Abstract
New models to study the intestine are key to understanding intestinal diseases and developing novel treatments. Intestinal organ-like culture systems (organoids and enteroids) have substantially advanced the study of the human gastrointestinal tract. Stem cell–derived cultures produce self-organizing structures that contain the multiple differentiated intestinal epithelial cell types including enterocytes, goblet, Paneth, and enteroendocrine cells. Understanding host–microbial interactions is one area in which these cultures are expediting major advancements. This review discusses how organoid and enteroid cultures are biologically and physiologically relevant systems to investigate the effects of commensal organisms and study the pathogenesis of human infectious diseases. These cultures can be established from many donors and they retain the genetic and biologic properties of the donors, which can lead to the discovery of host-specific factors that affect susceptibility to infection and result in personalized approaches to treat individuals. The continued development of these cultures to incorporate more facets of the gastrointestinal tract, including neurons, immune cells, and the microbiome, will unravel new mechanisms regulating host–microbial interactions with the long-term goal of translating findings into novel preventive or therapeutic treatments for gastrointestinal infections.
Keywords: Enteroids, Organoids, Infections, Microbiome, Host-Microbial Interactions
Abbreviations used in this paper: HBGA, histo-blood group antigen; IFN, interferon; IL, interleukin; 3D, three-dimensional
Summary.
Enteroid and organoid cultures have revolutionized the study of human small intestinal biology and are outstanding models to explore relationships between microbial organisms and the human host. Increasing the complexity of these cultures will contribute to a better understanding of gut function.
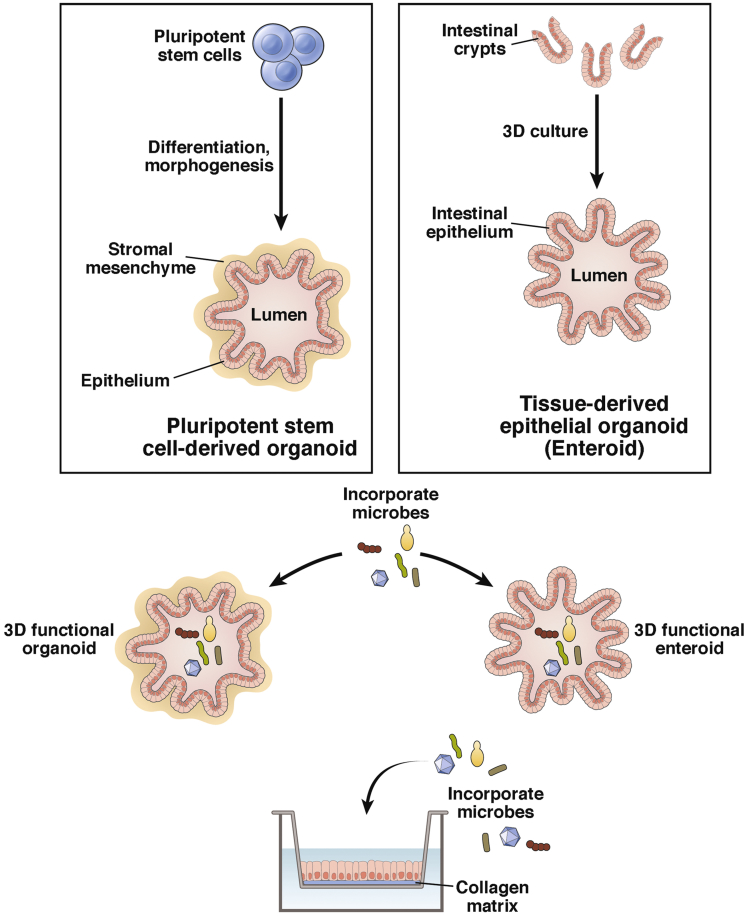
The human small intestine is a complex organ with an epithelial surface that provides a protective barrier against a diverse and hostile environment. New models to study the intestine will be a key component in developing novel treatments for many intestinal diseases. Although tissue engineering of the human small intestine is still in its infancy and no in vitro systems are yet available to study the human intestine as a whole, intestinal organ-like culture systems with long-term capacity for expansion in vitro have given rise to elegant models of the gastrointestinal epithelium. These cultures surpass and overcome limitations of the colonic adenocarcinoma cell lines used previously that retain altered cellular pathways of transformed cells and do not reflect cellular or host diversity. Two pivotal technologies substantially advanced production of novel human small intestinal cultures derived from different sources (Figure 1). Tissue-derived stem cells isolated from human intestinal biopsies or surgical specimens can be differentiated into epithelial only cultures.1, 2 Human pluripotent stem cells (either embryonic in origin or reprogrammed somatic cells) can be directed to give rise to intestinal epithelial cultures associated with mesenchyme.3 In both systems, the stem cells produce self-organizing cultures that contain the multiple differentiated intestinal epithelial cell types including enterocytes, goblet, Paneth, and enteroendocrine cells. In 2012, the intestinal stem cell consortium proposed nomenclature to distinguish epithelial only (enteroids) from epithelial/mesenchymal (organoids) cultures, and we use this terminology from here on.4 These cultures have launched a new era in the study of human gastrointestinal tract biology, physiology, and pathophysiology.
Figure 1.
Derivation of organoids and enteroids and modes of exposure to microbial organisms in three-dimensional or two-dimensional format. Modified from Mills and Estes.67
This review discusses how enteroid and organoid cultures are biologically and physiologically relevant systems to investigate the effects of commensal organisms and study the pathogenesis of human infectious diseases. The small intestinal epithelium is the primary site of replication for many microbial communities and gastrointestinal pathogens, yet fundamental knowledge of the molecular mechanisms of human intestinal epithelial cell–microbial interactions remains limited. Understanding host-microbial interactions is one area where these cultures are expediting major advancements. The unique interactions between human host cells and beneficial and pathogenic organisms are not reproduced in animal systems and unicellular transformed cell lines. Primary cell cultures and tissue explants can be used to pattern the human intestine, but their use is limited to short-term culture and continued access to the primary material. By contrast, enteroid and organoid cultures are revealing answers to longstanding questions concerning how microbial organisms interact with and induce responses in the human intestinal epithelium. Human-specific susceptibility factors including genetic risk factors, age, gender, and ethnicity that affect infection and pathogenesis can be elucidated. Both enteroid and organoid cultures grown from many donors resulting in “banks” will lead the way to personalized approaches to treat individuals who develop disease. The continued development of these cultures to include more facets of the gastrointestinal tract will facilitate further discoveries into host-microbial interactions with the long-term goal of translating findings into novel preventive or therapeutic treatments for gastrointestinal infections.
Organization of the Small Intestine
The human small intestine consists of different cell types and regional specialization that contribute to its biology and physiology. The apical side of the polarized epithelium faces the lumen of the gastrointestinal tract. The epithelium not only functions as a barrier to the outside environment but also has secretory and absorptive capabilities. Cellular heterogeneity is the basis for these different functions as goblet, enteroendocrine, and Paneth cells secrete mucous, neuroendocrine, and antimicrobial factors, respectively, whereas the enterocytes are primarily responsible for absorbing and secreting luminal components basolaterally into the portal circulation. The epithelium has 2 distinct zones, the crypt that houses the stem cells and Paneth cells and the villus that is composed of the differentiated secretory and absorptive cells and protrudes into the lumen of the intestine. The crypt has enormous capacity for self-renewal, which arises from multipotent stem cell populations that proliferate, differentiate, and migrate to become the differentiated villus cells. Adding to this complexity is regional specificity that provides unique functional properties in the 3 sections of small intestine (duodenum, jejunum, and ileum). Underlying the epithelium is the stroma consisting of mesenchymal, immune, neuronal, smooth muscle, and endothelial cells, which provide support, both physically and biologically, for the epithelium. The complex nature of the small intestine presents specific challenges for tissue engineering that range from supporting the diverse cell types that require vastly different factors to survive in vitro and communication pathways between the intestine and other organs that influence cellular function, to deriving an in vitro culture platform in which the biology of the human organ is fully recapitulated. Although not yet fully achieved, remarkable progress is being made.
Engineered Human Gastrointestinal Tissue
Both organoid and enteroid cultures are composed of heterogeneous cell populations that recapitulate the in vivo small intestinal epithelium. They self-organize into three-dimensional (3D) structures termed mini-guts that are maintained indefinitely by using in vitro culture techniques with propagation in extracellular support matrices such as Matrigel, a basement membrane analog that helps direct the polarity of the epithelium.3, 5, 6 The medium covering the Matrigel-embedded organoids and enteroids contains growth factors that promote stem cell proliferation including high concentrations of Wnt-3a and the WNT signal amplifier R-spondin, epidermal growth factor, and noggin, a secreted bone morphogenetic protein inhibitor. Withdrawal of some of these growth factors redirects the stem cells toward a differentiated cell state resulting in cultures that are composed of all the major differentiated cell types, in the appropriate frequencies, that comprise the human small intestinal epithelium in vivo. The epithelium exhibits polarization with defined apical and basolateral surfaces, and the cultures can generate several cell types including M cells,7, 8 Tuft cells,9 and Paneth cells5 that previously could not be generated in vitro. Furthermore, these epithelial cultures retain similar physiological functions of the original intestinal tissue.10, 11, 12 For example, they exhibit functional peptide and ion transport and secrete chloride in response to known secretagogues.3, 13, 14 Most important, the stem cells exhibit a low frequency of mutation,5 and these cultures can be expanded indefinitely and cryopreserved, which allows long-term storage and sharing between research groups to enable biological validation. Organoid and enteroid cultures have several advantages over transformed cell lines and animal models. They are able to be generated from human cells, simulate human diseases, can be used to study genetic effects on intestinal biology, are models that will theoretically lead to the establishment of personalized medicine approaches for clinical treatment, and potentially will be used in regenerative medicine. In addition, they can be genetically manipulated by using transfection, electroporation, and lentivirus transduction.13, 15, 16 These new experimental tools are applicable for both basic and translational research.
Small Intestinal Organoids
Intestinal organoids have several advantages as an experimental system. Because of their origin from induced pluripotent stem cells, the establishment of organoid cultures does not rely on the availability of intestinal tissue. In addition, organoid cultures have the advantage of also containing mesenchymal cells that interact closely with the epithelium.3, 17 This mixture of mesenchymal-epithelial cells mimics what is seen in vivo where the mesenchyme is a key factor in controlling the growth, differentiation, and functionality of the epithelium. Although the architectural complexity of the organoid cultures approaches what is seen in vivo, full maturation of the epithelium is not seen, and heterogeneous differentiation between organoids is often observed.3, 18, 19 Transcriptional profiling analyses indicates that human organoids exhibit a more fetal phenotype compared with mature adult small intestine,20, 21, 22 and the establishment of definitive regions has been limited to broadly proximal and distal small intestine.23 However, further maturation is achieved when organoids are implanted under the kidney capsule of a mouse where they recruit mouse stromal factors that help the organoids develop into a more adult-like phenotype.17, 21
Small Intestinal Enteroids
Enteroid cultures have different advantages as an experimental system. These cultures exhibit cellular differentiation and regional stem cell specificity similar to what is found in vivo.2 Villus-like cystic structures bud off from a single central lumen, with stem cells and Paneth cells at the bottom of the structure with the more mature enterocytes, goblet, and enteroendocrine cells found closer to the lumen. Enteroids retain regional specificity based on the origin of the tissue (duodenum, jejunum, ileum) and are physiologically active, exhibiting absorptive and secretory function, and the apical surface is coated with mucin secreted by the goblet cells.24 However, these cultures lack mesenchymal cells, and thus defined exogenously added factors are necessary to support their growth. Enteroids can be used to study a number of different somatic disorders, and many are able to replicate disease phenotypes such as cystic fibrosis25 and cancer2 in vitro. One of the challenges in working with both the 3D organoids and enteroids is access to the apical/luminal side of the cells that faces inward in the 3D structure. Several methods can be used to gain access to the interior compartment of the enclosed 3D structure including microinjection into the lumen directly or mechanical disruption of the enteroids.26, 27 Enteroids also can be plated as two-dimensional cultures in monolayers on culture dishes or on Transwells in which epithelial barrier function can be assessed28, 29 (Figure 1). In addition, monolayer cultures allow ease of access to both the apical and basolateral sides of the epithelium.29, 30, 31
Using Gastrointestinal Organoids and Enteroids to Study Host-Microbial Interactions
Microbiome
The microbiome lies on the apical side of the small intestinal epithelium and has a significant impact on gastrointestinal physiology and host health. Several diseases have been linked to alterations in the small intestinal microbiome including inflammatory bowel disease32 and obesity,33 suggesting there are important molecular and cellular mechanisms that are regulated by commensal organisms. In addition to directly interacting with receptors on the apical surface of the epithelium,34 the commensal organisms produce metabolic products that regulate host physiology35 and provide a means of communication to other organs in the body.36, 37 The microbiome, called a “new organ,” provides a variety of functions such as maintaining intestinal barrier integrity through regulation of tight junction proteins,38 modulating the host immune system,39 affecting host lipid metabolism,40 and inhibiting colonization with pathogenic organisms.40 The localization and density of microbial communities vary within regions of the small intestine (duodenum, jejunum, ileum) as well as from the lumen to the apical surface of the epithelium.41, 42 Although high-throughput DNA sequencing has made strides in determining the quantity and identity of the microbial communities, the molecular effects of specific organisms on the epithelium still remain undefined.
Enteroids and organoids offer many advantages when studying the microbiota. Although systems such as humanized and gnotobiotic mice have provided the foundation for understanding the impact of the microbiome in the context of healthy and disease states, there are significant differences in physiology, anatomy, and microbial composition between these animals and the human intestine.43 Better approaches are needed to make advances in understanding how human physiology is modulated by microbial communities. Host-specific microbiota grown from a single individual, cultivated classically or using novel bioreactors,44 and paired with epithelial cultures from the same individual’s intestine enables the study of patient-specific interventions for many gastrointestinal diseases. Despite these advantages, there have been few to no studies utilizing these culture systems to explore the mechanisms by which individual bacteria impact human epithelial physiology. One barrier is the anaerobic requirement of many bacteria. Anaerobic organisms injected into the lumen of 3D organoids have survived for 12 hours, suggesting the oxygen conditions in 3D cultures may facilitate interactions between commensal bacteria and the small intestine epithelium.45 Commensal Escherichia coli replicated, were completely contained within the lumen, and did not cause damage after microinjection into the lumen of organoids.46 Mucin was proposed as a defense mechanism to contain the non-pathogenic bacteria. Transcriptional analysis of the bacteria-exposed enteroids found upregulation of genes associated with gastrointestinal maturation.
Pathogens
In contrast to the organisms in the lumen of the intestine that maintain a homeostatic relationship with an intact intestinal epithelium and provide beneficial functions, pathogenic organisms use the epithelium as a site of attachment, invasion, and replication, leading to significant pathology and disease in the small intestine. Gastrointestinal infections (viral, bacterial, and parasitic) affect between 76 million (foodborne illness) and 135 million people each year.47 The development of treatments for gastrointestinal infections has been limited by a lack of understanding of the pathogenesis of many human intestinal organisms. Significant numbers of human small intestinal infections are not replicated well in vitro or in animals, limiting the identification and characterization of pathogen receptors, cellular entry pathways, mechanisms of epithelial barrier disruption, and the epithelial response to infection. Other areas that could benefit from increased research include studies on the induction of antimicrobial factors within the epithelium that prevent infection from occurring and the role of human genetic factors that determine susceptibility to infection and disease. New systems will advance the understanding of biological interactions between the epithelium, commensal bacteria, and pathogenic organisms and their relevance to human health.
Small intestinal organoids and enteroids are new approaches to address some of the outstanding questions for many human small intestinal pathogens. Because of the sheer numbers of cultures that can be established from individuals of different genetic backgrounds, organoids and enteroids can address genetically controlled susceptibility of human cells to infection with human pathogens. Factors influencing entry of the pathogen into the intestinal epithelium and cell types important for replication can be determined and evaluated. A better understanding of signaling pathways that comprise the human innate immune response of the epithelium, which is the first immune response triggered by pathogenic infection, is attainable in these systems. These pathways are important targets in the development of therapeutics to treat infected individuals.
Both organoids and enteroids have been used to study the pathogenesis of several enteric viruses (Table 1) including human rotavirus, a common small intestinal viral pathogen that causes dehydrating vomiting and diarrhea in young children.48 Initially, organoid cultures and later enteroid cultures were shown to support direct growth of human rotavirus strains.11, 49, 50, 51 As expected, enterocytes are the predominant cell type targeted by the virus in either culture system. Unexpectedly, rotavirus also infected mesenchymal cells in the organoids, demonstrating a previously unknown cell tropism. Enteroendocrine cells also are infected in the enteroid cultures, and rotavirus-infected or viral enterotoxin-treated enteroid cultures exhibit swelling indicative of a pathophysiological response likely reflecting chloride secretion that is associated with the disease symptom of diarrhea.11 Recent studies demonstrate that susceptibility to rotavirus infection and response to rotavirus vaccines may be mediated by host genetic factors such as the expression of histo-blood group antigens (HBGAs).52 Enteroids from different patients support varying levels of rotavirus replication mimicking differences in susceptibility between different individuals in the population. Exposure to one licensed rotavirus vaccine resulted in lower vaccine replication in specific enteroids when compared with other enteroids, indicating putative host factors that influence vaccine immunogenicity and efficacy.11 Organoid and enteroid cultures provide a new tool in which epidemiologic data on host susceptibility factors can be tested and validated. One explanation for the variation in host response may be the innate immune response mounted by the epithelium. Enteroids respond to human rotavirus infection with a robust innate immune response, with the predominant transcriptional pathway induced by human rotavirus infection being a type III interferon (IFN) response that activates IFN-stimulated genes.51 Enteroids have been used to model antiviral effects of both IFNs and ribavirin against patient-derived rotavirus strains.50 Sensitivity to treatment with these antivirals varied substantially on the basis of enteroid line.50 The importance of this response in limiting epithelial infection remains to be elucidated. Enteroids also can be used to evaluate antivirals for the treatment of rotavirus-infected children and chronically infected transplant patients,53, 54 and may provide new preclinical assays for understanding vaccine attenuation.
Table 1.
Insights Into Human Viral Infections of Human Intestinal Enteroids
| Virus | Enteroid type | Strains | Cells infected | Responses | Innate responses | Reference |
|---|---|---|---|---|---|---|
| Rotavirus | HIOs | Human rotavirus replication | Enterocytes, mesenchymal cells | 49 | ||
| Rotavirus | Differentiated HIEs from adults, all small intestinal segments in 3D cultures | Human rotavirus replicates more efficiently than animal rotavirus | Enterocytes, enteroendocrine cells; not stem cells | Swelling induced by viral infection and enterotoxin treatment | Predominant type III IFN response | 6, 11, 15 |
| Rotavirus | HIEs from adults | Human rotavirus replication | Antiviral testing | Selected innate response genes induced | 50, 53 | |
| Human norovirus | Differentiated HIEs from adults, all small intestinal segments on monolayers | Multiple human norovirus strains replicate; strain-specific requirements for replication; some require bile | Enterocytes; not stem cells | Inactivation and neutralization tested | Host-specific susceptibility to infection based on host HBGA expression | 6, 30 |
| Enteroviruses | HIEs from human fetal small intestine in 3D cultures | Strain-specific differences in relative replication efficiency with EV11 and CBV high and EV71 low | Strain-specific responses | 56 | ||
| Echovirus 11 | Good infections; cpe; cell death; mislocalization of occludin; infectious virus produced in levels similar to Caco-2 cells | Enteroendocrine cells; not goblet cells | Differential induction of 350 transcripts; cytokines, chemokines; IFN-stimulated genes | 56 | ||
| Coxsackievirus B | Good infections by immunofluorescence and viral RNA | Differential induction of 13 transcripts | 56 | |||
| Enterovirus 71 | Lower levels of replication | No significant induction of transcripts detected | 56 |
CBV, Coxsackievirus B; cpe, cytopathic effect; EV11, Echovirus 11; EV71, Enterovirus 71; HIE, human intestinal enteroid; HIO, human intestinal organoid.
Human noroviruses cause 699 million episodes of gastroenteritis and 219,000 deaths globally every year.55 For decades little progress had been made into preventive or therapeutic modalities for norovirus disease. In a landmark discovery, human noroviruses, previously uncultivatable in transformed epithelial cell cultures and small animal models, were demonstrated to replicate in human enteroid cultures.30 Robust and reproducible in vitro cultivation of human noroviruses in enterocytes was seen for multiple virus strains in differentiated cultures from all segments of the small intestine. In addition, norovirus replication in enteroids mimics epidemiologic data on differences in host susceptibility to infection; enteroids derived from individuals who lack a functional fucosyltransferase 2 enzyme to express specific HBGA receptors associated with susceptibility to infection did not support virus replication.30 Unexpectedly, strain-specific requirements were discovered, with the addition of bile being required to support replication in enteroids for some norovirus strains. This system can be used to evaluate and develop antivirals, inactivating agents, and to understand virus infectivity and replication. These studies demonstrate the potential of human enteroid cultures for studying and understanding the host factors and pathogenesis of previously non-cultivatable pathogens.
Other enteric viruses classified as enteroviruses, such as echovirus 11, coxsackievirus B, enterovirus 71, and poliovirus, infect and replicate to different levels in human enteroid cultures produced from human fetal small intestine.56 Transcriptional analysis of echovirus 11–infected enteroids showed differential induction of 350 transcripts, whereas only 13 transcripts were induced after coxsackievirus B infection. The transcripts induced by echovirus included immune factors such as cytokines, chemokines, and IFN-stimulated genes, possibly explaining lower levels of replication compared with coxsackievirus B. Like rotavirus, in addition to infecting enterocytes, echovirus 11 infected enteroendocrine cells. These studies contrast previous work in transformed cell lines that suggested enterovirus did not infect the intestinal epithelium but rather used it as a conduit to reach other target cells.57 Taken together, these studies of viral infections demonstrate human enteroid cultures provide important insights into viral pathogenesis that are not replicated in transformed cell lines.
The pathogenesis of human bacterial infections also has been studied in organoid and enteroid systems. Bacterial pathogens cause significant foodborne gastroenteritis, and many cause their effects in the small intestine. The transcriptional response of organoid cultures was examined after microinjection with salmonella strains58and included cytokine responses such as interleukin (IL)-23A, which is linked to protection of the intestinal barrier,59 and IL8, IL1B, tumor necrosis factor, and CXCL2, which are proinflammatory. Goblet and enteroendocrine cell-associated genes were also upregulated. Imaging approaches documented the attachment of salmonella on the apical surface of the organoid epithelium and classic vacuoles containing the pathogen. Enteroids were used to demonstrate adherence patterns when exposed to different strains of pathogenic E. coli.29 Similar to rotavirus, 3D human enteroids responded to luminal microinjection of enterotoxigenic E coli by swelling representing secretion of fluid.60 The Shiga toxins produced by enterohemorrhagic E coli stimulate toxin micropinocytosis and host actin remodeling.31 Studies using non-foodborne bacteria such as Clostridium difficile that cause infectious diarrhea demonstrated disruption of the epithelial barrier, alterations in transporter expression related to diarrhea, loss of polarity, and reduction of mucous production when organoid cultures were exposed to either the pathogen or its toxins.45, 61, 62 Vibrio cholera toxin elicited a faster increase in cyclic adenosine monophosphate in enteroids expressing blood group O antigens than those that expressed A antigen.63 These studies had an almost perfect parallel to clinical and epidemiologic data showing correlation between O blood group and risk for cholera. Overall, the use of these engineered gastrointestinal culture systems to study human bacterial infections demonstrates the potential for developing systems that allow host factors that contribute to disease pathogenesis caused by a microbial agent to be elucidated.
Approaches using organoid or enteroid cultures to study helminth and protozoa infections have lagged behind those used to study viruses and bacteria. Parasites are a worldwide health problem and cause significant morbidity and mortality affecting close to 3.5 billion people. There are many difficulties in propagating these organisms in vitro including host restriction and complicated life cycles. Major questions still remain for many gastrointestinal parasites including mechanisms of disease, loss of barrier function, and identification of virulence factors. In addition, little is known about host factors that affect infection. Human enteroid and organoid cultures have the potential to make major inroads into addressing some of these questions as they pertain to parasite infections. Once the systems are more established, these models could be used to screen new anti-parasitic drugs.
Challenges to Existing Engineered Gastrointestinal Tissues
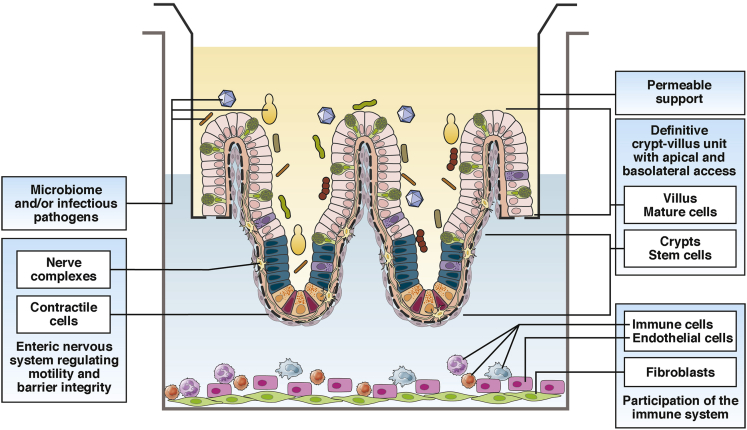
Organoid and enteroid cultures are simple to maintain, offer the unique capability of examining susceptibility to infections within certain genetic populations, and allow comparative analysis between these cultures and human tissue. Most importantly, these systems offer pathways to personalized medicine. Several areas to improve these systems for studies involving beneficial and pathogenic organisms include adding physical organization, immune cells, and neuronal signaling. Moving forward, the evolution of organoids and enteroid cultures will only serve to enhance these methods for use in studying the microbiome and infectious pathogens (Figure 2).
Figure 2.
Future platform development of enteroids and organoids to facilitate studies with microbial organisms.
Advances in culturing suggest that proper temporal organization in a crypt-villus–like unit in a two-dimensional format is an attainable goal12 and one that will be necessary to consider when examining the interactions of many microbial organisms with the epithelium. Currently, organoids and enteroids lack the definitive crypt-villus structure in monolayer format that facilitates the natural route of exposure to the apical surface. Of importance also will be incorporation of the mesenchyme (enteroids) and the mucosal muscular layer, the muscularis mucosa (enteroids and organoids), both of which participate in bidirectional signaling with the epithelium. Another hurdle in culturing these tissues is controlling the variability of reagents. Matrigel and non-commercially produced growth factors are expensive, have lot-to-lot variation, and are not fully defined. As culture conditions become more sophisticated, the development of defined reagents will ensure reproducibility of complex cellular systems.
Organoids and enteroids also lack components of the immune system. The complex interaction between the epithelium, the immune system, and microbes is key to understanding small intestinal physiology. Co-cultures of enteroids with macrophages have been successfully established,64 resulting in enhancement of epithelial barrier function and enteroid maturation. A coordinated response of these co-cultures to exposure with enterotoxigenic and enteropathogenic E coli infections was documented by observing intraepithelial macrophage projections, efficient phagocytosis, and stabilized barrier function. Organoids cultured with neutrophils recapitulate innate cellular responses when exposed to Shiga toxin–producing E coli including cytokine response, loss of epithelial integrity, and activation of stress responses that involve reactive oxygen species.46 These studies provide critical evidence that illustrates the importance of incorporating the immune system into these cultures in the context of studying microbial organisms.
The enteric nervous system also plays a key role in the physiology of infections through its regulation of movement, secretion, fluid flow across the epithelium, and epithelial barrier integrity.65 The myenteric and submucosal plexi have networks of neurons and glia that extend throughout the various intestinal compartments including where they play a critical role in modulating epithelial function. Because of its location and function, the enteric nervous system is thought to modulate responses to insults by triggering pathways that result in diarrhea and vomiting. However, the dissection of these contributions has been difficult, with no current way to study the enteric nervous system in vitro. Recent work by Workman et al66 utilizing a tissue-engineering approach resulted in the generation of normal intestinal enteric nervous system within an organoid culture. Importantly, these neurons functionally regulated waves of propagating contractions. This system shows tremendous promise to dissect the molecular aspects of enteric nervous system–epithelial interactions with both microbiome and pathogenic organisms.
As advances are made in organoid and enteroid cultures, validation will be an important consideration. Variability is an inevitable result of using human tissue as the source of these cultures. Transcriptional analyses of multiple jejunal enteroid cultures revealed that the transcriptional profile clusters first by individual. However, infection of these cultures with human rotavirus revealed a distinct and common transcriptional response,51 indicating that amid the human variability, common biological responses exist that are most likely relevant to understanding host-microbial interactions. Thus research directed toward determining host responses to microbial pathogens or commensals will benefit from validation across multiple organoid or enteroid lines. Analyses in cultures representing biologic replicates will be paramount to discovering and validating global host factors that can then be further tested in patients. Well-characterized biobanks of human intestinal organoids and enteroids generated from multiple individuals and shared among researchers will be required and will provide unique tools to define common host genes and responses involved in host-microbial interactions.
Summary
It is evident that the use of engineered human gastrointestinal organoid and enteroid models that recapitulate the complexity and cellularity of the small intestine is facilitating discoveries and providing new, fundamental knowledge on normal human intestinal physiology and pathophysiology and how the host responds to microbial pathogens and commensals. Application of these models ranges from patient-specific personalized medicine studies to pre-clinical studies for vaccines and therapeutics where the impact of differences in host susceptibility and response is a critical parameter for response assessment. Significant efforts continue to be made in the development of complex intestinal culture systems and will contribute to the long-term goal of developing integrated engineered human tissue models and human organs on a chip to understand gut function in health and disease.
Footnotes
Conflicts of interest The authors disclose the following: M.K. Estes is named as an inventor on patents related to cloning of the Norwalk virus genome and has received consultant's fees from Takeda Vaccines, Inc. The remaining authors disclose no conflicts.
Funding Supported by grants U18-TR000552, UH3-TR00003, U19-AI116497, RO1-AI080656, U01-DK103168, and P30-DK56338 from the National Institutes of Health and the Food Research Initiative Competitive grant 2011-68003-30395 from the U.S. Department of Agriculture, National Institute of Food and Agriculture.
References
- 1.Sato T., Vries R.G., Snippert H.J., van de Wetering M., Barker N., Stange D.E., van Es J.H., Abo A., Kujala P., Peters P.J., Clevers H. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature. 2009;459:262–265. doi: 10.1038/nature07935. [DOI] [PubMed] [Google Scholar]
- 2.Sato T., Stange D.E., Ferrante M., Vries R.G., van Es J.H., van den Brink S., Van Houdt W.J., Pronk A., Van G.J., Siersema P.D., Clevers H. Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett's epithelium. Gastroenterology. 2011;141:1762–1772. doi: 10.1053/j.gastro.2011.07.050. [DOI] [PubMed] [Google Scholar]
- 3.Spence J.R., Mayhew C.N., Rankin S.A., Kuhar M.F., Vallance J.E., Tolle K., Hoskins E.E., Kalinichenko V.V., Wells S.I., Zorn A.M., Shroyer N.F., Wells J.M. Directed differentiation of human pluripotent stem cells into intestinal tissue in vitro. Nature. 2011;470:105–109. doi: 10.1038/nature09691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Stelzner M., Helmrath M., Dunn J.C., Henning S.J., Houchen C.W., Kuo C., Lynch J., Li L., Magness S.T., Martin M.G., Wong M.H., Yu J., NIH Intestinal Stem Cell Consortium A nomenclature for intestinal in vitro cultures. Am J Physiol Gastrointest Liver Physiol. 2012;302:G1359–G1363. doi: 10.1152/ajpgi.00493.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sato T., van Es J.H., Snippert H.J., Stange D.E., Vries R.G., van den Born M., Barker N., Shroyer N.F., van de Wetering M., Clevers H. Paneth cells constitute the niche for Lgr5 stem cells in intestinal crypts. Nature. 2011;469:415–418. doi: 10.1038/nature09637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zou W.Y., Blutt S.E., Crawford S.E., Ettayebi K., Zeng X.L., Saxena K., Ramani S., Karandikar U.C., Zachos N.C., Estes M.K. Human intestinal enteroids: new models to study gastrointestinal virus infections. Methods Mol Biol. 2017;15:1–19. doi: 10.1007/7651_2017_1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.de Lau W., Kujala P., Schneeberger K., Middendorp S., Li V.S., Barker N., Martens A., Hofhuis F., DeKoter R.P., Peters P.J., Nieuwenhuis E., Clevers H. Peyer's patch M cells derived from Lgr5(+) stem cells require SpiB and are induced by RankL in cultured “miniguts”. Mol Cell Biol. 2012;32:3639–3647. doi: 10.1128/MCB.00434-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rouch J.D., Scott A., Lei N.Y., Solorzano-Vargas R.S., Wang J., Hanson E.M., Kobayashi M., Lewis M., Stelzner M.G., Dunn J.C., Eckmann L., Martin M.G. Development of functional microfold (M) cells from intestinal stem cells in primary human enteroids. PLoS One. 2016;11:e0148216. doi: 10.1371/journal.pone.0148216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Basak O., Beumer J., Wiebrands K., Seno H., van O.A., Clevers H. Induced quiescence of Lgr5+ stem cells in intestinal organoids enables differentiation of hormone-producing enteroendocrine cells. Cell Stem Cell. 2017;20:177–190. doi: 10.1016/j.stem.2016.11.001. [DOI] [PubMed] [Google Scholar]
- 10.Liu J., Walker N.M., Cook M.T., Ootani A., Clarke L.L. Functional Cftr in crypt epithelium of organotypic enteroid cultures from murine small intestine. Am J Physiol Cell Physiol. 2012;302:C1492–C1503. doi: 10.1152/ajpcell.00392.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Saxena K., Blutt S.E., Ettayebi K., Zeng X.L., Broughman J.R., Crawford S.E., Karandikar U.C., Sastri N.P., Conner M.E., Opekun A.R., Graham D.Y., Qureshi W., Sherman V., Foulke-Abel J., In J., Kovbasnjuk O., Zachos N.C., Donowitz M., Estes M.K. Human intestinal enteroids: a new model to study human rotavirus infection, host restriction, and pathophysiology. J Virol. 2015;90:43–56. doi: 10.1128/JVI.01930-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang Y., Gunasekara D.B., Reed M.I., DiSalvo M., Bultman S.J., Sims C.E., Magness S.T., Allbritton N.L. A microengineered collagen scaffold for generating a polarized crypt-villus architecture of human small intestinal epithelium. Biomaterials. 2017;128:44–55. doi: 10.1016/j.biomaterials.2017.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schwank G., Koo B.K., Sasselli V., Dekkers J.F., Heo I., Demircan T., Sasaki N., Boymans S., Cuppen E., van der Ent C.K., Nieuwenhuis E.E., Beekman J.M., Clevers H. Functional repair of CFTR by CRISPR/Cas9 in intestinal stem cell organoids of cystic fibrosis patients. Cell Stem Cell. 2013;13:653–658. doi: 10.1016/j.stem.2013.11.002. [DOI] [PubMed] [Google Scholar]
- 14.Foulke-Abel J., In J., Yin J., Zachos N.C., Kovbasnjuk O., Estes M.K., de J.H., Donowitz M. Human enteroids as a model of upper small intestinal ion transport physiology and pathophysiology. Gastroenterology. 2016;150:638–649. doi: 10.1053/j.gastro.2015.11.047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Koo B.K., Stange D.E., Sato T., Karthaus W., Farin H.F., Huch M., van Es J.H., Clevers H. Controlled gene expression in primary Lgr5 organoid cultures. Nat Methods. 2011;9:81–83. doi: 10.1038/nmeth.1802. [DOI] [PubMed] [Google Scholar]
- 16.Yilmaz O.H., Katajisto P., Lamming D.W., Gultekin Y., Bauer-Rowe K.E., Sengupta S., Birsoy K., Dursun A., Yilmaz V.O., Selig M., Nielsen G.P., Mino-Kenudson M., Zukerberg L.R., Bhan A.K., Deshpande V., Sabatini D.M. mTORC1 in the Paneth cell niche couples intestinal stem-cell function to calorie intake. Nature. 2012;486:490–495. doi: 10.1038/nature11163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Watson C.L., Mahe M.M., Munera J., Howell J.C., Sundaram N., Poling H.M., Schweitzer J.I., Vallance J.E., Mayhew C.N., Sun Y., Grabowski G., Finkbeiner S.R., Spence J.R., Shroyer N.F., Wells J.M., Helmrath M.A. An in vivo model of human small intestine using pluripotent stem cells. Nat Med. 2014;20:1310–1314. doi: 10.1038/nm.3737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.McCracken K.W., Cata E.M., Crawford C.M., Sinagoga K.L., Schumacher M., Rockich B.E., Tsai Y.H., Mayhew C.N., Spence J.R., Zavros Y., Wells J.M. Modelling human development and disease in pluripotent stem-cell-derived gastric organoids. Nature. 2014;516:400–404. doi: 10.1038/nature13863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Takasato M., Er P.X., Chiu H.S., Maier B., Baillie G.J., Ferguson C., Parton R.G., Wolvetang E.J., Roost M.S., Chuva de Sousa Lopes S.M., Little M.H. Kidney organoids from human iPS cells contain multiple lineages and model human nephrogenesis. Nature. 2015;526:564–568. doi: 10.1038/nature15695. [DOI] [PubMed] [Google Scholar]
- 20.McCracken K.W., Howell J.C., Wells J.M., Spence J.R. Generating human intestinal tissue from pluripotent stem cells in vitro. Nat Protoc. 2011;6:1920–1928. doi: 10.1038/nprot.2011.410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Finkbeiner S.R., Hill D.R., Altheim C.H., Dedhia P.H., Taylor M.J., Tsai Y.H., Chin A.M., Mahe M.M., Watson C.L., Freeman J.J., Nattiv R., Thomson M., Klein O.D., Shroyer N.F., Helmrath M.A., Teitelbaum D.H., Dempsey P.J., Spence J.R. Transcriptome-wide analysis reveals hallmarks of human intestine development and maturation in vitro and in vivo. Stem Cell Reports. 2015;4:1140–1155. doi: 10.1016/j.stemcr.2015.04.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Aurora M., Spence J.R. hPSC-derived lung and intestinal organoids as models of human fetal tissue. Dev Biol. 2016;420:230–238. doi: 10.1016/j.ydbio.2016.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Tsai Y.H., Nattiv R., Dedhia P.H., Nagy M.S., Chin A.M., Thomson M., Klein O.D., Spence J.R. In vitro patterning of pluripotent stem cell-derived intestine recapitulates in vivo human development. Development. 2017;144:1045–1055. doi: 10.1242/dev.138453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Middendorp S., Schneeberger K., Wiegerinck C.L., Mokry M., Akkerman R.D., van W.S., Clevers H., Nieuwenhuis E.E. Adult stem cells in the small intestine are intrinsically programmed with their location-specific function. Stem Cells. 2014;32:1083–1091. doi: 10.1002/stem.1655. [DOI] [PubMed] [Google Scholar]
- 25.Dekkers J.F., Wiegerinck C.L., de Jonge H.R., Bronsveld I., Janssens H.M., de Winter-de Groot K.M., Brandsma A.M., de Jong N.W., Bijvelds M.J., Scholte B.J., Nieuwenhuis E.E., van den Brink S., Clevers H., van der Ent C.K., Middendorp S., Beekman J.M. A functional CFTR assay using primary cystic fibrosis intestinal organoids. Nat Med. 2013;19:939–945. doi: 10.1038/nm.3201. [DOI] [PubMed] [Google Scholar]
- 26.Dutta D., Heo I., Clevers H. Disease modeling in stem cell-derived 3D organoid systems. Trends Mol Med. 2017;23:393–410. doi: 10.1016/j.molmed.2017.02.007. [DOI] [PubMed] [Google Scholar]
- 27.Bartfeld S., Clevers H. Organoids as model for infectious diseases: culture of human and murine stomach organoids and microinjection of Helicobacter pylori. J Vis Exp. 2015;105:53359. doi: 10.3791/53359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Moon C., VanDussen K.L., Miyoshi H., Stappenbeck T.S. Development of a primary mouse intestinal epithelial cell monolayer culture system to evaluate factors that modulate IgA transcytosis. Mucosal Immunol. 2014;7:818–828. doi: 10.1038/mi.2013.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.VanDussen K.L., Marinshaw J.M., Shaikh N., Miyoshi H., Moon C., Tarr P.I., Ciorba M.A., Stappenbeck T.S. Development of an enhanced human gastrointestinal epithelial culture system to facilitate patient-based assays. Gut. 2015;64:911–920. doi: 10.1136/gutjnl-2013-306651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Ettayebi K., Crawford S.E., Murakami K., Broughman J.R., Karandikar U., Tenge V.R., Neill F.H., Blutt S.E., Zeng X.L., Qu L., Kou B., Opekun A.R., Burrin D., Graham D.Y., Ramani S., Atmar R.L., Estes M.K. Replication of human noroviruses in stem cell-derived human enteroids. Science. 2016;353:1387–1393. doi: 10.1126/science.aaf5211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.In J., Foulke-Abel J., Zachos N.C., Hansen A.M., Kaper J.B., Bernstein H.D., Halushka M., Blutt S., Estes M.K., Donowitz M., Kovbasnjuk O. Enterohemorrhagic Escherichia coli reduce mucus and intermicrovillar bridges in human stem cell-derived colonoids. Cell Mol Gastroenterol Hepatol. 2016;2:48–62. doi: 10.1016/j.jcmgh.2015.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Forbes J.D., Van D.G., Bernstein C.N. The gut microbiota in immune-mediated inflammatory diseases. Front Microbiol. 2016;7:1081. doi: 10.3389/fmicb.2016.01081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Turnbaugh P.J., Ley R.E., Mahowald M.A., Magrini V., Mardis E.R., Gordon J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature. 2006;444:1027–1031. doi: 10.1038/nature05414. [DOI] [PubMed] [Google Scholar]
- 34.Earle K.A., Billings G., Sigal M., Lichtman J.S., Hansson G.C., Elias J.E., Amieva M.R., Huang K.C., Sonnenburg J.L. Quantitative imaging of gut microbiota spatial organization. Cell Host Microbe. 2015;18:478–488. doi: 10.1016/j.chom.2015.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Geva-Zatorsky N., Alvarez D., Hudak J.E., Reading N.C., Erturk-Hasdemir D., Dasgupta S., von Andrian U.H., Kasper D.L. In vivo imaging and tracking of host-microbiota interactions via metabolic labeling of gut anaerobic bacteria. Nat Med. 2015;21:1091–1100. doi: 10.1038/nm.3929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Nicholson J.K., Holmes E., Kinross J., Burcelin R., Gibson G., Jia W., Pettersson S. Host-gut microbiota metabolic interactions. Science. 2012;336:1262–1267. doi: 10.1126/science.1223813. [DOI] [PubMed] [Google Scholar]
- 37.Vogt S.L., Pena-Diaz J., Finlay B.B. Chemical communication in the gut: effects of microbiota-generated metabolites on gastrointestinal bacterial pathogens. Anaerobe. 2015;34:106–115. doi: 10.1016/j.anaerobe.2015.05.002. [DOI] [PubMed] [Google Scholar]
- 38.Bansal T., Alaniz R.C., Wood T.K., Jayaraman A. The bacterial signal indole increases epithelial-cell tight-junction resistance and attenuates indicators of inflammation. Proc Natl Acad Sci. 2010;107:228–233. doi: 10.1073/pnas.0906112107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Zelante T., Iannitti R.G., Cunha C., De Luca A., Giovannini G., Pieraccini G., Zecchi R., D'Angelo C., Massi-Benedetti C., Fallarino F., Carvalho A., Puccetti P., Romani L. Tryptophan catabolites from microbiota engage aryl hydrocarbon receptor and balance mucosal reactivity via interleukin-22. Immunity. 2013;39:372–385. doi: 10.1016/j.immuni.2013.08.003. [DOI] [PubMed] [Google Scholar]
- 40.Buffie C.G., Bucci V., Stein R.R., McKenney P.T., Ling L., Gobourne A., No D., Liu H., Kinnebrew M., Viale A., Littmann E., van den Brink M.R., Jenq R.R., Taur Y., Sander C., Cross J.R., Toussaint N.C., Xavier J.B., Pamer E.G. Precision microbiome reconstitution restores bile acid mediated resistance to Clostridium difficile. Nature. 2015;517:205–208. doi: 10.1038/nature13828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Albenberg L., Esipova T.V., Judge C.P., Bittinger K., Chen J., Laughlin A., Grunberg S., Baldassano R.N., Lewis J.D., Li H., Thom S.R., Bushman F.D., Vinogradov S.A., Wu G.D. Correlation between intraluminal oxygen gradient and radial partitioning of intestinal microbiota. Gastroenterology. 2014;147:1055–1063. doi: 10.1053/j.gastro.2014.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Vaishnava S., Yamamoto M., Severson K.M., Ruhn K.A., Yu X., Koren O., Ley R., Wakeland E.K., Hooper L.V. The antibacterial lectin RegIIIgamma promotes the spatial segregation of microbiota and host in the intestine. Science. 2011;334:255–258. doi: 10.1126/science.1209791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Treuting P.M., Clifford C.B., Sellers R.S., Brayton C.F. Of mice and microflora: considerations for genetically engineered mice. Vet Pathol. 2012;49:44–63. doi: 10.1177/0300985811431446. [DOI] [PubMed] [Google Scholar]
- 44.Auchtung J.M., Robinson C.D., Britton R.A. Cultivation of stable, reproducible microbial communities from different fecal donors using minibioreactor arrays (MBRAs) Microbiome. 2015;3:42. doi: 10.1186/s40168-015-0106-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Leslie J.L., Huang S., Opp J.S., Nagy M.S., Kobayashi M., Young V.B., Spence J.R. Persistence and toxin production by Clostridium difficile within human intestinal organoids result in disruption of epithelial paracellular barrier function. Infect Immun. 2015;83:138–145. doi: 10.1128/IAI.02561-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Karve S.S., Pradhan S., Ward D.V., Weiss A.A. Intestinal organoids model human responses to infection by commensal and Shiga toxin producing Escherichia coli. PLoS One. 2017;12:e0178966. doi: 10.1371/journal.pone.0178966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Sandler R.S., Everhart J.E., Donowitz M., Adams E., Cronin K., Goodman C., Gemmen E., Shah S., Avdic A., Rubin R. The burden of selected digestive diseases in the United States. Gastroenterology. 2002;122:1500–1511. doi: 10.1053/gast.2002.32978. [DOI] [PubMed] [Google Scholar]
- 48.Greenberg H.B., Estes M.K. Rotaviruses: from pathogenesis to vaccination. Gastroenterology. 2009;136:1939–1951. doi: 10.1053/j.gastro.2009.02.076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Finkbeiner S.R., Zeng X.L., Utama B., Atmar R.L., Shroyer N.F., Estes M.K. Stem cell-derived human intestinal organoidsas an infection model for rotaviruses. MBio. 2012;3 doi: 10.1128/mBio.00159-12. e00159-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Yin Y., Bijvelds M., Dang W., Xu L., van der Eijk A.A., Knipping K., Tuysuz N., Dekkers J.F., Wang Y., de J.J., Sprengers D., van der Laan L.J., Beekman J.M., Ten B.D., Metselaar H.J., de J.H., Koopmans M.P., Peppelenbosch M.P., Pan Q. Modeling rotavirus infection and antiviral therapy using primary intestinal organoids. Antiviral Res. 2015;123:120–131. doi: 10.1016/j.antiviral.2015.09.010. [DOI] [PubMed] [Google Scholar]
- 51.Saxena K., Simon L.M., Zeng X.L., Blutt S.E., Crawford S.E., Sastri N.P., Karandikar U.C., Ajami N.J., Zachos N.C., Kovbasnjuk O., Donowitz M., Conner M.E., Shaw C.A., Estes M.K. A paradox of transcriptional and functional innate interferon responses of human intestinal enteroids to enteric virus infection. Proc Natl Acad Sci. 2017;114:E570–E579. doi: 10.1073/pnas.1615422114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Hu L., Crawford S.E., Czako R., Cortes-Penfield N.W., Smith D.F., Le P.J., Estes M.K., Prasad B.V. Cell attachment protein VP8* of a human rotavirus specifically interacts with A-type histo-blood group antigen. Nature. 2012;485:256–259. doi: 10.1038/nature10996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Yin Y., Wang Y., Dang W., Xu L., Su J., Zhou X., Wang W., Felczak K., van der Laan L.J., Pankiewicz K.W., van der Eijk A.A., Bijvelds M., Sprengers D., de Jonge H., Koopmans M.P., Metselaar H.J., Peppelenbosch M.P., Pan Q. Mycophenolic acid potently inhibits rotavirus infection with a high barrier to resistance development. Antiviral Res. 2016;133:41–49. doi: 10.1016/j.antiviral.2016.07.017. [DOI] [PubMed] [Google Scholar]
- 54.Yin Y., Metselaar H.J., Sprengers D., Peppelenbosch M.P., Pan Q. Rotavirus in organ transplantation: drug-virus-host interactions. Am J Transplant. 2015;15:585–593. doi: 10.1111/ajt.13135. [DOI] [PubMed] [Google Scholar]
- 55.Cortes-Penfield N.W., Ramani S., Estes M.K., Atmar R.L. Prospects and challenges in the development of a norovirus vaccine. Clin Ther. 2017;39:1537–1549. doi: 10.1016/j.clinthera.2017.07.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Drummond C.G., Bolock A.M., Ma C., Luke C.J., Good M., Coyne C.B. Enteroviruses infect human enteroids and induce antiviral signaling in a cell lineage-specific manner. Proc Natl Acad Sci. 2017;114:1672–1677. doi: 10.1073/pnas.1617363114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Sicinski P., Rowinski J., Warchol J.B., Jarzabek Z., Gut W., Szczygiel B., Bielecki K., Koch G. Poliovirus type 1 enters the human host through intestinal M cells. Gastroenterology. 1990;98:56–58. doi: 10.1016/0016-5085(90)91290-m. [DOI] [PubMed] [Google Scholar]
- 58.Forbester J.L., Goulding D., Vallier L., Hannan N., Hale C., Pickard D., Mukhopadhyay S., Dougan G. Interaction of Salmonella enterica Serovar Typhimurium with intestinal organoids derived from human induced pluripotent stem cells. Infect Immun. 2015;83:2926–2934. doi: 10.1128/IAI.00161-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Macho-Fernandez E., Koroleva E.P., Spencer C.M., Tighe M., Torrado E., Cooper A.M., Fu Y.X., Tumanov A.V. Lymphotoxin beta receptor signaling limits mucosal damage through driving IL-23 production by epithelial cells. Mucosal Immunol. 2015;8:403–413. doi: 10.1038/mi.2014.78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Pattison A.M., Blomain E.S., Merlino D.J., Wang F., Crissey M.A., Kraft C.L., Rappaport J.A., Snook A.E., Lynch J.P., Waldman S.A. Intestinal enteroids model guanylate cyclase C-dependent secretion induced by heat-stable enterotoxins. Infect Immun. 2016;84:3083–3091. doi: 10.1128/IAI.00639-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Engevik M.A., Yacyshyn M.B., Engevik K.A., Wang J., Darien B., Hassett D.J., Yacyshyn B.R., Worrell R.T. Human Clostridium difficile infection: altered mucus production and composition. Am J Physiol Gastrointest Liver Physiol. 2015;308:G510–G524. doi: 10.1152/ajpgi.00091.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wang X., Yamamoto Y., Wilson L.H., Zhang T., Howitt B.E., Farrow M.A., Kern F., Ning G., Hong Y., Khor C.C., Chevalier B., Bertrand D., Wu L., Nagarajan N., Sylvester F.A., Hyams J.S., Devers T., Bronson R., Lacy D.B., Ho K.Y., Crum C.P., McKeon F., Xian W. Cloning and variation of ground state intestinal stem cells. Nature. 2015;522:173–178. doi: 10.1038/nature14484. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Kuhlmann F.M., Santhanam S., Kumar P., Luo Q., Ciorba M.A., Fleckenstein J.M. Blood group O-dependent cellular responses to cholera toxin: parallel clinical and epidemiological links to severe cholera. Am J Trop Med Hyg. 2016;95:440–443. doi: 10.4269/ajtmh.16-0161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Noel G., Baetz N.W., Staab J.F., Donowitz M., Kovbasnjuk O., Pasetti M.F., Zachos N.C. A primary human macrophage-enteroid co-culture model to investigate mucosal gut physiology and host-pathogen interactions. Sci Rep. 2017;7:45270. doi: 10.1038/srep45270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Furness J.B. The enteric nervous system and neurogastroenterology. Nat Rev Gastroenterol Hepatol. 2012;9:286–294. doi: 10.1038/nrgastro.2012.32. [DOI] [PubMed] [Google Scholar]
- 66.Workman M.J., Mahe M.M., Trisno S., Poling H.M., Watson C.L., Sundaram N., Chang C.F., Schiesser J., Aubert P., Stanley E.G., Elefanty A.G., Miyaoka Y., Mandegar M.A., Conklin B.R., Neunlist M., Brugmann S.A., Helmrath M.A., Wells J.M. Engineered human pluripotent-stem-cell-derived intestinal tissues with a functional enteric nervous system. Nat Med. 2017;23:49–59. doi: 10.1038/nm.4233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Mills M., Estes M.K. Physiologically relevant human tissue models for infectious diseases. Drug Discov Today. 2016;21:1540–1552. doi: 10.1016/j.drudis.2016.06.020. [DOI] [PMC free article] [PubMed] [Google Scholar]