Abstract
The relationship between intestinal stem cells (ISCs) and the surrounding niche environment is complex and dynamic. Key factors localized at the base of the crypt are necessary to promote ISC self-renewal and proliferation, to ultimately provide a constant stream of differentiated cells to maintain the epithelial barrier. These factors diminish as epithelial cells divide, migrate away from the crypt base, differentiate into the postmitotic lineages, and end their life span in approximately 7 days when they are sloughed into the intestinal lumen. To facilitate the rapid and complex physiology of ISC-driven epithelial renewal, in vivo gradients of growth factors, extracellular matrix, bacterial products, gases, and stiffness are formed along the crypt-villus axis. New bioengineered tools and platforms are available to recapitulate various gradients and support the stereotypical cellular responses associated with these gradients. Many of these technologies have been paired with primary small intestinal and colonic epithelial cells to re-create select aspects of normal physiology or disease states. These biomimetic platforms are becoming increasingly sophisticated with the rapid discovery of new niche factors and gradients. These advancements are contributing to the development of high-fidelity tissue constructs for basic science applications, drug screening, and personalized medicine applications. Here, we discuss the direct and indirect evidence for many of the important gradients found in vivo and their successful application to date in bioengineered in vitro models, including organ-on-chip and microfluidic culture devices.
Keywords: Intestinal Epithelial Cells, Stem Cell Niche, Gradients, Bioengineering
Abbreviations used in this paper: 3D, 3-dimensional; BMP, Bone morphogenetic protein; ECM, extracellular matrix; Eph, erythropoietin-producing human hepatocellular receptor; Ephrin, Eph family receptor interacting proteins; IFN-γ, interferon-γ; ISC, intestinal stem cell; NO, nitric oxide; SFCA, short-chain fatty acids; TA, transit amplifying; Wnt, wingless-related integration site
Summary.
Gradients of ligands, growth factors, receptors, extracellular matrix, metabolites, and gases along the crypt (colon) and crypt-villus (small intestine) axis drive maintenance of intestinal stem cells, orderly differentiation, and movement of epithelial cells from the intestinal stem cell niche to the luminal intestinal epithelium. Advances in biomaterials and microdevices enable reconstruction of this complex microenvironment, replicating the key architectural features and physiological functions of the in vivo intestinal epithelium.
The phrase stem cell niche refers to a specific anatomic tissue location that provides a microenvironment enabling intestinal stem cells (ISCs) to remain in an undifferentiated state and promote self-renewal.1, 2, 3 The intestinal epithelium represents one of the most well-characterized stem cell niches, with recent studies that use fluorescent reporter genes, lineage tracing transgenic mouse models, and single-cell transcriptomics defining epithelial cell signatures, behaviors, and function at unprecedented cellular resolution.1, 2, 4, 5, 6 The intestinal epithelium undergoes rapid and continuous stem cell–driven renewal during homeostasis, and the fine balance between ISC maintenance and lineage allocation must be finely regulated to maintain the epithelial barrier and intestinal health. In both the small intestine and colon, ISCs reside at the base of the crypts, which are microanatomic units of epithelial monolayers that invaginate into the luminal wall (Figure 1).2 In the small intestine, crypts are present in tightly packed arrays that feed cells into luminal protrusions called villi, which increase the surface area for nutrient absorption. In the colon, crypts also are present in densely packed arrays, but feed cells onto a flat luminal surface. Although there are functional differences between the small intestine and colon, remarkable similarities exist in the ordered arrangement of crypts, for example, the location of the stem cell zone at the base of the crypt, and the differentiation and migration pattern of epithelial cells.
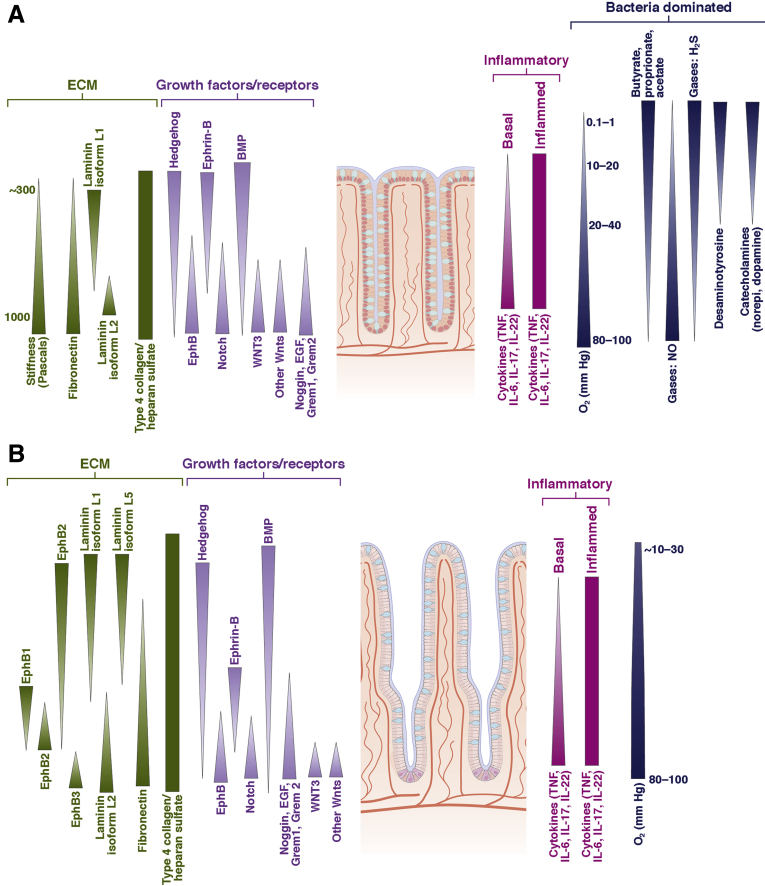
Figure 1.
Gradients in the intestine in vivo. (A) The large intestine possesses chemical gradients spanning the crypt long axis. These gradients include ECM proteins, growth factors and their receptors, inflammatory mediators, and microbial metabolites. Numerous gaseous gradients also are present including oxygen from the arterial system, NO from inflammatory cells and the vasculature, and microbial-derived gases such as H2S and S2O. (B) A multitude of gradients span the crypt/villus long axis in the small intestine. The concentration of ECM proteins, growth factors and their receptors, and inflammatory mediators varies along the length of the crypt/villus unit. The oxygen gradient is much shallower than that of the large intestine because of the absence of the vast numbers of oxygen-scavenging microbes. Because microbial numbers are greatly decreased relative to that in the large intestine, few microbial metabolite gradients have been characterized. (B and C) The shaded triangles show only the gradient direction because the quantitative shape of the gradient is unknown. EGF, epidermal growth factor; IL, interleukin; TNF, tumor necrosis factor.
ISCs divide to produce progenitor cells known as transit-amplifying (TA) cells, which reside above the ISCs within the crypt. The TA cells undergo several additional cell divisions as they migrate upward along the crypt axis and their progeny terminally differentiate into a variety of cell lineages. Absorptive enterocytes represent the majority of cells in the small intestine, while a host of secretory lineages including goblet, enteroendocrine, tuft, and M cells contribute to the functional epithelium. When these cells reach the villus tip in the small intestine or flat luminal surface in the colon, they undergo anoikis and exfoliate into the intestinal lumen to finish a self-renewal cycle that lasts approximately 3–5 days for mice and 5–7 days for human beings.2, 3 An exception to the upward migration of differentiated epithelial cells is the secretory Paneth cell in the small intestine and a Paneth-like cell (cKit+) cell in the colon, which remains at the crypt base intercalated among ISCs.7 These epithelial cells secrete growth factors and present ligands at the base of the crypt to support ISC maintenance-forming gradients of these molecules along the crypt long axis.4 Additional gradients, including ligands, other growth factors, receptors, extracellular matrices, metabolites, and gases, along the epithelial axis drive the ordered differentiation and movement of cells from the proliferative niche at the base of the crypt to the differentiated epithelium in contact with the intestinal lumen (Figure 1, Table 1).5, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19
Table 1.
Example Gradients of the Crypt or Crypt/Villus Axis
| Chemical | Supply (source)a | Removal (sink)a | Reference |
|---|---|---|---|
| EphB | ISCs and TA cells | – | 9 |
| EphrinB | Differentiated epithelial cells | – | 9 |
| Dll1/4 | Crypt/villus epithelial cells | – | 145 |
| BMP | Intravillus and intercrypt mesenchymal cells | Epithelial cells | 49 |
| Wnt3 | Paneth cells, myofibroblasts, mesenchymal cells | ISCs | 30, 36, 39 |
| Noggin | Mesenchymal cells below crypt | Crypt epithelial cells | 146 |
| EGF | Paneth cell | Epithelial cells | 12 |
| Grem1, Grem2 | Mesenchymal cells below crypt | Epithelial cells | 16 |
| Cytokines | Immune cells | Epithelial cells | 67 |
| O2 | Vasculature | Epithelial cells, stroma, luminal microbiota | 85, 86 |
| NO | Vasculature, immune cells | Reaction with oxygen | 85, 91 |
| Other gases (H2S, SO2, H2, others) | Luminal microbiota | – | 85, 94, 147, 148 |
| Butyrate | Luminal microbiota | Differentiated epithelial cells | 79 |
| Proprionate | Luminal microbiota | Blood stream/liver | 149, 150 |
| Acetate | Luminal microbiota | Blood stream | 149, 151 |
EGF, epidermal growth factor.
Nonexhaustive examples are provided.
Although rodent models laid the foundation for understanding ISC biology and the niche in vivo, 3-dimensional (3D) organoid and monolayer models of the small intestinal and colonic epithelium have fueled progress toward in vitro recapitulation of the ISC niche microenvironment to study both epithelial function and pathology.5, 20, 21, 22, 23, 24, 25, 26, 27 However, conventional organoid and monolayer culture systems do not fully recapitulate the microarchitecture of gut epithelium and cannot support the formation of gradients across geometric structures because of the nature of conventional culture systems. To develop high-fidelity, physiologically relevant in vitro models, new culture systems need to incorporate these in vivo gradients. Although there is clear evidence for factor gradients that drive gut epithelial dynamics, visualizing, measuring, and re-creating these gradients has historically been technically challenging. This review focuses on the direct and indirect evidence for in vivo gradients that impact ISC biology and gut epithelial dynamics, and then presents the current state-of-the-art technologies and platforms for the in vitro culture of gut epithelium, particularly as they relate to lab-on-chip and microfluidic culture devices.
In Vivo Factors and Gradients
Growth factor gradients commonly are associated with fundamental mechanisms that underlie ISC maintenance and differentiation. Paneth cells have been the focus of much attention as an ISC niche cell by secreting factors that set up gradients to regulate stemness. A number of studies have shown that Paneth cells are dispensable for ISC maintenance and suggest that other niche cells generate factor gradients that function similar to those set up by Paneth cells.28, 29 In this regard, the underlying mesenchyme releases diffusible factors and also deposits nondiffusible extracellular matrix (ECM) that can present ligands and bind factors to regulate ISC dynamics.30, 31 These studies have merely scratched the surface on a complex cellular and molecular balance that is required to maintain the ISC-driven renewal of the epithelium. The full complement of growth factors, ECM components, and cell types involved in regenerating the epithelial monolayer are not yet fully appreciated. Moreover, noncanonical gradients such as tissue stiffness, gases, and microbial metabolites likely play critical roles but are technically difficult to study in vivo. Understanding these physical properties is of substantial interest to those investigating the broad and diverse factors that regulate gut biology, and is essential for efforts to engineer functional intestinal and colonic tissues. The following section is a brief review of the current state of in vivo gradients and highlights gaps in knowledge as new avenues for investigation.
Key Pathways Regulating ISC Maintenance and Differentiation
Modern advances in understanding ISC biology are based largely on studies that define genetic pathways and mechanisms that govern ISC maintenance and differentiation. Among these are the wingless-related integration site (Wnt), bone morphogenetic protein (BMP), and Notch pathways, which classically are studied as key contributors to epithelial renewal in homeostasis, disease, and injury. Arguably, the Wnt/β-catenin signaling pathway has been a central focus of studies that have heavily influenced the current state in the field.4, 15, 20 Wnts are secreted ligands that bind their cognate receptors and function to regulate ISC maintenance and differentiation. Sox9 is largely a downstream Wnt target gene and shows a distinct expression gradient with higher expression at the base of the crypt in the ISC zone and lower expression through the TA zone, suggesting that Wnt signaling also is present in a gradient that mimics its downstream target genes.32, 33, 34, 35 In fact, 9 Wnts are expressed in the small intestine of mice and are regionally expressed along the crypt-villus axis.30 Contrary to popular assumptions, it appears that Wnt3 gradients may be formed not by simple diffusion, but rather by “plasma membrane dilution” as cells divide.36 A Wnt3-enhanced green fluorescent protein (EGFP) fusion transgenic mouse model enabled visualization of Wnt3 expression by proxy and showed high Wnt3 expression at Paneth cells, which produce Wnt3, and lower expression up the crypt axis (Table 1).36 Paneth cell–derived Wnt transfer involves direct contact between Paneth cells, which previously was suggested by in vitro ISC-Paneth cell co-cultures.37 However, it remains to be determined whether all 9 Wnts establish gradients from their cellular sources.38, 39 Complete understanding of Wnt gradient formation is challenging because there are many sources of Wnts and Wnt antagonists, including subepithelial myofibroblasts and nonmyofibroblast mesenchymal cells expressing Foxl1, Gli1, and CD34 within the ISC niche.13, 14, 40, 41, 42 In addition, potentiation of Wnt signaling by the R-spondin family of secreted factors recently was implicated as a major orchestrator of ISC maintenance and proliferation and suggests that R-spondin gradients also may play a role in ISC dynamics.43
Aside from direct regulation of stem cell maintenance and differentiation, Wnt gradients are essential for physiologic cell compartmentalization and migration by regulating proper cell sorting along the crypt-villus axis. Cell sorting in the epithelial monolayer is regulated in a Wnt-dependent manner through the erythropoietin-producing human hepatocellular receptor (Eph)/Eph family receptor interacting proteins (Ephrin) receptor/ligand pairs, in which expression of EphB2 and EphB3 receptors and their ligand Ephrin-B1 are expressed inversely (Figure 1, Table 1).9, 44, 45 Genetic disruption of the EphB2/B3 receptors results in defects in cell sorting and manifests as a loss of cell compartmentalization with proliferative and differentiated cells intermingled. For instance, loss of EphB3 alone results in defective Paneth cell sorting observed by scattering of Paneth cells along the crypt-villus axis.9
BMP gradients are found in an inverse orientation to Wnt gradients along the crypt axis, and correlate with reduced proliferative capacity and increased differentiation as cells migrate away from low BMP at the crypt base toward higher BMP at the crypt-villus junction (Figure 1, Table 1).16, 46 During development, hedgehog signaling occurs in localized regions, driving a BMP gradient to pattern differentiated cells along the developing villi.8, 47 BMP signaling is required for intestinal homeostasis because BMP inhibition causes abnormal villus morphogenesis, epithelial hyperplasia, and ectopic crypt formation.48 In the intestine, BMP2 and BMP4 are secreted by intravillus and intercrypt mesenchymal cells,49 whereas the BMP antagonists noggin and gremlin 1 are expressed mainly in mesenchymal cells beneath the crypt.46 Together these BMP agonist/antagonist gradients contribute to regulating the ratio of ISC self-renewal properties and differentiation along the crypt-villus axis (Figure 1).
ISCs are located in the crypt base and persistent Notch signaling is required for their maintenance.50 Differentiation of ISCs into secretory lineages occurs when Notch signaling is reduced. Notch is a membrane-bound receptor that is stimulated by ligands Jagged and Delta-like ligands (Dll1/4) found on adjacent cells (Table 1).51 Under homeostatic Notch signaling, the majority of epithelial cells differentiate toward the absorptive lineage. When Notch signaling is reduced, ISC proliferation is reduced and ISCs differentiate toward a secretory cell fate (Paneth, goblet, enteroendocrine, or tuft cells).50, 52 Importantly, opposing activities of Notch and Wnt signaling regulate the balance between ISC maintenance and secretory lineage allocation.10 Although strict gradients of Notch and its receptors have not been formally established along the crypt-villus axis, evidence suggests that Notch activity may regulate the Wnt gradient influence on cell fate and highlights the critical interplay between different ISC regulatory pathways and the ultimate impact of factor gradients.10
Extracellular Matrix
In close proximity to the ISC niche, ECM proteins and the biomechanical properties they provide have long been known to exert a powerful influence on ISC behavior and differentiation.53 Not surprisingly, gradients of ECM span the crypt axis, producing alterations in the matrix chemical and mechanical properties (Figure 1, Table 1). The crypt base is enriched for a number of ECM components within the basement membrane, including isoforms of fibronectins, laminins, collagens, and glycosaminoglycans, suggesting specific ECM component isoforms play a role in the regulation of ISC maintenance, proliferation, and differentiation.54, 55, 56, 57 ECM also is found diffusely throughout the lamina propria, providing supportive biomechanical properties for epithelial and mesenchymal cells along the crypt-villus axis because cell behavior has been shown to respond to and depend on ECM stiffness.58
A physical scaffold of ECM supports the ISC niche and is in contact with all of the cellular components. Both epithelial and mesenchymal compartments deposit this ECM; however, the ECM proteins secreted by specific cell types have not been fully characterized.31, 59 The basolateral membrane of the epithelium directly interacts through integrin binding with the basement ECM, a thin sheet of matrix composed mostly of laminins and collagen IV.59 The ECM found in the niche can impact cell behavior by modulating signaling pathways that control cellular proliferation, growth, and death. This influence can occur through physical anchoring of cells, regulating biomechanical stiffness, and creating reservoirs of soluble factors.31 The latter is particularly interesting because it is conceivable that factors can be focused to microniches to exert a particular effect. For example, heparan sulfate proteoglycans, found on many ECM proteins, stabilize growth factors such as Wnt.60 Heparan sulfate proteoglycans are found on collagen XVIII in the basement membrane of the ISC niche, and although not formally tested, may interact with Wnt to increase the Wnt concentration toward the base of the crypt.61
Laminins are the most abundant protein in the basement membrane and different laminins are regionally expressed along the crypt-villus axis. The localized distribution suggests a relationship between regional ECM composition and epithelial cell function. Laminin α2 is found surrounding the base of the crypts, whereas laminins α3 and α5 are found in the villi.59 Reduction of laminin α5 in the small intestine resulted in expression of colonic-type isoforms and transformation to colonic mucosal architecture, which was associated with increases in cell proliferation and migration.55 This study suggests that regionalization of ECM deposition fundamentally changes tissue morphology and cell fate. Other ECM proteins have been found to be distributed regionally, including tenascin, found at the base of the villus and surrounding the crypt, and fibronectin, surrounding the crypt.62
Laminins interact with integrins found on epithelial cells, anchoring the cells to the ECM, which is essential for ISC survival because apoptosis occurs because of a lack of epithelial β1 integrin interaction. Consequently, β1 integrin is found homogenously along the crypt-villus axis.59 Regional expression of the α subunit of integrins exists, with α2 enriched in the lower crypt and α3 enriched in the upper crypt and villus regions.59, 62 These integrin interactions with laminins suggest a role of ECM in ISC proliferation and maintenance, further highlighting the role of the niche environment on ISCs.
What remains to be fully understood are the complex mechanisms that regulate ECM deposition and ECM-dependent signaling mechanisms that regulate bidirectional interactions, or dynamic reciprocity, between the epithelium and the mesenchyme. Although ECM clearly is appreciated as playing an essential role in intestinal epithelial biology, a more comprehensive understanding of the ECM complexity is limited by constraints in ECM and isoform detection, a low-resolution working knowledge of the different cell types that deposit ECM, and a lack of animal models to test ECM-dependent mechanisms.
Stiffness Gradients
Mechanical cues from the local environment and ECM, deriving from physical properties such as stiffness, shape, and adhesion, guide ISC fate through regulated sensing pathways and biochemical signal transducers.63, 64, 65 Although gradients of stiffness have not been directly measured in the different stromal and matrix regions surrounding the crypt/villus because of technical limitations, inferences about their respective properties can and have been made. For example, the mucosa, which supports the epithelium and crypt-villus architecture, has been shown to have 0.25–0.5 times the collagen content of the submucosa, suggesting that a stiffness gradient may exist on the millimeter scale between these layers.66 In addition, in vitro culture systems have shown optimal ISC growth on stiff matrices and optimal differentiation on soft matrices.58 Extension of this gradient to the micron scale of the crypt-villus axis is plausible, although this has yet to be confirmed in the in vivo intestine.
Microbial Product Gradients
One of the main functions of the intestinal epithelium is to provide a barrier to luminal contents, including the large population of gut microbiota found in greatest abundance in the distal small intestine and colon.67 The co-evolution of animals and microbes has led to the majority of metabolic pathways within the host gut lumen involving, or even relying, on the gut microbiome, highlighting the importance of microbial product consideration when profiling the gradients of metabolites across the epithelium, especially in the colon.68, 69, 70
The assignment of chemicals found in the gut lumen to microbial vs host production suggests that primary amines, polyamines, and short-chain fatty acids (SFCAs) are largely produced by the microbiome.71, 72, 73 The SCFAs produced from the fermentation of dietary fiber by microbes are perhaps the most well-studied microbial metabolite because they regulate energy metabolism and impact ISC proliferation.74, 75, 76 The SCFA that has garnered the greatest attention is butyrate, which differentiated colonocytes use as their primary energy source and is essential for undergoing healthy mitochondrial respiration.77 In contrast, butyrate acts as an inhibitor of ISC proliferation, with typical luminal concentrations (approximately 70 mmol/L in human being) capable of inducing ISC apoptosis through a Foxo3-related mechanism.78, 79 During homeostasis, the physical structure of the intestinal crypt forms an effective barrier against butyrate from reaching ISCs at the base of the crypt, resulting in a sharp gradient between the crypt base and the intestinal lumen where differentiated cells are actively metabolizing butyrate (Table 1).79
Although not as rigorously characterized as SCFAs, other microbial metabolites also have been studied for their impact on the gut epithelium and on overall organismal health. Polyamines, which include putrescine, spermidine, and spermine, are produced by luminal microbiota and have been shown to be essential for growth and development of the small intestinal and colonic mucosa.71, 80, 81, 82 Free catecholamines, such as the neurotransmitters dopamine and norepinephrine, are produced within the gut lumen by microbes with high β-glucuronidase activity.83 Recently, the microbial product desaminotyrosine, produced from the digestion of plant flavonoids, was shown to enter the bloodstream and trigger type 1 interferon signaling, acting as a protective measure against influenza.84 These examples suggest that microbial metabolites produced in the gut lumen have both local impacts on epithelial proliferation and barrier function as well as far-reaching impacts on overall host health.
Gas Gradients
Oxygen is essential for epithelial cell survival; therefore, steep oxygen gradients exist across the intestinal mucosa and gut lumen. Partial pressures of oxygen reach 80 mm Hg deep in the submucosa, where intestinal cells are oxygenated by the oxygen-rich vasculature extending throughout intestinal tissue, but decrease to 0.1–1 mm Hg in the large intestinal lumen where anaerobic microbes thrive and facultative anaerobes act as oxygen scavengers (Table 1).85, 86, 87, 88, 89 Oxygen levels within the vasculature also may influence the microbiota composition, with more oxygen-tolerant microbes found closer to the intestinal epithelium. Production of nitric oxide by NO synthase within the vasculature and supporting stroma, potentially creating an NO gradient across the epithelium (Table 1).85, 90, 91, 92 Inversely, the luminal microbiota is the source of many gases including methane, H2, and hydrogen sulfide, which may form gradients impacting the intestinal epithelium and mucosa (Table 1).93, 94, 95 For example, hydrogen sulfide is increased in intestinal tissue from patients with ulcerative colitis, suggesting that this gas can diffuse into the epithelium.85, 96 The rapid diffusion of gases through tissue in combination with relatively short lifetimes may enable gradients to be rapidly modulated and act as first responders or sentinel molecules to changing intestinal conditions or insults.97
Engineered In Vitro Gradients
Growth Factor Gradients
Although it is well recognized that many growth factor and ECM gradients exist, few studies have re-created these gradients for proper epithelial polarization and recapitulation of the in vivo crypt-villus architecture in vitro. Primary ISCs cultured with exogenous factors such as Wnt, Notch, and R-spondin form organoids with crypt-like buds possessing increased ISC numbers.5 Because of their 3D morphology, it has been technically challenging to re-create spatial gradients to form separate proliferative and differentiated cell zones within organoids. The requirement for exogenous ISC supporting factors suggests that ISCs alone are unable to produce an adequate niche environment, including the gradients associated with a fully polarized crypt/villus structure.5, 98 The use of microfluidic systems has enabled a linear gradient of Wnt3A and R-spondin 1 to be placed over colonic murine organoids to create a distinct stem/proliferative and differentiated cell zone within a single organoid and shows that growth factors are integral in re-creating the cell compartments found in the crypt.99
Organoids typically are fully embedded within a hydrogel, possess an inaccessible lumen, show a nonphysiologic shape and size, and do not show the highly segregated spatial cell location seen within in vivo crypts. These limitations make conclusions from the manipulation of gradients in organoid systems challenging to interpret. Fortunately, a multitude of gradient-making systems exist for the task of applying controlled surface- and solution-based chemical gradients to cells.100 One of the most simple and robust gradient systems, the Transwell, represents a facile and robust strategy to form gradients across intestinal epithelial monolayers. A modified version has been paired with a shaped collagen scaffold to re-create arrays of polarized in vitro crypts and villi (Figure 2A and B).23, 101, 102, 103 Microwells (crypts) and posts (villi) were fabricated into a collagen scaffold to mimic the shape, size, and density of crypts in vivo. Primary human and murine epithelial cells were expanded as monolayers and then placed under a gradient of growth factors including Wnt, R-spondin, and Noggin, resulting in a polarized epithelium with stem/proliferative cells isolated to the base of the microwells and differentiated cells localized to the luminal surface or villi of the scaffolding. Importantly the epithelium in this culture system showed key features of the in vivo intestine, including the production of alkaline phosphatase by enterocytes and mucous production and secretion by goblet cells. Titrating the growth factor gradient moved the stem/proliferative cell regions up or down the in vitro crypt axis showing that the cells were fully responsive to the microenvironment.
Figure 2.
Gradients formed across intestinal tissue in vitro. (A) Generation of in vitro human, small intestine crypt-villus arrays. Left panel: Schematic of the gradient of growth factors (W, Wnt3A; R, R-spondin 3; N, noggin). Middle panels: Brightfield and fluorescence images of a polarized crypt-villus unit under the 3-growth factor gradient and opposing DAPT gradient. Mature enterocytes (red, alkaline phosphatase [ALP]) and proliferative cells (green, 5-ethynyl-20-deoxyuridine [EdU]) also are marked. DNA or nuclei are shown in blue. Right panel: Immunofluorescence staining (olfactomedin [Olfm4]/keratin 20 [KRT20]) of a cross-section through in vitro human small intestinal tissue under the combined growth factor and DAPT gradient. Scale bars: 100 μm. Reproduced by permission from Elsevier from Biomaterials, 2017: 128, 44-55. (B) Generation of in vitro human colon crypts. Left panel: Schematic of the gradient of growth factors (Wnt3A, R-spondin, noggin). Differentiated and stem/proliferative cells are shown in red and green, respectively. Middle panels: Brightfield and fluorescence side views of in vitro–formed crypts (EdU, green; ALP, red; DNA, blue). Right panel: Cross-section of in vitro crypt immunostained for KRT20 (red) and Olfm4 (green). Scale bars: 100 μm. (C) Modulation of in vitro human crypts by SCFAs. Left panel: Biochemical gradients applied to the tissue. Middle and right panels: Side view of representative crypts from the arrays under different SCFA gradients (EdU, green; ALP, red). Scale bar: 100 μm. Reproduced by permission under a Creative Commons Attribution-NonCommercial-No Derivatives License from Elsevier from Cellular and Molecular Gastroenterology Hepatology. (D) Bioengineered tissues on silk scaffolds mimic in vivo luminal oxygen levels. Left panels: Schematic of a 3D tissue and quantification of luminal partial pressure of oxygen (Po2) in the presence of confluent Caco-2 tumor cells. Right panels: Oxygen levels were detected in situ by using engineered oxygen-sensing fluorescent Yersinia pseudotuberculosis (red). The absence of green fluorescent protein (GFP) fluorescence and the presence of expressed mini singlet-oxygen generator (arrowheads) indicates the presence of anaerobic conditions. Reproduced under a Creative Commons Attribution 4.0 International License from Sci. Rep. 5, 13708. (E) Matrix mechanical properties control ISC proliferation. Upper panels: ISC colonies were formed within enzymatically cross-linked poly(ethylene glycol) (PEG) hydrogels modified with Arg (R)-Gly (G)-Asp (D) (RGD) and of varying stiffness. Yes-associated protein (YAP) immunofluorescence is shown in green. Scale bar: 50 μm. Lower panel: ISC colony-forming efficiency of ISCs embedded in degradable (DG) or nondegradable (N-DG) PEG gels of varying stiffness. GM6001 is a broad-spectrum matrix metalloproteinase inhibitor.
Reprinted with permission from Macmillan Publishers Ltd. Nature, 2016;538:560–564. DAPI, 4′,6-diamidino-2-phenylindole; DAPT, γ secretase inhibitor.
Microfluidic devices have been used extensively to create solution gradients of many molecules including growth factors with a range of device designs, parameters, and operating conditions.104, 105, 106 With respect to soluble gradients of proteins relevant to the intestine, a 2-chamber microfluidic chip with an intervening porous membrane yielded a bipolar gradient of Wnt3A and Dickkopf-related protein-1 that could be turned on or off on-demand and featured programmable gradient features.107 Although gradients can be created in microdevices, significant hurdles still exist in integrating primary cells. Although the devices generally are fabricated from biocompatible substances, the materials do not recapitulate the chemical or physical properties required to support primary cells, including surface properties such as stiffness and porosity.
Gradients of immobilized growth factors are more simple and often easier to operate than soluble gradients. These gradients also can be engineered to release proteins over time, yielding both temporal and/or spatial gradients to manipulate the stem cell microenvironment or direct differentiation. This is best exemplified in human mesenchymal stem cell systems, in which microparticles with controlled concentrations of BMP-2 and insulin-like growth factors are placed into alginate and silk scaffolds to form a gradient.108 Similarly, an opposing gradient of platelet-derived growth factor and BMP-2 was formed on a membrane, regulating adipose-derived stem cell differentiation.109 Lateral gradients of BMP-2 and/or BMP-7 applied to a biocompatible film using a microfluidic device were competent to alter myofibroblast gene expression and enzyme activity while covalent immobilization of Wnt3A on aldehyde-functionalized surfaces directs human mesenchymal stem cell differentiation.110, 111 In the future, select positioning of growth factor–producing cells with respect to intestinal epithelial cells also may be used to provide both soluble and surface-based gradients to more accurately mimic the in vivo conditions.
Surface-Bound Matrix Gradients
In vitro platforms have provided simplified and well-controlled approaches to study cell behaviors under a gradient of surface-bound cues.105, 112, 113 Gradients of collagen, laminin, or fibronectin have been formed on surfaces comprising poly(D,L-lactic acid), gold films, poly(ethylene glycol) hydrogel, and other surfaces.114, 115, 116 Although gradients are generated most easily on planar substrates, gradients on 3D scaffolds may be more physiologically relevant albeit technically more difficult. A gradient of BMP-7, transforming growth factor-β2, and vascular endothelial growth factor165 was formed along the longitudinal direction of polycaprolactone/pluronic F127 cylindric scaffolds by first creating a gradient of fibril-like polycaprolactones for the attachment of proteins.117 Although a multitude of cell types (both primary and tumor cells) have been paired with surface-bound protein gradients, none have included primary intestinal cells owing to their complex culture requirements.23, 24 A self-renewing monolayer culture system for primary intestinal epithelial cells recently was realized on a collagen hydrogel and now offers the possibility of acting as substrate for gradients of surface-bound proteins.23
Matrix Stiffness Gradients
Methods to create a continuous gradient of stiffness in hydrogels for cell culture have been reviewed recently.118 These strategies have created lateral stiffness gradients within the range of 1 kPa to 3.1 MPa using a variety of materials.119, 120, 121, 122, 123 Polyacrylamide gels have a unique advantage because their surface is easily conjugated with ECM proteins so that the ECM density is independent of substrate stiffness. In addition to planar gradients, stiffness variations along the longitudinal direction of a cylindrical hydrogel were created using a gradual freezing and thawing method.124 In a recent breakthrough, a synthetic hydrogel-based system of variable stiffness was developed to support organoid-based culture of ISCs.58 Intestinal cell expansion was optimal within matrices of intermediate stiffness (1.3 kPa), whereas stem cells embedded in soft matrices (300 Pa) proliferated poorly (Figure 2E). A platform possessing a gradient of stiffness but with a constant density of ECM will be critical to fully recapitulate all architectural and physiological features of the in vivo intestinal epithelium.
Microbial Product and Inflammatory Mediator Gradients
Reproducing the gradient profile of microbial products across organoids is technically difficult because these constituents are at their greatest concentration within the lumen, which in organoids is enclosed and not easily accessed through a hydrogel layer.76, 79, 125 In a tour de force effort, induced pluripotent stem cell (iPSC)-derived organoids were individually microinjected with Escherichia coli to investigate the interaction of the intestinal epithelium with the microbes.126 Colonization with E coli induced functional maturation of the immature organoids, increasing antimicrobial peptide production, mucus layer maturation, and improved barrier function. To circumvent the challenge of microinjecting single Matrigel (BD Biosciences, San Diego, CA)-embedded organoids, a strategy based on the preferred adhesion of cells to ECM rather than nonadhesive microstructures was developed, enabling cell self-organization around a pH-sensing particle.127 Refinements in strategies such as this may enable future delivery of analyte-sensing or drug-releasing microparticles and/or microbes to the lumen of organoids.
The use of a Transwell insert also provides a robust strategy to form microbial-product gradients across intestinal epithelial monolayers; however, the vast majority of these studies have focused on the response of tumor cells to microbial metabolites.128, 129, 130, 131 Although these models provide useful insights, recently established primary intestinal epithelial monolayer cultures in these inserts are expected to replace the intestinal cancer models.23, 26, 27, 132 For example, a mouse primary colonic monolayer was used to show that interferon-γ (IFN-γ) did not increase IgA-receptor expression or transcytosis as IFN-γ did in tumor cells.132
Perhaps the most straightforward strategy to establish a gradient of microbial products across an in vitro epithelium is to co-culture commensal microbes on the luminal surface of a monolayer.133 Because most intestinal microbes are obligate anaerobes, these model systems must supply an appropriate deoxygenated environment for the microbes in concert with an oxygen source to maintain intestinal epithelial cell viability. This demanding co-culture environment is most easily achieved using tumor models.134 A microfluidic model, the HuMix system (University of Luxembourg, Luxembourg City, Luxembourg), uses 3 stacked compartments, a deoxygenated microbial chamber, an apical cell chamber, and a basal oxygenated chamber to re-create mammalian cell-microbe cross-talk.135, 136 Mixtures of facultative (Lactobacillus rhamnosus GG) and obligate (Bacteroides caccae) anaerobes were co-cultured with mammalian cells, showing the potential of this technology.136
To truly understand the interactions of microbes and their gradients within intestinal tissue, primary tissue constructs with the architecture and tissue polarity of in vivo colon crypts must ultimately be used for gradient formation and microbial co-culture. Primary human crypts paired with a physiologic SCFA gradient along the crypt axis showed that butyrate, but not acetate or propionate, suppressed the proliferative cell population in a dose-dependent manner, diminishing the size of the ISC niche (Figure 2C).102 The SCFA gradients also enhanced epithelial cell differentiation into the absorptive colonocyte lineage as is shown to occur in vivo. The ability to recapitulate complex architectural features and physiologic responses shows the value of these emerging organ-on-chip systems.
The intestine and surrounding lymphoid system are a major site for immune system education regarding potential pathogenic microbes as well as a site for maturation of immune cells.102, 137, 138, 139 Gradients of tumor necrosis factor-α and IFN-γ applied to human in vitro crypt arrays reduced the alkaline phosphatase activity of differentiated cells as well as the size of the stem/proliferative cell compartment, profoundly altering crypt properties.102 Model systems to identify the differential impact of these inflammatory mediators and immune cells along the length of the crypt axis will be critical to understanding the impact of inflammation on the initiation or exacerbation of disease states.
Gas Gradients
Platforms capable of spatiotemporal oxygen control have the potential to enable the study of intestinal physiology, microbial ecology, and their interactions in vitro. Oxygen gradients with arbitrary profiles (eg, linear, exponential, and nonmonotonic) have been generated as stable or reconfigurable gradients.140, 141 Oxygen gradients compatible with an open-well culture also have permitted direct observation of cell behavior in response to varying oxygen levels.142 Vertical oxygen gradients over Caco-2 cells established by simultaneous perfusion of oxygenated and deoxygenated media through 2 separated microchannels permitted co-culture of Caco-2 cells with an obligate anaerobe under conditions representative of the gastrointestinal human–microbe interface.136 These types of controllable gradients may in the future be used to mimic hypoxic insults during loss of blood flow or epithelial integrity in the intestine.
Although gas control with microfluidics offers unique advantages, these systems typically require bulky or sophisticated off-chip equipment such as pumps, valves, and control systems. A spontaneous (cell-mediated) oxygen gradient was generated using cell respiration to deplete oxygen. Intraluminal oxygen gradients were spontaneously established when Caco-2 cells were cultured on a porous scaffolding containing a shaped, high-surface-area lumen (Figure 2D).143 In another example, rapid, predictable oxygen gradients were established by embedding cells in gelatin-based O2-controllable hydrogels.144 Although numerous platforms are available, the effect of gas gradients on primary intestinal epithelial cells and host-microbe interactions has not been studied extensively, presenting an opportunity for future research.
Conclusions
Despite the plethora of gradient-making technologies, only a limited number of these technologies and platforms have been applied successfully to primary intestinal epithelial models.101, 102, 103 Most intestine- (or gut)-on-a-chip models use tumor cell lines, which are unlikely to respond in a physiologically relevant way to in vitro gradients or to form a physiologically normal niche. Given the richness and diversity of gradients formed across the intestinal epithelium, tremendous opportunities exist in the pairing of primary intestinal culture systems with chemical and matrix gradients. These gradients may be shaped on surfaces, in solution, and within scaffolds, or generated using co-cultured cells such as bacteria or stroma. Modifications to the current organ-on-chip and microfluidic technologies to accommodate primary ISCs, epithelial cells, and other interacting cells will replicate the architecture and physiological functions of the in vivo intestinal epithelium in the near future.
Footnotes
Author contributions All authors participated in the writing of the review.
Conflicts of interest These authors disclose the following: Yuli Wang, Bailey Zwarycz, Scott T. Magness, and Nancy L. Allbritton have a financial interest in Altis Biosystems. The remaining authors disclose no conflicts.
Funding Supported by the National Institutes of Health, National Institute of Diabetes and Digestive and Kidney DiseasesR01DK109559 (N.L.A. and S.T.M.).
Contributor Information
Scott T. Magness, Email: magness@med.unc.edu.
Nancy L. Allbritton, Email: nlallbri@unc.edu, nlallbri@ncsu.edu.
Supplementary Material
Supplemental Graphical Summary.
References
- 1.Sailaja B.S., He X.C., Li L.H. The regulatory niche of intestinal stem cells. J Physiol. 2016;594:4827–4836. doi: 10.1113/JP271931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Barker N. Adult intestinal stem cells: critical drivers of epithelial homeostasis and regeneration. Nat Rev Mol Cell Biol. 2014;15:19–33. doi: 10.1038/nrm3721. [DOI] [PubMed] [Google Scholar]
- 3.Fuchs E., Chen T. A matter of life and death: self-renewal in stem cells. EMBO Rep. 2013;14:39–48. doi: 10.1038/embor.2012.197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Clevers H.C., Bevins C.L. Paneth cells: maestros of the small intestinal crypts. Ann Rev Physiol. 2013;75:289–311. doi: 10.1146/annurev-physiol-030212-183744. [DOI] [PubMed] [Google Scholar]
- 5.Sato T., Vries R.G., Snippert H.J., Van De Wetering M., Barker N., Stange D.E., Van Es J.H., Abo A., Kujala P., Peters P.J. Single Lgr5 stem cells build crypt villus structures in vitro without a mesenchymal niche. Nature. 2009;459:262–265. doi: 10.1038/nature07935. [DOI] [PubMed] [Google Scholar]
- 6.Barker N., van Es J.H., Kuipers J., Kujala P., van den Born M., Cozijnsen M., Haegebarth A., Korving J., Begthel H., Peters P.J., Clevers H. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature. 2007;449:1003–1007. doi: 10.1038/nature06196. [DOI] [PubMed] [Google Scholar]
- 7.Rothenberg M.E., Nusse Y., Kalisky T., Lee J.J., Dalerba P., Scheeren F., Lobo N., Kulkarni S., Sim S., Qian D., Beachy P.A., Pasricha P.J., Quake S.R., Clarke M.F. Identification of a cKit(+) colonic crypt base secretory cell that supports Lgr5(+) stem cells in mice. Gastroenterology. 2012;142:1195–1205. doi: 10.1053/j.gastro.2012.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Madison B.B., Braunstein K., Kuizon E., Portman K., Qiao X.T., Gumucio D.L. Epithelial hedgehog signals pattern the intestinal crypt-villus axis. Development. 2005;132:279–289. doi: 10.1242/dev.01576. [DOI] [PubMed] [Google Scholar]
- 9.Batlle E., Henderson J.T., Beghtel H., van den Born M.M.W., Sancho E., Huls G., Meeldijk J., Robertson J., van de Wetering M., Pawson T., Clevers H. b-Catenin and TCF mediate cell positioning in the intestinal epithelium by controlling the expression of EphB/EphrinB. Cell. 2002;111:251–263. doi: 10.1016/s0092-8674(02)01015-2. [DOI] [PubMed] [Google Scholar]
- 10.Tian H., Biehs B., Chiu C., Siebel C.W., Wu Y., Costa M., de Sauvage Frederic J., Klein Ophir D. Opposing activities of notch and wnt signaling regulate intestinal stem cells and gut homeostasis. Cell Rep. 2015;11:33–42. doi: 10.1016/j.celrep.2015.03.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Fevr T., Robine S., Louvard D., Huelsken J. Wnt/beta-catenin is essential for intestinal homeostasis and maintenance of intestinal stem cells. Mol Cell Biol. 2007;27:7551–7559. doi: 10.1128/MCB.01034-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sato T., van Es J.H., Snippert H.J., Stange D.E., Vries R.G., van den Born M., Barker N., Shroyer N.F., van de Wetering M., Clevers H. Paneth cells constitute the niche for Lgr5 stem cells in intestinal crypts. Nature. 2011;469:415–418. doi: 10.1038/nature09637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Kabiri Z., Greicius G., Madan B., Biechele S., Zhong Z.D., Zaribafzadeh H., Edison Aliyev J., Wu Y.H., Bunte R., Williams B.O., Rossant J., Virshup D.M. Stroma provides an intestinal stem cell niche in the absence of epithelial Wnts. Development. 2014;141:2206–2215. doi: 10.1242/dev.104976. [DOI] [PubMed] [Google Scholar]
- 14.Valenta T., Degirmenci B., Moor A.E., Herr P., Zimmerli D., Moor M.B., Hausmann G., Cantù C., Aguet M., Basler K. Wnt ligands secreted by subepithelial mesenchymal cells are essential for the survival of intestinal stem cells and gut homeostasis. Cell Rep. 2016;15:911–918. doi: 10.1016/j.celrep.2016.03.088. [DOI] [PubMed] [Google Scholar]
- 15.Pinto D., Gregorieff A., Begthel H., Clevers H. Canonical Wnt signals are essential for homeostasis of the intestinal epithelium. Genes Dev. 2003;17:1709–1713. doi: 10.1101/gad.267103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Qi Z., Li Y.H., Zhao B., Xu C., Liu Y., Li H.N., Zhang B.J., Wang X.Q., Yang X., Xie W., Li B.J., Han J.D.J., Chen Y.G. BMP restricts stemness of intestinal Lgr5(+) stem cells by directly suppressing their signature genes. Nat Commun. 2017;8:13824. doi: 10.1038/ncomms13824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Auclair B.A., Benoit Y.D., Rivard N., Mishina Y., Perreault N. Bone morphogenetic protein signaling is essential for terminal differentiation of the intestinal secretory cell lineage. Gastroenterology. 2007;133:887–896. doi: 10.1053/j.gastro.2007.06.066. [DOI] [PubMed] [Google Scholar]
- 18.Hou Q.H., Ye L.L., Huang L.L., Yu Q.H. The research progress on intestinal stem cells and its relationship with intestinal microbiota. Front Immunol. 2017;8:599. doi: 10.3389/fimmu.2017.00599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zheng L., Kelly C.J., Colgan S.P. Physiologic hypoxia and oxygen homeostasis in the healthy intestine. A review in the theme: cellular responses to hypoxia. Am J Physiol Cell Physiol. 2015;309:C350–C360. doi: 10.1152/ajpcell.00191.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Krausova M., Korinek V. Wnt signaling in adult intestinal stem cells and cancer. Cell Signal. 2014;26:570–579. doi: 10.1016/j.cellsig.2013.11.032. [DOI] [PubMed] [Google Scholar]
- 21.Sato T., Stange D.E., Ferrante M., Vries R.G.J., van Es J.H., van den Brink S., van Houdt W.J., Pronk A., van Gorp J., Siersema P.D., Clevers H. Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett's epithelium. Gastroenterology. 2011;141:1762–1772. doi: 10.1053/j.gastro.2011.07.050. [DOI] [PubMed] [Google Scholar]
- 22.van der Flier L.G., Haegebarth A., Stange D.E., van de Wetering M., Clevers H. OLFM4 is a robust marker for stem cells in human intestine and marks a subset of colorectal cancer cells. Gastroenterology. 2009;137:15–17. doi: 10.1053/j.gastro.2009.05.035. [DOI] [PubMed] [Google Scholar]
- 23.Wang Y., DiSalvo M., Gunasekara D.B., Dutton J., Proctor A., Lebhar M.S., Williamson I.A., Speer J., Howard R.L., Smiddy N.M., Bultman S.J., Sims C.E., Magness S.T., Allbritton N.L. Self-renewing monolayer of primary colonic or rectal epithelial cells. Cell Mol Gastroenterol Hepatol. 2017;4:165–182. doi: 10.1016/j.jcmgh.2017.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Yin X.L., Mead B.E., Safaee H., Langer R., Karp J.M., Levy O. Engineering stem cell organoids. Cell Stem Cell. 2016;18:25–38. doi: 10.1016/j.stem.2015.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.VanDussen K.L., Marinshaw J.M., Shaikh N., Miyoshi H., Moon C., Tarr P.I., Ciorba M.A., Stappenbeck T.S. Development of an enhanced human gastrointestinal epithelial culture system to facilitate patient-based assays. Gut. 2015;64:911–920. doi: 10.1136/gutjnl-2013-306651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kozuka K., He Y., Koo-McCoy S., Kumaraswamy P., Nie B., Shaw K., Chan P., Leadbetter M., He L., Lewis J.G. Development and characterization of a human and mouse intestinal epithelial cell monolayer platform. Stem Cell Rep. 2017;9:1976–1990. doi: 10.1016/j.stemcr.2017.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.In J., Foulke-Abel J., Zachos N.C., Hansen A.-M., Kaper J.B., Bernstein H.D., Halushka M., Blutt S., Estes M.K., Donowitz M. Enterohemorrhagic Escherichia coli reduces mucus and intermicrovillar bridges in human stem cell-derived colonoids. Cell Mol Gastroenterol Hepatol. 2016;2:48–62. doi: 10.1016/j.jcmgh.2015.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bastide P., Darido C., Pannequin J., Kist R., Robine S., Marty-Double C., Bibeau F., Scherer G., Joubert D., Hollande F., Blache P., Jay P. Sox9 regulates cell proliferation and is required for Paneth cell differentiation in the intestinal epithelium. J Cell Biol. 2007;178:635–648. doi: 10.1083/jcb.200704152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mori-Akiyama Y., van den Born M., van Es J.H., Hamilton S.R., Adams H.P., Zhang J., Clevers H., de Crombrugghe B. SOX9 is required for the differentiation of Paneth cells in the intestinal epithelium. Gastroenterology. 2007;133:539–546. doi: 10.1053/j.gastro.2007.05.020. [DOI] [PubMed] [Google Scholar]
- 30.Farin H.F., Van Es J.H., Clevers H. Redundant sources of Wnt regulate intestinal stem cells and promote formation of Paneth cells. Gastroenterology. 2012;143:1518–1529. doi: 10.1053/j.gastro.2012.08.031. [DOI] [PubMed] [Google Scholar]
- 31.Brizzi M.F., Tarone G., Defilippi P. Extracellular matrix, integrins, and growth factors as tailors of the stem cell niche. Curr Opin Cell Biol. 2012;24:645–651. doi: 10.1016/j.ceb.2012.07.001. [DOI] [PubMed] [Google Scholar]
- 32.Formeister E.J., Sionas A.L., Lorance D.K., Barkley C.L., Lee G.H., Magness S.T. Distinct SOX9 levels differentially mark stem/progenitor populations and enteroendocrine cells of the small intestine epithelium. Am J Physiol Gastrointest Liver Physiol. 2009;296:G1108–G1118. doi: 10.1152/ajpgi.00004.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Blache P., van de Wetering M., Duluc I., Domon C., Berta P., Freund J.N., Clevers H., Jay P. SOX9 is an intestine crypt transcription factor, is regulated by the Wnt pathway, and represses the CDX2 and MUC2 genes. J Cell Biol. 2004;166:37–47. doi: 10.1083/jcb.200311021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gracz A.D., Ramalingam S., Magness S.T. Sox9 expression marks a subset of CD24-expressing small intestine epithelial stem cells that form organoids in vitro. Am J Physiol Gastrointest Liver Physiol. 2010;298:G590–G600. doi: 10.1152/ajpgi.00470.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ramalingam S., Daughtridge G.W., Johnston M.J., Gracz A.D., Magness S.T. Distinct levels of Sox9 expression mark colon epithelial stem cells that form colonoids in culture. Am J Physiol Gastrointest Liver Physiol. 2012;302:G10–G20. doi: 10.1152/ajpgi.00277.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Farin H.F., Jordens I., Mosa M.H., Basak O., Korving J., Tauriello D.V., de Punder K., Angers S., Peters P.J., Maurice M.M., Clevers H. Visualization of a short-range Wnt gradient in the intestinal stem-cell niche. Nature. 2016;530:340–343. doi: 10.1038/nature16937. [DOI] [PubMed] [Google Scholar]
- 37.Gracz A.D., Williamson I.A., Roche K.C., Johnston M.J., Wang F., Wang Y., Attayek P.J., Balowski J., Liu X.F., Laurenza R.J., Gaynor L.T., Sims C.E., Galanko J.A., Li L., Allbritton N.L., Magness S.T. A high-throughput platform for stem cell niche co-cultures and downstream gene expression analysis. Nat Cell Biol. 2015;17:340–349. doi: 10.1038/ncb3104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Gregorieff A., Pinto D., Begthel H., Destree O., Kielman M., Clevers H. Expression pattern of Wnt signaling components in the adult intestine. Gastroenterology. 2005;129:626–638. doi: 10.1016/j.gastro.2005.06.007. [DOI] [PubMed] [Google Scholar]
- 39.Lei N.Y., Jabaji Z., Wang J., Joshi V.S., Brinkley G.J., Khalil H., Wang F., Jaroszewicz A., Pellegrini M., Li L., Lewis M., Stelzner M., Dunn J.C., Martin M.G. Intestinal subepithelial myofibroblasts support the growth of intestinal epithelial stem cells. PLoS One. 2014;9:e84651. doi: 10.1371/journal.pone.0084651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Aoki R., Shoshkes-Carmel M., Gao N., Shin S., May C.L., Golson M.L., Zahm A.M., Ray M., Wiser C.L., Wright C.V., Kaestner K.H. Foxl1-expressing mesenchymal cells constitute the intestinal stem cell niche. Cell Mol Gastroenterol Hepatol. 2016;2:175–188. doi: 10.1016/j.jcmgh.2015.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Koch S. Extrinsic control of Wnt signaling in the intestine. Differentiation. 2017;97:1–8. doi: 10.1016/j.diff.2017.08.003. [DOI] [PubMed] [Google Scholar]
- 42.Stzepourginski I., Nigro G., Jacob J.M., Dulauroy S., Sansonetti P.J., Eberl G., Peduto L. CD34+ mesenchymal cells are a major component of the intestinal stem cells niche at homeostasis and after injury. Proc Natl Acad Sci U S A. 2017;114:E506–E513. doi: 10.1073/pnas.1620059114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Yan K.S., Janda C.Y., Chang J., Zheng G.X.Y., Larkin K.A., Luca V.C., Chia L.A., Mah A.T., Han A., Terry J.M., Ootani A., Roelf K., Lee M., Yuan J., Li X., Bolen C.R., Wilhelmy J., Davies P.S., Ueno H., von Furstenberg R.J., Belgrader P., Ziraldo S.B., Ordonez H., Henning S.J., Wong M.H., Snyder M.P., Weissman I.L., Hsueh A.J., Mikkelsen T.S., Garcia K.C., Kuo C.J. Non-equivalence of Wnt and R-spondin ligands during Lgr5(+) intestinal stem-cell self-renewal. Nature. 2017;545:238–242. doi: 10.1038/nature22313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Holmberg J., Armulik A., Senti K.A., Edoff K., Spalding K., Momma S., Cassidy R., Flanagan J.G., Frisen J. Ephrin-A2 reverse signaling negatively regulates neural progenitor proliferation and neurogenesis. Genes Dev. 2005;19:462–471. doi: 10.1101/gad.326905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cortina C., Palomo-Ponce S., Iglesias M., Fernandez-Masip J.L., Vivancos A., Whissell G., Huma M., Peiro N., Gallego L., Jonkheer S., Davy A., Lloreta J., Sancho E., Batlle E. EphB-ephrin-B interactions suppress colorectal cancer progression by compartmentalizing tumor cells. Nat Genet. 2007;39:1376–1383. doi: 10.1038/ng.2007.11. [DOI] [PubMed] [Google Scholar]
- 46.Kosinski C., Li V.S., Chan A.S., Zhang J., Ho C., Tsui W.Y., Chan T.L., Mifflin R.C., Powell D.W., Yuen S.T., Leung S.Y., Chen X. Gene expression patterns of human colon tops and basal crypts and BMP antagonists as intestinal stem cell niche factors. Proc Natl Acad Sci U S A. 2007;104:15418–15423. doi: 10.1073/pnas.0707210104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Shyer A.E., Huycke T.R., Lee C., Mahadevan L., Tabin C.J. Bending gradients: how the intestinal stem cell gets its home. Cell. 2015;161:569–580. doi: 10.1016/j.cell.2015.03.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Batts L.E., Polk D.B., Dubois R.N., Kulessa H. Bmp signaling is required for intestinal growth and morphogenesis. Dev Dyn. 2006;235:1563–1570. doi: 10.1002/dvdy.20741. [DOI] [PubMed] [Google Scholar]
- 49.Haramis A.P., Begthel H., van den Born M., van Es J., Jonkheer S., Offerhaus G.J., Clevers H. De novo crypt formation and juvenile polyposis on BMP inhibition in mouse intestine. Science. 2004;303:1684–1686. doi: 10.1126/science.1093587. [DOI] [PubMed] [Google Scholar]
- 50.VanDussen K.L., Carulli A.J., Keeley T.M., Patel S.R., Puthoff B.J., Magness S.T., Tran I.T., Maillard I., Siebel C., Kolterud A., Grosse A.S., Gumucio D.L., Ernst S.A., Tsai Y.H., Dempsey P.J., Samuelson L.C. Notch signaling modulates proliferation and differentiation of intestinal crypt base columnar stem cells. Development. 2012;139:488–497. doi: 10.1242/dev.070763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kopan R., Ilagan M.X. The canonical Notch signaling pathway: unfolding the activation mechanism. Cell. 2009;137:216–233. doi: 10.1016/j.cell.2009.03.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Schroder N., Gossler A. Expression of Notch pathway components in fetal and adult mouse small intestine. Gene Expr Patterns. 2002;2:247–250. doi: 10.1016/s1567-133x(02)00060-1. [DOI] [PubMed] [Google Scholar]
- 53.Meran L., Baulies A., Li V.S.W. Intestinal stem cell niche: the extracellular matrix and cellular components. Stem Cells Int. 2017;2017:7970385. doi: 10.1155/2017/7970385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Benoit Y.D., Groulx J.F., Gagne D., Beaulieu J.F. RGD-dependent epithelial cell-matrix interactions in the human intestinal crypt. J Signal Trans. 2012;2012:248759. doi: 10.1155/2012/248759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Mahoney Z.X., Stappenbeck T.S., Miner J.H. Laminin α5 influences the architecture of the mouse small intestinal mucosa. J Cell Sci. 2008;121:2493–2502. doi: 10.1242/jcs.025528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Groulx J.F., Gagne D., Benoit Y.D., Martel D., Basora N., Beaulieu J.F. Collagen VI is a basement membrane component that regulates epithelial cell-fibronectin interactions. Matrix Biol. 2011;30:195–206. doi: 10.1016/j.matbio.2011.03.002. [DOI] [PubMed] [Google Scholar]
- 57.Lin G.N., Zhang X., Ren J., Pang Z.M., Wang C.H., Xu N., Xi R.W. Integrin signaling is required for maintenance and proliferation of intestinal stem cells in Drosophila. Dev Biol. 2013;377:177–187. doi: 10.1016/j.ydbio.2013.01.032. [DOI] [PubMed] [Google Scholar]
- 58.Gjorevski N., Sachs N., Manfrin A., Giger S., Bragina M.E., Ordonez-Moran P., Clevers H., Lutolf M.P. Designer matrices for intestinal stem cell and organoid culture. Nature. 2016;539:560–564. doi: 10.1038/nature20168. [DOI] [PubMed] [Google Scholar]
- 59.Teller I.C., Beaulieu J.F. Interactions between laminin and epithelial cells in intestinal health and disease. Expert Rev Mol Med. 2001;3:1–18. doi: 10.1017/S1462399401003623. [DOI] [PubMed] [Google Scholar]
- 60.Yamamoto S., Nakase H., Matsuura M., Honzawa Y., Matsumura K., Uza N., Yamaguchi Y., Mizoguchi E., Chiba T. Heparan sulfate on intestinal epithelial cells plays a critical role in intestinal crypt homeostasis via Wnt/beta-catenin signaling. Am J Physiol Gastrointest Liver Physiol. 2013;305:G241–G249. doi: 10.1152/ajpgi.00480.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Muragaki Y., Timmons S., Griffith C.M., Oh S.P., Fadel B., Quertermous T., Olsen B.R. Mouse Col18a1 is expressed in a tissue-specific manner as three alternative variants and is localized in basement membrane zones. Proc Natl Acad Sci U S A. 1995;92:8763–8767. doi: 10.1073/pnas.92.19.8763. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Beaulieu J.F. Differential expression of the VLA family of integrins along the crypt-villus axis in the human small intestine. J Cell Sci. 1992;102:427–436. doi: 10.1242/jcs.102.3.427. [DOI] [PubMed] [Google Scholar]
- 63.Guilak F., Cohen D.M., Estes B.T., Gimble J.M., Liedtke W., Chen C.S. Control of stem cell fate by physical interactions with the extracellular matrix. Cell Stem Cell. 2009;5:17–26. doi: 10.1016/j.stem.2009.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Trappmann B., Gautrot J.E., Connelly J.T., Strange D.G.T., Li Y., Oyen M.L., Stuart M.A.C., Boehm H., Li B.J., Vogel V., Spatz J.P., Watt F.M., Huck W.T.S. Extracellular-matrix tethering regulates stem-cell fate. Nat Mater. 2012;11:642–649. doi: 10.1038/nmat3339. [DOI] [PubMed] [Google Scholar]
- 65.Imajo M., Ebisuya M., Nishida E. Dual role of YAP and TAZ in renewal of the intestinal epithelium. Nat Cell Biol. 2015;17:7–19. doi: 10.1038/ncb3084. [DOI] [PubMed] [Google Scholar]
- 66.Sokolis D.P., Sassani S.G. Microstructure-based constitutive modeling for the large intestine validated by histological observations. J Mech Behav Biomed. 2013;21:149–166. doi: 10.1016/j.jmbbm.2013.02.016. [DOI] [PubMed] [Google Scholar]
- 67.Mowat A.M., Agace W.W. Regional specialization within the intestinal immune system. Nat Rev Immunol. 2014;14:667–685. doi: 10.1038/nri3738. [DOI] [PubMed] [Google Scholar]
- 68.Nicholson J.K., Holmes E., Kinross J., Burcelin R., Gibson G., Jia W., Pettersson S. Host-gut microbiota metabolic interactions. Science. 2012;336:1262–1267. doi: 10.1126/science.1223813. [DOI] [PubMed] [Google Scholar]
- 69.Martin F.P.J., Dumas M.E., Wang Y.L., Legido-Quigley C., Yap I.K.S., Tang H.R., Zirah S., Murphy G.M., Cloarec O., Lindon J.C., Sprenger N., Fay L.B., Kochhar S., van Bladeren P., Holmes E., Nicholson J.K. A top-down systems biology view of microbiome-mammalian metabolic interactions in a mouse model. Mol Syst Biol. 2007;3:112. doi: 10.1038/msb4100153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Wikoff W.R., Anfora A.T., Liu J., Schultz P.G., Lesley S.A., Peters E.C., Siuzdak G. Metabolomics analysis reveals large effects of gut microflora on mammalian blood metabolites. Proc Natl Acad Sci U S A. 2009;106:3698–3703. doi: 10.1073/pnas.0812874106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Matsumoto M., Kibe R., Ooga T., Aiba Y., Kurihara S., Sawaki E., Koga Y., Benno Y. Impact of intestinal microbiota on intestinal luminal metabolome. Sci Rep. 2012;2:233. doi: 10.1038/srep00233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Sridharan G.V., Choi K., Klemashevich C., Wu C., Prabakaran D., Pan L.B., Steinmeyer S., Mueller C., Yousofshahi M., Alaniz R.C., Lee K., Jayaraman A. Prediction and quantification of bioactive microbiota metabolites in the mouse gut. Nat Commun. 2014;5:5492. doi: 10.1038/ncomms6492. [DOI] [PubMed] [Google Scholar]
- 73.Matsumoto M., Ooga T., Kibe R., Aiba Y., Koga Y., Benno Y. Colonic absorption of low-molecular-weight metabolites influenced by the intestinal microbiome: a pilot study. PLoS One. 2017;12:e0169207. doi: 10.1371/journal.pone.0169207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.D'Argenio G., Mazzacca G. Short-chain fatty acid in the human colon - relation to inflammatory bowel diseases and colon cancer. Adv Exp Med Biol. 1999;472:149–158. doi: 10.1007/978-1-4757-3230-6_13. [DOI] [PubMed] [Google Scholar]
- 75.Rios-Covian D., Ruas-Madiedo P., Margolles A., Gueimonde M. de los Reyes-Gavilan CG, Salazar N. Intestinal short chain fatty acids and their link with diet and human health. Front Microbiol. 2016;7:185. doi: 10.3389/fmicb.2016.00185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Park J.-H., Kotani T., Konno T., Setiawan J., Kitamura Y., Imada S., Usui Y., Hatano N., Shinohara M., Saito Y. Promotion of intestinal epithelial cell turnover by commensal bacteria: role of short-chain fatty acids. PLoS One. 2016;11:e0156334. doi: 10.1371/journal.pone.0156334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Donohoe D.R., Garge N., Zhang X.X., Sun W., O'Connell T.M., Bunger M.K., Bultman S.J. The microbiome and butyrate regulate energy metabolism and autophagy in the mammalian colon. Cell Metab. 2011;13:517–526. doi: 10.1016/j.cmet.2011.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Louis P., Flint H.J. Development of a semiquantitative degenerate real-time PCR-based assay for estimation of numbers of butyryl-coenzyme A (CoA) CoA transferase genes in complex bacterial samples. Appl Environ Microb. 2007;73:2009–2012. doi: 10.1128/AEM.02561-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Kaiko G.E., Ryu S.H., Koues O.I., Collins P.L., Solnica-Krezel L., Pearce E.J., Pearce E.L., Oltz E.M., Stappenbeck T.S. The colonic crypt protects stem cells from microbiota-derived metabolites. Cell. 2016;165:1708–1720. doi: 10.1016/j.cell.2016.05.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Pegg A.E., Mccann P.P. Polyamine metabolism and function. Am J Physiol. 1982;243:C212–C221. doi: 10.1152/ajpcell.1982.243.5.C212. [DOI] [PubMed] [Google Scholar]
- 81.Loser C., Eisel A., Harms D., Folsch U.R. Dietary polyamines are essential luminal growth factors for small intestinal and colonic mucosal growth and development. Gut. 1999;44:12–16. doi: 10.1136/gut.44.1.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Matsumoto M., Kurihara S., Kibe R., Ashida H., Benno Y. Longevity in mice is promoted by probiotic-induced suppression of colonic senescence dependent on upregulation of gut bacterial polyamine production. PLoS One. 2011;6:e23652. doi: 10.1371/journal.pone.0023652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Asano Y., Hiramoto T., Nishino R., Aiba Y., Kimura T., Yoshihara K., Koga Y., Sudo N. Critical role of gut microbiota in the production of biologically active, free catecholamines in the gut lumen of mice. Am J Physiol Gastrointest Liver Physiol. 2012;303:G1288–G1295. doi: 10.1152/ajpgi.00341.2012. [DOI] [PubMed] [Google Scholar]
- 84.Steed A.L., Christophi G.P., Kaiko G.E., Sun L.L., Goodwin V.M., Jain U., Esaulova E., Artyomov M.N., Morales D.J., Holtzman M.J., Boon A.C.M., Lenschow D.J., Stappenbeck T.S. The microbial metabolite desaminotyrosine protects from influenza through type I interferon. Science. 2017;357:498–502. doi: 10.1126/science.aam5336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Espey M.G. Role of oxygen gradients in shaping redox relationships between the human intestine and its microbiota. Free Radic Biol Med. 2013;55:130–140. doi: 10.1016/j.freeradbiomed.2012.10.554. [DOI] [PubMed] [Google Scholar]
- 86.Albenberg L., Esipova T.V., Judge C.P., Bittinger K., Chen J., Laughlin A., Grunberg S., Baldassano R.N., Lewis J.D., Li H.Z., Thom S.R., Bushman F.D., Vinogradov S.A., Wu G.D. Correlation between intraluminal oxygen gradient and radial partitioning of intestinal microbiota. Gastroenterology. 2014;147:1055–1063. doi: 10.1053/j.gastro.2014.07.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Rivera-Chavez F., Zhang L.F., Faber F., Lopez C.A., Byndloss M.X., Olsan E.E., Xu G.G., Velazquez E.M., Lebrilla C.B., Winter S.E., Baumler A.J. Depletion of butyrate-producing Clostridia from the gut microbiota drives an aerobic luminal expansion of salmonella. Cell Host Microbe. 2016;19:443–454. doi: 10.1016/j.chom.2016.03.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Zeitouni N.E., Chotikatum S., von Köckritz-Blickwede M., Naim H.Y. The impact of hypoxia on intestinal epithelial cell functions: consequences for invasion by bacterial pathogens. Mol Cell Pediatr. 2016;3:14. doi: 10.1186/s40348-016-0041-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Kelly C.J., Zheng L., Campbell E.L., Saeedi B., Scholz C.C., Bayless A.J., Wilson K.E., Glover L.E., Kominsky D.J., Magnuson A., Weir T.L., Ehrentraut S.F., Pickel C., Kuhn K.A., Lanis J.M., Nguyen V., Taylor C.T., Colgan S.P. Crosstalk between microbiota-derived short-chain fatty acids and intestinal epithelial HIF augments tissue barrier function. Cell Host Microbe. 2015;17:662–671. doi: 10.1016/j.chom.2015.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Thomas D.D., Liu Z.P., Kantrow S.P., Lancaster J.R. The biological lifetime of nitric oxide: Implications for the perivascular dynamics of NO and O-2. Proc Natl Acad Sci U S A. 2001;98:355–360. doi: 10.1073/pnas.011379598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Kolios G., Valatas V., Ward S.G. Nitric oxide in inflammatory bowel disease: a universal messenger in an unsolved puzzle. Immunology. 2004;113:427–437. doi: 10.1111/j.1365-2567.2004.01984.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Lundberg J.O.N., Hellstrom P.M., Lundberg J.M., Alving K. Greatly increased luminal nitric-oxide in ulcerative-colitis. Lancet. 1994;344:1673–1674. doi: 10.1016/s0140-6736(94)90460-x. [DOI] [PubMed] [Google Scholar]
- 93.Strocchi A., Furne J.K., Ellis C.J., Levitt M.D. Competition for hydrogen by human faecal bacteria: evidence for the predominance of methane producing bacteria. Gut. 1991;32:1498–1501. doi: 10.1136/gut.32.12.1498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Nava G.M., Carbonero F., Croix J.A., Greenberg E., Gaskins H.R. Abundance and diversity of mucosa-associated hydrogenotrophic microbes in the healthy human colon. ISME J. 2012;6:57–70. doi: 10.1038/ismej.2011.90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Wolf P.G., Biswas A., Morales S.E., Greening C., Gaskins H.R. H2 metabolism is widespread and diverse among human colonic microbes. Gut Microbes. 2016;7:235–245. doi: 10.1080/19490976.2016.1182288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Jowett S.L., Seal C.J., Pearce M.S., Phillips E., Gregory W., Barton J.R., Welfare M.R. Influence of dietary factors on the clinical course of ulcerative colitis: a prospective cohort study. Gut. 2004;53:1479–1484. doi: 10.1136/gut.2003.024828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Colgan S.P., Taylor C.T. Hypoxia: an alarm signal during intestinal inflammation. Nat Rev Gastroenterol Hepatol. 2010;7:281–287. doi: 10.1038/nrgastro.2010.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Ootani A., Li X., Sangiorgi E., Ho Q.T., Ueno H., Toda S., Sugihara H., Fujimoto K., Weissman I.L., Capecchi M.R., Kuo C.J. Sustained in vitro intestinal epithelial culture within a Wnt-dependent stem cell niche. Nat Med. 2009;15:701–706. doi: 10.1038/nm.1951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Attayek P.J., Ahmad A.A., Wang Y., Williamson I., Sims C.E., Magness S.T., Allbritton N.L. In vitro polarization of colonoids to create an intestinal stem cell compartment. PLoS One. 2016;11:e0153795. doi: 10.1371/journal.pone.0153795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Somaweera H., Ibraguimov A., Pappas D. A review of chemical gradient systems for cell analysis. Anal Chim Acta. 2016;907:7–17. doi: 10.1016/j.aca.2015.12.008. [DOI] [PubMed] [Google Scholar]
- 101.Wang Y., Gunasekara D.B., Reed M.I., DiSalvo M., Bultman S.J., Sims C.E., Magness S.T., Allbritton N.L. A microengineered collagen scaffold for generating a polarized crypt-villus architecture of human small intestinal epithelium. Biomaterials. 2017;128:44–55. doi: 10.1016/j.biomaterials.2017.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Wang Y., Kim R., Gunasekara D.B., Reed M.I., DiSalvo M., Nguyen D.L., Bultman S.J., Sims C.E., Magness S.T., Allbritton N.L. Formation of human colonic crypt array by application of chemical gradients across a shaped epithelial monolayer. Cell Mol Gastroenterol Hepatol. 2018;5:113–130. doi: 10.1016/j.jcmgh.2017.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Wang Y.L., Gunasekara D.B., Attayek P.J., Reed M.I., DiSalvo M., Nguyen D.L., Dutton J.S., Lebhar M.S., Bultman S.J., Sims C.E., Magness S.T., Allbritton N.L. In vitro generation of mouse colon crypts. ACS Biomater Sci Eng. 2017;3:2502–2513. doi: 10.1021/acsbiomaterials.7b00368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Lee K., Silva E.A., Mooney D.J. Growth factor delivery-based tissue engineering: general approaches and a review of recent developments. J R Soc Interface. 2011;8:153–170. doi: 10.1098/rsif.2010.0223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Wu J.D., Mao Z.W., Tan H.P., Han L.L., Ren T.C., Gao C.Y. Gradient biomaterials and their influences on cell migration. Interface Focus. 2012;2:337–355. doi: 10.1098/rsfs.2011.0124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Kim S., Kim H.J., Jeon N.L. Biological applications of microfluidic gradient devices. Integr Biol. 2010;2:584–603. doi: 10.1039/c0ib00055h. [DOI] [PubMed] [Google Scholar]
- 107.Kim C., Kreppenhofer K., Kashef J., Gradl D., Herrmann D., Schneider M., Ahrens R., Guber A., Wedlich D. Diffusion-and convection-based activation of Wnt/β-catenin signaling in a gradient generating microfluidic chip. Lab Chip. 2012;12:5186–5194. doi: 10.1039/c2lc40172j. [DOI] [PubMed] [Google Scholar]
- 108.Wang X., Wenk E., Zhang X., Meinel L., Vunjak-Novakovic G., Kaplan D.L. Growth factor gradients via microsphere delivery in biopolymer scaffolds for osteochondral tissue engineering. J Control Release. 2009;134:81–90. doi: 10.1016/j.jconrel.2008.10.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Min H.K., Oh S.H., Lee J.M., Im G.I., Lee J.H. Porous membrane with reverse gradients of PDGF-BB and BMP-2 for tendon-to-bone repair: in vitro evaluation on adipose-derived stem cell differentiation. Acta Biomater. 2014;10:1272–1279. doi: 10.1016/j.actbio.2013.12.031. [DOI] [PubMed] [Google Scholar]
- 110.Almodóvar J., Guillot R., Monge C., Vollaire J., Selimović Š., Coll J.-L., Khademhosseini A., Picart C. Spatial patterning of BMP-2 and BMP-7 on biopolymeric films and the guidance of muscle cell fate. Biomaterials. 2014;35:3975–3985. doi: 10.1016/j.biomaterials.2014.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Lowndes M., Rotherham M., Price J.C., El Haj A.J., Habib S.J. Immobilized WNT proteins act as a stem cell niche for tissue engineering. Stem Cell Rep. 2016;7:126–137. doi: 10.1016/j.stemcr.2016.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Ricoult S.G., Kennedy T.E., Juncker D. Substrate-bound protein gradients to study haptotaxis. Front Bioeng Biotechnol. 2015;3:40. doi: 10.3389/fbioe.2015.00040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Benetti E.M., Gunnewiek M.K., van Blitterswijk C.A., Vancso G.J., Moroni L. Mimicking natural cell environments: design, fabrication and application of bio-chemical gradients on polymeric biomaterial substrates. J Mater Chem B. 2016;4:4244–4257. doi: 10.1039/c6tb00947f. [DOI] [PubMed] [Google Scholar]
- 114.Cai K.Y., Kong T.T., Wang L., Liu P., Yang W.H., Chen C. Regulation of endothelial cells migration on poly(D, L-lactic acid) films immobilized with collagen gradients. Colloids Surfaces B Biointerfaces. 2010;79:291–297. doi: 10.1016/j.colsurfb.2010.04.012. [DOI] [PubMed] [Google Scholar]
- 115.Gunawan R.C., Silvestre J., Gaskins H.R., Kenis P.J.A., Leckband D.E. Cell migration and polarity on microfabricated gradients of extracellular matrix proteins. Langmuir. 2006;22:4250–4258. doi: 10.1021/la0531493. [DOI] [PubMed] [Google Scholar]
- 116.Cosson S., Kobel S.A., Lutolf M.P. Capturing complex protein gradients on biomimetic hydrogels for cell-based assays. Adv Funct Mater. 2009;19:3411–3419. [Google Scholar]
- 117.Oh S.H., Kim T.H., Lee J.H. Creating growth factor gradients in three dimensional porous matrix by centrifugation and surface immobilization. Biomaterials. 2011;32:8254–8260. doi: 10.1016/j.biomaterials.2011.07.027. [DOI] [PubMed] [Google Scholar]
- 118.Xia T.T., Liu W.Q., Yang L. A review of gradient stiffness hydrogels used in tissue engineering and regenerative medicine. J Biomed Mater Res A. 2017;105:1799–1812. doi: 10.1002/jbm.a.36034. [DOI] [PubMed] [Google Scholar]
- 119.Sunyer R., Jin A.J., Nossal R., Sackett D.L. Fabrication of hydrogels with steep stiffness gradients for studying cell mechanical response. PLoS One. 2012;7:e46107. doi: 10.1371/journal.pone.0046107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Sunyer R., Conte V., Escribano J., Elosegui-Artola A., Labernadie A., Valon L., Navajas D., Garcia-Aznar J.M., Munoz J.J., Roca-Cusachs P., Trepat X. Collective cell durotaxis emerges from long-range intercellular force transmission. Science. 2016;353:1157–1161. doi: 10.1126/science.aaf7119. [DOI] [PubMed] [Google Scholar]
- 121.Hadden W.J., Young J.L., Holle A.W., McFetridge M.L., Kim D.Y., Wijesinghe P., Taylor-Weiner H., Wen J.H., Lee A.R., Bieback K., Vo B.N., Sampson D.D., Kennedy B.F., Spatz J.P., Engler A.J., Choi Y.S. Stem cell migration and mechanotransduction on linear stiffness gradient hydrogels. Proc Natl Acad Sci U S A. 2017;114:5647–5652. doi: 10.1073/pnas.1618239114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Koser D.E., Thompson A.J., Foster S.K., Dwivedy A., Pillai E.K., Sheridan G.K., Svoboda H., Viana M., Costa L.D., Guck J., Holt C.E., Franze K. Mechanosensing is critical for axon growth in the developing brain. Nat Neurosci. 2016;19:1592–1598. doi: 10.1038/nn.4394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Wang P.Y., Tsai W.B., Voelcker N.H. Screening of rat mesenchymal stem cell behaviour on polydimethylsiloxane stiffness gradients. Acta Biomater. 2012;8:519–530. doi: 10.1016/j.actbio.2011.09.030. [DOI] [PubMed] [Google Scholar]
- 124.Kim T.H., An D.B., Oh S.H., Kang M.K., Song H.H., Lee J.H. Creating stiffness gradient polyvinyl alcohol hydrogel using a simple gradual freezing-thawing method to investigate stem cell differentiation behaviors. Biomaterials. 2015;40:51–60. doi: 10.1016/j.biomaterials.2014.11.017. [DOI] [PubMed] [Google Scholar]
- 125.Lukovac S., Belzer C., Pellis L., Keijser B.J., de Vos W.M., Montijn R.C., Roeselers G. Differential modulation by Akkermansia muciniphila and Faecalibacterium prausnitzii of host peripheral lipid metabolism and histone acetylation in mouse gut organoids. MBio. 2014;5:e01438. doi: 10.1128/mBio.01438-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Hill D.R., Huang S., Fields C., Mukherjee D., Bons B., Thodla S., Dedhia P.H., Chin A.M., Tsai Y.-H., Nagy M.S. Bacterial colonization stimulates a complex physiological response in the immature human intestinal epithelium. eLife. 2017;6:e29132. doi: 10.7554/eLife.29132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Cerchiari A.E., Samy K.E., Todhunter M.E., Schlesinger E., Henise J., Rieken C., Gartner Z.J., Desai T.A. Probing the luminal microenvironment of reconstituted epithelial microtissues. Sci Rep. 2016;6:33148. doi: 10.1038/srep33148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Van Rymenant E., Abrankó L., Tumova S., Grootaert C., Van Camp J., Williamson G., Kerimi A. Chronic exposure to short-chain fatty acids modulates transport and metabolism of microbiome-derived phenolics in human intestinal cells. J Nutr Biochem. 2017;39:156–168. doi: 10.1016/j.jnutbio.2016.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Peng L., He Z., Chen W., Holzman I.R., Lin J. Effects of butyrate on intestinal barrier function in a Caco-2 cell monolayer model of intestinal barrier. Pediatr Res. 2007;61:37–41. doi: 10.1203/01.pdr.0000250014.92242.f3. [DOI] [PubMed] [Google Scholar]
- 130.Dossa A.Y., Escobar O., Golden J., Frey M.R., Ford H.R., Gayer C.P. Bile acids regulate intestinal cell proliferation by modulating EGFR and FXR signaling. Am J Physiol Gastrintest Liver Physiol. 2016;310:G81–G92. doi: 10.1152/ajpgi.00065.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Yoshimoto A.N., Bernardazzi C., Carneiro A.J.V., Elia C.C., Martinusso C.A., Ventura G.M., Castelo-Branco M.T., De Souza H.S. Hedgehog pathway signaling regulates human colon carcinoma HT-29 epithelial cell line apoptosis and cytokine secretion. PLoS One. 2012;7:e45332. doi: 10.1371/journal.pone.0045332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Moon C., VanDussen K.L., Miyoshi H., Stappenbeck T.S. Development of a primary mouse intestinal epithelial cell monolayer culture system to evaluate factors that modulate IgA transcytosis. Mucosal Immunol. 2014;7:818–828. doi: 10.1038/mi.2013.98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Kim H.J., Huh D., Hamilton G., Ingber D.E. Human gut-on-a-chip inhabited by microbial flora that experiences intestinal peristalsis-like motions and flow. Lab Chip. 2012;12:2165–2174. doi: 10.1039/c2lc40074j. [DOI] [PubMed] [Google Scholar]
- 134.von Martels J.Z.H., Sadabad M.S., Bourgonje A.R., Blokzijl T., Dijkstra G., Faber K.N., Harmsen H.J.M. The role of gut microbiota in health and disease: in vitro modeling of host-microbe interactions at the aerobe-anaerobe interphase of the human gut. Anaerobe. 2017;44:3–12. doi: 10.1016/j.anaerobe.2017.01.001. [DOI] [PubMed] [Google Scholar]
- 135.Ulluwishewa D., Anderson R.C., Young W., McNabb W.C., Baarlen P., Moughan P.J., Wells J.M., Roy N.C. Live Faecalibacterium prausnitzii in an apical anaerobic model of the intestinal epithelial barrier. Cell Microbiol. 2015;17:226–240. doi: 10.1111/cmi.12360. [DOI] [PubMed] [Google Scholar]
- 136.Shah P., Fritz J.V., Glaab E., Desai M.S., Greenhalgh K., Frachet A., Niegowska M., Estes M., Jäger C., Seguin-Devaux C., Zenhausern F., Wilmes P. A microfluidics-based in vitro model of the gastrointestinal human–microbe interface. Nat Commun. 2016;7:11535. doi: 10.1038/ncomms11535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Wood M.B., Rios D., Williams I.R. TNF-α augments RANKL-dependent intestinal M cell differentiation in enteroid cultures. Am J Physiol Cell Physiol. 2016;311:C498–C507. doi: 10.1152/ajpcell.00108.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Lindemans C.A., Calafiore M., Mertelsmann A.M., O’Connor M.H., Dudakov J.A., Jenq R.R., Velardi E., Young L.F., Smith O.M., Lawrence G. Interleukin-22 promotes intestinal-stem-cell-mediated epithelial regeneration. Nature. 2015;528:560–564. doi: 10.1038/nature16460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Noel G., Baetz N.W., Staab J.F., Donowitz M., Kovbasnjuk O., Pasetti M.F., Zachos N.C. A primary human macrophage-enteroid co-culture model to investigate mucosal gut physiology and host-pathogen interactions. Sci Rep. 2017;7:45270. doi: 10.1038/srep45270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Adler M., Polinkovsky M., Gutierrez E., Groisman A. Generation of oxygen gradients with arbitrary shapes in a microfluidic device. Lab Chip. 2010;10:388–391. doi: 10.1039/b920401f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Rexius-Hall M.L., Rehman J., Eddington D.T. A microfluidic oxygen gradient demonstrates differential activation of the hypoxia-regulated transcription factors HIF-1α and HIF-2α. Integr Biol. 2017;9:742–750. doi: 10.1039/c7ib00099e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Lo J.F., Sinkala E., Eddington D.T. Oxygen gradients for open well cellular cultures via microfluidic substrates. Lab Chip. 2010;10:2394–2401. doi: 10.1039/c004660d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Chen Y., Lin Y.N., Davis K.M., Wang Q.R., Rnjak-Kovacina J., Li C.M., Isberg R.R., Kumamoto C.A., Mecsas J., Kaplan D.L. Robust bioengineered 3D functional human intestinal epithelium. Sci Rep. 2015;5:13708. doi: 10.1038/srep13708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Lewis D.M., Blatchley M.R., Park K.M., Gerecht S. O-2-controllable hydrogels for studying cellular responses to hypoxic gradients in three dimensions in vitro and in vivo. Nat Protoc. 2017;12:1620–1638. doi: 10.1038/nprot.2017.059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Shimizu H., Okamoto R., Ito G., Fujii S., Nakata T., Suzuki K., Murano T., Mizutani T., Tsuchiya K., Nakamura T., Hozumi K., Watanabe M. Distinct expression patterns of Notch ligands, Dll1 and Dll4, in normal and inflamed mice intestine. PeerJ. 2014;2:e370. doi: 10.7717/peerj.370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.He X.C., Zhang J., Tong W.G., Tawfik O., Ross J., Scoville D.H., Tian Q., Zeng X., He X., Wiedemann L.M., Mishina Y., Li L. BMP signaling inhibits intestinal stem cell self-renewal through suppression of Wnt-beta-catenin signaling. Nat Genet. 2004;36:1117–1121. doi: 10.1038/ng1430. [DOI] [PubMed] [Google Scholar]
- 147.Cummings J.H., Macfarlane G.T. The control and consequences of bacterial fermentation in the human colon. J Appl Bacteriol. 1991;70:443–459. doi: 10.1111/j.1365-2672.1991.tb02739.x. [DOI] [PubMed] [Google Scholar]
- 148.Suarez F., Furne J., Springfield J., Levitt M. Insights into human colonic physiology obtained from the study of flatus composition. Am J Physiol. 1997;272:G1028–G1033. doi: 10.1152/ajpgi.1997.272.5.G1028. [DOI] [PubMed] [Google Scholar]
- 149.Wong J.M., de Souza R., Kendall C.W., Emam A., Jenkins D.J. Colonic health: fermentation and short chain fatty acids. J Clin Gastroenterol. 2006;40:235–243. doi: 10.1097/00004836-200603000-00015. [DOI] [PubMed] [Google Scholar]
- 150.Morrison D.J., Preston T. Formation of short chain fatty acids by the gut microbiota and their impact on human metabolism. Gut Microbes. 2016;7:189–200. doi: 10.1080/19490976.2015.1134082. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Pomare E.W., Branch W.J., Cummings J.H. Carbohydrate fermentation in the human colon and its relation to acetate concentrations in venous blood. J Clin Invest. 1985;75:1448–1454. doi: 10.1172/JCI111847. [DOI] [PMC free article] [PubMed] [Google Scholar]