plant 7: 377–387. [DOI] [PubMed] [Google Scholar]
- Shalit, A., Rozman, A., Goldshmidt, A., Alvarez, J.P., Bowman, J.L., Eshed, Y. and Lifschitz, E. (2009) The flowering hormone florigen functions as a general systemic regulator of growth and termination. Proc. Natl. Acad. Sci. USA 106: 8392–8397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soyk, S., Müller, N., Park, S.J., Schmalenbach, I., Jiang, K., Hayama, R., Zhang, L., Eck, J.V., Jiménez-Gómez, J.M. and Lippman, Z.B. (2017) Variation in the flowering gene SELF PRUNING 5G promotes day-neutrality and early yield in tomato. Nat. Genet. 49: 162–168. [DOI] [PubMed] [Google Scholar]
- Sumitomo, K., Higuchi, Y., Aoki, K., Miyamae, H., Oda, A., Ishiwata, M., Yamada, M., Nakayama, M. and Hisamatsu, T. (2012) Spectral sensitivity of flowering and FT-like gene expression in response to a night break treatment in the chrysanthemum cultivar ‘Reagan’. J. Hortic. Sci. Biotechnol. 87: 461–469. [Google Scholar]
- Sun, J., Wang, H., Ren, L., Chen, S., Chen, F. and Jiang, J. (2017) CmFTL2 is involved in the photoperiod- and sucrose-mediated control of flowering time in chrysanthemum. Hortic. Res. 4: 17001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tamaki, S., Matsuo, S., Wong, H.L., Yokoi, S. and Shimamoto, K. (2007) Hd3a protein is a mobile flowering signal in rice. Science 316: 1033–1036. [DOI] [PubMed] [Google Scholar]
- Tanaka, T. (1967) Studies on the regulation of Chrysanthemum flowering with special reference to plant regulators I. The inhibiting action of non-induced leaves on floral stimulus. J. Japan Soc. Hort. Sci. 36: 339–347. [Google Scholar]
- Taoka, K., Ohki, I., Tsuji, H., Furuita, K., Hayashi, K., Yanase, T., Yamaguchi, M., Nakashima, C., Purwestri, Y.A., Tamaki, S. et al. (2011) 14-3-3 proteins act as intracellular receptors for rice Hd3a florigen. Nature 476: 332–335. [DOI] [PubMed] [Google Scholar]
- Valverde, F., Mouradov, A., Soppe, W., Ravenscroft, D., Samach, A. and Coupland, G. (2004) Photoreceptor regulation of CONSTANS protein in photoperiodic flowering. Science 303: 1003–1006. [DOI] [PubMed] [Google Scholar]
- Wada, K.C., Yamada, M., Shiraya, T. and Takeno, K. (2010) Salicylic acid and the flowering gene FLOWERING LOCUS T homolog are involved in poor-nutrition stress-induced flowering of Pharbitis nil. J. Plant Physiol. 167: 447–452. [DOI] [PubMed] [Google Scholar]
- Wickland, D. and Hanzawa, Y. (2015) The FLOWERING LOCUS T/TERMINAL FLOWER 1 gene family: functional evolution and molecular mechanisms. Mol. Plant 8: 983–997. [DOI] [PubMed] [Google Scholar]
- Wigge, P.A., Kim, M.C., Jaeger, K.E., Busch, W., Schmid, M., Lohmann, J.U. and Weigel, D. (2005) Integration of spatial and temporal information during floral induction in Arabidopsis. Science 309: 1056–1059. [DOI] [PubMed] [Google Scholar]
- Xi, W., Liu, C., Hou, X. and Yu, H. (2010) MOTHER OF FT AND TFL1 regulates seed germination through a negative feedback loop modulating ABA signaling in Arabidopsis. Plant Cell 22: 1733–1748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue, W., Xing, Y., Weng, X., Zhao, Y., Tang, W., Wang, L., Zhou, H., Yu, S., Xu, C., Li, X. et al. (2008) Natural variation in Ghd7 is an important regulator of heading date and yield potential in rice. Nat. Genet. 40: 761–767. [DOI] [PubMed] [Google Scholar]
- Yamaguchi, A., Kobayashi, Y., Goto, K., Abe, M. and Araki, T. (2005) TWIN SISTER OF FT ( TSF) acts as a floral pathway integrator redundantly with FT. Plant Cell Physiol. 46: 1175–1189. [DOI] [PubMed] [Google Scholar]
- Yang, Y., Ma, C., Xu, Y., Wei, Q., Imtiaz, M., Lan, H., Gao, S., Cheng, L., Wang, M., Fei, Z. et al. (2014) A zinc finger protein regulates flowering time and abiotic stress tolerance in chrysanthemum by modulating gibberellin biosynthesis. Plant Cell 26: 2038–2054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanovsky, M. and Kay, S.A. (2002) Molecular basis of seasonal time measurement in Arabidopsis. Nature 419: 308–312. [DOI] [PubMed] [Google Scholar]
- Yoo, S.J., Chung, K.S., Jung, S.H., Yoo, S.Y., Lee, J.S. and Ahn, J.H. (2010) BROTHER OF FT AND TFL1 (BFT) has TFL1-like activity and functions redundantly with TFL1 in inflorescence meristem development in Arabidopsis. Plant J. 63: 241–253. [DOI] [PubMed] [Google Scholar]
- Yoo, S.Y., Kardailsky, I., Lee, J.S., Weigel, D. and Ahn, J.H. (2004) Acceleration of flowering by overexpression of MFT (MOTHER OF FT AND TFL1). Mol. Cells 17: 95–101. [PubMed] [Google Scholar]